Electrochemical Characterization and Inhibiting Mechanism on Calcium Leaching of Graphene Oxide Reinforced Cement Composites
Abstract
1. Introduction
2. Materials and Methods
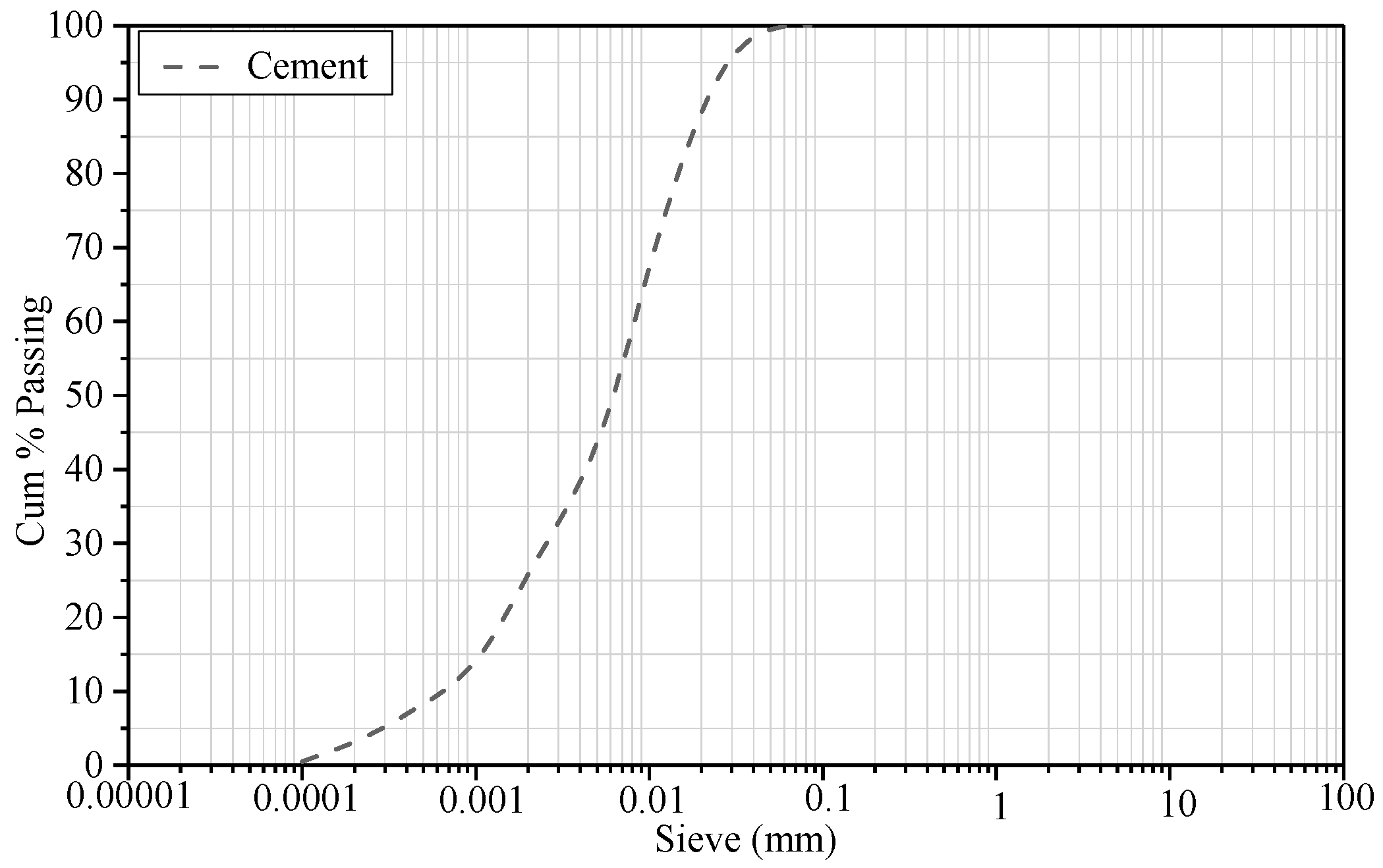
2.1. Raw Materials
2.2. Test Methods
2.2.1. Preparation and Characterization of GO Solution
2.2.2. Mix Proportioning and Sample Preparation
2.2.3. Leaching Solution Selection
2.2.4. Leaching Test
2.2.5. Leaching Depth Test
2.2.6. Compressive Strength Test
2.2.7. Electrochemical Impedance Spectroscopy (EIS) Measurement
2.2.8. Microstructural Analysis
3. Results and Discussion
