The Self-Assembly of Lignin and Its Application in Nanoparticle Synthesis: A Short Review
Abstract
1. Introduction
2. Forces and Interactions
2.1. Aggregation Propensity and the Effect of Reaction Conditions
2.1.1. Source and Extraction Processes of Lignin
2.1.2. Nature and pH of Solvent
2.1.3. Effect of Temperature
2.2. Noncovalent Interactions and Self-Assembly
π–π Stacking/Interactions
2.2.2. Hydrogen Bonding
2.2.3. Chain Entanglement
2.2.4. Van der Waals Forces
2.2.5. Hydrophobic Interactions
3. Nanoparticle Synthesis
4. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- Achyuthan, K.E.; Achyuthan, A.M.; Adams, P.D.; Dirk, S.M.; Harper, J.C.; Simmons, B.A.; Singh, A.K. Supramolecular Self-Assembled Chaos: Polyphenolic Lignin’s Barrier to Cost-Effective Lignocellulosic Biofuels. Molecules 2010, 15, 8641–8688. [Google Scholar] [CrossRef] [PubMed]
- Poletto, M.; Zattera, A.J. Materials produced from plant biomass: part III: degradation kinetics and hydrogen bonding in lignin. Mater. Res. 2013, 16, 1065–1070. [Google Scholar] [CrossRef]
- Westbye, P.; Köhnke, T.; Glasser, W.; Gatenholm, P. The influence of lignin on the self-assembly behaviour of xylan rich fractions from birch (Betula pendula). Cellulose 2007, 14, 603–613. [Google Scholar] [CrossRef]
- Beisl, S.; Miltner, A.; Friedl, A. Lignin from Micro-to Nanosize: Production Methods. Int. J. Mol. Sci. 2017, 18, 1244. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, P.; Lintinen, K.; Hirvonen, J.T.; Kostiainen, M.A.; Santos, H.A. Properties and chemical modifications of lignin: Towards lignin-based nanomaterials for biomedical applications. Prog. Mater Sci. 2018, 93, 233–269. [Google Scholar] [CrossRef]
- Zhao, W.; Simmons, B.; Singh, S.; Ragauskas, A.; Cheng, G. From lignin association to nano-/micro-particle preparation: extracting higher value of lignin. Green Chem. 2016, 18, 5693–5700. [Google Scholar] [CrossRef]
- Yang, W.; Fortunati, E.; Gao, D.; Balestra, G.M.; Giovanale, G.; He, X.; Torre, L.; Kenny, J.M.; Puglia, D. Valorization of acid isolated high yield lignin nanoparticles as innovative antioxidant/antimicrobial organic materials. ACS Sustainable Chem. Eng. 2018, 6, 3502–3514. [Google Scholar] [CrossRef]
- Nypeloe, T.E.; Carrillo, C.A.; Rojas, O.J. Lignin supracolloids synthesized from (W/O) microemulsions: use in the interfacial stabilization of Pickering systems and organic carriers for silver metal. Soft Matter 2015, 11, 2046–2054. [Google Scholar] [CrossRef]
- Qian, Y.; Zhang, Q.; Qiu, X.; Zhu, S. CO2-responsive diethylaminoethyl-modified lignin nanoparticles and their application as surfactants for CO2/N-2-switchable Pickering emulsions. Green Chem. 2014, 16, 4963–4968. [Google Scholar] [CrossRef]
- Mishra, P.K.; Wimmer, R. Aerosol assisted self-assembly as a route to synthesize solid and hollow spherical lignin colloids and its utilization in layer by layer deposition. Ultrason. Sonochem. 2017, 35, 45–50. [Google Scholar] [CrossRef]
