RF-Sputtered ZnO Nano-Coatings on Polyamide Thin-Film Composite Membranes for Tuned Nanofiltration Selectivity
Abstract
1. Introduction
2. Materials and Methods
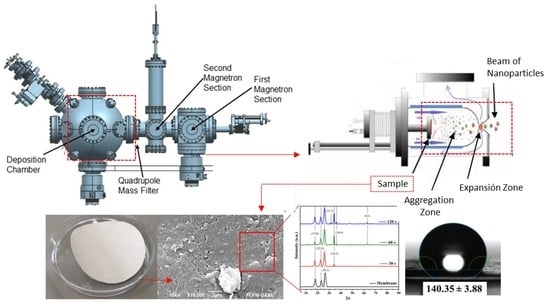
2.1. RF Sputtering Deposition of ZnO Nano-Coatings
2.2. Surface and Structural Characterization
2.3. Filtration Tests
3. Results
3.1. Physical and Chemical Characterization of ZnO-Modified Membranes
3.2. Water Permeate Flux Density of Modified Membranes
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Goh, P.S.; Lau, W.J.; Othman, M.H.D.; Ismail, A.F. Membrane fouling in desalination and its mitigation strategies. Desalination 2018, 425, 130–155. [Google Scholar] [CrossRef]
- Wadekar, S.S.; Vidic, R.D. Influence of Active Layer on Separation Potentials of Nanofiltration Membranes for Inorganic Ions. Environ. Sci. Technol. 2017, 51, 5658–5665. [Google Scholar] [CrossRef]
- Beratto-Ramos, A.; Jaramillo, K.; Zapata, P.; Romero, J.; Martínez, J.; Meléndrez, M.F.; Saravia, F.; Horn, H.; Borquez, R.; Pino-Soto, L. Characterization and evaluation of the recovery process of saturated reverse osmosis membranes by chemical oxidation. Desalination 2025, 594, 118273. [Google Scholar] [CrossRef]
- Zou, H.; Jin, Y.; Yang, J.; Dai, H.; Yu, X.; Xu, J. Synthesis and characterization of thin film composite reverse osmosis membranes via novel interfacial polymerization approach. Sep. Purif. Technol. 2010, 72, 256–262. [Google Scholar] [CrossRef]
- Tayeh, Y.A.; Alazaiza, M.Y.D.; Alzghoul, T.M.; Bashir, M.J.K. A comprehensive review of RO membrane fouling: Mechanisms, categories, cleaning methods and pretreatment technologies. J. Hazard. Mater. Adv. 2025, 18, 100684. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, R.; Liu, Y.; He, M.; Su, Y.; Gao, C.; Jiang, Z. Antifouling membrane surface construction: Chemistry plays a critical role. J. Membr. Sci. 2018, 551, 145–171. [Google Scholar] [CrossRef]
- Vargas-Figueroa, C.; Pino-Soto, L.; Beratto-Ramos, A.; Rivas, B.L.; Tapiero, Y.; Palacio, D.; Meléndrez, M.F.; Borquez, R. Surface Modification of Nanofiltration Membranes by Interpenetrating Polymer Networks and Their Evaluation in Water Desalination. ACS Appl. Polym. Mater. 2023, 5, 4910–4920. [Google Scholar] [CrossRef]
- Vargas-Figueroa, C.; Pino-Soto, L.; Beratto-Ramos, A.; Tapiero, T.; Rivas, B.L.; Berrio, M.E.; Meléndrez, M.F.; Bórquez, R. In-Situ modification of nanofiltration membranes using carbon nanotubes for water treatment. Membranes 2023, 13, 616. [Google Scholar] [CrossRef]
- Hilal, N.; Khayet, M.; Wright, C.J. (Eds.) Membrane Modification: Technology and Applications; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Luis, P. Introduction. In Fundamental Modelling of Membrane Systems; Luis, P., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–23. [Google Scholar][Green Version]
- Rajakumaran, R.; Boddu, V.; Kumar, M.; Shalaby, M.S.; Abdallah, H.; Chetty, R. Effect of ZnO morphology on GO-ZnO modified polyamide reverse osmosis membranes for desalination. Desalination 2019, 467, 245–256. [Google Scholar] [CrossRef]
- Yang, W.; Son, M.; Rossi, R.; Vrouwenvelder, J.S.; Logan, B.E. Adapting Aluminum-Doped Zinc Oxide for Electrically Conductive Membranes Fabricated by Atomic Layer Deposition. ACS Appl. Mater. Interfaces 2020, 12, 963–969. [Google Scholar] [CrossRef]
- Kacprzyńska-Gołacka, J.; Łożyńska, M.; Barszcz, W.; Sowa, S.; Wieciński, P. Microfiltration Membranes Modified with Zinc by Plasma Treatment. Membranes 2023, 13, 387. [Google Scholar] [CrossRef]
