Nanophosphor-Based Contrast Agents for Spectral X-ray Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
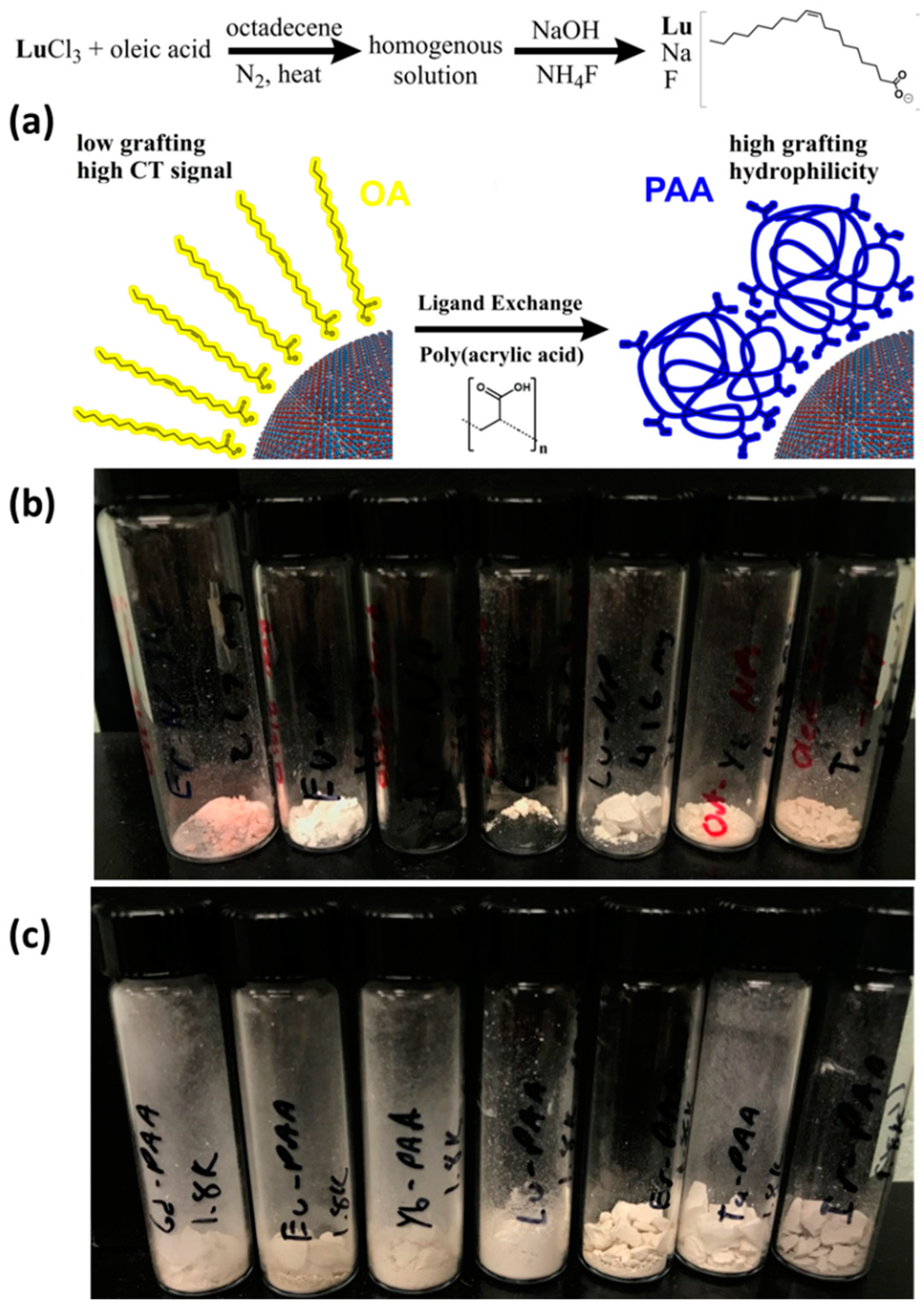
2.2. Synthesis of Oleic Acid Stabilized Nanophosphors (OA-Stabilized NPhs)
2.3. Water-Dispersible Poly(acrylic acid) Stabilized Nanophosphors (PAA-Stabilized NPhs)
2.4. Cell Studies
2.5. Gelatin Phantoms
2.6. Characterization
2.7. Imaging Studies
3. Results and Discussion
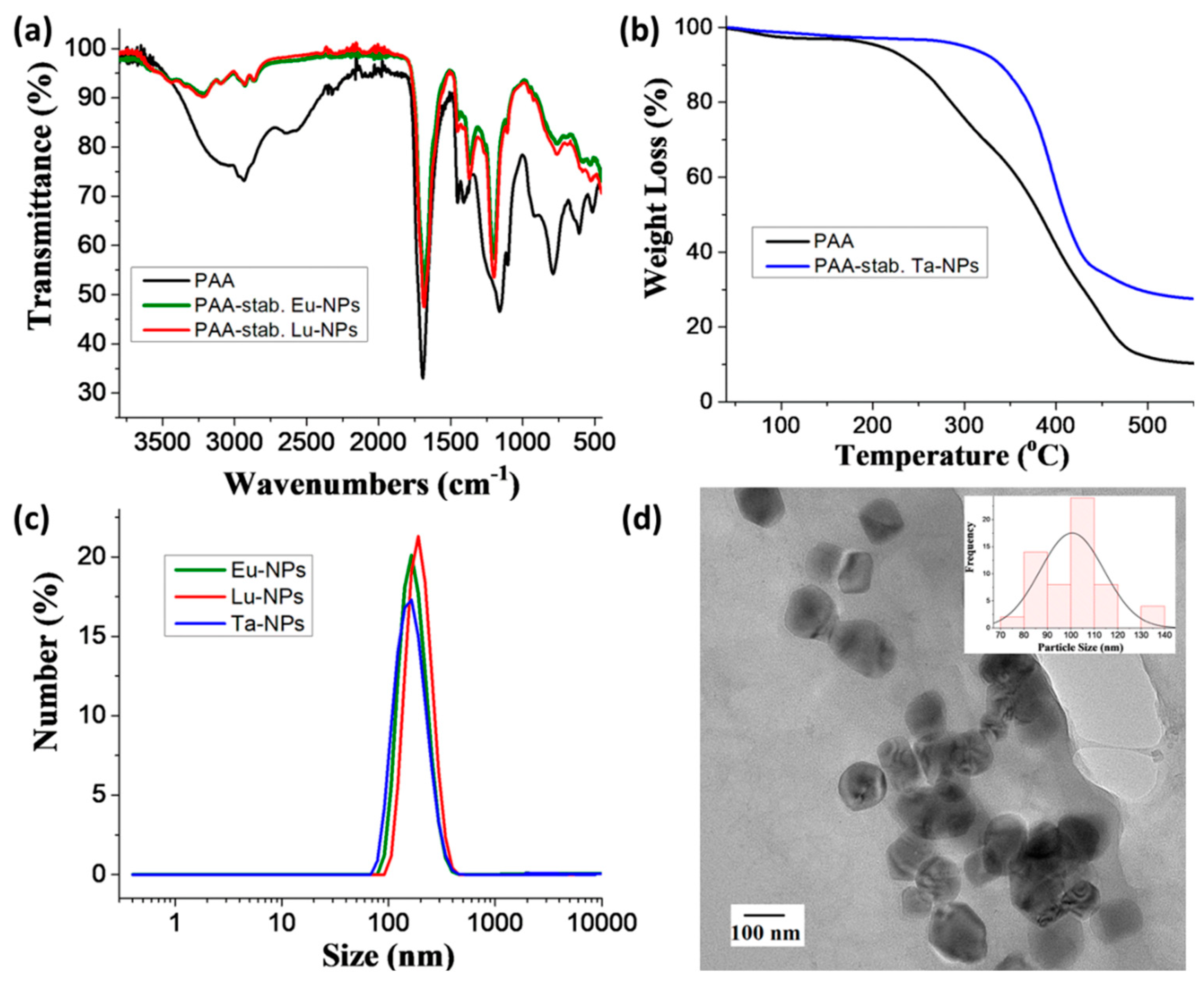
3.1. Synthesis and Characterization of OA-Stabilized NPhs
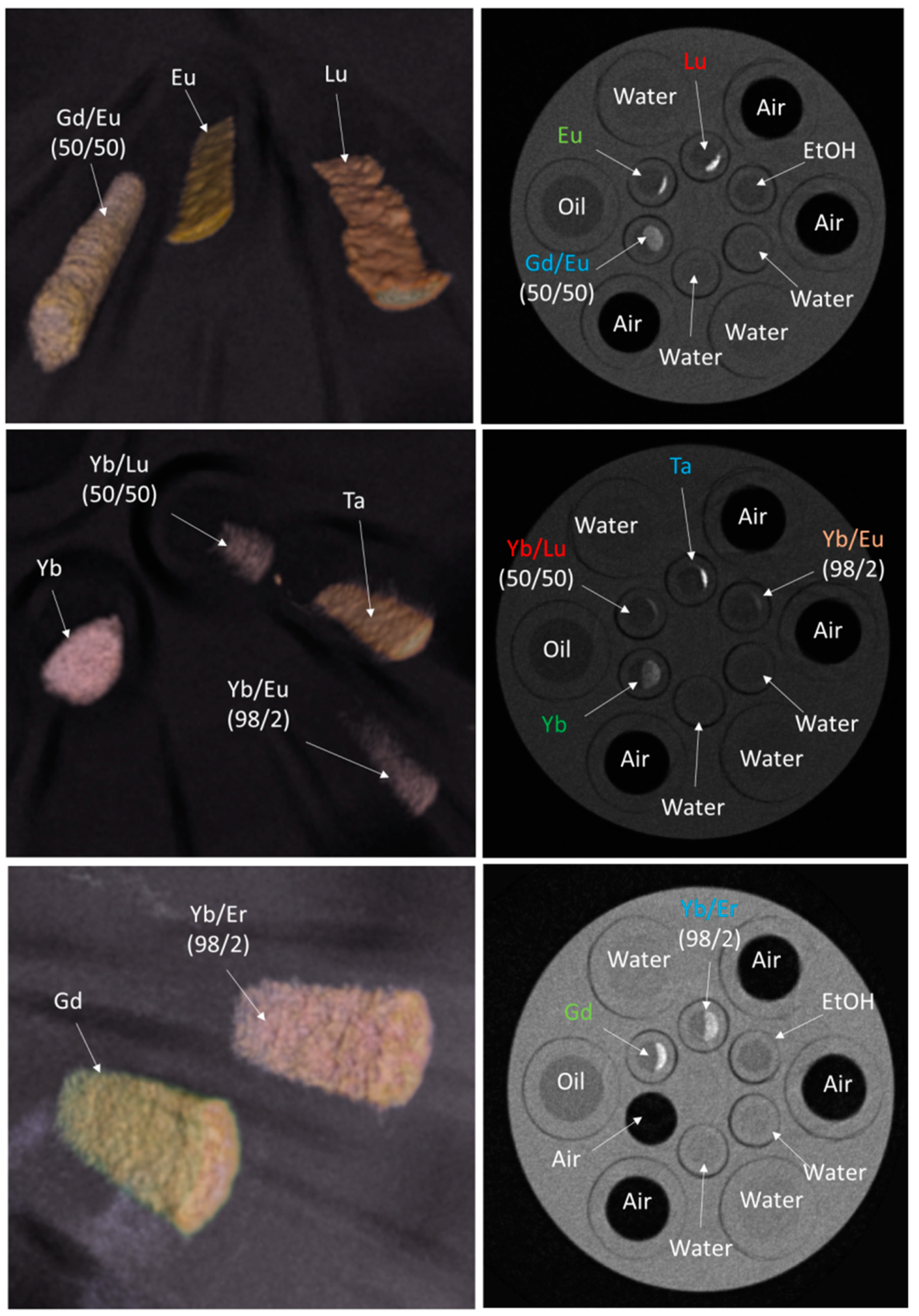
3.2. CT Imaging of OA-Stabilized NPh Contrast Agents
3.3. Ligand Exchange and Characterization of PAA-Stabilized NPhs
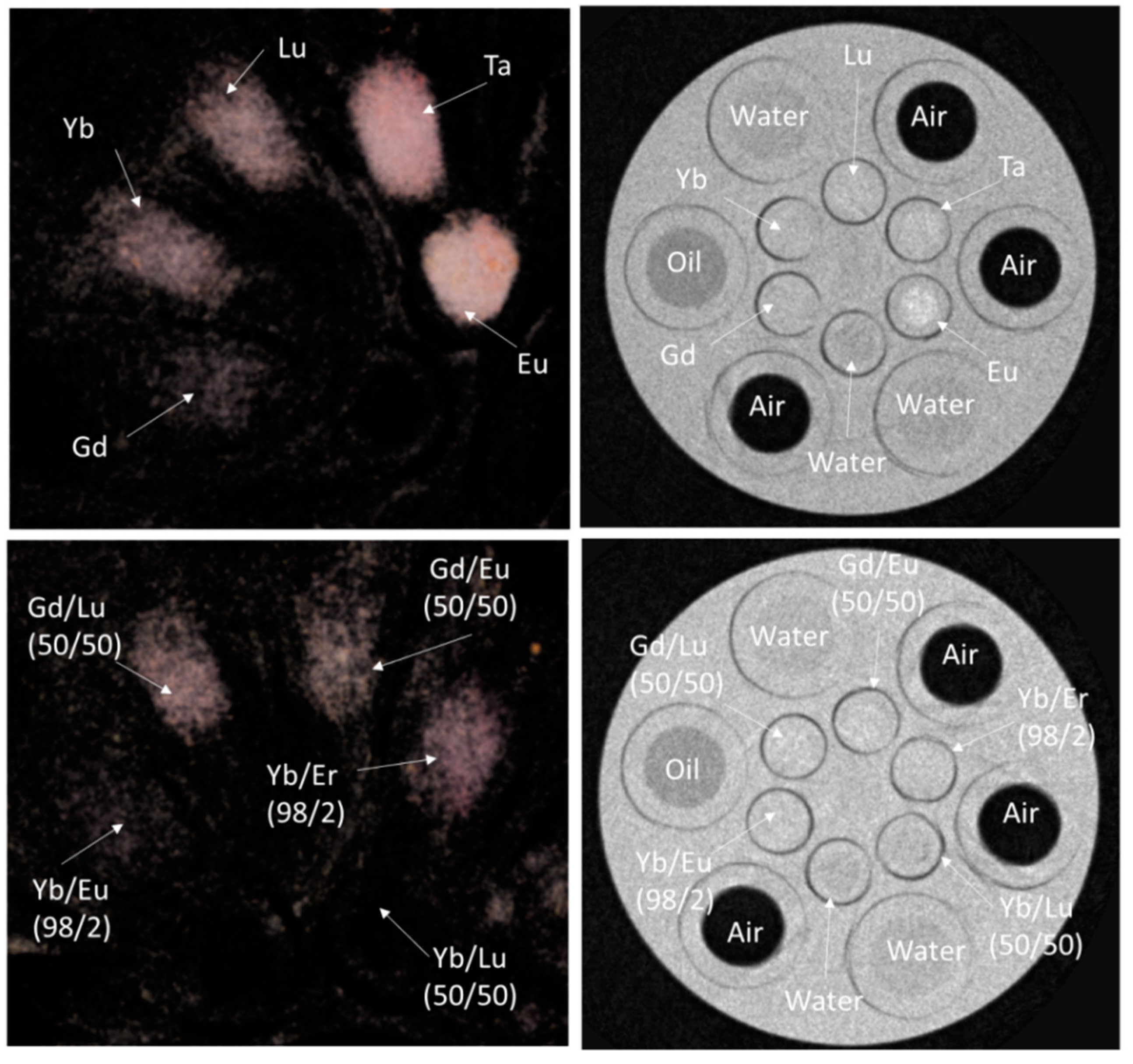
3.4. CT Imaging of PAA-Stabilized NPh Contrast Agents
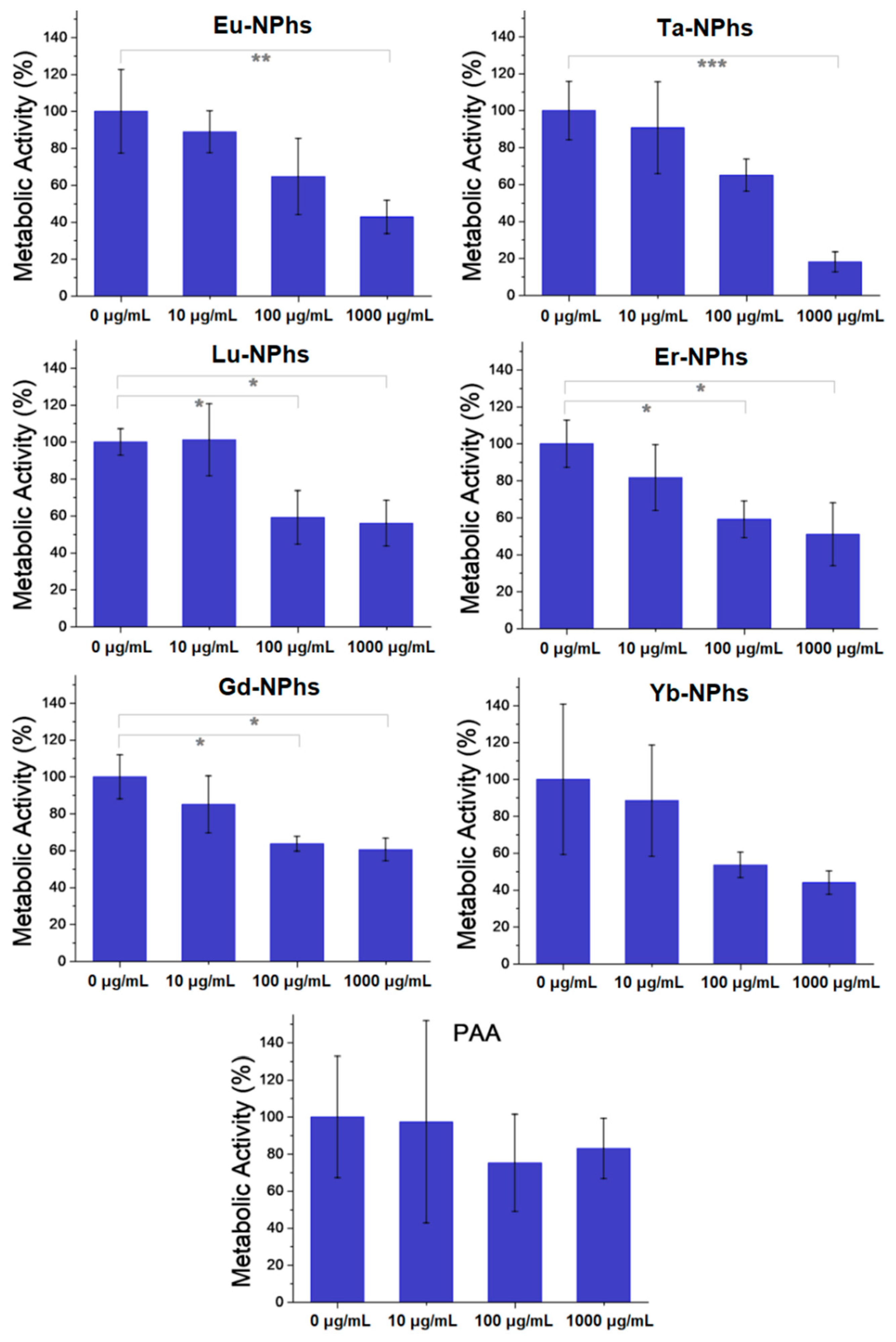
3.5. Cell Viability of PAA-Stabilized NPhs
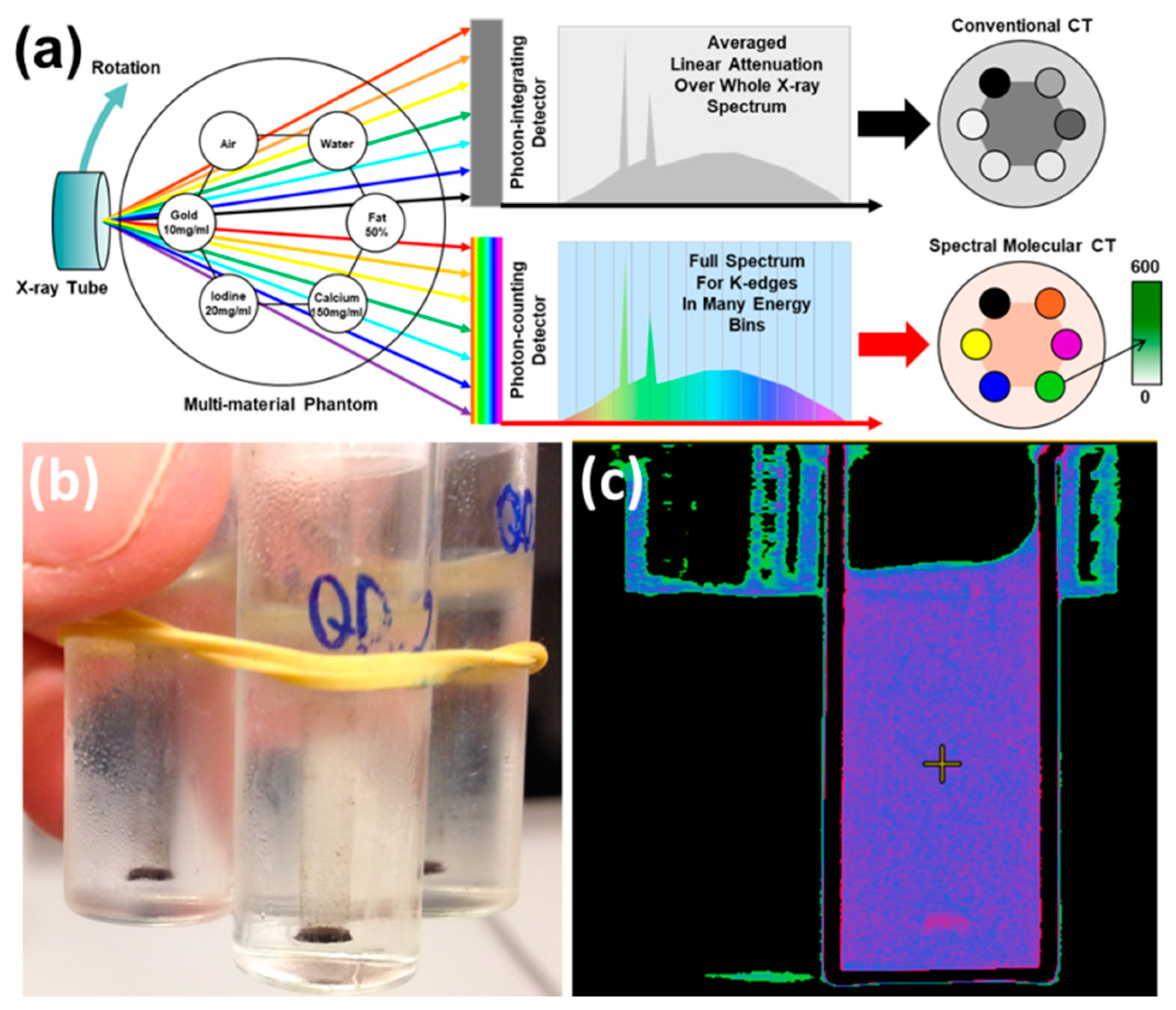
3.6. Contrast Efficacy of NPhs Embedded into Phantoms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Markou, A.; Liang, Y.; Lianidou, E. Prognostic, therapeutic, and diagnostic potential of microRNAs in lung cancer. Clin. Chem. Lab. Med. 2011, 49, 1591–1603. [Google Scholar] [CrossRef] [PubMed]
- Mastoraki, S.; Strati, A.; Tzanikou, E.; Chimonidou, M.; Politaki, E.; Voutsina, A.; Psyrri, A.; Georgoulias, V.; Lianidou, E. ESR1 methylation: A liquid biopsy–based epigenetic assay for the follow-up of patients with metastatic breast cancer receiving endocrine treatment. Clin. Cancer Res. 2018, 24, 1500–1510. [Google Scholar] [CrossRef] [PubMed]
- Schmid, D.; Park, C.G.; Hartl, C.A.; Subedi, N.; Cartwright, A.N.; Puerto, R.B.; Zheng, Y.; Maiarana, J.; Freeman, G.J.; Wucherpfennig, K.W.; et al. T cell-targeting nanoparticles focus delivery of immunotherapy to improve antitumor immunity. Nat. Commun. 2017, 8, 1747. [Google Scholar] [CrossRef] [PubMed]
