Nanoemulsion-Enabled Oral Delivery of Novel Anticancer ω-3 Fatty Acid Derivatives
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
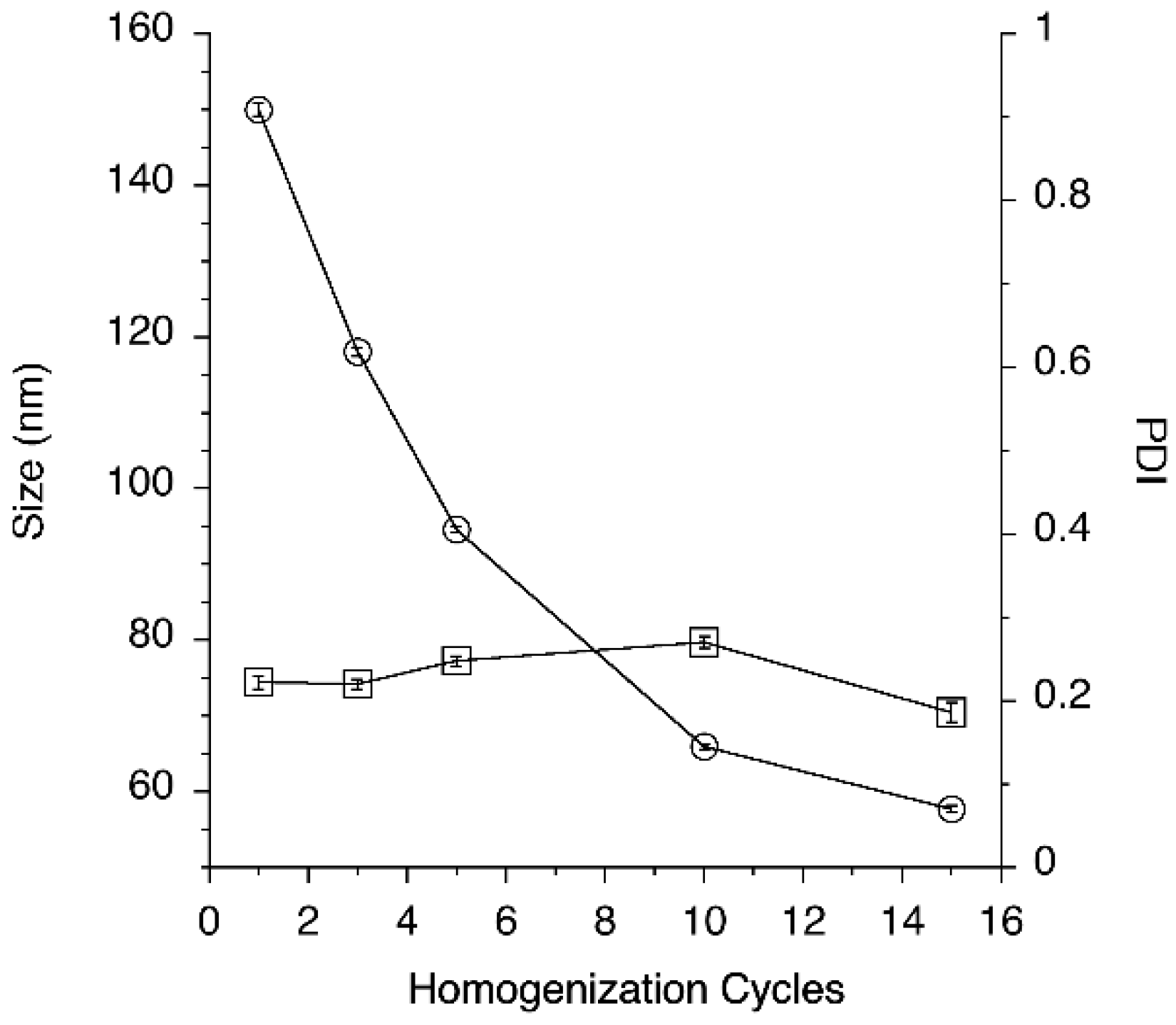
2.2.1. Study of the Impact of Process Parameters on Nanoemulsion Preparation
2.2.2. Preparation of ClFPh-CHA-Loaded Nanoemulsion Preparation
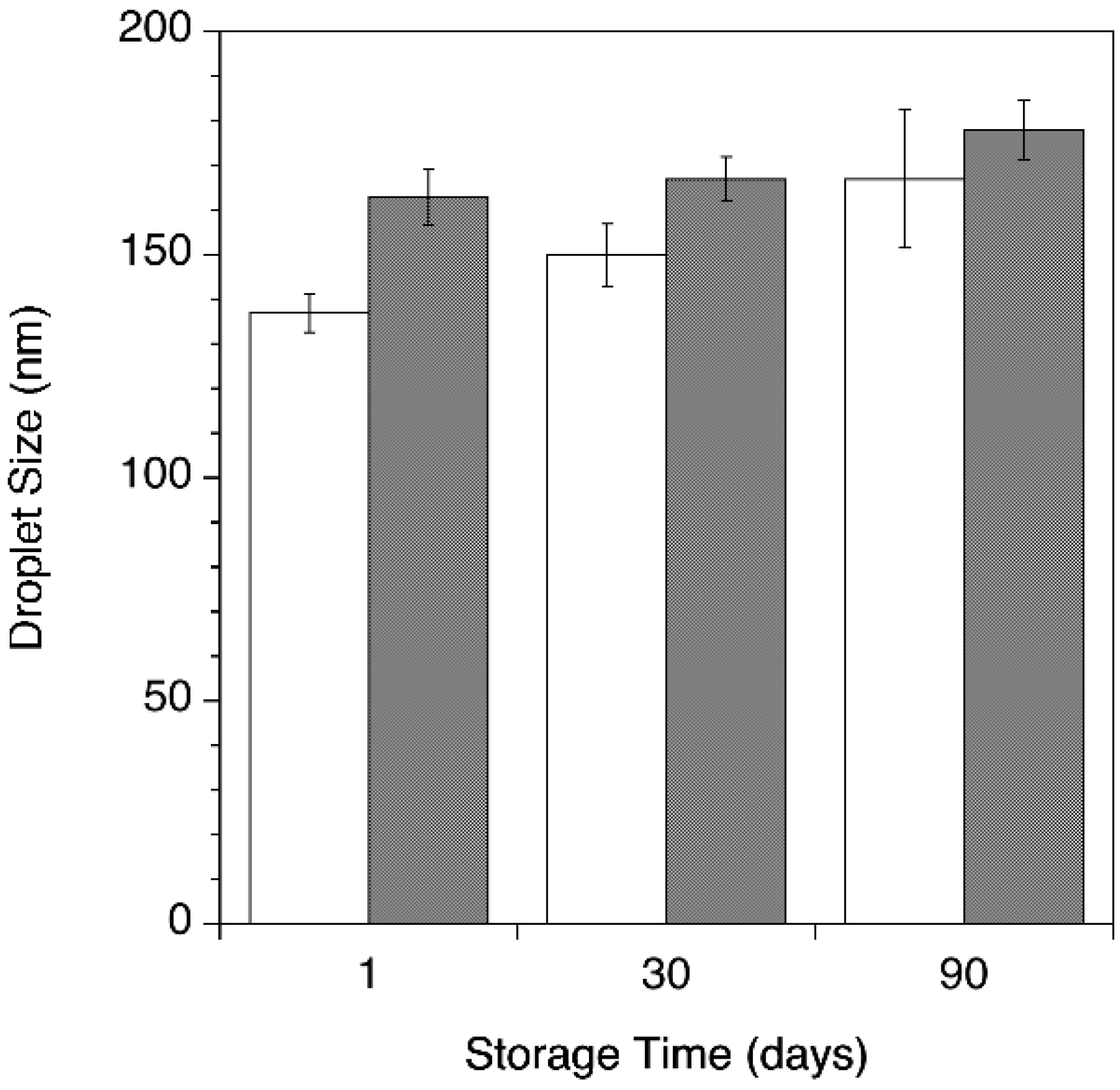
2.2.3. Nanoemulsion Characterization
2.2.4. HPLC/MS Analytical Method for ClFPh-CHA Quantification
2.2.5. In Vivo Studies
2.2.6. Statistics
3. Results
3.1. Optimization of Nanoemulsion Production Process
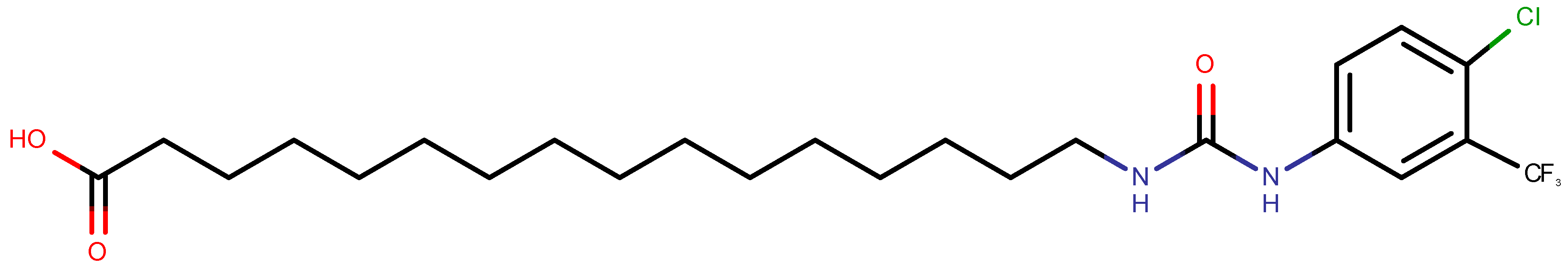
3.2. ClFPh-CHA-Loaded Nanoemulsion
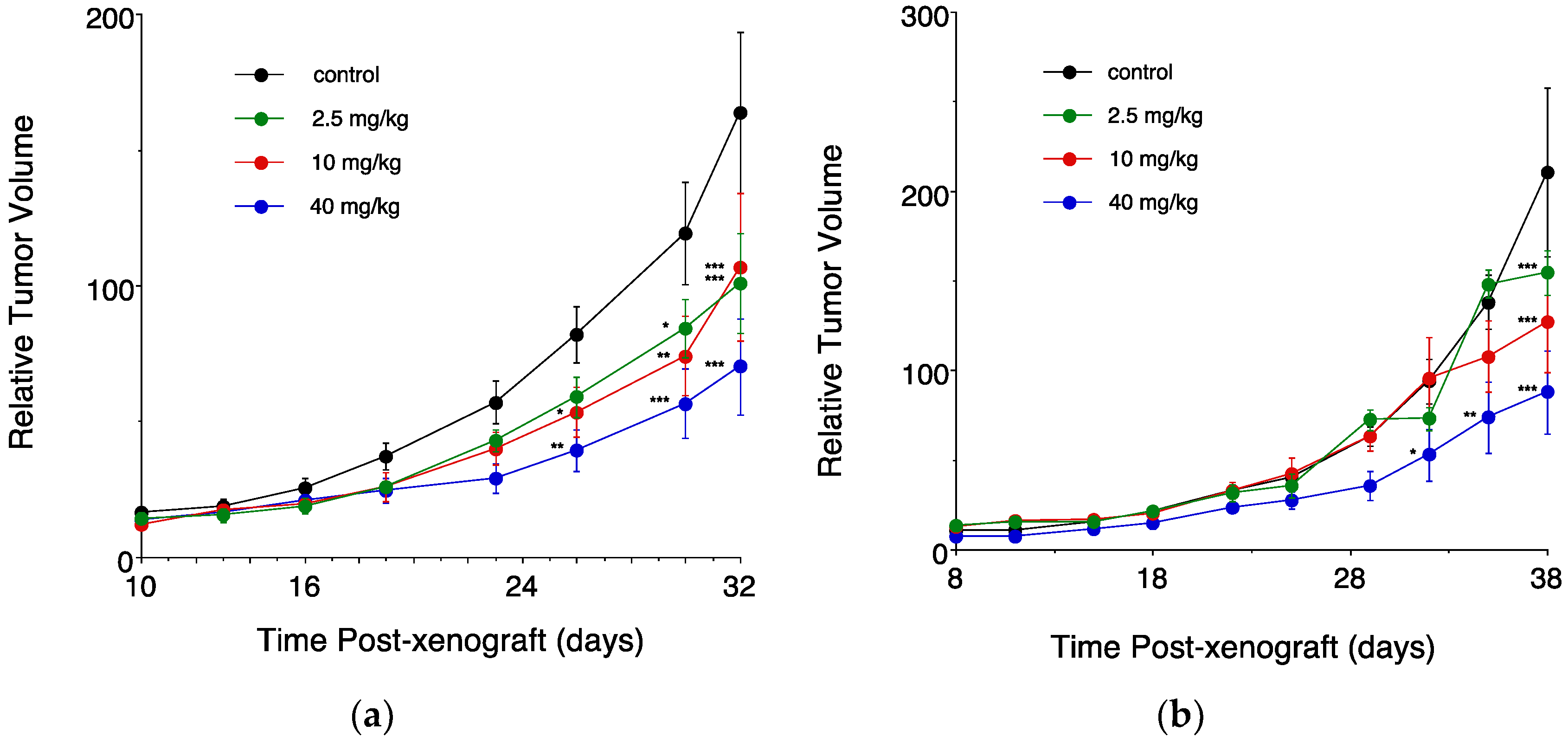
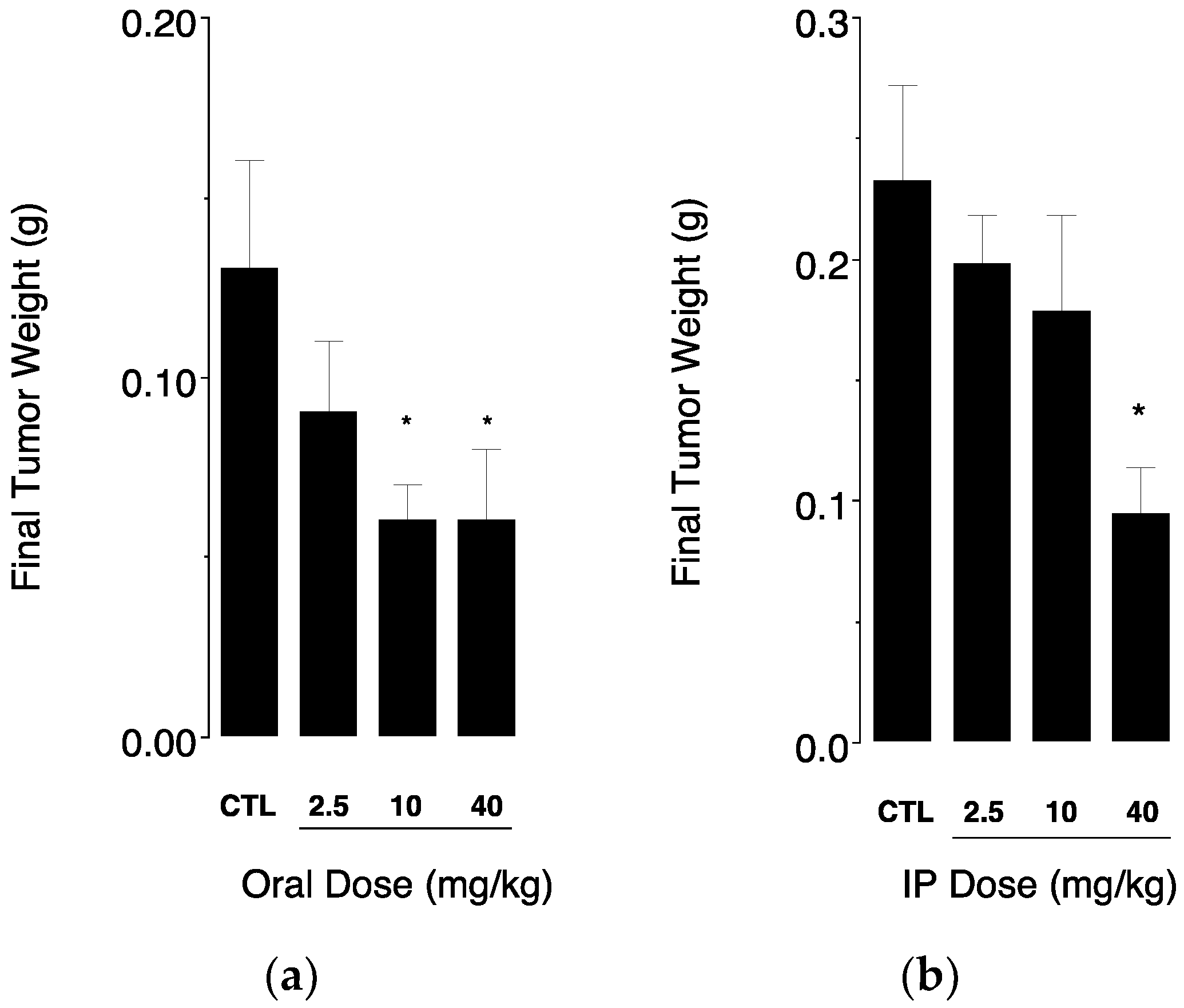
3.3. In Vivo Sudies in Mice Bearing an Intramammary Tumor Xenograft
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Foulkes, W.D.; Smith, I.E.; Reis-Filho, J.S. Triple-negative breast cancer. N. Engl. J. Med. 2010, 363, 1938–1948. [Google Scholar] [CrossRef] [PubMed]
- Calaf, G.M.; Urzua, U.; Termini, L.; Aguayo, F. Oxidative stress in female cancers. Oncotarget 2018, 9, 23824–23842. [Google Scholar] [CrossRef] [PubMed]
- Klemp, J.R. Breast cancer prevention across the cancer care continuum. Semin. Oncol. Nurs. 2015, 31, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.K.; Lee, S.B.; Won, J.; Choi, H.Y.; Kim, K.; Yang, G.-M.; Dayem, A.A.; Cho, S.-G. Correlation between oxidative stress, nutrition, and cancer initiation. Int. J. Mol. Sci. 2017, 18, 1544. [Google Scholar] [CrossRef] [PubMed]
