Functionalized Biochars as Supports for Ru/C Catalysts: Tunable and Efficient Materials for γ-Valerolactone Production
Abstract
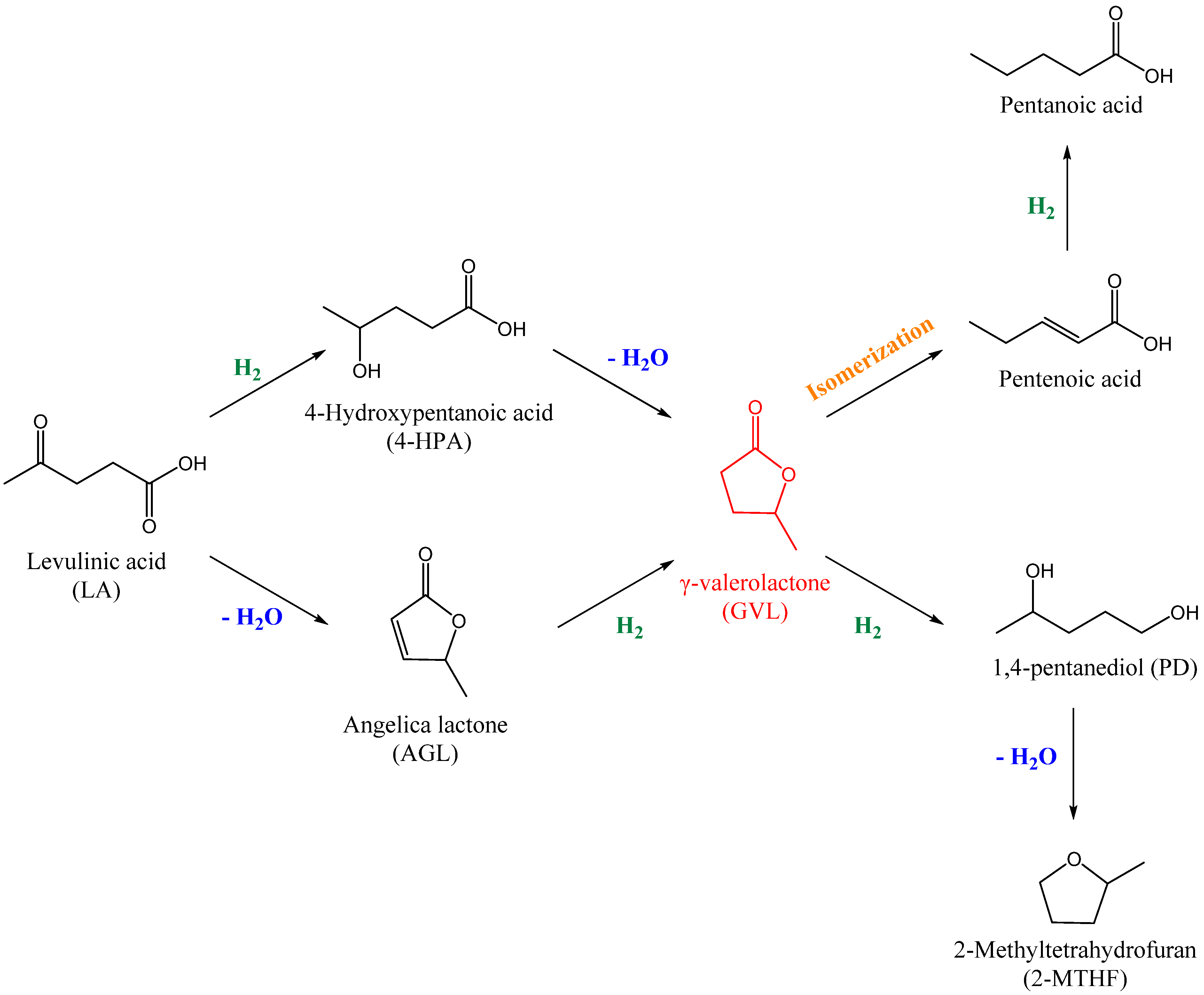
1. Introduction
2. Experimental
2.1. Cotton Stalks Biochars Elaboration
2.2. Metal Impregnation
2.3. Characterization Techniques
2.4. Catalytic Reaction and Analysis
3. Results and Discussion
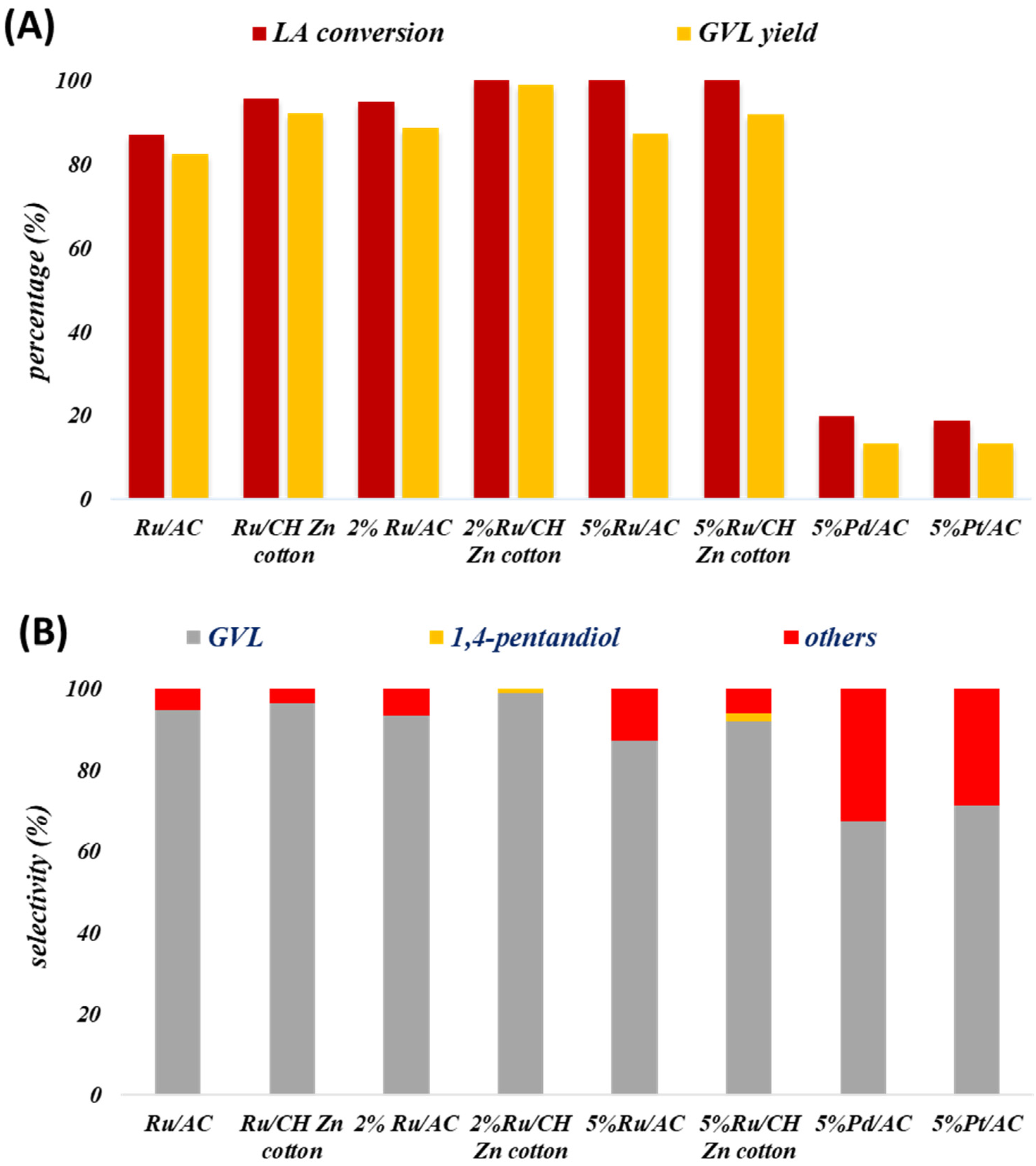
3.1. Catalyst Screening
3.2. Influence of the Support Nature
3.3. Time Effect
3.4. Temperature Effect
3.5. Pressure Effect
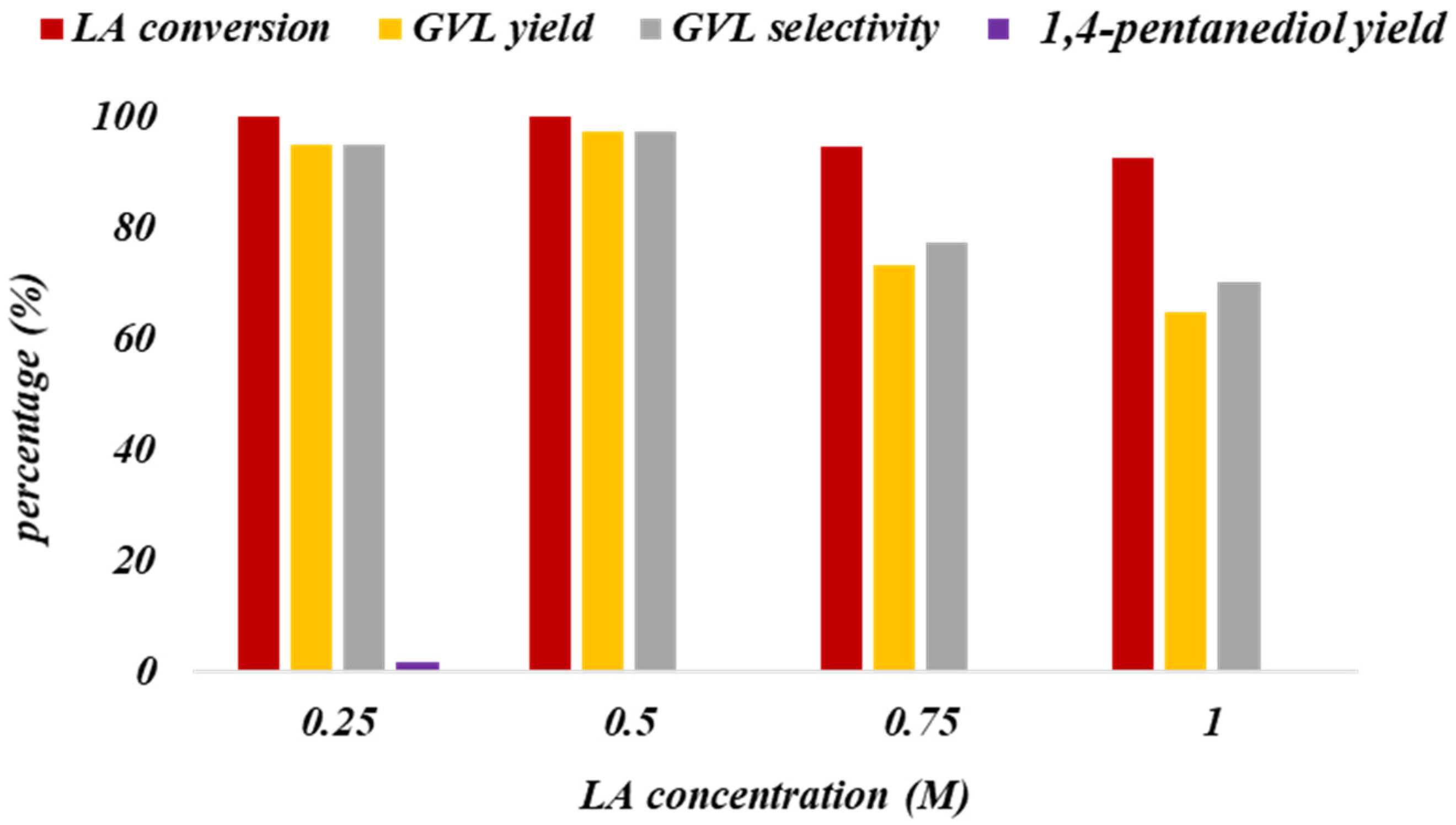
3.6. LA Concentration Effect
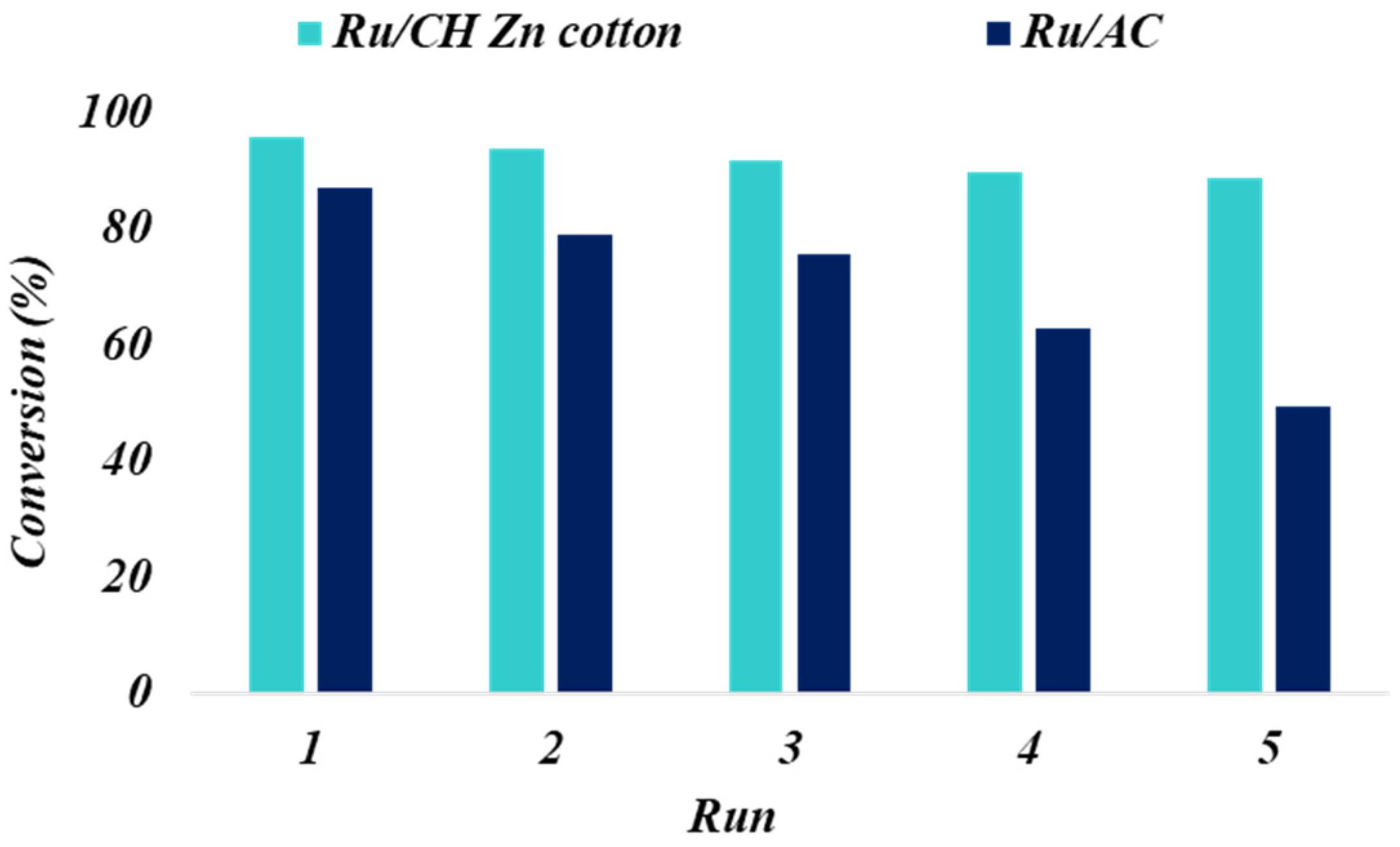
3.7. Catalyst Reuse
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dutta, S.; Bhat, N.S. Recent Advances in the Value Addition of Biomass-Derived Levulinic Acid: A Review Focusing on its Chemical Reactivity Patterns. ChemCatChem 2021, 13, 3202–3222. [Google Scholar] [CrossRef]
- Cai, B.; Zhang, Y.; Feng, J.; Huang, C.; Ma, T.; Pan, H. Highly efficient g-C3N4 supported ruthenium catalysts for the catalytic transfer hydrogenation of levulinic acid to liquid fuel γ-valerolactone. Renew. Energy 2021, 177, 652–662. [Google Scholar] [CrossRef]
- Kang, S.; Fu, J.; Zhang, G. From lignocellulosic biomass to levulinic acid: A review on acid-catalyzed hydrolysis. Renew. Sust. Energ. Rev. 2018, 94, 340–362. [Google Scholar] [CrossRef]