- Joseph, C.S.; Patel, R.; Neel, V.A.; Giles, R.H.; Yaroslavsky, A.N. Imaging of ex vivo nonmelanoma skin cancers in the optical and terahertz spectral regions optical and Terahertz skin cancers imaging. J. Biophotonics 2014, 7, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Melikechi, N.; Markushin, Y.; Connolly, D.; Lasue, J.; Ewusi-Annan, E.; Makrogiannis, S. Age specific classification of discrimination of blood plasma samples of healthy and ovarian cancer prone mice using laser-induced breakdown spectroscopy. Spectrochim. Acta Part B At. Spectrosc. 2016, 123, 33–41. [Google Scholar] [CrossRef]
- Gaudiuso, R.; EboEwusi-Annan, E.; Melikechi, N.; Sun, X.; Liu, B.; Campesato, L.F.; Merghoub, T. Using LIBS to diagnose melanoma in biomedical fluids deposited on solid substrates: Limits of direct spectral analysis and capability of machine learning. Spectrochim. Acta Part B At. Spectrosc. 2018, 146, 106–114. [Google Scholar] [CrossRef]
- Spirou, S.V.; Costa Lima, S.A.; Bouziotis, P.; Vranješ-Djurić, S.; Efthimiadou, E. Κ.; Laurenzana, A.; Barbosa, A.I.; Garcia-Alonso, I.; Jones, C.; Jankovic, D.; et al. Recommendations for in vitro and in vivo testing of magnetic nanoparticle hyperthermia combined with radiation therapy. Nanomaterials 2018, 8, 306. [Google Scholar] [CrossRef]
- Toniolo, G.; Efthimiadou, E.K.; Kordas, G.; Chatgilialoglu, C. Development of multi-layered and multi-sensitive polymeric nanocontainers for cancer therapy: In vitro evaluation. Sci. Rep. 2018, 8, 14704. [Google Scholar] [CrossRef]
- Kalender, W.A. X-ray computed tomography. Phys. Med. Biol. 2006, 51, R29–R43. [Google Scholar] [CrossRef]
- Momose, A.; Takeda, T.; Itai, Y.; Hirano, K. Phase-contrast X-ray computed tomography for observing biological soft tissues. Nat. Med. 1996, 2, 473–475. [Google Scholar] [CrossRef]
- Paulus, M.J.; Gleason, S.S.; Kennel, S.J.; Hunsicker, P.R.; Johnson, D.K. High resolution X-ray computed tomography: An emerging tool for small animal cancer research. Neoplasia 2000, 2, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Haller, C.; Hizoh, I. Radiocontrast-induced renal tubular cell apoptosis: Hypertonic versus oxidative stress. Investig. Radiol. 2002, 37, 428–434. [Google Scholar]
- Haller, C.; Hizoh, I. The cytotoxicity of iodinated radiocontrast agents on renal cells in vitro. Investig. Radiol. 2004, 39, 149–154. [Google Scholar] [CrossRef]