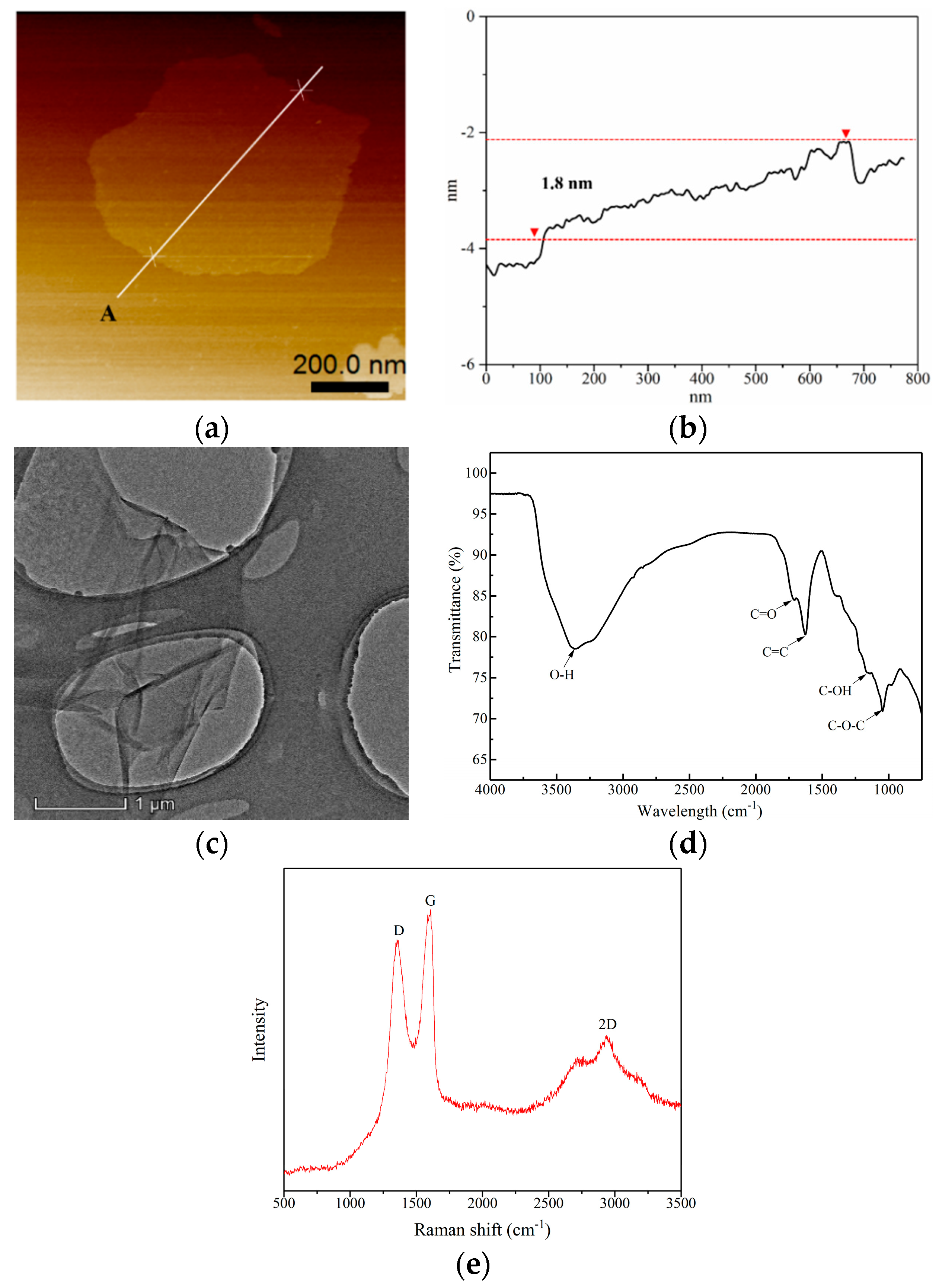
3.1. GO Characterization
3.2. Macroscopic Characterizations for Leached GO/Cement Composites
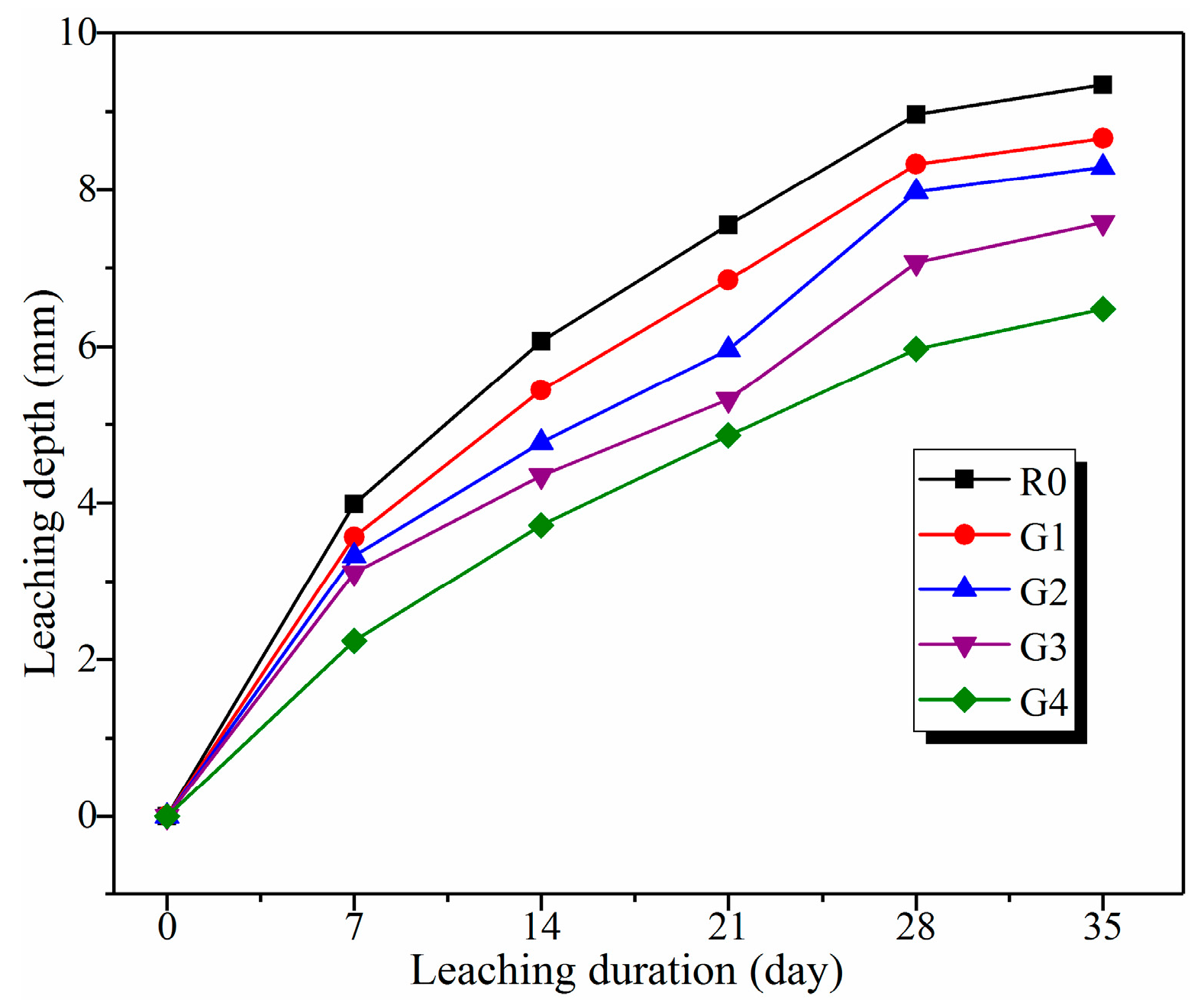
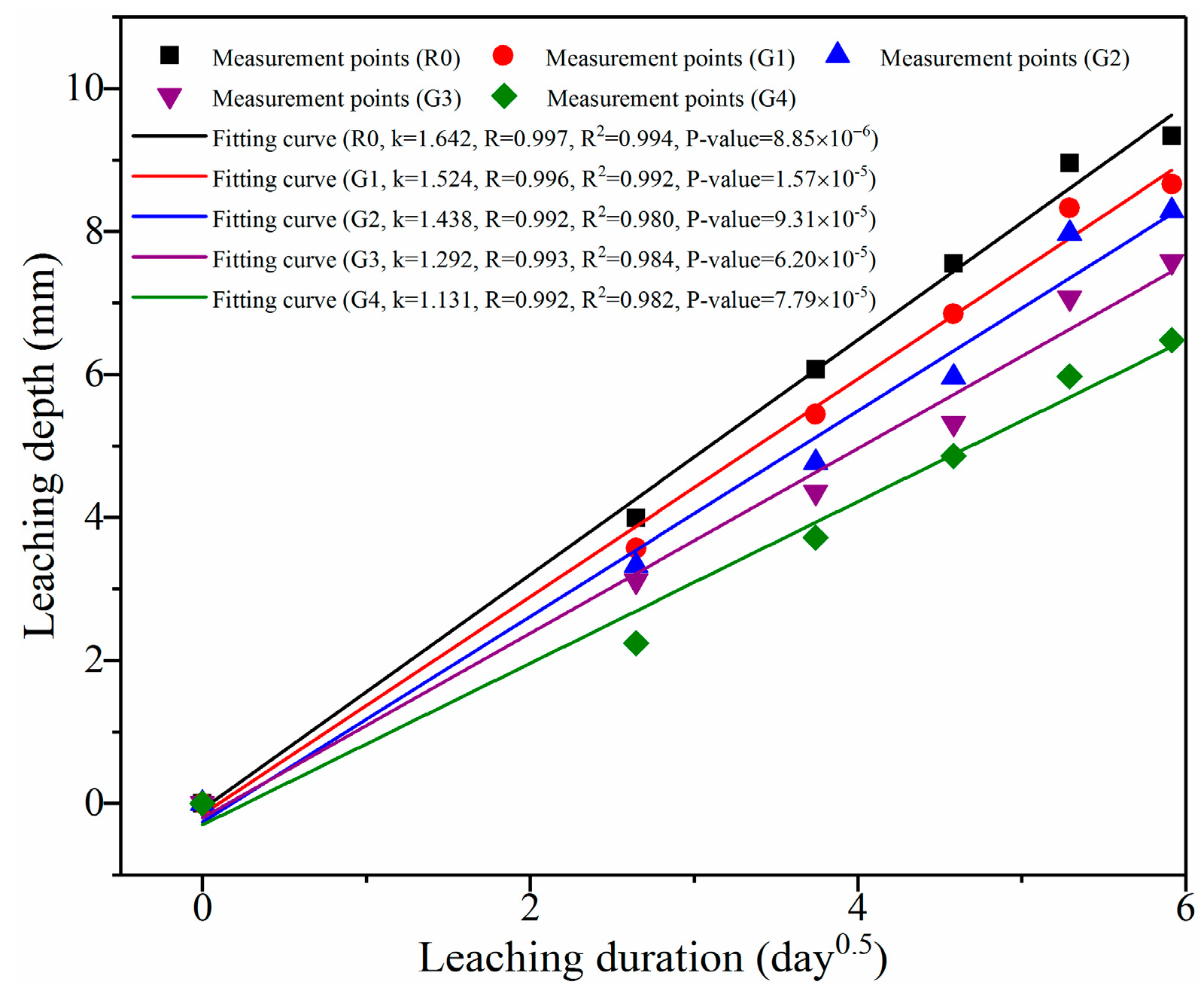
3.2.1. Leaching Depth Analysis
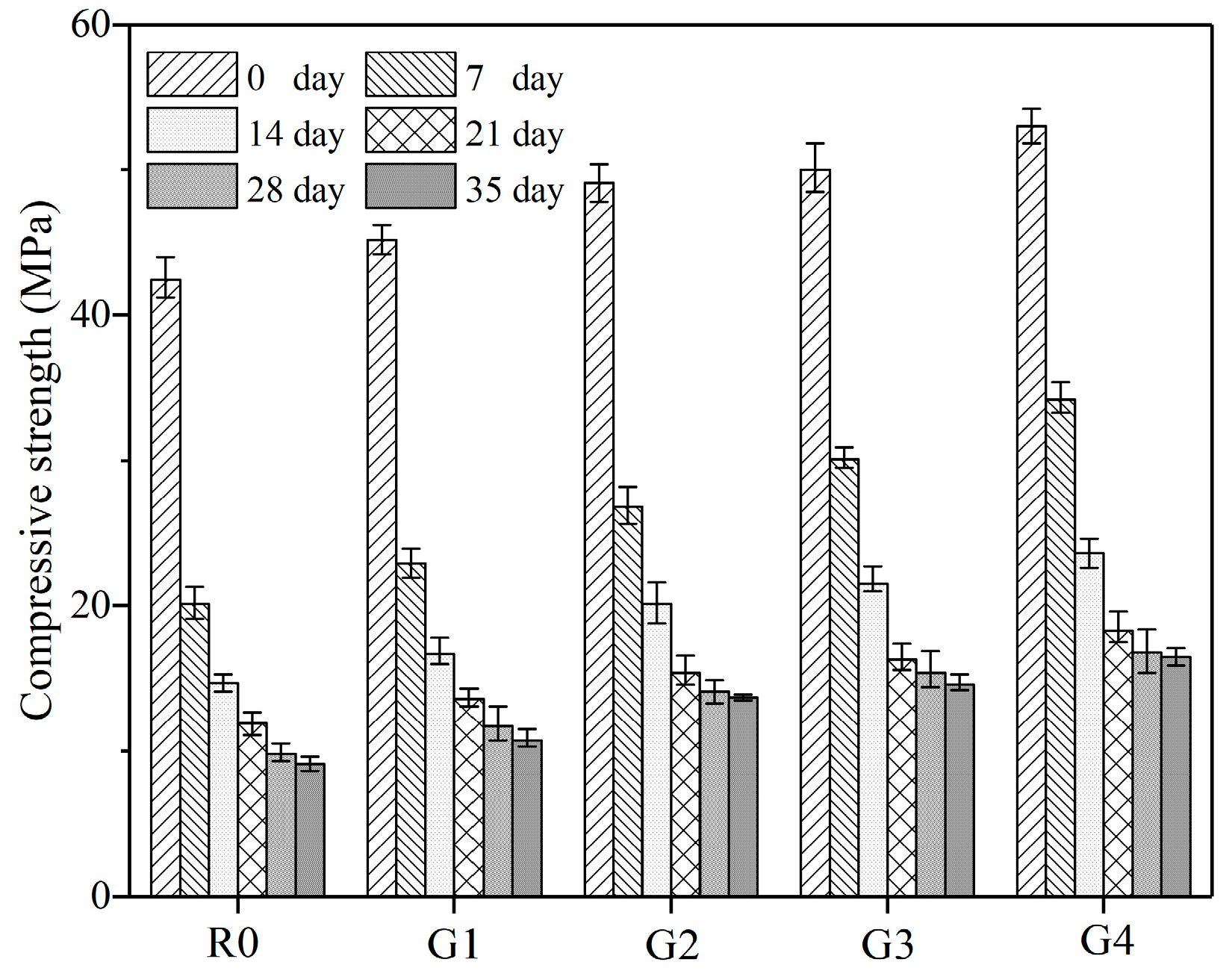
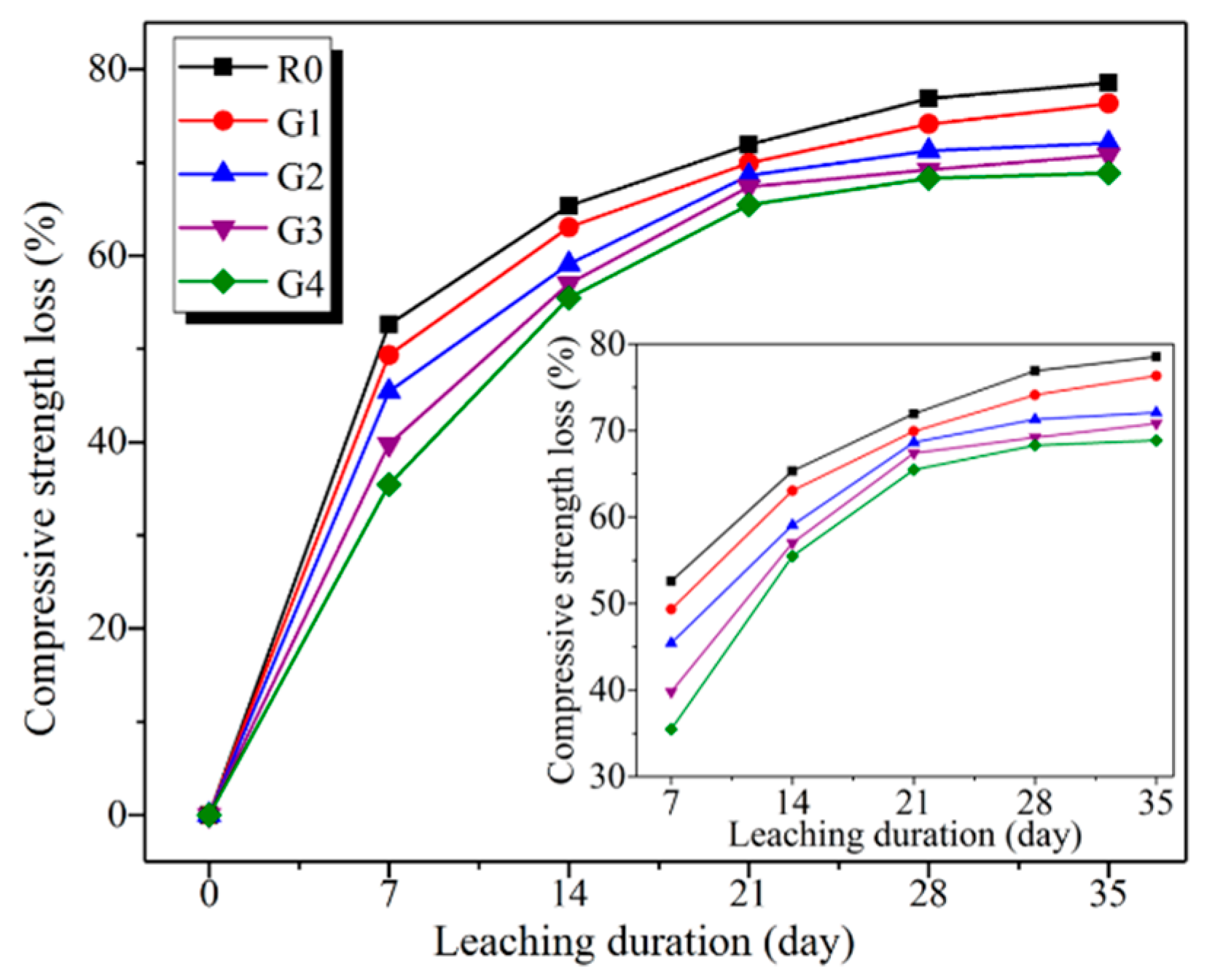
3.2.2. Compressive Strength Analysis
3.3. Electrochemical Characterizations for Leached GO/Cement Composites
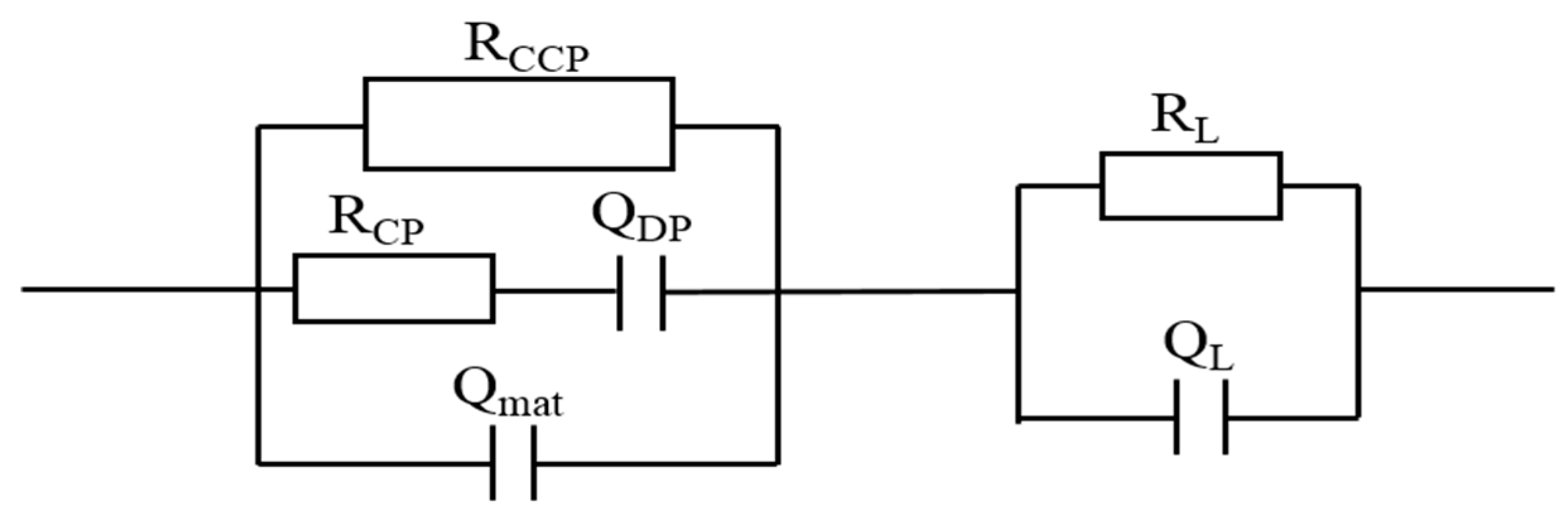
3.3.1. Modifications of Equivalent Circuit Model for Leached Samples
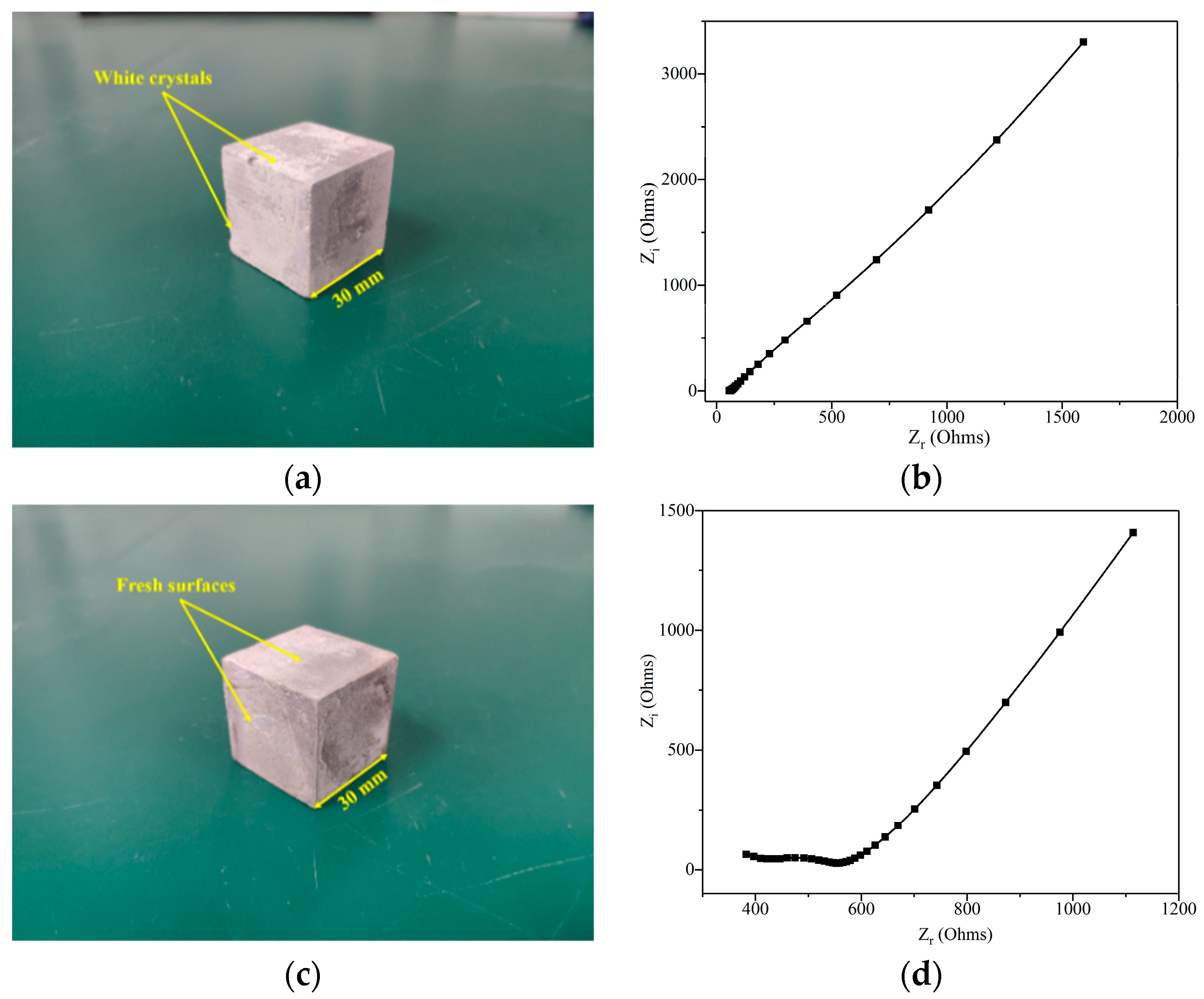