- Berquin, I.M.; Edwards, I.J.; Chen, Y.Q. Multi-targeted therapy of cancer by omega-3 fatty acids. Cancer Lett. 2008, 269, 363–377. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, N.R.; Kim, E.; Fan, Y.-Y.; Chapkin, R.S. Omega-3 fatty acids, membrane remodeling and cancer prevention. Mol. Aspects Med. 2018, in press. [Google Scholar] [CrossRef] [PubMed]
- Cui, P.H.; Rawling, T.; Bourget, K.; Kim, T.; Duke, C.C.; Doddareddy, M.R.; Hibbs, D.E.; Zhou, F.; Tattam, B.N.; Petrovic, N.; et al. Antiproliferative and Antimigratory Actions of Synthetic Long Chain n-3 Monounsaturated Fatty Acids in Breast Cancer Cells That Overexpress Cyclooxygenase-2. J. Med. Chem. 2012, 55, 7163–7172. [Google Scholar] [CrossRef] [PubMed]
- Dyari, H.R.E.; Rawling, T.; Bourget, K.; Murray, M. Synthetic ω-3 Epoxyfatty Acids As Antiproliferative and Pro-apoptotic Agents in Human Breast Cancer Cells. J. Med. Chem. 2014, 57, 7459–7464. [Google Scholar] [CrossRef] [PubMed]
- Dyari, H.R.E.; Rawling, T.; Chen, Y.; Sudarmana, W.; Bourget, K.; Dwyer, J.M.; Allison, S.E.; Murray, M. A novel synthetic analogue of v-3 17,18-epoxyeicosatetraenoic acid activates TNF receptor-1/ASK1/JNK signaling to promote apoptosis in human breast cancer cells. FASEB J. 2017, 31, 5246–5257. [Google Scholar] [CrossRef] [PubMed]
- Rawling, T.; Choucair, H.; Koolaji, N.; Bourget, K.; Allison, S.E.; Chen, Y.-J.; Dunstan, C.R.; Murray, M. A Novel Arylurea Fatty Acid That Targets the Mitochondrion and Depletes Cardiolipin to Promote Killing of Breast Cancer Cells. J. Med. Chem. 2017, 60, 8661–8666. [Google Scholar] [CrossRef] [PubMed]
