Electrical Property of Graphene and Its Application to Electrochemical Biosensing
Abstract
1. Introduction
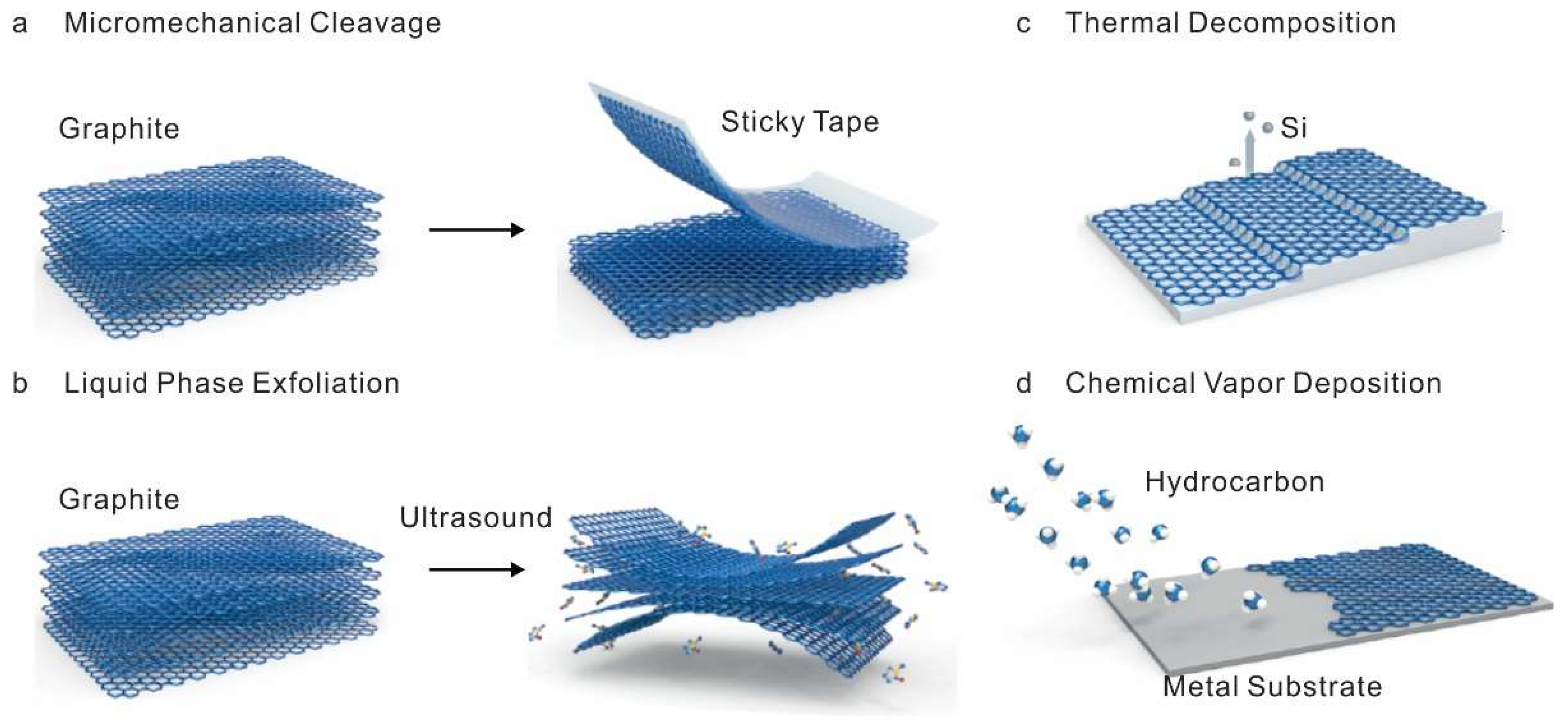
2. Synthesis of Graphene
2.1. Mechanical and Chemical Exfoliation Method
2.2. Thermal Decomposition and Chemical Vapor Deposition Method
3. Application to Electrochemical Sensing
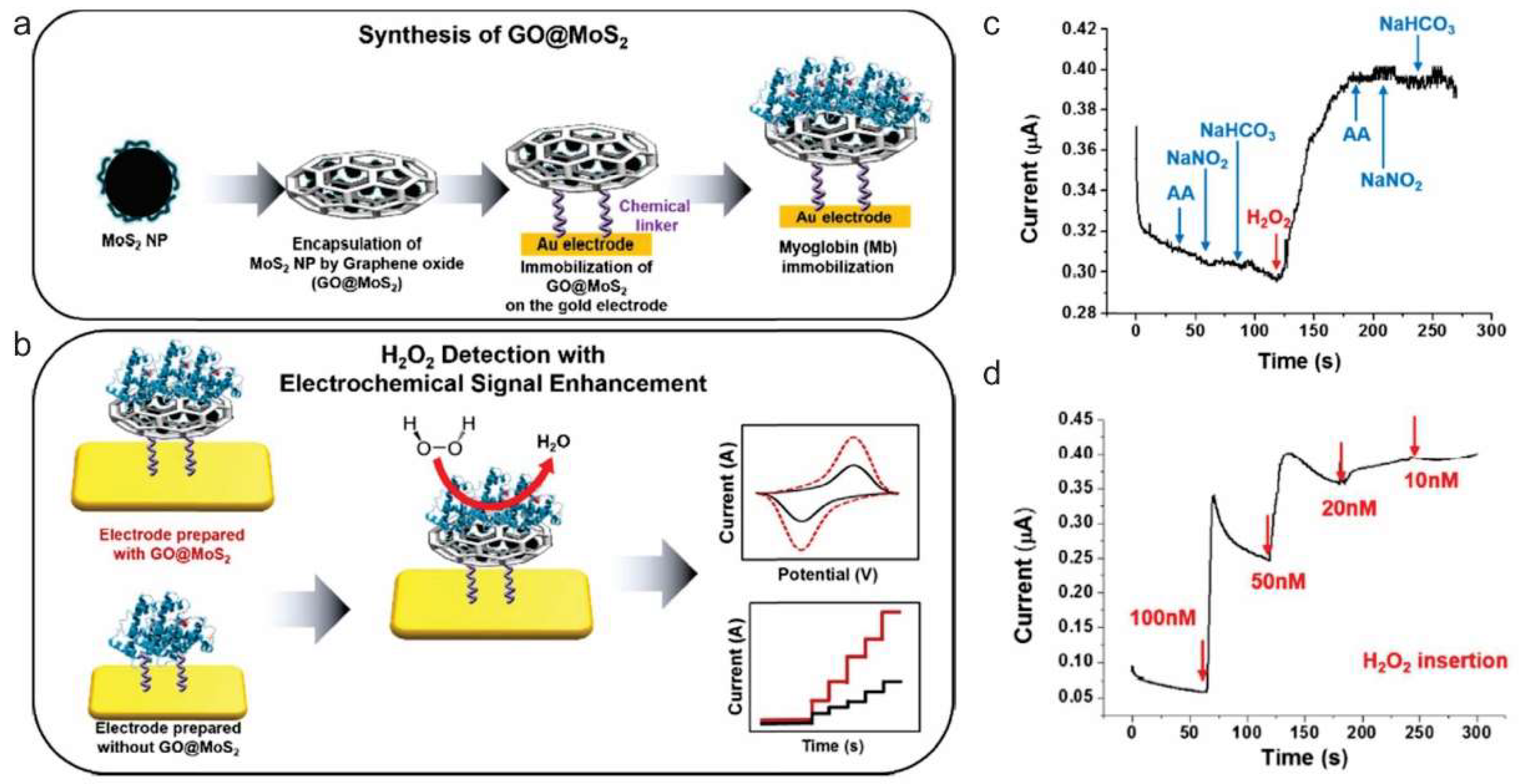
3.1. Small Molecule Sensing
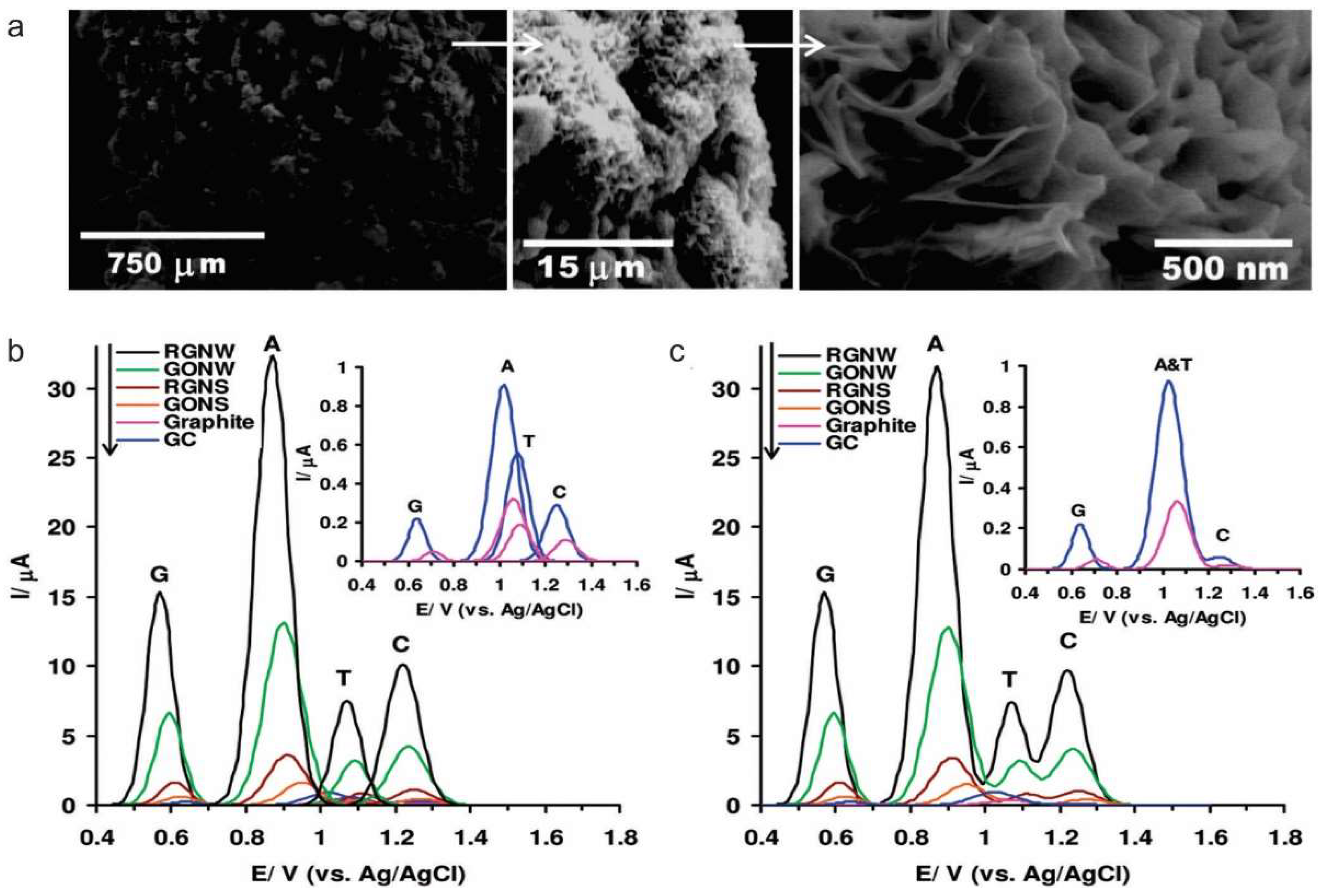
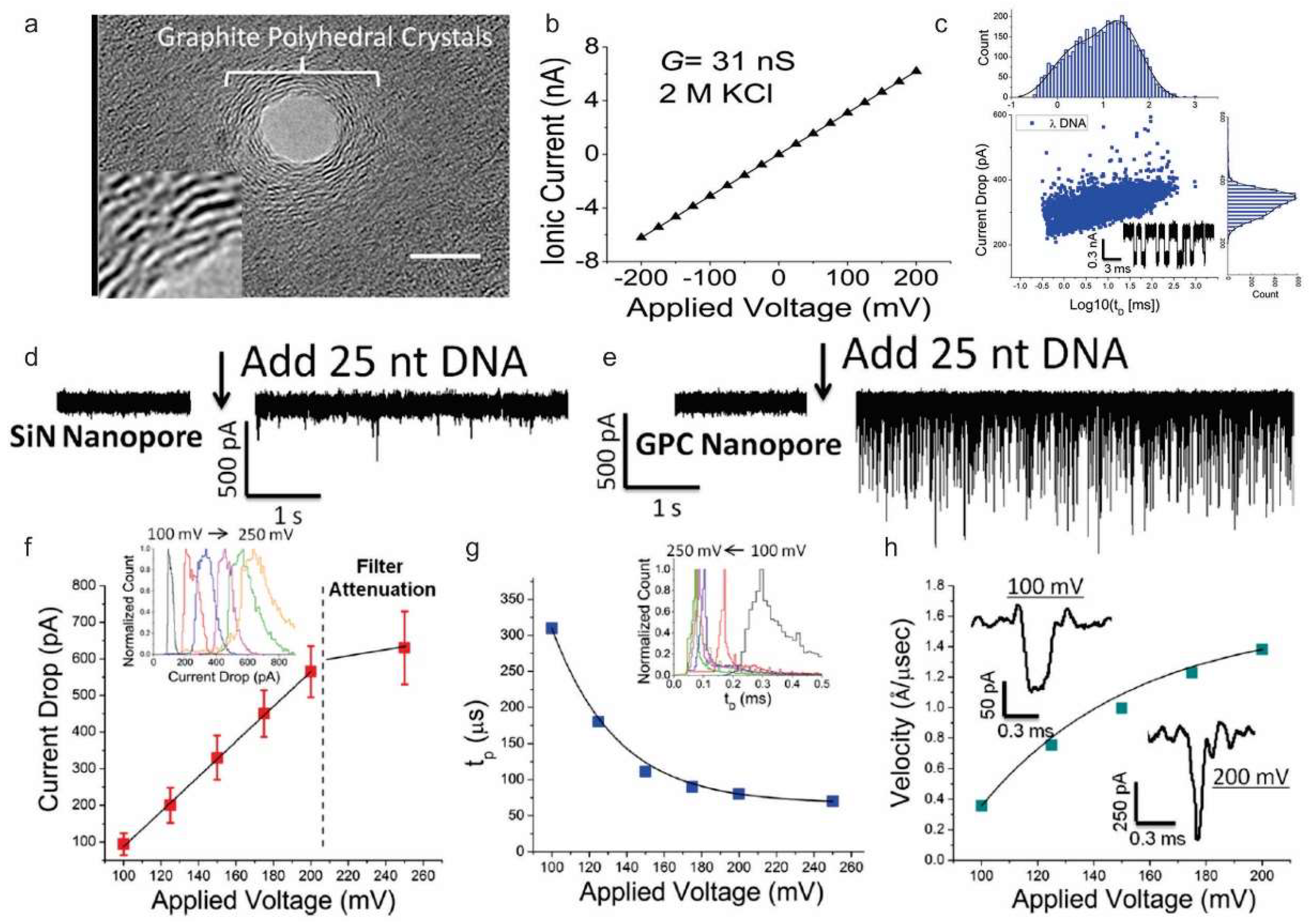
3.2. Nucleic Acid and Protein Sensing
3.3. Live Cell-based Sensing
4. Conclusions and Future Outlook
Funding
Conflicts of Interest
References
- Stoller, M.D.; Park, S.; Zhu, Y.; An, J.; Ruoff, R.S. Graphene-based ultracapacitors. Nano Lett. 2008, 8, 3498–3502. [Google Scholar] [CrossRef] [PubMed]
- Weiss, N.O.; Zhou, H.; Liao, L.; Liu, Y.; Jiang, S.; Huang, Y.; Duan, X. Graphene: An emerging electronic material. Adv. Mater. 2012, 24, 5782–5825. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Georgakilas, V.; Otyepka, M.; Bourlinos, A.B.; Chandra, V.; Kim, N.; Kemp, K.C.; Hobza, P.; Zboril, R.; Kim, K.S. Functionalization of graphene: Covalent and non-covalent approaches, derivatives and applications. Chem. Rev. 2012, 112, 6156–6214. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Luo, Y.; Zhu, C.; Li, H.; Du, D.; Lin, Y. Recent advances in electrochemical biosensors based on graphene two-dimensional nanomaterials. Biosens. Bioelectron. 2016, 76, 195–212. [Google Scholar] [CrossRef] [PubMed]
- Bollella, P.; Fusco, G.; Tortolini, C.; Sanzo, G.; Favero, G.; Gorton, L.; Antiochia, R. Beyond graphene: Electrochemical sensors and biosensors for biomarkers detection. Biosens. Bioelectron. 2017, 89 Pt 1, 152–166. [Google Scholar] [CrossRef]
- Rusling, J.F. Enzyme bioelectrochemistry in cast biomembrane-like films. Acc. Chem. Res. 1998, 31, 363–369. [Google Scholar] [CrossRef]
- Lawal, A.T. Synthesis and utilisation of graphene for fabrication of electrochemical sensors. Talanta 2015, 131, 424–443. [Google Scholar] [CrossRef]
- Pumera, M. Graphene in biosensing. Mater. Today 2011, 14, 308–315. [Google Scholar] [CrossRef]
- Ostrovsky, P.M.; Gornyi, I.V.; Mirlin, A.D. Electron transport in disordered graphene. Phys. Rev. B 2006, 74, 235443. [Google Scholar] [CrossRef]
- Schedin, F.; Geim, A.K.; Morozov, S.V.; Hill, E.W.; Blake, P.; Katsnelson, M.I.; Novoselov, K.S. Detection of individual gas molecules adsorbed on graphene. Nat. Mater. 2007, 6, 652–655. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Huang, P.J.; Liu, B.; Ying, Y.; Liu, J. Comparison of Graphene Oxide and Reduced Graphene Oxide for DNA Adsorption and Sensing. Langmuir 2016, 32, 10776–10783. [Google Scholar] [CrossRef] [PubMed]
- Hwang, M.T.; Landon, P.B.; Lee, J.; Choi, D.; Mo, A.H.; Glinsky, G.; Lal, R. Highly specific SNP detection using 2D graphene electronics and DNA strand displacement. Proc. Natl. Acad. Sci. USA 2016, 113, 7088–7093. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Z.; Barrow, C.J.; Yang, W. Molecularly engineered graphene surfaces for sensing applications: A review. Anal. Chim. Acta 2015, 859, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.H.; Wang, Y.Z.; Hu, S.S. Nanocomposites of graphene and graphene oxides: Synthesis, molecular functionalization and application in electrochemical sensors and biosensors. A review. Microchim. Acta 2017, 184, 1–44. [Google Scholar] [CrossRef]
