From Structure to Optics: The pH-Temperature Interplay in Aqueous Solution CdS Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
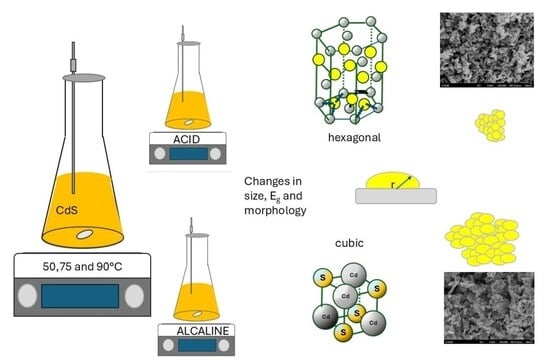
2.1. CdS Nanoparticles Synthesis
2.2. Characterization
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CdS | cadmium sulfide |
| SEM | scanning electron microscopy |
| XRD | X-ray diffraction |
| Cs | crystallite size |
| FWHM | full width at half maximum |
| Eg | band gap energy |
References
- Mandal, S.; Dutta, D.; Ali, S.I.; Ghosh, P.; Mandal, A.C. Role of vacancy type defects on microstructural and optoelectronic property of CdS wurtzite nanoparticles. J. Mol. Struct. 2024, 1304, 137734. [Google Scholar] [CrossRef]
- Lihitkar, P.B.; Kale, S.; Afre, R.A.; Pugliese, D. Structural, morphological and optical characterization of CdS-doped silica aerogels synthesized through sol-gel method. Mater. Res. Bull. 2025, 192, 113590. [Google Scholar]
- Chang, S.-K.; Abbasi, Q.-U.-I.; Abbasi, Z.; Khushbakht, F.; Ullah, I.; Rehman, F.U.; Hafeez, M. Rapid pH-dependent Photocatalytic Degradation of Methylene Blue by CdS Nanorods Synthesized through Hydrothermal Process. Arab. J. Chem. 2024, 17, 105422. [Google Scholar]
- Singh, V.; Chauhan, P. Structural and optical characterization of CdS nanoparticles prepared by chemical precipitation method. J. Phys. Chem. Solids 2009, 70, 1074–1079. [Google Scholar] [CrossRef]
- Sheng, C.K.; Alrababah, Y.M. Thermally driven structural phase transformation and dislocation density of CdS nanoparticles precipitated without surfactant in KOH alkaline medium. Kuwait J. Sci. 2022, 49, 1–10. [Google Scholar] [CrossRef]
- Sheng, C.K.; Alrababah, Y.M. pH-induced wurtzite-zinc blende heterogeneous phase formation, optical properties tuning and thermal stability improvement of green synthesized CdS nanoparticles. Heliyon 2023, 9, e15908. [Google Scholar] [CrossRef]
- Mahdi, M.A.; Hassan, J.J.; Ng, S.S.; Hassan, Z. Growth of CdS nanosheets and nanowires through the solvothermal method. J. Cryst. Growth 2012, 359, 43–48. [Google Scholar] [CrossRef]
- Mahdi, H.S.; Ali, M.M.; Dhabian, S.Z. Structural and optical properties of cadmium sulphide nanoparticles synthesized by green method using bay laurel leave extract. Int. J. Appl. Sci. Technol. 2024, 6, 287–295. [Google Scholar]
- Faisal, S.; Akhtar, N.; Ullah, H.; Akif, M.; Khan, I.; Ali, F.; Mohmand, N.Z.K. Antifungal and Antibacterial Activity of Lathyrus aphaca L. Mediated Green Synthesized CdS Nanoparticles and Their Characterization. Green Chem. Technol. 2025, 2, 10007. [Google Scholar] [CrossRef]
- Kumar, A.; Verma, A.; Singh, A.; Yadav, B.C. Wet chemical preparation of CdS nanoparticles for the photodetection application. Mater. Today Proc. 2023, 73, 337–341. [Google Scholar]
- Mohanraj, V.; Jayaprakash, R.; Chandrasekaran, J.; Robert, R.; Sangaiya, P. Influence of pH on particle size, bandgap and activation energy of CdS nanoparticles synthesized at constant frequency ultrasonic wave irradiation. Mater. Sci. Semicond. Process. 2017, 66, 131–139. [Google Scholar] [CrossRef]
