On the Structure of Ultrathin FeO Films on Ag(111)
Abstract
1. Introduction
2. Experimental and Theoretical Methods
3. Results and Discussion
3.1. Iron Deposition at Room Temperature and Post-Oxidation
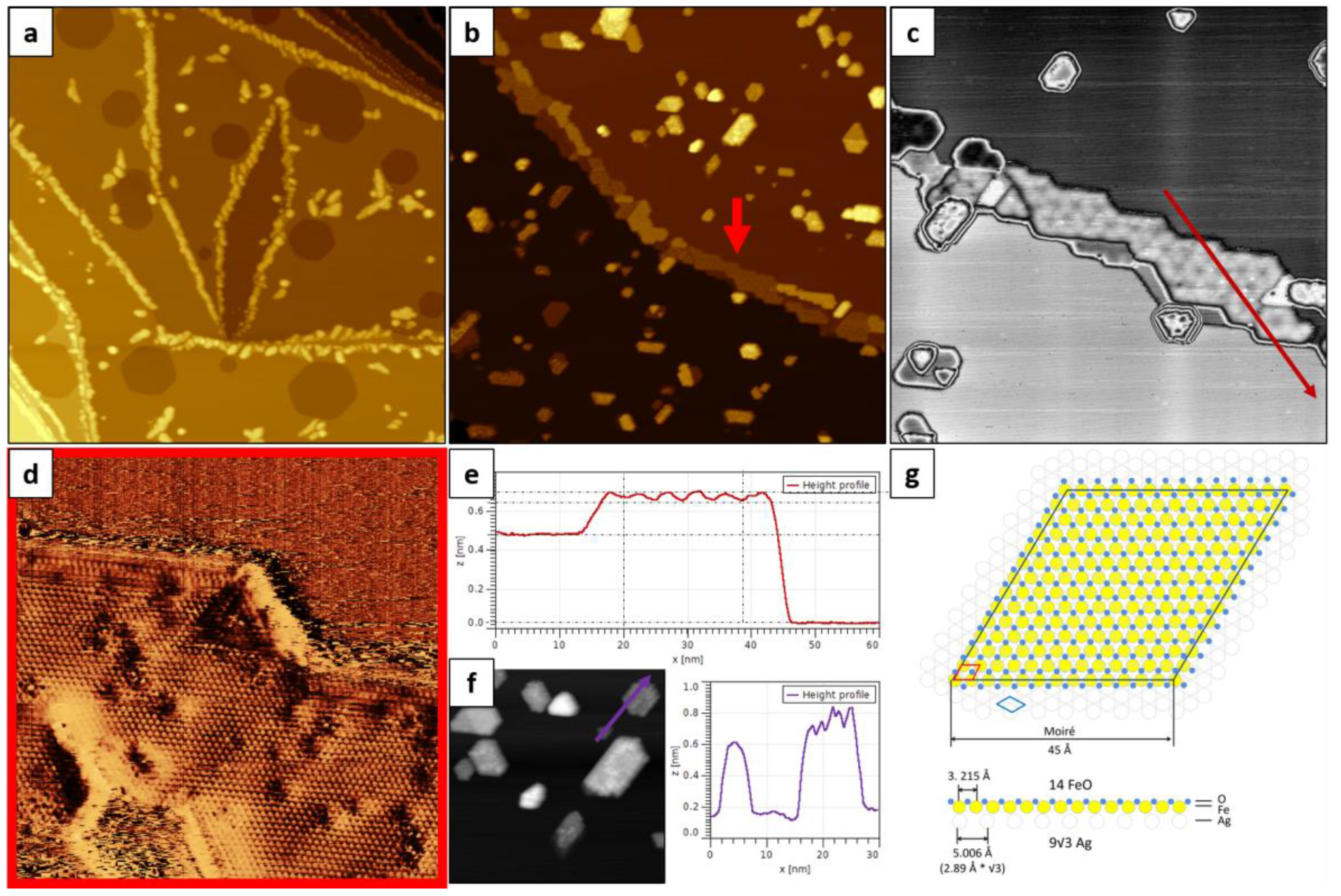
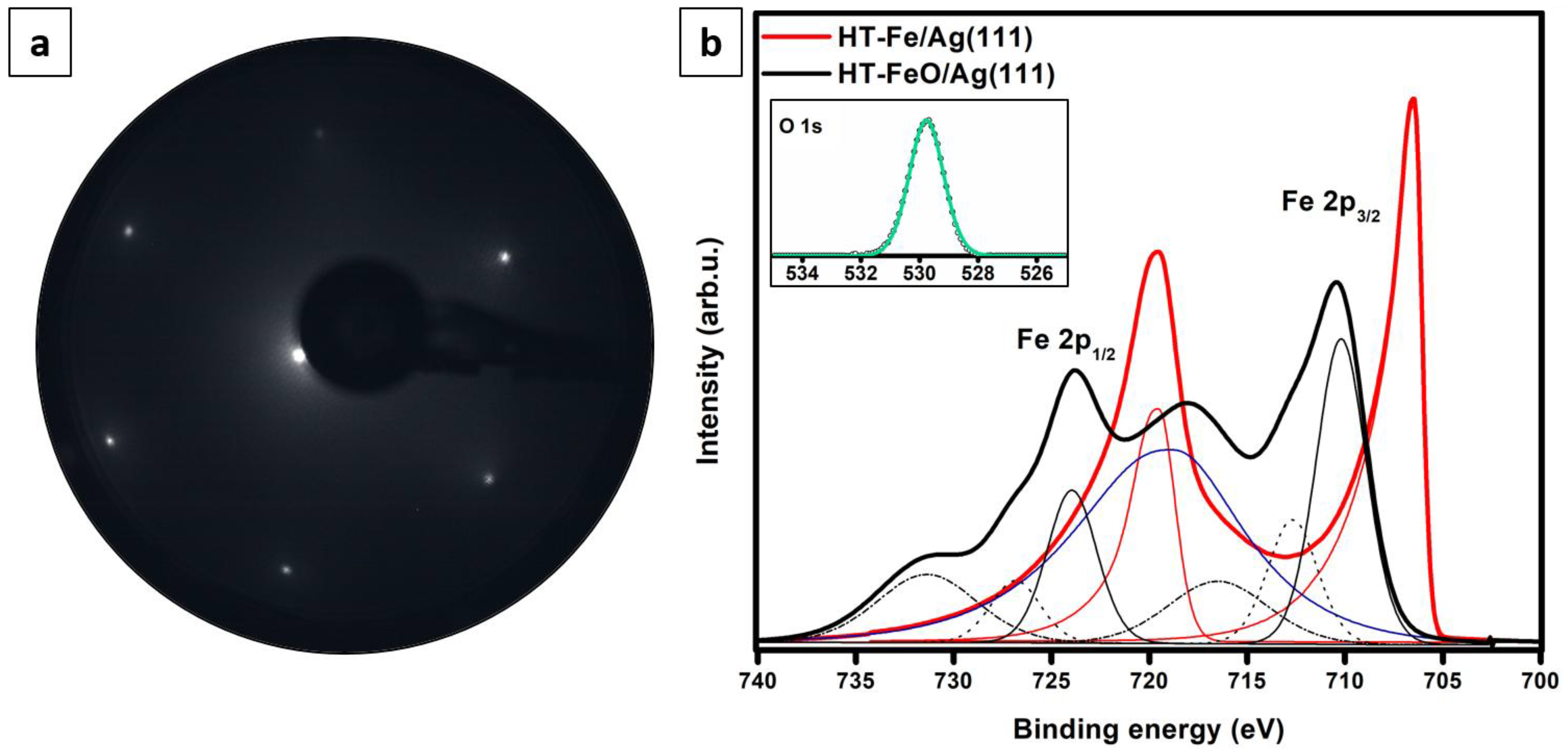
3.2. Iron Deposition at High Temperature and Post-Oxidation
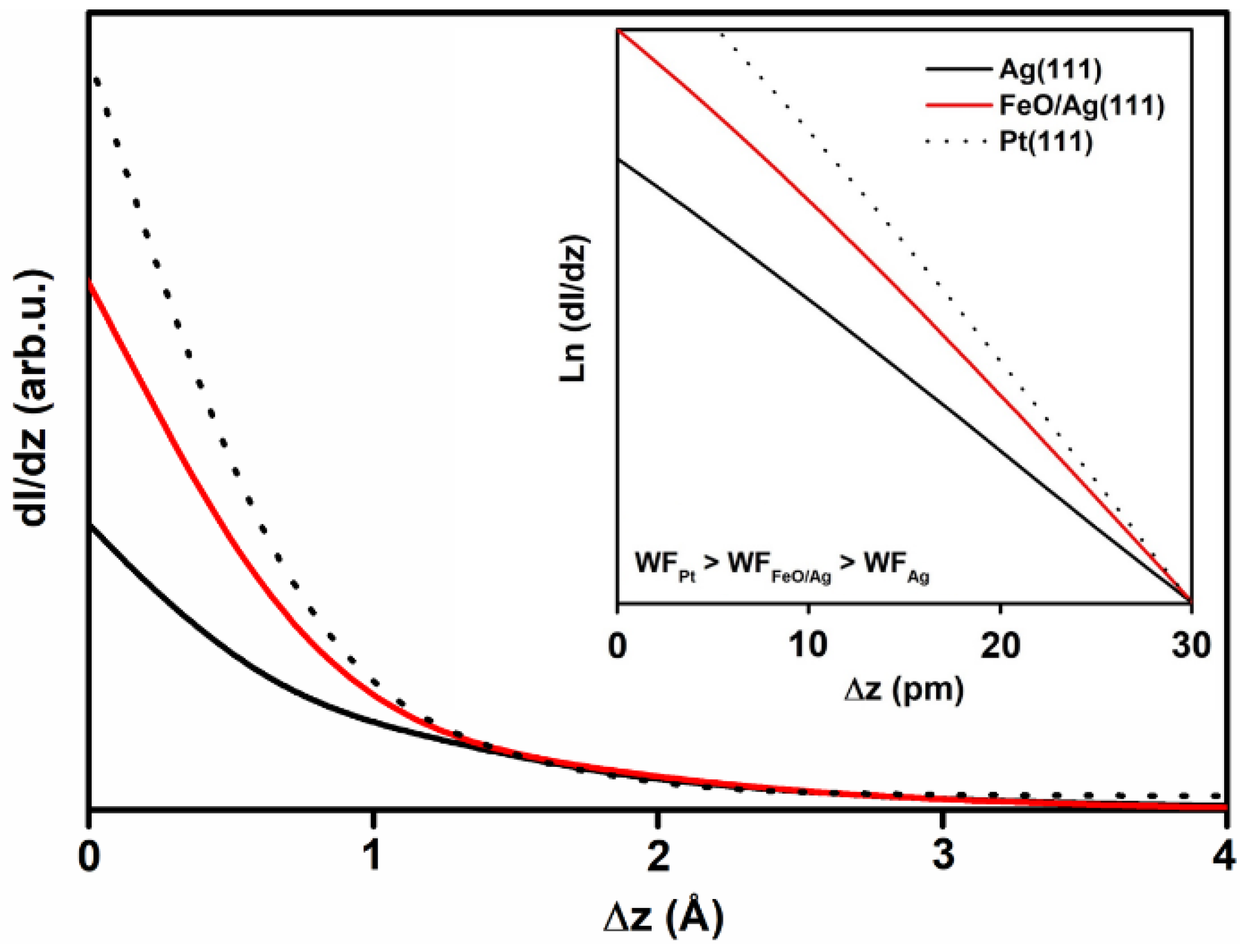
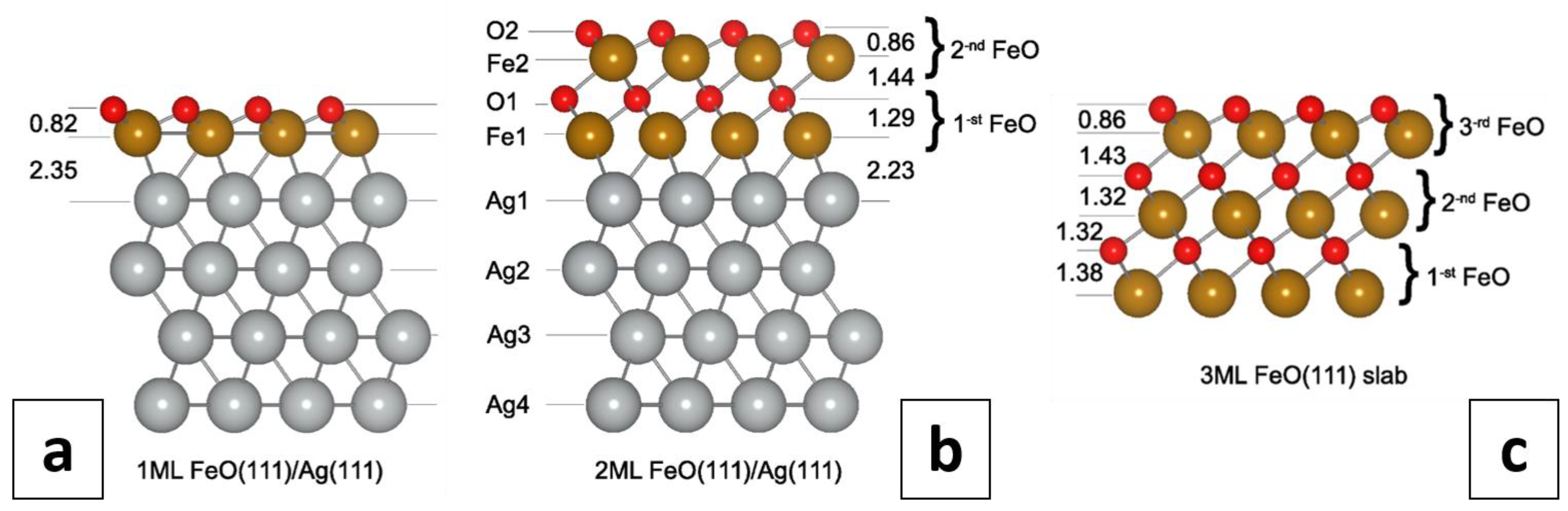
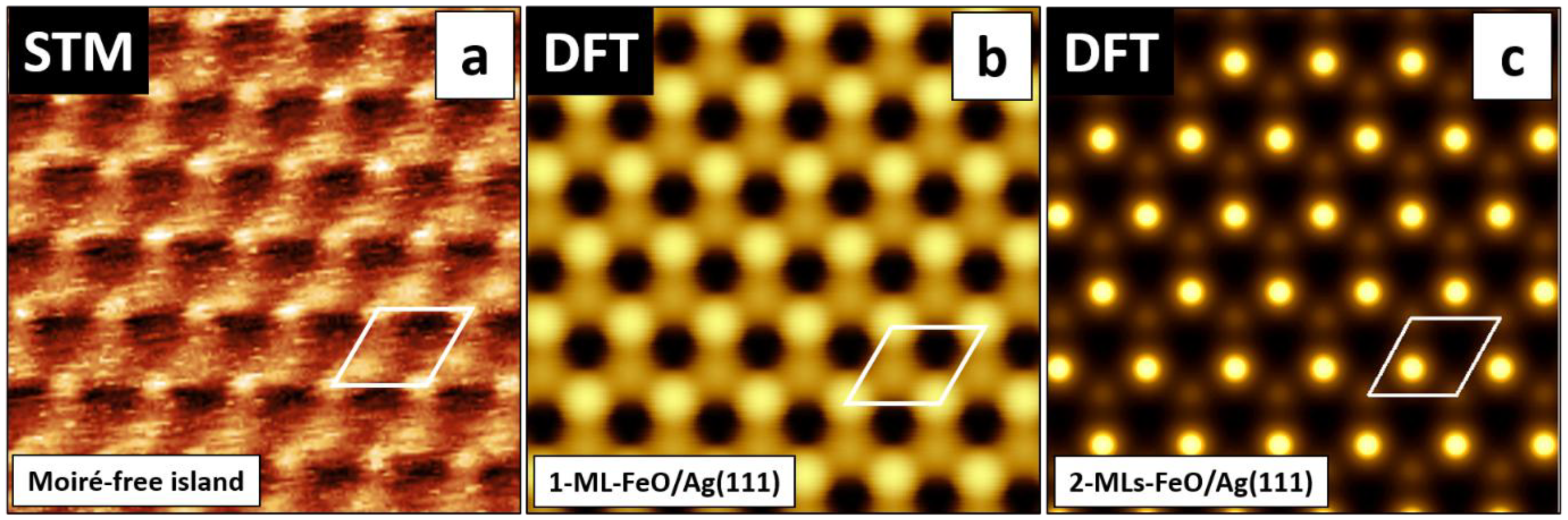
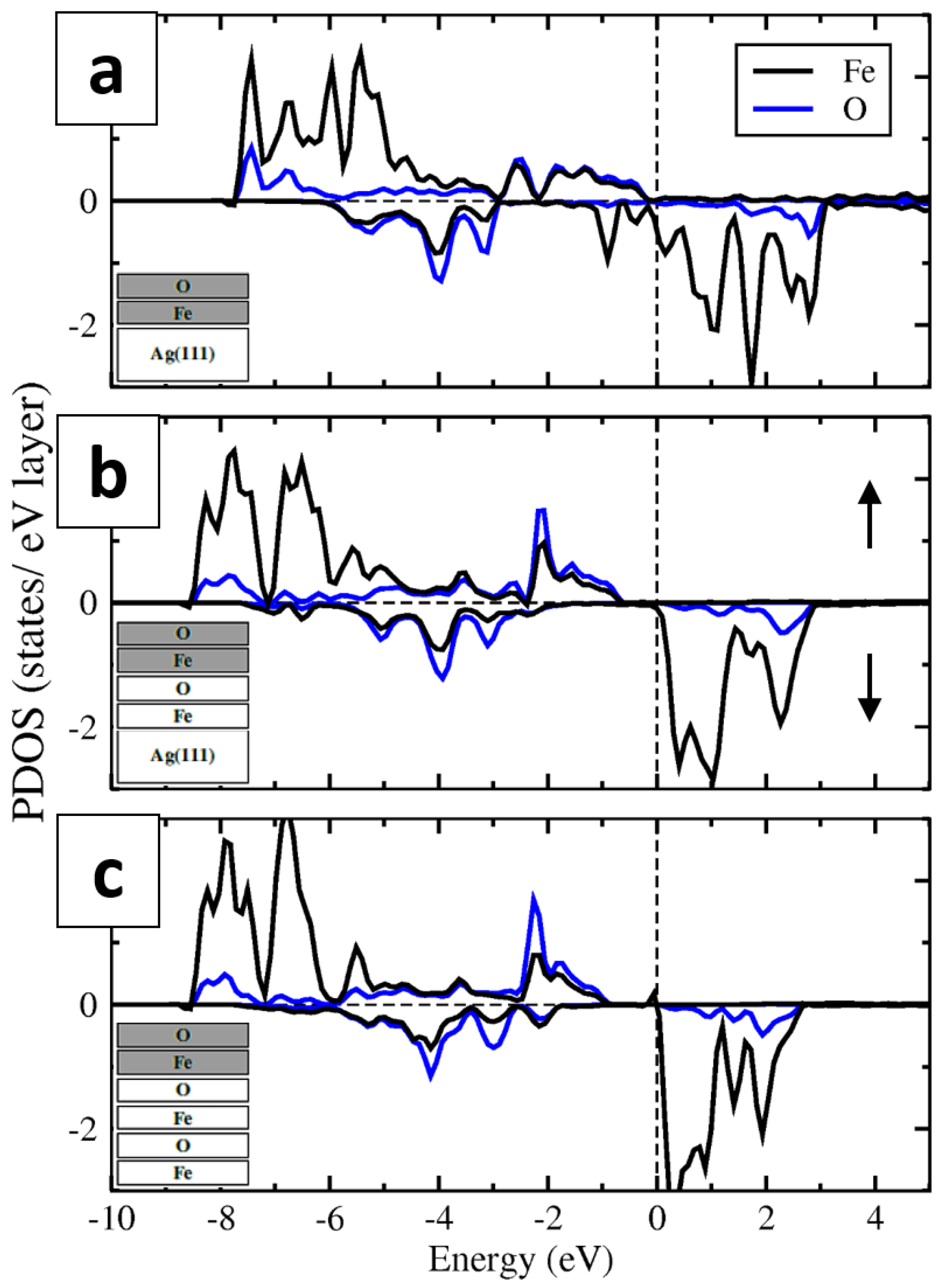
3.3. Calculations on a Conceptual Reconstruction-Free FeO/Ag(111)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pacchioni, G. Two-Dimensional Oxides: Multifunctional Materials for Advanced Technologies. Chem. Eur. J. 2012, 18, 10144–10158. [Google Scholar] [CrossRef] [PubMed]
- Peden, C.H.F.; Herman, G.S.; Ismagilov, I.Z.; Kay, B.D.; Henderson, M.A.; Kim, Y.-J.; Chambers, S.A. Model catalyst studies with single crystals and epitaxial thin oxide films. Catal. Today 1999, 51, 513–519. [Google Scholar] [CrossRef]