- Quintana, M.; Spyrou, K.; Grzelczak, M.; Browne, W.R.; Rudolf, P.; Prato, M. Functionalization of graphene via 1,3-dipolar cycloaddition. ACS Nano 2010, 4, 3527–3533. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Wang, E. Electrochemical biosensors on platforms of graphene. Chem. Commun. (Camb.) 2013, 49, 9526–9539. [Google Scholar] [CrossRef]
- Kuila, T.; Bose, S.; Mishra, A.K.; Khanra, P.; Kim, N.H.; Lee, J.H. Chemical functionalization of graphene and its applications. Prog. Mater. Sci. 2012, 57, 1061–1105. [Google Scholar] [CrossRef]
- Srivastava, R.K.; Srivastava, S.; Narayanan, T.N.; Mahlotra, B.D.; Vajtai, R.; Ajayan, P.M.; Srivastava, A. Functionalized multilayered graphene platform for urea sensor. ACS Nano 2012, 6, 168–175. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Fal’ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorso, F.; Lombardo, A.; Hasan, T.; Sun, Z.P.; Colombo, L.; Ferrari, A.C. Production and processing of graphene and 2d crystals. Mater. Today 2012, 15, 564–589. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Narita, A.; Müllen, K. Precision synthesis versus bulk-scale fabrication of graphenes. Nat. Rev. Chem. 2017, 2, 0100. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Katsnelson, M.I.; Grigorieva, I.V.; Dubonos, S.V.; Firsov, A.A. Two-dimensional gas of massless Dirac fermions in graphene. Nature 2005, 438, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Tan, Y.W.; Stormer, H.L.; Kim, P. Experimental observation of the quantum Hall effect and Berry’s phase in graphene. Nature 2005, 438, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Novoselov, K.S.; Jiang, Z.; Zhang, Y.; Morozov, S.V.; Stormer, H.L.; Zeitler, U.; Maan, J.C.; Boebinger, G.S.; Kim, P.; Geim, A.K. Room-temperature quantum Hall effect in graphene. Science 2007, 315, 1379. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.Z.; Thorpe, D.; Adamson, D.H.; Schniepp, H.C. Methods of graphite exfoliation. J. Mater. Chem. 2012, 22, 24992–25002. [Google Scholar] [CrossRef]
- Ciesielski, A.; Samori, P. Graphene via sonication assisted liquid-phase exfoliation. Chem. Soc. Rev. 2014, 43, 381–398. [Google Scholar] [CrossRef]
- Shen, J.; He, Y.; Wu, J.; Gao, C.; Keyshar, K.; Zhang, X.; Yang, Y.; Ye, M.; Vajtai, R.; Lou, J.; et al. Liquid Phase Exfoliation of Two-Dimensional Materials by Directly Probing and Matching Surface Tension Components. Nano Lett. 2015, 15, 5449–5454. [Google Scholar] [CrossRef]
- He, P.; Zhou, C.; Tian, S.; Sun, J.; Yang, S.; Ding, G.; Xie, X.; Jiang, M. Urea-assisted aqueous exfoliation of graphite for obtaining high-quality graphene. Chem. Commun. (Camb.) 2015, 51, 4651–4654. [Google Scholar] [CrossRef] [PubMed]
- Hummers Jr, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Ang, P.K.; Wang, S.; Bao, Q.; Thong, J.T.; Loh, K.P. High-throughput synthesis of graphene by intercalation-exfoliation of graphite oxide and study of ionic screening in graphene transistor. ACS Nano 2009, 3, 3587–3594. [Google Scholar] [CrossRef] [PubMed]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Feng, H.; Li, J. Graphene oxide: Preparation, functionalization, and electrochemical applications. Chem. Rev. 2012, 112, 6027–6053. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Kim, H.; Lee, Y.; Xu, X.; Park, J.S.; Zheng, Y.; Balakrishnan, J.; Lei, T.; Kim, H.R.; Song, Y.I.; et al. Roll-to-roll production of 30-inch graphene films for transparent electrodes. Nat. Nanotechnol. 2010, 5, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Forbeaux, I.; Themlin, J.M.; Debever, J.M. Heteroepitaxial graphite on6H−SiC(0001):Interface formation through conduction-band electronic structure. Phys. Rev. B 1998, 58, 16396–16406. [Google Scholar] [CrossRef]
