Selective Detection of Escherichia coli K12 and Staphylococcus aureus in Mixed Bacterial Communities Using a Single-Walled Carbon Nanotube (SWCNT)-Functionalized Electrochemical Immunosensor with Dielectrophoretic Concentration
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Instruments
2.2. Bacterial Sample Preparation
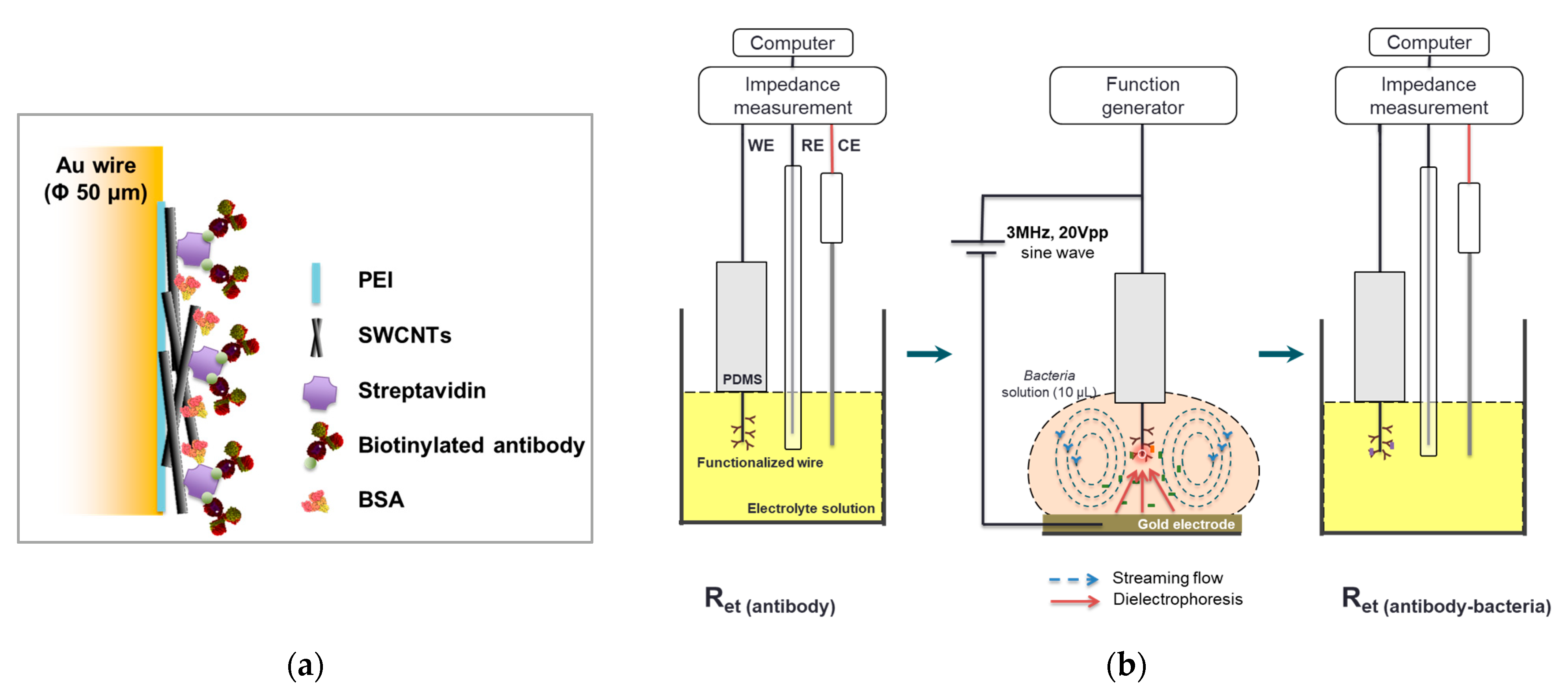
2.3. Functionalization of Microwire Surface
2.4. DEP-Based Concentration
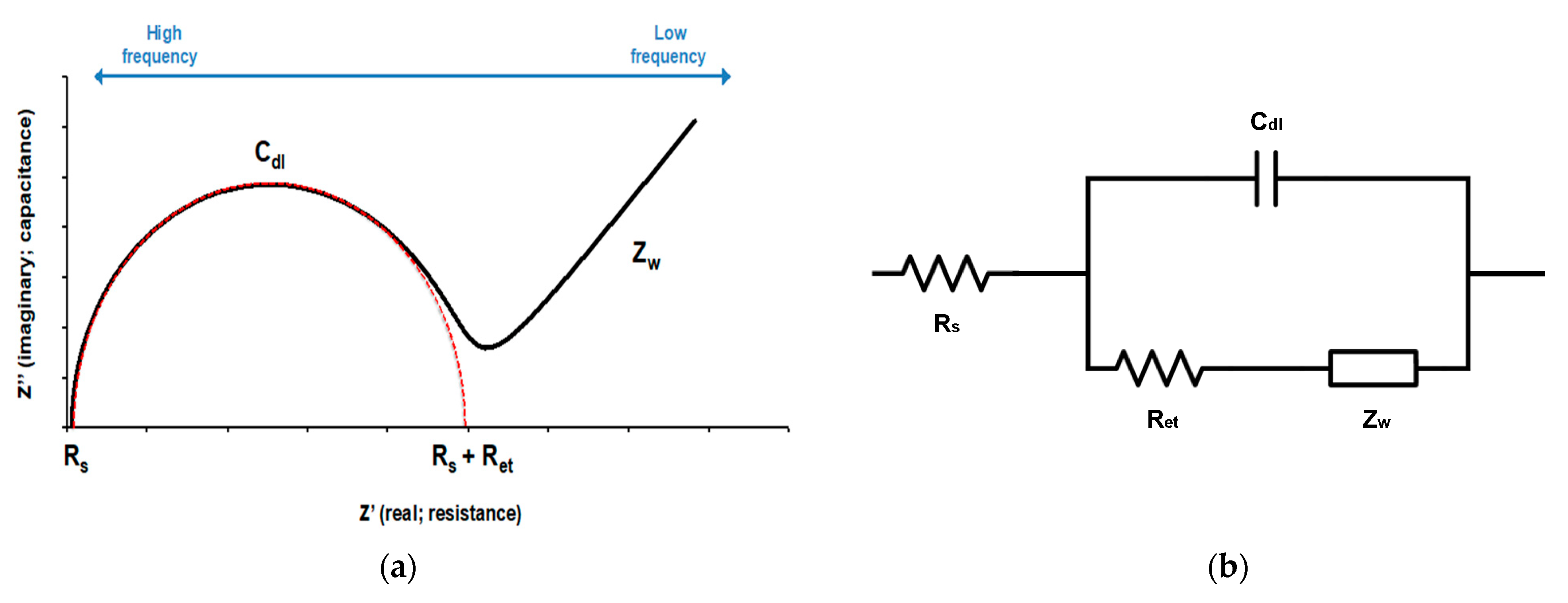
2.5. Impedance Measurement
2.6. Data Analysis
2.7. FESEM Visualization and Validation
3. Results and Discussion
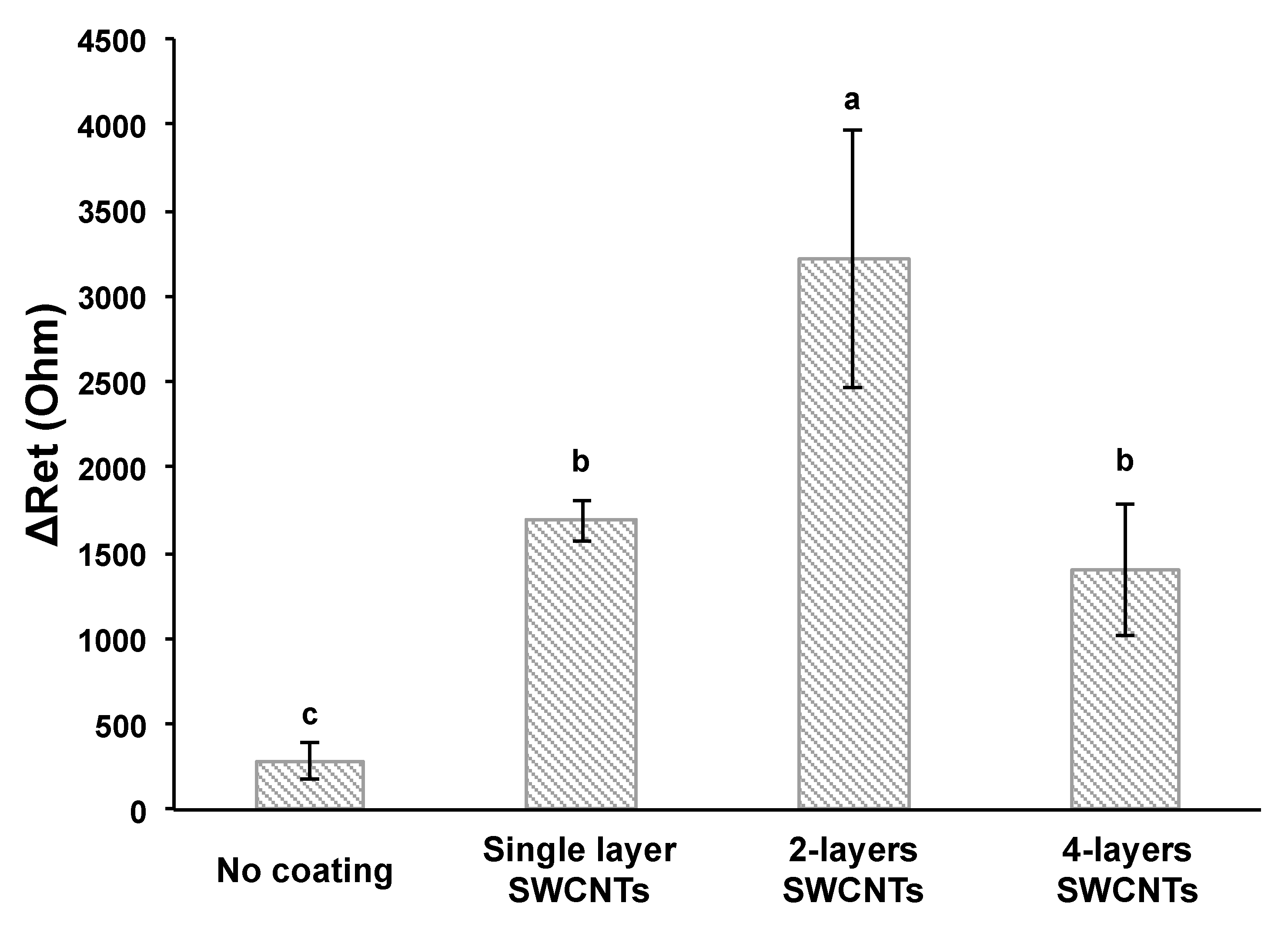
3.1. Effect of SWCNT Coating on Signal Enhancement
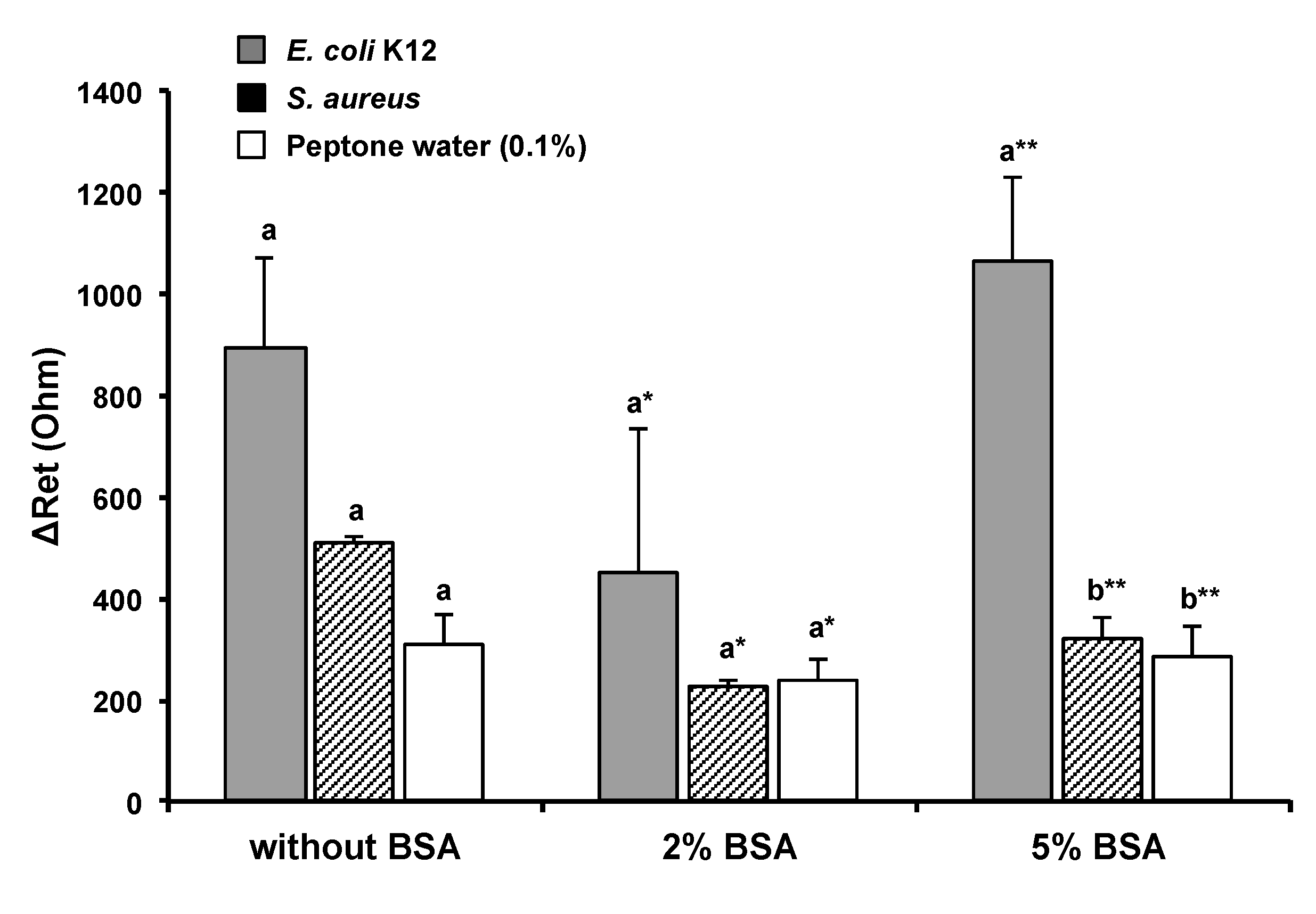
3.2. Effect of BSA Solution on Blocking Nonspecific Binding
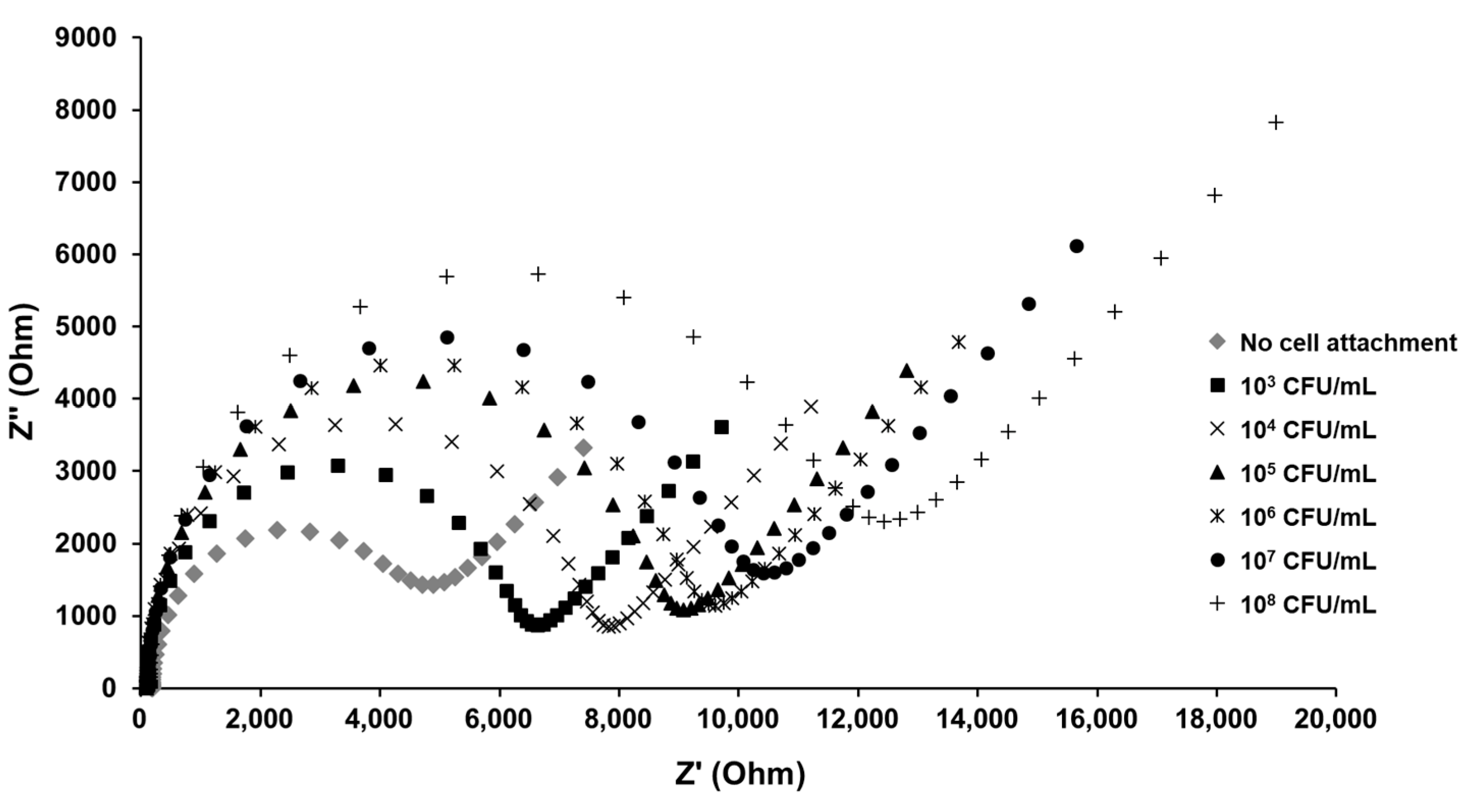
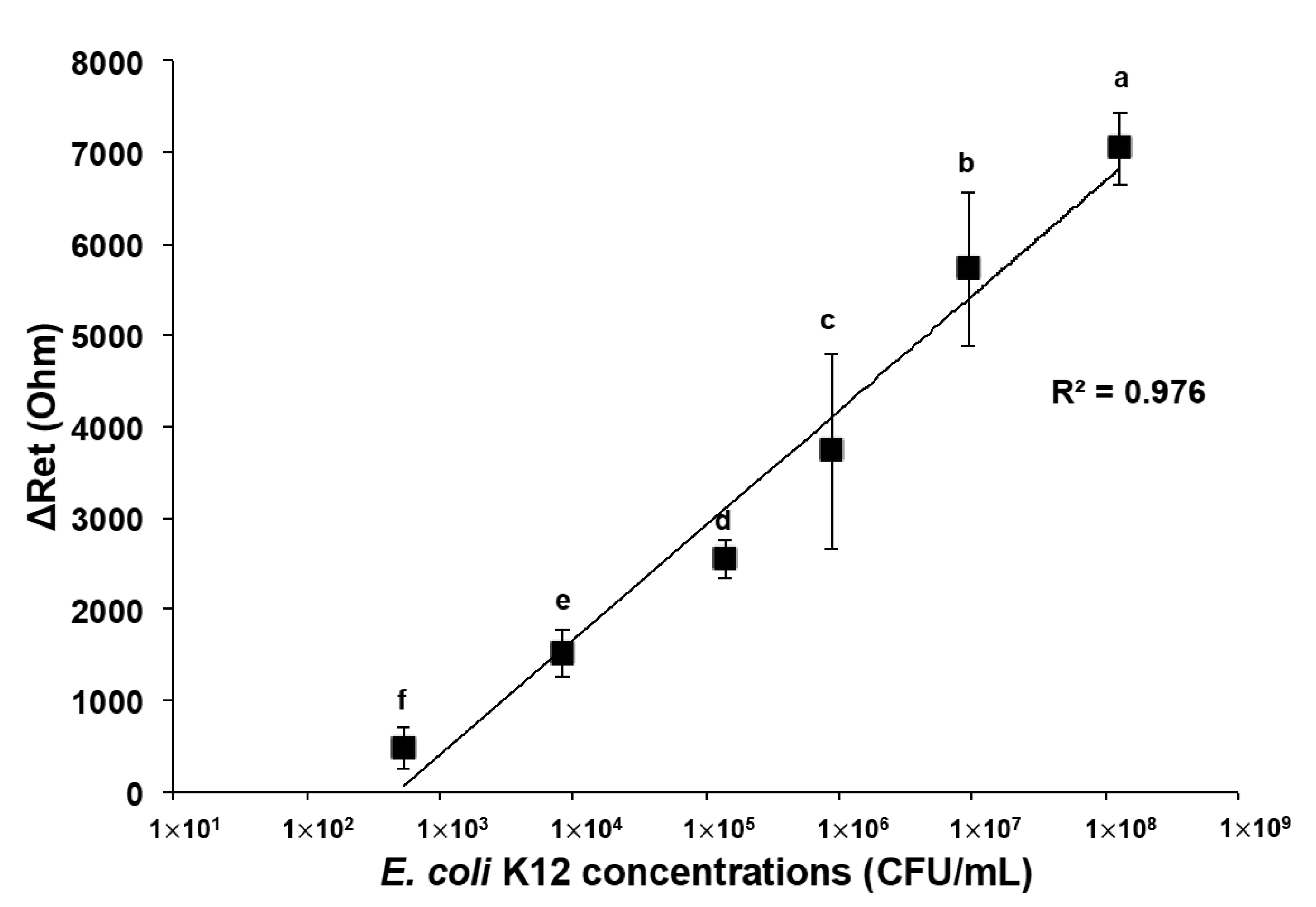
3.3. Detection of E. coli K12 in Pure and Mixed Solutions
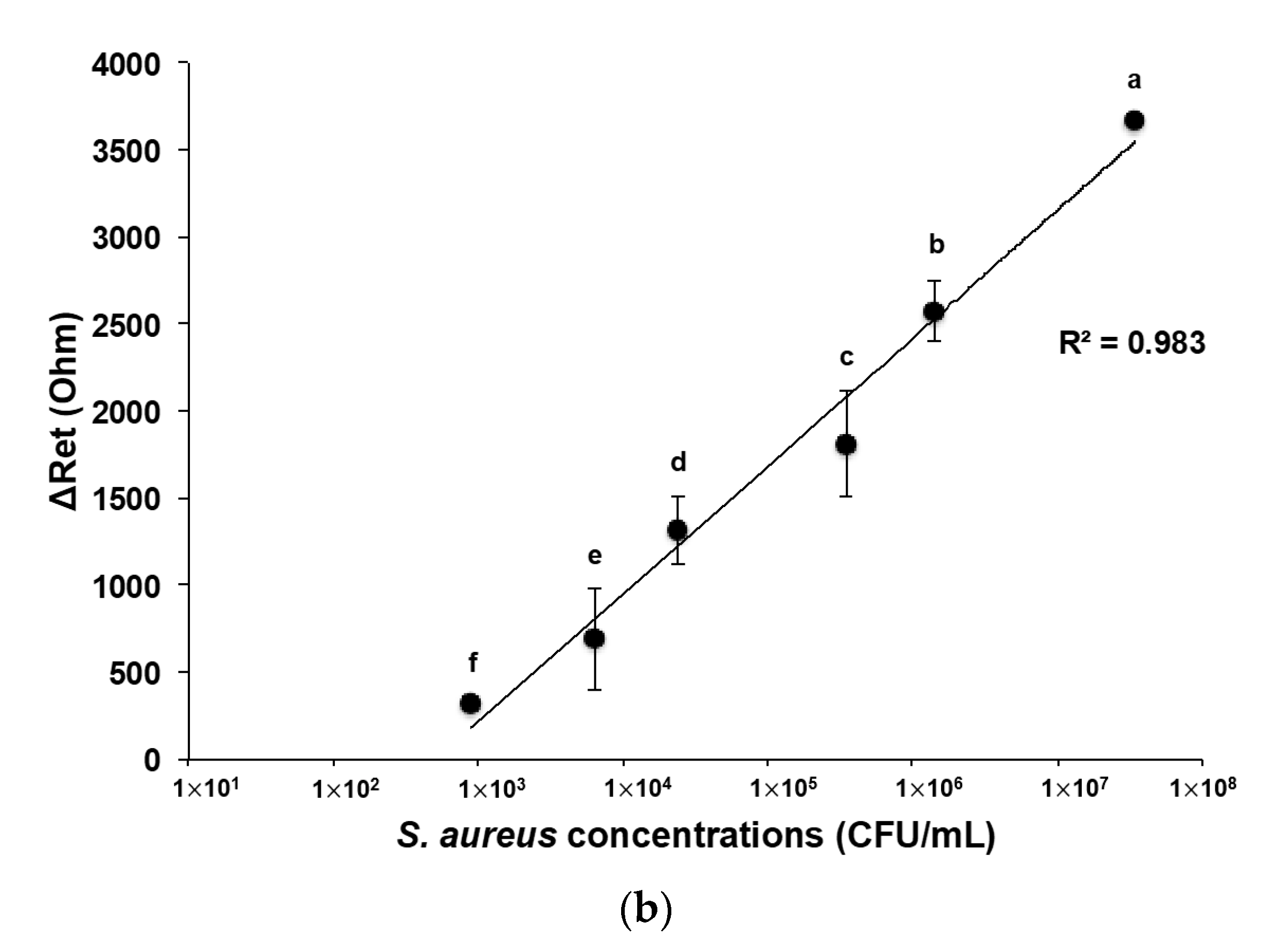
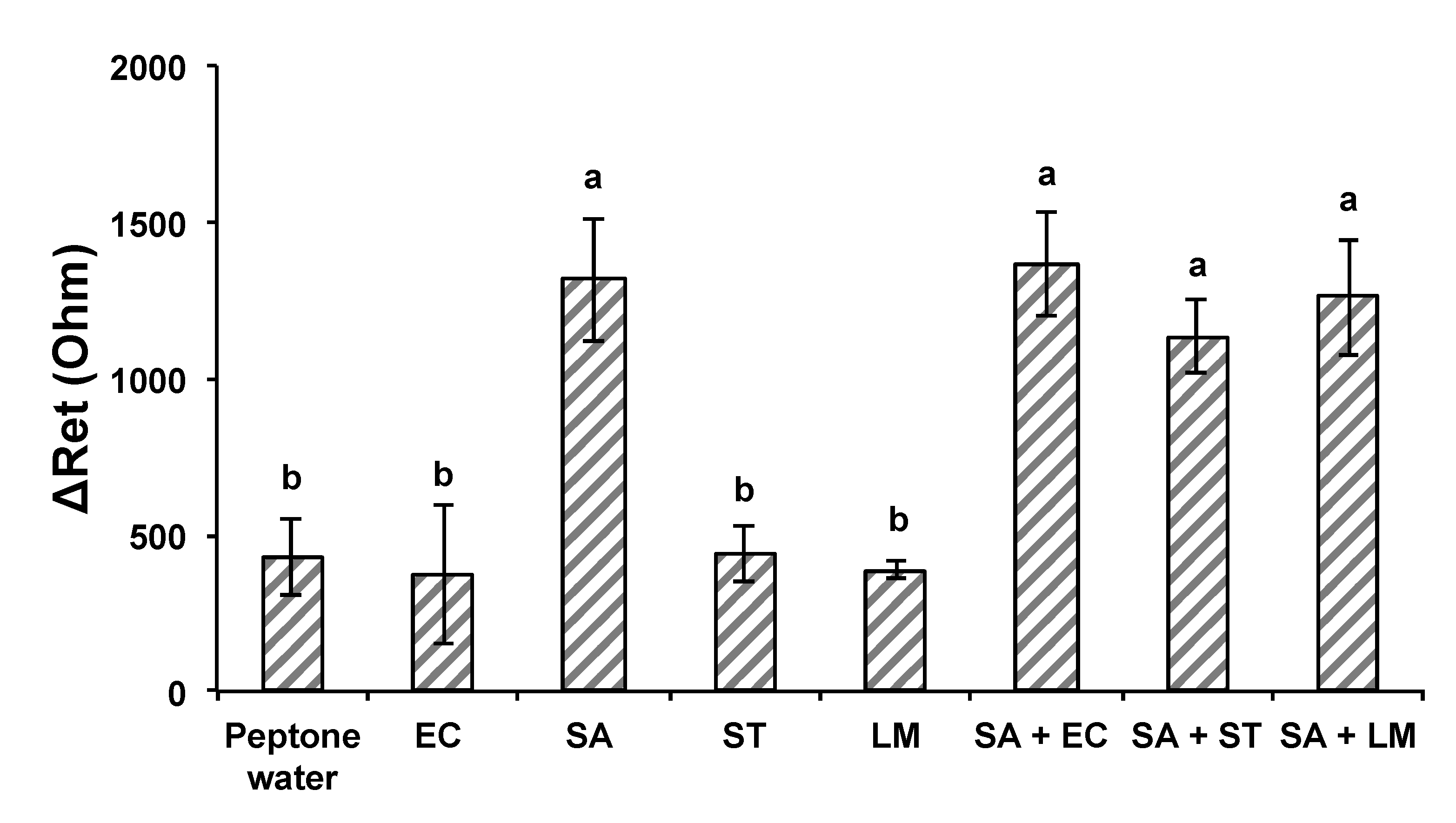
3.4. Developed Electrochemical Immunosensor for Detection of S. aureus in Pure and Mixed Solutions
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, Y.; Salazar, J.K. Culture-independent Rapid Detection Methods for Bacterial Pathogens and Toxins in Food Matrices. Compr. Rev. Food Sci. Food Saf. 2016, 15, 183–205. [Google Scholar] [CrossRef] [PubMed]