- Qian, Y.; Zhong, X.; Li, Y.; Qiu, X. Fabrication of uniform lignin colloidal spheres for developing natural broad-spectrum sunscreens with high sun protection factor. Ind. Crops Prod. 2017, 101, 54–60. [Google Scholar] [CrossRef]
- Yang, W.; Kenny, J.M.; Puglia, D. Structure and properties of biodegradable wheat gluten bionanocomposites containing lignin nanoparticles. Ind. Crops Prod. 2015, 74, 348–356. [Google Scholar] [CrossRef]
- Král, P.; Klímek, P.; Mishra, P.K.; Rademacher, P.; Wimmer, R. Preparation and Characterization of Cork Layered Composite Plywood Boards. BioResources 2014, 9, 1977–1985. [Google Scholar] [CrossRef]
- Gilca, I.A.; Ghitescu, R.E.; Puitel, A.C.; Popa, V.I. Preparation of lignin nanoparticles by chemical modification. Iran. Polym. J. 2014, 23, 355–363. [Google Scholar] [CrossRef]
- Klímek, P.; Wimmer, R.; Kumar Mishra, P.; Kúdela, J. Utilizing brewer’s-spent-grain in wood-based particleboard manufacturing. J. Cleaner Prod. 2017, 141, 812–817. [Google Scholar] [CrossRef]
- Cauley, A.N.; Wilson, J.N. Functionalized Lignin Biomaterials for Enhancing Optical Properties and Cellular Interactions of Dyes. Biomater. Sci. 2017, 5, 2114–2121. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qiu, X.; Qian, Y.; Xiong, W.; Yang, D. pH-responsive lignin-based complex micelles: preparation, characterization and application in oral drug delivery. Chem. Eng. J. 2017, 327, 1176–1183. [Google Scholar] [CrossRef]
- Liu, D.; Li, Y.; Qian, Y.; Xiao, Y.; Du, S.; Qiu, X. Synergistic Antioxidant Performance of Lignin and Quercetin Mixtures. ACS Sustainable Chem. Eng. 2017, 5, 8424–8428. [Google Scholar] [CrossRef]
- Siddiqui, L.; Mishra, H.; Mishra, P.K.; Iqbal, Z.; Talegaonkar, S. Novel 4-in-1 strategy to combat colon cancer, drug resistance and cancer relapse utilizing functionalized bioinspiring lignin nanoparticle. Med. Hypotheses 2018, 121, 10–14. [Google Scholar] [CrossRef]
- Lindström, T. The colloidal behaviour of kraft lignin. Colloid. Polym. Sci. 1980, 258, 168–173. [Google Scholar] [CrossRef]
- Lindströmn, T. The colloidal behaviour of kraft lignin. Colloid. Polym. Sci. 1979, 257, 277–285. [Google Scholar] [CrossRef]
- Norgren, M.; Edlund, H.; Wågberg, L. Aggregation of lignin derivatives under alkaline conditions. Kinetics and aggregate structure. Langmuir 2002, 18, 2859–2865. [Google Scholar] [CrossRef]
- Norgren, M.; Edlund, H.; Wågberg, L.; Lindström, B.; Annergren, G. Aggregation of kraft lignin derivatives under conditions relevant to the process, part I: phase behaviour. Colloids Surf. A Physicochem. Engin Aspects 2001, 194, 85–96. [Google Scholar] [CrossRef]
- Zhao, W.; Xiao, L.-P.; Song, G.; Sun, R.-C.; He, L.; Singh, S.; Simmons, B.A.; Cheng, G. From lignin subunits to aggregates: insights into lignin solubilization. Green Chem. 2017, 19, 3272–3281. [Google Scholar] [CrossRef]
