In-Situ Growth of NiAl-Layered Double Hydroxide on AZ31 Mg Alloy towards Enhanced Corrosion Protection
Abstract
1. Introduction
2. Methods
2.1. Materials and Reagents
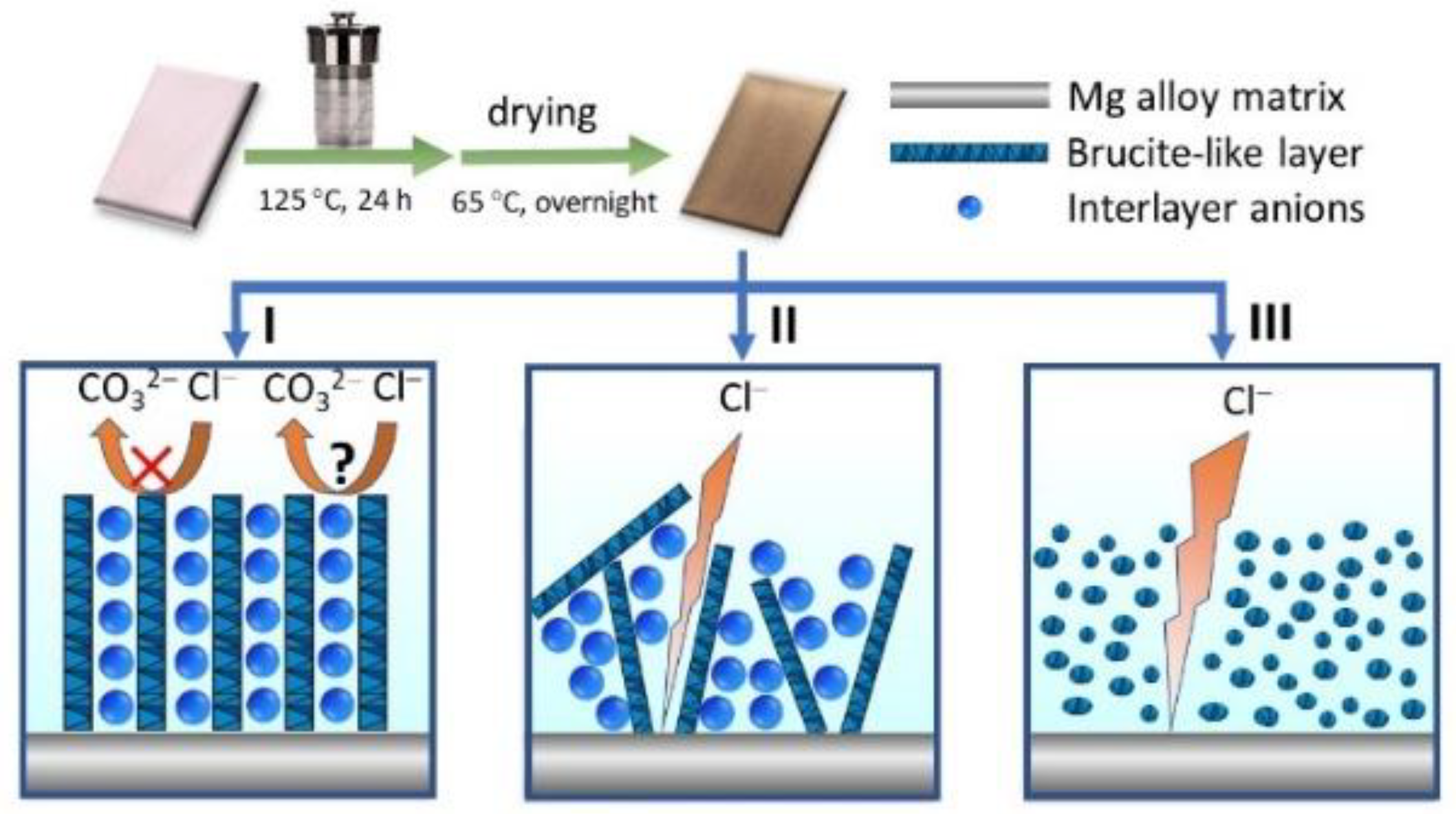
2.2. Preparation of NiAl-LDH Coatings on Mg Alloy
2.3. Characterization and Electrochemical Tests
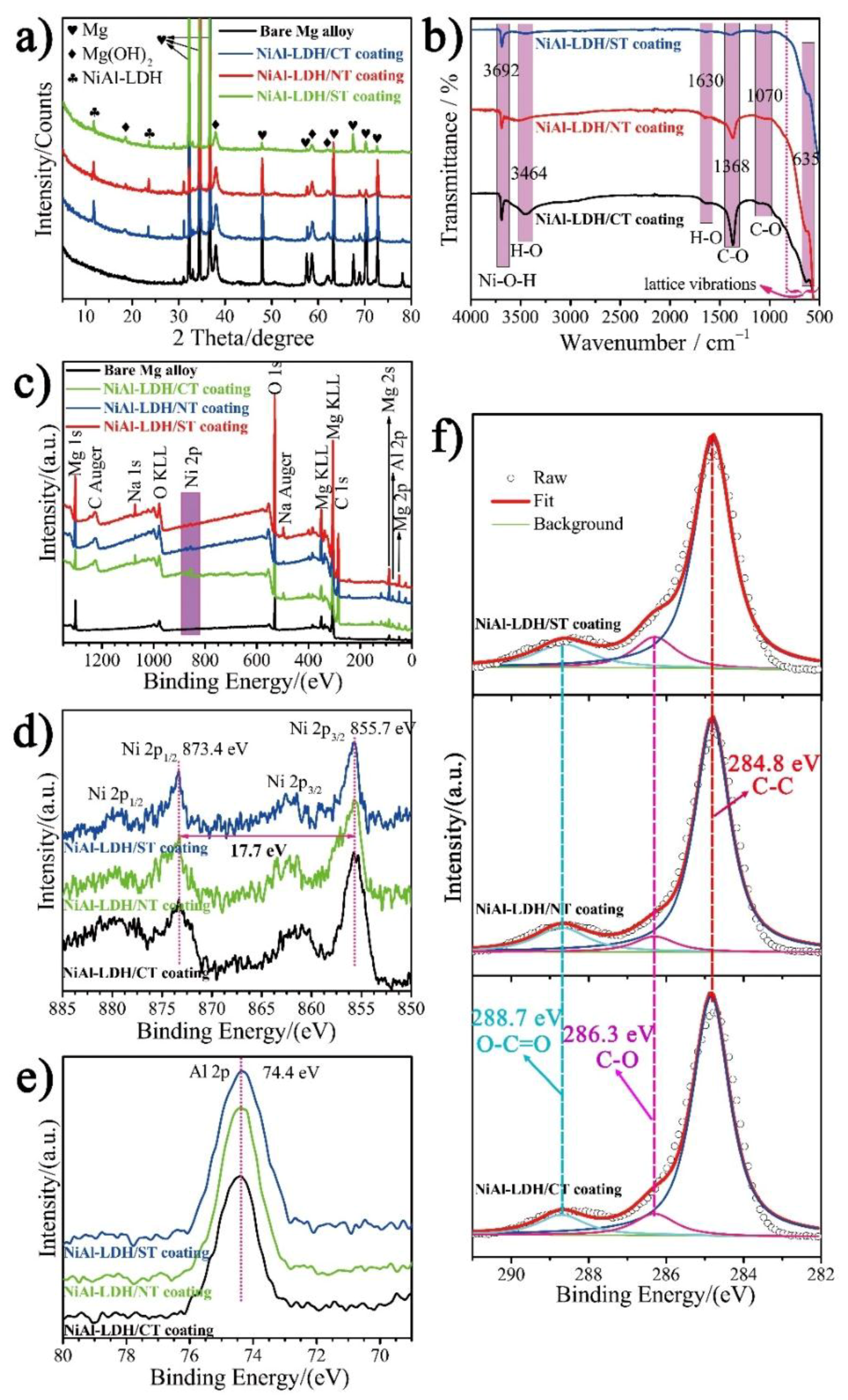
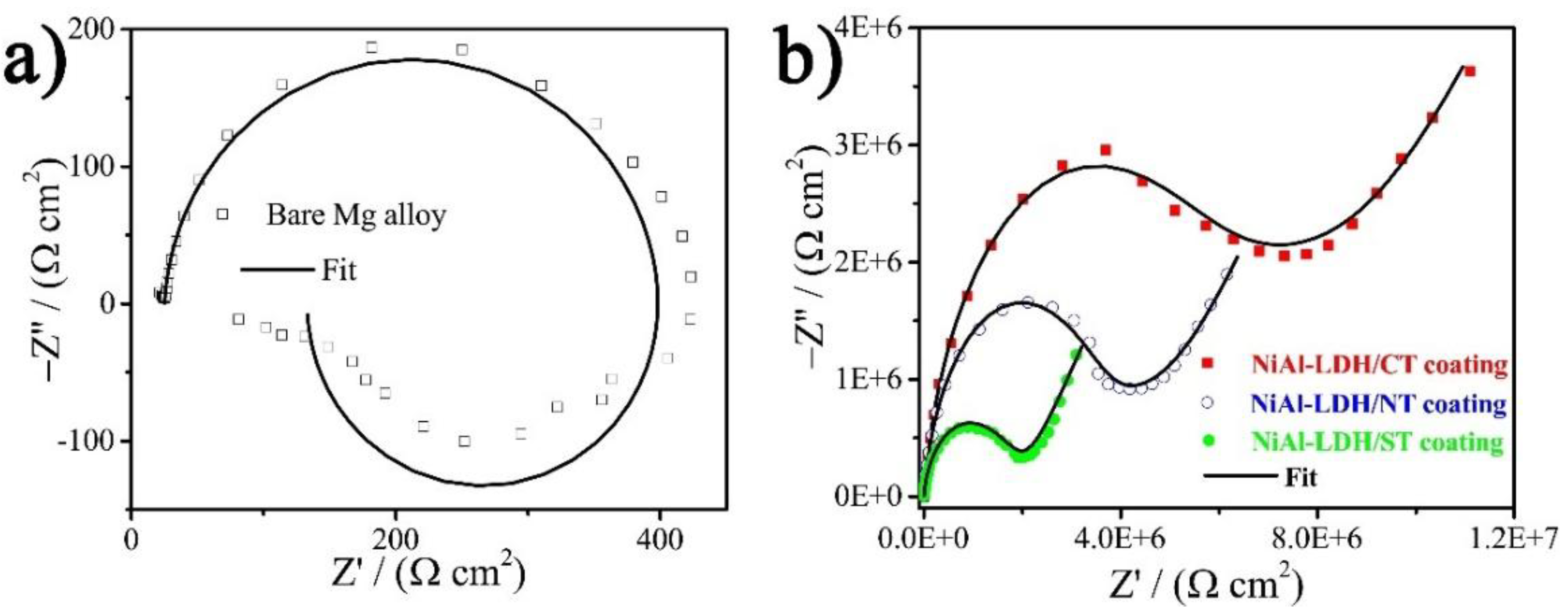
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Xie, Z.-H.; Li, D.; Skeete, Z.; Sharma, A.; Zhong, C.-J. Nanocontainer-Enhanced Self-Healing for Corrosion-Resistant Ni Coating on Mg Alloy. ACS Appl. Mater. Interfaces 2017, 9, 36247–36260. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xie, Z.-H.; Chen, H.; Hu, C.; Li, L.; Hu, B.; Song, Z.; Yan, D.; Yu, G. Electroless Deposition and Characterization of a Double-Layered Ni-B/Ni-P Coating on AZ91D Mg Alloy from Eco-Friendly Fluoride-Free Baths. Surf. Coat. Technol. 2018, 342, 178–189. [Google Scholar] [CrossRef]
- Xie, Z.H.; Chen, F.; Xiang, S.; Zhou, J.; Song, Z.; Yu, G. Studies of Several Pickling and Activation Processes for Electroless Ni-P Plating on AZ31 Magnesium Alloy. J. Electrochem. Soc. 2015, 162, D115–D123. [Google Scholar] [CrossRef]
- Rajabalizadeh, Z.; Seifzadeh, D. Application of Electroless Ni-P Coating on Magnesium Alloy via CrO3/HF Free Titanate Pretreatment. Appl. Surf. Sci. 2017, 422, 696–709. [Google Scholar] [CrossRef]