- Stevens, K.A.; Jaykus, L.-A. Bacterial Separation and Concentration from Complex Sample Matrices: A Review. Crit. Rev. Microbiol. 2004, 30, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Leonard, P.; Hearty, S.; Brennan, J.; Dunne, L.; Quinn, J.; Chakraborty, T.; O’kennedy, R. Advances in Biosensors for Detection of Pathogens in Food and Water. Enzyme Microb. Technol. 2003, 32, 3–13. [Google Scholar] [CrossRef]
- Zhao, X.; Lin, C.W.; Wang, J.; Oh, D.H. Advances in Rapid Detection Methods for Foodborne Pathogens. J. Microbiol. Biotechnol. 2014, 24, 297–312. [Google Scholar] [CrossRef]
- Yamada, K.; Choi, W.; Lee, I.; Cho, B.K.; Jun, S. Rapid Detection of Multiple Foodborne Pathogens Using a Nanoparticle-Functionalized Multi-Junction Biosensor. Biosens. Bioelectron. 2016, 77, 137–143. [Google Scholar] [CrossRef]
- Radke, S.M.; Alocilja, E.C. Design and Fabrication of a Microimpedance Biosensor for Bacterial Detection. IEEE Sens. J. 2004, 4, 434–440. [Google Scholar] [CrossRef]
- Tully, E.; Higson, S.P.; O’Kennedy, R. The Development of a “labeless” Immunosensor for the Detection of Listeria Monocytogenes Cell Surface Protein, Internalin B. Biosens. Bioelectron. 2008, 23, 906–912. [Google Scholar] [CrossRef]
- Velusamy, V.; Arshak, K.; Korostynska, O.; Oliwa, K.; Adley, C. An Overview of Foodborne Pathogen Detection: In the Perspective of Biosensors. Biotechnol. Adv. 2010, 28, 232–254. [Google Scholar] [CrossRef]
