Diazotization-Coupling Reaction-Based Determination of Tyrosine in Urine Using Ag Nanocubes by Surface-Enhanced Raman Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instrumentation
2.2. Preparation of AgNCs Substrates
2.3. SERS-based Diazotization-Coupling Reaction Test
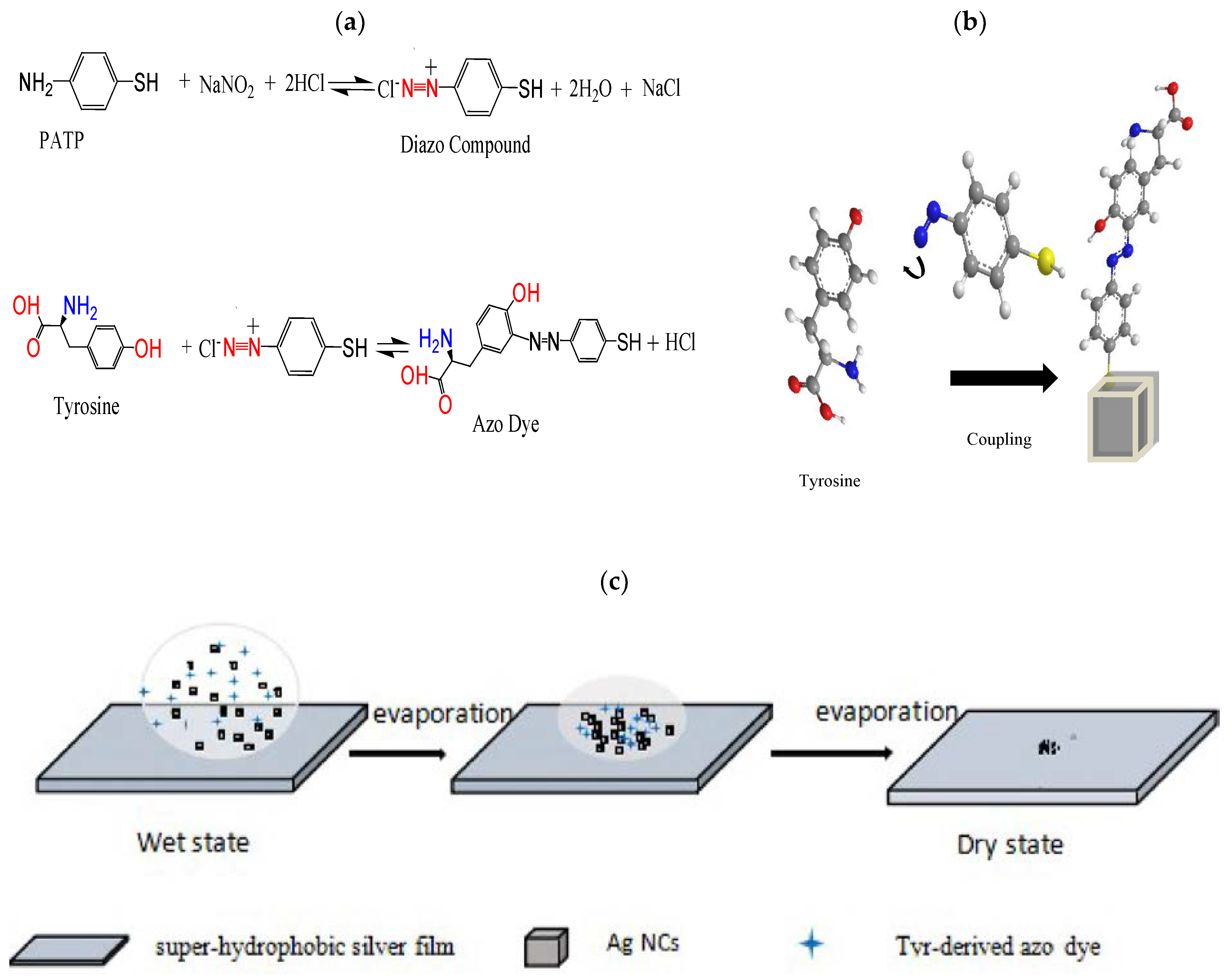
2.4. SERS-Based Diazotization-Coupling Reaction and Principle
3. Results and Discussion
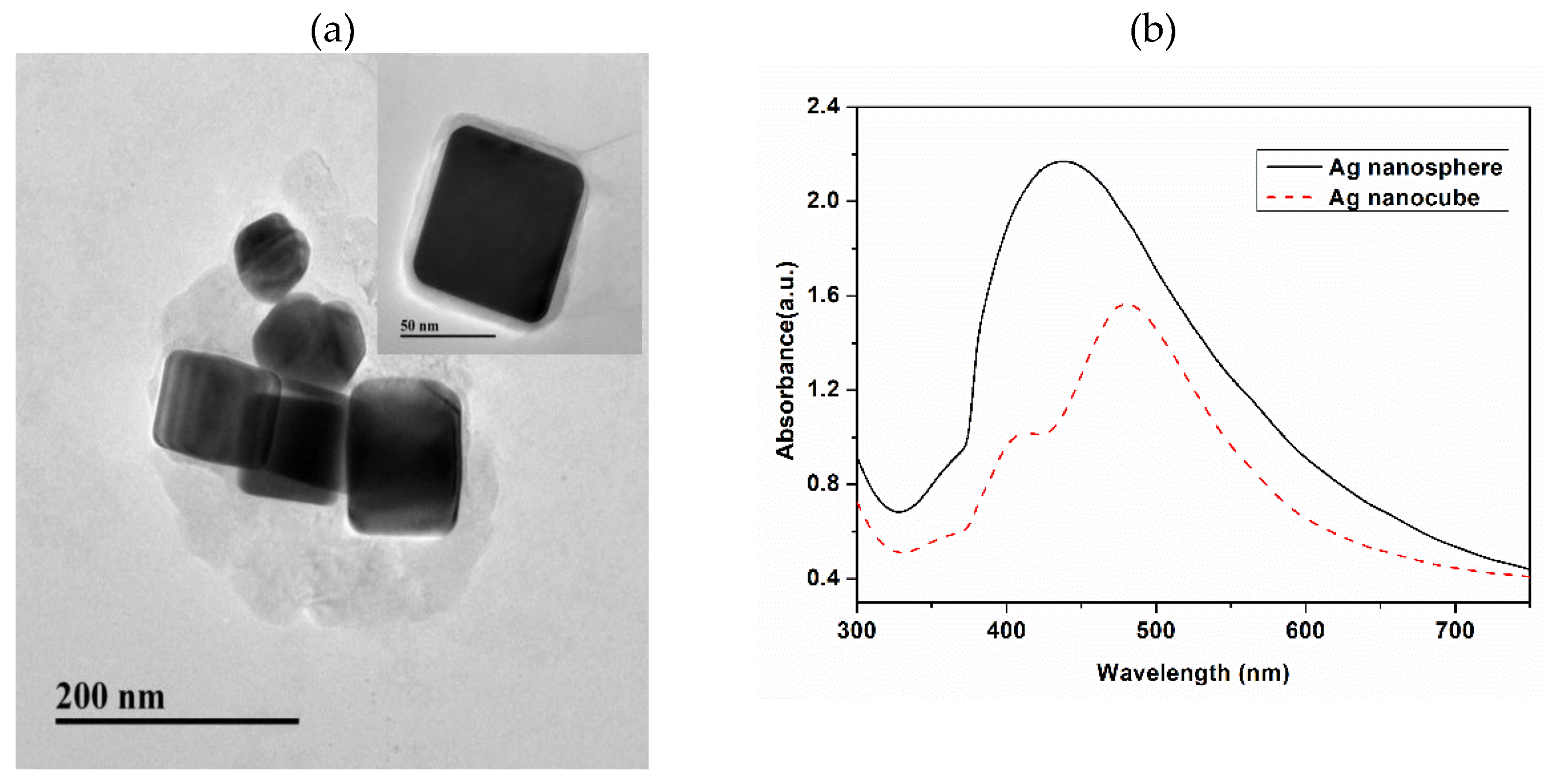
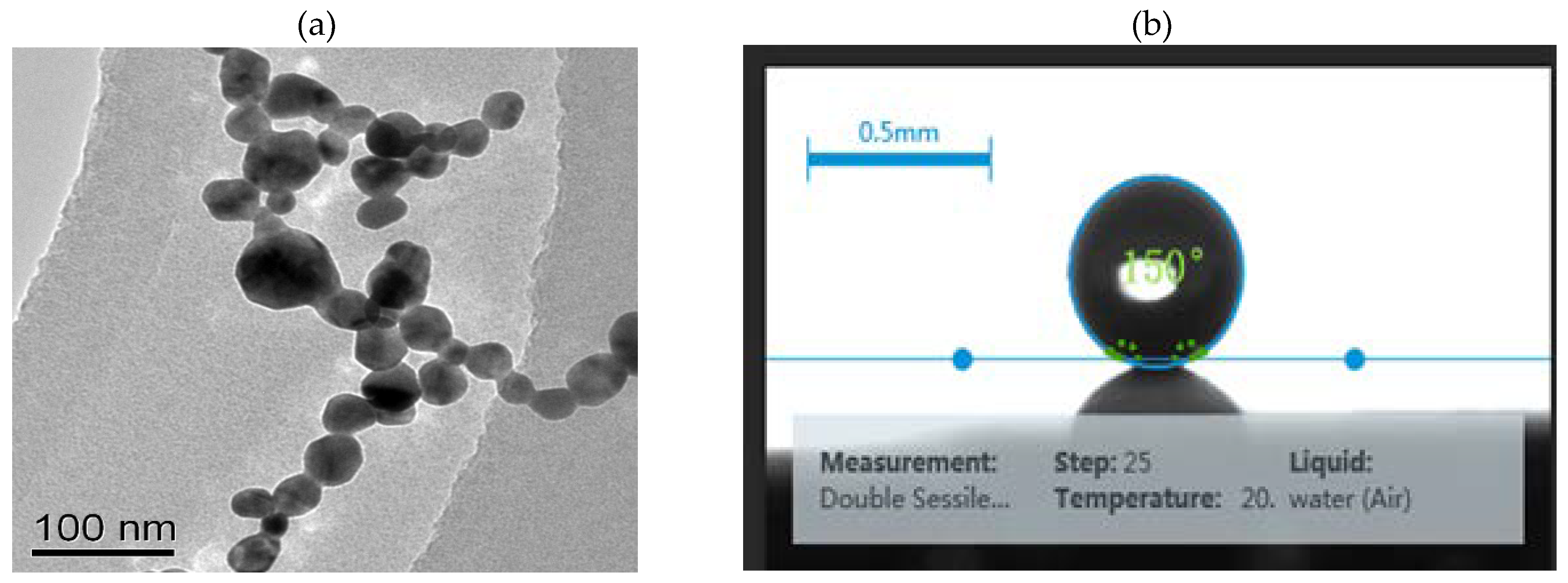
3.1. Characterization of the Prepared AgNCs
3.2. Detection of Azo Dye-Derived from Tyr by SERS
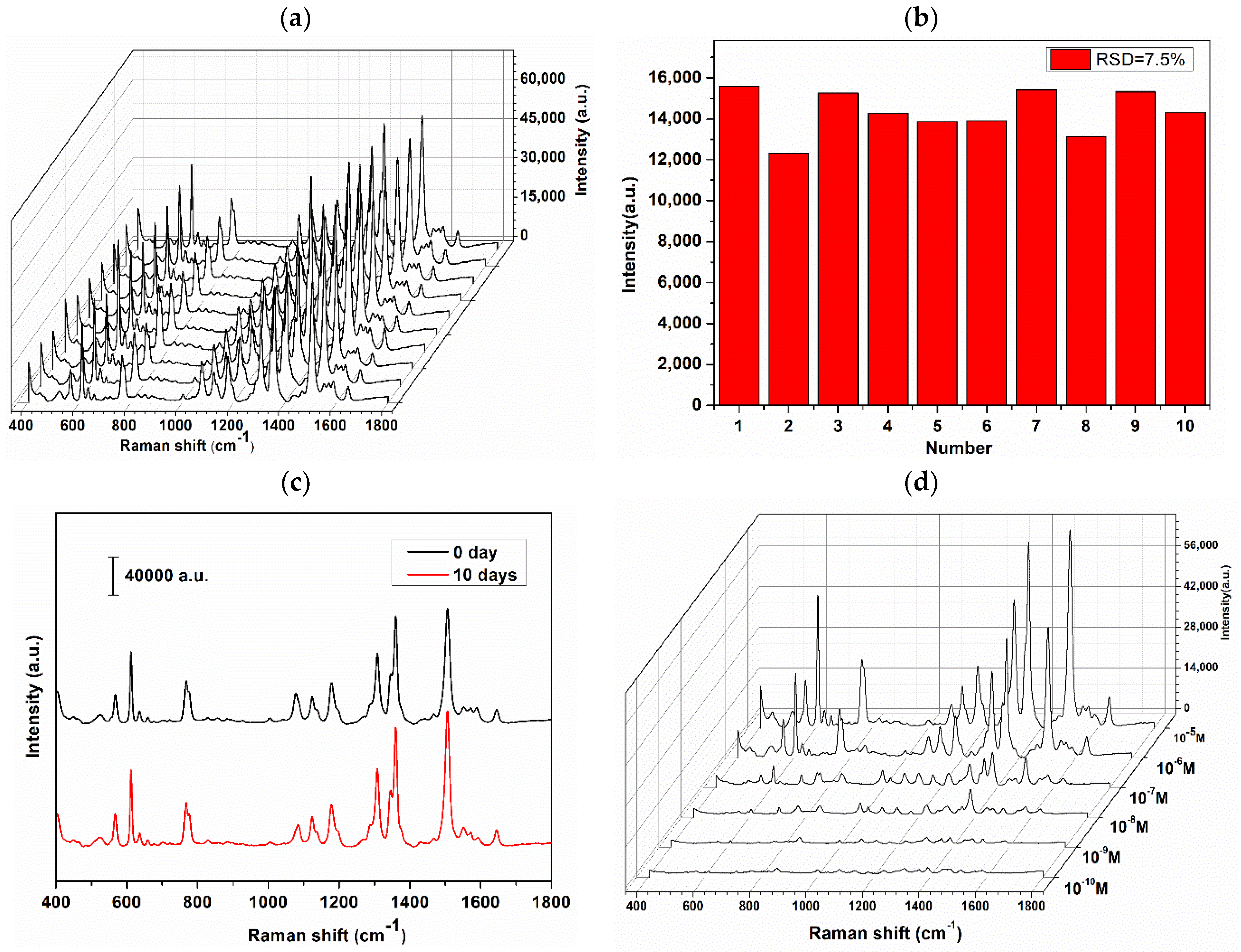
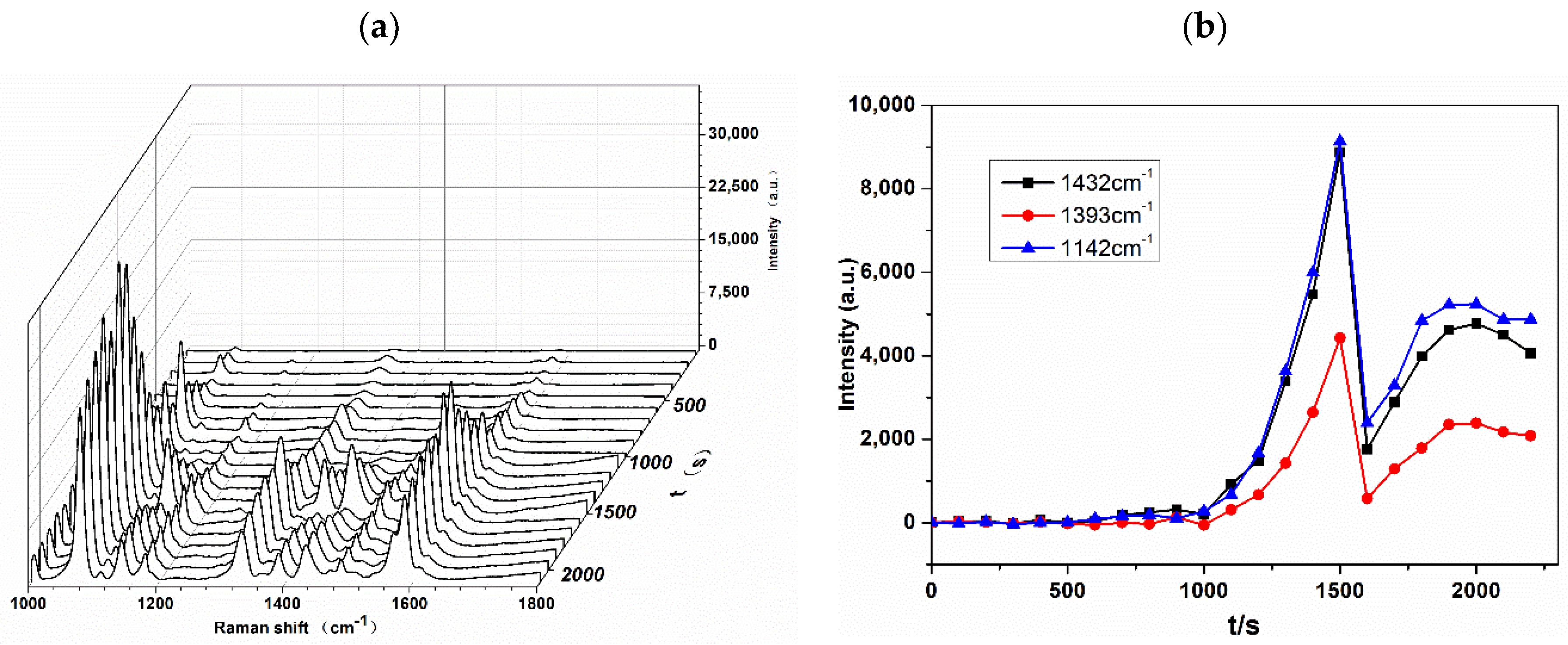
3.3. Detection of Azo Dye Derived from Tyr by D-SERS
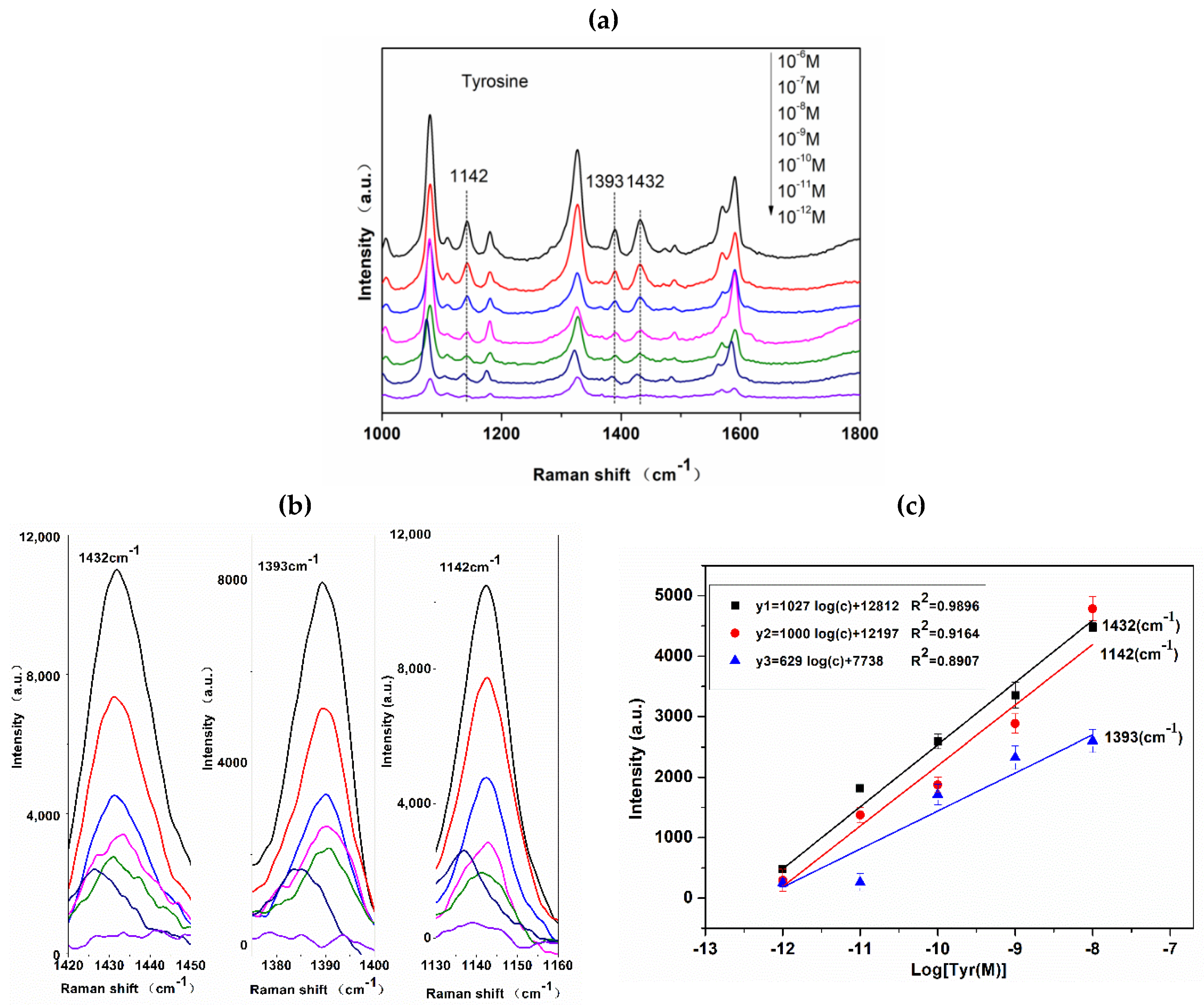
3.4. SERS Sensitivity of Azo Dye Derived from Tyr
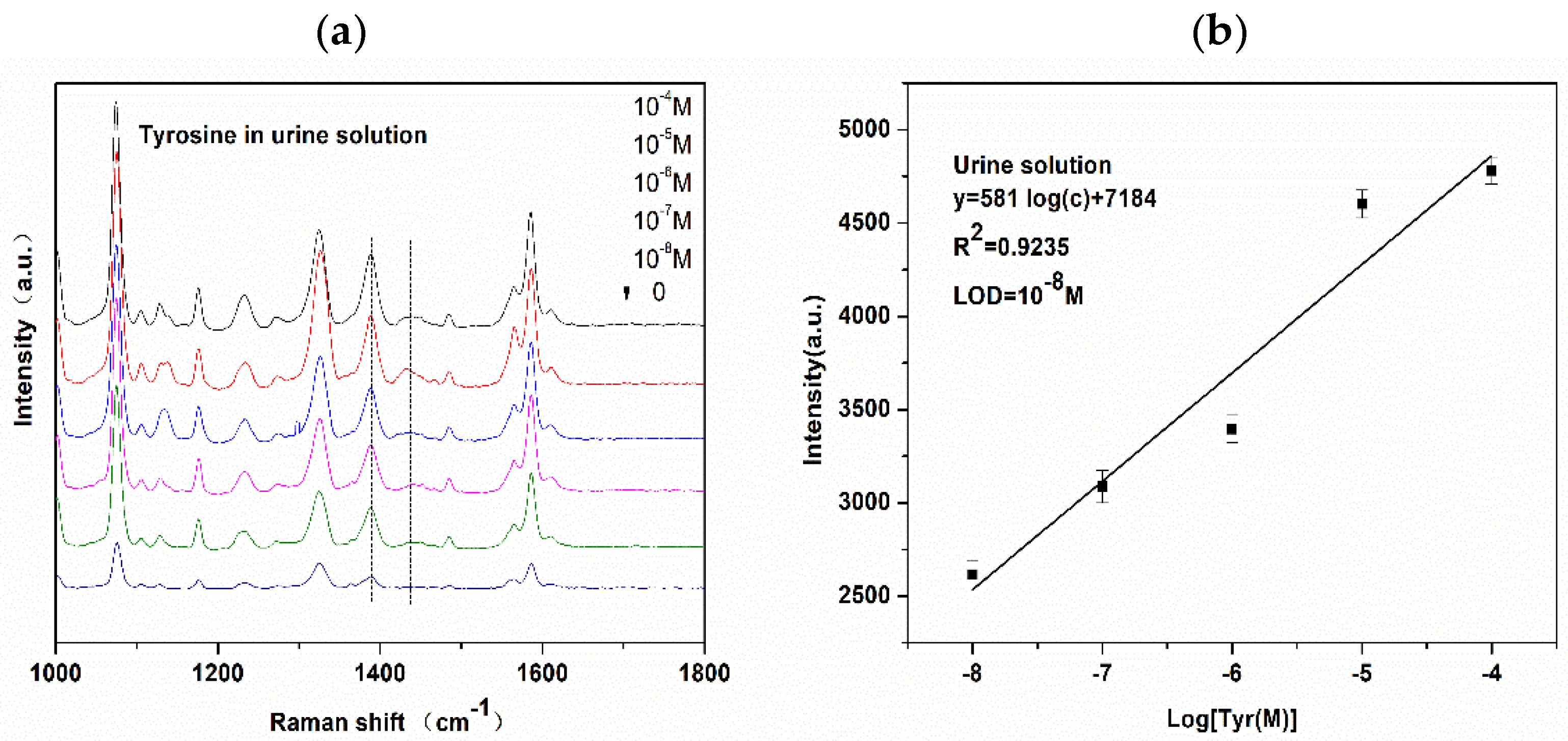
3.5. Application for Urine Detection
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ney, D.M.; Murali, S.G.; Stroup, B.M.; Nair, N.; Sawin, E.A.; Rohr, F.; Levy, H.L. Metabolomic changes demonstrate reduced bioavailability of tyrosine and altered metabolism of tryptophan via the kynurenine pathway with ingestion of medical foods in phenylketonuria. Mol. Genet. Metab. 2017, 121, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Bilandžija, H.; Ma, L.; Parkhurst, A.; Jeffery, W.R. A potential benefit of albinism in astyanax cavefish: Downregulation of the oca2 gene increases tyrosine and catecholamine levels as an alternative to melanin synthesis. PLoS ONE 2013, 8, e80823. