Upgrading of Extra-Heavy Crude Oils by Dispersed Injection of NiO–PdO/CeO2±δ Nanocatalyst-Based Nanofluids in the Steam
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Adsorption Experiments
2.2.2. Thermogravimetric Analyses
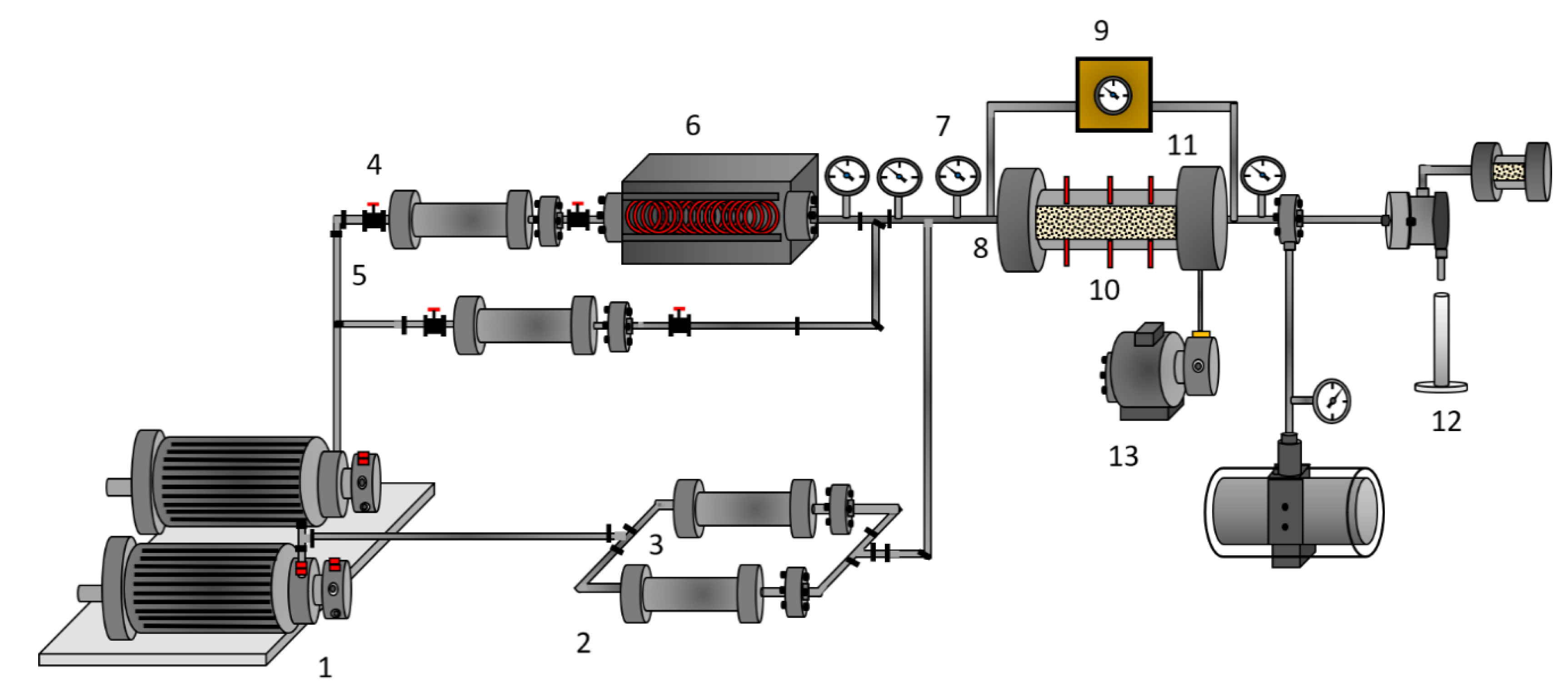
2.2.3. Oil Recovery
2.2.4. Oil Characterization before and after Upgrading
3. Modeling
3.1. Adsorption Model
3.2. Effective Activation Energy Estimation
3.3. Rheological Model
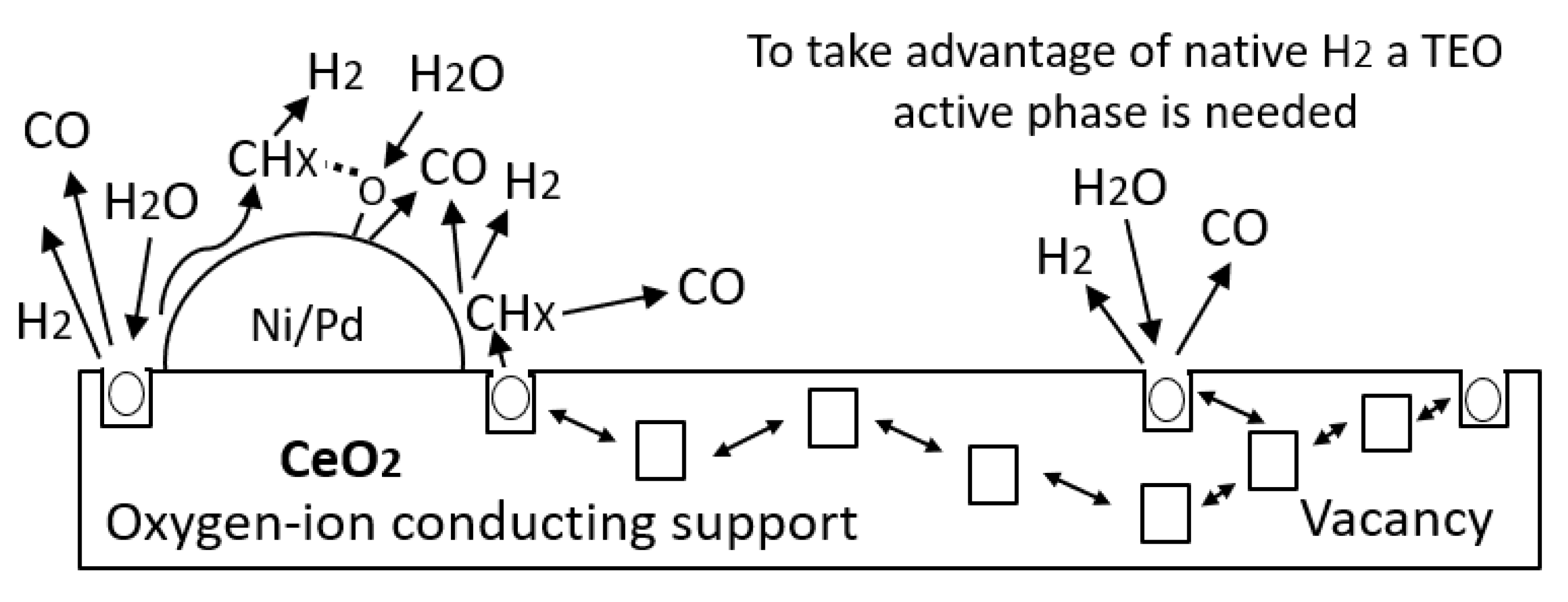
4. Results and Discussion
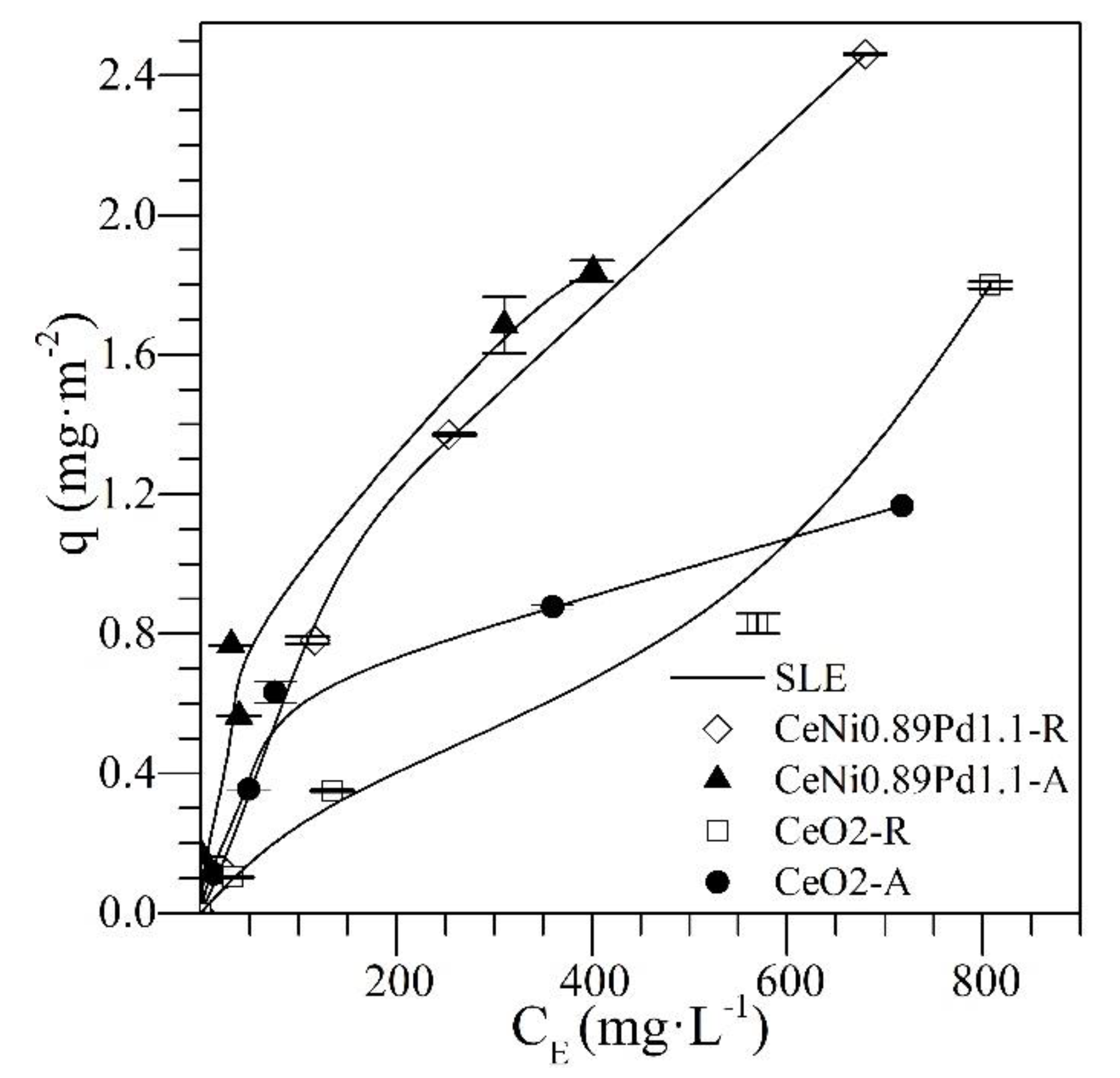
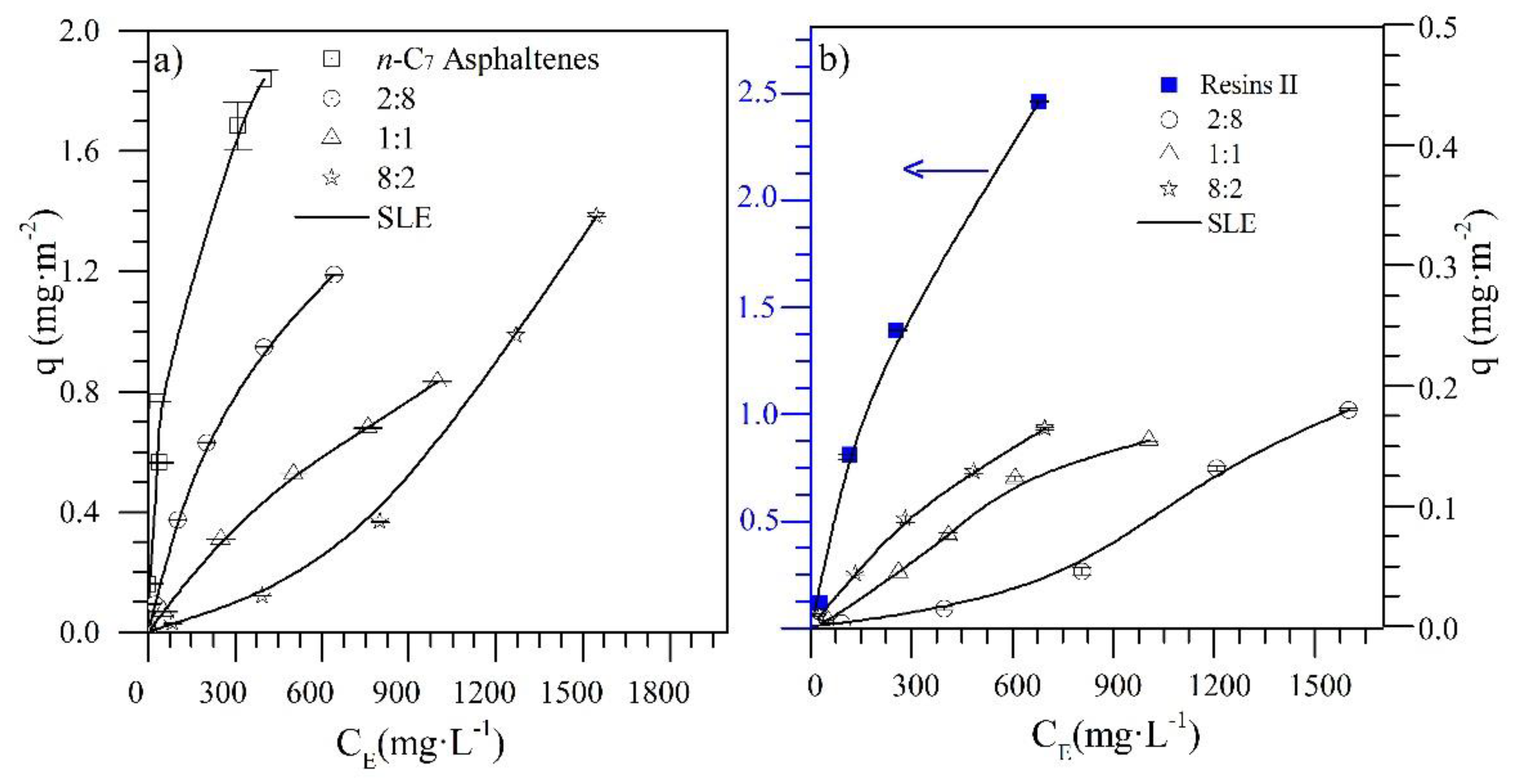
4.1. Adsorption Isotherms
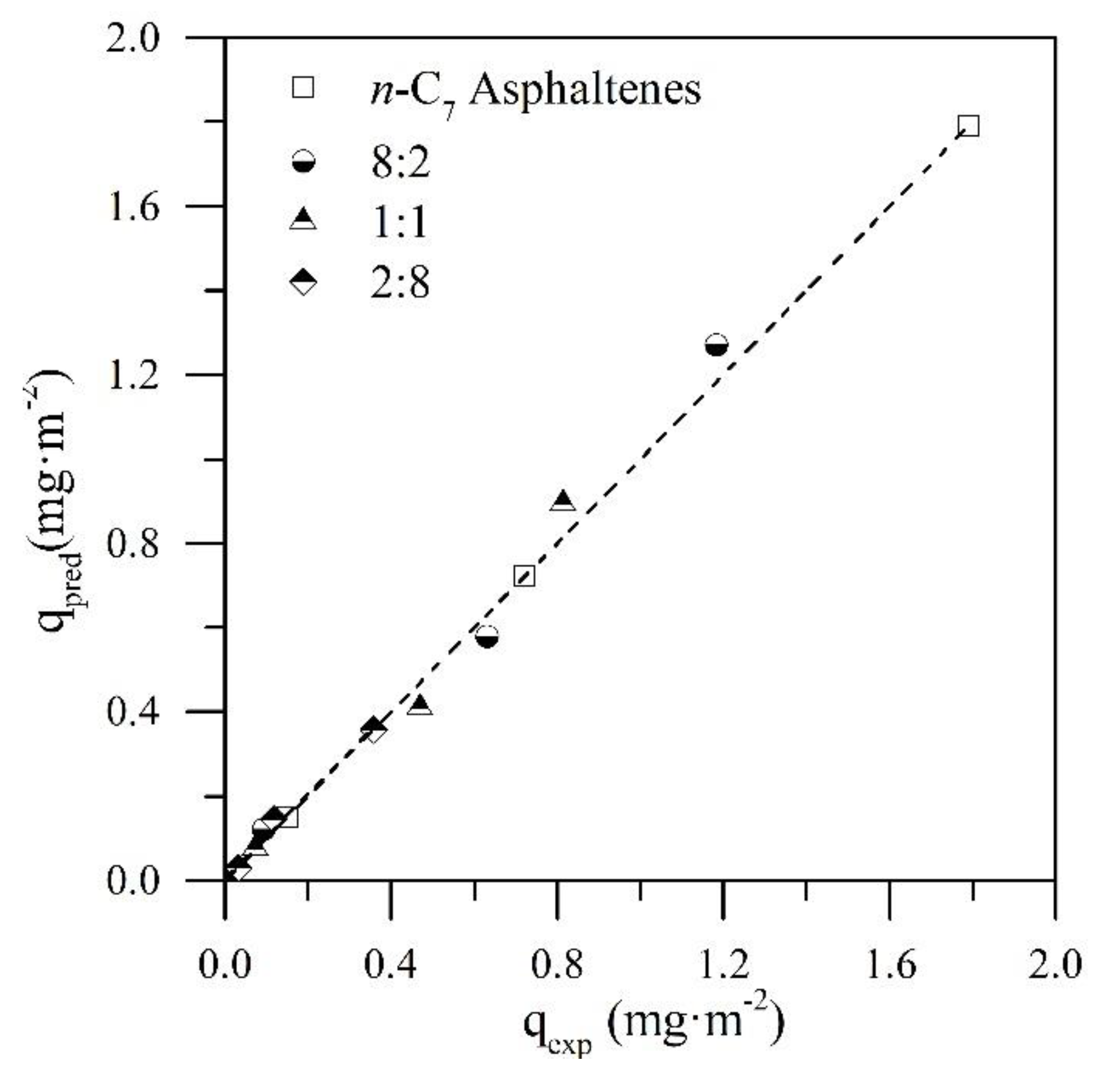
4.2. Prediction of the Adsorbed Amount of n-C7 Asphaltenes
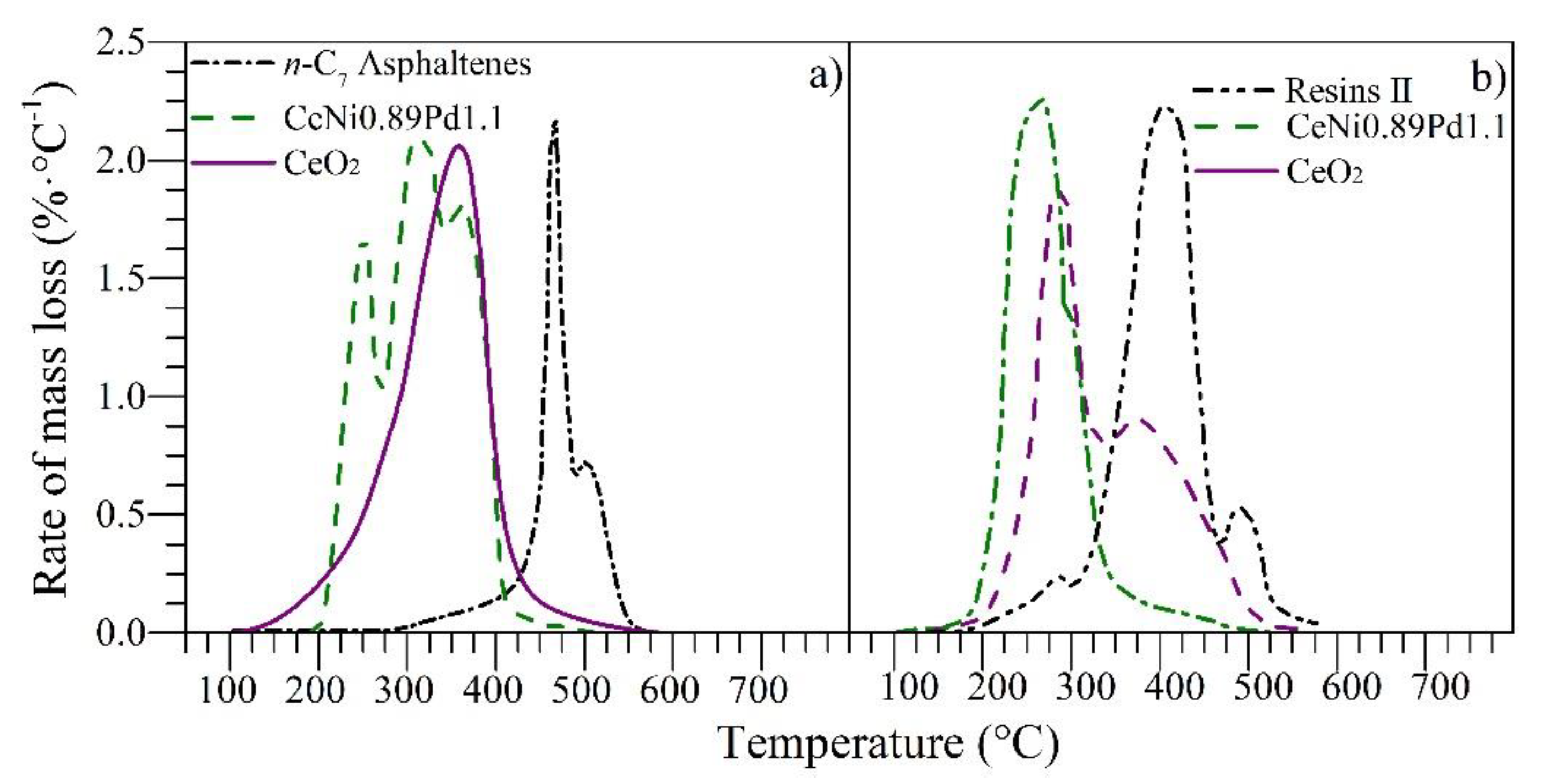
4.3. Thermogravimetric Experiments
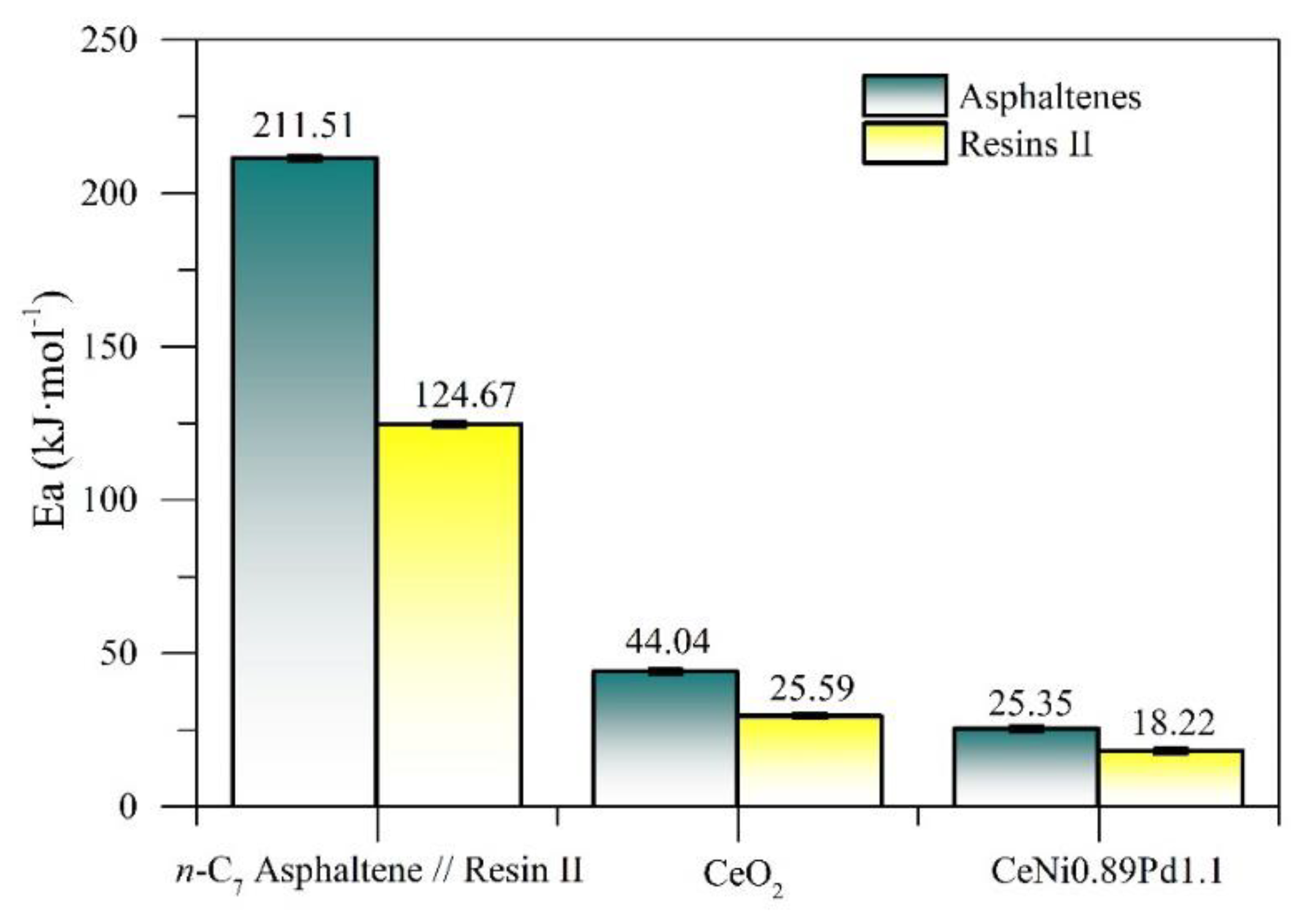
4.4. Effective Activation Energy
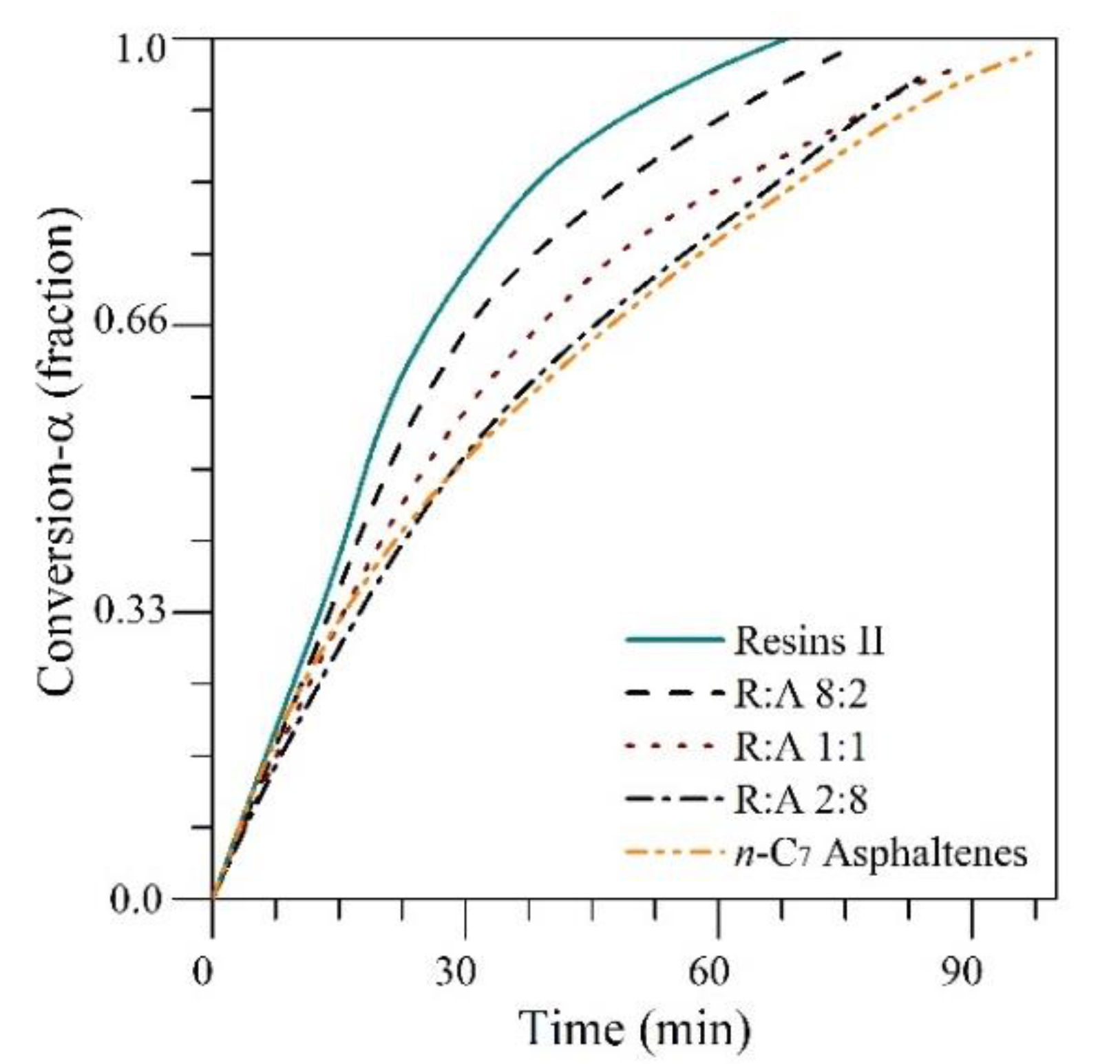
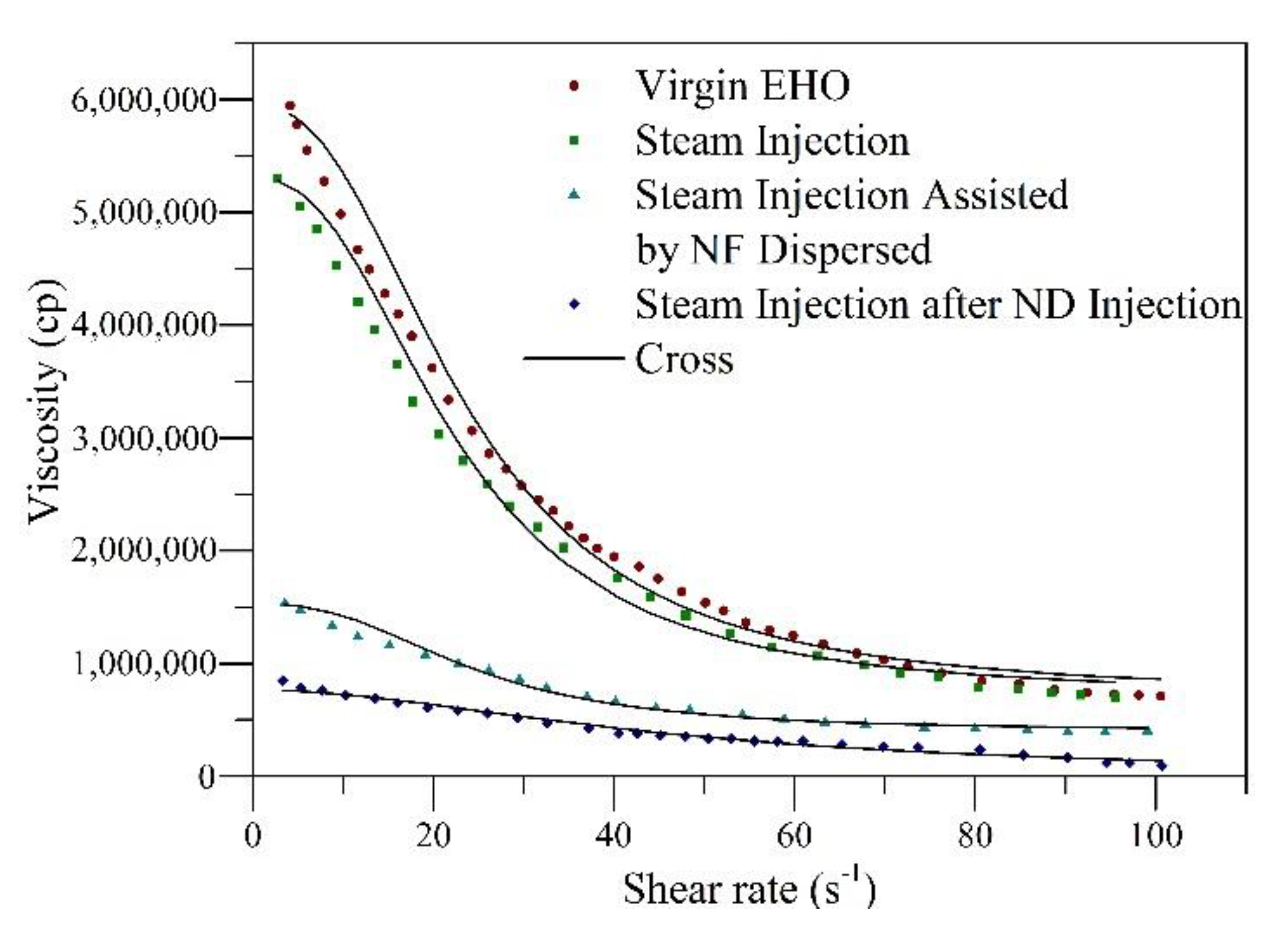
4.5. Dynamic Tests of Oil Recovery
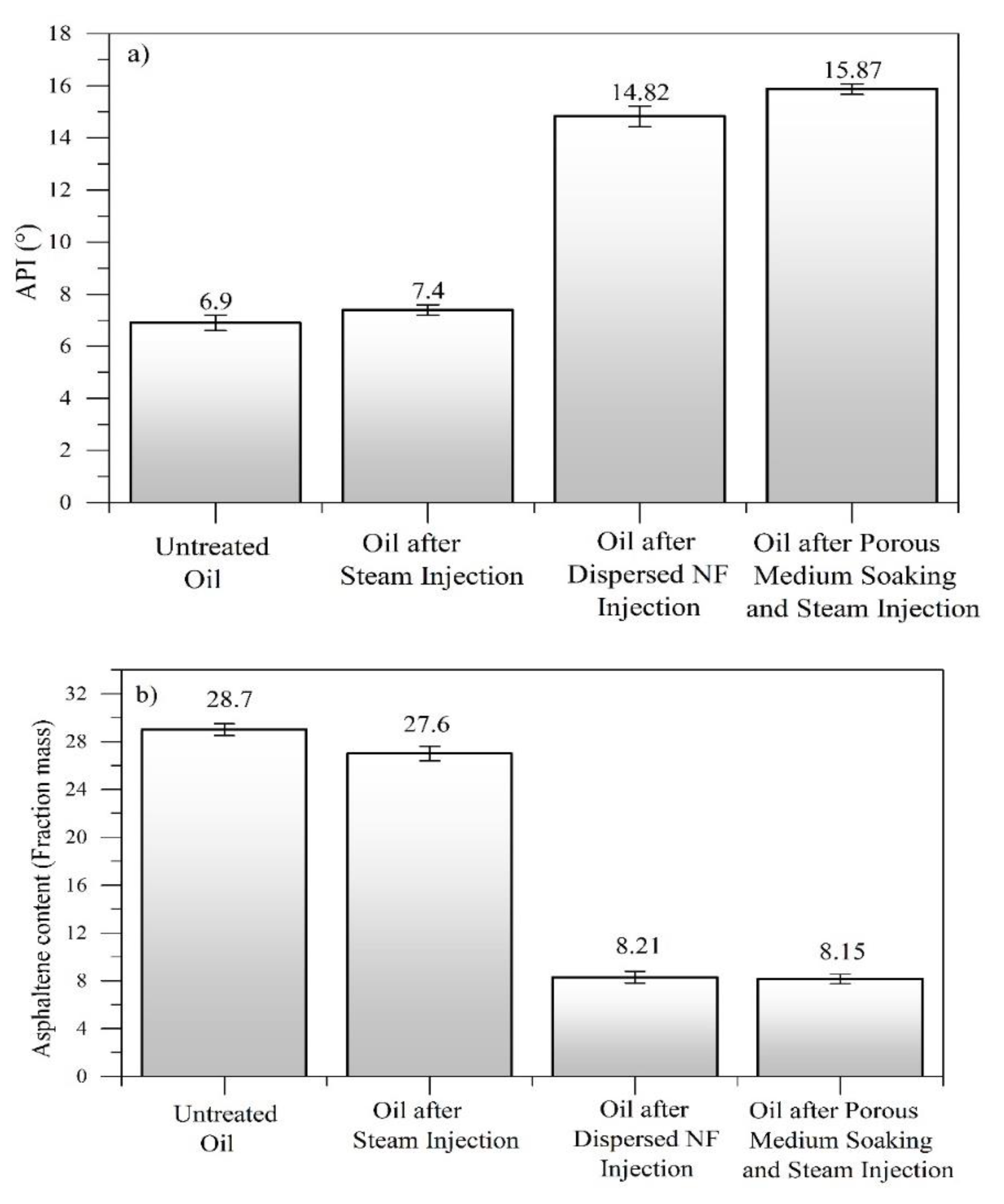
4.6. Crude Oil Characterization before and after Nanofluid Injection
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Patzek, T.; Koinis, M. Kern River steam-foam pilots. J. Petrol. Technol. 1990, 42, 496–503. [Google Scholar] [CrossRef]
- Yang, C.-Z.; Han, D.-K. Present status of EOR in the Chinese petroleum industry and its future. J. Petrol. Sci. Eng. 1991, 6, 175–189. [Google Scholar] [CrossRef]
- Thomas, S. Enhanced oil recovery-an overview. Oil Gas Sci. Technol. 2008, 63, 9–19. [Google Scholar] [CrossRef]
- Lu, C.; Liu, H.; Zhao, W. Visualized study of displacement mechanisms by injecting viscosity reducer and non-condensable gas to assist steam injection. J. Energy Inst. 2017, 90, 73–81. [Google Scholar] [CrossRef]
- Dong, X.; Liu, H.; Chen, Z.; Wu, K.; Lu, N.; Zhang, Q. Enhanced oil recovery techniques for heavy oil and oilsands reservoirs after steam injection. Appl. Energy 2019, 239, 1190–1211. [Google Scholar] [CrossRef]
- Rana, M.S.; Sámano, V.; Ancheyta, J.; Diaz, J. A review of recent advances on process technologies for upgrading of heavy oils and residua. Fuel 2007, 86, 1216–1231. [Google Scholar] [CrossRef]
- Metwally, M. Effect of gaseous additives on steam processes for Lindbergh field, Alberta. J. Can. Petrol. Technol. 1990, 29, 26–30. [Google Scholar] [CrossRef]
