Nanomaterials for the Removal of Heavy Metals from Wastewater
Abstract
:1. Introduction
2. Adsorption Isotherms and Kinetics
2.1. Adsorption Isotherms
2.1.1. Langmuir Model
2.1.2. Freundlich Model
2.1.3. Sips Model
2.2. Adsorption Kinetics
3. Nanomaterials for Removing Heavy Metals
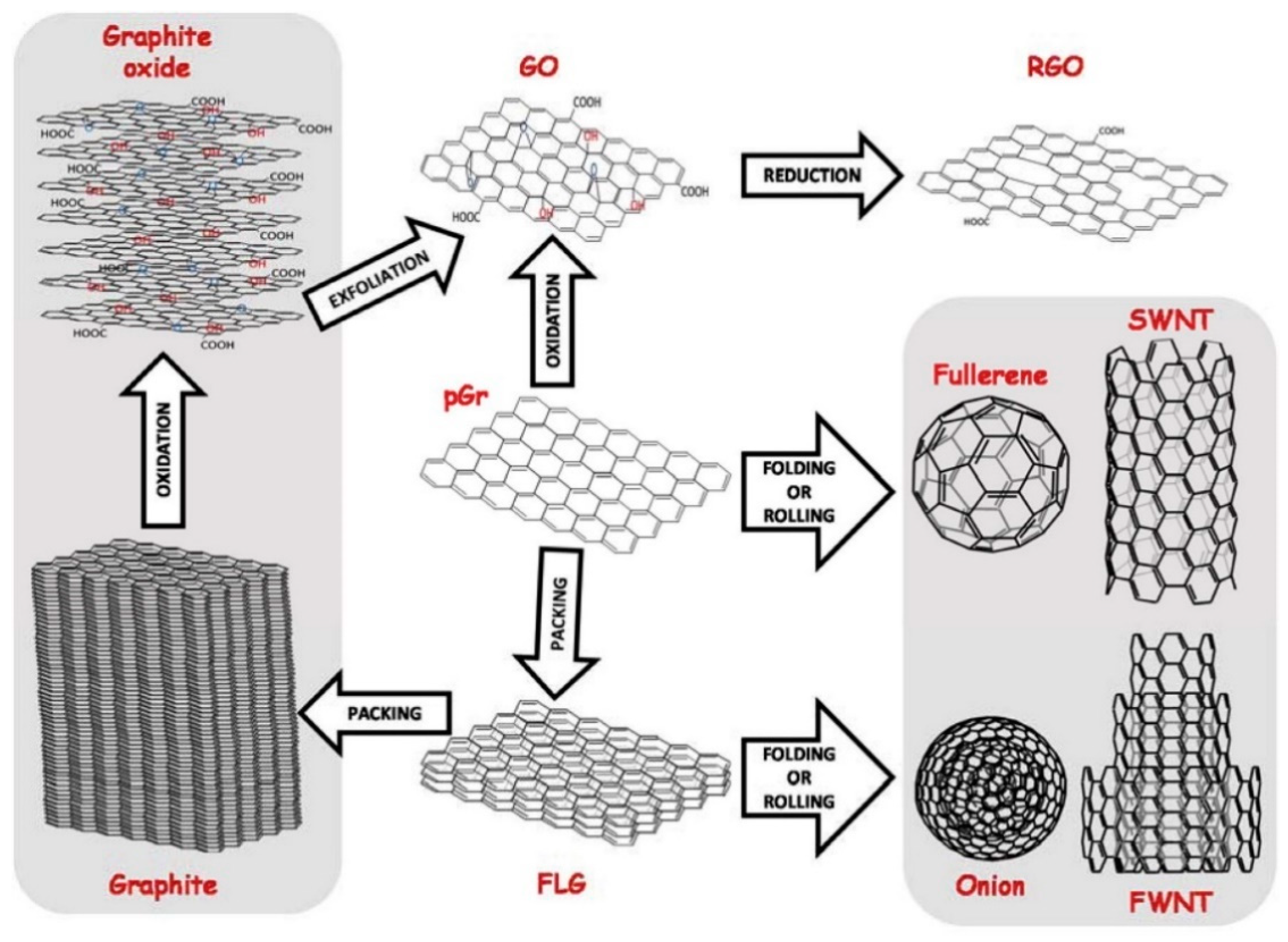
3.1. Carbon-Based Nanomaterials
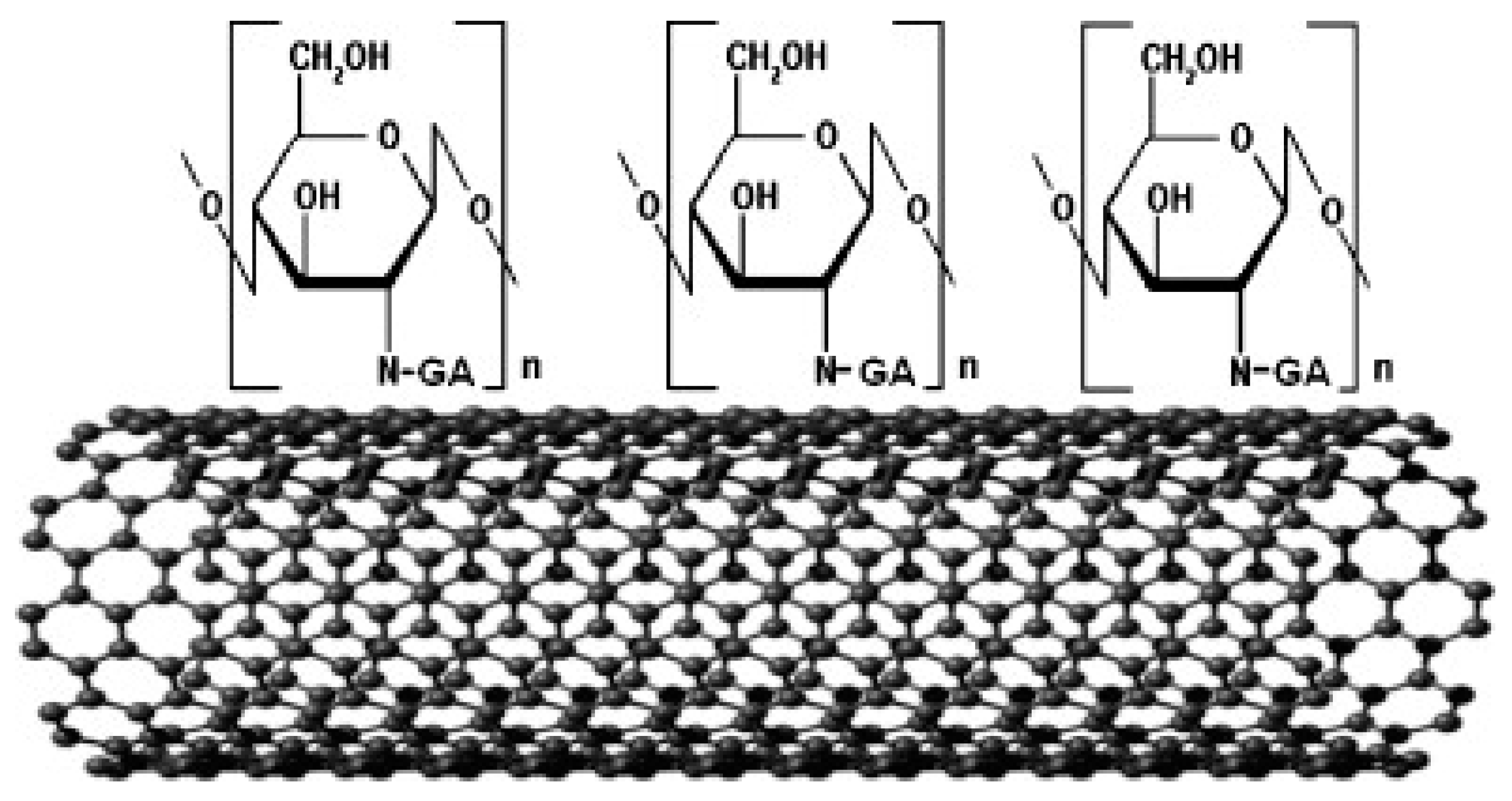
3.1.1. Carbon Nanotubes
3.1.2. Graphene Nanomaterials
3.2. Silica-Based Nanomaterials
3.3. Zero-Valent Metal-Based Nanomaterials
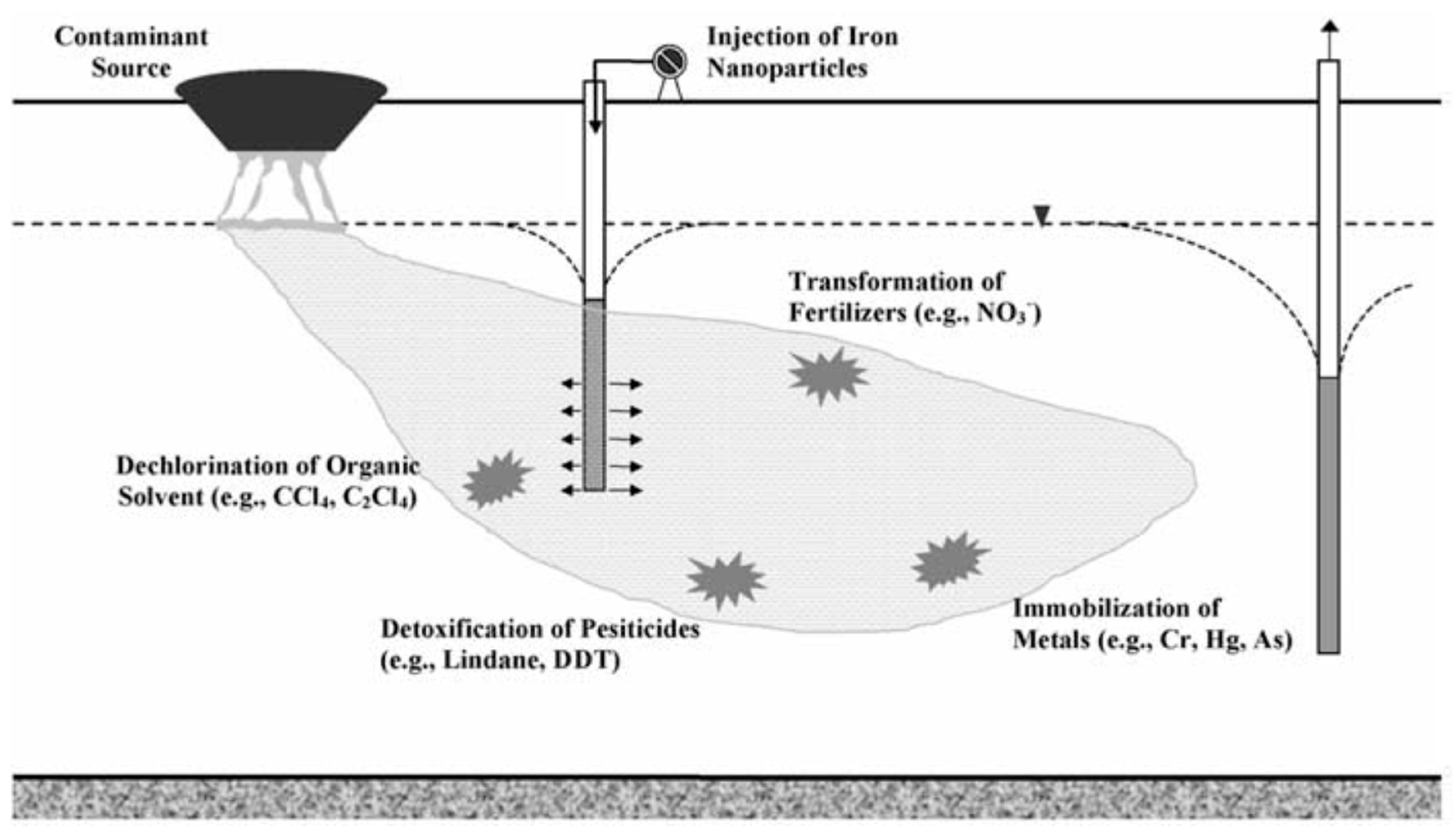
3.3.1. Zero-Valent Iron
3.3.2. Ag Nanoparticles
3.3.3. Au Nanoparticles
3.4. Metal Oxide-Based Nanomaterials
3.4.1. Iron Oxides-Based Nanomaterials
Goethite (α-FeOOH)
Hematite (α-Fe2O3)
Maghemite (γ-Fe2O3)
Magnetite (Fe3O4)
Hydrous Iron Oxides (HFO)
3.4.2. Manganese Oxides-Based Nanomaterials
3.4.3. Zinc Oxides-Based Nanomaterials
3.4.4. Titanium Oxides-Based Nanomaterials
3.4.5. Aluminum Oxides-Based Nanomaterials
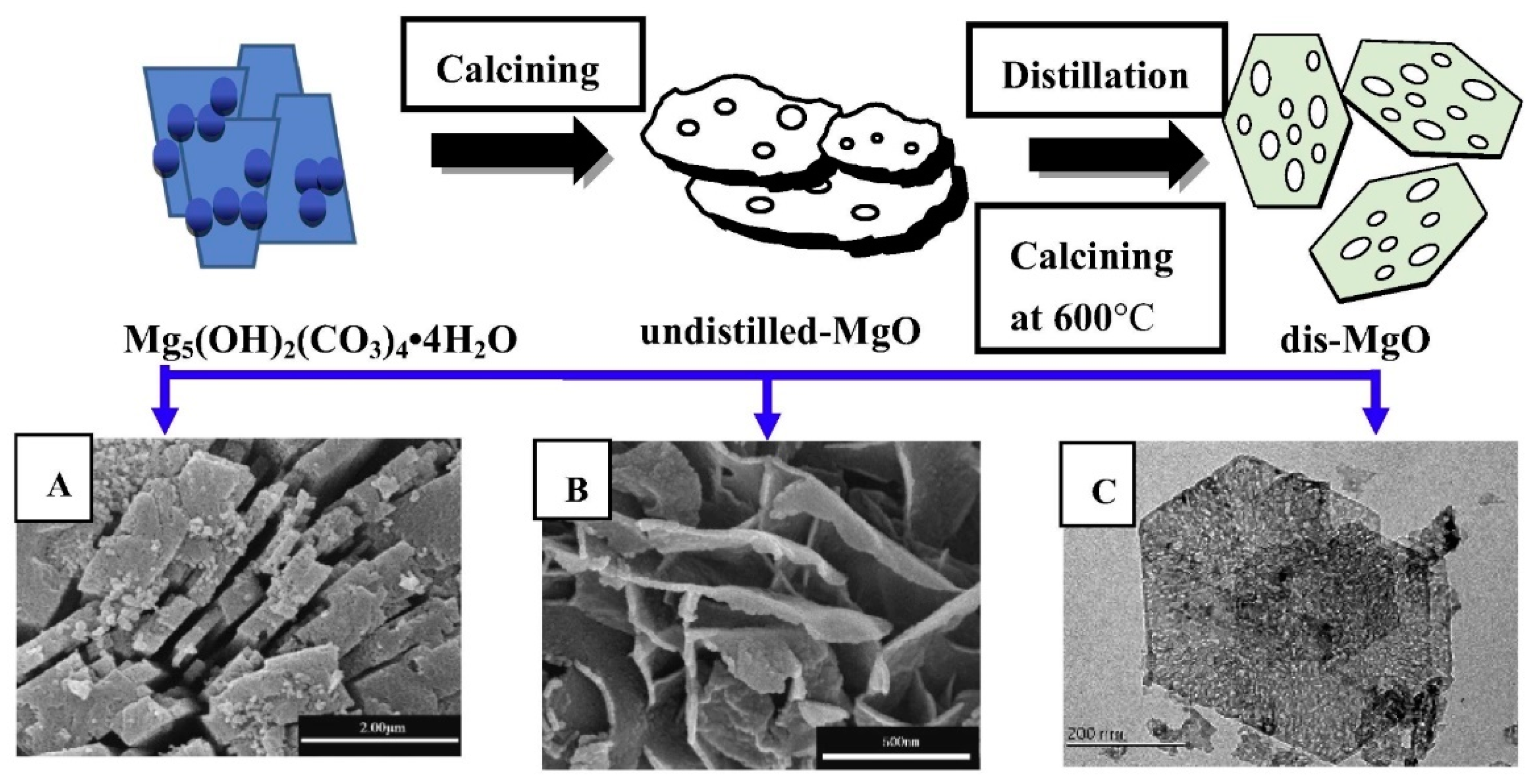
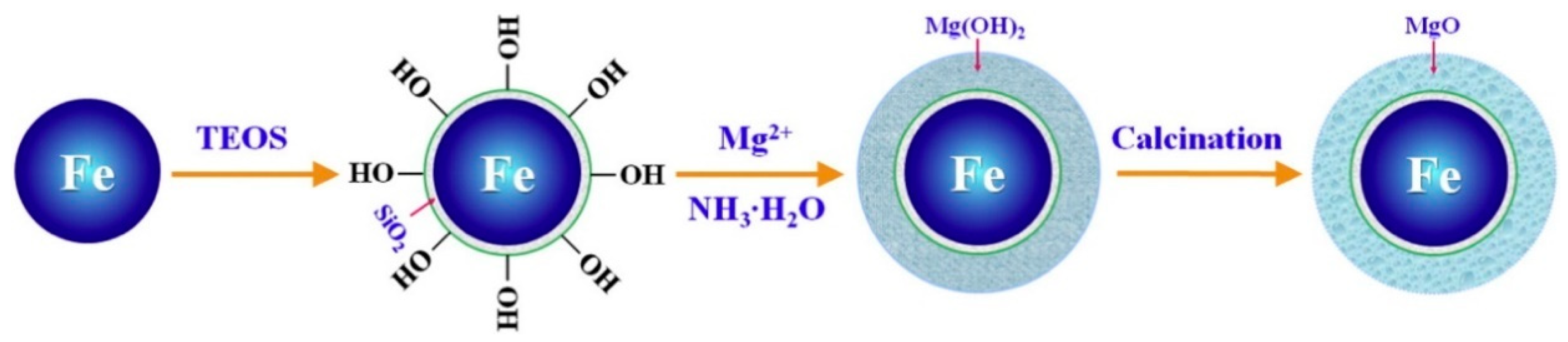
3.4.6. Magnesium Oxides-Based Nanomaterials
3.4.7. Cerium Oxides-Based Nanomaterials
3.4.8. Zirconium Oxides-Based Nanomaterials
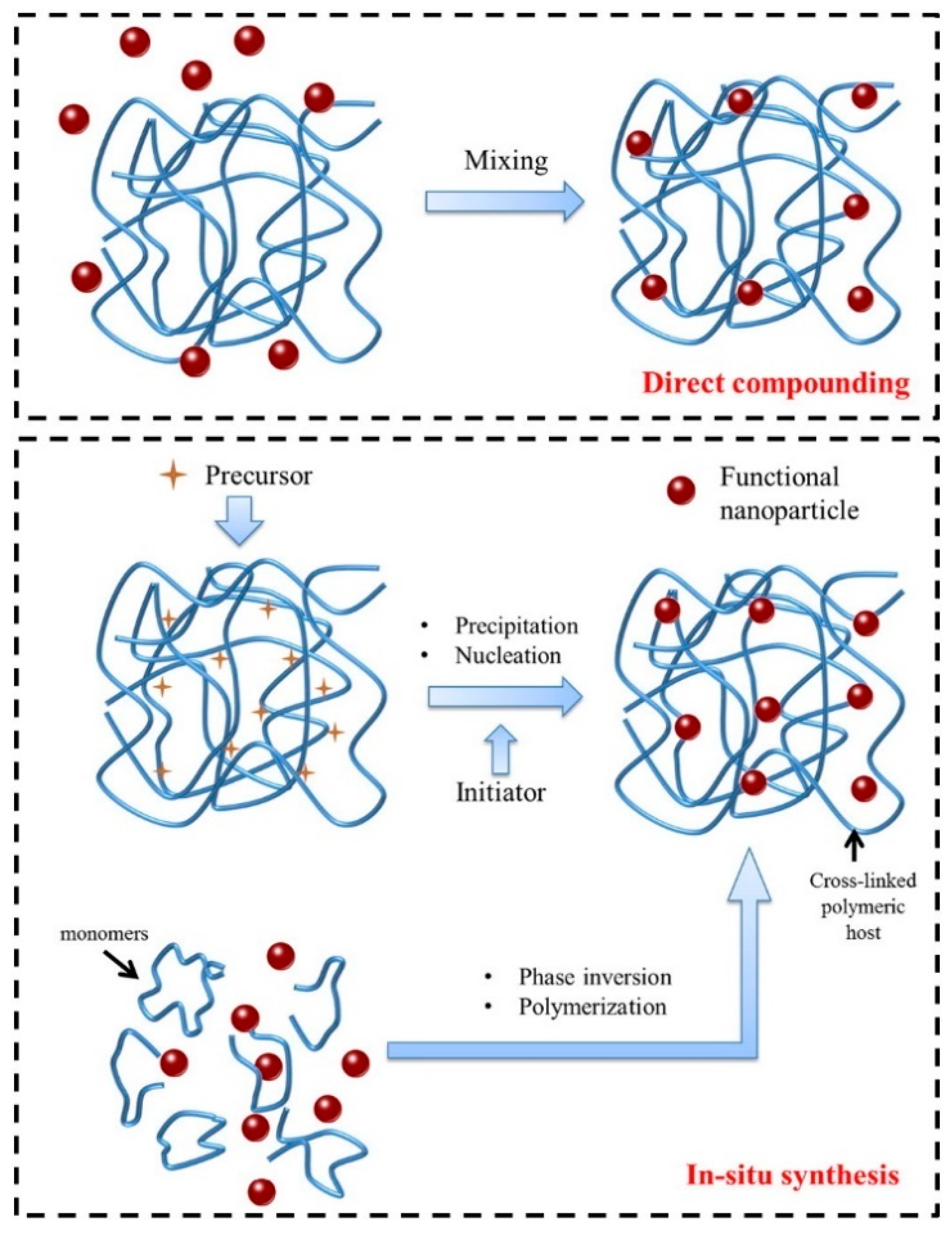
3.5. Nanocomposite Nanomaterials
3.5.1. Inorganic-Supported Nanocomposites
3.5.2. Organic Polymer-Supported Nanocomposites
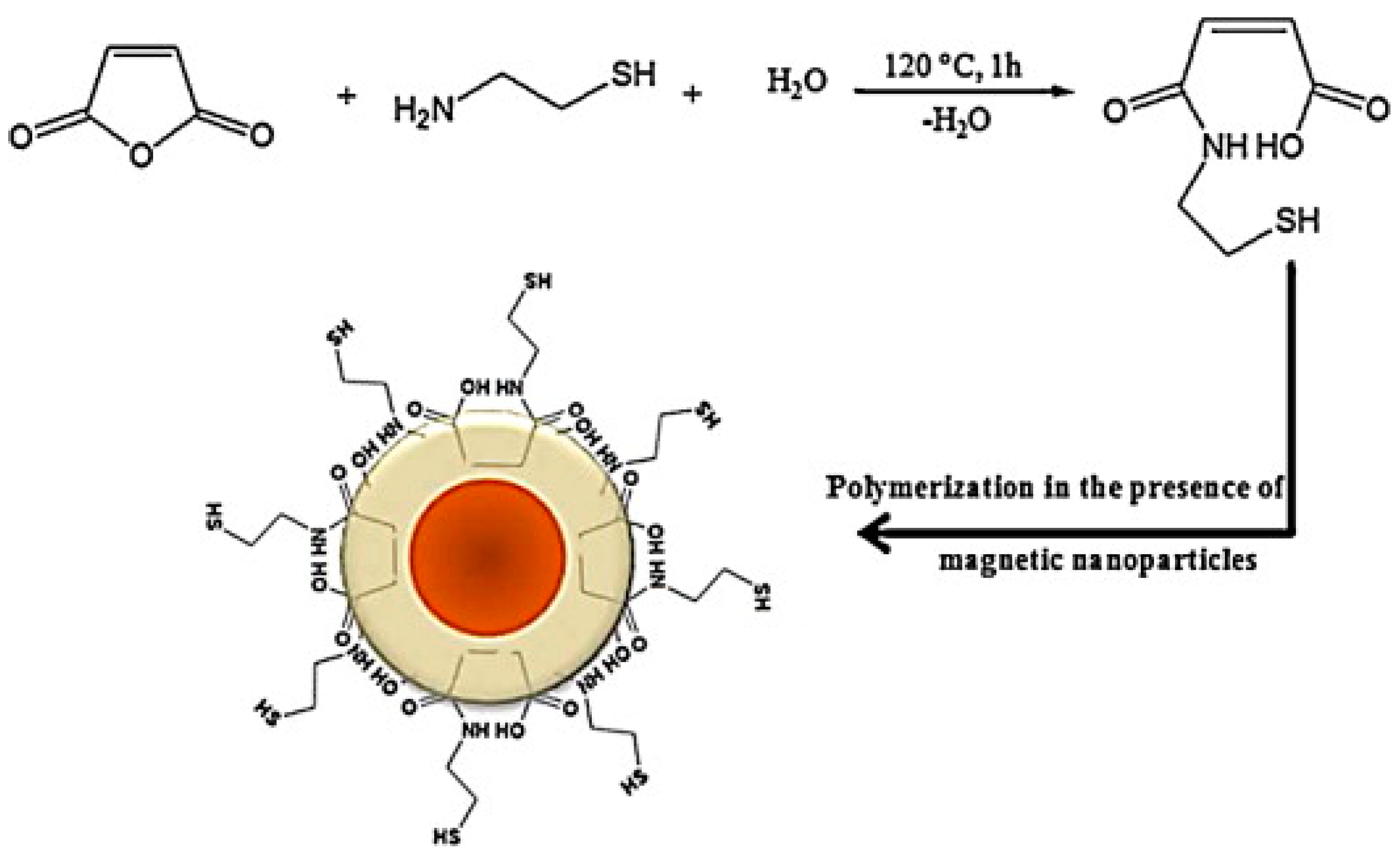
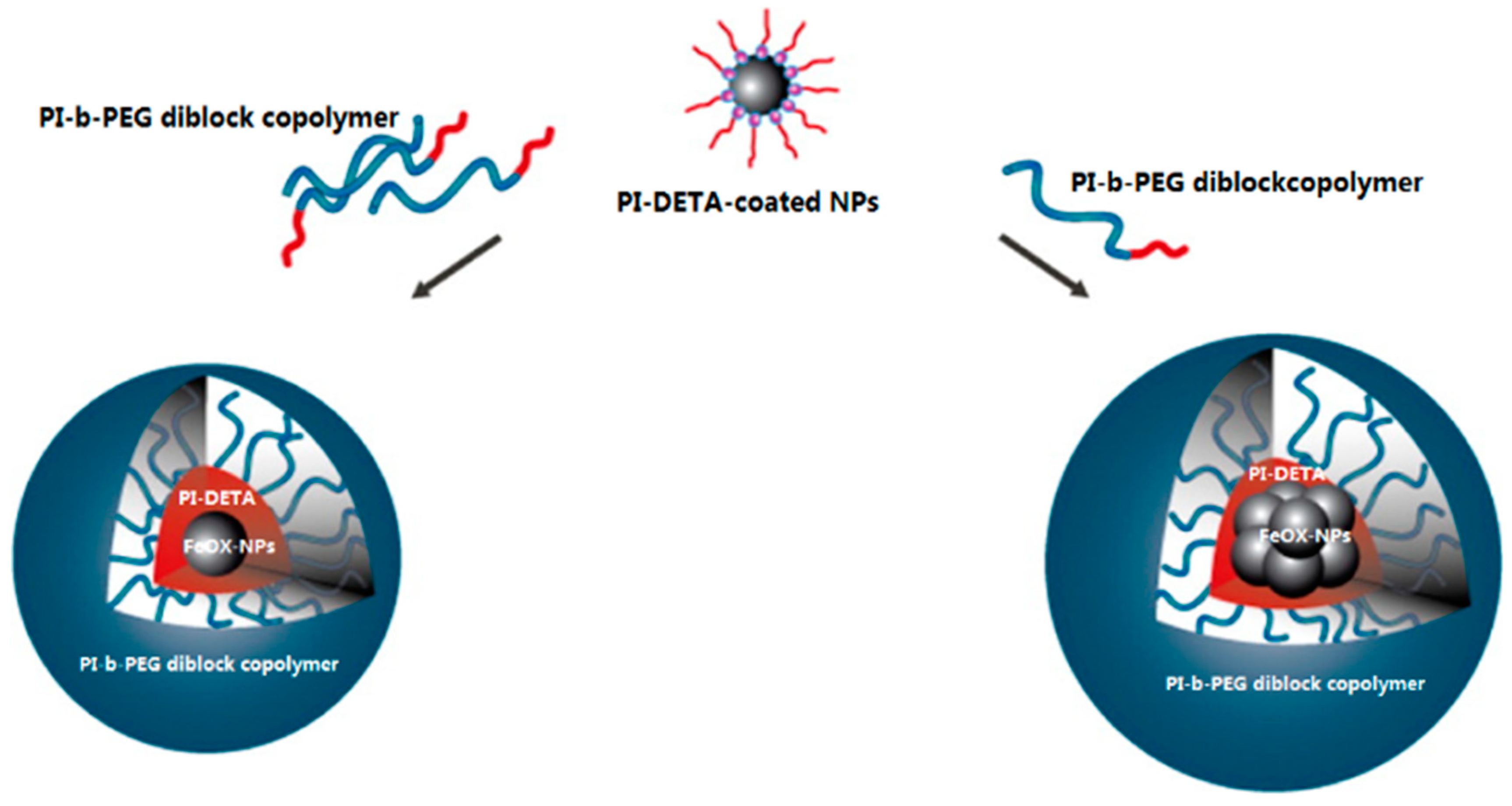
3.5.3. Magnetic Nanocomposites
4. Conclusions and Perspectives
Funding
Conflicts of Interest
References
- Cheraghi, M.; Lorestani, B.; Yousefi, N. Effect of waste water on heavy metal accumulation in Hamedan Province vegetables. Int. J. Bot. 2009, 5, 190–193. [Google Scholar] [CrossRef]
- Reglero, M.M.; Taggart, M.A.; Lidia, M.G.; Rafael, M. Heavy metal exposure in large game from a lead mining area: Effects on oxidative stress and fatty acid composition in liver. Environ. Pollut. 2009, 157, 1388–1395. [Google Scholar] [CrossRef] [PubMed]
- Gybina, A.A.; Prohaska, J.R. Copper deficiency results in AMP-activated protein kinase activation and acetylCoA carboxylase phosphorylation in rat cerebellum. Brain Res. 2008, 1204, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Kampa, M.; Castanas, E. Human health effects of air pollution. Environ. Pollut. 2008, 151, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Afroze, S.; Sen, T.K. A Review on Heavy Metal Ions and Dye Adsorption from Water by Agricultural Solid Waste Adsorbents. Water Air Soil Pollut. 2018, 229, 225. [Google Scholar] [CrossRef]
- Cocârţă, D.; Neamţu, S.; Deac, A.R. Carcinogenic risk evaluation for human health risk assessment from soils contaminated with heavy metals. Int. J. Environ. Sci. Technol. 2016, 13, 2025–2036. [Google Scholar] [CrossRef]
- González-Muñoz, M.J.; Rodríguez, M.A.; Luque, S.; Álvarez, J.R. Recovery of heavy metals from metal industry waste waters by chemical precipitation and nanofiltration. Desalination 2006, 200, 742–744. [Google Scholar] [CrossRef]
- Verbych, S.; Hilal, N.; Sorokin, G.; Leaper, M. Ion Exchange Extraction of Heavy Metal Ions from Wastewater. Separat. Sci. Technol. 2004, 39, 2031–2040. [Google Scholar] [CrossRef]
- Namasivayam, C.; Sangeetha, D. Recycling of agricultural solid waste, coir pith: Removal anions, heavy metals, organics and dyes from water by adsorption onto ZnCl2 activated coir pith carbon. J. Hazard. Mater. 2006, 135, 449–452. [Google Scholar] [CrossRef]
- Sudilovskiy, P.S.; Kagramanov, G.G.; Trushin, A.M.; Kolesnikov, V.A. Use of membranes for heavy metal cationic wastewater treatment: Flotation and membrane filtration. Clean Technol. Environ. Policy 2007, 9, 189–198. [Google Scholar] [CrossRef]
- Tran, T.K.; Leu, H.J.; Chiu, K.F.; Lin, C.Y. Electrochemical Treatment of Heavy Metal-containing Wastewater with the Removal of COD and Heavy Metal Ions: Electrochemical treatment of heavy metal containing wastewater. J. Chin. Chem. Soc. 2017, 64, 493–502. [Google Scholar] [CrossRef]
- Mavrov, V.; Erwe, T.; Blöcher, C.; Chmiel, H. Study of new integrated processes combining adsorption, membrane separation and flotation for heavy metal removal from wastewater **. Desalination 2003, 157, 97–104. [Google Scholar] [CrossRef]
- Blöcher, C.; Dorda, J.; Mavrov, V.; Chmiel, H.; Lazaridis, N.K.; Matis, K.A. Hybrid flotation—Membrane filtration process for the removal of heavy metal ions from wastewater. Water Res. 2003, 37, 4018–4026. [Google Scholar] [CrossRef]
- Khan, N.A.; Hasan, Z.; Jhung, S.H. Adsorptive removal of hazardous materials using metal-organic frameworks (MOFs): A review. J. Hazard. Mater. 2013, 244–245, 444–456. [Google Scholar] [CrossRef]
- Ricco, R.; Konstas, K.; Styles, M.J.; Richardson, J.J.; Babarao, R.; Suzuki, K.; Scopece, P.; Falcaro, P. Lead(II) uptake by aluminium based magnetic framework composites (MFCs) in water. J. Mater. Chem. A 2015, 3, 19822–19831. [Google Scholar] [CrossRef]
- Fatikow, S.; Eichhorn, V.; Bartenwerfer, M. Nanomaterials Enter the Silicon-Based CMOS Era: Nanorobotic Technologies for Nanoelectronic Devices. IEEE Nanotechnol. Mag. 2012, 6, 14–18. [Google Scholar] [CrossRef]
- Kumar, S.; Ahlawat, W.; Kumar, R.; Dilbaghi, N. Graphene, carbon nanotubes, zinc oxide and gold as elite nanomaterials for fabrication of biosensors for healthcare. Biosens. Bioelectron. 2015, 70, 498–503. [Google Scholar] [CrossRef]
- Pumera, M. Graphene-based nanomaterials for energy storage. Energy Environ. Sci. 2011, 4, 668–674. [Google Scholar] [CrossRef]
- Kharisov, B.I.; Kharissova, O.V.; Dias, H.V.R. Nanomaterials for Environmental Protection; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014. [Google Scholar]
- Buzea, C.; Pacheco, I. Nanomaterial and Nanoparticle: Origin and Activity; Springer International Publishing: Cham, Switzerland, 2017. [Google Scholar]
- Shi, Y.H.; Wang, C.Z.; Zi-Rong, X.U. The application and prospect of nanotechnology in animal husbandry. J. Northwest Sci-Tech Univ. Agric. For. (Nat. Sci. Ed.) 2006, 34, 49–52. [Google Scholar]
- Vunain, E.; Mishra, A.K.; Mamba, B.B. Dendrimers, mesoporous silicas and chitosan-based nanosorbents for the removal of heavy-metal ions: A review. Int. J. Biol. Macromol. 2016, 86, 570–586. [Google Scholar] [CrossRef]
- Lim, J.Y.; Mubarak, N.M.; Abdullah, E.C.; Nizamuddin, S.; Khalid, M.; Inamuddin. Recent trends in the synthesis of graphene and graphene oxide based nanomaterials for removal of heavy metals—A review. J. Ind. Eng. Chem. 2018, 66, 29–44. [Google Scholar] [CrossRef]
- Nujić, M.; Habuda-Stanić, M. Toxic Metal Ions in Drinking Water and Effective Removal Using Graphene Oxide Nanocomposite; Springer International Publishing: Cham, Switzerland, 2019; pp. 373–395. [Google Scholar]
- Ihsanullah; Abbas, A.; Al-Amer, A.M.; Laoui, T.; Al-Marri, M.J.; Nasser, M.S.; Khraisheh, M.; Atieh, M.A. Heavy metal removal from aqueous solution by advanced carbon nanotubes: Critical review of adsorption applications. Separat. Purif. Technol. 2016, 157, 141–161. [Google Scholar] [CrossRef]
- Ming, H.; Zhang, S.; Pan, B.; Zhang, W.; Lu, L.; Zhang, Q. Heavy metal removal from water/wastewater by nanosized metal oxides: A review. J. Hazard. Mater. 2012, 211–212, 317–331. [Google Scholar]
- Wang, X. Nanomaterials as Sorbents to Remove Heavy Metal Ions in Wastewater Treatment. J. Environ. Anal. Toxicol. 2012, 02, 154. [Google Scholar] [CrossRef]
- Lee, Z.L.; Zaini, M.A.A.; Tang, S.H. Porous Nanomaterials for Heavy Metal Removal; Springer International Publishing: Cham, Switzerland, 2019. [Google Scholar]
- Theodore, L.; Ricci, F. Mass Transfer Operations for the Practicing Engineer; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011. [Google Scholar]
- Putro, J.N.; Santoso, S.P.; Ismadji, S.; Ju, Y.H. Investigation of heavy metal adsorption in binary system by nanocrystalline cellulose—Bentonite nanocomposite: Improvement on extended Langmuir isotherm model. Microporous Mesoporous Mater. 2017, 246, 166–177. [Google Scholar] [CrossRef]
- Baseri, H.; Tizro, S. Treatment of nickel ions from contaminated water by magnetite based nanocomposite adsorbents: Effects of thermodynamic and kinetic parameters and modeling with Langmuir and Freundlich isotherms. Process Saf. Environ. Protect. 2017, 109, 465–477. [Google Scholar] [CrossRef]
