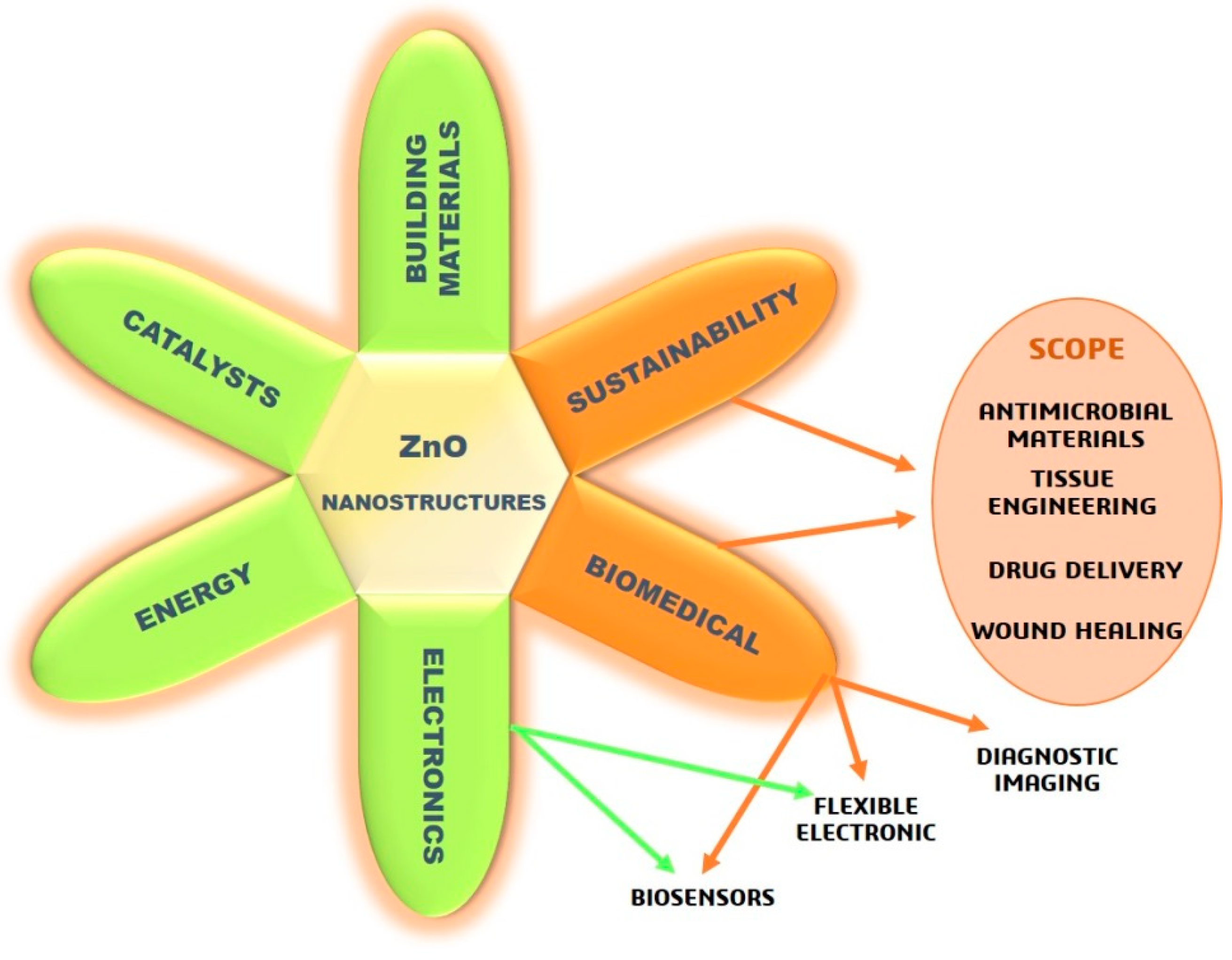
ZnO Nanostructures and Electrospun ZnO–Polymeric Hybrid Nanomaterials in Biomedical, Health, and Sustainability Applications
Abstract
1. Introduction
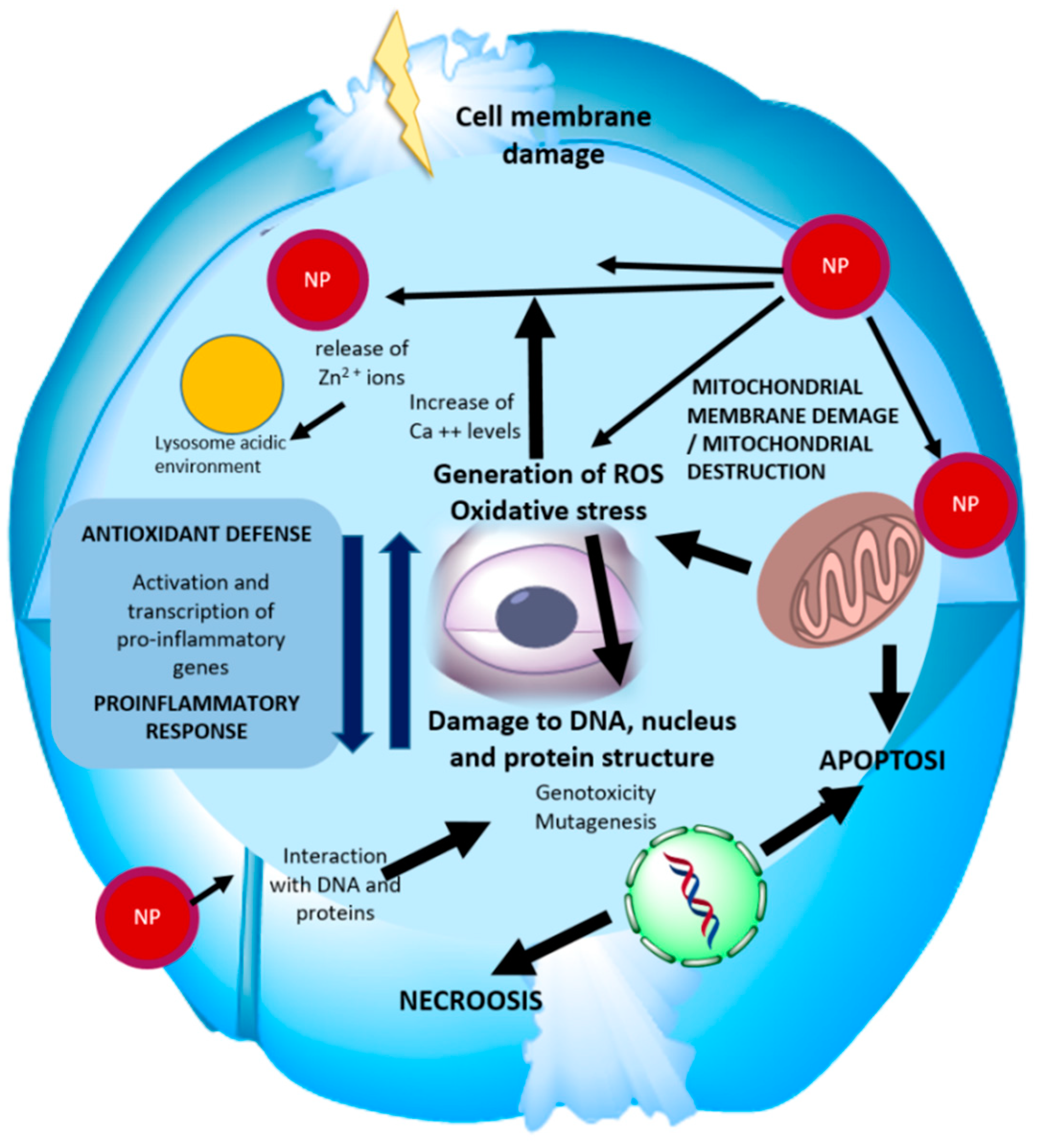
2. Toxicity Studies on ZnO Nanostructures In Vitro
2.1. ZnO Nanoparticles
2.2. Other Type of ZnO Nanostructures
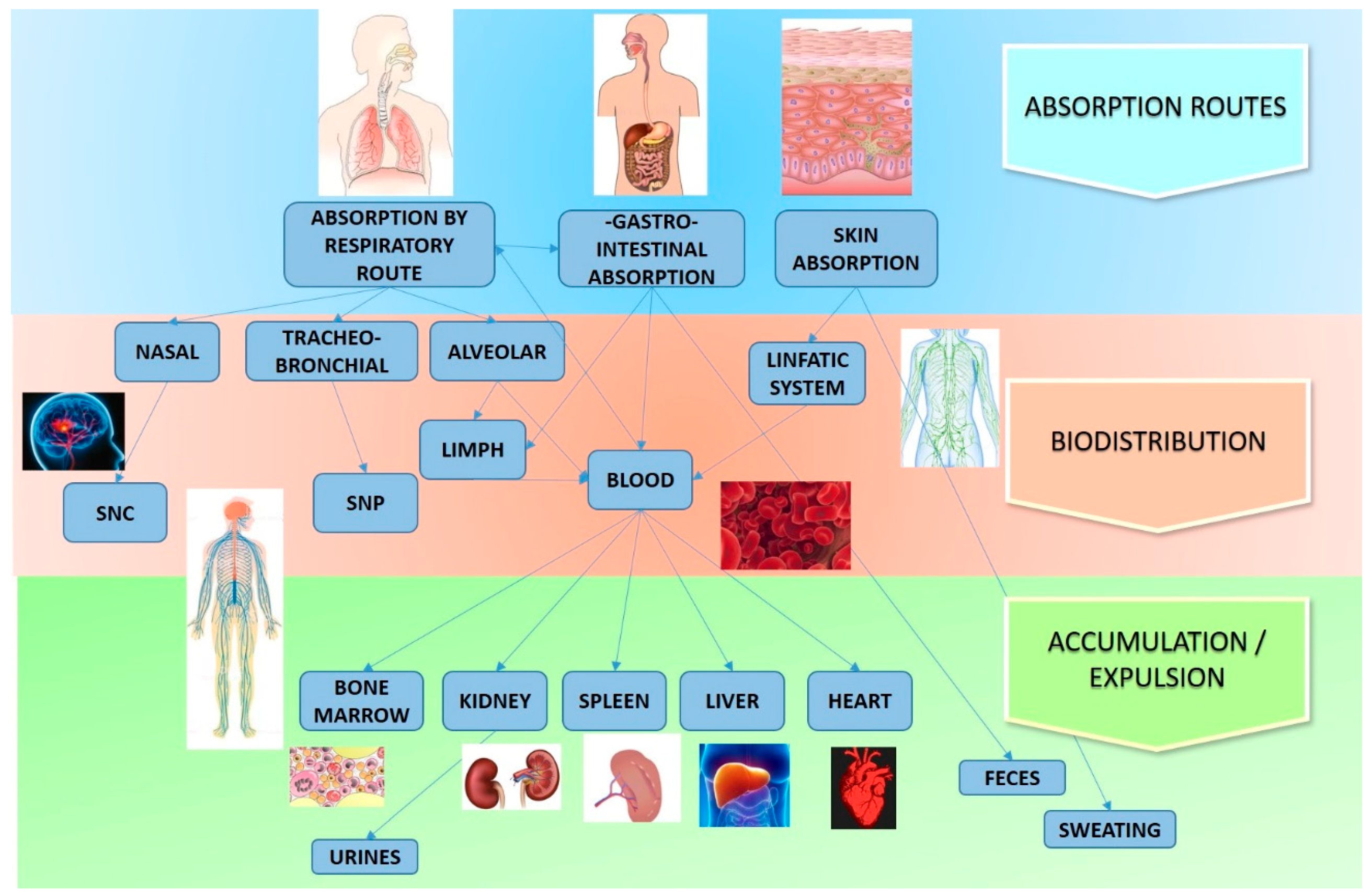
3. Toxicity Studies on ZnO Nanostructures In Vivo
- Most of the experiments were carried out on laboratory animals;
- In most cases, the doses were administered at one time and the concentration was significantly higher than the actual exposure conditions;
- There was no analysis of the long-term effects on the organism;
- There was no long-term study evaluating the effects due to exposure to small systemic concentrations.
- (1)
- The incidence of the shape and size of the particles; in fact, other studies reported that spherical and smaller nanoparticles are more likely to be taken up;
- (2)
- The use, in this study, of small doses, comparable to those used in clinical procedures, which were much lower than those generally used in literature;
- (3)
- The difference in conditions between in vitro and in vivo studies. The authors stressed the importance of the results obtained, but also the importance of carrying out further studies considering different routes of exposure, such as dermis, inhalation, etc.
4. New Approaches to Synthetize Safe ZnO Nanostructures for Biomedical Applications and Cancer Therapy
5. Influence of the Chemical and Physical Properties of the ZnO Nanostructures on Toxicity
6. ZnO–Polymeric Hybrid Electrospun Nanomaterials
6.1. Tissue-Engineering Applications
6.2. Wound-Healing Applications
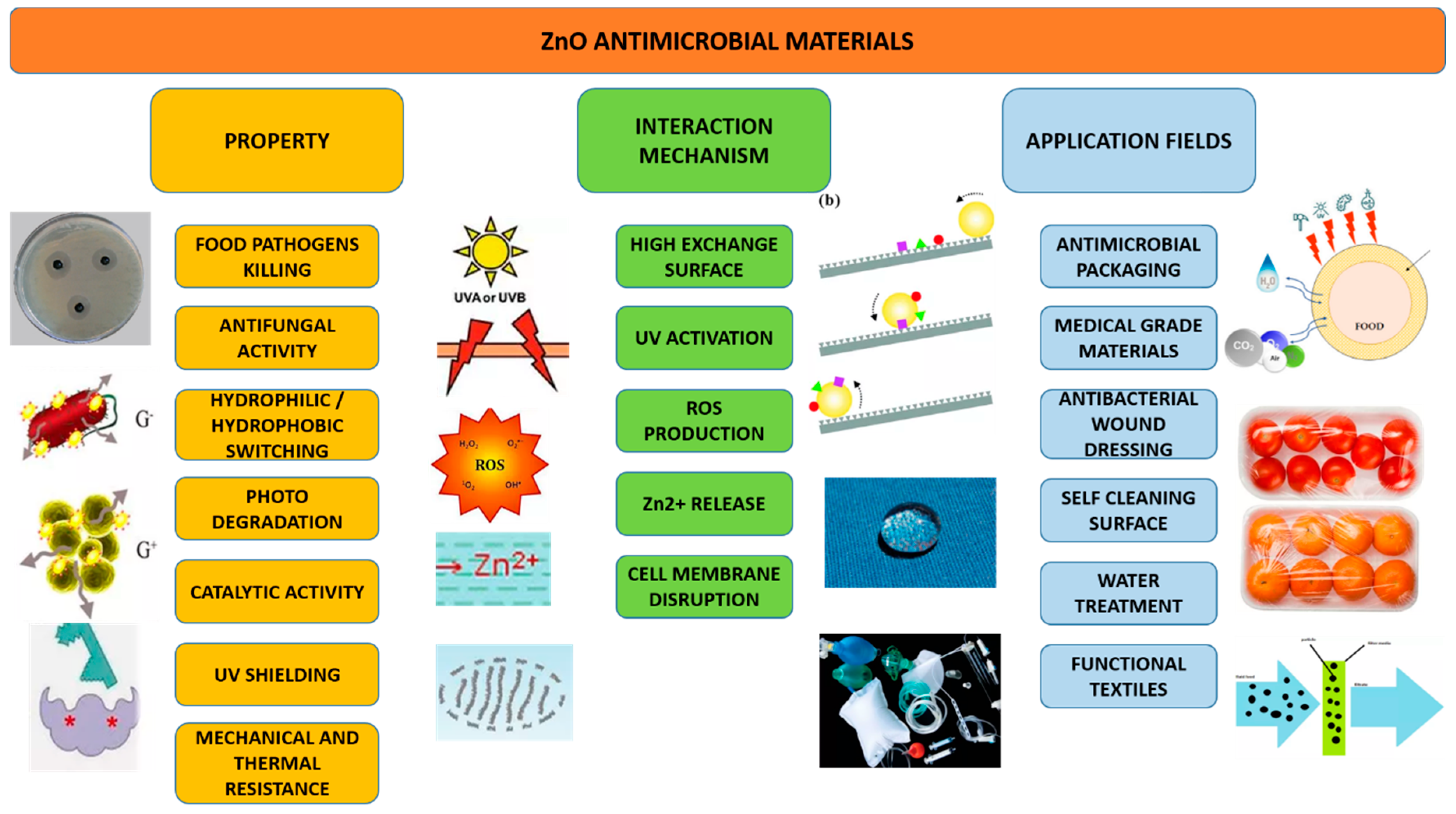
6.3. Antimicrobial Materials
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sousa, V.C.; Segadães, A.M. Combustion synthesized ZnO powders for varistor ceramics. Int. J. Inorg. Mater. 2002, 1, 235–241. [Google Scholar] [CrossRef]
- Lin, Y.; Chen, Y. Effect of ZnO nanoparticles doped graphene on static and dynamic mechanical properties of natural rubber composites. Compos. Part A Appl. Sci. Manuf. 2015, 70, 35–44. [Google Scholar] [CrossRef]
- Kandavelu, V.; Kastien, H. Photocatalytic degradation of isothiazolin-3-ones in water and emulsion paints containing nanocrystalline TiO2 and ZnO catalysts. Appl. Catal. B Environ. 2004, 48, 101–111. [Google Scholar] [CrossRef]
- Yebra, D.M.; Kiil, S. Dissolution rate measurements of sea water soluble pigments for antifouling paints: ZnO. Prog. Org. Coat. 2006, 56, 327–337. [Google Scholar] [CrossRef]
- Shi, L.E.; Li, Z.H. Synthesis, antibacterial activity, antibacterial mechanism and food applications of ZnO nanoparticles: A review. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2014, 31, 173–186. [Google Scholar] [CrossRef]
- Mu, L.; Sprando, R.L. Application of Nanotechnology in Cosmetics. Pharm. Res. 2010, 27, 1746–1749. [Google Scholar] [CrossRef]
- Lewicka, Z.A.; Yu, W.W. Photochemical behavior of nanoscale TiO2 and ZnO sunscreen ingredients. J. Photochem. Photobiol. A Chem. 2010, 263, 24–33. [Google Scholar] [CrossRef]
- Ozgur, Ü.; Hofstetter, D. ZnO devices and applications: A review of current status and future prospects. Proc. IEEE 2010, 98, 1255–1268. [Google Scholar] [CrossRef]
- Wang, X.; Song, J. Nanowire and nanobelt arrays of zinc oxide from synthesis to properties and to novel device. J. Mater. Chem. 2007, 17, 711–720. [Google Scholar] [CrossRef]
- Becheri, A.; Dürr, M. Synthesis and characterization of zinc oxide nanoparticles: Application to textiles as UV-absorbers. J. Nanoparticle Res. 2008, 10, 679–689. [Google Scholar] [CrossRef]
- Hasnidawani, J.N.; Azlina, H.N. Synthesis of ZnO nanostructures using sol-gel method. Procedia Chem. 2016, 19, 211–216. [Google Scholar] [CrossRef]
- Poornajar, M.; Marashi, P. Synthesis of ZnO nanorods via chemical bath deposition method: The effects of physicochemical factors. Ceram. Int. 2016, 42, 173–184. [Google Scholar] [CrossRef]
- Solis-Pomara, F.; Jaramillo, A. Rapid synthesis and photocatalytic activity of ZnO nanowires obtained through microwave-assisted thermal decomposition. Ceram. Int. 2016, 42, 18045–18052. [Google Scholar] [CrossRef]
- Ucer, K.B.; Pal, U. Synthesis and optical properties of ZnO nanostructures with different morphologies. Opt. Mater. (Amst) 2006, 29, 65–69. [Google Scholar]
- Azam, A.; Ahmed, A.S. Antimicrobial activity of metal oxide nanoparticles against Gram-positive and Gram-negative bacteria: A comparative study. Int. J. Nanomed. 2012, 7, 6003–6009. [Google Scholar] [CrossRef] [PubMed]
- Laurenti, M.; Cauda, V. ZnO Nanostructures for Tissue Engineering Applications. Nanomaterials 2017, 7, 374. [Google Scholar] [CrossRef] [PubMed]
