Improving Two-Step Prepared CH3NH3PbI3 Perovskite Solar Cells by Co-Doping Potassium Halide and Water in PbI2 Layer
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Device Fabrication
2.3. Characterizations
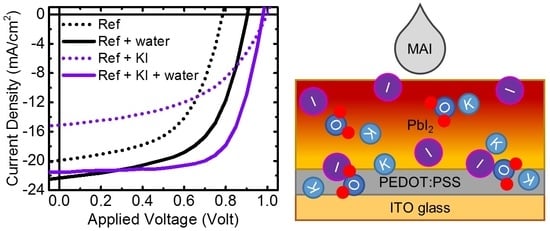
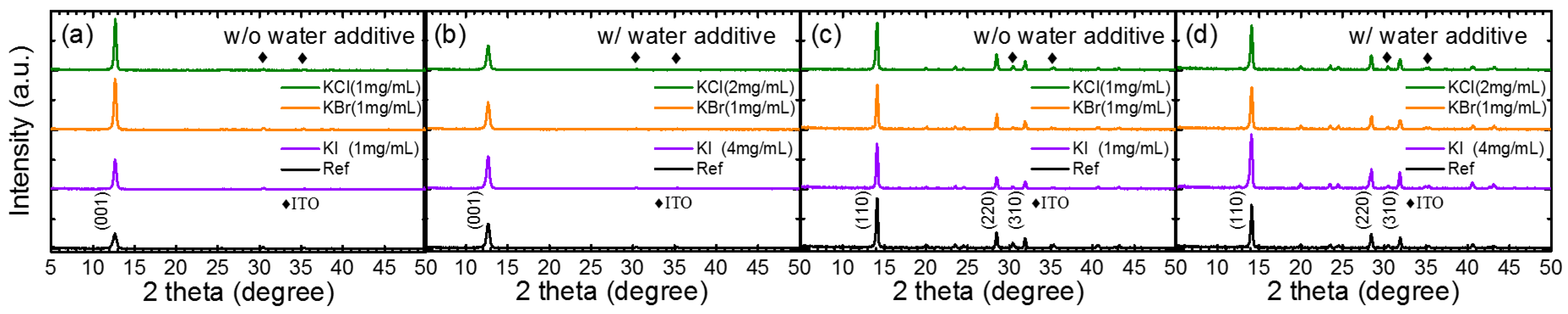
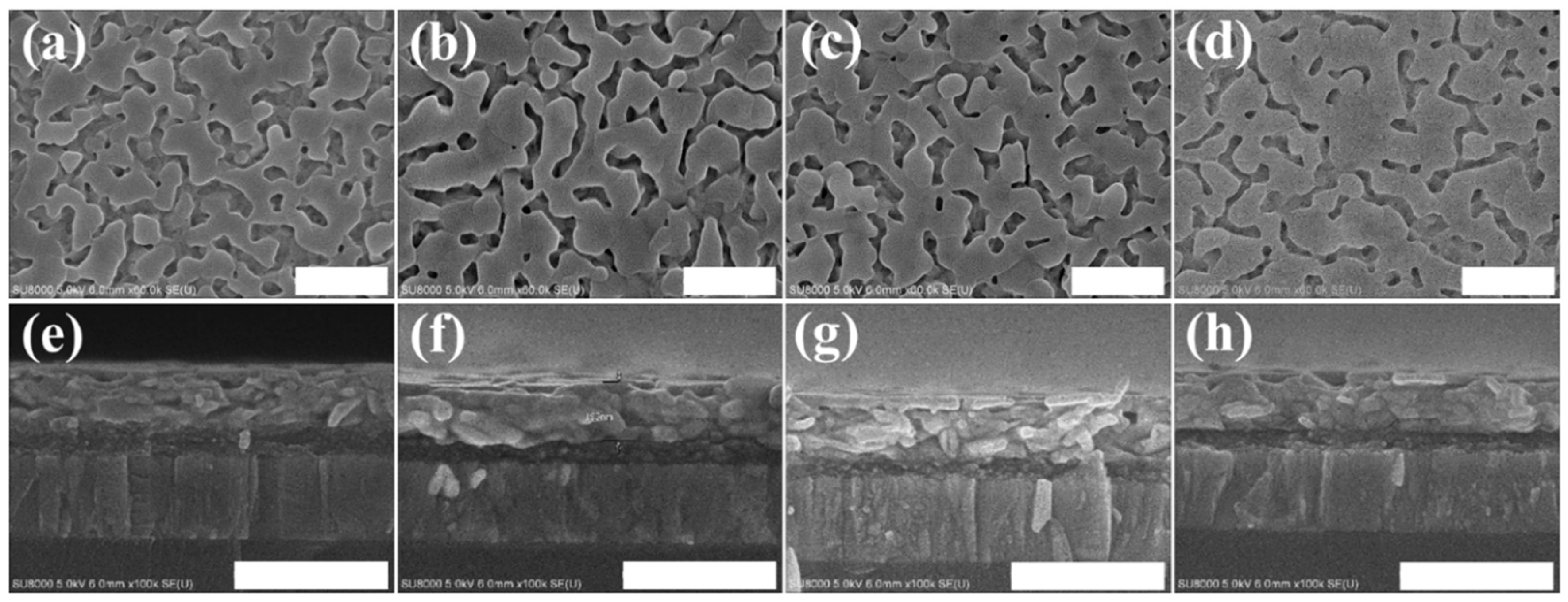
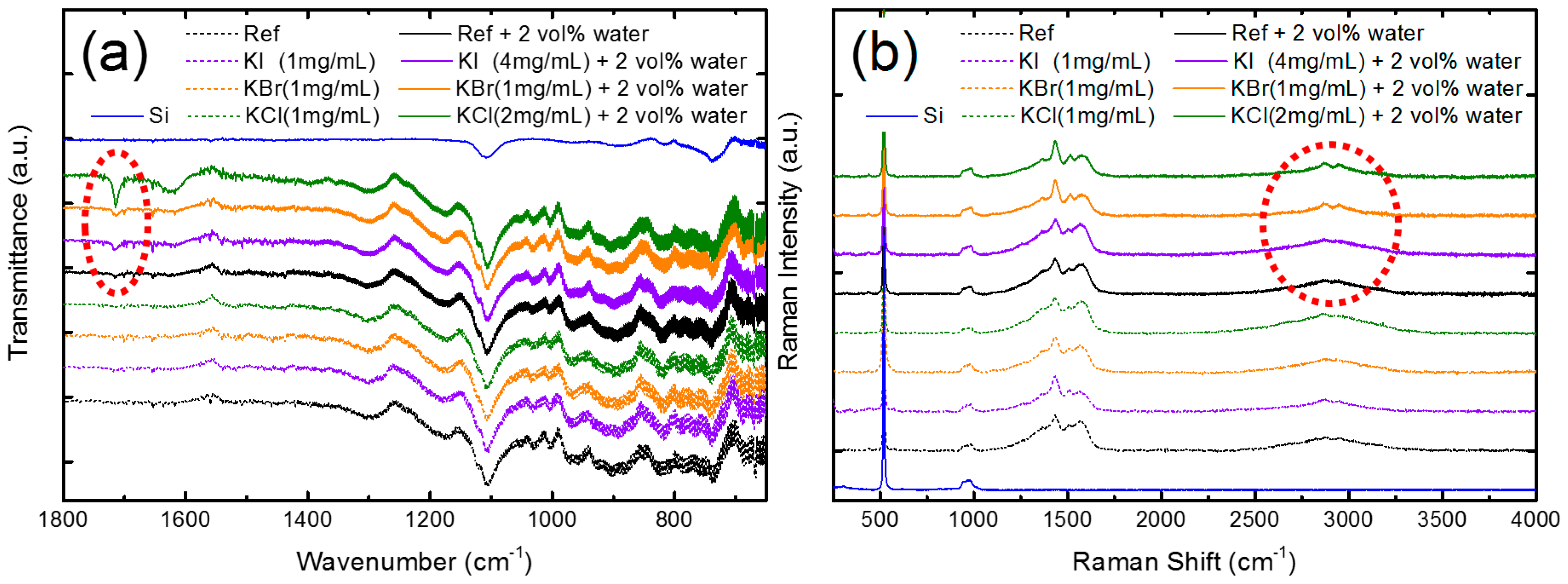
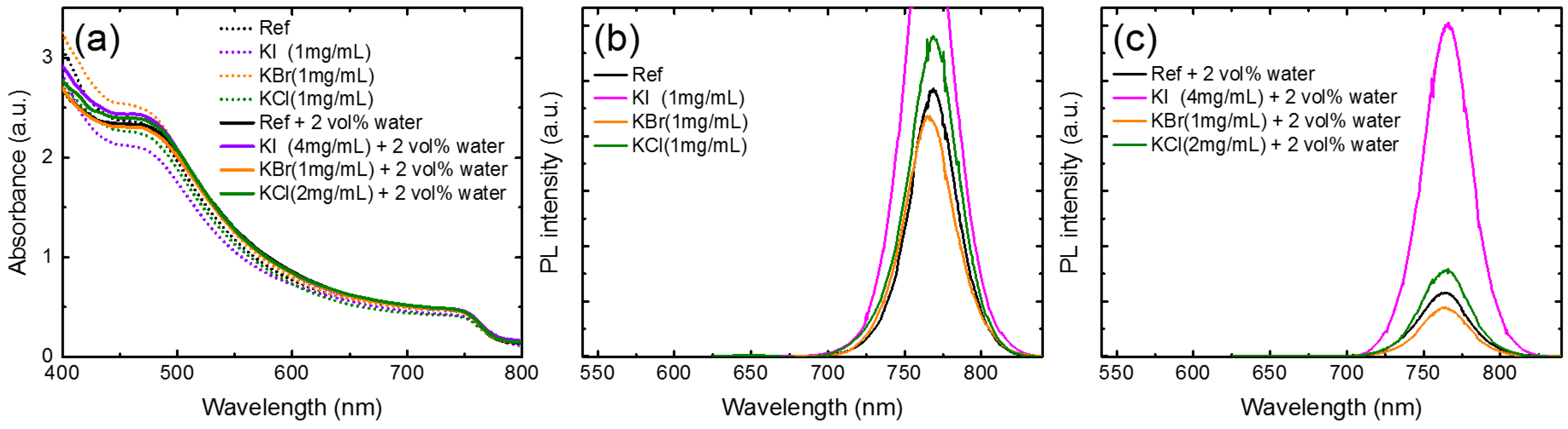
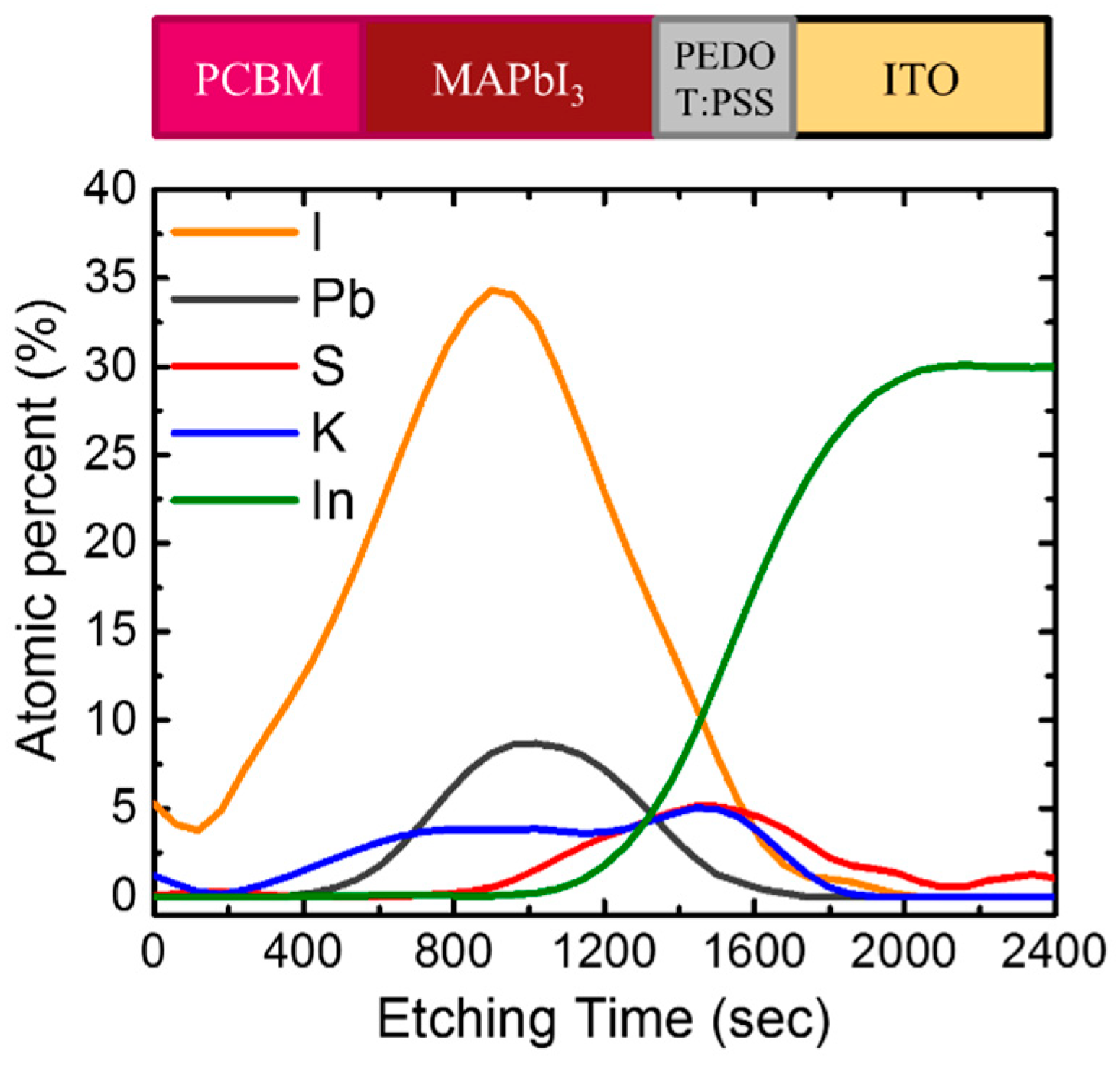
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal Halide Perovskites as Visible-Light Sensitizers for Photovoltaic Cells. J. Am. Chem. Soc. 2009, 131, 6050–6051. [Google Scholar] [CrossRef]
- Shin, S.S.; Yeom, E.J.; Yang, W.S.; Hur, S.; Kim, M.G.; Im, J.; Seo, J.; Noh, J.H.; Seok, S.I. Colloidally prepared La-doped BaSnO3 electrodes for efficient, photostable perovskite solar cells. Science 2017, 356, 167–171. [Google Scholar] [CrossRef]
- Saliba, M.; Matsui, T.; Seo, J.-Y.; Domanski, K.; Correa-Baena, J.-P.; Nazeeruddin, M.K.; Zakeeruddin, S.M.; Tress, W.; Abate, A.; Hagfeldt, A.; et al. Cesium-containing triple cation perovskite solar cells: Improved stability, reproducibility and high efficiency. Energy Environ. Sci. 2016, 9, 1989–1997. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.-H.; Nazeeruddin, M.K.; Grätzel, M.; Wu, C.-G. The synergistic effect of H2O and DMF towards stable and 20% efficiency inverted perovskite solar cells. Energy Environ. Sci. 2017, 10, 808–817. [Google Scholar] [CrossRef]
