Confined Crystallization of Pigment Red 146 in Emulsion Droplets and Its Mechanism
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Crystallization of PR 146 in Bulk Solution
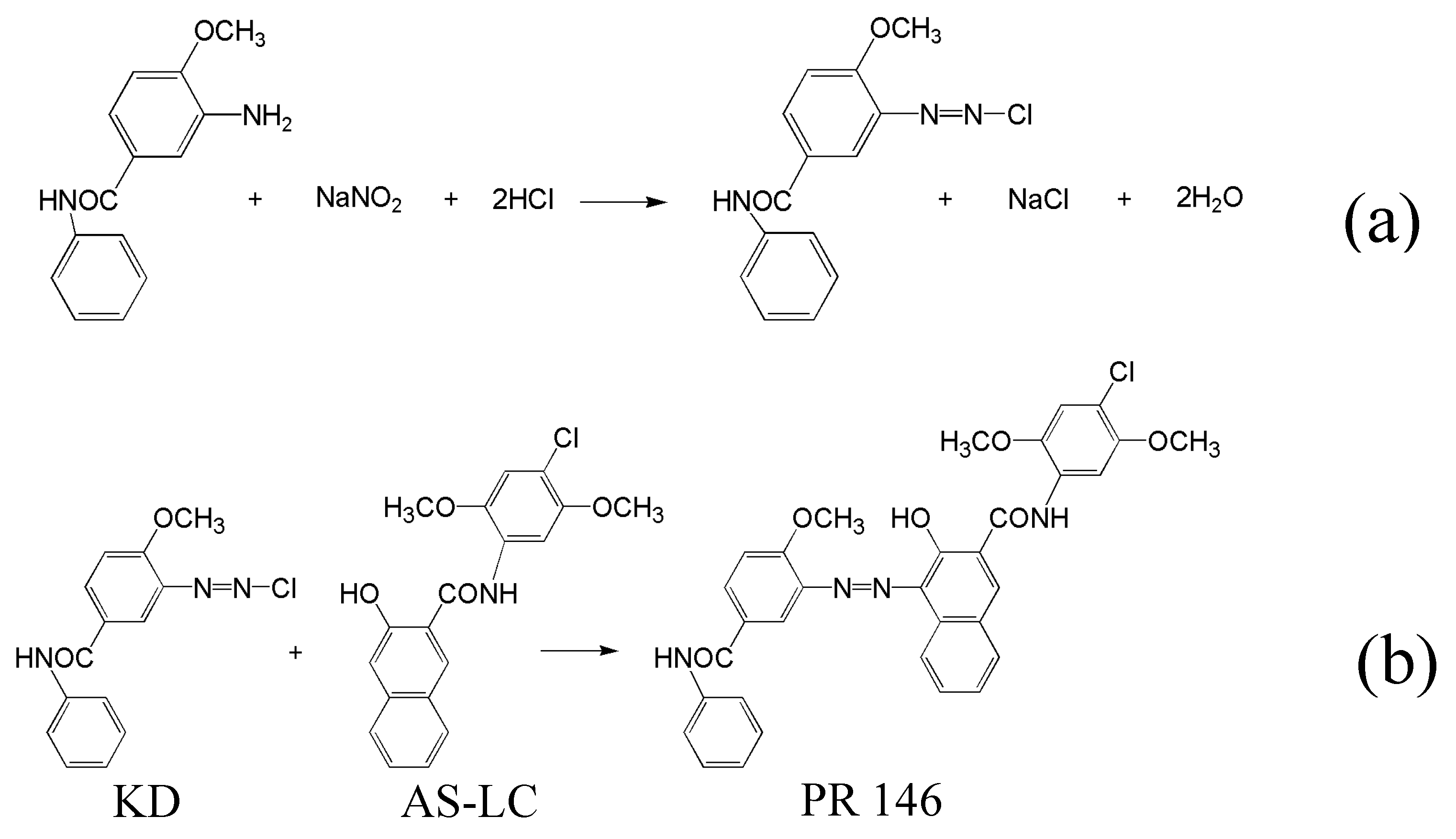
2.3. Crystallization of PR 146 in Macroemulsion
2.4. Crystallization of PR 146 in Miniemulsion
2.5. Characterization Methods
3. Results and Discussion
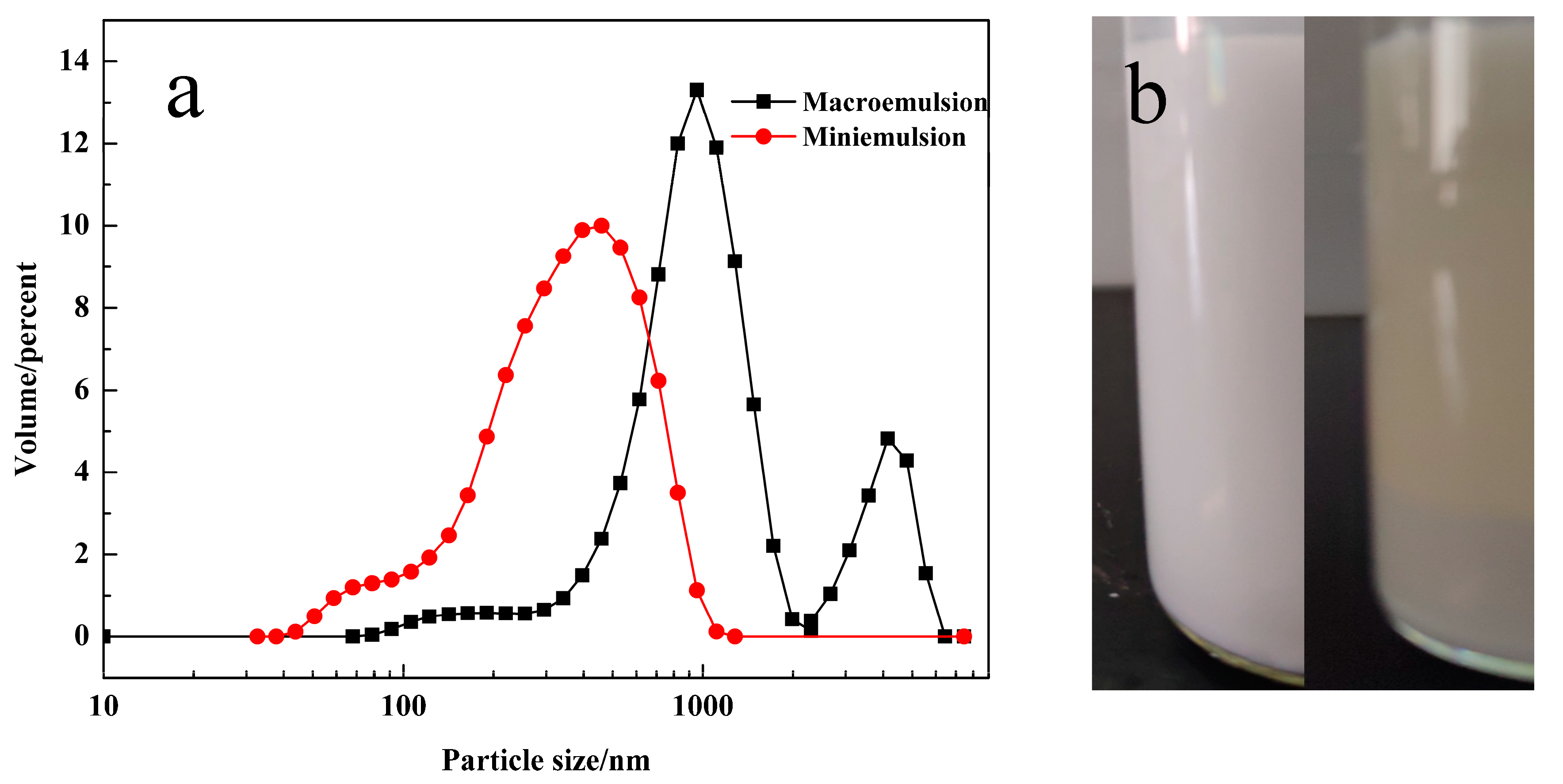
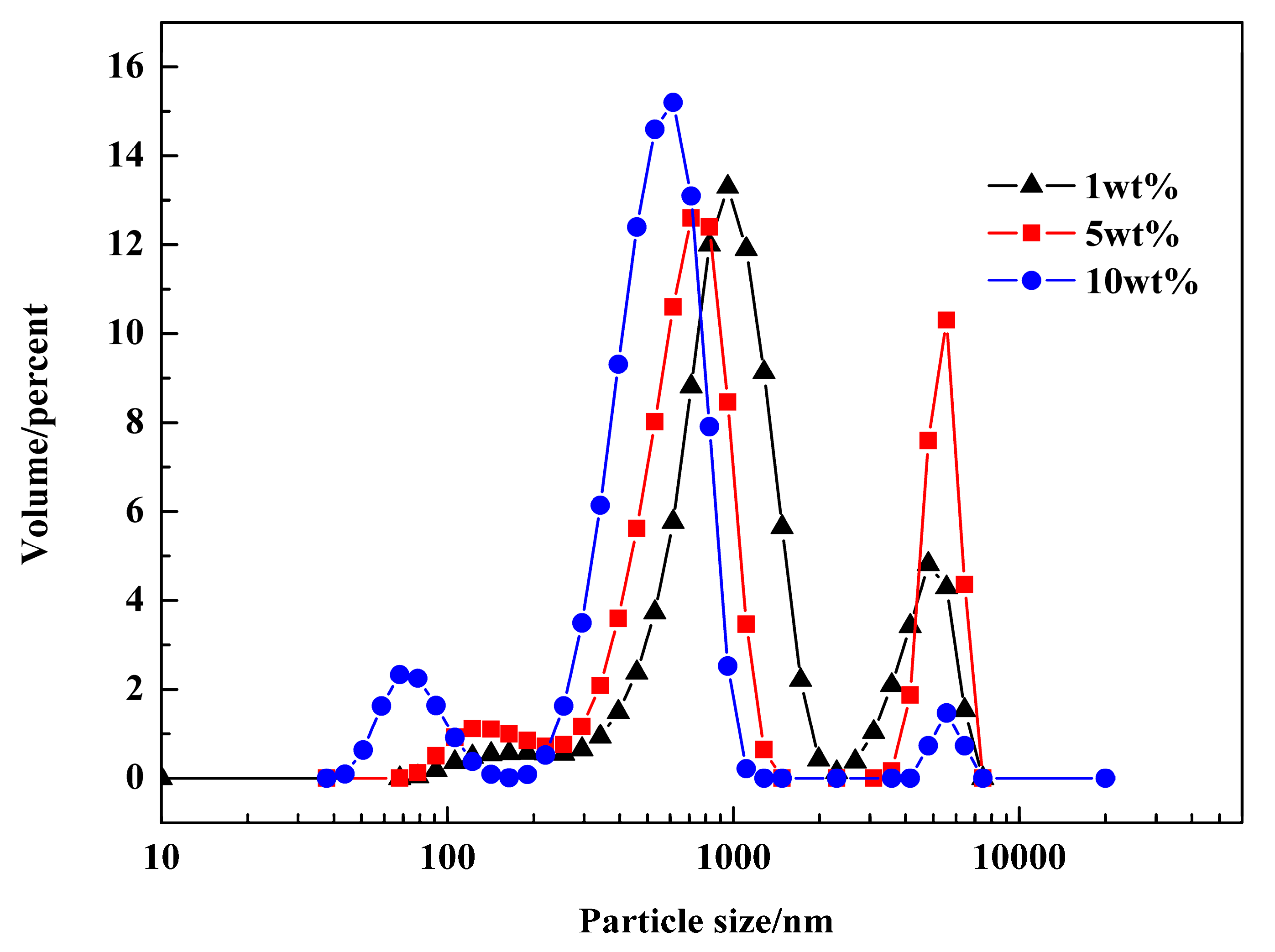
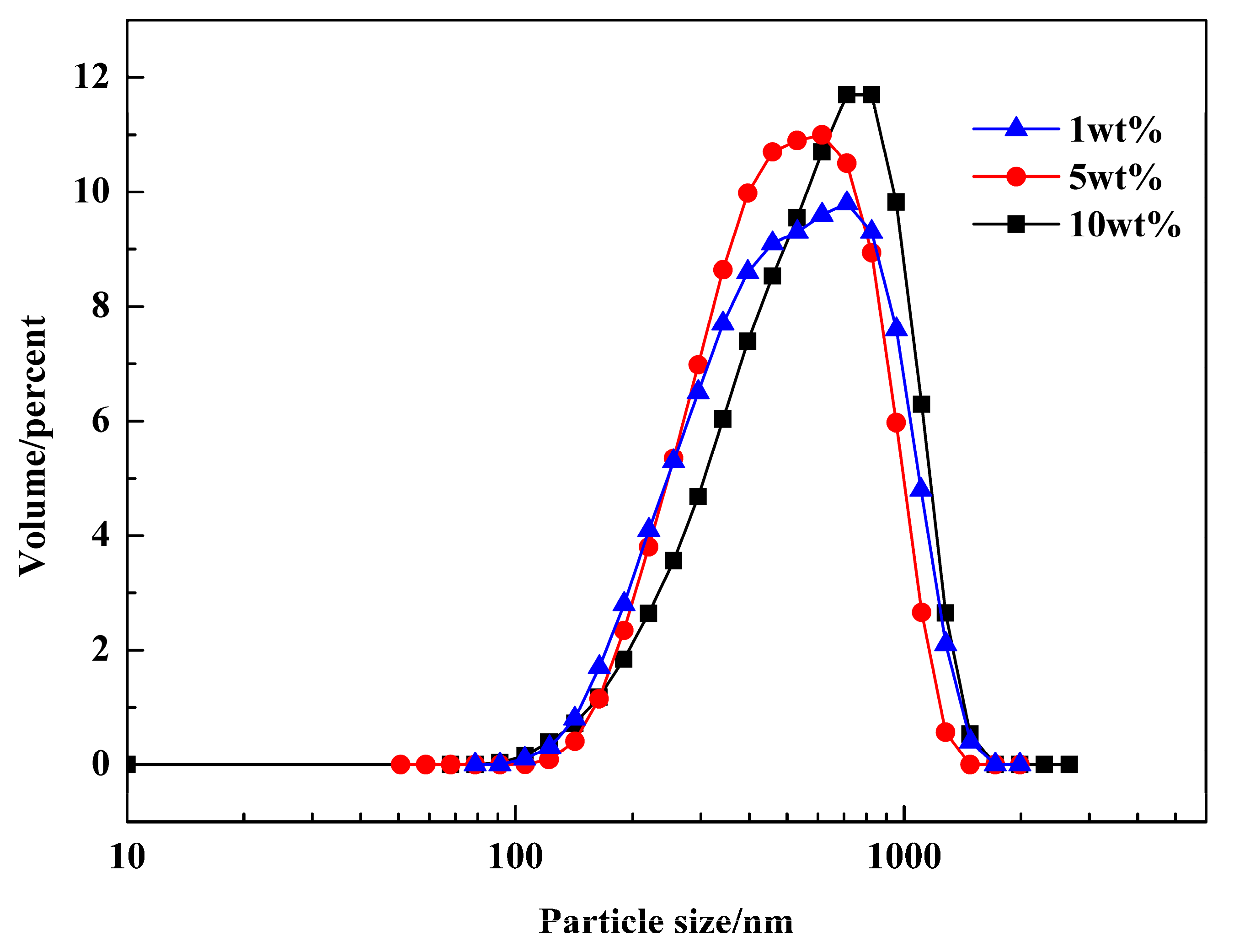
3.1. Analysis of the Water Droplets in Emulsions
3.2. Determination of the Reaction Conditions in Emulsions
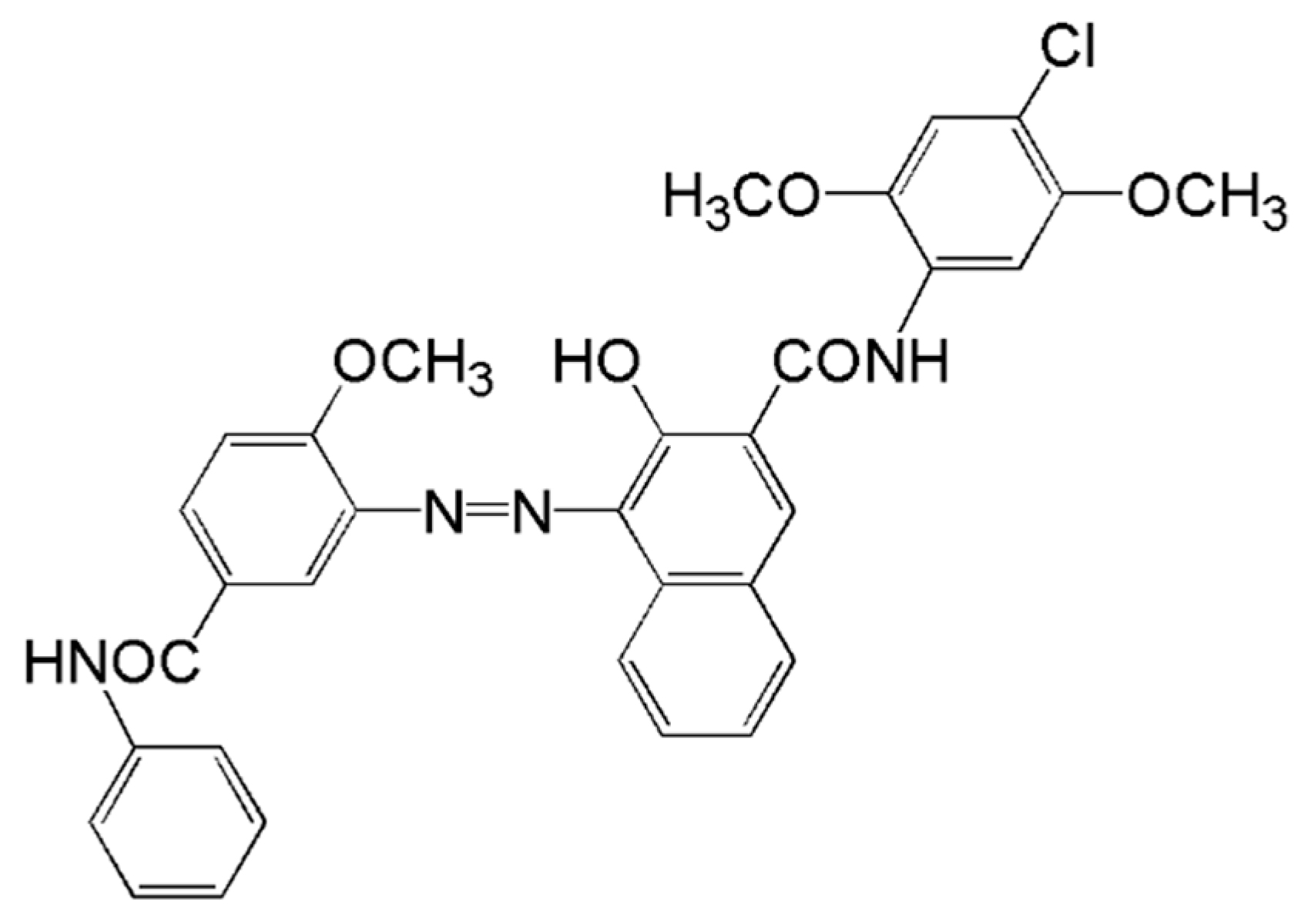
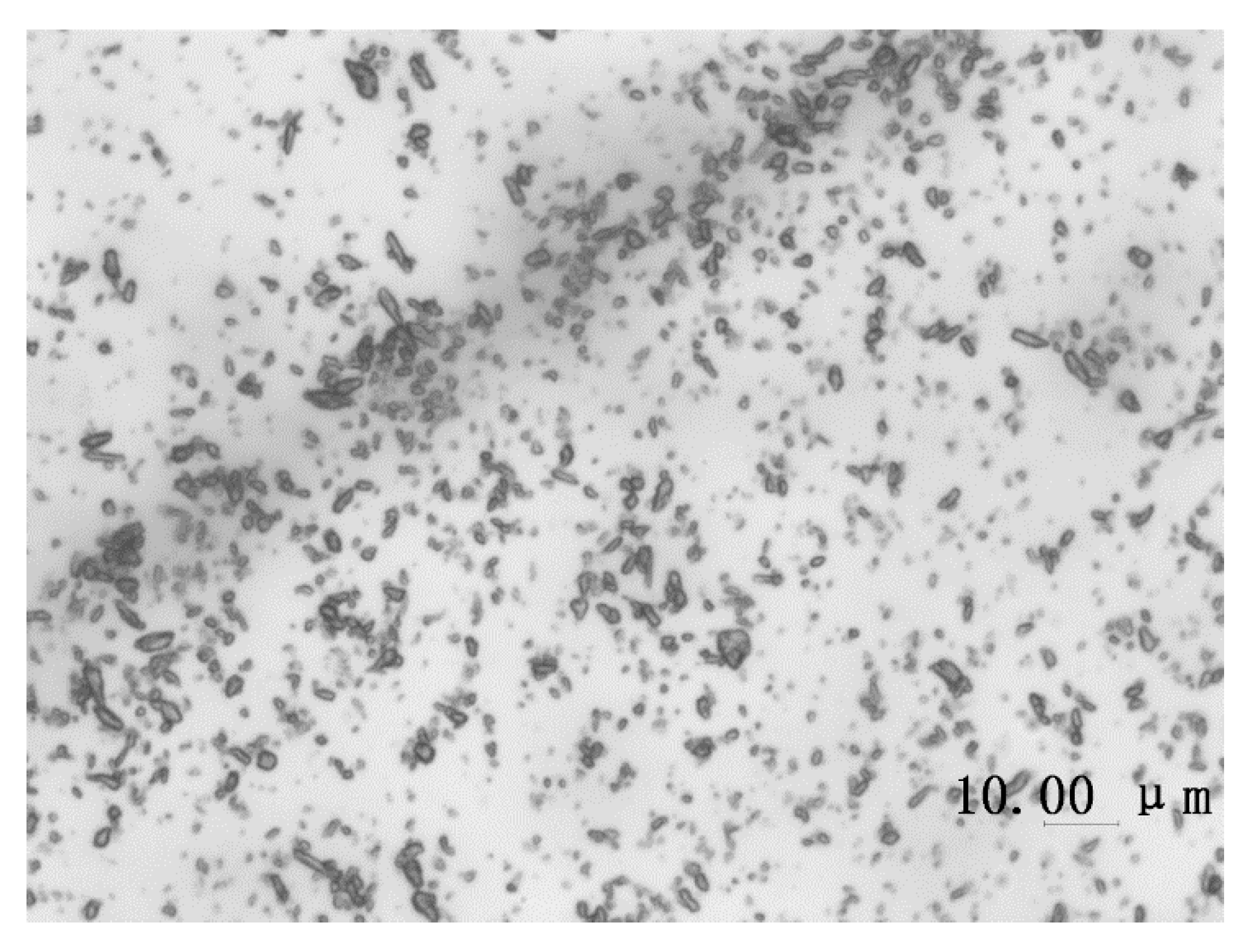
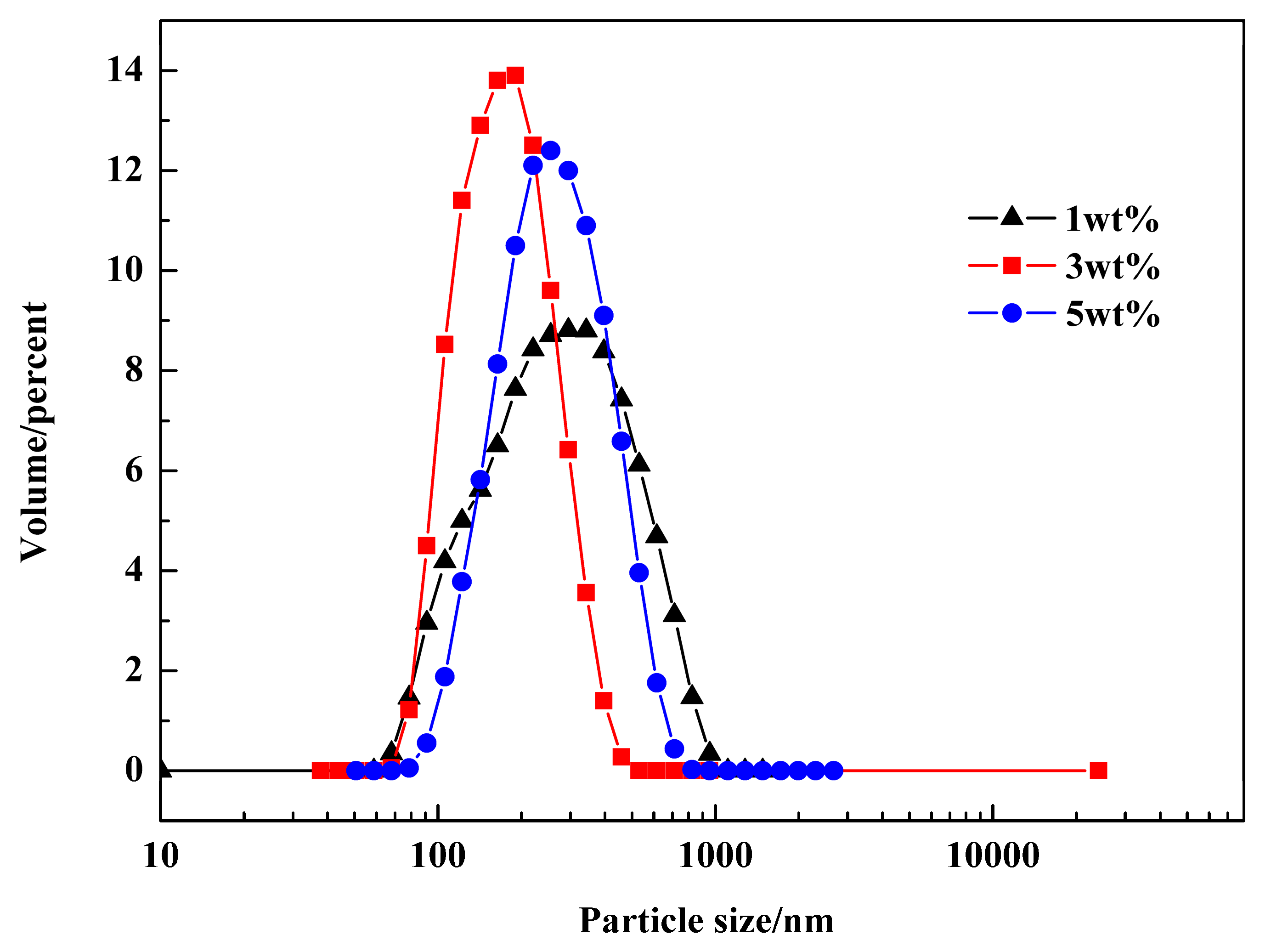
3.3. Confined Crystallization within Emulsion Droplets
3.4. Process Mechanism for Crystallization in Confined Space
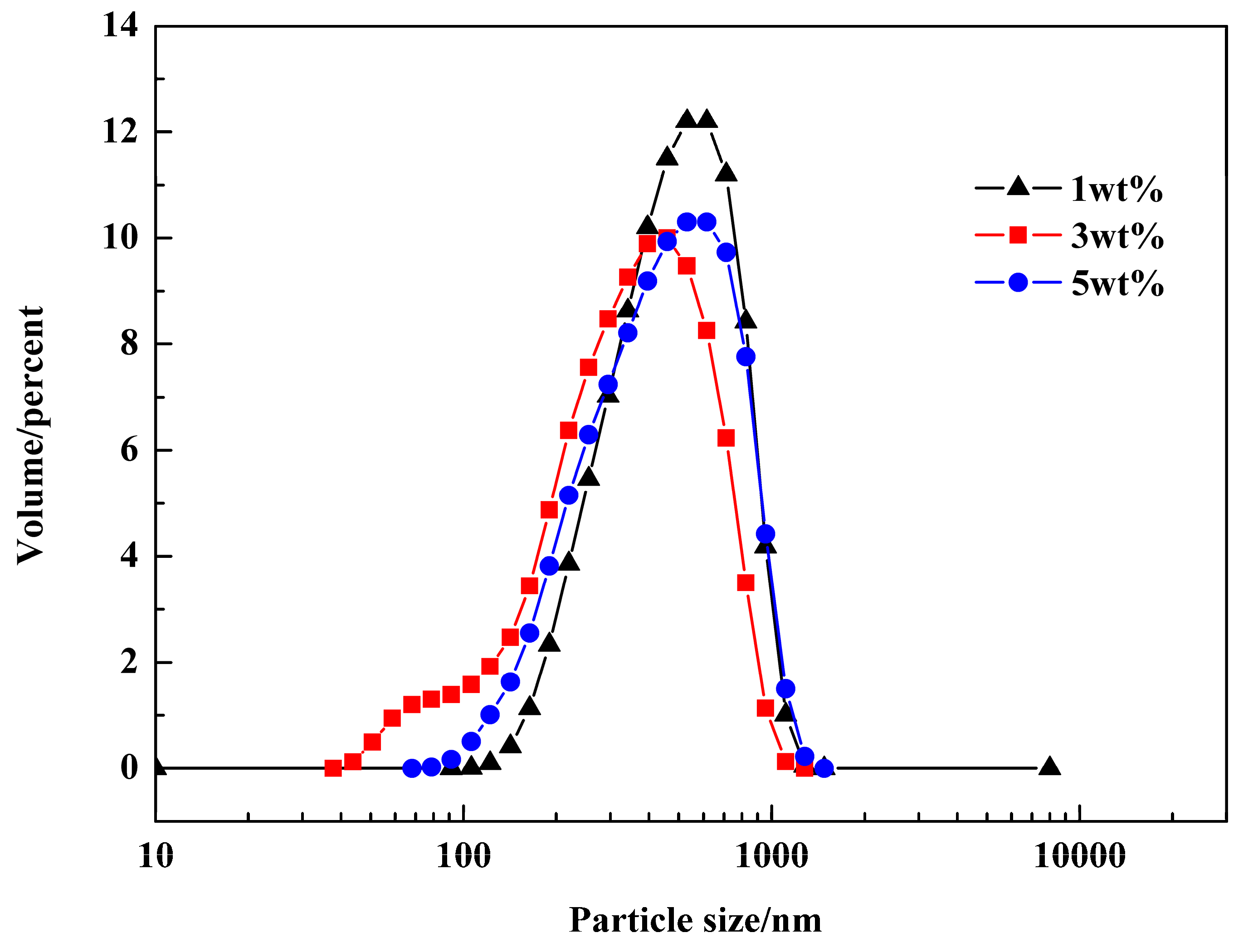
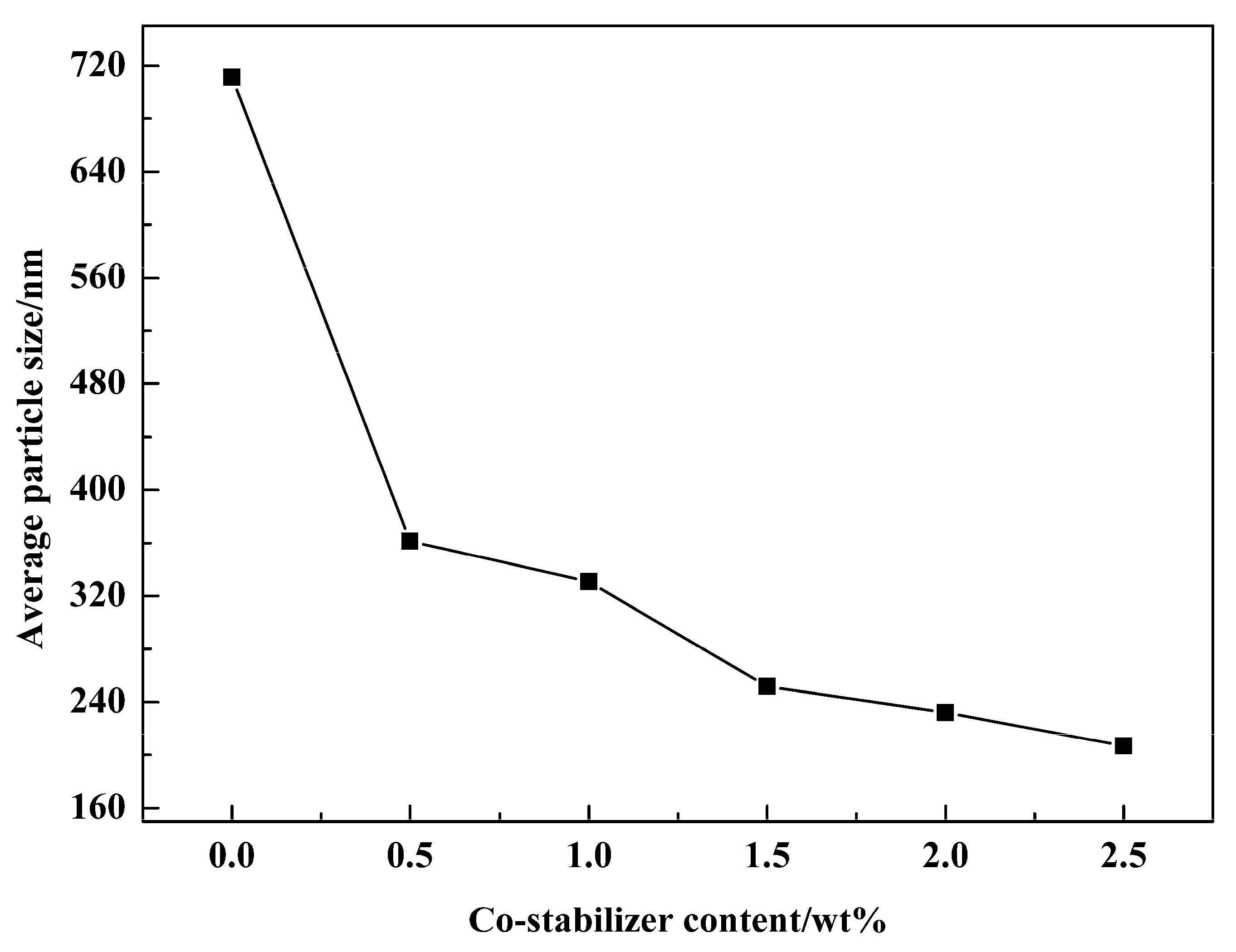
