Optimization of Malachite Green Removal from Water by TiO2 Nanoparticles under UV Irradiation
Abstract
1. Introduction
2. Results and Discussion
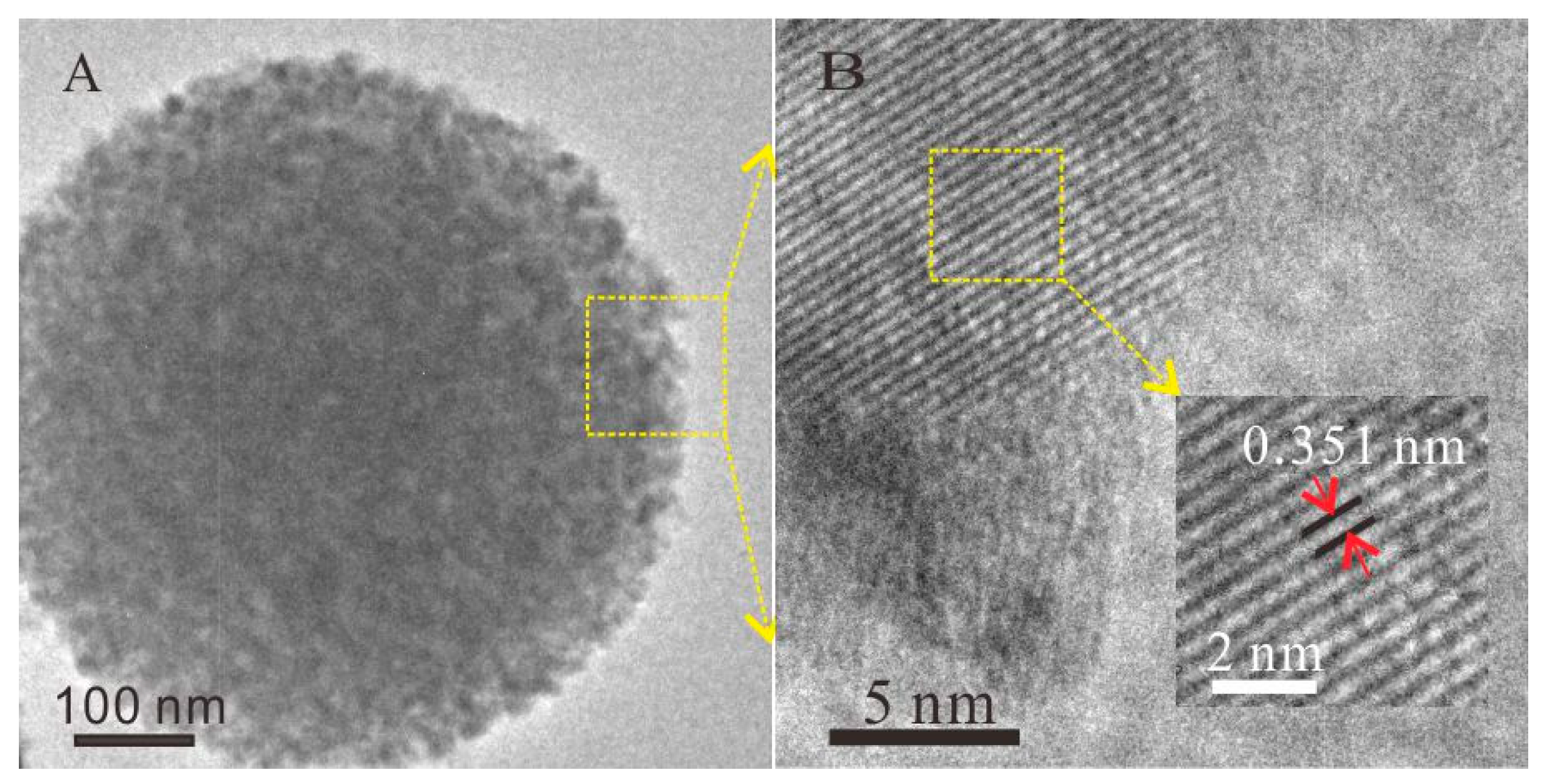
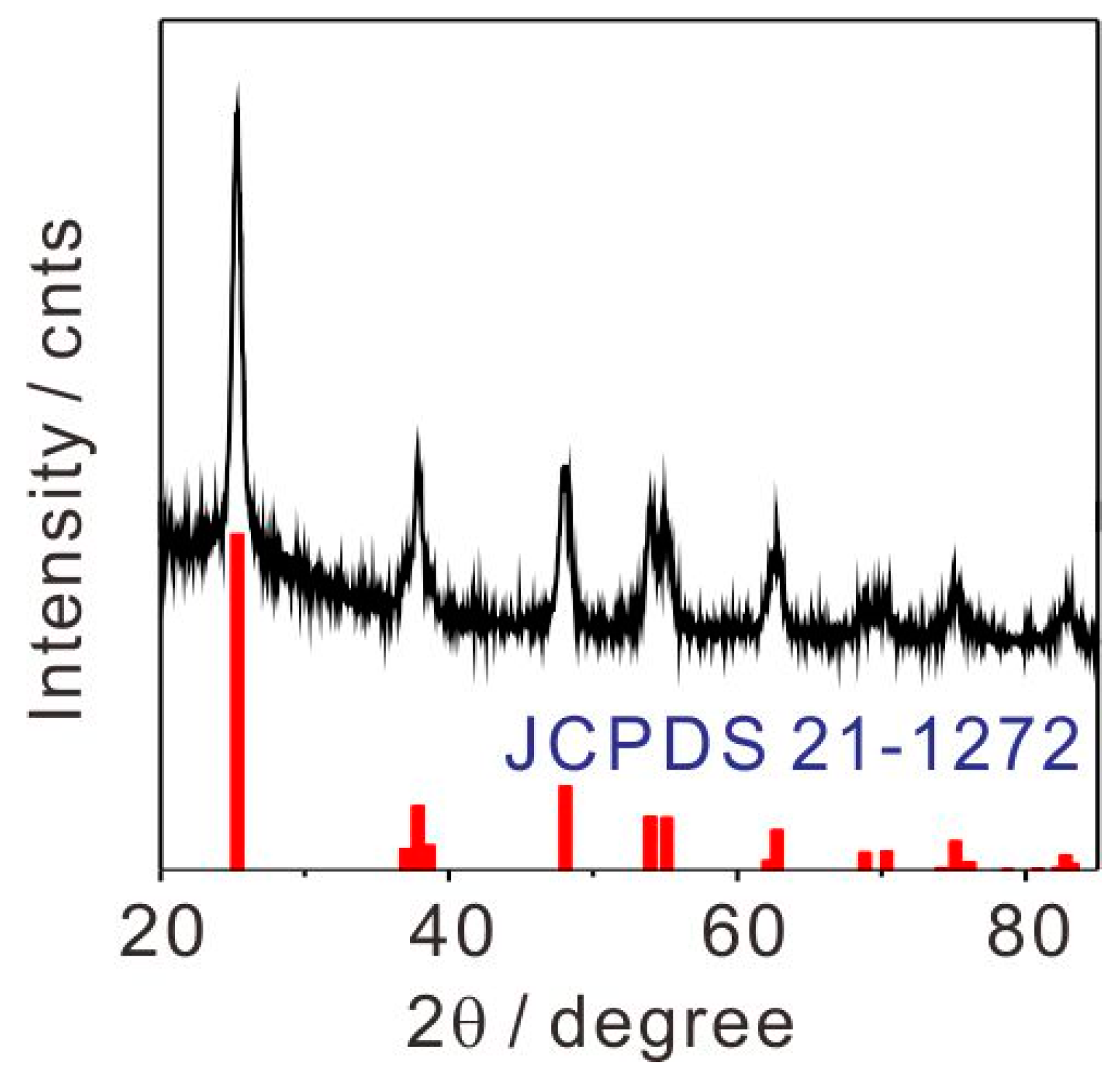
2.1. Characterization of TiO2
2.2. Kinetics of Photocatalytic Degradation
2.2.1. TiO2 Dosage Effect on MG Removal
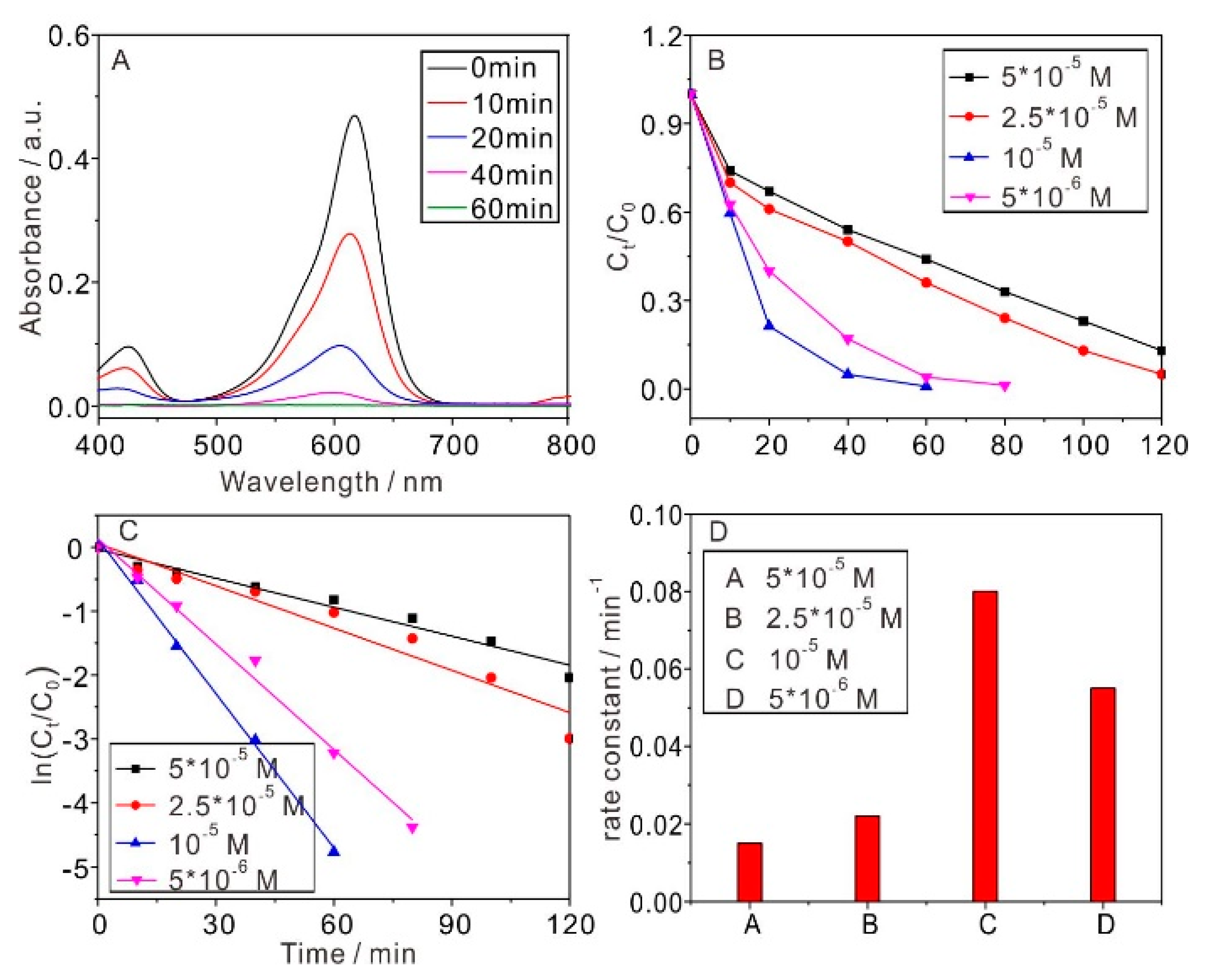
2.2.2. Initial Concentrations of MG Effect on the Efficiency of Photocatalytic Degradation
2.2.3. Effect of pH Values
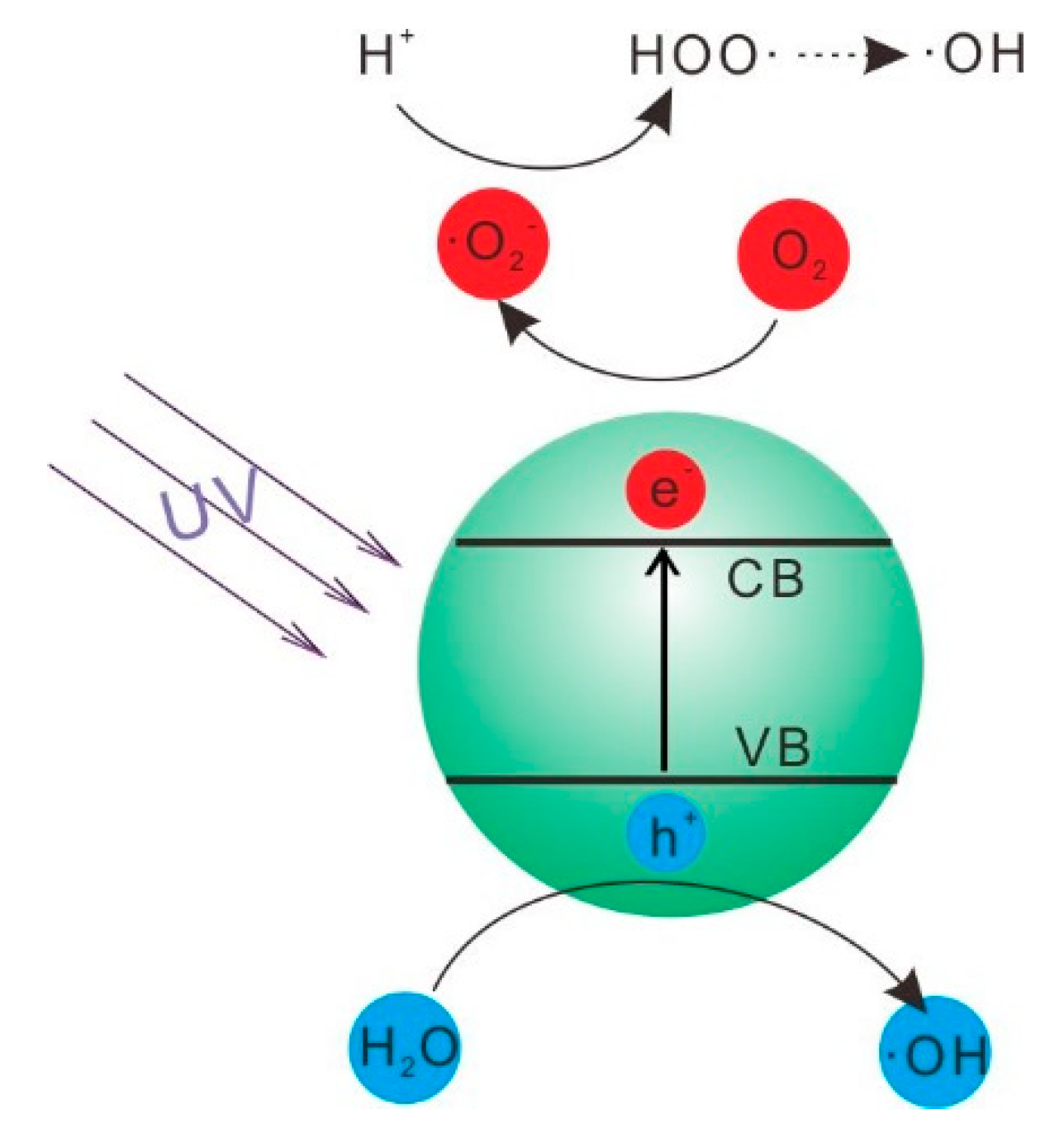
2.3. Mechanism of Photocatalytic Degradation
3. Materials and Methods
3.1. Chemicals and Materials
3.2. Preparation of TiO2
3.3. Photocatalytic Experiments
3.4. Apparatus
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Berger, E.; Haase, P.; Kuemmerlen, M.; Leps, M.; Schafer, R.B.; Sundermann, A. Water quality variables and pollution sources shaping stream macroinvertebrate communities. Sci. Total Environ. 2017, 587, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Moskovchenko, D.V.; Babushkin, A.G.; Ubaidulaev, A.A. Salt pollution of surface water in oil fields of Khanty-Mansi Autonomous Area-Yugra. Water Resour. 2017, 44, 128–138. [Google Scholar] [CrossRef]