- Adachi, H.; Hata, T.; Wasa, K. 5—Basic Process of Sputtering Deposition. In Handbook of Sputtering Technology, 2nd ed.; William Andrew Publishing: Oxford, UK, 2012; pp. 295–359. [Google Scholar]
- Meléndrez, M.F.; Solís-Pomar, F.; Nápoles, O.; Vázquez Robaina, O.; Gutierrez- Lazos, C.; Fundora, A.; Colin, A.; Pérez Tijerina, E. Preparation and Characterization of Nanostructured Titanium Nitride Thin Films at Room Temperature. Ceram. Int. 2016, 42, 7571–7575. [Google Scholar] [CrossRef]
- Martínez-Carreón, M.J.; Solís-Pomar, F.; Fundora, A.; Gutiérrez-Lazos, C.D.; Mejía-Rosales, S.; Fernández-Escamilla, H.N.; Guerrero-Sánchez, J.; Meléndrez, M.F.; Pérez-Tijerina, E. Synthesis of silver–palladium Janus nanoparticles using co-sputtering of independent sources: Experimental and theorical study. Beilstein J. Nanotechnol. 2024, 15, 808–816. [Google Scholar] [CrossRef]
- Wang, X.; Fu, C.; Zhang, C.; Qiu, Z.; Wang, B. A Comprehensive Review of Wetting Transition Mechanism on the Surfaces of Microstructures from Theory and Testing Methods. Materials 2022, 15, 4747. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.R.; Protasova, E.; Strand, S.; Bilstad, T. Implementation of Spiegler–Kedem and Steric Hindrance Pore Models for Analyzing Nanofiltration Membrane Performance for Smart Water Production. Membranes 2018, 8, 78. [Google Scholar] [CrossRef]
- Andalaft, J.; Schwarz, A.; Pino, L.; Fuentes, P.; Bórquez, R.; Aybar, M. Assessment and Modeling of Nanofiltration of Acid Mine Drainage. Ind. Eng. Chem. Res. 2018, 57, 14727–14739. [Google Scholar] [CrossRef]
- Chong, T.H.; Fane, A.G. Nanofiltration Module Design and Operation. In Nanofiltration; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2021; Chapter 3; pp. 95–135. [Google Scholar] [CrossRef]
- Ahmed, F.N. Modified Spiegler–Kedem Model to Predict the Rejection and Flux of Nanofiltration Processes at High NaCl Concentrations. Master’s Thesis, University of Ottawa, Ottawa, ON, Canada, 2013. [Google Scholar]
- Agboola, O.; Mokrani, T.; Sadiku, R. Porous and Fractal Analysis on the Permeability of Nanofiltration Membranes for the Removal of Metal Ions. J. Mater. Sci. 2016, 51, 2499–2511. [Google Scholar] [CrossRef]
- Nizar, B.M.; Lajnef, M.; Chaste, J.; Chtourou, R.; Herth, E. Highly C-Oriented (002) Plane ZnO Nanowires Synthesis. RSC Adv. 2023, 13, 15077–15085. [Google Scholar] [CrossRef] [PubMed]
- Jian, S.-J.; Chu, S.-Y.; Huang, T.-Y.; Water, W. Study of Preferred Orientation of Zinc Oxide Films on the 64° LiNbO3 Substrates and Their Applications as Liquid Sensors. J. Vac. Sci. Technol. A 2004, 22, 2424–2430. [Google Scholar] [CrossRef]
- Juholin, P.; Kääriäinen, M.-L.; Riihimäki, M.; Sliz, R.; Aguirre, J.L.; Pirilä, M.; Fabritius, T.; Cameron, D.; Keiski, R.L. Comparison of ALD Coated Nanofiltration Membranes to Unmodified Commercial Membranes in Mine Wastewater Treatment. Sep. Purif. Technol. 2018, 192, 69–77. [Google Scholar] [CrossRef]
- Nicolini, J.V.; Borges, C.P.; Ferraz, H.C. Selective rejection of ions and correlation with surface properties of nanofiltration membranes. Sep. Purif. Technol. 2016, 171, 238–247. [Google Scholar] [CrossRef]
- Rabeel, M.; Javed, S.; Khan, R.; Akram, M.A.; Rehman, S.; Kim, D.-k.; Khan, M.F. Controlling the Wettability of ZnO Thin Films by Spray Pyrolysis for Photocatalytic Applications. Materials 2022, 15, 3364. [Google Scholar] [CrossRef] [PubMed]
- Rosa, A.M.; da Silva, E.P.; Amorim, E.; Chaves, M.; Catto, A.C.; Lisboa-Filho, P.N.; Bortoleto, J.R.R. Growth evolution of ZnO thin films deposited by RF magnetron sputtering. J. Phys. Conf. Ser. 2012, 370, 012020. [Google Scholar] [CrossRef]