- Duan, C.; Zhao, J.; Qin, L.; Yang, L.; Zhou, Y. Ternary Ni-Co-Mo Oxy-Hydroxide Nanoflakes Grown on Carbon Cloth for Excellent Supercapacitor Electrodes. Mater. Lett. 2017, 208, 65–68. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.; Feng, X.; Ye, X.; Fu, J. Small-Sized Mg-Al LDH Nanosheets Supported on Silica Aerogel with Large Pore Channels: Textural Properties and Basic Catalytic Performance after Activation. Nanomaterials 2018, 8, 113. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Liu, L.; Fang, Y.; Zhang, X.; Lyu, M.; Wang, S. The Adsorption of Dextranase onto Mg/Fe-Layered Double Hydroxide: Insight into the Immobilization. Nanomaterials 2018, 8, 173. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Wu, L.; Tang, A.; Zhang, S.; Yuan, B.; Zheng, Z.; Pan, F. A Novel Approach to Fabricate Protective Layered Double Hydroxide Films on the Surface of Anodized Mg-Al alloy. Adv. Mater. Interfaces 2017, 4, 1700163. [Google Scholar] [CrossRef]
- Guo, L.; Wu, W.; Zhou, Y.; Zhang, F.; Zeng, R.; Zeng, J. Layered Double Hydroxide Coatings on Magnesium Alloys: A Review. J. Mater. Sci. Technol. 2018. [Google Scholar] [CrossRef]
- Wu, L.; Yang, D.; Zhang, G.; Zhang, Z.; Zhang, S.; Tang, A.; Pan, F. Fabrication and Characterization of Mg-M Layered Double Hydroxide Films on Anodized Magnesium Alloy AZ31. Appl. Surf. Sci. 2018, 431, 177–186. [Google Scholar] [CrossRef]
- Zhang, F.; Liu, Z.-G.; Zeng, R.-C.; Li, S.-Q.; Cui, H.-Z.; Song, L.; Han, E.-H. Corrosion Resistance of Mg-Al-LDH Coating on Magnesium Alloy AZ31. Surf. Coat. Technol. 2014, 258, 1152–1158. [Google Scholar] [CrossRef]