- Stubbs, N.; Lansdown, A.B.G. Zinc in wound healing: Theoretical, experimental, and clinical aspects. Wound Repair Regen. 2007, 15, 2–16. [Google Scholar]
- Cai, X.; Luo, Y. PH-Sensitive ZnO Quantum Dots-Doxorubicin Nanoparticles for Lung Cancer Targeted Drug Delivery. Acs Appl. Mater. Interfaces 2016, 8, 22442–22450. [Google Scholar] [CrossRef] [PubMed]
- Politi, J.; Rea, I. Versatile synthesis of ZnO nanowires for quantitative optical sensing of molecular biorecognition. Sens. Actuators B Chem. 2015, 220, 705–711. [Google Scholar] [CrossRef]
- Zang, Z.; Tang, X. Enhanced fluorescence imaging performance of hydrophobic colloidal ZnO nanoparticles by a facile method. J. Alloys Compd. 2015, 619, 98–101. [Google Scholar] [CrossRef]
- Ahmed, B.; Dwivedi, S. Mitochondrial and Chromosomal Damage Induced by Oxidative Stress in Zn2+ Ions, ZnO-Bulk and ZnO-NPs treated Allium cepa roots. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Ahtzaz, S.; Nasir, M. A study on the effect of zinc oxide and zinc peroxide nanoparticles to enhance angiogenesis-pro-angiogenic grafts for tissue regeneration applications. Mater. Des. 2017, 132, 409–418. [Google Scholar] [CrossRef]
- Zhang, R.; Huang, Q. ZnO nanostructures enhance the osteogenic capacity of SaOS-2 cells on acid-etched pure Ti. Mater. Lett. 2017, 215, 173–175. [Google Scholar] [CrossRef]
- Parnia, F.; Yazdani, J. Overview of Nanoparticle Coating of Dental Implants for Enhanced Osseointegration and Antimicrobial Purposes. J. Pharm. Pharm. Sci. 2017, 20, 148–160. [Google Scholar] [CrossRef] [PubMed]
- Wingett, D.; Louka, P. A role of ZnO nanoparticle electrostatic properties in cancer cell cytotoxicity. Nanotechnol. Sci. Appl. 2016, 9, 29–45. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Malik, P. Biopolymer reinforced nanocomposites: A comprehensive review. Mater. Today Commun. 2018, 16, 353–363. [Google Scholar] [CrossRef]
- Lin, R.; Hernandez, B.V. Metal organic framework based mixed matrix membranes: An overview on filler/polymer interfaces. J. Mater. Chem. A 2018, 6, 293–312. [Google Scholar] [CrossRef]
- Hemamalini, T.; Rengaswami, V. Comprehensive review on electrospinning of starch polymer for biomedical applications. Int. J. Biol. Macromol. 2018, 106, 712–718. [Google Scholar] [CrossRef]
- Haider, A.; Haider, S. A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology. Arab. J. Chem. 2018, 11, 1165–1188. [Google Scholar] [CrossRef]
- Ding, J.; Zhang, J. Electrospun polymer biomaterials. Prog. Polym. Sci. 2019, 90, 1–34. [Google Scholar] [CrossRef]
- Ponnamma, D.; Cabibihan, J.J. Synthesis, optimization and applications of ZnO/polymer nanocomposites. Mater. Sci. Eng. C 2019, 98, 1210–1240. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Li, X. Electrospun poly(vinylidene fluoride)-zinc oxide hierarchical composite fiber membrane as piezoelectric acoustoelectric nanogenerator. J. Mater. Sci. 2019, 54, 2754–2762. [Google Scholar] [CrossRef]
- Pascariu, P.; Homocianu, M. Preparation of La doped ZnO ceramic nanostructures by electrospinning–calcination method: Effect of La3+ doping on optical and photocatalytic properties. Appl. Surf. Sci. 2019, 476, 16–27. [Google Scholar] [CrossRef]
- Han, J.; Xiong, L. Bio-functional electrospun nanomaterials: From topology design to biological applications. Prog. Polym. Sci. 2019, 91, 1–28. [Google Scholar] [CrossRef]
- Ginestra, P.; Ceretti, E. Electrospinning of Poly-caprolactone for Scaffold Manufacturing: Experimental Investigation on the Process Parameters Influence. Procedia CIRP 2016, 49, 8–13. [Google Scholar] [CrossRef]
- Balen, R.; Vidotto, W. Structural, thermal, optical properties and cytotoxicity of PMMA/ZnO fibers and films: Potential application in tissue engineering. Appl. Surf. Sci. 2016, 385, 257–267. [Google Scholar] [CrossRef]
- Aziz, A.; Tiwale, N. Core–Shell Electrospun Polycrystalline ZnO Nanofibers for Ultra-Sensitive NO2 Gas Sensing. Acs Appl. Mater. Interfaces 2018, 10, 43817–43823. [Google Scholar] [CrossRef]
- Bafqi, M.S.S.; Bagherzadeh, R. Fabrication of composite PVDF-ZnO nanofiber mats by electrospinning for energy scavenging application with enhanced efficiency. J. Polym. Res. 2015, 22, 130. [Google Scholar] [CrossRef]
- Hou, J.; Wu, Y. Toxic effects of different types of zinc oxide nanoparticles on algae, plants, invertebrates, vertebrates and microorganisms. Chemosphere 2018, 193, 852–860. [Google Scholar] [CrossRef]
- Hanley, C.; Thurber, A. The influences of cell Type and ZnO nanoparticle size on immune cell cytotoxicity and cytokine induction. Nanoscale Res. Lett. 2009, 4, 1409–1420. [Google Scholar] [CrossRef]
- Hanley, C.; Layne, J. Preferential killing of cancer cells and activated human T cells using ZnO nanoparticles. Nanotechnology 2008, 19, 29. [Google Scholar] [CrossRef] [PubMed]
- Heng, B.C.; Zhao, X. Toxicity of zinc oxide (ZnO) nanoparticles on human bronchial epithelial cells (BEAS-2B) is accentuated by oxidative stress. Food Chem. Toxicol. 2010, 48, 1762–1766. [Google Scholar] [CrossRef] [PubMed]