- de Vries, A.; Custers, E.; Lub, J.; van den Bosch, S.; Nicolay, K.; Grüll, H. Block-copolymer-stabilized iodinated emulsions for use as CT contrast agents. Biomaterials 2010, 31, 6537–6544. [Google Scholar] [CrossRef] [PubMed]
- Popovtzer, R.; Agrawal, A.; Kotov, N.A.; Popovtzer, A.; Balter, J.; Carey, T.E.; Kopelman, R. Targeted gold nanoparticles enable molecular CT imaging of cancer. Nano Lett. 2008, 8, 4593–4596. [Google Scholar] [CrossRef] [PubMed]
- Janib, S.M.; Moses, A.S.; MacKay, J.A. Imaging and drug delivery using theranostic nanoparticles. Adv. Drug Deliv. Rev. 2010, 62, 1052–1063. [Google Scholar] [CrossRef]
- He, M.; Huang, P.; Zhang, C.; Hu, H.; Bao, C.; Gao, G.; He, R.; Cui, D. Dual phase-controlled synthesis of uniform lanthanide-doped NaGdF4 upconversion nanocrystals via an OA/ionic liquid two-phase system for in vivo dual-modality imaging. Adv. Funct. Mater. 2011, 21, 4470–4477. [Google Scholar] [CrossRef]
- Zhou, J.; Zhu, X.; Chen, M.; Sun, Y.; Li, F. Water-stable NaLuF4-based upconversion nanophosphors with long-term validity for multimodal lymphatic imaging. Biomaterials 2012, 33, 6201–6210. [Google Scholar] [CrossRef]
- Liu, Y.; Ai, K.; Liu, J.; Yuan, Q.; He, Y.; Lu, L. A high-performance ytterbium-based nanoparticulate contrast agent for in vivo X-ray computed tomography imaging. Angew. Chem. Int. Ed. 2012, 51, 1437–1442. [Google Scholar] [CrossRef]
- Davies, A.; Lewis, D.J.; Watson, S.P.; Thomas, S.G.; Pikramenou, Z. pH-controlled delivery of luminescent europium coated nanoparticles into platelets. Proc. Natl. Acad. Sci. USA 2012, 109, 1862–1867. [Google Scholar] [CrossRef]
- Lewis, D.G.; Pikramenou, Z. Lanthanide-coated gold nanoparticles for biomedical applications. Coord. Chem. Rev. 2014, 273–274, 213–225. [Google Scholar] [CrossRef]
- Heer, S.; Kömpe, K.; Güdel, H.U.; Haase, M. Highly efficient multicolour upconversion emission in transparent colloids of lanthanide-doped NaYF4 nanocrystals. Adv. Mater. 2004, 16, 2102–2105. [Google Scholar] [CrossRef]
- Kramer, K.W.; Biner, D.; Frei, G.; Güdel, H.U.; Hehlen, M.P.; Luthi, S.R. Hexagonal sodium yttrium fluoride based green and blue emitting upconversion phosphors. Chem. Mater. 2004, 16, 1244–1251. [Google Scholar] [CrossRef]