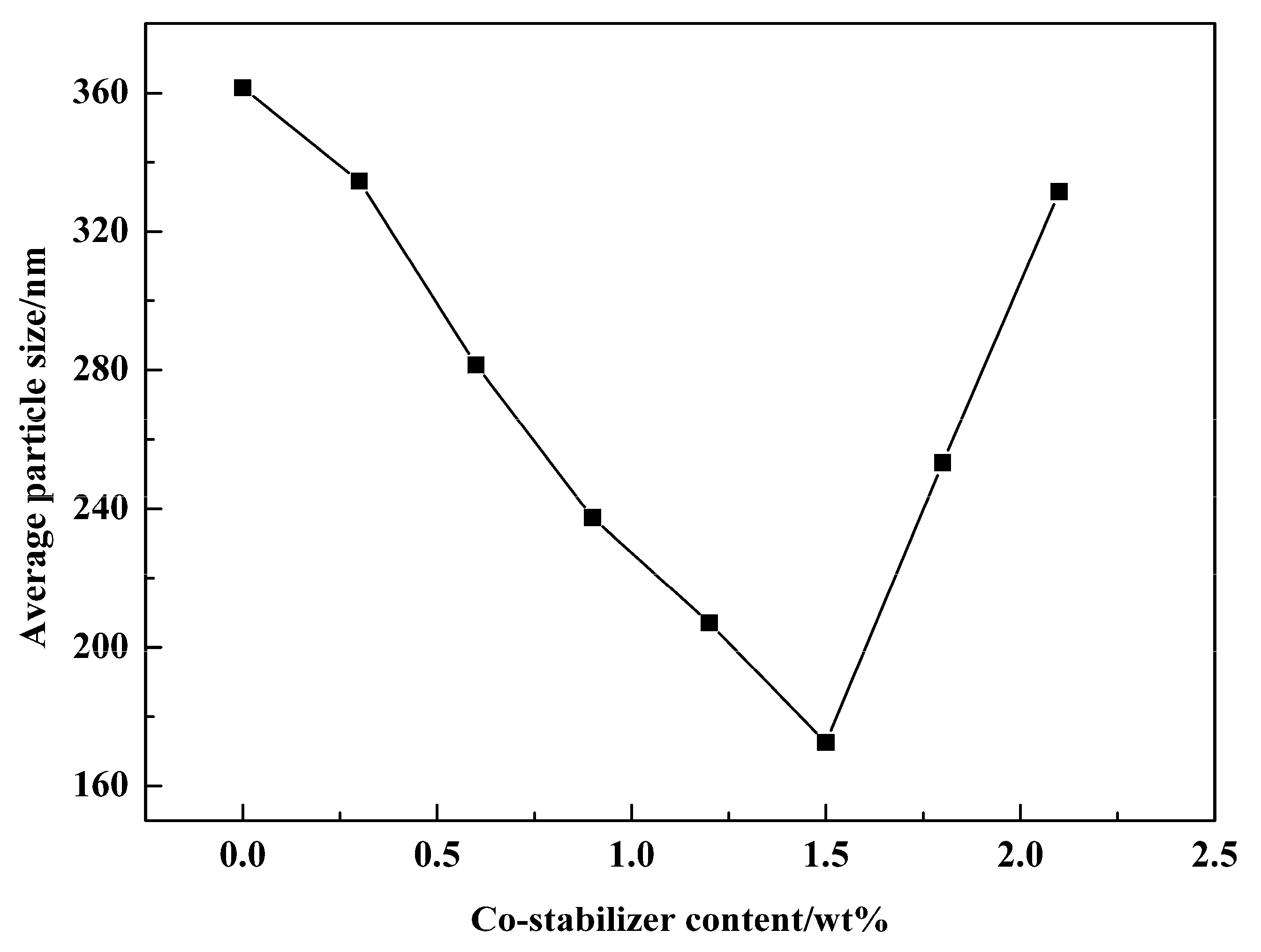
3.5. Effect of Emulsifier and Co-Stabilizer Dosage
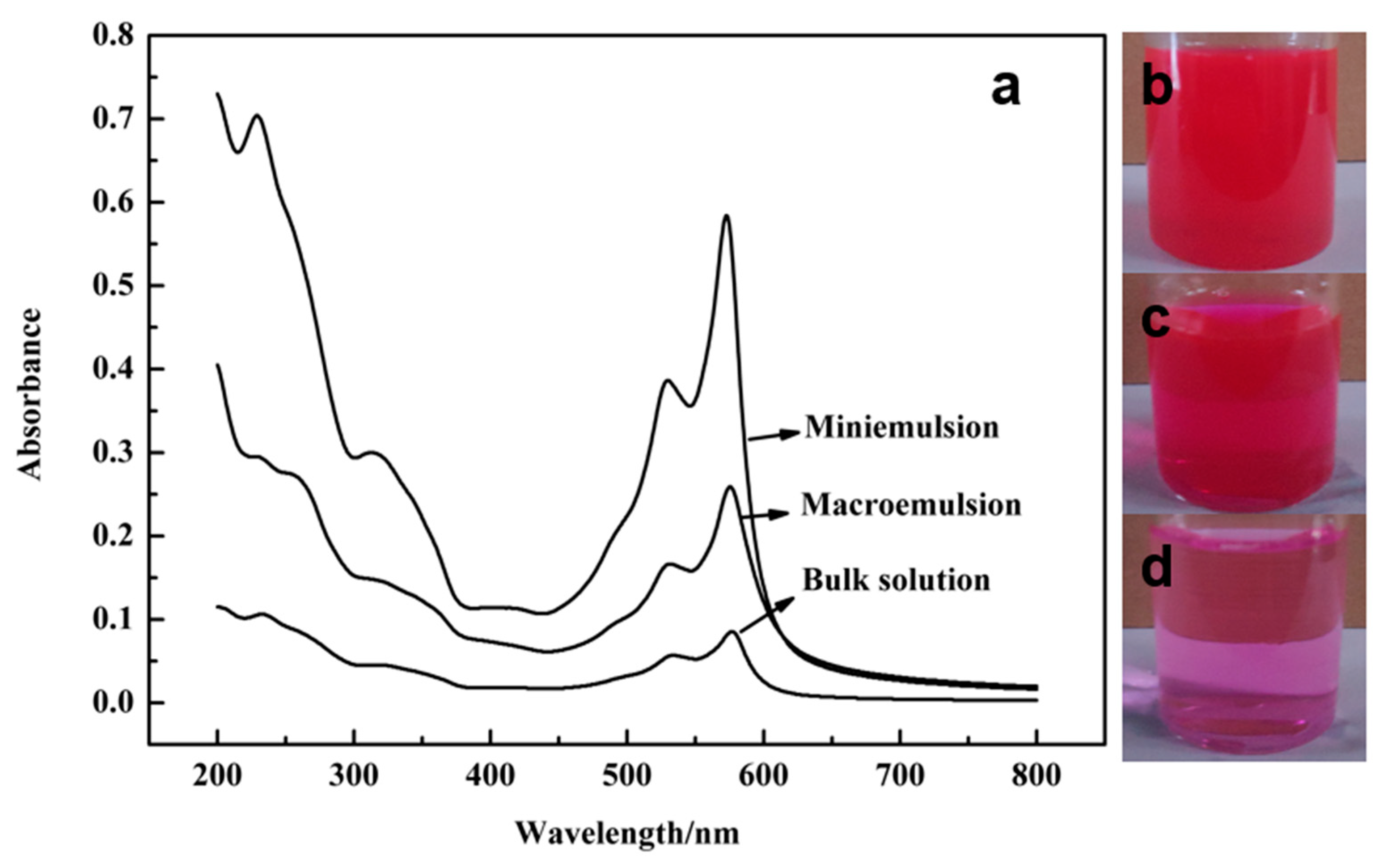
3.6. UV-Vis Spectra of the Samples
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Honarvar, M.G.; Latifi, M. Overview of wearable electronics and smart textiles. J. Text. Inst. 2016, 108, 631–652. [Google Scholar] [CrossRef]
- Mhetre, S.; Carr, W.; Radhakrishnaiah, P. On the relationship between ink-jet printing quality of pigment ink and the spreading behavior of ink drops. J. Text. Inst. 2010, 101, 423–430. [Google Scholar] [CrossRef]
- Li, J.; Fan, J.; Cao, R.; Zhang, Z.; Du, J.; Peng, X. Encapsulated dye/polymer nanoparticles prepared via miniemulsion polymerization for inkjet printing. ACS Omega 2018, 3, 7380–7387. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Q.; Chang, Q.; Hu, S.; Wang, X.; Yang, K. Effect of particle size on the blue chromate pigment CoAl2O4. Ceram. Sci. Technol. 2018, 9, 43–46. [Google Scholar]
