Using Graphene-Based Biosensors to Detect Dopamine for Efficient Parkinson’s Disease Diagnostics
Abstract
:1. Introduction
2. Analytical Performances of DA Graphene-Based Biosensors
3. Challenges and Perspectives towards POC Diagnostics of DA
4. Conclusions and Viewpoint
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kalia, L.V.; Lang, A.E. Parkinson’s disease. Lancet 2015, 386, 896–912. [Google Scholar] [CrossRef]
- Cheng, H.C.; Ulane, C.M.; Burke, R.E. Clinical progression in Parkinson disease and the neurobiology of axons. Ann. Neurol. 2010, 67, 715–725. [Google Scholar] [CrossRef] [PubMed]
- Eldrup, E.; Mogensen, P.; Jacobsen, J.; Pakkenberg, H.; Christensen, N.J. CSF and plasma concentrations of free norepinephrine, dopamine, 3,4-dihydroxyphenylacetic acid (DOPAC), 3,4-dihydroxyphenylalanine (DOPA), and epinephrine in Parkinson’s disease. Acta Neurol. Scand. 1995, 92, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, D.S.; Sullivan, P.; Holmes, C.; Kopin, I.J.; Basile, M.J.; Mash, D.C. Catechols in post-mortem brain of patients with Parkinson disease. Eur J. Neurol 2011, 18, 703–710. [Google Scholar] [CrossRef]
- Véronneau-Veilleux, F.; Ursino, M.; Robaey, P.; Lévesque, D.; Nekka, F. Nonlinear pharmacodynamics of levodopa through Parkinson’s disease progression. Chaos 2020, 30, 093146. [Google Scholar] [CrossRef] [PubMed]
- Thomas, I.; Alam, M.; Bergquist, F.; Johansson, D.; Memedi, M.; Nyholm, D.; Westin, J. Sensor-based algorithmic dosing suggestions for oral administration of levodopa/carbidopa microtablets for Parkinson’s disease: A first experience. J. Neurol. 2019, 266, 651–658. [Google Scholar] [CrossRef] [Green Version]
- Si, Y.; Park, Y.E.; Lee, J.E.; Lee, H.J. Nanocomposites of poly(l-methionine), carbon nanotube-graphene complexes and Au nanoparticles on screen printed carbon electrodes for electrochemical analyses of dopamine and uric acid in human urine solutions. Analyst 2020, 145, 3656–3665. [Google Scholar] [CrossRef]
- Goldstein, D.S.; Holmes, C.; Sharabi, Y. Cerebrospinal fluid biomarkers of central catecholamine deficiency in Parkinson’s disease and other synucleinopathies. Brain 2012, 135, 1900–1913. [Google Scholar] [CrossRef]
- Andersen, A.D.; Blaabjerg, M.; Binzer, M.; Kamal, A.; Thagesen, H.; Kjaer, T.W.; Stenager, E.; Gramsbergen, J.B.P. Cerebrospinal fluid levels of catecholamines and its metabolites in Parkinson’s disease: Effect of l-DOPA treatment and changes in levodopa-induced dyskinesia. J. Neurochem. 2017, 141, 614–625. [Google Scholar] [CrossRef] [Green Version]
- Davidson, D.F. Elevated urinary dopamine in adults and children. Ann. Clin. Biochem. 2005, 42, 200–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Senel, M.; Dervisevic, E.; Alhassen, S.; Dervisevic, M.; Alachkar, A.; Cadarso, V.J.; Voelcker, N.H. Microfluidic electrochemical sensor for cerebrospinal fluid and blood dopamine detection in a mouse model of parkinson’s disease. Anal. Chem. 2020, 92, 12347–12355. [Google Scholar] [CrossRef] [PubMed]
- Asif, M.; Liu, H.; Aziz, A.; Wang, H.; Wang, Z.; Ajmal, M.; Xiao, F.; Liu, H. Core-shell iron oxide-layered double hydroxide: High electrochemical sensing performance of H2O2 biomarker in live cancer cells with plasma therapeutics. Biosens. Bioelectron. 2017, 97, 352–359. [Google Scholar] [CrossRef]
