Self-assembly Synthesis of Molecularly Imprinted Polymers for the Ultrasensitive Electrochemical Determination of Testosterone
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Synthesis and Characterization of Testosterone-imprinted Electronically Conductive Polymer (TIECP) films
2.3. Electrochemical Characterization of TIECP-coated Electrodes
2.4. The Determination of Testosterone in Human Urine Samples
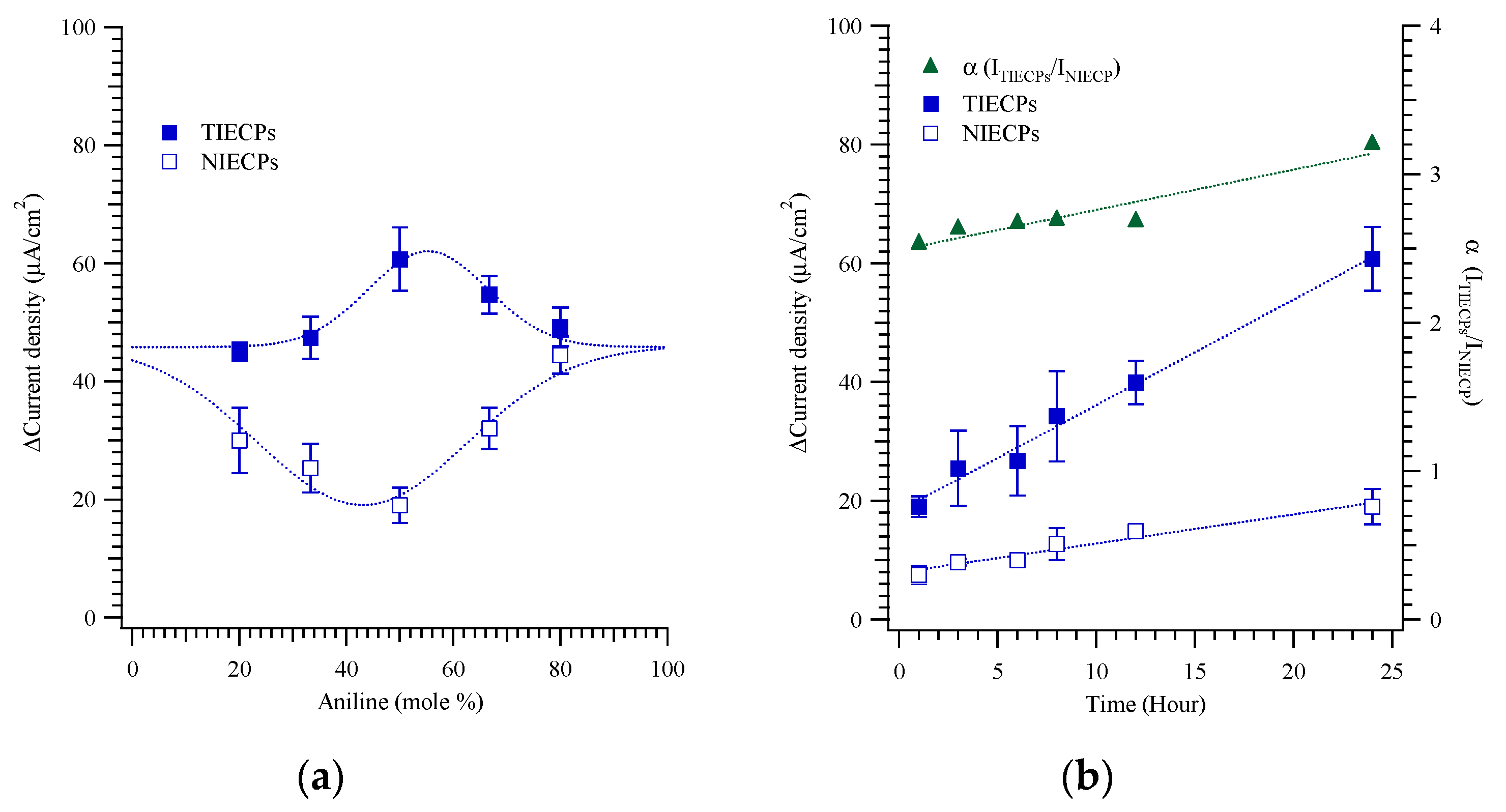
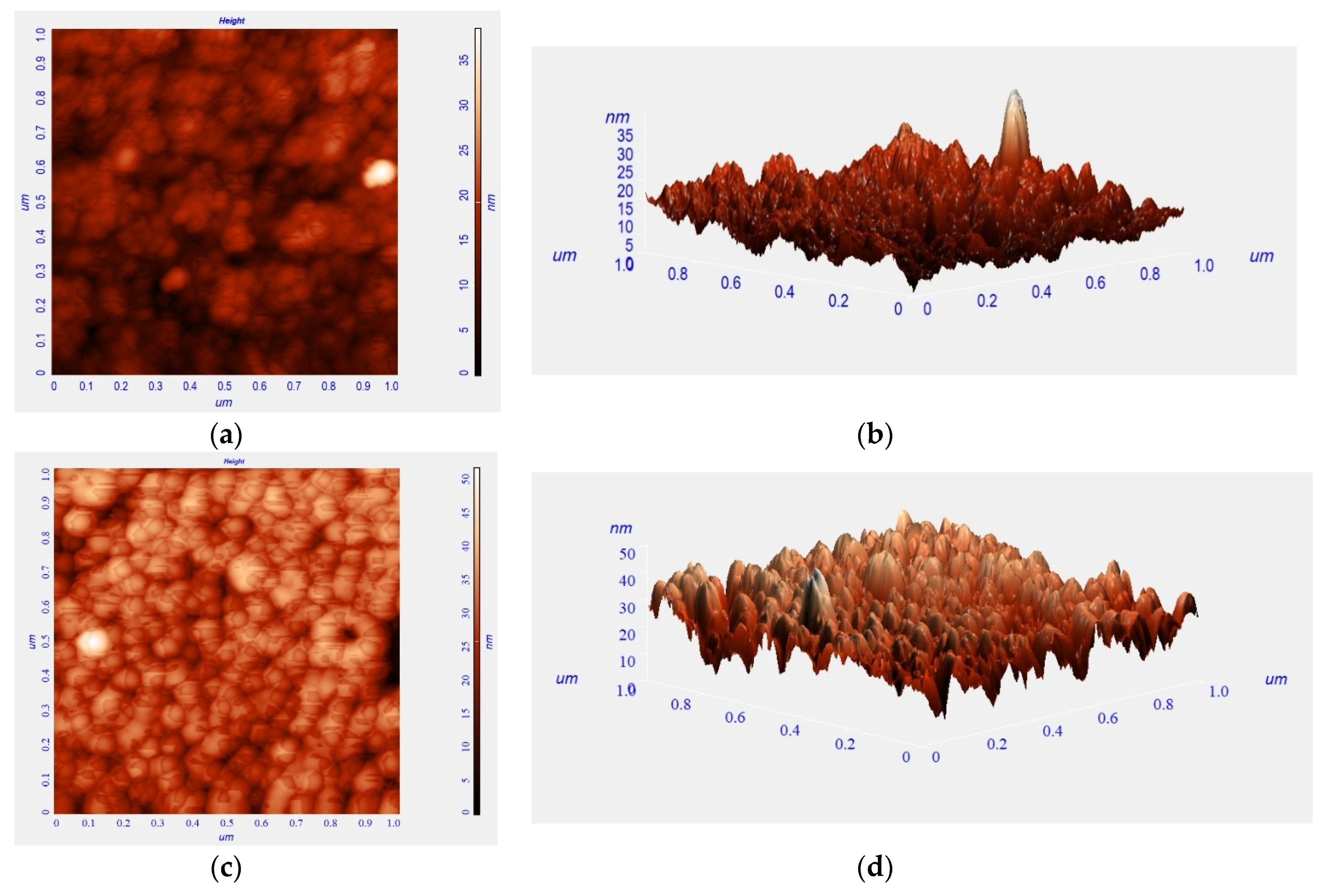
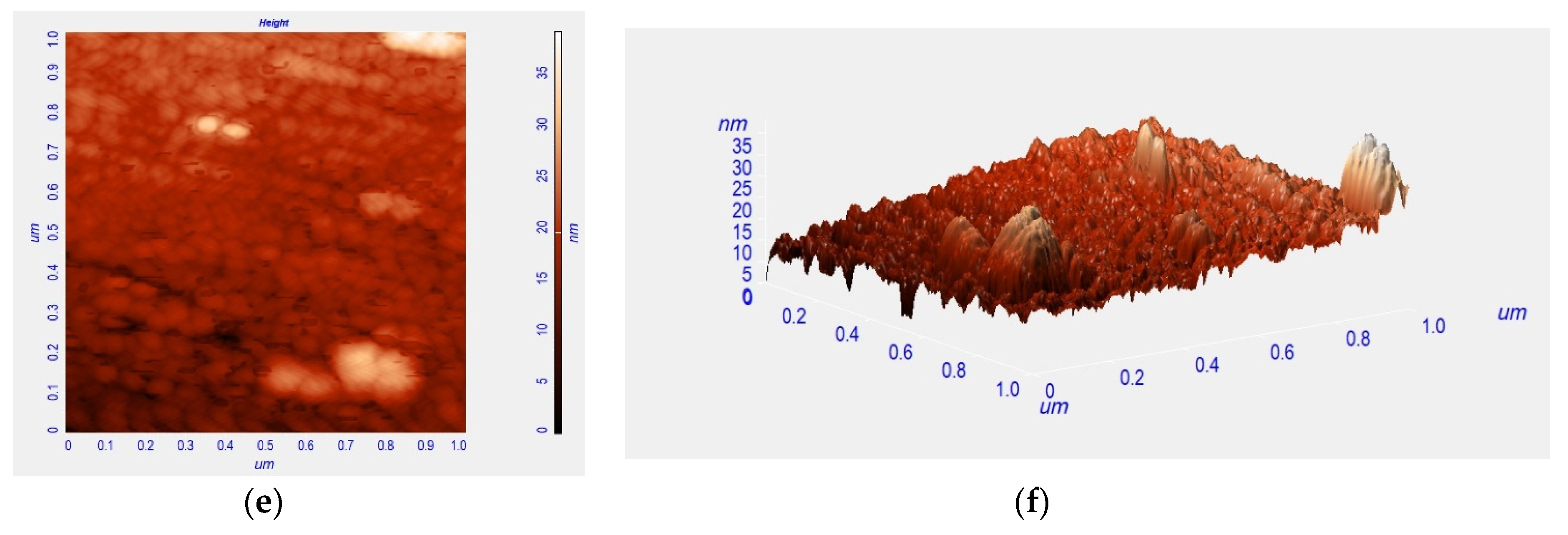
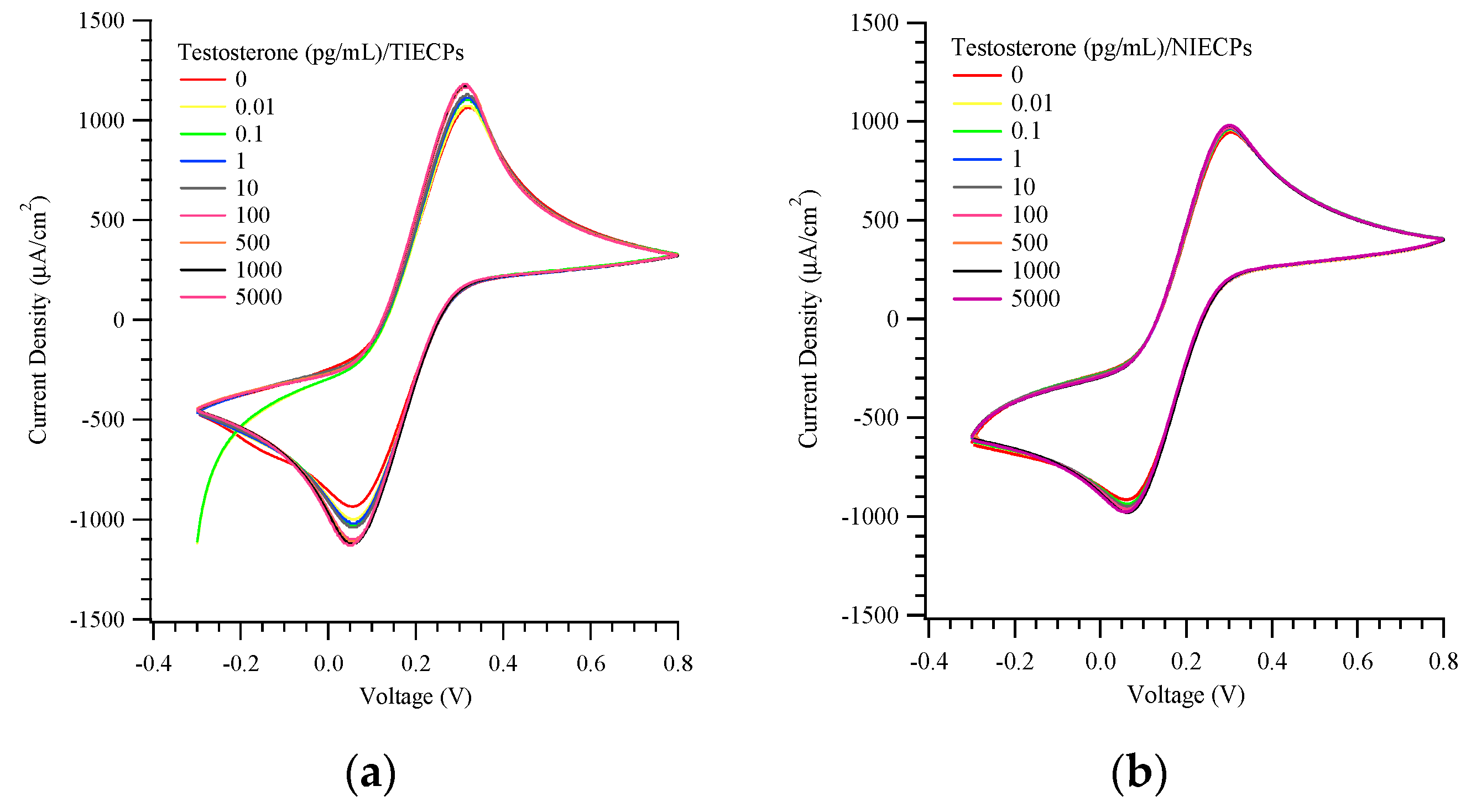
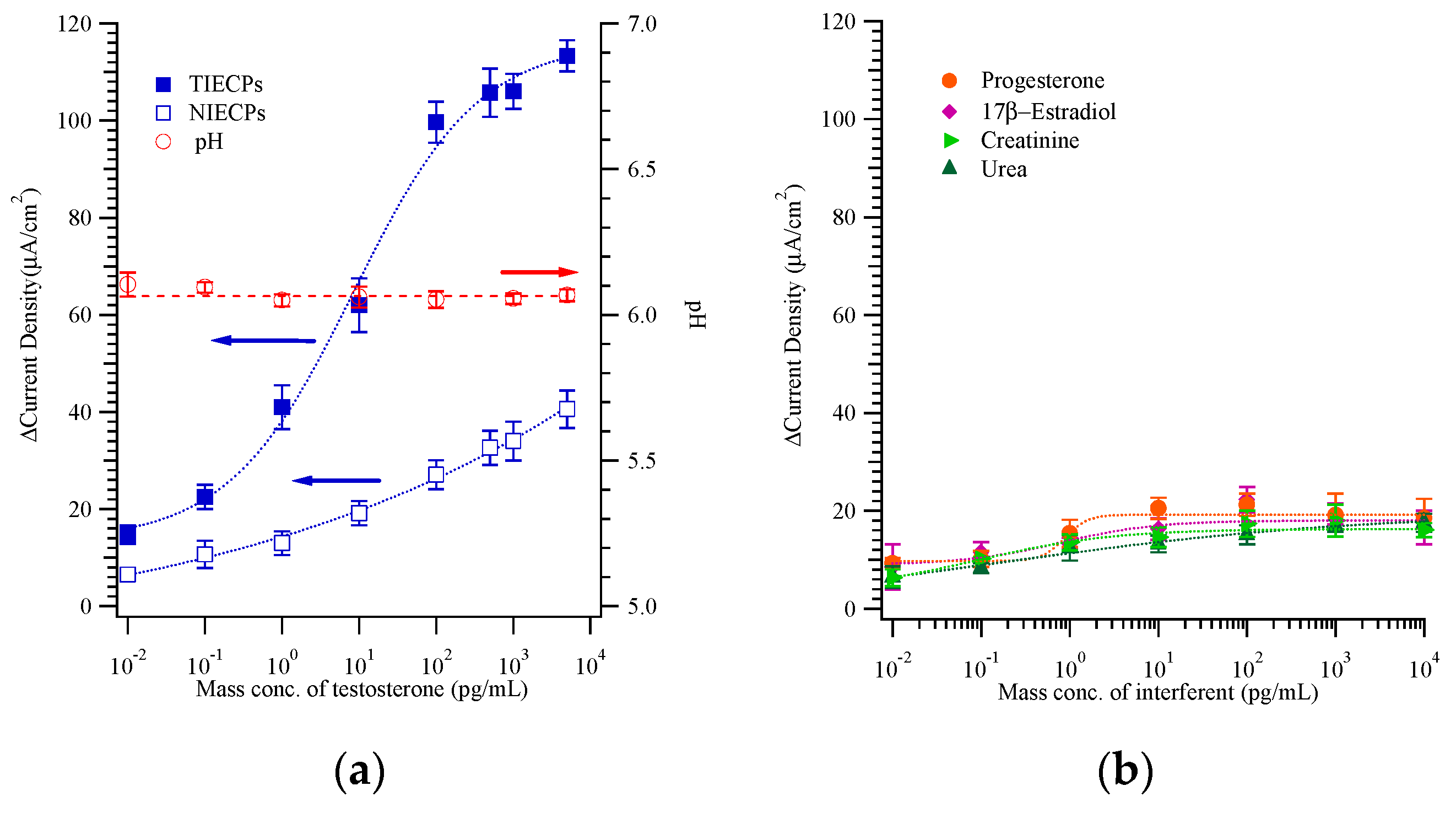
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Feldman, H.A.; Longcope, C.; Derby, C.A.; Johannes, C.B.; Araujo, A.B.; Coviello, A.D.; Bremner, W.J.; McKinlay, J.B. Age trends in the level of serum testosterone and other hormones in middle-aged men: Longitudinal results from the massachusetts male aging study. J. Clin. Endocrinol. Metab. 2002, 87, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, S.; Storer, T.W.; Javanbakht, M.; Berman, N.; Yarasheski, K.E.; Phillips, J.; Dike, M.; Indrani, S.-H.; Shen, R.; Hays, R.D.; et al. Testosterone replacement and resistance exercise in hiv-infected men with weight loss and low testosterone levels. JAMA 2000, 283, 763–770. [Google Scholar] [CrossRef]
- Carrero, J.J.; Qureshi, A.R.; Parini, P.; Arver, S.; Lindholm, B.; Bárány, P.; Heimbürger, O.; Stenvinkel, P. Low serum testosterone increases mortality risk among male dialysis patients. J. Am. Soc. Nephrol. 2009, 20, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Seidman, S.N.; Araujo, A.B.; Roose, S.P.; Devanand, D.P.; Xie, S.; Cooper, T.B.; McKinlay, J.B. Low testosterone levels in elderly men with dysthymic disorder. Am. J. Psychiatry 2002, 159, 456–459. [Google Scholar] [CrossRef] [PubMed]