- Kotta, S.; Khan, A.W.; Pramod, K.; Ansari, S.H.; Sharma, R.K.; Ali, J. Exploring oral nanoemulsions for bioavailability enhancement of poorly water-soluble drugs. Expert Opin. Drug Deliv. 2012, 9, 585–598. [Google Scholar] [CrossRef] [PubMed]
- Čerpnjak, K.; Zvonar, A.; Gasperlin, M.; Vrečer, F. Lipid-based systems as a promising approach for enhancing the bioavailability of poorly water-soluble drugs. Acta Pharm. 2013, 63, 427–445. [Google Scholar] [CrossRef] [PubMed]
- Comfort, C.; Garrastazu, G.; Pozzoli, M.; Sonvico, F. Opportunities and Challenges for the Nasal Administration of Nanoemulsions. Curr. Top. Med. Chem. 2015, 15, 356–368. [Google Scholar] [CrossRef] [PubMed]
- Solans, C.; Izquierdo, P.; Nolla, J.; Azemar, N.; Garcia-Celma, M.J. Nano-emulsions. Curr. Opin. Colloid Interface Sci. 2005, 10, 102–110. [Google Scholar] [CrossRef]
- Raphael, A.P.; Garrastazu, G.; Sonvico, F.; Prow, T.W. Formulation design for topical drug and nanoparticle treatment of skin disease. Ther. Deliv. 2015, 6, 197–216. [Google Scholar] [CrossRef] [PubMed]
- Telò, I.; Favero, E.D.; Cantù, L.; Frattini, N.; Pescina, S.; Padula, C.; Santi, P.; Sonvico, F.; Nicoli, S. Gel-like TPGS-Based Microemulsions for Imiquimod Dermal Delivery: Role of Mesostructure on the Uptake and Distribution into the Skin. Mol. Pharm. 2017, 14, 3281–3289. [Google Scholar] [CrossRef] [PubMed]
- Singh, Y.; Meher, J.G.; Raval, K.; Khan, F.A.; Chaurasia, M.; Jain, N.K.; Chourasia, M.K. Nanoemulsion: Concepts, development and applications in drug delivery. J. Control. Release 2017, 252, 28–49. [Google Scholar] [CrossRef] [PubMed]