- Asif, M.; Aziz, A.; Ashraf, G.; Iftikhar, T.; Sun, Y.; Xiao, F.; Liu, H. Unveiling microbiologically influenced corrosion engineering to transfigure damages into benefits: A textile sensor for H2O2 detection in clinical cancer tissues. Chem. Eng. J. 2022, 427, 131398. [Google Scholar] [CrossRef]
- Asif, M.; Haitao, W.; Shuang, D.; Aziz, A.; Zhang, G.; Xiao, F.; Liu, H. Metal oxide intercalated layered double hydroxide nanosphere: With enhanced electrocatalyic activity towards H2O2 for biological applications. Sens. Actuators B Chem. 2017, 239, 243–252. [Google Scholar] [CrossRef]
- Atcherley, C.W.; Laude, N.D.; Monroe, E.B.; Wood, K.M.; Hashemi, P.; Heien, M.L. Improved Calibration of voltammetric sensors for studying pharmacological effects on dopamine transporter kinetics in vivo. ACS Chem. Neurosci. 2015, 6, 1509–1516. [Google Scholar] [CrossRef] [PubMed]
- Asif, M.; Aziz, A.; Wang, H.; Wang, Z.; Wang, W.; Ajmal, M.; Xiao, F.; Chen, X.; Liu, H. Superlattice stacking by hybridizing layered double hydroxide nanosheets with layers of reduced graphene oxide for electrochemical simultaneous determination of dopamine, uric acid and ascorbic acid. Mikrochim. Acta 2019, 186, 61. [Google Scholar] [CrossRef]
- Li, B.R.; Hsieh, Y.J.; Chen, Y.X.; Chung, Y.T.; Pan, C.Y.; Chen, Y.T. An ultrasensitive nanowire-transistor biosensor for detecting dopamine release from living PC12 cells under hypoxic stimulation. J. Am. Chem. Soc. 2013, 135, 16034–16037. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Alharbi, A.; Kiani, R.; Shahrjerdi, D. Quantitative principles for precise engineering of sensitivity in graphene electrochemical sensors. Adv. Mater. 2019, 31, e1805752. [Google Scholar] [CrossRef] [PubMed]
- Kujawska, M.; Jourdes, M.; Witucki, Ł.; Karaźniewicz-Łada, M.; Szulc, M.; Górska, A.; Mikołajczak, P.Ł.; Teissedre, P.-L.; Jodynis-Liebert, J. Pomegranate Juice Ameliorates Dopamine Release and Behavioral Deficits in a Rat Model of Parkinson’s Disease. Brain Sci. 2021, 11, 1127. [Google Scholar] [CrossRef]
- Ji, D.; Liu, Z.; Liu, L.; Low, S.S.; Lu, Y.; Yu, X.; Zhu, L.; Li, C.; Liu, Q. Smartphone-based integrated voltammetry system for simultaneous detection of ascorbic acid, dopamine, and uric acid with graphene and gold nanoparticles modified screen-printed electrodes. Biosens. Bioelectron. 2018, 119, 55–62. [Google Scholar] [CrossRef]
- Cernat, A.; Ştefan, G.; Tertis, M.; Cristea, C.; Simon, I. An overview of the detection of serotonin and dopamine with graphene-based sensors. Bioelectrochemistry 2020, 136, 107620. [Google Scholar] [CrossRef]
- Butler, D.; Moore, D.; Glavin, N.R.; Robinson, J.A.; Ebrahimi, A. Facile Post-deposition Annealing of Graphene Ink Enables Ultrasensitive Electrochemical Detection of Dopamine. ACS Appl. Mater. Interfaces 2021, 13, 11185–11194. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, S.K.; Chauhan, R.; Yadav, P.; Ghosh, S.; Mahapatro, A.K.; Singh, J.; Basu, T. Bi-enzyme functionalized electrochemically reduced transparent graphene oxide platform for triglyceride detection. Biomater. Sci. 2019, 7, 1598–1606. [Google Scholar] [CrossRef]