- Danish, M.; Hashim, R.; Mohamad Ibrahim, M.N.; Rafatullah, M.; Sulaiman, O.; Ahmad, T.; Shamsuzzoha, M.; Ahmad, A. Sorption of Copper(II) and Nickel(II) Ions from Aqueous Solutions Using Calcium Oxide Activated Date (Phoenix dactylifera) Stone Carbon: Equilibrium, Kinetic, and Thermodynamic Studies. J. Chem. Eng. Data 2011, 56, 3607–3619. [Google Scholar] [CrossRef]
- Repo, E.; Warchol, J.K.; Kurniawan, T.A.; Sillanpää, M.E.T. Adsorption of Co(II) and Ni(II) by EDTA- and/or DTPA-modified chitosan: Kinetic and equilibrium modeling. Chem. Eng. J. 2010, 161, 73–82. [Google Scholar] [CrossRef]
- Wei, S.; Chen, S.; Shi, S.; Li, X.; Zhang, X.; Hu, W.; Wang, H. Adsorption of Cu(II) and Pb(II) onto diethylenetriamine-bacterial cellulose. Carbohydr. Polym. 2009, 75, 110–114. [Google Scholar]
- Sha, L.; Guo, X.; Feng, N.; Tian, Q. Adsorption of Cu and Cd from aqueous solution by mercapto-acetic acid modified orange peel. Colloids Surf. B Biointerfaces 2009, 73, 10–14. [Google Scholar] [CrossRef]
- Ho, Y.-S.; Chiu, W.-T.; Wang, C.-C. Regression analysis for the sorption isotherms of basic dyes on sugarcane dust. Bioresour. Technol. 2005, 96, 1285–1291. [Google Scholar] [CrossRef]
- Ramanaiah, S.V.; Mohan, S.V.; Sarma, P.N. Adsorptive removal of fluoride from aqueous phase using waste fungus (Pleurotus ostreatus 1804) biosorbent: Kinetics evaluation. Ecol. Eng. 2007, 31, 47–56. [Google Scholar] [CrossRef]
- Freundlich, H. Over the adsorption in the solution. J. Phys. Chem. 1906, 57, 385–470. [Google Scholar]
- Sips, R. On the Structure of a Catalyst Surface. J. Chem. Phys. 2004, 16, 1024–1026. [Google Scholar] [CrossRef]
- Al-Asheh, S.; Banat, F.; Al-Omari, R.; Duvnjak, Z. Predictions of binary sorption isotherms for the sorption of heavy metals by pine bark using single isotherm data. Chemosphere 2000, 41, 659–665. [Google Scholar] [CrossRef]
- Ho, Y.S.; Porter, J.F.; Mckay, G. Equilibrium Isotherm Studies for the Sorption of Divalent Metal Ions onto Peat: Copper, Nickel and Lead Single Component Systems. Water Air Soil Pollut. 2002, 141, 1–33. [Google Scholar] [CrossRef]
- Repo, E.; Warchoł, J.K.; Bhatnagar, A.; Sillanpää, M. Heavy metals adsorption by novel EDTA-modified chitosan–silica hybrid materials. J. Colloid Interface Sci. 2011, 358, 261–267. [Google Scholar] [CrossRef]
- Matouq, M.; Jildeh, N.; Qtaishat, M.; Hindiyeh, M.; Syouf, M.Q.A. The adsorption kinetics and modeling for heavy metals removal from wastewater by Moringa pods. J. Environ. Chem. Eng. 2015, 3, 775–784. [Google Scholar] [CrossRef]
- ÇAvuş, S.; GuRdag, G.L. Noncompetitive Removal of Heavy Metal Ions from Aqueous Solutions by Poly[2-(acrylamido)-2-methyl-1-propanesulfonic acid-co-itaconic acid] Hydrogel. Ind. Eng. Chem. Res. 2016, 48, 2652–2658. [Google Scholar] [CrossRef]
- Farhan, A.M.; Salem, N.M.; Al-Dujaili, A.H.; Awwad, A.M. Biosorption Studies of Cr(VI) Ions from Electroplating Wastewater by Walnut Shell Powder. Am. J. Environ. Eng. 2012, 2, 188–195. [Google Scholar] [CrossRef]
- Dresselhaus, M.S.; Terrones, M. Carbon-Based Nanomaterials from a Historical Perspective. Proc. IEEE 2013, 101, 1522–1535. [Google Scholar] [CrossRef]
- Rodrigues, D.F. Carbon-Based Nanomaterials for Removal of Chemical and Biological Contaminants from Water: A Review of Mechanisms and Applications. Carbon 2015, 91, 122–143. [Google Scholar]
- Popov, V.N. Carbon nanotubes: Properties and application. Mater. Sci. Eng. R 2004, 43, 61–102. [Google Scholar] [CrossRef]
- Gupta, V.K.; Moradi, O.; Tyagi, I.; Agarwal, S.; Sadegh, H.; Shahryari-Ghoshekandi, R.; Makhlouf, A.S.H.; Goodarzi, M.; Garshasbi, A. Study on the removal of heavy metal ions from industry waste by carbon nanotubes: Effect of the surface modification: A review. Crit. Rev. Environ. Sci. Technol. 2016, 46, 93–118. [Google Scholar] [CrossRef]
- Martel, R.; Schmidt, T.; Shea, H.R.; Hertel, T.; Avouris, P. Single- and Multi-Wall Carbon Nanotube Field-Effect Transistors. Appl. Phys. Lett. 1998, 73, 2447–2449. [Google Scholar] [CrossRef]
- Yu, G.; Yang, L.; Jiang, G.; Patel, M.; Bafana, A.; Wang, X.; Qiu, B.; Jeffryes, C.; Wei, S.; Guo, Z. Carbon nanotubes, graphene, and their derivatives for heavy metal removal. Adv. Compos. Hybrid Mater. 2018, 1, 56–78. [Google Scholar] [CrossRef]
- Lu, H.; Wang, J.; Stoller, M.; Wang, T.; Ying, B.; Hao, H. An overview of nanomaterials for water and wastewater treatment. Adv. Mater. Sci. Eng. 2016, 2016, 1–10. [Google Scholar] [CrossRef]
- Yadav, D.K.; Srivastava, S. Carbon nanotubes as adsorbent to remove heavy metal ion (Mn+7) in wastewater treatment. Mater. Today Proc. 2017, 4, 4089–4094. [Google Scholar] [CrossRef]
- Pu, Y.; Yang, X.; Hong, Z.; Wang, D.; Yu, S.; Jie, H. Adsorption and desorption of thallium(I) on multiwalled carbon nanotubes. Chem. Eng. J. 2013, 219, 403–410. [Google Scholar] [CrossRef]
- Tang, W.W.; Zeng, G.M.; Gong, J.L.; Liu, Y.; Wang, X.Y.; Liu, Y.Y.; Liu, Z.F.; Chen, L.; Zhang, X.R.; Tu, D.Z. Simultaneous adsorption of atrazine and Cu(II) from wastewater by magnetic multi-walled carbon nanotube. Chem. Eng. J. 2012, 211–212, 470–478. [Google Scholar] [CrossRef]
- Kabbashi, N.A.; Atieh, M.A.; Al-Mamun, A.; Mirghami, M.E.S.; Alam, M.D.Z.; Yahy, N. Kinetic adsorption of application of carbon nanotubes for Pb(II) removal from aqueous solution. J. Environ. Sci. 2009, 21, 539–544. [Google Scholar] [CrossRef]
- Tuzen, M.; Soylak, M. Multiwalled carbon nanotubes for speciation of chromium in environmental samples. J. Hazard. Mater. 2007, 147, 219–225. [Google Scholar] [CrossRef]
- Sandeep, A.; Mota, J.P.B.; Massoud, R.A.; Rood, M.J. Theoretical and experimental investigation of morphology and temperature effects on adsorption of organic vapors in single-walled carbon nanotubes. J. Phys. Chem. B 2006, 110, 7640–7647. [Google Scholar]
- Kumar, R.; Khan, M.A.; Haq, N. Application of Carbon Nanotubes in Heavy Metals Remediation. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1000–1035. [Google Scholar] [CrossRef]
- El-Sheikh, A.H.; Al-Degs, Y.S.; Al-As’Ad, R.M.; Sweileh, J.A. Effect of oxidation and geometrical dimensions of carbon nanotubes on Hg(II) sorption and preconcentration from real waters. Desalination 2011, 270, 214–220. [Google Scholar] [CrossRef]
- Alomar, M.K.; Alsaadi, M.A.; Hayyan, M.; Akib, S.; Ibrahim, M.; Hashim, M.A. Allyl triphenyl phosphonium bromide based DES-functionalized carbon nanotubes for the removal of mercury from water. Chemosphere 2017, 167, 44–52. [Google Scholar] [CrossRef]
- Xu, J.; Cao, Z.; Zhang, Y.; Yuan, Z.; Lou, Z.; Xu, X.; Wang, X. A review of functionalized carbon nanotubes and graphene for heavy metal adsorption from water: Preparation, application, and mechanism. Chemosphere 2018, 195, 351. [Google Scholar] [CrossRef]
- Zhan, Y.; Hai, H.; Yi, H.; Long, Z.; Wan, X.; Zeng, G. Novel amino-functionalized Fe3O4/carboxylic multi-walled carbon nanotubes: One-pot synthesis, characterization and removal for Cu(II). Russ. J. Appl. Chem. 2016, 89, 1894–1902. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Fal’Ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef]
- Gao, W.; Majumder, M.; Alemany, L.B.; Narayanan, T.N.; Ibarra, M.A.; Pradhan, B.K.; Ajayan, P.M. Engineered graphite oxide materials for application in water purification. ACS Appl. Mater. Interfaces 2011, 3, 1821–1826. [Google Scholar] [CrossRef]
- Avouris, P.; Dimitrakopoulos, C. Graphene: Synthesis and applications. Mater. Today 2012, 15, 86–97. [Google Scholar] [CrossRef]
- Suárez-Iglesias, O.; Collado, S.; Oulego, P.; Díaz, M. Graphene-Family Nanomaterials in Wastewater Treatment Plants. Chem. Eng. J. 2017, 313, 121–135. [Google Scholar] [CrossRef]
- Xu, L.; Wang, J. The application of graphene-based materials for the removal of heavy metals and radionuclides from water and wastewater. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1042–1105. [Google Scholar] [CrossRef]
- Hou, W.; Yuan, X.; Yan, W.; Huang, H.; Zeng, G.; Yan, L.; Wang, X.; Lin, N.; Yu, Q. Adsorption characteristics and behaviors of graphene oxide for Zn(II) removal from aqueous solution. Appl. Surf. Sci. 2013, 279, 432–440. [Google Scholar] [CrossRef]
- Zhao, G.; Li, J.; Ren, X.; Chen, C.; Wang, X. Few-layered graphene oxide nanosheets as superior sorbents for heavy metal ion pollution management. Environ. Sci. Technol. 2011, 45, 10454–10462. [Google Scholar] [CrossRef]
