RETRACTED: Nanostructured Nickel Nitride with Reduced Graphene Oxide Composite Bifunctional Electrocatalysts for an Efficient Water-Urea Splitting
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Ni3N and Reduced Graphene Oxide Composite on Ni Foam
2.3. Apparatus
2.4. Electrochemical Measurements
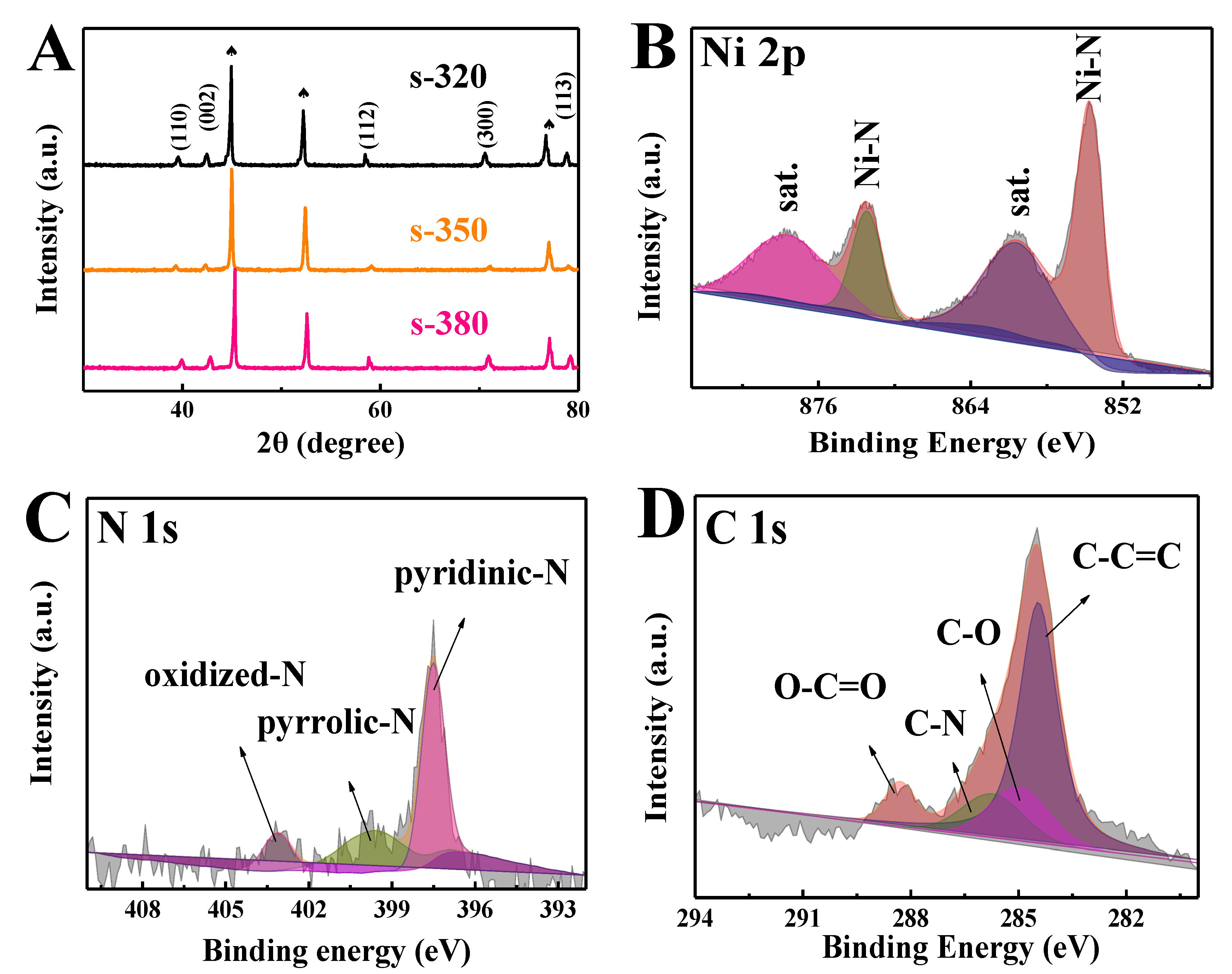
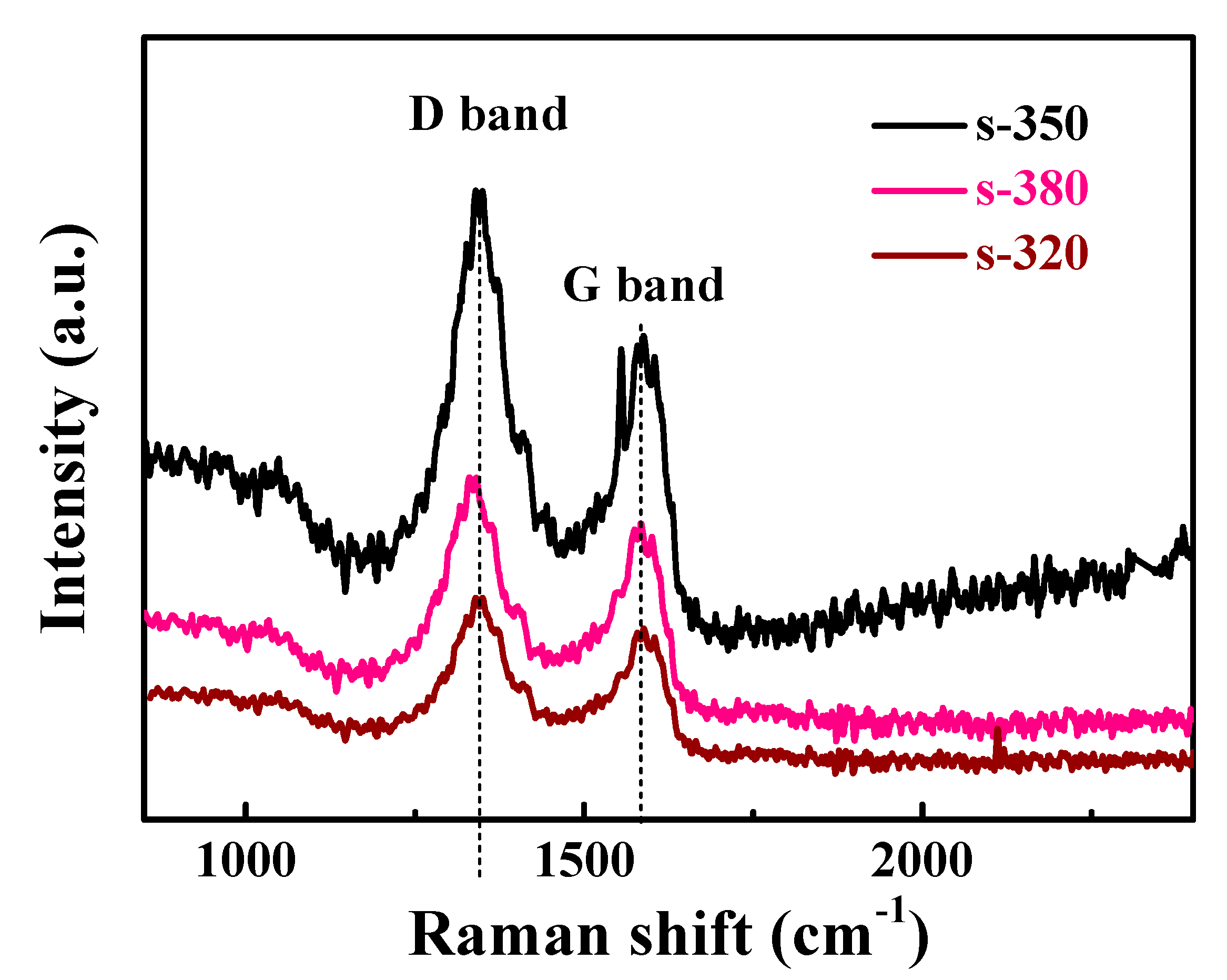
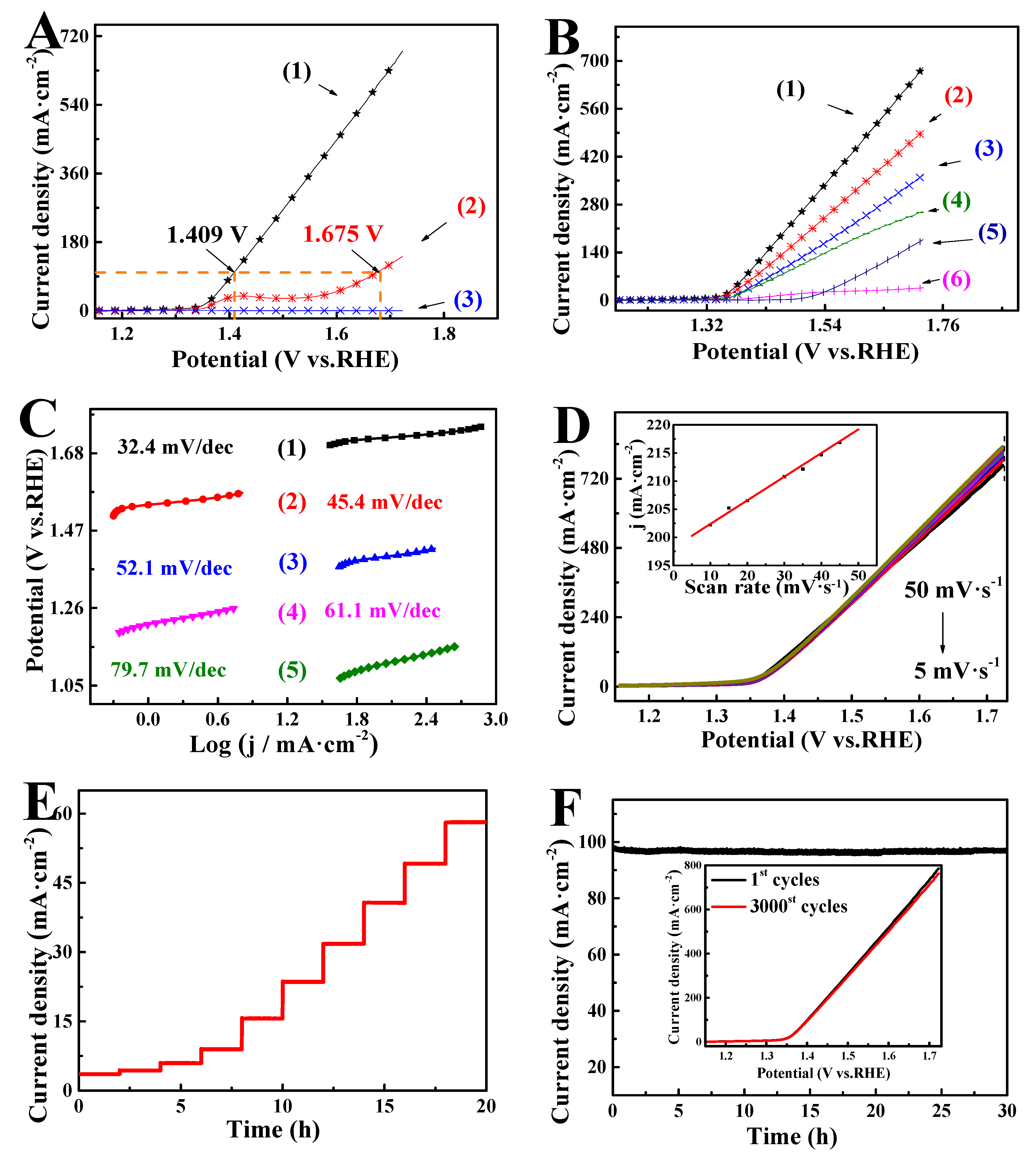
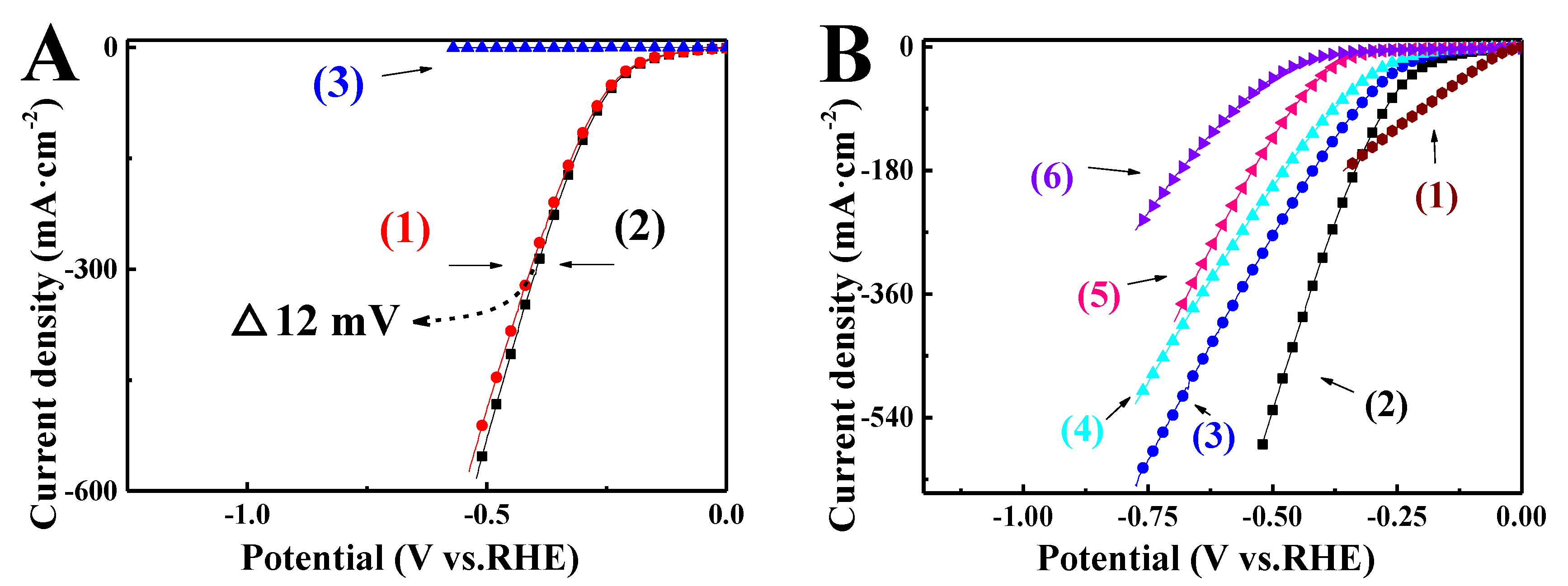
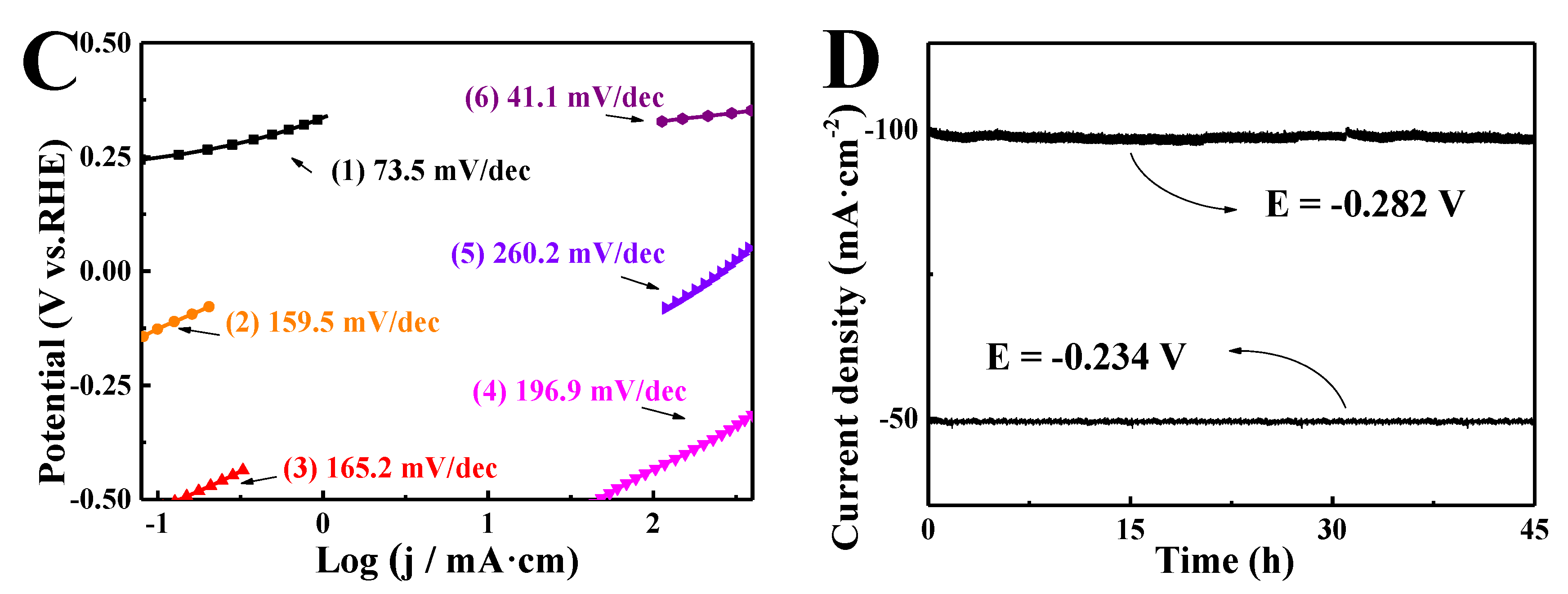
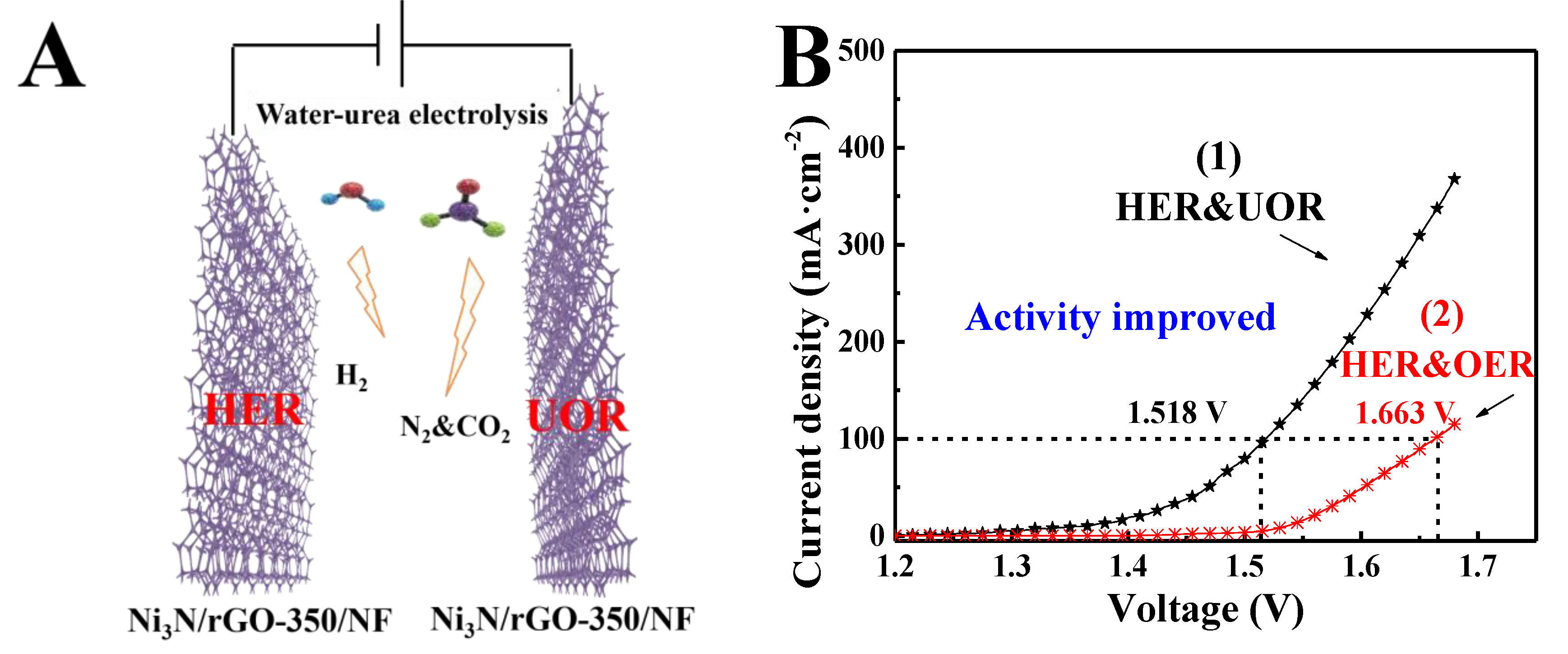
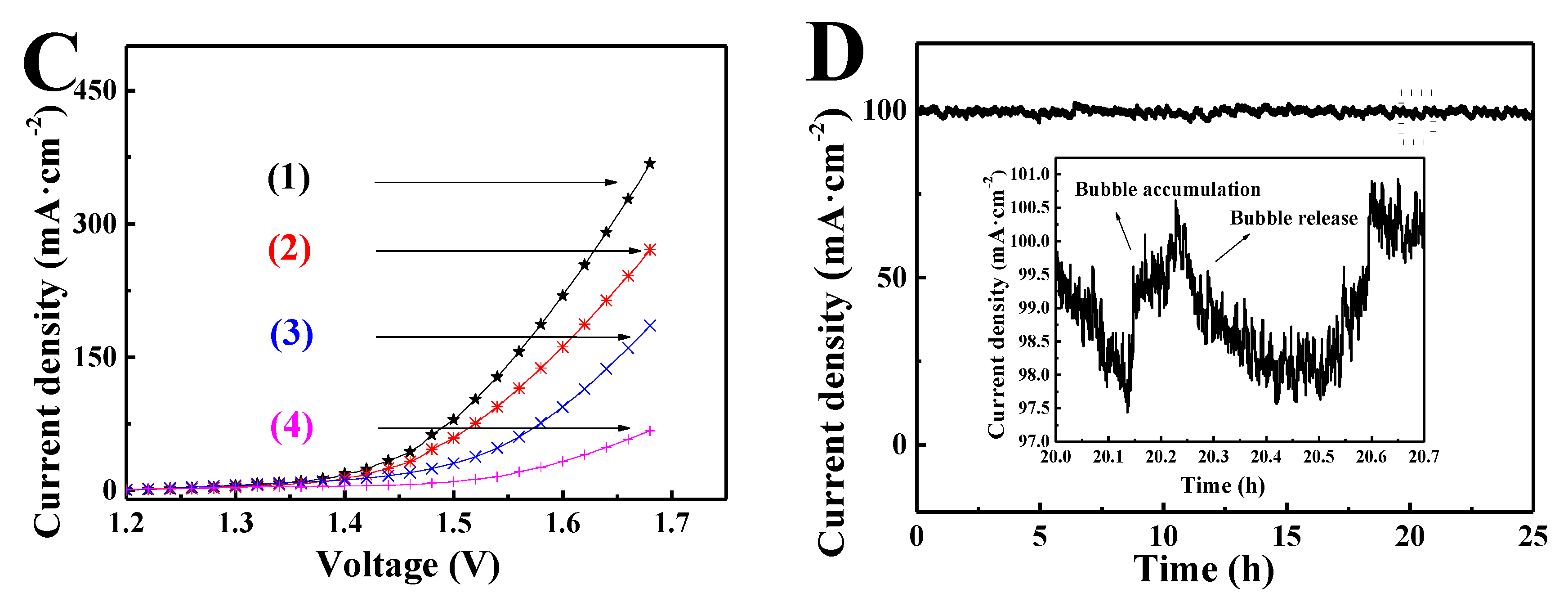
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Yang, F.; Huang, S.; Zhang, B.; Hou, L.; Ding, Y.; Bao, W.; Li, Y. Facile Synthesis of Well-Dispersed Ni2P on N-Doped Nanomesh Carbon Matrix as a High-Efficiency Electrocatalyst for Alkaline Hydrogen Evolution Reaction. Nanomaterials 2019, 9, 1022–1034. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Xiong, T.; Zhao, L.; Wang, F.; Liu, H.; Hu, R.; Zhou, J.; Zhou, W.; Chen, S. Ultrathin N-Doped Mo2C Nanosheets with Exposed Active Sites as Efficient Electrocatalyst for Hydrogen Evolution Reactions. ACS Nano 2017, 11, 12509–12518. [Google Scholar] [CrossRef] [PubMed]
- Long, X.; Li, J.; Xiao, S.; Yan, K.; Wang, Z.; Chen, H.; Yang, S. A Strongly Coupled Graphene and FeNi Double Hydroxide Hybrid as an Excellent Electrocatalyst for the Oxygen Evolution Reaction. Angew. Chem. Inter. Ed. 2014, 53, 7584–7588. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Cheng, H.; Lv, H.; Wang, J.; Liu, L.; Liu, S.; Wu, X.; Chu, W.; Wu, C.; Xie, Y. Controllable Surface Reorganization Engineering on Cobalt Phosphide Nanowire Arrays for Efficient Alkaline Hydrogen Evolution Reaction. Adv. Mater. 