- Sheetal, K.B.; Basu, T. Study on binding phenomenon of lipase enzyme with tributyrin on the surface of graphene oxide array using surface plasmon resonance. Thin Solid Films 2018, 645, 10–18. [Google Scholar] [CrossRef]
- Bhardwaj, S.K.; Yadav, P.; Ghosh, S.; Basu, T.; Mahapatro, A.K. Biosensing Test-Bed Using Electrochemically Deposited Reduced Graphene Oxide. ACS Appl. Mater. Interfaces 2016, 8, 24350–24360. [Google Scholar] [CrossRef]
- Bhardwaj, S.K.; Basu, T.; Mahapatro, A.K. Triglyceride detection using reduced graphene oxide on ITO surface. Integr. Ferroelectr. 2017, 184, 92–98. [Google Scholar] [CrossRef]
- Minta, D.; Moyseowicz, A.; Gryglewicz, S.; Gryglewicz, G. A Promising Electrochemical Platform for Dopamine and Uric Acid Detection Based on a Polyaniline/Iron Oxide-Tin Oxide/Reduced Graphene Oxide Ternary Composite. Molecules 2020, 25, 5869. [Google Scholar] [CrossRef]
- Wang, N.; Liu, Y.; Fu, Y.; Yan, F. AC Measurements Using Organic Electrochemical Transistors for Accurate Sensing. ACS Appl. Mater. Interfaces 2018, 10, 25834–25840. [Google Scholar] [CrossRef]
- Hui, X.; Xuan, X.; Kim, J.; Park, J.Y. A highly flexible and selective dopamine sensor based on Pt-Au nanoparticle-modified laser-induced graphene. Electrochimica Acta 2019, 328, 135066. [Google Scholar] [CrossRef]
- Vilian, A.T.E.; An, S.; Choe, S.R.; Kwak, C.H.; Huh, Y.S.; Lee, J.; Han, Y.K. Fabrication of 3D honeycomb-like porous polyurethane-functionalized reduced graphene oxide for detection of dopamine. Biosens. Bioelectron. 2016, 86, 122–128. [Google Scholar] [CrossRef]
- Wang, L.; Yang, R.; Qu, L.; Harrington, P.B. Electrostatic repulsion strategy for high-sensitive and selective determination of dopamine in the presence of uric acid and ascorbic acid. Talanta 2020, 210, 120626. [Google Scholar] [CrossRef]
- Yue, H.Y.; Huang, S.; Chang, J.; Heo, C.; Yao, F.; Adhikari, S.; Gunes, F.; Liu, L.C.; Lee, T.H.; Oh, E.S.; et al. ZnO nanowire arrays on 3D hierachical graphene foam: Biomarker detection of Parkinson’s disease. ACS Nano 2014, 8, 1639–1646. [Google Scholar] [CrossRef]
- Aziz, A.; Asif, M.; Azeem, M.; Ashraf, G.; Wang, Z.; Xiao, F.; Liu, H. Self-stacking of exfoliated charged nanosheets of LDHs and graphene as biosensor with real-time tracking of dopamine from live cells. Anal. Chim. Acta 2019, 1047, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Yang, Q.; Zhang, W.; Xiao, G.; Wang, M.; Cheng, L.; Zhou, X.; Zhao, M.; Ji, J.; Zhang, J.; et al. Silver nanoclusters and carbon dots based light-addressable sensors for multichannel detections of dopamine and glutathione and its applications in probing of parkinson’s diseases. Talanta 2020, 219, 121290. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, C.; Thirumalraj, B.; Chen, S.M.; Chen, H.A. A simple preparation of graphite/gelatin composite for electrochemical detection of dopamine. J. Colloid Interface Sci. 2017, 487, 149–155. [Google Scholar] [CrossRef]
- Rani, G.P.J.; Babu, K.J.; Kumar, G.G.; Rajan, A.J. Watsonia meriana flower like Fe3O4/reduced graphene oxide nanocomposite for the highly sensitive and selective electrochemical sensing of dopamine. J. Alloys Compd. 2016, 688, 500–512. [Google Scholar] [CrossRef]