- Berger, C.; Song, Z.; Li, X.; Wu, X.; Brown, N.; Naud, C.; Mayou, D.; Li, T.; Hass, J.; Marchenkov, A.N.; et al. Electronic confinement and coherence in patterned epitaxial graphene. Science 2006, 312, 1191–1196. [Google Scholar] [CrossRef]
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.H.; Kim, P.; Choi, J.Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710. [Google Scholar] [CrossRef]
- Li, X.; Cai, W.; An, J.; Kim, S.; Nah, J.; Yang, D.; Piner, R.; Velamakanni, A.; Jung, I.; Tutuc, E.; et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 2009, 324, 1312–1314. [Google Scholar] [CrossRef]
- Li, X.; Cai, W.; Colombo, L.; Ruoff, R.S. Evolution of graphene growth on Ni and Cu by carbon isotope labeling. Nano Lett. 2009, 9, 4268–4272. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Ren, W.; Xu, H.; Jin, L.; Wang, Z.; Ma, T.; Ma, L.P.; Zhang, Z.; Fu, Q.; Peng, L.M.; et al. Repeated growth and bubbling transfer of graphene with millimetre-size single-crystal grains using platinum. Nat. Commun. 2012, 3, 699. [Google Scholar] [CrossRef] [PubMed]
- Banszerus, L.; Schmitz, M.; Engels, S.; Dauber, J.; Oellers, M.; Haupt, F.; Watanabe, K.; Taniguchi, T.; Beschoten, B.; Stampfer, C. Ultrahigh-mobility graphene devices from chemical vapor deposition on reusable copper. Sci. Adv. 2015, 1, e1500222. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Schwarzschild, M.A. The epidemiology of Parkinson’s disease: Risk factors and prevention. Lancet Neurol. 2016, 15, 1257–1272. [Google Scholar] [CrossRef]
- Logroscino, G.; Piccininni, M.; Marin, B.; Nichols, E.; Abd-Allah, F.; Abdelalim, A.; Alahdab, F.; Asgedom, S.W.; Awasthi, A.; Chaiah, Y.; et al. Global, regional, and national burden of motor neuron diseases 1990-2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 1083–1097. [Google Scholar] [CrossRef]
- Trounson, A.; DeWitt, N.D. Pluripotent stem cells progressing to the clinic. Nat. Rev. Mol. Cell Biol. 2016, 17, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Fox, I.J.; Daley, G.Q.; Goldman, S.A.; Huard, J.; Kamp, T.J.; Trucco, M. Stem cell therapy. Use of differentiated pluripotent stem cells as replacement therapy for treating disease. Science 2014, 345, 1247391. [Google Scholar] [CrossRef]
- Connolly, B.S.; Lang, A.E. Pharmacological treatment of Parkinson disease: A review. JAMA 2014, 311, 1670–1683. [Google Scholar] [CrossRef]
- Choi, J.H.; Lee, J.H.; Oh, B.K.; Choi, J.W. Localized surface plasmon resonance-based label-free biosensor for highly sensitive detection of dopamine. J. Nanosci. Nanotechnol. 2014, 14, 5658–5661. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, T.; Choi, J.W. Nano-Biosensor for Monitoring the Neural Differentiation of Stem Cells. Nanomaterials (Basel) 2016, 6, 224. [Google Scholar] [CrossRef] [PubMed]
- Ping, J.; Wu, J.; Wang, Y.; Ying, Y. Simultaneous determination of ascorbic acid, dopamine and uric acid using high-performance screen-printed graphene electrode. Biosens. Bioelectron. 2012, 34, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.J.; Lin, Q.Q.; Gong, P.W.; Liu, B.; Wang, J.Q.; Yang, S.R. A new enzymatic immobilization carrier based on graphene capsule for hydrogen peroxide biosensors. Electrochim. Acta 2015, 151, 186–194. [Google Scholar] [CrossRef]