- Salager, J.-L.; Forgiarini, A.; Márquez, L.; Peña, A.; Pizzino, A.; Rodriguez, M.P.; Rondón-González, M. Using emulsion inversion in industrial processes. Adv. Colloid Interface Sci. 2004, 108–109, 259–272. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Marshall-Breton, C.; Leser, M.E.; Sher, A.A.; McClements, D.J. Fabrication of ultrafine edible emulsions: Comparison of high-energy and low-energy homogenization methods. Food Hydrocolloids 2012, 29, 398–406. [Google Scholar] [CrossRef]
- Faustino-Rocha, A.; Oliveira, P.A.; Pinho-Oliveira, J.; Teixeira-Guedes, C.; Soares-Maia, R.; da Costa, R.G.; Colaço, B.; Pires, M.J.; Colaço, J.; Ferreira, R.; et al. Estimation of rat mammary tumor volume using caliper and ultrasonography measurements. Lab. Anim. (NY) 2013, 42, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, P.; Feng, J.; Esquena, J.; Tadros, T.F.; Dederen, J.C.; Garcia, M.J.; Azemar, N.; Solans, C. The influence of surfactant mixing ratio on nano-emulsion formation by the pit method. J. Colloid Interface Sci. 2005, 285, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Montenegro, L.; Carbone, C.; Puglisi, G. Vehicle effects on in vitro release and skin permeation of octylmethoxycinnamate from microemulsions. Int. J. Pharm. 2011, 405, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Sajjadi, S. Effect of mixing protocol on formation of fine emulsions. Chem. Eng. Sci. 2006, 61, 3009–3017. [Google Scholar] [CrossRef]