- Vurens, G.H.; Salmeron, M.; Samorjai, G.A. Structure, composition and chemisorption studies of thin ordered iron oxide films on platinum (111). Surf. Sci. 1988, 201, 129–144. [Google Scholar] [CrossRef]
- Weiss, W.; Ranke, W. Surface chemistry and catalysis on well-defined epitaxial iron-oxide layers. Progr. Surf. Sci. 2002, 70, 1–151. [Google Scholar] [CrossRef]
- Zeuthen, H.; Kudernatsch, W.; Peng, G.; Merte, L.R.; Ono, L.K.; Lammich, L.; Bai, Y.; Grabow, L.C.; Mavrikakis, M.; Wendt, S.; et al. Structure of Stoichiometric and Oxygen-Rich Ultrathin FeO(111) Films Grown on Pd(111). J. Phys. Chem. C 2013, 117, 15155–15163. [Google Scholar] [CrossRef]
- Ketteler, G.; Ranke, W. Heteroepitaxial Growth and Nucleation of Iron Oxide Films on Ru(0001). J. Phys. Chem. B 2003, 107, 4320–4333. [Google Scholar] [CrossRef]
- Khan, N.A.; Matranga, C. Nucleation and growth of Fe and FeO nanoparticles and films on Au(111). Surf. Sci. 2008, 602, 932–942. [Google Scholar] [CrossRef]