- Saboorian-Jooybari, H.; Dejam, M.; Chen, Z. Heavy oil polymer flooding from laboratory core floods to pilot tests and field applications: Half-century studies. J. Petrol. Sci. Eng. 2016, 142, 85–100. [Google Scholar] [CrossRef]
- Franco, C.; Cardona, L.; Lopera, S.; Mejía, J.; Cortés, F. Heavy oil upgrading and enhanced recovery in a continuous steam injection process assisted by nanoparticulated catalysts. In Proceedings of the SPE Improved Oil Recovery Conference, Tulsa, OK, USA, 11–13 April 2016. [Google Scholar]
- Abdrafikova, I.; Kayukova, G.; Petrov, S.; Ramazanova, A.; Musin, R.; Morozov, V. Conversion of extra-heavy Ashal’chinskoe oil in hydrothermal catalytic system. Pet. Chem. 2015, 55, 104–111. [Google Scholar] [CrossRef]
- Demirbas, A.; Alidrisi, H.; Balubaid, M. API gravity, sulfur content, and desulfurization of crude oil. Petrol. Sci. Technol. 2015, 33, 93–101. [Google Scholar] [CrossRef]
- Hashemi, R.; Nassar, N.N.; Almao, P.P. Nanoparticle technology for heavy oil in-situ upgrading and recovery enhancement: Opportunities and challenges. Appl. Energy 2014, 133, 374–387. [Google Scholar] [CrossRef]
- Wang, T.; Yang, W.; Wang, J.; Kalitaani, S.; Deng, Z. Low temperature oxidation of crude oil: Reaction progress and catalytic mechanism of metallic salts. Fuel 2018, 225, 336–342. [Google Scholar] [CrossRef]
- Hashemi, R.; Nassar, N.N.; Pereira Almao, P. Enhanced heavy oil recovery by in situ prepared ultradispersed multimetallic nanoparticles: A study of hot fluid flooding for Athabasca bitumen recovery. Energy Fuels 2013, 27, 2194–2201. [Google Scholar] [CrossRef]
- Cardona Rojas, L. Efecto de Nanopartículas en Procesos con Inyección de Vapor a Diferentes Calidades. Master’s Thesis, Universidad Nacional de Colombia-Sede Medellín, Medellín, Colombia, March 2018. [Google Scholar]
- Medina, O.E.; Gallego, J.; Arias-Madrid, D.; Cortés, F.B.; Franco, C.A. Optimization of the load of transition metal oxides (Fe2O3, Co3O4, NiO and/or PdO) onto CeO2 nanoparticles in catalytic steam decomposition of n-C7 asphaltenes at low temperatures. Nanomaterials 2019, 9, 401. [Google Scholar] [CrossRef] [PubMed]
- Medina, O.E.; Gallego, J.; Restrepo, L.G.; Cortés, F.B.; Franco, C.A. Influence of the Ce4+/Ce3+ Redox-couple on the cyclic regeneration for adsorptive and catalytic performance of NiO-PdO/CeO2±δ nanoparticles for n-C7 asphaltene steam gasification. Nanomaterials 2019, 9, 734. [Google Scholar] [CrossRef] [PubMed]