- Li, L.; Lin, Z.Z.; Peng, A.H.; Zhong, H.P.; Chen, X.M.; Huang, Z.Y. Biomimetic ELISA detection of malachite green based on magnetic molecularly imprinted polymers. J. Chromatogr. B 2016, 1035, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Stead, S.L.; Ashwin, H.; Johnston, B.; Tarbin, J.A.; Sharman, M.; Kay, J.; Keely, B.J. An RNA-aptamer-based assay for the detection and analysis of malachite green and leucomalachite green residues in fish tissue. Anal. Chem. 2010, 82, 2652–2660. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, E.J.; Tong, W.G. Sensitive detection of malachite green and crystal violet by nonlinear laser wave mixing and capillary electrophoresis. J. Chromatogr. B 2016, 1020, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Plakas, S.M.; ElSaid, K.R.; Stehly, G.R.; Gingerich, W.H.; Allen, J.L. Uptake, tissue distribution, and metabolism of malachite green in the channel catfish (Ictalurus punctatus). Can. J. Fish. Aquat. Sci. 1996, 53, 1427–1433. [Google Scholar] [CrossRef]
- Safarik, I.; Safarikova, M. Detection of low concentrations of malachite green and crystal violet in water. Water Res. 2002, 36, 196–200. [Google Scholar] [CrossRef]