- Waddill, G.D.; Ozturk, O. Epitaxial growth of iron oxide films on Ag(111). Surf. Sci. 2005, 575, 35–50. [Google Scholar] [CrossRef]
- Lopes, E.L.; Abreu, G.J.P.; Paniago, R.; Soares, E.A.; de Carvalho, V.E.; Pfannes, H.-D. Atomic geometry determination of FeO(001) grown on Ag(001) by low energy electron diffraction. Surf. Sci. 2007, 601, 1239–1245. [Google Scholar] [CrossRef]
- Schlueter, C.; Lübbe, M.; Gigler, A.M.; Moritz, W. Growth of iron oxides on Ag(111)—Reversible Fe2O3/Fe3O4 transformation. Surf. Sci. 2011, 605, 1986–1993. [Google Scholar] [CrossRef]
- Abreu, G.J.P.; Paniago, R.; Pfannes, H.-D. Growth of ultra-thin FeO(100) films on Ag(100): A combined XPS, LEED and CEMS study. J. Magn. Magn. Mater. 2014, 349, 235–239. [Google Scholar] [CrossRef]
- Bruns, D.; Kiesel, I.; Jentsch, S.; Lindemann, S.; Otte, C.; Schemme, T.; Kuschel, T.; Wollschläger, J. Structural analysis of FeO(111)/Ag(001): Undulation of hexagonal oxide monolayers due to square lattice metal substrates. J. Phys. Condens. Matter 2014, 26, 315001. [Google Scholar] [CrossRef] [PubMed]
- Genuzio, F.; Sala, A.; Schmidt, Th.; Menzel, D.; Freund, H.-J. Interconversion of α-Fe2O3 and Fe3O4 Thin Films: Mechanisms, Morphology, and Evidence for Unexpected Substrate Participation. J. Phys. Chem. C 2014, 118, 29068–29076. [Google Scholar] [CrossRef]
- Merte, L.R.; Shipilin, M.; Ataran, S.; Blomberg, S.; Zhang, C.; Mikkelsen, A.; Gustafson, J.; Lundgren, E. Growth of Ultrathin Iron Oxide Films on Ag(100). J. Phys. Chem. C 2015, 119, 2572–2582. [Google Scholar] [CrossRef]
- Genuzio, F.; Sala, A.; Schmidt, Th.; Menzel, D.; Freund, H.-J. Phase transformations in thin iron oxide films: Spectromicroscopic study of velocity and shape of the reaction fronts. Surf. Sci. 2016, 648, 177–187. [Google Scholar] [CrossRef]
