The Effect of the Antioxidant Activity of Plant Extracts on the Properties of Gold Nanoparticles
Abstract
1. Introduction
2. Experimental
2.1. Chemicals and Reagents
2.2. Apparatus
2.3. Preparation of Leaf Extracts
2.4. Determination of AOA of the Leaf Extracts
2.5. Synthesis of AuNPs
2.6. UV-Vis Spectrophotometric Measurements
2.7. High Resolution Transmission Electron Microscopy Measurements
2.8. Dynamic Light Scattering Measurements
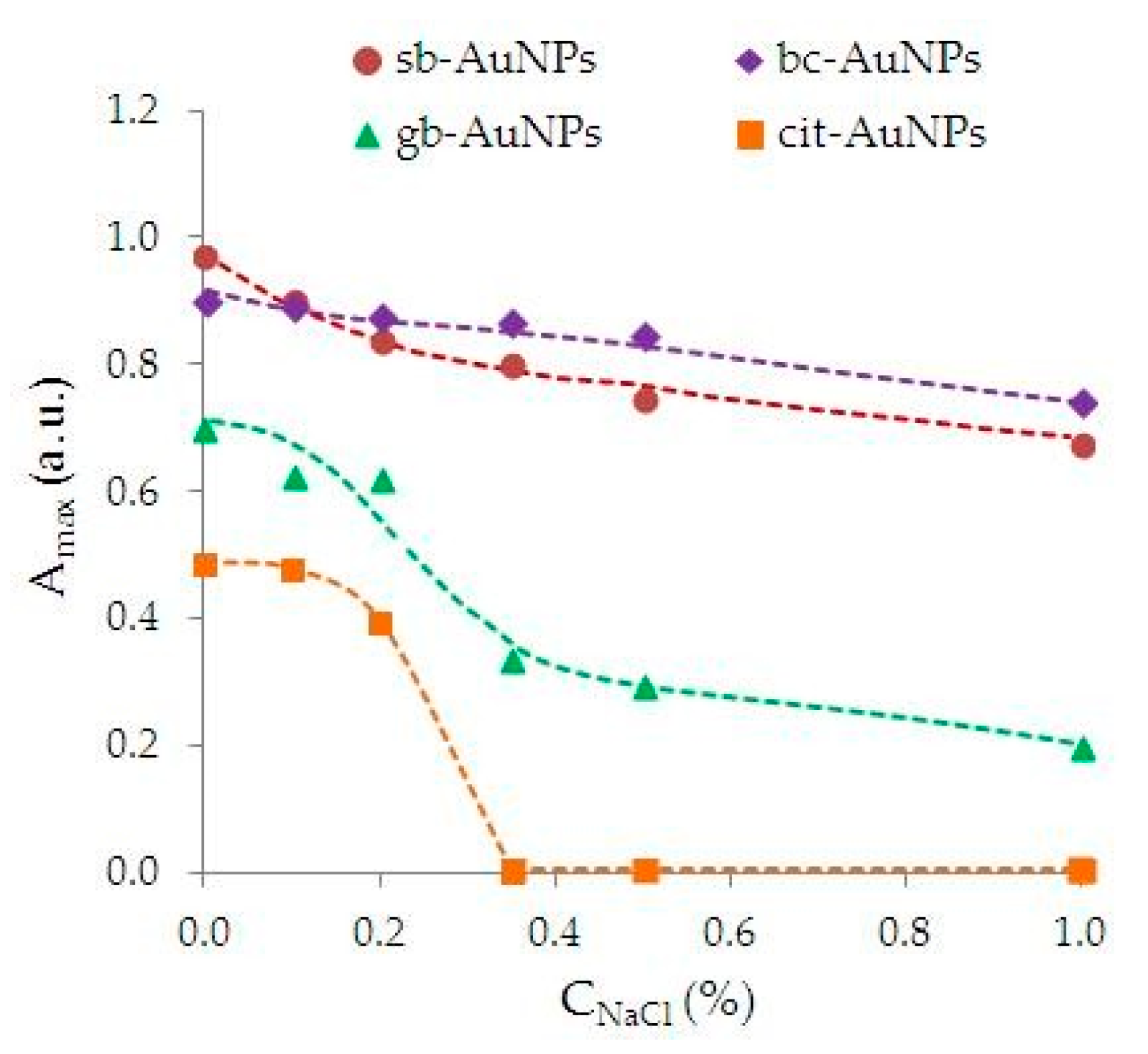
2.9. Assessment of the Aggregate Stability of AuNPs Suspensions
2.10. Data Treatment
3. Results and Discussions
