Nanomaterials in Plants: A Review of Hazard and Applications in the Agri-Food Sector
Abstract
1. Introduction
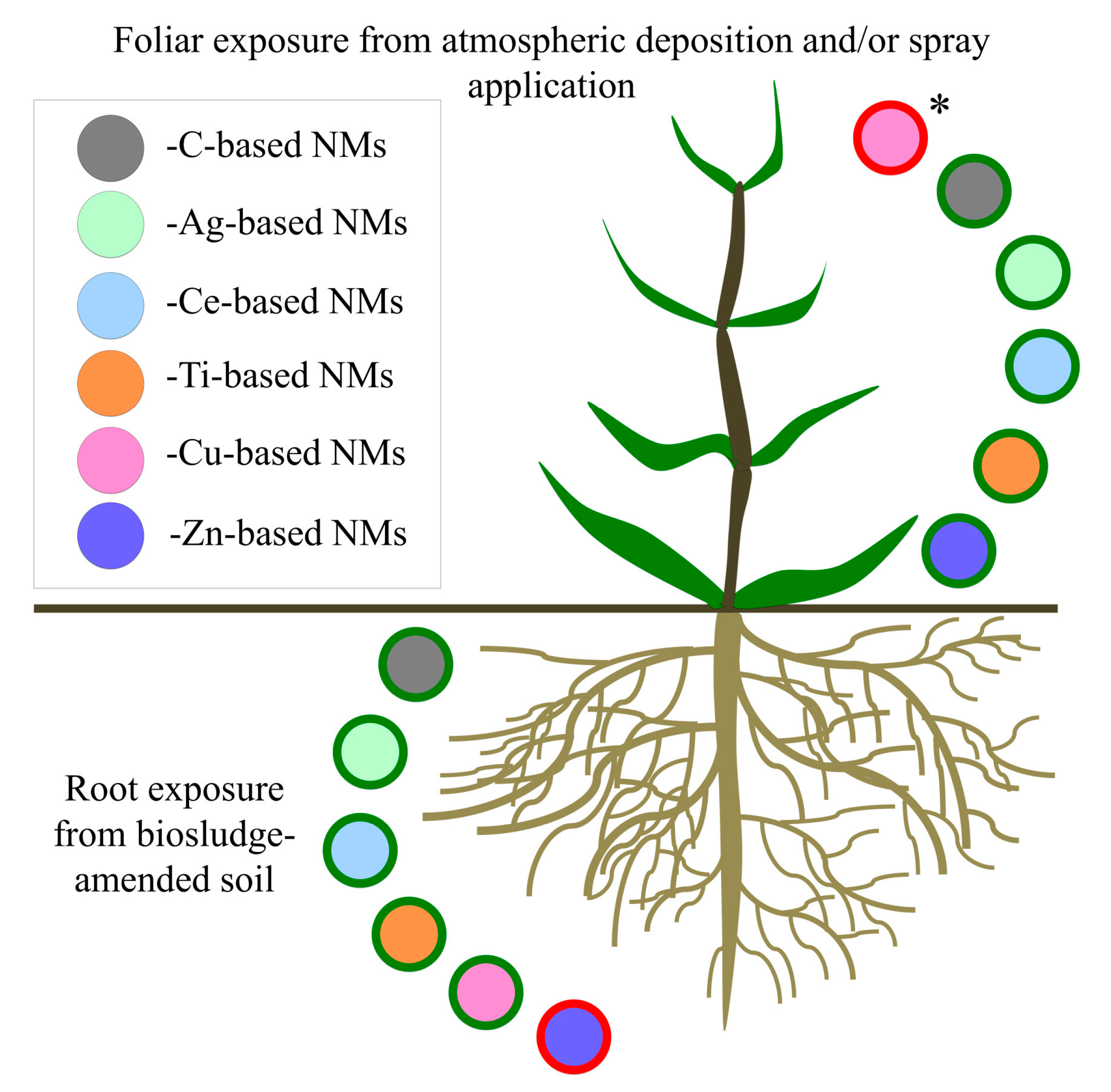
2. NM Interactions with Plants
3. Unintentional NM Exposure and Impacts on Agricultural Crop Yield and Nutritional Value
4. Co-Exposure to ENMs and Pollutants and Effects on Bioaccumulation and Phytotoxicity
5. NM Biotransformation in Plants
6. Applications of ENMs in the Agri-Food Sector
7. Hazard Potential of ENMs to Plants: General Perspective
8. Conclusions and Future Perspectives
- NMs do not pose risks to plant safety and agronomic characteristics, such as yield and nutritional quality, except at extremely high, environmentally unrealistic concentrations;
- NM dissolution appears to be a significant driver of toxicity due to the increased bioavailability of ions;
- NM co-exposures may enhance or diminish the risks posed by other toxic pollutants;
- NMs at low concentrations and/or applied during the early stages of plant growth (e.g., as seed coatings) provide beneficial effects with limited introduction into the environment or edible plant segments, justifying such uses from a nanosafety perspective.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Organisation for Economic Co-operation and Development. Task Force on the Safety of Novel Foods and Feeds. In Proceedings of the Organisation for Economic Co-operation and Development 25th Meeting of the Working Group for the Safety of Novel Foods and Feeds, Paris, France, 26–27 June 2018. [Google Scholar]
- Wang, Y.; Sun, C.; Zhao, X.; Cui, B.; Zeng, Z.; Wang, A.; Liu, G.; Cui, H. The Application of Nano-TiO2 Photo Semiconductors in Agriculture. Nanoscale Res. Lett. 2016, 11, 529. [Google Scholar] [CrossRef] [PubMed]
- Parisi, C.; Vigani, M.; Rodríguez-Cerezo, E. Agricultural Nanotechnologies: What are the current possibilities? Nano Today 2015, 10, 124–127. [Google Scholar] [CrossRef]
- Schwab, F.; Zhai, G.; Kern, M.; Turner, A.; Schnoor, J.L.; Wiesner, M.R. Barriers, pathways and processes for uptake, translocation and accumulation of nanomaterials in plants—Critical review. Nanotoxicology 2016, 10, 257–278. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.J.; Herth, S. Plant nanotoxicology. Trends Plant. Sci. 2011, 16, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; White, J.C.; Dhankher, O.P.; Xing, B. Metal-Based Nanotoxicity and Detoxification Pathways in Higher Plants. Environ. Sci. Technol. 2015, 49, 7109–7122. [Google Scholar] [CrossRef] [PubMed]
- Morales-Díaz, A.; Ortega-Ortíz, H.; Juárez-Maldonado, A.; Cadenas-Pliego, G.; González-Morales, S.; Benavides-Mendoza, A. Application of nanoelements in plant nutrition and its impact in ecosystems. Adv. Nat. Sci. Nanosci. Nanotechnol. 2017, 8, 013001. [Google Scholar] [CrossRef]
- Gardea-Torresdey, J.L.; Rico, C.M.; White, J.C. Trophic Transfer, Transformation, and Impact of Engineered Nanomaterials in Terrestrial Environments. Environ. Sci. Technol. 2014, 48, 2526–2540. [Google Scholar] [CrossRef] [PubMed]
- Xiong, T.; Dumat, C.; Dappe, V.; Vezin, H.; Schreck, E.; Shahid, M.; Pierart, A.; Sobanska, S. Copper Oxide Nanoparticle Foliar Uptake, Phytotoxicity, and Consequences for Sustainable Urban Agriculture. Environ. Sci. Technol. 2017, 51, 5242–5251. [Google Scholar] [CrossRef]
- Drobne, D.; Novak, S.; Talaber, I.; Lynch, I.; Kokalj, A.; Drobne, D.; Novak, S.; Talaber, I.; Lynch, I.; Kokalj, A.J. The Biological Fate of Silver Nanoparticles from a Methodological Perspective. Materials 2018, 11, 957. [Google Scholar] [CrossRef]
- Kranjc, E.; Mazej, D.; Regvar, M.; Drobne, D.; Remškar, M. Foliar surface free energy affects platinum nanoparticle adhesion, uptake, and translocation from leaves to roots in arugula and escarole. Environ. Sci. Nano 2017, 5, 520–532. [Google Scholar] [CrossRef]
- Kořenková, L.; Šebesta, M.; Urík, M.; Kolenčík, M.; Kratošová, G.; Bujdoš, M.; Vávra, I.; Dobročka, E. Physiological response of culture media-grown barley (Hordeum vulgare L.) to titanium oxide nanoparticles. Acta Agric. Scand. Sect. B Soil Plant. Sci. 2017, 67, 285–291. [Google Scholar] [CrossRef]
- Du, W.; Gardea-Torresdey, J.L.; Xie, Y.; Yin, Y.; Zhu, J.; Zhang, X.; Ji, R.; Gu, K.; Peralta-Videa, J.R.; Guo, H. Elevated CO2 levels modify TiO2 nanoparticle effects on rice and soil microbial communities. Sci. Total Environ. 2017, 578, 408–416. [Google Scholar] [CrossRef] [PubMed]
- García-Gómez, C.; Obrador, A.; González, D.; Babín, M.; Fernández, M.D. Comparative effect of ZnO NPs, ZnO bulk and ZnSO4 in the antioxidant defences of two plant species growing in two agricultural soils under greenhouse conditions. Sci. Total Environ. 2017, 589, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Xue, W.; Han, Y.; Tan, J.; Wang, Y.; Wang, G.; Wang, H. Effects of Nanochitin on the Enhancement of the Grain Yield and Quality of Winter Wheat. J. Agric. Food Chem. 2017, 66, 6637–6645. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Zhao, L.; Keller, A.A. Interactions, Transformations, and Bioavailability of Nano-Copper Exposed to Root Exudates. Environ. Sci. Technol. 2017, 51, 9774–9783. [Google Scholar] [CrossRef]
- Rossi, L.; Sharifan, H.; Zhang, W.; Schwab, A.P.; Ma, X. Mutual effects and in planta accumulation of co-existing cerium oxide nanoparticles and cadmium in hydroponically grown soybean (Glycine max L. Merr). Environ. Sci. Nano 2018, 5, 150–157. [Google Scholar] [CrossRef]
- Noori, A.; White, J.C.; Newman, L.A. Mycorrhizal fungi influence on silver uptake and membrane protein gene expression following silver nanoparticle exposure. J. Nanoparticle Res. 2017, 19, 66. [Google Scholar] [CrossRef]
- Siani, N.G.; Fallah, S.; Pokhrel, L.R.; Rostamnejadi, A. Natural amelioration of Zinc oxide nanoparticle toxicity in fenugreek (Trigonella foenum-gracum) by arbuscular mycorrhizal (Glomus intraradices) secretion of glomalin. Plant. Physiol. Biochem. 2017, 112, 227–238. [Google Scholar] [CrossRef]
- Zhao, Q.; Ma, C.; White, J.C.; Dhankher, O.P.; Zhang, X.; Zhang, S.; Xing, B. Quantitative evaluation of multi-wall carbon nanotube uptake by terrestrial plants. Carbon 2017, 114, 661–670. [Google Scholar] [CrossRef]