- Lamirand, A.D.; Grenier, S.; Langlais, V.; Ramos, A.Y.; Tolentino, H.C.N.; Torelles, X.; De Santis, M. Magnetite epitaxial growth on Ag(001): Selected orientation, seed layer, and interface sharpness. Surf. Sci. 2016, 647, 33–38. [Google Scholar] [CrossRef]
- Mehar, V.; Merte, L.R.; Choi, J.; Shipilin, M.; Lundgren, E.; Weaver, J.F. Adsorption of NO on FeOx Films Grown on Ag(111). J. Phys. Chem. C 2016, 120, 9282–9291. [Google Scholar] [CrossRef]
- Merte, L.R.; Heard, C.J.; Zhang, F.; Choi, J.; Shipilin, M.; Gustafson, J.; Weaver, J.F.; Grönbeck, H.; Lundgren, E. Tuning the Reactivity of Ultrathin Oxides: NO Adsorption on Monolayer FeO(111). Angew. Chem. Int. Ed. 2016, 55, 9267–9271. [Google Scholar] [CrossRef] [PubMed]
- Shipilin, M.; Lundgren, E.; Gustafson, J.; Zhang, C.; Bertram, F.; Nicklin, C.; Heard, C.J.; Grönbeck, H.; Zhang, F.; Choi, J.; et al. Fe Oxides on Ag Surfaces: Structure and Reactivity. Top. Catal. 2017, 60, 492–502. [Google Scholar] [CrossRef]
- Gwyddion. Available online: http://gwyddion.net (accessed on 9 October 2018).