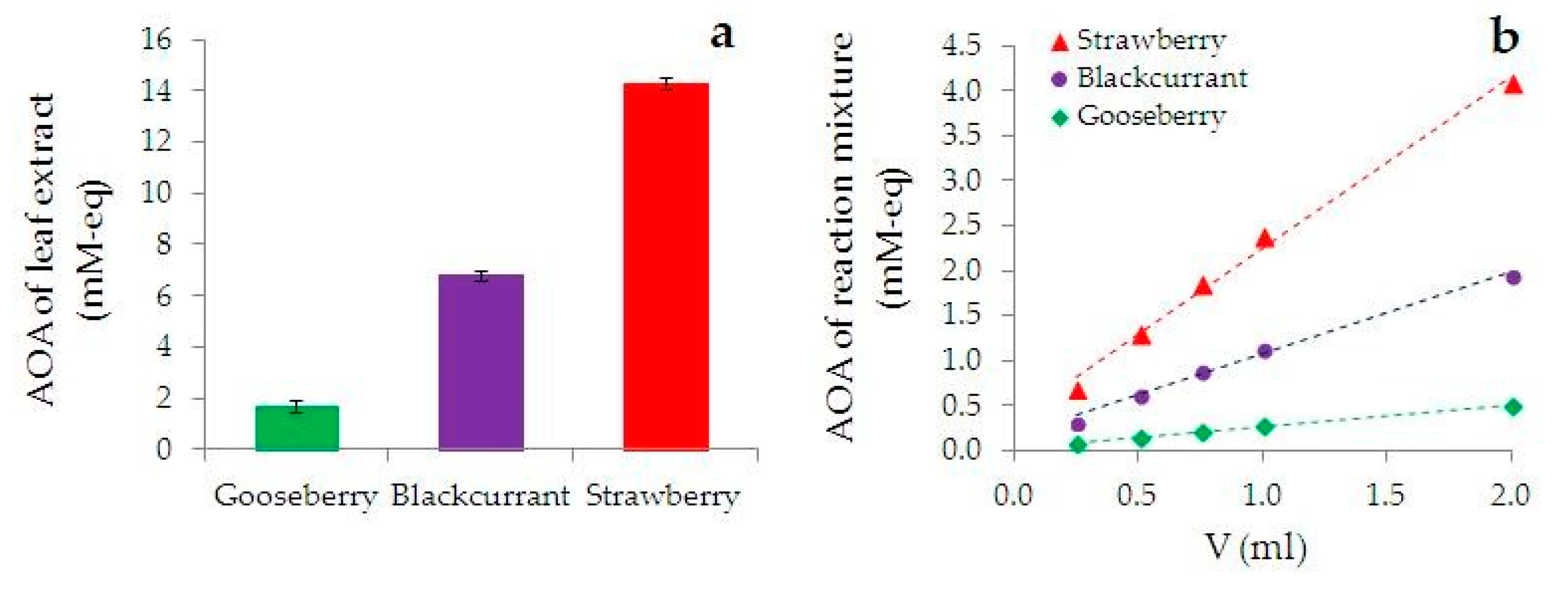
3.1. Characterization of AOA of the Leaf Extracts and Reaction Mixtures by HPM
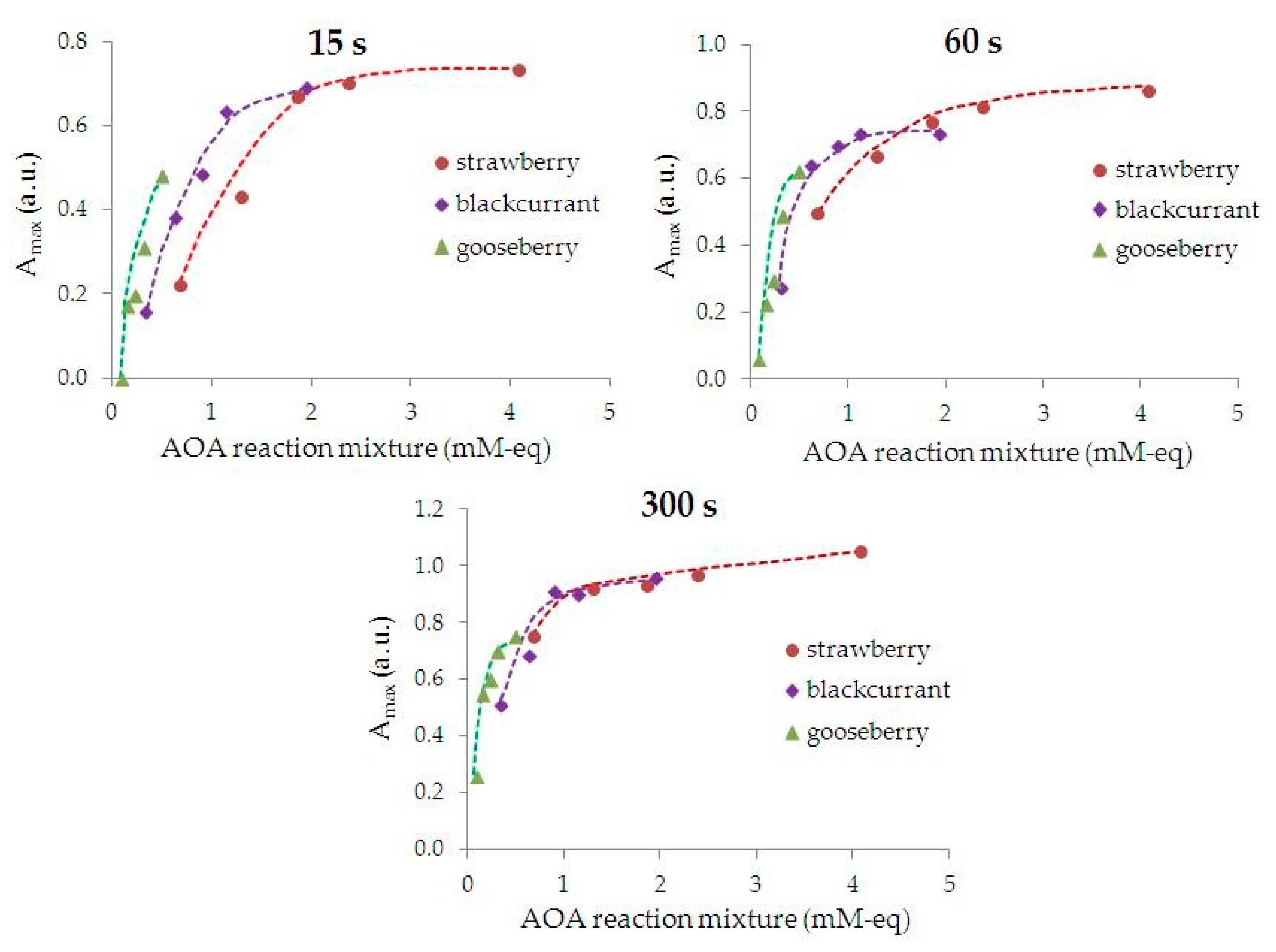
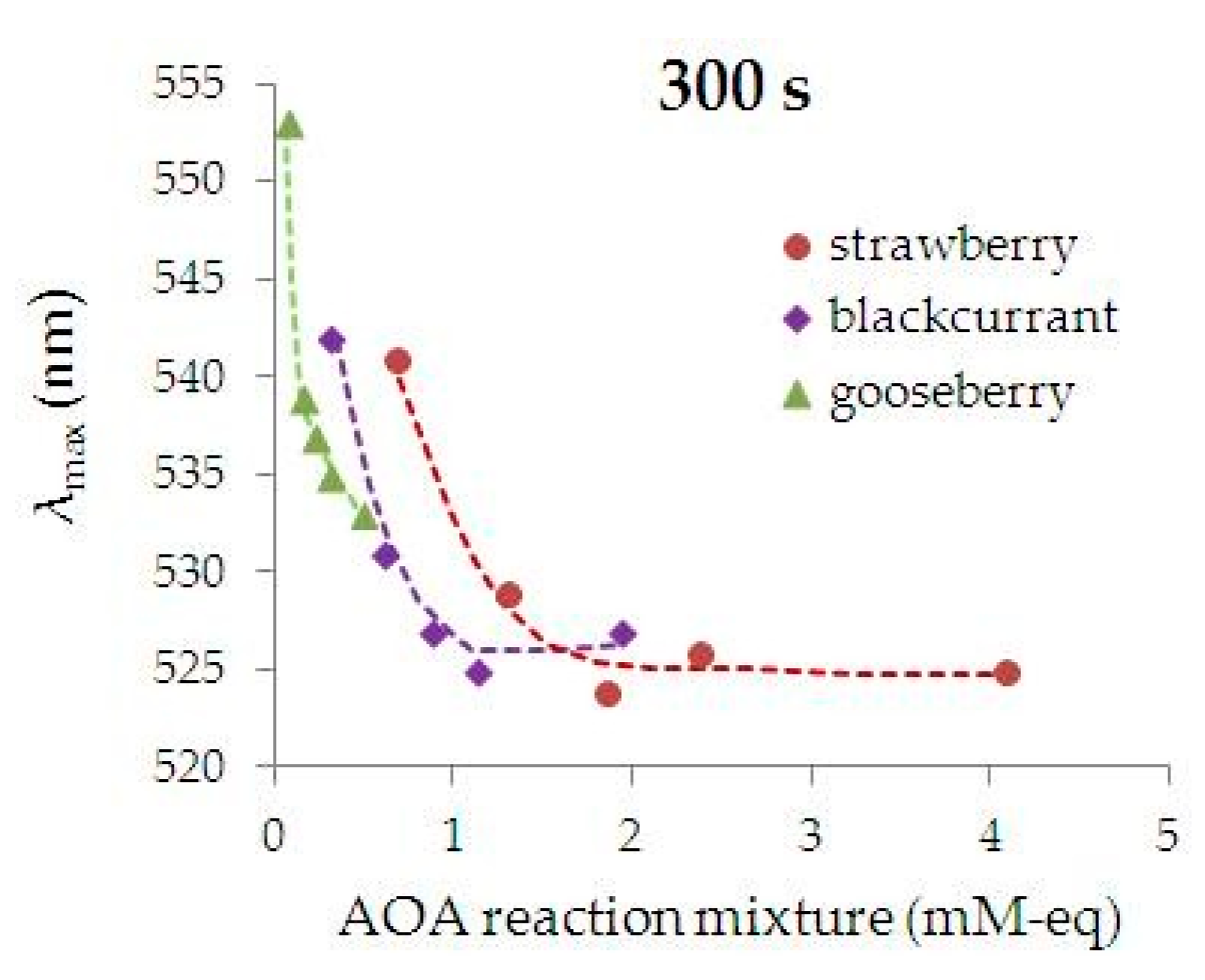
3.2. Characterization of the Kinetics and Phytosynthesis Completeness Using UV-Vis Spectrophotometry
- −
- the rate in section (II) of the kinetic curve is significantly higher than in section (III);
- −
- an increase in AOA of the reaction mixture leads to an increase in the phyto-AuNPs’ suspension formation rate at the growth stage (II), for example, the phyto-AuNPs’ suspension formation rate increases by 4.5 times with an increase in AOA of the reaction mixture of gooseberry from 0.08 to 0.49 mM-eq;
- −
- the higher AOA of the extract from the plant, the greater the rate of the phyto-AuNPs’ suspension formation. Thus, an increase in AOA of the extract in the row of gooseberry (Ribes uva-crispa) < blackcurrant (Ribes nigrum) < strawberry (Fragaria vesca), leads to an increase in the rate of the phyto-AuNPs’ suspension formation. Thus, the rate of formation of AuNPs suspensions in reaction mixtures containing 0.25 mL of gooseberry, currant and strawberry extract is 1.0; 2.2 and 5.7 ms−1, respectively.
