Synthesis, Characterization, and Nanomedical Applications of Conjugates between Resorcinarene-Dendrimers and Ibuprofen
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Resorcinarene with Poliaminoamide Dendritic Arm Dendrimers
2.3. Synthesis of Ibuprofen Conjugates of First and Second Generations
2.4. Characterization of the Conjugates
2.5. Cell Lines
2.6. Hydrolytic Release Studies
2.7. In Vitro Cellular Uptake of the Conjugates of Ibuprofen
2.8. In Vitro Cytotoxicity Studies
3. Results
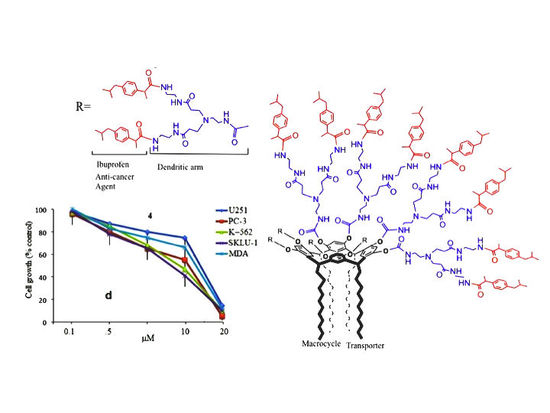
3.1. Synthesis of Ibuprofen–Resorcinarene-Dendrimer Conjugates
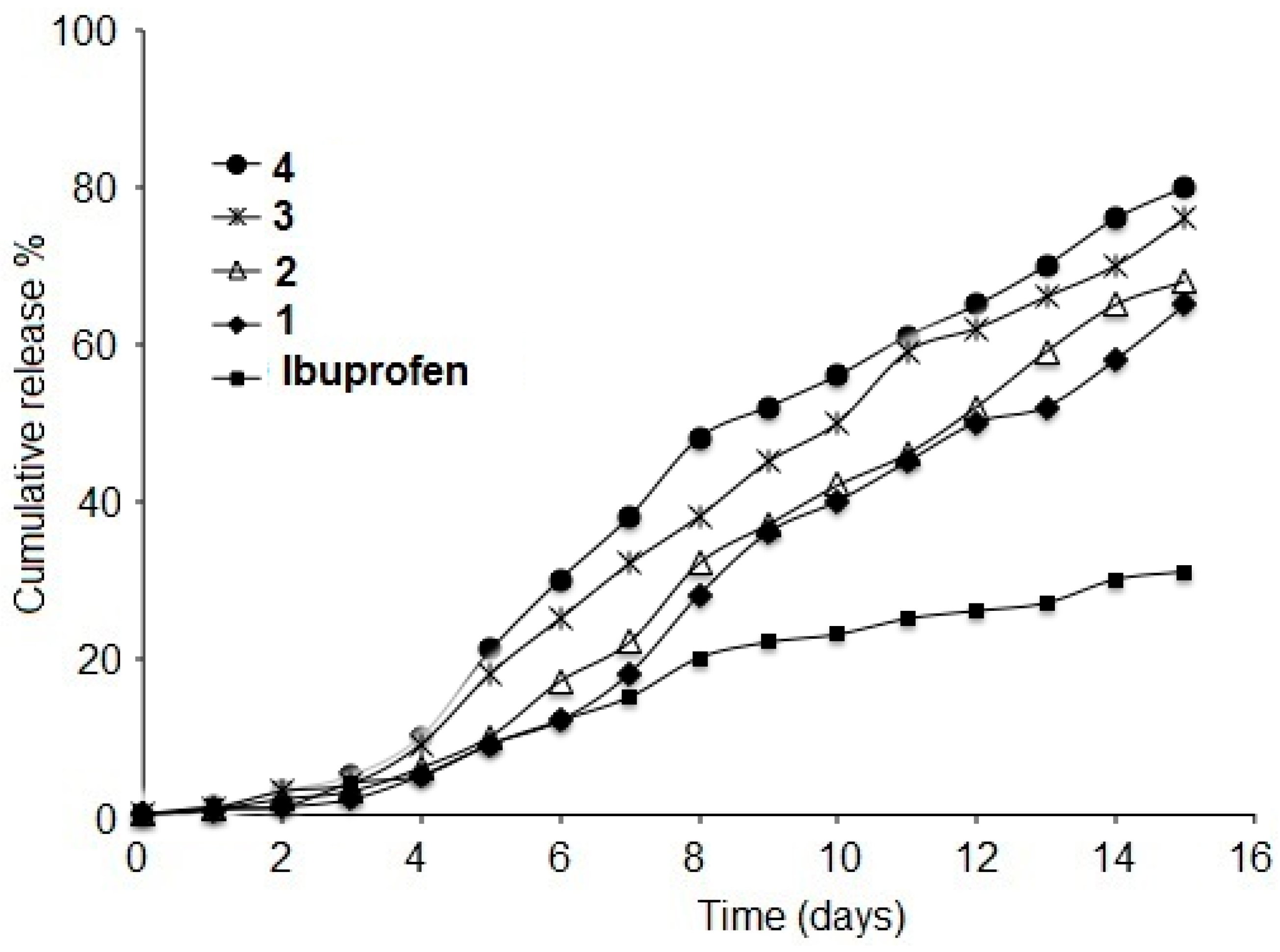
3.2. Hydrolytic Release of Ibuprofen and Ibuprofen Conjugates
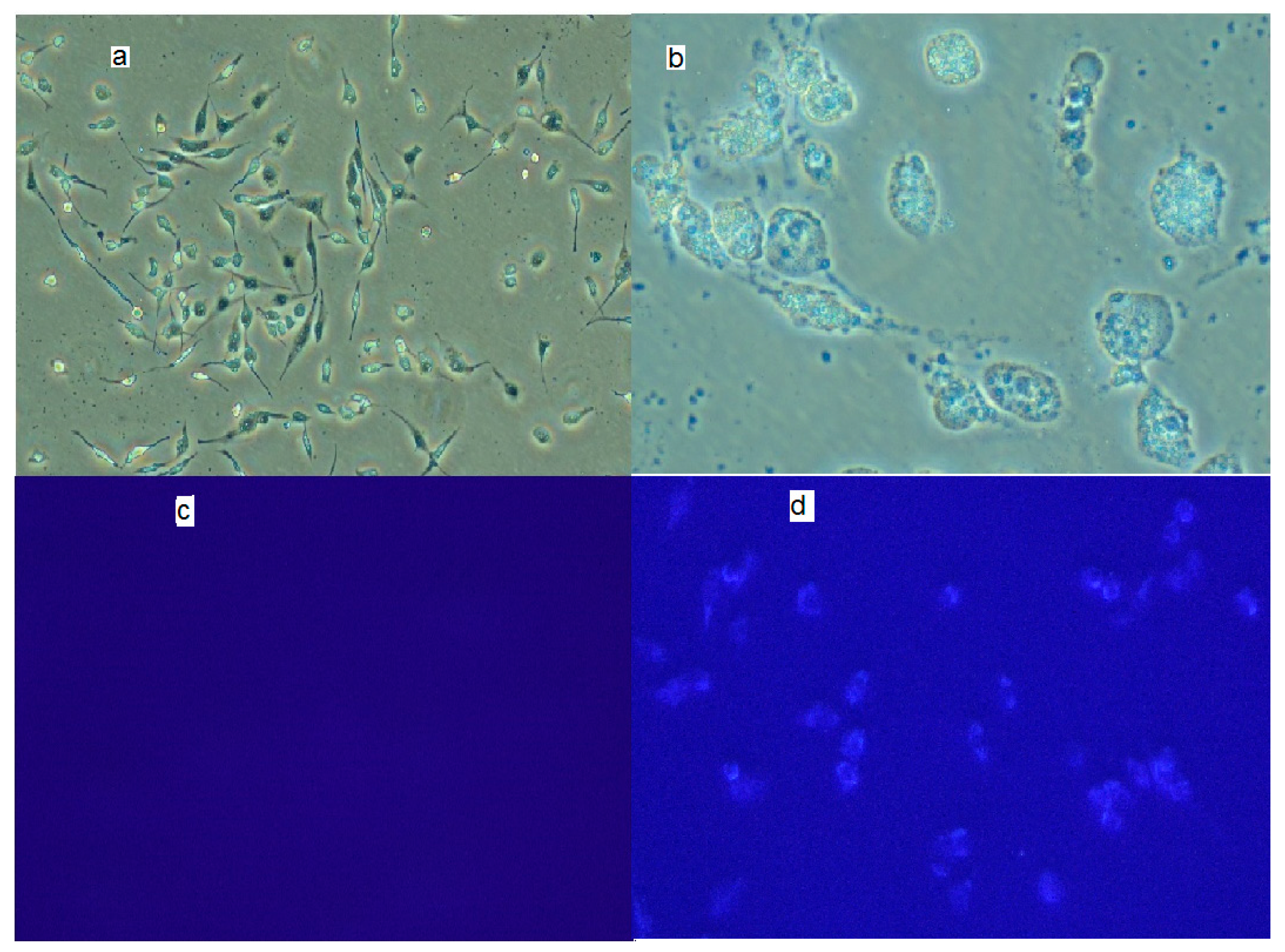
3.3. The In Vitro Cellular Uptake of the Conjugates of Ibuprofen
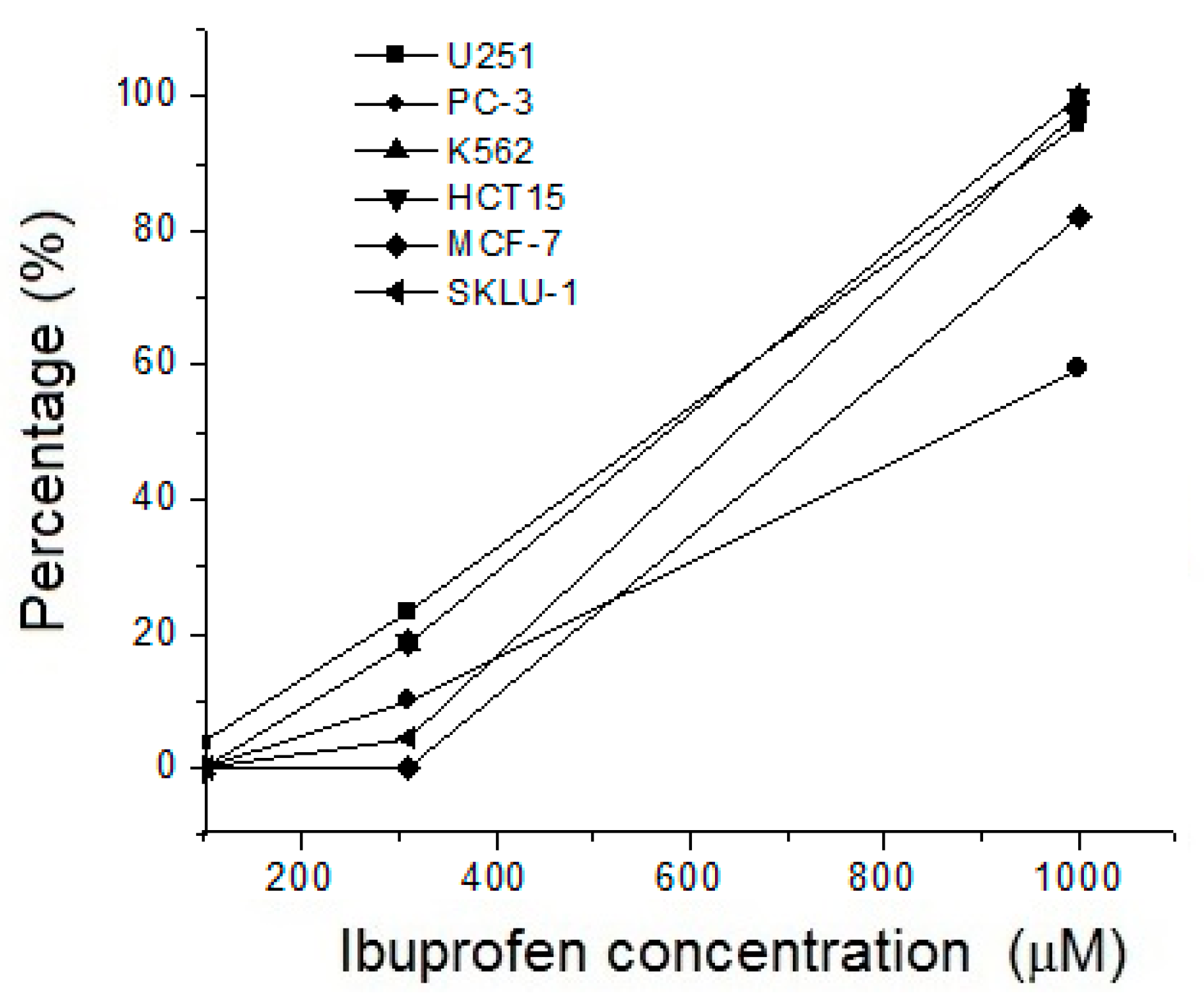
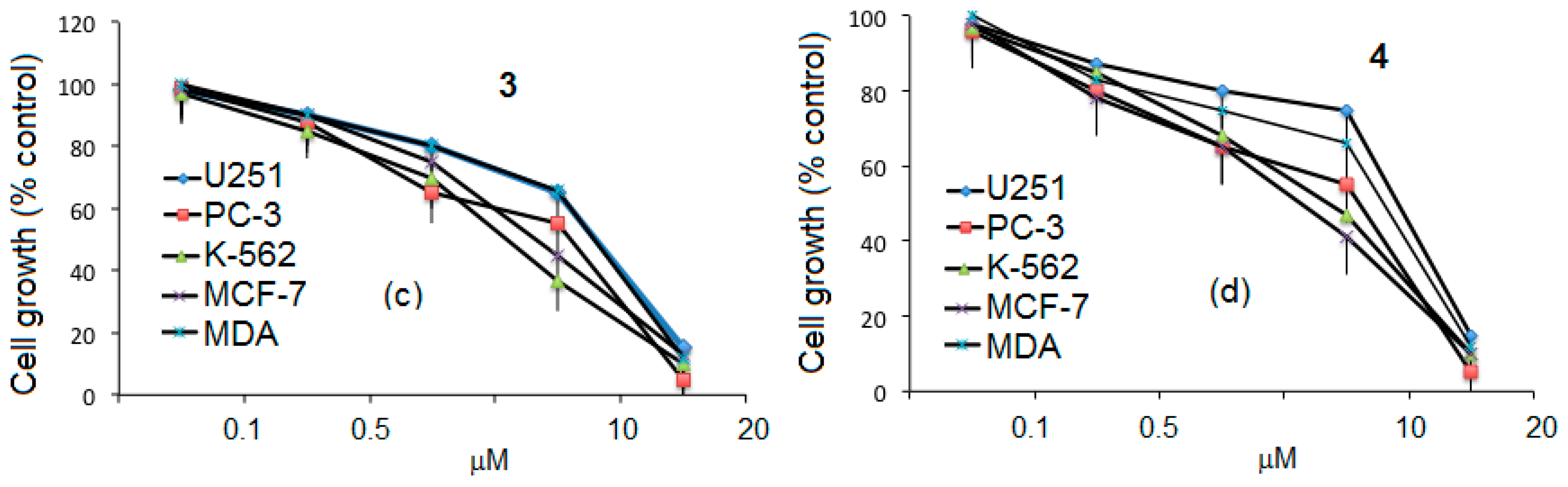
3.4. Cytotoxic Activity of Ibuprofen
3.5. Cytotoxicity of Ibuprofen Conjugates
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Garavito, R.M.; Malkowski, M.G.; DeWitt, D.L. The structures of prostaglandin endoperoxide H synthases-1 and -2, Prostaglandin. Other Lipid Mediat. 2002, 68–69, 129–152. [Google Scholar] [CrossRef]
- Kawail, S.; Kojima, F.; Kusunoki, N. Recent Advances in Nonsteroidal Anti Inflammatory Drugs. Allergol. Int. 2005, 54, 209–215. [Google Scholar] [CrossRef]
