Fabrication and Performance of ZnO Doped Tantalum Oxide Multilayer Composite Coatings on Ti6Al4V for Orthopedic Application
Abstract
1. Introduction
2. Materials and Experimental Design
2.1. Preparation of Coatings
2.2. Characterization of Coatings
2.3. Scratch Test
2.4. Contact Angle Measurement
2.5. Electrochemical Measurements
2.6. Antibacterial Experiment
3. Results and Discussion
3.1. Microstructural Characterization of the Coatings
3.2. Adhesion Strength
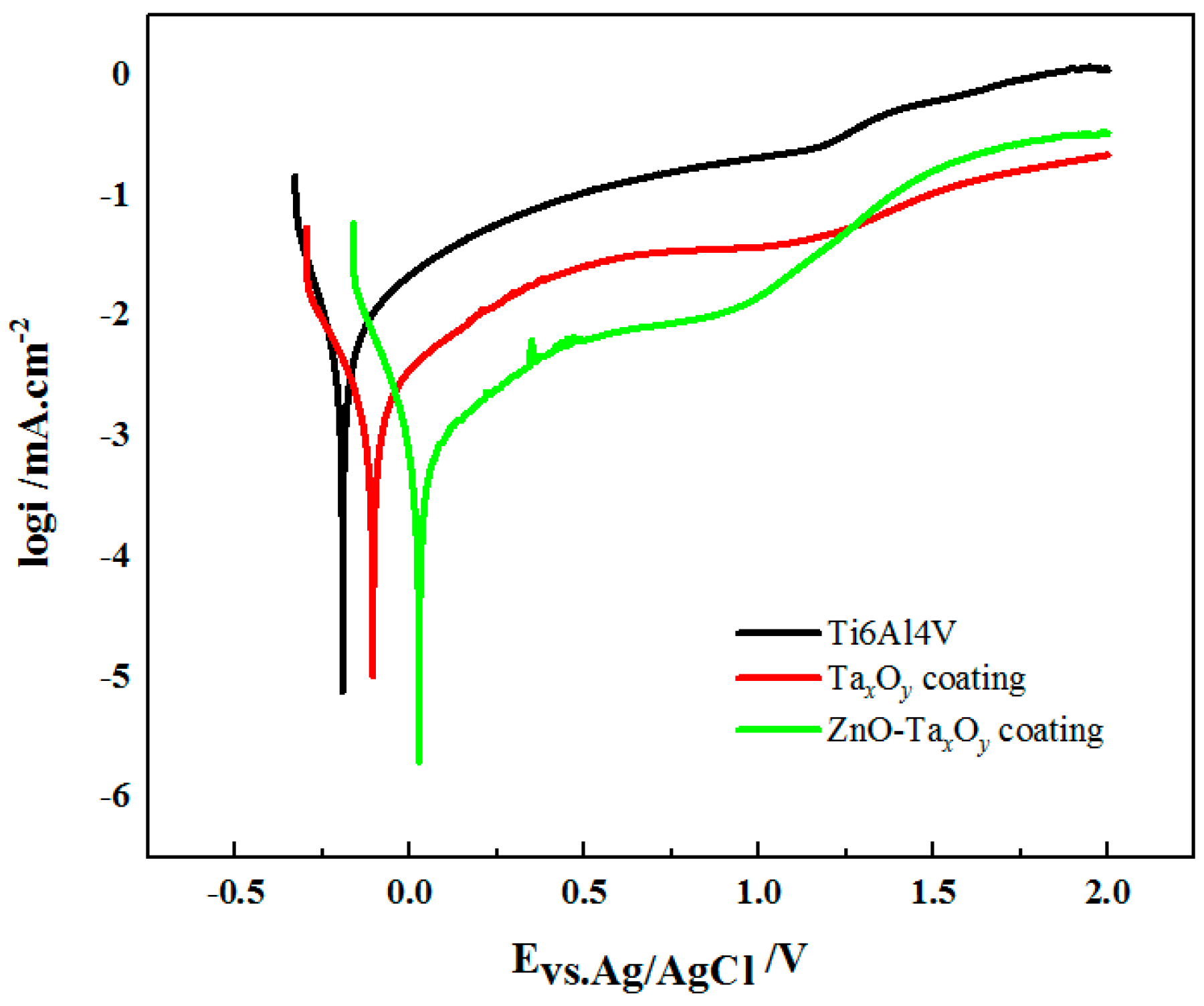
3.3. Corrosion Behavior
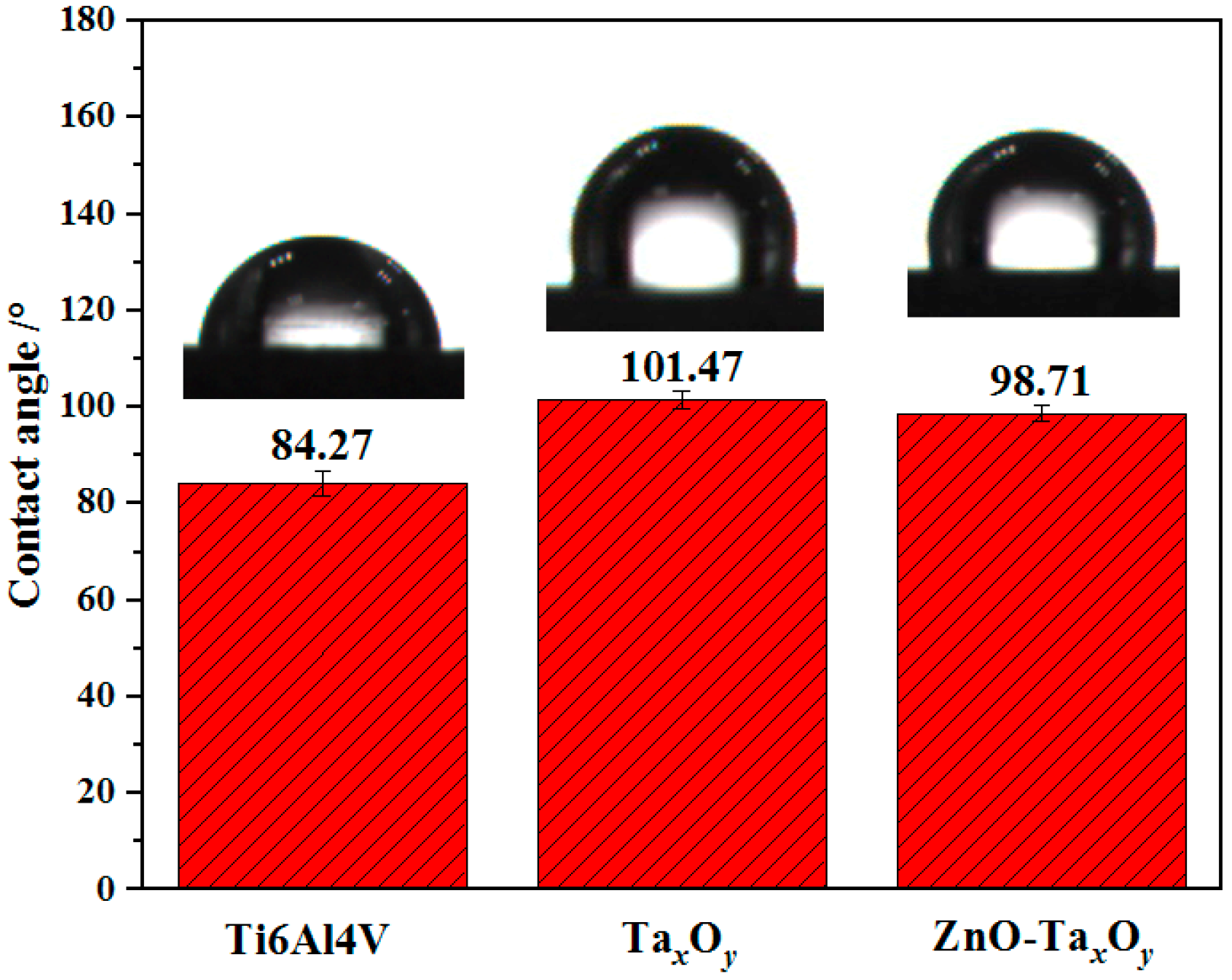
3.4. Wettability
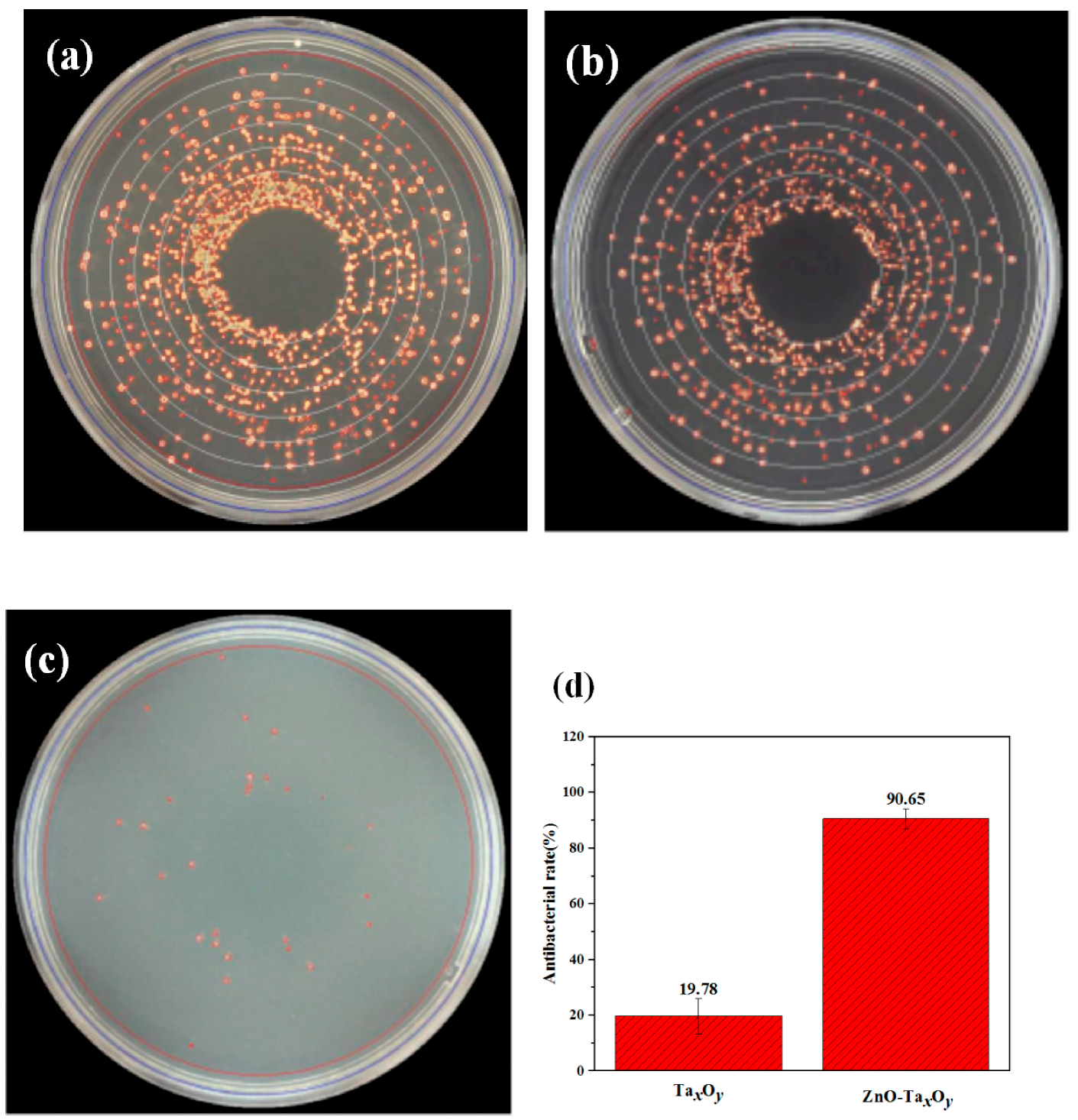
3.5. Antibacterial Property
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Atar, E.; Kayali, E.S.; Cimenoglu, H. Characteristics and Wear Performance of Borided Ti6Al4V alloy. Surf. Coat. Technol. 2008, 202, 4538–4549. [Google Scholar] [CrossRef]
- Rack, H.J.; Qazi, J.I. Titanium Alloys for Biomedical Applications. Mater. Sci. Eng. C 2006, 26, 1269–1277. [Google Scholar] [CrossRef]
- Gurrappa, I. Characterization of Titanium Alloy Ti6Al4V for Chemical, Marine and Industrial Applications. Mater. Charact. 2003, 51, 131–139. [Google Scholar] [CrossRef]
- Kamachi Mudali, U.; Sridhar, T.M.; Raj, B. Corrosion of Bio Implants. Sadhana 2003, 28, 601–637. [Google Scholar] [CrossRef]
- Okazaki, Y.; Gotoh, E. Comparison of Metal Release from Various Metallic Biomaterials in Vitro. Biomaterials 2005, 26, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Woodman, J.L.; Jacobs, J.J.; Galante, J.O.; Urban, R.M. Metal Ion Release from Titanium-Based Prosthetic Segmental Replacements of Long Bones in Baboons: A Long-Term Study. J. Orthop. Res. 1984, 1, 421–430. [Google Scholar] [CrossRef]
- Sargeant, A.; Goswami, T. Hip Implants—Paper VI—Ion Concentrations. Mater. Des. 2007, 28, 155–171. [Google Scholar] [CrossRef]
- Raphel, J.; Holodniy, M.; Goodman, S.B.; Heilshorn, S.C. Multifunctional coatings to simultaneously promote osseointegration and prevent infection of orthopaedic implants. Biomaterials 2016, 84, 301–314. [Google Scholar] [CrossRef]
- He, Q.; Liu, J.; Liang, J.; Liu, X.; Li, W.; Liu, Z.; Ding, Z.; Tuo, D. Towards Improvements for Penetrating the Blood–Brain Barrier—Recent Progress from a Material and Pharmaceutical Perspective. Cells 2018, 7, 24. [Google Scholar] [CrossRef]
- He, Q.; Liu, J.; Liu, X.; Li, G.; Deng, P.; Liang, J. Preparation of Cu2O-Reduced Graphene Nanocomposite Modified Electrodes towards Ultrasensitive Dopamine Detection. Sensors 2018, 18, 199. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.Y.; Xu, J.L.; Liu, Z.H.; Deng, L.; Sun, B.; Liu, S.D.; Wang, L.; Liu, H.Y. Preparation, corrosion resistance and hemocompatibility of thesuperhydrophobic TiO2 coatings on biomedical Ti-6Al-4V alloys. Appl. Surf. Sci. 2015, 347, 591–595. [Google Scholar] [CrossRef]