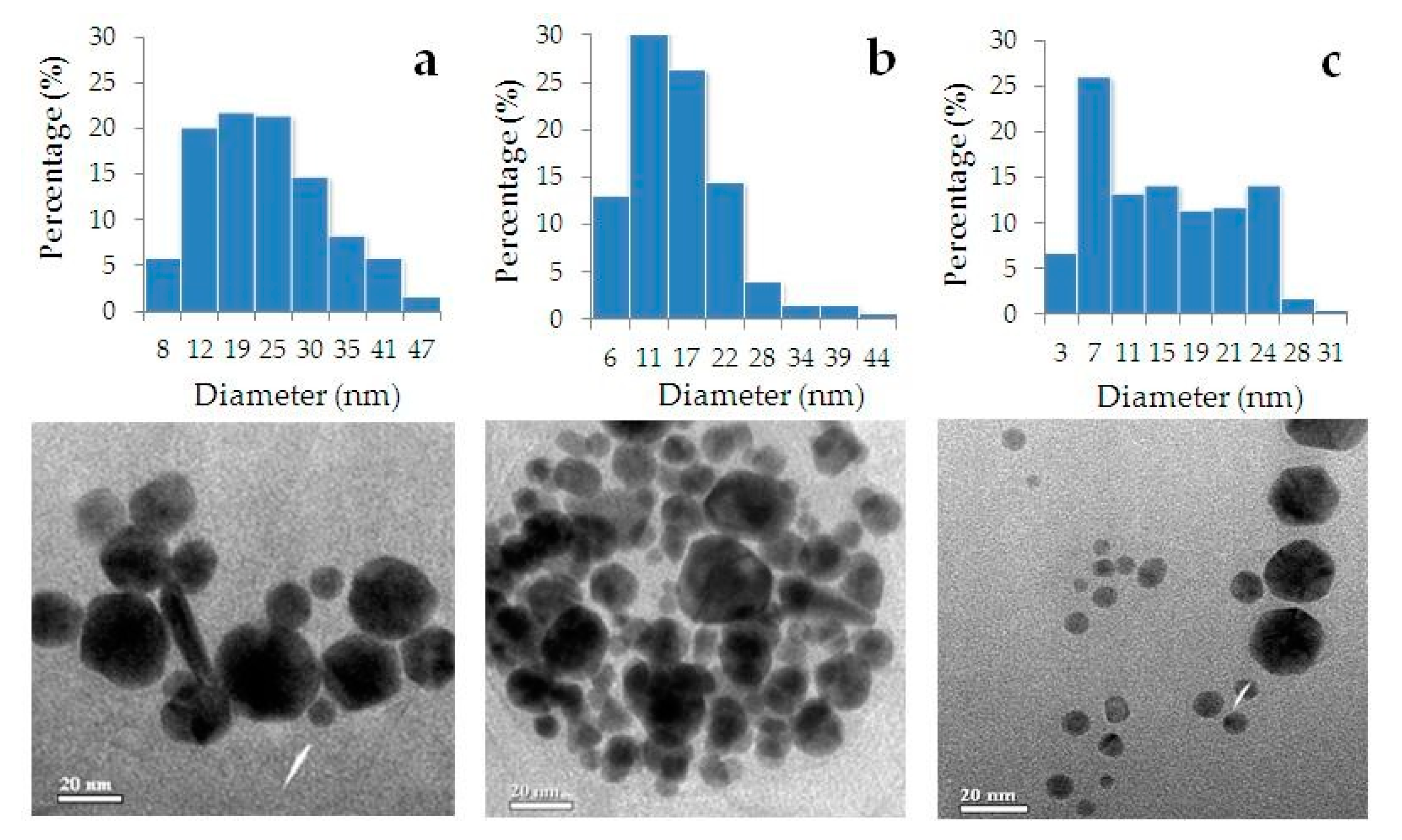
3.3. Characteristics of Phyto-AuNPs
4. Conclusions
- −
- the rate of phyto-AuNPs formation (phytosynthesis rate) increases;
- −
- the size of phyto-AuNPs decreases;
- −
- the fraction of small phyto-AuNPs (d ≤ 5 nm) increases and the fraction of large phyto-AuNPs (d ≥ 31–50 nm) decreases;
- −
- the stability of phyto-AuNPs suspensions increases.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kizling, M.; Dzwonek, M.; Wieckowska, A.; Bilewicz, R. Gold nanoparticles in bioelectrocatalysis—The role of nanoparticle size. Curr. Opin. Electrochem. 2018, 12, 113–120. [Google Scholar] [CrossRef]
- Kim, B.H.; Hackett, M.J.; Park, J.; Hyeon, T. Synthesis, characterization, and application of ultrasmall nanoparticles. Chem. Mater. 2014, 26, 59–71. [Google Scholar] [CrossRef]
- Brainina, K.; Stozhko, N.; Bukharinova, M.; Vikulova, E. Nanomaterials: Electrochemical properties and application in sensors. Phys. Sci. Rev. 2018, 3, 8050. [Google Scholar] [CrossRef]
- Jindal, B. The effect of particle shape on cellular interaction and drug delivery applications of micro-and nanoparticles. Int. J. Pharm. 2017, 532, 450–465. [Google Scholar] [CrossRef]
- Ullah, R.; Bakht, J.; Shah, M.R.; Shafi, M. Bioinspired synthesis and characterization of gold nano-particles from medicinally important Periploca hydaspidis and their in vitro antioxidant and antimicrobial activity. Pak. J. Pharm. Sci. 2019, 32, 1069–1080. [Google Scholar]
- Kumar, P.V.; Kala, S.M.J.; Prakash, K.S. Green synthesis of gold nanoparticles using Croton Caudatus Geisel leaf extract and their biological studies. Mater. Lett. 2019, 236, 19–22. [Google Scholar] [CrossRef]
- Usman, I.; Aziz, A.A.; Abu, N.O. Application of green synthesis of gold nanoparticles: A review. J. Teknol. 2019, 81, 171–182. [Google Scholar] [CrossRef]
- Khan, S.; Bakht, J.; Syed, F. Green synthesis of gold nanoparticles using acer pentapomicum leaves extract its characterization, antibacterial, antifungal and antioxidant bioassay. Dig. J. Nanomater. Biostruct. 2018, 13, 579–589. [Google Scholar]
- Divakaran, D.; Lakkakula, J.R.; Thakur, M.; Kumawat, M.K.; Srivastava, R. Dragon fruit extract capped gold nanoparticles: Synthesis and their differential cytotoxicity effect on breast cancer cells. Mater. Lett. 2019, 236, 498–502. [Google Scholar] [CrossRef]
- Ahn, E.-Y.; Hwang, S.J.; Choi, M.J.; Cho, S.; Lee, H.-J.; Park, Y. Upcycling of jellyfish (Nemopilema nomurai) sea wastes as highly valuable reducing agents for green synthesis of gold nanoparticlesand their antitumor and anti-inflammatory activity. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1127–1136. [Google Scholar] [CrossRef]
- Ahmad, N.; Bhatnagar, S.; Saxena, R.; Iqbal, D.; Ghosh, A.K.; Dutta, R. Biosynthesis and characterization of gold nanoparticles: Kinetics, in vitro and in vivo study. Mater. Sci. Eng. C 2017, 78, 553–564. [Google Scholar] [CrossRef] [PubMed]
- Umamaheswari, C.; Lakshmanan, A.; Nagarajan, N.S. Phyto-mediated synthesis, biological and catalytic activity studies of gold nanoparticles. IET Nanobiotechnol. 2018, 12, 166–174. [Google Scholar] [CrossRef]
- Filip, G.A.; Moldovan, B.; Baldea, I.; Olteanu, D.; Suharoschi, R.; Decea, N.; Cismaru, C.M.; Gal, E.; Cenariu, M.; Clichici, S.; et al. UV-light mediated green synthesis of silver and gold nanoparticlesusing Cornelian cherry fruit extract and their comparative effects in experimental inflammation. J. Photochem. Photobiol. B 2019, 191, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, K.S.; Husen, A. Recent advances in plant-mediated engineered gold nanoparticles and their application in biological system. J. Trace Elem. Med. Biol. 2017, 40, 10–23. [Google Scholar] [CrossRef]
- Chellapandian, C.; Ramkumar, B.; Puja, P.; Shanmuganathan, R.; Pugazhendhi, A.; Kumar, P. Gold nanoparticles using red seaweed Gracilaria verrucosa: Green synthesis, characterization and biocompatibility studies. Process Biochem. 2019, 80, 58–63. [Google Scholar] [CrossRef]
- Timoszyk, A. A review of the biological synthesis of gold nanoparticles using fruit extracts: Scientific potential and application. Bull. Mater. Sci. 2018, 41, 154. [Google Scholar] [CrossRef]
- Kumar, K.P.; Paul, W.; Sharma, C.P. Green synthesis of gold nanoparticles with Zingiber officinale extract: Characterization and blood compatibility. Process Biochem. 2011, 46, 2007–2013. [Google Scholar] [CrossRef]
- Xiang, Z.; Wang, K.J.; Zhang, W.Z.; Teh, S.W.; Peli, A.; Mok, P.L.; Higuchi, A.; Kumar, S.S. Gold Nanoparticles Inducing Osteogenic Differentiation of Stem Cells: A Review. J. Clust. Sci. 2018, 29, 1–7. [Google Scholar] [CrossRef]