- Yan, K.; Jarvis, C.; Gu, J.; Yan, Y. Production and catalytic transformation of levulinic acid: A platform for speciality chemicals and fuels. Renew. Sust. Energ. Rev. 2015, 51, 986–997. [Google Scholar] [CrossRef]
- Horváth, I.T.; Mehdi, H.; Fábos, V.; Boda, L.; Mika, L.T. γ-Valerolactone—A sustainable liquid for energy and carbon-based chemicals. Green Chem. 2008, 10, 238–242. [Google Scholar] [CrossRef]
- Liguori, F.; Moreno-Marrodan, C.; Barbaro, P. Environmentally Friendly Synthesis of γ-Valerolactone by Direct Catalytic Conversion of Renewable Sources. ACS Catal. 2015, 5, 1882–1894. [Google Scholar] [CrossRef]
- Du, Y.; Chen, X.; Qi, J.; Wang, P.; Liang, C. Synthesis of Valeric Acid by Selective Electrocatalytic Hydrogenation of Biomass-Derived Levulinic Acid. Catalysts 2020, 10, 692. [Google Scholar] [CrossRef]
- Lv, J.; Rong, Z.; Sun, L.; Liu, C.; Lu, A.H.; Wang, Y.; Qu, J. Catalytic conversion of biomass-derived levulinic acid into alcohols over nanoporous Ru catalyst. Catal. Sci. Technol. 2018, 8, 975–979. [Google Scholar] [CrossRef]
- Chen, L.; Liu, Y.; Gu, C.; Feng, G.; Zhang, X.; Liu, J.; Zhang, Q.; Wang, C.; Ma, L. Selective Production of 2-Butanol from Hydrogenolysis of Levulinic Acid Catalyzed by the Non-precious NiMn Bimetallic Catalyst. ACS Sustain. Chem. Eng. 2021, 9, 46, 15603–15611. [Google Scholar] [CrossRef]