- Zhang, W.; Zheng, J.; Shi, J.; Lin, Z.; Huang, Q.; Zhang, H.; Wei, C.; Chen, J.; Hu, S.; Hao, A. Nafion covered core-shell structured Fe3O4@graphene nanospheres modified electrode for highly selective detection of dopamine. Anal. Chim. Acta 2015, 853, 285–290. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, X.; Wang, M.; Huang, H.; Ma, J. A facile electrochemical sensor based on well-dispersed graphene-molybdenum disulfide modified electrode for highly sensitive detection of dopamine. J. Electroanal. Chem. 2017, 786, 1–7. [Google Scholar] [CrossRef]
- Bagheri, H.; Pajooheshpour, N.; Jamali, B.; Amidi, S.; Hajian, A.; Khoshsafar, H. A novel electrochemical platform for sensitive and simultaneous determination of dopamine, uric acid and ascorbic acid based on Fe3O4SnO2Gr ternary nanocomposite. Microchem. J. 2017, 131, 120–129. [Google Scholar] [CrossRef]
- Sakthinathan, S.; Lee, H.F.; Chen, S.M.; Tamizhdurai, P. Electrocatalytic oxidation of dopamine based on non-covalent functionalization of manganese tetraphenylporphyrin/reduced graphene oxide nanocomposite. J. Colloid Interface Sci. 2016, 468, 120–127. [Google Scholar] [CrossRef]
- Aydoğdu, T.G. Development of electrochemical sensor for detection of ascorbic acid, dopamine, uric acid and l-tryptophan based on Ag nanoparticles and poly(l-arginine)-graphene oxide composite. J. Electroanal. Chem. 2017, 807, 19–28. [Google Scholar] [CrossRef]
- Haldorai, Y.; Vilian, A.E.; Rethinasabapathy, M.; Huh, Y.S.; Han, Y.-K. Electrochemical determination of dopamine using a glassy carbon electrode modified with TiN-reduced graphene oxide nanocomposite. Sens. Actuators B Chem. 2017, 247, 61–69. [Google Scholar] [CrossRef]
- Wang, D.; Xu, F.; Hu, J.; Lin, M. Phytic acid/graphene oxide nanocomposites modified electrode for electrochemical sensing of dopamine. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 71, 1086–1089. [Google Scholar] [CrossRef]
- Daemi, S.; Ashkarran, A.A.; Bahari, A.; Ghasemi, S. Gold nanocages decorated biocompatible amine functionalized graphene as an efficient dopamine sensor platform. J. Colloid Interface Sci. 2017, 494, 290–299. [Google Scholar] [CrossRef]
- Liu, Y.; She, P.; Gong, J.; Wu, W.; Xu, S.; Li, J.; Zhao, K.; Deng, A. A novel sensor based on electrodeposited Au–Pt bimetallic nano-clusters decorated on graphene oxide (GO)–electrochemically reduced GO for sensitive detection of dopamine and uric acid. Sens. Actuators B Chem. 2015, 221, 1542–1553. [Google Scholar] [CrossRef]
- Mathew, G.; Dey, P.; Das, R.; Chowdhury, S.D.; Paul Das, M.; Veluswamy, P.; Neppolian, B.; Das, J. Direct electrochemical reduction of hematite decorated graphene oxide α-Fe. Biosens. Bioelectron. 2018, 115, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zheng, X.; Li, Z.; Zheng, J. A facile one-pot synthesis of carbon nitride dots-reduced graphene oxide nanocomposites for simultaneous enhanced detecting of dopamine and uric acid. Analyst 2016, 141, 4757–4765. [Google Scholar] [CrossRef]
- Yue, H.Y.; Zhang, H.J.; Huang, S.; Lu, X.X.; Gao, X.; Song, S.S.; Wang, Z.; Wang, W.Q.; Guan, E.H. Highly sensitive and selective dopamine biosensor using Au nanoparticles-ZnO nanocone arrays/graphene foam electrode. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 108, 110490. [Google Scholar] [CrossRef]