- Khanna, R.; Kokubo, T.; Matsushita, T.; Takadama, H. Fabrication of dense α-alumina layer on Ti-6Al-4V alloy hybrid for bearing surfaces of artificial hip joint. Mater. Sci. Eng. C 2016, 69, 1229–1239. [Google Scholar] [CrossRef]
- Chellappa, M.; Vijayalakshmi, U. Electrophoretic deposition of silica and its composite coatings on Ti-6A1-4V, and its in vitro corrosion behaviour for biomedical applications. Mater. Sci. Eng. C 2017, 71, 879–890. [Google Scholar] [CrossRef]
- Berni, M.; Lopomo, N.; Marchiori, G.; Gambardella, A.; Boi, M.; Bianchi, M.; Visani, A.; Pavan, P.; Russo, A.; Marcacci, M. Tribological characterization of zirconia coatings deposited on Ti6Al4V components for orthopedic applications. Mater. Sci. Eng. C 2016, 62, 643–655. [Google Scholar] [CrossRef]
- Xu, J.; Hu, W.; Xie, Z.H.; Munroe, P. Reactive-sputter-deposited β-Ta2O5 and TaON nanoceramic coatings on Ti-6Al-4V alloy against wear and corrosion damage. Surf. Coat. Technol. 2016, 296, 171–184. [Google Scholar] [CrossRef]
- Hauert, R.; Falub, C.V.; Thorwarth, G.; Thorwarth, K.; Affolter, C.; Stiefel, M.; Podleska, L.E.; Taeger, G. Retrospective lifetime estimation of failed and explanted diamond-like carbon coated hip joint balls. Acta Biomater. 2012, 8, 3170–3176. [Google Scholar] [CrossRef]
- Kwok, C.T.; Wong, P.K.; Cheng, F.T.; Man, H.C. Characterization and corrosion behavior of hydroxyapatite coatings on Ti6Al4V fabricated by electrophoretic deposition. Appl. Surf. Sci. 2009, 255, 6736–6744. [Google Scholar] [CrossRef]
- Rahmati, B.; Sarhan, A.A.D.; Jeffrey Basirun, W.; Abas, W.A.B.W. Ceramic tantalum oxide thin film coating to enhance the corrosion and wear characteristics of Ti-6Al-4V alloy. J. Alloys Compd. 2016, 676, 369–376. [Google Scholar] [CrossRef]
- Hu, W.; Xu, J.; Lu, X.L.; Hu, D.S.; Tao, H.L.; Munroe, P.; Xie, Z.H. Corrosion and wear behaviours of a reactive-sputter-deposited Ta2O5 nanoceramic coating. Appl. Surf. Sci. 2016, 368, 177–190. [Google Scholar] [CrossRef]
- Xu, J.; Bao, X.K.; Fu, T.; Lyu, Y.H.; Munroe, P.; Xie, Z.H. In vitro biocompatibility of a nanocrystalline β-Ta2O5 coating for orthopaedic implants. Ceram. Int. 2018, 44, 4660–4675. [Google Scholar] [CrossRef]
- Chang, Y.Y.; Huang, H.L.; Chen, H.J.; Lai, C.H.; Wen, C.Y. Antibacterial properties and cytocompatibility of tantalum oxide coatings. Surf. Coat. Technol. 2014, 259, 193–198. [Google Scholar] [CrossRef]