- Mafé, S.; Manzanares, J.; Kontturi, A.-K.; Kontturi, K. Temperature effects on counterion binding to spherical polyelectrolytes: the charge-discharge transition of lignosulfonate. Bioelectrochem. Bioenerg. 1995, 38, 367–375. [Google Scholar] [CrossRef]
- Norgren, M.; Lindström, B. Physico-Chemical Characterization of a Fractionated Kraft Lignin. Holzforschung 2005, 54, 528. [Google Scholar] [CrossRef]
- Hita, I.; Heeres, H.J.; Deuss, P.J. Insight into structure–reactivity relationships for the iron-catalyzed hydrotreatment of technical lignins. Bioresour. Technol. 2018, 267, 93–101. [Google Scholar] [CrossRef]
- Constant, S.; Wienk, H.L.J.; Frissen, A.E.; de Peinder, P.; Boelens, R.; van Es, D.S.; Grisel, R.J.H.; Weckhuysen, B.M.; Huijgen, W.J.J.; Gosselink, R.J.A.; et al. New insights into the structure and composition of technical lignins: A comparative characterisation study. Green Chem. 2016, 18, 2651–2665. [Google Scholar]
- Renders, T.; Van den Bosch, S.; Koelewijn, S.-F.; Schutyser, W.; Sels, B. Lignin-first biomass fractionation: the advent of active stabilisation strategies. Energy Environ. Sci. 2017, 10, 1551–1557. [Google Scholar] [CrossRef]
- Dence, C.W. The determination of lignin. In Methods in lignin chemistry; Springer-Verlag: New York, NY, USA, 1992; pp. 33–61. [Google Scholar]
- Lupoi, J.S.; Singh, S.; Parthasarathi, R.; Simmons, B.A.; Henry, R.J. Recent innovations in analytical methods for the qualitative and quantitative assessment of lignin. Renewable Sustainable Energy Rev. 2015, 49, 871–906. [Google Scholar] [CrossRef]
- Ragauskas, A.J.; Williams, C.K.; Davison, B.H.; Britovsek, G.; Cairney, J.; Eckert, C.A.; Frederick, W.J.; Hallett, J.P.; Leak, D.J.; Liotta, C.L. The path forward for biofuels and biomaterials. Science 2006, 311, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Lancefield, C.S.; Wienk, H.L.; Boelens, R.; Weckhuysen, B.M.; Bruijnincx, P.C. Identification of a diagnostic structural motif reveals a new reaction intermediate and condensation pathway in kraft lignin formation. Chem. Sci. 2018, 9, 6348–6360. [Google Scholar] [CrossRef] [PubMed]
- Shen, Q.; Zhang, T.; Zhu, M.-F. A comparison of the surface properties of lignin and sulfonated lignins by FTIR spectroscopy and wicking technique. Colloids Surf. A Physicochem. Eng. Aspects 2008, 320, 57–60. [Google Scholar] [CrossRef]
- Dawy, M.; Shabaka, A.A.; Nada, A.M.A. Molecular structure and dielectric properties of some treated lignins. Polym. Degrad. Stab. 1998, 62, 455–462. [Google Scholar] [CrossRef]
- Ratnaweera, D.R.; Saha, D.; Pingali, S.V.; Labbé, N.; Naskar, A.K.; Dadmun, M. The impact of lignin source on its self-assembly in solution. RSC Adv. 2015, 5, 67258–67266. [Google Scholar] [CrossRef]
- Fiţigău, I.; Peter, F.; Boeriu, C. Structural analysis of lignins from different sources. Int. J. Chem. Mol. Nucl. Mater. Metall. Eng. 2013, 7, 167–172. [Google Scholar]