- Anand, K.; Tiloke, C.; Pragalathan, N.; Chuturgoon, A.A. Chuturgoon Phytonanotherapy for management of diabetes using green synthesis nanoparticles. J. Photochem. Photobiol. B 2017, 173, 626–639. [Google Scholar] [CrossRef]
- Sathishkumar, P.; Gu, F.L.; Zhan, Q.; Palvannan, T.; Yusof, A.R.M. Flavonoids mediated ‘Green’ nanomaterials: A novel nanomedicine system to treat various diseases—Current trends and future perspective. Mater. Lett. 2018, 210, 26–30. [Google Scholar] [CrossRef]
- Rajasekharreddy, P.; Huang, C.; Busi, S.; Rajkumari, J.; Tai, M.-H.; Liu, G. Green Synthesized nanomaterials as theranostic platforms for cancer yreatment: Principles, challenges and the road ahead. Curr. Med. Chem. 2019, 26, 1311–1327. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Sau, S.; Madhuri, D.; Bollu, V.S.; Madhusudana, K.; Sreedhar, B.; Banerjee, R.; Patra, C.R. Green synthesis and characterization of monodispersed gold nanoparticles: Toxicity study, delivery of doxorubicin and its biodistribution in mouse model. J. Biomed. Nanotechnol. 2016, 12, 165–181. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Annu; Ikram, S.; Yudha, S.S. Biosynthesis of gold nanoparticles: A green approach. J. Photochem. Photobiol. B 2016, 161, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Khoshnamvand, M.; Ashtiani, S.; Huo, C.; Saeb, S.P.; Liu, J. Use of Alcea rosea leaf extract for biomimetic synthesis of gold nanoparticles with innate free radical scavenging and catalytic activities. J. Mol. Struct. 2019, 1179, 749–755. [Google Scholar] [CrossRef]
- Italiano, F.; Agostiano, A.; Belviso, B.D.; Caliandro, R.; Carrozzini, B.; Comparelli, R.; Melillo, M.T.; Mesto, E.; Tempestae, G.; Trotta, M. Interaction between the photosynthetic anoxygenic microorganism Rhodobacter sphaeroides and soluble gold compounds. From toxicity to gold nanoparticle synthesis. Colloids Surf. B 2018, 172, 362–371. [Google Scholar] [CrossRef]
- Karthik, R.; Govindasamy, M.; Chen, S.-M.; Mani, V.; Lou, B.-S.; Devasenathipathy, R.; Hou, Y.-S.; Elangovan, A. Green synthesized gold nanoparticles decorated graphene oxide for sensitive determination of chloramphenicol in milk, powdered milk, honey and eye drops. J. Colloid Interface Sci. 2016, 475, 46–56. [Google Scholar] [CrossRef]
- Li, L.; Zhang, Z.J. Biosynthesis of Gold Nanoparticles Using Green Alga Pithophora oedogonia with Their Electrochemical Performance for Determining Carbendazim in Soil. Int. J. Electrochem. Sci. 2016, 11, 4550–4559. [Google Scholar] [CrossRef]
- Karuppiah, C.; Palanisamy, S.; Chen, S.-M.; Emmanuel, R.; Muthupandi, K.; Prakash, P. Green synthesis of gold nanoparticles and its application for the trace level determination of painter’s colic. RSC Adv. 2015, 5, 16284–16291. [Google Scholar] [CrossRef]
- Karthik, R.; Chen, S.M.; Elangovan, A.; Muthukrishnan, P.; Shanmugam, R.; Lou, B.S. Phyto mediated biogenic synthesis of gold nanoparticles using Cerasus serrulata and its utility in detecting hydrazine, microbial activity and DFT studies. J. Colloid Interface Sci. 2016, 468, 163–175. [Google Scholar] [CrossRef]
- Stozhko, N.; Bukharinova, M.; Galperin, L.; Brainina, K. Nanostructured sensor based on gold nanoparticles and nafion for determination of uric acid. Biosensors 2018, 8, 21. [Google Scholar] [CrossRef]
- Saratale, R.G.; Karuppusamy, I.; Saratalec, G.D.; Pugazhendhid, A.; Kumare, G.; Parka, Y.; Ghodakef, G.S.; Bharagavag, R.N.; Banuh, J.R.; Shinc, H.S. A comprehensive review on green nanomaterials using biological systems: Recent perception and their future applications. Colloids Surf. B 2018, 170, 20–35. [Google Scholar] [CrossRef] [PubMed]
- Iravani, S.; Korbekandi, H.; Zolfaghari, B. Phytosynthesis of Nanoparticles. In Nanotechnology and Plant Sciences; Siddiqui, M.H., Al-Whaibi, M.H., Mohammad, F., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 203–258. [Google Scholar] [CrossRef]
- Sutan, N.A.; Manolescu, D.S.; Fierascu, I.; Neblea, A.M.; Sutan, C.; Ducu, C.; Soare, L.C.; Negrea, D.; Avramescu, S.M.; Fierascu, R.C. Phytosynthesis of gold and silver nanoparticles enhance in vitro antioxidant and mitostimulatory activity of Aconitum toxicum Reichenb. rhizomes alcoholic extracts. Mater. Sci. Eng. C 2018, 93, 746–758. [Google Scholar] [CrossRef] [PubMed]
- Din, M.I.; Arshad, F.; Hussain, Z.; Mukhtar, M. Green adeptness in the synthesis and stabilization of copper nanoparticles: Catalytic, antibacterial, cytotoxicity, and antioxidant activities. Nanoscale Res. Lett. 2017, 12, 638. [Google Scholar] [CrossRef] [PubMed]
