Development of an Electrochemical Immunosensor for Specific Detection of Visceral Leishmaniasis Using Gold-Modified Screen-Printed Carbon Electrodes
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Biomolecules
2.2. Devices
2.3. Indirect ELISA
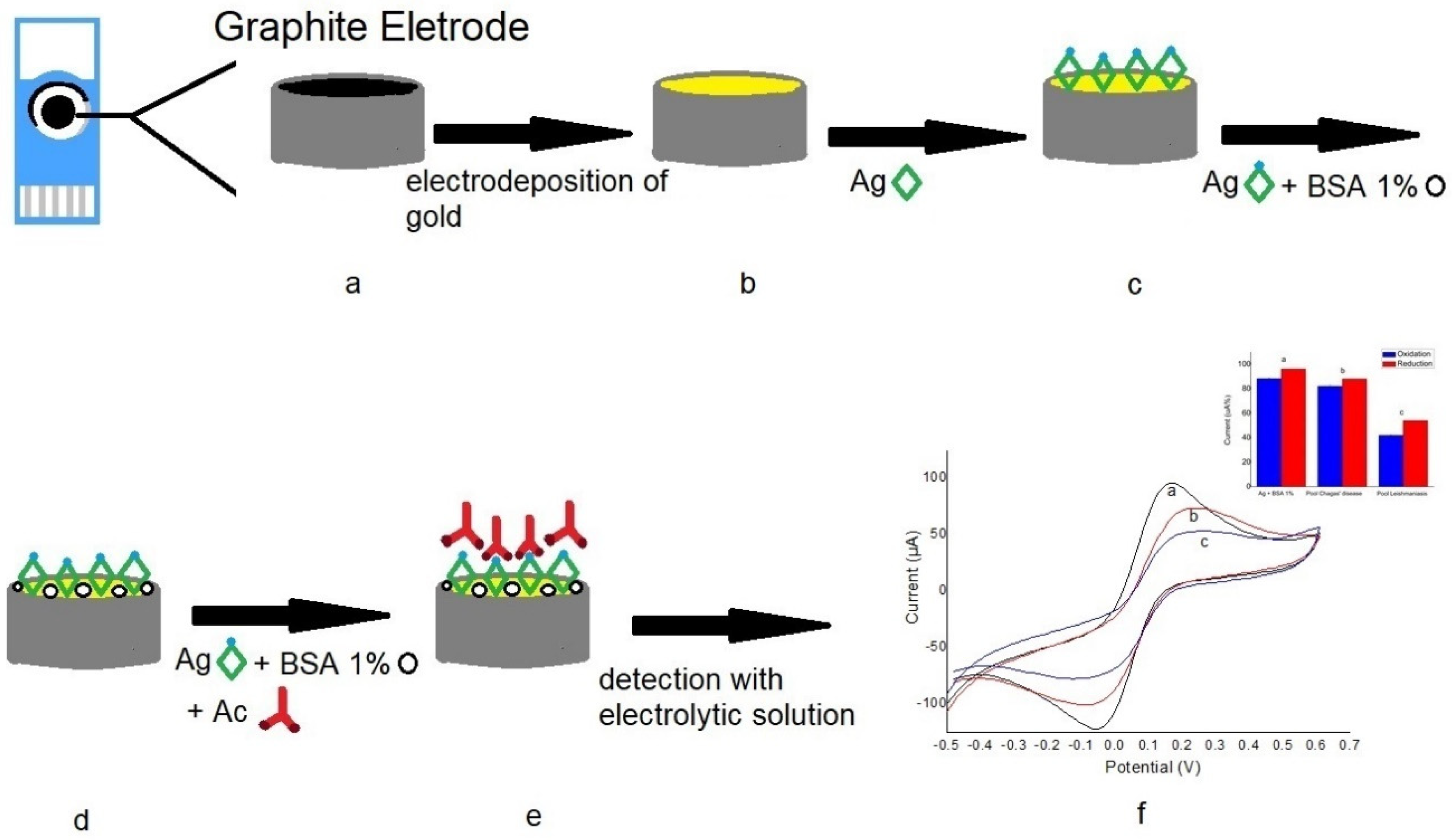
2.4. Electrodeposition of Gold Nanoparticles on Carbon Electrodes
2.5. Immunosensor
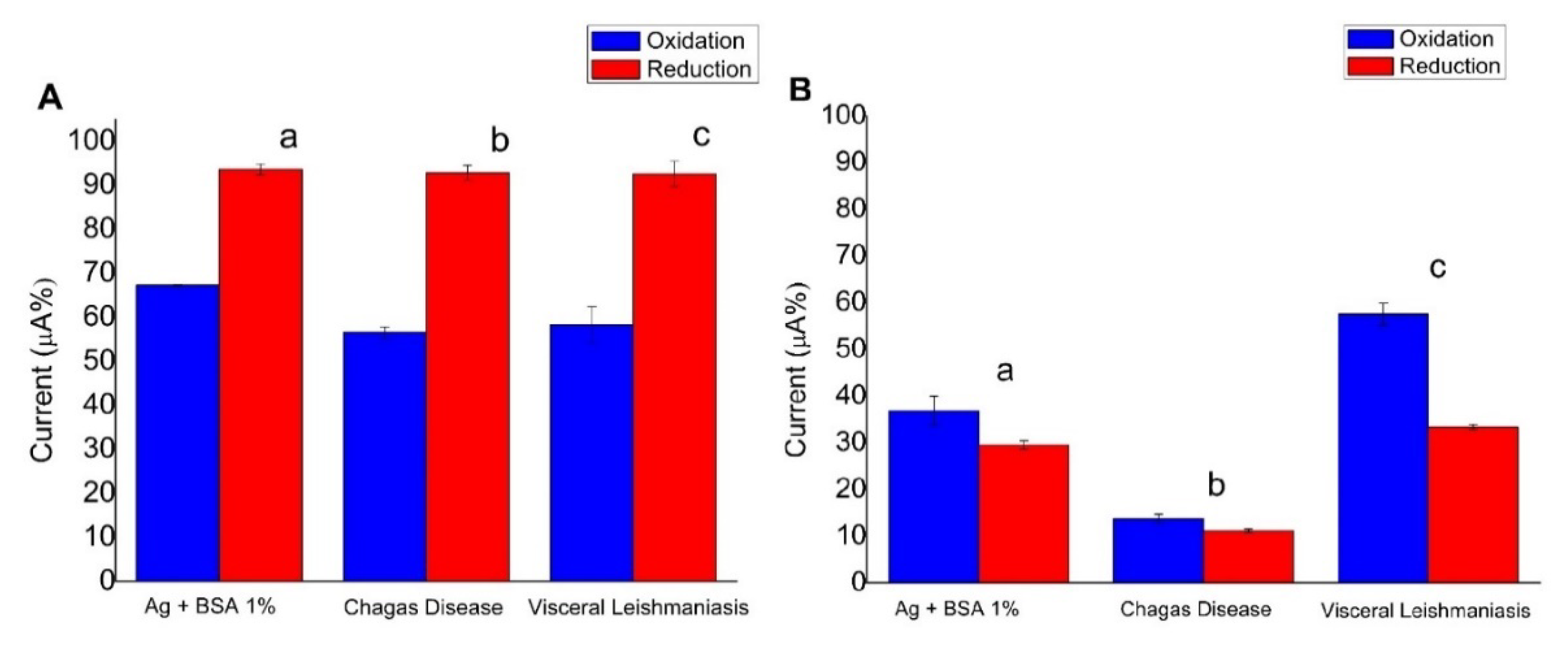
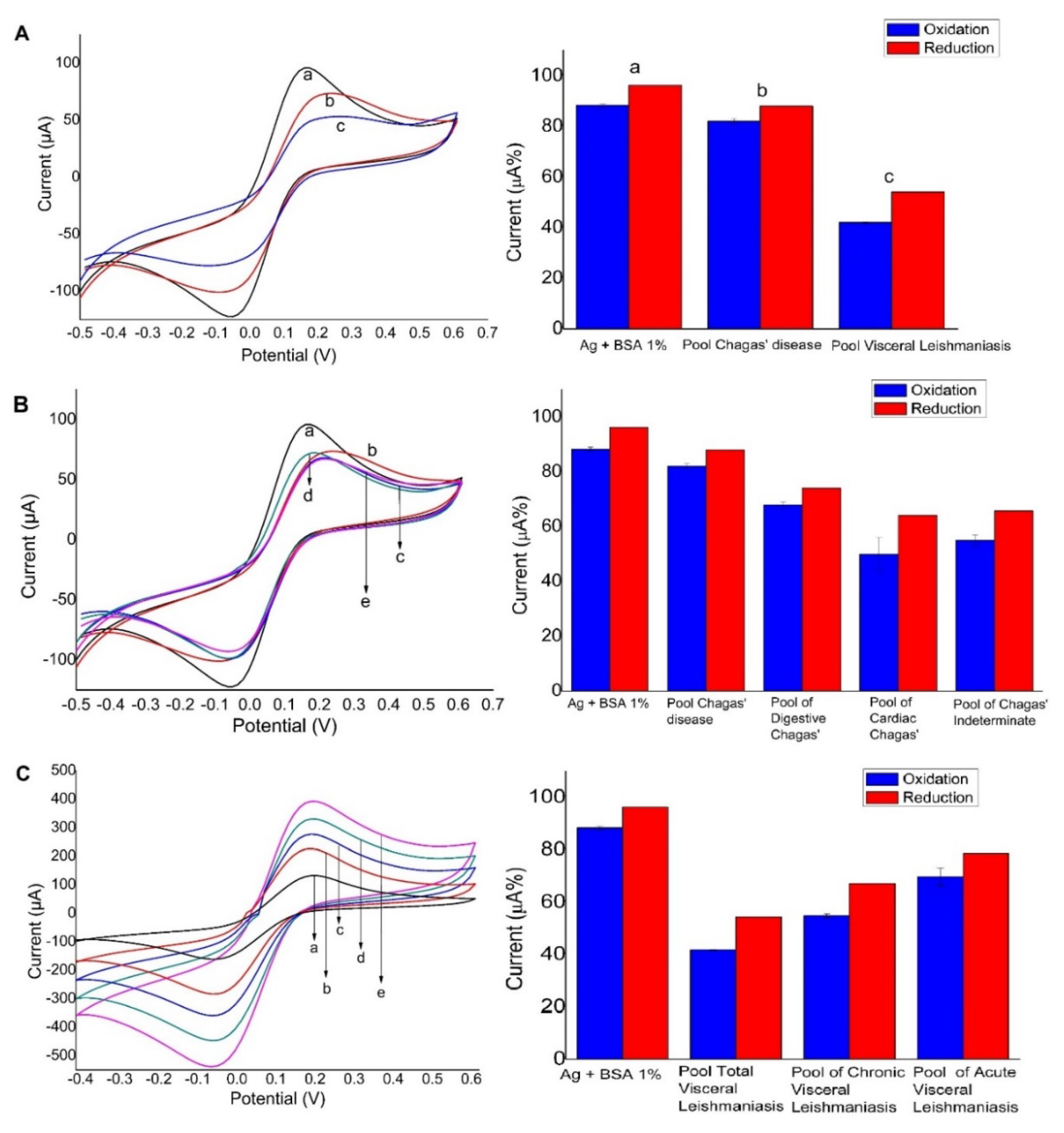
2.6. Specificity
2.7. Sensor Stability
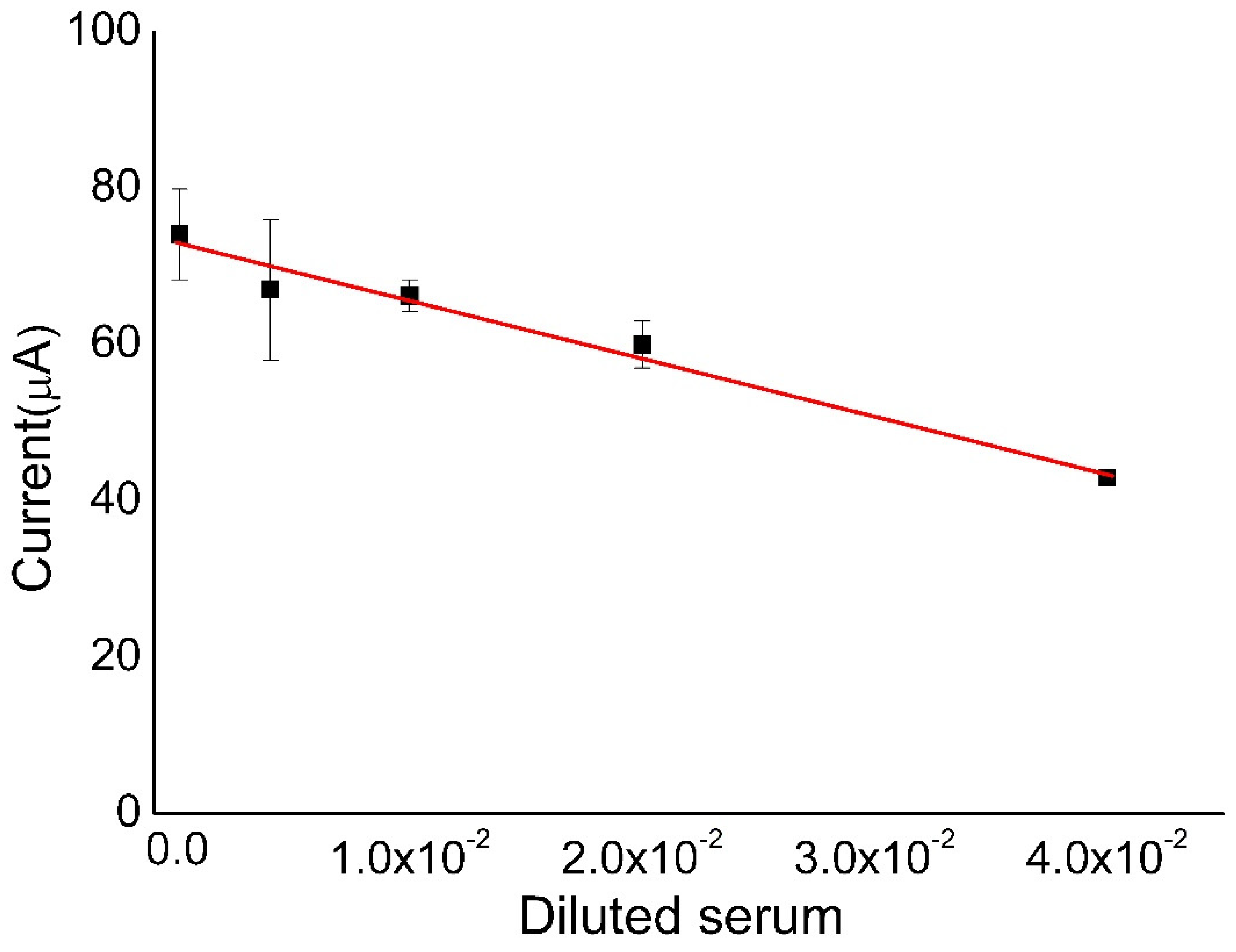
2.8. Calibration Curve
2.9. Statistical Analysis
3. Results and Discussion
3.1. Screen-Printed Electrode
3.2. Gold-Modified Electrode Used as a Platform