- Miller-Chou, B.A.; Koenig, J.L. A review of polymer dissolution. Prog. Polym. Sci. 2003, 28, 1223–1270. [Google Scholar] [CrossRef]
- Wang, J.; Li, Y.; Qiu, X.; Liu, D.; Yang, D.; Liu, W.; Qian, Y. Dissolution of lignin in green urea aqueous solution. Appl. Surf. Sci. 2017, 425, 736–741. [Google Scholar] [CrossRef]
- Qian, Y.; Deng, Y.; Qiu, X.; Li, H.; Yang, D. Formation of uniform colloidal spheres from lignin, a renewable resource recovered from pulping spent liquor. Green Chem. 2014, 16, 2156–2163. [Google Scholar] [CrossRef]
- Xiong, F.; Han, Y.; Wang, S.; Li, G.; Qin, T.; Chen, Y.; Chu, F. Preparation and formation mechanism of size-controlled lignin nanospheres by self-assembly. Ind. Crops Prod. 2017, 100, 146–152. [Google Scholar] [CrossRef]
- Xiong, F.; Han, Y.; Wang, S.; Li, G.; Qin, T.; Chen, Y.; Chu, F. Preparation and Formation Mechanism of Renewable Lignin Hollow Nanospheres with a Single Hole by Self-Assembly. ACS Sustainable Chem. Eng. 2017, 5, 2273–2281. [Google Scholar] [CrossRef]
- Bikova, T.; Treimanis, A.; Rossinska, G.; Telysheva, G. On-line study of lignin behaviour in dilute alkaline solution by the SEC-UV method. Holzforschung 2004, 58, 489–494. [Google Scholar] [CrossRef]
- Garver, T.M.; Callaghan, P.T. Hydrodynamics of kraft lignins. Macromolecules 1991, 24, 420–430. [Google Scholar] [CrossRef]
- Salentinig, S.; Schubert, M. Softwood Lignin Self-Assembly for Nanomaterial Design. Biomacromolecules 2017, 18, 2649–2653. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Zhao, W.; Singh, S.; Simmons, B.; Cheng, G. On the solution structure of kraft lignin in ethylene glycol and its implication for nanoparticle preparation. Nanoscale Adv. 2019, 1, 299–304. [Google Scholar] [CrossRef]
- Sameni, J.; Krigstin, S.; Sain, M. Solubility of Lignin and Acetylated Lignin in Organic Solvents. BioResources 2017, 12, 1548–1565. [Google Scholar] [CrossRef]
- Evstigneev, E. Factors affecting lignin solubility. Russ. J. Appl. Chem. 2011, 84, 1040–1045. [Google Scholar] [CrossRef]
- Borisova, O.V.; Billon, L.; Cernochova, Z.; Lapp, A.; Stepanek, P.; Borisov, O.V. Effect of Temperature on Self-Assembly of Amphiphilic Block-Gradient Copolymers of Styrene and Acrylic Acid. Macromol. Symp. 2015, 348, 25–32. [Google Scholar] [CrossRef]
- Guerra, A.; Gaspar, A.R.; Contreras, S.; Lucia, L.A.; Crestini, C.; Argyropoulos, D.S. On the propensity of lignin to associate: A size exclusion chromatography study with lignin derivatives isolated from different plant species. Phytochemistry 2007, 68, 2570–2583. [Google Scholar] [CrossRef]
- Dhotel, A.; Chen, Z.; Delbreilh, L.; Youssef, B.; Saiter, J.-M.; Tan, L. Molecular Motions in Functional Self-Assembled Nanostructures. Int. J. Mol. Sci. 2013, 14, 2303–2333. [Google Scholar] [CrossRef]
- Martinez, C.R.; Iverson, B.L. Rethinking the term “pi-stacking”. Chem. Sci. 2012, 3, 2191–2201. [Google Scholar] [CrossRef]
