Identification of Nanocellulose Retention Characteristics in Porous Media
Abstract
:1. Introduction
2. Materials and Methods
2.1. Porous Media
2.2. Brine
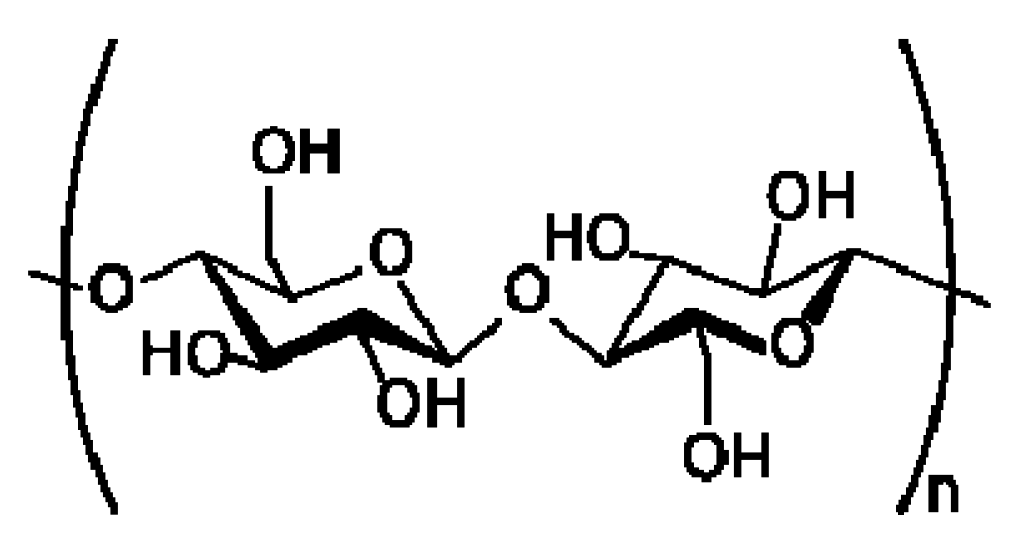
2.3. Nanocellulose
3. Experimental Methods
3.1. Atomic-Force Microscopy (AFM)
3.2. Nanocellulose Aggregate Size and Zeta Potential Measurements
3.3. Batch Adsorption Experiment