- Horcas, I.; Fernandez, R.; Gomez-Rodriguez, J.M.; Colchero, J.; Gomez-Herrero, J.; Baro, A.M. WSXM: A software for scanning probe microscopy and a tool for nanotechnology. Rev. Sci. Instrum. 2007, 78, 013705. [Google Scholar] [CrossRef] [PubMed]
- Hedman, J.; Klasson, M.; Nilsson, R.; Nordling, C.; Sorokina, M.F.; Kljushnikov, O.I.; Nemnonov, S.A.; Trapeznikov, V.A.; Zyryanov, V.G. The Electronic Structure of Some Palladium Alloys Studied by ESCA and X-ray Spectroscopy. Phys. Scr. 1971, 4, 195–201. [Google Scholar] [CrossRef]
- Kresse, G.; Hafner, J. Ab initio molecular dynamics for open-shell transition metals. Phys. Rev. B 1993, 48, 13115–13118. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169–11186. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1996, 6, 15–50. [Google Scholar] [CrossRef]
- Blöchl, P.E. Projector augmented-wave method. Phys. Rev. B 1994, 50, 17953–17979. [Google Scholar] [CrossRef]
- Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 1999, 59, 1758–1775. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [PubMed]
- Methfessel, M.; Paxton, A.T. High-precision sampling for Brillouin-zone integration in metals. Phys. Rev. B 1989, 40, 3616–3621. [Google Scholar] [CrossRef]
- Dudarev, S.L.; Botton, G.A.; Savrasov, S.Y.; Humphreys, C.J.; Sutton, A.P. Electron-energy-loss spectra and the structural stability of nickel oxide: An LSDA+U study. Phys. Rev. B 1998, 57, 1505–1509. [Google Scholar] [CrossRef]
- Giordano, L.; Pacchioni, G.; Goniakowski, J.; Nilius, N.; Rienks, E.D.L.; Freund, H.-J. Interplay between structural, magnetic, and electronic properties in a FeO∕Pt(111) ultrathin film. Phys. Rev. B 2007, 76, 075416. [Google Scholar] [CrossRef]
- Kokalj, A.; Dal Corso, A.; de Gironcoli, S.; Baroni, S. Adsorption of ethylene on the Ag(001) surface. Surf. Sci. 2002, 507–510, 62–68. [Google Scholar] [CrossRef]
- Müller, M.; Diller, K.; Maurer, R.J.; Reuter, K. Interfacial charge rearrangement and intermolecular interactions: Density-functional theory study of free-base porphine adsorbed on Ag(111) and Cu(111). J. Chem. Phys. 2016, 144, 024701. [Google Scholar] [CrossRef] [PubMed]
- Lide, D.R. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA; London, UK; New York, NY, USA, 2003. [Google Scholar]
- Neugebauer, J.; Scheffler, M. Adsorbate-substrate and adsorbate-adsorbate interactions of Na and K adlayers on Al(111). Phys. Rev. B 1992, 46, 16067–16080. [Google Scholar] [CrossRef]
- Chelvayohan, M.; Mee, C.H.B. Work function measurements on (110), (100) and (111) surfaces of silver. J. Phys. C Solid State Phys. 1982, 15, 2305–2312. [Google Scholar] [CrossRef]
- Otálvaro, D.; Veening, T.; Brocks, G. Self-Assembled Monolayer Induced Au(111) and Ag(111) Reconstructions: Work Functions and Interface Dipole Formation. J. Phys. Chem. C 2012, 116, 7826–7837. [Google Scholar] [CrossRef]
- Ossowski, T. Private communication, 2016.