- Ahmed, A.; Rushworth, J.V.; Hirst, N.A.; Millner, P.A. Biosensors for Whole-Cell Bacterial Detection. Clin. Microbiol. Rev. 2014, 27, 631–646. [Google Scholar] [CrossRef]
- Lu, L.; Chee, G.; Yamada, K.; Jun, S. Electrochemical Impedance Spectroscopic Technique with a Functionalized Microwire Sensor for Rapid Detection of Foodborne Pathogens. Biosens. Bioelectron. 2013, 42, 492–495. [Google Scholar] [CrossRef]
- Joung, C.K.; Kim, H.N.; Lim, M.C.; Jeon, T.J.; Kim, H.Y.; Kim, Y.R. A Nanoporous Membrane-Based Impedimetric Immunosensor for Label-Free Detection of Pathogenic Bacteria in Whole Milk. Biosens. Bioelectron. 2013, 44, 210–215. [Google Scholar] [CrossRef]
- Jain, S. Development of an Antibody Functionalized Carbon Nanotube Biosensor for Foodborne Bacterial Pathogens. J. Biosens. Bioelectron. 2012, 01, 002. [Google Scholar] [CrossRef]
- Weber, J.E.; Pillai, S.; Ram, M.K.; Kumar, A.; Singh, S.R. Electrochemical Impedance-Based DNA Sensor Using a Modified Single Walled Carbon Nanotube Electrode. Mater. Sci. Eng. C 2011, 31, 821–825. [Google Scholar] [CrossRef]
- Putzbach, W.; Ronkainen, N.J. Immobilization Techniques in the Fabrication of Nanomaterial-Based Electrochemical Biosensors: A Review. Sensors 2013, 13, 4811–4840. [Google Scholar] [CrossRef]