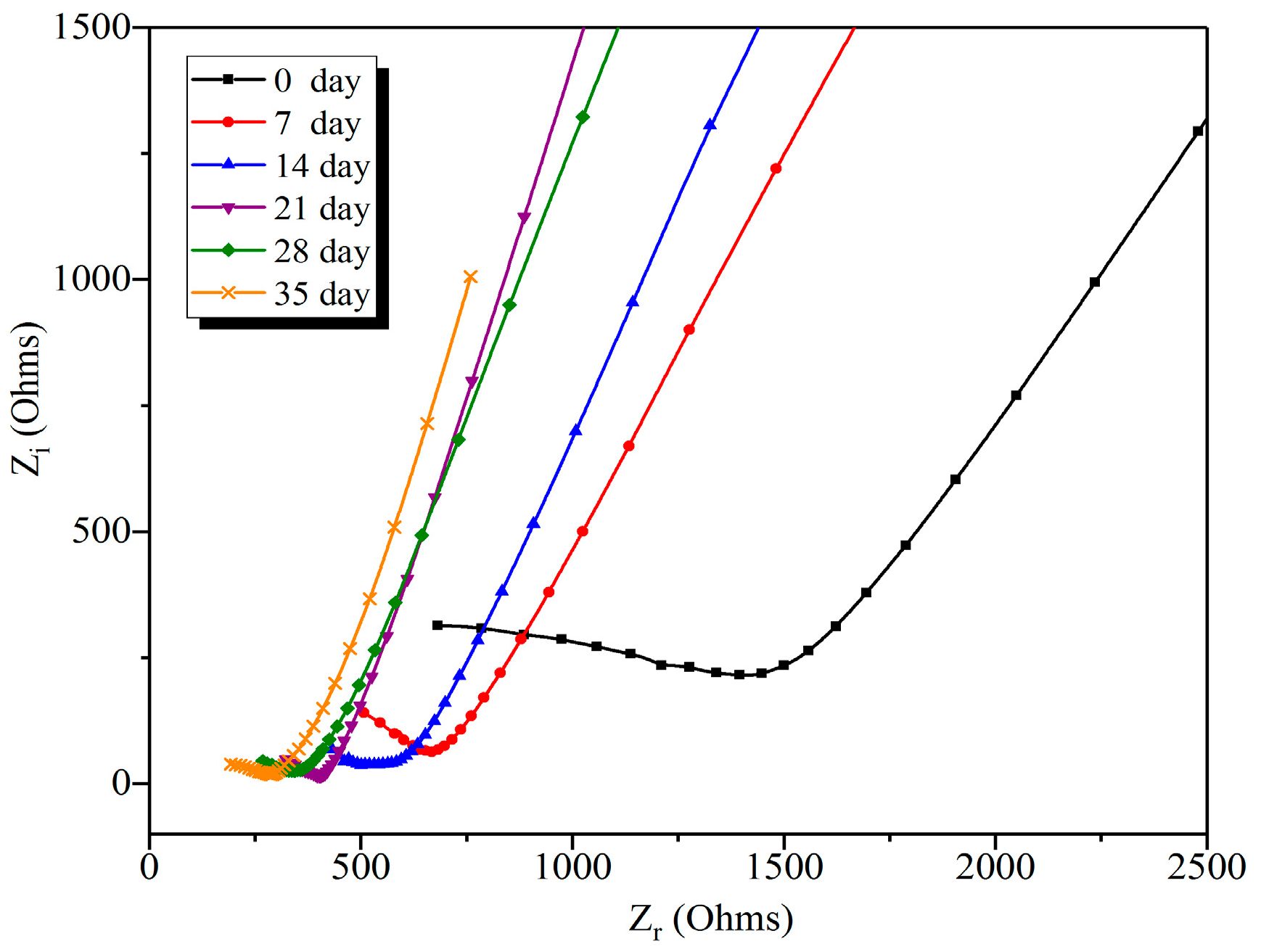
3.3.2. Effect of Leaching Duration on the Nyquist Curve of Leached Samples
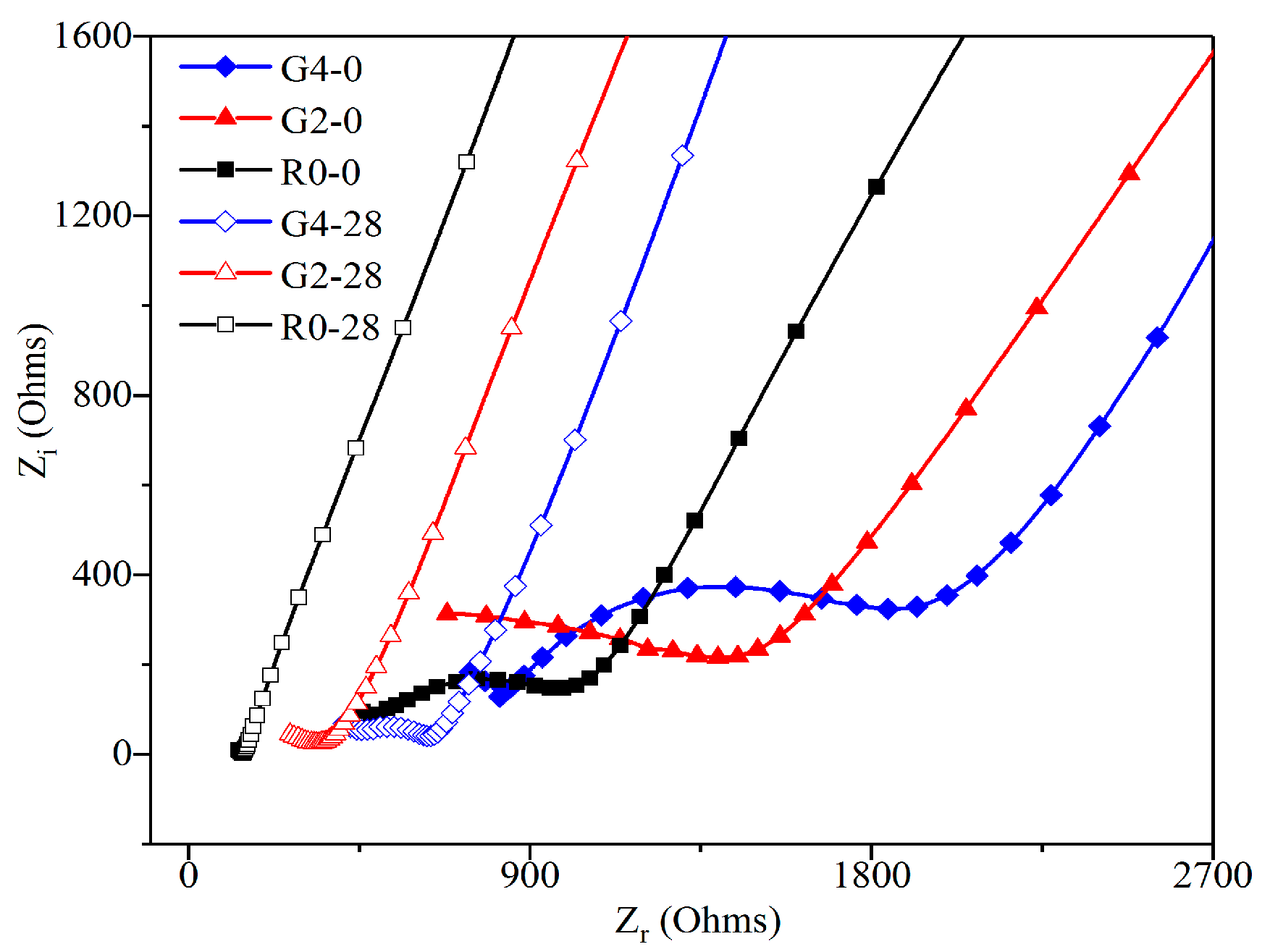
3.3.3. Effect of GO Addition on the Nyquist Curve of Leached Samples
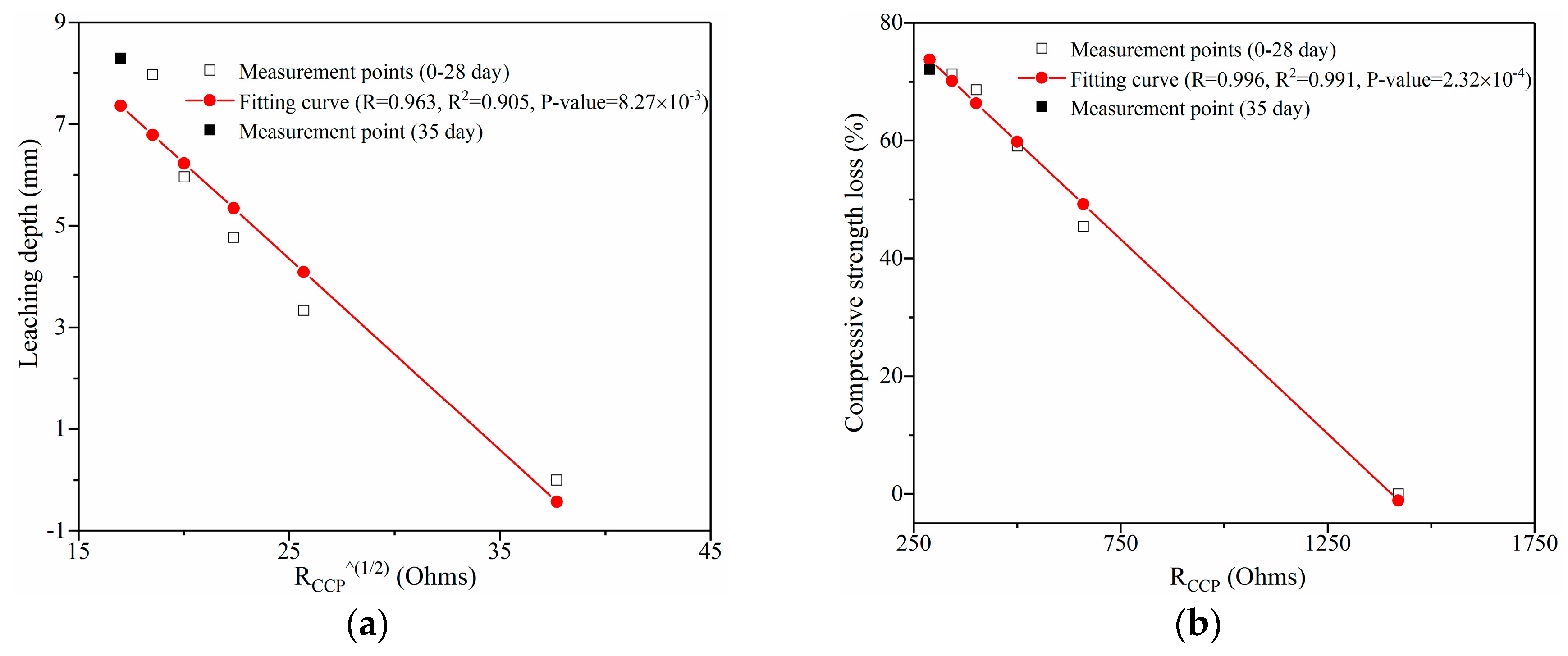
3.3.4. Model Impedance RCCP Analysis and its Fitting Results
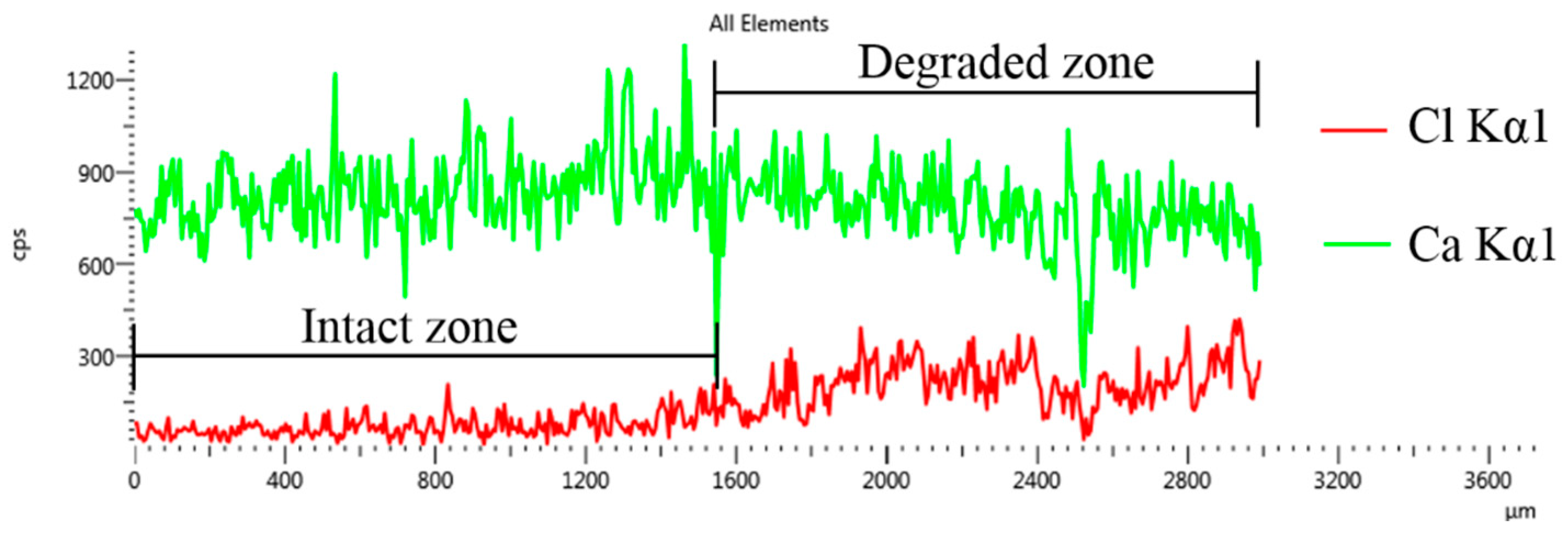
3.4. Microstructural Characterization for Leached GO/Cement Composites
3.5. Inhibiting Mechanism of GO on Calcium Leaching of Cement Composites
4. Conclusions