- Panieri, E.; Gogvadze, V.; Norberg, E.; Venkatesh, R.; Orrenius, S.; Zhivotovsky, B. Reactive oxygen species generated in different compartments induce cell death, survival, or senescence. Free Radic. Biol. Med. 2013, 57, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ye, Y.J.; Lu, X.P.; Wu, Y.; Sun, L.L.; Tan, H.L.; Xu, F.G.; Song, Y.H. Prussian blue nanocubes on nitrobenzene-functionalized reduced graphene oxide and its application for H2O2 biosensing. Electrochim. Acta 2013, 114, 223–232. [Google Scholar] [CrossRef]
- Yoon, J.; Lee, T.; Bapurao, G.B.; Jo, J.; Oh, B.K.; Choi, J.W. Electrochemical H2O2 biosensor composed of myoglobin on MoS2 nanoparticle-graphene oxide hybrid structure. Biosens. Bioelectron. 2017, 93, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Ni, Y.; Kokot, S. A novel electrochemical biosensor based on the hemin-graphene nano-sheets and gold nano-particles hybrid film for the analysis of hydrogen peroxide. Anal. Chim. Acta 2013, 788, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Simplicio, J. Hemin monomers in micellar sodium lauryl sulfate. A spectral and equilibrium study with cyanide. Biochemistry 1972, 11, 2525–2528. [Google Scholar] [CrossRef]
- Shao, Y.; Zhang, S.; Engelhard, M.H.; Li, G.; Shao, G.; Wang, Y.; Liu, J.; Aksay, I.A.; Lin, Y. Nitrogen-doped graphene and its electrochemical applications. J. Mater. Chem. 2010, 20, 7491. [Google Scholar] [CrossRef]
- Ensafi, A.A.; Alinajafi, H.A.; Jafari-Asl, M.; Rezaei, B.; Ghazaei, F. Cobalt ferrite nanoparticles decorated on exfoliated graphene oxide, application for amperometric determination of NADH and H2O2. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 60, 276–284. [Google Scholar] [CrossRef]
- Aydogdu Tig, G. Highly sensitive amperometric biosensor for determination of NADH and ethanol based on Au-Ag nanoparticles/poly(L-Cysteine)/reduced graphene oxide nanocomposite. Talanta 2017, 175, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Du, T.; Jiang, H.; Wang, X. Synergistic effect of pyrroloquinoline quinone and graphene nano-interface for facile fabrication of sensitive NADH biosensor. Biosens. Bioelectron. 2017, 89 Pt 1, 422–429. [Google Scholar] [CrossRef]
- Balamurugan, J.; Thanh, T.D.; Kim, N.H.; Lee, J.H. Facile fabrication of FeN nanoparticles/nitrogen-doped graphene core-shell hybrid and its use as a platform for NADH detection in human blood serum. Biosens. Bioelectron. 2016, 83, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Ouyang, X.; Ding, Y.; Luo, L.; Xu, D.; Ning, Y. Electrochemical preparation of nickel and copper oxides-decorated graphene composite for simultaneous determination of dopamine, acetaminophen and tryptophan. Talanta 2016, 146, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Liu, J.; Liu, M.H.; Yu, F.; Zhang, L.; Tang, H.; Ye, B.C.; Lai, L.F. Fabrication of ultra-sensitive and selective dopamine electrochemical sensor based on molecularly imprinted polymer modified graphene@carbon nanotube foam. Electrochem. Commun. 2016, 64, 42–45. [Google Scholar] [CrossRef]
- Thanh, T.D.; Balamurugan, J.; Lee, S.H.; Kim, N.H.; Lee, J.H. Effective seed-assisted synthesis of gold nanoparticles anchored nitrogen-doped graphene for electrochemical detection of glucose and dopamine. Biosens. Bioelectron. 2016, 81, 259–267. [Google Scholar] [CrossRef]
- Thanh, T.D.; Balamurugan, J.; Hwang, J.Y.; Kim, N.H.; Lee, J.H. In situ synthesis of graphene-encapsulated gold nanoparticle hybrid electrodes for non-enzymatic glucose sensing. Carbon 2016, 98, 90–98. [Google Scholar] [CrossRef]
- Chaiyo, S.; Mehmeti, E.; Siangproh, W.; Hoang, T.L.; Nguyen, H.P.; Chailapakul, O.; Kalcher, K. Non-enzymatic electrochemical detection of glucose with a disposable paper-based sensor using a cobalt phthalocyanine-ionic liquid-graphene composite. Biosens. Bioelectron. 2018, 102, 113–120. [Google Scholar] [CrossRef]