- Prabu, H.J.; Johnson, I. Plant-mediated biosynthesis and characterization of silver nanoparticles by leaf extracts of Tragia involucrata, Cymbopogon citronella, Solanum verbascifolium and Tylophora ovata. Karbala Int. J. Mod. Sci. 2015, 1, 237–246. [Google Scholar] [CrossRef]
- Ahmed, K.B.A.; Subramanian, S.; Sivasubramanian, A.; Veerappan, G.; Veerappan, A. Preparation of gold nanoparticles using Salicornia brachiata plant extract and evaluation of catalytic and antibacterial activity. Spectrochim. Acta Part A 2014, 130, 54–58. [Google Scholar] [CrossRef]
- Nakkala, J.R.; Bhagat, E.; Suchiang, K.; Sadras, S.R. Comparative study of antioxidant and catalytic activity of silver and gold nanoparticles synthesized from Costus pictus leaf extract. J. Mater. Sci. Technol. 2015, 31, 986–994. [Google Scholar] [CrossRef]
- Mohammadi, S.; Pourseyedi, S.; Amini, A. Green synthesis of silver nanoparticles with a long lasting stability using colloidal solution of cowpea seeds (Vigna sp. L). J. Environ. Chem. Eng. 2016, 4, 2023–2032. [Google Scholar] [CrossRef]
- Kumar, B.; Smita, K.; Cumbal, L.; Debut, A. Green synthesis of silver nanoparticles using Andean blackberry fruit extract. Saudi J. Biol. Sci. 2017, 24, 45–50. [Google Scholar] [CrossRef]
- Palomo, J.M.; Filice, M. Biosynthesis of metal nanoparticles: Novel efficient heterogeneous nanocatalysts. Nanomaterials 2016, 6, 84. [Google Scholar] [CrossRef]
- Vijayaraghavan, K.; Ashokkumar, T. Plant-mediated biosynthesis of metallic nanoparticles: A review of literature, factors affecting synthesis, characterization techniques and applications. J. Environ. Chem. Eng. 2017, 5, 4866–4883. [Google Scholar] [CrossRef]
- Mittal, A.K.; Chisti, Y.; Banerjee, U.C. Synthesis of metallic nanoparticles using plant extracts. Biotechnol. Adv. 2013, 31, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, K.B.; Sakthivel, N. Phytosynthesis of gold nanoparticles using leaf extract of Coleus amboinicus Lour. Mater. Charact. 2010, 61, 1232–1238. [Google Scholar] [CrossRef]
- Mata, R.; Nakkala, J.R.; Sadras, S.R. Polyphenol stabilized colloidal gold nanoparticles from Abutilon indicum leaf extract induces apoptosis in HT-29 colon cancer cells. Colloids Surf. B 2016, 143, 499–510. [Google Scholar] [CrossRef]
- Lee, H.A.; Castro-Aceituno, V.; Abbai, R.; Moon, S.S.; Kim, Y.-J.; Simu, S.Y.; Yang, D.C. Rhizome of Anemarrhena asphodeloides as mediators of the eco-friendly synthesis of silver and gold spherical, face-centred cubic nanocrystals and its anti-migratory and cytotoxic potential in normal and cancer cell lines. Artif. Cells Nanomed. Biotechnol. 2018, 46, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Chahardoli, A.; Karimi, N.; Sadeghi, F.; Fattahi, A. A Green approach for synthesis of gold nanoparticles from Nigella arvensis leaf extract and evaluation of their antibacterial, antioxidant, cytotoxicity and catalytic activities. Artif Cells Nanomed. Biotechnol. 2018, 46, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Patil, M.P.; Kim, G.-D. Eco-friendly approach for nanoparticles synthesis and mechanism behind antibacterial activity of silver and anticancer activity of gold nanoparticles. Appl. Microbiol. Biotechnol. 2017, 101, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Koperuncholan, M. Bioreduction of chloroauric acid (HAuCl4) for the synthesis of gold nanoparticles (GNPs): A special empathies of pharmacological activity. Int. J. Phytopharm. 2015, 5, 72–80. [Google Scholar] [CrossRef]
- Khademi-Azandehi, P.; Moghaddam, J. Green synthesis, characterization and physiological stability of gold nanoparticles from Stachys lavandulifolia Vahl extract. Particuology 2015, 19, 22–26. [Google Scholar] [CrossRef]
- Gopinath, V.; Priyadarshini, S.; Mubarak Ali, D.; Loke, M.F.; Thajuddin, N.; Alharbi, N.S.; Yadavalli, T.; Alagiri, M.; Vadivelu, J. AntiHelicobacter pylori, cytotoxicity and catalytic activity of biosynthesized gold nanoparticles: Multifaceted application. Arab. J. Chem. 2019, 12, 33–40. [Google Scholar] [CrossRef]
- Anand, R.; Gengan, M.; Phulukdaree, A.; Chuturgoon, A. Agroforestry waste Moringa oleifera petals mediated green synthesis of gold nanoparticles and their anti-cancer and catalytic activity. J. Ind. Eng. Chem. 2015, 21, 1105–1111. [Google Scholar] [CrossRef]
- Park, Y.; Hong, Y.N.; Weyers, A.; Kim, Y.S.; Linhardt, R.J. Polysaccharides and phytochemicals: A natural reservoir for the green synthesis of gold and silver nanoparticles. IET Nanobiotechnol. 2011, 5, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Guo, Q.; Yuan, J.; Zeng, J. Biosynthesis of gold nanoparticles using a kind of flavonol: Dihydromyricetin. Colloids Surf. A 2014, 441, 127–132. [Google Scholar] [CrossRef]