- Baker, H.; Burger, H.; De Kretser, D.; Dulmanis, A.; Hudson, B.; O’Connor, S.; Paulsen, C.; Purcell, N.; Rennie, G.; Seah, C. A study of the endocrine manifestations of hepatic cirrhosis. QJM 1976, 45, 145–178. [Google Scholar] [PubMed]
- Diaz-Arjonilla, M.; Schwarcz, M.; Swerdloff, R.S.; Wang, C. Obesity, low testosterone levels and erectile dysfunction. Int. J. Impot. Res. 2008, 21, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.-C.; Lee, G.-B.; Chien, F.-C.; Chen, S.-J.; Chen, W.-J.; Yang, M.-C. A microfluidic system with integrated molecular imprinting polymer films for surface plasmon resonance detection. J. Micromech. Microeng. 2006, 16, 1251. [Google Scholar] [CrossRef]
- Wei, Q.Q.; Wei, T.X. A novel method to prepare spr sensor chips based on photografting molecularly imprinted polymer. Chin. Chem. Lett. 2011, 22, 721–724. [Google Scholar] [CrossRef]
- Zhang, Q.; Jing, L.; Zhang, J.; Ren, Y.; Wang, Y.; Wang, Y.; Wei, T.; Liedberg, B. Surface plasmon resonance sensor for femtomolar detection of testosterone with water-compatible macroporous molecularly imprinted film. Anal. Biochem. 2014, 463, 7–14. [Google Scholar] [CrossRef]
- Tan, Y.; Jing, L.; Ding, Y.; Wei, T. A novel double-layer molecularly imprinted polymer film based surface plasmon resonance for determination of testosterone in aqueous media. Appl. Surf. Sci. 2015, 342, 84–91. [Google Scholar] [CrossRef]
- Qiu, L.; Liu, W.; Huang, M.; Zhang, L. Preparation and application of solid-phase microextraction fiber based on molecularly imprinted polymer for determination of anabolic steroids in complicated samples. J. Chromatogr. A 2010, 1217, 7461–7470. [Google Scholar] [CrossRef] [PubMed]