- Xiao, G.; Song, Y.; Zhang, Y.; Xing, Y.; Zhao, H.; Xie, J.; Xu, S.; Gao, F.; Wang, M.; Xing, G.; et al. Microelectrode Arrays Modified with Nanocomposites for Monitoring Dopamine and Spike Firings under Deep Brain Stimulation in Rat Models of Parkinson’s Disease. ACS Sens. 2019, 4, 1992–2000. [Google Scholar] [CrossRef]
- Biji, R.S.a.P. Preparation of copper (I) oxide nanohexagon decorated reduced graphene oxide nanocomposite and its application in electrochemical sensing of dopamine. Mater. Sci. Eng. B 2016, 210, 10–18. [Google Scholar] [CrossRef]
- Ahammad, A.J.S.; Odhikari, N.; Shah, S.S.; Hasan, M.M.; Islam, T.; Pal, P.R.; Ahmed Qasem, M.A.; Aziz, M.A.Y. Porous tal palm carbon nanosheets: Preparation, characterization and application for the simultaneous determination of dopamine and uric acid. Nanoscale Adv. 2019, 1, 613. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Li, L.; Ma, W.; Chen, X.; Zhang, Y. Electrodeposited reduced graphene oxide incorporating polymerization of l-lysine on electrode surface and its application in simultaneous electrochemical determination of ascorbic acid, dopamine and uric acid. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 70, 241–249. [Google Scholar] [CrossRef]
- Jiang, J.; Ding, D.; Wang, J.; Lin, X.; Diao, G. Three-dimensional nitrogen-doped graphene-based metal-free electrochemical sensors for simultaneous determination of ascorbic acid, dopamine, uric acid, and acetaminophen. Analyst 2021, 146, 964–970. [Google Scholar] [CrossRef]
- Kim, D.; Lee, S.; Piao, Y. Electrochemical determination of dopamine and acetaminophen using activated graphene-Nafion modified glassy carbon electrode. J. Electroanal. Chem. 2017, 794, 221–228. [Google Scholar] [CrossRef]
- Ammara Ejaz and Yuri Joo and Seungwon, J. Fabrication of 1,4-bis(aminomethyl)benzene and cobalt hydroxide @ graphene oxide for selective detection of dopamine in the presence of ascorbic acid and serotonin. Sens. Actuators B Chem. 2017, 240, 297–307. [Google Scholar] [CrossRef]
- Li, Y.-H.; Ji, Y.; Ren, B.-B.; Jia, L.-N.; Cai, Q.; Liu, X.-S. Palladium-doped graphene-modified nano-carbon ionic liquid electrode: Preparation, characterization and simultaneous voltammetric determination of dopamine and uric acid. J. Iran. Chem. Soc. 2019, 16, 1903–1912. [Google Scholar] [CrossRef]
- Gao, X.; Yue, H.; Song, S.; Huang, S.; Li, B.; Lin, X.; Guo, E.; Wang, B.; Guan, E.; Zhang, H.; et al. 3-Dimensional hollow graphene balls for voltammetric sensing of levodopa in the presence of uric acid. Mikrochim. Acta 2018, 185, 91. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Li, J.; Shi, K.; Wang, Q.; Zhao, X.; Xiong, Z.; Zou, X.; Wang, Y. Graphene coated by polydopamine/multi-walled carbon nanotubes modified electrode for highly selective detection of dopamine and uric acid in the presence of ascorbic acid. J. Electroanal. Chem. 2016, 770, 56–61. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Y.-C.; Ma, L.-X. One-pot facile fabrication of graphene-zinc oxide composite and its enhanced sensitivity for simultaneous electrochemical detection of ascorbic acid, dopamine and uric acid. Sens. Actuators B Chem. 2016, 227, 488–496. [Google Scholar] [CrossRef]