- Wu, L.; Lin, Z.Z.; Zhong, H.P.; Peng, A.H.; Chen, X.M.; Huang, Z.Y. Rapid detection of malachite green in fish based on CdTe quantum dots coated with molecularly imprinted silica. Food Chem. 2017, 229, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Yang, R.; Wang, C.W.; Xiao, R.; Long, F. Reusable nanosilver-coated magnetic particles for ultrasensitive SERS-based detection of malachite green in water samples. Sci. Rep. 2016, 6, 22870. [Google Scholar] [CrossRef] [PubMed]
- Sivashanmugan, K.; Liao, J.D.; Liu, B.H.; Yao, C.K.; Luo, S.C. Ag nanoclusters on ZnO nanodome array as hybrid SERS-active substrate for trace detection of malachite green. Sens. Actuators B Chem. 2015, 207, 430–436. [Google Scholar] [CrossRef]
- Hashemzadeh, F.; Rahimi, R.; Ghaffarinejad, A. Mesoporous nanostructures of Nb2O5 obtained by an EISA route for the treatment of malachite green dye-contaminated aqueous solution under UV and visible light irradiation. Ceram. Int. 2014, 40, 9817–9829. [Google Scholar] [CrossRef]
- Yin, Y.Y.; Li, C.; Song, C.W.; Tao, P.; Sun, M.H.; Pan, Z.L.; Wang, T.H.; Shao, M.H. The design of coal-based carbon membrane coupled with the electric field and its application on the treatment of malachite green (MG) aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2016, 506, 629–636. [Google Scholar] [CrossRef]