- Zhang, Y.-W.; Sun, X.; Si, R.; You, L.-P.; Yan, C.-H. Single-crystalline and monodisperse LaF3 triangular nanoplates from a single-source precursor. J. Am. Chem. Soc. 2005, 127, 3260–3261. [Google Scholar] [CrossRef] [PubMed]
- Boyer, J.C.; Vetrone, F.; Cuccia, L.A.; Capobianco, J.A. Synthesis of colloidal upconverting NaYF4 nanocrystals doped with Er3+, Yb3+ and Tm3+, Yb3+ via thermal decomposition of lanthanide trifluoroacetate precursors. J. Am. Chem. Soc. 2006, 128, 7444–7445. [Google Scholar] [CrossRef] [PubMed]
- Nyk, M.; Kumar, R.; Ohulchanskyy, T.Y.; Bergey, E.J.; Prasad, P.N. High contrast in vitro and in vivo photoluminescence bioimaging using near infrared to near infrared up-conversion in Tm3+ and Yb3+ doped fluoride nanophosphors. Nano Lett. 2008, 11, 3834–3838. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Chen, H.; Hu, H.; Yu, M.; Li, F.; Zhang, Q.; Zhou, Z.; Yi, T.; Huang, C.N. Versatile synthesis strategy for carboxylic acid-functionalized upconverting nanophosphors as biological labels. J. Am. Chem. Soc. 2008, 130, 3023–3029. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Chen, Z.; Tian, Q.; Cao, T.; Xu, C.; Li, F. High contrast upconversion luminescence targeted imaging in vivo using peptide-labeled nanophosphors. Anal. Chem. 2009, 81, 8687–8694. [Google Scholar] [CrossRef]
- Xiong, L.; Yang, T.; Yang, Y.; Xu, C.; Li, F. Long-term in vivo biodistribution imaging and toxicity of polyacrylic acid-coated upconversion nanophosphors. Biomaterials 2010, 27, 7078–7084. [Google Scholar] [CrossRef]
- Chen, G.; Shen, J.; Ohulchanskyy, T.Y.; Patel, N.J.; Kutikov, A.; Li, Z.; Song, J.; Pandey, R.K.; Agren, H.; Prasad, P.N.; et al. (a-NaYbF4:Tm3+)/CaF2 core/shell nanoparticles with efficient near-infrared to near-infrared upconversion for high-contrast deep tissue bioimaging. ACS Nano 2012, 6, 8280–8287. [Google Scholar] [CrossRef]