- Lange, J.; Price, R.; Ayou, P.; Louis, J.; Petrus, L.; Clarke, L.; Gosselink, H. Valeric Biofuels: A Platform of Cellulosic Transportation Fuels. Angew. Chem. Int. Ed. 2010, 49, 4479–4483. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Ruiz, J.C.; Pineda, A.; Balu, A.M.; Luque, R.; Campelo, J.M.; Romero, A.A.; Ramos-Fernández, J.M. Catalytic transformations of biomass derived acids into advanced biofuels. Catal Today 2012, 195, 162–168. [Google Scholar] [CrossRef]
- Koley, P.; Srinivasa Rao, B.; Chandra Shit, S.; Sabri, Y.; Mondal, J.; Tardio, J.; Lingaiah, N. One-pot conversion of levulinic acid into gamma-valerolactone over a stable Ru tungstosphosphoric acid catalyst. Fuel 2021, 289, 119900. [Google Scholar] [CrossRef]
- Shao, Y.; Ba, S.; Sun, K.; Gao, G.; Fan, M.; Wang, J.; Fan, H.; Zhang, L.; Hu, X. Selective production of γ-valerolactone or 1,4-pentanediol from levulinic acid/esters over Co-based catalyst: Importance of the synergy of hydrogenation sites and acidic sites. Chem. Eng. J. 2022, 429, 132433. [Google Scholar] [CrossRef]
- Xie, Z.; Chen, B.; Wu, H.; Liu, M.; Liu, H.; Zhang, J.; Yang, G.; Han, B. Highly efficient hydrogenation of levulinic acid into 2-methyltetrahydrofuran over Ni–Cu/Al2O3–ZrO2 bifunctional catalysts. Green Chem. 2019, 21, 606–613. [Google Scholar] [CrossRef]
- Yan, K.; Yang, Y.; Chai, J.; Lu, Y. Catalytic reactions of gamma-valerolactone: A platform to fuels and value-added chemicals. Appl. Catal. B 2015, 179, 292–304. [Google Scholar] [CrossRef]
