Applications of Nanomaterials Based on Magnetite and Mesoporous Silica on the Selective Detection of Zinc Ion in Live Cell Imaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
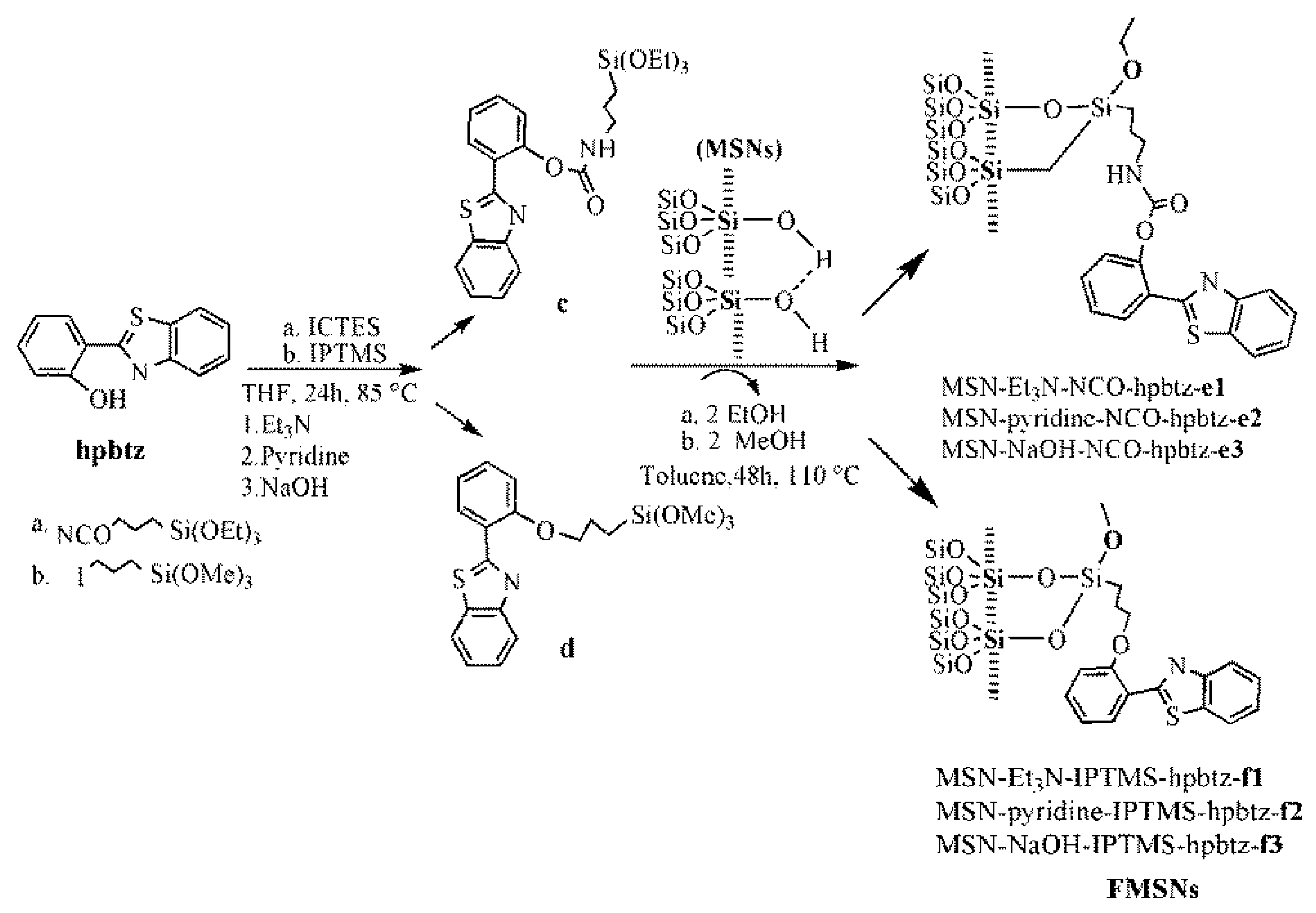
2.2. Preparation of Functionalized Mesoporous Silica Nanoparticles (FMSNs)
2.2.1. Synthesis of Mesoporous Silica Nanoparticles (MSNs)
2.2.2. Synthesis of e1–e3 in the Presence of Different Bases
2.2.3. Synthesis of f1–f3 in the Presence of Different Bases
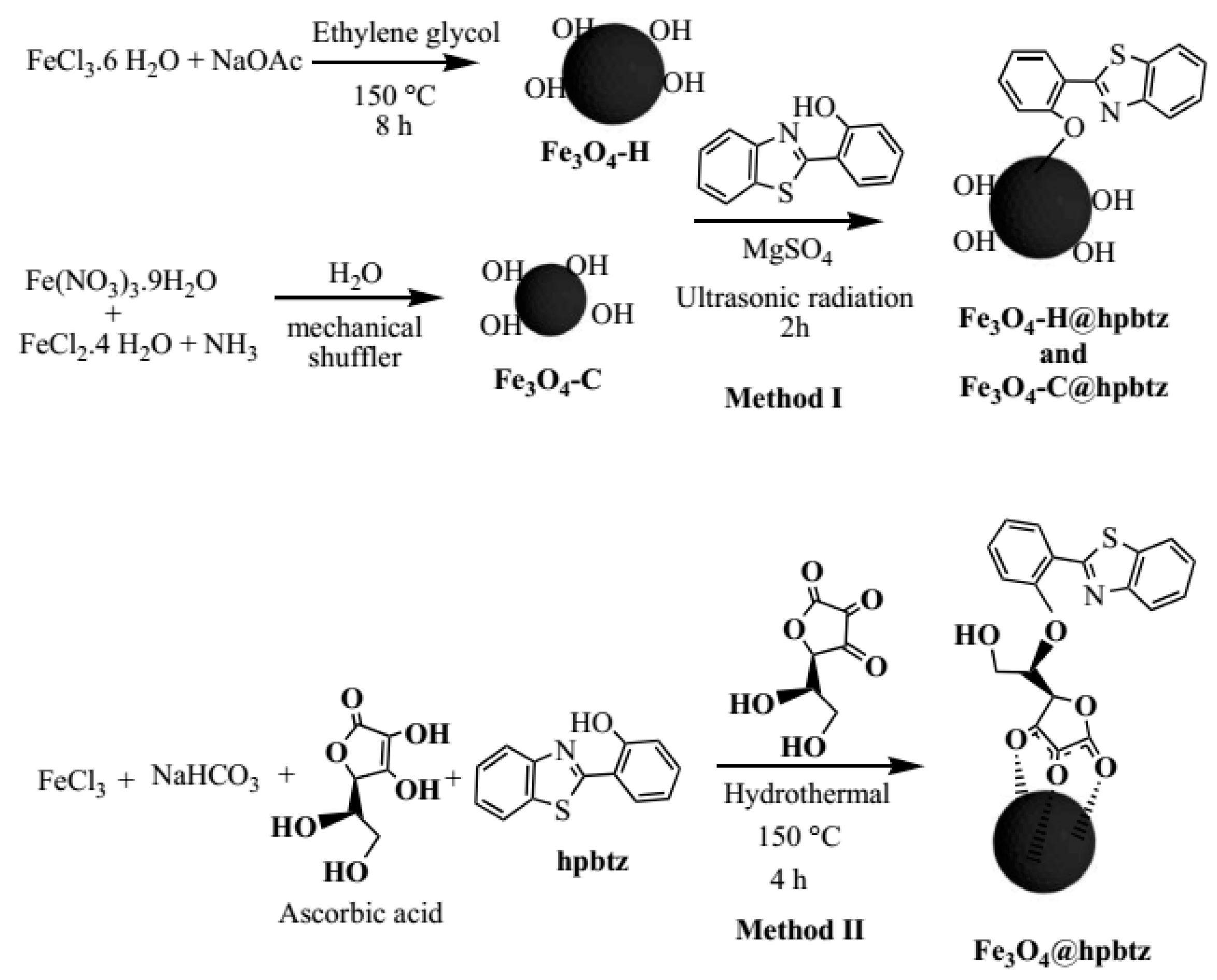
2.3. Detailed Synthesis Methodology of MNPs and FMNPs
2.3.1. Synthesis of Magnetite Nanoparticles under Hydrothermal Condition (Fe3O4-H)
2.3.2. Synthesis of Magnetite Nanoparticles by Coprecipitation (Fe3O4-C)
2.3.3. Preparation of Fe3O4-H@hpbtz and Fe3O4-C@hpbtz (Method I).
2.3.4. Preparation of Fe3O4@hpbtz (Method II)
2.4. Fluorescence Spectral Measurements
2.5. In Vitro Cytotoxicity Study (MTT Assay)
2.6. Fluorescence Imaging Experiments
2.7. Characterization Instruments and Conditions
3. Results and Discussion
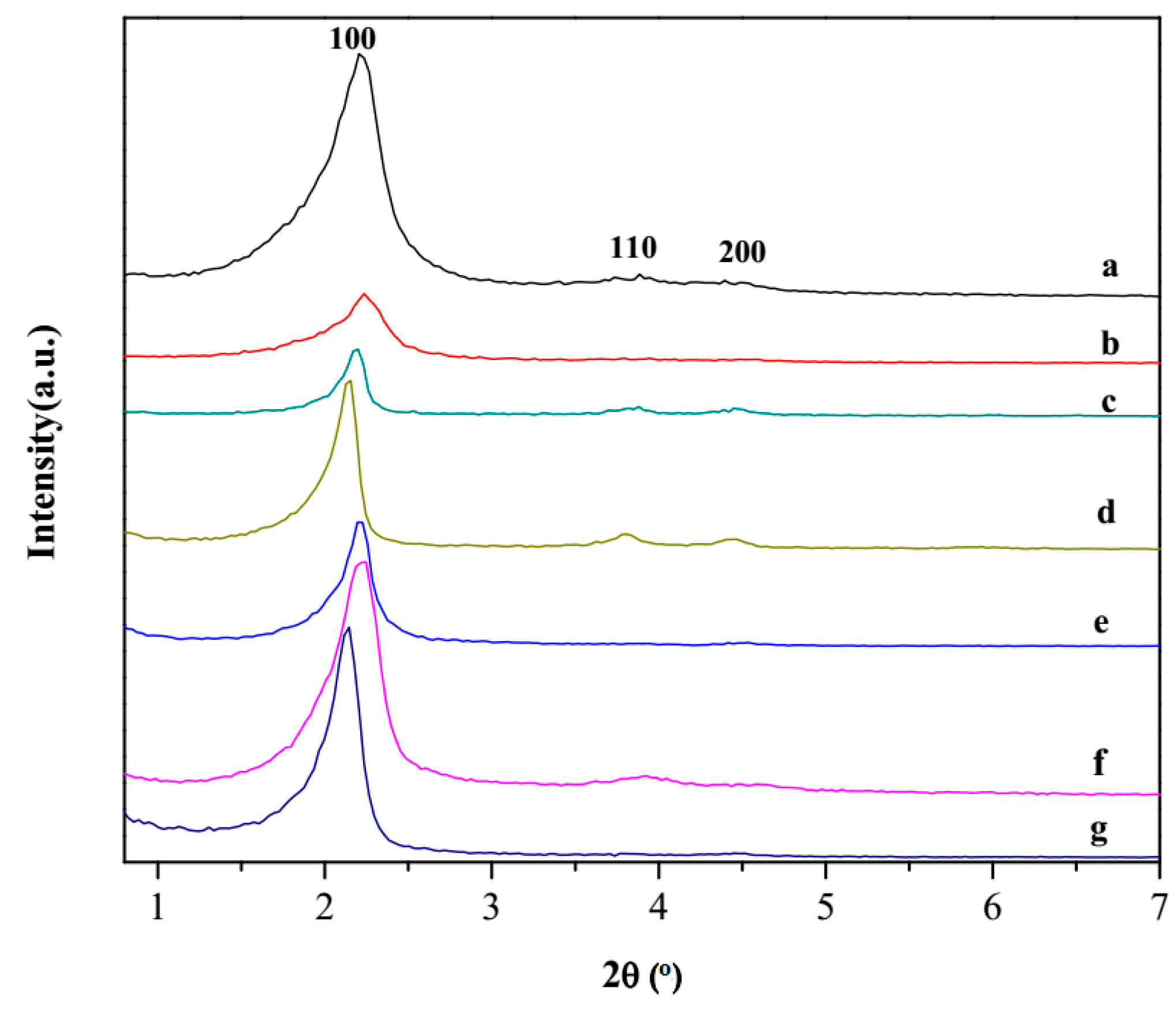
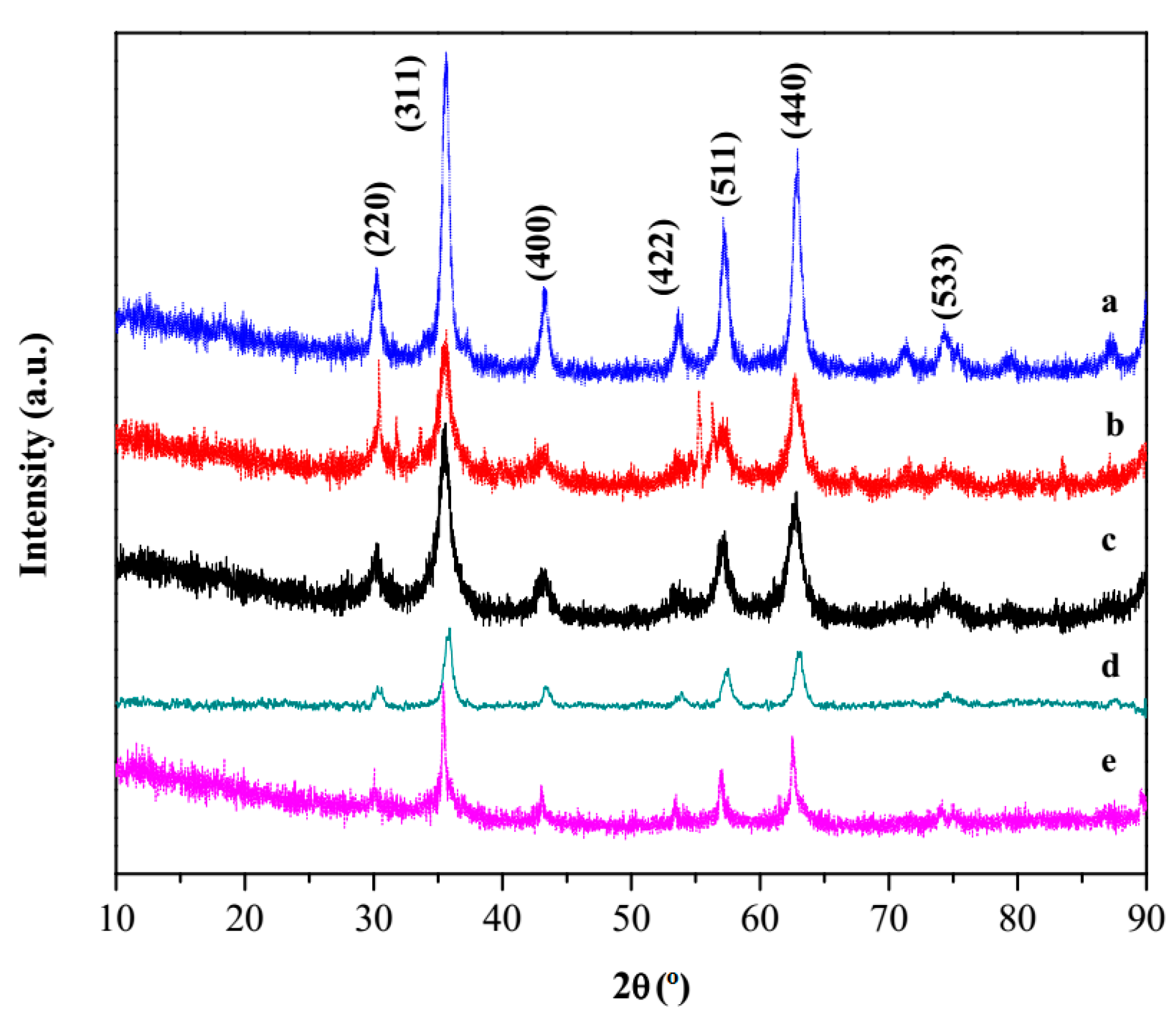
3.1. Synthesis and Characterization of Nanomaterials
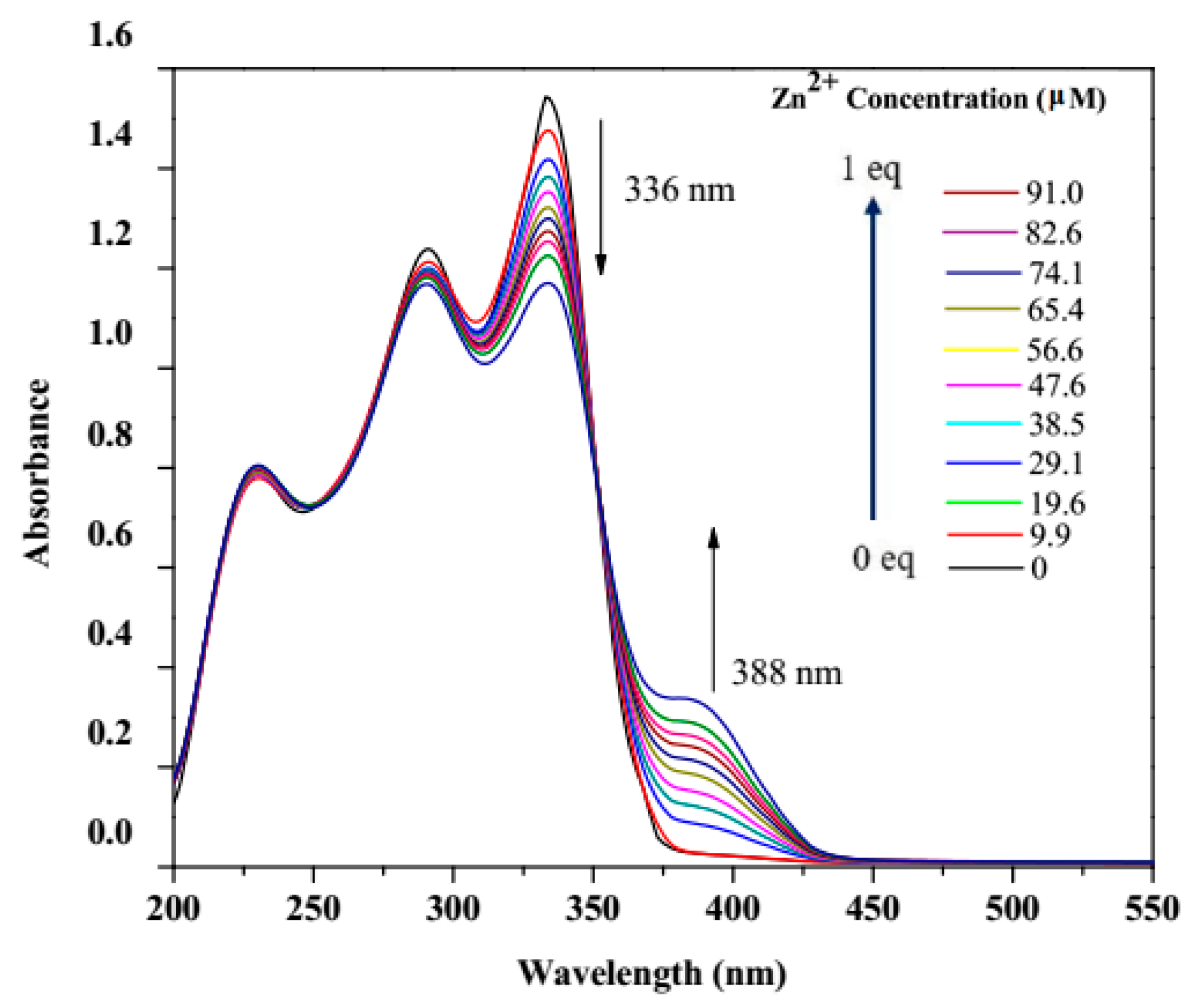
3.2. UV–Vis Titration
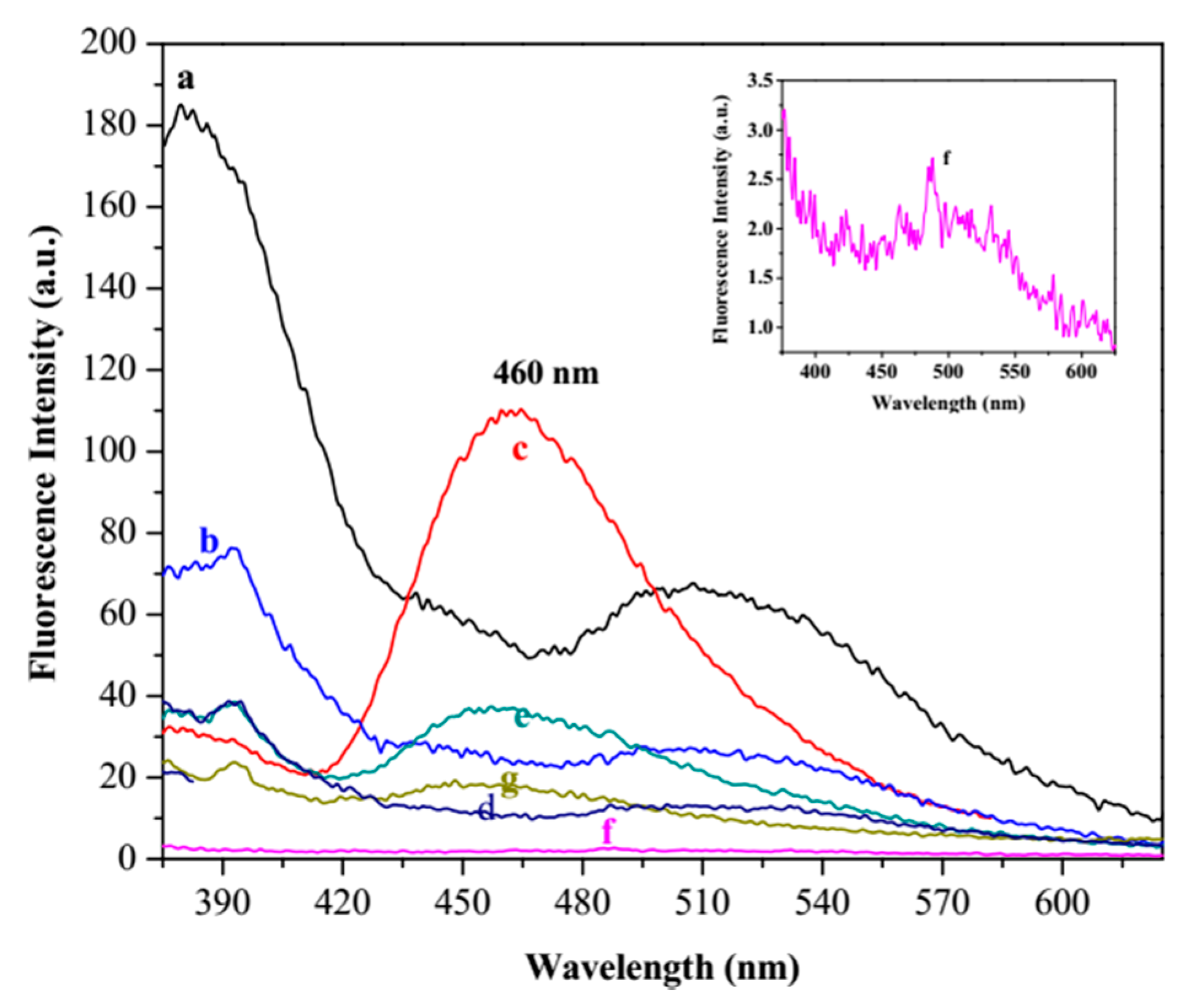
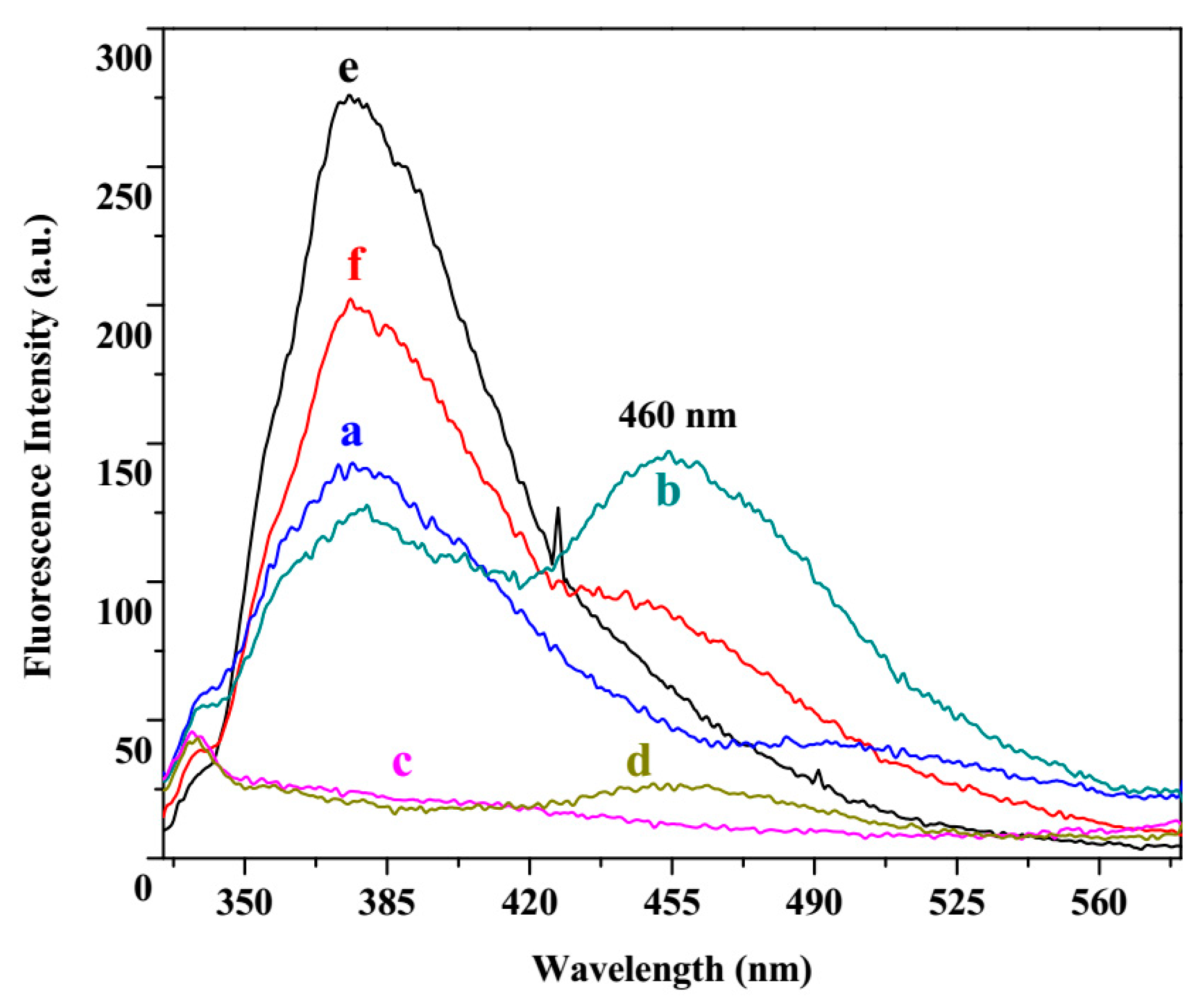
3.3. Fluorescence Study
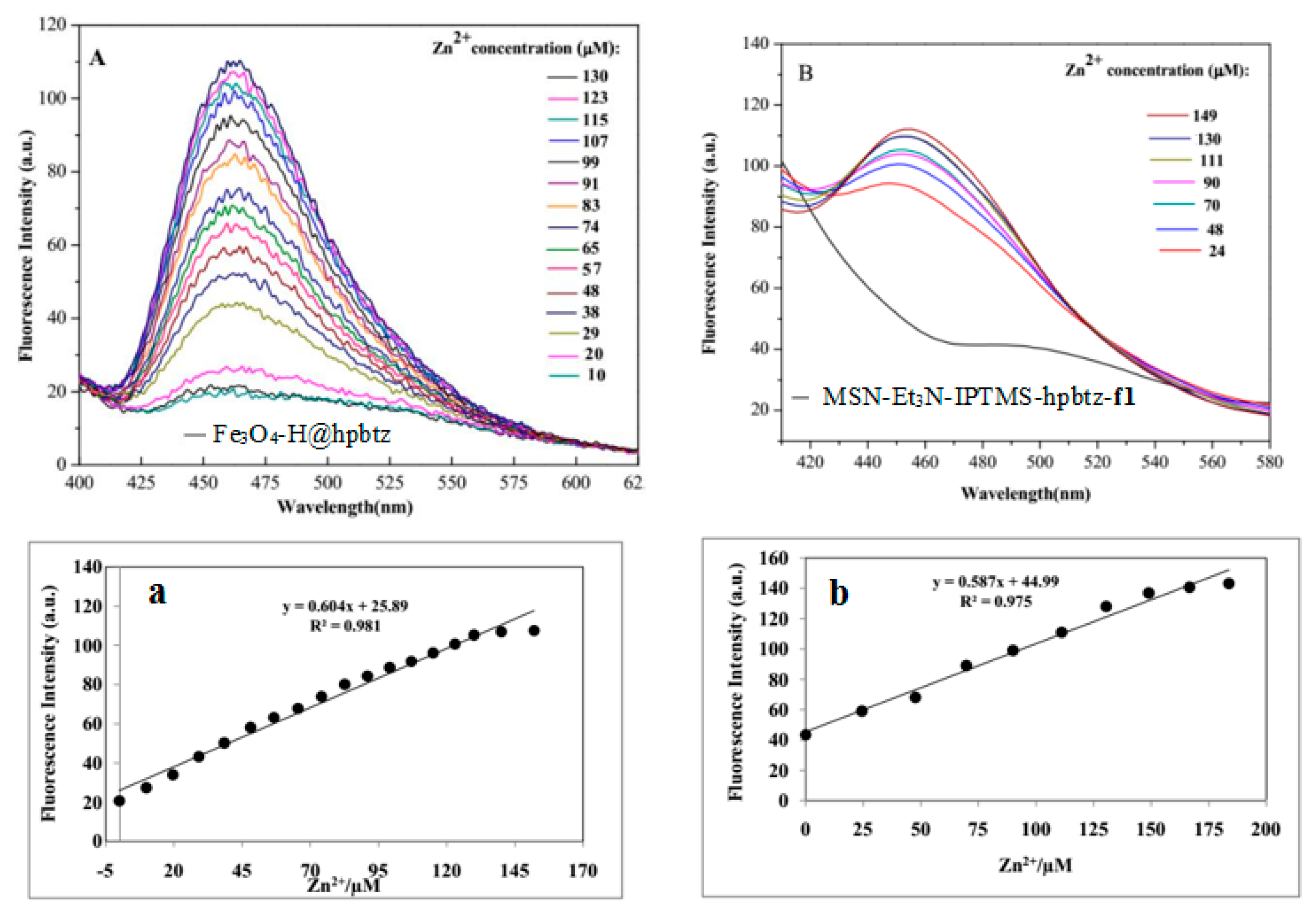
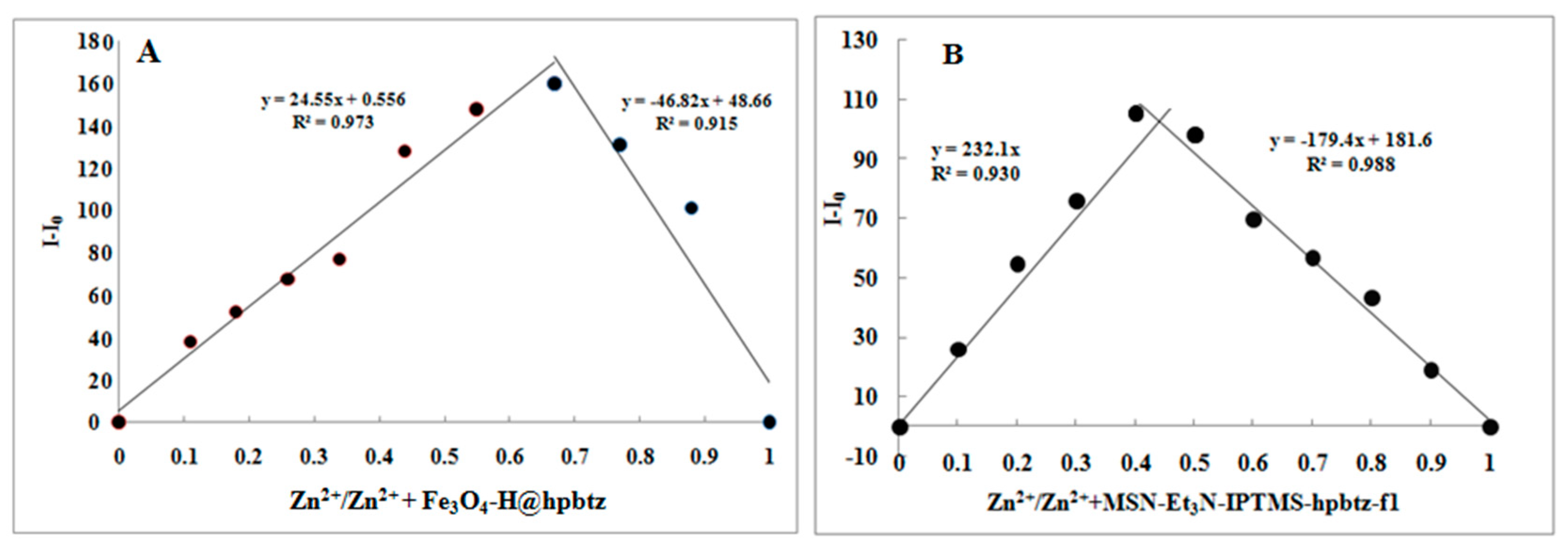
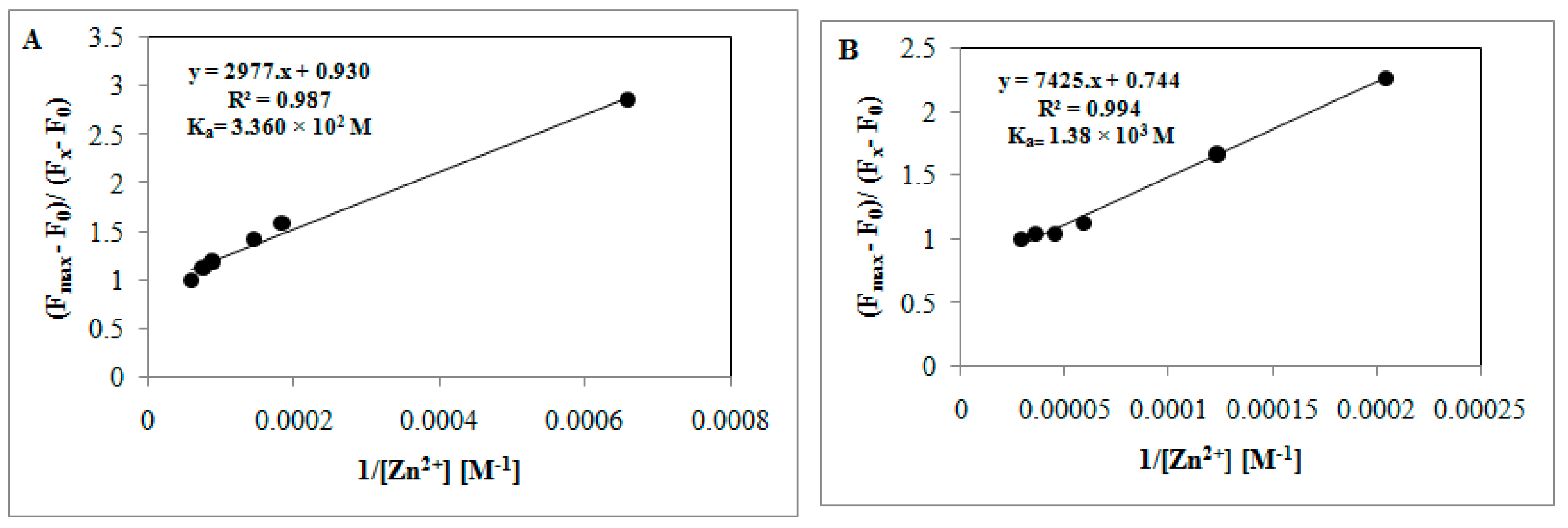
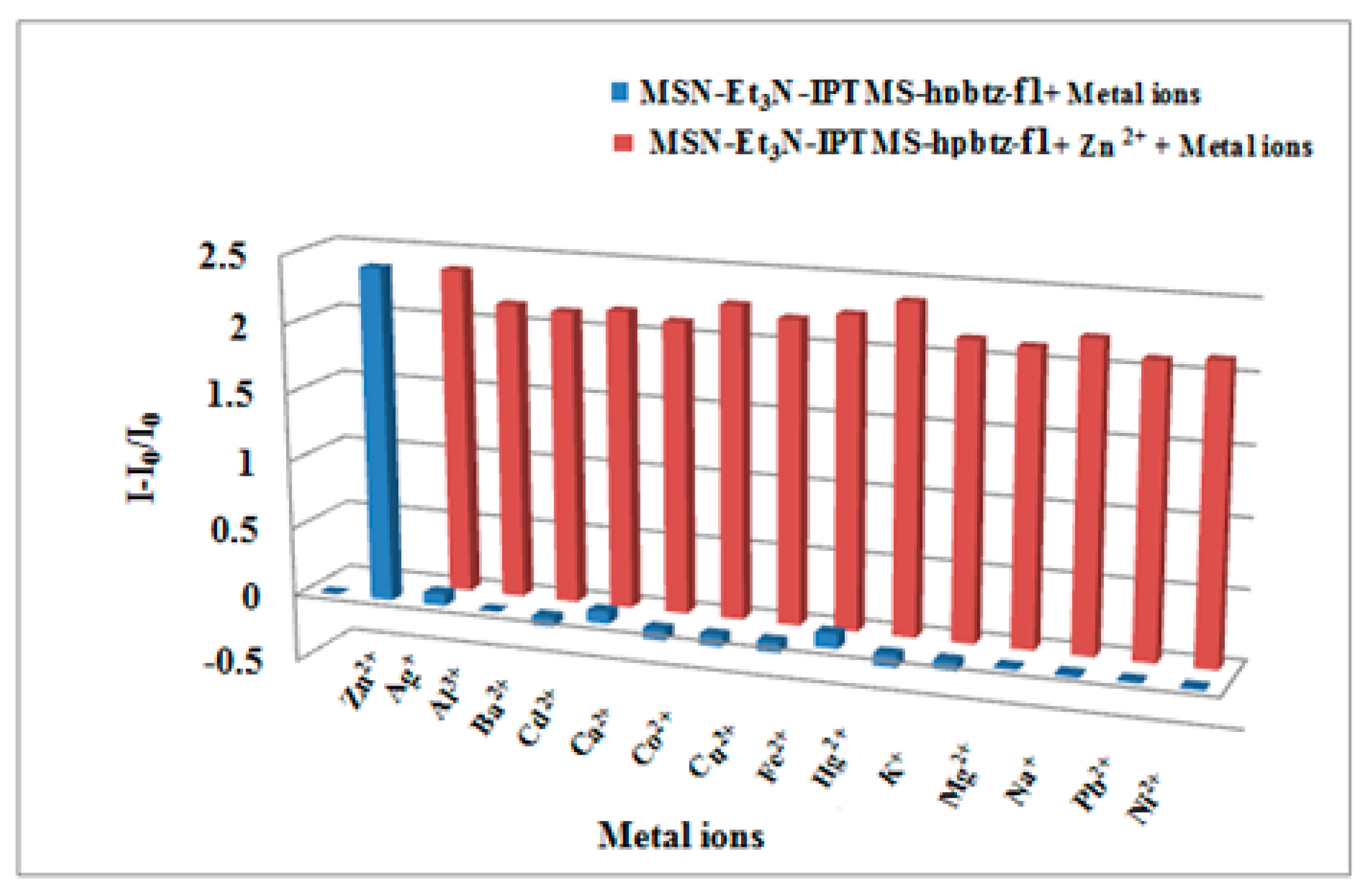
3.4. Fluorescence Titration and Binding Studies
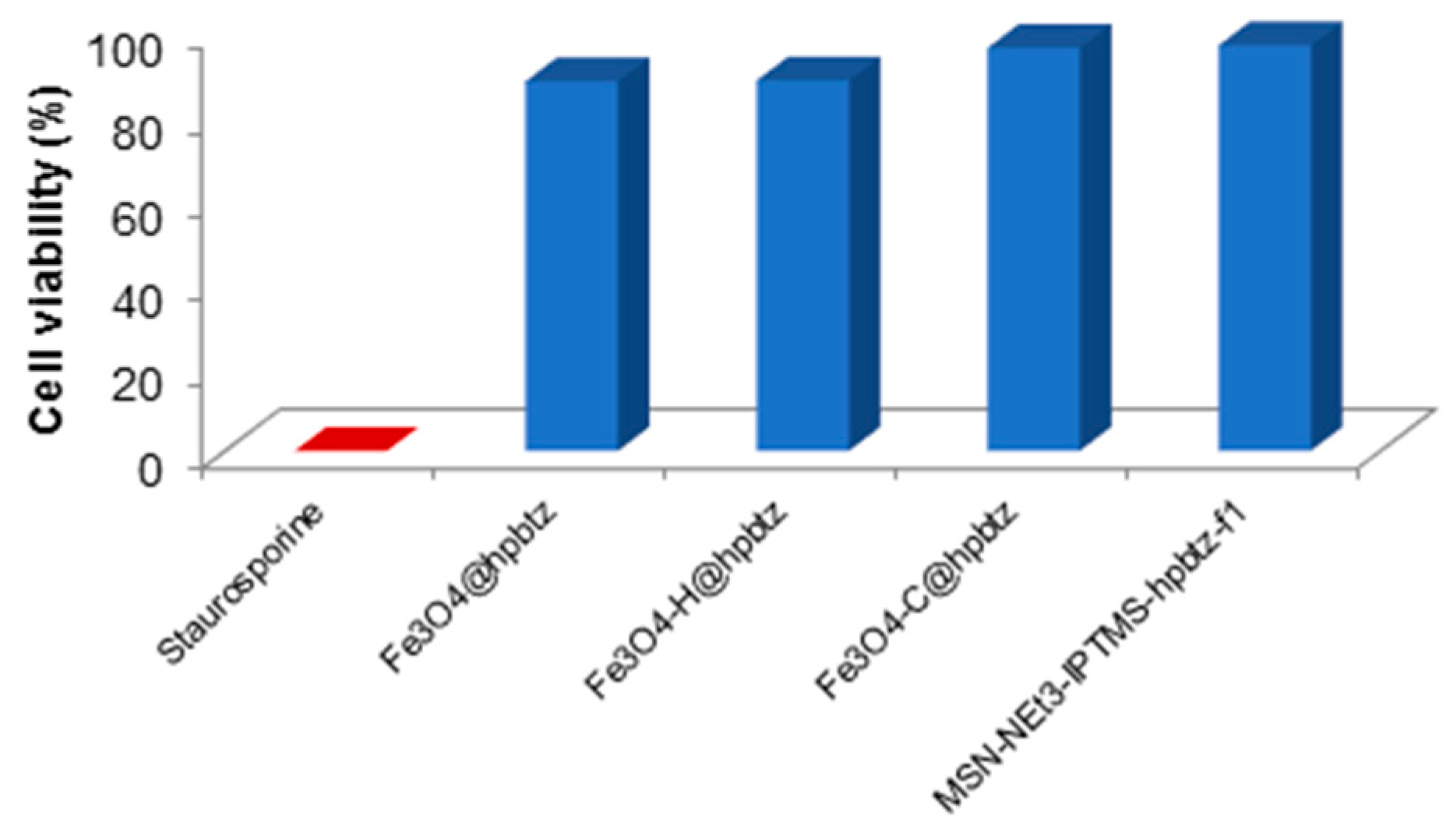
3.5. Influence of FMNPs and FMSNs on Cell Viability of HEK293 Cell Line
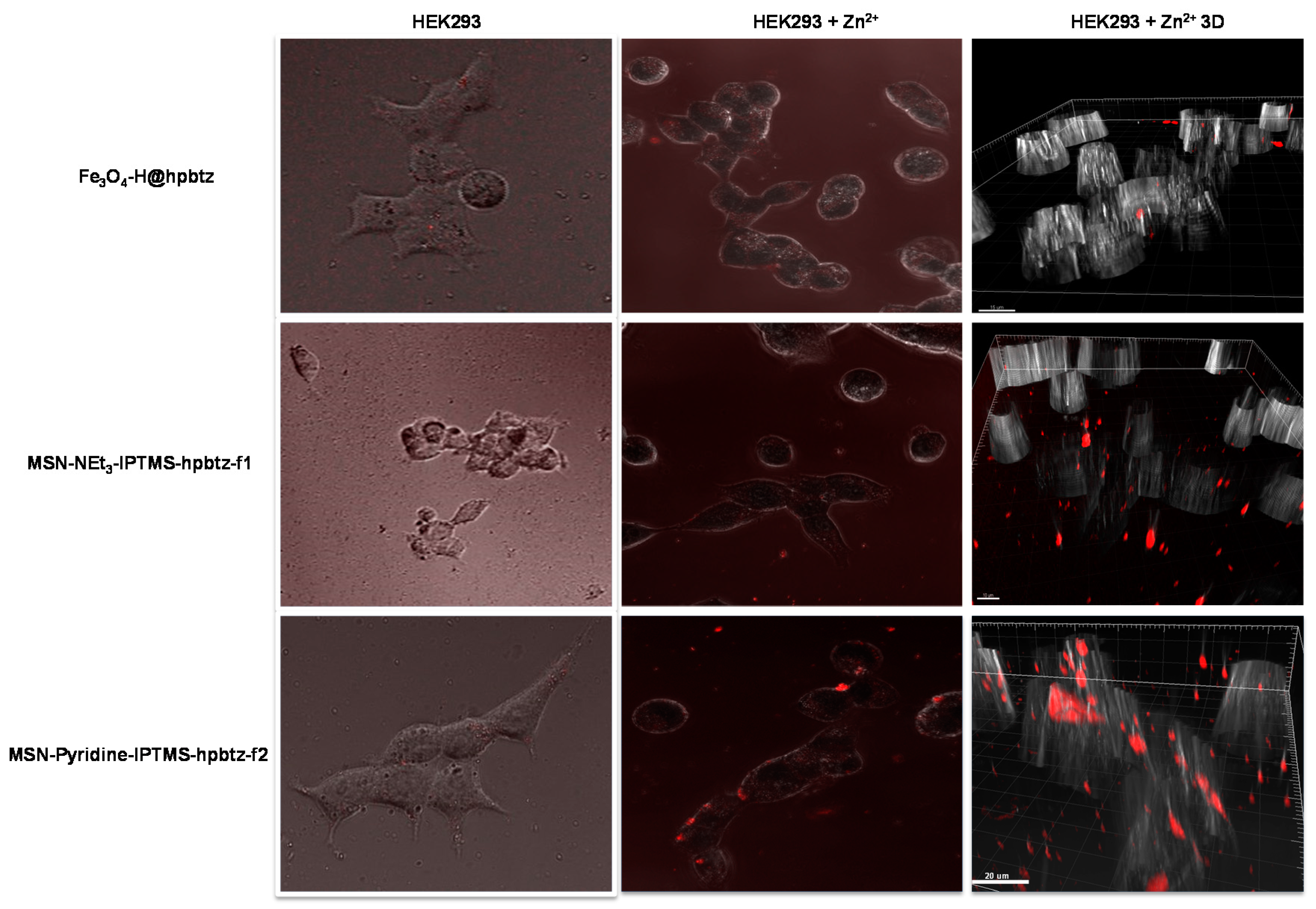
3.6. Live-Cell Imaging
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Greenough, M.A.; Camakaris, J.; Bush, A.I. Metal dyshomeostasis and oxidative stress in Alzheimer’s disease. Neurochem. Int. 2013, 62, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P. Thematic Minireview Series: Metals in Biology 2012. J. Biol. Chem. 2012, 287, 13508–13509. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, J.J.R.F.; Williams, R.J.P. The Biological Chemistry of the Elements, 2nd ed.; Oxford University Press: New York, NY, USA, 2001; ISBN 978-0-19-850848-9. [Google Scholar]
- Fukada, T.; Yamasaki, S.; Nishida, K.; Murakami, M.; Hirano, T. Zinc homeostasis and signaling in health and diseases. J. Biol. Inorg. Chem. 2011, 16, 1123–1134. [Google Scholar] [CrossRef] [PubMed]
- Fukada, T.; Kambe, T. Zinc Signals in Cellular Functions and Disorders; Springer: Tokyo, Japan, 2014. [Google Scholar]