- Duran, N.M.; Savassa, S.M.; Lima, R.G.; de Almeida, E.; Linhares, F.S.; van Gestel, C.A.M.; Pereira de Carvalho, H.W. X-ray Spectroscopy Uncovering the Effects of Cu Based Nanoparticle Concentration and Structure on Phaseolus vulgaris Germination and Seedling Development. J. Agric. Food Chem. 2017, 65, 7874–7884. [Google Scholar] [CrossRef]
- Pagano, L.; Pasquali, F.; Majumdar, S.; Torre-Roche, R.D.; Zuverza-Mena, N.; Villani, M.; Zappettini, A.; Marra, R.E.; Isch, S.M.; Marmiroli, M.; et al. Exposure of Cucurbita pepo to binary combinations of engineered nanomaterials: Physiological and molecular response. Environ. Sci. Nano 2017, 4, 1579–1590. [Google Scholar] [CrossRef]
- Pradas Del Real, A.E.; Vidal, V.; Carrière, M.; Castillo-Michel, H.; Levard, C.; Chaurand, P.; Sarret, G. Silver Nanoparticles and Wheat Roots: A Complex Interplay. Environ. Sci. Technol. 2017, 51, 5774–5782. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Stowers, C.; Rossi, L.; Zhang, W.; Lombardini, L.; Ma, X. Physiological effects of cerium oxide nanoparticles on the photosynthesis and water use efficiency of soybean (Glycine max L. Merr). Environ. Sci. Nano 2017, 4, 1086–1094. [Google Scholar] [CrossRef]
- Deng, Y.; Eitzer, B.; White, J.C.; Xing, B. Impact of multiwall carbon nanotubes on the accumulation and distribution of carbamazepine in collard greens (Brassica oleracea). Environ. Sci. Nano 2017, 4, 149–159. [Google Scholar] [CrossRef]
- Medina-Velo, I.A.; Dominguez, O.E.; Ochoa, L.; Barrios, A.C.; Hernández-Viezcas, J.A.; White, J.C.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Nutritional quality of bean seeds harvested from plants grown in different soils amended with coated and uncoated zinc oxide nanomaterials. Environ. Sci. Nano 2017, 4, 2336–2347. [Google Scholar] [CrossRef]
- Gao, X.; Avellan, A.; Laughton, S.N.; Vaidya, R.; Rodrigues, S.M.; Casman, E.A.; Lowry, G.V. CuO nanoparticle dissolution and toxicity to wheat (Triticum aestivum) in rhizosphere soil. Environ. Sci. Technol. 2018, 52, 2888–2897. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; He, X.; Zhang, P.; Zhang, Z.; Ding, Y.; Zhang, J.; Wang, G.; Xie, C.; Luo, W.; Zhang, J.; et al. Xylem and Phloem Based Transport of CeO2 Nanoparticles in Hydroponic Cucumber Plants. Environ. Sci. Technol. 2017, 51, 5215–5221. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, K.M.; Bhati, A.; Singh, A.; Sonker, A.K.; Sarkar, S.; Sonkar, S.K. Sustainable Changes in the Contents of Metallic Micronutrients in First Generation Gram Seeds Imposed by Carbon Nano-onions: Life Cycle Seed to Seed Study. ACS Sustain. Chem. Eng. 2017, 5, 2906–2916. [Google Scholar] [CrossRef]
- Lahiani, M.H.; Nima, Z.A.; Villagarcia, H.; Biris, A.S.; Khodakovskaya, M.V. Assessment of Effects of the Long-Term Exposure of Agricultural Crops to Carbon Nanotubes. J. Agric. Food Chem. 2017, 66, 6654–6662. [Google Scholar] [CrossRef]
- Rui, M.; Ma, C.; Tang, X.; Yang, J.; Jiang, F.; Pan, Y.; Xiang, Z.; Hao, Y.; Rui, Y.; Cao, W.; et al. Phytotoxicity of Silver Nanoparticles to Peanut (Arachis hypogaea L.): Physiological Responses and Food Safety. ACS Sustain. Chem. Eng. 2017, 5, 6557–6567. [Google Scholar] [CrossRef]
- Yang, J.; Jiang, F.; Ma, C.; Rui, Y.; Rui, M.; Adeel, M.; Cao, W.; Xing, B. Alteration of Crop Yield and Quality of Wheat upon Exposure to Silver Nanoparticles in a Life Cycle Study. J. Agric. Food Chem. 2018, 66, 2589–2597. [Google Scholar] [CrossRef] [PubMed]
- Das, P.; Barua, S.; Sarkar, S.; Chatterjee, S.K.; Mukherjee, S.; Goswami, L.; Das, S.; Bhattacharya, S.; Karak, N.; Bhattacharya, S.S. Mechanism of toxicity and transformation of silver nanoparticles: Inclusive assessment in earthworm-microbe-soil-plant system. Geoderma 2018, 314, 73–84. [Google Scholar] [CrossRef]
- Rico, C.M.; Johnson, M.G.; Marcus, M.A.; Andersen, C.P. Intergenerational responses of wheat (Triticum aestivum L.) to cerium oxide nanoparticles exposure. Environ. Sci. Nano 2017, 4, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Djanaguiraman, M.; Nair, R.; Giraldo, J.P.; Prasad, P.V.V. Cerium Oxide Nanoparticles Decrease Drought-Induced Oxidative Damage in Sorghum Leading to Higher Photosynthesis and Grain Yield. ACS Omega 2018, 3, 14406–14416. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Xu, C.; Liu, Q.; Sun, L.; Luo, Y.; Shi, J. Fate and Transformation of CuO Nanoparticles in the Soil–Rice System during the Life Cycle of Rice Plants. Environ. Sci. Technol. 2017, 51, 4907–4917. [Google Scholar] [CrossRef] [PubMed]
- Rawat, S.; Pullagurala, V.L.R.; Hernandez-Molina, M.; Sun, Y.; Niu, G.; Hernandez-Viezcas, J.A.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Impacts of copper oxide nanoparticles on bell pepper (Capsicum annum L.) plants: A full life cycle study. Environ. Sci. Nano 2018, 5, 83–95. [Google Scholar] [CrossRef]
- Zhao, L.; Huang, Y.; Adeleye, A.S.; Keller, A.A. Metabolomics Reveals Cu(OH)2 Nanopesticide-Activated Anti-oxidative Pathways and Decreased Beneficial Antioxidants in Spinach Leaves. Environ. Sci. Technol. 2017, 51, 10184–10194. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Tan, W.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L.; Ji, R.; Yin, Y.; Guo, H. Interaction of metal oxide nanoparticles with higher terrestrial plants: Physiological and biochemical aspects. Plant Physiol. Biochem. 2017, 110, 210–225. [Google Scholar] [CrossRef] [PubMed]
- Zahra, Z.; Waseem, N.; Zahra, R.; Lee, H.; Badshah, M.A.; Mehmood, A.; Choi, H.K.; Arshad, M. Growth and Metabolic Responses of Rice (Oryza sativa L.) Cultivated in Phosphorus-Deficient Soil Amended with TiO2 Nanoparticles. J. Agric. Food Chem. 2017, 65, 5598–5606. [Google Scholar] [CrossRef] [PubMed]
- Dimkpa, C.O.; Singh, U.; Bindraban, P.S.; Elmer, W.H.; Gardea-Torresdey, J.L.; White, J.C. Exposure to Weathered and Fresh Nanoparticle and Ionic Zn in Soil Promotes Grain Yield and Modulates Nutrient Acquisition in Wheat (Triticum aestivum L.). J. Agric. Food Chem. 2018, 66, 9645–9656. [Google Scholar] [CrossRef] [PubMed]