- Bieniaś, J.; Surowska, B.; Stoch, A.; Matraszek, H.; Walczak, M. The influence of SiO2 and SiO2-TiO2 intermediate coatings on bond strength of titanium and Ti6Al4V alloy to dental porcelain. Dental Materials 2009, 25, 1128–1135. [Google Scholar]
- Huang, A.P.; Chu, P.K. Crystallization improvement of Ta2O5 thin films by the addition of water vapor. J. Cryst. Growth 2005, 274, 73–77. [Google Scholar] [CrossRef]
- Huang, C.J. Room-temperature formation of tantalum oxide films by liquid phase deposition. Thin Solid Film 2005, 478, 332–337. [Google Scholar] [CrossRef]
- Ispas, A.; Adolphi, B.; Bund, A.; Endres, F. On the electrodeposition of tantalum from three different ionic liquids with the bis(trifluoromethyl sulfonyl)amide anion. Phys. Chem. Chem. Phys. 2010, 12, 1793–1803. [Google Scholar] [CrossRef] [PubMed]
- Kurnia, F.; Hadiyawarman; Jung, C.U.; Jung. R., J.; Liu, C.L. Composition dependence of unipolar resistance switching in TaOx thin films. Phys. Status Solidi RRL 2011, 7, 253–255. [Google Scholar] [CrossRef]
- Kukli, K.; Aarik, J.; Aidla, A.; Kohan, O.; Uustare, T.; Sammelselg, V. Properties of tantalum oxide thin films grown by atomic layer deposition. Thin Solid Film 1995, 260, 135–142. [Google Scholar] [CrossRef]
- Arnould, C.; Korányi, T.I.; Delhalle, J.; Mekhalif, Z. Fabrication of tantalum oxide/carbon nanotubes thin film composite on titanium substrate. J. Colloid Interface Sci. 2010, 344, 390–394. [Google Scholar] [CrossRef]
- Wolf, M.J.; Roitsch, S.; Mayer, J.; Nijmeijer, A.; Bouwmeester, H.J.M. Fabrication of ultrathin films of Ta2O5 by a sol-gel method. Thin Solid Film 2013, 527, 354–357. [Google Scholar] [CrossRef]
- Liu, X.Y.; Chu, P.K.; Ding, C.X. Surface modification of titanium, titanium alloys, and related materials for biomedical applications. Mater. Sci. Eng. R Rep. 2004, 47, 49–121. [Google Scholar] [CrossRef]
- Wei, A.X.; Ge, Z.X.; Zhao, X.H.; Liu, J.; Zhao, Y. Electrical and optical properties of tantalum oxide thin films prepared by reactive magnetron sputtering. J. Alloys Compd. 2011, 509, 9758–9763. [Google Scholar] [CrossRef]
- Qi, K.Z.; Cheng, B.; Yu, J.G.; Ho, W.K. Review on the improvement of the photocatalytic and antibacterial activities of ZnO. J. Alloys Compd. 2017, 727, 792–820. [Google Scholar] [CrossRef]
- Güy, N.; Özacar, M. The influence of noble metals on photocatalytic activity of ZnO for Congo red degradation. Int. J. Hydrogen Energy 2016, 41, 20100–20112. [Google Scholar] [CrossRef]
- Podasca, V.E.; Buruiana, T.; Buruiana, E.C. UV-cured polymeric films containing ZnO and silver nanoparticles with UV-vis light-assisted photocatalytic activity. Appl. Surf. Sci. 2016, 377, 262–273. [Google Scholar] [CrossRef]
