A Review on Recently Developed Antibacterial Composites of Inorganic Nanoparticles and Non-Hydrogel Polymers for Biomedical Applications
Abstract
1. Introduction
2. Fields of Application for NPs/Polymer Composites
3. Nanoparticles for Composites
3.1. Type and Composition of Filler
3.2. Origination of Nanoparticles
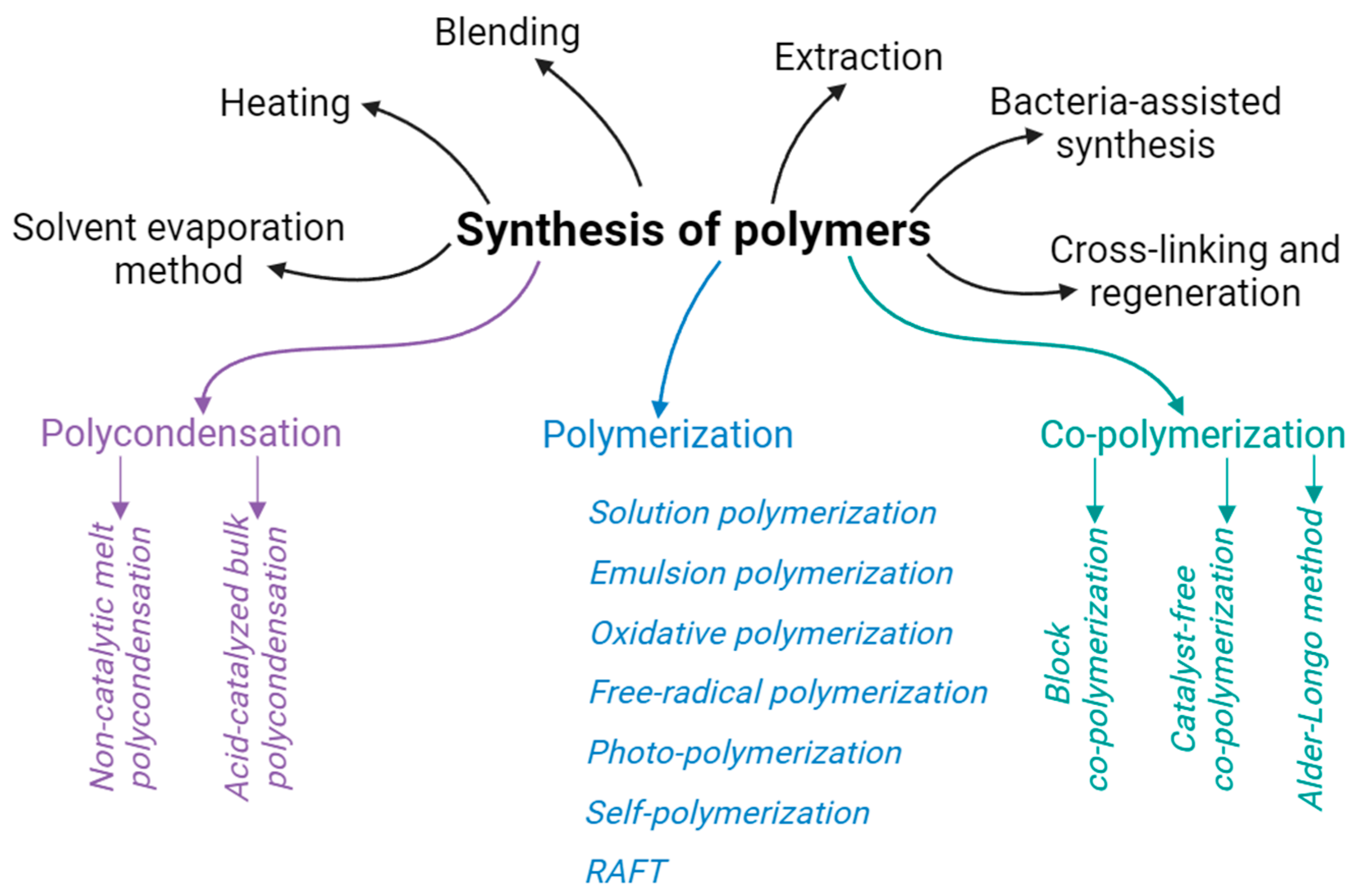
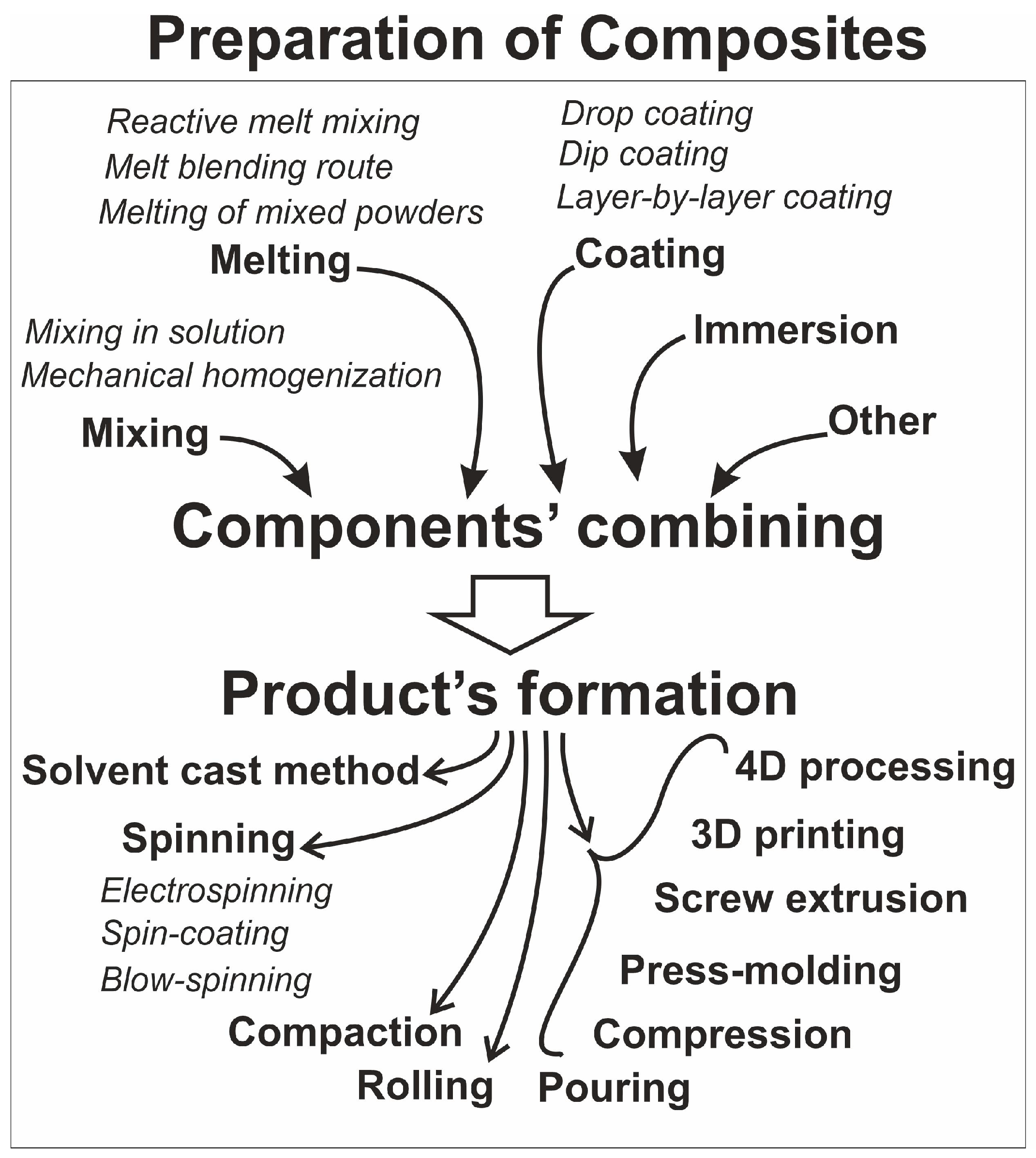
4. Polymers for Composites
4.1. Polymer Matrix Composition
4.2. Origination of the Polymer Matrix
5. Antibacterial Activity of NPs/Polymer Composites
5.1. Comparative Antibacterial Activity of the Composites
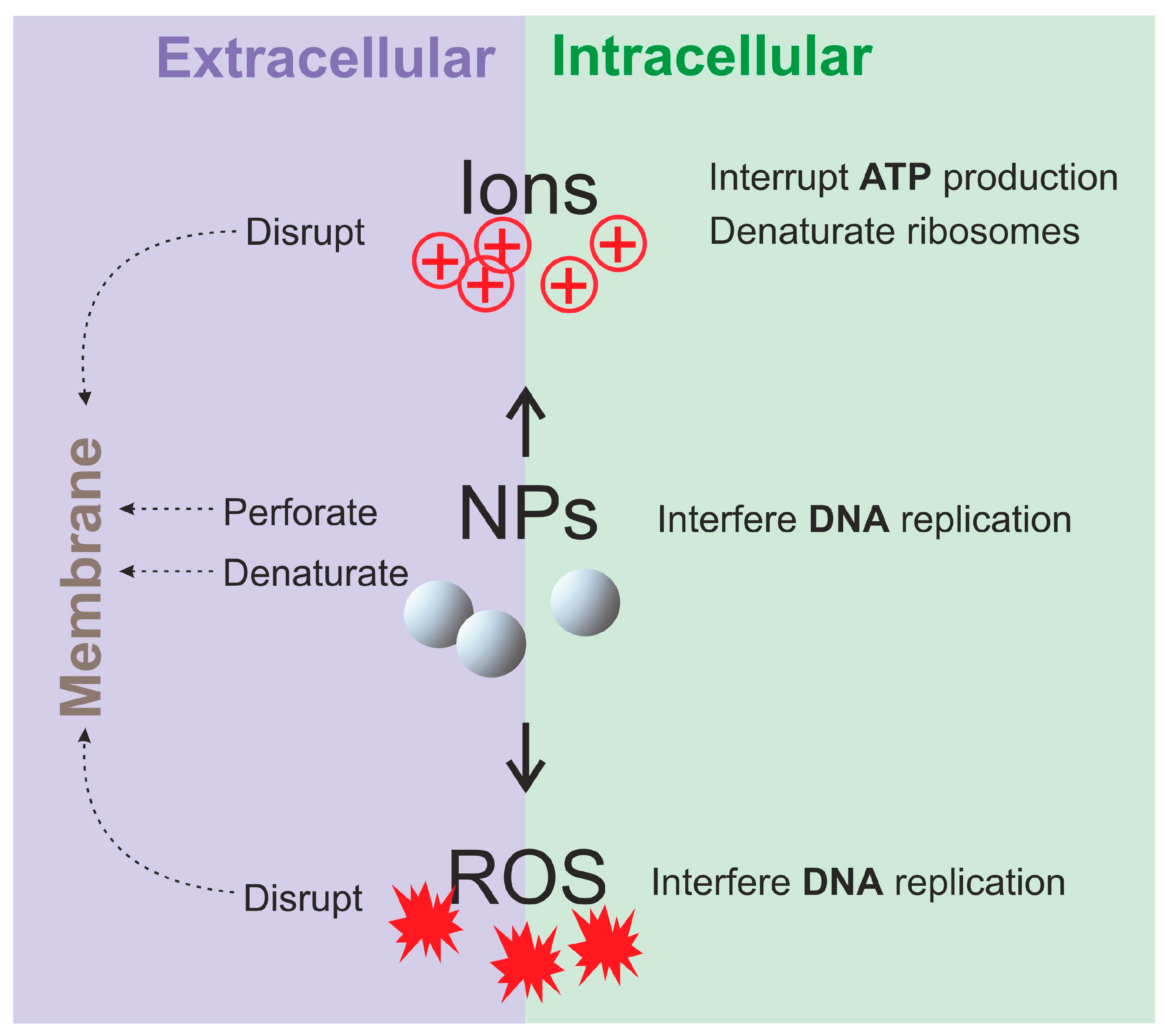
5.2. Antibacterial Mechanisms of the Composites
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ghazzy, A.; Naik, R.R.; Shakya, A.K. Metal–Polymer Nanocomposites: A Promising Approach to Antibacterial Materials. Polymers 2023, 15, 2167. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.-K.; Cheng, T.-M.; Chu, H.-L.; Tan, S.-H.; Kuo, J.-C.; Hsu, P.-H.; Su, C.-Y.; Chen, H.-M.; Lee, C.-M.; Kuo, T.-R. Metabolic Mechanism Investigation of Antibacterial Active Cysteine-Conjugated Gold Nanoclusters in Escherichia coli. ACS Sustain. Chem. Eng. 2019, 7, 15479–15486. [Google Scholar] [CrossRef]
- Arumugam, M.; Murugesan, B.; Chinnalagu, D.; Balasekar, P.; Cai, Y.; Sivakumar, P.M.; Rengasamy, G.; Chinniah, K.; Mahalingam, S. Electrospun Silk Fibroin and Collagen Composite Nanofiber Incorporated with Palladium and Platinum Nanoparticles for Wound Dressing Applications. J. Polym. Environ. 2024, 32, 2797–2817. [Google Scholar] [CrossRef]
- Balcucho, J.; Narváez, D.M.; Tarazona, N.A.; Castro-Mayorga, J.L. Microbially Synthesized Polymer-Metal Nanoparticles Composites as Promising Wound Dressings to Overcome Methicillin-Resistance Staphylococcus aureus Infections. Polymers 2023, 15, 920. [Google Scholar] [CrossRef]
- Chen, J.; Fan, L.; Yang, C.; Wang, S.; Zhang, M.; Xu, J.; Luo, S. Facile synthesis of Ag nanoparticles-loaded chitosan antibacterial nanocomposite and its application in polypropylene. Int. J. Biol. Macromol. 2020, 161, 1286–1295. [Google Scholar] [CrossRef]
- Cobos, M.; De-La-Pinta, I.; Quindós, G.; Fernández, M.; Fernández, M. Synthesis, Physical, Mechanical and Antibacterial Properties of Nanocomposites Based on Poly(vinyl alcohol)/Graphene Oxide–Silver Nanoparticles. Polymers 2020, 12, 723. [Google Scholar] [CrossRef]
- Elabbasy, M.T.; Algahtani, F.D.; Othman, M.S.; Ahmad, K.; Maysara, S.; Al-Najjar, M.A.A.; El-Morsy, M.A.; Menazea, A.A. Laser deposited ultra-thin silver nanoparticles on CMC-PVA blend film as sheet for wound dressings. Mater. Chem. Phys. 2024, 318, 129246. [Google Scholar] [CrossRef]
- Hashemi, S.S.; Mohammadi, A.A.; Kian, M.; Rafati, A.; Ghaedi, M.; Ghafari, B. Fabrication and evaluation of the regenerative effect of a polycaprolactone/chitosan nanofibrous scaffold containing bentonite nanoparticles in a rat model of deep second-degree burn injury. Iran. J. Basic Med. Sci. 2024, 27, 223–232. [Google Scholar] [CrossRef]
- Ibrahim, M.A.; Nasr, G.M.; Ahmed, R.M.; Kelany, N.A. Physical characterization, biocompatibility, and antimicrobial activity of polyvinyl alcohol/sodium alginate blend doped with TiO2 nanoparticles for wound dressing applications. Sci. Rep. 2024, 14, 5391. [Google Scholar] [CrossRef]
- Iswarya, S.; Bharathi, M.; Hariram, N.; Theivasanthi, T.; Gopinath, S.C.B. Solid polymer electrolyte and antimicrobial performance of Polyvinyl alcohol/Silver nanoparticles composite film. Results Chem. 2024, 7, 101431. [Google Scholar] [CrossRef]
- Kalaycıoğlu, Z.; Kahya, N.; Adımcılar, V.; Kaygusuz, H.; Torlak, E.; Akın-Evingür, G.; Erim, F.B. Antibacterial nano cerium oxide/chitosan/cellulose acetate composite films as potential wound dressing. Eur. Polym. J. 2020, 133, 109777. [Google Scholar] [CrossRef]
- Karami, R.; Moradipour, P.; Arkan, E.; Zarghami, R.; Rashidi, K.; Darvishi, E. Biocompatible nano-bandage modified with silver nanoparticles based on herbal for burn treatment. Polym. Bull. 2023, 81, 8285–8314. [Google Scholar] [CrossRef]
- Khaje, K.; Ghaee, A.; Ghaie, F.; Hosseini, I.; Seifi, S. Gelatin-based scaffold incorporating Ag nanoparticles decorated polydopamine nanoparticles for skin tissue engineering. Int. J. Polym. Mater. Polym. Biomater. 2024, 1–12. [Google Scholar] [CrossRef]
- Kumar, M.; Dhiman, S.K.; Bhat, R.; Saran, S. In situ green synthesis of AgNPs in bacterial cellulose membranes and antibacterial properties of the composites against pathogenic bacteria. Polym. Bull. 2023, 81, 6957–6978. [Google Scholar] [CrossRef]
- Sanchez, J.A.; Ibrahim, A.; Materon, L.; Parsons, J.G.; Alcoutlabi, M. Centrifugally spun PVP/ZnO composite fibers with different surfactants and their use as antibacterial agents. J. Appl. Polym. Sci. 2023, 140, e54314. [Google Scholar] [CrossRef]
- Shao, K.; Li, X.; Tan, B.; Wang, Q.; Guo, Z.; Liu, T.; Dong, L. Polyvinyl alcohol composite gel membrane modified by natural polysaccharide and waterborne polyurethane. Polym. Eng. Sci. 2023, 63, 3731–3742. [Google Scholar] [CrossRef]
- Amoee, S.; Monad, N.; Hamidinezhad, H.; Karimian, M. Fabrication and investigation antibacterial properties of green cross-linked PVA/GO/L-Arg-Cu electrospun nanofiber. Polym. Bull. 2024, 81, 10805–10839. [Google Scholar] [CrossRef]
- Bagheri, M.; Validi, M.; Gholipour, A.; Makvandi, P.; Sharifi, E. Chitosan nanofiber biocomposites for potential wound healing applications: Antioxidant activity with synergic antibacterial effect. Bioeng. Transl. Med. 2021, 7, e10254. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Kader, M.F.H.; Elabbasy, M.T.; Ahmed, M.K.; Menazea, A.A. Structural, morphological features, and antibacterial behavior of PVA/PVP polymeric blends doped with silver nanoparticles via pulsed laser ablation. J. Mater. Res. Technol. 2021, 13, 291–300. [Google Scholar] [CrossRef]
- El-Kader, M.F.H.A.; Elabbasy, M.T.; Adeboye, A.A.; Menazea, A.A. Nanocomposite of PVA/PVP blend incorporated by copper oxide nanoparticles via nanosecond laser ablation for antibacterial activity enhancement. Polym. Bull. 2021, 79, 9779–9795. [Google Scholar] [CrossRef]
- Guo, C.; Cheng, F.; Liang, G.; Zhang, S.; Jia, Q.; He, L.; Duan, S.; Fu, Y.; Zhang, Z.; Du, M. Copper-based polymer-metal–organic framework embedded with Ag nanoparticles: Long-acting and intelligent antibacterial activity and accelerated wound healing. Chem. Eng. J. 2022, 435, 134915. [Google Scholar] [CrossRef]
- Korniienko, V.; Husak, Y.; Diedkova, K.; Varava, Y.; Grebnevs, V.; Pogorielova, O.; Bērtiņš, M.; Korniienko, V.; Zandersone, B.; Ramanaviciene, A.; et al. Antibacterial Potential and Biocompatibility of Chitosan/Polycaprolactone Nanofibrous Membranes Incorporated with Silver Nanoparticles. Polymers 2024, 16, 1729. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xu, X.; Ma, X.; Cui, M.; Wang, X.; Chen, J.; Zhu, J.; Chen, J. Antimicrobial Nonisocyanate Polyurethane Foam Derived from Lignin for Wound Healing. ACS Appl. Bio. Mater. 2024, 7, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Huang, T.; Li, X.; Wang, J.; Lv, T.; Tan, W.; Gao, F.; Zhang, J.; Zhou, B. Synergistic antibacterial and wound-healing applications of an imidazole-based porous organic polymer encapsulated silver nanoparticles composite. Microporous Mesoporous Mater. 2022, 337, 111925. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, Y.; Zheng, S.; Zhong, L.; Xue, J. Preparation and properties of conductive bacterial cellulose-based graphene oxide-silver nanoparticles antibacterial dressing. Carbohydr. Polym. 2021, 257, 117671. [Google Scholar] [CrossRef]
- Zhang, Z.; Shi, L.; Chu, L.; Chen, P.; Sun, P.; Chen, Z.; Wei, L.; Zhou, B. Crown ether-based porous organic polymer encapsulated Ag2[Fe(CN)5NO] composite towards ultra-low dose efficient sterilization and wound healing application. Mater. Today Chem. 2023, 34, 101794. [Google Scholar] [CrossRef]
- Zhang, F.; Yao, Q.; Niu, Y.; Chen, X.; Zhou, H.; Bai, L.; Kong, Z.; Li, Y.; Cheng, H. In Situ Fabrication of Silver Nanoparticle-Decorated Polymeric Vesicles for Antibacterial Applications. Chem. Open 2024, 13, e202300223. [Google Scholar] [CrossRef]
- Jia, H.; Zeng, X.; Fan, S.; Cai, R.; Wang, Z.; Yuan, Y.; Yue, T. Silver nanoparticles anchored magnetic self-assembled carboxymethyl cellulose-ε-polylysine hybrids with synergetic antibacterial activity for wound infection therapy. Int. J. Biol. Macromol. 2022, 210, 703–715. [Google Scholar] [CrossRef]
- Zhu, G.; Sun, Z.; Hui, P.; Chen, W.; Jiang, X. Composite Film with Antibacterial Gold Nanoparticles and Silk Fibroin for Treating Multidrug-Resistant E. coli-Infected Wounds. ACS Biomater. Sci. Eng. 2020, 7, 1827–1835. [Google Scholar] [CrossRef]
- Dilmani, S.A.; Koç, S.; Erkut, T.S.; Gümüşderelioğlu, M. Polymer-clay nanofibrous wound dressing materials containing different boron compounds. J. Trace Elem. Med. Biol. 2024, 83, 127408. [Google Scholar] [CrossRef]