- Kean, W.F.; Buchanan, W.W. The use of NSAIDs in rheumatic disorders 2005: A global perspective. Inflammopharmacology 2005, 13, 343–370. [Google Scholar] [CrossRef] [PubMed]
- Shacter, E.; Weitzman, S.A. Chronic inflammation and cancer. Oncology 2002, 16, 217–226. [Google Scholar] [PubMed]
- Bonelli, P.; Tuccillo, F.; Calemma, R.; Pezzetti, F.; Borrelli, A.; Martinelli, R.; De Rosa, A.; Esposito, D.; Palaia, R.; Castello, G. Changes in the gene expression profile of gastric cancer cells in response to ibuprofen: A gene pathway analysis. Pharm. J. 2010, 11, 412–428. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Zhou, W.; Sangha, S.; Albert, A.; Chang, A.J.; Liu, T.C.; Wolfe, M.M. Effects of nonselective cyclooxygenase inhibition with low-dose ibuprofen on tumor growth, oncogenesis, metastasis, and survival in a mouse model of colorectal cancer. Clin. Cancer Res. 2005, 11, 1618–1628. [Google Scholar] [CrossRef] [PubMed]
- Janssen, A.; Schiffmann, S.; Birod, K.; Maier, T.J.; Wobst, I.; Geisslinger, G.; Grösch, S. p53 is important for the anti-proliferative effect of ibuprofen in colon carcinoma cells. Biochem. Biophys. Res. Commun. 