- Zuo, C.; Vak, D.; Angmo, D.; Ding, L.; Gao, M. One-step roll-to-roll air processed high efficiency perovskite solar cells. Nano Energy 2018, 46, 185–192. [Google Scholar] [CrossRef]
- Bi, C.; Wang, Q.; Shao, Y.; Yuan, Y.; Xiao, Z.; Huang, J. Non-wetting surface-driven high-aspect-ratio crystalline grain growth for efficient hybrid perovskite solar cells. Nat. Commun. 2015, 6, 7747. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.T.; Sai, H.; Tan, K.W.; Smilgies, D.-M.; Zhang, W.; Snaith, H.J.; Wiesner, U.; Estroff, L.A. Crystallization Kinetics of Organic–Inorganic Trihalide Perovskites and the Role of the Lead Anion in Crystal Growth. J. Am. Chem. Soc. 2015, 137, 2350–2358. [Google Scholar] [CrossRef]
- Fei, C.; Guo, L.; Li, B.; Zhang, R.; Fu, H.; Tian, J.; Cao, G. Controlled growth of textured perovskite films towards high performance solar cells. Nano Energy 2016, 27, 17–26. [Google Scholar] [CrossRef]
- Song, X.; Wang, W.; Sun, P.; Ma, W.; Chen, Z.-K. Additive to regulate the perovskite crystal film growth in planar heterojunction solar cells. Appl. Phys. Lett. 2015, 106, 033901. [Google Scholar] [CrossRef]