- Yang, T.T.; Xu, J.K.; Lu, L.M.; Zhu, X.F.; Gao, Y.S.; Xing, H.K.; Yu, Y.F.; Ding, W.C.; Liu, Z. Copper nanoparticle/graphene oxide/single wall carbon nanotube hybrid materials as electrochemical sensing platform for nonenzymatic glucose detection. J. Electroanal. Chem. 2016, 761, 118–124. [Google Scholar] [CrossRef]
- Hu, Y.; Li, F.; Han, D.; Wu, T.; Zhang, Q.; Niu, L.; Bao, Y. Simple and label-free electrochemical assay for signal-on DNA hybridization directly at undecorated graphene oxide. Anal. Chim. Acta 2012, 753, 82–89. [Google Scholar] [CrossRef]
- Akhavan, O.; Ghaderi, E.; Rahighi, R. Toward single-DNA electrochemical biosensing by graphene nanowalls. ACS Nano 2012, 6, 2904–2916. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, I.; Singh, M.; Pandey, C.M.; Sumana, G. Electrochemical genosensor based on graphene oxide modified iron oxide–chitosan hybrid nanocomposite for pathogen detection. Sens. Actuators B Chem. 2015, 206, 276–283. [Google Scholar] [CrossRef]
- Esteban-Fernandez de Avila, B.; Araque, E.; Campuzano, S.; Pedrero, M.; Dalkiran, B.; Barderas, R.; Villalonga, R.; Kilic, E.; Pingarron, J.M. Dual functional graphene derivative-based electrochemical platforms for detection of the TP53 gene with single nucleotide polymorphism selectivity in biological samples. Anal. Chem. 2015, 87, 2290–2298. [Google Scholar] [CrossRef] [PubMed]
- Freedman, K.J.; Ahn, C.W.; Kim, M.J. Detection of long and short DNA using nanopores with graphitic polyhedral edges. ACS Nano 2013, 7, 5008–5016. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; Huang, J.Y.; Bao, T.; Zhou, J.; Xia, H.X.; Zhang, X.H.; Wang, S.F.; Zhao, Y.D. Increased electrocatalyzed performance through hairpin oligonucleotide aptamer-functionalized gold nanorods labels and graphene-streptavidin nanomatrix: Highly selective and sensitive electrochemical biosensor of carcinoembryonic antigen. Biosens. Bioelectron. 2016, 83, 142–148. [Google Scholar] [CrossRef] [PubMed]
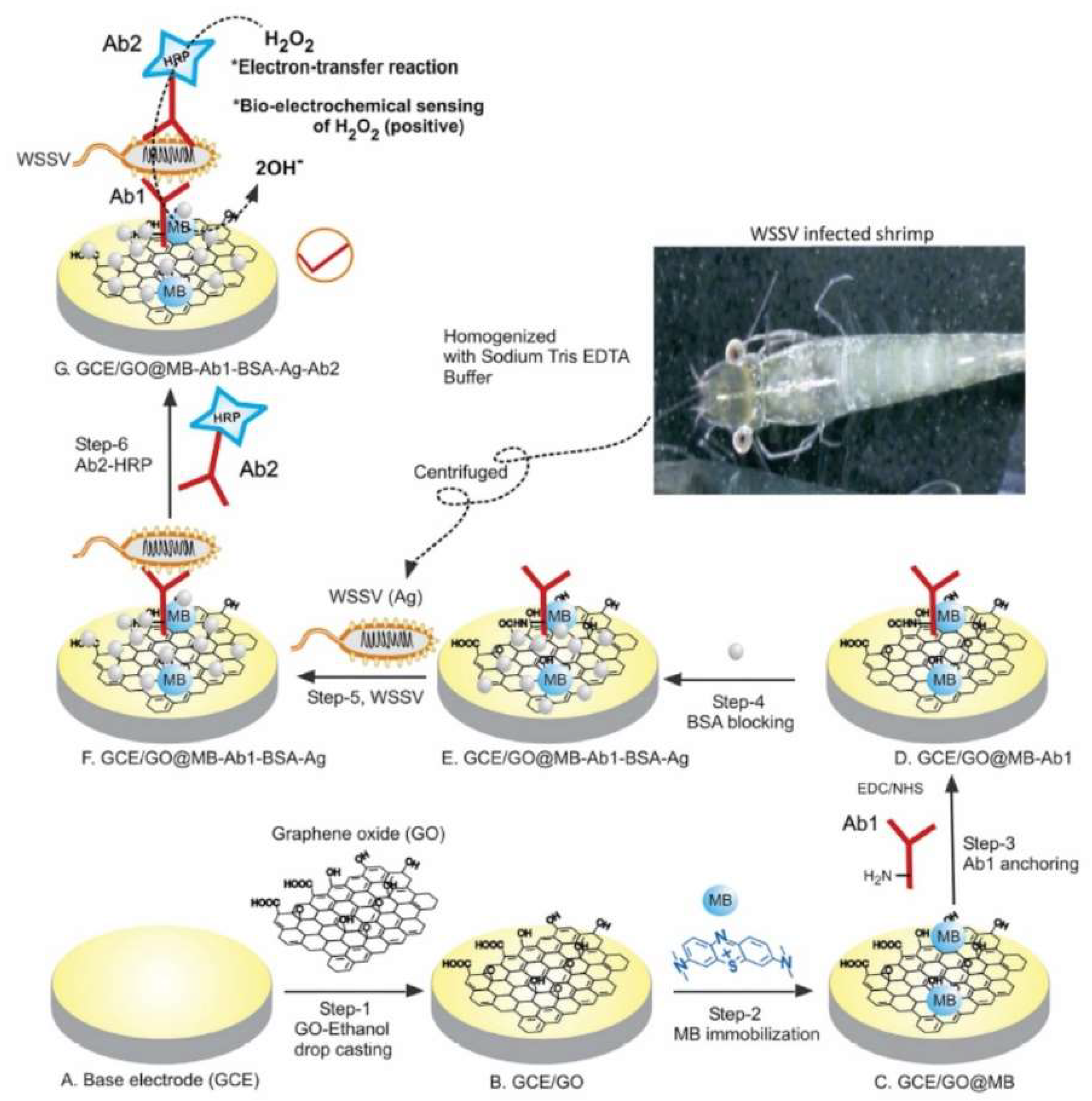
- Natarajan, A.; Devi, K.S.; Raja, S.; Senthil Kumar, A. An Elegant Analysis of White Spot Syndrome Virus Using a Graphene Oxide/Methylene Blue based Electrochemical Immunosensor Platform. Sci. Rep. 2017, 7, 46169. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Hou, C.; Huo, D.; Bao, J.; Fa, H.; Shen, C. An electrochemical DNA biosensor based on nitrogen-doped graphene/Au nanoparticles for human multidrug resistance gene detection. Biosens. Bioelectron. 2016, 85, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Teymourian, H.; Salimi, A.; Khezrian, S. Development of a New Label-free, Indicator-free Strategy toward Ultrasensitive Electrochemical DNA Biosensing Based on Fe3O4 Nanoparticles/Reduced Graphene Oxide Composite. Electroanalysis 2017, 29, 409–414. [Google Scholar] [CrossRef]
- Shahrokhian, S.; Salimian, R. Ultrasensitive detection of cancer biomarkers using conducting polymer/electrochemically reduced graphene oxide-based biosensor: Application toward BRCA1 sensing. Sens. Actuators B-Chem. 2018, 266, 160–169. [Google Scholar] [CrossRef]
- Huang, J.Y.; Zhao, L.; Lei, W.; Wen, W.; Wang, Y.J.; Bao, T.; Xiong, H.Y.; Zhang, X.H.; Wang, S.F. A high-sensitivity electrochemical aptasensor of carcinoembryonic antigen based on graphene quantum dots-ionic liquid-nafion nanomatrix and DNAzyme-assisted signal amplification strategy. Biosens. Bioelectron. 2018, 99, 28–33. [Google Scholar] [CrossRef]