- Ceferov, S.; Abbasov, A. Synthesis and physical properties of cadmium sulphur (cds) nanoparticles. Luminis Appl. Sci. Eng. 2025, 2, 64–71. [Google Scholar] [CrossRef]
- Gaur, R.; Jeevanandam, P. Evolution of different morphologies of CdS nanoparticles by thermal decomposition of bis(thiourea)cadmium chloride in various solvents. J. Nanoparticle Res. 2015, 17, 156. [Google Scholar] [CrossRef]
- Gaur, R.; Jeevanandam, P. Effect of anions on the morphology of CdS nanoparticles prepared via thermal decomposition of different cadmium thiourea complexes in a solvent and in the solid state. New J. Chem. 2015, 39, 9442–9453. [Google Scholar] [CrossRef]
- Jie, L.; Gao, X.; Cao, X.; Wu, S.; Long, X.; Ma, Q.; Su, J. A review of CdS photocatalytic nanomaterials: Morphology, synthesis methods, and applications. Mater. Sci. Semicond. Process. 2024, 176, 108288. [Google Scholar] [CrossRef]
- Sharma, S.; Dadhwal, R.; Banerjee, R. Nanoparticle assisted phytoremediation: An eco-friendly approach for removal of heavy metals from the environment. J. Environ. Sci. 2026, 159, 705–720. [Google Scholar] [CrossRef]
- Le, Y.; Wang, H. Remediation of wastewater by using CdS-based biohybrids: Challenges and enhancement strategies. Bioresour. Technol. 2025, 426, 132379. [Google Scholar] [CrossRef]
- Priya, N.S.; Kamala, S.S.P.; Anbarasu, V.; Azhagan, S.A.; Saravanakumar, R. Characterization of CdS thin films and nanoparticles by a simple Chemical Bath Technique. Mater. Lett. 2018, 220, 161–164. [Google Scholar] [CrossRef]
- Fu, D.; Zhang, Y.; Liu, J.; Lu, Z. pH effects on the second-order nonlinear optical properties of surface modified CdS nanoparticles. Mater. Chem. Phys. 2002, 77, 285–288. [Google Scholar] [CrossRef]
- Yang, H.; Jin, Z.; Fan, K.; Liu, D.; Lu, G. The roles of Ni nanoparticles over CdS nanorods for improved photocatalytic stability and activity. Superlattices Microstruct. 2017, 111, 687–695. [Google Scholar] [CrossRef]
- Higareda-Sánchez, A.; Mis-Fernández, R.; Rimmaudo, I.; Camacho-Espinosa, E.; Peña, J.L. Evaluation of pH and deposition mechanisms effect on tin sulfide thin films deposited by chemical bath deposition. Superlattices Microstruct. 2021, 151, 106831. [Google Scholar] [CrossRef]
- Landin, R.O.; Sandoval-Paz, M.G.; Ortuño-López, M.B.; Sotelo-Lerma, M.; Ramírez-Bon, R. Observations on the influence of pH control on the properties of chemically deposited CdS films in an ammonia-free system. J. Phys. Chem. Solids 2009, 70, 1034–1041. [Google Scholar] [CrossRef]
- Rienke, P.C.; Bentjen, S.B. Deposition of Cadmium Sulfide Films by Descomposition of Thiourea in Basic Solutions. Chem. Mater. 1993, 5, 43–53. [Google Scholar]
- Khalizov, A.; Wang, L.; Hu, M.; Xu, W. Nucleation and Growth of Nanoparticles in the Atmosphere. Chem. Rev. 2012, 112, 1957–2011. [Google Scholar]