- Heng, B.C.; Zhao, X. Evaluation of the cytotoxic and inflammatory potential of differentially shaped zinc oxide nanoparticles. Arch. Toxicol. 2011, 85, 1517–1528. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhang, Y. Toxicity of ZnO Nanoparticles to Macrophages Due to Cell Uptake and Intracellular Release of Zinc Ions. J. Nanosci. Nanotechnol. 2014, 14, 5688–5696. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Bi, H. Zinc oxide nanoparticles decrease the expression and activity of plasma membrane calcium ATPase, disrupt the intracellular calcium homeostasis in rat retinal ganglion cells. Int. J. Biochem. Cell Biol. 2013, 45, 1849–1859. [Google Scholar] [CrossRef]
- Moratin, H.; Scherzad, A. Toxicological Characterization of ZnO Nanoparticles in Malignant and Non-Malignant Cells. Environ. Mol. Mutagenesis. 2018, 59, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Reshma, V.G.; Mohanan, P.V. Cellular interactions of zinc oxide nanoparticles with human embryonic kidney (HEK 293) cells. Colloids Surf. B Biointerfaces 2017, 157, 182–190. [Google Scholar]
- Okyay, T.O.; Bala, R.K. Antibacterial properties and mechanisms of toxicity of sonochemically grown ZnO nanorods. Rsc Adv. 2015, 5, 2568–2575. [Google Scholar] [CrossRef]
- Singh, A.; Singh, S. ZnO nanowire-coated hydrophobic surfaces for various biomedical applications. Bull. Mater. Sci. 2018, 41, 94. [Google Scholar] [CrossRef]
- Girigoswami, A.; Ramalakshm, M. ZnO Nanoflower petals mediated amyloid degradation-An in vitro electrokinetic potential approach. Mater. Sci. Eng. C 2019, 101, 169–178. [Google Scholar] [CrossRef]
- Bahramian, R.; Eshghi, H. Influence of annealing temperature on morphological, optical and UV detection properties of ZnO nanowires grown by chemical bath deposition. Mater. Des. 2016, 107, 269–276. [Google Scholar] [CrossRef]
- Paino, I.M.M.; Gonçalves, F.J. Zinc Oxide Flower-Like Nanostructures That Exhibit Enhanced Toxicology Effects in Cancer Cells. Acs Appl. Mater. Interfaces 2016, 8, 32699–32705. [Google Scholar] [CrossRef] [PubMed]
- Müller, K.H.; Kulkarni, J. PH-dependent toxicity of high aspect ratio ZnO nanowires in macrophages due to intracellular dissolution. Acs Nano 2010, 4, 6767–6779. [Google Scholar] [CrossRef] [PubMed]
- Gopikrishnan, R.; Zhang, K. Epitaxial growth of the Zinc Oxide nanorods, their characterization and in vitro biocompatibility studies. J. Mater. Sci. Mater. Med. 2011, 22, 2301–2309. [Google Scholar] [CrossRef] [PubMed]
- Ahamed, M.; Akhtar, M.J. ZnO nanorod-induced apoptosis in human alveolar adenocarcinoma cells via p53, survivin and bax/bcl-2 pathways: Role of oxidative stress. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 904–913. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Y. Cytotoxicity of ZnO Nanowire Arrays on Excitable Cells. Nanomaterials 2017, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Papavlassopoulos, H.; Mishra, Y.K. Toxicity of Functional Nano-Micro Zinc Oxide Tetrapods: Impact of Cell Culture Conditions, Cellular Age and Material Properties. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Tian, L.; Lin, B. Neurotoxicity induced by zinc oxide nanoparticles: Age-related differences and interaction. Sci. Rep. 2015, 5, 1–12. [Google Scholar] [CrossRef]
- Ansar, S.; Abudawood, M. Exposure to Zinc Oxide Nanoparticles Induces Neurotoxicity and Proinflammatory Response: Amelioration by Hesperidin. Biol. Trace Elem. Res. 2017, 175, 360–366. [Google Scholar] [CrossRef]
- Liu, X.Q.; Zhang, H.F. Regulation of neuroendocrine cells and neuron factors in the ovary by zinc oxide nanoparticles. Toxicol. Lett. 2016, 256, 19–32. [Google Scholar] [CrossRef]
- Han, Z.; Yan, Q. Cytotoxic effects of ZnO nanoparticles on mouse testicular cells. Int. J. Nanomed. 2016, 11, 5187–5203. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Liu, C.; Chen, X.; Yang, Z. Zinc oxide nanoparticles induce renal toxicity through reactive oxygen species. Food Chem. Toxicol. 2016, 90, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Sravan Bollu, V.; Soren, G. Genotoxic and Histopathological Evaluation of Zinc Oxide Nanorods in Vivo in Swiss Albino Mice. J. Evol. Med. Dent. Sci. 2016, 5, 6186–6192. [Google Scholar] [CrossRef]
- Pietkiewicz, M.W.; Tokarska, K. Safe-by-design’ ligand coated-ZnO nanocrystals engineered by an organometallic approach: Unique physicochemical properties and low negative toxicological effect toward lung cells. Chem. Eur. J. 2018, 24, 4033–4042. [Google Scholar] [CrossRef] [PubMed]
- Chun, H.S.; Park, D. Two zinc-aminoclays’ in-vitro cytotoxicity assessment in HeLa cells and in-vivo embryotoxicity assay in zebrafish. Ecotoxicol. Environ. Saf. 2017, 137, 103–112. [Google Scholar] [CrossRef]