3.3. Stability
3.4. Calibration Curve
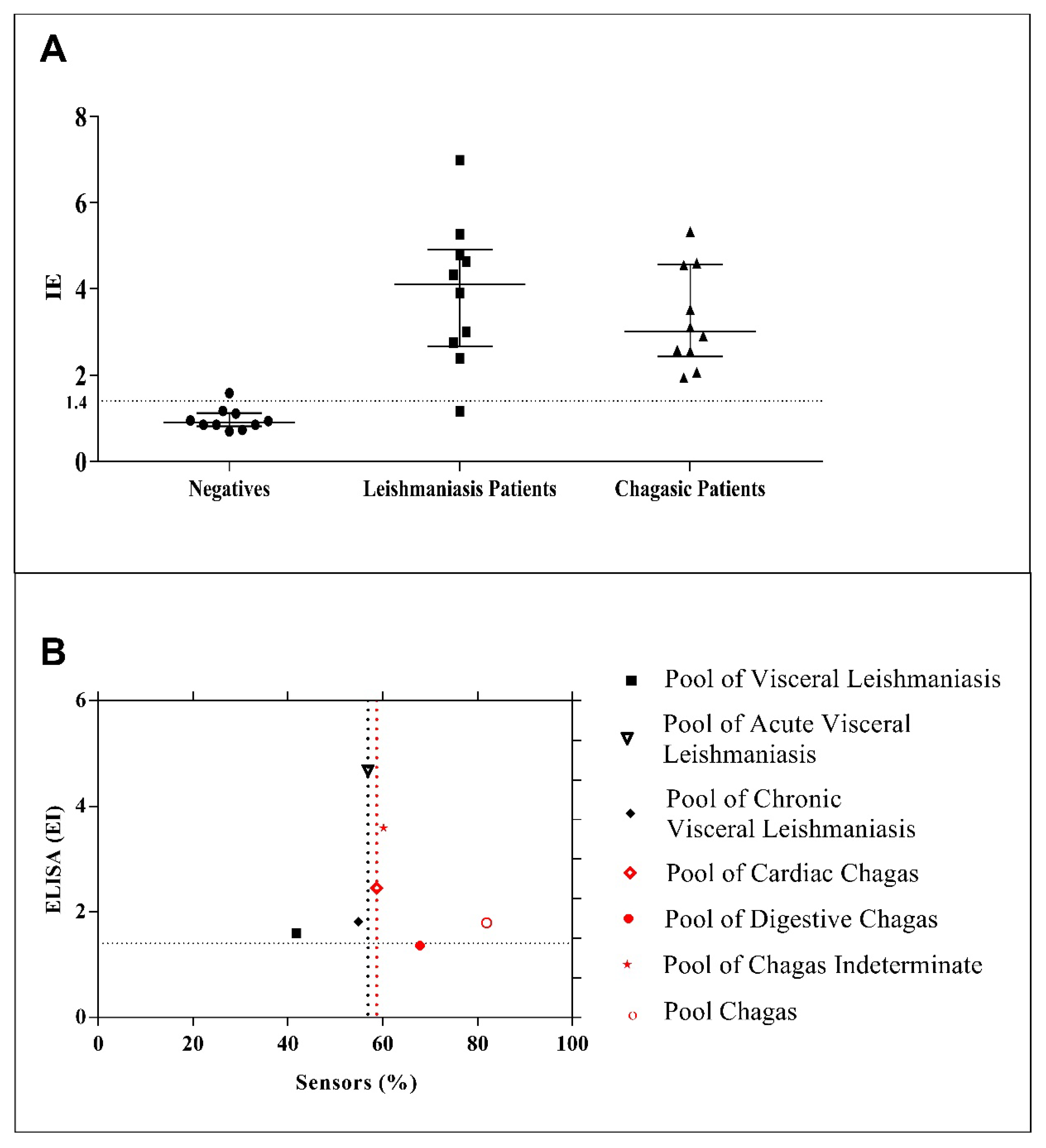
3.5. Comparison with Indirect ELISA
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Convit, J.; Ulrich, M.; Fernandez, C.T.; Tapia, F.J.; Caceres-Dittmar, G.; Castes, M.; Rondon, A.J. The clinical and immunological spectrum of American cutaneous leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 1993, 87, 444–448. [Google Scholar] [CrossRef]
- Castellano, L.R.; Correia-Filho, D.; Agiro, L.; Dessein, H.; Prata, A.; Dessein, A.; Rodrigues, V. Th1/Th2 immune responses are associated with active cutaneous leishmaniasis and clinical cure is associated with strong interferon-γ production. Hum. Immonol. 