- Zulfiqar, A.; Morgan, G.; Turner, N.W. Detection of multiple steroidal compounds in synthetic urine using comprehensive gas chromatography-mass spectrometry (gc[times]gc-ms) combined with a molecularly imprinted polymer clean-up protocol. Analyst 2014, 139, 4955–4963. [Google Scholar] [CrossRef] [PubMed]
- Gavrilović, I.; Mitchell, K.; Brailsford, A.D.; Cowan, D.A.; Kicman, A.T.; Ansell, R.J. A molecularly imprinted receptor for separation of testosterone and epitestosterone, based on a steroidal cross-linker. Steroids 2011, 76, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Liu, W.; Chen, J.; Jia, J.; Ma, Z.; Shi, Q.; Gao, Y.; Wang, X.; Xu, S.; Wang, K.; et al. Preparation and evaluation of polydopamine imprinting layer coated multi-walled carbon nanotubes for the determination of testosterone in prostate cancer lncap cells. Anal. Methods 2015, 7, 8326–8334. [Google Scholar] [CrossRef]
- Kadhem, A.; Xiang, S.; Nagel, S.; Lin, C.-H.; Fidalgo de Cortalezzi, M. Photonic molecularly imprinted polymer film for the detection of testosterone in aqueous samples. Polymers 2018, 10, 349. [Google Scholar] [CrossRef]
- Kopperi, M.; Riekkola, M.-L. Non-targeted evaluation of selectivity of water-compatible class selective adsorbents for the analysis of steroids in wastewater. Anal. Chim. Acta 2016, 920, 47–53. [Google Scholar] [CrossRef]
- Tse Sum Bui, B.; Merlier, F.; Haupt, K. Toward the use of a molecularly imprinted polymer in doping analysis: Selective preconcentration and analysis of testosterone and epitestosterone in human urine. Anal. Chem. 2010, 82, 4420–4427. [Google Scholar] [CrossRef]