- Bader, R.F.W. Atoms in Molecules: A Quantum Theory; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Henkelman, G.; Arnaldsson, A.; Jónsson, H. A fast and robust algorithm for Bader decomposition of charge density. Comput. Mater. Sci. 2006, 36, 354–360. [Google Scholar] [CrossRef]
- Tersoff, T.; Hamann, D.R. Theory of the scanning tunneling microscope. Phys. Rev. B 1985, 31, 805–813. [Google Scholar] [CrossRef]
- Buchner, F.; Kellner, I.; Steinrück, H.-P.; Marbach, H. Modification of the Growth of Iron on Ag(111) by Predeposited Organic Monolayers. Z. Phys. Chem. 2009, 223, 131–144. [Google Scholar] [CrossRef]
- Bocquet, F.; Maurel, C.; Roussel, J.-M.; Abel, M.; Koudia, M.; Porte, L. Segregation-mediated capping of Volmer-Weber Cu islands grown onto Ag(111). Phys. Rev. B 2005, 71, 075405. [Google Scholar] [CrossRef]
- Jeong, H.-C.; Williams, E.D. Steps on surfaces: Experiment and theory. Surf. Sci. Rep. 1999, 34, 171–294. [Google Scholar] [CrossRef]
- Graat, P.C.J.; Somers, M.A.J. Simultaneous determination of composition and thickness of thin iron-oxide films from XPS Fe 2p spectra. Appl. Surf. Sci. 1996, 100/101, 36–40. [Google Scholar] [CrossRef]
- Yamashita, T.; Hayes, P. Analysis of XPS spectra of Fe and Fe ions in oxide materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
- García, L.M.; Bernal-Villamil, I.; Oujja, M.; Carrasco, E.; Gargallo-Caballero, R.; Castillejo, M.; Marco, J.F.; Gallego, S.; de la Figuera, J. Unconventional properties of nanometric FeO(111) films on Ru(0001): Stoichiometry and surface structure. J. Mater. Chem. C 2016, 4, 1850–1859. [Google Scholar] [CrossRef]
- Grosvenor, A.P.; Kobe, B.A.; Biesinger, M.C.; McIntyre, N.S. Investigation of multiplet splitting of Fe 2p XPS spectra and bonding in iron compounds. Surf. Interface Anal. 2004, 36, 1564–1574. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Payne, B.P.; Grosvenor, A.P.; Lau, L.W.M.; Gerson, A.R.; Smart, R.S.C. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Appl. Surf. Sci. 2011, 257, 2717–2730. [Google Scholar] [CrossRef]
- McIntyre, N.S.; Zetaruk, D.G. X-ray photoelectron spectroscopic studies of iron oxides. Anal. Chem. 1977, 49, 1521–1529. [Google Scholar] [CrossRef]
- Ratner, B.D.; Castner, D.G. Surface Analysis—The Principal Techniques, 2nd ed.; Vickerman, J.C., Gilmore, I.S., Eds.; Wiley-VCH: Weinheim, Germany, 2011. [Google Scholar]
- Rubio-Zuazo, L.; Chainani, A.; Taguchi, M.; Malterre, D.; Serrano, A.; Castro, G.R. Electronic structure of FeO, γ-Fe2O3, and Fe3O4 epitaxial films using high-energy spectroscopies. Phys. Rev. B 2018, 97, 235148. [Google Scholar] [CrossRef]
- Scofield, J.H. Hartree-Slater subshell photoionization cross-sections at 1254 and 1487 eV. J. Electr. Spectrosc. Relat. Phenom. 1976, 8, 129–137. [Google Scholar] [CrossRef]
- Davey, W.P. Precision Measurements of the Lattice Constants of Twelve Common Metals. Phys. Rev. 1925, 25, 753–761. [Google Scholar] [CrossRef]
- Longworth, G.; Jain, R. Mossbauer effect study of iron implanted silver alloys as a function of dose and annealing temperature. J. Phys. F Metal Phys. 1978, 8, 993–1007. [Google Scholar] [CrossRef]
- Jaouen, T.; Tricot, S.; Delhaye, G.; Lépine, B.; Sébilleau, D.; Jézéquel, G.; Schieffer, P. Layer-Resolved Study of Mg Atom Incorporation at the MgO/Ag(001) Buried Interface. Phys. Rev. Lett. 2013, 111, 027601. [Google Scholar] [CrossRef] [PubMed]
- Pal, J.; Smerieri, M.; Celasco, E.; Savio, L.; Vattuone, L.; Rocca, M. Morphology of Monolayer MgO Films on Ag(100): Switching from Corrugated Islands to Extended Flat Terraces. Phys. Rev. Lett. 2014, 112, 126102. [Google Scholar] [CrossRef] [PubMed]
- Steurer, W.; Surnev, S.; Fortunelli, A.; Netzer, F.P. Scanning tunneling microscopy imaging of NiO(100)(1 × 1) islands embedded in Ag(100). Surf. Sci. 2012, 606, 803–807. [Google Scholar] [CrossRef]
- Binnig, G.; Rohrer, H.; Gerber, Ch.; Weibel, E. Tunneling through a controllable vacuum gap. Appl. Phys. Lett. 1982, 40, 178–180. [Google Scholar] [CrossRef]
- Schuster, R.; Barth, J.V.; Wintterlin, J.; Behm, R.J.; Ertl, G. Distance dependence and corrugation in barrier-height measurements on metal surfaces. Ultramicroscopy 1992, 42–44, 533–540. [Google Scholar] [CrossRef]