2009, 70, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Cabeza, B.; Cecílio, P.; Robalo, A.L.; Silvestre, R.; Carrilho, E.; Moreno, J.; San Martin, J.V.; Vasconcellos, R.; Cordeiro-da -Silva, A. Interleukin-27 Early Impacts Leishmania infantum Infection in Mice and Correlates with Active Visceral Disease in Humans. Front Immunol. 2016, 7, 478. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Rebolledo, G.A.; Drier-Jonas, S.; Jiménez-Arellanes, M.A. Natural compounds and extracts from Mexican medicinal plants with anti-leishmaniasis activity: An update. Asian Pac. J. Trop. Med. 2017, 10, 1105–1110. [Google Scholar] [CrossRef]
- Gollob, K.J.; Viana, A.G.; Dutra, W.O. Immunoregulation in human American leishmaniasis: Balancing pathology and protection. Parasite Immunol. 2014, 36, 367–376. [Google Scholar] [CrossRef]
- Matos, H.J.d.; Pinto, A.Y.d.N.; Miranda, A.M.M.; Silva, F.L.C.; Ramos, F.L.P. Cross-reactivity in serological tests between Chagas disease and visceral leishmaniasis in endemic regions for both diseases. Rev. Pan-Amaz. Saúde 2015, 6, 65–68. [Google Scholar] [CrossRef]
- Pan American Health Organization, Key facts on Neglected Infectious Diseases. Leishmaniasis. 2017. Available online: https://www.paho.org/hq/dmdocuments/2017/2017-cha-leishmaniasis-factsheet-work.pdf (accessed on 12 June 2020).