2018, 30, 1703322–1703328. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, Y.; Yan, F.; Zhu, C.; Gao, P.; Zhang, X.; Chen, Y. Self-Supported NiMo-Based Nanowire Arrays as Bifunctional Electrocatalysts for Full Water Splitting. J. Mater. Chem. A 2018, 6, 8479–8487. [Google Scholar] [CrossRef]
- Long, X.; Lin, H.; Zhou, D.; An, Y.; Yang, S. Enhancing Full Water-Splitting Performance of Transition Metal Bifunctional Electrocatalysts in Alkaline Solutions by Tailoring CeO2-Transition Metal Oxides–Ni Nanointerfaces. ACS Energy Lett. 2018, 3, 290–296. [Google Scholar] [CrossRef]
- Ma, W.; Ma, R.; Wang, C.; Liang, J.; Liu, X.; Zhou, K.; Sasaki, T. A Superlattice of Alternately Stacked Ni–Fe Hydroxide Nanosheets and Graphene for Efficient Splitting of Water. ACS Nano 2015, 9, 1977–1984. [Google Scholar] [CrossRef]
- Hu, S.; Feng, C.; Wang, S.; Liu, J.; Wu, H.; Zhang, L.; Zhang, J. Ni3N/NF as Bifunctional Catalysts for Both Hydrogen Generation and Urea Decomposition. ACS Appl. Mater. Interfaces 2019, 11, 13168–13175. [Google Scholar] [CrossRef]
- Hellstern, T.R.; Kibsgaard, J.; Tsai, C.; Palm, D.W.; King, L.A.; Abild-Pedersen, F.; Jaramillo, T.F. Investigating Catalyst-Support Interactions to Improve the Hydrogen Evolution Reaction Activity of Thiomolybdate [Mo3S13]2− Nanoclusters. ACS Catal. 2017, 7, 7126–7130. [Google Scholar] [CrossRef]
- Kim, J.S.; Kim, B.; Kim, H.; Kang, K. Recent Progress on Multimetal Oxide Catalysts for the Oxygen Evolution Reaction. Adv. Energy Mater. 2018, 8, 1702774. [Google Scholar] [CrossRef]