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Tang, L.; Niessner, R.; Ying, Y.; Haisch, C. Nitrite-triggered surface plasmon-assisted catalytic conversion of p-aminothiophenol to p,p’-dimercaptoazobenzene on gold nanoparticle: Surface-enhanced Raman scattering investigation and potential for nitrite detection. Anal. Chem. 2015, 87, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Moein, M.M.; El-Beqqali, A.; Abdel-Rehim, A.; Jeppsson-Dadoun, A.; Abdel-Rehim, M. Preparation of monolithic molecularly imprinted polymer sol-gel packed tips for high-throughput bioanalysis: Extraction and quantification of L-tyrosine in human plasma and urine samples utilizing liquid chromatography and tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 967, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Ya, M.A.; Tan, T.; Yan, A.P.; Guo, L.; Wan, Y.Q. Determination of tyrosine, 4-hydroxyphenyllactic acid and 4-hydroxyphenylacetic acid in human urine by ultra-performance liquid chromatography coupled with fluorescence detection. J. Anal. Sci. 2015, 31, 333–338. [Google Scholar]
- Alonso, M.S.; Zamora, L.L.; Calatayud, J.M. Determination of tyrosine through a FIA-direct chemiluminescence procedure. Talanta 2003, 60, 369–376. [Google Scholar] [CrossRef]
- Huang, X.; Liu, Y.; Yung, B.; Xiong, Y.; Chen, X. Nanotechnology-enhanced no-wash biosensors for in vitro diagnostics of cancer. ACS Nano 2017, 11, 5238–5292. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Chen, R.; Lin, J.; Pan, J.; Chen, G.; Li, Y.; Cheng, M.; Huang, Z.; Chen, J.; Zeng, H. Nasopharyngeal cancer detection based on blood plasma surface-enhanced Raman spectroscopy and multivariate analysis. Biosens. Bioelectron. 2010, 25, 2414–2419. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Feng, S.; Pan, J.; Chen, Y.; Lin, J.; Chen, G.; Zeng, S.X.; Chen, R. Colorectal cancer detection by gold nanoparticle based surface-enhanced Raman spectroscopy of blood serum and statistical analysis. Opt. Express 2011, 19, 13565–13577. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Qu, L.; Zhai, W.; Xue, J.; Fossey, J.S.; Long, Y. Facile on-site detection of substituted aromatic pollutants in water using thin layer chromatography combined with surface-enhanced Raman spectroscopy. Environ. Sci. Technol. 2011, 45, 4046–4052. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Liu, Z.; Zhong, H.; Yang, H.; Zhou, Y.; Hou, Y.; Long, J.; Lin, J.; Guo, Z. Melanin-associated synthesis of SERS-active nanostructures and the application for monitoring of intracellular melanogenesis. Nanomaterials 2017, 7, 70. [Google Scholar] [CrossRef] [PubMed]
- Katrin, K.; Harald, K.; Kamran, N.; Charles, B.; Karen, E.; Ramachandra, R. Surface-enhanced raman spectroscopy in single living cells using gold nanoparticles. Appl. Spectrosc. 2002, 56, 150–154. [Google Scholar]
- Gao, J.; Sanchez-Purra, M.; Huang, H.; Wang, S.; Chen, Y.; Yu, X.; Luo, Q.; Hamad-Schifferli, K.; Liu, S. Synthesis of different-sized gold nanostars for Raman bioimaging and photothermal therapy in cancer nanotheranostics. Sci. China Chem. 2017, 60, 1219–1229. [Google Scholar] [CrossRef]