- Wright, W.R.H.; Palkovits, R. Development of Heterogeneous Catalysts for the Conversion of Levulinic Acid to γ-Valerolactone. ChemSusChem 2012, 5, 1657–1667. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Lin, L.; Liu, S. Synthesis of γ-Valerolactone by Hydrogenation of Biomass-Derived Levulinic Acid over Ru/C Catalyst. Energy Fuels 2009, 23, 3853–3858. [Google Scholar] [CrossRef]
- Anjali, K.; Venkatesha, N.J.; Christopher, J.; Sakthivel, A. Rhodium Porphyrin Molecule-Based Catalysts for the Hydrogenation of Biomass Derived Levulinic Acid to Biofuel Additive γ-Valerolactone. New J. Chem. 2020, 44, 11064–11075. [Google Scholar] [CrossRef]
- Wang, S.; Huang, H.; Dorcet, V.; Roisnel, T.; Bruneau, C.; Fischmeister, C. Efficient Iridium Catalysts for Base-Free Hydrogenation of Levulinic Acid. Organometallics 2017, 36, 3152–3162. [Google Scholar] [CrossRef]
- Siddiqui, N.; Pendem, C.; Goyal, R.; Khatun, R.; Khan, T.S.; Samanta, C.; Chiang, K.; Shah, K.; Haider, M.A.; Bal, R. Study of γ-valerolactone production from hydrogenation of levulinic acid over nanostructured Pt-hydrotalcite catalysts at low temperatura. Fuel 2022, 323, 124272. [Google Scholar] [CrossRef]
- Yan, K.; Lafleur, T.; Wu, G.; Liao, J.; Ceng, C.; Xie, X. Highly selective production of value-added γ-valerolactone from biomass-derived levulinic acid using the robust Pd nanoparticles. Appl. Catal. A Gen. 2013, 468, 52–58. [Google Scholar] [CrossRef]