Phenol-Sulfuric Acid Method
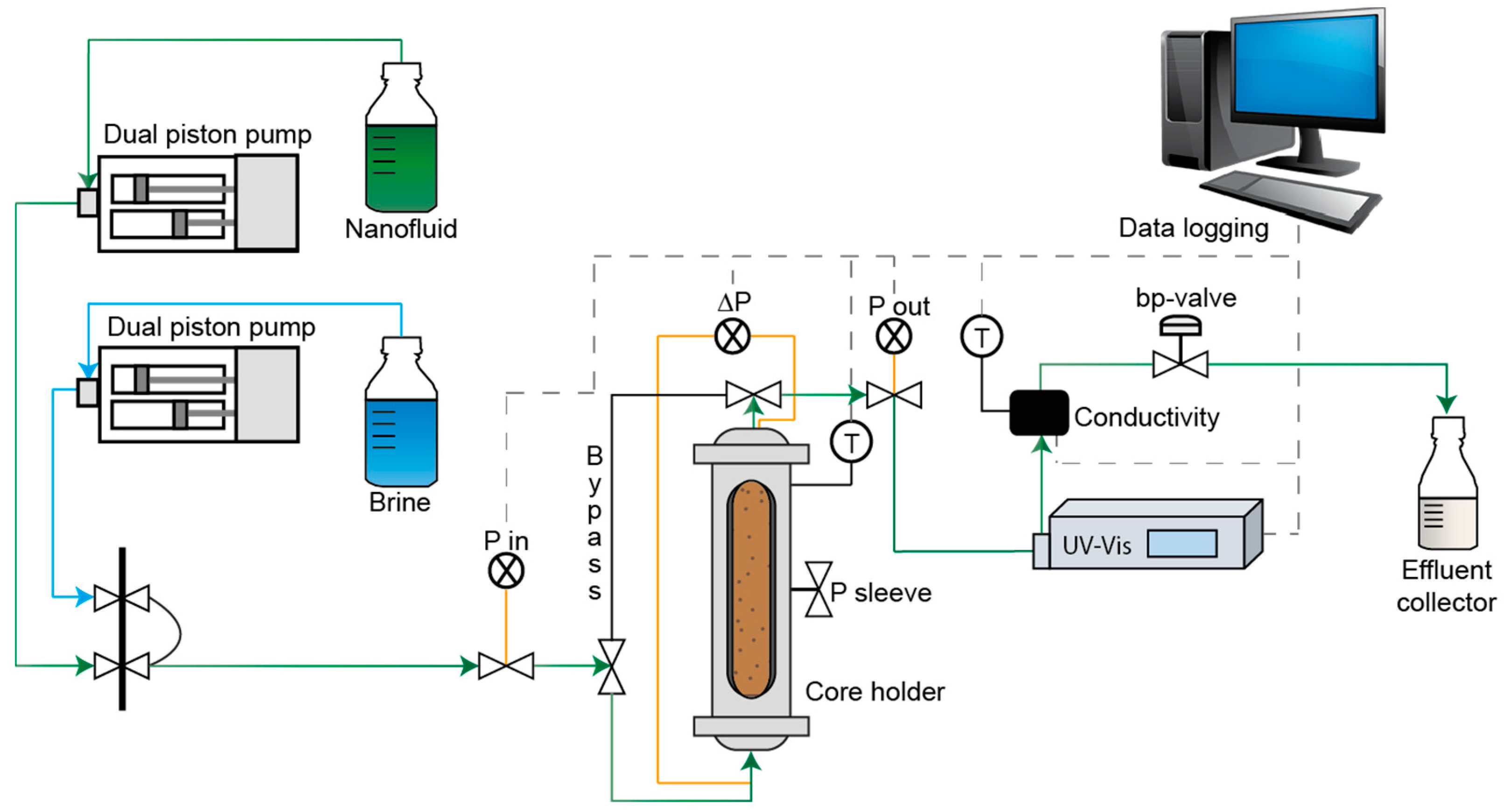
3.4. Sandpack Retention Flooding Experiments
3.4.1. Preparation of Sandpacks
3.4.2. Sandpack Flooding Procedure
3.5. Observing Nanocellulose Retention in Sandpack
3.6. Berea Sandstone Retention Flooding Experiments
3.6.1. Preparation of Berea Sandstone Core Plugs
3.6.2. Core Flooding Procedure
