Preparation and Characterization of Zirconia-Coated Nanodiamonds as a Pt Catalyst Support for Methanol Electro-Oxidation
Abstract
:1. Introduction
2. Experimental
2.1. Preparation of ZrO2/ND
2.2. Characterization of ZrO2/ND
2.3. Electrodeposition of Pt Catalysts
2.4. Electrochemical Measurement
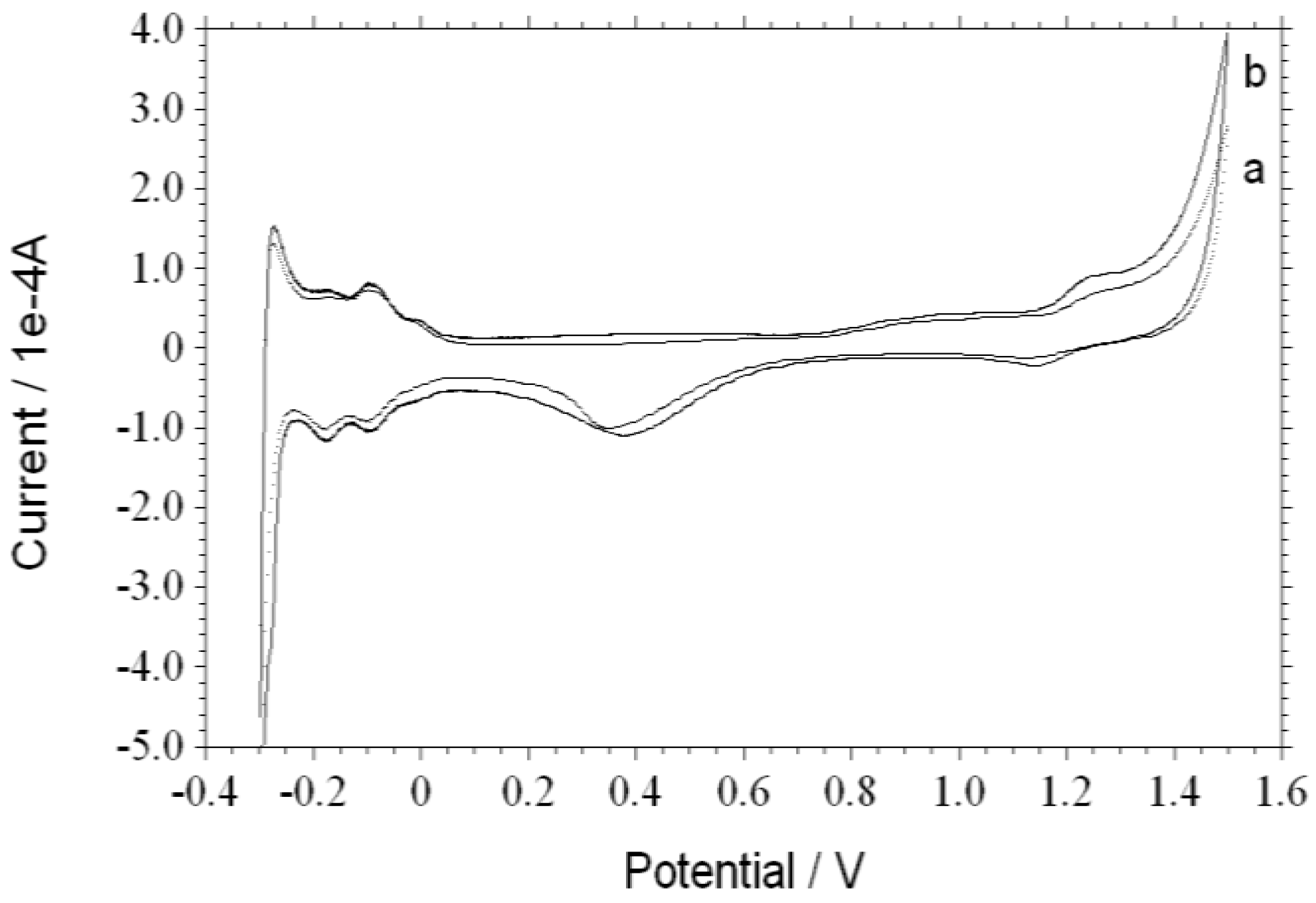
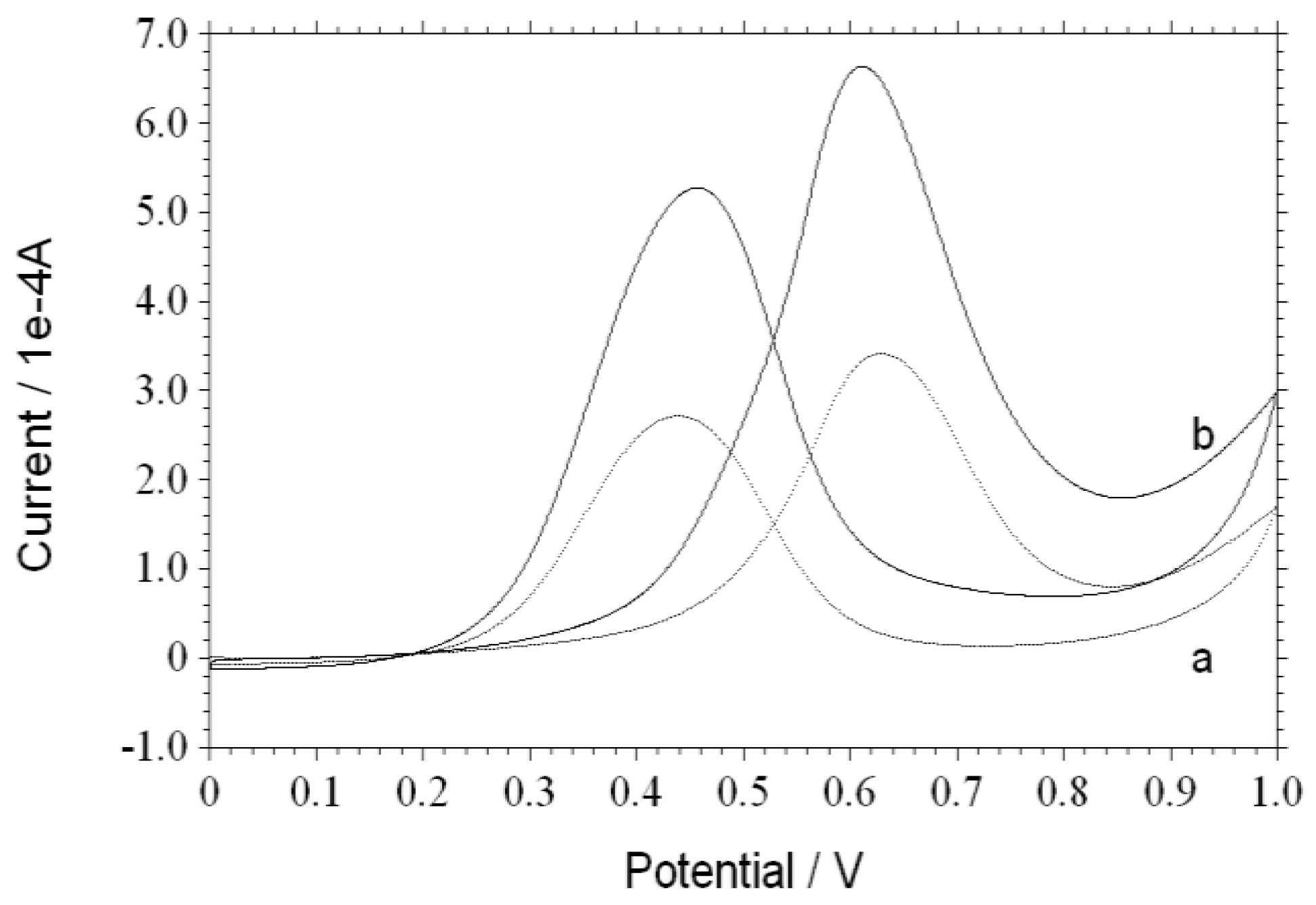
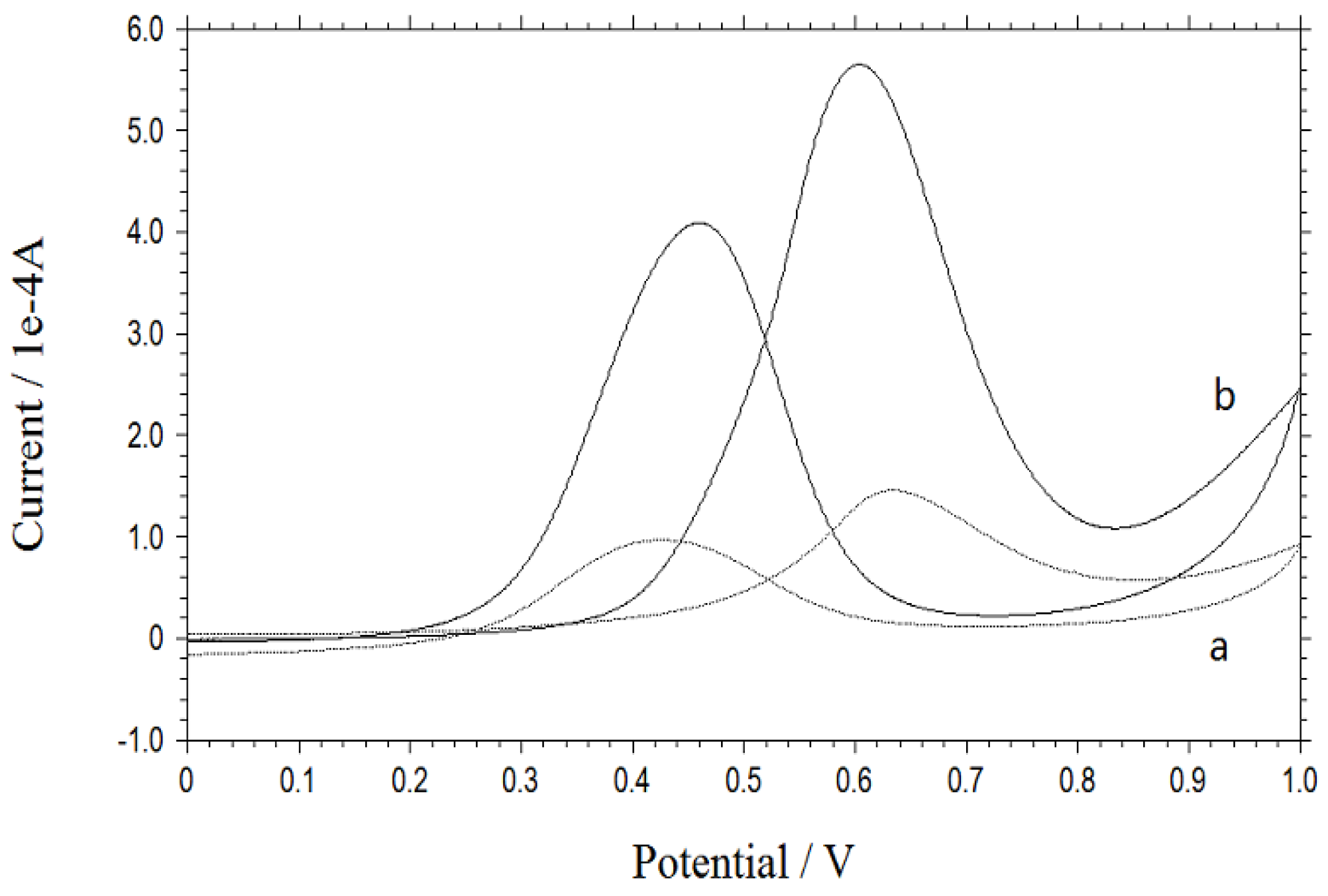
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zheng, J.F.; Bi, W.H.; Dong, X.; Zhu, J.H.; Mao, H.C.; Li, S.H.; Zhang, S.B. High performance tetra-sulfonated poly(p-phenylene-co-aryl ether ketone) membranes with microblock moieties for passive direct methanol fuel cells. J. Membr. Sci. 2016, 517, 47–56. [Google Scholar] [CrossRef]
- Kim, J.; Jang, J.S.; Peck, D.H.; Lee, B.; Yoon, S.H.; Jung, D.H. Methanol-tolerant platinum-palladium catalyst supported on nitrogen-doped carbon nanofiber for high concentration direct methanol fuel cells. Nanomaterials 2016, 6, 148. [Google Scholar] [CrossRef]
- Rabissi, C.; Brightman, E.; Hinds, G.; Casalegno, A. In operando investigation of anode overpotential dynamics in direct methanol fuel cells. Int. J. Hydrogen Energy 2016, 41, 18221–18225. [Google Scholar] [CrossRef]
- Wang, N.; Xu, Y.; Han, Y.; Gao, C.Z.; Cao, X. Mesoporous Pd@M (M = Pt, Au) microrods as excellent electrocatalysts for methanol oxidation. Nano Energy 2015, 17, 111–119. [Google Scholar] [CrossRef]
- Xu, C.B.; Shi, M.Q.; Kang, L.Z.; Ma, C.A. Novel core–shell Pt/WC@TiO2 electro-catalyst for methanol oxidation. Mater. Lett. 2013, 91, 183–186. [Google Scholar] [CrossRef]