- Wu, Y.; Xie, F.; Chen, H.; Yang, X.; Su, H.; Cai, M.; Zhou, Z.; Noda, T.; Han, L. Thermally Stable MAPbI3 Perovskite Solar Cells with Efficiency of 19.19% and Area over 1 cm2 achieved by Additive Engineering. Adv. Mater. 2017, 29, 1701073. [Google Scholar] [CrossRef]
- Ke, W.; Xiao, C.; Wang, C.; Saparov, B.; Duan, H.-S.; Zhao, D.; Xiao, Z.; Schulz, P.; Harvey, S.P.; Liao, W.; et al. Employing Lead Thiocyanate Additive to Reduce the Hysteresis and Boost the Fill Factor of Planar Perovskite Solar Cells. Adv. Mater. 2016, 28, 5214–5221. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wang, J.; Zhang, X.; Wang, H.; Cui, X.; Yuan, S.; Lu, H.; Tu, L.; Zhan, Y.; Zheng, L. Boosting the performance of perovskite solar cells through a novel active passivation method. J. Mater. Chem. A 2018, 6, 15853–15858. [Google Scholar] [CrossRef]
- Tang, Z.; Bessho, T.; Awai, F.; Kinoshita, T.; Maitani, M.M.; Jono, R.; Murakami, T.N.; Wang, H.; Kubo, T.; Uchida, S.; et al. Hysteresis-free perovskite solar cells made of potassium-doped organometal halide perovskite. Sci. Rep. 2017, 7, 12183. [Google Scholar] [CrossRef] [PubMed]
- Nam, J.K.; Chai, S.U.; Cha, W.; Choi, Y.J.; Kim, W.; Jung, M.S.; Kwon, J.; Kim, D.; Park, J.H. Potassium Incorporation for Enhanced Performance and Stability of Fully Inorganic Cesium Lead Halide Perovskite Solar Cells. Nano Lett. 2017, 17, 2028–2033. [Google Scholar] [CrossRef]