- Wang, B.; Wang, H.; Wang, Z.; Tang, J.; Yuan, X.; Zhang, Y.; Chen, H.; Yu, W.; Song, M. Preparation of AgBrNPs@copolymer-decorated chitosan with synergistic antibacterial activity. Mater. Today Commun. 2023, 37, 107482. [Google Scholar] [CrossRef]
- Guimarães, M.L.; da Silva, F.A.G.; da Costa, M.M.; de Oliveira, H.P. Coating of conducting polymer-silver nanoparticles for antibacterial protection of Nile tilapia skin xenografts. Synth. Met. 2022, 287, 117055. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, Z.; Wang, J.; Sun, X.; Yang, X.; Liu, G. Mussel-inspired electroactive, antibacterial and antioxidative composite membranes with incorporation of gold nanoparticles and antibacterial peptides for enhancing skin wound healing. J. Biol. Eng. 2024, 18, 3. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Ma, C.; Zhou, C.; Li, Z.; Li, R. Preparation and Properties of Water-Resistant Antibacterial Curcumin/Silver Composite Nanofiber. Fibers Polym. 2023, 24, 3821–3832. [Google Scholar] [CrossRef]
- Miralaei, N.; Mohammadimehr, M.; Farazin, A.; Ghasemi, A.H.; Bargozini, F. Design, fabrication, evaluation, and in vitro study of green biomaterial and antibacterial polymeric biofilms of polyvinyl alcohol/tannic acid/CuO/SiO2. J. Mech. Behav. Biomed. Mater. 2023, 14, 106219. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, K.; Dezvare, Y.; Kamyab, S.; Amirian, J.; Brangule, A.; Bandere, D. Development of Composite Sponge Scaffolds Based on Carrageenan (CRG) and Cerium Oxide Nanoparticles (CeO2 NPs) for Hemostatic Applications. Biomimetics 2023, 8, 409. [Google Scholar] [CrossRef]
- Asha, A.B.; Ounkaew, A.; Peng, Y.-Y.; Gholipour, M.R.; Ishihara, K.; Liu, Y.; Narain, R. Bioinspired antifouling and antibacterial polymer coating with intrinsic self-healing property. Biomater. Sci. 2023, 11, 128–139. [Google Scholar] [CrossRef]
- Chen, K.; Wang, F.; Liu, S.; Wu, X.; Xu, L.; Zhang, D. In situ reduction of silver nanoparticles by sodium alginate to obtain silver-loaded composite wound dressing with enhanced mechanical and antimicrobial property. Int. J. Biol. Macromol. 2020, 148, 501–509. [Google Scholar] [CrossRef]
- Chen, Z.; Sun, J.; Zhan, Z.; Yuan, Y.; Tian, X.; Jin, J.; Wu, W.; Ayikanbaier, K. Multifunctional flexible strain sensor based on three-dimensional core-shell structures of silver nanoparticles/natural rubber. J. Appl. Polym. Sci. 2024, 141, e55436. [Google Scholar] [CrossRef]
- Haladu, S.A.; Elsayed, K.A.; Olanrewaju Alade, I.; Alheshibri, M.; Al Baroot, A.; Ali, S.A.; Kotb, E.; Manda, A.A.; Ul-Hamid, A.; Dafalla, H.D.M.; et al. Laser-assisted fabrication of silver quantum dots/polyaspartate polymer composite for antimicrobial applications. Opt. Laser Technol. 2022, 152, 108122. [Google Scholar] [CrossRef]
- Li, H.; Luo, R.; Qu, J. Poly(methyl methacrylate-co-butyl acrylate) copolymer/Ag nanocomposites prepared by latex mixing for multifunctional coatings. Polym. Compos. 2023, 45, 2795–2808. [Google Scholar] [CrossRef]
- Liu, F.; Cheng, X.; Xiao, L.; Wang, Q.; Yan, K.; Su, Z.; Wang, L.; Ma, C.; Wang, Y. Inside-outside Ag nanoparticles-loaded polylactic acid electrospun fiber for long-term antibacterial and bone regeneration. Int. J. Biol. Macromol. 2021, 167, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Quintero-Quiroz, C.; Botero, L.E.; Zárate-Triviño, D.; Acevedo-Yepes, N.; Escobar, J.S.; Pérez, V.Z.; Cruz Riano, L.J. Synthesis and characterization of a silver nanoparticle-containing polymer composite with antimicrobial abilities for application in prosthetic and orthotic devices. Biomater. Res. 2020, 24, 13. [Google Scholar] [CrossRef] [PubMed]
- Ragab, H.M.; Diab, N.S.; AlElaimi, M.; Alghamdi, A.M.; Farea, M.O.; Farea, A. Fabrication and Characterization of Silver Nanoparticle-Doped Chitosan/Carboxymethyl Cellulose Nanocomposites for Optoelectronic and Biological Applications. ACS Omega 2024, 9, 22112–22122. [Google Scholar] [CrossRef]
- Sun, D.; Turner, J.; Jiang, N.; Zhu, S.; Zhang, L.; Falzon, B.G.; McCoy, C.P.; Maguire, P.; Mariotti, D.; Sun, D. Atmospheric pressure microplasma for antibacterial silver nanoparticle/chitosan nanocomposites with tailored properties. Compos. Sci. Technol. 2020, 186, 107911. [Google Scholar] [CrossRef]
- Teper, P.; Oleszko-Torbus, N.; Bochenek, M.; Hajduk, B.; Kubacki, J.; Jałowiecki, Ł.; Płaza, G.; Kowalczuk, A.; Mendrek, B. Hybrid nanolayers of star polymers and silver nanoparticles with antibacterial activity. Colloids Surf. B Biointerfaces 2022, 213, 112404. [Google Scholar] [CrossRef]
- Vidakis, N.; Michailidis, N.; David, C.; Papadakis, V.; Argyros, A.; Sagris, D.; Spiridaki, M.; Mountakis, N.; Nasikas, N.K.; Petousis, M. Polyvinyl alcohol as a reduction agent in material extrusion additive manufacturing for the development of pharmaceutical-grade polypropylene/silver nanocomposites with antibacterial properties. Mater. Today Commun. 2024, 39, 109366. [Google Scholar] [CrossRef]
- Wang, B.; Guo, W.; Liu, X.; He, Y.; Song, P.; Wang, R. Fabrication of silver-decorated popcorn-like polymeric nanoparticles for enhanced antibacterial activity. Appl. Surf. Sci. 2020, 522, 146318. [Google Scholar] [CrossRef]
- Wang, B.-B.; Quan, Y.-H.; Xu, Z.-M.; Zhao, Q. Preparation of highly effective antibacterial coating with polydopamine/chitosan/silver nanoparticles via simple immersion. Prog. Org. Coat. 2020, 149, 105967. [Google Scholar] [CrossRef]
- Wang, X.; Yan, H.; Hang, R.; Shi, H.; Wang, L.; Ma, J.; Liu, X.; Yao, X. Enhanced anticorrosive and antibacterial performances of silver nanoparticles/polyethyleneimine/MAO composite coating on magnesium alloys. J. Mater. Res. Technol. 2021, 11, 2354–2364. [Google Scholar] [CrossRef]
- Wang, J.-L.; Wang, S.; Zhong, P.; Chen, Y.; Zhao, Z.; Liu, W. Multimechanism Long-Term Antibacterial Coating with Simultaneous Antifouling, Contact-Active, and Release-Kill Properties. ACS Appl. Polym. Mater. 2024, 6, 5726–5737. [Google Scholar] [CrossRef]
- Xiong, C.; Wang, T.; Zhang, Y.; Duan, C.; Zhang, Z.; Zhou, Q.; Xiong, Q.; Zhao, M.; Wang, B.; Ni, Y. Multifunctional Conductive Material Based on Intelligent Porous Paper Used in Conjunction with a Vitrimer for Electromagnetic Shielding, Sensing, Joule Heating, and Antibacterial Properties. ACS Appl. Mater. Interfaces 2023, 15, 33763–33773. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, V.V.; Chausov, D.N.; Serov, D.A.; Kozlov, V.A.; Ivashkin, P.I.; Pishchalnikov, R.Y.; Uvarov, O.V.; Vedunova, M.V.; Semenova, A.A.; Lisitsyn, A.B.; et al. A Novel Biodegradable Composite Polymer Material Based on PLGA and Silver Oxide Nanoparticles with Unique Physicochemical Properties and Biocompatibility with Mammalian Cells. Materials 2021, 14, 6915. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Kang, L.; Zhang, B.; Liu, L.; Sun, H.; Shen, X.; Su, F.; Li, S. Biodegradable nanocomposites prepared from poly(butylene adipate terephthalate) and silver nanoparticles for applications as disposable medical equipments. Polym. Adv. Technol. 2023, 35, e6212. [Google Scholar] [CrossRef]
- Arif, W.; Rana, N.; Saleem, I.; Tanweer, T.; Khan, M.; Alshareef, S.; Sheikh, H.; Alaryani, F.; Al-Kattan, M.; Alatawi, H.; et al. Antibacterial Activity of Dental Composite with Ciprofloxacin Loaded Silver Nanoparticles. Molecules 2022, 27, 7182. [Google Scholar] [CrossRef]
- Cao, B.; Zhang, J.; Ma, Y.; Wang, Y.; Li, Y.; Wang, R.; Cao, D.; Yang, Y.; Zhang, R. Dual-Polymer Functionalized Melanin-AgNPs Nanocomposite with Hydroxyapatite Binding Ability to Penetrate and Retain in Biofilm Sequentially Treating Periodontitis. Small 2024, 2400771. [Google Scholar] [CrossRef]
- Datta, L.P.; Dutta, D.; Mukherjee, R.; Das, T.K.; Biswas, S. Polyoxometalate-Polymer Directed Macromolecular Architectonics of Silver Nanoparticles as Effective Antimicrobials. Chem. Asian J. 2024, 19, e202400344. [Google Scholar] [CrossRef]
- Mogharbel, A.T.; Ibarhiam, S.F.; Alqahtani, A.M.; Attar, R.M.S.; Alshammari, K.F.; Bamaga, M.A.; Al-Qahtani, S.D.; El-Metwaly, N.M. Plasma-assisted in-situ preparation of silver nanoparticles and polypyrrole toward superhydrophobic, antimicrobial and electrically conductive nonwoven fabrics from recycled polyester waste. J. Ind. Eng. Chem. 2023, 127, 356–364. [Google Scholar] [CrossRef]
- Abdelaziz, D.; Hefnawy, A.; Al-Wakeel, E.; El-Fallal, A.; El-Sherbiny, I.M. New biodegradable nanoparticles-in-nanofibers based membranes for guided periodontal tissue and bone regeneration with enhanced antibacterial activity. J. Adv. Res. 2021, 28, 51–62. [Google Scholar] [CrossRef]
- Guo, W.; Liu, W.; Xu, L.; Feng, P.; Zhang, Y.; Yang, W.; Shuai, C. Halloysite nanotubes loaded with nano silver for the sustained-release of antibacterial polymer nanocomposite scaffolds. J. Mater. Sci. Technol. 2020, 46, 237–247. [Google Scholar] [CrossRef]