- Arshad, F.; Selvaraj, M.; Zain, J.; Banat, F.; Haija, M.A. Polyethylenimine modified graphene oxide hydrogel composite as an efficient adsorbent for heavy metal ions. Separat. Purif. Technol. 2018, 209, 870–880. [Google Scholar] [CrossRef]
- Vilela, D.; Parmar, J.; Zeng, Y.; Zhao, Y.; Sánchez, S. Graphene-Based Microbots for Toxic Heavy Metal Removal and Recovery from Water. Nano Lett. 2016, 16, 2860–2866. [Google Scholar] [CrossRef]
- Fan, Z.; Bo, W.; He, S.; Man, R. Preparation of Graphene-Oxide/Polyamidoamine Dendrimers and Their Adsorption Properties toward Some Heavy Metal Ions. J. Chem. Eng. Data 2014, 59, 1719–1726. [Google Scholar]
- Li, X.; Zhou, H.; Wu, W.; Wei, S.; Xu, Y.; Kuang, Y. Studies of heavy metal ion adsorption on Chitosan/Sulfydryl-functionalized graphene oxide composites. J. Colloid Interface Sci. 2015, 448, 389–397. [Google Scholar] [CrossRef]
- Yilong, W.; Song, L.; Bingdi, C.; Fangfang, G.; Shuili, Y.; Yulin, T. Synergistic removal of Pb(II), Cd(II) and humic acid by Fe3O4@mesoporous silica-graphene oxide composites. PLoS ONE 2013, 8, e65634. [Google Scholar]
- Musico, Y.L.F.; Santos, C.M.; Dalida, M.L.P.; Rodrigues, D.F. Improved removal of lead(II) from water using a polymer-based graphene oxide nanocomposite. J. Mater. Chem. A 2013, 1, 3789–3796. [Google Scholar] [CrossRef]
- Kumar, S.; Nair, R.R.; Pillai, P.B.; Gupta, S.N.; Iyengar, M.A.; Sood, A.K. Graphene oxide-MnFe2O4 magnetic nanohybrids for efficient removal of lead and arsenic from water. ACS Appl. Mater. Interfaces 2014, 6, 17426. [Google Scholar] [CrossRef]
- Cui, L.; Wang, Y.; Liang, G.; Hu, L.; Yan, L.; Qin, W.; Du, B. EDTA functionalized magnetic graphene oxide for removal of Pb(II), Hg(II) and Cu(II) in water treatment: Adsorption mechanism and separation property. Chem. Eng. J. 2015, 281, 1–10. [Google Scholar] [CrossRef]
- Tan, M.; Liu, X.; Li, W.; Li, H. Enhancing Sorption Capacities for Copper(II) and Lead(II) under Weakly Acidic Conditions by l-Tryptophan-Functionalized Graphene Oxide. J. Chem. Eng. Data 2015, 60, 1469–1475. [Google Scholar] [CrossRef]
- Hai, T.X.; Jian, H.C.; Xue, S.; Yi, H.H.; Zhen, B.S.; Shi, R.H.; Wen, W.; Li, S.X.; Hong, X.G.; Wen, B.W. NH2-rich polymer/graphene oxide use as a novel adsorbent for removal of Cu(II) from aqueous solution. Chem. Eng. J. 2015, 263, 280–289. [Google Scholar]
- Chauke, V.P.; Maity, A.; Chetty, A. High-performance towards removal of toxic hexavalent chromium from aqueous solution using graphene oxide-alpha cyclodextrin-polypyrrole nanocomposites. J. Mol. Liquids 2015, 211, 71–77. [Google Scholar] [CrossRef]
- Zhang, K.; Li, H.; Xu, X.; Yu, H. Synthesis of reduced graphene oxide/NiO nanocomposites for the removal of Cr(VI) from aqueous water by adsorption. Microporous Mesoporous Mater. 2018, 255, 7–14. [Google Scholar] [CrossRef]
- Zhao, D.; Chen, L.L.; Xu, M.; Feng, S.; Chen, C. Amino Siloxane Oligomer Modified Graphene Oxide Composite for the Efficient Capture of U(VI) and Eu(III) from Aqueous Solution. ACS Sustain. Chem. 2017, 5, 10290–10297. [Google Scholar] [CrossRef]
- Najafabadi, H.H.; Irani, M.; Rad, L.R.; Haratameh, A.H.; Haririan, I. Removal of Cu2+, Pb2+ and Cr6+ from aqueous solutions using a chitosan/graphene oxide composite nanofibrous adsorbent. RSC Adv. 2015, 5, 22390. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Fekry, N.A.; El-Latif, M.M.A. Nanocomposites of nanosilica-immobilized-nanopolyaniline and crosslinked nanopolyaniline for removal of heavy metals. Chem. Eng. J. 2016, 304, 679–691. [Google Scholar] [CrossRef]
- Kotsyuda, S.S.; Tomina, V.V.; Zub, Y.L.; Furtat, I.M.; Melnyk, I.V. Bifunctional silica nanospheres with 3-aminopropyl and phenyl groups. Synthesis approach and prospects of their applications. Appl. Surf. Sci. 2017, 420, 782–791. [Google Scholar] [CrossRef]
- Najafi, M.; Yousefi, Y.; Rafati, A.A. Synthesis, characterization and adsorption studies of several heavy metal ions on amino-functionalized silica nano hollow sphere and silica gel. Separat. Purif. Technol. 2012, 85, 193–205. [Google Scholar] [CrossRef]
- Pogorilyi, R.P.; Melnyk, I.A.; Zub, Y.L.; Carlson, S.; Daniel, G.; Svedlindh, P.; Seisenbaeva, G.A.; Kessler, V.G. New product from old reaction: Uniform magnetite nanoparticles from iron-mediated synthesis of alkali iodides and their protection from leaching in acidic media. RSC Adv. 2014, 4, 22606–22612. [Google Scholar] [CrossRef]
- Srinivasan, N.R.; Shankar, P.A.; Bandyopadhyaya, R. Plasma treated activated carbon impregnated with silver nanoparticles for improved antibacterial effect in water disinfection. Carbon 2013, 57, 1–10. [Google Scholar] [CrossRef]
- Bokare, V.; Jung, J.L.; Chang, Y.Y.; Chang, Y.S. Reductive dechlorination of octachlorodibenzo-p-dioxin by nanosized zero-valent zinc: Modeling of rate kinetics and congener profile. J. Hazard. Mater. 2013, 250–251, 397–402. [Google Scholar] [CrossRef]
- O’Carroll, D.; Sleep, B.; Krol, M.; Boparai, H.; Kocur, C. Nanoscale zero valent iron and bimetallic particles for contaminated site remediation. Adv. Water Resour. 2013, 51, 104–122. [Google Scholar] [CrossRef]
- Liu, T.; Wang, Z.L.; Sun, Y. Manipulating the morphology of nanoscale zero-valent iron on pumice for removal of heavy metals from wastewater. Chem. Eng. J. 2015, 263, 55–61. [Google Scholar] [CrossRef]
- Zhen, Z.; Wei, H.Z.; Li, L.W.; Hua, X.X. Synchronous Treatment of Heavy Metal Ions and Nitrate by Zero-valent Iron. Huan Jing Ke Xue 2009, 30, 775–779. [Google Scholar]
- Seyedi, S.M.; Rabiee, H.; Shahabadi, S.M.S.; Borghei, S.M. Synthesis of Zero-valent Iron Nanoparticles via Electrical Wire Explosion for Efficient Removal of Heavy Metals: Water. Clean—Soil Air Water 2016, 45, 1600139. [Google Scholar] [CrossRef]
- Cundy, A.B.; Hopkinson, L.; Whitby, R.L. Use of iron-based technologies in contaminated land and groundwater remediation: A review. Sci. Total Environ. 2008, 400, 42–51. [Google Scholar] [CrossRef]
- Huang, P.; Ye, Z.; Xie, W.; Chen, Q.; Li, J.; Xu, Z.; Yao, M. Rapid magnetic removal of aqueous heavy metals and their relevant mechanisms using nanoscale zero valent iron (nZVI) particles. Water Res. 2013, 47, 4050–4058. [Google Scholar] [CrossRef]
- Wei, W.; Hua, Y.; Li, S.; Yan, W.; Zhang, W.X. Removal of Pb(II) and Zn(II) using lime and nanoscale zero-valent iron (nZVI): A comparative study. Chem. Eng. J. 2016, 304, 79–88. [Google Scholar]
- Li, X.Q.; Zhang, W.X. Sequestration of Metal Cations with Zerovalent Iron Nanoparticles—A Study with High Resolution X-ray Photoelectron Spectroscopy (HR-XPS). J. Phys. Chem. C 2007, 111, 6939–6946. [Google Scholar] [CrossRef]
- Tratnyek, P.G.; Sarathy, V.; Nurmi, J.; Baer, D.R.; Amonette, J.E.; Chan, L.C.; Penn, R.L.; Reardon, E.J. Aging of Iron Nanoparticles in Water: Effects on Structure and Reactivity. J. Phys. Chem. C 2008, 112, 2286–2293. [Google Scholar]
- Fu, F.; Dionysiou, D.D.; Hong, L. The use of zero-valent iron for groundwater remediation and wastewater treatment: A review. J. Hazard. Mater. 2014, 267, 194–205. [Google Scholar] [CrossRef]
- Huang, D.L.; Chen, G.M.; Zeng, G.M.; Xu, P.; Yan, M.; Lai, C.; Zhang, C.; Li, N.J.; Cheng, M.; He, X.X. Synthesis and Application of Modified Zero-Valent Iron Nanoparticles for Removal of Hexavalent Chromium from Wastewater. Water Air Soil Pollut. 2015, 226, 375. [Google Scholar] [CrossRef]
- Su, Y.; Adeleye, A.S.; Huang, Y.; Sun, X.; Dai, C.; Zhou, X.; Zhang, Y.; Keller, A.A. Simultaneous removal of cadmium and nitrate in aqueous media by nanoscale zerovalent iron (nZVI) and Au doped nZVI particles. Water Res. 2014, 63, 102–111. [Google Scholar] [CrossRef]
- Zarime, N.A.; Wan, Z.W.Y.; Jamil, H. Removal of heavy metals using bentonite supported nano-zero valent iron particles. In Proceedings of the Ukm Fst Postgraduate Colloquium: University Kebangsaan Malaysia, Faculty of Science & Technology Postgraduate Colloquium, Univ Kebangsaan Malaysia, Fac Sci & Technol, Selangor, Malaysia, 12–13 July 2017. [Google Scholar]
- Zhang, W. Nanoscale Iron Particles for Environmental Remediation: An Overview. J. Nanopart. Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- Morris, T.; Copeland, H.; Mclinden, E.; Wilson, S.; Szulczewski, G. The Effects of Mercury Adsorption on the Optical Response of Size-Selected Gold and Silver Nanoparticles. Langmuir 2002, 18, 7261–7264. [Google Scholar] [CrossRef]
- Fan, Y.; Liu, Z.; Wang, L.E.; Zhan, J. Synthesis of Starch-Stabilized Ag Nanoparticles and Hg2+Recognition in Aqueous Media. Nano Scale Res. Lett. 2009, 4, 1230–1235. [Google Scholar] [CrossRef]