- Dimkpa, C.O.; White, J.C.; Elmer, W.H.; Gardea-Torresdey, J. Nanoparticle and Ionic Zn Promote Nutrient Loading of Sorghum Grain under Low NPK Fertilization. J. Agric. Food Chem. 2017, 65, 8552–8559. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Jing, X.; Adams, C.A.; Shi, Z.; Sun, Y. Decreased ZnO nanoparticle phytotoxicity to maize by arbuscular mycorrhizal fungus and organic phosphorus. Environ. Sci. Pollut. Res. 2018, 25, 23736–23747. [Google Scholar] [CrossRef] [PubMed]
- Medina-Velo, I.A.; Zuverza-Mena, N.; Tamez, C.; Ye, Y.; Hernandez-Viezcas, J.A.; White, J.C.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Minimal Transgenerational Effect of ZnO Nanomaterials on the Physiology and Nutrient Profile of Phaseolus vulgaris. ACS Sustain. Chem. Eng. 2018, 6, 7924–7930. [Google Scholar] [CrossRef]
- Dimkpa, C.O.; Bindraban, P.S.; Fugice, J.; Agyin-Birikorang, S.; Singh, U.; Hellums, D. Composite micronutrient nanoparticles and salts decrease drought stress in soybean. Agron. Sustain. Dev. 2017, 37, 5. [Google Scholar] [CrossRef]
- Rui, M.; Ma, C.; White, J.C.; Hao, Y.; Wang, Y.; Tang, X.; Yang, J.; Jiang, F.; Ali, A.; Rui, Y.; et al. Metal oxide nanoparticles alter peanut (Arachis hypogaea L.) physiological response and reduce nutritional quality: A life cycle study. Environ. Sci. Nano 2018, 5, 2088–2102. [Google Scholar] [CrossRef]
- Deng, R.; Lin, D.; Zhu, L.; Majumdar, S.; White, J.C.; Gardea-Torresdey, J.L.; Xing, B. Nanoparticle interactions with co-existing contaminants: Joint toxicity, bioaccumulation and risk. Nanotoxicology 2017, 11, 591–612. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Mu, L.; Hu, X. Integrating proteomics, metabolomics and typical analysis to investigate the uptake and oxidative stress of graphene oxide and polycyclic aromatic hydrocarbons. Environ. Sci. Nano 2018, 5, 115–129. [Google Scholar] [CrossRef]
- Liu, J.; Simms, M.; Song, S.; King, R.S.; Cobb, G.P. Physiological Effects of Copper Oxide Nanoparticles and Arsenic on the Growth and Life Cycle of Rice (Oryza sativa japonica ‘Koshihikari’). Environ. Sci. Technol. 2018, 52, 13728–13737. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Liu, H.; Chen, G.; Zhao, Q.; Eitzer, B.; Wang, Z.; Cai, W.; Newman, L.A.; White, J.C.; Dhankher, O.P.; et al. Effects of titanium oxide nanoparticles on tetracycline accumulation and toxicity in Oryza sativa. Environ. Sci. Nano 2017, 4, 1827–1839. [Google Scholar] [CrossRef]
- Soares, C.; Branco-Neves, S.; de Sousa, A.; Azenha, M.; Cunha, A.; Pereira, R.; Fidalgo, F. SiO2 nanomaterial as a tool to improve Hordeum vulgare L tolerance to nano-NiO stress. Sci. Total Environ. 2018, 622, 517–525. [Google Scholar] [CrossRef]
- Rossi, L.; Zhang, W.; Ma, X. Cerium oxide nanoparticles alter the salt stress tolerance of Brassica napus L. by modifying the formation of root apoplastic barriers. Environ. Pollut. Bark. Essex 1987 2017, 229, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Rossi, L.; Zhang, W.; Schwab, A.P.; Ma, X. Uptake, Accumulation, and in Planta Distribution of Coexisting Cerium Oxide Nanoparticles and Cadmium in Glycine max L Merr. Environ. Sci. Technol. 2017, 51, 12815–12824. [Google Scholar] [CrossRef] [PubMed]
- Servin, A.D.; Pagano, L.; Castillo-Michel, H.; De la Torre-Roche, R.; Hawthorne, J.; Hernandez-Viezcas, J.A.; Loredo-Portales, R.; Majumdar, S.; Gardea-Torresday, J.; Dhankher, O.P.; et al. Weathering in soil increases nanoparticle CuO bioaccumulation within a terrestrial food chain. Nanotoxicology 2017, 11, 98–111. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yi, Y.; Fang, Z.; Tsang, E.P. Effects of biochar on phytotoxicity and translocation of polybrominated diphenyl ethers in Ni/Fe bimetallic nanoparticle-treated soil. Environ. Sci. Pollut. Res. 2018, 25, 2570–2579. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.X.; Wang, G.Z.; Zhang, Y.X.; Zhao, H.J. Hydroxyapatite nanoparticles in root cells: Reducing the mobility and toxicity of Pb in rice. Environ. Sci. Nano 2018, 5, 398–407. [Google Scholar] [CrossRef]
- Jośko, I.; Oleszczuk, P.; Skwarek, E. Toxicity of combined mixtures of nanoparticles to plants. J. Hazard. Mater. 2017, 331, 200–209. [Google Scholar] [CrossRef] [PubMed]
- Maurer-Jones, M.A.; Gunsolus, I.L.; Murphy, C.J.; Haynes, C.L. Toxicity of Engineered Nanoparticles in the Environment. Anal. Chem. 2013, 85, 3036–3049. [Google Scholar] [CrossRef] [PubMed]
- Cruz, T.N.M.; Savassa, S.M.; Gomes, M.H.F.; Rodrigues, E.S.; Duran, N.M.; Almeida, E.; Martinelli, A.P.; Carvalho, H.W.P. Shedding light on the mechanisms of absorption and transport of ZnO nanoparticles by plants via in vivo X-ray spectroscopy. Environ. Sci. Nano 2017, 4, 2367–2376. [Google Scholar] [CrossRef]
- Layet, C.; Auffan, M.; Santaella, C.; Chevassus-Rosset, C.; Montes, M.; Ortet, P.; Barakat, M.; Collin, B.; Legros, S.; Bravin, M.N.; et al. Evidence that Soil Properties and Organic Coating Drive the Phytoavailability of Cerium Oxide Nanoparticles. Environ. Sci. Technol. 2017, 51, 9756–9764. [Google Scholar] [CrossRef] [PubMed]
- Spielman-Sun, E.; Lombi, E.; Donner, E.; Howard, D.; Unrine, J.M.; Lowry, G.V. Impact of Surface Charge on Cerium Oxide Nanoparticle Uptake and Translocation by Wheat (Triticum aestivum). Environ. Sci. Technol. 2017, 51, 7361–7368. [Google Scholar] [CrossRef] [PubMed]
- Savassa, S.M.; Duran, N.M.; Rodrigues, E.S.; de Almeida, E.; van Gestel, C.A.M.; Bompadre, T.F.V.; de Carvalho, H.W. Effects of ZnO Nanoparticles on Phaseolus vulgaris Germination and Seedling Development Determined by X-ray Spectroscopy. ACS Appl. Nano Mater. 2018, 1, 6414–6426. [Google Scholar] [CrossRef]
- Wang, P.; Lombi, E.; Sun, S.; Scheckel, K.G.; Malysheva, A.; McKenna, B.A.; Menzies, N.W.; Zhao, F.J.; Kopittke, P.M. Characterizing the uptake, accumulation and toxicity of silver sulfide nanoparticles in plants. Environ. Sci. Nano 2017, 4, 448–460. [Google Scholar] [CrossRef]
- Rico, C.M.; Johnson, M.G.; Marcus, M.A. Cerium oxide nanoparticles transformation at the root–soil interface of barley (Hordeum vulgare L.). Environ. Sci. Nano 2018, 5, 1807–1812. [Google Scholar] [CrossRef]