- Zhang, M.-R.; Jiang, Q.-M.; Wang, Z.-G.; Zhang, S.-H.; Hou, F.; Pan, G.-B. Three-dimensional gallium nitride nanoflowers supports decorated by gold or silver nanoparticles to fabricate surface-enhanced Raman scattering substrates. Sens. Actuators B 2017, 253, 652–659. [Google Scholar] [CrossRef]
- Kundu, S.; Dai, W.; Chen, Y.; Ma, L.; Yue, Y.; Sinyukov, A.M.; Liang, H. Shape-selective catalysis and surface enhanced Raman scattering studies using Ag nanocubes, nanospheres and aggregated anisotropic nanostructures. J. Colloid Interface Sci. 2017, 498, 248–262. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Zhang, L.; Zhou, J. Silver nanocube-mediated sensitive immunoassay based on surface-enhanced Raman scattering assisted by etched silicon nanowire arrays. Analyst 2014, 139, 5893–5900. [Google Scholar] [CrossRef] [PubMed]
- Ben-Jaber, S.; Peveler, W.J.; Quesada-Cabrera, R.; Sol, C.W.O.; Papakonstantinou, I.; Parkin, I.P. Sensitive and specific detection of explosives in solution and vapour by surface-enhanced Raman spectroscopy on silver nanocubes. Nanoscale 2017, 9, 16459–16466. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhang, L.; Zhou, X. Synthesis of silver nanocubes as a SERS substrate for the determination of pesticide paraoxon and thiram. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2014, 121, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Hasna, K.; Lakshmi, K.; Ezhuthachan Jayaraj, M.K.; Kumar, K.R.; Matham, M.V. Development of high-sensitive, reproducible colloidal surface-enhanced Raman spectroscopy active substrate using silver nanocubes for potential biosensing applications. J. Nanophotonics 2016, 10, 20–29. [Google Scholar] [CrossRef]