- Pradhan, N.; Pal, A.; Pal, T. Silver nanoparticle catalyzed reduction of aromatic nitro compounds. Colloids Surf. A 2002, 196, 247–257. [Google Scholar] [CrossRef]
- Sumesh, E.; Bootharaju, M.S.; Anshup; Pradeep, T. A practical silver nanoparticle-based adsorbent for the removal of Hg2+ from water. J. Hazard. Mater. 2011, 189, 450–457. [Google Scholar] [CrossRef]
- Zhang, Y.; Bing, W.; Hui, X.; Hui, L.; Wang, M.; He, Y.; Pan, B. Nanomaterials-enabled water and wastewater treatment. Nanoimpact 2016, 3–4, 22–39. [Google Scholar] [CrossRef]
- Lisha, K.P.; Anshup; Pradeep, T. Towards a practical solution for removing inorganic mercury from drinking water using gold nanoparticles. Gold Bull. 2009, 42, 144–152. [Google Scholar] [CrossRef]
- Ojea-Jiménez, I.; López, X.; Arbiol, J.; Puntes, V. Citrate-coated gold nanoparticles as smart scavengers for mercury(II) removal from polluted waters. ACS Nano 2012, 6, 2253. [Google Scholar] [CrossRef]
- Sneed; Cannon, M. Comprehensive inorganic chemistry. Nature 1958, 181, 1428. [Google Scholar] [CrossRef]
- Saharan, P.; Chaudhary, G.R.; Mehta, S.K.; Umar, A. Removal of water contaminants by iron oxide nanomaterials. J. Nanosci. Nanotechnol. 2014, 14, 627–643. [Google Scholar] [CrossRef]
- Birch, F. Density and Composition of Mantle and Core. J. Geophys. Res. 1964, 69, 4377–4388. [Google Scholar] [CrossRef]
- Massalimov, I.A.; Il’Yasova, R.R.; Musavirova, L.R.; Samsonov, M.R.; Mustafin, A.G. Use of micrometer hematite particles and nanodispersed goethite as sorbent for heavy metals. Russ. J. Appl. Chem. 2014, 87, 1456–1463. [Google Scholar] [CrossRef]
- Sun, Y.B.; Wang, Q.; Yang, S.T.; Sheng, G.D.; Guo, Z.Q. Characterization of nano-iron oxyhydroxides and their application in UO22+ removal from aqueous solutions. J. Radioanal. Nuclear Chem. 2011, 290, 643–648. [Google Scholar] [CrossRef]
- Chen, Y.H.; Li, F.A. Kinetic study on removal of copper(II) using goethite and hematite nano-photocatalysts. J. Colloid Interface Sci. 2010, 347, 277–281. [Google Scholar] [CrossRef]
- Khezami, L.; M’Hamed, M.O.; Lemine, O.M.; Bououdina, M.; Bessadokjemai, A. Milled goethite nanocrystalline for selective and fast uptake of cadmium ions from aqueous solution. Desalinat. Water Treat. 2016, 57, 1–9. [Google Scholar] [CrossRef]
- Leiviskä, T.; Khalid, M.K.; Sarpola, A.; Tanskanen, J. Removal of vanadium from industrial wastewater using iron sorbents in batch and continuous flow pilot systems. J. Environ. Manag. 2017, 190, 231–242. [Google Scholar] [CrossRef]
- Forbes, E.A.; Posner, A.M.; Quirk, J.P. The specific adsorption of divalent Cd, Co, Cu, Pb, AND Zn on goethite. Eur. J. Soil Sci. 2010, 27, 154–166. [Google Scholar] [CrossRef]
- Li, Y.; Fan, Q.; Wu, W. Sorption of Th(IV) on goethite: Effects of pH, ionic strength, FA and phosphate. J. Radioanal. Nuclear Chem. 2011, 289, 865–871. [Google Scholar]
- Tadic, M.; Panjan, M.; Damnjanovic, V.; Milosevic, I. Magnetic properties of hematite (α-Fe2O3) nanoparticles prepared by hydrothermal synthesis method. Appl. Surf. Sci. 2014, 320, 183–187. [Google Scholar] [CrossRef]
- Chen, L.; Wang, G.; Mathur, G.N.; Varadan, V.K. Size and Shape Dependence of the Electrochemical Properties of Hematite Nanoparticles and Their Applications in Lithium Ion Batteries. Proc. SPIE 2012, 38. [Google Scholar] [CrossRef]
- Guo, H.; Barnard, A.S. Can Hematite Nanoparticles be an Environmental Indicator? Energy Environ. Sci. 2013, 6, 561–569. [Google Scholar] [CrossRef]
- Cuong, N.D.; Hoa, N.D.; Hoa, T.T.; Khieu, D.Q.; Quang, D.T.; Quang, V.V.; Hieu, N.V. Nanoporous hematite nanoparticles: Synthesis and applications for benzylation of benzene and aromatic compounds. J. Alloys Compd. 2014, 582, 83–87. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Ali, S.M.; El-Dek, S.I.; Galal, A. Magnetite–hematite nanoparticles prepared by green methods for heavy metal ions removal from water. Mater. Sci. Eng. B 2013, 178, 744–751. [Google Scholar] [CrossRef]
- Adegoke, H.I.; Amooadekola, F.; Fatoki, O.S.; Ximba, B.J. Adsorption of Cr(VI) on synthetic hematite (α-Fe2O3) nanoparticles of different morphologies. Korean J. Chem. Eng. 2014, 31, 142–154. [Google Scholar] [CrossRef]
- Dickson, D.; Liu, G.; Cai, Y. Adsorption kinetics and isotherms of arsenite and arsenate on hematite nanoparticles and aggregates. J. Environ. Manag. 2017, 186, 261–267. [Google Scholar] [CrossRef]
- Shipley; Heather, J.; Engates; Karen, E.; Grover; Valerie, A. Removal of Pb(II), Cd(II), Cu(II), and Zn(II) by hematite nanoparticles: Effect of sorbent concentration, pH, temperature, and exhaustion. Environ. Sci. Pollut. Res. Int. 2013, 20, 1727–1736. [Google Scholar] [CrossRef]
- Kefeni, K.K.; Msagati, T.A.M.; Nkambule, T.T.I.; Mamba, B.B. Synthesis and application of hematite nanoparticles for acid mine drainage treatment. J. Environ. Chem. Eng. 2018, 6, 1865–1874. [Google Scholar] [CrossRef]
- Akhtarkhavari, A. Removal, preconcentration and determination of Mo(VI) from water and wastewater samples using maghemite nanoparticles. Colloids Surf. A 2009, 346, 52–57. [Google Scholar]
- Jing, H.; Guohua, C.; Lo, I.M.C. Removal and recovery of Cr(VI) from wastewater by maghemite nanoparticles. Water Res. 2005, 39, 4528–4536. [Google Scholar]
- Etale, A.; Tutu, H.; Drake, D.C. The effect of silica and maghemite nanoparticles on remediation of Cu(II)-, Mn(II)- and U(VI)-contaminated water by Acutodesmus sp. J. Appl. Phycol. 2016, 28, 251–260. [Google Scholar] [CrossRef]
- Tuutijärvi, T.; Lu, J.; Sillanpää, M.; Chen, G. As(V) adsorption on maghemite nanoparticles. J. Hazard. Mater. 2009, 166, 1415–1420. [Google Scholar] [CrossRef]
- Akhbarizadeh, R.; Shayestefar, M.R.; Darezereshki, E. Competitive Removal of Metals from Wastewater by Maghemite Nanoparticles: A Comparison Between Simulated Wastewater and AMD. Mine Water Environ. 2014, 33, 89–96. [Google Scholar] [CrossRef]
- Rajput, S.; Singh, L.P.; Jr, C.U.P.; Mohan, D. Lead (Pb2+) and copper (Cu2+) remediation from water using Superparamagnetic maghemite (γ-Fe2O3) nanoparticles synthesized by Flame Spray Pyrolysis (FSP). J. Colloid Interface Sci. 2017, 492, 176–190. [Google Scholar] [CrossRef]
- Madrakian, T.; Afkhami, A.; Zadpour, B.; Ahmadi, M. New synthetic mercaptoethylamino homopolymer-modified maghemite nanoparticles for effective removal of some heavy metal ions from aqueous solution. J. Ind. Eng. Chem. 2015, 21, 1160–1166. [Google Scholar] [CrossRef]
- Takafuji, M.; Ide, S.; Ihara, H.; Xu, Z.H. Preparation of poly(1-vinylimidazole)-grafted magnetic nanoparticles and their application for removal of metal ions. Chem. Mater. 2004, 16, 1977–1983. [Google Scholar] [CrossRef]
- Song, J.; Kong, H.; Jang, J. Adsorption of heavy metal ions from aqueous solution by polyrhodanine-encapsulated magnetic nanoparticles. J. Colloid Interface Sci. 2011, 359, 505–511. [Google Scholar] [CrossRef]
- Chávez-Guajardo, A.E.; Medina-Llamas, J.C.; Maqueira, L.; Andrade, C.A.S.; Alves, K.G.B.; Melo, C.P.D. Efficient removal of Cr(VI) and Cu(II) ions from aqueous media by use of polypyrrole/maghemite and polyaniline/maghemite magnetic nanocomposites. Chem. Eng. J. 2015, 281, 826–836. [Google Scholar] [CrossRef]
- Hao, Y.M.; Man, C.; Hu, Z.B. Effective removal of Cu(II) ions from aqueous solution by amino-functionalized magnetic nanoparticles. J. Hazard. Mater. 2010, 184, 392–399. [Google Scholar] [CrossRef]
- Watts, M.P.; Coker, V.S.; Parry, S.A.; Pattrick, R.A.D.; Thomas, R.A.P.; Kalin, R.; Lloyd, J.R. Biogenic nano-magnetite and nano-zero valent iron treatment of alkaline Cr(VI) leachate and chromite ore processing residue. Appl. Geochem. 2015, 54, 27–42. [Google Scholar] [CrossRef]
- Shan, C.; Ma, Z.; Tong, M.; Ni, J. Removal of Hg(II) by poly(1-vinylimidazole)-grafted Fe3O4 @SiO2 magnetic nanoparticles. Water Res. 2015, 69, 252–260. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Abdelwahab, M.S.; Abdou, A.E.H. Enhanced removal of lead and cadmium from water by Fe3O4-cross linked-O-phenylenediamine nano-composite. Sep. Sci. Technol. 2016, 51, 237–247. [Google Scholar] [CrossRef]
- Giraldo, L.; Erto, A.; Moreno-Piraján, J.C. Magnetite nanoparticles for removal of heavy metals from aqueous solutions: Synthesis and characterization. Adsorption 2013, 19, 465–474. [Google Scholar] [CrossRef]