- Fraceto, L.F.; Grillo, R.; Medeiros, D.A.G.; Scognamiglio, V.; Rea, G.; Bartolucci, C. Nanotechnology in Agriculture: Which Innovation Potential Does It Have? Front. Environ. Sci. 2016, 4, 20. [Google Scholar] [CrossRef]
- Prasad, R.; Bhattacharyya, A.; Nguyen, Q.D. Nanotechnology in Sustainable Agriculture: Recent Developments, Challenges, and Perspectives. Front. Microbiol. 2017, 8, 1014. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, S.M.; Demokritou, P.; Dokoozlian, N.; Hendren, C.O.; Karn, B.; Mauter, M.S.; Sadik, O.A.; Safarpour, M.; Unrine, J.M.; Viers, J.; et al. Nanotechnology for sustainable food production: Promising opportunities and scientific challenges. Environ. Sci. Nano 2017, 4, 767–781. [Google Scholar] [CrossRef]
- Singh, T.; Shukla, S.; Kumar, P.; Wahla, V.; Bajpai, V.K.; Rather, I.A. Application of Nanotechnology in Food Science: Perception and Overview. Front. Microbiol. 2017, 8, 2517. [Google Scholar] [CrossRef] [PubMed]
- Kah, M.; Hofmann, T. Nanopesticide research: Current trends and future priorities. Environ. Int. 2014, 63, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Baddar, Z.E.; Unrine, J.M. Functionalized-ZnO-Nanoparticle Seed Treatments to Enhance Growth and Zn Content of Wheat (Triticum aestivum) Seedlings. J. Agric. Food Chem. 2018, 66, 12166–12178. [Google Scholar] [CrossRef]
- Cadena, M.B.; Preston, G.M.; Van der Hoorn, R.A.L.; Flanagan, N.A.; Townley, H.E.; Thompson, I.P. Enhancing cinnamon essential oil activity by nanoparticle encapsulation to control seed pathogens. Ind. Crop. Prod. 2018, 124, 755–764. [Google Scholar] [CrossRef]
- Kumar, V.; Sachdev, D.; Pasricha, R.; Maheshwari, P.H.; Taneja, N.K. Zinc-Supported Multiwalled Carbon Nanotube Nanocomposite: A Synergism to Micronutrient Release and a Smart Distributor To Promote the Growth of Onion Seeds in Arid Conditions. ACS Appl. Mater. Interfaces 2018, 10, 36733–36745. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Yang, D.; Cui, J. A graphene oxide/silver nanoparticle composite as a novel agricultural antibacterial agent against Xanthomonas oryzae pv. oryzae for crop disease management. New J. Chem. 2017, 41, 13692–13699. [Google Scholar] [CrossRef]
- Oliveira, J.L.; Campos, E.V.R.; Pereira, A.E.S.; Pasquoto, T.; Lima, R.; Grillo, R.; Andrade, D.J.; Santos, F.A.D.; Fraceto, L.F. Zein Nanoparticles as Eco-Friendly Carrier Systems for Botanical Repellents Aiming Sustainable Agriculture. J. Agric. Food Chem. 2018, 66, 1330–1340. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.E.S.; Silva, P.M.; Oliveira, J.L.; Oliveira, H.C.; Fraceto, L.F. Chitosan nanoparticles as carrier systems for the plant growth hormone gibberellic acid. Coll. Surf. B Biointerfaces 2017, 150, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, A.; Barmota, H.; Bala, A. Antifungal evaluation studies of copper sulfide nano-aquaformulations and its impact on seed quality of rice (Oryzae sativa). Appl. Nanosci. 2017, 7, 681–689. [Google Scholar] [CrossRef]
- Xiong, L.; Wang, P.; Hunter, M.N.; Kopittke, P.M. Bioavailability and movement of hydroxyapatite nanoparticles (HA-NPs) applied as a phosphorus fertiliser in soils. Environ. Sci. Nano 2018, 5, 2888–2898. [Google Scholar] [CrossRef]
- Borgatta, J.; Ma, C.; Hudson-Smith, N.; Elmer, W.; Plaza Pérez, C.D.; De La Torre-Roche, R.; Zuverza-Mena, N.; Haynes, C.L.; White, J.C.; Hamers, R.J. Copper Based Nanomaterials Suppress Root Fungal Disease in Watermelon (Citrullus lanatus): Role of Particle Morphology, Composition and Dissolution Behavior. ACS Sustain. Chem. Eng. 2018, 6, 14847–14856. [Google Scholar] [CrossRef]
- Ashfaq, M.; Verma, N.; Khan, S. Carbon nanofibers as a micronutrient carrier in plants: Efficient translocation and controlled release of Cu nanoparticles. Environ. Sci. Nano 2017, 4, 138–148. [Google Scholar] [CrossRef]
- Choudhary, R.C.; Kumaraswamy, R.V.; Kumari, S.; Sharma, S.S.; Pal, A.; Raliya, R.; Biswas, P.; Saharan, V. Cu-chitosan nanoparticle boost defense responses and plant growth in maize (Zea mays L.). Sci. Rep. 2017, 7, 9754. [Google Scholar] [CrossRef]
- Taran, N.; Storozhenko, V.; Svietlova, N.; Batsmanova, L.; Shvartau, V.; Kovalenko, M. Effect of Zinc and Copper Nanoparticles on Drought Resistance of Wheat Seedlings. Nanoscale Res. Lett. 2017, 12, 60. [Google Scholar] [CrossRef]
- Wang, X.; Cai, A.; Wen, X.; Jing, D.; Qi, H.; Yuan, H. Graphene oxide-Fe3O4 nanocomposites as high-performance antifungal agents against Plasmopara viticola. Sci. China Mater. 2017, 60, 258–268. [Google Scholar] [CrossRef]
- Zhao, P.; Yuan, W.; Xu, C.; Li, F.; Cao, L.; Huang, Q. Enhancement of Spirotetramat Transfer in Cucumber Plant Using Mesoporous Silica Nanoparticles as Carriers. J. Agric. Food Chem. 2018, 66, 11592–11600. [Google Scholar] [CrossRef] [PubMed]
- Giese, B.; Klaessig, F.; Park, B.; Kaegi, R.; Steinfeldt, M.; Wigger, H.; Gleich, A.; Gottschalk, F. Risks, Release and Concentrations of Engineered Nanomaterial in the Environment. Sci. Rep. 2018, 8, 1565. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.Y.; Gottschalk, F.; Hungerbühler, K.; Nowack, B. Comprehensive probabilistic modelling of environmental emissions of engineered nanomaterials. Environ. Pollut. Bark. Essex 1987 2014, 185, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chang, C.H.; Ji, Z.; Bouchard, D.C.; Nisbet, R.M.; Schimel, J.P.; Gardea-Torresdey, J.L.; Holden, P.A. Agglomeration Determines Effects of Carbonaceous Nanomaterials on Soybean Nodulation, Dinitrogen Fixation Potential, and Growth in Soil. ACS Nano 2017, 11, 5753–5765. [Google Scholar] [CrossRef]
- Lazareva, A.; Keller, A.A. Estimating Potential Life Cycle Releases of Engineered Nanomaterials from Wastewater Treatment Plants. ACS Sustain. Chem. Eng. 2014, 2, 1656–1665. [Google Scholar] [CrossRef]
- Hao, Y.; Ma, C.; Zhang, Z.; Song, Y.; Cao, W.; Guo, J.; Zhou, G.; Rui, Y.; Liu, L.; Xing, B. Carbon nanomaterials alter plant physiology and soil bacterial community composition in a rice-soil-bacterial ecosystem. Environ. Pollut. 2018, 232, 123–136. [Google Scholar] [CrossRef]