- Maroto, A.; Balasubramanian, K.; Burghard, M.; Kern, K. Functionalized Metallic Carbon Nanotube Devices for PH Sensing. ChemPhysChem 2007, 8, 220–223. [Google Scholar] [CrossRef]
- Kim, J.H.; Hiraiwa, M.; Lee, H.B.; Lee, K.H.; Cangelosi, G.A.; Chung, J.H. Electrolyte-Free Amperometric Immunosensor Using a Dendritic Nanotip. RSC Adv. 2013, 3, 4281–4287. [Google Scholar] [CrossRef]
- Yamada, K.; Kim, C.T.; Kim, J.H.; Chung, J.H.; Lee, H.G.; Jun, S. Single Walled Carbon Nanotube-Based Junction Biosensor for Detection of Escherichia Coli. PLoS ONE 2014, 9, e0105767. [Google Scholar] [CrossRef]
- Hamada, R.; Takayama, H.; Shonishi, Y.; Mao, L.; Nakano, M.; Suehiro, J. A Rapid Bacteria Detection Technique Utilizing Impedance Measurement Combined with Positive and Negative Dielectrophoresis. Sens. Actuators B Chem. 2013, 181, 439–445. [Google Scholar] [CrossRef]
- Suehiro, J.; Ohtsubo, A.; Hatano, T.; Hara, M. Selective Detection of Bacteria by a Dielectrophoretic Impedance Measurement Method Using an Antibody-Immobilized Electrode Chip. Sens. Actuators B Chem. 2006, 119, 319–326. [Google Scholar] [CrossRef]
- Castillo-Fernandez, Ó.; Uria, N.; Muñoz, F.X.; Bratov, A. Cell Concentration Systems for Enhanced Biosensor Sensitivity. In Biosensors—Micro and Nanoscale Applications; InTech Open: London, UK, 2015. [Google Scholar]