2008, 365, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Andrews, J.; Djakiew, D.; Krygier, S.; Andrews, P. Superior effectiveness of ibuprofen compared with other NSAIDs for reducing the survival of human prostate cancer cells. Cancer Chemother. Pharmacol. 2002, 50, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Attiga, F.A.; Fernandez, P.M.; Weeraratna, A.T.; Manyak, M.J.; Patierno, S.R. Inhibitors of Prostaglandin Synthesis Inhibit Human Prostate Tumor Cell Invasiveness and Reduce the Release of Matrix Metalloproteinases. Cancer Res. 2000, 60, 4629–4637. [Google Scholar] [PubMed]
- Redpath, M.; Marques, C.; Dibden, C.; Waddon, A.; Lalla, R.; MacNeil, S. Ibuprofen and hydrogel-released ibuprofen in the reduction of inflammation induced migration in melanoma cells. Br. J. Dermatol. 2009, 161, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Lu, J.; Huang, Y.; Zhao, W.; Li, J.; Gao, X.; Venkataramanan, R.; Sun, M.; Stolz, D.D.; Zhang, L.; et al. PEG-farnesylthiosalicylate conjugate as a nanomicellar carrier for delivery of paclitaxel. Bioconjug. Chem. 2013, 24, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Nie, T.; Wong, C.C.; Alston, N.; Aro, P.; Constantinides, P.P.; Rigas, B. Phospho-ibuprofen (MDC-917) incorporated in nanocarriers: Anti-cancer activity in vitro and in vivo. Br. J. Pharmacol. 2012, 166, 991–1001. [Google Scholar] [CrossRef] [PubMed]