- Anton, N.; Vandamme, T.F. Nano-emulsions and micro-emulsions: Clarifications of the critical differences. Pharm. Res. 2011, 28, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Scott, K.F.; Graham, G.G.; Bryant, K.J. Secreted phospholipase A2 enzymes as therapeutic targets. Expert Opin. Ther. Targets 2003, 7, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Gazzerro, P.; Proto, M.C.; Gangemi, G.; Malfitano, A.M.; Ciaglia, E.; Pisanti, S.; Santoro, A.; Laezza, C.; Bifulco, M. Pharmacological actions of statins: A critical appraisal in the management of cancer. Pharmacol. Rev. 2012, 64, 102–146. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Hraiki, A.; Bebawy, M.; Pazderka, C.; Rawling, T. Anti-tumor activities of lipids and lipid analogues and their development as potential anticancer drugs. Pharmacol. Ther. 2015, 150, 109–128. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, S.; Bouchemal, K.; Ponchel, G. Oral delivery of anticancer drugs I: General considerations. Drug Discov. Today 2013, 18, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Thanki, K.; Gangwal, R.P.; Sangamwar, A.T.; Jain, S. Oral delivery of anticancer drugs: Challenges and opportunities. J. Control. Release 2013, 170, 15–40. [Google Scholar] [CrossRef] [PubMed]
- Pouton, C.W. Formulation of poorly water-soluble drugs for oral administration: Physicochemical and physiological issues and the lipid formulation classification system. Eur. J. Pharm. Sci. 2006, 29, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Stegemann, S.; Leveiller, F.; Franchi, D.; de Jong, H.; Lindén, H. When