- Yan, L.; Zhou, M.; Zhang, X.; Huang, L.; Chen, W.; Roy, V.A.L.; Zhang, W.; Chen, X. A Novel Type of Aqueous Dispersible Ultrathin-Layered Double Hydroxide Nanosheets for in Vivo Bioimaging and Drug Delivery. ACS Appl. Mater. Interfaces 2017, 9, 34185–34193. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Wang, D.; Tian, Y.; Cao, H.; Qiao, Y.; Liu, X. Sealing the Pores of PEO Coating with Mg-Al Layered Double Hydroxide: Enhanced Corrosion Resistance, Cytocompatibility and Drug Delivery Ability. Sci. Rep. 2017, 7, 8167. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Li, H.; Wang, D.; Tian, P.; Tian, Y.; Yuan, G.; Xu, D.; Liu, X. Enhanced Corrosion Resistance and Biocompatibility of Magnesium Alloy by Mg-Al-Layered Double Hydroxide. ACS Appl. Mater. Interfaces 2016, 8, 35033–35044. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Xue, J.; Xie, L.; Shen, J.; He, G.; Chen, H. Construction of Magnetically Separable NiAl LDH/Fe3O4-RGO Nanocomposites with Enhanced Photocatalytic Performance under Visible Light. Phys. Chem. Chem. Phys. 2018, 20, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Wang, D.; Cao, H.; Liu, X. Loading 5-Fluorouracil into Calcined Mg/Al Layered Double Hydroxide on AZ31 Via Memory Effect. Mater. Lett. 2018, 213, 383–386. [Google Scholar] [CrossRef]
- Zhou, M.; Yan, L.; Ling, H.; Diao, Y.; Pang, X.; Wang, Y.; Gao, K. Design and Fabrication of Enhanced Corrosion Resistance Zn-Al Layered Double Hydroxides Films Based Anion-Exchange Mechanism on Magnesium Alloys. Appl. Surf. Sci. 2017, 404, 246–253. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, L.; Tang, A.; Weng, B.; Atrens, A.; Ma, S.; Liu, L.; Pan, F. Sealing of Anodized Magnesium Alloy AZ31 with MgAl Layered Double Hydroxides Layers. RSC Adv. 2018, 8, 2248–2259. [Google Scholar] [CrossRef]