- Freire, M.L.; Machado de Assis, T.; Oliveira, E.; Moreira de Avelar, D.; Siqueira, I.C.; Barral, A.; Rabello, A.; Cota, G. Performance of serological tests available in Brazil for the diagnosis of human visceral leishmaniasis. PLoS Negl. Trop. Dis. 2019, 13, e0007484. [Google Scholar] [CrossRef]
- Lemos, A.J.G.; Balvedi, R.P.A.; Rodovalho, V.R.; Resende, L.O.; Castro, A.C.H.; Cuadros-Orellana, S.; Madurro, J.M.; Brito-Madurro, A.G. Immunosensor assembled on polymeric nanostructures for clinical diagnosis of C-reactive protein. Microchem. J. 2017, 133, 572–576. [Google Scholar] [CrossRef]
- Pereira, S.V.; Bertolino, F.A.; Fernández-Baldo, M.A.; Messina, G.A.; Salinas, E.; Sanz, M.I.; Raba, J. A microfluidic device based on a screen-printed carbon electrode with electrodeposited gold nanoparticles for the detection of IgG anti-Trypanosoma cruzi antibodies. Analyst 2011, 136, 4745–4751. [Google Scholar] [CrossRef]
- Moreno, E.C.; Gonçalves, A.V.; Chaves, A.V.; Melo, M.N.; Lambertucci, J.R.; Andrade, A.S.R.; Negrão-Corrêa, D.; Antunes, C.M.D.F.; Carneiro, M. Inaccuracy of enzyme-linked immunosorbent assay using soluble and recombinant antigens to detect asymptomatic infection by Leishmania infantum. PLoS Negl. Trop. Dis. 2009, 3, 1–7. [Google Scholar] [CrossRef]
- Balvedi, R.P.A.; Castro, A.C.H.; Madurro, J.M.; Brito-Madurro, A.G. Detection of a specific biomarker for Epstein-Barr virus using a polymer-based genosensor. Int. J. Mol. Sci. 2014, 15, 9051–9066. [Google Scholar] [CrossRef] [PubMed]
- Moradi, M.; Sattarahmady, N.; Rahi, A.; Hatam, G.R.; Sorkhabadi, S.M.R.; Heli, H. A label-free, PCR-free and signal-on electrochemical DNA biosensor for Leishmania major based on gold nanoleaves. Talanta 2016, 161, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, H.; Gartzonika, C.; Levidiotou, S. Laboratory diagnosis of human visceral leishmaniasis. J. Vector Borne Dis. 2016, 53, 8–16. [Google Scholar] [PubMed]
- Luciano, R.M.; Lucheis, S.B.; Troncarelli, M.Z.; Luciano, D.M.; Langoni, H. Leishmania sp. And Trypanossoma cruzi antigen cross reaction evaluation in dogs by indirect immunofluorescence diagnosis (RIIF). Braz. J. Vet. Res. Anim. Sci. 2009, 46, 181–187. [Google Scholar] [CrossRef][Green Version]
- Burns, J.M.; Shreffler, W.G.; Benson, D.R.; Ghalib, H.W.; Badaro, R.; Reed, S.G. Molecular characterization of a kinesin-related antigen of Leishmania Chagasic that detects specific antibody in African and American visceral leishmaniasis. Proc. Natl. Acad. Sci. USA 1993, 90, 775–779. [Google Scholar] [CrossRef] [PubMed]
- Maia, Z.; Lírio, M.; Mistro, S.; Mendes, C.M.C.; Mehta, S.R.; Badaro, R. Comparative study of rK39 Leishmania antigen for serodiagnosis of visceral leishmaniasis: Systematic review with meta-analysis. PLoS Negl. Trop. Dis. 2012, 6, e1484. [Google Scholar] [CrossRef]
- Saghrouni, F.; Gaïed-Meksi, S.; Fathallah, A.; Amri, F.; Ach, H.; Guizani, I.; Saïd, M. Ben Immunochromatographic rK39 strip test in the serodiagnosis of visceral leishmaniasis in Tunisia. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 1273–1278. [Google Scholar] [CrossRef]
- Segatto, M.; Ribeiro, L.S.; Costa, D.L.; Costa, C.H.N.; de Oliveira, M.R.; Carvalho, S.F.G.; Macedo, A.M.; Valadares, H.M.S.; Dietze, R.; de Brito, C.F.A.; et al. Genetic diversity of Leishmania infantum field populations from Brazil. Mem. Inst. Oswaldo Cruz 2012, 107, 39–47. [Google Scholar] [CrossRef][Green Version]
- Arraes, S.M.A.A.; Marini, M.T.; Martello, D.; Silveira, T.G.V.; Lonardoni, M.V.C.; Nanni, M.R. Serological investigation of subclinical cutaneous leishmaniasis cases following an outbreak in an endemic area. Rev. Soc. Bras. Med. Trop. 2008, 41, 205–208. [Google Scholar] [CrossRef][Green Version]