- Zeng, H.; Zhang, Z. Lipid-coated ZnO nanoparticles as lymphatic-targeted drug carriers: Study on cell-specific toxicity in vitro and lymphatic targeting in vivo. J. Mater. Chem. B 2015, 3, 5249–5260. [Google Scholar] [CrossRef]
- Vimala, K.; Shanthi, K. Synergistic effect of chemo-photothermal for breast cancer therapy using folic acid (FA) modified zinc oxide nanosheet. J. Colloid Interface Sci. 2017, 488, 92–108. [Google Scholar] [CrossRef]
- Padmanabhan, J.; Kyriakides, T.R. Nanomaterials, Inflammation, and Tissue Engineering. Wires Nanomater. Nanobiotechnol. 2015, 7, 355–370. [Google Scholar] [CrossRef]
- Bhowmick, S.; Rother, S. Biomimetic electrospun scaffolds from main extracellular matrix components for skin tissue engineering application – The role of chondroitin sulfate and sulfated hyaluronan. Mater. Sci. Eng. C 2017, 79, 15–22. [Google Scholar] [CrossRef]
- Jiang, Y.C.; Jiang, L. Electrospun polycaprolactone/gelatin composites with enhanced cell–matrix interactions as blood vessel endothelial layer scaffolds. Mater. Sci. Eng. C 2017, 71, 901–908. [Google Scholar] [CrossRef]
- Bose, S.; Roy, M. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554. [Google Scholar] [CrossRef] [PubMed]
- Trombetta, R.; Inzana, J.A. 3D Printing of Calcium Phosphate Ceramics for Bone Tissue Engineering and Drug Delivery. Ann. Biomed. Eng. 2017, 45, 23–44. [Google Scholar] [CrossRef] [PubMed]
- Felice, B.; Sanchez, M.A. Controlled degradability of PCL-ZnO nanofibrous scaffolds for bone tissue engineering and their antibacterial activity. Mater. Sci. Eng. C 2018, 93, 724–738. [Google Scholar] [CrossRef] [PubMed]
- Park, J.K.; Kim, Y.J. The Topographic Effect of Zinc Oxide Nanoflowers on Osteoblast Growth and Osseointegration. Adv. Mater. 2010, 22, 4857–4861. [Google Scholar] [CrossRef] [PubMed]
- Padovani, G.C.; Feitosa, V.P. Advances in Dental Materials through Nanotechnology: Facts, Perspectives and Toxicological Aspects. Trends Biotechnol. 2015, 33, 621–636. [Google Scholar] [CrossRef] [PubMed]
- Ciofani, G.; Genchi, G.G. ZnO nanowire arrays as substrates for cell proliferation and differentiation. Mater. Sci. Eng. C 2012, 32, 341–347. [Google Scholar] [CrossRef]
- Errico, V.; Arrabito, G. High-Density ZnO Nanowires as a Reversible Myogenic–Differentiation Switch. Acs Appl. Mater. Interfaces 2018, 10, 14097–14107. [Google Scholar] [CrossRef] [PubMed]
- Follmann, D.M.; Naves, A.F. Hybrid Materials and Nanocomposites as Multifunctional Biomaterials. Curr. Pharm. Des. 2017, 23, 3794–3813. [Google Scholar] [CrossRef]
- Jun, I.; Han, H.S. Electrospun Fibrous Scaffolds for Tissue Engineering: Viewpoints on Architecture and Fabrication. Int. J. Mol. Sci. 2018, 19, 745. [Google Scholar] [CrossRef]
- Fang, J.; Wang, X. Functional applications of electrospun nanofibers. In Nanofibers—Production, Properties and Functional Applications; InTech—Open Access Publisher: London, UK, 2011; pp. 287–326. [Google Scholar]
- Therona, S.A.; Zussmanab, E. Experimental investigation of the governing parameters in the electrospinning of polymer solutions. Polymer 2004, 45, 2017–2030. [Google Scholar] [CrossRef]
- Soliman, S.; Pagliari, S. Multiscale three-dimensional scaffolds for soft tissue engineering via multimodal electrospinning. Acta Biomater. 2006, 6, 1227–1237. [Google Scholar] [CrossRef] [PubMed]
- Wannatong, L.; Sirivat, A. Effects of solvents on electrospun polymeric fibers: Preliminary study on polystyrene. Polym. Int. 2004, 53, 1851–1859. [Google Scholar] [CrossRef]
- Luo, C.J.; Nangrejo, M. A novel method of selecting solvents for polymer electrospinning. Polymer 2010, 51, 1654–1662. [Google Scholar] [CrossRef]
- Liverani, L.; Boccaccini, A.R. Versatile Production of Poly(Epsilon-Caprolactone) Fibers by Electrospinning Using Benign Solvents. Nanomaterials 2016, 6, 75. [Google Scholar] [CrossRef] [PubMed]
- Dotivala, A.C.; Puthuveetil, K.P. Shear Force Fiber Spinning: Process Parameter and Polymer Solution Property Consideration. Polymers 2019, 11, 294. [Google Scholar] [CrossRef]
- Shin, D.; Kim, J. Experimental study on jet impact speed in near-field electrospinning for precise patterning of nanofiber. J. Manuf. Process. 2018, 36, 231–237. [Google Scholar] [CrossRef]
- Hekmati, A.H.; Rashidi, A. Effect of needle length, electrospinning distance, and solution concentration on morphological properties of polyamide-6 electrospun nanowebs. Text. Res. J. 2013, 83. [Google Scholar] [CrossRef]
- Asghari, F.; Samiei, M. Biodegradable and biocompatible polymers for tissue engineering application: A review. Artif. Cells Nanomed. Biotechnol. 2017, 45, 185–192. [Google Scholar] [CrossRef]
- Malikmammadov, E.; Tanir, T.E. PCL and PCL-based materials in biomedical applications. J. Biomater. Sci. Polym. Ed. 2018, 29, 863–893. [Google Scholar] [CrossRef] [PubMed]