- Maret, W. Metals on the move: Zinc ions in cellular regulation and in the coordination dynamics of zinc proteins. BioMetals 2011, 24, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Kambe, T.; Tsuji, T.; Hashimoto, A.; Itsumura, N. The physiological, biochemical, and molecular roles of zinc transporters in zinc homeostasis and metabolism. Physiol. Rev. 2015, 95, 749–784. [Google Scholar] [CrossRef] [PubMed]
- Park, J.G.; Qin, Y.; Galati, D.F.; Palmer, A.E. New Sensors for Quantitative Measurement of Mitochondrial Zn2+. ACS Chem. Biol. 2012, 7, 1636–1640. [Google Scholar] [CrossRef] [PubMed]
- Chabosseau, P.; Tuncay, E.; Meur, G.; Bellomo, E.A.; Hessels, A.M.; Hughes, S.; Johnson, P.R.V.; Bugliani, M.; Marchetti, P.; Turan, B.; et al. Mitochondrial and ER-Targeted eCALWY Probes Reveal High Levels of Free Zn2+. ACS Chem. Biol. 2014, 9, 2111–2120. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xiao, Y.; Zhang, Z.; Qian, X.; Yang, Y.; Xu, Q. A pH-resistant Zn(II) sensor derived from 4-aminonaphthalimide: Design, synthesis and intracellular applications. J. Mater. Chem. 2005, 15, 2836–2839. [Google Scholar] [CrossRef]
- Sumner, J.P.; Aylott, J.W.; Monson, E.; Kopelman, R. A fluorescent PEBBLE nanosensor for intracellular free zinc. Analyst 2002, 127, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Rosenzweig, Z. Luminescent CdS Quantum Dots as Selective Ion Probes. Anal. Chem. 2002, 74, 5132–5138. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.; Son, H.; Lim, J.M.; Kweon, H.-S.; Lee, I.S.; Kang, D.; Jung, J.H. Functionalized Fe3O4 Nanoparticles for Detecting Zinc Ions in Living Cells and Their Cytotoxicity. Chem. Eur. J. 2012, 18, 5843–5847. [Google Scholar] [CrossRef] [PubMed]
- Weissleder, R.; Kelly, K.; Sun, E.Y.; Shtatland, T.; Josephson, L. Cell-specific targeting of nanoparticles by multivalent attachment of small molecules. Nat. Biotechnol. 2005, 23, 1418–1423. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Gao, J.; Ai, H.; Chen, X. Applications and Potential Toxicity of Magnetic Iron Oxide Nanoparticles. Small 2013, 9, 1533–1545. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Wen, T.; Cristina, A.; Samia, S.; Khandhar, A.; Krishnan, K.M. Magnetic Nanoparticles: Material Engineering and Emerging Applications in Lithography and Biomedicine. J. Mater. Sci. 2016, 51, 513–553. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, P.; Dong, Z.; Dong, Z.; Ma, Z.; Jiang, J.; Li, R.; Ma, J. Highly Sensitive Fluorescence Probe Based on Functional SBA-15 for Selective Detection of Hg2+. Nanoscale Res. Lett. 2010, 5, 1468–1473. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Liu, W.; Ge, J.; Wu, J.; Zhang, W.; Meng, X.; Wang, P. A novel fluorogenic hybrid material for selective sensing of thiophenols. J. Mater. Chem. 2011, 21, 13561–13568. [Google Scholar] [CrossRef]