- Cardona, L.; Arias-Madrid, D.; Cortés, F.; Lopera, S.; Franco, C. Heavy oil upgrading and enhanced recovery in a steam injection process assisted by NiO-and PdO-Functionalized SiO2 nanoparticulated catalysts. Catalysts 2018, 8, 132. [Google Scholar] [CrossRef]
- Nassar, N.N.; Franco, C.A.; Montoya, T.; Cortés, F.B.; Hassan, A. Effect of oxide support on Ni–Pd bimetallic nanocatalysts for steam gasification of n-C7 asphaltenes. Fuel 2015, 156, 110–120. [Google Scholar] [CrossRef]
- Alamolhoda, S.; Vitale, G.; Hassan, A.; Nassar, N.N.; Almao, P.P. Synergetic effects of cerium and nickel in Ce-Ni-MFI catalysts on low-temperature water-gas shift reaction. Fuel 2019, 237, 361–372. [Google Scholar] [CrossRef]
- Wang, X.; Chen, J.; Zeng, J.; Wang, Q.; Li, Z.; Qin, R.; Wu, C.; Xie, Z.; Zheng, L. The synergy between atomically dispersed Pd and cerium oxide for enhanced catalytic properties. Nanoscale 2017, 9, 6643–6648. [Google Scholar] [CrossRef]
- Vignatti, C.I.; Avila, M.S.; Apesteguia, C.R.; Garetto, T.F. Study of the water-gas shift reaction over Pt supported on CeO2–ZrO2 mixed oxides. Catal. Today 2011, 171, 297–303. [Google Scholar] [CrossRef]
- Franco, C.A.; Montoya, T.; Nassar, N.N.; Pereira-Almao, P.; Cortés, F.B. Adsorption and subsequent oxidation of colombian asphaltenes onto nickel and/or palladium oxide supported on fumed silica nanoparticles. Energy Fuels 2013, 27, 7336–7347. [Google Scholar] [CrossRef]
- Yi, S.; Babadagli, T.; Li, H.A. Use of nickel nanoparticles for promoting aquathermolysis reaction during cyclic steam stimulation. SPE J. 2018, 23, 145–156. [Google Scholar] [CrossRef]
- Afzal, S.; Nikookar, M.; Ehsani, M.R.; Roayaei, E. An experimental investigation of the catalytic effect of Fe2O3 nanoparticle on steam injection process of an Iranian reservoir. Iran. J. Oil Gas Sci. Technol. 2014, 3, 27–36. [Google Scholar]
- Franco, C.A.; Lozano, M.M.; Acevedo, S.; Nassar, N.N.; Cortés, F.B. Effects of resin I on asphaltene adsorption onto nanoparticles: A novel method for obtaining asphaltenes/resin isotherms. Energy Fuels 2015, 30, 264–272. [Google Scholar] [CrossRef]