- Kasthuri, J.; Veerapandian, S.; Rajendiran, N. Biological synthesis of silver and gold nanoparticles using apiin as reducing agent. Colloids Surf. B 2009, 68, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Brainina, K.; Stozhko, N.; Bukharinova, M.; Khamzina, E.; Vidrevich, M. Potentiometric method of plant microsuspensions antioxidant activity determination. Food Chem. 2019, 278, 653–658. [Google Scholar] [CrossRef] [PubMed]
- Brainina, K.Z.; Varzakova, D.P.; Kazakov, Y.E.; Vidrevich, M.B. Noninvasive electrochemical antioxidant activity estimation: Saliva analysis. Biointerface Res. Appl. Chem. 2018, 8, 3381–3387. [Google Scholar]
- Brainina, K.Z.; Ivanova, A.V.; Sharafutdinova, E.N.; Lozovskaya, E.L.; Shkarina, E.I. Potentiometry as a method of antioxidant activity investigation. Talanta 2007, 71, 13–18. [Google Scholar] [CrossRef]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. A Study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc. 1951, 11, 55–75. [Google Scholar] [CrossRef]
- Frens, G. Controlled Nucleation for the Regulation of the Particle Size in Monodisperse Gold Suspensions. Nat. Phys. Sci. 1973, 241, 20–22. [Google Scholar] [CrossRef]
- McFarland, A.D.; Haynes, C.L.; Mirkin, C.A.; Van Duyne, R.P.; Godwin, H.A. Color my nanoworld. J. Chem. Educ. 2004, 81, 544A. [Google Scholar] [CrossRef]
- Brainina, K.Z.; Galperin, L.G.; Vikulova, E.V.; Stozhko, N.Y.; Murzakaev, A.M.; Timoshenkova, O.R.; Kotov, Y.A. Gold nanoparticles electrooxidation: Comparison of theory and experiment. J. Solid State Electrochem. 2011, 15, 1049–1056. [Google Scholar] [CrossRef]
- Shankar, S.S.; Bhargava, S.; Sastry, M. Synthesis of gold nanospheres and nanotriangles by the Turkevich approach. J. Nanosci. Nanotechnol. 2005, 5, 1721–1727. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.; Singh, A.; Ahmad, A.; Sastry, M. Role of halide ions and temperature on the morphology of biologically synthesized gold nanotriangles. Langmuir 2006, 22, 736–741. [Google Scholar] [CrossRef] [PubMed]
- Haiss, W.; Thanh, N.T.K.; Aveyard, J.; Fernig, D.G. Fernig Determination of size and concentration of gold nanoparticles from UV-Vis spectra. Anal. Chem. 2007, 79, 4215–4221. [Google Scholar] [CrossRef] [PubMed]
- Suchomel, P.; Kvitek, L.; Prucek, R.; Panacek, A.; Halder, A.; Vajda, S.; Zboril, R. Simple size-controlled synthesis of Au nanoparticles and their size-dependent catalytic activity. Sci. Rep. 2018, 8, 4589. [Google Scholar] [CrossRef] [PubMed]
- Anwar, N.; Khan, A.; Shah, M.; Azam, A.; Zaman, K.; Parven, Z. The green synthesis of fine particles of gold using an aqueous extract of Monotheca buxifolia (Flac.). Russ. J. Phys. Chem. A 2016, 90, 2625–2632. [Google Scholar] [CrossRef]
- Alzoubi, F.Y.; Alzouby, J.Y.; Alqadi, M.K.; Alshboul, H.A.; Aljarrah, K.M. Synthesis and characterization of colloidal gold nanoparticles controlled by the pH and ionic strength. Chin. J. Phys. 2015, 53, 1008011–1008019. [Google Scholar] [CrossRef]
- Zhong, Z.; Chen, F.; Subramanian, A.S.; Lin, J.; Highfield, J.; Gedanken, A. Assembly of Au colloids into linear and spherical aggregates and effect of ultrasound irradiation on structure. J. Mater. Chem. 2006, 16, 489–495. [Google Scholar] [CrossRef]
- Radziuk, D.; Grigoriev, D.; Zhang, W.; Su, D.; Mohwald, H.; Shchukin, D. Ultrasound-assisted fusion of preformed gold nanoparticles. J. Phys. Chem. C 2010, 114, 1835–1843. [Google Scholar] [CrossRef]
- Watzky, M.A.; Finke, R.G. Transition metal nanocluster formation kinetic and mechanistic studies. A new mechanism when hydrogen is the reductant: Slow, continuous nucleation and fast autocatalytic surface growth. J. Am. Chem. Soc. 1997, 119, 10382–10400. [Google Scholar] [CrossRef]
- Watzky, M.A.; Finke, R.G. Gold Nanoparticle formation kinetics and mechanism: A critical analysis of the “redox crystallization” mechanism. ACS Omega 2018, 3, 1555–1563. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, H.; Lin, W.; Lin, L.; Gao, Y.; Yang, F.; Du, M.; Fang, W.; Huang, J.; Sun, D.; et al. Quantitative nucleation and growth kinetics of gold nanoparticles via model-assisted dynamic spectroscopic approach. J. Colloid Interface Sci. 2013, 407, 8–16. [Google Scholar] [CrossRef]
- Gold Nanoparticles: Properties and Applications. Available online: https://www.sigmaaldrich.com/technical-documents/articles/materials-science/nanomaterials/gold-nanoparticles.html (accessed on 28 June 2019).