- Ge, L.; Wang, W.; Sun, X.; Hou, T.; Li, F. Affinity-Mediated Homogeneous Electrochemical Aptasensor on a Graphene Platform for Ultrasensitive Biomolecule Detection via Exonuclease-Assisted Target-Analog Recycling Amplification. Anal. Chem. 2016, 88, 2212–2219. [Google Scholar] [CrossRef] [PubMed]
- Pandey, C.M.; Tiwari, I.; Singh, V.N.; Sood, K.N.; Sumana, G.; Malhotra, B.D. Highly sensitive electrochemical immunosensor based on graphene-wrapped copper oxide-cysteine hierarchical structure for detection of pathogenic bacteria. Sens. Actuators B-Chem. 2017, 238, 1060–1069. [Google Scholar] [CrossRef]
- Jijie, R.; Kahlouche, K.; Barras, A.; Yamakawa, N.; Bouckaert, J.; Gharbi, T.; Szunerits, S.; Boukherroub, R. Reduced graphene oxide/polyethylenimine based immunosensor for the selective and sensitive electrochemical detection of uropathogenic Escherichia coli. Sens. Actuators B-Chem. 2018, 260, 255–263. [Google Scholar] [CrossRef]
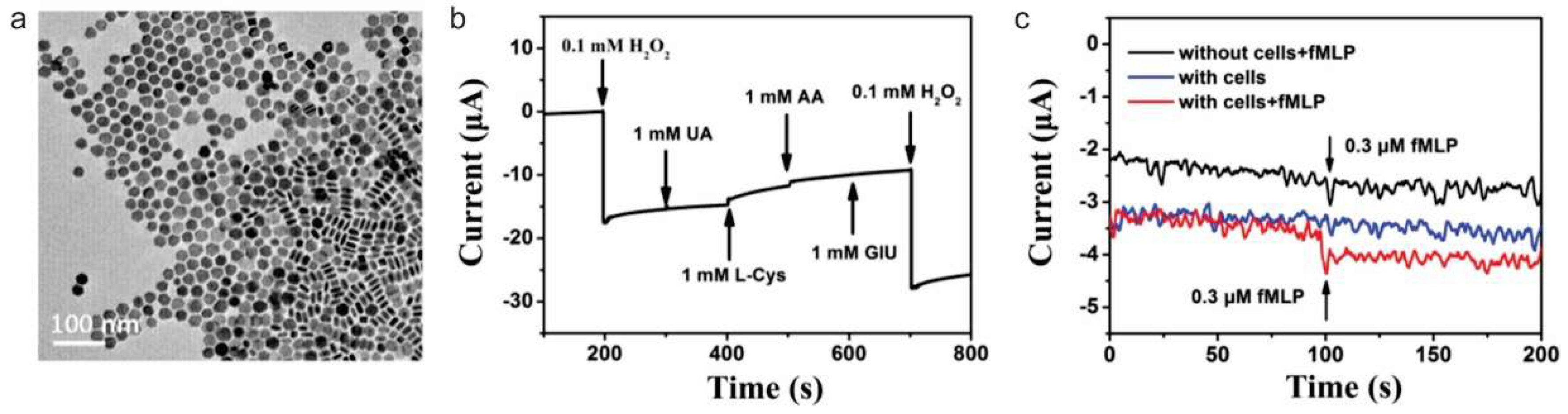
- Wu, P.; Qian, Y.D.; Du, P.; Zhang, H.; Cai, C.X. Facile synthesis of nitrogen-doped graphene for measuring the releasing process of hydrogen peroxide from living cells. J. Mater. Chem. 2012, 22, 6402–6412. [Google Scholar] [CrossRef]
- Sun, Y.; Luo, M.; Meng, X.; Xiang, J.; Wang, L.; Ren, Q.; Guo, S. Graphene/Intermetallic PtPb Nanoplates Composites for Boosting Electrochemical Detection of H2O2 Released from Cells. Anal. Chem. 2017, 89, 3761–3767. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xiao, J.; Lv, Q.; Wang, L.; Dong, X.; Asif, M.; Ren, J.; He, W.; Sun, Y.; Xiao, F.; et al. In Situ Electrochemical Sensing and Real-Time Monitoring Live Cells Based on Freestanding Nanohybrid Paper Electrode Assembled from 3D Functionalized Graphene Framework. ACS Appl. Mater. Interfaces 2017, 9, 38201–38210. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, X.; Guo, Z.; Hu, Z.; Xue, Z.; Lu, X. Horseradish peroxidase supported on porous graphene as a novel sensing platform for detection of hydrogen peroxide in living cells sensitively. Biosens. Bioelectron. 2017, 87, 101–107. [Google Scholar] [CrossRef]
- Li, J.; Xie, J.; Gao, L.; Li, C.M. Au nanoparticles-3D graphene hydrogel nanocomposite to boost synergistically in situ detection sensitivity toward cell-released nitric oxide. ACS Appl. Mater. Interfaces 2015, 7, 2726–2734. [Google Scholar] [CrossRef]
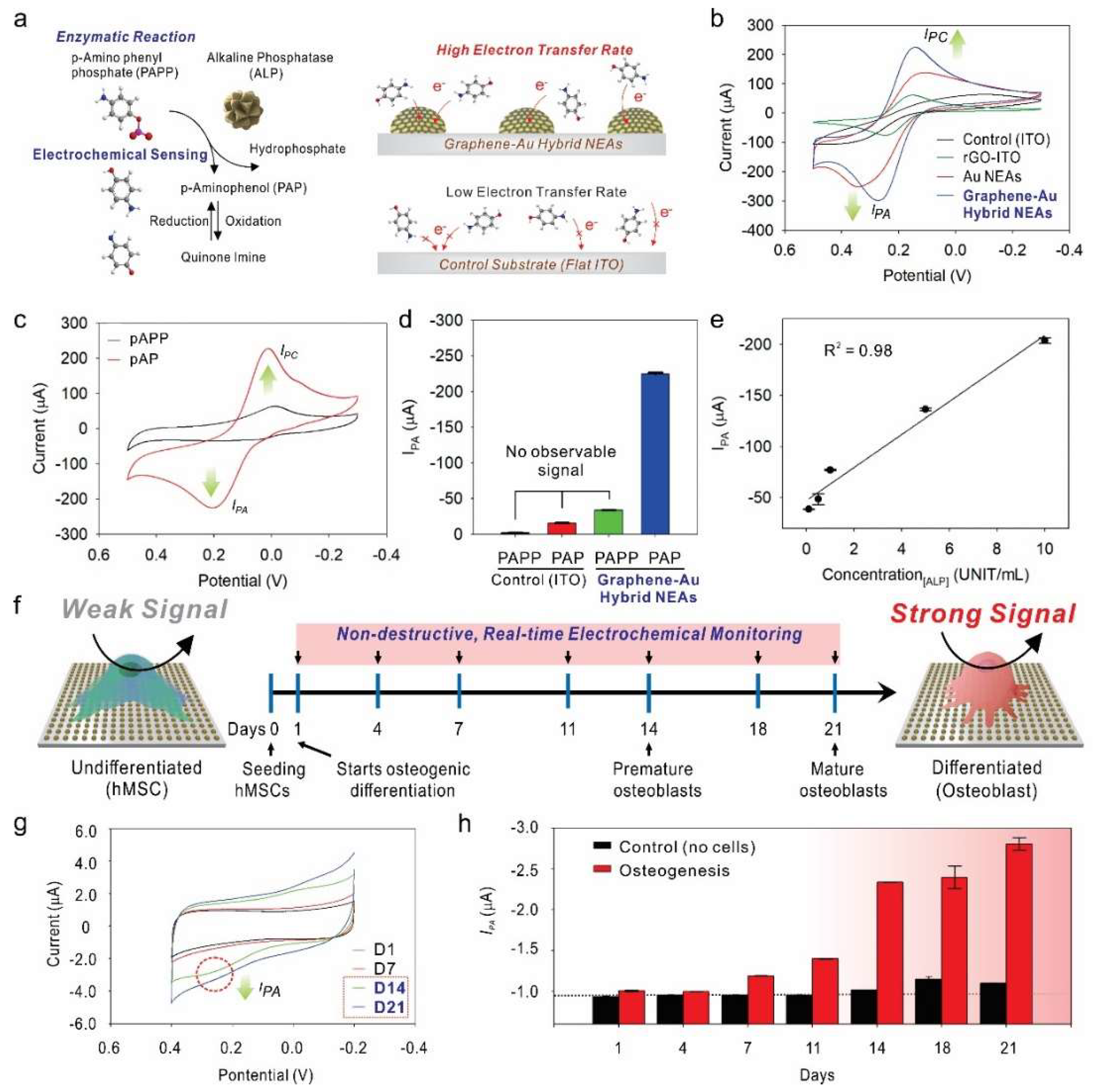
- Lee, J.H.; Choi, H.K.; Yang, L.; Chueng, S.D.; Choi, J.W.; Lee, K.B. Nondestructive Real-Time Monitoring of Enhanced Stem Cell Differentiation Using a Graphene-Au Hybrid Nanoelectrode Array. Adv. Mater. 2018, 30, e1802762. [Google Scholar] [CrossRef]
- Rathnam, C.; Chueng, S.D.; Yang, L.; Lee, K.B. Advanced Gene Manipulation Methods for Stem Cell Theranostics. Theranostics 2017, 7, 2775–2793. [Google Scholar] [CrossRef]
- Zhang, Y.; Bai, X.; Wang, X.; Shiu, K.K.; Zhu, Y.; Jiang, H. Highly sensitive graphene-Pt nanocomposites amperometric biosensor and its application in living cell H2O2 detection. Anal. Chem. 2014, 86, 9459–9465. [Google Scholar] [CrossRef] [PubMed]
- Dou, B.; Li, J.; Jiang, B.; Yuan, R.; Xiang, Y. DNA-Templated In Situ Synthesis of Highly Dispersed AuNPs on Nitrogen-Doped Graphene for Real-Time Electrochemical Monitoring of Nitric Oxide Released from Live Cancer Cells. Anal. Chem. 2019, 91, 2273–2278. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Liao, C.; Liu, Y.; Ye, B.C.; Liu, B. Iron Phthalocyanine Decorated Nitrogen-Doped Graphene Biosensing Platform for Real-Time Detection of Nitric Oxide Released from Living Cells. Anal. Chem. 2018, 90, 4438–4444. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.B.; Liu, Y.L.; Wang, W.J.; Zhang, H.W.; Qin, Y.; Guo, S.; Zhang, X.W.; Fu, L.; Huang, W.H. Biomimetic Graphene-Based 3D Scaffold for Long-Term Cell Culture and Real-Time Electrochemical Monitoring. Anal. Chem. 2018, 90, 1136–1141. [Google Scholar] [CrossRef] [PubMed]