- Zhao, L.; Hu, Q.; Huang, Y.; Keller, A.A. Response at Genetic, Metabolic, and Physiological Levels of Maize (Zea mays) Exposed to a Cu(OH)2 Nanopesticide. ACS Sustain. Chem. Eng. 2017, 5, 8294–8301. [Google Scholar] [CrossRef]
- Paterson, G.; Macken, A.; Thomas, K.V. The need for standardized methods and environmental monitoring programs for anthropogenic nanoparticles. Anal. Methods 2011, 3, 1461–1467. [Google Scholar] [CrossRef]
- Ruotolo, R.; Maestri, E.; Pagano, L.; Marmiroli, M.; White, J.C.; Marmiroli, N. Plant response to metal-containing engineered nanomaterials: An omics-based perspective. Environ. Sci. Technol. 2018. [Google Scholar] [CrossRef]
- Chen, L.; Yang, S.; Liu, Y.; Mo, M.; Guan, X.; Huang, L.; Sun, C.; Yang, S.T.; Chang, X.L. Toxicity of graphene oxide to naked oats (Avena sativa L) in hydroponic and soil cultures. RSC Adv. 2018, 8, 15336–15343. [Google Scholar] [CrossRef]
- Zhang, H.; Du, W.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L.; White, J.C.; Keller, A.; Guo, H.; Ji, R.; Zhao, L. Metabolomics Reveals How Cucumber (Cucumis sativus) Reprograms Metabolites To Cope with Silver Ions and Silver Nanoparticle-Induced Oxidative Stress. Environ. Sci. Technol. 2018, 52, 8016–8026. [Google Scholar] [CrossRef] [PubMed]
- Salehi, H.; Chehregani, A.; Lucini, L.; Majd, A.; Gholami, M. Morphological, proteomic and metabolomic insight into the effect of cerium dioxide nanoparticles to Phaseolus vulgaris L. under soil or foliar application. Sci. Total Environ. 2018, 616, 1540–1551. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Hu, Q.; Huang, Y.; Fulton, A.N.; Hannah-Bick, C.; Adeleye, A.S.; Keller, A.A. Activation of antioxidant and detoxification gene expression in cucumber plants exposed to a Cu(OH)2 nanopesticide. Environ. Sci. Nano 2017, 4, 1750–1760. [Google Scholar] [CrossRef]
| NM | Size | Plant Species | Exposure/Medium | Duration | Results | Reference |
|---|---|---|---|---|---|---|
| Carbon-Based NMs | ||||||
| CNO | 20–40 nm | Gram | Sprouted seed; 0, 10, 20, and 30 µg/mL water. Transfer to soil after 10 days. | 10 days; harvested after ~4 months |
| [29] |
| Chitin | 80–200 nm long, 30–50 nm wide | Winter wheat—MSW and LSW cultivars | Seed, root; 0, 0.002, 0.006, and 0.02 g/kg sandy soil | Full life-cycle |
| [15] |
| MWCNTs | 15–40 nm wide | Barley Maize Soybean | Root; 50 µg/mL deionized water with nutrient solution | 20 weeks |
| [30] |
| Metal-based NMs | ||||||
| Ag | 20 nm | Peanut | Seed and root; 50, 500, and 2000 mg/kg sandy soil | 98 days |
| [31] |
| Ag | 5.6 nm | Wheat | Seed and root; 20, 200, and 2000 mg/kg soil | 4 months |
| [32] |
| Ag with PEG coating | 7–14 nm | Tomato | Root; 10 mg/kg soil | 56–62 days |
| [33] |
| CeO2 | 8 ± 1 nm | Wheat | S1 plants grown to maturity (0, 125, and 500 mg/kg soil); seeds grown in factorial combinations (1, 125, and 500 mg/kg) (S2 plants). | 90 days |
| [34] |
| CeO2 | 15 ± 5 nm | Sorghum | Foliar: 0 and 2 mg/plant applied 60 days after sowing, at which time one group was subjected to drought conditions for 21 days. Soil medium used. | >21 days (until maturity) |
| [35] |
| CuO | 43 ± 9 nm | Rice | Root; 50, 100, 500, and 1,000 mg/kg soil | 7, 21, 60, and 88 days |
| [36] |
| CuO | 20–100 nm | Bell pepper | Root; 0, 125, 250, and 500 mg/kg soil | 90 days |
| [37] |
| CuO | 40–60 nm | Lettuce Cabbage | Leaf; 0, 10, and 250 mg/plant (applied as dry particles to adaxial surfaces); plants grown in soil medium | 5, 10, and 15 days |
| [9] |
| Cu(OH)2 | (~50 -> 1,000 nm) | Spinach | Leaf; 0, 1.8, and 18 mg/plant; plants grown in artificial growth medium | 7 days |
| [38] |
| TiO2 | 20–100 nm | Rice | Root; 50 and 200 mg/kg soil under background or elevated CO2 (370 and 570 µmol mol−1, respectively) | 130 days |
| [39] |
| TiO2 | 20 nm | Rice | Seed, root: 0, 25, 50, 150, 250, 500, and 750 mg/kg P-deficient soil | Full life-cycle |
| [40] |
| ZnO | 18 nm | Winter wheat | Root; Fresh soil with 6 mg/kg soil and used soil with 5.98 mg/kg (previously used to grow sorghum and aged for 6 months) | Grown to maturity |
| [41] |
| ZnO | 18 nm | Sorghum | Root; 6 mg/kg soil Leaf; 100 mL treatment with same amount of Zn as in soil. Low or high soil N, P, and K for root and leaf exposures | Not provided |
| [42] |
| ZnO | <100 nm | Bean Tomato | Root; 3, 20, 100, and 225 mg/kg acidic (pH 5.4) or calcareous (pH 8.3) soil | 90 days |
| [14] |
| ZnO | 30 nm | Maize | Root; 0 and 500 mg/kg soil with and without organic P (0, 20, and 50 mg/kg) and AMF (Funneliformis mosseae; with and without) | 9 weeks |
| [43] |
| ZnO (bare and hydrophobically-coated) | 93.8 nm (bare) 84.1 nm (coated) | Bean | Root; S1 plants grown in soil with 125, 250, and 500 mg/kg soil. S2 plants grown in soil without NMs | Grown to maturity |
| [44] |
| ZnO (bare and hydrophobically-coated) | 10–300 nm | Bean | Seed and root; bare and hydrophobically-coated NMs (62.5, 125, 250, and 500 mg/kg) in natural soil (NS) and organic matter-enriched soil (ES) | >45 days, until maturity |
| [26] |
| ZnO B2O3 CuO | <100 nm <100 nm <50 nm | Soybean | Leaf; 20 mL with all 3 NMs (1.77 g ZnO/L, 0.80 g CuO/L, and 0.92 g B203/L water) followed by a 14 day drought period | 19 weeks |
| [45] |
| Fe2O3 CuO TiO2 | 20 nm 40 nm 5 nm | Peanut | Seed and root; 50 and 500 mg/kg soil | 145 days |
| [46] |
| NM | Size | Plant Species | Exposure/Medium | Duration | Results | Reference |
|---|---|---|---|---|---|---|
| GO | 2.0 ± 0.5 nm wide, 0.5–5 µm long | Rice | Root; 0.01, 0.1, and 1.0 mg/L ½-strength culture solution with or without 10 µg/L PAHs | 7 days |
| [48] |
| pCNTs cCNTs | <8 nm wide; 10–30 µm in length | Collard greens | Root; 50 mg/L hydroponic medium and 500 mg/kg soil with carbamazepine (100 µg/L and 100 µg/kg, respectively) | 28 days (hydroponics); 42 days (soil) |
| [25] |
| MWCNTs | 36.5 ± 12.7 nm width; 350 nm length | Rice Maize Soybean | Root; 2.25 mg/L with SPAOMs (0 and 0.325 mM) in ½-strength Hoagland solution. | 1 day |
| [20] |
| PVP-CeO2 | 41.7 ± 5.2 nm | Soybean | Root; 0 and 500 mg/kg sand with 25% Hoagland solution with Cd (0, 0.25, and 1 mg/kg sand) | 30 days |
| [53] |