- Shim, K.; Abdellatif, M.; Choi, E.; Kim, D. Nanostructured ZnO films on stainless steel are highly safe and effective for antimicrobial applications. Appl. Microbiol. Biotechnol. 2017, 101, 2801–2809. [Google Scholar] [CrossRef] [PubMed]
- Ohtsu, N.; Kakuchi, Y.; Ohtsuki, T. Antibacterial effect of zinc oxide/hydroxyapatite coatings prepared by chemical solution deposition. Appl. Surf. Sci. 2018, 445, 596–600. [Google Scholar] [CrossRef]
- Sohrabnezhad, S.; Seifi, A. The green synthesis of Ag/ZnO in montmorillonite with enhanced photocatalytic activity. Appl. Surf. Sci. 2016, 386, 33–40. [Google Scholar] [CrossRef]
- Li, J.H.; Hong, R.Y.; Li, M.Y.; Li, H.Z.; Zheng, Y.; Ding, J. Effects of ZnO nanoparticles on the mechanical and antibacterial properties of polyurethane coatings. Prog. Org. Coat. 2009, 64, 504–509. [Google Scholar] [CrossRef]
- Yusa, K.; Yamamoto, O.; Fukuda, M.; Koyota, S.; Koizumi, Y.; Sugiyama, T. In vitro prominent bone regeneration by release zinc ion from Zn-modified implant. Biochem. Biophys. Res. Commun. 2011, 412, 273–278. [Google Scholar] [CrossRef]
- Kawamura, H.; Ito, A.; Miyakawa, S.; Layrolle, P.; Ojima, K.; Ichinose, N.; Tateishi, T. Stimulatory effect of zinc-releasing calcium phosphate implant on bone formation in rabbit femora. J. Biomed. Mater. Res. 2000, 50, 184–190. [Google Scholar] [CrossRef]
- Botequim, D.; Maia, J.; Lino, M.M.F.; Lopes, L.M.F.; Simões, P.N.; Ilharco, L.M.; Ferreira, L. Nanoparticles and surfaces presenting antifungal, antibacterial and antiviral properties. Langmuir 2012, 28, 7646–7656. [Google Scholar] [CrossRef]
- Hu, H.; Zhang, W.; Qiao, Y.; Jiang, X.; Liu, X.; Ding, C. Antibacterial activity and increased bone marrow stem cell functions of Zn-incorporated TiO2 coatings on titanium. Acta Biomater. 2012, 8, 904–915. [Google Scholar] [CrossRef]
- Rahmati, B.; Zalnezhad, E.; Sarhand, A.A.D.; Kamiab, Z.; Nasiri Tabrizi, B.; Abasc, W.A.B.W. Enhancing the adhesion strength of tantalum oxide ceramic thin film coating on biomedical Ti-6A1-4V alloy by thermal surface treatment. Ceram. Int. 2015, 41, 13055–13063. [Google Scholar] [CrossRef]
- Zhang, W.G.; Liu, W.M.; Liu, Y.; Wang, C.T. Tribological behaviors of single and dual sol-gel ceramic films on Ti-6Al-4V. Ceram. Int. 2009, 35, 1513–1520. [Google Scholar] [CrossRef]
- Knotek, O.; Loffler, F.; Kramer, G. Process and advantage of multicomponent and multilayer PVD coatings. Surf. Coat. Technol. 1993, 59, 14–20. [Google Scholar] [CrossRef]