- Huang, X.; Ge, M.; Wang, H.; Liang, H.; Meng, N.; Zhou, N. Functional modification of polydimethylsiloxane nanocomposite with silver nanoparticles-based montmorillonite for antibacterial applications. Colloids Surf. A Physicochem. Eng. Asp. 2022, 642, 128666. [Google Scholar] [CrossRef]
- Jatoi, A.W.; Kim, I.S.; Ni, Q.-Q. Cellulose acetate nanofibers embedded with AgNPs anchored TiO2 nanoparticles for long term excellent antibacterial applications. Carbohydr. Polym. 2019, 207, 640–649. [Google Scholar] [CrossRef] [PubMed]
- Jatoi, A.W.; Ogasawara, H.; Kim, I.S.; Ni, Q.-Q. Cellulose acetate/multi-wall carbon nanotube/Ag nanofiber composite for antibacterial applications. Mater. Sci. Eng. C 2020, 110, 110679. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.U.A.; Abd Razak, S.I.; Mehboob, H.; Abdul Kadir, M.R.; Anand, T.J.S.; Inam, F.; Shah, S.A.; Abdel-Haliem, M.E.F.; Amin, R. Synthesis and Characterization of Silver-Coated Polymeric Scaffolds for Bone Tissue Engineering: Antibacterial and In Vitro Evaluation of Cytotoxicity and Biocompatibility. ACS Omega 2021, 6, 4335–4346. [Google Scholar] [CrossRef] [PubMed]
- Lipovka, A.; Fatkullin, M.; Shchadenko, S.; Petrov, I.; Chernova, A.; Plotnikov, E.; Menzelintsev, V.; Li, S.; Qiu, L.; Cheng, C.; et al. Textile Electronics with Laser-Induced Graphene/Polymer Hybrid Fibers. ACS Appl. Mater. Interfaces 2023, 15, 38946–38955. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Ling, J.; Yang, L.-Y.; Ouyang, X.-k.; Wang, N. Chitosan-based carbon nitride-polydopamine-silver composite dressing with antibacterial properties for wound healing. Carbohydr. Polym. 2023, 303, 120436. [Google Scholar] [CrossRef]
- Sarıipek, F.B.; Sevgi, F.; Dursun, S. Preparation of poly(ε-caprolactone) nanofibrous mats incorporating graphene oxide-silver nanoparticle hybrid composite by electrospinning method for potential antibacterial applications. Colloids Surf. A Physicochem. Eng. Asp. 2022, 653, 129969. [Google Scholar] [CrossRef]
- Shuai, C.; Xu, Y.; Feng, P.; Wang, G.; Xiong, S.; Peng, S. Antibacterial polymer scaffold based on mesoporous bioactive glass loaded with in situ grown silver. Chem. Eng. J. 2019, 374, 304–315. [Google Scholar] [CrossRef]
- Shuai, C.; Liu, G.; Yang, Y.; Qi, F.; Peng, S.; Yang, W.; He, C.; Wang, G.; Qian, G. A strawberry-like Ag-decorated barium titanate enhances piezoelectric and antibacterial activities of polymer scaffold. Nano Energy 2020, 74, 104825. [Google Scholar] [CrossRef]
- Su, X.; Zhai, Y.; Jia, C.; Xu, Z.; Luo, D.; Pan, Z.; Xiang, H.; Yu, S.; Zhu, L.; Zhu, M. Improved Antibacterial Properties of Polylactic Acid-Based Nanofibers Loaded with ZnO–Ag Nanoparticles through Pore Engineering. ACS Appl. Mater. Interfaces 2023, 15, 42920–42929. [Google Scholar] [CrossRef]
- Wang, B.; Moon, J.R.; Ryu, S.; Park, K.D.; Kim, J.H. Antibacterial 3D graphene composite gel with polyaspartamide and tannic acid containing in situ generated Ag nanoparticle. Polym. Compos. 2020, 41, 2578–2587. [Google Scholar] [CrossRef]
- Zhong, Q.; Long, H.; Hu, W.; Shi, L.; Zan, F.; Xiao, M.; Tan, S.; Ke, Y.; Wu, G.; Chen, H. Dual-Function Antibacterial Micelle via Self-Assembling Block Copolymers with Various Antibacterial Nanoparticles. ACS Omega 2020, 5, 8523–8533. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Ji, H.; Liu, L.; Feng, S.; Fu, Y.; Yang, Y.; Lü, C. Mussel-inspired coordination functional polymer brushes-decorated rGO-stabilized silver nanoparticles composite for antibacterial application. Polym. Chem. 2020, 11, 2822–2830. [Google Scholar] [CrossRef]
- Burmistrov, D.E.; Simakin, A.V.; Smirnova, V.V.; Uvarov, O.V.; Ivashkin, P.I.; Kucherov, R.N.; Ivanov, V.E.; Bruskov, V.I.; Sevostyanov, M.A.; Baikin, A.S.; et al. Bacteriostatic and Cytotoxic Properties of Composite Material Based on ZnO Nanoparticles in PLGA Obtained by Low Temperature Method. Polymers 2021, 14, 49. [Google Scholar] [CrossRef] [PubMed]
- Chausov, D.N.; Burmistrov, D.E.; Kurilov, A.D.; Bunkin, N.F.; Astashev, M.E.; Simakin, A.V.; Vedunova, M.V.; Gudkov, S.V. New Organosilicon Composite Based on Borosiloxane and Zinc Oxide Nanoparticles Inhibits Bacterial Growth, but Does Not Have a Toxic Effect on the Development of Animal Eukaryotic Cells. Materials 2021, 14, 6281. [Google Scholar] [CrossRef]
- Galli, R.; Hall, M.C.; Breitenbach, E.R.; Colpani, G.L.; Zanetti, M.; de Mello, J.M.M.; Silva, L.L.; Fiori, M.A. Antibacterial polyethylene–Ethylene vinyl acetate polymeric blend by incorporation of zinc oxide nanoparticles. Polym. Test. 2020, 89, 106554. [Google Scholar] [CrossRef]
- Hezma, A.M.; Rajeh, A.; Mannaa, M.A. An insight into the effect of zinc oxide nanoparticles on the structural, thermal, mechanical properties and antimicrobial activity of Cs/PVA composite. Colloids Surf. A Physicochem. Eng. Asp. 2019, 581, 123821. [Google Scholar] [CrossRef]
- Ishak, M.Q.H.; Shankar, P.; Turabayev, M.E.; Kondo, T.; Honda, M.; Gurbatov, S.O.; Okamura, Y.; Iwamori, S.; Kulinich, S.A. Biodegradable Polymer Nanosheets Incorporated with Zn-Containing Nanoparticles for Biomedical Applications. Materials 2022, 15, 8101. [Google Scholar] [CrossRef]
- Khlifi, K.; Atallah, M.S.; Cherif, I.; Karkouch, I.; Barhoumi, N.; Attia-Essaies, S. Synthesis of ZnO nanoparticles and study of their influence on the mechanical properties and antibacterial activity of PMMA/ZnO composite for orthotic devices. Surf. Interfaces 2023, 41, e6212. [Google Scholar]
- Norouzi, M.A.; Montazer, M.; Harifi, T.; Karimi, P. Flower buds like PVA/ZnO composite nanofibers assembly: Antibacterial, in vivo wound healing, cytotoxicity and histological studies. Polym. Test. 2021, 93, 106914. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, X.; Fang, X.; Sun, L.; Wang, X.; Chen, H.; Zhu, N. Antimicrobial hydrocolloid composite sponge with on-demand dissolving property, consisting mainly of zinc oxide nanoparticles, hydroxypropyl chitosan, and polyvinyl alcohol. J. Polym. Eng. 2023, 43, 810–819. [Google Scholar] [CrossRef]
- Banerjee, A.; Roy, P.; Chakraborty, J.; Majumder, M. Environmentally degradable curcumin/zinc oxide nanoparticles-incorporated polycaprolactone films for use as top-sheets in feminine sanitary hygiene napkins. Mater. Today Commun. 2024, 40, 109452. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Li, C.; Cao, J.; He, E.; Wu, X.; Wang, F.; Wang, L. Facile preparation PCL/modified nano ZnO organic-inorganic composite and its application in antibacterial materials. J. Polym. Res. 2020, 27, 78. [Google Scholar] [CrossRef]
- Abouelnaga, A.M.; El Nahrawy, A.M. Spectroscopic investigation, dielectric and antimicrobial properties of chitin-cellulose@ZnO/CuO conductive nanocomposites. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2024, 320, 124646. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Lin, C.; Wang, Y.; Ma, J.; Wang, X.; Yao, X.; Tang, B. Preparation of Zn doped mesoporous silica nanoparticles (Zn-MSNs) for the improvement of mechanical and antibacterial properties of dental resin composites. Dent. Mater. 2020, 36, 794–807. [Google Scholar] [CrossRef]
- Toh-ae, P.; Lee-Nip, R.; Nakaramontri, Y. Releasing of zinc ions from modified zinc oxide surfaces for improvement chemical crosslinks and antibacterial properties of acrylonitrile butadiene rubber films. Express Polym. Lett. 2023, 17, 944–963. [Google Scholar] [CrossRef]
- Abdelghany, A.M.; Elamin, N.Y.; Younis, S.; Ayaad, D.M. Polyvinyl pyrrolidone/carboxymethyl cellulose (PVP/CMC) polymer composites containing CuO nanoparticles synthesized via laser ablation in liquids. J. Mol. Liq. 2024, 403, 124857. [Google Scholar] [CrossRef]
- Fahmy, T.; Sarhan, A. Investigation of optical properties and antibacterial activity of chitosan copper nanoparticle composites. Mater. Technol. 2022, 37, 2400–2413. [Google Scholar] [CrossRef]
- Glazkova, E.; Bakina, O.; Rodkevich, N.; Mosunov, A.; Evstigneev, M.; Evstigneev, V.; Klimenko, V.; Lerner, M. Antibacterial Properties of PMMA Functionalized with CuFe2O4/Cu2O/CuO Nanoparticles. Coatings 2022, 12, 957. [Google Scholar] [CrossRef]
- Shah, B.A.; Sardar, A.; Peng, W.; Din, S.T.U.; Hamayoun, S.; Li, S.; Yuan, B. Photoresponsive CuS@polyaniline nanocomposites: An excellent synthetic bactericide against several multidrug-resistant pathogenic strains. Inorg. Chem. Front. 2023, 10, 6339–6356. [Google Scholar] [CrossRef]
- Jardón-Maximino, N.; Cadenas-Pliego, G.; Ávila-Orta, C.A.; Comparán-Padilla, V.E.; Lugo-Uribe, L.E.; Pérez-Alvarez, M.; Tavizón, S.F.; Santillán, G.d.J.S. Antimicrobial Property of Polypropylene Composites and Functionalized Copper Nanoparticles. Polymers 2021, 13, 1694. [Google Scholar] [CrossRef] [PubMed]
- Govindasamy, G.A.; Sreekantan, S.; Saharudin, K.A.; Poliah, R.; Ong, M.T.; Thavamany, P.J.; Sahgal, G.; Tan, A.A. Composition-Dependent Physicochemical and Bactericidal Properties of Dual Cu-TiO2 Nanoparticles Incorporated in Polypropylene. BioNanoScience 2024, 14, 770–782. [Google Scholar] [CrossRef]