- Sanchez-Ramirez, E.A.; Hernandez-Perez, M.A.; Aguilar-Hernandez, J.R. Study on the introduction of Se into CdS thin films: Influence on the kinetics of the deposition and the structural and optical properties. Appl. Surf. Sci. 2015, 347, 35–39. [Google Scholar] [CrossRef]
- Levit, S.L.; Stwodah, R.M.; Tang, C. Rapid, Room Temperature Nanoparticle Drying and Low-Energy Reconstitution via Electrospinning. J. Pharm. Sci. 2017, 107, 807–813. [Google Scholar]
- Olasanmi, O.; Akinsola, S.; Yusuf, K.; Aregbesola, E. Comparative studies for determining the optical band gap energy of CuSe thin films. Proc. Niger. Soc. Phys. Sci. 2025, 2, 191. [Google Scholar] [CrossRef]
- Garoufalis, C.S.; Barnasas, A.; Stamatelatos, A.; Karoutsos, V.; Grammatikopoulos, S.; Poulopoulos, P.; Baskoutas, S. A study of quantum confinement effects in ultrathin NiO films performed by experiment and theory. Materials 2018, 11, 949. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, J.; Wang, W.; Huang, X.; Yuan, Q.; Hong, R.; Cha, L. Growth process and properties of CdS thin films prepared by chemical bath deposition at different pH values. J. Mater. Sci. Mater. Electron. 2018, 29, 7637–7643. [Google Scholar]
- Fernando, D.; Khan, M.; Vasquez, Y. Control of the crystalline phase and morphology of CdS deposited on microstructured surfaces by chemical bath deposition. Mater. Sci. Semicond. Process. 2015, 30, 174–180. [Google Scholar] [CrossRef]
- Lima-lima, H.; Portillo-Moreno, O.; de Oca, L.M.E.M.; Lozada-Morales, R.; Zelaya-Angel, O. Análisis de reacciones en la transición de CdS (semiconductor) a CdCO3 (aislante) en formato de películas delgadas obtenidas mediante DBQ. Superf. Vacío 2008, 21, 21–26. [Google Scholar]
- Oliva, A.I.; Martín-Várguez, P.E.; González-Panzo, I.J.; González-Chan, I.J. Papel del diagrama de distribución de especies en el depósito de capas delgadas semiconductoras por baño químico. Rev. Mex. Ing. Química 2016, 15, 209–220. [Google Scholar]
- Ruvalcaba-Manzo, S.G.; Castillo, S.J.; Ochoa-Landin, R.; Flores-Acosta, M.; Ramirez-Bon, R. optical, structural, and morphological characterization of cadmium carbonate thin films by CBD two formulations. Opt. Mater. 2020, 109, 110295. [Google Scholar] [CrossRef]
- Balayeva, N.O.; Mamiyev, Z.Q. Synthesis and studies of CdS and ZnS-PE/NBR modified thermoplastic elastomeric copolymer nanocomposite films. Mater. Lett. 2016, 162, 121–125. [Google Scholar] [CrossRef]
- Kotkata, M.F.; Masoud, A.E.; Mohamed, M.B.; Mahmoud, E.A. Synthesis and structural characterization of CdS nanoparticles. Phys. E Low-Dimens. Syst. Nanostructures 2009, 41, 1457–1465. [Google Scholar] [CrossRef]
- Singh, V.; Sharma, P.K.; Chauhan, P. Synthesis of CdS nanoparticles with enhanced optical properties. Mater. Charact. 2011, 62, 43–52. [Google Scholar] [CrossRef]
- Moreno-Regino, V.D.; Castañeda-de-la-Hoya, F.M.; Torres-Castanedo, C.G.; Márquez-Marín, J.; Castanedo-Pérez, R.; Torres-Delgado, G.; Zelaya-Ángel, O. Structural, optical, electrical and morphological properties of CdS films deposited by CBD varying the complexing agent concentration. Results Phys. 2019, 13, 102238. [Google Scholar] [CrossRef]