- McRae, E.M. Kasha in Physical Process in Radiation Biology. Academic Press: New York, NY, USA, 1964. [Google Scholar]
- Deng, Y.; Feng, X.; Yang, D.; Yi, C.; Qiu, X. Pi-Pi stacking of the aromatic groups in lignosulfonates. BioResources 2012, 7, 1145–1156. [Google Scholar]
- Lievonen, M.; Valle-Delgado, J.J.; Mattinen, M.-L.; Hult, E.-L.; Lintinen, K.; Kostiainen, M.A.; Paananen, A.; Szilvay, G.R.; Setälä, H.; Österberg, M. A simple process for lignin nanoparticle preparation. Green Chem. 2016, 18, 1416–1422. [Google Scholar] [CrossRef]
- Whitesides, G.M.; Boncheva, M. Beyond molecules: Self-assembly of mesoscopic and macroscopic components. Proc. Natl. Acad. Sci. U.S.A. 2002, 99, 4769–4774. [Google Scholar] [CrossRef] [PubMed]
- Kubo, S.; Kadla, J.F. Hydrogen bonding in lignin: a Fourier transform infrared model compound study. Biomacromolecules 2005, 6, 2815–2821. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.P.; Eriksson, K.-E.L. The significance of intra-molecular hydrogen bonding in the ß-O-4 linkage of lignin. J. Mol. Struct. 1996, 384, 1–7. [Google Scholar] [CrossRef]
- Kadla, J.F.; Kubo, S. Miscibility and Hydrogen Bonding in Blends of Poly(ethylene oxide) and Kraft Lignin. Macromolecules 2003, 36, 7803–7811. [Google Scholar] [CrossRef]
- Vainio, U.; Maximova, N.; Hortling, B.; Laine, J.; Stenius, P.; Simola, L.K.; Gravitis, J.; Serimaa, R. Morphology of Dry Lignins and Size and Shape of Dissolved Kraft Lignin Particles by X-ray Scattering. Langmuir 2004, 20, 9736–9744. [Google Scholar] [CrossRef]
- Suzuki, M.; Hanabusa, K. Polymer organogelators that make supramolecular organogels through physical cross-linking and self-assembly. Chem. Soc. Rev. 2010, 39, 455–463. [Google Scholar] [CrossRef]
- Choi, B.; Yu, J.; Paley, D.W.; Trinh, M.T.; Paley, M.V.; Karch, J.M.; Crowther, A.C.; Lee, C.-H.; Lalancette, R.A.; Zhu, X.; et al. van der Waals Solids from Self-Assembled Nanoscale Building Blocks. Nano Lett. 2016, 16, 1445–1449. [Google Scholar] [CrossRef]
- Gao, H.-Y.; Wagner, H.; Held, P.A.; Du, S.; Gao, H.-J.; Studer, A.; Fuchs, H. In-plane Van der Waals interactions of molecular self-assembly monolayer. Appl. Phys. Lett. 2015, 106, 081606. [Google Scholar] [CrossRef]
- Lindström, T.; Westman, L. The colloidal behaviour of kraft lignin. Colloid. Polym. Sci. 1982, 260, 594–598. [Google Scholar] [CrossRef]
- Youssefian, S.; Rahbar, N. Molecular Origin of Strength and Stiffness in Bamboo Fibrils. Sci. Rep. 2015, 5, 11116. [Google Scholar] [CrossRef] [PubMed]
- Meyer, E.E.; Rosenberg, K.J.; Israelachvili, J. Recent progress in understanding hydrophobic interactions. Proc. Natl. Acad. Sci. 2006, 103, 15739–15746. [Google Scholar] [CrossRef]
- Leskinen, T.; Witos, J.; Valle Delgado, J.J.; Lintinen, K.S.; Kostiainen, M.A.; Wiedmer, S.K.; Österberg, M.; Mattinen, M.-L. Adsorption of proteins on colloidal lignin particles for advanced biomaterials. Biomacromolecules 2017, 18, 2767–2776. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Clarke, K.; Li, K. Interactive forces between lignin and cellulase as determined by atomic force microscopy. Biotechnol. Biofuels 2014, 7, 65. [Google Scholar]