- Karbowniczek, J.E.; Berniak, K.; Knapczyk-Korczak, J.; Williams, G.; Bryant, J.A.; Nikoi, N.D.; Banzhaf, M.; de Cogan, F.; Stachewicz, U. Strategies of nanoparticles integration in polymer fibers to achieve antibacterial effect and enhance cell proliferation with collagen production in tissue engineering scaffolds. J. Colloid Interface Sci. 2023, 650, 1371–1381. [Google Scholar] [CrossRef] [PubMed]
- Sebak, M.A.; Qahtan, T.F.; Asnag, G.M.; Abdallah, E.M. The Role of TiO2 Nanoparticles in the Structural, Thermal and Electrical Properties and Antibacterial Activity of PEO/PVP Blend for Energy Storage and Antimicrobial Application. J. Inorg. Organomet. Polym. Mater. 2022, 32, 4715–4728. [Google Scholar] [CrossRef]
- Balan, R.; Gayathri, V. In-vitro and antibacterial activities of novel POT/TiO2/PCL composites for tissue engineering and biomedical applications. Polym. Bull. 2021, 79, 4269–4286. [Google Scholar] [CrossRef]
- Du, Y.; Chen, M.; Wang, B.; Chai, Y.; Wang, L.; Li, N.; Zhang, Y.; Liu, Z.; Guo, C.; Jiang, X.; et al. TiO2/Polystyrene Nanocomposite Antibacterial Material as a Hemoperfusion Adsorbent for Efficient Bilirubin Removal and Prevention of Bacterial Infection. ACS Biomater. Sci. Eng. 2024, 10, 1494–1506. [Google Scholar] [CrossRef]
- Babers, N.; El-Sherbiny, M.G.D.; El-Shazly, M.; Kamel, B.M. Mechanical and antibacterial properties of hybrid polymers composite reinforcement for biomedical applications. J. Biomater. Sci. Polym. Ed. 2023, 35, 85–108. [Google Scholar] [CrossRef]
- Shaalan, M.; Vykydalová, A.; Švajdlenková, H.; Kroneková, Z.; Marković, Z.M.; Kováčová, M.; Špitálský, Z. Antibacterial activity of 3D printed thermoplastic elastomers doped with carbon quantum dots for biomedical applications. Polym. Bull. 2024, 81, 13009–13025. [Google Scholar] [CrossRef]
- Joy, A.; Megha, M.; Mohan, C.C.; Thomas, J.; Bhat, S.G.; Muthuswamy, S. Novel polycaprolactone-based biomimetic grafts enriched with graphene oxide and cerium oxide: Exploring improved osteogenic potential. Mater. Today Chem. 2024, 3, 102031. [Google Scholar] [CrossRef]
- Kafali, M.; Şahinoğlu, O.B.; Tufan, Y.; Orsel, Z.C.; Aygun, E.; Alyuz, B.; Saritas, E.U.; Erdem, E.Y.; Ercan, B. Antibacterial properties and osteoblast interactions of microfluidically synthesized chitosan–SPION composite nanoparticles. J. Biomed. Mater. Res. Part A 2023, 111, 1662–1677. [Google Scholar] [CrossRef]
- Sadeghi-Ghadi, Z.; Behjou, N.; Ebrahimnejad, P.; Mahkam, M.; Goli, H.R.; Lam, M.; Nokhodchi, A. Improving Antibacterial Efficiency of Curcumin in Magnetic Polymeric Nanocomposites. J. Pharm. Innov. 2022, 18, 13–28. [Google Scholar] [CrossRef]
- Guo, W.; Zhou, B.; Zou, Y.; Lu, X. 4D Printed Poly(l-lactide)/(FeCl3–TA/MgO) Composite Scaffolds with Near-Infrared Light-Induced Shape-Memory Effect and Antibacterial Properties. Adv. Eng. Mater. 2023, 26, 2301381. [Google Scholar] [CrossRef]
- Ahangaran, F.; Navarchian, A.H. Towards the development of self-healing and antibacterial dental nanocomposites via incorporation of novel acrylic microcapsules. Dent. Mater. 2022, 38, 858–873. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xu, Z.; Guo, Y.; Zhang, H.; Qiu, Y.; Li, J.; Ma, D.; Li, Z.; Zhen, P.; Liu, B.; et al. Novel core–shell CHX/ACP nanoparticles effectively improve the mechanical, antibacterial and remineralized properties of the dental resin composite. Dent. Mater. 2021, 37, 636–647. [Google Scholar] [CrossRef]
- Yao, S.; Qin, L.; Wang, Z.; Zhu, L.; Zhou, C.; Wu, J. Novel nanoparticle-modified multifunctional microcapsules with self-healing and antibacterial activities for dental applications. Dent. Mater. 2022, 38, 1301–1315. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Z.; Wang, T.; Tang, W.; Li, T.; Xu, H.; Sun, H.; Lin, Y.; Tonin, B.S.H.; Ye, Z.; et al. Bioactive Dental Resin Composites with MgO Nanoparticles. ACS Biomater. Sci. Eng. 2023, 9, 4632–4645. [Google Scholar] [CrossRef]
- Benedini, L.; Laiuppa, J.; Santillán, G.; Baldini, M.; Messina, P. Antibacterial alginate/nano-hydroxyapatite composites for bone tissue engineering: Assessment of their bioactivity, biocompatibility, and antibacterial activity. Mater. Sci. Eng. C 2020, 115, 111101. [Google Scholar] [CrossRef]
- Ahmed, S.; Hussain, R.; Khan, A.; Batool, S.A.; Mughal, A.; Nawaz, M.H.; Irfan, M.; Wadood, A.; Avcu, E.; Rehman, M.A.U. 3D Printing Assisted Fabrication of Copper–Silver Mesoporous Bioactive Glass Nanoparticles Reinforced Sodium Alginate/Poly(vinyl alcohol) Based Composite Scaffolds: Designed for Skin Tissue Engineering. ACS Appl. Bio. Mater. 2023, 6, 5052–5066. [Google Scholar] [CrossRef]
- Shalini, A.; Deepa, K.; Meenakshi, S.; Al-Ansari, M.M.; Alhumaid, L.; Rajendran, K.; Dixit, S.; Lo, H.M. A study on antibacterial and anti-inflammatory activity of xylitol-based polymeric nano-bioactive glass nanocomposites. Polym. Adv. Technol. 2024, 35, e6414. [Google Scholar] [CrossRef]
- Astashev, M.E.; Sarimov, R.M.; Serov, D.A.; Matveeva, T.A.; Simakin, A.V.; Ignatenko, D.N.; Burmistrov, D.E.; Smirnova, V.V.; Kurilov, A.D.; Mashchenko, V.I.; et al. Antibacterial behavior of organosilicon composite with nano aluminum oxide without influencing animal cells. React. Funct. Polym. 2022, 170, 105143. [Google Scholar] [CrossRef]
- Bashal, A.H.; Khalil, K.D.; Abu-Dief, A.M.; El-Atawy, M.A. Cobalt oxide-chitosan based nanocomposites: Synthesis, characterization and their potential pharmaceutical applications. Int. J. Biol. Macromol. 2023, 253, 126856. [Google Scholar] [CrossRef] [PubMed]
- Chacko, S.K.; Balakrishnan, R.; Kalarikkal, N.; Thomas, N.G. Ternary Fiber Mats of PVDF-HFP/Cellulose/LiFe5O8 Nanoparticles with Enhanced Electric, Magnetoelectric, and Antibacterial Properties: A Promising Approach for Magnetic and Electric Field-Responsive Antibacterial Coatings. ACS Appl. Polym. Mater. 2024, 6, 1429–1438. [Google Scholar] [CrossRef]
- Raja, T.; Al-Otibi, F.O.; Alharbi, R.I.; Mohanavel, V.; Velmurugan, P.; Karthikeyan, S.; Perumal, M.; Basavegowda, N. A novel study of biological and structural analysis on Cissus quadrangularis fiber-reinforced CaO particulates epoxy composite for biomedical application. J. Mater. Res. Technol. 2023, 27, 692–702. [Google Scholar] [CrossRef]
- Silva, C.; Bobillier, F.; Canales, D.; Antonella Sepúlveda, F.; Cament, A.; Amigo, N.; Rivas, L.M.; Ulloa, M.T.; Reyes, P.; Ortiz, J.A.; et al. Mechanical and Antimicrobial Polyethylene Composites with CaO Nanoparticles. Polymers 2020, 12, 2132. [Google Scholar] [CrossRef] [PubMed]
- Sathiyaseelan, A.; Zhang, X.; Lin, J.; Wang, M.-H. In situ, synthesis of chitosan fabricated tellurium nanoparticles for improved antimicrobial and anticancer applications. Int. J. Biol. Macromol. 2024, 258, 128778. [Google Scholar] [CrossRef]
- Tamta, A.; Kumar, R.; Gouri, V.; Joshi, R.; Chandra, B.; Kandpal, N.D. Synthesis, characterization and biological activities of NiO-cellulose nanocomposite. Curr. Chem. Lett. 2024, 13, 593–602. [Google Scholar] [CrossRef]
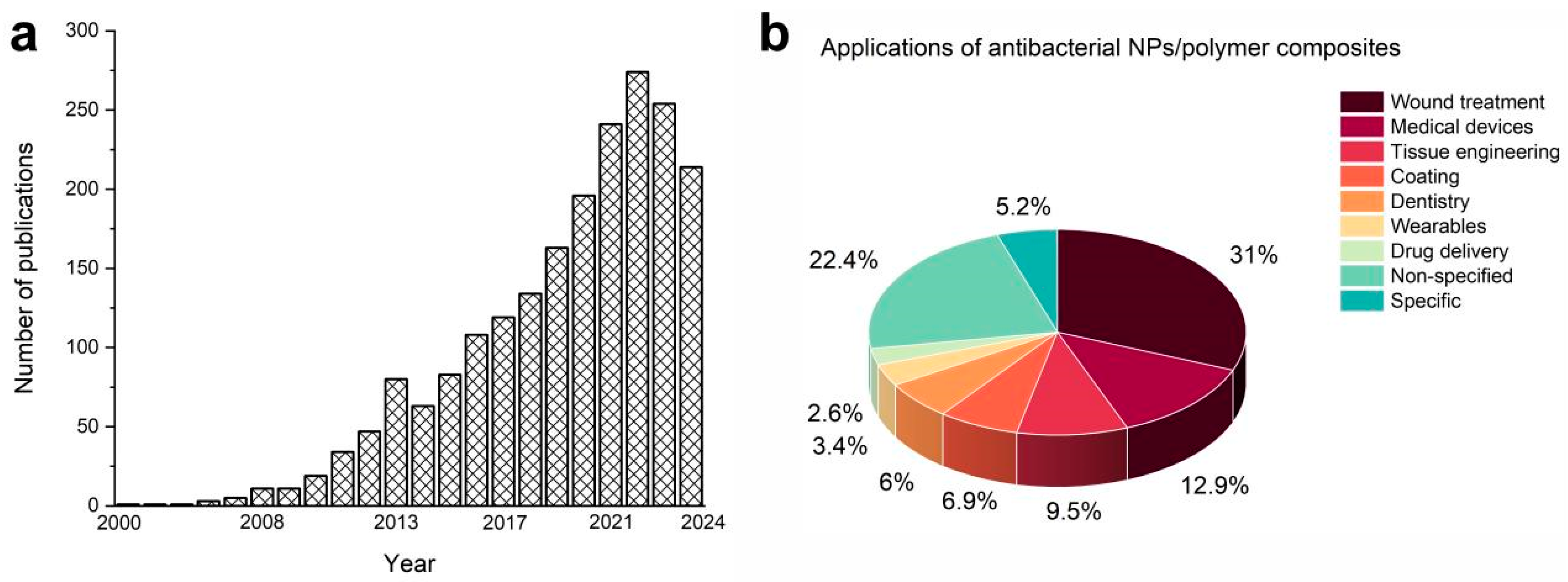

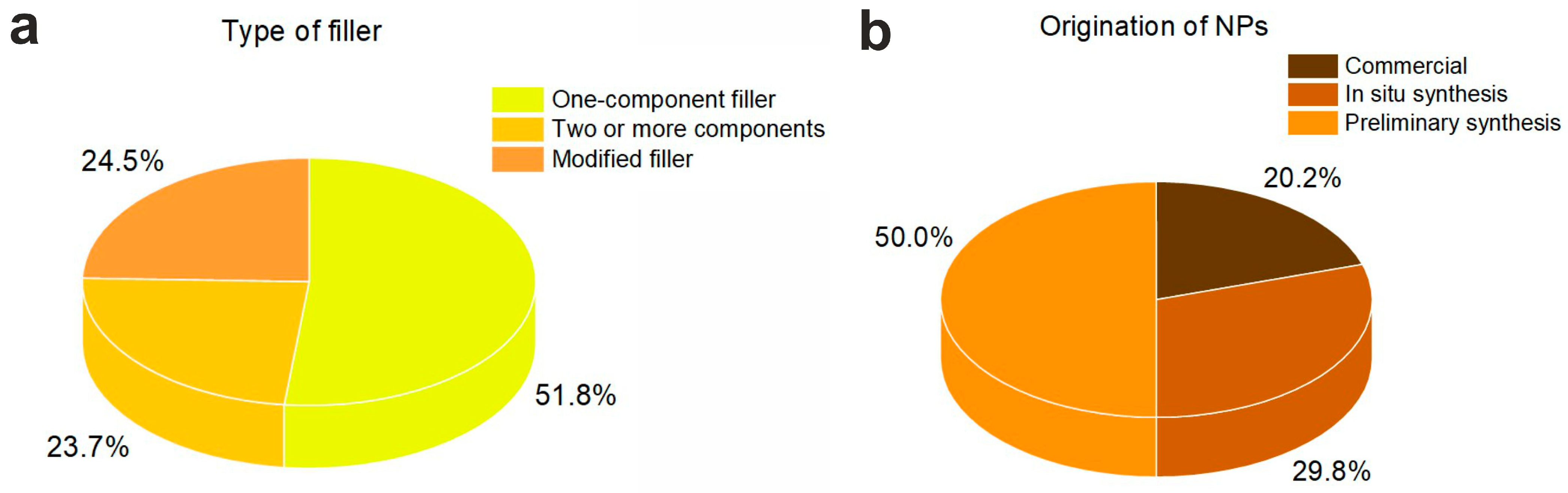
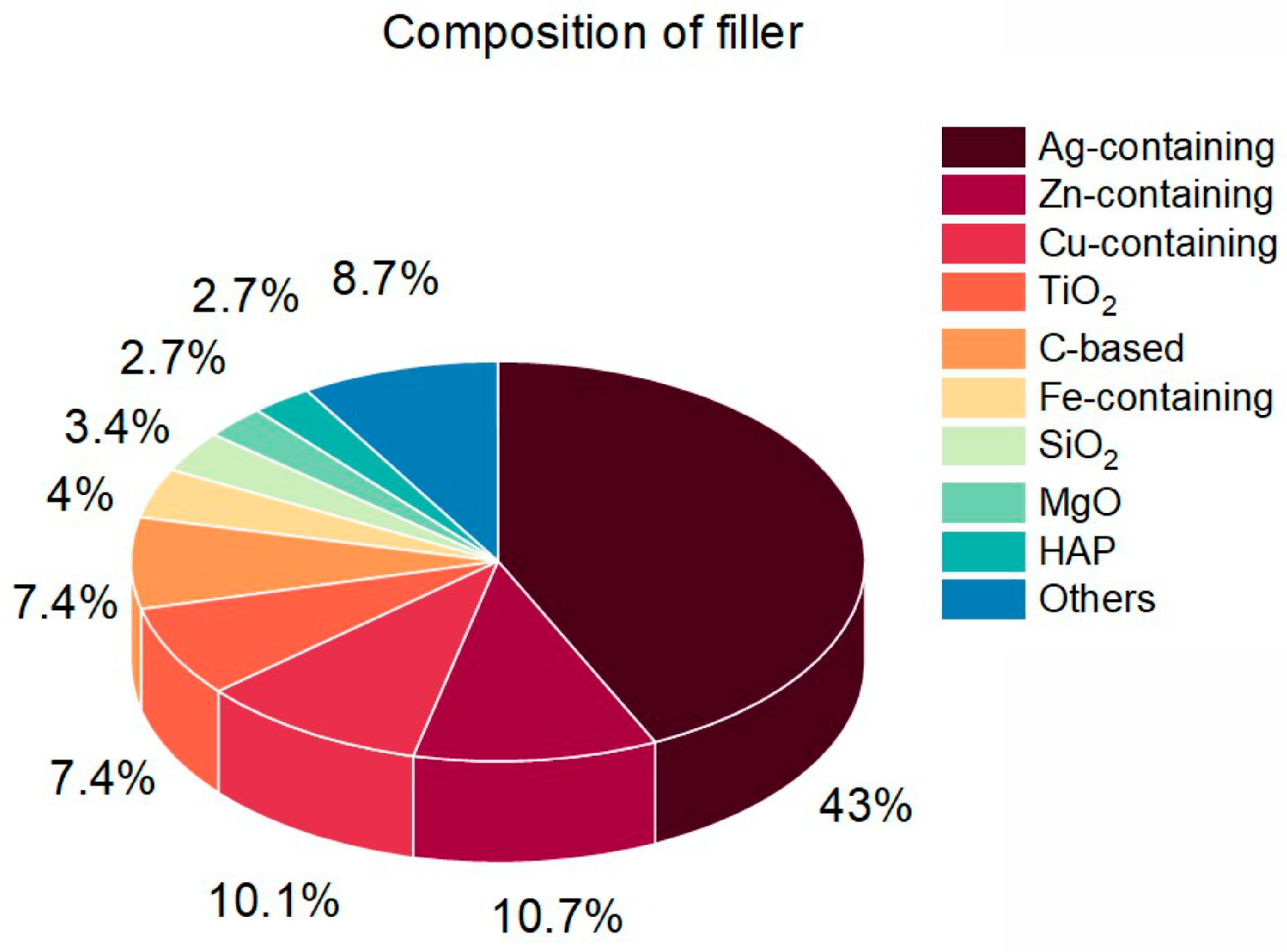
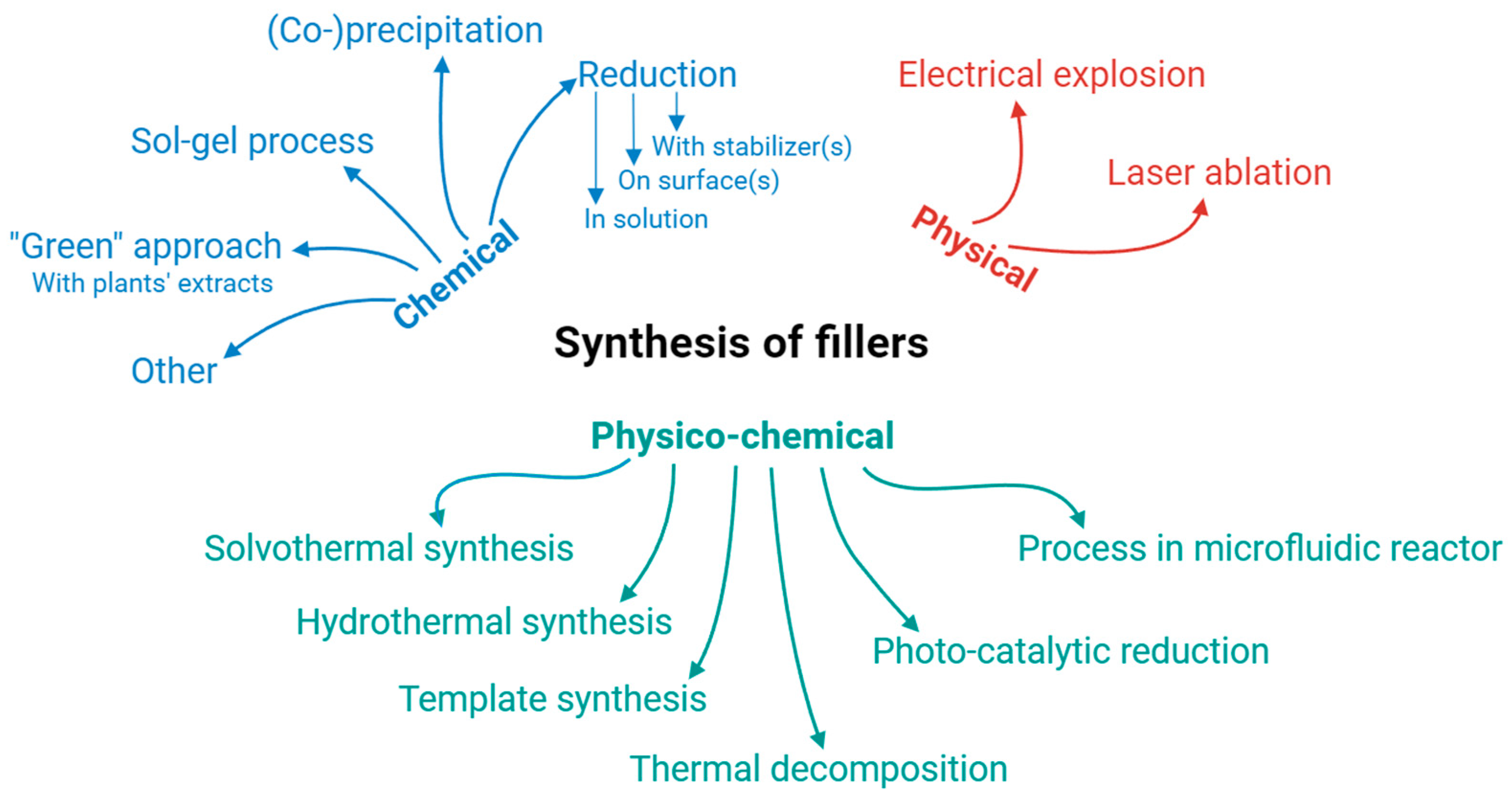
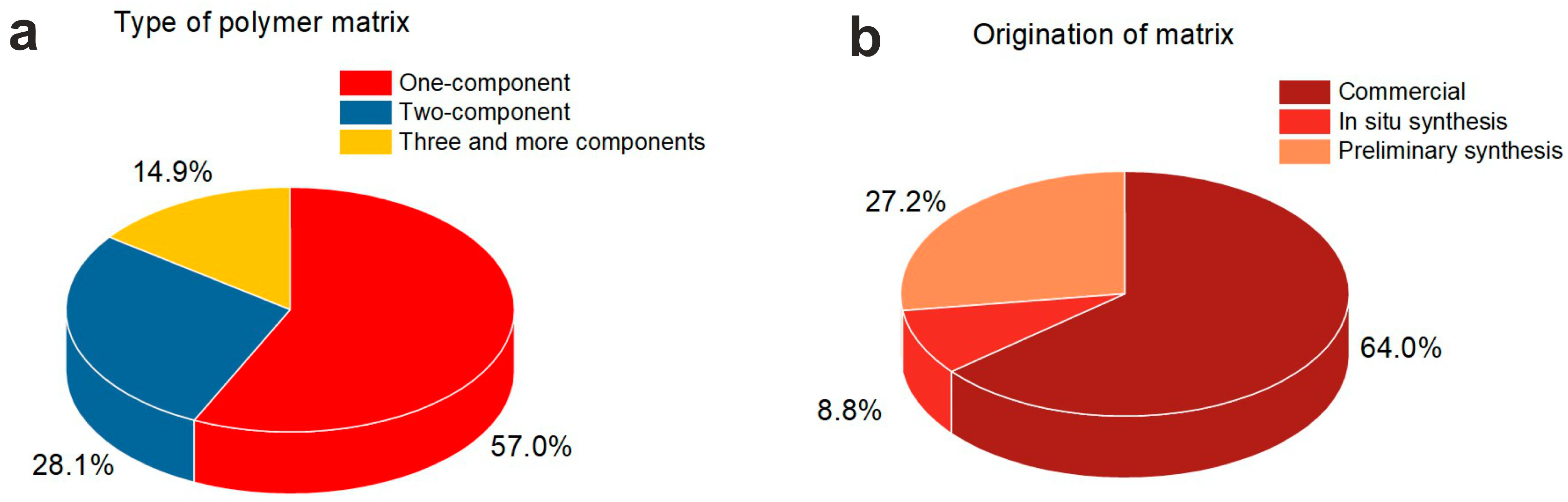
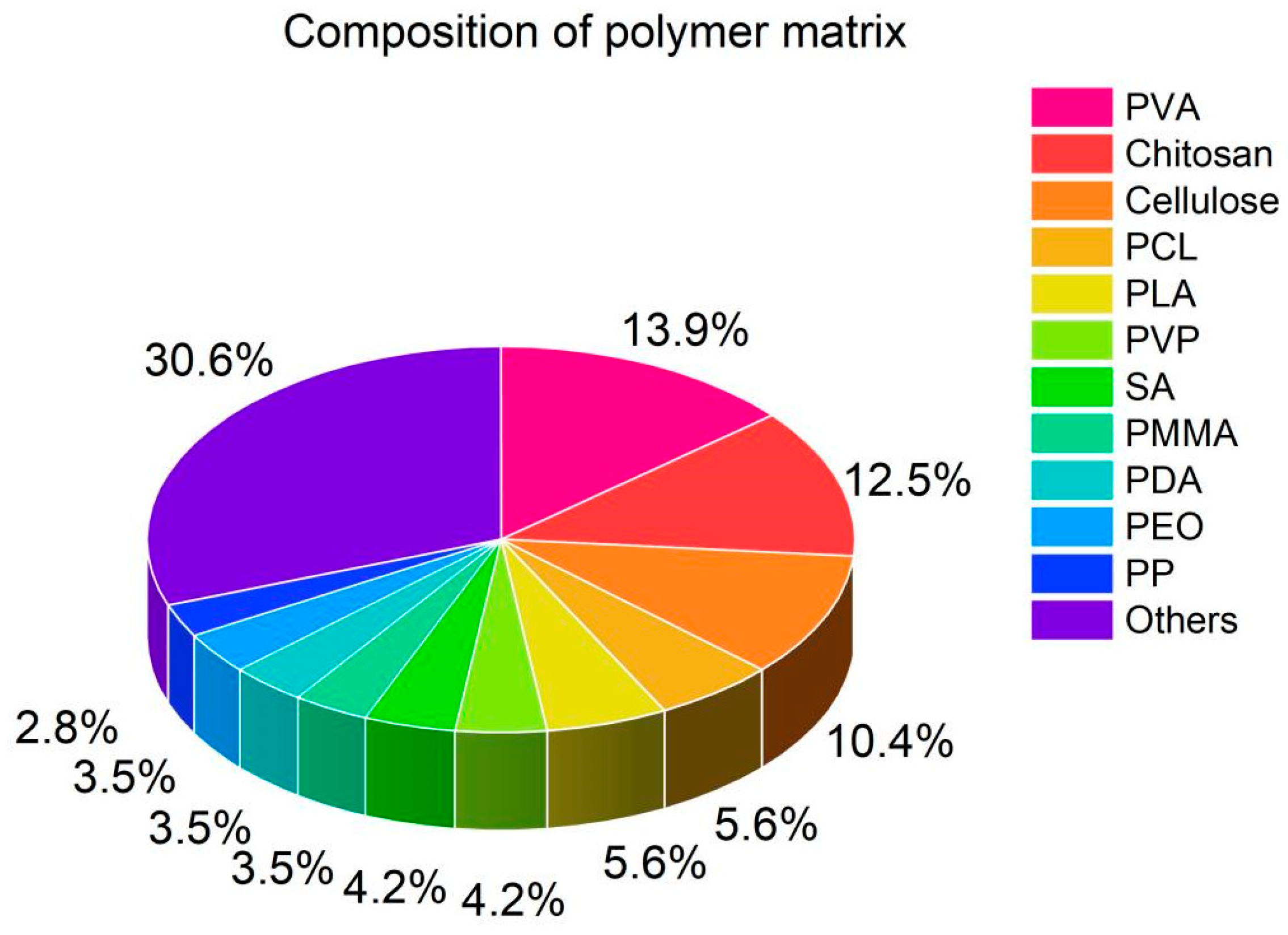

| Reference | Polymer | Filler | Composite | Application | |||||
|---|---|---|---|---|---|---|---|---|---|
| Composition | Origination | Composition | Origination | Size | Composition | Synthesis | Product | ||
| Ag-based NPs | |||||||||
| [37] | (1) Amine-rich aminomalononitrile (AMN); (2) Homopolymer poly(B5AMA); (3) Zwitterionic compound 2-methacryloyloxyethyl phosphorylcholine (MPC); (4) statistical co-polymer poly(MPC-st-B5AMA) | All the components were separately synthesized via polymerization | AgNPs | Synthesized in situ via chemical reduction | - | AMN/B5/MPC/Ag | Components were applied to the surface one by one | Self-healing coating on glass substrates | Coating |
| [38] | Polyvinyl alcohol (PVA); sodium alginate (SA); carboxymethyl chitosan (CMCS) | Commercial | Ag NPs | Synthesized with SA as reductant and stabilizer | 12 nm | PVA/SA/CMCS/ SA- AgNPs | Mixing in solution, immersion into CaCl2 solution for cross-linking | Gel | Wound dressing |
| [34] | Polyvinyl alcohol/polyacrylic acid (PVA/PAA) | Commercial | AgNPs | Synthesized in situ via chemical reduction | 20–160 nm | PVA/ PAA/Cur/AgNPs | Mixing in solution, electrospinning | Nanofibers | Wound medical dressing |
| [39] | Highly elastic natural rubber (NR); polydopamine (PDA) | Self-polymerization of dopamine in the NR (commercial) emulsion | AgNPs | Synthesized in situ via chemical reduction by PDA | between 80 and 120 nm | PDA/NR/AgNPs | Mixing in solution | Film | Flexible wearable strain sensors |