- Pohl, H.A. Dielectrophoresis: The Behavior of Neutral Matter in Nonuniform Electric Fields. In Cambridge Monographs on Physics; Cambridge University Press: Cambridge, UK, 1978; Chapters 1–3. [Google Scholar]
- Fernández-Morales, F.H.; Duarte, J.E.; Samitier-Martí, J. Bacterial Handling under the Influence of Non-Uniform Electric Fields: Dielectrophoretic and Electrohydrodynamic Effects. An. Acad. Bras. Ciênc. 2008, 80, 627–638. [Google Scholar] [CrossRef]
- Kim, S.; Lu, L.; Chung, J.H.; Lee, K.; Li, Y.; Jun, S. A Microwire Sensor for Rapid Detection of Escherichia Coli K-12 in Fresh Produce. Innov. Food Sci. Emerg. Technol. 2011, 12, 617–622. [Google Scholar] [CrossRef]
- He, X.; Hu, C.; Guo, Q.; Wang, K.; Li, Y.; Shangguan, J. Rapid and Ultrasensitive Salmonella Typhimurium Quantification Using Positive Dielectrophoresis Driven On-Line Enrichment and Fluorescent Nanoparticles Label. Biosens. Bioelectron. 2013, 42, 460–466. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Lee, J.; Kim, Y.; Kim, B.; Lee, S. Analysis of Cell Separation Efficiency in Dielectrophoresis-Activated Cell Sorter. In Proceedings of the 2008 3rd IEEE International Conference on Nano/Micro Engineered and Molecular Systems, Sanya, China, 6–9 January 2008; pp. 965–969. [Google Scholar]