- Guan, B.Y.; Yu, L.; Lou, X.W. General Synthesis of Multishell Mixed-Metal Oxyphosphide Particles with Enhanced Electrocatalytic Activity in the Oxygen Evolution Reaction. Angew. Chem. Int. Ed. 2017, 56, 2386–2389. [Google Scholar] [CrossRef] [PubMed]
- Chi, J.; Yu, H.M.; Qin, B.W.; Fu, L.; Jia, J.; Yi, B.L.; Shao, Z.G. Vertically Aligned FeOOH/NiFe Layered Double Hydroxides Electrode for Highly Efficient Oxygen Evolution Reaction. ACS Appl. Mater. Interfaces 2017, 9, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Hunter, B.M.; Gray, H.B.; Muller, A.M. Earth-Abundant Heterogeneous Water Oxidation Catalysts. Chem. Rev. 2016, 116, 14120–14136. [Google Scholar]
- Ji, X.; Zhang, R.; Shi, X.; Asiri, A.M.; Zheng, B.; Sun, X. Fabrication of Hierarchical Cop Nanosheet@Microwire Arrays Via Space-Confined Phosphidation toward High-Efficiency Water Oxidation Electrocatalysis under Alkaline Conditions. Nanoscale 2018, 10, 7941–7945. [Google Scholar] [CrossRef]
- Zhao, J.; Li, X.; Cui, G.; Sun, X. Highly-Active Oxygen Evolution Electrocatalyzed by an Fe-Doped NiCr2O4 Nanoparticle Film. Chem. Commun. 2018, 54, 5462–5465. [Google Scholar] [CrossRef]
- Xiao, C.; Li, S.; Zhang, X. MnO2/MnCo2O4/Ni Heterostructure with Quadruple Hierarchy: A Bifunctional Electrode Architecture for Overall Urea Oxidation. J. Mater. Chem. A 2017, 5, 7825–7832. [Google Scholar] [CrossRef]
- Gwak, J.; Choun, M.; Lee, J. Alkaline Ammonia Electrolysis on Electrodeposited Platinum for Controllable Hydrogen Production. ChemSusChem 2015, 9, 403–408. [Google Scholar] [CrossRef]
- Lin, C.; Gao, Z.; Zhang, F.; Yang, J.; Liu, B.; Jin, J. In Situ Growth of Single-Layered α-Ni(OH)2 Nanosheets on a Carbon Cloth for Highly Efficient Electrocatalytic Oxidation of Urea. J. Mater. Chem. A 2018, 6, 13867–13873. [Google Scholar] [CrossRef]