- Zan, X.; Bai, H.; Wang, C.; Zhao, F.; Duan, H. Graphene Paper Decorated with a 2D Array of Dendritic Platinum Nanoparticles for Ultrasensitive Electrochemical Detection of Dopamine Secreted by Live Cells. Chemistry 2016, 22, 5204–5210. [Google Scholar] [CrossRef] [PubMed]
- Asif, M.; Aziz, A.; Wang, H.; Wang, Z.; Wang, W.; Ajmal, M.; Xiao, F.; Chen, X.; Liu, H. Superlattice stacking by hybridizing layered double hydroxide nanosheets with layers of reduced graphene oxide for electrochemical simultaneous determination of dopamine, uric acid and ascorbic acid. Mikrochim. Acta 2019, 186, 61. [Google Scholar] [CrossRef] [PubMed]
- Qi, M.; Zhang, Y.; Cao, C.; Zhang, M.; Liu, S.; Liu, G. Decoration of Reduced Graphene Oxide Nanosheets with Aryldiazonium Salts and Gold Nanoparticles toward a Label-Free Amperometric Immunosensor for Detecting Cytokine Tumor Necrosis Factor-alpha in Live Cells. Anal. Chem. 2016, 88, 9614–9621. [Google Scholar] [CrossRef] [PubMed]
| Physicochemical Property | Estimated Value | Ref. |
|---|---|---|
| High surface area | ~2630 m2g−1 | [1] |
| Excellent electrical conductivity | ~1738 siemens/m | [2] |
| Strong mechanical strength | Young’ Modulus ~1100 GPa, Fracture strength ~125 GPa | [3] |
| Thermal conductivity | 5000 Wm−1K−1 | [4] |
| Ease of functionalization | π–π stacking interaction Electrostatic interaction | [5] |
| Electrode Materials | Target | Linear Range | Detection Limit | Ref. |
|---|---|---|---|---|
| Graphene capsule/horseradish peroxidase | H2O2 | 0.01–12 mM | 3.3 μM | [53] |
| Prussian blue nanocubes/nitrobenzene/reduced graphene oxide | H2O2 | 1.2 μM–15.25 mM | 0.4 μM | [55] |
| Myoglobin (Mb)/MoS2 nanoparticle/graphene oxide | H2O2 | - | 20 nM | [56] |
| Hemin porphyrin/graphene/gold nanoparticle | H2O2 | 0.3 μM–1.8 mM | 0.11 μM | [57] |
| Cobalt ferrite nanoparticles decorated exfoliated graphene oxide | H2O2 NADH | 0.9–900 μM 0.50–100 μM | 0.54 μM 0.38 μM | [60] |
| Au-Ag nanoparticles/poly(L-Cysteine)/reduced graphene oxide | NADH ethanol | 0.083 µM–1.05 mM 0.017 µM–1.845 mM | 9.0 nM 5.0 µM | [61] |
| Graphene-pyrroloquinoline quinone | NADH | 0.32 µM–220 µM | 0.16 µM | [62] |
| FeN nanoparticles/nitrogen-doped graphene core-shell | NADH | 0.4 µM–718 μM | 25 nM | [63] |
| Screen-printed graphene | Dopamine Ascorbic acid Uric acid | 0.5 µM–2000 μM 4.0 µM–4500 μM 0.8 µM–2500 μM | 0.12 μM 0.95 μM 0.20 μM | [52] |
| Nickel and copper oxides-decorated graphene | Dopamine | 0.5 µM–20 μM | 0.17 μM | [64] |
| Molecularly imprinted polymer modified graphene/carbon nanotube | Dopamine | 2.0 fM–1.0 pM | 667 aM | [65] |
| Gold nanoparticle-anchored nitrogen-doped graphene | Dopamine glucose | 30 nM–48 μM 40 μM–16.1 mM | 10 nM 12 μM | [66] |
| Graphene-encapsulated gold nanoparticle | glucose | 6 μM–28.5 mM | 1 μM | [67] |
| Cobalt phthalocyanine–ionic liquid–graphene | glucose | 0.01–1.3 mM and 1.3–5.0 mM | 0.67 µM | [68] |
| Copper nanoparticle/graphene oxide/single wall carbon nanotube | glucose | 1 μM–4.538 mM | 0.34 μM | [69] |
| Electrode Materials | Target | Linear Range | Detection Limit | Ref. |
|---|---|---|---|---|