- Li, X.L.; Zhang, Y.; Jing, L.Y.; He, X.H. Novel N-doped CNTs stabilized Cu2O nanoparticles as adsorbent for enhancing removal of Malachite Green and tetrabromobisphenol A. Chem. Eng. J. 2016, 292, 326–339. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, C.C.; Zhao, D.; Ma, W.H.; Zhao, J.C. Change of adsorption modes of dyes on fluorinated TiO2 and its effect on photocatalytic degradation of dyes under visible irradiation. Langmuir 2008, 24, 7338–7345. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, S.; Shishido, T.; Teramura, K.; Tanaka, T. Photocatalytic oxidation of alcohols over TiO2 covered with Nb2O5. ACS Catal. 2012, 2, 175–179. [Google Scholar] [CrossRef]
- Grabowska, E.; Diak, M.; Marchelek, M.; Zaleska, A. Decahedral TiO2 with exposed facets: Synthesis, properties, photoactivity and applications. Appl. Catal. B Environ. 2014, 156, 213–235. [Google Scholar] [CrossRef]
- Mansouri, F.; Kalankesh, R.L.; Hasankhani, H. The comparison of photo catalytic degradation of dissolved organic carbon (DOC) from water by UV/TiO2 in the presence and absence of iron ion. Glob. NEST J. 2016, 18, 392–401. [Google Scholar]