- Patel, S.; Showers, D.; Vedantam, P.; Tzeng, T.R.; Qian, S.; Xuan, X. Microfluidic Separation of Live and Dead Yeast Cells Using Reservoir-Based Dielectrophoresis. Biomicrofluidics 2012, 6, 034102. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Jun, S. Evaluation of a Microwire Sensor Functionalized to Detect Escherichia Coli Bacterial Cells. Biosens. Bioelectron. 2012, 36, 257–261. [Google Scholar] [CrossRef]
- Okuno, J.; Maehashi, K.; Kerman, K.; Takamura, Y.; Matsumoto, K.; Tamiya, E. Label-Free Immunosensor for Prostate-Specific Antigen Based on Single-Walled Carbon Nanotube Array-Modified Microelectrodes. Biosens. Bioelectron. 2007, 22, 2377–2381. [Google Scholar] [CrossRef]
- Gomes-Filho, S.L.R.; Dias, A.; Silva, M.M.S.; Silva, B.V.M.; Dutra, R.F. A Carbon Nanotube-Based Electrochemical Immunosensor for Cardiac Troponin T. Microchem. J. 2013, 109, 10–15. [Google Scholar] [CrossRef]
- Tlili, A.; Abdelghani, A.; Ameur, S.; Jaffrezic-Renault, N. Impedance Spectroscopy and Affinity Measurement of Specific Antibody-Antigen Interaction. Mater. Sci. Eng. C 2006, 26, 546–550. [Google Scholar] [CrossRef]
- Punbusayakul, N.; Talapatra, S.; Ajayan, P.M.; Surareungchai, W. Label-free as-grown double wall carbon nanotubes bundles for Salmonella typhimuriumimmunoassay. Chem. Cent. J. 2013, 7, 1–8. [Google Scholar] [CrossRef]
- Ruan, C.; Yang, L.; Li, Y. Immunobiosensor Chips for Detection of Escherichia c Oli O157: H7 Using Electrochemical Impedance Spectroscopy. Anal. Chem. 2002, 74, 4814–4820. [Google Scholar] [CrossRef]