| [7] | Carboxymethyl cellulose (CMC); polyvinyl alcohol (PVA) | Commercial | AgNPs | Synthesized via pulsed laser deposition in situ in polymer solution | - | PVA- CMC/ AgNPs | Pulsed laser ablation pf Ag target in CMC-PVA blend | Film | Wound dressing |
| [19] | Polyvinyl alcohol (PVA); Polyvinyl pyrrolidone (PVP) | Commercial | AgNPs | Synthesized via laser ablation in situ | 45.3 to 81.3 nm | PVA/ PVP/ AgNPs | Pulsed laser ablation pf Ag target in PVA/PVP blend | Film | Wound healing/ dressing |
| [32] | Polypyrrole (PPy); polyaniline (PANI) | Polymerization | AgNPs | Biosynthesis with citronella oil | 35–50 nm max at 45.64 nm | TS/PPy/ AgNPs TS/PANI/AgNPs | Nile tilapia skin (TS) was covered step-by-step with polymers and NPs | Coating | Xenograft for burn treatment; active wound dressing system |
| [21] | Copper-based polymer–MOF (polyCu-MOF) | Synthesized using polyether containing specific units as building block, 4,4′-bipyridine as co-ligand, and copper ions as metal-coordinated centers | AgNPs | Synthesized in situ via chemical reduction | - | polyCu-MOF@ AgNPs | Chemical reduction of Ag ions on the polyCu-MOF matrix | Scaffold | Wound healing |
| [40] | Polyaspartate (PASP) | Polycondensation of L-aspartic acid | silver quantum dots (Ag QDs) | Synthesized via laser ablation in situ | 5 nm | PASP/ AgQDs | Pulsed laser ablation pf Ag target in PASP solution | Powder | Drug delivery |
| [10] | Polyvinyl alcohol (PVA) | Commercial | AgNPs | Synthesized via chemical reduction | ~116 to 208 nm | PVA/ Ag NPs | Mixing in solution | Film | Wound dressing |
| [12] | Inner layer: PVA, chitosan, gelatin; Outer layer: polyacrylonitrile (PAN) | Commercial | AgNPs | Green synthesis with Capsella bursa-pastoris extract | 72.57 nm | Inner layer: PVA, chitosan gelatin/AgNP outer layer: polyacrylonitrile (PAN)/AgNPs | Mixing in solution, electrospinning | Two-layer nanofiber mats | Wound dressing |
| [22] | Polycaprolactone (PCL); chitosan (CH); poly(ethylene oxide) (PEO) | Commercial | AgNPs | Synthesized via photocatalyzed reduction | 27 ± 4.3 nm | PCL/CH/PEO/AgNPs | Layer-by-layer electrospinning PCL, then CH/PEO; after that drop-coated with AgNPs colloid | Composite membrane | Wound treatment; regenerative medicine |
| [14] | Bacterial cellulose (BC) | Obtained using Komagataeibacter intermedius (MBS-88) | AgNPs | Synthesized in situ | 5–25 nm 17.5 nm | BC/AgNPs | BC membrane poured with silver undergone hydrothermal reduction | Mats | Wound dressing |
| [41] | Random co-polymers of methyl methacrylate and butyl acrylate (MMA-co-BA) | Emulsion polymerization | AgNPs | Synthesized via eco-friendly reduction | 5–30 nm | MMA-co-BA/AgNPs | Mixing in solution | Multifunctional film with photo-activated healing | Coating |
| [23] | Lignin-based nonisocyanate polyurethane foam | Polymerization and blending | AgNPs | - | - | Lignin-based polyurethane foam AgNPs | Polymerization in the presence of AgNPs | Foam | Wound healing |
| [42] | Polylactic acid (PLLA); polydopamine (PDA); chitosan (CS) | PLLA and CS commercial; PDA polymerized in situ | AgNPs | Commercial | - | PLLA/Ag@PDA@CS/Ag | Electrospinning, dip coating, electrospinning | Fiber | Bone repair material |
| [24] | Imidazole-based porous organic polymer (IM-POP) | Co-polymerization via catalyst-free Schiff base chemistry | AgNPs | Synthesized in situ | - | IM-POP-Ag | - | Particles | Wound- healing |
| [43] | Silicon elastomer matrix Ecoflex 00-50 (Smooth-On); Polysorbate 80 (Tween 80) as a surfactant | Polymerized in situ | AgNPs | Synthesized via chemical reduction | 5–50 nm | Ecoflex 00-50/AgNPs | Elastomer matrix polymerized in the presence of AgNPs | Composite matrix | Prosthetic and orthotic devices |
| [44] | Carboxymethyl cellulose (CMC); chitosan (CS) | Commercial | AgNPs | Synthesized from AgNO | 5 to 23 nm | CS/CMC/AgNPs | Mixing in solution | Thin films | Non- specified |
| [45] | Chitosan (CS) | Commercial | AgNPs | Synthesized in situ | - | AgNP/CS | In situ silver reduction; plasma treatment; solvent-casting process | Film | Coating; wound dressing |
| [46] | P(DMAEMA-coOEGMA-OH) | Synthesized via polymerization | AgNPs | Synthesized in situ | 32 ± 3 nm | P(DMAEMA-coOEGMA-OH)/AgNPs | The AgNPs were formed within the layers via the reduction by amine groups of PDMAEMA | Star nanolayers | Coating |
| [47] | Polypropylene (PP); polyvinyl alcohol (PVA) | Commercial | AgNPs | Synthesized in situ via reduction | - | PP/AgNO3/PVA | Reactive melt mixing method; MEX 3D printing | Filament | Non- specified |
| [48] | Popcorn-like polymeric nanoparticles (PPNPs) | Obtained by cross-linking and regeneration of polyglycidyl methacrylate (PGMA), styrene (St) and methacrylic acid (MAA) | AgNPs | Synthesized in situ | 3-45 nm on A-PPNPs 25–130 nm on PPNPs | Ag@PPNPs Ag@A-PPNPs (aminated) | AgNPs obtained in situ on PPNPs via reduction | Popcorn-like particles | Coating |
| [49] | Polydopamine (PDA); chitosan (CS) | CS commercial; PDA polymerized in situ | AgNPs | Synthesized in situ | - | AgNPs/ PDA/CS | Substrate coated with PDA/CS, than AgNPs reduced | Coating on urinary catheter | Biomedical devices |
| [50] | Polyethyleneimine (PEI) | Commercial | AgNPs | Synthesized via chemical reduction | 8 nm | Ag NPs/ PEI/MAO | PEI/MAO immersed into AgNPs dispersion | Coating on the Mg alloy | Medical devices |
| [31] | Poly(glycidyl butyl amine) (PGBA); chitosan (CS) | PGBA prepared by solution polymerization and decorated in solution by purified commercial CS | AgBrNPs | Synthesized in situ | below 20 nm | AgBr@ PGBA- C/N | AgBr chemically synthesized in the presence of matrix | Porous sheet-like structures | Non- specified |
| [51] | Polydopamine (PDA); polyhexamethylene guanidine (PHMG); polyurethane (PU) | PDA polymerized in situ; PHMG and PU commercial | AgNPs | Synthesized in situ | - | PU-PDA-Ag-PDA-PHMG | Step-by-step immersion of PU into solutions: polymerization of PDA, then reduction of Ag, and covering with PHMG | Composite coating on polyurethane | Reducing catheter- associated infections |
| [52] | Cellulose paper (CP); polydopamine (PDA); Vitrimer glass polymer (V) | CP commercial; PDA and V synthesized via polymerization | AgNPs | Synthesized in situ via reduction | - | CP@PDA@Ag-V | Growing AgNPs inside CP modified with PDA and conjunction with V | Paper-based composite | Wearable devices |
| [26] | Crown ether-based porous organic polymer (CEP-POP) | Polymerization via Alder–Longo method | Silver nitroprusside nanoparticles (AgNNPs) Ag2[Fe(CN)5NO] | Synthesized in situ | - | CEP-POP/AgNNPs | Obtained via “one-pot two-step” method | Nanorod-like powder | Wound healing |
| [27] | Polymeric vesicles from DIPEMA, GlyMA, and mPEG-CPADB | Synthesized via RAFT polymerization | AgNPs | Synthesized in situ | - | AgNP-decorated vesicles | AgNPs obtained in situ on cross-linked vesicle membranes via reduction polyvinylpyrrolidone (PVP) | Polymeric vesicles | Wound healing |
| [53] | Poly(lactic-co-glycolic acid) (PLGA) | Commercial | Ag2O | Synthesized via laser ablation in liquid | 20 to 40 nm | PLGA/ Ag2O NPs | Obtained via low-temperature technology | Bulk composite | Prosthetics |
| [54] | Poly(butylene adipate terephthalate) (PBAT) | Commercial | AgNPs polyvinylpyrrolidone (PVP) | AgNPs chemically synthesized in the presence of commercial PVP | 20–50 nm | PBAT/ AgNPs | Mixing in solution; solution casting method | Film | Disposable medical equipment |
| [29] | Silk fibroin (SF); poly(ethylene oxide) (PEO) | SF extracted from Bombyx mori cocoons; PEO commercial | DAPT-Au NPs | Reduced in the presence of 4,6-diamino-2-pyrimidinethiol (DAPT) | 2.44 nm | DAPT- Au NPs/ SF/PEO | Mixing in solution | Mixed-matrix membrane | Wound dressing |
| [55] | Nexcomp-META BIOMED | Commercial | Ciprofloxacin-loaded AgNPs CIP-AgNPs | Synthesized via chemical reduction and loaded with CIP | Ag 33.5 nm CIP-Ag 98.32 nm | Nexcomp-META BIOMED/AgNPs Nexcomp-META BIOMED/CIP-AgNPs | NPs were incorporated into resin composite at room temperature and were homogenized manually | Disc | Dentistry |
| [56] | PAPMA-b-PDMAEMA (D); PAPMA-b-PPMA (P) | Reversible addition–fragmentation chain transfer (RAFT) polymerization | MNP-AgNPs | Silver was chemically reduced in the presence of melanin nanoparticles (MNP) | 15.2 nm | P/D-MNP-Ag | Conjugation of functional polymers P and D with MNP-Ag under alkaline conditions | Dual-polymer functionalized melanin-AgNPs | Oral biofilm eradication systems for dentistry |
| [5] | Polypropylene (PP) | Commercial | Ag NPs-CS | Synthesized via chemical reduction with chitosan | 5.9 nm | Ag-NPs-CS/PP | Obtained via the melt blending route by using a twin-screw extruder | Bulk composite | Non- specified |
| [57] | Polyoxometalate (POM) | Commercial | Tyrosine-based polymer stabilized Ag NPs (DTP-AgNPs) | DTP prepared by RAFT polymerization; AgNPs were obtained via chemical reduction in the presence of DTP | - | DTP-AgNPs/POM | Mixing in solution | Powder | Antibacterial agent against Shigella and Shigella flexneri 2a |