- Roessl, E.; Proksa, R. K-edge imaging in X-ray computed tomography using multi-bin photon counting detectors. Phys. Med. Biol. 2007, 52, 4679–4696. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Wei, B.; Cong, W.; Wang, G. Optimization of K-edge imaging with spectral CT. Med. Phys. 2012, 39, 6572–6579. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Yu, H.; Bennett, J.; Ronaldson, P.; Zainon, R.; Butler, A.; Butler, P.; Wei, B.; Wang, G. Energy-discriminative performance of a spectral micro-CT system. J. X-ray Sci. Technol. 2013, 21, 335–345. [Google Scholar]
- Taguchi, K.; Iwanczyk, J.S. Vision 20/20: Single photon counting X-ray detectors in medical imaging. Med. Phys. 2013, 40, 100901. [Google Scholar] [CrossRef]
- Cammin, J.; Kappler, S.; Weidinger, T.; Taguchi, K. Photon-counting CT: Modeling and compensating of spectral distortion effects. In Proceedings of the SPIE, Orlando, FL, USA, 21–26 February 2015; Volume 9412, p. 941250. [Google Scholar]
- Cammin, J.; Kappler, S.; Weidinger, T.; Taguchi, K. Evaluation of models of spectral distortions in photon-counting detectors for computed tomography. J. Med. Imaging 2016, 3, 023503. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lin, T.H.; Cheng, P.; Shinozaki, D.M. A general cone-beam reconstruction algorithm. IEEE Trans. Med. Imaging. 1993, 12, 486–496. [Google Scholar] [CrossRef]
- Xu, Q.; Yu, H.; Mou, X.; Zhang, L.; Hsieh, J.; Wang, G. Low-dose X-ray CT reconstruction via Dictionary Learning. IEEE Trans. Med. Imaging. 2012, 31, 1682–1697. [Google Scholar]
- Feng, P.; Cong, W.X.; Wei, B.A.; Wang, G. Analytic comparison between X-Ray fluorescence CT and K-edge CT. IEEE Trans. Biomed. Eng. 2014, 61, 975–985. [Google Scholar] [CrossRef]
- Meng, B.; Cong, W.X.; Xi, Y.; De Man, B.; Wang, G. Energy window optimization for X-ray K-edge tomographic imaging. IEEE Trans. Biomed. Eng. 2016, 63, 1623–1630. [Google Scholar] [CrossRef]
- Zhang, Y.; Mou, X.; Wang, G.; Yu, H. Tensor-based dictionary learning for spectral CT reconstruction. IEEE Trans. Med. Imaging 2017, 36, 142–154. [Google Scholar] [CrossRef]
- Meng, B.; Cong, W.; Xi, Y.; De Man, B.; Yang, J.; Wang, G. Model and reconstruction of a K-edge contrast agent distribution with an X-ray photon-counting detector. Opt. Express 2017, 25, 9378–9392. [Google Scholar] [CrossRef] [PubMed]