- Li, D.; Chen, F.; Xie, Z.-H.; Shan, S.; Zhong, C.-J. Enhancing Structure Integrity and Corrosion Resistance of Mg Alloy by a Two-Step Deposition to Avoid F Ions Etching to Nano-SiO2 Reinforcement. J. Alloy. Compd. 2017, 705, 70–78. [Google Scholar] [CrossRef]
- Xie, Z.-H.; Shan, S. Nanocontainers-Enhanced Self-Healing Ni Coating for Corrosion Protection of Mg Alloy. J. Mater. Sci. 2018, 53, 3744–3755. [Google Scholar] [CrossRef]
- Zeng, R.-C.; Liu, Z.-G.; Zhang, F.; Li, S.-Q.; Cui, H.-Z.; Han, E.-H. Corrosion of Molybdate Intercalated Hydrotalcite Coating on AZ31 Mg Alloy. J. Mater. Chem. A 2014, 2, 13049–13057. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, J.; Dai, N.; Qin, P.; Attar, H.; Zhang, L.-C. Corrosion Behaviour of Selective Laser Melted Ti-TiB Biocomposite in Simulated Body Fluid. Electrochim. Acta 2017, 232, 89–97. [Google Scholar] [CrossRef]
- Gan, R.; Wang, D.; Xie, Z.-H.; He, L. Improving Surface Characteristic and Corrosion Inhibition of Coating on Mg Alloy by Trace Stannous (II) Chloride. Corros. Sci. 2017, 123, 147–157. [Google Scholar] [CrossRef]
- Dai, N.; Zhang, J.; Chen, Y.; Zhang, L.-C. Heat Treatment Degrading the Corrosion Resistance of Selective Laser Melted Ti-6Al-4V Alloy. J. Electrochem. Soc. 2017, 164, C428–C434. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, J.; Gu, X.; Dai, N.; Qin, P.; Zhang, L.-C. Distinction of Corrosion Resistance of Selective Laser Melted Al-12Si Alloy on Different Planes. J. Alloy. Compd. 2018, 747, 648–658. [Google Scholar] [CrossRef]