- Kurtoglua, Y.E.; Mishraa, M.K.; Kannanb, S.; Kannana, R.M. Drug release characteristics of PAMAM dendrimer-drug conjugates with different linkers. Int. J. Pharm. 2010, 384, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Huang, Y.; Chen, Y.; Xu, J.; Li, S.; Guo, X. PEG-Fmoc-Ibuprofen Conjugate as a Dual Functional Nanomicellar Carrier for Paclitaxel. Bioconjug. Chem. 2016, 27, 2198–2205. [Google Scholar] [CrossRef] [PubMed]
- Menjoge, A.R.; Kannan, R.M.; Tomalia, D.A. Dendrimer-based drug and imaging conjugates: Design considerations for nanomedical applications. Drug Discov. Today 2010, 15, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Najlah, M.; Freeman, S.; Attwood, D.; D’Emanuele, A. In vitro evaluation of dendrimer prodrugs for oral drug delivery. Int. J. Pharm. 2007, 336, 183–190. [Google Scholar] [CrossRef] [PubMed]
- McMahon, H.T.; Boucrot, E. Molecular mechanism and physiological functions of clathrin-mediated endocytosis. Nat. Rev. Mol. Cell Biol. 2011, 12, 517–533. [Google Scholar] [CrossRef] [PubMed]
- Cortez-Maya, S.; Hernández-Ortega, S.; Ramírez Apan, T.; Lijanova, I.V.; Martínez-García, M. Synthesis of 5-Aryl-1,4-Benzodiazepine Derivatives Attached in Resorcinaren-PAMAM Dendrimers and their Anti-Cancer Activity. Biorgan. Med. Chem. 