- Wang, S.C.; Liu, K.Y.; Huang, J.L. Tantalum oxide film prepared by reactive magnetron sputtering deposition for all-solid-state electrochromic device. Thin Solid Film 2011, 520, 1454–1459. [Google Scholar] [CrossRef]
- Moreira, H.; Costa-Barbosa, A.; Marques, S.M.; Sampaio, P.; Carvalho, S. Evaluation of cell activation promoted by tantalum and tantalum oxide coatings deposited by reactive DC magnetron sputtering. Surf. Coat. Technol. 2017, 330, 260–269. [Google Scholar] [CrossRef]
- He, Q.; Liu, J.; Liu, X.; Li, G.; Deng, P.; Liang, J. Manganese dioxide Nanorods/electrochemically reduced graphene oxide nanocomposites modified electrodes for cost-effective and ultrasensitive detection of Amaranth. Colloids Surf. B Biointerfaces 2018, 172, 565. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Liu, J.; Liu, X.; Li, G.; Chen, D.; Deng, P.; Liang, J. A promising sensing platform toward dopamine using MnO2 nanowires/electro-reduced graphene oxide composites. Electrochim. Acta 2019, 296, 683. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- He, Q.; Liu, J.; Liu, X.; Li, G.; Chen, D.; Deng, P.; Liang, J. Fabrication of Amine-Modified Magnetite-Electrochemically Reduced Graphene Oxide Nanocomposite Modified Glassy Carbon Electrode for Sensitive Dopamine Determination. Nanomaterials 2018, 8, 194. [Google Scholar] [CrossRef]
- He, Q.; Li, G.; Liu, X.; Liu, J.; Deng, P.; Chen, D. Morphologically Tunable MnO2 Nanoparticles Fabrication, Modelling and Their Influences on Electrochemical Sensing Performance toward Dopamine. Catalysts 2018, 8, 323. [Google Scholar] [CrossRef]
- Liu, R.; Tang, Y.; Zeng, L.; Zhao, Y.; Ma, Z.; Sun, Z.; Xiang, L.; Ren, L.; Yang, K. In vitro and in vivo studies of anti-bacterial copper-bearing titanium alloy for dental application. Dent. Mater. 2018, 34, 1112–1126. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.K.; Zheng, X.B.; Xie, Y.T.; Ding, C.X.; Ruan, H.J.; Fan, C.Y. Anti-bacterial and cytotoxic properties of plasma sprayed silver-containing HA coatings. J. Mater. Sci. Mater. Med. 2008, 19, 3603–3609. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.H.; Liu, H.Y. Structures of tantalum pentoxide thin films formed by reactive sputtering of Ta metal. Thin Solid Film 1995, 258, 56–63. [Google Scholar] [CrossRef]
- Chen, H.X.; Ding, J.J.; Shi, F.; Li, Y.F.; Guo, W.G. Optical properties of Ti-doped ZnO films synthesized via magnetron sputtering. J. Alloys Compd. 2012, 534, 59–63. [Google Scholar] [CrossRef]
- Jimmy Wu, S.J.; Houng, B.; Huang, B.S. Effect of growth and annealing temperatures on crystallization of tantalum pentoxide thin film prepared by RF magnetron sputtering method. J. Alloys Compd. 2009, 475, 488–493. [Google Scholar]