- Abdi-Jalebi, M.; Andaji-Garmaroudi, Z.; Cacovich, S.; Stavrakas, C.; Philippe, B.; Richter, J.M.; Alsari, M.; Booker, E.P.; Hutter, E.M.; Pearson, A.J.; et al. Maximizing and stabilizing luminescence from halide perovskites with potassium passivation. Nature 2018, 555, 497. [Google Scholar] [CrossRef]
- Son, D.-Y.; Kim, S.-G.; Seo, J.-Y.; Lee, S.-H.; Shin, H.; Lee, D.; Park, N.-G. Universal Approach toward Hysteresis-Free Perovskite Solar Cell via Defect Engineering. J. Am. Chem. Soc. 2018, 140, 1358–1364. [Google Scholar] [CrossRef]
- Wu, C.-G.; Chiang, C.-H.; Tseng, Z.-L.; Nazeeruddin, M.K.; Hagfeldt, A.; Gratzel, M. High efficiency stable inverted perovskite solar cells without current hysteresis. Energy Environ. Sci. 2015, 8, 2725–2733. [Google Scholar] [CrossRef]
- Xiu, G.; Meng, L.; Xiao-Bo, S.; Heng, M.; Zhao-Kui, W.; Liang-Sheng, L. Controllable Perovskite Crystallization by Water Additive for High-Performance Solar Cells. Adv. Funct. Mater. 2015, 25, 6671–6678. [Google Scholar] [CrossRef]
- Boopathi, K.M.; Mohan, R.; Huang, T.-Y.; Budiawan, W.; Lin, M.-Y.; Lee, C.-H.; Ho, K.-C.; Chu, C.-W. Synergistic improvements in stability and performance of lead iodide perovskite solar cells incorporating salt additives. J. Mater. Chem. A 2016, 4, 1591–1597. [Google Scholar] [CrossRef]