- Sousa, S.C.L.; Mendes, A.D.O.; Fiadeiro, P.T.; Ramos, A.M.M. Dynamic interactions of pigment-based inks on chemically modified papers and their influence on inkjet print quality. Ind. Eng. Chem. Res. 2014, 53, 4660–4668. [Google Scholar] [CrossRef]
- Güngör, G.L.; Kara, A.; Blosi, M.; Gardini, D.; Guarini, G.; Zanelli, C.; Dondi, M. Micronizing ceramic pigments for inkjet printing: Part I. Grindability and particle size distribution. Ceram. Int. 2015, 41, 6498–6506. [Google Scholar] [CrossRef]
- Elgammal, M.; Schneider, R.; Gradzielski, M. Development of self-curable hybrid pigment inks by miniemulsion polymerization for inkjet printing of cotton fabrics. Dyes Pigment. 2016, 133, 467–478. [Google Scholar] [CrossRef]
- Hakeim, O.A.; Diab, H.A.; Adams, J. Preparation and characterization of UV curable-encapsulated phthalocyanine blue pigment. Prog. Org. Coat. 2015, 84, 70–78. [Google Scholar] [CrossRef]
- Jovaní, M.; Domingo, M.; Machado, T.R.; Longo, E.; Beltrán-Mir, H.; Cordoncillo, E. Pigments based on Cr and Sb doped TiO2 prepared by microemulsion-mediated solvothermal synthesis for inkjet printing on ceramics. Dyes Pigment. 2015, 116, 106–113. [Google Scholar] [CrossRef]
- Nsib, F.; Ayed, N.; Chevalier, Y. Selection of dispersants for the dispersion of C.I. Pigment Violet 23 in organic medium. Dyes Pigment. 2007, 74, 133–140. [Google Scholar] [CrossRef]
- Xiao, Z.; Qin, M.; Liu, J.; Li, Q. Hydrophobic dye/polymer composite colorants synthesized by miniemulsion solvent evaporation technique. Dyes Pigment. 2014, 100, 41–49. [Google Scholar]
- Muñoz-Espí, R.; Mastai, Y.; Gross, S.; Landfester, K. Colloidal systems for crystallization processes from liquid phase. CrystEngComm 2013, 15, 2175–2191. [Google Scholar] [CrossRef]