3.7. Determining Cellulose Retention from Mass Balance
4. Results and Discussion
4.1. Atomic-Force Microscopy (AFM)
4.2. Particle Size and Zeta Potential
4.3. Batch Adsorption Experiments
4.4. Sandpack Flooding Results
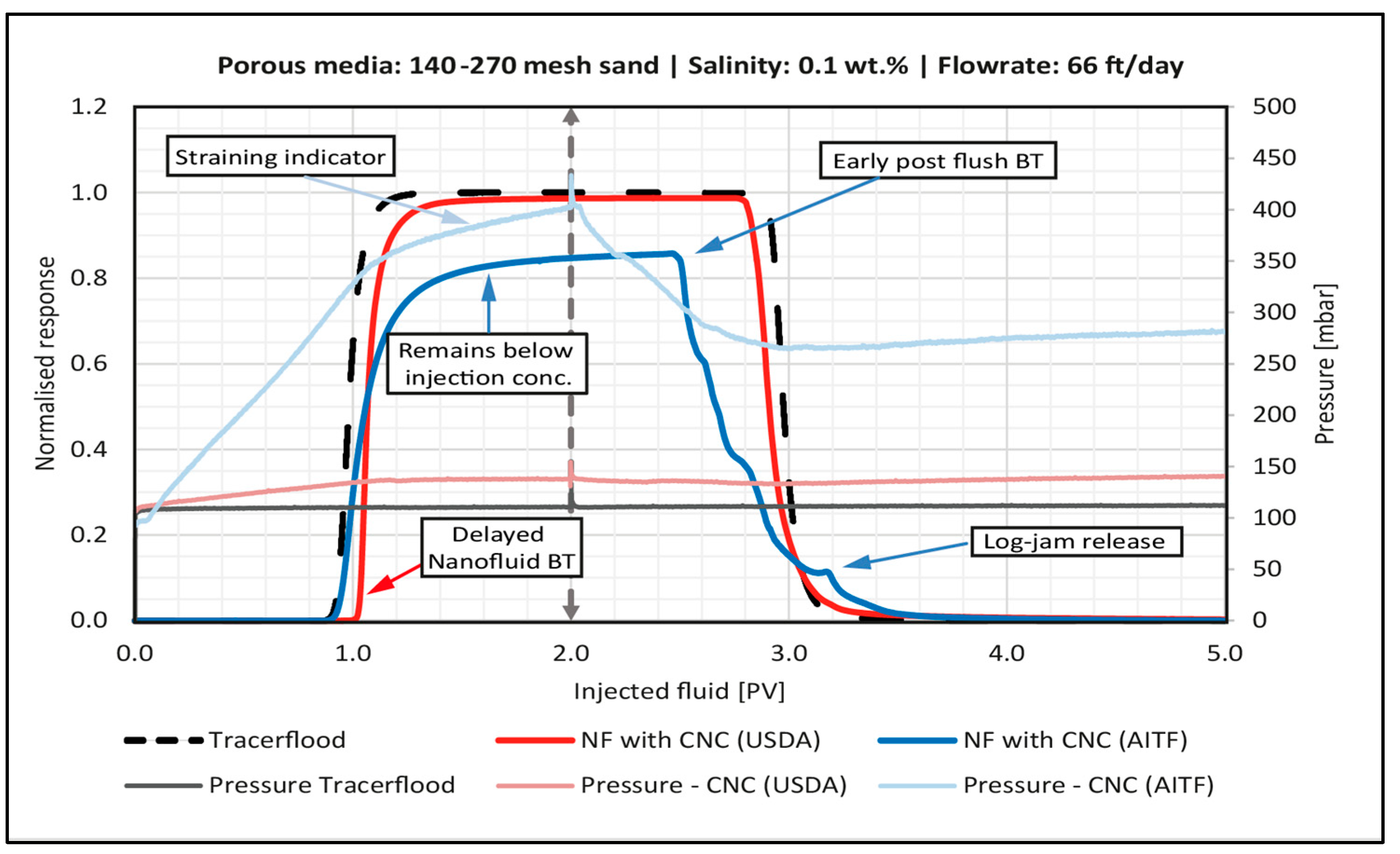
4.4.1. Effect of Particle Type
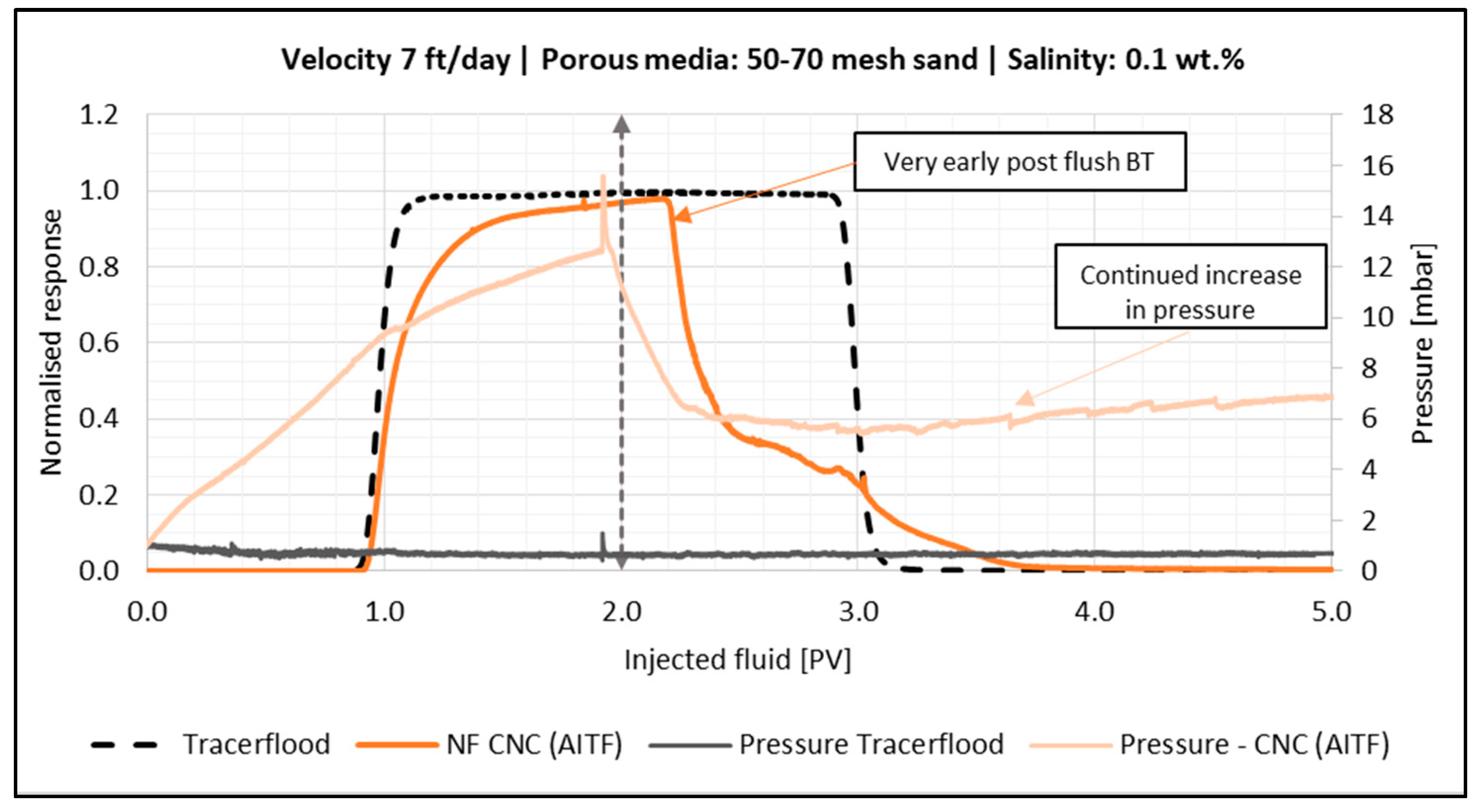
4.4.2. Effect of Flow Velocity
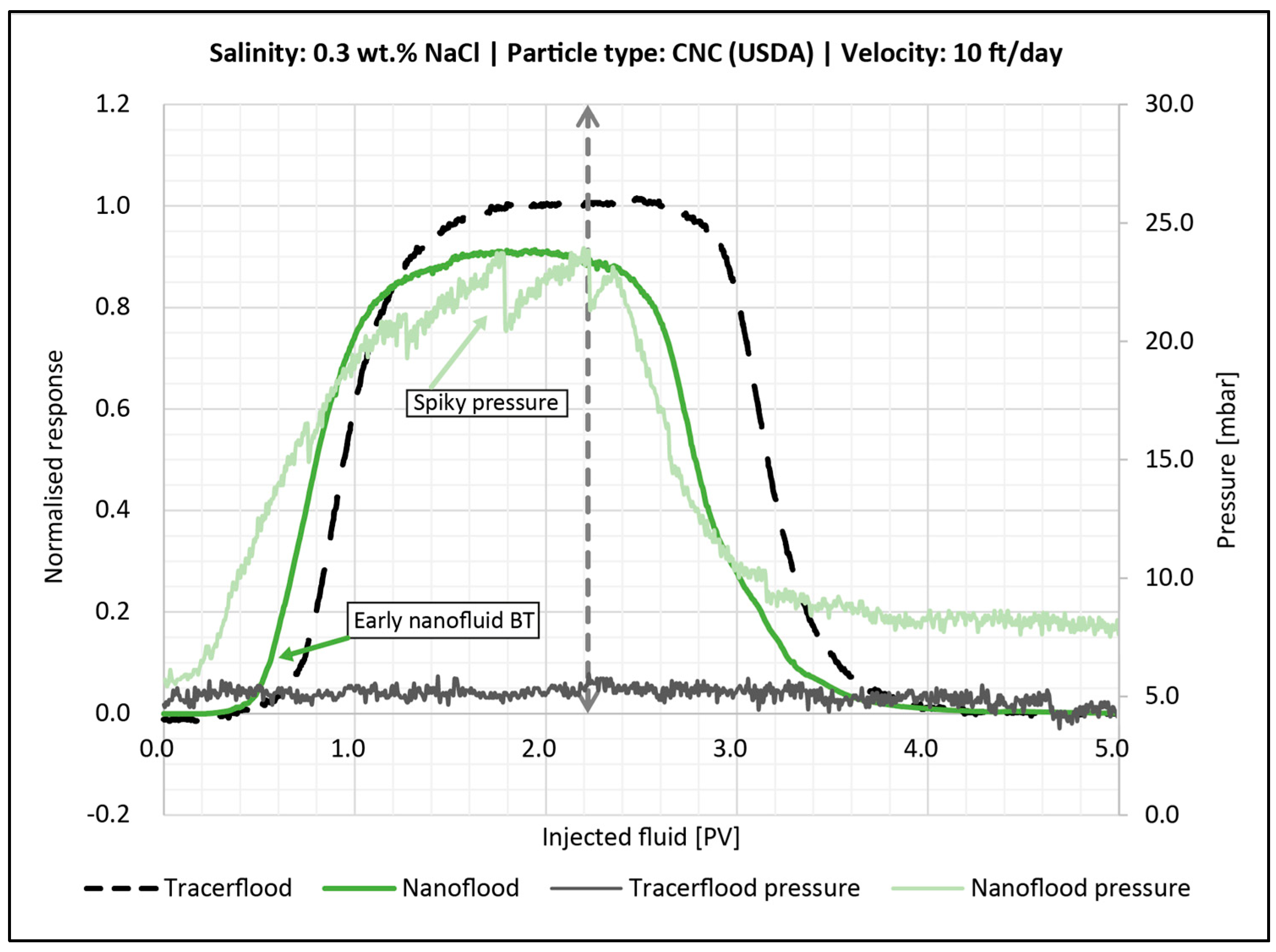
4.4.3. Effect of Salinity
4.4.4. Effect of Sand Grain Size
4.5. Direct Observation of Sandpack Retention
4.6. Berea Sandstone Core Flooding Results
4.6.1. Effect of 0.1 wt. % NaCl
4.6.2. Effect of 0.3 wt. % NaCl
5. Conclusions
- In the static adsorption experiments, it was found that neither of the two nanocellulose types seemed to adsorb significantly on the sand grains.