- Bass, K.K.; McAnally, R.E.; Zhou, S.; Djurovich, P.I.; Thompson, M.E.; Melot, B.C. Influence of moisture on the preparation, crystal structure, and photophysical properties of organohalide perovskites. Chem. Commun. 2014, 50, 15819–15822. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Yang, Y.; Hong, Z.; Song, T.-B.; Meng, L.; Liu, Y.; Jiang, C.; Zhou, H.; Chang, W.-H.; Li, G.; et al. Moisture assisted perovskite film growth for high performance solar cells. Appl. Phys. Lett. 2014, 105, 183902. [Google Scholar] [CrossRef]
- Sison, E.S.; Rahman, M.D.; Durham, D.L.; Hermanowski, J.; Ross, M.F.; Jennison, M.J. Dielectric and chemical characteristics of electron-beam-cured photoresist. In Proceedings of the SPIE’s 1995 Symposium on Microlithography, Santa Clara, CA, USA, 9 June 1995; p. 14. [Google Scholar]
- Cao, X.; Li, C.; Li, Y.; Fang, F.; Cui, X.; Yao, Y.; Wei, J. Enhanced performance of perovskite solar cells by modulating the Lewis acid–base reaction. Nanoscale 2016, 8, 19804–19810. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.B.; Li, Y.H.; Fang, F.; Cui, X.; Yao, Y.W.; Wei, J.Q. High quality perovskite films fabricated from Lewis acid–base adduct through molecular exchange. RSC Adv. 2016, 6, 70925–70931. [Google Scholar] [CrossRef]
- Winther-Jensen, B.; West, K. Vapor-Phase Polymerization of 3,4-Ethylenedioxythiophene: A Route to Highly Conducting Polymer Surface Layers. Macromolecules 2004, 37, 4538–4543. [Google Scholar] [CrossRef]