- Asuha, S.; Zhou, X.G.; Zhao, S. Adsorption of methyl orange and Cr(Vi) on mesoporous TiO2 prepared by hydrothermal method. J. Hazard. Mater. 2010, 181, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.H.; Cai, W.Y.; Tang, X.H.; Yang, L.B. Sensitively monitoring photodegradation process of organic dye molecules by surface-enhanced Raman spectroscopy based on Fe3O4@SiO2@TiO2@Ag particle. Analyst 2014, 139, 5509–5515. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.; Kumaresan, Y.; Yoon, S.Y.; Um, S.H.; Kwon, I.K.; Jung, G.Y. Design of gold nanoparticles-decorated SiO2@TiO2 core/shell nanostructures for visible light-activated photocatalysis. RSC Adv. 2017, 7, 7469–7475. [Google Scholar] [CrossRef]
- Cai, X.G.; He, J.Y.; Chen, L.; Chen, K.; Li, Y.L.; Zhang, K.S.; Jin, Z.; Liu, J.Y.; Wang, C.M.; Wang, X.G.; et al. A 2D-g-C3N4 nanosheet as an eco-friendly adsorbent for various environmental pollutants in water. Chemosphere 2017, 171, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.M.; Liu, H.L.; Han, Z.Z.; Yang, L.B.; Liu, J.H. Non-ultraviolet photocatalytic kinetics of NaYF4:Yb,Tm@TiO2/Ag core@comby shell nanostructures. J. Mater. Chem. A 2015, 3, 14642–14650. [Google Scholar] [CrossRef]