- Hashemi, R.; Nassar, N.N.; Pereira Almao, P. In situ upgrading of Athabasca bitumen using multimetallic ultradispersed nanocatalysts in an oil sands packed-bed column: Part 1. Produced liquid quality enhancement. Energy Fuels 2013, 28, 1338–1350. [Google Scholar] [CrossRef]
- Franco, C.A.; Zabala, R.D.; Zapata, J.; Mora, E.; Botero, O.; Candela, C.; Castillo, A. Inhibited gas stimulation to mitigate condensate banking and maximize recovery in cupiagua field. SPE Prod. Oper. 2013, 28, 154–167. [Google Scholar] [CrossRef]
- Restrepo, A.; Ocampo, A.; Lopera Castro, S.H.; Diaz, M.P.; Clavijo, J.; Marin, J. GaStim concept—A novel technique for well stimulation. Part I: Understanding the Physics. In Proceedings of the SPE Latin America and Caribbean Petroleum Engineering Conference, Mexico City, Mexico, 16–18 April 2012. [Google Scholar] [CrossRef]
- Medina, O.E.; Gallego, J.; Rodriguez, E.; Franco, C.A.; Cortés, F.B. Effect of pressure on the oxidation kinetics of Asphaltenes. Energy Fuels 2019, 33, 10734–10744. [Google Scholar] [CrossRef]
- Delannoy, L.; El Hassan, N.; Musi, A.; Le To, N.N.; Krafft, J.-M.; Louis, C. Preparation of supported gold nanoparticles by a modified incipient wetness impregnation method. J. Phys. Chem. B 2006, 110, 22471–22478. [Google Scholar] [CrossRef]
- Nassar, N.N.; Montoya, T.; Franco, C.A.; Cortés, F.B.; Pereira-Almao, P. A new model for describing the adsorption of asphaltenes on porous media at a high pressure and temperature under flow conditions. Energy Fuels 2015, 29, 4210–4221. [Google Scholar] [CrossRef]
- Cortés, F.B.; Montoya, T.; Acevedo, S.; Nassar, N.N.; Franco, C.A. Adsorption-desorption of n-c7 asphaltenes over micro-and nanoparticles of silica and its impact on wettability alteration. CT&F-Ciencia Tecnología y Futuro 2016, 6, 89–106. [Google Scholar]
- Hurtado, Y.; Beltrán, C.; Zabala, R.D.; Lopera, S.H.; Franco, C.A.; Nassar, N.N.; Cortés, F.B. Effects of surface acidity and polarity of SiO2 nanoparticles on the foam stabilization applied to natural gas flooding in tight gas-condensate reservoirs. Energy Fuels 2018, 32, 5824–5833. [Google Scholar] [CrossRef]
- Betancur, S.a.; Carrasco-Marín, F.; Franco, C.A.; Cortés, F.B. Development of composite materials based on the interaction between nanoparticles and surfactants for application in chemical enhanced oil recovery. Ind. Eng. Chem. Res. 2018, 57, 12367–12377. [Google Scholar] [CrossRef]