| Substrate | LDH Coatings | Corrosive Medium | Ecorr V vs. SCE | jcorr (A cm−2) | Ref. |
|---|---|---|---|---|---|
| Mg alloy | MgAl-NO3− | Phosphate buffer saline | −1.53 | 3.63 × 10−7 | [14] |
| Anodized AZ31 Mg alloy | MgAl-NO3− | 3.5 wt % NaCl solution | −0.47 | 9.48 × 10−7 | [8] |
| Anodized AZ31 Mg alloy | MgAl-VO3− | 3.5 wt % NaCl solution | −0.40 | 2.48 × 10−7 | [8] |
| Anodized AZ31 Mg alloy | MgAl-NO3− | 3.5 wt % NaCl solution | −1.34 | 1.18 × 10−7 | [10] |
| AZ31 Mg alloy | MgAl-5-fluorouracil | Phosphate buffer saline | −1.12 | 3.34 × 10−5 | [13] |
| Plasma electrolytic oxidation pretreated AZ31 Mg alloy | MgAl-5-fluorouracil | Phosphate buffer saline | −1.20 | 3.92 × 10−6 | [13] |
| AZ31 Mg alloy | MgAl-5-fluorouracil | Phosphate buffer saline | −1.42 | 3.27 × 10−5 | [16] |
| Anodized AZ31 Mg alloy | MgFe-NO3− | 3.5 wt % NaCl solution | −1.44 | 1.09 × 10−6 | [10] |
| Anodized AZ31 Mg alloy | MgCr-NO3− | 3.5 wt % NaCl solution | −1.47 | 2.16 × 10−6 | [10] |
| AZ91D Mg alloy | ZnAl-VO3− | 3.5 wt % NaCl solution | −1.30 | 2.21 × 10−6 | [17] |
| AZ91D Mg alloy | ZnAl-Cl− | 3.5 wt % NaCl solution | −1.39 | 2.52 × 10−6 | [17] |
| AZ91D Mg alloy | ZnAl-NO3− | 3.5 wt % NaCl solution | −1.42 | 1.33 × 10−5 | [17] |
| AZ31 Mg alloy | MgAl-CO32− | 3.5 wt % NaCl solution | −0.36 | 8.40 × 10−7 | [18] |
| Anodized AZ31 | MgAl-CO32− | 3.5 wt % NaCl solution | −0.29 | 3.50 × 10−7 | [18] |
| Samples | Qf/10−9 (S sn cm−2) | Rf (MΩ cm2) | Qdl/10−9 (S sn cm−2) | Rct (MΩ cm2) | W/10−7 (S s0.5 cm−2) | |Z|f = 0.1 Hz (MΩ cm2) | χ2/10−3 |
|---|---|---|---|---|---|---|---|