- Yamada, Y.; Sinsarp, A.; Sasaki, M.; Yamamoto, S. Local Tunneling Barrier Height Measurement on Au(111). Jpn. J. Appl. Phys. 2003, 42, 4898–4900. [Google Scholar] [CrossRef]
- Rienks, E.D.L.; Nilius, N.; Rust, H.-P.; Freund, H.-J. Surface potential of a polar oxide film: FeO on Pt(111). Phys. Rev. B 2005, 71, 241404. [Google Scholar] [CrossRef]
- Hulse, J.; Küppers, J.; Wandelt, K.; Ertle, G. UV-photoelectron spectroscopy from xenon adsorbed on heterogeneous metal surfaces. Appl. Surf. Sci. 1980, 6, 453–463. [Google Scholar] [CrossRef]
- Salmeron, M.; Ferrer, S.; Jazzar, M.; Samorjai, G.A. Photoelectron-spectroscopy study of the electronic structure of Au and Ag overlayers on Pt(100), Pt(111), and Pt(997) surfaces. Phys. Rev. B 1983, 28, 6758–6765. [Google Scholar] [CrossRef]
- Derry, G.N.; Ji-Zhong, Z. Work function of Pt(111). Phys. Rev. B 1989, 39, 1940. [Google Scholar] [CrossRef]
- Kim, Y.J.; Westphal, C.; Ynzunza, R.X.; Galloway, H.C.; Salmeron, M.; van Hove, M.A.; Fadley, C.S. Interlayer interactions in epitaxial oxide growth: FeO on Pt(111). Phys. Rev. B 1997, 55, R13448–R13451. [Google Scholar] [CrossRef]
- Noguera, C. Polar oxide surfaces. J. Phys. Condens. Matter 2000, 12, R367–R410. [Google Scholar] [CrossRef]
- Rienks, E.D.L.; Nilius, N.; Giordano, L.; Goniakowski, J.; Pacchioni, G.; Felicissimo, M.P.; Risse, Th.; Rust, H.-P.; Freund, H.-J. Local zero-bias anomaly in tunneling spectra of a transition-metal oxide thin film. Phys. Rev. B 2007, 75, 205443. [Google Scholar] [CrossRef]
- Goniakowski, J.; Finocchi, F.; Noguera, C. Polarity of oxide surfaces and nanostructures. Rep. Prog. Phys. 2008, 71, 016501. [Google Scholar] [CrossRef]
- Goniakowski, J.; Noguera, C. Polarization and rumpling in oxide monolayers deposited on metallic substrates. Phys. Rev. B 2009, 79, 155433. [Google Scholar] [CrossRef]
- McCammon, C.A. Magnetic properties of FexO (x > 0.95): Variation of Néel temperature. J. Magn. Magn. Mater. 1992, 104–107, 1937–1938. [Google Scholar] [CrossRef]
- Lu, S.-Z.; Qin, Z.-H.; Cao, G.-Y. Nanostructured double-layer FeO as nanotemplate for tuning adsorption of titanyl phthalocyanine molecules. Appl. Phys. Lett. 2014, 104, 253104. [Google Scholar]
- Lu, S.-Z.; Qin, Z.-H.; Guo, Q.-M.; Cao, G.-Y. Work function mediated by deposition of ultrathin polar FeO on Pt(111). Appl. Surf. Sci. 2017, 392, 849–853. [Google Scholar] [CrossRef]
- The experimental results were partially presented by V. Babačić, Y. Wang and M. Lewandowski at the DPG Spring Meeting 2018, Berlin, Germany, 11–16 March 2018, the NanoTech Poland 2018 Conference, Poznań, Poland, 6–9 June 2018, the AMPERE NMR School 2018, Zakopane, Poland, 10–16 June 2018, 34th European Conference on Surface Science, Aarhus, Denmark, 26–31 August 2018 and the 3rd International Meeting on Materials Science for Energy Related Applications—3IMMSERA, Belgrade, Serbia, 25–26 September 2018.
- The theoretical results were partially presented by T. Pabisiak and A. Kiejna at the 8th International Workshop on Surface Physics, Trzebnica, Poland, 26–30 June 2017.


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewandowski, M.; Pabisiak, T.; Michalak, N.; Miłosz, Z.; Babačić, V.; Wang, Y.; Hermanowicz, M.; Palotás, K.; Jurga, S.; Kiejna, A. On the Structure of Ultrathin FeO Films on Ag(111). Nanomaterials 2018, 8, 828. https://doi.org/10.3390/nano8100828
Lewandowski M, Pabisiak T, Michalak N, Miłosz Z, Babačić V, Wang Y, Hermanowicz M, Palotás K, Jurga S, Kiejna A. On the Structure of Ultrathin FeO Films on Ag(111). Nanomaterials. 2018; 8(10):828. https://doi.org/10.3390/nano8100828
Chicago/Turabian StyleLewandowski, Mikołaj, Tomasz Pabisiak, Natalia Michalak, Zygmunt Miłosz, Višnja Babačić, Ying Wang, Michał Hermanowicz, Krisztián Palotás, Stefan Jurga, and Adam Kiejna. 2018. "On the Structure of Ultrathin FeO Films on Ag(111)" Nanomaterials 8, no. 10: 828. https://doi.org/10.3390/nano8100828
APA StyleLewandowski, M., Pabisiak, T., Michalak, N., Miłosz, Z., Babačić, V., Wang, Y., Hermanowicz, M., Palotás, K., Jurga, S., & Kiejna, A. (2018). On the Structure of Ultrathin FeO Films on Ag(111). Nanomaterials, 8(10), 828. https://doi.org/10.3390/nano8100828