- CNC (USDA) and CNC (AITF) nanoparticles were different in size, which contributed to different behavior when flooded with 0.1 wt. % NaCl in the sandpacks.
- Salinity had the largest effect on retention, since it changed the aggregated particle size in the bulk solution. For both porous media (sandpack and core plug), a higher retention was observed when salinity was increased.
- Retention and permeability reduction in sandpacks increased as the velocity decreased, and the same trend was seen by decreasing the sand grain size.
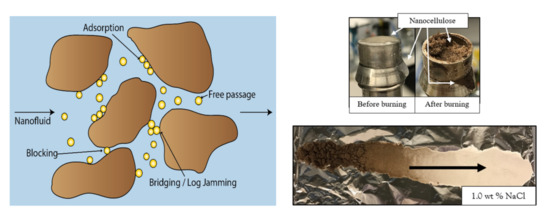
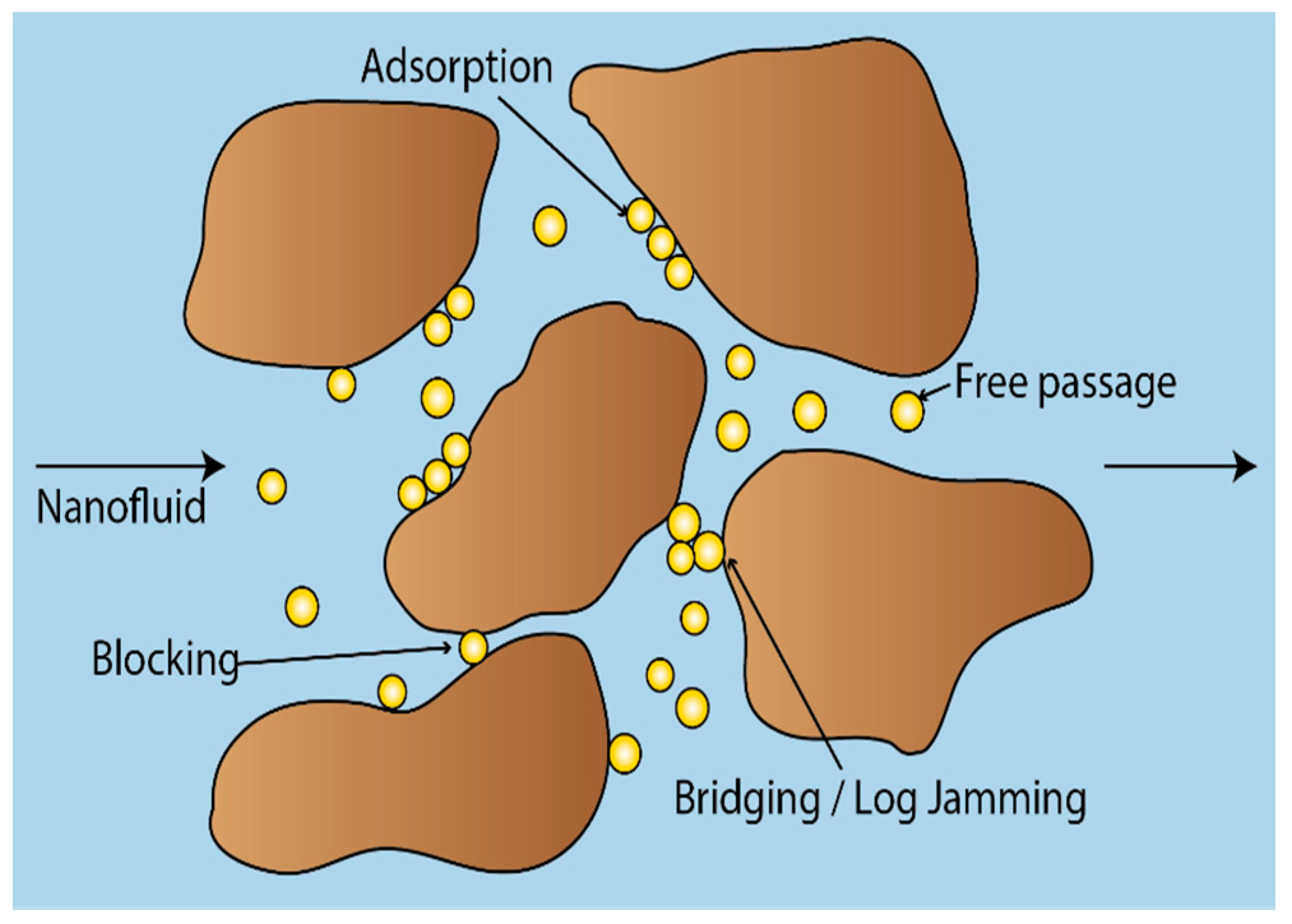
- In general, for the sandpack experiments, smaller particles seemed to be dominated by adsorption on sand grains, whereas larger particles were retained in the sandpack by blocking and log-jamming. For the core floods, it also seemed like adsorption was the dominating mechanism in the low salinity flood, while bridging and log-jamming of particles appeared to be the major factor for the medium salinity flood.
- The sand from the sandpack floods were baked after each experiment, which provided qualitative data and could further support the findings from the calculations and breakthrough curves.
- In the flow experiments using core plugs, it was evident that the nanocellulose could not enter some of the pore volume.
- For polymers, 200 μg/g appears to be a maximum value for retention in order for the solution to still be considered viable. By comparing this value against the CNC (USDA) floods in this study, only the flood with 1.0 wt. % NaCl exceeds this criterion where the amount of retained particles was 2.2 mg/g. For CNC (AITF), both the floods with low velocity (7 ft/day) and the flood in the small grain size pack (140–270 mesh) exceeded the maximum retention value. Furthermore, it was not possible to get a mass balance at 0.3 wt. % NaCl for CNC (AITF) due to severe aggregation. Thus, it is likely that the retention value was higher than 200 μg/g for this particle at this salinity.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kamal, M.S.; Sultan, A.S.; Al-Mubaiyedh, U.A.; Hussein, I.A. Review on Polymer Flooding: Rheology, Adsorption, Stability, and Field Applications of Various Polymer Systems. Polym. Rev. 2015, 55, 491–530. [Google Scholar] [CrossRef]