- Kamble, P.A.; Vinod, C.P.; Rathod, V.K.; Kantam, M.L. Hydrogenation of levulinic acid to gamma-valerolactone over nickel supported organoclay catalyst. Catal. Today 2023, 408, 36–49. [Google Scholar] [CrossRef]
- Yan, K.; Chen, A. Selective Hydrogenation of Furfural and Levulinic Acid to Biofuels on the Ecofriendly Cu–Fe Catalyst. Fuel 2014, 115, 101–108. [Google Scholar] [CrossRef]
- Ndolomingo, M.J.; Meijboom, R. Noble and Base-Metal Nanoparticles Supported on Mesoporous Metal Oxides: Efficient Catalysts for the Selective Hydrogenation of Levulinic Acid to γ-Valerolactone. Catal. Lett. 2019, 149, 2807–2822. [Google Scholar] [CrossRef]
- Seretis, A.; Diamantopoulou, P.; Thanou, I.; Tzevelekidis, P.; Fakas, C.; Lilas, P.; Papadogianakis, G. Recent Advances in Ruthenium-Catalyzed Hydrogenation Reactions of Renewable Biomass-Derived Levulinic Acid in Aqueous Media. Front. Chem. 2020, 8, 221. [Google Scholar] [CrossRef]
- Šivec, R.; Grilc, M.; Huš, M.; Likozar, B. Multiscale Modeling of (Hemi)cellulose Hydrolysis and Cascade Hydrotreatment of 5-Hydroxymethylfurfural, Furfural, and Levulinic Acid. Ind. Eng. Chem. Res. 2019, 58, 16018–16032. [Google Scholar] [CrossRef]
- Werpy, T.; Petersen, G. Top Value Added Chemicals from Biomass: Volume I—Results of Screening for Potential Candidates from Sugars and Synthesis Gas, United States. Available online: https://www.osti.gov/biblio/15008859/ (accessed on 2 February 2021).