- Doué, M.; Bichon, E.; Dervilly-Pinel, G.; Pichon, V.; Chapuis-Hugon, F.; Lesellier, E.; West, C.; Monteau, F.; Le Bizec, B. Molecularly imprinted polymer applied to the selective isolation of urinary steroid hormones: An efficient tool in the control of natural steroid hormones abuse in cattle. J. Chromatogr. A 2012, 1270, 51–61. [Google Scholar] [CrossRef]
- Bousoumah, R.; Antignac, J.P.; Camel, V.; Grimaldi, M.; Balaguer, P.; Courant, F.; Bichon, E.; Morvan, M.-L.; Le Bizec, B. Development of a molecular recognition based approach for multi-residue extraction of estrogenic endocrine disruptors from biological fluids coupled to liquid chromatography-tandem mass spectrometry measurement. Anal. Bioanal. Chem. 2015, 407, 8713–8723. [Google Scholar] [CrossRef]
- Gadzała-Kopciuch, R.; Ričanyová, J.; Buszewski, B. Isolation and detection of steroids from human urine by molecularly imprinted solid-phase extraction and liquid chromatography. J. Chromatogr. B 2009, 877, 1177–1184. [Google Scholar] [CrossRef]
- Ričanyová, J.; Gadzała-Kopciuch, R.; Reiffova, K.; Bazel, Y.; Buszewski, B. Molecularly imprinted adsorbents for preconcentration and isolation of progesterone and testosterone by solid phase extraction combined with hplc. Adsorption 2010, 16, 473–483. [Google Scholar] [CrossRef]
- Ambrosini, S.; Shinde, S.; De Lorenzi, E.; Sellergren, B. Glucuronide directed molecularly imprinted solid-phase extraction: Isolation of testosterone glucuronide from its parent drug in urine. Analyst 2012, 137, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Augustine, A.; Mathew, B. Synthesis of carbon nanotube incorporated molecular imprinted polymer with binding affinity towards testosterone. ISRN Polym. Sci. 2014, 2014, 7. [Google Scholar] [CrossRef]