- Sato, C.M.; Sanchez, M.C.A.; Celeste, B.J.; Duthie, M.S.; Guderian, J.; Reed, S.G.; De Brito, M.E.F.; Campos, M.B.; De Souza Encarnação, H.V.; Guerra, J.; et al. Use of recombinant antigens for sensitive serodiagnosis of American tegumentary leishmaniasis caused by different leishmania species. J. Clin. Microbiol. 2017, 55, 495–503. [Google Scholar] [CrossRef]
- Ferreira, M.P.; Roselino, A.M.F.; Nascimento, M.M.P.; Machado Aires, J.; Figueiredo, J.F.C. Sensitivity of an immunoenzymatic test for the detection of anti-l. braziliensis antibodies compared to other tests used for the diagnosis of american cutaneous leishmaniasis. Rev. Inst. Med. Trop. S. Paulo 2006, 48, 1–3. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Laurenti, M. Correlation between parasitological and serological diagnosis in canine american visceral leishmaniasis. BEPA Bol. Epidemiológico Paul. 2009, 6, 13–23. [Google Scholar]
- Pereira, A.C.; Santos, A.D.S.; Kubota, L.T. Trends in amperometrics electrodes modification for electroanalytical applications. Quim. Nova 2002, 25, 1012–1021. [Google Scholar]
- Freire, R.S.; Pessoa, C.A.; Kubota, L.T. Self-assembled monolayers applications for the development of electrochemical sensors. Quim. Nova 2003, 26, 381–389. [Google Scholar] [CrossRef][Green Version]
- Fonseca, R.A.S.; Ramos-Jesus, J.; Kubota, L.T.; Dutra, R.F. A nanostructured piezoelectric immunosensor for detection of human cardiac troponin T. Sensors 2011, 11, 10785–10797. [Google Scholar] [CrossRef]
- Pérez-Fernández, B.; Costa-García, A.; Muñiz, A.D.L.E. Electrochemical (Bio)Sensors for Pesticides Detection Using Screen-Printed Electrodes. Biosensors 2020, 10, 32–58. [Google Scholar]
- Shkodra, B.; Demelash Abera, B.; Cantarella, G.; Douaki, A.; Avancini, E.; Petti, L.; Lugli, P. Flexible and Printed Electrochemical Immunosensor Coated with Oxygen Plasma Treated SWCNTs for Histamine Detection. Biosensors 2020, 10, 35. [Google Scholar] [CrossRef]
- Cancelliere, R.; Carbone, K.; Pagano, M.; Cacciotti, I.; Micheli, L. Biochar from brewers’ spent grain: A green and low-cost smart material to modify screen-printed electrodes. Biosensors 2019, 9, 139. [Google Scholar] [CrossRef]
- Chiticaru, E.A.; Pilan, L.; Damian, C.M.; Vasile, E.; Burns, J.S.; Ionita, M. Influence of graphene oxide concentration when fabricating an electrochemical biosensor for DNA detection. Biosensors 2019, 9, 113. [Google Scholar] [CrossRef]
- Martínez-García, G.; Pérez-Julián, E.; Agüí, L.; Cabré, N.; Joven, J.; Yáñez-Sedeño, P.; Pingarrón, J.M. An electrochemical enzyme biosensor for 3-hydroxybutyrate detection using screen-printed electrodes modified by reduced graphene oxide and thionine. Biosensors 2017, 7, 50. [Google Scholar] [CrossRef]
- Ahmad, N.M.; Abdullah, J.; Yusof, N.A.; Ab Rashid, A.H.; Rahman, S.A.; Hasan, M.R. Amperometric biosensor based on zirconium oxide/polyethylene glycol/tyrosinase composite film for the detection of phenolic compounds. Biosensors 2016, 6, 31. [Google Scholar] [CrossRef]
- Badalyan, A.; Dierich, M.; Stiba, K.; Schwuchow, V.; Leimkühler, S.; Wollenberger, U. Electrical wiring of the aldehyde oxidoreductase PaoABC with a polymer containing osmium redox centers: Biosensors for benzaldehyde and GABA. Biosensors 2014, 4, 403–421. [Google Scholar] [CrossRef] [PubMed]
- Setterington, E.B.; Alocilja, E.C. Electrochemical biosensor for rapid and sensitive detection of magnetically extracted bacterial pathogens. Biosensors 2012, 2, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Rama, E.C.; Costa-García, A. Screen-printed Electrochemical Immunosensors for the Detection of Cancer and Cardiovascular Biomarkers. Electroanalysis 2016, 28, 1700–1715. [Google Scholar] [CrossRef]