- Munchow, E.A.; Albuquerque, M.T.P. Development and characterization of novel ZnO-loaded electrospun membranes for periodontal regeneration. Dent. Mater. 2015, 31, 1038–1051. [Google Scholar] [CrossRef] [PubMed]
- Sezer, U.A.; Ozturk, K. Zero valent zinc nanoparticles promote neuroglial cell proliferation: A biodegradable and conductive filler candidate for nerve regeneration. J. Mater. Sci. Mater. Med. 2017, 28. [Google Scholar]
- Augustine, R.; Dominic, E.A. Investigation of angiogenesis and its mechanism using zinc oxide nanoparticle-loaded electrospun tissue engineering scaffolds. Rsc Adv. 2014, 4, 51528–51536. [Google Scholar] [CrossRef]
- Li, Y.; Sun, L. The Investigation of ZnO/Poly(vinylidene fluoride) Nanocomposites with Improved Mechanical, Piezoelectric, and Antimicrobial Properties for Orthopedic Applications. J. Biomed. Nanotechnol. 2018, 14, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Collins, G. Neurite extension of primary neurons on electrospun piezoelectric scaffolds. Acta Biomater. 2011, 7, 3877–3886. [Google Scholar] [CrossRef] [PubMed]
- Augustine, R.; Dan, P. Electrospun poly(vinylidene fluoride-trifluoroethylene)/zinc oxide nanocomposite tissue engineering scaffolds with enhanced cell adhesion and blood vessel formation. Nano Res. 2017, 10, 3358–3376. [Google Scholar] [CrossRef]
- Amna, T.; Hassan, M.S. Zinc oxide-doped poly(urethane) spider web nanofibrous scaffold via one-step electrospinning: A novel matrix for tissue engineering. Appl. Microbiol. Biotechnol. 2013, 97, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Amna, T.; Hassan, M.S. Electrospun nanofibers of ZnO-TiO2 hybrid: Characterization and potential as an extracellular scaffold for supporting myoblasts. Surf. Interface Anal. 2014, 46, 72–76. [Google Scholar] [CrossRef]
- Zhu, P.; Weng, Z. Biomedical Applications of Functionalized ZnO Nanomaterials: From Biosensors to Bioimaging. Adv. Mater. Interfaces 2016, 3. [Google Scholar] [CrossRef]
- Parham, S.; Wicaksono, D.H.B. Antimicrobial Treatment of Different Metal Oxide Nanoparticles: A Critical Review. J. Chin. Med. Soc. 2016, 63, 385–393. [Google Scholar] [CrossRef]
- Häffner, S.M.; Malmsten, M. Membrane interactions and antimicrobial effects of inorganic nanoparticles. Adv. Colloid Interface Sci. 2017, 248, 105–128. [Google Scholar] [CrossRef] [PubMed]
- Eutimio, M.; Monroy, E. Enhanced healing and anti-inflammatory effects of a carbohydrate polymer with zinc oxide in patients with chronic venous leg ulcers: Preliminary results. Arch. Med. Sci. 2018, 14, 336–344. [Google Scholar] [CrossRef]
- Kantipudi, S.; Sunkara, J.R. Enhanced wound healing activity of Ag–ZnO composite NPs in Wistar Albino rats. Iet Digit. Libr. 2018, 12, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Gao, J. Enhanced antibacterial and wound healing activities of microporous chitosan-Ag/ZnO composite dressing. Carbohydr. Polym. 2017, 156, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Khorasani, M.T.; Joorabloo, A. Incorporation of ZnO nanoparticles into heparinised polyvinyl alcohol/chitosan hydrogels for wound dressing application. Int. J. Biol. Macromol. 2018, 114, 1203–1215. [Google Scholar] [CrossRef] [PubMed]
- Zhai, M.; Xu, Y. Keratin-chitosan/n-ZnO nanocomposite hydrogel for antimicrobial treatment of burn wound healing: Characterization and biomedical application. J. Photochem. Photobiol. B Biol. 2018, 180, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Chhabra, H.; Deshpande, R. A nano zinc oxide doped electrospun scaffold improves wound healing in a rodent model. Rsc Adv. 2016, 6, 1428–1439. [Google Scholar] [CrossRef]
- Shalumon, K.T.; Anulekha, K.H. Sodium alginate/poly(vinyl alcohol)/nano ZnO composite nanofibers for antibacterial wound dressings. Int. J. Biol. Macromol. 2011, 49, 247–254. [Google Scholar] [CrossRef]
- Augustine, R.; Dominic, E.A. Electrospun polycaprolactone membranes incorporated with ZnO nanoparticles as skin substitutes with enhanced fibroblast proliferation and wound healing. Rsc Adv. 2014, 4, 24777–24785. [Google Scholar] [CrossRef]
- Abdalkarim, S.Y.H.; Yu, H.Y. Electrospun poly(3-hydroxybutyrate-co-3-hydroxy-valerate)/cellulose reinforced nanofibrous membranes with ZnO nanocrystals for antibacterial wound dressings. Cellulose 2017, 24, 2925–2938. [Google Scholar] [CrossRef]
- Ahmed, R.; Tariq, M. Novel electrospun chitosan/polyvinyl alcohol/zinc oxide nanofibrous mats with antibacterial and antioxidant properties for diabetic wound healing. Int. J. Biol. Macromol. 2018, 120, 385–393. [Google Scholar] [CrossRef] [PubMed]
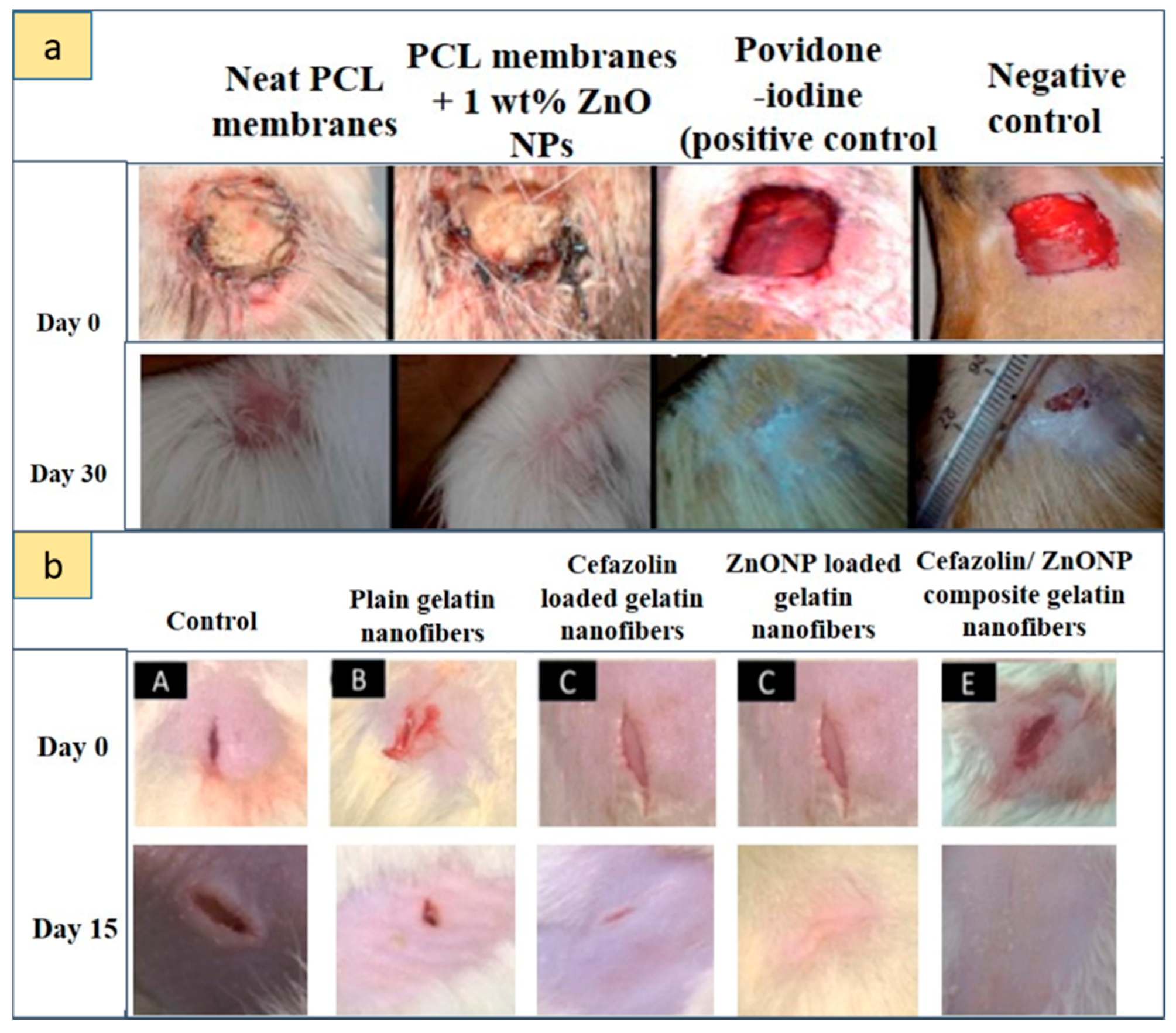
- Rath, G.; Hussain, T. Development and characterization of cefazolin loaded zinc oxide nanoparticles composite gelatin nanofiber mats for postoperative surgical wounds. Mater. Sci. Eng. C 2016, 58, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Zhang, J. Anti-fouling potential evaluation of PVDF membranes modified with ZnO against polysaccharide. Chem. Eng. J. 2016, 304, 65–174. [Google Scholar] [CrossRef]