- Tohidi, T.; Novini, N.Y.; Jamshidi-Ghaleh, K. Effect of Gamma irradiation on optical characteristics of Fe-doped CdS thin films prepared using chemical bath deposition. Opt. Mater. 2024, 151, 115394. [Google Scholar] [CrossRef]
- Liu, Q.Q.; Shi, J.H.; Li, Z.Q.; Zhang, D.W.; Li, X.D.; Sun, Z.; Zhang, L.Y.; Huang, S.M. Morphological and stoichiometric study of chemical bath deposited CdS films by varying ammonia concentration. Phys. B Condens. Matter 2010, 405, 4360–4365. [Google Scholar] [CrossRef]
- Kariper, A.; Güneri, E.; Göde, F.; Gümüş, C.; Özpozan, T. The structural, electrical and optical properties of CdS thin films as a function of pH. Mater. Chem. Phys. 2011, 129, 183–188. [Google Scholar] [CrossRef]



| T (°C) | pH | Peaks Position CdS 2θ | Cs | Eg |
|---|---|---|---|---|
| 50 | 6.8 | 25.06°, 26.85°, 28.48°, 44.15°, 48.31°, 51.89° | 10 | 2.34 |
| 9.4 | 25.26°, 26.92°, 44.34°, 52.23° | 9 | 2.29 | |
| 10.1 | 25.26°, 44.10°, 52.04°, 54.86° | 11 | 2.31 | |
| 75 | 4.7 | 25.06°, 26.70°, 28.34°, 43.95°, 48.13°, 52.14° | 10 | 2.39 |
| 6.8 | 25.02°, 26.80°, 28.44°, 44.06°, 48.22°, 52.04° | 10 | 2.36 | |
| 9.4 | 26.63°, 44.06°, 52.14° | 7 | 2.27 | |
| 10.1 | 26.72°, 44.15°, 52.33° | 9 | 2.34 | |
| 90 | 4.7 | 25.06°, 26.62°, 28.28°, 36.58°, 44.06°, 48.10°, 52.26° | 10 | 2.39 |
| 6.8 | 25.30°, 26.63°, 28.47°, 44.10°, 52.04° | 10 | 2.30 | |
| 9.4 | 25.30°, 26.63°, 28.29°, 44.06°, 52.24° | 8 | 2.21 | |
| 10.1 | 26.72°, 44.15°, 52.33° | 10 | 2.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sanchez-Ramirez, E.A.; Arellano-Piña, R.; Hernandez-Perez, M.A.; Bello-Teodoro, S.; Garcia-Aguirre, K.K.; Sastré-Hernández, J.; Aguilar-Hernandez, J.R. From Structure to Optics: The pH-Temperature Interplay in Aqueous Solution CdS Nanoparticles. Nanomaterials 2026, 16, 3. https://doi.org/10.3390/nano16010003
Sanchez-Ramirez EA, Arellano-Piña R, Hernandez-Perez MA, Bello-Teodoro S, Garcia-Aguirre KK, Sastré-Hernández J, Aguilar-Hernandez JR. From Structure to Optics: The pH-Temperature Interplay in Aqueous Solution CdS Nanoparticles. Nanomaterials. 2026; 16(1):3. https://doi.org/10.3390/nano16010003
Chicago/Turabian StyleSanchez-Ramirez, Elvia Angelica, Ramón Arellano-Piña, M. A. Hernandez-Perez, Simón Bello-Teodoro, Karol Karla Garcia-Aguirre, J. Sastré-Hernández, and J. R. Aguilar-Hernandez. 2026. "From Structure to Optics: The pH-Temperature Interplay in Aqueous Solution CdS Nanoparticles" Nanomaterials 16, no. 1: 3. https://doi.org/10.3390/nano16010003
APA StyleSanchez-Ramirez, E. A., Arellano-Piña, R., Hernandez-Perez, M. A., Bello-Teodoro, S., Garcia-Aguirre, K. K., Sastré-Hernández, J., & Aguilar-Hernandez, J. R. (2026). From Structure to Optics: The pH-Temperature Interplay in Aqueous Solution CdS Nanoparticles. Nanomaterials, 16(1), 3. https://doi.org/10.3390/nano16010003