- Hočevar, B.; Grilc, M.M.; Likozar, B. Aqueous Dehydration, Hydrogenation, and Hydrodeoxygenation Reactions of Bio-Based Mucic Acid over Ni, NiMo, Pt, Rh, and Ru on Neutral or Acidic Catalyst Supports. Catalysts 2019, 9, 286. [Google Scholar] [CrossRef]
- Bounoukta, C.E.; Megías-Sayago, C.; Rendón, N.; Ammari, F.; Penkova, A.; Ivanova, S.; Centeno, M.A.; Odriozola, J.A. Selective hydrodeoxygenation of levulinic acid to γ-valerolactone over Ru supported on functionalized carbon nanofibers. Sustainable Energy Fuels 2023, 7, 857. [Google Scholar] [CrossRef]
- Ye, L.; Han, Y.; Feng, J.; Lu, X. A review about GVL production from lignocellulose: Focusing on the full components utilization. Ind. Crop. Prod. 2020, 144, 112031. [Google Scholar] [CrossRef]
- Luo, W.; Deka, U.; Beale, A.M.; Eck, E.R.H.; Bruijnincx, P.C.A.; Weckhuysen, B.M. Ruthenium-catalyzed hydrogenation of levulinic acid: Influence of the support and solvent on catalyst selectivity and stability. J. Catal. 2013, 301, 175–186. [Google Scholar] [CrossRef]
- Kuwahara, Y.; Kaburagi, W.; Osada, Y.; Fujitani, T.; Yamashita, H. Catalytic transfer hydrogenation of biomass-derived levulinic acid and its esters to γ-valerolactone over ZrO2 catalyst supported on SBA-15 silica. Catal. Today 2017, 281, 418–428. [Google Scholar] [CrossRef]
- Maumela, M.; Marx, S.; Meijboom, R. Heterogeneous Ru Catalysts as the Emerging Potential Superior Catalysts in the Selective Hydrogenation of Bio-Derived Levulinic Acid to γ-Valerolactone: Effect of Particle Size, Solvent, and Support on Activity, Stability, and Selectivity. Catalysts 2021, 11, 292. [Google Scholar] [CrossRef]
- Ruiz-Bernal, Z.; Lillo-Ródenas, M.Á.; Román-Martínez, M.d.C. Ru Catalysts Supported on Commercial and Biomass-Derived Activated Carbons for the Transformation of Levulinic Acid into γ-Valerolactone under Mild Conditions. Catalysts 2021, 11, 559. [Google Scholar] [CrossRef]
- Cattaneo, S.; Stucchi, M.; Veith, G.M.; Prati, L.; Wang, D.; Wang, W.; Villa, A. Ru supported on micro and mesoporous carbons as catalysts for biomass-derived molecules hydrogenation. Catal. Today 2020, 357, 143–151. [Google Scholar] [CrossRef]
- Molleti, J.; Tiwari, M.S.; Yadav, G.D. Novel synthesis of Ru/OMS catalyst by solvent-free method: Selective hydrogenation of levulinic acid to γ-valerolactone in aqueous medium and kinetic modelling. Chem. Eng. J. 2018, 334, 2488–2499. [Google Scholar] [CrossRef]
- Santos, J.L.; Megías-Sayago, C.; Ivanova, S.; Centeno, M.Á.; Odriozola, J.A. Functionalized biochars as supports for Pd/C catalysts for efficient hydrogen production from formic acid. Appl. Catal. B Environ. 2021, 282, 119615. [Google Scholar] [CrossRef]
- Lozano-Castello, D.; Calo, J.M.; Cazorla-Amorós, D.; Linares-Solano, A. Carbon activation with KOH as explored by temperature programmed techniques, and the effects of hydrogen. Carbon 2007, 45, 2529–2536. [Google Scholar] [CrossRef]
- Guo, Y.; Rockstraw, D.A. Physical and chemical properties of carbons synthesized from xylan, cellulose and Kraft lignin by H3PO4 activation. Carbon 2006, 44, 1464–1475. [Google Scholar] [CrossRef]