- Moger, J.; Gribbon, P.; Sewing, A.; Winlove, C.P. Feasibility study using surface-enhanced Raman spectroscopy for the quantitative detection of tyrosine and serine phosphorylation. BBA Gen. Subj. 2007, 1770, 912–918. [Google Scholar] [CrossRef] [PubMed]
- Topkaya, S.N.; Ozyurt, V.H.; Cetin, A.E.; Otles, S. Nitration of tyrosine and its effect on DNA hybridization. Biosens. Bioelectron. 2018, 102, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.L.; Tsai, B.C.; Yang, J. Silver nanoparticle-treated filter paper as a highly sensitive surface-enhanced Raman scattering (SERS) substrate for detection of tyrosine in aqueous solution. Anal. Chim. Acta 2011, 708, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Sui, H.; Wang, Y.; Yu, Z.; Cong, Q.; Han, X.X.; Zhao, B. A rapid and ultrasensitive SERRS assay for histidine and tyrosine based on azo coupling. Talanta 2016, 159, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Han, X.X.; Pienpinijtham, P.; Zhao, B.; Ozaki, Y. Coupling reaction-based ultrasensitive detection of phenolic estrogens using surface-enhanced resonance Raman scattering. Anal. Chem. 2011, 83, 8582–8588. [Google Scholar] [CrossRef] [PubMed]