- Wang, C.; Du, J.; Wang, H.; Zou, C.; Jiang, F.; Yang, P.; Du, Y. A facile electrochemical sensor based on reduced graphene oxide and Au nanoplates modified glassy carbon electrode for simultaneous detection of ascorbic acid, dopamine and uric acid. Sens. Actuators B Chem. 2014, 204, 302–309. [Google Scholar] [CrossRef]
- Zhu, X.; Liang, Y.; Zuo, X.; Hu, R.; Xiao, X.; Nan, J. Novel water-soluble multi-nanopore graphene modified glassy carbon electrode for simultaneous determination of dopamine and uric acid in the presence of ascorbic acid. Electrochimica Acta 2014, 143, 366–373. [Google Scholar] [CrossRef]
- Ahlem, T.; Amina, R.; Gaëlle, C. A Simple Fluorescent Aptasensing Platform Based on Graphene Oxide for Dopamine Determination. Appl. Biochem. Biotechnol. 2021. [Google Scholar] [CrossRef]
- Cheng, R.; Ge, C.; Qi, L.; Zhang, Z.; Ma, J.; Huang, H.; Pan, T.; Dai, Q.; Dai, L. Label-Free Graphene Oxide Förster Resonance Energy Transfer Sensors for Selective Detection of Dopamine in Human Serums and Cells. J. Phys. Chem. C 2018, 122, 13314–13321. [Google Scholar] [CrossRef]
- Wang, Y.; Kang, K.; Wang, S.; Kang, W.; Cheng, C.; Niu, L.M.; Guo, Z. A novel label-free fluorescence aptasensor for dopamine detection based on an Exonuclease III- and SYBR Green I- aided amplification strategy. Sens. Actuators B Chem. 2020, 305, 127348. [Google Scholar] [CrossRef]
- Suzuki, Y. Development of Magnetic Nanobeads Modified by Artificial Fluorescent Peptides for the Highly Sensitive and Selective Analysis of Oxytocin. Sensors (Basel) 2020, 20, 5956. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.; DeRosa, M.C. Retention of function in the DNA homolog of the RNA dopamine aptamer. Biochem. Biophys. Res. Commun. 2009, 388, 732–735. [Google Scholar] [CrossRef]
- Zhou, X.; Ma, P.; Wang, A.; Yu, C.; Qian, T.; Wu, S.; Shen, J. Dopamine fluorescent sensors based on polypyrrole/graphene quantum dots core/shell hybrids. Biosens. Bioelectron. 2015, 64, 404–410. [Google Scholar] [CrossRef]
- Steckl, A.J.; Ray, P. Stress Biomarkers in Biological Fluids and Their Point-of-Use Detection. ACS Sens. 2018, 3, 2025–2044. [Google Scholar] [CrossRef]
- Campuzano, S.; Pedrero, M.; Yáñez-Sedeño, P.; Pingarrón, J.M. New challenges in point of care electrochemical detection of clinical biomarkers. Sens. Actuators B Chem. 2021, 345, 130349. [Google Scholar] [CrossRef]
- Shen, X.; Ju, F.; Li, G.; Ma, L. Smartphone-Based Electrochemical Potentiostat Detection System Using PEDOT: PSS/Chitosan/Graphene Modified Screen-Printed Electrodes for Dopamine Detection. Sensors 2020, 20, 2781. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Feng, L.; Xiong, L.; Li, S.; Xu, Q.; Pan, X.; Xiao, Y. Multifunctional nanoscale lanthanide metal-organic framework based ratiometric fluorescence paper microchip for visual dopamine assay. Nanoscale 2021, 13, 11188–11196. [Google Scholar] [CrossRef]