- Leaching depth of the samples decreased with the increase of GO content. After leaching for 35 days, leaching depths of R0, G1, G2, G3, and G4 were 9.34, 8.66, 8.29, 7.58, and 6.48 mm, respectively. Furthermore, the leaching coefficient of R0 was above 1.45 times than that of G4. These results demonstrate that GO addition can effectively mitigate calcium leaching of cement composites.
- After leaching for 35 days, the compressive strength loss of R0, G1, G2, G3, and G4 was 78.54%, 76.33%, 72.10%, 70.80%, and 68.87%, respectively. In particular, the rapid loss of compressive strength at early age was attributed to the additional capillary pores produced by the dissolution of calcium hydroxide.
- A modified circuit model was proposed to explain the impedance response of the samples exposed to calcium leaching. After leaching for 35 days, loss rate of model impedance RCCP of leached samples with 0, 0.05, 0.1, 0.15, and 0.2 wt.% GO addition was 94.85%, 84.07%, 79.66%, 75.34%, and 68.75%, respectively. In addition, coupling leaching depth and compressive strength loss were accurately predicted by the model parameter RCCP, which indicated that EIS method can be applied to monitor the performance change of the leached infrastructure incorporating GO.
- The leached samples can be divided into two regions: the degraded zone and the intact zone, which is consistent with the model. After leaching for 14 days, the number of leaching products and micro-pores in the degraded zone of the samples decreased with the increase of GO content, indicating that GO addition can refine the microstructure of leached cement composites.
- GO addition can significantly mitigate calcium leaching of cement composites. This is due to the fact that GO can absorb Ca2+ ions in cement pore solution, as well as improve the microstructure of GO/cement composites.