| [13] | Gelatin | Commercial | PDA-Ag NPs | PDA was polymerized, than silver was reduced on its surface | 138 nm 383 ± 14 nm (DLS data) | Gelatin/ PDA- Ag NPs | Mixing in solution | Scaffold | Skin tissue engineering |
| [58] | Polyester | Commercial | AgNPs/Ppyr NPs | Synthesized in situ | 6–25 nm | AgNPs/ Ppyr- polyester | Polypyrrole redox-polymerized and silver reduced in the presence of fabric | Nonwoven fabric | Wearable electronics and protective coats |
| [59] | Polylactic acid (PLA); cellulose acetate (CA); poly(caprolactone) (PCL) | Commercial | Hydroxyapatite (HANPs); AgNPs | HANPs synthesized via chemical precipitation; AgNPs obtained via green synthesis with Callistemon viminalis extract | HANPs length 25–75 nm; width 8–35 nm; AgNPs 54–79 nm | PLA/CA/PCL/ HANPs/ AgNPs | Mixing in solution, electrospinning | Nanofibers | Bone regeneration |
| [18] | Chitosan (CS); polyethylene oxide (PEO) | Commercial | AgNPs ZnO NPs | Commercial | - | AgNPs/ ZnO NPs/ Chitosan/PEO | Mixing in solution, electrospinning | Nanofibrous mats | Wound dressing and healing |
| [4] | Poly(3-hydroxyoctanoate)-co-(3-hydroxyhexanoate) (PHO) | Synthesized in situ using Pseudomonas putida KT2440 bacteria | AgNPs CuONPs | CuONPs commercial; AgNPs synthesized in situ via reduction | - | PHO- AgNPs/ CuO NPs | PHO-AgNPs obtained in situ and mixed with CuO NPs, | Film | Wound dressing |
| [6] | Poly(vinyl alcohol) (PVA) | Commercial | GO AgNPs | AgNPs chemically reduced in the presence of GO | AgNPs 3.1 ± 0.8 nm | PVA/GO–AgNPs | Mixing in solution, solution casting approach | Film | Wound dressing |
| [60] | Poly-l-lactic acide (PLLA) | Commercial | Halloysite nanotubes filled with Ag | Nano-Ag loaded into HNTs via CH3COOAg vacuum negative-pressure suction, injection, and thermal decomposition | 50 nm × 1 μm | PLLA/HNT@ Ag NPs | Mixing in solution | Powder for SLS scaffolds | Drug sustained- release |
| [61] | Polydimethylsiloxane (PDMS) | Commercial | AgNPs-lecithin modified montmorillonite gNPs@LEC-Mt | Mt commercial; AgNPs synthesized via chemical reduction in the presence of LEC-Mt | 8–15 nm | PDMS/ AgNPs@LEC-Mt | Polymer solution intercalation method | Film | Medical devices |
| [62] | Cellulose acetate (CA) | Commercial | TiO2 NPs AgNPs | Commercial TiO2 covered DOPA and AgNPs reduced on it | TiO2 20–100 nm (36.12 nm) AgNPs 4–12 nm (5.9 nm) | CA/TiO2/AgNP | Mixing in solution, electrospinning | Nanofibers | Non- specified |
| [63] | Cellulose acetate (CA) | Commercial | CNT/Ag | AgNPs reduced in the presence of oxidized commercial CNTs | - | CA/CNT/Ag | Mixing in solution, electrospinning | Nanofiber mats | Non- specified |
| [28] | Sodium carboxymethyl cellulose (CMC); ε-polylysine hydrochloride (EPL) | Commercial | Fe3O4 Ag NPs | Synthesized in situ via chemical reduction | - | FCE/ AgNPs | NPs were step-by-step reduced on the polymer matrix | Powder | Wound infection therapy |
| [64] | Arabinoxylans; acrylic acid (AAc) | Arabinoxylan extracted from P. ovata husk; polymeric materials were synthesized via the free radical polymerization in situ | Graphene oxide (GO); hydroxyapatite (HAp); aluminum oxide (Al2O3); AgNPs | Commercial GO, HAp, and Al2O3 loaded into polymer in situ; AgNPs reduced in situ on polymer surface | - | AgNPs-Arabinoxylan/PAA/GO/ Al2O3 | Synthesized via the free radical polymerization with filler and subsequent covering with AgNPs | Silver-coated scaffolds | Bone tissue engineering |
| [65] | Textile composition (88% nylon and 12% Spandex) | Commercial | rGO AgNPs | GO commercial; AgNPs synthesized in situ | - | Polymer fiber/rGO/AgNPs | GO placed on the textile via drop coating; silver nitrate applied there and laser processed | Fiber | Textile sensor for wearables and smart clothes |
| [66] | Chitosan | Commercial | Polydopamine (PDA); carbon nitride C3N4; Ag NPs | PDA polymerized in the presence commercial C3N4; AgNPs reduced on them after that | - | C3N4- PDA-Ag@CS | Mixing in solution | Film | Wound healing |
| [67] | Poly(ε-caprolactone) PCL | Commercial | GO-AgNPs | GO produced by modified Hummer’s method; AgNPs reduced in the presence of GO | 5 ± 0.5 nm | PCL-GO-Ag | Mixing in solution, electrospinning | Nanofibrous mat | Non- specified |
| [68] | Polyglycolic acid (PGA); poly-L-lactic acid (PLLA) | Commercial | Mesoporous bioactive glass (MBG); AgNPs | MBG obtained via sol-gel process, and functionalized with polydopamine; AgNPs reduced on pMBG surface | AgNPs 5 nm | PLLA/ PGA/Ag@pMBG | Mixing in solution; selective laser sintering to prepare scaffold | Scaffolds | Treatment of infected bone defects |
| [69] | Polyvinylidene fluoride (PVDF) | Commercial | Ag-pBT NPs | Barium titanate (BaTiO3) powder modified with dopamine and AgNPs reduced on its surface | AgNPs 10 to 45 nm 24.5 nm | Ag-pBT NPs/ PVDF | 3D scaffolds built layer-by-layer using self-developed SLS system | Scaffolds | Orthopedics |
| [70] | Polylactic acid (PLA) | PLA made of commercial PLLA and PLDA | ZnO–Ag NPs | Simultaneous chemical synthesis | ZnO rods of 20 nm × 30–80 nm Ag NPs of 5–15 nm | PLA/ZnO-Ag NPs | Solution blow spinning process | Porous fibers | Personal protection and medical care |
| [71] | PolyAspAm(EDA/EA)/TA: polyaspartamide (PolyAspAm); thylenediamine (EDA); thanolamine (EA); tannic acid (TA) | Prepared by acid-catalyzed bulk polycondensation | AgNPs; 3D graphene | GO commercial; AgNPs synthesized in situ | less than 50 nm | PolyAspAm(EA/EDA)/TA-GO-AgNP | Mixing in solution; AgNPs obtained in situ via reduction | Nanocomposite aerogel | Medical devices |
| [25] | Bacterial cellulose (BC) | Prepared using G. xylinus | Graphene (GO); AgNPs; polydopamine (pDA) | GO produced by Hummer’s method; AgNPs reduced in situ | AgNPs less than 50 nm | Ag-pDA (rGO)/BC | BC (GO) formed by blending and naturally combining; then immersed in DA and soaked in AgNO3 | Film | Wound healing |
| [72] | Triblock co-polymer PEG–PHBV–PEG (PPP) | Copolymerization of poly(ethylene glycol), b-poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and poly(ethylene glycol) | ZnO NPs Ag–ZnO NPs | Synthesized via chemical precipitation and reduction | length 183.3 nm width 66.7 nm | PPP/ZnO NPs PPP/Ag–ZnO NPs | Mixing in solution; solution casting technique | Powder aggregations or film | Non- specified |
| [73] | Catechol-terminated co-polymer (CCP) | Synthesized via RAFT polymerization | rGO AgNPs | rGO commercial; AgNP synthesized in situ | AgNPs 10 nm | CCP-rGO-Ag | GO decorated with CCP by mussel-inspired chemistry route; CCP-rGO covered with AgNPs via reduction | Powder | Non- specified |
| Zn-based NPs | |||||||||
| [74] | Co-polymers of poly(lactic-co-glycolic acid) PLGA | Commercial | ZnO NPs | Synthesized via laser ablation in liquid | 45–60 nm | PLGA/ ZnO NPs | Obtained via low-temperature technology | Film | Prostheses; biomedical devices |
| [75] | Borosiloxane (Bs) | Obtained from PDMS and boric acid (BA) heating above 200 °C | ZnO NPs | Synthesized via laser ablation in liquid | 90 nm | Bs/ ZnO NPs | Mixing in solution | Film | Prostheses and biomedical devices |
| [76] | Low-density polyethylene (LDPE); ethylene vinyl acetate (EVA) | Commercial | ZnO NPs | Synthesized via chemical precipitation | 20–30 nm | LDPE/ PVA/ ZnO NPs | Mixing in solution using mono-screw extruder | Bulk composite | Non- specified |
| [77] | Chitosan (Cs); polyvinyl alcohol (PVA) | Commercial | ZnO NPs | Synthesized via sol–gel method | - | CS/PVA-nano ZnO | Mixing in solution; solution casting approach | Film | Non- specified |
| [78] | Poly(L-lactic acid) (PLLA) | Commercial | ZnCl2 NPs ZnO NPs | Synthesized via laser ablation in different liquids | 30–70 nm | PLLA/ ZnCl2 NPs PLLA/ ZnO NPs | Mixing in solution; spin-coating | Nanosheets | Wound healing |
| [79] | Polymethylmethacrylate (PMMA) | Commercial | ZnO | Synthesized via “oxalate route” chemical process | - | PMMA/ ZnO | Mixing in solution; slurry composites pour into cylindrical molds | Orthoses | Orthotic devices |
| [80] | Polyvinyl alcohol (PVA) | Commercial | ZnO NPs | Synthesized in situ | 50–150 nm | PVA/ ZnO NPs | ZnO chemically obtained in PVA solution; electrospinning | Nanofibers | Wound healing and tissue reconstruc- tion |
| [81] | Hydroxypropyl chitosan (HPCs); polyvinyl alcohol (PVA) | Commercial | ZnO-NPs | Commercial | 30 nm | HPCs/ PVA/ ZnO-NPs | Mixing in solution | Hydrocolloid composite sponge | Wound- healing |
| [82] | Polycaprolactone (PCL) | Commercial | ZnO loaded with curcumin (ZC-NPs) | Commercial ZnO was loaded with curcumin | <100 nm | ZCPCL | Mixing in solution, electrospinning | Mats | Top-sheets in feminine sanitary hygiene napkins |