- Liu, T.; Li, G.; Zhang, N.; Chen, Y. An inorganic–organic hybrid optical sensor for heavy metal ion detection based on immobilizing 4-(2-pyridylazo)-resorcinol on functionalized HMS. J. Hazard. Mater. 2012, 201–202, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Stein, A.; Melde, B.J.; Schroden, R.C. Hybrid Inorganic–Organic MesoporousSilicates—Nanoscopic Reactors Coming of Age. Adv. Mater. 2000, 12, 1403–1419. [Google Scholar] [CrossRef]
- Cui, M.-C.; Li, Z.-J.; Tang, R.-K.; Liu, B.-L. Synthesis and evaluation of novel benzothiazole derivatives based on the bithiophene structure as potential radiotracers for β-amyloid plaques in Alzheimer’s disease. Bioorg. Med. Chem. 2010, 18, 2777–2784. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekharappa, A.P.; Badiger, S.E.; Dubey, P.K.; Panigrahi, S.K.; Manukonda, S.R.V.V. Design and synthesis of novel 2-substituted benzothiazole compounds as PTP1B inhibitors. Lett. Drug Des. Discov. 2014, 11, 444–453. [Google Scholar] [CrossRef]
- Gill, R.K.; Rawal, K.; Bariwal, J. Recent Advances in the Chemistry and Biology of Benzothiazoles. Arch. Pharm. 2015, 348, 155–178. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Xu, L.; Zhou, J.; Xu, Y.; Zhu, W.; Qian, X. A highly selective fluorescent probe for fast detection of hydrogen sulfide in aqueous solution and living cells. Chem. Commun. 2012, 48, 10871–10873. [Google Scholar] [CrossRef] [PubMed]
- Santra, M.; Roy, B.; Ahn, K.H. A “Reactive” Ratiometric Fluorescent Probe for Mercury Species. Org. Lett. 2011, 13, 3422–3425. [Google Scholar] [CrossRef] [PubMed]
- Maity, D.; Kumar, V.; Govindaraju, T. Reactive Probes for Ratiometric Detection of Co2+ and Cu+ Based on Excited-State Intramolecular Proton Transfer Mechanism. Org. Lett. 2012, 14, 6008–6011. [Google Scholar] [CrossRef] [PubMed]
- Roh, S.G.; Kim, Y.H.; Seo, K.D.; Lee, D.H.; Kim, H.K.; Park, Y.I. Synthesis, Photophysical, and Electroluminescent Device Properties of Zn(II)-Chelated Complexes Based on Functionalized Benzothiazole Derivatives. Adv. Funct. Mater. 2009, 19, 1663–1671. [Google Scholar] [CrossRef]
- Gogoi, A.; Samanta, S.; Das, G. A benzothiazole containing CHEF based fluorescence turn-ON sensor for Zn2+ and Cd2+ and subsequent sensing of H2PO4− and P4O74− in physiological pH. Sens. Actuators B 2014, 202, 788–794. [Google Scholar] [CrossRef]
- Wang, R.; Deng, L.; Fu, M.; Cheng, J.; Li, J. Novel ZnII complexes of 2-(2-hydroxyphenyl)benzothiazoles ligands: Electroluminescence and application as host materials for phosphorescent organic light-emitting diodes. J. Mater. Chem. 2012, 22, 23454–23460. [Google Scholar] [CrossRef]