- Yang, J.K.; Lee, S.M. Removal of Cr(Vi) and humic acid by using TiO2 photocatalysis. Chemosphere 2006, 63, 1677–1684. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.H.; Leal, M.; Li, Q.L. Degradation of natural organic matter by TiO2 photocatalytic oxidation and its effect on fouling of low-pressure membranes. Water Res. 2008, 42, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Zhu, B.S.; Jia, Y.; Jin, Z.; Sun, B.; Luo, T.; Yu, X.Y.; Kong, L.T.; Huang, X.J.; Liu, J.H. Controlled synthesis of natroalunite microtubes and spheres with excellent fluoride removal performance. Chem. Eng. J. 2015, 271, 240–251. [Google Scholar] [CrossRef]
- Wu, S.B.; Zhang, K.S.; Wang, X.L.; Jia, Y.; Sun, B.; Luo, T.; Meng, F.L.; Jin, Z.; Lin, D.Y.; Shen, W.; et al. Enhanced adsorption of cadmium ions by 3D sulfonated reduced graphene oxide. Chem. Eng. J. 2015, 262, 1292–1302. [Google Scholar] [CrossRef]
- Wang, X.D.; Caruso, R.A. Enhancing photocatalytic activity of titania materials by using porous structures and the addition of gold nanoparticles. J. Mater. Chem. 2011, 21, 20–28. [Google Scholar] [CrossRef]
- Mills, A.; LeHunte, S. An overview of semiconductor photocatalysis. J. Photochem. Photobiol. A 1997, 108, 1–35. [Google Scholar] [CrossRef]