- Lozano, M.M.; Franco, C.A.; Acevedo, S.A.; Nassar, N.N.; Cortés, F.B. Effects of resin I on the catalytic oxidation of n-C 7 asphaltenes in the presence of silica-based nanoparticles. RSC Adv. 2016, 6, 74630–74642. [Google Scholar] [CrossRef]
- ASTM D36. Standard Test Method for Softening Point of Bitumen (Ring-and-Ball Apparatus); Annual Book of ASTM Standards; ASTM International: West Conshohocken, PA, USA, 2006. [Google Scholar]
- Franco, C.A.; Montoya, T.; Nassar, N.N.; Cortés, F.B. NiO and PdO Supported on fumed silica nanoparticles for adsorption and catalytic steam gasification of colombian n-C7 asphaltenes. In Handbook on Oil Production Research; Nova Science Publishers: Hauppauge, NY, USA, 2014; pp. 101–145. [Google Scholar]
- Whitaker, S. Flow in porous media I: A theoretical derivation of Darcy’s law. Transp. Porous Med. 1986, 1, 3–25. [Google Scholar] [CrossRef]
- Incropera, F.P.; Lavine, A.S.; Bergman, T.L.; DeWitt, D.P. Fundamentals of Heat and Mass Transfer; Wiley: Hoboken, NJ, USA, 2007. [Google Scholar]
- Bansal, V.; Krishna, G.; Chopra, A.; Sarpal, A. Detailed hydrocarbon characterization of RFCC feed stocks by NMR spectroscopic techniques. Energy Fuels 2007, 21, 1024–1029. [Google Scholar] [CrossRef]
- Montoya, T.; Coral, D.; Franco, C.A.; Nassar, N.N.; Cortés, F.B. A novel solid–liquid equilibrium model for describing the adsorption of associating asphaltene molecules onto solid surfaces based on the “chemical theory”. Energy Fuels 2014, 28, 4963–4975. [Google Scholar] [CrossRef]
- Nassar, N.N.; Hassan, A.; Luna, G.; Pereira-Almao, P. Kinetics of the catalytic thermo-oxidation of asphaltenes at isothermal conditions on different metal oxide nanoparticle surfaces. Catal. Today 2013, 207, 127–132. [Google Scholar] [CrossRef]
- Schneider, P. Adsorption isotherms of microporous-mesoporous solids revisited. Appl. Catal. A 1995, 129, 157–165. [Google Scholar] [CrossRef]
- Bates, M.K.; Jia, Q.; Doan, H.; Liang, W.; Mukerjee, S. Charge-transfer effects in Ni–Fe and Ni–Fe–Co mixed-metal oxides for the alkaline oxygen evolution reaction. ACS Catal. 2015, 6, 155–161. [Google Scholar] [CrossRef]
- Dong, Y.-B.; Jin, G.-X.; Smith, M.D.; Huang, R.-Q.; Tang, B.; zur Loye, H.-C. [Ag2(C33H26N2O2)(H2O)2(SO3CF3)2]·0.5C6H6: A Luminescent Supramolecular Silver (I) Complex Based on Metal−Carbon and Metal−Heteroatom Interactions. Inorg. Chem. 2002, 41, 4909–4914. [Google Scholar] [CrossRef]