poor solubility becomes an issue: From early stage to proof of concept. Eur. J. Pharm. Sci. 2007, 31, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Mazzaferro, S.; Bouchemal, K.; Ponchel, G. Oral delivery of anticancer drugs III: Formulation using drug delivery systems. Drug Discov. Today 2013, 18, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Guan, P.; Lu, Y.; Qi, J.; Niu, M.; Lian, R.; Hu, F.; Wu, W. Enhanced oral bioavailability of cyclosporine A by liposomes containing a bile salt. Int. J. Nanomed. 2011, 6, 965–974. [Google Scholar]
- Harde, H.; Das, M.; Jain, S. Solid lipid nanoparticles: An oral bioavailability enhancer vehicle. Expert Opin. Drug Deliv. 2011, 8, 1407–1424. [Google Scholar] [CrossRef] [PubMed]
- Dening, T.J.; Rao, S.; Thomas, N.; Prestidge, C.A. Silica encapsulated lipid-based drug delivery systems for reducing the fed/fasted variations of ziprasidone in vitro. Eur. J. Pharm. Biopharm. 2016, 101, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, S.; Buttini, F.; Rossi, A.; Bettini, R.; Colombo, P.; Ponchel, G.; Sonvico, F.; Colombo, G. Ex vivo permeation of tamoxifen and its 4-OH metabolite through rat intestine from lecithin/chitosan nanoparticles. Int. J. Pharm. 2015, 491, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Shukla, D.; Mishra, B.; Singh, S. Lipid—An emerging platform for oral delivery of drugs with poor bioavailability. Eur. J. Pharm. Biopharm. 2009, 73, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Qi, J.; Wu, W. Absorption, Disposition and Pharmacokinetics of Nanoemulsions. Curr. Drug Metab. 2012, 13, 396–417. [Google Scholar] [CrossRef] [PubMed]