- Antonello, A.; Benedetti, C.; Perez-Pla, F.F.; Kokkinopoulou, M.; Kirchhoff, K.; Fischer, V.; Landfester, K.; Gross, S.; Muñoz-Espí, R. Colloidally confined crystallization of highly efficient ammonium phosphomolybdate catalysts. ACS Appl. Mater. Interfaces 2018, 10, 23174–23186. [Google Scholar] [CrossRef] [PubMed]
- Antonello, A.; Jakob, G.; Dolcet, P.; Momper, R.; Kokkinopoulou, M.; Landfester, K.; Muñoz-Espí, R.; Gross, S. Synergy of miniemulsion and solvothermal conditions for the low temperature crystallization of magnetic nanostructured transition metal ferrites. Chem. Mater. 2017, 29, 985–997. [Google Scholar] [CrossRef]
- Varol, H.S.; Álvarez-Bermúdez, O.; Dolcet, P.; Kuerbanjiang, B.; Muñoz-Espí, R. Crystallization at nanodroplet interfaces in emulsion systems: A soft-template strategy for preparing porous and hollow nanoparticles. Langmuir 2016, 32, 13116–13123. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Cha, J.; Stucky, G.D.; Morse, D.E. Silicatein alpha: cathepsin L-like protein in sponge biosilica. Proc. Natl. Acad. Sci. USA 1998, 95, 6234–6238. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Shen, M.; Möhwald, H.J. Polyelectrolyte multilayer nanoreactors toward the synthesis of diverse nanostructured materials. Prog. Polym. Sci. 2004, 29, 987–1019. [Google Scholar] [CrossRef]
- Singh, I.; Landfester, K.; Chandra, A.; Munoz-Espi, R. A new approach for crystallization of copper(II) oxide hollow nanostructures with superior catalytic and magnetic response. Nanoscale 2015, 7, 19250–19258. [Google Scholar] [CrossRef] [PubMed]
- Shavel, A.; Gaponik, N.; Eychmüller, A. The assembling of semiconductor nanocrystals. Eur. J. Inorg. Chem. 2010, 36, 3613–3623. [Google Scholar]