- Ding, Y.; Li, Y.; Xue, Y.Y.; Miao, B.; Li, S.; Jiang, Y.; Chen, Y. Atomically Thick Ni(OH)2 Nanomeshes for Urea Electrooxidation. Nanoscale 2019, 11, 1058–1064. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, G.; Yu, M.; Wang, X.; Lv, J.; Yang, F. Free-Standing 3D Porous N-Doped Graphene Aerogel Supported Platinum Nanocluster for Efficient Hydrogen Production from Ammonia Electrolysis. ACS Sustain. Chem. Eng. 2018, 6, 8437–8446. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, D.; Hao, S.; Xie, L.; Qu, F.; Du, G.; Sun, X. Electrochemical Hydrazine Oxidation Catalyzed by Iron Phosphide Nanosheets Array toward Energy-Efficient Electrolytic Hydrogen Production from Water. ChemistrySelect 2017, 2, 3401–3407. [Google Scholar] [CrossRef]
- Forslund, R.P.; Mefford, J.T.; Hardin, W.G.; Alexander, C.T.; Johnston, K.P.; Stevenson, K.J. Nanostructured LaNiO3 Perovskite Electrocatalyst for Enhanced Urea Oxidation. ACS Catal. 2016, 6, 5044–5050. [Google Scholar] [CrossRef]
- Xiong, G.; He, P.; Wang, D.; Zhang, Q.; Chen, T.; Fisher, T.S. Hierarchical Ni-Co Hydroxide Petals on Mechanically Robust Graphene Petal Foam for High-Energy Asymmetric Supercapacitors. Adv. Funct. Mater. 2016, 26, 5460–5470. [Google Scholar] [CrossRef]
- Ji, J.; Zhang, L.L.; Ji, H.; Li, Y.; Zhao, X.; Bai, X.; Ruoff, R.S. Nanoporous Ni(OH)2 Thin Film on 3D Ultrathin-Graphite Foam for Asymmetric Supercapacitor. ACS Nano 2013, 7, 6237–6243. [Google Scholar] [CrossRef]
- Yang, Q.H.; Guo, S.Y.; Wang, L.X.; Li, X.J. Dry Sliding Friction and Wear Behavior of WC/WC-Ni Particles Laser Cladding Reinforced by Nano TiC on Mold Steel Surface. Adv. Mater. Res. 2011, 189, 3721–3725. [Google Scholar] [CrossRef]