- Rajeshkumar, S.; Bharath, L.V. Mechanism of plant-mediated synthesis of silver nanoparticles—A review on biomolecules involved, characterisation and antibacterial activity. Chem. Biol. Interact. 2017, 273, 219–227. [Google Scholar] [CrossRef]
- Tantra, R.; Schulze, P.; Quincey, P. Effect of nanoparticle concentration on zeta-potential measurement results and reproducibility. Particuology 2010, 8, 279–285. [Google Scholar] [CrossRef]




| Extract | Extract Aliquot in the Reaction Mixture, mL | Antioxidant Activity of the Reaction Mixture, mM-eq | ν, ms–1, (Section II) | ν, ms–1, (Section III) |
|---|---|---|---|---|
| Gooseberry (Ribes uva-crispa) | 0.25 | 0.08 | 1.0 | – |
| 0.50 | 0.15 | 1.3 | – | |
| 0.75 | 0.22 | 1.4 | – | |
| 1.0 | 0.28 | 4.1 | 0.9 | |
| 2.0 | 0.49 | 4.5 | 0.4 | |
| Blackcurrant (Ribes nigrum) | 0.25 | 0.32 | 2.2 | 0 |
| 0.50 | 0.62 | 6.3 | 0.2 | |
| 0.75 | 0.89 | 6.9 | 0.8 | |
| 1.0 | 1.13 | 7.1 | 0.7 | |
| 2.0 | 1.94 | 7.9 | 0.7 | |
| Strawberry (Fragaria vesca) | 0.25 | 0.68 | 5.7 | 0.4 |
| 0.50 | 1.30 | 6.0 | 0.9 | |
| 0.75 | 1.86 | 7.0 | 0.6 | |
| 1.0 | 2.38 | 7.6 | 0.7 | |
| 2.0 | 4.08 | 8.2 | 0.7 |
| phyto-AuNPs | Percentage (%) | |||
|---|---|---|---|---|
| Spheres | Triangular Plates | Rhomboid Plates | Rods | |
| gs-AuNPs (n = 264) | 90.2 | 6.8 | 2.6 | 0.4 |
| bc-AuNPs (n = 227) | 91.2 | 5.9 | 1.9 | 1.0 |
| sb-AuNPs (n = 241) | 90.9 | 5.1 | 2.3 | 1.7 |
| phyto-AuNPs | TEM | UV-Vis-Spectrophotometry | DLS | |||
|---|---|---|---|---|---|---|
| d, nm * | PI | d, nm ** | dH, nm * | PI | ζ, mV | |
| gb-AuNPs | 23 ± 10 | 0.17 | 25 ± 3 | 42 ± 1 | 0.29 | –16 ± 3 |
| bc-AuNPs | 15 ± 7 | 0.21 | 11 ± 2 | 38 ± 1 | 0.33 | –17 ± 4 |
| sb-AuNPs | 14 ± 7 | 0.24 | 10 ± 1 | 30 ± 1 | 0.28 | –26 ± 1 |
| Spherical phyto-AuNPs | Percentage (%) | ||||
|---|---|---|---|---|---|
| Up to 5 nm | 6–10 nm | 11–15 nm | 16–30 nm | 31–50 nm | |
| gs-AuNPs (n = 238) | 0 | 13.9 | 17.6 | 48.3 | 20.2 |
| bc-AuNPs (n = 201) | 1.5 | 38.8 | 23.9 | 33.3 | 2.5 |
| sb-AuNPs (n = 219) | 13.7 | 25.1 | 20.1 | 40.6 | 0.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stozhko, N.Y.; Bukharinova, M.A.; Khamzina, E.I.; Tarasov, A.V.; Vidrevich, M.B.; Brainina, K.Z. The Effect of the Antioxidant Activity of Plant Extracts on the Properties of Gold Nanoparticles. Nanomaterials 2019, 9, 1655. https://doi.org/10.3390/nano9121655
Stozhko NY, Bukharinova MA, Khamzina EI, Tarasov AV, Vidrevich MB, Brainina KZ. The Effect of the Antioxidant Activity of Plant Extracts on the Properties of Gold Nanoparticles. Nanomaterials. 2019; 9(12):1655. https://doi.org/10.3390/nano9121655
Chicago/Turabian StyleStozhko, Natalia Yu., Maria A. Bukharinova, Ekaterina I. Khamzina, Aleksey V. Tarasov, Marina B. Vidrevich, and Khiena Z. Brainina. 2019. "The Effect of the Antioxidant Activity of Plant Extracts on the Properties of Gold Nanoparticles" Nanomaterials 9, no. 12: 1655. https://doi.org/10.3390/nano9121655
APA StyleStozhko, N. Y., Bukharinova, M. A., Khamzina, E. I., Tarasov, A. V., Vidrevich, M. B., & Brainina, K. Z. (2019). The Effect of the Antioxidant Activity of Plant Extracts on the Properties of Gold Nanoparticles. Nanomaterials, 9(12), 1655. https://doi.org/10.3390/nano9121655