| Graphene Oxide/probe DNA | HIV-1 gene (cDNA) | 1 pM–1 μM | 0.11 pM | [70] |
| Reduced graphene nanowalls | dsDNA | 0.1 fM–10 mM | 9.4 zM | [71] |
| Graphene oxide modified iron Oxide/chitosan/probe DNA | Escherichia coli Ο157:¨Η7 gene (cDNA) | 10 fM–1 μM | 10 fM | [72] |
| Screen-printed carbon/reduced graphene oxide/Carboxy-methyl-cellulose/probe DNA | p53 tumor suppressor gene (cDNA) | 10 nM–0.1 μM | 2.9 nM | [73] |
| Nitrogen-doped graphene/Au nanoparticles/probe DNA | multidrug resistance gene | 10 fM–100 nM | 3.12 fM | [77] |
| Fe3O4 Nanoparticles/reduced graphene oxide | HIV-1 gene (cDNA) | 10 aM–100 pM | - | [78] |
| Glassy carbon/reduced graphene oxide/polypyrrole–3–carboxylic acid | Breast cancer 1 gene | 1 pM–0.1 μM | 0.3 pM | [79] |
| Gold nanorods/graphene/ hairpin-shaped DNA aptamer | Carcinoembryonic antigen | 5 pg·mL−1–50 ng·mL−1 | 1.5 pg·mL−1 | [75] |
| Graphene quantum dot-ionic liquid-nafion/hairpin aptamer | Carcinoembryonic antigen | 0.5 fg·mL−1–0.5 ng mL−1 | 0.34 fg·mL−1 | [80] |
| Graphene/glassy carbon/aptamer | Carcinoembryonic antigen | 80 ag·mL−1–950 fg·mL−1 | 80 ag·mL−1 | [81] |
| Glassy carbon/graphene oxide methylene blue/Antibody | White spot syndrome virus | 1.36 × 10−3–107 copies·µL−1 | 103 copies·µL−1 | [76] |
| Graphene-wrapped copper oxide/cysteine | E. coli O157:H7 | 10 CFU·mL−1–108 CFU·mL−1 | 3.8 CFU·mL−1 | [82] |
| Gold/reduced graphene oxide/polyethylenimine | E. coli | 10 CFU·mL−1–104 CFU·mL−1 | 10 CFU·mL−1 | [83] |
| Electrode Materials | Target | Linear Range | Detection Limit | Ref. |
|---|---|---|---|---|
| Nitrogen doped graphene | H2O2 | 0.5 μM–1.2 mM | 0.05 μM | [84] |
| Graphene/PtPb-nanoplate | H2O2 | 2 nM–2516 μM | 2 nM | [85] |
| Gold nanoflowers modified ionic liquid functionalized graphene framework | H2O2 | 0.5 μM–2.3 mM | 100 nM | [86] |
| HRP supported Porous graphene | H2O2 | 2.77 μM –835 μM | 26.7 pM | [87] |
| Graphene-Pt nanocomposites | H2O2 | 0.5 μM–0.475 mM | 0.2 μM | [91] |
| GNP deposited 3D graphene hydrogel | NO | 200 nM –6 μM | 9 nM | [88] |
| GNP/calf thymus DNA/nitrogen-doped graphene | NO | 2 nM–500 nM | 0.8 nM | [92] |
| Iron phthalocyanine decorated nitrogen-doped graphene on ITO | NO | 0.18 μM–400 μM | 0.18 μM | [93] |
| 3-aminophenylboronic acid functionalized graphene foam network | H2S | 0.2 μM–10 μM | 50 nM | [94] |
| Dendritic Pt nanoparticles decorated freestanding graphene paper | DA | 87 nM–100 μM | 5 nM | [95] |
| Zn-NiAl layered double hydroxide on reduced graphene oxide | DA | 1 nM–1 μM | 0.1 nM | [96] |
| Aryldiazonium Salts and GNP decorated reduced graphene oxide | TNF-α | 0.1–150 pg·mL−1 | 0.1 pg·mL−1 | [97] |
| Graphene-Au hybrid nanoelectrode array | ALP | 0.1–10 unit·mL−1 | 0.03 unit·mL−1 | [89] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.-H.; Park, S.-J.; Choi, J.-W. Electrical Property of Graphene and Its Application to Electrochemical Biosensing. Nanomaterials 2019, 9, 297. https://doi.org/10.3390/nano9020297
Lee J-H, Park S-J, Choi J-W. Electrical Property of Graphene and Its Application to Electrochemical Biosensing. Nanomaterials. 2019; 9(2):297. https://doi.org/10.3390/nano9020297
Chicago/Turabian StyleLee, Jin-Ho, Soo-Jeong Park, and Jeong-Woo Choi. 2019. "Electrical Property of Graphene and Its Application to Electrochemical Biosensing" Nanomaterials 9, no. 2: 297. https://doi.org/10.3390/nano9020297
APA StyleLee, J.-H., Park, S.-J., & Choi, J.-W. (2019). Electrical Property of Graphene and Its Application to Electrochemical Biosensing. Nanomaterials, 9(2), 297. https://doi.org/10.3390/nano9020297