- Gwiazda, M.; Bhardwaj, S.K.; Kijeńska-Gawrońska, E.; Swieszkowski, W.; Sivasankaran, U.; Kaushik, A. Impedimetric and Plasmonic Sensing of Collagen I Using a Half-Antibody-Supported, Au-Modified, Self-Assembled Monolayer System. Biosensors 2021, 11, 227. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Shi, W.; Li, J.; Bao, N.; Yu, C.; Gu, H. Determination of salivary uric acid by using poly(3,4-ethylenedioxythipohene) and graphene oxide in a disposable paper-based analytical device. Anal. Chim. Acta 2020, 1103, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Lingervelder, D.; Koffijberg, H.; Kusters, R.; IJzerman, M.J. Point-of-care testing in primary care: A systematic review on implementation aspects addressed in test evaluations. Int. J. Clin. Pract. 2019, 73, e13392. [Google Scholar] [CrossRef] [Green Version]
- Nehra, M.; Uthappa, U.T.; Kumar, V.; Kumar, R.; Dixit, C.; Dilbaghi, N.; Mishra, Y.K.; Kumar, S.; Kaushik, A. Nanobiotechnology-assisted therapies to manage brain cancer in personalized manner. J. Control. Release 2021, 338, 224–243. [Google Scholar] [CrossRef] [PubMed]
- Khunger, A.; Kaur, N.; Mishra, Y.K.; Chaudhary, G.R.; Kaushik, A. Perspective and prospects of 2D MXenes for smart biosensing. Mater. Lett. 2021, 304, 130656. [Google Scholar] [CrossRef]
- Fuletra, I.; Chansi; Nisar, S.; Bharadwaj, R.; Saluja, P.; Bhardwaj, S.K.; Asokan, K.; Basu, T. Self-assembled gold nano islands for precise electrochemical sensing of trace level of arsenic in water. Groundw. Sustain. Dev. 2021, 12, 100528. [Google Scholar] [CrossRef]
- Pal, K.; Asthana, N.; A Aljabali, A.; Bhardwaj, S.K.; Kralj, S.; Penkova, A.; Thomas, S.; Zaheer, T.; de Souza, F.G. A critical review on multifunctional smart materials ‘nanographene’ emerging avenue: Nano-imaging and biosensor applications. Crit. Rev. Solid State Mater. Sci. 2021, 1–17. [Google Scholar] [CrossRef]
- Sharma, P.K.; Kim, E.-S.; Mishra, S.; Ganbold, E.; Seong, R.-S.; Kaushik, A.K.; Kim, N.-Y. Ultrasensitive and Reusable Graphene Oxide-Modified Double-Interdigitated Capacitive (DIDC) Sensing Chip for Detecting SARS-CoV-2. ACS Sens. 2021, 6, 3468–3476. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Casas, B.; Galdámez-Martínez, A.; Gutiérrez-Flores, J.; Baca Ibañez, A.; Kumar Panda, P.; Santana, G.; de la Vega, H.A.; Suar, M.; Gutiérrez Rodelo, C.; Kaushik, A.; et al. Bio-Acceptable 0D and 1D ZnO nanostructures for cancer diagnostics and treatment. Mater. Today 2021. [Google Scholar] [CrossRef]
- Kaushik, A.; Khan, R.; Solanki, P.; Gandhi., S.; Gohel, H.; Mishra, Y.K. From Nanosystems to a Biosensing Prototype for an Efficient Diagnostic: A Special Issue in Honor of Professor Bansi, D. Malhotra. Biosensors 2021, 11, 359. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.P.; Ruotolo, A.; Khan, R.; Mishra, Y.K.Y.; Kaushik, N.K.; Kim, N.-Y.; Kaushik, A.K. Perspectives on 2D-borophene flatland for smart bio-sensing. Mater. Lett. 2021, 308, 31089. [Google Scholar] [CrossRef]
- Bhardwaj, S.K.; Mujawar, M.; Mishra, Y.K.; Hickman, N.; Chavali, M.; Kaushik, A. Bio-inspired graphene-based nano-systems for biomedical applications. Nanotechnology 2021, 32, 502001. [Google Scholar] [CrossRef] [PubMed]
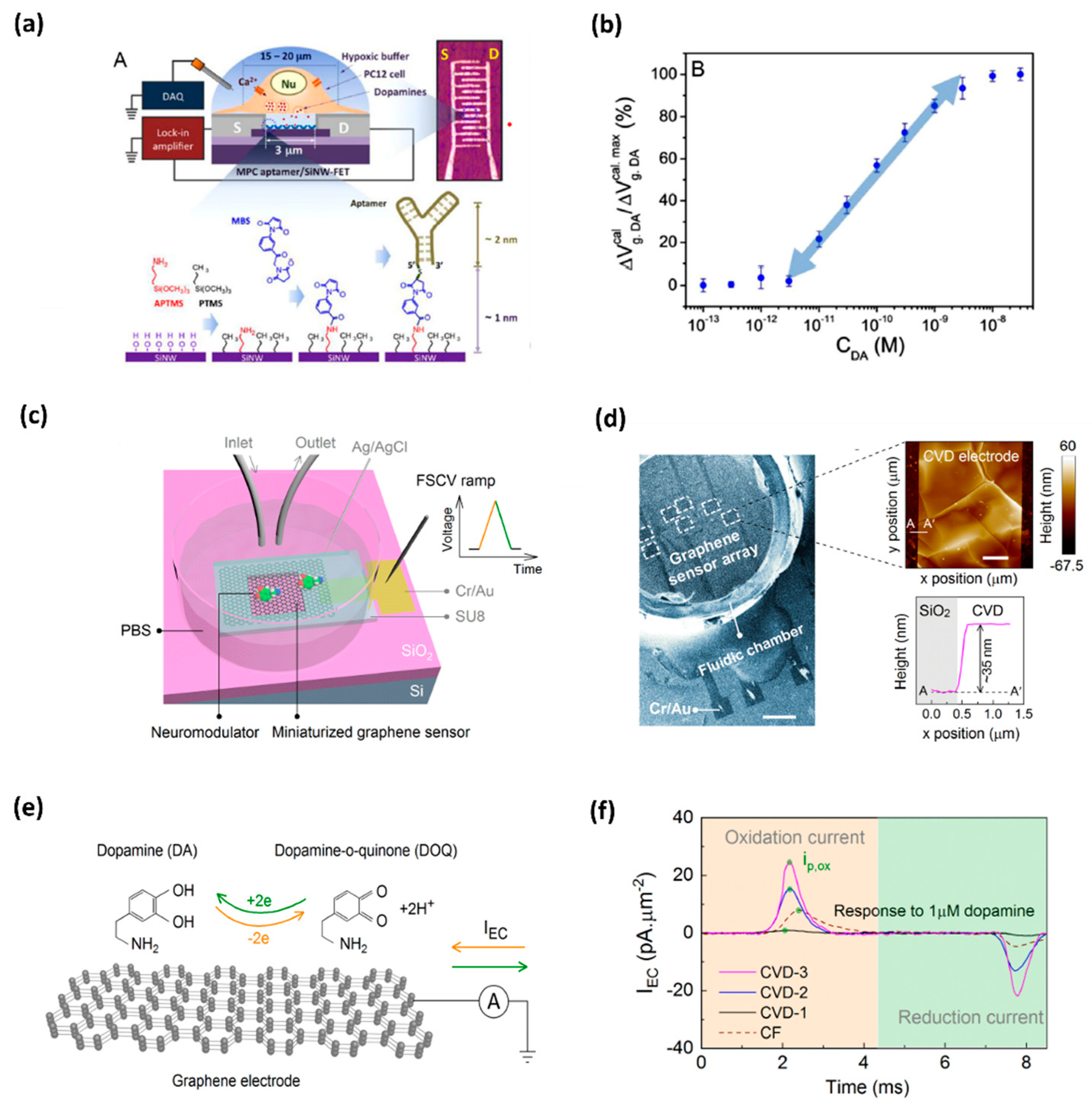
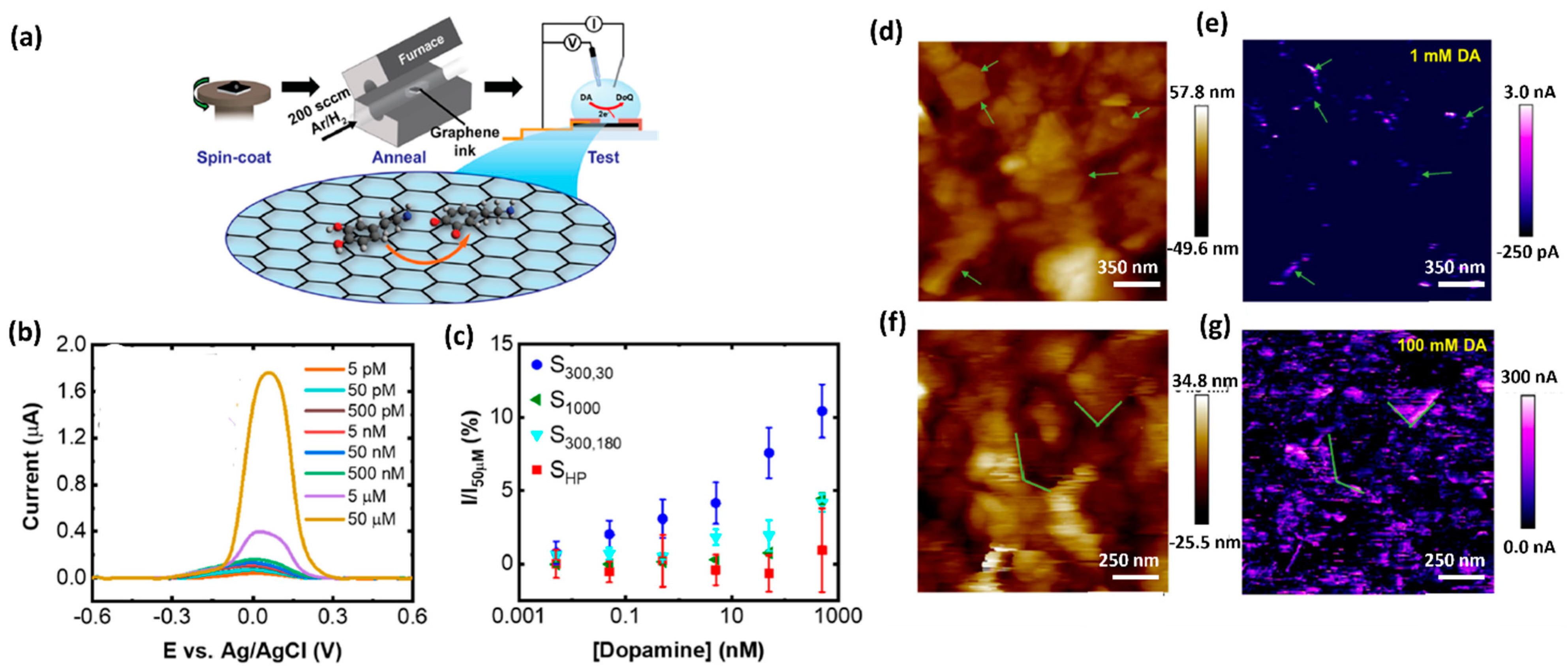
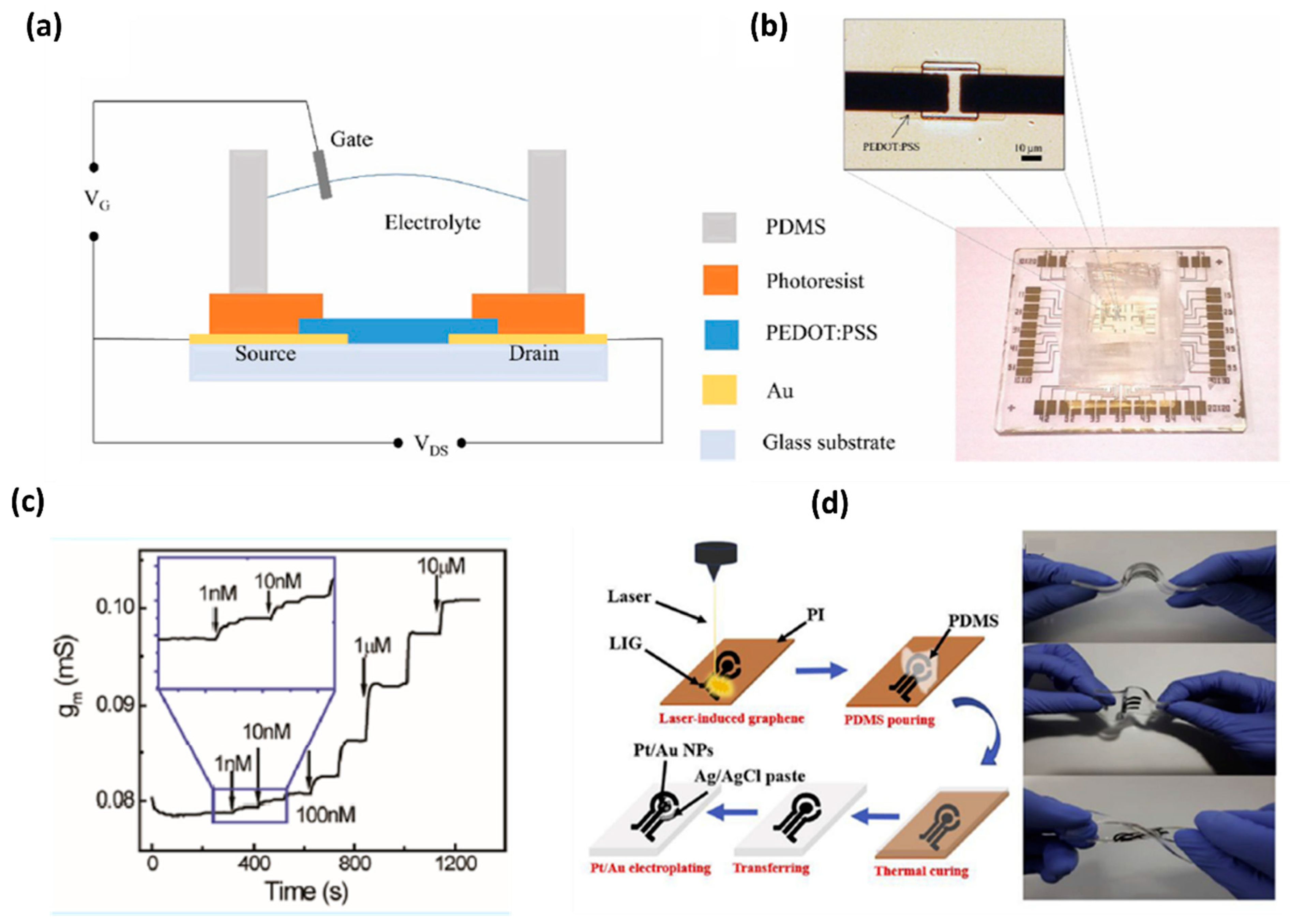

| Graphene Functionalization | LOD (μM) | Biological Samples | Interference Compound | Reference |
|---|---|---|---|---|
| 3D RGO-PU | 1.0 × 10−6 | (h) urine, serum | AA, UA, Glu, CA, 4-NP, Trp, Tyr, GSH | [30] |