- Sui, H.; Wang, Y.; Zhang, X.; Wang, X.; Cheng, W.; Su, H.; Wang, X.; Sun, X.; Han, X.X.; Zhao, B.; et al. Ultrasensitive detection of thyrotropin-releasing hormone based on azo coupling and surface-enhanced resonance Raman spectroscopy. Analyst 2016, 141, 5181–5188. [Google Scholar] [CrossRef] [PubMed]
- Angelis, F.D.; Gentile, F.; Mecarini, F.; Das, G.; Moretti, M.; Candeloro, P.; Coluccio, M.L.; Cojoc, G.; Accardo, A.; Liberale, C. Breaking the diffusion limit with super-hydrophobic delivery of molecules to plasmonic nanofocusing SERS structures. Nat. Photonics 2011, 5, 682–687. [Google Scholar] [CrossRef]
- Yan, X.; Li, P.; Zhou, B.; Tang, X.; Li, X.; Weng, S.; Yang, L.; Liu, J. Optimal hotspots of dynamic surfaced-enhanced raman spectroscopy for drugs quantitative detection. Anal. Chem. 2017, 89, 4875–4881. [Google Scholar] [CrossRef] [PubMed]
- Kuttner, C.; Mayer, M.; Dulle, M.; Moscoso, A.; Lopez-Romero, J.M.; Forster, S.; Fery, A.; Perez-Juste, J.; Contreras-Caceres, R. Seeded growth synthesis of gold nanotriangles: Size control, SAXS analysis, and SERS performance. ACS Appl. Mater. Interfaces 2018, 10, 11152–11163. [Google Scholar] [CrossRef] [PubMed]
- Lend, N.; La, B. A new method for fast preparation of highly Surface-Enhanced Raman Scattering (SERS) active silver colloids at room temperature by reduction of silver nitrate with hydroxylamine hydrochloride. J. Phys. Chem. B 2003, 107, 5723–5727. [Google Scholar]
- Khlebtsov, B.; Khanadeev, V.; Khlebtsov, N. Surface-enhanced Raman scattering inside Au@Ag core/shell nanorods. Nano Res. 2016, 9, 2303–2318. [Google Scholar] [CrossRef]