- Salas, C.; Rojas, O.J.; Lucia, L.A.; Hubbe, M.A.; Genzer, J. On the surface interactions of proteins with lignin. ACS Appl. Mater. Interfaces 2013, 5, 199–206. [Google Scholar] [CrossRef]
- Sammond, D.W.; Yarbrough, J.M.; Mansfield, E.; Bomble, Y.J.; Hobdey, S.E.; Decker, S.R.; Taylor, L.E.; Resch, M.G.; Bozell, J.J.; Himmel, M.E.; et al. Predicting Enzyme Adsorption to Lignin Films by Calculating Enzyme Surface Hydrophobicity. J. Biol. Chem. 2014, 289, 20960–20969. [Google Scholar] [CrossRef] [PubMed]
- Ekeberg, D.; Gretland, K.S.; Gustafsson, J.; Bråten, S.M.; Fredheim, G.E. Characterisation of lignosulphonates and kraft lignin by hydrophobic interaction chromatography. Anal. Chim. Acta 2006, 565, 121–128. [Google Scholar] [CrossRef]
- Mansfield, S.D. Solutions for dissolution—engineering cell walls for deconstruction. Curr. Opin. Biotechnol. 2009, 20, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.K.; Giagli, K.; Tsalagkas, D.; Mishra, H.; Talegaonkar, S.; Gryc, V.; Wimmer, R. Changing Face of Wood Science in Modern Era: Contribution of Nanotechnology. Recent. Pat. Nanotechnol. 2018, 12, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, L.A. Lignification and lignin topochemistry—an ultrastructural view. Phytochemistry 2001, 57, 859–873. [Google Scholar] [CrossRef]
- Chen, Y.; Sarkanen, S. Macromolecular lignin replication: A mechanistic working hypothesis. Phytochem. Rev. 2003, 2, 235–255. [Google Scholar] [CrossRef]
- Gang, D.R.; Costa, M.A.; Fujita, M.; Dinkova-Kostova, A.T.; Wang, H.-B.; Burlat, V.; Martin, W.; Sarkanen, S.; Davin, L.B.; Lewis, N.G. Regiochemical control of monolignol radical coupling: A new paradigm for lignin and lignan biosynthesis. Chem. Biol. 1999, 6, 143–151. [Google Scholar] [CrossRef]
- Karmanov, A.P.; Monakov, Y.B. Lignin. Structural organisation and fractal properties. Russ. Chem. Rev. 2003, 72, 715–734. [Google Scholar] [CrossRef]
- Radotic, K.; Tasic, M.; Jeremic, M.; Budimlija, Z.; Simic-Krstic, J.; Polzovic, A.; Bozovic, Z. Fractal dimension of lignin structure at the molecular level. Iugoslav. Physiol. Pharmacol. Acta 1998, 34, 215–220. [Google Scholar]
- Karmanov, A.; Kuznetsov, S.; Monakov, Y.B. Modeling of lignin biosynthesis in vitro. The strange attractor. mezhdunarodnaya kniga 1995, 342, 193–196. [Google Scholar]
- Rowe, J.M.; Johnston, K.P. Precipitation Technologies for Nanoparticle Production. In Formulating Poorly Water Soluble Drugs; AAPS Advances in the Pharmaceutical Sciences Series; Springer: New York, NY, USA, 2012; pp. 501–568. [Google Scholar]
- Frangville, C.; Rutkevicius, M.; Richter, A.P.; Velev, O.D.; Stoyanov, S.D.; Paunov, V.N. Fabrication of Environmentally Biodegradable Lignin Nanoparticles. Chem. Phys. Chem. 2012, 13, 4235–4243. [Google Scholar] [CrossRef]