- Yoo, J.; Cha, M.; Lee, J. Bacterial Concentration Detection with Dielectrophoresis and Capacitive Measurement. In Proceedings of the NSTI Nanotechnology Conference and Trade Show, Boston, MA, USA, 11 February 2008; pp. 1–5. [Google Scholar]



| Concentration | In Pure Sample (CFU/mL) | |
| Bacteria | Target | Nontarget |
| E. coli K12 | 8.53 × 103 | |
| S. aureus | 1.41 × 104 | |
| S. typhimurium | 1.60 × 104 | |
| L. monocytogenes | 3.40 × 104 | |
| Concentration | In Mixed Sample (CFU/mL) | |
| Bacteria | Target | Nontarget |
| E. coli K12 + S. aureus | 1.67 × 104 | 9.76 × 104 |
| E. coli K12 + S. typhimurium | 1.15 × 104 | 3.30 × 103 |
| E. coli K12 + L. monocytogenes | 1.90 × 104 | 9.00 × 102 |
| Concentration | In Pure Sample (CFU/mL) | |
| Bacteria | Target | Non-Target |
| E. coli K12 | 1.96 × 104 | |
| S. aureus | 2.39 × 104 | |
| S. typhimurium | 3.70 × 104 | |
| L. monocytogenes | 1.59 × 104 | |
| Concentration | In Mixed Sample (CFU/mL) | |
| Bacteria | Target | Nontarget |
| S. aureus + E. coli K12 | 5.65 × 104 | 2.33 × 104 |
| S. aureus + S. typhimurium | 6.08 × 103 | 9.08 × 103 |
| S. aureus + L. monocytogenes | 9.83 × 103 | 2.20 × 104 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, I.; So, H.; Kim, J.; Auh, J.-H.; Wall, M.M.; Li, Y.; Ho, K.; Jun, S. Selective Detection of Escherichia coli K12 and Staphylococcus aureus in Mixed Bacterial Communities Using a Single-Walled Carbon Nanotube (SWCNT)-Functionalized Electrochemical Immunosensor with Dielectrophoretic Concentration. Nanomaterials 2023, 13, 985. https://doi.org/10.3390/nano13060985