- Hu, H.; Wang, Z.; Pan, L. Synthesis of monodisperse Fe3O4 @silica core–shell microspheres and their application for removal of heavy metal ions from water. J. Alloys Compod. 2010, 492, 656–661. [Google Scholar] [CrossRef]
- Tan, Y.Q.; Chen, M.; Hao, Y.M. High efficient removal of Pb(II) by amino-functionalized Fe3O4 magnetic nano-particles. Chem. Eng. J. 2012, 191, 104–111. [Google Scholar] [CrossRef]
- Shi, J.; Li, H.Y.; Lu, H.G.; Zhao, X.W. Use of Carboxyl Functional Magnetite Nanoparticles as Potential Sorbents for the Removal of Heavy Metal Ions from Aqueous Solution. J. Chem. Eng. Data 2015, 60, 2035–2041. [Google Scholar] [CrossRef]
- Pan, S.D.; Shen, H.Y.; Xu, Q.H.; Luo, J.; Hu, M.Q. Surface mercapto engineered magnetic Fe3O4 nanoadsorbent for the removal of mercury from aqueous solutions. J. Colloid Interface Sci. 2012, 365, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Christian, S.; Robin, E.; Robert, Z.; Artur, F.; Hauke, K.; Christopher, W.; Johannes, O.; Jan-Philip, M.; Theo, S.; Kornelius, N. Polymer-assisted self-assembly of superparamagnetic iron oxide nanoparticles into well-defined clusters: Controlling the collective magnetic properties. Langmuir ACS J. Surf. Colloids 2014, 30, 11190–11196. [Google Scholar]
- Baghani, A.N.; Mahvi, A.H.; Gholami, M.; Rastkari, N.; Delikhoon, M. One-Pot synthesis, characterization and adsorption studies of amine-functionalized magnetite nanoparticles for removal of Cr(VI) and Ni(II) ions from aqueous solution: Kinetic, isotherm and thermodynamic studies. J. Environ. Health Sci. 2016, 14, 11. [Google Scholar] [CrossRef] [PubMed]
- Madrakian, T.; Afkhami, A.; Zolfigol, M.A.; Ahmadi, M.; Koukabi, N. Application of Modified Silica Coated Magnetite Nanoparticles for Removal of Iodine from Water Samples. Nano-Micro Lett. 2012, 4, 57–63. [Google Scholar] [CrossRef]
- Adeli, M.; Yamini, Y.; Faraji, M. Removal of copper, nickel and zinc by sodium dodecyl sulphate coated magnetite nanoparticles from water and wastewater samples. Arab. J. Chem. 2017, 10, S514–S521. [Google Scholar] [CrossRef]
- Magnet, C.; Lomenech, C.; Hurel, C.; Reilhac, P.; Giulieri, F.; Chaze, A.M.; Persello, J.; Kuzhir, P. Adsorption of nickel ions by oleate-modified magnetic iron oxide nanoparticles. Environ. Sci. Pollut. R 2017, 24, 7423–7435. [Google Scholar] [CrossRef] [PubMed]
- Madrakian, T.; Afkhami, A.; Rezvani-jalal, N.; Ahmadi, M. Removal and preconcentration of lead(II), cadmium(II) and chromium(III) ions from wastewater samples using surface functionalized magnetite nanoparticles. J. Iran Chem. Soc. 2014, 11, 489–498. [Google Scholar] [CrossRef]
- Behbahani, N.S.; Rostamizadeh, K.; Yaftian, M.R.; Zamani, A.; Ahmadi, H. Covalently modified magnetite nanoparticles with PEG: Preparation and characterization as nano-adsorbent for removal of lead from wastewater. J. Environ. Health Sci. 2014, 12, 103. [Google Scholar] [CrossRef] [PubMed]
- Rahbar, N.; Jahangiri, A.; Boumi, S.; Khodayar, M.J. Mercury Removal from Aqueous Solutions with Chitosan-Coated Magnetite Nanoparticles Optimized Using the Box-Behnken Design. Jundishapur J. Nat. Pharm. Prod. 2014, 9, e15913. [Google Scholar] [CrossRef] [PubMed]
- Bagtash, M.; Yamini, Y.; Tahmasebi, E.; Zolgharnein, J.; Dalirnasab, Z. Magnetite nanoparticles coated with tannic acid as a viable sorbent for solid-phase extraction of Cd2+, Co2+ and Cr3+. Microchim. Acta 2016, 183, 449–456. [Google Scholar] [CrossRef]
- Huang, X.; Yang, J.; Wang, J.; Bi, J.; Xie, C.; Hao, H. Design and synthesis of core–shell Fe3O4 @PTMT composite magnetic microspheres for adsorption of heavy metals from high salinity wastewater. Chemosphere 2018, 206, 513. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Boonfueng, T.; Xu, Y.; Axe, L.; Tyson, T.A. Modeling Pb sorption to microporous amorphous oxides as discrete particles and coatings. J. Colloid Interface Sci. 2005, 281, 39–48. [Google Scholar] [CrossRef]
- Hui, Q.; Shujuan, Z.; Bingcai, P.; Weiming, Z.; Lu, L. Effect of sulfate on Cu(II) sorption to polymer-supported nano-iron oxides: Behavior and XPS study. J. Colloid Interface Sci. 2012, 366, 37–43. [Google Scholar]
- Singh, S.K.; Subramanian, V.; Gibbs, R.J. Hydrous FE and MN oxides—Scavengers of heavy metals in the aquatic environment. Crit. Rev. Environ. Control 1984, 14, 33–90. [Google Scholar] [CrossRef]
- Hui, Q.; Zhang, S.; Pan, B.; Zhang, W.; Lu, L. Oxalate-promoted dissolution of hydrous ferric oxide immobilized within nanoporous polymers: Effect of ionic strength and visible light irradiation. Chem. Eng. J. 2013, 232, 167–173. [Google Scholar]
- Zhang, Y.; Li, Z. Heavy metals removal using hydrogel-supported nanosized hydrous ferric oxide: Synthesis, characterization, and mechanism. Sci. Total Environ. 2017, 580, 776–786. [Google Scholar] [CrossRef] [PubMed]
- Byungryul, A.; Steinwinder, T.R.; Dongye, Z. Selective removal of arsenate from drinking water using a polymeric ligand exchanger. Water Res. 2005, 39, 4993–5004. [Google Scholar]
- Huo, L.; Zeng, X.; Su, S.; Bai, L.; Wang, Y. Enhanced removal of As(V) from aqueous solution using modified hydrous ferric oxide nanoparticles. Sci. Rep. 2017, 7, 40765. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, R.A.; Chen, K.Y. Adsorption of mercury(II) by hydrous manganese oxides. Environ. Sci. Technol. 1973, 7, 1028–1034. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, J.; Ramkumar, J.; Shukla, R.; Tyagi, A.K. Sorption characteristics of nano manganese oxide: Efficient sorbent for removal of metal ions from aqueous streams. J. Radioanal. Nuclear Chem. 2013, 297, 49–57. [Google Scholar] [CrossRef]
- Wang, X.; Huang, K.; Chen, Y.; Liu, J.; Chen, S.; Cao, J.; Mei, S.; Zhou, Y.; Jing, T. Preparation of dumbbell manganese dioxide/gelatin composites and their application in the removal of lead and cadmium ions. J. Hazard. Mater. 2018, 350, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Huangfu, X.; Jin, J.; Lu, X.; Wang, Y.; Liu, Y.; Pang, S.Y.; Cheng, H.; Xiang, Z.; Ma, J. Adsorption and Oxidation of Thallium(I) by a Nanosized Manganese Dioxide. Water Air Soil Pollut. 2015, 226, 2272. [Google Scholar] [CrossRef]
- Abdullah, J.A.; Lafi, A.G.A.; Amin, Y.; Alnama, T. A Styrofoam-nano manganese oxide based composite: Preparation and application for the treatment of wastewater. Appl. Radiat. Isotopes 2018, 136, 73. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.J.; Lee, C.S.; Chang, Y.Y.; Chang, Y.S. Hierarchically structured manganese oxide-coated magnetic nanocomposites for the efficient removal of heavy metal ions from aqueous systems. Appl. Mater. Interfaces 2013, 5, 9628–9634. [Google Scholar] [CrossRef] [PubMed]
- Lisha, K.P.; Maliyekkal, S.M.; Pradeep, T. Manganese dioxide nanowhiskers: A potential adsorbent for the removal of Hg(II) from water. Chem. Eng. J. 2010, 160, 432–439. [Google Scholar] [CrossRef]
- Sposito, G. On the surface complexation model of the oxide-aqueous solution interface. J. Colloid Interface Sci. 1983, 91, 329–340. [Google Scholar] [CrossRef]
- Wan, S.; Wu, J.; Zhou, S.; Rui, W.; Gao, B.; Feng, H. Enhanced lead and cadmium removal using biochar-supported hydrated manganese oxide (HMO) nanoparticles: Behavior and mechanism. Sci. Total Environ. 2018, 616–617, 1298–1306. [Google Scholar] [CrossRef]
- Kumar, K.Y.; Muralidhara, H.B.; Nayaka, Y.A.; Balasubramanyam, J.; Hanumanthappa, H. Low-cost synthesis of metal oxide nanoparticles and their application in adsorption of commercial dye and heavy metal ion in aqueous solution. Powder Technol. 2013, 246, 125–136. [Google Scholar] [CrossRef]
- Rafiq, Z.; Nazir, R.; Durr-e-Shahwar; Shah, M.R.; Ali, S. Utilization of magnesium and zinc oxide nano-adsorbents as potential materials for treatment of copper electroplating industry wastewater. J. Environ. Chem. Eng. 2014, 2, 642–651. [Google Scholar] [CrossRef]
- Hadadian, M.; Goharshadi, E.K.; Fard, M.M.; Ahmadzadeh, H. Synergistic effect of graphene nanosheets and zinc oxide nanoparticles for effective adsorption of Ni(II) ions from aqueous solutions. Appl. Phys. A 2018, 124, 239. [Google Scholar] [CrossRef]
- Sheela, T.; Nayaka, Y.A.; Viswanatha, R.; Basavanna, S.; Venkatesha, T.G. Kinetics and thermodynamics studies on the adsorption of Zn(II), Cd(II) and Hg(II) from aqueous solution using zinc oxide nanoparticles. Powder Technol. 2012, 217, 163–170. [Google Scholar] [CrossRef]