- Ignasiak, T.; Kemp-Jones, A.; Strausz, O. The molecular structure of Athabasca asphaltene. Cleavage of the carbon-sulfur bonds by radical ion electron transfer reactions. J. Org. Chem. 1977, 42, 312–320. [Google Scholar] [CrossRef]
- Leon, O.; Rogel, E.; Espidel, J.; Torres, G. Asphaltenes: Structural characterization, self-association, and stability behavior. Energy Fuels 2000, 14, 6–10. [Google Scholar] [CrossRef]
- Praharaj, S.; Nath, S.; Ghosh, S.K.; Kundu, S.; Pal, T. Immobilization and recovery of Au nanoparticles from anion exchange resin: Resin-bound nanoparticle matrix as a catalyst for the reduction of 4-nitrophenol. Langmuir 2004, 20, 9889–9892. [Google Scholar] [CrossRef] [PubMed]
- Aguilera-Mercado, B.; Herdes, C.; Murgich, J.; Müller, E. Mesoscopic simulation of aggregation of asphaltene and resin molecules in crude oils. Energy Fuels 2006, 20, 327–338. [Google Scholar] [CrossRef]
- Brewer, L.; Wengert, P.R. Erratum to: Transition metal alloys of extraordinary stability; An example of generalized Lewis-acid-base interactions in metallic systems. Metall. Trans. 1973, 4, 83–104. [Google Scholar] [CrossRef]
- Murgich, J.; Rodríguez, J.; Aray, Y. Molecular recognition and molecular mechanics of micelles of some model asphaltenes and resins. Energy Fuels 1996, 10, 68–76. [Google Scholar] [CrossRef]
- León, O.; Contreras, E.; Rogel, E.; Dambakli, G.; Acevedo, S.; Carbognani, L.; Espidel, J. Adsorption of native resins on asphaltene particles: A correlation between adsorption and activity. Langmuir 2002, 18, 5106–5112. [Google Scholar] [CrossRef]
- Jacobs, G.; Ricote, S.; Graham, U.M.; Patterson, P.M.; Davis, B.H. Low temperature water gas shift: Type and loading of metal impacts forward decomposition of pseudo-stabilized formate over metal/ceria catalysts. Catal. Today 2005, 106, 259–264. [Google Scholar] [CrossRef]
- Alvarez-Ramirez, F.; Ramirez-Jaramillo, E.; Ruiz-Morales, Y. Calculation of the interaction potential curve between asphaltene-asphaltene, asphaltene-resin, and resin-resin systems using density functional theory. Energy Fuels 2006, 20, 195–204. [Google Scholar] [CrossRef]
- Merino-Garcia, D.; Andersen, S.I. Thermodynamic characterization of asphaltene-resin interaction by microcalorimetry. Langmuir 2004, 20, 4559–4565. [Google Scholar] [CrossRef]