- González-Sálamo, J.; Socas-Rodríguez, B.; Hernández-Borges, J.; del Mar Afonso, M.; Rodríguez-Delgado, M.Á. Evaluation of two molecularly imprinted polymers for the solid-phase extraction of natural, synthetic and mycoestrogens from environmental water samples before liquid chromatography with mass spectrometry. J. Sep. Sci. 2015, 38, 2692–2699. [Google Scholar] [CrossRef] [PubMed]
- Sanglar, C.; Jansen, T.; Silaghi, M.C.; Mernier, J.; Mignon, P.; Chermette, H. Study of prepolymerization complex formation in the synthesis of steroid-based molecularly imprinted polymers. Anal. Chem. 2012, 84, 4481–4488. [Google Scholar] [CrossRef]
- Panagiotopoulou, M.; Beyazit, S.; Nestora, S.; Haupt, K.; Tse Sum Bui, B. Initiator-free synthesis of molecularly imprinted polymers by polymerization of self-initiated monomers. Polymer 2015, 66, 43–51. [Google Scholar] [CrossRef]
- Adali-Kaya, Z.; Tse Sum Bui, B.; Falcimaigne-Cordin, A.; Haupt, K. Molecularly imprinted polymer nanomaterials and nanocomposites: Atom-transfer radical polymerization with acidic monomers. Angew. Chem. Int. Ed. 2015, 54, 5192–5195. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, Y.; Shen, X.; Chang, Z.; Tang, L.; Dong, W.-F.; Li, M.; He, J.-J. Ultrasensitive detection of testosterone using microring resonator with molecularly imprinted polymers. Sensors 2015, 15, 31558–31565. [Google Scholar] [CrossRef]
- Kellens, E.; Bové, H.; Vandenryt, T.; Lambrichts, J.; Dekens, J.; Drijkoningen, S.; D’Haen, J.; De Ceuninck, W.; Thoelen, R.; Junkers, T. Micro-patterned molecularly imprinted polymer structures on functionalized diamond-coated substrates for testosterone detection. Biosens. Bioelectron. 2018, 118, 58–65. [Google Scholar] [CrossRef]