- Raney, K.H.; Ayirala, S.; Chin, R.W.; Verbeek, P. Surface and Subsurface Requirements for Successful Implementation of Offshore Chemical Enhanced Oil Recovery. SPE Prod. Oper. 2012, 27, 294–305. [Google Scholar] [CrossRef]
- Sheng, J.J.; Leonhardt, B.; Azri, N. Status of Polymer-Flooding Technology. J. Can. Pet. Technol. 2015, 54, 116–126. [Google Scholar] [CrossRef]
- Chang, H.L. Polymer Flooding Technology Yesterday, Today, and Tomorrow. J. Pet. Technol. 1978, 30, 1113–1128. [Google Scholar] [CrossRef]
- Lake, L.W. Enhanced Oil Recovery; Prentice-Hall, Inc.: Upper Saddle River, NJ, USA, 1989. [Google Scholar]
- Wellington, S.L. Biopolymer solution viscosity stabilization-polymer degradation and antioxidant use. Soc. Pet. Eng. J. 1983, 23, 901–912. [Google Scholar] [CrossRef]
- Dufresne, A. Nanocellulose: From Nature to High Performance Tailored Materials; Walter de Gruyter: Berlin, Germany, 2012. [Google Scholar]
- Klemm, D.; Kramer, F.; Moritz, S.; Lindström, T.; Ankerfors, M.; Gray, D.; Dorris, A. Nanocelluloses: A new family of nature-based materials. Angew. Chem. Int. Ed. 2011, 50, 5438–5466. [Google Scholar] [CrossRef] [PubMed]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef] [PubMed]
- Molnes, S.N.; Mamonov, A.; Paso, K.G.; Strand, S.; Syverud, K. Investigation of a new application for cellulose nanocrystals: A study of the enhanced oil recovery potential by use of a green additive. Cellulose 2018, 25, 2289–2301. [Google Scholar] [CrossRef]
- Wei, B.; Li, Q.; Jin, F.; Li, H.; Wang, C. The Potential of a Novel Nanofluid in Enhancing Oil Recovery. Energy Fuels 2016, 30, 2882–2891. [Google Scholar] [CrossRef]
- Molnes, S.N.; Torrijos, I.P.; Strand, S.; Paso, K.G.; Syverud, K. Sandstone injectivity and salt stability of cellulose nanocrystals (CNC) dispersions—Premises for use of CNC in enhanced oil recovery. Ind. Crop. Prod. 2016, 93, 152–160. [Google Scholar] [CrossRef]
- Ju, B.; Fan, T. Experimental study and mathematical model of nanoparticle transport in porous media. Powder Technol. 2009, 192, 195–202. [Google Scholar] [CrossRef]
- Manichand, R.N.; Seright, R. Field vs. laboratory polymer-retention values for a polymer flood in the Tambaredjo field. SPE Reserv. Eval. Eng. 2014, 17, 314–325. [Google Scholar] [CrossRef]
- Green, D.W.; Willhite, G.P. Enhanced Oil Recovery; Henry, L., Ed.; Doherty Memorial Fund of AIME, Society of Petroleum Engineers: Richardson, TX, USA, 1998. [Google Scholar]