- Yang, J.; Dai, J.; Chen, C.C.; Zhao, J.C. Effects of hydroxyl radicals and oxygen species on the 4-chlorophenol degradation by photoelectrocatalytic reactions with TiO2-film electrodes. J. Photochem. Photobiol. A 2009, 208, 66–77. [Google Scholar] [CrossRef]
- Li, X.H.; Chen, G.Y.; Yang, L.B.; Jin, Z.; Liu, J.H. Multifunctional Au-coated TiO2 nanotube arrays as recyclable SERS substrates for multifold organic pollutants detection. Adv. Funct. Mater. 2010, 20, 2815–2824. [Google Scholar] [CrossRef]
- Muggli, D.S.; McCue, J.T.; Falconer, J.L. Mechanism of the photocatalytic oxidation of ethanol on TiO2. J. Catal. 1998, 173, 470–483. [Google Scholar] [CrossRef]
- Tang, Y.N.; Di, W.H.; Zhai, X.S.; Yang, R.Y.; Qin, W.P. NIR-responsive photocatalytic activity and mechanism of NaYF4:Yb,Tm@TiO2 core-shell nanoparticles. ACS Catal. 2013, 3, 405–412. [Google Scholar] [CrossRef]
- Zhang, Y.X.; Yu, X.Y.; Jia, Y.; Jin, Z.; Liu, J.H.; Huang, X.J. A facile approach for the synthesis of Ag-coated Fe3O4@TiO2 core/shell microspheres as highly efficient and recyclable photocatalysts. Eur. J. Inorg. Chem. 2011, 2011, 5096–5104. [Google Scholar] [CrossRef]
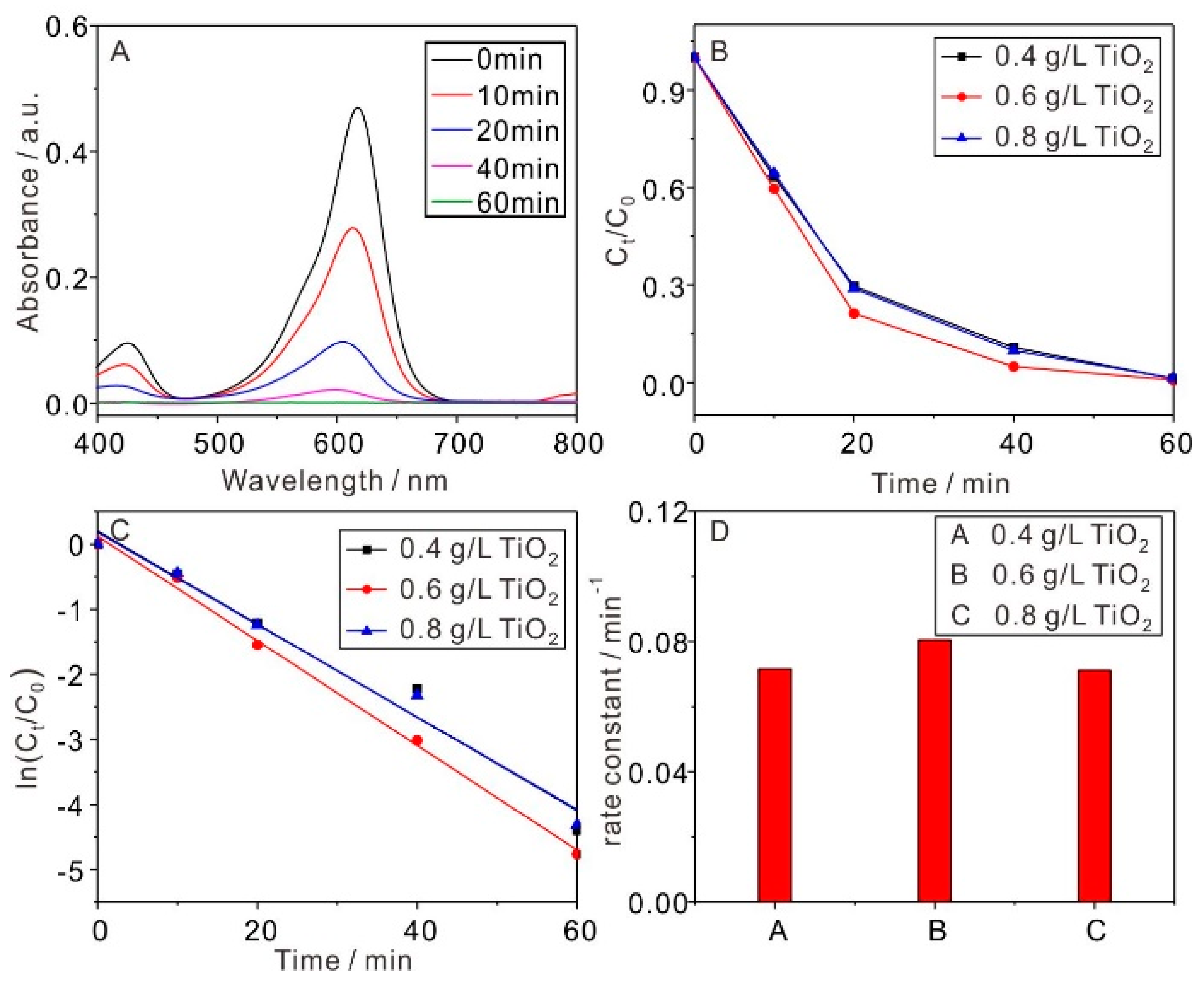

| TiO2 Dosage (g/L) | k (min−1) | R2 |
|---|---|---|