- Garcia-Soto, M.J.; Haupt, K.; Gonzato, C. Synthesis of molecularly imprinted polymers by photo-iniferter polymerization under visible light. Polym. Chem. 2017, 8, 4830–4834. [Google Scholar] [CrossRef]
- Janata, J.; Josowicz, M. Conducting polymers in electronic chemical sensors. Nat. Mater. 2003, 2, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.S.; Pietrzyk-Le, A.; D’Souza, F.; Kutner, W. Electrochemically synthesized polymers in molecular imprinting for chemical sensing. Anal. Bioanal. Chem. 2012, 402, 3177–3204. [Google Scholar] [CrossRef]
- Malitesta, C.; Mazzotta, E.; Picca, R.A.; Poma, A.; Chianella, I.; Piletsky, S.A. Mip sensors–the electrochemical approach. Anal. Bioanal. Chem. 2012, 402, 1827–1846. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.H.; Wen, T.C. Polyaniline derivative with external and internal doping via electrochemical copolymerization of aniline and 2,5-diaminobenzenesulfonic acid on iro2-coated titanium electrode. J. Electrochem. Soc. 1994, 141, 2624–2632. [Google Scholar] [CrossRef]
- Lee, M.-H.; Thomas, J.L.; Su, Z.-L.; Zhang, Z.-X.; Lin, C.-Y.; Huang, Y.-S.; Yang, C.-H.; Lin, H.-Y. Doping of transition metal dichalcogenides in molecularly imprinted conductive polymers for the ultrasensitive determination of 17β-estradiol in eel serum. Biosens. Bioelectron. 2019, 150, 111901. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.-H.; Chih, Y.-K.; Cheng, H.-E.; Chen, C.-H. Nanofibers of self-doped polyaniline. Polymer 2005, 46, 10688–10698. [Google Scholar] [CrossRef]