| PVP-CeO2 | 41.7 ± 5.2 nm | Soybean | Roots; 100 mg/L tap water with and without 1.0 mg Cd2+/L tap water | 8 days |
| [17] |
| PVP- CeO2 | 52.6 nm (average) | Rapeseed | Root; 0 and 500 mg/kg dry sand and NaCl (0 and 50 mM) | 3 weeks |
| [52] |
| CuO | 23–37 nm | Rice | Seed; 0, 0.1, 1.0, 10, 50, and 100 mg/L in 20% Hoagland solution for 18 days Root; soil medium with and without as (0 and 10 mg/kg) | 131 days |
| [49] |
| CuO | 40 nm | Lettuce | Root; Pristine and weathered (mixed with soil 70 days prior to use) NMs at 0 and 400 mg /kg soil co-contaminated with chlordane (concentration not provided) | 70 days |
| [54] |
| BC Ni/Fe BC-supported Ni/Fe | 28.4 nm (Ni/Fe NMs) | Chinese cabbage | Seed and root; 0 and 30 mg/kg soil contaminated with BDE209 | 20 days |
| [55] |
| nHAP | 20 ± 5 nm | Rice | Root; 100 mg/L Hoagland solution (5 days) followed by movement into solution with Pb(NO3)2 (0, 15, and 25 µM; 14 days) | 19 days |
| [56] |
| SiO2 | 25 nm | Barley | Seed and root; 3 mg/kg artificial soil with or without 120 mg NiO NMs/kg | 14 days |
| [51] |
| TiO2 (≥99% anatase) | 10–25 nm | Rice | Root; 500, 1,000, and 2,000 mg/L with tetracycline (TC; 0, 5, 10, and 20 mg/L) in ½-strength Hoagland solution | 10 days |
| [50] |
| ZnO CuO Cr2O3TiO2 Fe2O3 | 100 ± 25 nm 50 ± 10 nm 100 ± 30 nm 25 ± 6 nm 50 ± 15 nm | Cress Flax Wheat Cucumber | Seed; dispersions contain each NM singly or in combinations (ZnO or CuO NMs with Cr2O3, TiO2, or Fe2O3 NMs) at 10, 100, and 1,000 mg/L redistilled water | 3 days |
| [57] |
| ZnO CeO2La2O3CuO CdS QDs | <100 nm <25 nm 10-100 nm 40 nm <5 nm | Zucchini | Root; vermiculate with dispersion containing each NM individually and in binary combinations (500 mg NMs/L; 100 mg QDs/L) | 21 days |
| [22] |
| NM | Size | Plant Species | Exposure/Medium | Duration | NM Biotransformations | Reference |
|---|---|---|---|---|---|---|
| Metal-Based NMs | ||||||
| Ag Ag2S | 52 ± 1 nm 42 ± 5 nm | Wheat | Root; 30 µM Ag or Ag2S NMs in ¼-strength Hoagland solution | 3 weeks |
| [23] |
| Ag2S | 59 nm | Wheat Cucumber | Root; 20 mg/L nutrient solution | 7 days |
| [63] |
| CeO2 | 25.2 ± 2.3 nm | Cucumber | Root; split root hydroponics system (ultrapure water)—one half placed in 200 and 2000 mg CeO2/L; other half in ultrapure water | 3 days |
| [28] |
| CeO2 | 8 ± 1 nm | Wheat | S1 plants were grown to maturity (0, 125, and 500 mg/kg soil) and the seeds were cultivated in factorial combinations (1, 125, and 500 mg/kg) to maturity (S2 plants). | 90 days |
| [34] |
| CeO2 | Length: 67 ± 8 nm Diameter: 8 ± 1 nm | Barley | Root; 250 mg/kg soil | 60 days |
| [64] |
| CeO2 (bare and citrate-coated) | 3 ± 1 (bare) 3.9 ± 1.8 nm (citrate coated) | Fescue Tomato | Root; 1, 15, or 50 mg bare or citrate coated CeO/kg of either sandy soil with low NOM or clay-rich soil with high NOM | Not provided |
| [60] |
| CeO2 (3 surface coatings) | 4 nm | Wheat | Root; 20 mg/L ¼-strength Hoagland solution containing NMs functionalized with neutral, positive, or negative charge | 34 h |
| [61] |
| CuO | 25, 40, and <80 nm | Bean | Seed: 1, 10, 100, and 1000 mg Cu/L of aqueous medium for each size separately | 5 days |
| [21] |
| CuO | 43 ± 9 nm | Rice | Root; 50, 100, 500, and 1000 mg/kg soil | 7, 21, 60, and 88 days |
| [36] |
| CuO | 40 nm | Lettuce | Root; Pristine and weathered (mixed with soil 70 days prior to use) NMs at 0 and 400 mg /kg soil | 70 days |
| [54] |
| CuO | 40–60 nm | Lettuce Cabbage | Leaf; 0, 10, and 250 mg/plant (applied as dry particles to adaxial surfaces); plants grown in soil medium | 5, 10, or 15 days |
| [9] |
| ZnO | 20, 40, and 60 nm | Bean | Seed; 1, 10, 100, 1,000, and 5,000 mg/L deionized water for each NM size | 20 min; harvested after 5 days |
| [62] |
| ZnO | 20, 40, 60, and 300 nm | Bean | Root; 100 and 1000 mg/L aqueous medium with 20, 40, and 60 nm NMs (no surfactant) and 20, 40, and 300 nm (with surfactant) | 48 h |
| [59] |
| NM | Plant Species | Exposure/Medium | Duration | Aim of Application | Nano-/Commercial Advantage? | Reference |
|---|---|---|---|---|---|---|
| Carbon-Based NMs | ||||||
| ALG/CS CS/TPP | Bean | Seed; 1 h (ALG/CS in 11 mM CaCl2 and CS/TPP in 0.1% TPP) with or without encapsulation of gibberellic acid (GA3; 0.05%, 0.037%, 0.025%, and 0.012% in distilled water). | 1 h; harvested 7 days later |
| Yes | [75] |
| CNO | Gram | Sprouted seed; 0, 10, 20, and 30 µg/mL water. Transfer to soil after 10 days. | 10 days; harvested at maturity (~4 months) |
| NA | [29] |
| Zein NM-GRL, NM-R-CTL | Bean Tomato | Seed; Zein NMs, NM-GRL, and NM-R-CTL (0.05, 0.5, and 5 mg/mL agar medium) | 5 days |
| Yes | [74] |
| Metal-based NMs | ||||||
| BC Ni/Fe BC-supported Ni/Fe | Chinese cabbage | Seed and root; 0 and 30 mg/kg soil contaminated with decabromodiphenyl ether (BDE209) | 20 days |
| Yes | [55] |
| Cu-CNFs | Gram | Seed and root; 10–500 µg/mL aqueous medium | 20 days |
| Yes | [79] |
| Cu-chitosan | Maize | Seed; 0.01, 0.04, 0.08, 0.12, and 0.16%, w/v (4 h) Foliar; corresponding concentration until 35 days old | 95 days |
| Yes | [80] |
| CuO | Bean | Seed: 1, 10, 100, and 1000 mg Cu/L of aqueous medium for each size separately (25, 40, and <80 nm) | 5 days |
| Yes | [21] |
| CuO | Wheat | Root: ~500 mg/kg soil CuO NMs were either ‘fresh’ or ‘aged’ (added to soil 28 days before exposure) | 14 days |
| Yes | [27] |
| CuO NPs Cu3(PO4)2•3H2O nanosheets | Watermelon | Greenhouse experiments: Foliar: (1) dipped (0.6–0.8 mL; 10, 50, 100, 250, 500, and 1000 mg/L water) (2) sprayed 1 time (50, 500 mg/L); 3) sprayed 2 times (20, 200 mg/L) Root: 500 and 1000 mg/L Plants cultivated in soilless mix with Fusarium oxysporum. Field experiments: Foliar: 400 mg/L; with and without F. oxysporum in soil | 5 weeks |
| Yes | [78] |
| CuS (3 surface coatings) | Rice | Seed; fungi-infested seeds placed in dispersions containing CuS NMs with 3 coatings: PVP, GABA 4-aminobutyric acid), and citrate (tri-sodium citrate) at 3, 5, 7, 10, and 15 µg/mL | 1-2 h; harvested 10 days later |
| Yes | [76] |
| Cu/Zn | Winter wheat Stolichna and Acveduc ecotypes | Seed; 1:100 ratio of solution to water, followed by planting in sand medium with water. 8 days after emergence: plants subjected to drought conditions or normally watered for 3 days. | 4 h; harvested 11 days after seedling emergence |