- Ferrandon, M.; Kropf, A.J.; Krause, T. Bimetallic Ni-Rh catalysts with low amounts of Rh for the steam and autothermal reforming of n-butane for fuel cell applications. Appl. Catal. A 2010, 379, 121–128. [Google Scholar] [CrossRef]
- L’vov, B.V. Thermochemical approach to the mechanism and kinetics of NiO reduction with hydrogen. Russ. J. Appl. Chem. 2010, 83, 778–786. [Google Scholar] [CrossRef]
- Zheng, Y.; Qiao, J.; Yuan, J.; Shen, J.; Wang, A.; Gong, P.; Niu, L. Three-dimensional NiCu Layered Double Hydroxide Nanosheets Array on Carbon Cloth for Enhanced Oxygen Evolution. Electrochim. Acta 2018, 282, 735–742. [Google Scholar] [CrossRef]
- Lu, Z.; Qian, L.; Tian, Y.; Li, Y.; Sun, X.; Duan, X. Ternary NiFeMn Layered Double Hydroxides as Highly-Efficient Oxygen Evolution Catalysts. Chem. Commun. 2016, 52, 908–911. [Google Scholar] [CrossRef]
- Liu, L.; Ahmad, W.; Tao, J.; Tu, F.; Cheng, Z.; Gao, Y. NiO-NF/MWCNT nanocomposite catalyst as a counter electrode for high performance dye-sensitized solar cells. Appl. Surf. Sci. 2015, 331, 333–338. [Google Scholar]
- Hu, J.; Ou, Y.; Li, Y.; Gao, D.; Zhang, Y.; Xiao, P. FeCo2S4 Nanosheet Arrays Supported on Ni Foam: An Efficient and Durable Bifunctional Electrocatalyst for Overall Water-Splitting. Appl. Surf. Sci. 2018, 428, 148–153. [Google Scholar] [CrossRef]
- Li, J.; Zhou, C.; Mu, J. In Situ Synthesis of Molybdenum Carbide/N-Doped Carbon Hybrids as an Efficient Hydrogen-Evolution Electrocatalyst. RSC Adv. 2018, 8, 17202–17208. [Google Scholar] [CrossRef]
- He, B.; Zhou, M.; Hou, Z. Facile Synthesis of Ni3S2/rGO Nanosheets Composite on Nickel Foam as Efficient Electrocatalyst for Hydrogen Evolution Reaction in Alkaline Media. J. Mater. Res. 2018, 33, 1–9. [Google Scholar] [CrossRef]