- Yang, C.-H.; Huang, L.-R.; Chih, Y.-K.; Chung, S.-L. Simultaneous molecular-layer assembly and copolymerization of aniline and o-aminobenzenesulfonic acid for application in electrochromic devices. J. Phys. Chem. C 2007, 111, 3786–3794. [Google Scholar] [CrossRef]
- Lee, M.-H.; O’Hare, D.; Guo, H.-Z.; Yang, C.-H.; Lin, H.-Y. Electrochemical sensing of urinary progesterone with molecularly imprinted poly (aniline-co-metanilic acid) s. J. Mater. Chem. B 2016, 4, 3782–3787. [Google Scholar] [CrossRef]
- Syed, A.A.; Dinesan, M.K. Review: Polyaniline—A novel polymeric material. Talanta 1991, 38, 815–837. [Google Scholar] [CrossRef]
- Anand, M.S.; Sruthi, P.U.; Banshi, D.G. A localized and propagating spr, and molecular imprinting based fiber-optic ascorbic acid sensor using an in situ polymerized polyaniline–ag nanocomposite. Nanotechnology 2016, 27, 345501. [Google Scholar]
- Yang, C.-H.; Wang, T.-L.; Shieh, Y.-T. Molecular assembled crosslinked self-doped polyaniline nano-thin films in application of electrochromic devices. Electrochem. Commun. 2009, 11, 335–338. [Google Scholar] [CrossRef]
- Yang, C.-H.; Huang, L.-R.; Chih, Y.-K.; Lin, W.-C.; Liu, F.-J.; Wang, T.-L. Molecular assembled self-doped polyaniline copolymer ultra-thin films. Polymer 2007, 48, 3237–3247. [Google Scholar] [CrossRef]
- Huang, C.-Y.; Tsai, T.-C.; Thomas, J.L.; Lee, M.-H.; Liu, B.-D.; Lin, H.-Y. Urinalysis with molecularly imprinted poly(ethylene-co-vinyl alcohol) potentiostat sensors. Biosens. Bioelectron. 2009, 24, 2611–2617. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-Y.; O’Hare, D.; Chao, I.J.; Wei, H.-W.; Liang, Y.-F.; Liu, B.-D.; Lee, M.-H.; Lin, H.-Y. Integrated potentiostat for electrochemical sensing of urinary 3-hydroxyanthranilic acid with molecularly imprinted poly(ethylene-co-vinyl alcohol). Biosens. Bioelectron. 2015, 67, 208–213. [Google Scholar] [CrossRef]