- Xu, J.; Wang, C.; Wang, T.; Wang, Y.; Kang, Q.; Liu, Y.; Tian, Y. Mechanisms for low-temperature direct bonding of Si/Si and quartz/quartz via VUV/O3 activation. RSC Adv. 2018, 8, 11528–11535. [Google Scholar] [CrossRef]
- Wilamowska, M.; Kujawa, M.; Michalska, M.; Lipińska, L.; Lisowska-Oleksiak, A. Electroactive polymer/graphene oxide nanostructured composites; evidence for direct chemical interactions between PEDOT and GOx. Synth. Met. 2016, 220, 334–346. [Google Scholar] [CrossRef]
- Chang, J.; Lin, Z.; Zhu, H.; Isikgor, F.H.; Xu, Q.-H.; Zhang, C.; Hao, Y.; Ouyang, J. Enhancing the photovoltaic performance of planar heterojunction perovskite solar cells by doping the perovskite layer with alkali metal ions. J. Mater. Chem. A 2016, 4, 16546–16552. [Google Scholar] [CrossRef]

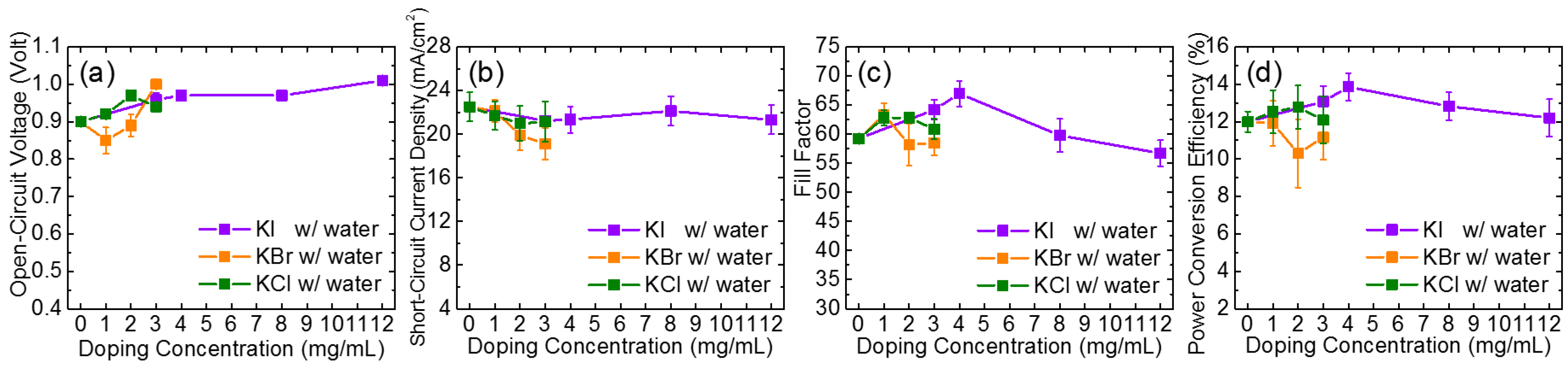

| Samples | VOC (V) | JSC (mA/cm2) | FF | PCE (%) | RSH (Ω·cm2) | RS (Ω·cm2) |
|---|---|---|---|---|---|---|
| Ref | 0.79 ± 0.03 | 19.7 ± 1.4 | 56.3 ± 1.6 | 8.8 ± 1.1 | 340 ± 99 | 7.2 ± 0.7 |
| KI (1 mg/mL) | 0.99 ± 0.02 | 15.1 ± 0.8 | 50.1 ± 1.5 | 7.5 ± 0.6 | 262 ± 40 | 8.8 ± 1.6 |
| KBr (1 mg/mL) | 0.79 ± 0.05 | 19.8 ± 3.9 | 52.2 ± 4.8 | 8.2 ± 2.5 | 220 ± 70 | 11.7 ± 3.3 |
| KCl (1 mg/mL) | 0.96 ± 0.03 | 16.0 ± 1.0 | 52.4 ± 2.1 | 8.0 ± 0.6 | 337 ± 137 | 12.5 ± 1.8 |
| Ref + water | 0.90 ± 0.01 | 22.5 ± 1.3 | 59.2 ± 0.6 | 12.0 ± 0.6 | 168 ± 40 | 3.6 ± 0.1 |