- Liu, D.; Liu, T.; Zhang, L.; Qu, F.; Du, G.; Asiri, A.M.; Sun, X. High-Performance Urea Electrolysis Towards Less Energy-Intensive Electrochemical Hydrogen Production Using a Bifunctional Catalyst Electrode. J. Mater. Chem. A 2017, 5, 3208–3213. [Google Scholar] [CrossRef]
- Silva, D.A.; Pires, M.; Haye, E.; Zubiaur, A.; Job, N.; Pireaux, J.J.; Houssiau, L.; Busby, Y. Defective Pt–Ni/graphene nanomaterials by simultaneous or sequential treatments of organometallic precursors by low-pressure oxygen plasma. Plasma Process. Polym. 2019, 16, 1800203. [Google Scholar]
- Gao, D.; Zhang, J.; Wang, T.; Xiao, W.; Tao, K. Metallic Ni3N Nanosheets with Exposed Active Surface Sites for Efficient Hydrogen Evolution. J. Mater. Chem. A 2016, 4, 17363–17372. [Google Scholar] [CrossRef]
- Min, S.; Zhao, C.; Chen, G.; Qian, X. One-pot hydrothermal synthesis of reduced graphene oxide/Ni(OH)2 films on nickel foam for high performance supercapacitors. Electrochim. Acta 2014, 115, 155–164. [Google Scholar] [CrossRef]
- Yang, R.; Shi, Z.; Zhang, L.; Shi, D.; Zhang, G. Observation of RamanG-Peak Split for Graphene Nanoribbons with Hydrogen-Terminated Zigzag Edges. Nano Lett. 2011, 11, 4083–4088. [Google Scholar] [CrossRef]
- McCrory, C.C.; Jung, S.; Peters, J.C.; Jaramillo, T.F. Benchmarking heterogeneous electrocatalysts for the oxygen evolution reaction. J. Am. Chem. Soc. 2013, 135, 16977–16987. [Google Scholar] [CrossRef]
- Chen, S.; Duan, J.; Jaroniec, M.; Qiao, S. Three-Dimensional N-Doped Graphene Hydrogel/NiCo Double Hydroxide Electrocatalysts for Highly Efficient Oxygen Evolution. Angew. Chem. Int. Ed. 2013, 52, 13567–13570. [Google Scholar] [CrossRef]