- Mohan, S.; Srivastava, P.; Maheshwari, S.N.; Sundar, S.; Prakash, R. Nano-structured nickel oxide-based DNA biosensor for detection of visceral leishmaniasis (Kala-azar). Analyst 2011, 136, 2845–2851. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Matharu, Z.; Sumana, G.; Solanki, P.R.; Kim, C.G.; Malhotra, B.D. Antibody immobilized cysteamine functionalized-gold nanoparticles for aflatoxin detection. Thin Solid Film. 2010, 519, 1213–1218. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Gontijo, C.M.F.; Melo, M.N. Visceral Leishmaniasis in Brazil: Current status, challenges, and prospects. Rev. Bras. Epidemiol. 2004, 7, 338–349. [Google Scholar] [CrossRef]
- Scott, P.; Pearce, E.; Natovitz, P.; Sher, A. Vaccination against cutaneous leishmaniasis in a murine model. I. Induction of protective immunity with a soluble extract of promastigotes. J. Immunol. 1987, 139, 221–227. [Google Scholar]
- Dai, X.; Nekraseova, O.; Hyde, M.E.; Compton, R.G. Anodic stripping voltammetry of arsenic (III) using gold nanoparticle-modified electrodes. Anal. Chem. 2004, 76, 5924–5929. [Google Scholar] [CrossRef]
- Chikae, M.; Fukuda, T.; Kerman, K.; Idegami, K.; Miura, Y.; Tamiya, E. Amyloid-β detection with saccharide immobilized gold nanoparticle on carbon electrode. Bioelectrochemistry 2008, 74, 118–123. [Google Scholar] [CrossRef]
- Regiart, M.; Pereira, S.V.; Bertolino, F.A.; Garcia, C.D.; Raba, J.; Aranda, P.R. An electrochemical immunosensor for anti-T. cruzi IgM antibodies, a biomarker for congenital Chagas disease, using a screen-printed electrode modified with gold nanoparticles and functionalized with shed acute phase antigen. Microchim. Acta 2016, 183, 1203–1210. [Google Scholar] [CrossRef]
- Bollella, P.; Gorton, L.; Ludwig, R.; Antiochia, R. A third-generation glucose biosensor based on cellobiose dehydrogenase immobilized on a glassy carbon electrode decorated with electrodeposited gold nanoparticles: Characterization and application in human saliva. Sensors 2017, 17, 1912. [Google Scholar] [CrossRef] [PubMed]
- Almeida, E.S.; Richter, E.M.; Munoz, R.A.A. On-site fuel electroanalysis: Determination of lead, copper and mercury in fuel bioethanol by anodic stripping voltammetry using screen-printed gold electrodes. Anal. Chim. Acta 2014, 837, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, S.M.; Squissato, A.L.; Rocha, D.P.; Vasconcellos, M.L.S.; Ferreira, R.D.; Richter, E.M.; Munoz, R.A.A. Improved anodic stripping voltammetric detection of zinc on a disposable screen-printed gold electrode. Ionics 2020, 26, 2611–2621. [Google Scholar] [CrossRef]
- Vasconcellos, M.L.S.; Rocha, D.P.; Castro, S.V.F.; Silva, L.R.G.; Munoz, R.A.A.; Freitas, M.B.J.G.; Ferreira, R.Q. Electroanalytical Method for Determination of Trace Metals in Struvite Using Electrochemically Treated Screen-Printed Gold Electrodes. J. Braz. Chem. Soc. 2020, 31, 1873–1882. [Google Scholar] [CrossRef]
- Cordeiro, T.A.R.; Gonçalves, M.V.C.; Franco, D.L.; Reis, A.B.; Martins, H.R.; Ferreira, L.F. Label-free electrochemical impedance immunosensor based on modified screen-printed gold electrodes for the diagnosis of canine visceral leishmaniasis. Talanta 2019, 195, 327–332. [Google Scholar] [CrossRef]
- Janegitz, B.C.; Medeiros, R.A.; Rocha-Filho, R.C.; Fatibello-Filho, O. Direct electrochemistry of tyrosinase and biosensing for phenol based on gold nanoparticles electrodeposited on a boron-doped diamond electrode. Diam. Relat. Mater. 2012, 25, 128–133. [Google Scholar] [CrossRef]
- Chu, X.; Zhao, Z.L.; Shen, G.L.; Yu, R.Q. Quartz crystal microbalance immunoassay with dendritic amplification using colloidal gold immunocomplex. Sens. Actuators B Chem. 2006, 114, 696–704. [Google Scholar] [CrossRef]
- Godínez, L.A. Substratos modificados con monocapas autoensambladas: Dispositivos para fabricar sensores y estudiar procesos químicos y fisicoquímicos interfaciales. J. Mex. Chem. Soc. 1999, 43, 219–229. [Google Scholar]
- Galal, A.; Atta, N.F.; El-Ads, E.H. Probing cysteine self-assembled monolayers over gold nanoparticles-Towards selective electrochemical sensors. Talanta 2012, 93, 264–273. [Google Scholar] [PubMed]