- Pascariu, P.; Olaru, L. Photocatalytic activity of ZnO nanostructures grown on electrospun CAB ultrafine fibers. Appl. Surf. Sci. 2018, 455, 61–69. [Google Scholar] [CrossRef]
- Pant, B.; Ojha, G.P.O. Fly-ash-incorporated electrospun zinc oxide nanofibers: Potential material for environmental remediation. Environ. Pollut. 2019, 245, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Kunjuzwa, N.; Nthunya, L.N. Chapter 5-The use of nanomaterials in the synthesis of nanofiber membranes and their application in water treatment. In Advanced Nanomaterials for Membrane Synthesis and its Applications; Elsevier: Amsterdam, The Netherlands, 2019; pp. 101–125. [Google Scholar]
- Mai, Z.; Xiong, Z. Multifunctionalization of cotton fabrics with polyvinylsilsesquioxane/ZnO composite coatings. Carbohydr. Polym. 2018, 199, 516–525. [Google Scholar] [CrossRef]
- Song, K.; Wu, Q. 20-Electrospun nanofibers with antimicrobial properties. In Electrospun Nanofibers; Woodhead Publishing Series in Textiles; Woodhead Publishing: Cambridge, UK, 2017; pp. 551–569. [Google Scholar]
- Mallakpour, S.; Behranvand, V. Nanocomposites based on biosafe nano ZnO and different polymeric matrixes for antibacterial, optical, thermal and mechanical applications. Eur. Polym. J. 2016, 84, 377–403. [Google Scholar] [CrossRef]
- Li, X.; Xing, Y. Antimicrobial activities of ZnO powder coated PVC film to inactivate food pathogens. Int. J. Food Sci. Technol. 2009, 44, 2161–2168. [Google Scholar] [CrossRef]
- Ejaz, M.; Arfat, Y.A. Zinc oxide nanorods/clove essential oil incorporated Type B gelatin composite films and its applicability for shrimp packaging. Food Packag. Shelf Life 2018, 15, 113–121. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Soares, N.F.F. Zinc Oxide Nanoparticles: Synthesis, Antimicrobial Activity and Food Packaging Applications. Food Bioprocess. Technol. 2012, 5, 1447–1464. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, L. The Antibacterial Polyamide 6-ZnO Hierarchical Nanofibers Fabricated by Atomic Layer Deposition and Hydrothermal Growth. Nanoscale Res. Lett. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Prone, G.P.; Bermudez, P.S. Enhanced antibacterial nanocomposite mats by coaxial electrospinning of polycaprolactone fibers loaded with Zn-based nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 1695–1706. [Google Scholar] [CrossRef] [PubMed]
- Anitha, S.; Brabu, B. Optical, bactericidal and water repellent properties of electrospun nano-composite membranes of cellulose acetate and ZnO. Carbohydr. Polym. 2013, 97, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Kima, H.; Joshi, M.K. Polydopamine-assisted immobilization of hierarchical zinc oxide nanostructures on electrospun nanofibrous membrane for photocatalysis and antimicrobial activity. J. Colloid Interface Sci. 2018, 513, 566–574. [Google Scholar] [CrossRef] [PubMed]
- Malwal, D.; Gopinath, P. Efficient adsorption and antibacterial properties of electrospun CuO-ZnO composite nanofibers for water remediation. J. Hazard. Mater. 2017, 321, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, Y. Hydrophobic Ethylcellulose/Gelatin Nanofibers Containing Zinc Oxide Nanoparticles for Antimicrobial Packaging. J. Agric. Food Chem. 2018, 66, 9498–9506. [Google Scholar] [CrossRef] [PubMed]



| Ref | C0 | Well Type | Cell Line | ZnO NStr Type | ZnO NStr Dimensions | Cell Viability Assay |
|---|---|---|---|---|---|---|
| [41] | 1 × 105 cells/well | 96-well plates | T-cell | NPs | 4–20 nm | L/D |
| [41] | 5 × 104 cells/well | 96-well plates | Jurkat | NPs | 4–20 nm | L/D |
| [42] | 5 × 104 cells/well | 12-well culture dish | BEAS-2B | NPs | ~10 nm | WST |
| [53] | - | 96-well plates | HMMs | NWs | 120 nm × 2–5 µm | NR |
| [44] | 5 × 104 cells/cm2 | Flat 96-well plates | NHDF | TRPs | ~37 nm | MTT |
| [62] | 1 × 104 cells/mL in each well | 96-well plates | Mouse podocytes | NPs | 20–80 nm | MTT |
| [61] | 1 × 104 cells/well | 96-well plates | LCs | NPs | 70 nm | MTT |
| [61] | 1 × 104 cells/well | 96-well plates | SCs | NPs | MTT | |
| [47] | 1 × 104 cells/well | 96-well plates | HEK 293 | NPs | 25–40 nm | NR |
| [64] | 1 × 104 cells/well | 96-well plates | A549 | NCs | 4.7 ± 0.8 nm | MTT |
| [64] | 1 × 104 cells/well | 96-well plates | MRC-5 | MTT | ||
| [46] | 1 × 105 cells/well | 96-well round-bottom plates | FaDu | NPs | 20 nm | MTT |
| [46] | 1 × 105 cells/well | 96-well round-bottom plates | BMSC | NPs | TB | |
| [65] | 104 cells/mL | 96-well plates | HeLa | NPs | ~50 nm | WST |
| [55] | 1 × 104 cells well | 96-well plates | A549 (2) | NRs | diameter ≈ 52 nm | MTT |
| Type of System | Ref | Description of the System | ZnO Concentration | Cell Line/Bacteria | In Vivo Experiments | Main Results |