- He, C.Y.; Tao, J.Z. Pt loaded two-dimensional TaC-nanosheet/graphene hybrid as an efficient and durable electrocatalyst for direct methanol fuel cells. J. Power Sources 2016, 324, 317–324. [Google Scholar] [CrossRef]
- Lo, A.Y.; Yu, N.Y.; Huang, S.J.; Hung, C.T.; Liu, S.H.; Lei, Z.B.; Kuo, C.T.; Liu, S.B. Fabrication of CNTs with controlled diameters and their applications as electrocatalyst supports for DMFC. Diam. Relat. Mater. 2011, 20, 343–350. [Google Scholar] [CrossRef]
- Wen, Z.; Liu, J.; Li, J. Core/shell Pt/C nanoparticles embedded in mesoporous carbon as a methanol-tolerant cathode catalyst in direct methanol fuel cells. Adv. Mater. 2008, 20, 743–747. [Google Scholar] [CrossRef]
- Akbari, E.; Buntat, A.; Nikoukar, A.; Kheirandish, A.; Khaledian, M.; Afroozeh, A. Sensor Application in Direct Methanol Fuel Cell (DMFCs). Renew. Sustain. Energy Rev. 2016, 60, 1125–1139. [Google Scholar] [CrossRef]
- Lee, S.H.; Kakati, N.; Jee, S.H.; Maiti, J.; Yoon, Y.S. Hydrothermal synthesis of PtRu nanoparticles supported on graphene sheets for methanol oxidation in direct methanol fuel cell. Mater. Lett. 2011, 65, 3281–3284. [Google Scholar] [CrossRef]
- Mondal, S.; Malik, S. Easy synthesis approach of Pt-nanoparticles on polyaniline surface: An efficient electro-catalyst for methanol oxidation reaction. J. Power Sources 2016, 328, 271–279. [Google Scholar] [CrossRef]
- Wang, J.; Swain, G.M. Fabrication and evaluation of platinum/diamond composite electrodes for electrocatalysis: Preliminary studies of the oxygen-reduction reaction. J. Electrochem. Soc. 2003, 150, E24–E32. [Google Scholar] [CrossRef]