- Lu, J.J.; Lu, Y.M.; Tasi, S.I.; Hsiung, T.L.; Wang, H.P.; Jang, L.Y. Conductivity enhancement and semiconductor-metal transition in Ti-doped ZnO films. Opt. Mater. 2007, 29, 1548–1552. [Google Scholar] [CrossRef]
- Simpson, R.; White, R.G.; Watts, J.F.; Baker, M.A. XPS investigation of monatomic and cluster argon ion sputtering of tantalum pentoxide. Appl. Surf. Sci. 2017, 405, 79–87. [Google Scholar] [CrossRef]
- Ivanov, M.V.; Perevalov, T.V.; Aliev, V.S.; Gritsenko, V.A.; Kaichev, V.V. Electronic structure of Ta2O5 with oxygen vacancy: Ab initio calculations and comparison with experiment. J. Appl. Phys. 2011, 110, 024115. [Google Scholar] [CrossRef]
- Li, H.; Muraki, Y.; Karahashi, K.; Hamaguchi, S. Suboxide/subnitride formation on Ta masks during magnetic material etching by reactive plasmas. J. Vac. Sci. Technol. A 2015, 33, 040602. [Google Scholar] [CrossRef]
- Wang, R.; He, X.J.; Gao, Y.E.; Zhang, X.Y.; Yao, X.H.; Tang, B. Antimicrobial property, cytocompatibility and corrosion resistance of Zn-doped ZrO2/TiO2 coatings on Ti6Al4V implants. Mater. Sci. Eng. C 2017, 75, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Farnoush, H.; Aldic, A.; Cimenoglu, H. Functionally graded HA-TiO2 nanostructured composite coating on Ti-6Al-4V substrate via electrophoretic deposition. Surf. Coat. Technol. 2015, 265, 7–15. [Google Scholar] [CrossRef]
- Bonu, V.; Jeevitha, M.; Kumar, V.P.; Bysakhl, S.; Barshilia, H.C. Ultra-thin multilayered erosion resistant Ti/TiN coatings with stress absorbing layers. Appl. Surf. Sci. 2019, 478, 872–881. [Google Scholar] [CrossRef]
- Farnoush, H.; Mohandesi, J.A.; Çimenoğlu, H. Micro-scratch and corrosion behavior of functionally graded HA-TiO2 nanostructured composite coatings fabricated by electrophoretic deposition. J. Mech. Behav. Biomed. Mater. 2015, 46, 31–40. [Google Scholar] [CrossRef]
- Moldovan, M.; Weyant, C.M.; Johnson, D.L.; Faber, K.T. Tantalum Oxide Coatings as Candidate Environmental Barriers. J. Therm. Spray Technol. 2004, 13, 51–56. [Google Scholar] [CrossRef]
- Behera, R.R.; Das, A.; Pamu, D.; Pandey, L.M.; Sankara, M.R. Mechano-tribological properties and in vitro bioactivity of biphasic calcium phosphate coating on Ti-6Al-4V. J. Mech. Behav. Biomed. Mater. 2018, 86, 143–157. [Google Scholar] [CrossRef]
- Luo, F.; Gao, K.; Pang, X.; Yang, H.; Qiao, L.; Wang, Y. Characterization of the mechanical properties and failure modes of hard coatings deposited by RF magnetron sputtering. Surf. Coat. Technol. 2008, 202, 3354–3359. [Google Scholar] [CrossRef]