- Huang, J.A.; Zhao, Y.Q.; Zhang, X.J.; He, L.F.; Wong, T.L.; Chui, Y.S.; Zhang, W.J.; Lee, S.T. Ordered Ag/Si nanowires array: Wide-range surface-enhanced Raman spectroscopy for reproducible biomolecule detection. Nano Lett. 2013, 13, 5039–5045. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Li, J.; Zhang, L.; Wang, B.; Zhou, J. Microwave assisted in situ synthesis of Ag–NaCMC films and their reproducible surface-enhanced Raman scattering signals. J. Alloys Compd. 2014, 602, 94–100. [Google Scholar] [CrossRef]
- Zhao, L.; Huang, Y.; Wu, D.; Ren, B. Surface-enhanced raman spectroscopy and plasmon-assisted photocatalysis of p-aminothiophenol. Acta Chim. Sin. 2014, 72, 1125. [Google Scholar] [CrossRef]
- Zhang, K.; Hu, Y.; Li, G. Diazotization-coupling reaction-based selective determination of nitrite in complex samples using shell-isolated nanoparticle-enhanced Raman spectroscopy. Talanta 2013, 116, 712–718. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Feng, L.; Xiao, D.; Li, N.; Li, Y.; Cao, D.; Shi, Z.; Cui, Z.; Lu, N. Silver nanoislands over silica spheres platform: Enriching trace-amount analytes for ultrasensitive and reproducible SERS detections. Nanoscale 2017, 9, 16749–16754. [Google Scholar] [CrossRef] [PubMed]
- Subaihi, A.; Almanqur, L.; Muhamadali, H.; AlMasoud, N.; Ellis, D.I.; Trivedi, D.K.; Hollywood, K.A.; Xu, Y.; Goodacre, R. Rapid, accurate, and quantitative detection of propranolol in multiple human biofluids via surface-enhanced raman scattering. Anal. Chem. 2016, 88, 10884–10892. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Pang, S.; He, L.; Nugen, S.R. Highly sensitive and selective detection of nitrite ions using Fe3O4@SiO2/Au magnetic nanoparticles by surface-enhanced Raman spectroscopy. Biosens. Bioelectron. 2016, 85, 726–733. [Google Scholar] [CrossRef] [PubMed]


| Samples | True concentration (mol/L) | Recovery (%) |
|---|---|---|
| Tyr in the urine | 2.50 × 10−6 | 135.2 |
| 2.50 × 10−7 | 110.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, Y.; Lu, D.; You, R.; Liu, J.; Huang, L.; Su, J.; Feng, S. Diazotization-Coupling Reaction-Based Determination of Tyrosine in Urine Using Ag Nanocubes by Surface-Enhanced Raman Spectroscopy. Nanomaterials 2018, 8, 400. https://doi.org/10.3390/nano8060400
Lu Y, Lu D, You R, Liu J, Huang L, Su J, Feng S. Diazotization-Coupling Reaction-Based Determination of Tyrosine in Urine Using Ag Nanocubes by Surface-Enhanced Raman Spectroscopy. Nanomaterials. 2018; 8(6):400. https://doi.org/10.3390/nano8060400
Chicago/Turabian StyleLu, Yudong, Dechan Lu, Ruiyun You, Jialing Liu, Luqiang Huang, Jingqian Su, and Shangyuan Feng. 2018. "Diazotization-Coupling Reaction-Based Determination of Tyrosine in Urine Using Ag Nanocubes by Surface-Enhanced Raman Spectroscopy" Nanomaterials 8, no. 6: 400. https://doi.org/10.3390/nano8060400
APA StyleLu, Y., Lu, D., You, R., Liu, J., Huang, L., Su, J., & Feng, S. (2018). Diazotization-Coupling Reaction-Based Determination of Tyrosine in Urine Using Ag Nanocubes by Surface-Enhanced Raman Spectroscopy. Nanomaterials, 8(6), 400. https://doi.org/10.3390/nano8060400