- Holt, K.B. Undoped diamond nanoparticles: origins of surface redox chemistry. Phys. Chem. Chem. Phys. 2010, 12, 2048–2058. [Google Scholar] [CrossRef] [PubMed]
- Horbatenko, Y.; Yousaf, M.; Lee, J.; Choi, T.H.; Ruoff, R.S.; Park, N. Synergetic interplay between pressure and surface chemistry for the conversion of sp2-bonded carbon layers into sp3-bonded carbon films. Carbon 2016, 106, 158–163. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Miyazaki, T.; Takeuchi, D.; Yamasaki, S. Direct first-principles simulation of a high-performance electron emitter: lithium-oxide-coated diamond surface. J. Appl. Phys. 2014, 116, 124309. [Google Scholar] [CrossRef]
- Holt, K.B.; Caruana, D.J.; Millán-Barrios, E.J. Electrochemistry of Undoped Diamond Nanoparticles: Accessing Surface Redox States. J. Am. Chem. Soc. 2009, 131, 11272–11273. [Google Scholar] [CrossRef] [PubMed]
- Bian, L.Y.; Wang, Y.H.; Zang, J.B.; Yu, J.K.; Huang, H. Electrodeposition of Pt nanoparticles on undoped nanodiamond powder for methanol oxidation electrocatalysts. J. Electroanal. Chem. 2010, 644, 85–88. [Google Scholar] [CrossRef]
- Ji, Y.Y.; Ying, Y.; Pan, Y.X.; Li, M.Z.; Guo, X.Y.; Wu, Y.P.; Wen, Y.; Yang, H.F. Palladium networks decorated by cuprous oxide for remarkably enhanced electrocatalytic activity of methanol oxidation reaction with high CO-tolerance. J. Power Sources 2016, 329, 115–122. [Google Scholar] [CrossRef]
- Zhou, Y.; Gao, Y.F.; Liu, Y.C.; Liu, J.R. High efficiency Pt-CeO2/carbon nanotubes hybrid composite as an anode electrocatalyst for direct methanol fuel cells. J. Power Sources 2010, 195, 1605–1609. [Google Scholar] [CrossRef]