- Jayatissa, A.H.; Ahmed, O.; Manu, B.R.; Schroeder, A.M. Tribological Behaviour of Sputter Coated ZnO Thin Films. Recent Prog. Mater. 2021, 3, 003. [Google Scholar] [CrossRef]
- Li, N.; Wang, W.; Zhu, L.; Cui, W.; Chen, X.; Zhang, B.; Zhang, Z. A novel electro-cleanable PAN-ZnO nanofiber membrane with superior water flux and electrocatalytic properties for organic pollutant degradation. Chem. Eng. J. 2021, 421, 127857. [Google Scholar] [CrossRef]
- Baig, U.; Al-Kuhaili, M.F.; Dastageer, M.A. Photo-responsive Zinc Oxide-coated alumina ceramic membrane with super-wettable and self-cleaning features fabricated by single step RF magnetron sputtering for oily water treatment. Process Saf. Environ. Prot. 2023, 175, 541–553. [Google Scholar] [CrossRef]
- Salih, A.K.; Aditya, L.; Matar, F.; Nghiem, L.D.; Ton-That, C. Improved flux and anti-fouling performance of a photocatalytic ZnO membrane on porous stainless steel substrate for microalgae harvesting. J. Membr. Sci. 2024, 694, 122405. [Google Scholar] [CrossRef]
- Al-Zoubi, H.; Hilal, N.; Darwish, N.A.; Mohammad, A.W. Rejection and modelling of sulphate and potassium salts by nanofiltration membranes: Neural network and Spiegler–Kedem model. Desalination 2007, 206, 42–60. [Google Scholar] [CrossRef]
- Mikulášek, P.; Kopecký, V.; Kušnierik, O. Characterization of nanofiltration membranes used for the separation of aqueous dye-salt solutions. Environ. Prot. Eng. 2005, 31, 169–176. [Google Scholar]








| Membrane | Scan Area (μm2) | Rq (RMS) (nm) | Ra (nm) | Wenzel Factor, r (-) |
|---|---|---|---|---|
| Unmodified | 10 × 10 | 90.22 | 71.42 | 1.059 |
| ZnO-coated (30 s) | 10 × 10 | 230.7 | 201.1 | 1.195 |
| ZnO-coated (60 s) | 10 × 10 | 249.1 | 202.8 | 1.155 |
| ZnO-coated (120 s) | 10 × 10 | 174.0 | 144.2 | 1.096 |
| Membrane | σ (-) | P (m·s−1) | k (m·s−1) |
|---|---|---|---|
| NF90 | 0.83 | 4.86 × 10−7 | 0.011 |
| ZnO 30 s | 0.86 | 2.58 × 10−7 | 0.011 |
| ZnO 60 s | 0.90 | 7.25 × 10−8 | 0.011 |
| ZnO 120 s | 0.92 | 2.85 × 10−7 | 0.011 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Vargas, C.; Palacio, D.A.; Ramírez, J.; Pérez-Tijerina, E.; Solís-Pomar, F.; Fundara-Cruz, A.; Bórquez, R.; Jaramillo, A.F.; Oñate, Á.; Pino-Soto, L.; et al. RF-Sputtered ZnO Nano-Coatings on Polyamide Thin-Film Composite Membranes for Tuned Nanofiltration Selectivity. Nanomaterials 2026, 16, 598. https://doi.org/10.3390/nano16100598
Vargas C, Palacio DA, Ramírez J, Pérez-Tijerina E, Solís-Pomar F, Fundara-Cruz A, Bórquez R, Jaramillo AF, Oñate Á, Pino-Soto L, et al. RF-Sputtered ZnO Nano-Coatings on Polyamide Thin-Film Composite Membranes for Tuned Nanofiltration Selectivity. Nanomaterials. 2026; 16(10):598. https://doi.org/10.3390/nano16100598
Chicago/Turabian StyleVargas, Catalina, Daniel A. Palacio, Jesús Ramírez, Eduardo Pérez-Tijerina, Francisco Solís-Pomar, Abel Fundara-Cruz, Rodrigo Bórquez, Andrés F. Jaramillo, Ángelo Oñate, Luis Pino-Soto, and et al. 2026. "RF-Sputtered ZnO Nano-Coatings on Polyamide Thin-Film Composite Membranes for Tuned Nanofiltration Selectivity" Nanomaterials 16, no. 10: 598. https://doi.org/10.3390/nano16100598
APA StyleVargas, C., Palacio, D. A., Ramírez, J., Pérez-Tijerina, E., Solís-Pomar, F., Fundara-Cruz, A., Bórquez, R., Jaramillo, A. F., Oñate, Á., Pino-Soto, L., & Melendrez, M. F. (2026). RF-Sputtered ZnO Nano-Coatings on Polyamide Thin-Film Composite Membranes for Tuned Nanofiltration Selectivity. Nanomaterials, 16(10), 598. https://doi.org/10.3390/nano16100598