| PFSG/GCE | 0.0008 | (h) serum | AA, UA | [31] |
| ZnO NWAs/GF | 0.001 | (PD) serum | AA, UA | [32] |
| NiAl LDH/G LBL | 0.002 | SH-SY 5Y cells | AA, UA | [33] |
| Au NPs-CNT-G-pMet-SPCE | 0.0029 | (h) urine | UA | [7] |
| AgNCs/AgNPs/GO | 0.00353 | brain homogenate of PD mice | GSH | [34] |
| GR/GLN | 0.0045 | (h) urine, serum | AA, UA, Glu | [35] |
| Fe3O4/rGO/GCE | 0.005 | (h) urine | UA, CA, Glu, AA, NaCl, AP | [36] |
| Fe3O4@GNs/Nafion/GCE | 0.00713 | (h) urine, plasma | AA, UA | [37] |
| graphene-MoS2/GCE | 0.007 | (b) serum | AA, UA, CA, Glu, cysteine, Na+, K+, Mg2+, Ca2+, Cl− | [38] |
| Fe3O4-SnO2-G/CPE | 0.0071 | (h) urine, serum | AA, UA | [39] |
| RGO/Mn-TPP/GCE | 0.008 | (h) serum | AA, UA | [40] |
| Ag NPs/GO/P(Arg)/GCE | 0.01 | (h) urine | U, CA, Glu, Na+, K+, L-lysine, L-cysteine | [41] |
| TiN-RGO/GCE | 0.012 | (h) urine | AA, UA, Glu, LA | [42] |
| PA/GO/GCE | 0.016 | (h) urine | AA, UA | [43] |
| GNCs/CMG/GCE | 0.02 | (h) serum | AA | [44] |
| Au–Pt/GO–ERGO | 0.0207 | (h) serum | AA, 5-HT, UA, AP, EP, NEP, CA, Glu, H2O2, NaCl, KCl, KNO3, Na2SO4, ZnCl2, CaCl2, (b) serum albumin, immunoglobulin | [45] |
| α-Fe2O3@erGO/GCE | 0.024 | (h) serum | AA, UA, Glu, U, H2O2, NaCl, KCl | [46] |
| CNDs-RGO/GCE | 0.03 | (h) serum | UA | [47] |
| Au-ZnO NCAs/GF | 0.04 | (h) urine | UA | [48] |
| Pt/rGO/MEA | 0.05 | (r) CPU | AA, UA, Glu, U, 5-HT, DOPAC | [49] |
| rGO–Cu2O/GCE | 0.05 | (h) urine, blood | AA, UA | [50] |
| PANI/Fe2O3-SnO2/rGO/PFSG/GCE | 0.076 | (a) urine | UA | [27] |
| PTPCNs/GCE | 0.078 | DA injection and urine | UA | [51] |
| ERGO/PLL/GCE | 0.10 | (h) urine | AA, UA | [52] |
| 3D-NG | 0.26 | (h) urine | AA, UA, AP | [53] |
| GO/Au NPs | 0.29 | (a) urine | UA, AA | [20] |
| AG-NA/GCE | 0.33 | (h) urine | AP | [54] |
| GO-BAMB-Co(OH)2 | 0.4 | (h) urine | AA, 5-HT | [55] |
| Pd-GR/nano-CILE | 0.5 | (h) urine, serum | UA | [56] |
| 3D HGB/ITO | 1.0 * | (h) plasma | UA | [57] |
| Pdop@GR/MWCNTs | 1.0 | (h) urine, serum | AA, UA | [58] |
| RGO–ZnO/GCE | 1.08 | (h) urine, plasma | AA, UA | [59] |
| Au/RGO/GCE | 1.4 | (r) serum | AA, UA, CA, NaCl, KCl, NaNO3, CaCl2, Glu, cysteine | [60] |
| mp-GR/GCE | 1.5 | (h) serum | UA | [61] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kujawska, M.; Bhardwaj, S.K.; Mishra, Y.K.; Kaushik, A. Using Graphene-Based Biosensors to Detect Dopamine for Efficient Parkinson’s Disease Diagnostics. Biosensors 2021, 11, 433. https://doi.org/10.3390/bios11110433
Kujawska M, Bhardwaj SK, Mishra YK, Kaushik A. Using Graphene-Based Biosensors to Detect Dopamine for Efficient Parkinson’s Disease Diagnostics. Biosensors. 2021; 11(11):433. https://doi.org/10.3390/bios11110433
Chicago/Turabian StyleKujawska, Małgorzata, Sheetal K. Bhardwaj, Yogendra Kumar Mishra, and Ajeet Kaushik. 2021. "Using Graphene-Based Biosensors to Detect Dopamine for Efficient Parkinson’s Disease Diagnostics" Biosensors 11, no. 11: 433. https://doi.org/10.3390/bios11110433
APA StyleKujawska, M., Bhardwaj, S. K., Mishra, Y. K., & Kaushik, A. (2021). Using Graphene-Based Biosensors to Detect Dopamine for Efficient Parkinson’s Disease Diagnostics. Biosensors, 11(11), 433. https://doi.org/10.3390/bios11110433