| [83] | Polycaprolactone (PCL) | Commercial | ZnO NPs (CA) | Commercial ZnO modified with citric acid monohydrate | 20 to 40 nm 99.71 nm (ZnO-CA) | PCL/ZnO-CA NPs | Mixing in solution; solvent-casting process | Membrane | Non- specified |
| [15] | Polyvinylpyrrolidone (PVP) | Commercial | ZnO NPs ZnO-CTAB NPs ZnO-HTAB NPs | Commercial and chemically synthesized ZnO NPs were modified with Cetyltrimethylammonium bromide (CTAB) and Hexadecyltrimethylammonium bromide (HTAB) | - | PVP/ZnO NPs PVP/ZnO-CTAB NPs PVP/ZnO-HTAB NPs | Mixing in solution and centrifugally spun using centrifuge equipped with a spinneret | Nanofibers | Wound dressing |
| [84] | Chitin–cellulose (CH-CE) | Commercial | ZnO, CuO | Synthesized preliminary via sol–gel synthesis | 15–23 nm | CH-CE@ZnO/CuO | Mixing in solution, solution cast method | Conductive nanocomposite films with adjusted semiconductors properties | Non- specified |
| [85] | Dental resin | Obtained from commercial Bis-GMA and TEGDMA via photopolymerization | Zn doped mesoporous silica nanoparticles (Zn-MSNs) | Synthesized via sol–gel method | about 138 nm | Zn-MSNs/Dental resin | Fillers mixed with resin | Dental resin | Dentistry |
| [86] | Acrylonitrile butadiene rubber (NBR) | Commercial | ZnO NRs; ZnO NPs; ZnO NPs with CaCO3; ZnO NPs with TiO2 | Commercial | 3 μm 20 nm | NBR/ ZnO | Latex processing technique | Films | Medical devices |
| Cu-based NPs | |||||||||
| [87] | Polyvinyl pyrrolidone/ carboxymethyl cellulose (PVP/CMC) | Commercial | CuO NPs | Synthesized preliminary via laser ablation | <10 nm | CuONPs/PVP/ CMC | Mixing in solution | PVP/CMC polymer blend films modified with CuO nanoparticles | Coating |
| [20] | Polyvinyl alcohol (PVA); polyvinyl pyrrolidone (PVP) | Commercial | CuO NPs | Synthesized via laser ablation in situ | - | PVA/ PVP/CuO NPs | Pulsed laser ablation pf Cu target in PVA/PVP blend | Film | Wound healing |
| [88] | Chitosan (Cs) | Commercial | CuNPs | Commercial | ~8.11 nm | Cs/ CuNPs | Mixing in solution; solution casting approach | Film | Non- specified |
| [89] | Polymethyl methacrylate (PMMA) | Commercial | CuFe2O4/Cu2O/CuO | Synthesized via electrical explosion of twisted copper and iron wires | 10 to 100 nm | PMMA- CuFe2O4/ Cu2O/ CuO NPs | Mixing in solution; solution casting approach | Film | Non- specified |
| [90] | Polyaniline (PANI) | Synthesized by in situ oxidative polymerization | CuS NPs | Obtained via solvothermal synthesis | - | CuS@ PANI | PANI polymerized in the presence of CuS NPs | Core–shell particles | Non- specified |
| [91] | Isotactic polypropylene (iPP) | Commercial | CuNPs-PEI/GABA | CuNPs functionalized with polyethyleneimine and 4-aminobutyric acid | 27.0 nm | iPP/ CuNPs | Mixing in solution | Plates | Non- specified |
| [17] | Polyvinyl alcohol (PVA) | Commercial | GO/L-Arg/CuO GO/L-Arg-Cu | GO prepared by Hammers’ method and modified with L-Arginine; Cu-containig particles synthsized via green synthesis | - | PVA/GO/L-Arg/ CuO PVA/GO/L-Arg-Cu | Mixing in solution, additional cross-linking by citric acid (CA) | Green cross-linked electrospun nanofber | Wound healing |
| [92] | Polypropylene (PP) | Commercial | Cu-TiO2 | Obtained via soft-chemical hydrothermal technique | 35 nm length 20 nm diameter | Cu-TiO2-PP | Mixing in solution; compressed by compression molding machine | Film | Non- specified |
| [35] | Polyvinyl alcohol (PVA); tannic acid (TA) | Commercial | CuO/SiO2 | Simultaneously synthesized via chemical approach | Between 50 and 90 nm | PVA/TA/CuO/SiO2 | Mixing in solution | Biofilm | Rapid wound dressing |
| TiO2 NPs | |||||||||
| [9] | Polyvinyl alcohol (PVA); sodium alginate (SA) | Commercial | TiO2 NPs | Green synthesis using Aloe vera leaves extract | - | PVA/SA/TiO2 NPs | Mixing in solution; solvent-casting process | Film | Wound dressing |
| [93] | Poly(3-hydroxybuty-rate-co-3-hydroxyvalerate) (PHBV) | Commercial | TiO2 NPs | Commercial | - | PHBV/ TiO2 NPs | Mixing in solution, electrospinning | Nanofiber scaffolds | Non- specified |
| [94] | Poly(ethylene oxide) (PEO); polyvinylpyrrolidone (PVP) | Commercial | TiO2 NPs | Chemically synthesized | 12.1 to 19.19 nm | PEO/PVP/TiO2 NPs | Mixing in solution; solvent-casting process | Film | Non- specified |
| [16] | Polyvinyl alcohol (PVA); sodium alginate (SA); sodium carboxymethyl cellulose (CMC-Na); waterborne polyurethane (WPU); polyvinylpyrrolidone (PVP) | Commercial components were blended in water | nTiO2 | Commercial | 20–30 nm | PVA/SA/CMC/ WPU/ PVP/ nTiO2 | Mixing in solution | Transparent protective gel membrane | Wound protection |
| [95] | Polycaprolactone (PCL) | Commercial | TiO2 modified with poly-ortho-toluidine (POT) | Commercial TiO2 modified with POT via oxidative polymerization | - | POT- TiO2/PCL | Mixing in solution, solution cast method | Film | Tissue engineering |
| [96] | Polystyrene (PS); poly(vinyl alcohol) (PVA) | PS synthesized in situ; PVA commercial | Vinyltriethoxysilane (VTES) modified TiO2 | VTES and TiO2 commercial | 30 nm | V-TiO2/ PS/PVA | PS polymerized in the presence of V-TiO2; PVA solution was added | Microspheres | Hemoperfusion adsorbent for efficient bilirubin removal |
| [97] | High-density polyethylene (HDPE) | Commercial | TiO2 hydroxyapatite (HAp) | Commercial with additional treatment | 10–14 nm | HDPE/ HAp/ TiO2 | Powders mixing and melting; compaction technique | Composite sheets | Non- specified |
| Other NPs | |||||||||
| [98] | Thermoplastic elastomers TPE88 (TPE) | Commercial | Hydrophobic carbon quantum dots (hCQDs) | Chemical synthesis using polymerization | - | TPE/ hCQDs | TPE filament 3D printed and immersed into hCQDs solution | Filament | Medical tools and devices |
| [99] | Polycaprolactone (PCL) | Commercial | Graphene oxide (GO); CeO2 | GO synthesized via modified Hummer’s technique; CeO2 obtained via hydrothermal method | - | CL-GO- CeO2 | Mixing in solution; solvent-casting process | Film | Bone tissue engineering |
| [100] | Chitosan (Ch) | Commercial | SPION | Co-precipitation in situ using microfluidic reactor | 6.8 ± 0.6 nm | Ch- SPION | Composite obtained using microfluidic reactor | Powder | MRI imaging |
| [101] | Sodium alginate-containing curcumin | Commercial | Fe3O4NPs | Synthesized via co-precipitation | 390±14.76 nm (DLS) 359 nm (SEM) | SA- Curcumin/Fe3O4 NPs | Mixing in solution | Particles | Drug delivery |
| [102] | Poly(L-lactide) (PLLA) | Commercial | MgO NP surface modified by FeCl3 and tannic acid (TA) (FeCl3–TA/MgO) | Commercial MgO modified with commercial FeCl3 and TA | 20 nm | PLLA/ FeCl3– TA/MgO | Mixing in solution; 4D processing | Porous scaffolds | Bone tissue engineering |
| [103] | Dental resin of triethylene glycol dimethacrylate (TEGDMA), polymethyl methacrylate (PMMA), acrylic matrix | Solvent evaporation method | MPS-grafted SiO2 NPs | Commercial SiO2 modified with MPS (3-methacryloxypropyl trimethoxysilane) | 15–20 nm specific surface area of 210–240 m2/g | MPS- SiO2NPs/ Dental resin | Mixing and photo initiated polymerization | Self-healing dental composite | Dentistry |
| [104] | Dental resin | Obtained from Bis-GMA and TEGDMA | core–shell chlorhexidine/amorphous calcium phosphate (CHX/ACP) NPs silanization silica fillers | Synthesized via vesicle templating technology | 98.48 ± 17.61 nm | Dental resin/ CHX/ ACP/ SiO2 | Fillers mixed evenly with pure resin | Composite resin | Dentistry |
| [105] | Microcapsules (MCs) contained: poly(urea-formaldehyde) (PUF) shells; N, N-dihydroxyethyl-p-toluidine (DHEPT); triethylene glycol dimethacrylate (TEGDMA) | Synthesized in situ | Nanoantibacterial inorganic fillers (NIFs): nanosilica; quaternary ammonium salts (QAS) | QAS chemically synthesized and conjugated with SiO2 | - | MCs/ NIFs | In situ polymerization and liquid-phase precipitation | Microcapsules for self-healing of dental resin | Dentistry |
| [106] | Dental resin matrix | Obtained from commercial Bis-GMA and TEGDMA via photopolymerization | MgONPs SiO2 | Commercial | ∼50 nm ∼2 μm | Bis-GMA/TEGDMA/MgONPs/SiO2 | Mixing under yellow light | Photoactivating paste dental resin composite | Repairing dental defects |
| [107] | Sodium alginate (ALG) | Polymerized in situ | CIP-loaded nanohydroxyapatite (HANPs) | Synthesized and loaded with CIP | length 52 nmdiameter 8 nm | CIP- HANPs/ALG | HANPs added to ALG solution as cross-linking agent | Powder | Bone filler |
| [108] | Sodium alginate (Na-ALG)/polyvinyl alcohol (PVA) | Commercial | Mesoporous bioactive glass NPs doped with Ag and Cu | Synthesized preliminary via Stöber process (sol–gel synthesis) | - | Na-ALG/PVA/Cu–Ag MBGNs | Mixing in solution, 3D printing | Skin scaffolds | Patient- specific implants |
| [109] | Poly (xylitol maleate adipate) (PXMA) | Synthesized by melt polycondensation without catalyst | Nanobioactive glass (n-BG) SiO2–CaO–P2O5 | Synthesized via sol–gel method | 13–24 nm | n-BG/ PXMA | Mixing in solution | Film | Bone construction |