- Chaki, N.K.; Vijayamohanan, K. Self-assembled monolayers as a tunable platform for biosensor applications. Biosens. Bioelectron. 2002, 17, 1–12. [Google Scholar] [CrossRef]
- Gómez-Velasco, A.; León-Cortés, J.L.; Gordillo-Marroquín, C.; Sánchez-Pérez, H.J.; Alocilja, E.C.; Muñoz-Jiménez, S.G.; Enríquez-Ríos, N.; Bencomo-Alerm, A.; Jonapá-Gómez, L.; Gómez-Bustamante, A. Use of magnetic nanoparticles and a biosensor for the diagnosis and monitoring of emergent, re-emergent and neglected tropical infectious diseases. Rev. Enf. Emerg. 2019, 18, 23–31. [Google Scholar]
- Cios, K.J.; William Moore, G.; Moore, G.W. Unique features of medical data mining and knowledge discovery; Ethical; Security and legal aspects of medical data mining. Artif. Intell. Med. 2002, 26, 1–24. [Google Scholar] [CrossRef]
- Ortolani, C.; Ispano, M.; Pastorello, E.A.; Ansaloni, R.; Magri, G.C. Comparison of results of skin prick tests (with fresh foods and commercial food extracts) and RAST in 100 patients with oral allergy syndrome. J. Allergy Clin. Immunol. 1989, 83, 683–690. [Google Scholar] [CrossRef]
- Assis, T.S.M.; Azevedo-da-Silva, A.L.F.; Oliveira, D.; Cota, G.; Werneck, G.L.; Rabello, A. Budgetary impact of diagnostic tests for visceral leishmaniasis in Brazil. Cad. Saúde Publica 2017, 33, 1–8. [Google Scholar] [CrossRef][Green Version]
- Faria, A.R.; de Andrade, H.M. Diagnosis of canine visceral leishmaniasis: Major technological advances and few practical applications. Rev. Pan-Amaz Saúde 2012, 3, 47–57. [Google Scholar] [CrossRef]
| Method | Material | Sensitivity/Specificity | Reference |
|---|---|---|---|
| IFAT | Serum | 88–92%/83–88% | [57] |
| Kalazar Detect | Serum | 84–88.1%/91% | [57] |
| IT LEISHBio-Rad | Blood/Serum | 92–93%/92–98% | [57] |
| PCR | Blood | 93%/96% | [8,57,58] |
| DAT-LPC | Blood | 99%/98% | [57] |
| RIFI | Serum | 0–100%/80% | [9,10] |
| Immunochromatographic tests | Serum | 87%/94% | [11,12,13] |
| ELISA | Serum | 80–99%/81–100% | [14,15,16,17,18] |
| Electrochemical | DNA extracted of blood | Not explained | [22] |
| ELISA | Serum | 92.3%/44% | This work |
| Electrochemical | Serum | 100%/100% | This work |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, B.R.; Barbosa, Y.O.; Andrade, C.M.R.; Pereira, L.Q.; Simão, G.F.; de Oliveira, C.J.; Correia, D.; Oliveira, R.T.S., Jr.; da Silva, M.V.; Silva, A.C.A.; et al. Development of an Electrochemical Immunosensor for Specific Detection of Visceral Leishmaniasis Using Gold-Modified Screen-Printed Carbon Electrodes. Biosensors 2020, 10, 81. https://doi.org/10.3390/bios10080081
Martins BR, Barbosa YO, Andrade CMR, Pereira LQ, Simão GF, de Oliveira CJ, Correia D, Oliveira RTS Jr., da Silva MV, Silva ACA, et al. Development of an Electrochemical Immunosensor for Specific Detection of Visceral Leishmaniasis Using Gold-Modified Screen-Printed Carbon Electrodes. Biosensors. 2020; 10(8):81. https://doi.org/10.3390/bios10080081
Chicago/Turabian StyleMartins, Beatriz R., Yanne O. Barbosa, Cristhianne M. R. Andrade, Loren Q. Pereira, Guilherme F. Simão, Carlo J. de Oliveira, Dalmo Correia, Robson T. S. Oliveira, Jr., Marcos V. da Silva, Anielle C. A. Silva, and et al. 2020. "Development of an Electrochemical Immunosensor for Specific Detection of Visceral Leishmaniasis Using Gold-Modified Screen-Printed Carbon Electrodes" Biosensors 10, no. 8: 81. https://doi.org/10.3390/bios10080081
APA StyleMartins, B. R., Barbosa, Y. O., Andrade, C. M. R., Pereira, L. Q., Simão, G. F., de Oliveira, C. J., Correia, D., Oliveira, R. T. S., Jr., da Silva, M. V., Silva, A. C. A., Dantas, N. O., Rodrigues, V., Jr., Muñoz, R. A. A., & Alves-Balvedi, R. P. (2020). Development of an Electrochemical Immunosensor for Specific Detection of Visceral Leishmaniasis Using Gold-Modified Screen-Printed Carbon Electrodes. Biosensors, 10(8), 81. https://doi.org/10.3390/bios10080081