- Acevedo, S.; Zuloaga, C.; Rodríguez, P. Aggregation-dissociation studies of asphaltene solutions in resins performed using the combined freeze fracture-transmission electron microscopy technique. Energy Fuels 2008, 22, 2332–2340. [Google Scholar] [CrossRef]
- Kuang, W.; Rives, A.; Tayeb, B.O.B.; Fournier, M.; Hubaut, R. Isomerization of n-Hexane over silica-supported heteropoly acids promoted by the reduced Ce-Ni Oxides. J. Colloid Interf. Sci. 2002, 248, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Hosseinpour, M.; Fatemi, S.; Ahmadi, S.J. Catalytic cracking of petroleum vacuum residue in supercritical water media: Impact of α-Fe2O3 in the form of free nanoparticles and silica-supported granules. Fuel 2015, 159, 538–549. [Google Scholar] [CrossRef]
- Lamonier, C.; Ponchel, A.; D’huysser, A.; Jalowiecki-Duhamel, L. Studies of the cerium-metal-oxygen-hydrogen system (metal = Cu, Ni). Catal. Today 1999, 50, 247–259. [Google Scholar] [CrossRef]
- Pinc, W.; Yu, P.; O’Keefe, M.; Fahrenholtz, W. Effect of gelatin additions on the corrosion resistance of cerium based conversion coatings spray deposited on Al 2024-T3. Surf. Coat. Technol. 2009, 23, 3533–3540. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Ikeda, N.; Hattori, H.; Tanabe, K. Surface and catalytic properties of cerium oxide. J. Catal. 1981, 67, 324–330. [Google Scholar] [CrossRef]
- Taborda, E.A.; Franco, C.A.; Lopera, S.H.; Alvarado, V.; Cortés, F.B. Effect of nanoparticles/nanofluids on the rheology of heavy crude oil and its mobility on porous media at reservoir conditions. Fuel 2016, 184, 222–232. [Google Scholar] [CrossRef]


| Elemental Mass Fraction Concentration (%) | |||||
|---|---|---|---|---|---|
| Fraction | Carbon | Hydrogen | Nitrogen | Oxygen | Sulphur |
| Resin II | 82.10 | 9.00 | 0.54 | 2.26 | 6.10 |
| n-C7 Asphaltenes | 81.70 | 7.80 | 0.33 | 3.57 | 6.60 |
| Property | Value |
|---|---|
| Mineralogy | 99% Silica |
| Length | 67 cm |
| Diameter | 3.8 cm |
| Porosity | 26.5% |
| Porous Volume | 194 cm3 |
| Initial oil saturation state | 77.4% |
| Initial water saturation state | 22.6% |
| Sample | n-C7 Asphaltenes | Resin II | |||||||
|---|---|---|---|---|---|---|---|---|---|
| R:A | (mg·g−1) × 10−2 | (g·g−1) × 10−2 | qm (mg·m−2) | % RSM | (mg·g−1) × 10−2 | (g·g−1) × 10−2 | qm (mg·m−2) | % RSM | |