- Xiong, X.; Yu, I.K.M.; Cao, L.; Tsang, D.C.W.; Zhang, S. A review of biochar-based catalysts for chemical synthesis, biofuel production, and pollution control. Bioresour. Technol. 2017, 246, 254–270. [Google Scholar] [CrossRef]
- Xu, J.; Cao, Z.; Zhang, Y.; Yuan, Z.; Lou, Z.; Xu, X. A review of functionalized carbon nanotubes and graphene for heavy metal adsorption from water: Preparation, application, and mechanism. Chemosphere 2018, 195, 351–364. [Google Scholar] [CrossRef]
- Feng, J.; Gu, X.; Xue, Y.; Han, Y. Production of γ-valerolactone from levulinic acid over a Ru/C catalyst using formic acid as the sole hydrogen source. Sci. Total Environ. 2018, 633, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.L.; Mäki-Arvela, P.; Monzón, A.; Murzin, D.Y.; Centeno, M.Á. Metal catalysts supported on biochars: Part I synthesis and characterization. Appl. Catal. B Environ. 2020, 268, 118423. [Google Scholar] [CrossRef]
- Kim, Y.; Oh, J.I.; Vithanage, M.; Park, Y.K.; Lee, J.; Kwon, E.E. Modification of biochar properties using CO2. Chem. Eng. J. 2009, 372, 383–389. [Google Scholar] [CrossRef]
- Yan, W.; Mahurin, S.M.; Chen, B.; Overbury, S.H.; Dai, S. Effect of Supporting Surface Layers on Catalytic Activities of Gold Nanoparticles in CO Oxidation. J. Phys. Chem. B 2005, 109, 15489–15496. [Google Scholar] [CrossRef] [PubMed]
- Mamun, O.; Walker, E.; Faheem, M.; Bond, J.Q.; Heyden, A. Theoretical Investigation of the Hydrodeoxygenation of Levulinic Acid to γ-Valerolactone over Ru(0001). ACS Catal. 2007, 7, 215–228. [Google Scholar] [CrossRef]
- Pavlova, A.; Meijer, E.J. Understanding the Role of Water in Aqueous Ruthenium-Catalyzed Transfer Hydrogenation of Ketones. ChemPhysChem 2012, 13, 3492–3496. [Google Scholar] [CrossRef]
- Piskun, A.S.; van de Bovenkamp, H.H.; Rasrendra, C.B.; Winkelman, J.G.M.; Heeres, H.J. Kinetic modeling of levulinic acid hydrogenation to γ-valerolactone in water using a carbon supported Ru catalyst. Appl. Catal. A Gen. 2016, 525, 158–167. [Google Scholar] [CrossRef]
- Wei, Z.; Li, X.; Deng, J.; Wang, J.; Li, H.; Wang, Y. Improved catalytic activity and stability for hydrogenation of levulinic acid by Ru/N-doped hierarchically porous carbon. Mol. Catal. 2018, 448, 100–107. [Google Scholar] [CrossRef]
- Wei, Z.; Lou, J.; Su, C.; Guo, D.; Liu, Y.; Deng, S. An Efficient and Reusable Embedded Ru Catalyst for the Hydrogenolysis of Levulinic Acid to γ-Valerolactone. ChemSusChem 2017, 10, 1720–1732. [Google Scholar] [CrossRef]
- Liu, X.; Lan, G.; Boyjoo, Y.; Qian, L.; Gu, S.; Price, C.A.H.; Wang, L.; Li, Y.; Lu, I.J. N-doped carbon spheres impregnated with highly monodispersed ruthenium nanoparticles as a hydrogenation catalyst. Chem. Eng. J. 2019, 374, 895–903. [Google Scholar] [CrossRef]
- Galletti, A.M.R.; Antonetti, C.; Luise, V.D.; Martinelli, M. A sustainable process for the production of γ-valerolactone by hydrogenation of biomass-derived levulinic acid. Green Chem. 2012, 14, 688–694. [Google Scholar] [CrossRef]





| Samples | C | H | N | S | O * | C/H |