|---|---|---|---|---|---|---|
| ZnO NStr/ZnO array for experimental purposes | [74] | ZnO NFls arrays on Si substrate | Zinc nitrate solution 25 mM | MC3T3-E1 osteoblast culture | Implantation on calvarial bone defects of Sprague Dawley rats | Formation of lamellipodia and filopodia |
| [74] | ZnO NWs arrays incubated with a collagen solution | PC12 and H9C2 | _ | Adhesion, proliferation, and differentiation of two different electrically excitable mammalian cell lines | ||
| [77] | ZnO NWs arrays on a glass substrate | Mesoangioblasts | _ | - Reversibly locked differentiation - No cell damage - Differentiation capabilities completely recovered upon cell removal from the nanowire substrate and re-plating on standard culture glass | ||
| ZnO/PCL electrospun scaffold | [93] | PCL+ZnO NPs | 0.5–6 wt.% | HDFa | Implantation in guinea pigs | - Proangiogenic properties of ZnO/PCL fibers - Increase in the formation of mature blood vessels and highly branched capillary network |
| [91] | PCL and PCL/gelatin + ZnO NPs | 0, 5, 15, 30 wt.% | Pg, Fn, hDPSCs, AllCells LLC, Alameda, CA. | _ | - Potential application in periodontal regeneration - Good antibacterial properties | |
| [92] | PCL matrix + zero-valent Zn NPs | 5, 10, 15, 20 wt% | Neuroglioblastoma cells, human primary fibroblasts | _ | Small concentrations of Zn NPs promoted neuronal cell proliferation with relative non-toxicity for fibroblasts | |
| ZnO–polymeric (other polymers) electrospun implantable scaffold | [97] | ZnO–PU scaffold | 5 wt.% | mouse fibroblast | _ | Fibroblast viability, adhesion, and proliferation |
| [96] | (PVDF–TrFE) + ZnO NP scaffold | 0, 0.5, 1, 2, 4 wt.% | Red blood cells, White blood cells, platelet, hMSCs), HUVECs | Subcutaneous implantation in Wistar rats | - Tissue regeneration due to the piezoelectric properties of the composite components - Biocompatibility of the system in vitro - Angiogenic properties in vivo | |
| [94] | β-phase PVDF + ZnO NPs | 0.5, 1, 2 mg/mL | Human osteoblasts, S. aureus, methicillin-resistant S. aureus, E. coli. | _ | - Improvement of the elongation modulus at break and load stress - Greater osteoblast density and antibacterial properties of the piezoelectrically excited scaffold | |
| [98] | 1D ZnO-dopedTiO2 fabricated using colloidal gel | 1 and 10 μg/mL of ZnO/TO2 | C2C12 myoblast cells | _ | Beneficial effect on the adhesion, proliferation, and growth of myoblasts | |
| [36] | PMMA + ZnO NPs fibers and films | 0, 1, 3, 5, 10, 15 wt.%. | Fibroblast cells (L929) | _ | - Good proliferation of fibroblast cells - Thermal stability - Luminescence with emission in the near-UV range |
| Type of System | Ref | Description of the System | ZnO Concentration | Cell Line/Bacteria | In Vivo Experiments | Main Results |
|---|---|---|---|---|---|---|
| Electrospun fibrous membranes | [108] | Sodium alginate/poly(vinyl alcohol) fibrous mat + ZnO NPs | 0.5, 1, 2.5 wt.% | L929 fibroblasts cells, S. aureus, E. coli | - Fibers with 0.5 and 1% ZnO concentrations are less toxic - Inhibition for both the bacteria - Toxicity increase at the high ZnO concentration. | |
| [109] | PCL + ZnO NPs | 1, 2, 4 wt.% | Membranes implanted subcutaneously in guinea pigs | - ZnO enhanced the cell adhesion, migration, and proliferation -No significant sign of inflammation - In vivo implant enhanced the wound healing without any scar formation | ||
| [110] | Cellulose nanocrystal (CNC)–ZnO in poly(3-hydroxybutyrate-co-3-hydroxy-valerate) | CNC–ZnO suspension at 0, 3, 5, 10, 15 wt.% | E. coli and S. aureus | - Improvement in tensile strength and in Young’s modulus - High thermal stability -Good antibacterial activity | ||
| [111] | Chitosan/PVA/ZnO NP nanofibrous membranes | E. coli, P. aeruginosa, B. subtilis, S. aureus | Subcutaneous wounds in diabetes-induced rabbits | - High antibacterial and antioxidant potential - ZnO accelerated wound healing in vivo | ||
| Spongy hydrogels | [104] | Ag/ZnO into chitosan sponge | Immersion in 0.1, 0.2, 0.5, and 1.0 mg/mL of Ag/ZnO and in 0.5 mg/mL of ZnO solution | S. aureus, E. coli, P. aeruginosa, human normal hepatocyte (L02) | BALB/c mice: wound with a length of 7 mm on the back | - Evaluation of the porosity, swelling, blood clotting, and in vitro antibacterial activity -Low toxicity in vitro - Enhanced wound healing, re-epithelialization, and collagen deposition in vivo |
| [105] | Hydrogels of heparinized PVA/chitosan/ZnO NPs | Mouse fibroblast cells (L-929), E. coli, S. aureus | - Heparin release rate decreased by adding ZnO NPs - Good antibacterial protection of wounds | |||
| [106] | Porous keratin–chitosan/n-ZnO hydrogel | ZnO nanopowder 0, 0.5, 1 wt.% | fibroblasts cells (NIH 3T3), E. coli, S. aureus | Sprague-Dawley rats: skin wound of 1.5 cm2 in the dorsum of the rat | - Biocompatibility in vitro. - Increased wound curing in vivo with quicker skin cell construction and collagen development | |