| Sample No. | ARCHITECT ci 8200 System Testosterone (ng/mL) | TIECP Sensors | Accuracy (%) | |
|---|---|---|---|---|
| ΔCurrent (μA/cm2) | Avg. conc. (ng/mL) | |||
| 1 | 0.79 ± 0.02 | 33.85 ± 0.25 | 0.64 ± 0.03 | 81.0 |
| 2 | 1.51 ± 0.08 | 40.65 ± 0.75 | 1.28 ± 0.13 | 84.8 |
| 3 | 2.32 ± 0.01 | 47.10 ± 1.90 | 2.27 ± 0.50 | 97.8 |
| 4 | 0.33 ± 0.09 | 27.35 ± 1.15 | 0.28 ± 0.07 | 84.8 |
| 5 | 3.04 ± 0.18 | 50.05 ± 1.95 | 2.88 ± 0.62 | 94.7 |
| 6 | 9.13 ± 1.33 | 65.15 ± 2.95 | 8.99 ± 2.68 | 98.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, K.-H.; O’Hare, D.; Thomas, J.L.; Guo, H.-Z.; Yang, C.-H.; Lee, M.-H. Self-assembly Synthesis of Molecularly Imprinted Polymers for the Ultrasensitive Electrochemical Determination of Testosterone. Biosensors 2020, 10, 16. https://doi.org/10.3390/bios10030016
Liu K-H, O’Hare D, Thomas JL, Guo H-Z, Yang C-H, Lee M-H. Self-assembly Synthesis of Molecularly Imprinted Polymers for the Ultrasensitive Electrochemical Determination of Testosterone. Biosensors. 2020; 10(3):16. https://doi.org/10.3390/bios10030016
Chicago/Turabian StyleLiu, Kai-Hsi, Danny O’Hare, James L. Thomas, Han-Zhang Guo, Chien-Hsin Yang, and Mei-Hwa Lee. 2020. "Self-assembly Synthesis of Molecularly Imprinted Polymers for the Ultrasensitive Electrochemical Determination of Testosterone" Biosensors 10, no. 3: 16. https://doi.org/10.3390/bios10030016
APA StyleLiu, K.-H., O’Hare, D., Thomas, J. L., Guo, H.-Z., Yang, C.-H., & Lee, M.-H. (2020). Self-assembly Synthesis of Molecularly Imprinted Polymers for the Ultrasensitive Electrochemical Determination of Testosterone. Biosensors, 10(3), 16. https://doi.org/10.3390/bios10030016