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Joshaghani, A.; Balapour, M.; Ramezanianpour, A.A. Effect of controlled environmental conditions on mechanical, microstructural and durability properties of cement mortar. Constr. Build. Mater. 2018, 164, 134–149. [Google Scholar] [CrossRef]
- Ortega, J.M.; Sánchez, I.; Climent, M.A. Impedance spectroscopy study of the effect of environmental conditions in the microstructure development of OPC and slag cement mortars. Arch. Civ. Mech. Eng. 2017, 10, 569–583. [Google Scholar] [CrossRef]
- Ramezanianpour, A.A. Effect of curing on the compressive strength, resistance to chloride-ion penetration and porosity of concretes incorporating slag, fly ash or silica fume. Cem. Concr. Compos. 1995, 17, 125–133. [Google Scholar] [CrossRef]
- Williams, M.; Ortega, J.; Sánchez, I.; Cabeza, M.; Climent, M. Non-Destructive Study of the Microstructural Effects of Sodium and Magnesium Sulphate Attack on Mortars Containing Silica Fume Using Impedance Spectroscopy. Appl. Sci. 2017, 7, 648. [Google Scholar] [CrossRef]
- Liu, L.; Wang, X.; Zhou, J.; Chu, H.; Shen, D.; Chen, H.; Qin, S. Investigation of pore structure and mechanical property of cement paste subjected to the coupled action of freezing/thawing and calcium leaching. Cem. Concr. Res. 2018, 109, 133–146. [Google Scholar] [CrossRef]
- Forster, A.M.; Szadurski, E.M.; Banfill, P.F.G. Deterioration of natural hydraulic lime mortars, I: Effects of chemically accelerated leaching on physical and mechanical properties of uncarbonated materials. Constr. Build. Mater. 2014, 72, 199–207. [Google Scholar] [CrossRef]
- Tang, Y.J.; Zuo, X.B.; He, S.L.; Ayinde, O.; Yin, G.J. Influence of slag content and water-binder ratio on leaching behavior of cement pastes. Constr. Build. Mater. 2016, 129, 61–69. [Google Scholar] [CrossRef]
- Mainguy, M.; Tognazzi, C.; Torrenti, J.M.; Adenot, F. Modelling of leaching in pure cement paste and mortar. Cem. Concr. Res. 2000, 30, 83–90. [Google Scholar] [CrossRef]
- Faucon, P.; Bescop, P.L.; Adenot, F.; Bonville, P.; Jacquinot, J.F.; Pineau, F.; Felix, B. Leaching of cement: Study of the surface layer. Cem. Concr. Res. 1996, 26, 1707–1715. [Google Scholar] [CrossRef]
- Gaitero, J.J.; Campillo, I.; Guerrero, A. Reduction of the calcium leaching rate of cement paste by addition of silica nanoparticles. Cem. Concr. Res. 2008, 38, 1112–1118. [Google Scholar] [CrossRef]
- Arribas, I.; Vegas, I.; García, V.; Villa, R.V.D.L.; Martínez-Ramírez, S.; Frías, M. The deterioration and environmental impact of binary cements containing thermally activated coal mining waste due to calcium leaching. J. Clean. Prod. 2018, 183, 887–897. [Google Scholar] [CrossRef]
- Gaitero, J.J.; Zhu, W.; Campillo, I. Multi-Scale Study of Calcium Leaching in Cement Pastes with Silica Nanoparticles. In Nanotechnology in Construction; Springer: Berlin/Heidelberg, Germany, 2009; Volume 3. [Google Scholar]
- Muthu, M.; Santhanam, M. Effect of reduced graphene oxide, alumina and silica nanoparticles on the deterioration characteristics of Portland cement paste exposed to acidic environment. Cem. Concr. Compos. 2018, 91, 118–137. [Google Scholar] [CrossRef]
- Singh, L.P.; Bhattacharyya, S.K.; Mishra, G.; Ahalawat, S. Reduction of calcium leaching in cement hydration process using nanomaterials. Mater. Process. Rep. 1999, 27, 233–238. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Todd, A.D.; Bielawski, C.W. The Chemistry of Graphene Oxide. Chem. Soc. Rev. 2009, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Monasterio, M.; Cui, H.; Han, N. Experimental study of the effects of graphene oxide on microstructure and properties of cement paste composite. Compos. Part A Appl. Sci. Manuf. 2017, 102, 263–272. [Google Scholar] [CrossRef]
- Qureshi, T.S.; Panesar, D.K.; Sidhureddy, B.; Chen, A.; Wood, P.C. Nano-cement composite with graphene oxide produced from epigenetic graphite deposit. Compos. Part B Eng. 2019, 159, 248–258. [Google Scholar] [CrossRef]
- Alharbi, Y.; An, J.; Cho, B.H.; Khawaji, M.; Chung, W.; Nam, B.H. Mechanical and sorptivity characteristics of edge-oxidized graphene oxide (EOGO)-cement composites: Dry- and wet-mix design methods. Nanomaterials 2018, 8, 718. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, S.; Zheng, D.; Yang, H.; Cui, H.; Tang, W.; Li, D. Effect of graphene oxide (GO) on the morphology and microstructure of cement hydration products. Nanomaterials 2017, 7, 429. [Google Scholar] [CrossRef]
- Long, W.-J.; Ye, T.H.; Gu, Y.-C.; Li, H.-D.; Xing, F. Inhibited effect of graphene oxide on calcium leaching of cement pastes. Constr. Build. Mater. 2019, 202, 177–188. [Google Scholar] [CrossRef]
- Jebli, M.; Jamin, F.; Pelissou, C.; Malachanne, E.; Garcia-Diaz, E.; Youssoufi, M.S.E. Leaching effect on mechanical properties of cement-aggregate interface. Cem. Concr. Compos. 2018, 87, 10–19. [Google Scholar] [CrossRef]
- Jain, J.; Neithalath, N. Analysis of calcium leaching behavior of plain and modified cement pastes in pure water. Cem. Concr. Compos. 2009, 31, 176–185. [Google Scholar] [CrossRef]
- Wan, K.; Xu, Q.; Lin, L.; Wei, S. 3D porosity distribution of partly calcium leached cement paste. Constr. Build. Mater. 2013, 48, 11–15. [Google Scholar] [CrossRef]
- Song, Z.; Liu, Y.; Jiang, L.; Guo, M.; Chen, J.; Wang, W.; Xu, N. Determination of calcium leaching behavior of cement pastes exposed to ammonium chloride aqueous solution via an electrochemical impedance spectroscopic approach. Constr. Build. Mater. 2019, 196, 267–276. [Google Scholar] [CrossRef]
- Dong, B.; Qiu, Q.; Gu, Z.; Xiang, J.; Huang, C.; Yuan, F.; Feng, X.; Wei, L. Characterization of carbonation behavior of fly ash blended cement materials by the electrochemical impedance spectroscopy method. Cem. Concr. Compos. 2016, 65, 118–127. [Google Scholar] [CrossRef]
- Long, W.-J.; Gu, Y.-c.; Xing, F.; Khayat, K.H. Microstructure development and mechanism of hardened cement paste incorporating graphene oxide during carbonation. Cem. Concr. Compos. 2018, 94, 72–84. [Google Scholar] [CrossRef]
- 175-2007, G.T. Common Portland Cement; Chinese National Standard: Beijing, China, 2007.
- 223-2007, J.T. Polycarboxylates High Performance Water-Reducing Admixture; Chinese Construction Industry Standard: Beijing, China, 2007.
- Long, W.J.; Li, H.D.; Fang, C.L.; Xing, F. Uniformly dispersed and re-agglomerated graphene oxide-based cement pastes: A comparison of rheological properties, mechanical properties and microstructure. Nanomaterials 2018, 8, 31. [Google Scholar] [CrossRef]
- 50081-2002, G.T. Standard for Test Method of Mechanical Properties on Oridinary Concrete; Chinese National Standard: Beijing, China, 2003.
- Ryu, J.S.; Otsuki, N.; Minagawa, H. Long-term forecast of Ca leaching from mortar and associated degeneration. Cem. Concr. Res. 2002, 32, 1539–1544. [Google Scholar] [CrossRef]
- Wan, K.; Li, Y.; Sun, W. Experimental and modelling research of the accelerated calcium leaching of cement paste in ammonium nitrate solution. Constr. Build. Mater. 2013, 40, 832–846. [Google Scholar] [CrossRef]
- Tang, Y.J.; Zuo, X.B.; Yin, G.J.; He, S.L.; Ayinde, O. Influence of slag on leaching behavior of cement mortar lined in ductile iron pipe under a flowing solution. Mater. Des. 2016, 114, 612–622. [Google Scholar] [CrossRef]
- Haga, K.; Sutou, S.; Hironaga, M.; Tanaka, S.; Nagasaki, S. Effects of porosity on leaching of Ca from hardened ordinary Portland cement paste. Cem. Concr. Res. 2005, 35, 1764–1775. [Google Scholar] [CrossRef]
- Cai, X.; He, Z.; Shao, Y.; Sun, H. Macro- and micro- characteristics of cement binders containing high volume fly ash subject to electrochemical accelerated leaching. Constr. Build. Mater. 2016, 116, 25–35. [Google Scholar] [CrossRef]
- GB/T17671-1999. Method of Testing Cements-Determination of Strength; Chinese National Standard: Beijing, China, 1999.