- Paulpandi, R.Q.; Ramasamy, S.; Paulraj, M.S.; Baños, F.G.D.; Villora, G.; Cerón-Carrasco, J.P.; Pérez-Sanchez, H.; Enoch, I.V.M.V. Enhanced Zn2+ ion-sensing behavior of a benzothiazole derivative on encapsulation by bcyclodextrin. RSC Adv. 2016, 6, 15670–15677. [Google Scholar] [CrossRef]
- Jin, Y.; Wang, S.; Zhang, Y.; Song, B. Highly selective fluorescent chemosensor based on benzothiazole for detection of Zn2+. Sens. Actuators B 2016, 225, 167–173. [Google Scholar] [CrossRef]
- Bansal, D.; Gupta, R. Chemosensors containing appended benzothiazole group(s): Selective binding of Cu2+ and Zn2+ Ions by two related receptors. Dalton Trans. 2016, 45, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, Q.; Qi, F.; Li, L.; Yu, H.-D.; Liu, Z.; Huang, W. Benzothiazole-Pyrimidine-Based BODIPY Analogues: Promising Luminophores with Fluorescence Sensing and Imaging Ability and Asymmetrization-Induced Solid-State Emission. Dalton Trans. 2016, 45, 17274–17280. [Google Scholar] [CrossRef] [PubMed]
- Erdemir, S.; Tabakci, B. Selective and Sensitive Fluorescein-Benzothiazole Based Fluorescent Sensor for Zn2+ Ion in Aqueous Media. J. Fluoresc. 2017, 27, 2145–2152. [Google Scholar] [CrossRef] [PubMed]
- Mintova, S.; De Waele, V.; Hözl, M.; Schmidhammer, U.; Mihailova, B.; Riedle, E.; Bein, T. Photochemistry of 2-(2′-Hydroxyphenyl)benzothiazole Encapsulated in Nanosized Zeolites. J. Phys. Chem. A 2004, 108, 10640–10648. [Google Scholar] [CrossRef]
- Grando, S.R.; Pessoa, C.M.; Gallas, M.R.; Costa, T.M.H.; Rodembusch, F.S.; Benvenutti, E.V. Modulation of the ESIPT Emission of Benzothiazole Type Dye Incorporated in Silica-Based Hybrid Materials. Langmuir 2009, 25, 13219–13223. [Google Scholar] [CrossRef] [PubMed]
- Amirnasr, M.; Sadeghi Erami, R.; Meghdadi, S. A fluorescent carboxamide ligand, having combined ionophore/fluorophore moieties, exhibiting “On-Off” switching toward Zn2+ ion. Sens. Actuators B 2016, 233, 355–360. [Google Scholar] [CrossRef]
- Sohrabi, M.; Amirnasr, M.; Farrokhpour, H.; Meghdadi, S. A single chemosensor with combined ionophore/fluorophore moieties acting as a fluorescent “Off-On” Zn2+ sensor and a colorimetric sensor for Cu2+: Experimental, logic gate behavior and TD-DFT calculations. Sens. Actuators B 2017, 31, 255–267. [Google Scholar] [CrossRef]
- Zhao, Y.; Trewyn, B.G.; Slowing, I.I.; Lin, V.S.-Y. Mesoporous Silica Nanoparticle-Based Double Drug Delivery System for Glucose-Responsive Controlled Release of Insulin and Cyclic AMP. J. Am. Chem. Soc. 2009, 131, 8398–8400. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Qi, D.; Deng, C.; Zhang, X.; Zhao, D. Superparamagnetic High-Magnetization Microspheres with an Fe3O4@SiO2 Core and Perpendicularly Aligned Mesoporous SiO2 Shell for Removal of Microcystins. J. Am. Chem. Soc. 2008, 130, 28–29. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Li, J.; Brougham, D.F.; Fox, E.K.; Feliu, N.; Bushmelev, A.; Schmidt, A.; Mertens, N.; Kiessling, F.; Valldor, M.; et al. Water-Soluble Superparamagnetic Magnetite Nanoparticles with Biocompatible Coating for Enhanced Magnetic Resonance Imaging. ACS Nano 2011, 5, 6315–6324. [Google Scholar] [CrossRef]