| [36] | Carrageenan (CRG) | Commercial | CeO2 NPs | Commercial | 15–30 nm | CRG- CeO2 | Mixing in solution with glutaraldehyde | Sponge | Wound healing, blood clotting agent |
| [11] | Cellulose acetate (CelAc); chitosan (Chit) | Commercial | CeO2 | Commercial | <25 nm | CelAc/Chit/ CeO2 | Mixing in solution | Film | Wound dressing |
| [110] | Borsiloxane (BS) | Obtained from hydroxyl-terminated polydimethylsiloxane (PDMS) and boric acid (BA) heating above 200 °C | Al2O3 | Synthesized preliminary via laser ablation in liquid | Average hydrodynamic diameter ~45 nm | Al2O3NPs/BS | Mixing in solution | Films | For prostheses and biomedical devices |
| [111] | Chitosan | Commercial | CoO | Commercial | - | CS-CoO | Solution casting approach | Film | Potential pharma- ceutical applications |
| [30] | Polylactic acid (PLA) | Commercial | Montmorillonite (MMT) NPswith adsorbed B compounds | Commercial boron nitride (BN), zinc borate (ZB), phenylboronic acid (PBA), and MMT mixed | 200–300 nm | MMT/Bs/PLA | Mixing in solution, electrospinning | Micron/submicron fibers | Wound dressing |
| [112] | Vinylidene fluoride-co-hexafluoropropylene (PVDF-HFP); microcrystalline cellulose (MCC) | Commercial | LiFe5O8 (LFO) | Chemical synthesis | - | PVDF- HFP/ MCC/ LFO | Mixing in solution, electrospinning | Fiber mats | Coating |
| [33] | PDA-modifed poly(lactic-co-glycolic) acid (PLGA) | Polymerization of dopamine on the PLGA membrane (commercial) | AuNPs; antibacterial peptides (Os) | AuNPs synthesized in situ via chemical reduction; Os commercial | - | Os/ Au-PDA@PLGA | Sequential immersion of membrane into solution for Au reduction and for Os attachment | Membrane | Wound repair |
| [3] | Silk fibroin (SF)and collagen (CL) | Silk fibroin obtained via extraction; collagen commercial | Pd Pt | Synthesized in situ via chemical reduction | 8.64 nm 7.36 nm | SF/CL/ Pd–Pt | Mixing in solution, electrospinning | Composite nanofber scafolds | Wound dressing |
| [8] | Polycaprolactone (PCL); chitosan (CHT) | Commercial | Bentonite (Ben) | Commercial | - | PCL/ CHT/Ben | Mixing in solution, electrospinning | 3D scaffolds | Wound dressing |
| [113] | Cissus quadrangularis fiber (CQF); bisphenol F type LY556 epoxy resin; Araldite HY951 hardener | Commercial | CaO NPs | Commercial | - | Epoxy resin/CaO | Mixing, compressing | Laminate | Non- specified |
| [114] | Low-density polyethylene (LDPE) | Commercial | CaO NPs CaO-OA | CaO chemically synthesized and modified with oleic acid (OA) | 25 and 55 nm | LDPE/ CaO NPs | Mixing in nitrogen atmosphere and press-molded at 170 °C | Bulk composite | Non- specified |
| [115] | Chitosan (CS) | Commercial | Te NPs | Synthesized in situ | 37.48 ± 14.56 nm | CS-Te NPs | Te NPs obtained in the presence of CS | Particles | Non- specified |
| [116] | Cellulose | Commercial | NiO NPs | Synthesized in situ | 20–30 nm | Cellulose/NiO NPs | NiO chemically synthesized in the presence of cellulose | Powder | Treatment of visceral leishma- niasis |
| Polymer | State | Toxicity | Cost | Hydrophilicity | Solubility in Water | Mechanic Characteristics | Stability |
|---|---|---|---|---|---|---|---|
| PVA | Semi- crystalline | Non-toxic | Low cost | Hydrophilic | Soluble in hot water | Superior mechanical properties; high tensile strength and flexibility | Low thermal stability |
| Cs | Semi- crystalline | Non-toxic | Expensive | Hydrophilic and hydrophobic | Water- soluble in acidic media | Limited mechanical and barrier properties; low strength | Poor stability |
| CE | Semi- crystalline | Non-toxic | Low cost | Extensively hydrophilic | Insoluble in water | Superior mechanical properties; low flexibility | Sustainable |
| PCL | Semi- crystalline | Non-toxic | Commercially available | Hydrophobic | Long-term degradation in the presence of water | Good mechanical properties; flexible; tough | Good thermal stability |
| PLA | Amorphous; semi-crystalline; highly crystalline | Non-toxic | High cost | Hydrophobic | Insoluble in water | High strength but brittle; poor toughness; shape-memory effect | Relatively low thermal stability. Chemically inert |
| PVP | Amorphous | Low-toxic | Low cost | Hydrophilic and hydrophobic | Water-soluble | Poor mechanical stability | Chemically inert, temperature-resistant, pH-stable |
| SA | Amorphous; semi- crystalline | Low-toxic | Relatively low cost | Hydrophilic | Outstan- ding water absorption and holding ability | Poor mechanical strength | Relatively low stability. pH- sensitive |
| PMMA | Amorphous | Non-toxic | Low cost of production | Hydrophobic | Insoluble in water | Good mechanical properties; strong, tough, and lightweight | Good physical, chemical and thermal stability; superior environmental stability |
| PDA | Non-toxic | High-cost | Moderately hydrophilic | Water- soluble | Poor thermal stability; durable | ||
| PEO | Semi- crystalline | Non-toxic | Low-cost | Hydrophilic | Water- soluble | Strong mechanical properties; strength and resistance to impact and stress | High chemical and electrochemical stability; thermal stability; durability |
| PP | Increased crystallinity | Generally considered safe for use | Comparatively low cost | Hydrophobic | Insoluble in water | Satisfactory mechanical strength; flexible material; outstanding fatigue resistance | High thermal and chemical stability; superior chemical resistance |
| Polymer | Degradability | Biocompatibility | Bioactivity | Surface Groups | Other |
|---|---|---|---|---|---|
| PVA | Biodegradable under both aerobic and anaerobic conditions | Biocompatible | Synthetic polymer; transparent | ||
| Cs | Biodegradable | Exceptional biocompatibility | Unique bioactivity; Antibacterial and antioxidant properties; bioadhesivity | Reactive groups | Natural origination; semi-synthetic polymer |
| CE | Biodegradable | Biocompatible | Hydroxyl groups | Renewable polymer; natural origination | |
| PCL | Biodegradable | Excellent biocompatibility | Non-immunogenic | Synthetic polymer; low melting point; good blend ability; easy to functionalize | |
| PLA | Slow biodegradation rate | Biocompatible | Low cell adhesion | Lack of reactive side-chain groups | Can be synthesized from renewable resources; good layer adhesion. good printability; widely used in 3D printing |
| PVP | Biodegradable | Excellent biocompatibility | Inert; excreted rapidly and completely through kidney | Hydrophilic and hydrophobic functional groups | Synthetic polymer; film-forming capability; great stabilizer; protective agent; good electrical properties; good transparency; adhesive properties; undergoes cross-linking |
| SA | Good biodegradability | Good biocompatibility | Well tolerated by the immune system | Abundant hydroxyl and carboxyl groups | Natural origination; renewable; excellent gelling and thickening properties |
| PMMA | Non-degradable | Good biocompatibility | Easily hydrolyzed ester groups | Synthetic polymer; malleability; transmits up to 92% of visible light | |
| PDA | Great biodegradability | Promotes cell adhesion and proliferation on substrates; promotes adhesion of implants onto tissue; stimulates bone formation around the implant; scavenges harmful ROS | Abundant functional groups; large volumes of catechol, quinine, and amine groups | Synthetic eumelanin polymer; coating material; functionalizing and modifying agent; redox activities; photoprotective and superparamagnetic properties | |
| PEO | Resistant to degradation | Excellent biocompatibility | Minimal health risks | Synthetic polymer; film-forming capability; high ionic conductivity | |
| PP | Non-biodegradable | Biocompatible | Synthetic polymer; thermoplastic; Recyclable; superior electrical insulation; transparent |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shabalina, A.V.; Kozlov, V.A.; Popov, I.A.; Gudkov, S.V. A Review on Recently Developed Antibacterial Composites of Inorganic Nanoparticles and Non-Hydrogel Polymers for Biomedical Applications. Nanomaterials 2024, 14, 1753. https://doi.org/10.3390/nano14211753
Shabalina AV, Kozlov VA, Popov IA, Gudkov SV. A Review on Recently Developed Antibacterial Composites of Inorganic Nanoparticles and Non-Hydrogel Polymers for Biomedical Applications. Nanomaterials. 2024; 14(21):1753. https://doi.org/10.3390/nano14211753
Chicago/Turabian StyleShabalina, Anastasiia V., Valeriy A. Kozlov, Ivan A. Popov, and Sergey V. Gudkov. 2024. "A Review on Recently Developed Antibacterial Composites of Inorganic Nanoparticles and Non-Hydrogel Polymers for Biomedical Applications" Nanomaterials 14, no. 21: 1753. https://doi.org/10.3390/nano14211753
APA StyleShabalina, A. V., Kozlov, V. A., Popov, I. A., & Gudkov, S. V. (2024). A Review on Recently Developed Antibacterial Composites of Inorganic Nanoparticles and Non-Hydrogel Polymers for Biomedical Applications. Nanomaterials, 14(21), 1753. https://doi.org/10.3390/nano14211753