- Trevaskis, N.L.; Charman, W.N.; Porter, C.J.H. Lipid-based delivery systems and intestinal lymphatic drug transport: A mechanistic update. Adv. Drug Deliv. Rev. 2008, 60, 702–716. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Yang, Q.; Bagby, T.R.; Forrest, M.L. Lymphatic drug delivery using engineered liposomes and solid lipid nanoparticles. Adv. Drug Deliv. Rev. 2011, 63, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Dees, E.C.; Wang, A.Z. Nanoparticle-Delivered Chemotherapy: Old Drugs in New Packages. Oncology (Huntingt) 2017, 31, 198–208. [Google Scholar]
| Time | 1 Day | 30 Days | 60 Days | 90 Days | ||||
|---|---|---|---|---|---|---|---|---|
| T (°C) | Size (nm) | PDI | Size (nm) | PDI | Size (nm) | PDI | Size (nm) | PDI |
| 25 | Creaming | - | - | - | - | - | - | |
| 45 | 101.5 ± 1.1 | 0.17 ± 0.02 | 125.9 ± 2.4 | 0.20 ± 0.00 | 113.8 ± 1.2 | 0.23 ± 0.01 | 104.5 ± 0.4 | 0.17 ± 0.01 |
| 70 | 133.1 ± 8.0 | 0.29 ± 0.04 | 135.4 ± 8.3 | 0.29 ± 0.01 | 243.4 ± 29.7 | 0.28 ± 0.08 | 554.5 ± 11.2 | 0.44 ± 0.01 |
| 85 | 137.1 ± 3.0 | 0.16 ± 0.01 | 144.6 ± 3.1 | 0.16 ± 0.01 | 136.9 ± 0.8 | 0.13 ± 0.02 | 150.5 ± 0.3 | 0.24 ± 0.01 |
| Time | 1 Day | 30 Days | 60 Days | 90 Days | ||||
|---|---|---|---|---|---|---|---|---|
| Surf. (%) 1 | Size (nm) | PDI | Size (nm) | PDI | Size (nm) | PDI | Size (nm) | PDI |
| 8 | 162.4 ± 7.7 | 0.28 ± 0.02 | Coalescence | - | - | - | - | |
| 9 | 150.0 ± 1.1 | 0.20 ± 0.02 | 156.5 ± 3.2 | 0.15 ± 0.02 | 164.4 ± 3.2 | 0.25 ± 0.04 | 151.1 ± 1.8 | 0.19 ± 0.01 |