- Ghiloufi, I.; Mir, L.E. Preparation and characterization of nanoporous resin for heavy metal removal from aqueous solution. J. Water Supply Res. Technol.-Aqua. 2015, 64, 316–325. [Google Scholar] [CrossRef]
- Somu, P.; Paul, S. Casein based biogenic-synthesized zinc oxide nanoparticles simultaneously decontaminate heavy metals, dyes, and pathogenic microbes: A rational strategy for wastewater treatment. J. Chem. Technol. Biotechnol. 2018. [Google Scholar] [CrossRef]
- Anandan, S.; Kathiravan, K.; Murugesan, V.; Ikuma, Y. Anionic IO3- non-metal doped TiO2 nanoparticles for the photocatalytic degradation of hazardous pollutant in water. Catal. Commun. 2009, 10, 1014–1019. [Google Scholar] [CrossRef]
- Skubal, L.R.; Meshkov, N.K.; Rajh, T.; Thurnauer, M. Cadmium removal from water using thiolactic acid-modified titanium dioxide nanoparticles. J. Photochem. Photobiol. A Chem. 2002, 148, 393–397. [Google Scholar] [CrossRef]
- Seisenbaeva, G.A.; Daniel, G.; Nedelec, J.M.; Gun'Ko, Y.K.; Kessler, V.G. High surface area ordered mesoporous nano-titania by a rapid surfactant-free approach. J. Mater. Chem. 2012, 22, 20374–20380. [Google Scholar] [CrossRef]
- Malhat, F.M.; Youssef, A. Selective removal of heavy metal from drinking water using titanium oxide nanowire. Macromol. Symposia 2014, 337, 96–101. [Google Scholar]
- Deedar, N.; Irfan, A.; Qazi, I.A. Evaluation of the adsorption potential of titanium dioxide nanoparticles for arsenic removal. J. Environ. Sci. 2009, 21, 402–408. [Google Scholar]
- Baysal, A.; Kuznek, C.; Ozcan, M. Starch coated titanium dioxide nanoparticles as a challenging sorbent to separate and preconcentrate some heavy metals using graphite furnace atomic absorption spectrometry. Int. J. Environ. Anal. Chem. 2018, 98. [Google Scholar] [CrossRef]
- Mahmoud, M.E.; Ali, S.A.A.A.; Elweshahy, S.M.T. Microwave functionalization of titanium oxide nanoparticles with chitosan nanolayer for instantaneous microwave sorption of Cu(II) and Cd(II) from water. Int. J. Biol. Macromol. 2018, 111, 393–399. [Google Scholar] [CrossRef]
- Du, P.; Cameiro, J.T.; Moulijn, J.A.; Mul, G. A novel photocatalytic monolith reactor for multiphase heterogeneous photocatalysis. Appl. Catal. A Gen. 2008, 334, 119–128. [Google Scholar] [CrossRef]
- Giles, D.E.; Mohapatra, M.; Issa, T.B.; Anand, S.; Singh, P. Iron and aluminium based adsorption strategies for removing arsenic from water. J. Environ. Manag. 2011, 92, 3011–3022. [Google Scholar] [CrossRef]
- Prabhakar, R.; Samadder, S.R. Low cost and easy synthesis of aluminium oxide nanoparticles for arsenite removal from groundwater: A complete batch study. J. Mol. Liquids 2018, 250, 192–201. [Google Scholar] [CrossRef]
- Xie, Y.; Kocaefe, D.; Kocaefe, Y.; Cheng, J.; Liu, W. The Effect of Novel Synthetic Methods and Parameters Control on Morphology of Nano-alumina Particles. Nanoscale Res. Lett. 2016, 11, 259. [Google Scholar] [CrossRef]
- Saadi, Z.; Saadi, R.; Fazaeli, R. Fixed-bed adsorption dynamics of Pb(II) adsorption from aqueous solution using nanostructured γ-alumina. J. Nanostruct. Chem. 2013, 3, 1–8. [Google Scholar] [CrossRef]
- Tabesh, S.; Davar, F.; Reza Loghman-Estarki, M. Preparation of γ-Al2O3 Nanoparticles Using Modified Sol-Gel Method and Its Use for the Adsorption of Lead and Cadmium Ions. J. Alloys Compd. 2018, 730, 441–449. [Google Scholar] [CrossRef]
- Stietiya, M.H.; Wang, J.J. Zinc and cadmium adsorption to aluminum oxide nanoparticles affected by naturally occurring ligands. J. Environ. Qual. 2014, 43, 498. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhan, C.; Kong, B.; Zhu, X.; Liu, J.; Xu, W.; Cai, W.; Wang, H. Self-curled coral-like γ-Al2O3 nanoplates for use as an adsorbent. J. Colloid Interface Sci. 2015, 453, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Patra, A.K.; Dutta, A.; Bhaumik, A. Self-assembled mesoporous γ-Al2O3 spherical nanoparticles and their efficiency for the removal of arsenic from water. J. Hazard. Mater. 2012, 201, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi, S.; Jalali, M.; Afkhami, A. Heavy metals removal from aqueous solutions by Al2O3 nanoparticles modified with natural and chemical modifiers. Clean Technol. Environ. Policy 2015, 17, 85–102. [Google Scholar] [CrossRef]
- Poursani, A.S.; Nilchi, A.; Hassani, A.H.; Shariat, M.; Nouri, J. A novel method for synthesis of nano-γ-Al2O3: Study of adsorption behavior of chromium, nickel, cadmium and lead ions. Int. J. Environ. Sci. Technol. 2015, 12, 2003–2014. [Google Scholar] [CrossRef]
- Cai, Y.; Li, C.; Dan, W.; Wei, W.; Tan, F.; Wang, X.; Wong, P.K.; Qiao, X. Highly active MgO nanoparticles for simultaneous bacterial inactivation and heavy metal removal from aqueous solution. Chem. Eng. J. 2016, 312, 158–166. [Google Scholar] [CrossRef]
- Stoimenov, P.K.; Klinger, R.L.; Marchin, G.L.; Klabunde, K.J. Metal oxide nanoparticles as bactericidal agents. Langmuir 2002, 18, 6679–6686. [Google Scholar] [CrossRef]
- Mahdavi, S.; Jalali, M.; Afkhami, A. Heavy metals removal from aqueous solutions using TiO2, MgO, and Al2O3 nanoparticles. Chem. Eng. Commun. 2013, 200, 448–470. [Google Scholar] [CrossRef]
- Madzokere, T.C.; Karthigeyan, A. Heavy Metal Ion Effluent Discharge Containment Using Magnesium Oxide (MgO) Nanoparticles. Mater. Today 2017, 4, 9–18. [Google Scholar] [CrossRef]
- Xiong, C.; Wang, W.; Tan, F.; Luo, F.; Chen, J.; Qiao, X. Investigation on the efficiency and mechanism of Cd(II) and Pb(II) removal from aqueous solutions using MgO nanoparticles. J. Hazard. Mater. 2015, 299, 664–674. [Google Scholar] [CrossRef]
- Jing, F.; Zou, L.; Wang, Y.; Li, B.; He, X.; Fan, Z.; Ren, Y.; Lv, Y.; Zhang, M.; Dan, C. Synthesis of high surface area, mesoporous MgO nanosheets with excellent adsorption capability for Ni(II) via a distillation treating. J. Colloid Interface Sci. 2015, 438, 259–267. [Google Scholar]
- Umar, A.; Kumar, R.; Akhtar, M.S.; Kumar, G.; Kim, S.H. Growth and properties of well-crystalline cerium oxide (CeO2) nanoflakes for environmental and sensor applications. J. Colloid Interface Sci. 2015, 454, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Anupriya, K.; Vivek, E.; Subramanian, B. Facile synthesis of ceria nanoparticles by precipitation route for UV blockers. J. Alloys Compd. 2014, 590, 406–410. [Google Scholar] [CrossRef]
- Recillas, S.; García, A.; González, E.; Casals, E.; Puntes, V.; Sánchez, A.; Font, X. Use of CeO2, TiO2 and Fe3O4 nanoparticles for the removal of lead from water Toxicity of nanoparticles and derived compounds. Desalination 2011, 277, 213–220. [Google Scholar] [CrossRef]
- Recillas, S.; Colón, J.; Casals, E.; González, E.; Puntes, V.; Sánchez, A.; Font, X. Chromium VI adsorption on cerium oxide nanoparticles and morphology changes during the process. J. Hazard. Mater. 2010, 184, 425–431. [Google Scholar] [CrossRef]
- Mishra, P.K.; Saxena, A.; Rawat, A.S.; Dixit, P.K.; Rai, P.K. Surfactant-Free One-Pot Synthesis of Low-Density Cerium Oxide Nanoparticles for Adsorptive Removal of Arsenic Species. Environ. Prog. Sustain. Energy 2018, 37, 221–231. [Google Scholar] [CrossRef]
- Ayawanna, J.; Teoh, W.T.; Niratisairak, S.; Sato, K. Gadolinia-modified ceria photocatalyst for removal of lead(II) ions from aqueous solutions. Mater. Sci. Semicond. Process. 2015, 40, 136–139. [Google Scholar] [CrossRef]
- Ayawanna, J.; Sato, K. Photoelectrodeposition Effect of Lanthanum oxide-Modified Ceria Particles on the Removal of Lead(II) Ions from Water. Catal. Today 2017, 321–322, 128–134. [Google Scholar] [CrossRef]
- Meepho, M.; Sirimongkol, W.; Ayawanna, J. Samaria-doped Ceria Nanopowders for Heavy Metal Removal from Aqueous Solution. Mater. Chem. Phys. 2018, 214, 56–65. [Google Scholar] [CrossRef]
- Jiang, C.; Xiao, D.A. Nanosized Zirconium Dioxide Particles as an Efficient Sorbent for Lead Removal in Waters. Adv. Mater. Res. 2014, 926–930, 166–169. [Google Scholar] [CrossRef]
- Seisenbaeva, G.A.; Geoffrey, D.; Kessler, V.G.; Jean-Marie, N. General facile approach to transition-metal oxides with highly uniform mesoporosity and their application as adsorbents for heavy-metal-ion sequestration. Chemistry 2015, 20, 10732–10736. [Google Scholar] [CrossRef] [PubMed]