- Zhang, G.; Seright, R. Effect of concentration on HPAM retention in porous media. SPE J. 2014, 19, 373–380. [Google Scholar] [CrossRef]
- Martin, F.; Hatch, M.; Shepitka, J.; Ward, J. Improved water-soluble polymers for enhanced recovery of oil. In Proceedings of the SPE Oilfield and Geothermal Chemistry Symposium, Denver, CO, USA, 1–3 June 1983. [Google Scholar]
- Lotsch, T.; Muller, T.; Pusch, G. The effect of inaccessible pore volume on polymer coreflood experiments. In Proceedings of the SPE Oilfield and Geothermal Chemistry Symposium, Phoenix, AZ, USA, 9–11 March 1985. [Google Scholar]
- Huh, C.; Lange, E.A.; Cannella, W.J. Polymer Retention in Porous Media. In Proceedings of the SPE/DOE Enhanced Oil Recovery Symposium, Tulsa, OK, USA, 22–25 April 1990. [Google Scholar]
- Zhang, T.; Murphy, M.J.; Yu, H.; Bagaria, H.G.; Yoon, K.Y.; Nielson, B.M.; Bielawski, C.W.; Johnston, K.P.; Huh, C.; Bryant, S.L. Investigation of Nanoparticle Adsorption During Transport in Porous Media. SPE J. 2014, 20, 667–677. [Google Scholar] [CrossRef]
- Dong, X.M.; Revol, J.F.; Gray, D.G. Effect of microcrystallite preparation conditions on the formation of colloid crystals of cellulose. Cellulose 1998, 5, 19–32. [Google Scholar] [CrossRef]
- Sartor, M. Dynamic Light Scattering; University of California: San Diego, CA, USA, 2003; pp. 2–21. [Google Scholar]
- Zhang, T. Modeling of Nanoparticle Transport in Porous Media. Ph.D. Thesis, Department of Petroleum and Geosystems Engineering, University of Texas at Austin, Austin, TX, USA, 2012; p. 311. Available online: http://hdl.handle.net/2152/ETD-UT-2012-08-6044 (accessed on 26 September 2017).
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Chung, D.H. Transport of Nanoparticle during Drainage and Imbibition Displacements in Porous Media. Master’s Thesis, University of Texas at Austin, Austin, TX, USA, 2013. Available online: http://hdl.handle.net/2152/22366 (accessed on 4 January 2018).
- Heggset, E.B.; Chinga-Carrasco, G.; Syverud, K. Temperature stability of nanocellulose dispersions. Carbohydr. Polym. 2017, 57, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Dziuba, C. Investigation of Single Phase NanoCellulose Transport through Porous Media. Master’s Thesis, University of Calgary, Calgary, AB, Canada, 2017. Available online: http://hdl.handle.net/11023/3773 (accessed on 5 June 2017).