- Hyde, F.W.; Alberg, M.; Smith, K. Comparison of fluorinated polymers against stainless steel, glass and polypropylene in microbial biofilm adherence and removal. J. Ind. Microbiol. Biotechnol. 1997, 19, 142. [Google Scholar] [CrossRef] [PubMed]
- Lassen, B.; Holmberg, K.; Brink, C.; Carlén, A.; Olsson, J. Binding of salivary proteins and oral bacteria to hydrophobic and hydrophilic surfaces in vivo and in vitro. Colloid Polym. Sci. 1994, 272, 1143–1150. [Google Scholar] [CrossRef]
- Welin-Klintstrom, S.; Askendal, A.; Elwing, H. Surfactant and Protein Interactions on Wettability Gradient Surfaces. J. Colloid Interface Sci. 1993, 158, 188–194. [Google Scholar] [CrossRef]
- Zhou, W.; Zhong, X.; Wu, X.; Yuan, L.; Zhao, Z.; Wang, H.; Xia, Y.; Feng, Y.; He, J.; Chen, W. The effect of surface roughness and wettability of nanostructured TiO2 film on TCA-8113 epithelial-like cells. Surf. Coat. Technol. 2006, 200, 6155–6160. [Google Scholar] [CrossRef]
- Cheng, Y.L.; Cao, L.; He, G.; Yao, G.; Song, X.P.; Sun, Z.Q. Preparation, microstructure and photoelectrical properties of Tantalum-doped zinc oxide transparent conducting films. J. Alloys Compd. 2014, 608, 85–89. [Google Scholar] [CrossRef]
- Zhang, L.L.; Jiang, Y.H.; Ding, Y.L.; Daskalakis, N.; Jeuken, L.; Povey, M.; O’Neill, A.J.; York, D.W. Mechanistic Investigation in to Antibacterial Behaviour of Suspensions of ZnO Nanoparticles Against E. coli. J. Nanopart. Res. 2010, 12, 1625–1636. [Google Scholar] [CrossRef]
- Zhang, L.L.; Jiang, Y.H.; Ding, Y.L.; Povey, M.; York, D. Investigation into the antibacterial behaviour of suspensions of ZnO nanoparticles (ZnO nanofluids). J. Nanopart. Res. 2007, 9, 479–489. [Google Scholar] [CrossRef]
- Pandey, A.; Midhal, S.; Sharma, R.K.; Maurya, R.; Nigam, V.K.; Ghoshb, S.; Balani, K. Antioxidant and antibacterial hydroxyapatite-based biocomposite for orthopedic applications. Mater. Sci. Eng. C 2018, 88, 13–24. [Google Scholar] [CrossRef]
- Ramaswamy, Y.; Wu, C.; Zhou, H.; Zreiqat, H. Biological response of human bone cells to zinc-modified Ca-Si-based ceramics. Acta Biomater. 2008, 4, 1487–1497. [Google Scholar] [CrossRef] [PubMed]
- Saha, N.; Keskinbora, K.; Suvaci, E.; Basu, B. Sintering, microstructure, mechanical and antimicrobial properties of HAp-ZnO biocomposites. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 95B, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.H.; Zhang, W.; Wang, D.N.; Feng, W.; Liu, Y.; Lin, Z.; Du, K.Q.; Deng, C.F. Effect of Zn content on cytoactivity and bacteriostasis of micro-arc oxidation coatings on pure titanium. Surf. Coat. Technol. 2013, 228, S428–S432. [Google Scholar] [CrossRef]