- Yalçınkaya, Ö.; Kalfa, O.M.; Türker, A.R. Chelating agent free-solid phase extraction (CAF-SPE) of Co(II), Cu(II) and Cd(II) by new nano hybrid material (ZrO2/B2O3). J. Hazard. Mater. 2011, 195, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Jie, T.; Zhang, Z.; Nie, G.; Zhao, H.; Peng, Q.; Jiao, T. Unique and outstanding cadmium sequestration by polystyrene-supported nanosized zirconium hydroxides: A case study. RSC Adv. 2015, 5, 55445–55452. [Google Scholar] [CrossRef]
- Hua, M.; Jiang, Y.; Wu, B.; Pan, B.; Zhao, X.; Zhang, Q. Fabrication of a new hydrous Zr(IV) oxide-based nanocomposite for enhanced Pb(II) and Cd(II) removal from waters. ACS Appl. Mater. Interfaces 2013, 5, 12135. [Google Scholar] [CrossRef] [PubMed]
- Hayati, B.; Maleki, A.; Najafi, F.; Daraei, H.; Gharibi, F.; Mckay, G. Synthesis and characterization of PAMAM/CNT nanocomposite as a super-capacity adsorbent for heavy metal (Ni2+, Zn2+, As3+, Co2+) removal from wastewater. J. Mol. Liquids 2016, 224, 1032–1040. [Google Scholar] [CrossRef]
- Hotze, E.M.; Phenrat, T.; Lowry, G.V. Nanoparticle aggregation: Challenges to understanding transport and reactivity in the environment. J. Environ. Qual. 2010, 39, 1909–1924. [Google Scholar] [CrossRef] [PubMed]
- Tounsadi, H.; Khalidi, A.; Abdennouri, M.; Barka, N. Activated carbon from Diplotaxis Harra biomass: Optimization of preparation conditions and heavy metal removal. J. Taiwan Inst. Chem. Eng. 2016, 59, 348–358. [Google Scholar] [CrossRef]
- Parlayici, S.; Eskizeybek, V.; Avcı, A.; Pehlivan, E. Removal of chromium(VI) using activated carbon-supported-functionalized carbon nanotubes. J. Nanostruct. Chem. 2015, 5, 255–263. [Google Scholar] [CrossRef]
- Kang, A.J.; Baghdadi, M.; Pardakhti, A. Removal of cadmium and lead from aqueous solutions by magnetic acid-treated activated carbon nanocomposite. Desalinat. Water Treat. 2015, 3994, 1–17. [Google Scholar]
- Jayaweera, H.D.A.C.; Siriwardane, I.; Silva, K.M.N.D.; Silva, R.M.D. Synthesis of multifunctional activated carbon nanocomposite comprising biocompatible flake nano hydroxyapatite and natural turmeric extract for the removal of bacteria and lead ions from aqueous solution. Chem. Central J. 2018, 12, 18. [Google Scholar] [CrossRef]
- Fernando, M.S.; Silva, R.M.D.; Silva, K.M.N.D. Synthesis, characterization, and application of nano hydroxyapatite and nanocomposite of hydroxyapatite with granular activated carbon for the removal of Pb2+ from aqueous solutions. Appl. Surf. Sci. 2015, 351, 95–103. [Google Scholar] [CrossRef]
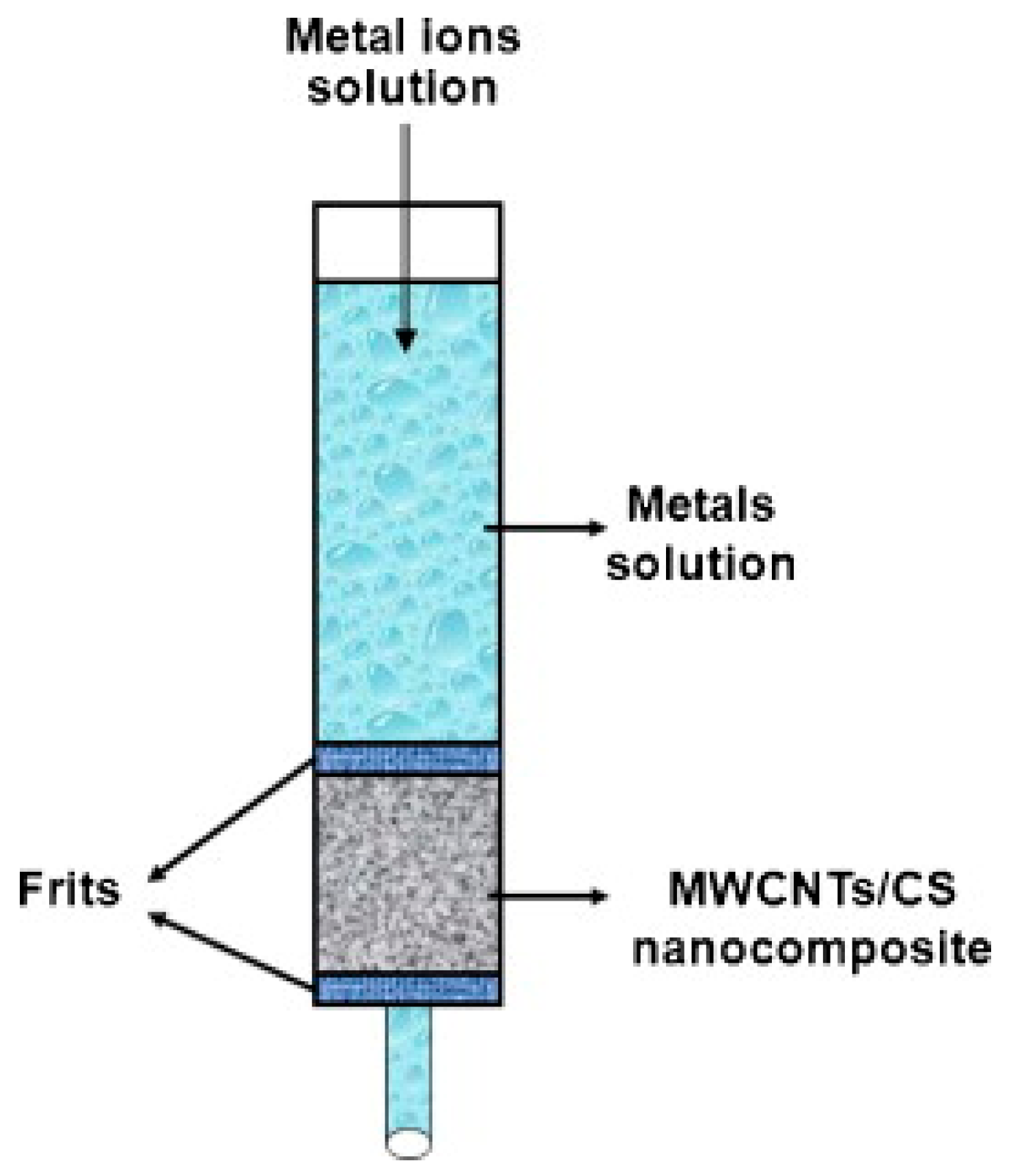
- Salam, M.A.; Makki, M.S.I.; Abdelaal, M.Y.A. Preparation and characterization of multi-walled carbon nanotubes/chitosan nanocomposite and its application for the removal of heavy metals from aqueous solution. J. Alloys Compd. 2011, 509, 2582–2587. [Google Scholar] [CrossRef]
- Xie, Y.; Qiang, H.; Liu, M.; Ke, W.; Wan, Q.; Deng, F.; Long, L.; Zhang, X.; Wei, Y. Mussel Inspired Functionalization of Carbon Nanotubes for Heavy Metal Ion Removal. RSC Adv. 2015, 5, 68430–68438. [Google Scholar] [CrossRef]
- Cheng, Z.; Sui, J.; Jing, L.; Tang, Y.; Wei, C. Efficient removal of heavy metal ions by thiol-functionalized superparamagnetic carbon nanotubes. Chem. Eng. J. 2012, 210, 45–52. [Google Scholar]
- Kosa, S.A.; Al-Zhrani, G.; Salam, M.A. Removal of heavy metals from aqueous solutions by multi-walled carbon nanotubes modified with 8-hydroxyquinoline. Chem. Eng. J. 2012, 181–182, 159–168. [Google Scholar] [CrossRef]
- Mamba, G.; Mbianda, X.Y.; Govender, P.P.; Mamba, B.B.; Krause, R.W. Application of multiwalled carbon nanotube-cyclodextrin polymers in the removal of heavy metals from water. J. Appl. Sci. 2010, 10. [Google Scholar] [CrossRef]
- Hayati, B.; Maleki, A.; Najafi, F.; Gharibi, F.; Mckay, G.; Gupta, V.K.; Marzban, N. Heavy metal adsorption using PAMAM/CNT nanocomposite from aqueous solution in batch and continuous fixed bed systems. Chem. Eng. J. 2018, 346, 258–270. [Google Scholar] [CrossRef]
- Donat, R.; Akdogan, A.; Erdem, E.; Cetisli, H. Thermodynamics of Pb2+ and Ni2+ adsorption onto natural bentonite from aqueous solutions. J. Colloid Interface Sci. 2005, 286, 43–52. [Google Scholar] [CrossRef]
- Eskandari, M.; Zakeri Khatir, M.; Khodadadi Darban, A.; Meshkini, M. Decreasing Ni, Cu, Cd, and Zn heavy metal magnetite-bentonite nanocomposites and adsorption isotherm study. Mater. Res. Express 2018, 5, 045030. [Google Scholar] [CrossRef]
- Ma, J.; Su, G.; Zhang, X.; Huang, W. Adsorption of Heavy Metal Ions from Aqueous Solutions by Bentonite Nanocomposites. Water Environ. Res. 2016, 88, 741–746. [Google Scholar] [CrossRef]
- Alswata, A.A.; Ahmad, M.B.; Al-Hada, N.M.; Kamari, H.M.; Hussein, M.Z.B.; Ibrahim, N.A. Preparation of Zeolite/Zinc Oxide Nanocomposites for toxic metals removal from water. Results Phys. 2017, 7, 723–731. [Google Scholar] [CrossRef]
- Zendehdel, M.; Shoshtari-Yeganeh, B.; Cruciani, G. Removal of heavy metals and bacteria from aqueous solution by novel hydroxyapatite/zeolite nanocomposite, preparation, and characterization. J. Iran. Chem. Soc. 2016, 13, 1915–1930. [Google Scholar] [CrossRef]
- Lee, S.M.; Kim, W.G.; Laldawngliana, C.; Tiwari, D. Removal Behavior of Surface Modified Sand for Cd(II) and Cr(VI) from Aqueous Solutions. J. Chem. Eng. Data 2010, 55, 3089–3094. [Google Scholar] [CrossRef]
- Kaşgöz, H.; Durmuş, A.; Kaşgöz, A. Enhanced swelling and adsorption properties of AAm-AMPSNa/clay hydrogel nanocomposites for heavy metal ion removal. Polym. Adv. Technol. 2010, 19, 213–220. [Google Scholar] [CrossRef]
- Zhao, G.; Huang, X.; Tang, Z.; Huang, Q.; Wang, X.K. Polymer-based nanocomposites for heavy metal ions removal from aqueous solution: A review. Polym. Chem. 2018, 9, 3562–3582. [Google Scholar] [CrossRef]
- Lu, F.; Astruc, D. Nanomaterials for removal of toxic elements from water. Coord. Chem. Rev. 2018, 356, 147–164. [Google Scholar] [CrossRef]
- Manju, G.N.; Krishnan, K.A.; Vinod, V.P.; Anirudhan, T.S. An investigation into the sorption of heavy metals from wastewaters by polyacrylamide-grafted iron(III) oxide. J. Hazard. Mater. 2002, 91, 221–238. [Google Scholar] [CrossRef]
- Rajakumar, K.; Dinesh, K.S.; Sivanesan, S.; Sai, R.L. Effective removal of heavy metal ions using Mn2O3 doped polyaniline nanocomposite. J. Nanosci. Nanotechnol. 2014, 14, 2937–2946. [Google Scholar] [CrossRef]
- Afshar, A.; Sadjadi, S.A.S.; Mollahosseini, A.; Eskandarian, M.R. Polypyrrole-polyaniline/Fe3O4 magnetic nanocomposite for the removal of Pb(II) from aqueous solution. Korean J. Chem. Eng. 2016, 33, 1–9. [Google Scholar] [CrossRef]
- Cai, J.; Lei, M.; He, J.R.Z.; Chen, T.; Liu, S.; Fu, S.H.; Li, T.T.; Liu, G.; Fei, P. Electrospun Composite Nanofiber Mats of Cellulose@Organically Modified Montmorillonite for Heavy Metal Ion Removal: Design, Characterization, Evaluation of Absorption Performance. Compos. Part A 2017, 92, 10–16. [Google Scholar] [CrossRef]
- Abhishek, K.; Meeta, G.; Jain, V.K. A novel reusable nanocomposite for complete removal of dyes, heavy metals and microbial load from water based on nanocellulose and silver nano-embedded pebbles. Environ. Technol. 2015, 36, 706–714. [Google Scholar]
- Saad, A.H.A.; Azzam, A.M.; El-Wakeel, S.T.; Mostafa, B.B.; El-Latif, M.B.A. Removal of toxic metal ions from wastewater using ZnO@Chitosan core-shell nanocomposite. Environ. Nanotechnol. Monitor. Manag. 2018, 9, 67–75. [Google Scholar] [CrossRef]
- Esmat, M.; Farghali, A.A.; Khedr, M.H.; El-Sherbiny, I.M. Alginate-Based Nanocomposites for Efficient Removal of Heavy Metal Ions. Int. J. Biol. Macromol. 2017, 102, 272–283. [Google Scholar] [CrossRef]
- Gokila, S.; Gomathi, T.; Sudha, P.N.; Anil, S. Removal of the heavy metal ion chromiuim(VI) using Chitosan and Alginate nanocomposites. Int. J. Biol. Macromol. 2017, 104, 1459–1468. [Google Scholar] [CrossRef]
- Lofrano, G.; Carotenuto, M.; Libralato, G.; Domingos, R.F.; Markus, A.; Dini, L.; Gautam, R.K.; Baldantoni, D.; Rossi, M.; Sharma, S.K. Polymer functionalized nanocomposites for metals removal from water and wastewater: An overview. Water Res. 2016, 92, 22–37. [Google Scholar] [CrossRef]
- Jingfu, L.; Zongshan, Z.; Guibin, J.; Cuiling, R. Coating Fe3O4 Magnetic Nanoparticles with Humic Acid for High Effi cient Removal of Heavy Metals in Water. Environ. Sci. Technol. 2008, 42, 6949–6954. [Google Scholar]
- Lü, T.; Qi, D.; Dong, Z.; Lü, Y.; Zhao, H. A facile method for emulsified oil-water separation by using polyethylenimine-coated magnetic nanoparticles. J. Nanopart. Res. 2018, 20, 88. [Google Scholar] [CrossRef]