| CeNi0.89Pd1.1 | Individual | 6.02 | 3.91 | 0.22 | 0.01 | 3.26 | 0.48 | 0.19 | 0.1 |
| CeO2 | Individual | 10.12 | 20.7 | 0.12 | 0.02 | 4.03 | 7.62 | 18.57 | 0.1 |
| CeNi0.89Pd1.1 | 2:8 | 6.70 | 4.32 | 0.16 | 0.01 | 1552 | 8.81 | 25.66 | 0.2 |
| 1:1 | 10.61 | 4.66 | 0.11 | 0.02 | 985.1 | 7.66 | 0.22 | 0.1 | |
| 8:2 | 1343 | 5.77 | 27.17 | 0.01 | 3.33 | 2.33 | 0.25 | 0.2 | |
| R:A Ratio | Slope | Intercept | |
|---|---|---|---|
| 2:8 | 1.04 | 0.007 | 0.99 |
| 1:1 | 1.06 | −0.002 | 0.98 |
| 8:2 | 1.04 | −0.007 | 0.99 |
| Stage | Swr (%) | Sor (%) |
|---|---|---|
| Steam Injection | 62.3 | 24.0 |
| Steam injection assisted by ND | 81.5 | 14.0 |
| Steam injection after ND injection | 94.6 | 7.0 |
| Parameters | Virgin EHO | EHO after Steam Injection | EHO during Nanofluid Dispersed Injection | EHO Post Nanofluid Dispersed Injection |
|---|---|---|---|---|
| × 106 | 5.95 | 5.31 | 0.55 | 0.27 |
| × 106 | 7.10 | 7.04 | 1.60 | 0.98 |
| × 10−2 | 4.48 | 4.44 | 4.32 | 4.01 |
| 0.87 | 0.83 | 0.45 | 0.38 | |
| 0.34 | 0.29 | 0.21 | 0.33 | |
| DVR (%) | - | 10 | 75 | 85 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Medina, O.E.; Caro-Vélez, C.; Gallego, J.; Cortés, F.B.; Lopera, S.H.; Franco, C.A. Upgrading of Extra-Heavy Crude Oils by Dispersed Injection of NiO–PdO/CeO2±δ Nanocatalyst-Based Nanofluids in the Steam. Nanomaterials 2019, 9, 1755. https://doi.org/10.3390/nano9121755
Medina OE, Caro-Vélez C, Gallego J, Cortés FB, Lopera SH, Franco CA. Upgrading of Extra-Heavy Crude Oils by Dispersed Injection of NiO–PdO/CeO2±δ Nanocatalyst-Based Nanofluids in the Steam. Nanomaterials. 2019; 9(12):1755. https://doi.org/10.3390/nano9121755
Chicago/Turabian StyleMedina, Oscar E., Cristina Caro-Vélez, Jaime Gallego, Farid B. Cortés, Sergio H. Lopera, and Camilo A. Franco. 2019. "Upgrading of Extra-Heavy Crude Oils by Dispersed Injection of NiO–PdO/CeO2±δ Nanocatalyst-Based Nanofluids in the Steam" Nanomaterials 9, no. 12: 1755. https://doi.org/10.3390/nano9121755
APA StyleMedina, O. E., Caro-Vélez, C., Gallego, J., Cortés, F. B., Lopera, S. H., & Franco, C. A. (2019). Upgrading of Extra-Heavy Crude Oils by Dispersed Injection of NiO–PdO/CeO2±δ Nanocatalyst-Based Nanofluids in the Steam. Nanomaterials, 9(12), 1755. https://doi.org/10.3390/nano9121755