- An, C.; Wang, Y.; Li, L.; Qiu, F.; Xu, Y.; Xu, C.; Huang, Y.; Jiao, L.; Yuan, H. Effects of Highly Crumpled Graphene Nanosheets on the Electrochemical Performances of Pseudocapacitor Electrode Materials. Electrochim. Acta 2014, 133, 180–187. [Google Scholar] [CrossRef]
- Chen, T.; Liu, D.; Lu, W.; Wang, K.; Du, G.; Asiri, A.M.; Sun, X. Three-Dimensional Ni2P Nanoarray: An Efficient Catalyst Electrode for Sensitive and Selective Nonenzymatic Glucose Sensing with High Specificity. Anal. Chem. 2016, 88, 7885–7889. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lin, C.; Huang, D.; Chen, J.; Jiang, L.; Wang, M.; Jin, J. Optimizing the Volmer Step by Single-Layer Nickel Hydroxide Nanosheets in Hydrogen Evolution Reaction of Platinum. ACS Catal. 2015, 5, 3801–3806. [Google Scholar] [CrossRef]
- Lu, X.; Zhao, C. Electrodeposition of hierarchically structured three-dimensional nickel–iron electrodes for efficient oxygen evolution at high current densities. Nat. Commun. 2015, 6, 6616–6622. [Google Scholar] [CrossRef]
- Qiao, L.; Zhou, M.; Li, Y.; Zhang, A.; Deng, J.; Liao, M.; Zhang, S. Enhancing Electrochemical Hydrogen Generation by Platinum-Modification of p-Type Silicon Wires Array under Visible Light. J. Electrochem. Soc. 2014, 161, H458–H463. [Google Scholar] [CrossRef]
- Sivanantham, A.; Ganesan, P.; Shanmugam, S. Bifunctional Electrocatalysts: Hierarchical NiCo2S4 Nanowire Arrays Supported on Ni Foam: An Efficient and Durable Bifunctional Electrocatalyst for Oxygen and Hydrogen Evolution Reactions. Adv. Funct. Mater. 2016, 26, 4660. [Google Scholar] [CrossRef]