- Mirrezaie, N.; Nikazar, M.; Hasan Zadeh, M. Synthesis of Magnetic Nanocomposite Fe3O4 Coated Polypyrrole (PPy) for Chromium(VI) Removal. Adv. Mater. Res. 2013, 829, 649–653. [Google Scholar] [CrossRef]
- Zhang, W.; Shi, X.; Zhang, Y.; Gu, W.; Li, B.; Xian, Y. Synthesis of water-soluble magnetic graphene nanocomposites for recyclable removal of heavy metal ions. J. Mater. Chem. A 2013, 1, 1745–1753. [Google Scholar] [CrossRef]
- Elmi, F.; Hosseini, T.; Taleshi, M.S.; Taleshi, F. Kinetic and thermodynamic investigation into the lead adsorption process from wastewater through magnetic nanocomposite Fe3O4/CNT. Nanotechnol. Environ. Eng. 2017, 2, 13. [Google Scholar] [CrossRef]
- Huang, L.; He, M.; Chen, B.; Hu, B. Magnetic Zr-MOFs nanocomposites for rapid removal of heavy metal ions and dyes from water. Chemosphere 2018, 199, 435–444. [Google Scholar] [CrossRef]
- Ge, L.; Wang, W.; Peng, Z.; Tan, F.; Wang, X.; Chen, J.; Qiao, X. Facile fabrication of Fe@MgO magnetic nanocomposites for efficient removal of heavy metal ion and dye from water. Powder Technol. 2018, 326, 393–401. [Google Scholar] [CrossRef]
- Lu, Z.; Wang, X.Q.; Miao, Y.M.; Chen, Z.Q.; Qiang, P.F.; Cui, L.Q.; Jing, H.; Guo, Y.Q. Magnetic ferroferric oxide nanoparticles induce vascular endothelial cell dysfunction and inflammation by disturbing autophagy. J. Hazard. Mater. 2016, 304, 186–195. [Google Scholar]
- Shen, Y.; Huang, Z.; Liu, X.; Qian, J.; Xu, J.; Yang, X.; Sun, A.; Ge, J. Iron-induced myocardial injury: An alarming side effect of superparamagnetic iron oxide nanoparticles. J. Cell. Mol. Med. 2015, 19, 2032. [Google Scholar] [CrossRef]



| No. | Adsorbent | Adsorbate | Maximum Adsorption Capacity (mg·g−1) | Refs. |
|---|---|---|---|---|
| 1 | Functionalized GOCA beads | Pb (II), Hg (II) and Cd (II) | 602, 374 and 181 | [71] |
| 2 | GO/PAMAMs | Pb (II), Cd (II), Cu (II) and Mn (II) | 568.18, 253.81, 68.68 and 18.29 | [73] |
| 3 | CS/GO-SH | Cu (II), Pb (II) and Cd (II) | 425, 447 and 177 | [74] |
| 4 | MMSP-GO | Pb (II) and Cd (II) | 333 and 167 | [75] |
| 5 | PVK–GO | Pb (II) | 887.98 | [76] |
| 6 | MnFe2O4/GO | Pb (II), As (III) and As (V) | 673, 146 and 207 | [77] |
| 7 | EDTA-mGO | Pb (II), Hg (II) and Cu (II) | 508.4, 268.4 and 301.2 | [78] |
| 8 | GO/L-Trp | Cu (II) and Pb (II) | 588 and 222 | [79] |
| 9 | PAH-GO | Cu (II) | 349.04 | [80] |
| 10 | GO-αCD-PPY NC | Cr (VI) | 666.67 | [81] |
| 11 | RGO/NiO | Cr (VI) | 198 | [82] |
| 12 | PAS–GO | U (VI) and Eu (III) | 310.63 and 243.90 | [83] |
| 13 | Chitosan/GO | Pb (II), Cu (II) and Cr (VI) | 461.3, 423.8 and 310.4 | [84] |
| Metal | E0 (V) | |
|---|---|---|
| Barium (Ba) | −2.90 | |
| Zinc (Zn) | −0.76 | |
| Iron (Fe) | −0.41 | |
| Cadmium (Cd) | −0.40 | |
| Nickel (Ni) | −0.24 | |
| Lead (Pb) | −0.13 | |
| Copper (Cu) | 0.34 | |
| Silver (Ag) | 0.80 | |
| Mercury (Hg) | 0.86 | |
| Chromium (Cr) | 1.36 |
| No. | Adsorbent | Adsorbate | Maximum Adsorption Capacity (mg·g−1) | Refs. |
|---|---|---|---|---|
| 1 | DESs-CNTs | Hg (II) | 186.97 | [61] |
| 2 | amino-functionalized Fe3O4/MWCNTs | Cu (II) | 30.49 | [63] |
| 3 | GO | Zn (II) | 246 | [69] |
| 4 | few-layered GO nanosheets | Cd (II) and Co (II) | 106.3 and 68.2 | [70] |
| 5 | functionalized GOCA beads | Pb (II), Hg (II) and Cd (II) | 602, 374 and 181 | [71] |
| 6 | GOx-microbots | Pb (II) | - | [72] |
| 7 | Bifunctional silica nanospheres | Cu (II) and methylene blue. | 139.8 and 99.0 | [86] |
| 8 | NH2–SNHS | Pb (II), Cd (II) and Ni (II) | 96.79, 40.73 and 31.29 | [87] |
| 9 | Sil-Phy-NPANI | Cu (II), Cd (II), Hg (II) and Pb (II) | 108, 90, 120 and 186 | [85] |
| 10 | Sil-Phy-CrossNPANI | Cu (II), Cd (II), Hg (II) and Pb (II) | 105, 118, 271 and 300 | [85] |
| 11 | SDS-nZVI | Cr (VI) | 253.68 | [101] |
| 12 | Au-doped nZVI | Cd (II) | 188 | [102] |
| 13 | B-nZVI | Pb (II), Cu (II), Cd (II), Co (II), Ni (II) and Zn (II) | 50.25, 70.20, 14.25, 12.90, 16.50 and 34.95 | [103] |
| 14 | Ag@MSA | Hg (II) | 800 | [108] |
| 15 | Au | Hg (0) | 4065 | [110] |
| 16 | citrate-coated Au nanoparticles | Hg (II) | - | [111] |
| 17 | α-FeOOH | Cu (II) | 149.25 | [117] |
| 18 | goethite nanocrystalline powders | Cd (II) | 167 | [118] |
| 19 | nano-hematite | Cr (VI) | 6.33–200 | [127] |
| 20 | nano-hematite | Pb (II), Cd (II), Cu (II) and Zn (II) | _ | [129] |
| 21 | superparamagnetic hematite nanoparticles | Al (III), Mg (II), Mn (II), Zn (II), Ni (II) | - | [130] |
| 22 | maghemite nanoparticles | Cu (II), Ni (II), Mn (II), Cd (II) and Cr (VI) | - | [135] |
| 23 | maghemite nanoparticle | Pb (II) and Cu (II) | 68.9 and 34.0 | [136] |
| 24 | MAMNPs | Ag (I), Hg (II), Pb (II) and Cd (II) | 260.55, 237.60, 118.51 and 91.55 | [137] |
| 25 | magnetite nanoparticles | Pb (II), Cu (II), Zn (II), Mn (II) | 37.3, 10.8, 10.5 and 7.69 | [145] |
| 26 | amino functionalized Fe3O4 nanoparticles | Cr (VI) and Ni (II) | 232.51 and 222.12 | [151] |
| 27 | Fe3O4 @PTMT | Pb (II), Hg (II) and Cd (II) | 533.13, 603.16 and 216.59 | [159] |
| 28 | HFO-P(TAA/HEA) | Pb (II), Cu (II), Cd (II) and Ni (II) | 303.8, 107.5, 149.8 and 85.87 | [164] |
| 29 | HFO nanoparticles | As | 92 | [165] |
| 30 | HFO-CMC | As (V) | 355 | [166] |
| 31 | manganese dioxide/gelatin | Pb (II) and Cd (II) | 318.7 and 105.1 | [169] |
| 32 | nanoscale manganese dioxide | Tl (I) | 672 | [170] |
| 33 | ZnO nanoparticles | Zn (II), Cd (II) and Hg (II) | 357, 387 and 714 | [179] |
| 34 | casein-capped ZnO nanoparticles | Cd (II), Pd (II) and Co (II) | 156.74, 194.93 and 67.93 | [181] |
| 35 | nanosized TiO2 | Cr (VI) | 12.6 | [184] |
| 36 | TiO2 nanowire | Pb (II), Cu (II), Fe (III), Cd (II) and Zn (II) | - | [185] |
| 37 | Starch-coated TiO2 nanoparticles | Cd (II), Co (II), Cu (II), Ni (II) and Pb (II) | - | [187] |
| 38 | TiO2-chitosan nanoparticles | Cu (II) and Cd (II) | - | [188] |
| 39 | γ-Al2O3 nanoparticles | Pb (II) and Cd (II) | 47.08 and 17.22 | [194] |
| 40 | Al2O3 nanoparticles | Zn (II) and Cd (II) | - | [195] |
| 41 | TiO2 nanoparticles | Cd (II), Cu (II), Ni (II) and Pb (II) | 120.1, 50.2, 39.3 and 21.7 | [202] |
| 42 | Al2O3 nanoparticles | Cd (II), Cu (II), Ni (II) and Pb (II) | 118.9, 47.9, 35.9 and 41.2 | [202] |
| 43 | MgO nanoparticles | Cd (II), Cu (II), Ni (II) and Pb (II) | 135, 149.1, 149.9 and 148.6 | [202] |
| 44 | MgO nanoparticles | Cu (II) | - | [203] |
| 45 | MgO nanoparticles | Cd (II) and Pb (II) | 2294 and 2614 | [204] |
| 46 | mesoporous MgO nanosheets | Ni (II) | 1684.25 | [205] |
| 47 | CeO2 nanoparticles | Cr (VI) | 121.95 | [209] |
| 48 | CeO2 nanoparticles | As (III) and As (V) | 71.9 and 36.8 | [210] |
| 49 | CeO2 nanopowder | Pb (II), Cu (II) and Zn (II) | 23,-,- | [213] |
| 50 | mesoporous ZrO2 | Cr (VI) | 73.0 | [215] |
| 51 | ZrO2/B2O3 nanocomposite | Co (II), Cu (II) and Cd (II) | 32.2, 46.5 and 109.9 | [216] |
| 52 | HZO-PS | Cd (II) | - | [217] |
| 53 | NZP | Pb (II) and Cd (II) | 319.4 and 214.7 | [218] |
| 54 | CNTs/chitosan | Cu (II), Zn (II), Cd (II), and Ni (II) | - | [226] |
| 55 | CNT/PAMAM | As (III), Co (II) and Zn (II) | 432, 494 and 470 | [231] |
| 56 | HAp/NaP | Pb (II) and Cd (II) | 55.55 and 40.16 | [236] |
| 57 | polypyrrole-polyaniline/Fe3O4 | Pb (II) | 243.9 | [243] |
| 58 | NC-AgNPs | Pb (II) and Cr (III) | 9.42 and 8.93 | [245] |
| 59 | ZOCS | Pb (II), Cd (II) and Cu (II) | 476.1, 135.1 and 117.6 | [246] |
| 60 | chitosan/alginate nanocomposite | Cr (VI) | 108.8 | [248] |
| 61 | Fe3O4@SiO2/Zr-MOFs | Pb (II) | 102 | [255] |
| 62 | Fe@MgO | Pb (II) | 1476.4 | [256] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Hou, B.; Wang, J.; Tian, B.; Bi, J.; Wang, N.; Li, X.; Huang, X. Nanomaterials for the Removal of Heavy Metals from Wastewater. Nanomaterials 2019, 9, 424. https://doi.org/10.3390/nano9030424
Yang J, Hou B, Wang J, Tian B, Bi J, Wang N, Li X, Huang X. Nanomaterials for the Removal of Heavy Metals from Wastewater. Nanomaterials. 2019; 9(3):424. https://doi.org/10.3390/nano9030424
Chicago/Turabian StyleYang, Jinyue, Baohong Hou, Jingkang Wang, Beiqian Tian, Jingtao Bi, Na Wang, Xin Li, and Xin Huang. 2019. "Nanomaterials for the Removal of Heavy Metals from Wastewater" Nanomaterials 9, no. 3: 424. https://doi.org/10.3390/nano9030424
APA StyleYang, J., Hou, B., Wang, J., Tian, B., Bi, J., Wang, N., Li, X., & Huang, X. (2019). Nanomaterials for the Removal of Heavy Metals from Wastewater. Nanomaterials, 9(3), 424. https://doi.org/10.3390/nano9030424