|---|---|---|---|---|---|---|
| Ccotton | 56.59 | 1.14 | 0.54 | 0.52 | 41.21 | 49.73 |
| CH cotton | 77.84 | 2.31 | 1.28 | 0.53 | 18.04 | 33.65 |
| CZnCotton | 58.17 | 1.74 | 1.25 | 0.23 | 38.61 | 33.45 |
| CH ZnCotton | 83.02 | 1.55 | 1.92 | 0.23 | 13.28 | 53.69 |
| AC | 83.86 | 0.59 | 0.14 | 0.43 | 13.44 | 143.89 |
| Samples | BET Surface Area (m2/g) | Average Pore Size (nm) | Pore Volume (cm3/g) | Surfacemicro (m2/g) | Surfacemeso (m2/g) |
|---|---|---|---|---|---|
| Ru/CCotton | 103 | 5.8 | 0.03 | 71 | 21 |
| Ru/CH Cotton | 436 | 3.88 | 0.32 | 384 | 33 |
| Ru/C Zn cotton | 676 | 4.92 | 0.49 | 236 | 432 |
| Ru/CH Zn cotton | 1422 | 3.05 | 0.92 | 14 | 1210 |
| Ru/AC | 969 | 6.3 | 0.91 | 405 | 423 |
| Samples | C Crystal Size, Lc | Particles Size TEM, nm | Raman I(D)/I(G) | Metal wt.% ICP | Dispersion (%) | TON * |
|---|---|---|---|---|---|---|
| Ru/C cotton | 12 | n.a. | 1.1 | 0.98 | n.a. | - |
| Ru/CH Cotton | 13 | 7.1 | 0.94 | 1 | 31.4 | 9957 |
| Ru/CZn Cotton | 15 | 3.1 | 1.1 | 1.1 | 68.8 | 6953 |
| Ru/C H Zn cotton | 12 | 2.5 | 0.96 | 1.2 | 84.3 | 5472 |
| Ru/AC | 13 | 3.9 | 1.18 | 0.96 | 55.3 | 7873 |
| Catalyst | LA/Ru * | t, min | T, °C | P H2, bars | TOF **, h−1 | Conv. % | Yield, % | Ref. |
|---|---|---|---|---|---|---|---|---|
| 3%Ru/NHPC | - | 180 | 50 | 10 | 338 | 99 | 99 | [49] |
| 5%Ru/NOMC | 350 | 180 | 120 | 13 | 438 | 92 | 91 | [50] |
| 1%Ru/N-CS | 5000 | 60 | 70 | 40 | 86.2 | 82 | 82 | [51] |
| 1%Ru/NCS | 10,000 | 60 | 70 | 40 | 9851 | 51 | 51 | [51] |
| 2%Ru/r-GO | - | - | 40 | 40 | 5520 | 52 | 52 | [48] |
| 5 %Ru/AC+A70 | 350 | 180 | 70 | 5 | 558.2 | 98 | 97 | [52] |
| 1%Ru/CNF-ILs | 2500 | 120 | 100 | 10 | 9888 | 96 | 90 | [50] |
| Ru/CH Zn cotton | 5000 | 120 | 100 | 10 | 5203 | 95 | 92 | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bounoukta, C.E.; Megías-Sayago, C.; Navarro, J.C.; Ammari, F.; Ivanova, S.; Centeno, M.Á.; Odriozola, J.A. Functionalized Biochars as Supports for Ru/C Catalysts: Tunable and Efficient Materials for γ-Valerolactone Production. Nanomaterials 2023, 13, 1129. https://doi.org/10.3390/nano13061129
Bounoukta CE, Megías-Sayago C, Navarro JC, Ammari F, Ivanova S, Centeno MÁ, Odriozola JA. Functionalized Biochars as Supports for Ru/C Catalysts: Tunable and Efficient Materials for γ-Valerolactone Production. Nanomaterials. 2023; 13(6):1129. https://doi.org/10.3390/nano13061129
Chicago/Turabian StyleBounoukta, Charf Eddine, Cristina Megías-Sayago, Juan Carlos Navarro, Fatima Ammari, Svetlana Ivanova, Miguel Ángel Centeno, and Jose Antonio Odriozola. 2023. "Functionalized Biochars as Supports for Ru/C Catalysts: Tunable and Efficient Materials for γ-Valerolactone Production" Nanomaterials 13, no. 6: 1129. https://doi.org/10.3390/nano13061129
APA StyleBounoukta, C. E., Megías-Sayago, C., Navarro, J. C., Ammari, F., Ivanova, S., Centeno, M. Á., & Odriozola, J. A. (2023). Functionalized Biochars as Supports for Ru/C Catalysts: Tunable and Efficient Materials for γ-Valerolactone Production. Nanomaterials, 13(6), 1129. https://doi.org/10.3390/nano13061129