| NiAl-LDH/CT coating | 2.1 ± 0.029 | 6.2 ± 0.17 | 62.8 ± 7.6 | 3.5 ± 0.45 | 3.5 ± 0.17 | 11.6 | 0.31 |
| NiAl-LDH/NT coating | 1.5 ± 0.021 | 3.7 ± 0.14 | 113.5 ± 9.8 | 1.8 ± 0.22 | 3.5 ± 0.35 | 6.7 | 0.22 |
| NiAl-LDH/ST coating | 0.84 ± 0.087 | 1.4 ± 0.15 | 24.5 ± 2.6 | 0.72 ± 0.076 | 6.9 ± 0.27 | 3.4 | 1.5 |
| Samples | Ecorr (mV) | jcorr (nA cm−2) | βa (mV dec−1) | −βc (mV dec−1) |
|---|---|---|---|---|
| Bare Mg alloy | −1450 ± 52 | 3242 ± 425 | 331 | 95 |
| NiAl-LDH/CT coating | −674 ± 42 | 1.06 ± 0.59 | 190 | 171 |
| NiAl-LDH/NT coating | −625 ± 96 | 3.24 ± 0.25 | 244 | 184 |
| NiAl-LDH/ST coating | −577 ± 51 | 5.75 ± 1.03 | 270 | 188 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, X.; Jiang, Z.; Li, L.; Xie, Z.-H. In-Situ Growth of NiAl-Layered Double Hydroxide on AZ31 Mg Alloy towards Enhanced Corrosion Protection. Nanomaterials 2018, 8, 411. https://doi.org/10.3390/nano8060411
Ye X, Jiang Z, Li L, Xie Z-H. In-Situ Growth of NiAl-Layered Double Hydroxide on AZ31 Mg Alloy towards Enhanced Corrosion Protection. Nanomaterials. 2018; 8(6):411. https://doi.org/10.3390/nano8060411
Chicago/Turabian StyleYe, Xin, Zimin Jiang, Linxin Li, and Zhi-Hui Xie. 2018. "In-Situ Growth of NiAl-Layered Double Hydroxide on AZ31 Mg Alloy towards Enhanced Corrosion Protection" Nanomaterials 8, no. 6: 411. https://doi.org/10.3390/nano8060411
APA StyleYe, X., Jiang, Z., Li, L., & Xie, Z.-H. (2018). In-Situ Growth of NiAl-Layered Double Hydroxide on AZ31 Mg Alloy towards Enhanced Corrosion Protection. Nanomaterials, 8(6), 411. https://doi.org/10.3390/nano8060411




