Electrical and Structural Properties of Si1−xGex Nanowires Prepared from a Single-Source Precursor
Abstract
1. Introduction
2. Materials and Methods
CVD Process and Thin Film Characterization
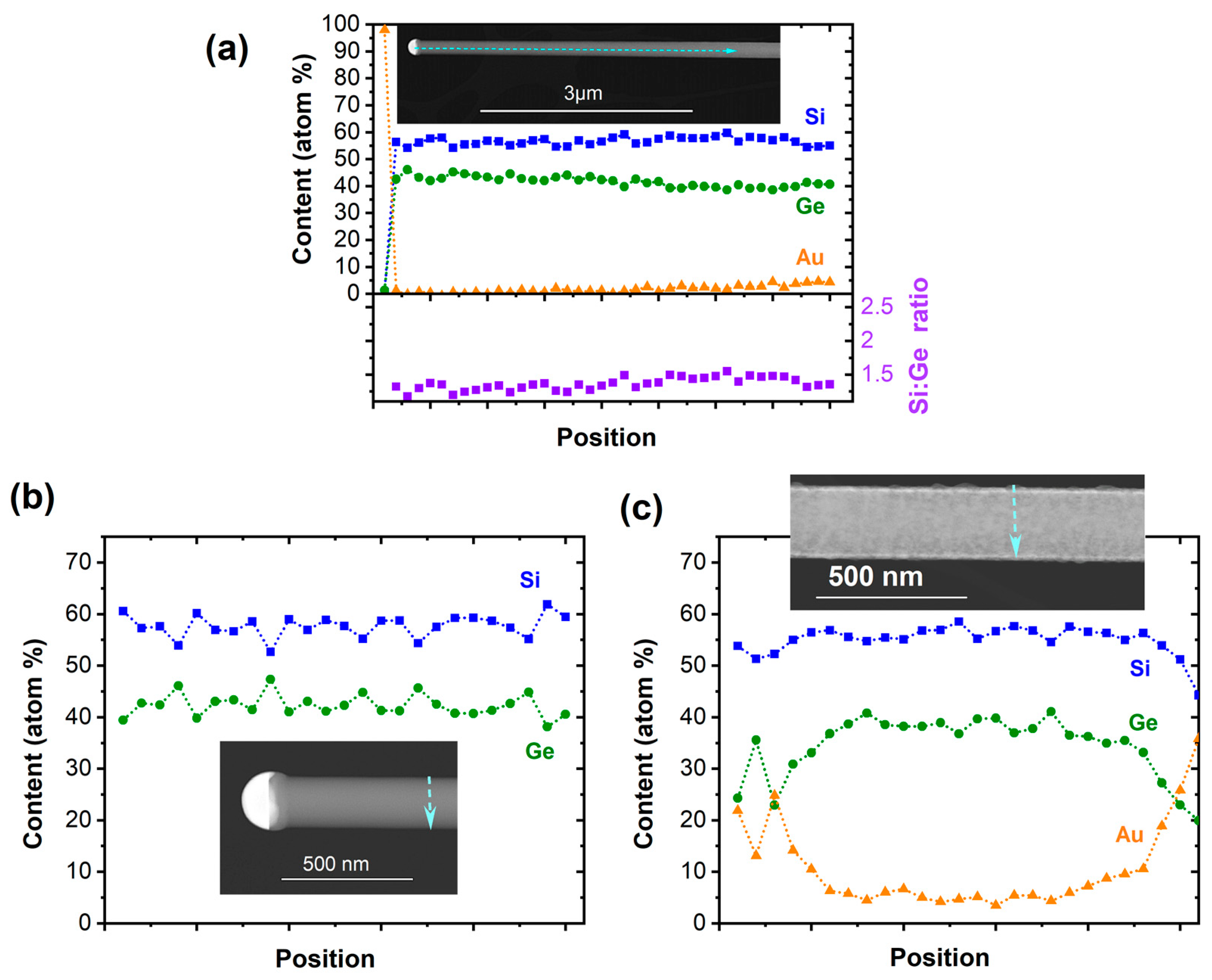
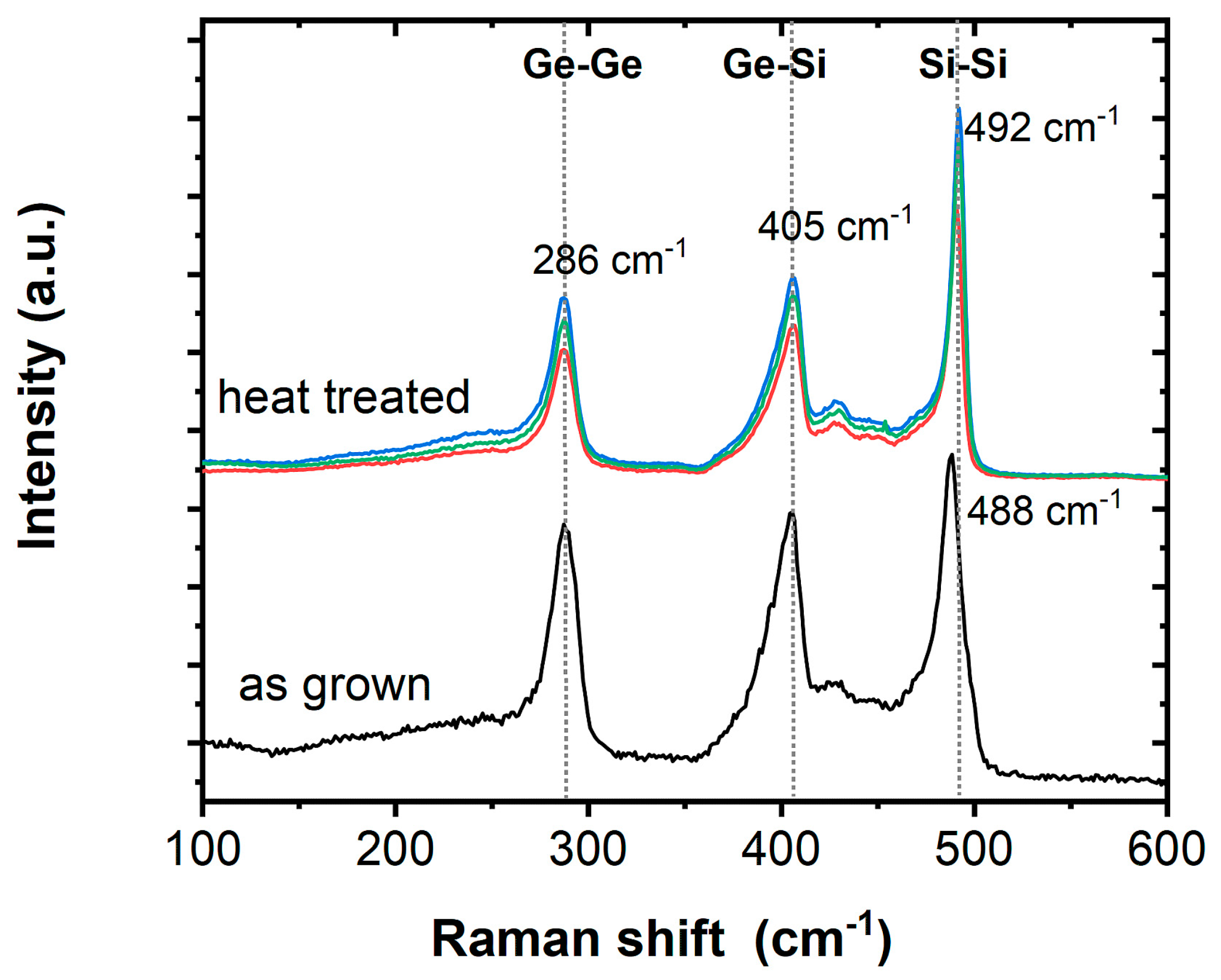
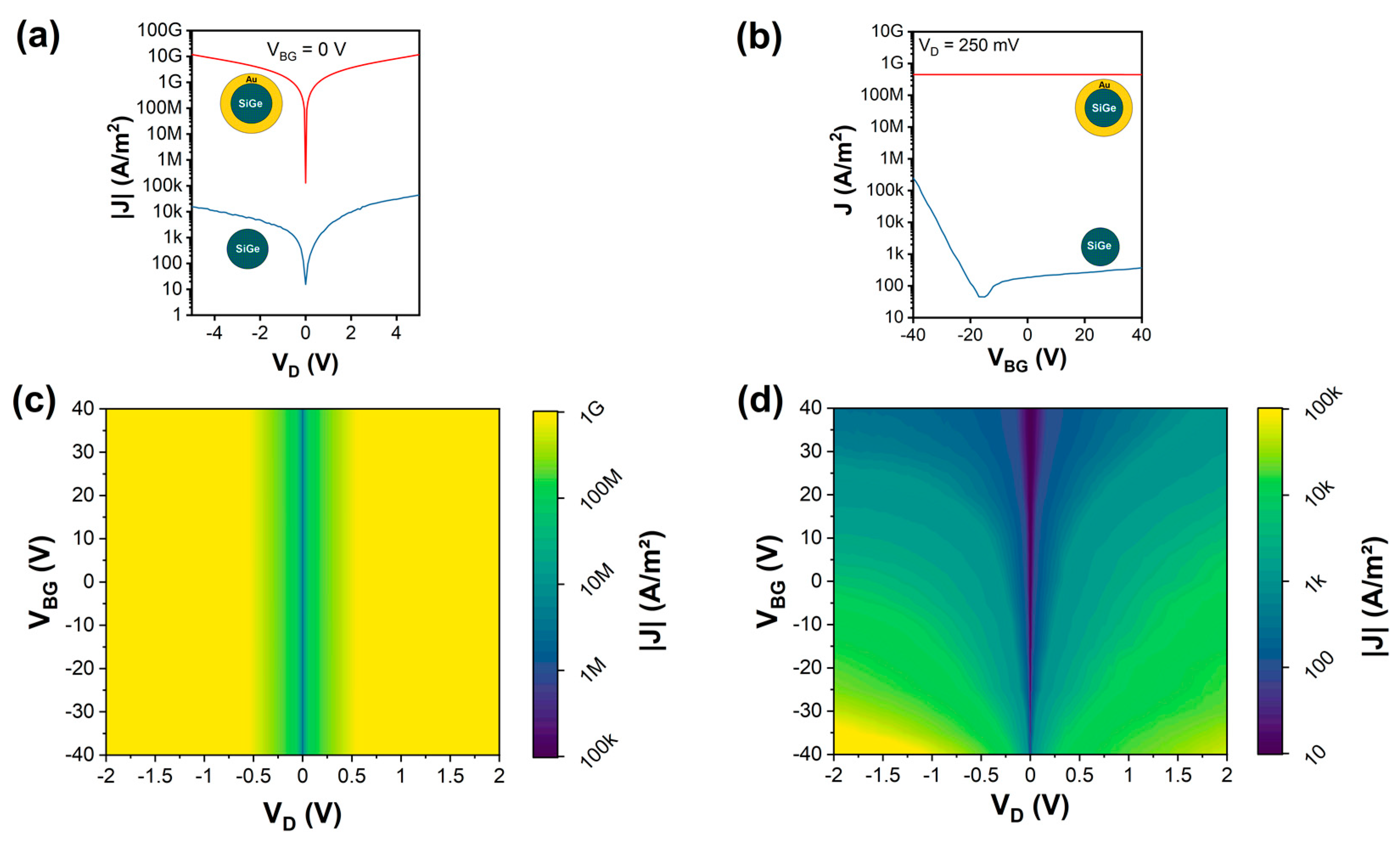
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jia, C.; Lin, Z.; Huang, Y.; Duan, X. Nanowire Electronics: From Nanoscale to Macroscale. Chem. Rev. 2019, 119, 9074–9135. [Google Scholar] [CrossRef]
- Hrachowina, L.; Chen, Y.; Barrigón, E.; Wallenberg, R.; Borgström, M.T. Realization of axially defined GaInP/InP/InAsP triple-junction photovoltaic nanowires for high-performance solar cells. Mater. Today Energy 2022, 27, 101050. [Google Scholar] [CrossRef]
- Fedeli, J.M.; Nicoletti, S. Mid-Infrared (Mid-IR) Silicon-Based Photonics. Proc. IEEE 2018, 106, 2302–2312. [Google Scholar] [CrossRef]