| NA | [81] |
| GO-Ag | Rice | Seed; Ag NMs and GO-Ag at 1.25, 2.5, 5, and 10 µg/mL ultrapure water. Innoculation with bacterial leaf blight (Xanthomonas oryzae pv. Oryzae [Xoo]). | 6 days |
| Yes | [73] |
| GO Fe3O4 GO- Fe3O4 NMs | Grapevine | Leaf; Plants infected with Plasmopara viticola were sprayed with GO, Fe3O4, and GO-Fe3O4 at 0 and 250 µg/mL | 7 days |
| Yes | [82] |
| HA(+) HA(−) HA(0) | Sunflower | Root; 150 mg/kg of each type of HA in two types of P-deficient soil (Ultisol and Vertisol). | 35 days |
| Yes (Ultisol soil) No (Vertisol soil) | [77] |
| CNAD-MSNPs | Common pea | Seed; coated with alginate or alginate-CNAD-MSNPs (2 mg/mL MS agar medium); inoculated with Pseudomonas syringae pv. Pisi (pea blight; OD600 = 0.025) | 20 days; harvested after 4 weeks in soil |
| Yes | [71] |
| MSNs | Cucumber | Leaf; 0.5 mL of suspension (200 and 1000 mg/L deionized water) applied to the middle leaf after emergence of the 5th leaf | 14 days |
| Yes | [83] |
| ZnO | Bean | Seed; 1, 10, 100, 1000, and 5000 mg/L deionized water for three different sizes (20, 40, and 60 nm) | 20 min; harvested after 5 days |
| Yes | [62] |
| ZnO (bare, with a Zn3(PO4)2 shell, DEX-coated, and DEX-(SO4) coated) | Wheat | Seed; 100, 500, and 1000 mg Zn/L deionized water | 24 h; harvested when >65% of control seeds had radicle root at least 20 mm long |
| Yes | [70] |
| ZnO MWCNTs ZnO/MWCNTs nanocomposite | Onion | Seed; MWCNTs and ZnO/MWCNTs at 0, 2, 5, 10, 15, 20, and 40 µg/mL and ZnO at 20 µg/mL; seeds germinated under varying watering schedules (every 2nd or 4th days or after the 6th, or 8th day) | 20 h; harvested after 12 days |
| Yes, for the nano-composite under arid conditions | [72] |
| NM Material | Plant | Exposure Period/Route/Medium | NOAEL | LOAEL | Measured Adverse Effect(s) | Reference |
|---|---|---|---|---|---|---|
| Carbon-Based ENMs | ||||||
| C60 | Rice | 30 day exposure in soil | NA | 50 mg/kg | • Reduced root and shoot lengths; increased SOD activity | [88] |
| CB | Soybean | Up to 41 days root exposure in soil | 1000 mg/kg | 0.1 and 100 mg/kg | • Reduced plant growth, root nodulation, and N2 fixation potential. | [86] |
| Chitin | Wheat (MSW and LSW cultivars) | Full life-cycle root exposure in sandy soil | 0.02 g/kg | NA | [15] | |
| CNOs | Gram | 10 day sprouted seed exposure in water before transplantation to soil | 30 µg/mL | NA | [29] | |
| CNTs (carboxylated) | Collard greens | 42 days root exposure in soil | 500 mg/kg | NA | [25] | |
| GNPs | Soybean | Up to 41 days root exposure in soil | 100 mg/kg | mg/kg | • Reduced plant growth. | [86] |
| GO | Oats | 15 days in vermiculite | 40 mg/L | 200 mg/L | • Reduced chlorophyll contents and increased MDA activity | [92] |
| rGO | Rice | 30 day exposure in soil | NA | 50 mg/kg | • Reduced root and shoot lengths and shoot dry weight; increased SOD and POD activities | [88] |
| MWCNTs | Rice | 30 day exposure in soil | NA | 50 mg/kg | • Reduced root and shoot lengths; reduced SOD and POD activities | [88] |
| MWCNTs | Soybean | Up to 41 days root exposure in soil | NA | 0.1 mg/kg | • Reduced plant growth. | [86] |
| Expected environmental concentrations: | ||||||
| CNTs (EU averages) | ||||||
| surface water: 0.23 ng/L | ||||||
| WWTP effluent: 4.0 ng/L | ||||||
| WWTP biosludge: 0.15 mg/kg [86] | ||||||
| Carbon black | ||||||
| WWTP effluent: as low as 3.28–287.5 µg/L in London and as high as 5.91-673 µg/L in New York | ||||||
| WWTP biosludge: as low as 530–2250 mg/kg in Shanghai and as high as 1220–5240 mg/kg in New York [88] | ||||||
| Silver-based NMs | ||||||
| Ag | Cucumber | 7 day foliar exposure | NA | 4 mg/plant | • Increased MDA contents; visible leaf yellowing | [93] |
| Ag (2 nm) | Tomato | 2 weeks root exposure (uninoculated with mycorrhizal fungi) in soil | NA | 12 mg/kg | • Decreased shoot dry weight (12–36 mg/kg) | [18] |
| Ag (2 nm) | Tomato | 2 weeks root exposure (inoculated with mycorrhizal fungi) in soil | 12 mg/kg | 24 mg/kg | • Decreased shoot dry weight (24–36 mg/kg) | [18] |
| Ag (15 nm) | Tomato | 2 weeks root exposure (uninoculated with mycorrhizal fungi) in soil | 12 mg/kg | 24 mg/kg | • Decreased shoot dry weight (24–36 mg/kg) | [18] |
| Ag (15 nm) | Tomato | 2 weeks root exposure (inoculated with mycorrhizal fungi) in soil | 36 mg/kg | NA | [18] | |
| Ag with PEG coating | Tomato | 56–62 days root exposure in soil | NA | 10 mg/kg | • Reduced fruit yield and chlorophyll contents; increased oxidative stress parameters | [33] |
| Ag | Peanut | 98 days root exposure in soil | NA | 50 mg/kg | • Reduced growth and yield; increased antioxidant enzyme activities | [31] |
| Ag | Wheat | 4 month root exposure in soil | NA | 20 mg/kg | • Reduced growth | [32] |
| Expected environmental concentrations (EU averages): | ||||||
| Sewage treatment effluent: 1–104 ng//L | ||||||
| Surface (fresh) water: 0.03–3 ng/L | ||||||
| Sludge-treated soils (100% degradation after one year): 20–1661 ng/kg | ||||||
| Sludge-treated soils (100% persistence): 464–24,995 ng/kg [85] | ||||||
| Cerium-based NMs | ||||||
| CeO2 | Bean | 15 day root exposure in soil | NA | 250 mg/kg | • Reduced total chlorophyll and proline contents | [94] |
| CeO2 | Bean | 15 day foliar exposure | NA | 250 mg/plant | • Reduced anthocyanin, POD, and proline contents; reduced stomatal density | [94] |
| CeO2 | wheat | 90 days root exposure in soil | NA | 125 mg/kg exposure in 1st and 2nd generations | • Reduced grain nutrient quality | [34] |
| CeO2 | Soybean | 3 weeks root exposure in soil | 100 mg/kg | 500 mg/kg | • Reduced photosynthesis rate | [24] |
| PVP- CeO2 | Soybean | 3 weeks root exposure in soil | 100 mg/kg | 500 mg/kg | • Reduced photosynthesis rate | [24] |
| Expected environmental concentrations (EU averages): | ||||||
| Sewage treatment effluent: 20–889 ng/L | ||||||
| Sludge-treated soils (100% degradation after one year): 528–19,012 ng/kg | ||||||
| Sludge-treated soils (100% persistence): 11,212–560,423 ng/kg [85] | ||||||
| Copper-based NMs | ||||||