- Singh, D.; McMillan, J.M.; Kabanov, A.V.; Sokolsky-Papkov, M.; Gendelman, H.E. Bench-to-bedside translation of magnetic nanoparticles. Nanomedicine 2014, 9, 501–516. [Google Scholar] [CrossRef] [PubMed]
- Wani, W.A.; Prashar, S.; Shreaz, S.; Gómez-Ruiz, S. Nanostructured materials functionalized with metal complexes: In search of alternatives for administering anticancer metallodrugs. Coord. Chem. Rev. 2016, 312, 67–98. [Google Scholar] [CrossRef]
- Pérez-Quintanilla, D.; Gómez-Ruiz, S.; Žižak, Ž.; Sierra, I.; Prashar, S.; del Hierro, I.; Fajardo, M.; Juranić, Z.D.; Kaluđerović, G.N. A New Generation of Anticancer Drugs: Mesoporous Materials Modified with Titanocene Complexes. Chem. Eur. J. 2009, 15, 5588–5597. [Google Scholar] [CrossRef] [PubMed]
- Ceballos-Torres, J.; Virag, P.; Cenariu, M.; Prashar, S.; Fajardo, M.; Fischer-Fodor, E.; Gómez-Ruiz, S. Anti-cancer Applications of Titanocene-Functionalised Nanostructured Systems: An Insight into Cell Death Mechanisms. Chem. Eur. J. 2014, 20, 10811–10828. [Google Scholar] [CrossRef] [PubMed]
- Ceballos-Torres, J.; Prashar, S.; Fajardo, M.; Chicca, A.; Gertsch, J.; Pinar, A.B.; Gómez-Ruiz, S. Ether-Substituted Group 4 Metallocene Complexes: Cytostatic Effects and Applications in Ethylene Polymerization. Organometallics 2015, 34, 2522–2532. [Google Scholar] [CrossRef]
- Gómez-Ruiz, S.; García-Peñas, A.; Prashar, S.; Rodríguez-Diéguez, A.; Fischer-Fodor, E. Anticancer Applications of Nanostructured Silica-Based Materials Functionalized with Titanocene Derivatives: Induction of Cell Death Mechanism through TNFR1 Modulation. Materials 2018, 11, 224. [Google Scholar] [CrossRef] [PubMed]
- Bulatović, M.Z.; Maksimović-Ivanić, D.; Bensing, C.; Gómez-Ruiz, S.; Steinborn, D.; Schmidt, H.; Mojić, M.; Korać, A.; Golić, I.; Pérez-Quintanilla, D.; et al. Organotin(IV)-Loaded Mesoporous Silica as a Biocompatible Strategy in Cancer Treatment. Angew. Chem. Int. Ed. 2014, 53, 5982–5987. [Google Scholar] [CrossRef] [PubMed]
- Bensing, C.; Mojić, M.; Gómez-Ruiz, S.; Carralero, S.; Dojčinović, B.; Maksimović-Ivanić, D.; Mijatović, S.; Kaluđerović, G.N. Evaluation of functionalized mesoporous silica SBA-15 as a carrier system for Ph3Sn(CH2)3OH against A2780 ovarian carcinoma cell line. Dalton Trans. 2016, 45, 18984–18993. [Google Scholar] [CrossRef] [PubMed]
- Harris, L.A.; Goff, J.D.; Carmichael, A.Y.; Riffle, J.S.; Harburn, J.J.; St Pierre, T.G.; Saunders, M. Magnetite Nanoparticle Dispersions Stabilized with Triblock Copolymers. Chem. Mater. 2003, 15, 1367–1377. [Google Scholar] [CrossRef]