- Chen, C.M.; Zhang, Q.; Yang, M.G.; Huang, C.H.; Yang, Y.G.; Wang, M.Z. Structural evolution during annealing of thermally reduced graphene nanosheets for application in supercapacitors. Carbon 2012, 50, 3572–3584. [Google Scholar] [CrossRef]
- Du, H.; Gao, H.J.; Pang, S.D. Improvement in concrete resistance against water and chloride ingress by adding graphene nanoplatelet. Cem. Concr. Res. 2016, 83, 114–123. [Google Scholar] [CrossRef]
- Du, H.; Pang, S.D. Enhancement of barrier properties of cement mortar with graphene nanoplatelet. Cem. Concr. Res. 2015, 76, 10–19. [Google Scholar] [CrossRef]
- Tang, Y.-J.; Zuo, X.-B.; Yin, G.-J.; Davoudi, H.; Li, X.-N. Influence of calcium leaching on chloride diffusivity in cement-based materials. Constr. Build. Mater. 2018, 174, 310–319. [Google Scholar] [CrossRef]
- Wan, K.; Lin, L.; Wei, S. Solid–liquid equilibrium curve of calcium in 6 mol/L ammonium nitrate solution. Cem. Concr. Res. 2013, 53, 44–50. [Google Scholar] [CrossRef]
- Hou, D.; Li, D.; Hua, P.; Jiang, J.; Zhang, G. Statistical modelling of compressive strength controlled by porosity and pore size distribution for cementitious materials. Cem. Concr. Compos. 2019, 96, 11–20. [Google Scholar] [CrossRef]
- Song, G. Equivalent circuit model for AC electrochemical impedance spectroscopy of concrete. Cem. Concr. Res. 2000, 30, 1723–1730. [Google Scholar] [CrossRef]
- Li, X.; Korayem, A.H.; Li, C.; Liu, Y.; He, H.; Sanjayan, J.G.; Duan, W.H. Incorporation of graphene oxide and silica fume into cement paste: A study of dispersion and compressive strength. Constr. Build. Mater. 2016, 123, 327–335. [Google Scholar] [CrossRef]
- Zhou, C.; Li, F.; Hu, J.; Ren, M.; Wei, J.; Yu, Q. Enhanced mechanical properties of cement paste by hybrid graphene oxide/carbon nanotubes. Constr. Build. Mater. 2017, 134, 336–345. [Google Scholar] [CrossRef]
- Chuah, S.; Li, W.; Chen, S.J.; Sanjayan, J.G.; Duan, W.H. Investigation on dispersion of graphene oxide in cement composite using different surfactant treatments. Constr. Build. Mater. 2018, 161, 519–527. [Google Scholar] [CrossRef]
- Lin, C.; Wei, W.; Yun, H.H. Catalytic behavior of graphene oxide for cement hydration process. J. Phys. Chem. Solids 2016, 89, 128–133. [Google Scholar] [CrossRef]
- Lei, W.; Lin, L.; Bin, G.; Rafael, M.O.C.; Ming, Z.; Hao, C.; Zuhao, Z.; Hao, W. Aggregation kinetics of graphene oxides in aqueous solutions: Experiments, mechanisms, and modeling. Langmuir 2013, 29, 15174–15181. [Google Scholar]
- Snyder, K.A.; Feng, X.; Keen, B.D.; Mason, T.O. Estimating the electrical conductivity of cement paste pore solutions from OH, K and Na concentrations. Cem. Concr. Res. 2003, 33, 793–798. [Google Scholar] [CrossRef]
- Buckley, L.J.; Carter, M.A.; Wilson, M.A.; Scantlebury, J.D. Methods of obtaining pore solution from cement pastes and mortars for chloride analysis. Cem. Concr. Res. 2007, 37, 1544–1550. [Google Scholar] [CrossRef]
- Pilvar, A.; Ramezanianpour, A.A.; Rajaie, H. New method development for evaluation concrete chloride ion permeability. Constr. Build. Mater. 2015, 93, 790–797. [Google Scholar] [CrossRef]
- Pan, Z.; He, L.; Qiu, L.; Korayem, A.H.; Li, G.; Zhu, J.W.; Collins, F.; Li, D.; Duan, W.H.; Wang, M.C. Mechanical properties and microstructure of a graphene oxide–cement composite. Cem. Concr. Compos. 2015, 58, 140–147. [Google Scholar] [CrossRef]



| Chemical Composition | Ingredient | CaO | SiO2 | Al2O3 | Fe2O3 | MgO | SO3 | K2O | Na2O | LOI | |||||
| Content (mass %) | 64.42 | 20.52 | 5.62 | 3.78 | 2.11 | 2.10 | 0.28 | 0.20 | 0.87 | ||||||
| Physical properties | Specific surface area (m2/g) | ρ0 (g/cm3) | Setting time (min) | Flexural strength (MPa) | Compressive strength (MPa) | ||||||||||
| 0.581 | 3.00 | Initial | Final | 3 d | 28 d | 3 d | 28 d | ||||||||
| 112 | 145 | 6.50 | 9.20 | 34.80 | 58.00 | ||||||||||
| Appearance | Solid Content (mass %) | pH | Viscosity (Pa·s) | Absorbance Ratio A230/A600 | Carbon (mass %) | Molar Ratio (O/C) |
|---|---|---|---|---|---|---|
| Brown paste | 43 ± 1 | ≥1.2 | ≥2000 | ≥45 | 47 ± 5 | 0.6 ± 1 |
| Designation | State | Density (g/m3) | pH | Solid Content (mass %) |
|---|---|---|---|---|
| RCM–3 | liquid | 1.102 | 5.0 | 49.98 |
| CP–WRM50 | liquid | 1.114 | 4.5 | 50.79 |
| Sample | Cement (g) | Water (g) | W/C Ratio | GO (g) | PCE/GO |
|---|---|---|---|---|---|
| R0 | 100 | 40 | 0.4 | 0 | – |
| G1 | 100 | 40 | 0.4 | 0.05 | 3.0 |
| G2 | 100 | 40 | 0.4 | 0.1 | 3.0 |
| G3 | 100 | 40 | 0.4 | 0.15 | 3.0 |
| G4 | 100 | 40 | 0.4 | 0.2 | 3.0 |
| Leaching Duration (day) | R0 | G1 | G2 | G3 | G4 |
|---|---|---|---|---|---|
| 0 | 970 | 1287 | 1421 | 1545 | 1843 |
| 7 | 320 | 556 | 660 | 801 | 1012 |
| 14 | 255 | 421 | 500 | 625 | 883 |
| 21 | 182 | 289 | 401 | 481 | 710 |
| 28 | 143 | 255 | 343 | 440 | 635 |
| 35 | 50 | 205 | 289 | 381 | 576 |
| Leaching Duration | Measured vs. Calculated Depth | GO Content | ||||
|---|---|---|---|---|---|---|
| 0% | 0.05% | 0.1% | 0.15% | 0.2% | ||
| 35 days | Measured depth (mm) | 9.34 | 8.66 | 8.29 | 7.58 | 6.48 |
| Predicted depth (mm) | 10.19 | 8.10 | 7.36 | 6.61 | 5.82 | |
| Variation (%) | 9.10 | 6.47 | 11.22 | 12.80 | 10.19 | |
| Leaching Duration | Measured vs. Calculated Loss | GO Content | ||||
|---|---|---|---|---|---|---|
| 0% | 0.05% | 0.1% | 0.15% | 0.2% | ||
| 35 days | Measured loss | 78.54 | 76.33 | 72.10 | 70.80 | 68.87 |
| Predicted loss | 82.90 | 76.70 | 73.74 | 71.71 | 71.25 | |
| Variation | 5.55 | 0.48 | 2.22 | 1.29 | 3.46 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Long, W.-J.; Ye, T.-H.; Li, L.-X.; Feng, G.-L. Electrochemical Characterization and Inhibiting Mechanism on Calcium Leaching of Graphene Oxide Reinforced Cement Composites. Nanomaterials 2019, 9, 288. https://doi.org/10.3390/nano9020288
Long W-J, Ye T-H, Li L-X, Feng G-L. Electrochemical Characterization and Inhibiting Mechanism on Calcium Leaching of Graphene Oxide Reinforced Cement Composites. Nanomaterials. 2019; 9(2):288. https://doi.org/10.3390/nano9020288
Chicago/Turabian StyleLong, Wu-Jian, Tao-Hua Ye, Li-Xiao Li, and Gan-Lin Feng. 2019. "Electrochemical Characterization and Inhibiting Mechanism on Calcium Leaching of Graphene Oxide Reinforced Cement Composites" Nanomaterials 9, no. 2: 288. https://doi.org/10.3390/nano9020288
APA StyleLong, W.-J., Ye, T.-H., Li, L.-X., & Feng, G.-L. (2019). Electrochemical Characterization and Inhibiting Mechanism on Calcium Leaching of Graphene Oxide Reinforced Cement Composites. Nanomaterials, 9(2), 288. https://doi.org/10.3390/nano9020288