| CuO (aged) | Wheat | 2 weeks root exposure in soil | NA | 500 mg/kg | • Shorter root length | [27] |
| CuO (aged) | Lettuce | 70 days root exposure in soil | NA | 400 mg/kg | • Decreased biomass | [54] |
| CuO (unaged) | Wheat | 2 weeks root exposure in soil | 500 mg/kg | NA | [27] | |
| CuO (unaged) | Lettuce | 70 days root exposure in soil | NA | 400 mg/kg | • Decreased biomass | [54] |
| CuO | Rice | Up to 88 days root exposure in soil | 100 mg/kg | 500 mg/kg | • Decreased growth and yield | [36] |
| CuO | Bell pepper | 90 days root exposure in soil | 250 mg/kg | 500 mg/kg | • Reduced Zn contents in fruits and leaves | [37] |
| CuO | Cabbage | Up to 15 days foliar exposure | 10 mg/plant | 250 mg/plant | • Decreased gas and water exchange from blocked stomata and reduced dry weight | [9] |
| CuO | Lettuce | Up to 15 days foliar exposure | 10 mg/plant | 250 mg/plant | • Decreased gas and water exchange from blocked stomata and reduced dry weight | [9] |
| CuO | Peanut | 145 days seed and root exposure in soil | NA | 50 mg/kg | • Decreased total amino acid contents and altered fatty acid profile in peanut grains | [46] |
| Cu(OH)2 | Spinach | 7 days foliar exposure | 1.8 mg/plant | 18 mg/plant | • Reduced contents of antioxidant compounds and amino acids | [38,89] |
| Cu(OH)2 | Corn | 7 days foliar exposure | 10 mg/plant | 100 mg/plant | • Reduced leaf biomass and photosynthetic pigments | [89] |
| Cu(OH)2 | Cucumber | 7 days foliar exposure | 25 mg/plant | NA | [95] | |
| Expected environmental concentrations: | ||||||
| WWTP effluent: Cu + CuOx: as low as >0.001–0.02 µg/L in London and as high as >0.001–0.03 µg/L in New York and Shanghai | ||||||
| WWTP biosolids: Cu + CuOx: as low as >0.0–0.12 mg/kg in Shanghai to as high as 0.01–0.24 mg/kg in New York [88] | ||||||
| Titanium-based NMs | ||||||
| TiO2 | Rice | 130 days root exposure in soil (low CO2 conditions) | 200 mg/kg | NA | [39] | |
| TiO2 | Rice | 130 days root exposure in soil (high CO2 conditions) | NA | 50 mg/kg | • Decreased plant biomass and yield | [39] |
| TiO2 | Peanut | 145 days seed and root exposure in soil | 50 mg/kg | 500 mg/kg | • Decreased total amino acid contents in peanut grains | [46] |
| TiO2 | Rice | Full life-cycle root exposure in soil | 750 mg/kg | NA | [40] | |
| Expected environmental concentrations (EU averages): | ||||||
| WWTP effluent: 16 μg/L | ||||||
| WWTP sludge: 170 mg/kg [86] | ||||||
| Zinc-based NMs | ||||||
| ZnO | Bean | Up to 90 days root exposure in acidic soil | NA | 3 mg/kg | • Decreased chlorophyll b and protein contents and altered antioxidant enzyme activity levels in leaves (increased GPOD activity at 15 and 30 days) | [14] |
| ZnO | Bean | Up to 90 days root exposure in calcareous soil | NA | 3mg/kg | • Increased GPOD activity at 15 days | [14] |
| ZnO | Tomato | Up to 90 days root exposure in acidic soil | NA | 3 mg/kg | • Increased MDA (indicative of lipid peroxidation) at 90 days. Decreased GPOD and increased CAT activities at 15 days. Plants died at ≥100 mg/kg | [14] |
| ZnO | Tomato | Up to 90 days root exposure in calcareous soil | NA | 3 mg/kg | • Increased CAT activity at 15 and 30 days. | [14] |
| ZnO | Wheat | Full life-cycle exposure in fresh soil | 6 mg/kg | NA | [41] | |
| ZnO | Wheat | Full life-cycle exposure in weathered soil | 5.98 mg/kg | NA | [41] | |
| ZnO | Sorghum | Exposure time not provided. Foliar exposure | 100 mL equivalent amount of Zn applied to roots | NA | [42] | |
| ZnO | Sorghum | Exposure time not provided. Root exposure in soil | 6 mg/kg | NA | [42] | |
| ZnO | Maize | 9 weeks root exposure in soil without organic P or AMF | 500 mg/kg | NA | [43] | |
| ZnO | Maize | 9 weeks root exposure in soil with organic P, but without AMF | 500 mg/kg | NA | [43] | |
| ZnO | Maize | 9 weeks root exposure in soil without organic P, but with AMF | 500 mg/kg | NA | [43] | |
| ZnO | Maize | 9 weeks root exposure in soil with both organic P and AMF | 500 mg/kg | NA | [43] | |
| ZnO | Zucchini | 21 days root exposure in vermiculate with dispersion | 500 mg/L | NA | [22] | |
| ZnO | Fenugreek (no inoculation with Rhizobium melliloti) | 60 days root exposure in sand medium | NA | 125 mg/kg | • Decreased nodule biomass | [19] |
| ZnO | Fenugreek (inoculated with Rhizobium melliloti) | 60 days root exposure in sand medium | 125 mg/kg | 250 mg/kg | • Decreased nodule biomass | [19] |
| ZnO (bare) | Bean | >45 days root exposure in soil for S1 plants; unexposed S2 plants analyzed | 500 mg/kg | NA | • Reduced Ni content in bean grains | [44] |
| ZnO (hydrophobically-coated) | Bean | >45 days root exposure in soil for S1 plants; unexposed S2 plants analyzed | NA | 125 mg/kg | [44] | |
| ZnO (bare) | Bean | >45 days root exposure until maturity in natural soil | NA | 125 mg/kg | [26] | |
| ZnO (hydrophobically-coated) | Bean | >45 days root exposure until maturity in natural soil | 500 mg/kg | NA | [26] | |
| ZnO (bare) | Bean | >45 days root exposure until maturity in organic-matter enriched soil | 500 mg/kg | NA | [26] | |
| ZnO (hydrophobically-coated) | Bean | >45 days root exposure until maturity in organic-matter enriched soil | 500 mg/kg | NA | [26] | |
| Expected environmental concentrations (EU averages): | ||||||
| WWTP effluent: 2.3 μg/L | ||||||
| WWTP sludge: 24 mg/kg [86] | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kranjc, E.; Drobne, D. Nanomaterials in Plants: A Review of Hazard and Applications in the Agri-Food Sector. Nanomaterials 2019, 9, 1094. https://doi.org/10.3390/nano9081094
Kranjc E, Drobne D. Nanomaterials in Plants: A Review of Hazard and Applications in the Agri-Food Sector. Nanomaterials. 2019; 9(8):1094. https://doi.org/10.3390/nano9081094
Chicago/Turabian StyleKranjc, Eva, and Damjana Drobne. 2019. "Nanomaterials in Plants: A Review of Hazard and Applications in the Agri-Food Sector" Nanomaterials 9, no. 8: 1094. https://doi.org/10.3390/nano9081094
APA StyleKranjc, E., & Drobne, D. (2019). Nanomaterials in Plants: A Review of Hazard and Applications in the Agri-Food Sector. Nanomaterials, 9(8), 1094. https://doi.org/10.3390/nano9081094