- Tortora, M.; Cavalieri, F.; Mosesso, P.; Ciaffardini, F.; Melone, F.; Crestini, C. Ultrasound Driven Assembly of Lignin into Microcapsules for Storage and Delivery of Hydrophobic Molecules. Biomacromolecules 2014, 15, 1634–1643. [Google Scholar] [CrossRef]
- Bartzoka, E.D.; Lange, H.; Thiel, K.; Crestini, C. Coordination Complexes and One-Step Assembly of Lignin for Versatile Nanocapsule Engineering. ACS Sustainable Chem. Eng. 2016, 4, 5194–5203. [Google Scholar] [CrossRef]
- Li, H.; Deng, Y.; Liang, J.; Dai, Y.; Li, B.; Ren, Y.; Qiu, X.; Li, C. Direct Preparation of Hollow Nanospheres with Kraft Lignin: A Facile Strategy for Effective Utilization of Biomass Waste. BioResources 2016, 11, 3073–3083. [Google Scholar] [CrossRef]
- Gilca, I.A.; Pupa, V.I.; Crestini, C. Obtaining lignin nanoparticles by sonication. Ultrason. Sonochem. 2015, 23, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Rao, X.; Liu, Y.; Zhang, Q.; Chen, W.; Liu, Y.; Yu, H. Assembly of Organosolv Lignin Residues into Submicron Spheres: The Effects of Granulating in Ethanol/Water Mixtures and Homogenization. ACS Omega 2017, 2, 2858–2865. [Google Scholar] [CrossRef]
- Li, H.; Deng, Y.; Liu, B.; Ren, Y.; Liang, J.; Qian, Y.; Qiu, X.; Li, C.; Zheng, D. Preparation of Nanocapsules via the Self-Assembly of Kraft Lignin: A Totally Green Process with Renewable Resources. ACS Sustainable Chem. Eng. 2016, 4, 1946–1953. [Google Scholar] [CrossRef]
- Yiamsawas, D.; Baier, G.; Thines, E.; Landfester, K.; Wurm, F.R. Biodegradable lignin nanocontainers. RSC Adv. 2014, 4, 11661–11663. [Google Scholar] [CrossRef]
- Li, H.; Deng, Y.; Wu, H.; Ren, Y.; Qiu, X.; Zheng, D.; Li, C. Self-assembly of kraft lignin into nanospheres in dioxane-water mixtures. Holzforschung 2016, 70, 725–731. [Google Scholar] [CrossRef]
- Myint, A.A.; Lee, H.W.; Seo, B.; Son, W.-S.; Yoon, J.; Yoon, T.J.; Park, H.J.; Yu, J.; Yoon, J.; Lee, Y.-W. One pot synthesis of environmentally friendly lignin nanoparticles with compressed liquid carbon dioxide as an anti-solvent. Green Chem. 2016, 18, 2129–2146. [Google Scholar] [CrossRef]
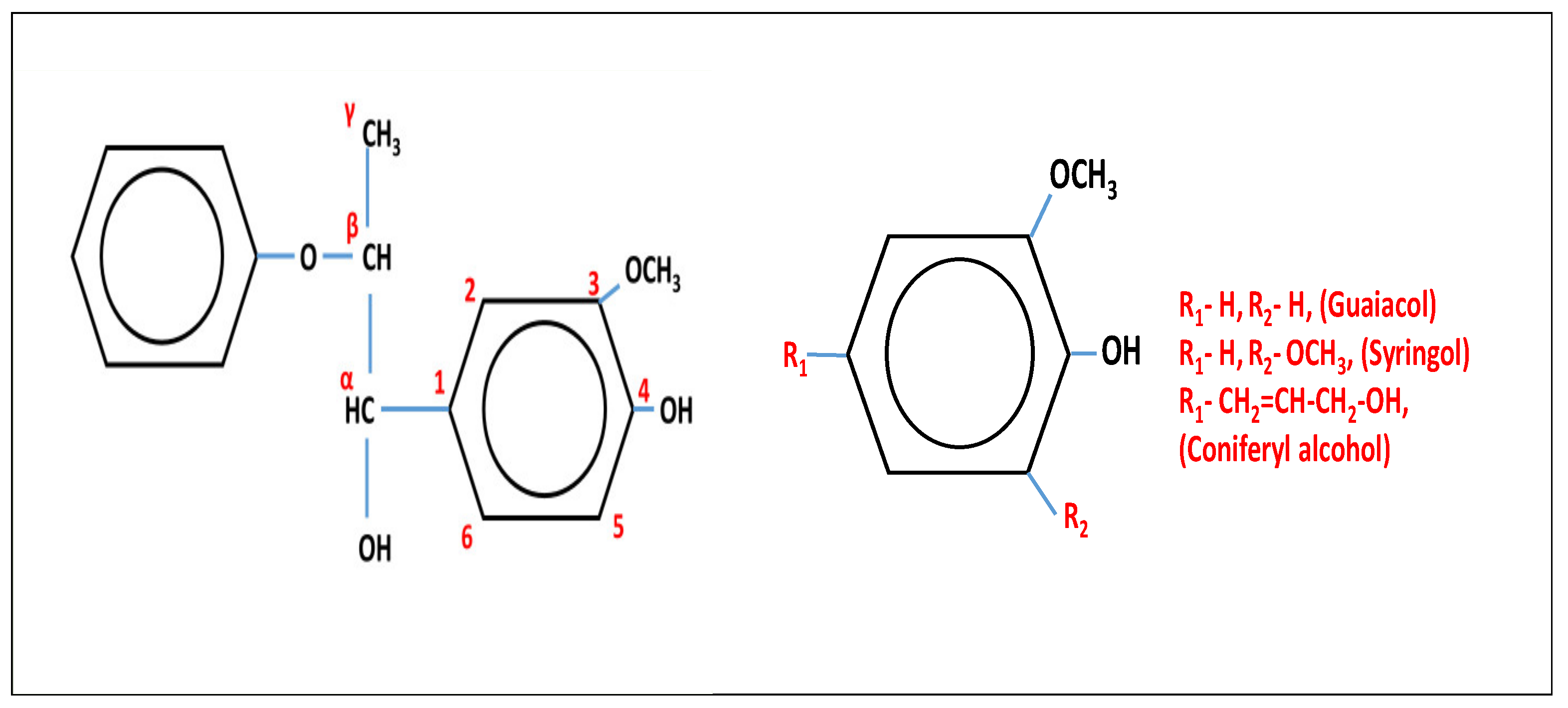
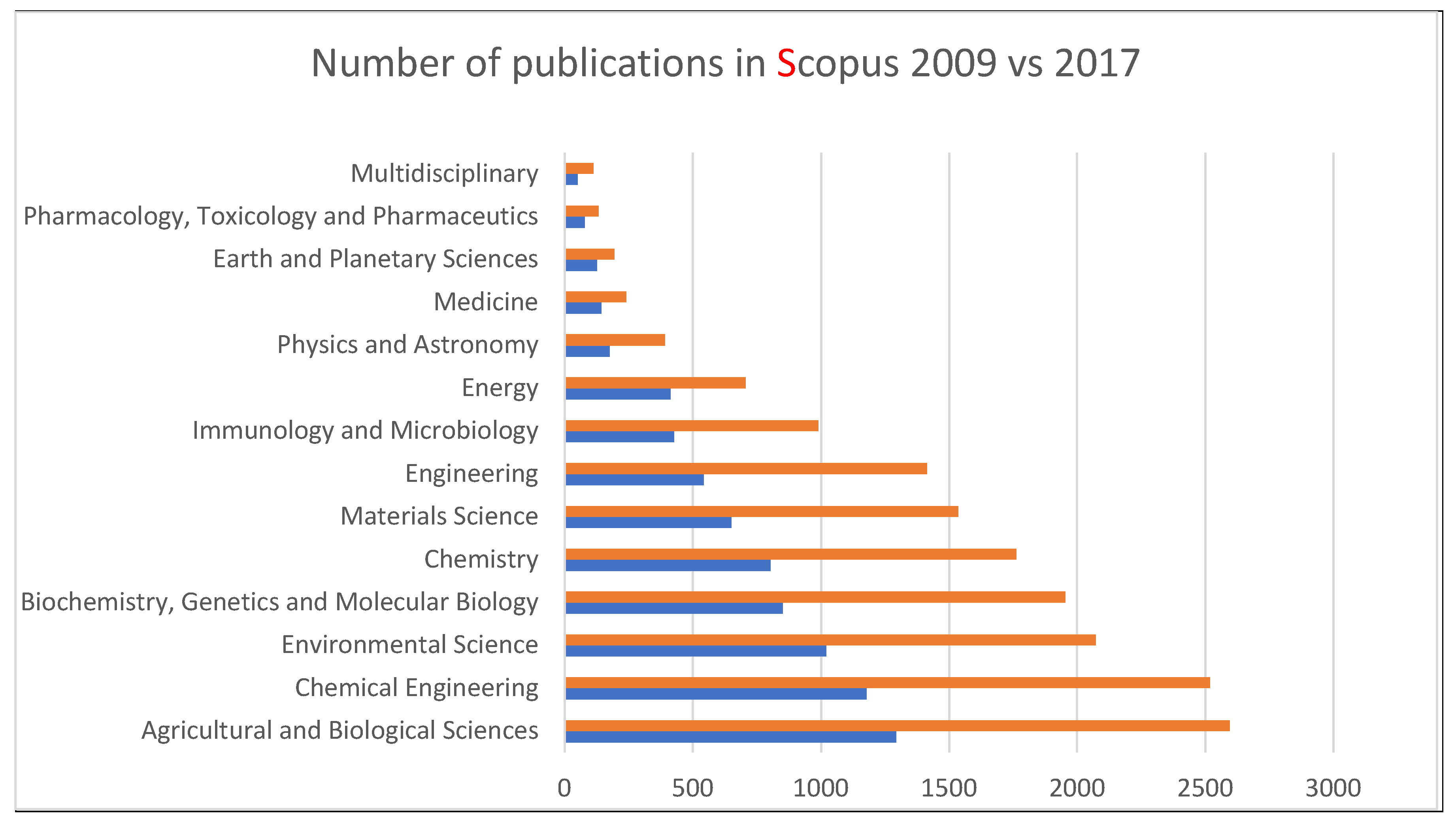
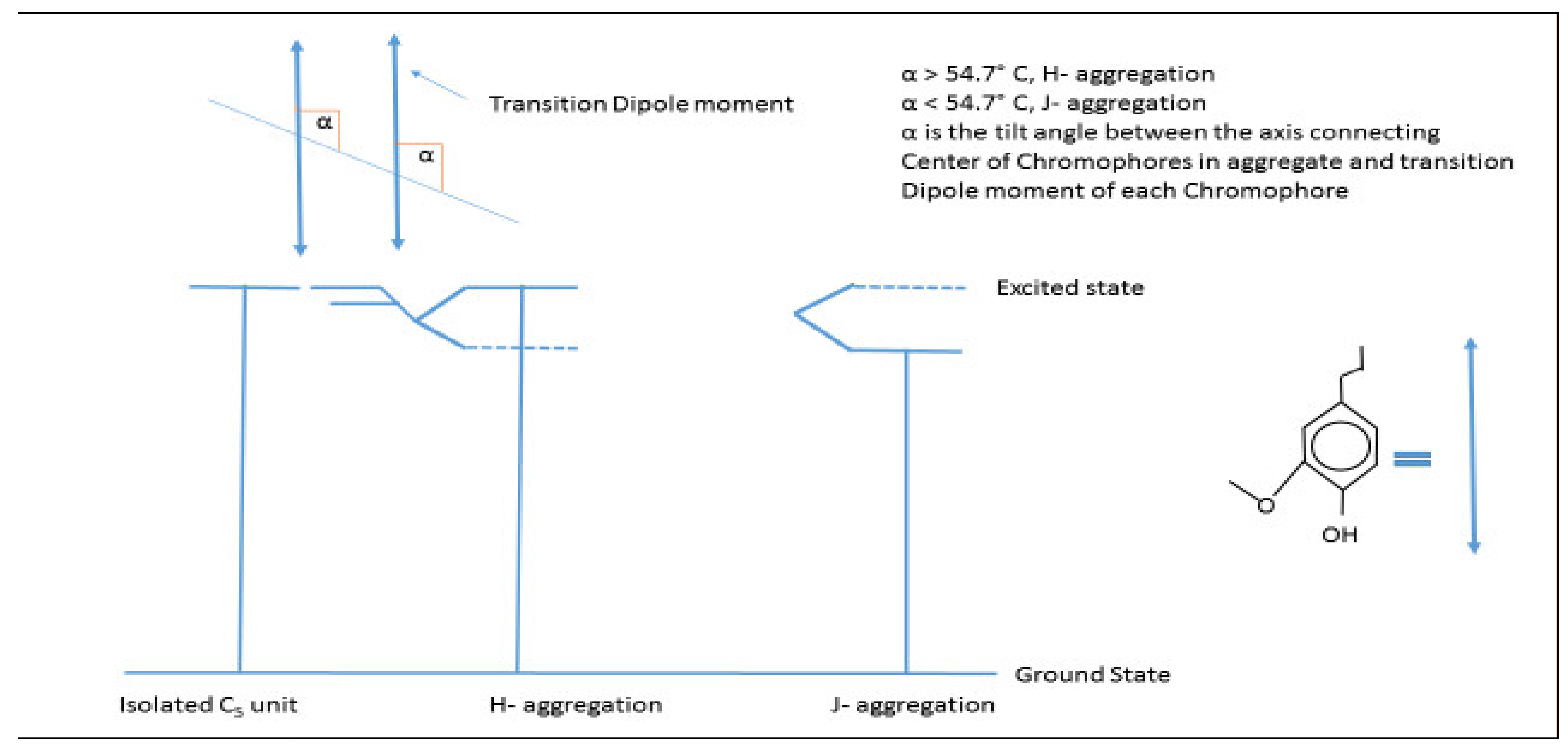
| Type | Source | Phenolic | Aliphatic | Carboxylic | Total |
|---|---|---|---|---|---|
| Soda lignin | Wheat straw | 2.29 | 2.1 | 1.10 | 5.49 a |
| P1000 soda lignin | Sarkanda grass and wheat straw | 1.85 | 1.79 | 1.11 | 4.75 a |
| Alcell/organosolv # | Mixed hardwoods (maple, birch and poplar) | 1.91 | 1.11 | 0.29 | 3.31 a |
| Kraft/indulin AT # | Softwood | 2.31 | 2.41 | 0.54 | 5.26 a |
| Alkali pretreated | Wheat straw | 1.58 | 1.07 | 0.92 | 3.57 a |
| Biolignin/organic acid extracted | Wheat straw | 1.0 | 3.9 | - | 4.9 b |
| Organosolv # | Hardwood | 4.0 | 1.5 | 0.1 | 5.6 b |