| 0.4 | 0.0716 | 0.964 |
| 0.6 | 0.0805 | 0.995 |
| 0.8 | 0.0711 | 0.976 |
| Concentration of MG (M) | k (min−1) | R2 |
|---|---|---|
| 5 × 10−5 | 0.0151 | 0.965 |
| 2.5 × 10−5 | 0.022 | 0.935 |
| 10−5 | 0.0805 | 0.995 |
| 5 × 10−6 | 0.055 | 0.989 |
| pH Values | k (min−1) | R2 |
|---|---|---|
| 4 | 0.01 | 0.961 |
| 6 | 0.039 | 0.983 |
| 8 | 0.048 | 0.995 |
| 10 | 0.124 | 0.737 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, Y.; Ni, M.; Li, S. Optimization of Malachite Green Removal from Water by TiO2 Nanoparticles under UV Irradiation. Nanomaterials 2018, 8, 428. https://doi.org/10.3390/nano8060428
Ma Y, Ni M, Li S. Optimization of Malachite Green Removal from Water by TiO2 Nanoparticles under UV Irradiation. Nanomaterials. 2018; 8(6):428. https://doi.org/10.3390/nano8060428
Chicago/Turabian StyleMa, Yongmei, Maofei Ni, and Siyue Li. 2018. "Optimization of Malachite Green Removal from Water by TiO2 Nanoparticles under UV Irradiation" Nanomaterials 8, no. 6: 428. https://doi.org/10.3390/nano8060428
APA StyleMa, Y., Ni, M., & Li, S. (2018). Optimization of Malachite Green Removal from Water by TiO2 Nanoparticles under UV Irradiation. Nanomaterials, 8(6), 428. https://doi.org/10.3390/nano8060428