- Sun, S.; Zeng, H.; Robinson, D.B.; Raoux, S.; Rice, P.M.; Wang, S.X.; Li, G. Monodisperse MFe2O4 (M = Fe, Co, Mn) Nanoparticles. J. Am. Chem. Soc. 2004, 126, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Shen, H.; von Hagen, R.; Pan, J.; Belkoura, L.; Mathur, S. Microwave assisted fast and facile synthesis of SnO2 quantum dots and their printing applications. Chem. Commun. 2010, 46, 6509–6511. [Google Scholar] [CrossRef] [PubMed]
- Naik, B.; Hazra, S.; Prasad, V.S.; Ghosh, N.N. Synthesis of Ag nanoparticles within the pores of SBA-15: An efficient catalyst for reduction of 4-nitrophenol. Catal. Commun. 2011, 12, 1104–1108. [Google Scholar] [CrossRef]
- Zhao, S.; Gao, Y.; Li, J.; Zhang, G.; Sun, R.; Wong, C.-P. In situ synthesis of silver nanostructures on magnetic Fe3O4@organosilicon microparticles for rapid hydrogenation catalysis. RSC Adv. 2015, 5, 56974–56981. [Google Scholar] [CrossRef]
- Toledo-Antonio, J.A.; Gutiérrez-Baez, R.; Sebastian, P.J.; Vàzquez, A. Thermal stability and structural deformation of rutile SnO2 nanoparticles. J. Solid State Chem. 2003, 174, 241–248. [Google Scholar] [CrossRef]
- Daniele, P.G.; Prenesti, E.; De Concetta, S.; Sammartando, S. Formation and Stability of Proton-AmineInorganic Anion Complexes in Aqueous Solution. J Solut. Chem. 1995, 24, 325–341. [Google Scholar] [CrossRef]
- Gil, V.M.S.; Oliveira, N.C. On the Use of the Method of Continuous Variations. J. Chem. Educ. 1990, 67, 473–478. [Google Scholar] [CrossRef]
- Raj, T.; Saluja, P.; Singh, N. A new class of pyrene based multifunctional chemosensors for differential sensing of metals in different media: Selective recognition of Zn2+ in organic and Fe3+ in aqueous medium. Sens. Actuators B 2015, 206, 98–106. [Google Scholar] [CrossRef]
- Xiao, D.; Xi, L.; Yang, W.; Hongbing, F.; Fang, Y.; Yao, J. Size-Tunable Emission from 1,3-Diphenyl-5-(2-anthryl)-2-pyrazoline Nanoparticles. J. Am. Chem. Soc. 2003, 125, 14816–14824. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wu, Y.; Wei, G.; Cheng, Y.; Zhu, C. A highly selective and sensitive polymer-based OFF-ON fluorescent sensor for Hg2+ detection incorporating salen and perylenyl moieties. J. Mater. Chem. 2012, 22, 478–482. [Google Scholar] [CrossRef]
- Cantürk, C.; Üçüncü, M.; Emrullahoğlu, M. A BODIPY-based fluorescent probe for the differential recognition of Hg(II) and Au(III) ions. RSC Adv. 2015, 5, 30522–30525. [Google Scholar] [CrossRef]
- Hirano, T.; Kikuchi, K.; Urano, Y.; Higuchi, T.; Nagano, T. Highly Zinc-Selective Fluorescent Sensor Molecules Suitable for Biological Applications. J. Am. Chem. Soc. 2000, 122, 12399–12400. [Google Scholar] [CrossRef]