- Choi, M.; Han, C.; Kim, I.T.; Lee, J.J.; Lee, H.K.; Shim, J. Effect of the Nanosized TiO2 Particles in Pd/C Catalysts as Cathode Materials in Direct Methanol Fuel Cells. J. Nanosci. Nanotechnol. 2011, 11, 6420–6424. [Google Scholar] [CrossRef] [PubMed]
- Assaud, L.; Brazeau, N.; Barr, M.K.S.; Hanbücken, M.; Ntais, S.; Baranova, E.A.; Santinacci, L. Atomic layer deposition of Pd nanoparticles on TiO2 nanotubes for ethanol electrooxidation: synthesis and electrochemical properties. ACS Appl. Mater. Interfaces 2015, 7, 24533–24542. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.Q.; Li, Y.Z.; Ma, Y.H.; Zhao, X.; Zhang, Q.H. Platinum-antimony doped tin oxide nanoparticles supported on carbon black as anode catalysts for direct methanol fuel cells. J. Power Sources 2011, 196, 6228–6231. [Google Scholar] [CrossRef]
- Wang, R.F.; Wang, K.L.; Wang, H.; Wang, Q.Z.; Key, J.L.; Linkov, V.; Ji, S. Nitrogen-doped carbon coated ZrO2 as a support for Pt nanoparticles in the oxygen reduction reaction. Int. J. Hydrogen Energy 2013, 38, 5783–5788. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, H.M.; Zhong, H.X.; Hu, J.W.; Xu, D.Y.; Shao, Z.G. A novel sintering resistant and corrosion resistant Pt4ZrO2/C catalyst for high temperature PEMFCs. Electrochim. Acta 2006, 51, 5710–5714. [Google Scholar] [CrossRef]
- Taghizadeh, M.T.; Vatanparast, M. Ultrasonic-assisted synthesis of ZrO2 nanoparticles and their application to improve the chemical stability of Nafion membrane in proton exchange membrane (PEM) fuel cells. J. Colloid Interface Sci. 2016, 483, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zang, J.B.; Shan, S.X.; Huang, H.; Wang, Y.H. Synthesis and Characterization of Core-Shell Structural MWNT-Zirconia Nanocomposites. Nano Lett. 2008, 8, 4070–4074. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.H.; Zhao, Y.L.; Lu, R.; Dong, L.; Zang, J.B.; Lu, J.; Xu, X.P. Nano Titania Modified Nanodiamonds as Stable Electrocatalyst Supports for Direct Methanol Fuel Cells. J. Electrochem. Soc. 2015, 162, F211–F215. [Google Scholar] [CrossRef]
- Zhao, Y.L.; Wang, Y.H.; Zang, J.B.; Lu, J.; Xu, X.P. A novel support of nano titania modified graphitized nanodiamond for Pt electrocatalyst in direct methanol fuel cell. Int. J. Hydrogen Energy 2015, 40, 4540–4547. [Google Scholar] [CrossRef]


© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lu, J.; Zang, J.; Wang, Y.; Xu, Y.; Xu, X. Preparation and Characterization of Zirconia-Coated Nanodiamonds as a Pt Catalyst Support for Methanol Electro-Oxidation. Nanomaterials 2016, 6, 234. https://doi.org/10.3390/nano6120234
Lu J, Zang J, Wang Y, Xu Y, Xu X. Preparation and Characterization of Zirconia-Coated Nanodiamonds as a Pt Catalyst Support for Methanol Electro-Oxidation. Nanomaterials. 2016; 6(12):234. https://doi.org/10.3390/nano6120234
Chicago/Turabian StyleLu, Jing, Jianbing Zang, Yanhui Wang, Yongchao Xu, and Xipeng Xu. 2016. "Preparation and Characterization of Zirconia-Coated Nanodiamonds as a Pt Catalyst Support for Methanol Electro-Oxidation" Nanomaterials 6, no. 12: 234. https://doi.org/10.3390/nano6120234
APA StyleLu, J., Zang, J., Wang, Y., Xu, Y., & Xu, X. (2016). Preparation and Characterization of Zirconia-Coated Nanodiamonds as a Pt Catalyst Support for Methanol Electro-Oxidation. Nanomaterials, 6(12), 234. https://doi.org/10.3390/nano6120234