- Chen, Z.; Yu, F.; Zeng, X.; Zhang, Z. Preparation, characterization and thermal properties of nanocapsules containing phase change material n-dodecanol by miniemulsion polymerization with polymerizable emulsifier. Appl. Energy 2012, 91, 7–12. [Google Scholar] [CrossRef]
- Fei, X.; Cao, L.; Liu, Y.J.; Modified, C.I. Pigment Red 170 with a core-shell structure: Preparation, characterization and computational study. Dyes Pigment. 2016, 125, 192–200. [Google Scholar] [CrossRef]

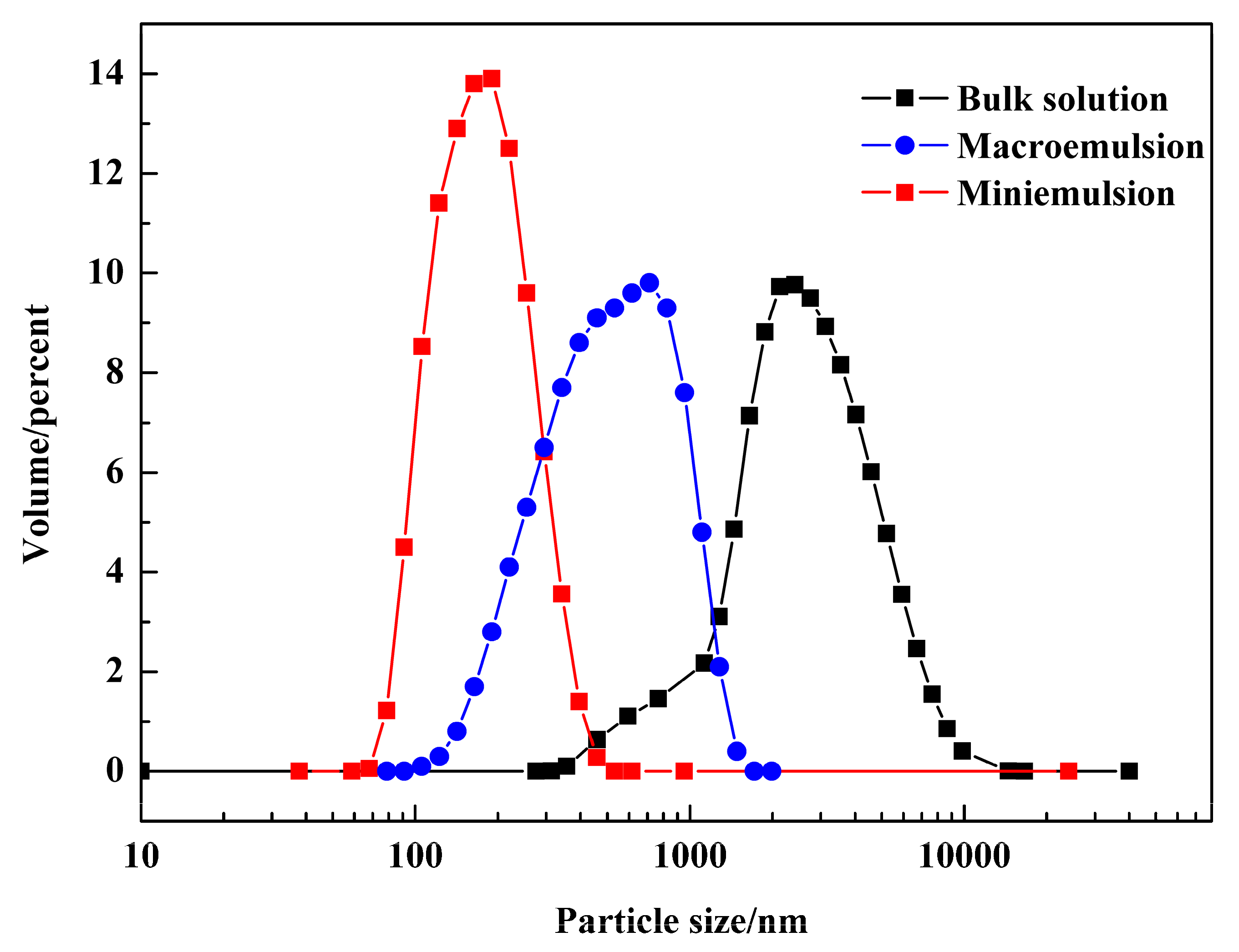
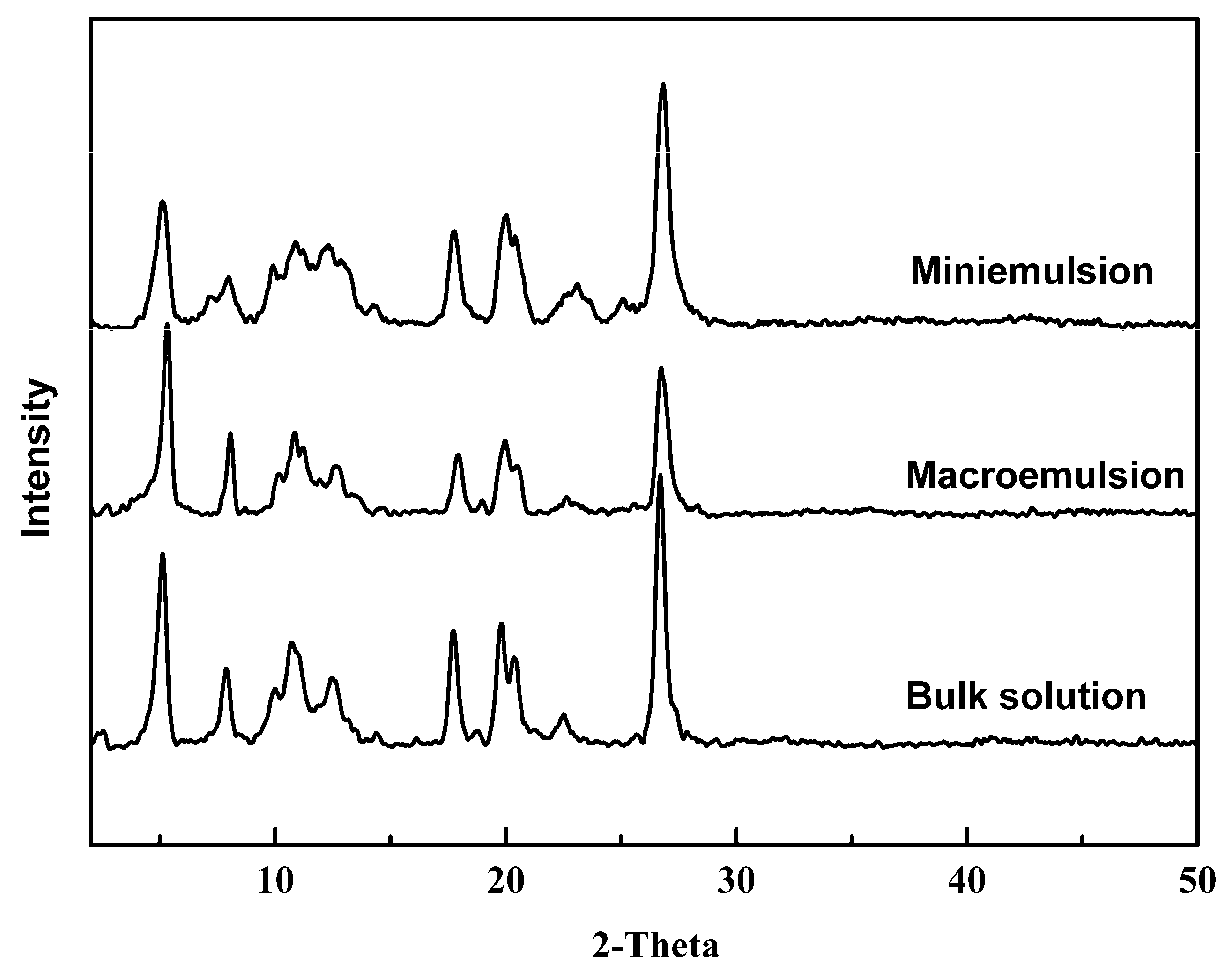

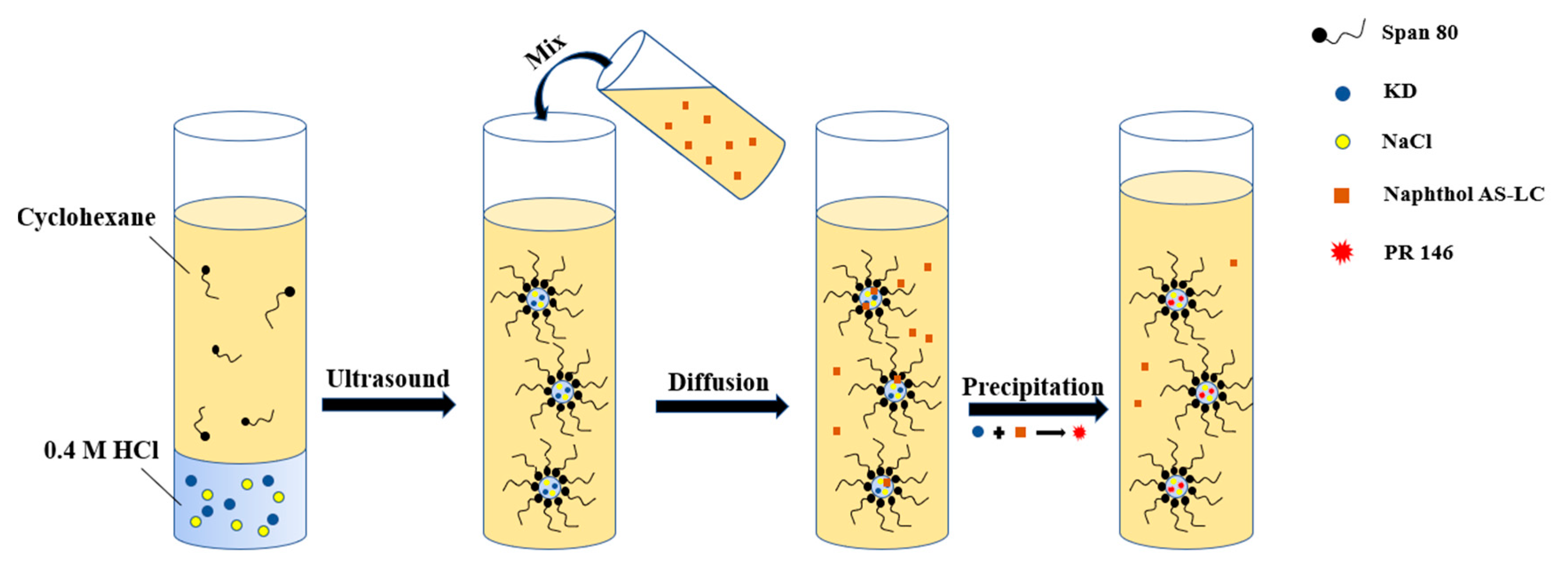
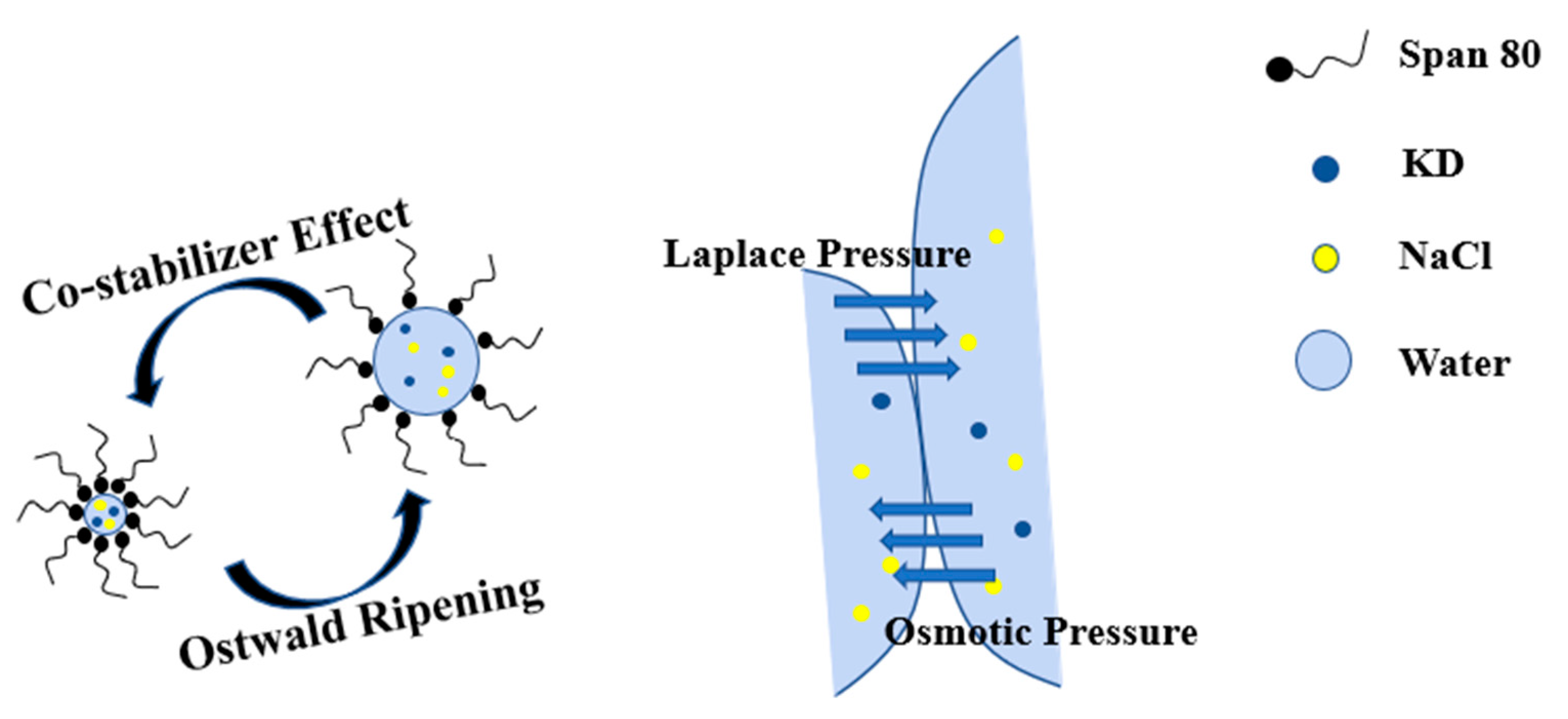
| Crystallization Method | Average Particle Size/nm | Crystallite Size/nm |
|---|---|---|
| Bulk solution | 2840.0 | 17.2 |
| Macroemulsion | 361.4 | 19.6 |
| Miniemulsion | 172.5 | 15.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, X.; Wang, Y.; Li, X.; Chen, X.; Lv, D.; Xie, C.; Yin, Q.; Zhang, X.; Hao, H. Confined Crystallization of Pigment Red 146 in Emulsion Droplets and Its Mechanism. Nanomaterials 2019, 9, 379. https://doi.org/10.3390/nano9030379
Meng X, Wang Y, Li X, Chen X, Lv D, Xie C, Yin Q, Zhang X, Hao H. Confined Crystallization of Pigment Red 146 in Emulsion Droplets and Its Mechanism. Nanomaterials. 2019; 9(3):379. https://doi.org/10.3390/nano9030379
Chicago/Turabian StyleMeng, Xianze, Yongli Wang, Xin Li, Xue Chen, Dongjun Lv, Chuang Xie, Qiuxiang Yin, Xuling Zhang, and Hongxun Hao. 2019. "Confined Crystallization of Pigment Red 146 in Emulsion Droplets and Its Mechanism" Nanomaterials 9, no. 3: 379. https://doi.org/10.3390/nano9030379
APA StyleMeng, X., Wang, Y., Li, X., Chen, X., Lv, D., Xie, C., Yin, Q., Zhang, X., & Hao, H. (2019). Confined Crystallization of Pigment Red 146 in Emulsion Droplets and Its Mechanism. Nanomaterials, 9(3), 379. https://doi.org/10.3390/nano9030379