- Li, M.; Zhu, L.Z.; Lin, D.H. Toxicity of ZnO Nanoparticles to Escherichia Coli: Mechanism and the Influence of Medium Components. Environ. Sci. Technol. 2011, 45, 1977–1983. [Google Scholar] [CrossRef]
- Song, W.; Zhang, J.; Guo, J.; Zhang, J.; Ding, F.; Li, L.; Sun, Z. Role of the Dissolved Zinc lon and Reactive Oxygen Species in Cytotoxicity of ZnO Nanoparticles. Toxicol. Lett. 2010, 199, 389–397. [Google Scholar] [CrossRef] [PubMed]
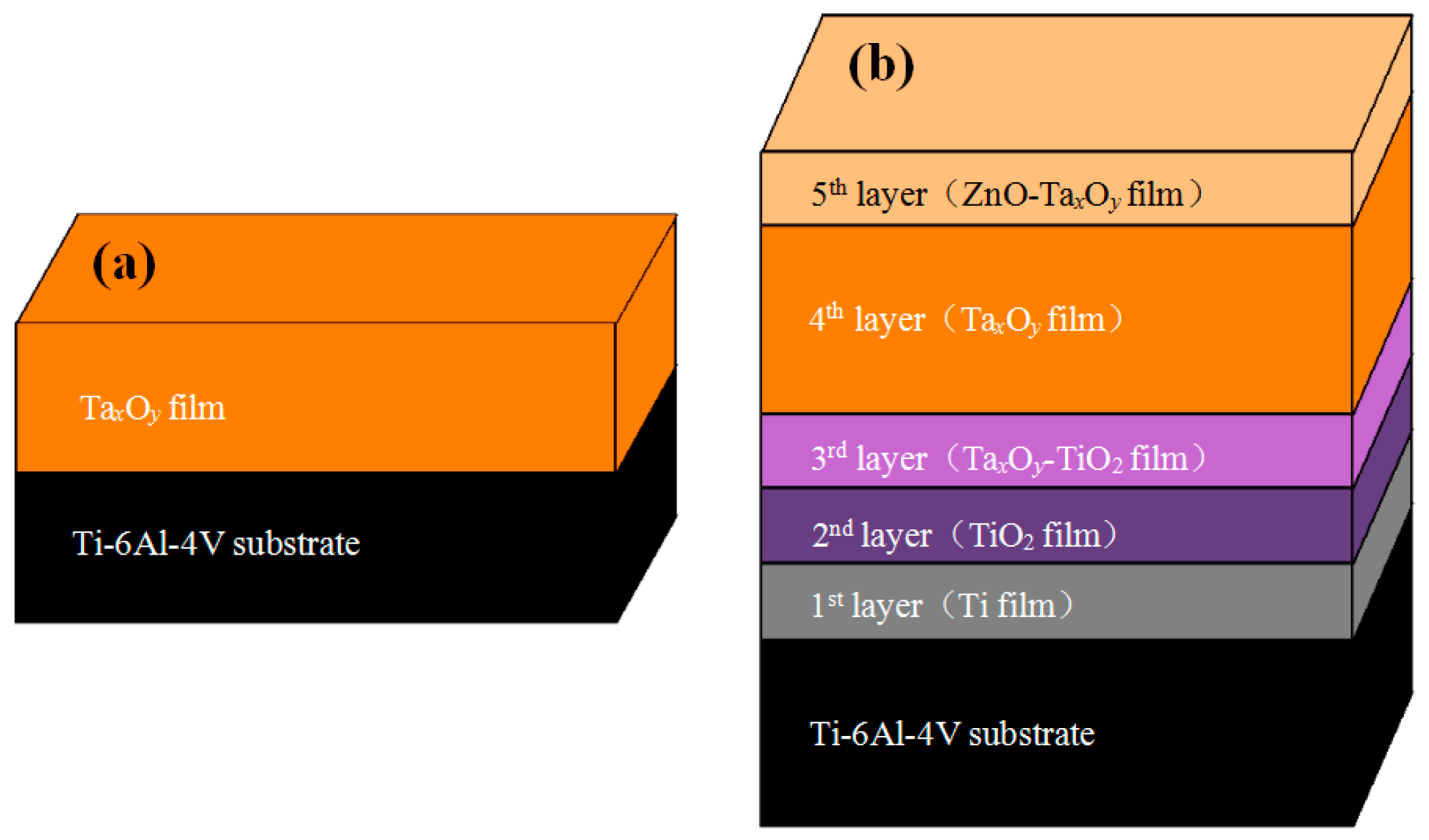


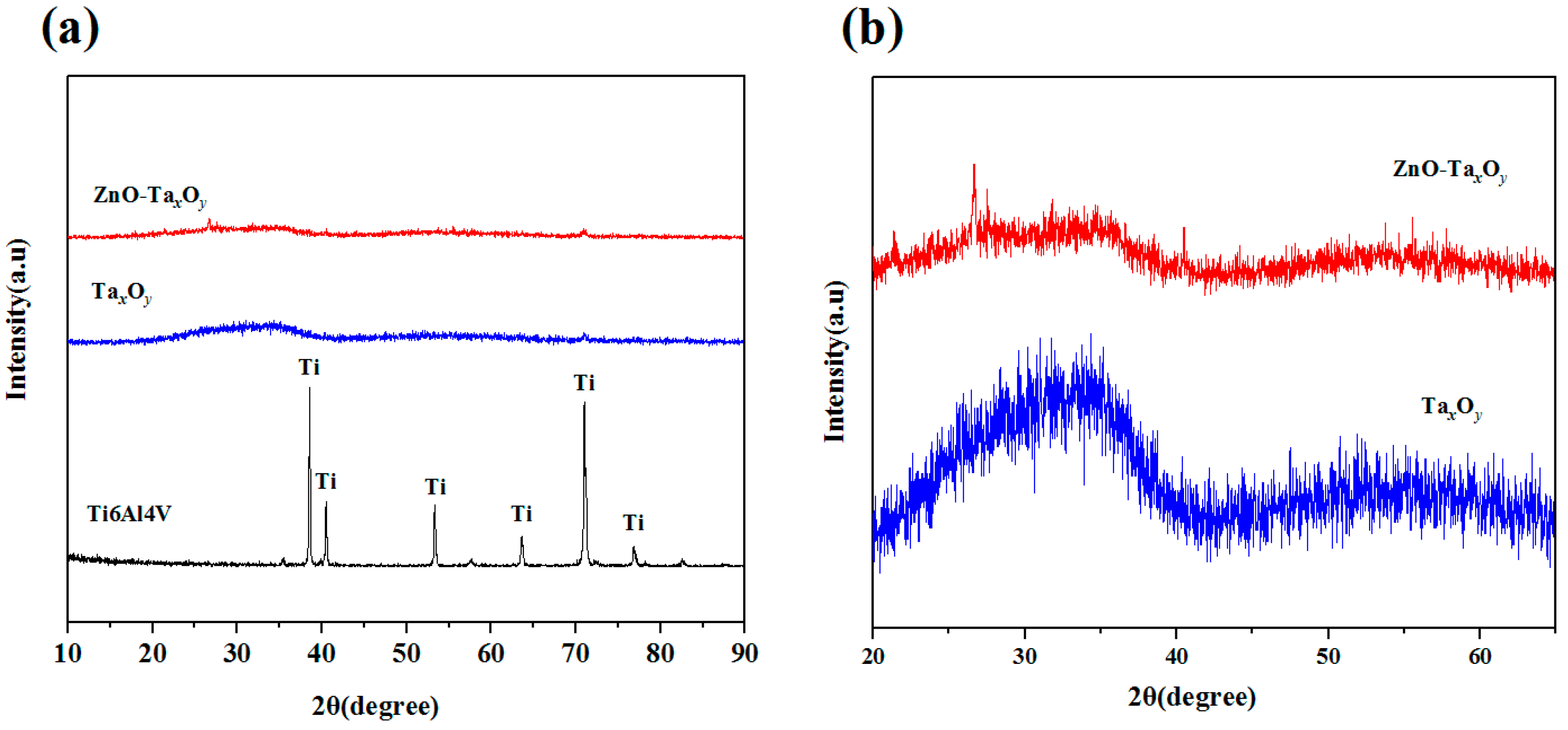

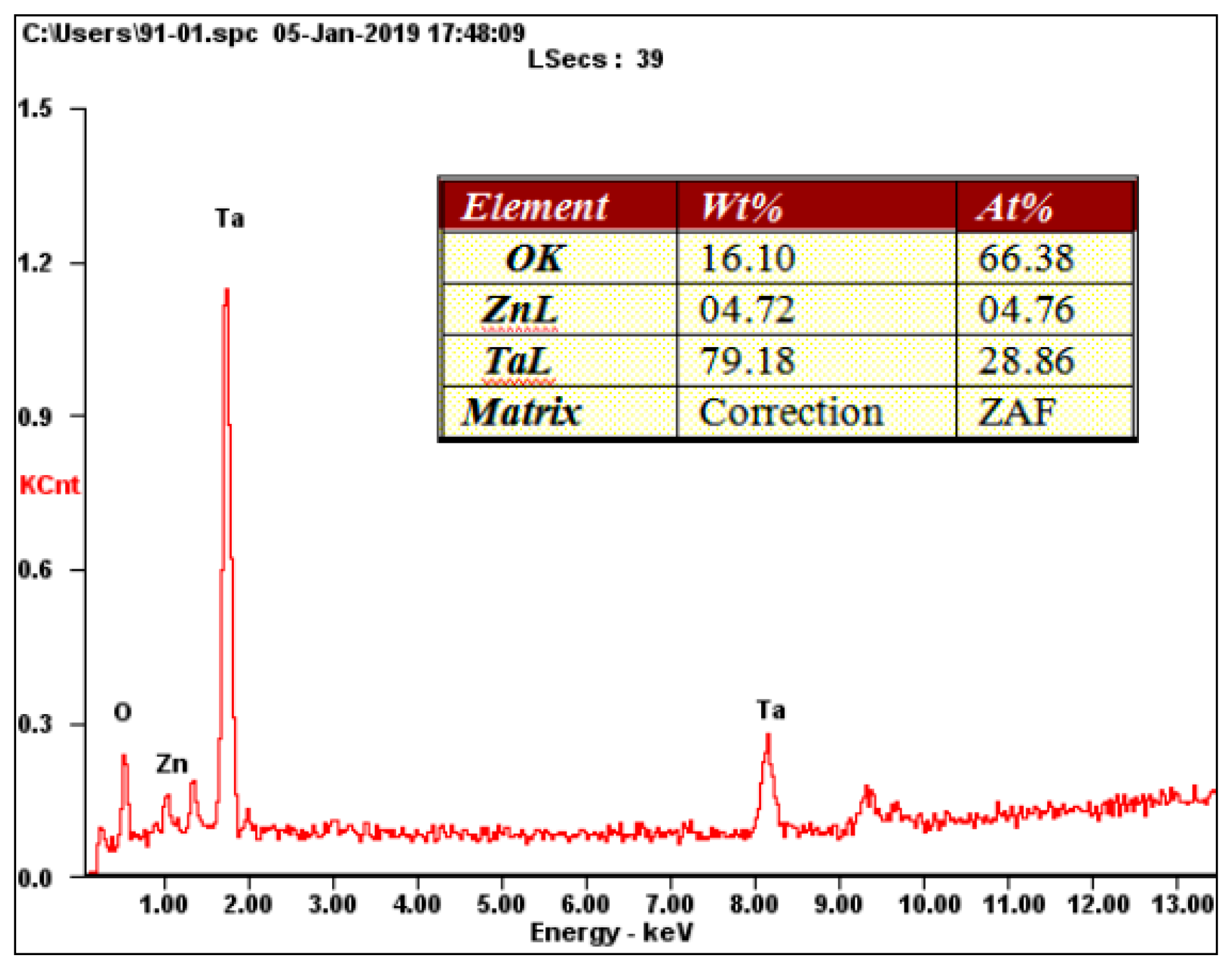
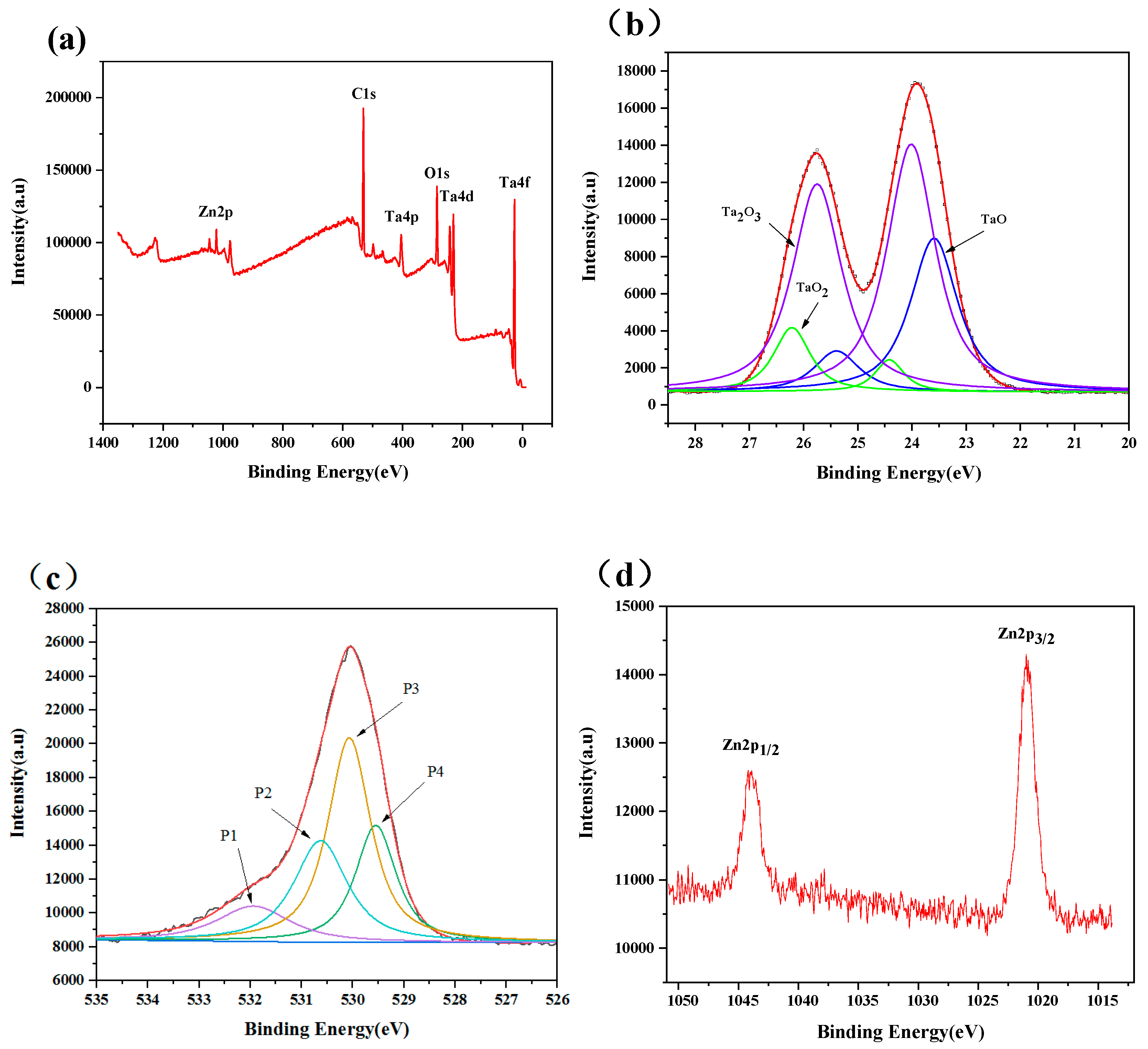
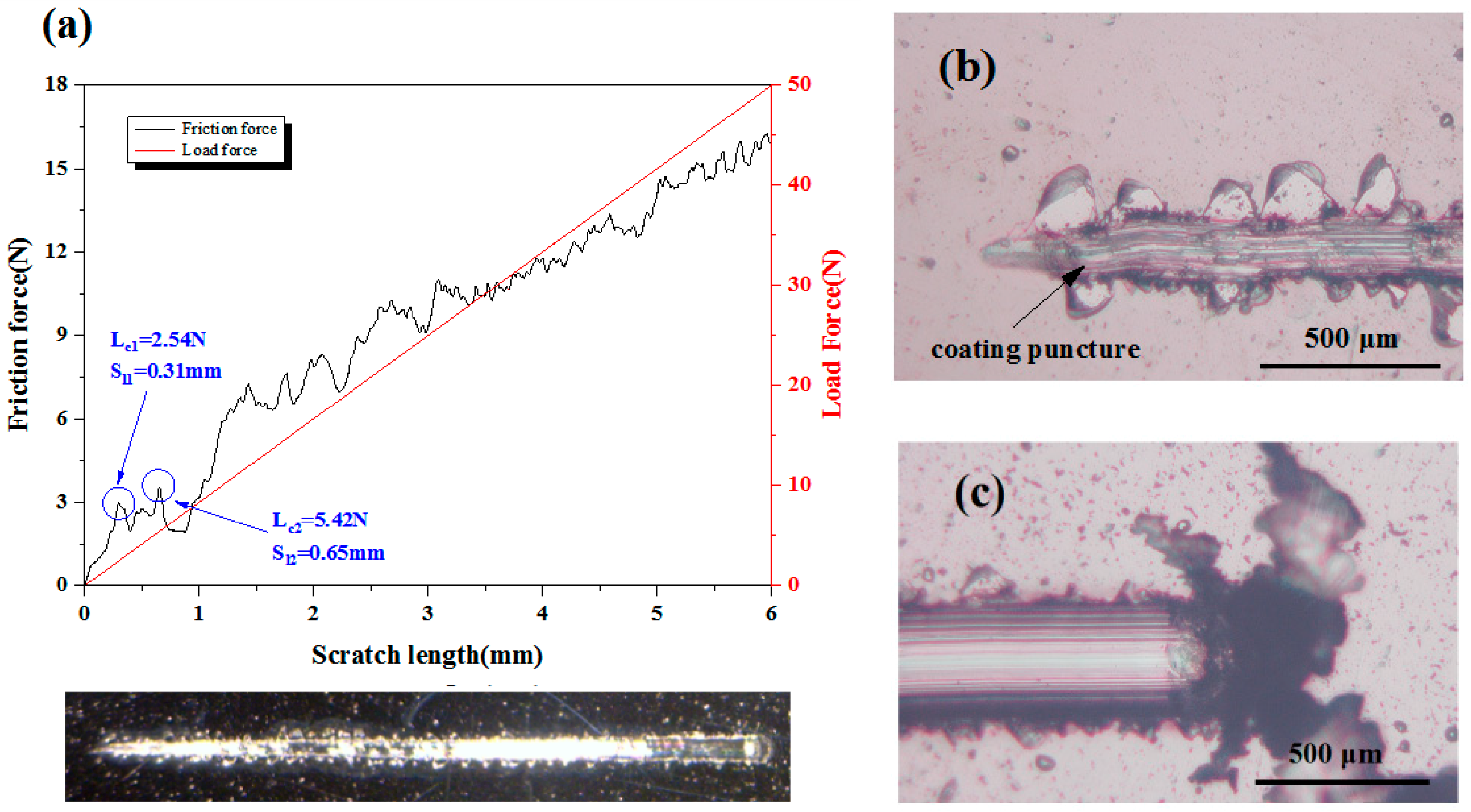
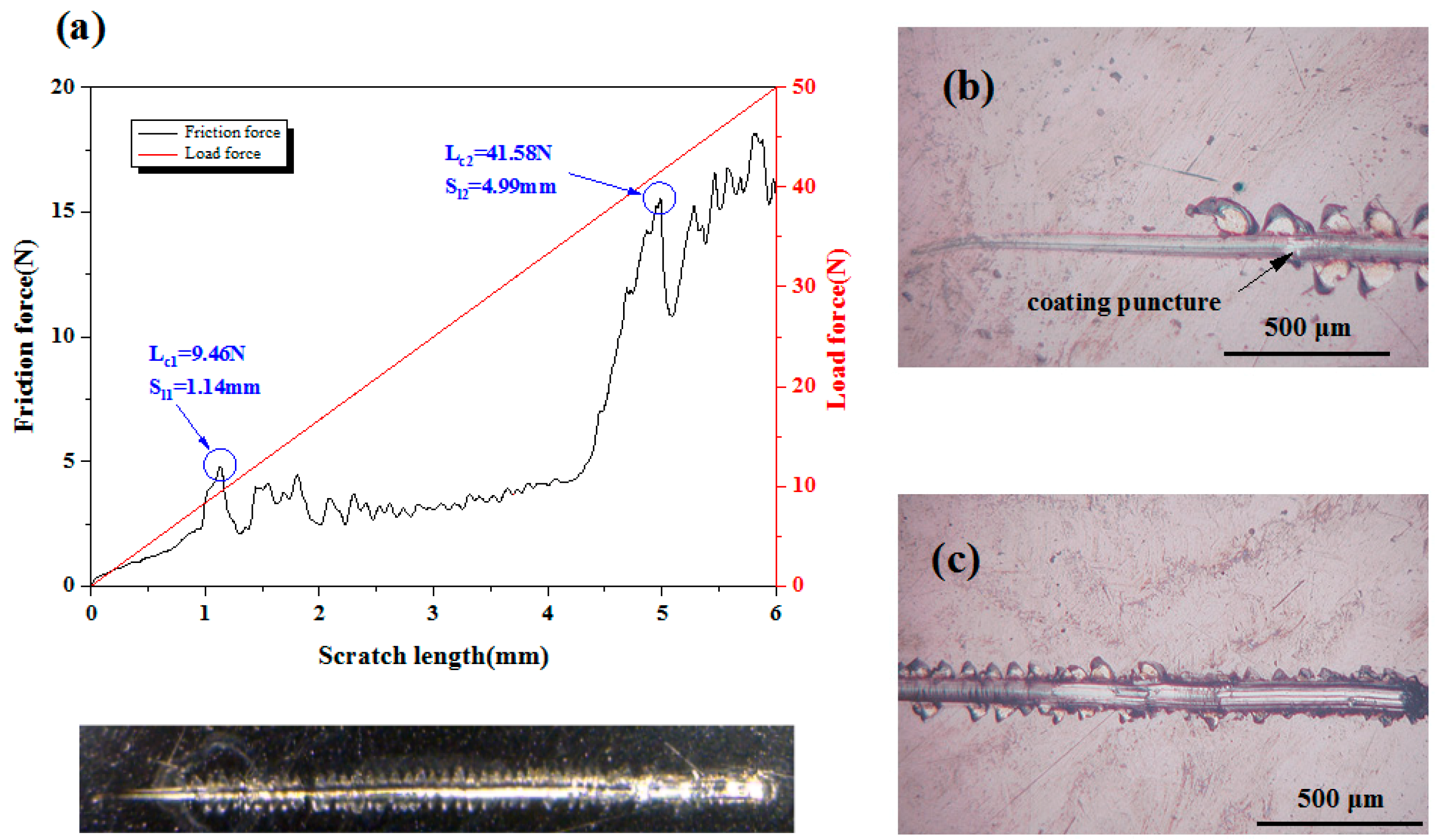
| Coating Code | Layer Number | Coating Material | Sputtering Mode | Sputtering Power (W) | Deposition Time (min) | Gas flow (sccm) | ||
|---|---|---|---|---|---|---|---|---|
| Ar | O2 | |||||||
| ZnO-TaxOy | 1st layer | Ti | RF sputtering | 200 | 15 | 16 | / | |
| 2nd layer | TiO2 | RF reaction sputtering | 200 | 15 | 16 | 4 | ||
| 3rd layer | TaxOy-TiO2 | TaxOy | DC reaction sputtering | 250 | 15 | 24 | 6 | |
| TiO2 | RF reaction sputtering | 200 | ||||||
| 4th layer | TaxOy | DC reaction sputtering | 250 | 120 | 16 | 4 | ||
| 5th layer | ZnO-TaxOy | ZnO | RF sputtering | 150 | 15 | 16 | 4 | |
| TaxOy | DC reaction sputtering | 250 | ||||||
| TaxOy | / | TaxOy | DC reaction sputtering | 250 | 120 | 16 | 4 | |
| Sample | Ti6Al4V | TaxOy | ZnO-TaxOy |
|---|---|---|---|
| Ecorr (V vs. Ag/AgCl) | –0.19 ± 0.02 | −0.11 ± 0.01 | 0.02 ± 0.01 |
| Icorr (μA/cm2) | 7.07 ± 0.012 | 3.85 ± 0.003 | 1.12 ± 0.004 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, Z.; He, Q.; Ding, Z.; Liao, C.; Chen, D.; Ou, L. Fabrication and Performance of ZnO Doped Tantalum Oxide Multilayer Composite Coatings on Ti6Al4V for Orthopedic Application. Nanomaterials 2019, 9, 685. https://doi.org/10.3390/nano9050685
Ding Z, He Q, Ding Z, Liao C, Chen D, Ou L. Fabrication and Performance of ZnO Doped Tantalum Oxide Multilayer Composite Coatings on Ti6Al4V for Orthopedic Application. Nanomaterials. 2019; 9(5):685. https://doi.org/10.3390/nano9050685
Chicago/Turabian StyleDing, Ziyu, Quanguo He, Zeliang Ding, Cuijiao Liao, Dongchu Chen, and Ling Ou. 2019. "Fabrication and Performance of ZnO Doped Tantalum Oxide Multilayer Composite Coatings on Ti6Al4V for Orthopedic Application" Nanomaterials 9, no. 5: 685. https://doi.org/10.3390/nano9050685
APA StyleDing, Z., He, Q., Ding, Z., Liao, C., Chen, D., & Ou, L. (2019). Fabrication and Performance of ZnO Doped Tantalum Oxide Multilayer Composite Coatings on Ti6Al4V for Orthopedic Application. Nanomaterials, 9(5), 685. https://doi.org/10.3390/nano9050685