2012, 20, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Monks, A.; Scudiero, D.; Skehan, P.; Shoemaker, R.; Paul, K.; Vistica, D.; Hose, C.; Langley, J.; Cronise, P.; Vaigro-Wolff, A.; et al. Feasibility of a high-flux anticancer drug screen using a diverse panel of cultured human tumor cell lines. J. Natl. Cancer Inst. 1991, 83, 757–766. [Google Scholar] [CrossRef] [PubMed]




| Cell Lines | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sample | U251 | PC-3 | K-562 | HCT-15 | MFC-7 | SKLU-1 | MDA-MB-231 | HGF |
| 1 | 100 | 100 | 100 | 80.93 | 94.22 | 70.28 | 100 | 13.68 |
| 2 | 89.16 | 100 | 85.43 | 73.26 | 100 | 82.06 | 100 | 2.65 |
| 3 | 100 | 100 | 100 | 75.20 | 100 | 61.04 | 100 | 5.90 |
| 4 | 81.78 | 92.56 | 100 | 79.16 | 82 | 71 | 96.64 | 2.54 |
| Ibuprofen | 3.61 | 7.5 | 6.3 | 6.0 | 19.63 | 5.18 | 18.67 | 9.52 |
| Cisplatin | 87.49 | 42.65 | 79.15 | 32.42 | 52.58 | 81.35 | 43.29 | ND |
| Cell Line/(μM ± SD) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sample | U251 | PC-3 | K-562 | HCT-15 | MFC-7 | SKLU-1 | MDA | HGF |
| 1 | 3.1 ± 0.1 | 19.9 ± 1.5 | 7.3 ± 0.6 | 11.0 ± 0.7 | 4.2 ± 0.3 | 3.6 ± 0.4 | 6.8 ± 0.9 | 32.3 ± 4.3 |
| 2 | 6.7 ± 0.6 | 15.9 ± 1.4 | 3.4 ± 0.8 | 11.3 ± 0.7 | 7.9 ± 0.7 | 12.9 ± 1.1 | 3.6 ± 1.5 | 22.3 ± 4.3 |