| Kraft # | Softwood | 3.4 | 1.7 | 0.0 | 5.1 b |
| Type of Lignin Used and Structure Formed | Method Used | Mechanism Suggested | Reported by |
|---|---|---|---|
| Ultrasonic Assisted Synthesis | |||
| Dioxane soluble fragment of alkali lignin, hollow colloid | Ultrasonic spray-freezing | Peripheral precipitation | [10] |
| Kraft lignin, microcapsules | Ultrasound driven self- association | Ultrasound-mediated cross-linking | [82] |
| Kraft lignin, nanocapsules | Ultrasonication of microemulsion | Ultrasound-induced self-assembly and complexation | [83] |
| Kraft lignin, hollow nanospheres | Ultrasonication assisted solubilization, anti-solvent addition | Self-assembly at two different phases | [84] |
| Wheatgrass lignin and Sarkanda grass lignin | Ultrasonication of aqueous suspension | Side chain cleavage/depolymerization and oxidative coupling/polymerization | [85] |
| Homogenization assisted synthesis | |||
| Organosolv lignin residue, submicron spheres | Homogenization enhanced nucleation | Hydrophilic-lipophilic aggregation | [86] |
| Change of pH and/or solvent | |||
| Enzymatic hydrolytic lignin, single hole nanospheres | Solvent-anti-solvent precipitation method | Layer by layer self- assembly outside to inside | [42] |
| Kraft lignin, nanocapsules | Solvent-anti-solvent precipitation method | Spontaneous distribution of hydrophilic-lipophilic sequences | [87] |
| Softwood Kraft lignin, lignin Nanoparticles | Anti-solvent addition using dialysis bag | Chemical precipitation | [55] |
| lignin nanocontainer | Interfacial polyaddition | Polyaddition reaction at interface | [88] |
| Enzymatic hydrolytic lignin, solid nanospheres | Solvent–anti-solvent precipitation method | Layer by layer self-assembly inside to outside | [41] |
| Kraft lignin, nanospheres | Solvent-anti-solvent precipitation method | Chemical precipitation | [89] |
| Low sulfonated lignin (indulin AT) | Solvent-anti-solvent and base-acid precipitation | Chemical precipitation | [81] |
| Kraft lignin | DMF (solvent)-compressed CO2 (anti-solvent) | Expansion by anti-solvent | [90] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mishra, P.K.; Ekielski, A. The Self-Assembly of Lignin and Its Application in Nanoparticle Synthesis: A Short Review. Nanomaterials 2019, 9, 243. https://doi.org/10.3390/nano9020243
Mishra PK, Ekielski A. The Self-Assembly of Lignin and Its Application in Nanoparticle Synthesis: A Short Review. Nanomaterials. 2019; 9(2):243. https://doi.org/10.3390/nano9020243
Chicago/Turabian StyleMishra, Pawan Kumar, and Adam Ekielski. 2019. "The Self-Assembly of Lignin and Its Application in Nanoparticle Synthesis: A Short Review" Nanomaterials 9, no. 2: 243. https://doi.org/10.3390/nano9020243
APA StyleMishra, P. K., & Ekielski, A. (2019). The Self-Assembly of Lignin and Its Application in Nanoparticle Synthesis: A Short Review. Nanomaterials, 9(2), 243. https://doi.org/10.3390/nano9020243