- Stasiuk, G.J.; Minuzzi, F.; Sae-Heng, M.; Rivas, C.; Juretschke, H.-P.; Piemonti, L.; Allegrini, P.R.; Laurent, D.; Duckworth, A.R.; Beeby, A.; et al. Dual-Modal Magnetic Resonance/Fluorescent Zinc Probes for Pancreatic β-Cell Mass Imaging. Chem. Eur. J. 2015, 21, 5023–5033. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, J.; DeGraff, W.G.; Gazdar, A.F.; Minna, J.D.; Mitchell, J.B. Evaluation of a Tetrazolium-based Semiautomated Colorimetrie Assay: Assessment of Chemosensitivity Testing. Cancer Res. 1987, 47, 936–942. [Google Scholar] [PubMed]
- Yang, H.; Liu, C.; Yang, D.; Zhang, H.; Xi, Z. Comparative study of cytotoxicity, oxidative stress and genotoxicity induced by four typical nanomaterials: The role of particle size, shape and composition. J. Appl. Toxicol. 2009, 29, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Colomer, A.; Pinazo, A.; García, M.T.; Mitjans, M.; Vinardell, M.P.; Infante, M.R.; Martínez, V.; Perez, L. pH-Sensitive Surfactants from Lysine: Assessment of Their Cytotoxicity and Environmental Behavior. Langmuir 2012, 28, 5900–5912. [Google Scholar] [CrossRef] [PubMed]
- Bossy-Wetzel, E.; Talantova, M.V.; Lee, W.D.; Scholzke, M.N.; Harrop, A.; Mathews, E.; Götz, T.; Han, J.; Ellisman, M.H.; Perkins, G.A.; et al. Crosstalk between Nitric Oxide and Zinc Pathways to Neuronal Cell Death Involving Mitochondrial Dysfunction and p38-Activated K+ Channels. Neuron 2004, 41, 351–365. [Google Scholar] [CrossRef]
- Lobner, D.; Canzoniero, L.M.; Manzerra, P.; Gottron, F.; Ying, H.; Knudson, M.; Tian, M.; Dugan, L.L.; Kerchner, G.A.; Sheline, C.T.; et al. Zinc-induced neuronal death in cortical neurons. Cell. Mol. Biol. 2000, 46, 797–806. [Google Scholar] [PubMed]
- Choi, D.W.; Koh, J.Y. Zinc and brain injury. Annu. Rev. Neurosci. 1998, 21, 347–375. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhou, J. Zinc and Myocardial Ischemia/Reperfusion Injury. BioMetals 2013, 26, 863–878. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-M.; Venkatesan, P.; Wu, S.-P. A sensitive and selective fluorescent sensor for Zinc(II) and its application to living cell imaging. Sens. Actuators B 2014, 203, 719–725. [Google Scholar] [CrossRef]
- Zastrow, M.L.; Radford, R.J.; Chyan, W.; Anderson, C.T.; Zhang, D.Y.; Loas, A.; Tzounopoulos, T.; Lippard, S.J. Reaction-Based Probes for Imaging Mobile Zinc in Live Cells and Tissues. ACS Sens. 2016, 1, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Wu, Y.-Z.; Hu, X.; Xu, X.; Sun, J.; Geng, R.; Huang, T.; Liu, X.; Ma, Y.; Chen, Y.; et al. Multicolor bioimaging with biosynthetic zinc nanoparticles and their application in tumor detection. Sci. Rep. 2017, 4, 45313. [Google Scholar] [CrossRef] [PubMed]