| 3 | 3.3 ± 0.2 | 4.4 ± 0.3 | 5.2 ± 0.5 | 5.9 ± 0.2 | 5.5 ± 0.4 | 5.9 ± 0.1 | 4.3 ± 0.4 | 20.3 ± 4.3 |
| 4 | 3.5 ± 0.5 | 11.4 ± 0.2 | 3.0 ± 0.5 | 6.3 ± 0.4 | 3.3 ± 0.3 | 4.2 ± 0.7 | 3.1 ± 0.5 | Not active |
| Cisplatin | 10.8 ± 0.7 | 17.5 ± 1.4 | 1.2 ± 0.08 | 12.3 ± 0.8 | 17.4 ± 0.8 | 12.2 ± 0.9 | 11.2 ± 0.9 | 12.2 ± 0.9 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pedro-Hernández, L.D.; Martínez-Klimova, E.; Cortez-Maya, S.; Mendoza-Cardozo, S.; Ramírez-Ápan, T.; Martínez-García, M. Synthesis, Characterization, and Nanomedical Applications of Conjugates between Resorcinarene-Dendrimers and Ibuprofen. Nanomaterials 2017, 7, 163. https://doi.org/10.3390/nano7070163
Pedro-Hernández LD, Martínez-Klimova E, Cortez-Maya S, Mendoza-Cardozo S, Ramírez-Ápan T, Martínez-García M. Synthesis, Characterization, and Nanomedical Applications of Conjugates between Resorcinarene-Dendrimers and Ibuprofen. Nanomaterials. 2017; 7(7):163. https://doi.org/10.3390/nano7070163
Chicago/Turabian StylePedro-Hernández, Luis D., Elena Martínez-Klimova, Sandra Cortez-Maya, Sonia Mendoza-Cardozo, Teresa Ramírez-Ápan, and Marcos Martínez-García. 2017. "Synthesis, Characterization, and Nanomedical Applications of Conjugates between Resorcinarene-Dendrimers and Ibuprofen" Nanomaterials 7, no. 7: 163. https://doi.org/10.3390/nano7070163