- Getzin, M.; Garfield, J.J.; Rundle, D.S.; Krueger, U.; Gkikas, M.; Wang, G. Increased separability of K-edge nanoparticles by photon-counting detectors for spectral micro-CT. J. X-ray Sci. Technol. 2018, 26, 707–726. [Google Scholar] [CrossRef] [PubMed]
- Cormode, D.P.; Si-Mohamed, S.; Bar-Ness, D.; Sigovan, M.; Naha, P.C.; Balegamire, J.; Lavenne, F.; Coulon, P.; Roessl, E.; Bartels, M.; et al. Multicolor spectral photon-counting computed tomography: In vivo dual contrast imaging with a high count rate scanner. Sci. Rep. 2017, 7, 7–4784. [Google Scholar] [CrossRef] [PubMed]
- Si-Mohamed, S.; Cormode, D.P.; Bar-Ness, D.; Sigovan, M.; Naha, P.C.; Langlois, J.B.; Chalabreysse, L.; Coulon, P.; Blevis, I.; Roessl, E.; et al. Evaluation of spectral photon counting computed tomography K-edge imaging for determination of gold nanoparticle biodistribution in vivo. Nanoscale 2017, 9, 18246–18257. [Google Scholar] [CrossRef] [PubMed]
- Nara, M.; Tanokura, M. Infrared spectroscopic study of the metal-coordination structures of calcium-binding proteins. Biochem. Biophys. Res. Commun. 2008, 369, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Cotton, F.A.; Wilkinson, G.; Murillo, C.A.; Bochmann, M. Advanced Inorganic Chemistry, 6th ed.; Wiley: New York, NY, USA, 1988. [Google Scholar]
- Liu, B.; Chen, Y.; Li, C.; He, F.; Hou, Z.; Huang, S.; Zhu, H.; Chen, X.; Lin, J. Poly(acrylic acid) modification of Nd3+-sensitized upconversion nanophosphors for highly efficient UCL imaging and pH-responsive drug delivery. Adv. Funct. Mater. 2015, 25, 4717–4729. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Zhu, Y. One-pot synthesis and strong near-infrared upconversion luminescence of poly(acrylic acid)-functionalized YF3:Yb3+/Er3+ nanocrystals. Nano. Res. 2010, 3, 317–325. [Google Scholar] [CrossRef]
- Celebi, S.; Erdamar, A.K.; Sennaroglu, A.; Kurt, A.; Yagci, H.A. Synthesis and characterization of poly(acrylic acid) stabilized cadmium sulfide quantum dots. J. Phys. Chem. B 2007, 111, 12668–12675. [Google Scholar] [CrossRef]