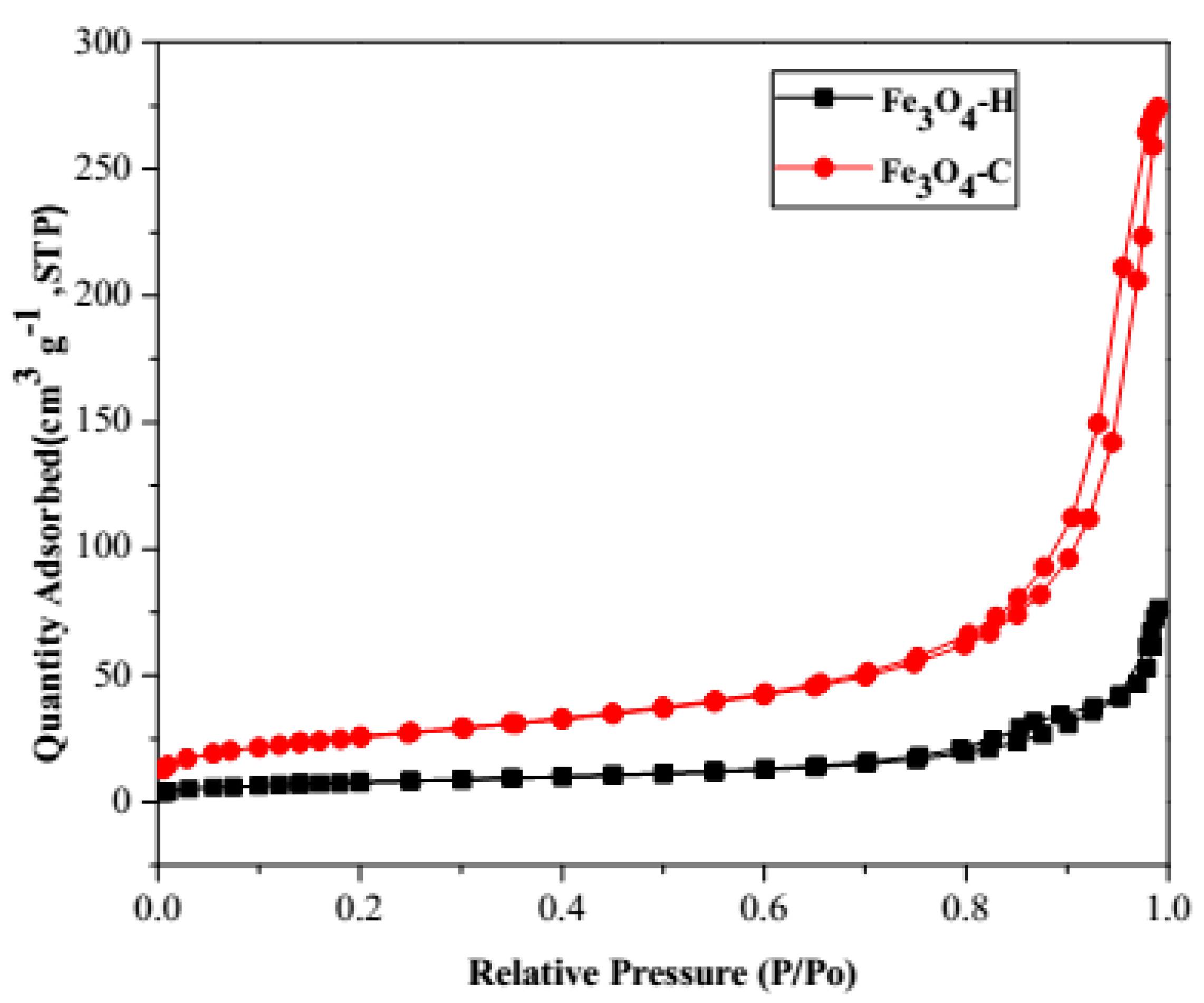
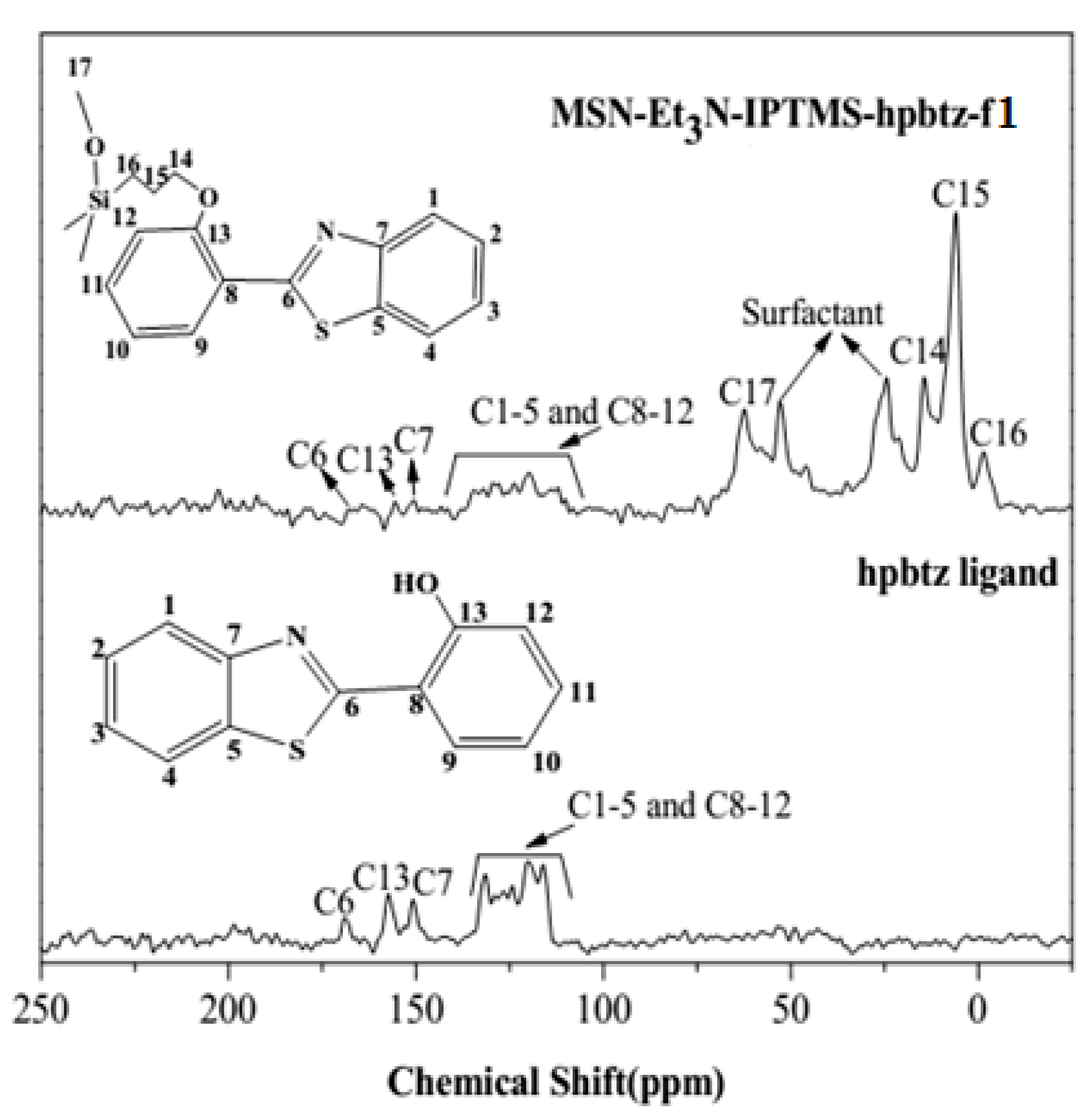
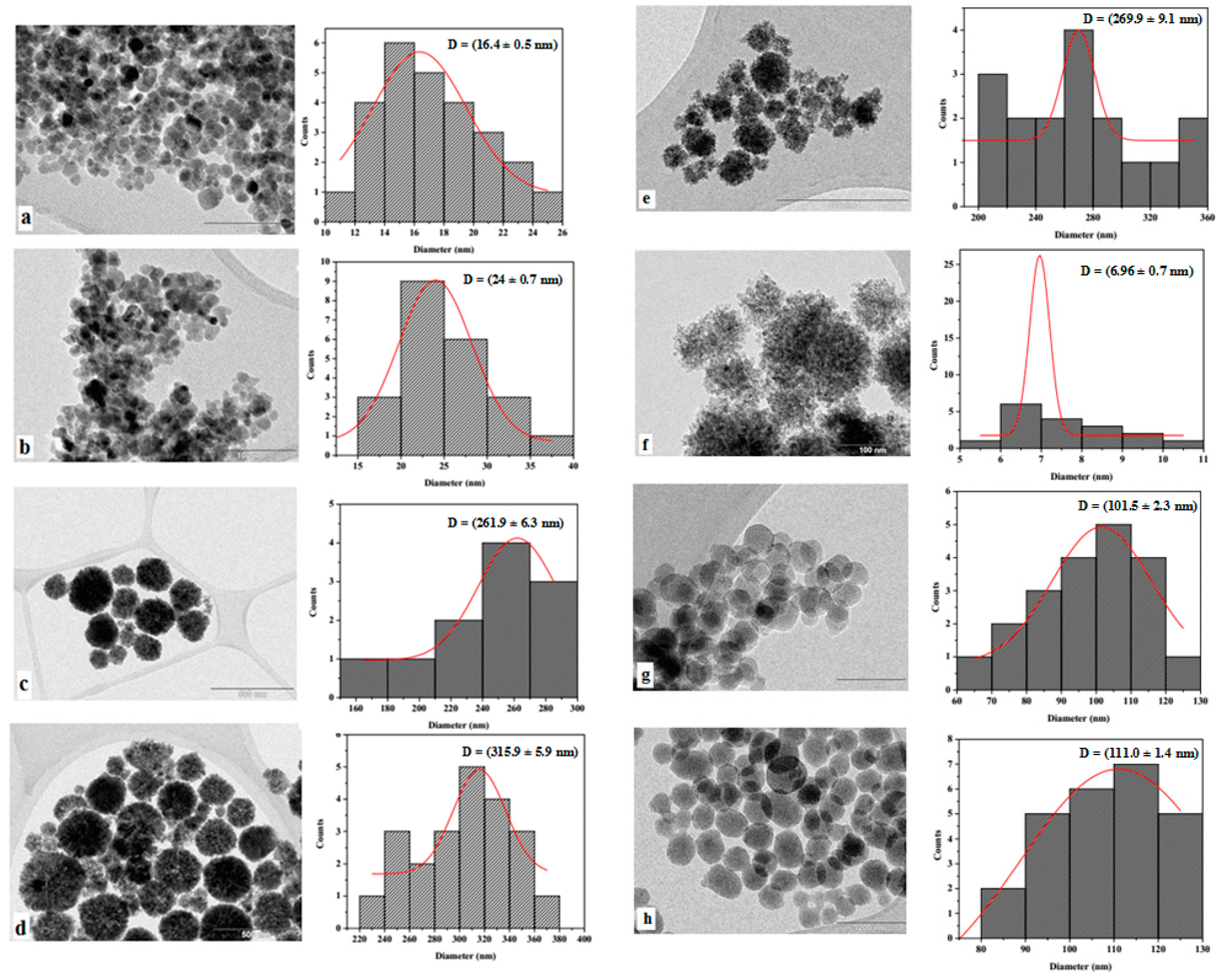



| Material | SBET (m2 g−1) | Pore Volume (cm3 g−1) | Pore Size (Å) |
|---|---|---|---|
| Fe3O4-H | 27.96 | 0.072 | 161.69 |
| Fe3O4-C | 92.53 | 0.319 | 175.31 |
| MSNs | 1028.37 | 0.970 | 41.49 |
| MSN-Et3N-IPTMS-hpbtz-f1 | 665.91 | 0.682 | 57.20 |
| Material | Fe, Si Content (%) | S (%) | hpbtz (m mol g−1) |
|---|---|---|---|
| Fe3O4-H | Fe (61.4) | - | - |
| Fe3O4-C | Fe (74.2) | - | - |
| Fe3O4-H@hpbtz | Fe (75.2) | 0.307 | 0.096 |
| Fe3O4-C@hpbtz | Fe (60.3) | 0.543 | 0.170 |
| Fe3O4@hpbtz | Fe (64.5) | 0.107 | 0.033 |
| MSNs | Si (22.7) | - | - |
| MSN-Et3N-IPTMS-hpbtz-f1 | Si (28) | 0.178 | 0.056 |
| MSN-pyridine-IPTMS-hpbtz-f2 | Si (19.0) | 0.051 | 0.016 |
| MSN-NaOH-IPTMS-hpbtz-f3 | Si (17.0) | 0.057 | 0.018 |
| MSN-Et3N-NCO-hpbtz-e1 | Si (9.70) | 0.010 | 0.003 |
| MSN-pyridine-NCO-hpbtz-e2 | Si (11,6) | 0.025 | 0.008 |
| MSN-NaOH-NCO-hpbtz-e3 | Si (8.97) | 0.021 | 0.006 |
| Material | % Weight Loss (20–190 °C) | % Weight Loss (190–450 °C) |
|---|---|---|
| Fe3O4-H(a) | 1.43 | 1.31 |
| Fe3O4-H@hpbtz(b) | 1.11 | 2.54 |
| Fe3O4-C(c) | 1.02 | 1.21 |
| Fe3O4-C@hpbtz(d) | 6.07 | 5.41 |
| Fe3O4@hpbtz(e) | 2.61 | 8.73 |
| MSN(f) | 6.60 | 1.44 1 |
| MSN-NEt3-IPTMS-hpbtz-f1(g) | 3.90 | 18.54 1 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Erami, R.S.; Ovejero, K.; Meghdadi, S.; Filice, M.; Amirnasr, M.; Rodríguez-Diéguez, A.; De La Orden, M.U.; Gómez-Ruiz, S. Applications of Nanomaterials Based on Magnetite and Mesoporous Silica on the Selective Detection of Zinc Ion in Live Cell Imaging. Nanomaterials 2018, 8, 434. https://doi.org/10.3390/nano8060434
Erami RS, Ovejero K, Meghdadi S, Filice M, Amirnasr M, Rodríguez-Diéguez A, De La Orden MU, Gómez-Ruiz S. Applications of Nanomaterials Based on Magnetite and Mesoporous Silica on the Selective Detection of Zinc Ion in Live Cell Imaging. Nanomaterials. 2018; 8(6):434. https://doi.org/10.3390/nano8060434
Chicago/Turabian StyleErami, Roghayeh Sadeghi, Karina Ovejero, Soraia Meghdadi, Marco Filice, Mehdi Amirnasr, Antonio Rodríguez-Diéguez, María Ulagares De La Orden, and Santiago Gómez-Ruiz. 2018. "Applications of Nanomaterials Based on Magnetite and Mesoporous Silica on the Selective Detection of Zinc Ion in Live Cell Imaging" Nanomaterials 8, no. 6: 434. https://doi.org/10.3390/nano8060434
APA StyleErami, R. S., Ovejero, K., Meghdadi, S., Filice, M., Amirnasr, M., Rodríguez-Diéguez, A., De La Orden, M. U., & Gómez-Ruiz, S. (2018). Applications of Nanomaterials Based on Magnetite and Mesoporous Silica on the Selective Detection of Zinc Ion in Live Cell Imaging. Nanomaterials, 8(6), 434. https://doi.org/10.3390/nano8060434