| 10 | 137.1 ± 3.0 | 0.16 ± 0.01 | 144.6 ± 3.1 | 0.16 ± 0.01 | 136.9 ± 0.8 | 0.13 ± 0.02 | 150.5 ± 0.3 | 0.24 ± 0.01 |
| 11 | 120.3 ± 2.3 | 0.10 ± 0.03 | 130.7 ± 6.2 | 0.14 ± 0.02 | 125.0 ± 1.3 | 0.13 ± 0.02 | 122.5 ± 1.1 | 0.17 ± 0.01 |
| 12 | 122.3 ± 1.5 | 0.10 ± 0.02 | 129.0 ± 3.3 | 0.12 ± 0.02 | 126.4 ± 1.1 | 0.18 ± 0.01 | 124.9 ± 0.5 | 0.17 ± 0.01 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garrastazu Pereira, G.; Rawling, T.; Pozzoli, M.; Pazderka, C.; Chen, Y.; Dunstan, C.R.; Murray, M.; Sonvico, F. Nanoemulsion-Enabled Oral Delivery of Novel Anticancer ω-3 Fatty Acid Derivatives. Nanomaterials 2018, 8, 825. https://doi.org/10.3390/nano8100825
Garrastazu Pereira G, Rawling T, Pozzoli M, Pazderka C, Chen Y, Dunstan CR, Murray M, Sonvico F. Nanoemulsion-Enabled Oral Delivery of Novel Anticancer ω-3 Fatty Acid Derivatives. Nanomaterials. 2018; 8(10):825. https://doi.org/10.3390/nano8100825
Chicago/Turabian StyleGarrastazu Pereira, Gabriela, Tristan Rawling, Michele Pozzoli, Curtis Pazderka, Yongjuan Chen, Colin R. Dunstan, Michael Murray, and Fabio Sonvico. 2018. "Nanoemulsion-Enabled Oral Delivery of Novel Anticancer ω-3 Fatty Acid Derivatives" Nanomaterials 8, no. 10: 825. https://doi.org/10.3390/nano8100825
APA StyleGarrastazu Pereira, G., Rawling, T., Pozzoli, M., Pazderka, C., Chen, Y., Dunstan, C. R., Murray, M., & Sonvico, F. (2018). Nanoemulsion-Enabled Oral Delivery of Novel Anticancer ω-3 Fatty Acid Derivatives. Nanomaterials, 8(10), 825. https://doi.org/10.3390/nano8100825