- Iatrou, H.; Dimas, K.; Gkikas, M.; Tsimblouli, C.; Sofianopoulou, S. Polymersomes from polypeptide containing triblock Co- and terpolymers for drug delivery against pancreatic cancer: Asymmetry of the external hydrophilic blocks. Macromol. Biosci. 2014, 14, 1222–1238. [Google Scholar] [CrossRef]
- Stephan, M.T.; Moon, J.J.; Um, S.H.; Bershteyn, A.; Irvine, D.J. Therapeutic cell engineering with surface-conjugated synthetic nanoparticles. Nat. Med. 2010, 16, 1035–1041. [Google Scholar] [CrossRef]
- Moon, J.J.; Huang, B.; Irvine, D.J. Engineering nano- and microparticles to tune immunity. Adv. Mater. 2012, 24, 3724–3746. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Chen, L.; Thompson, D.M.; Montoya, L.D. Effect of particle size on in vitro cytotoxicity of titania and alumina nanoparticles. J. Exp. Nanosci. 2012, 9, 625–638. [Google Scholar] [CrossRef]
- Watanabe, M.; Yoneda, M.; Morohashi, A.; Hori, Y.; Okamoto, D.; Sato, A.; Kurioka, D.; Nittami, T.; Hirokawa, Y.; Shiraishi, T.; et al. Effects of Fe3O4 magnetic nanoparticles on A549 cells. Int. J. Mol. Sci. 2013, 8, 15546–15560. [Google Scholar] [CrossRef] [PubMed]
- Gkikas, M.; Peponis, T.; Mesar, T.; Hong, C.; Avery, R.K.; Roussakis, E.; Yoo, H.-J.; Parakh, A.; Patino, M.; Sahani, D.V.; et al. Systemically administered hemostatic nanoparticles for identification and treatment of internal bleeding. ACS Biomater. Sci. Eng. 2019, 55, 2563–2576. [Google Scholar] [CrossRef]
| Yb% | Gd% | Er% | Eu% | Lu% | Ta% | I% | Atten. Coeff. 1 (cm−1) | Hmg 2 | D (g/mL) | K-edge (keV) |
|---|---|---|---|---|---|---|---|---|---|---|
| 100 | - | - | - | - | - | - | 0.206 ± 0.015 | + | 6.9 | 61.3 |
| - | 100 | - | - | - | - | - | 0.268 ± 0.024 | + | 7.9 | 50.3 |
| - | - | 100 | - | - | - | - | - | - | 6.9 | 57.5 |
| - | - | - | 100 | - | - | - | 0.450 ± 0.107 | - | 5.2 | 48.5 |
| - | - | - | - | 100 | - | - | 0.657 ± 0.087 | - | 9.8 | 63.3 |
| - | - | - | - | - | 100 | - | 1.020 ± 0.140 | - | 16.7 | 67.4 |
| - | - | - | - | - | - | 100 3 | 0.234 ± 0.019 | + | 4.9 | 33.2 |
| 98 | - | 2 | - | - | - | - | 0.181 ± 0.014 | + | - | - |
| 98 | - | - | 2 | - | - | - | 0.173 ± 0.023 | - | - | - |
| 50 | - | - | 50 | - | - | 0.225 ± 0.021 | - | - | - | |