- Benck, J.D.; Chen, Z.; Kuritzky, L.Y.; Forman, A.J.; Jaramillo, T.F. Amorphous Molybdenum Sulfide Catalysts for Electrochemical Hydrogen Production: Insights into the Origin of Their Catalytic Activity. ACS Catal. 2012, 2, 1916–1923. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.H.; Jia, S.P.; Xu, H.Q.; Zang, J.B.; Liu, J.; Xu, X.P. A hybrid of NiMo-Mo2C/C as non-noble metal electrocatalyst for hydrogen evolution reaction in an acidic solution. Electrochim. Acta 2016, 222, 747–754. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, F.; Zhao, D.; Zhang, L.; Fan, L.; Zhang, X.; Hu, S. RETRACTED: Nanostructured Nickel Nitride with Reduced Graphene Oxide Composite Bifunctional Electrocatalysts for an Efficient Water-Urea Splitting. Nanomaterials 2019, 9, 1583. https://doi.org/10.3390/nano9111583
Wang F, Zhao D, Zhang L, Fan L, Zhang X, Hu S. RETRACTED: Nanostructured Nickel Nitride with Reduced Graphene Oxide Composite Bifunctional Electrocatalysts for an Efficient Water-Urea Splitting. Nanomaterials. 2019; 9(11):1583. https://doi.org/10.3390/nano9111583
Chicago/Turabian StyleWang, Feng, Dongsheng Zhao, Linbao Zhang, Liming Fan, Xiutang Zhang, and Shengnan Hu. 2019. "RETRACTED: Nanostructured Nickel Nitride with Reduced Graphene Oxide Composite Bifunctional Electrocatalysts for an Efficient Water-Urea Splitting" Nanomaterials 9, no. 11: 1583. https://doi.org/10.3390/nano9111583
APA StyleWang, F., Zhao, D., Zhang, L., Fan, L., Zhang, X., & Hu, S. (2019). RETRACTED: Nanostructured Nickel Nitride with Reduced Graphene Oxide Composite Bifunctional Electrocatalysts for an Efficient Water-Urea Splitting. Nanomaterials, 9(11), 1583. https://doi.org/10.3390/nano9111583