Lee I, So H, Kim J, Auh J-H, Wall MM, Li Y, Ho K, Jun S. Selective Detection of Escherichia coli K12 and Staphylococcus aureus in Mixed Bacterial Communities Using a Single-Walled Carbon Nanotube (SWCNT)-Functionalized Electrochemical Immunosensor with Dielectrophoretic Concentration. Nanomaterials. 2023; 13(6):985. https://doi.org/10.3390/nano13060985
Chicago/Turabian StyleLee, Inae, Heejin So, Jungyoon Kim, Joong-Hyuck Auh, Marisa M. Wall, Yong Li, Kacie Ho, and Soojin Jun. 2023. "Selective Detection of Escherichia coli K12 and Staphylococcus aureus in Mixed Bacterial Communities Using a Single-Walled Carbon Nanotube (SWCNT)-Functionalized Electrochemical Immunosensor with Dielectrophoretic Concentration" Nanomaterials 13, no. 6: 985. https://doi.org/10.3390/nano13060985
APA StyleLee, I., So, H., Kim, J., Auh, J.-H., Wall, M. M., Li, Y., Ho, K., & Jun, S. (2023). Selective Detection of Escherichia coli K12 and Staphylococcus aureus in Mixed Bacterial Communities Using a Single-Walled Carbon Nanotube (SWCNT)-Functionalized Electrochemical Immunosensor with Dielectrophoretic Concentration. Nanomaterials, 13(6), 985. https://doi.org/10.3390/nano13060985