- Xu, H.-N.; Tang, Y.-Y.; Ouyang, X.-K. Shear-Induced Breakup of Cellulose Nanocrystal Aggregates. Langmuir 2016, 33, 235–242. [Google Scholar] [CrossRef] [PubMed]










| Sand | Grain Size | Porosity | Permeability | Pore Volume |
|---|---|---|---|---|
| [Mesh] | [μm] | [%] | [D] | [mL] |
| 140–270 | 53–105 | 39 | 3 | 23.6 |
| 50–70 | 210–297 | 37 | 35 | 21.9 |
| 16–30 | 595–1190 | 33 | 282 | 19.7 |
| Core | Porosity | Permeability | Pore Volume |
|---|---|---|---|
| [Number] | [%] | [mD] | [mL] |
| 1 | 16.7 | 803 | 19.3 |
| 2 | 16.0 | 922 | 18.5 |
| Variable Tested | Values Selected | |
|---|---|---|
| Sandpack Floods | Core Floods | |
| Salinity (wt. %) | 0.1 | 0.1 |
| 0.3 | 0.3 | |
| 1.0 | - | |
| Particle Type | CNC (USDA) | CNC (USDA) |
| CNC (AITF) | - | |
| Grain Size (mesh) | 140–270 | n/a |
| 50–70 | ||
| 16–30 | ||
| Velocity (ft/day) | 7 | 10 |
| 66 | ||
| 521 | ||
| Parameter | 0.5 wt. % CNC (USDA) | 0.5 wt. % CNC (AITF) | |||||
|---|---|---|---|---|---|---|---|
| Salinity (wt. %) | 0 | 0.1 | 0.3 | 1.0 | 0 | 0.1 | 0.3 |
| Avg. Aggregate Size (nm) | 53 ± 3 | 67 ± 3 | 298 ± 12 | 913 ± 132 | 166 ± 15 | 321 ± 29 | 1347 ± 186 |
| Zeta Potential (mv) | - | −30.3 ± 1.8 | −21.2 ± 2.4 | −11.0 ± 1.3 | - | −23.6 ± 2.2 | −15.7 ± 1.0 |
| Parameter Changed | Retention [mg/g Rock] | Permeability Reduction [%] | ||
|---|---|---|---|---|
| CNC (USDA) | CNC (AITF) | CNC (USDA) | CNC (AITF) | |
| Velocity | ||||
| 7 ft/day | - | 0.60 | - | 90 |
| 66 ft/day * | 0.14 | 0.15 | 6 | 13 |
| 521 ft/day | - | 0.18 | - | 8 |
| Salinity | ||||
| 0.1 wt. % NaCl * | 0.14 | 0.15 | 6 | 13 |
| 0.3 wt. % NaCl | 0.19 | - | 24 | 97 |
| 1.0 wt. % NaCl | 2.20 | - | 97 | - |
| Grain size | ||||
| 140–270 mesh | 0.20 | 0.69 | 22 | 66 |
| 50–70 mesh * | 0.14 | 0.15 | 6 | 13 |
| 16–30 mesh | 0.05 | 0.07 | 5 | 14 |
| Salinity | Retention | Permeability Reduction |
|---|---|---|
| [mg/g Rock] | [%] | |
| 0.1 wt. % NaCl | 0.015 | 18 |
| 0.3 wt. % NaCl | 0.12 | 19 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aadland, R.C.; Dziuba, C.J.; Heggset, E.B.; Syverud, K.; Torsæter, O.; Holt, T.; Gates, I.D.; Bryant, S.L. Identification of Nanocellulose Retention Characteristics in Porous Media. Nanomaterials 2018, 8, 547. https://doi.org/10.3390/nano8070547
Aadland RC, Dziuba CJ, Heggset EB, Syverud K, Torsæter O, Holt T, Gates ID, Bryant SL. Identification of Nanocellulose Retention Characteristics in Porous Media. Nanomaterials. 2018; 8(7):547. https://doi.org/10.3390/nano8070547
Chicago/Turabian StyleAadland, Reidun C., Carter J. Dziuba, Ellinor B. Heggset, Kristin Syverud, Ole Torsæter, Torleif Holt, Ian D. Gates, and Steven L. Bryant. 2018. "Identification of Nanocellulose Retention Characteristics in Porous Media" Nanomaterials 8, no. 7: 547. https://doi.org/10.3390/nano8070547
APA StyleAadland, R. C., Dziuba, C. J., Heggset, E. B., Syverud, K., Torsæter, O., Holt, T., Gates, I. D., & Bryant, S. L. (2018). Identification of Nanocellulose Retention Characteristics in Porous Media. Nanomaterials, 8(7), 547. https://doi.org/10.3390/nano8070547