| KI (4 mg/mL) + water | 0.97 ± 0.01 | 21.3 ± 1.2 | 67.0 ± 2.2 | 13.9 ± 0.7 | 555 ± 39 | 5.5 ± 0.7 |
| KBr (1 mg/mL) + water | 0.85 ± 0.04 | 22.1 ± 1.0 | 63.4 ± 1.9 | 11.9 ± 1.2 | 264 ± 46 | 3.8 ± 0.3 |
| KCl (2 mg/mL) + water | 0.97 ± 0.01 | 21.0 ± 1.6 | 62.8 ± 0.8 | 12.8 ± 1.2 | 1217 ± 106 | 4.7 ± 0.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, H.-T.; Cheng, Y.-T.; Leu, C.-C.; Wu, S.-H.; Shih, C.-F. Improving Two-Step Prepared CH3NH3PbI3 Perovskite Solar Cells by Co-Doping Potassium Halide and Water in PbI2 Layer. Nanomaterials 2019, 9, 666. https://doi.org/10.3390/nano9050666
Wu H-T, Cheng Y-T, Leu C-C, Wu S-H, Shih C-F. Improving Two-Step Prepared CH3NH3PbI3 Perovskite Solar Cells by Co-Doping Potassium Halide and Water in PbI2 Layer. Nanomaterials. 2019; 9(5):666. https://doi.org/10.3390/nano9050666
Chicago/Turabian StyleWu, Hsuan-Ta, Yu-Ting Cheng, Ching-Chich Leu, Shih-Hsiung Wu, and Chuan-Feng Shih. 2019. "Improving Two-Step Prepared CH3NH3PbI3 Perovskite Solar Cells by Co-Doping Potassium Halide and Water in PbI2 Layer" Nanomaterials 9, no. 5: 666. https://doi.org/10.3390/nano9050666
APA StyleWu, H.-T., Cheng, Y.-T., Leu, C.-C., Wu, S.-H., & Shih, C.-F. (2019). Improving Two-Step Prepared CH3NH3PbI3 Perovskite Solar Cells by Co-Doping Potassium Halide and Water in PbI2 Layer. Nanomaterials, 9(5), 666. https://doi.org/10.3390/nano9050666