| 50 | - | - | - | 50 | - | - | 0.141 ± 0.031 | - | - | - |
| - | 50 | - | 50 | - | - | - | 0.227 ± 0.025 | + | - | - |
| Yb% | Gd% | Er% | Eu% | Lu% | Ta% | I% | Atten. Coeff. 1 (cm−1) | Hmg 2 | K-edge (keV) | PAA (%) wt Loss 3 |
|---|---|---|---|---|---|---|---|---|---|---|
| 100 | - | - | - | - | - | - | 0.027 ± 0.020 | + | 61.3 | 68.1 ± 0.4 |
| - | 100 | - | - | - | - | - | 0.028 ± 0.021 | + | 50.3 | 74.3 ± 1.0 |
| - | - | 100 | - | - | - | - | 0.067 ± 0.019 | + | 57.5 | 71.8 ± 1.3 |
| - | - | - | 100 | - | - | - | 0.086 ± 0.021 | + | 48.5 | 72.0 ± 1.3 |
| - | - | - | - | 100 | - | - | 0.036 ± 0.014 | + | 63.3 | 71.3 ± 0.2 |
| - | - | - | - | - | 100 | - | 0.029 ± 0.013 | + | 67.4 | 68.9 ± 0.3 |
| - | - | - | - | - | - | 100 4 | 0.234 ± 0.019 | + | 33.2 | - |
| 98 | - | 2 | - | - | - | - | 0.033 ± 0.015 | + | - | - |
| 98 | - | - | 2 | - | - | - | 0.005 ± 0.013 | + | - | - |
| 50 | - | - | 50 | - | - | 0.041 ± 0.021 | + | - | - | |
| 50 | - | - | - | 50 | - | - | 0.009 ± 0.013 | + | - | - |
| - | 50 | - | 50 | - | - | - | 0.043 ± 0.021 | + | - | - |
| - | - | - | - | 100 5 | - | - | 0.303 ± 0.017 | + | 63.3 | 18.1 ± 0.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, K.; Getzin, M.; Garfield, J.J.; Suvarnapathaki, S.; Camci-Unal, G.; Wang, G.; Gkikas, M. Nanophosphor-Based Contrast Agents for Spectral X-ray Imaging. Nanomaterials 2019, 9, 1092. https://doi.org/10.3390/nano9081092
Smith K, Getzin M, Garfield JJ, Suvarnapathaki S, Camci-Unal G, Wang G, Gkikas M. Nanophosphor-Based Contrast Agents for Spectral X-ray Imaging. Nanomaterials. 2019; 9(8):1092. https://doi.org/10.3390/nano9081092
Chicago/Turabian StyleSmith, Kevin, Matthew Getzin, Josephine J. Garfield, Sanika Suvarnapathaki, Gulden Camci-Unal, Ge Wang, and Manos Gkikas. 2019. "Nanophosphor-Based Contrast Agents for Spectral X-ray Imaging" Nanomaterials 9, no. 8: 1092. https://doi.org/10.3390/nano9081092
APA StyleSmith, K., Getzin, M., Garfield, J. J., Suvarnapathaki, S., Camci-Unal, G., Wang, G., & Gkikas, M. (2019). Nanophosphor-Based Contrast Agents for Spectral X-ray Imaging. Nanomaterials, 9(8), 1092. https://doi.org/10.3390/nano9081092