| Gel and gelatin nanofibers or ointments | [112] | Cefazolin + ZnO NPs electrospun gelatin nanofiber mats | 1:1 w/w combination of cefazolin and ZnO NPs (1–64 μg/mL) | In vitro release studies + antibacterial property for S. aureus | Wistar rats: 2-cm-long incision | - Therapeutic approaches for post-operative wound - Determination of minimum inhibitory concentration - Hybrid antibacterial nature of ZnO NPs and cefazolin - Accelerated wound healing |
| [29] | AgNPs and Ag–ZnO NPs formulated into gel using Carbapol 934 | 0.1 g of NPs | Adult male albino Wistar rats: excision wound (4 cm length and 2 mm depth) | - Wound-healing properties of Ag–ZnO NPs in vivo - Rapid healing within 10 days when compared with pure AgNPs and standard drug dermazin |
| Type of System | Ref | Application | Description of the System | ZnO Concentration | Cell Line/Bacteria | Main Results |
|---|---|---|---|---|---|---|
| ZnO Coating | [120] | Antimicrobial activity against food pathogens | ZnO (ZnO nanoparticle suspension)-coated Polyvinyl chloride film | 93.75 and 187.5 ug/cm2 | E. coli, S. aureus, fungal Aspergillus flavus and Penicillium citrinum | - Antimicrobial activities of Polyvinyl chloride-based films to inactivate food pathogens - Effective antibacterial activity for S. aureus - No antifungal activity |
| [127] | Advanced functional textile | ZnO NP-coated polyvinylsilsesquioxane (PVSQ) composite | 0, 0.3, 0.5, 1, 2, 3 g | E. coli and S. aureus | - Excellent UV shielding and stable superhydrophobic properties - Enhanced mechanical properties and thermal stability - Larger resistivity of the E. coli compared to the S. aureus | |
| Electrospun nanocomposite membranes | [125] | Hydrophobic–bactericidal materials | ZnO NPs embedded on CA fibrous membrane | 0.2 mol of zinc acetate dihydrate | Staphylococcus aureus, E. coli, Klebsiella pneumoniae, Citrobacter freundii | - Hydrophobic nature of the surface - Strong antibacterial activity |
| [123] | Antibacterial application | PA-6 nanofiber modified with ZnO using ALD + hydrothermal reaction | 100–150 cycles of ALD with ZnO seed layers (14.6 nm) | S. aureus | Efficient in suppression of bacteria survivorship | |
| [127] | Removal of biological/organic contaminants for water treatment and purification | CuO–ZnO–PVA nanofibers | 50, 100, 150, 200, 250, 300, 350 ug/mL | E. coli and S. aureus | - Enhanced adsorption efficiency and antibacterial properties - Excellent adsorption capacity for congo red dye | |
| [126] | Photocatalysis and antimicrobial activity for organic pollutant degradation and waste water purification | Hierarchical ZnO NR deposited on PU nanofiber | E. coli | - High photocatalytic/antimicrobial activity at the low-intensity UV LED device with good reusability - Measure of the degradation of the methylene blue (MB) solution | ||
| [124] | Antibacterial nanocomposite wound dressings | ZnO NP–PCL uniaxial or coaxial fiber structure | ZnO NPs 9, 12, 15 and 25 wt.% relative to PCL | E. coli, S. aureus | - Inhibition of planktonic and biofilm bacterial growth - Increased antibacterial properties for coaxial fibers and for exposure to UV-A light prior to bacteria inoculation | |
| Gelatin composite films | [121] | Active shrimp packaging | ZnO NRs/clove essential oil incorporated into type B gelatin composite films | NR loading concentration 2% w/w of gelatin | Listeria monocytogenes and Salmonella typhimurium | - Film with low flexibility and high mechanical resistance- Oxygen and UV barrier property increased with ZnO NR incorporation - Composite films loaded with 50% clove essential oil and with ZnO NRs showed maximum antibacterial activity |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrone, E.; Araneo, R.; Notargiacomo, A.; Pea, M.; Rinaldi, A. ZnO Nanostructures and Electrospun ZnO–Polymeric Hybrid Nanomaterials in Biomedical, Health, and Sustainability Applications. Nanomaterials 2019, 9, 1449. https://doi.org/10.3390/nano9101449
Ferrone E, Araneo R, Notargiacomo A, Pea M, Rinaldi A. ZnO Nanostructures and Electrospun ZnO–Polymeric Hybrid Nanomaterials in Biomedical, Health, and Sustainability Applications. Nanomaterials. 2019; 9(10):1449. https://doi.org/10.3390/nano9101449
Chicago/Turabian StyleFerrone, Eloisa, Rodolfo Araneo, Andrea Notargiacomo, Marialilia Pea, and Antonio Rinaldi. 2019. "ZnO Nanostructures and Electrospun ZnO–Polymeric Hybrid Nanomaterials in Biomedical, Health, and Sustainability Applications" Nanomaterials 9, no. 10: 1449. https://doi.org/10.3390/nano9101449
APA StyleFerrone, E., Araneo, R., Notargiacomo, A., Pea, M., & Rinaldi, A. (2019). ZnO Nanostructures and Electrospun ZnO–Polymeric Hybrid Nanomaterials in Biomedical, Health, and Sustainability Applications. Nanomaterials, 9(10), 1449. https://doi.org/10.3390/nano9101449