- Li, Y.; Wang, G.; Akbari-Saatlu, M.; Procek, M.; Radamson, H.H. Si and SiGe Nanowire for Micro-Thermoelectric Generator: A Review of the Current State of the Art. Front. Mater. 2021, 8, 611078. [Google Scholar] [CrossRef]
- Lee, E.K.; Yin, L.; Lee, Y.; Lee, J.W.; Lee, S.J.; Lee, J.; Cha, S.N.; Whang, D.; Hwang, G.S.; Hippalgaonkar, K.; et al. Large Thermoelectric Figure-of-Merits from SiGe Nanowires by Simultaneously Measuring Electrical and Thermal Transport Properties. Nano Lett. 2012, 12, 2918–2923. [Google Scholar] [CrossRef] [PubMed]
- Stokes, K.; Geaney, H.; Flynn, G.; Sheehan, M.; Kennedy, T.; Ryan, K.M. Direct Synthesis of Alloyed Si1−xGex Nanowires for Performance-Tunable Lithium Ion Battery Anodes. ACS Nano 2017, 11, 10088–10096. [Google Scholar] [CrossRef]
- Özgün, A.; Garipcan, B. Silicon nanowires in biomedicine. In Silicon Nanomaterials Sourcebook; Sattler, K.D., Ed.; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Güniat, L.; Caroff, P.; Fontcuberta i Morral, A. Vapor Phase Growth of Semiconductor Nanowires: Key Developments and Open Questions. Chem. Rev. 2019, 119, 8958–8971. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yuan, W.; Kang, W.; Ye, Y.; Pan, Q.; Zhang, X.; Ke, Y.; Wang, C.; Qiu, Z.; Tang, Y. A review on silicon nanowire-based anodes for next-generation high-performance lithium-ion batteries from a material-based perspective. Sustain. Energy Fuels 2020, 4, 1577–1594. [Google Scholar] [CrossRef]
- Amato, M.; Palummo, M.; Rurali, R.; Ossicini, S. Silicon–Germanium Nanowires: Chemistry and Physics in Play, from Basic Principles to Advanced Applications. Chem. Rev. 2014, 114, 1371–1412. [Google Scholar] [CrossRef] [PubMed]
- Barth, S.; Hernandez-Ramirez, F.; Holmes, J.D.; Romano-Rodriguez, A. Synthesis and applications of one-dimensional semiconductors. Prog. Mater. Sci. 2010, 55, 563–627. [Google Scholar] [CrossRef]
- Schmidt, V.; Wittemann, J.V.; Gösele, U. Growth, Thermodynamics, and Electrical Properties of Silicon Nanowires. Chem. Rev. 2010, 110, 361–388. [Google Scholar] [CrossRef] [PubMed]
- Barth, S.; Seifner, M.S.; Maldonado, S. Metastable Group IV Allotropes and Solid Solutions: Nanoparticles and Nanowires. Chem. Mater. 2020, 32, 2703–2741. [Google Scholar] [CrossRef]
- Singh, N.; Buddharaju, K.D.; Manhas, S.K.; Agarwal, A.; Rustagi, S.C.; Lo, G.Q.; Balasubramanian, N.; Kwong, D.L. Si, SiGe Nanowire Devices by Top–Down Technology and Their Applications. IEEE Trans. Electron Devices 2008, 55, 3107–3118. [Google Scholar] [CrossRef]
- Harame, D.; Buquet, J.; Caymax, M.; Cressler, J.; Koester, S.; Masini, G.; Mijazaki, S.; Reznicek, A.; Rim, K.; Takagi, S.; et al. SiGe, Ge, and Related Compounds: Materials, Processing, and Devices; Electrochemical Society (ECS): Pennington, NJ, USA, 2008; Volume 16. [Google Scholar]
- Olesinski, R.W.; Abbaschian, G.J. The Ge–Si (Germanium-Silicon) system. Bull. Alloy Phase Diagr. 1984, 5, 180–183. [Google Scholar] [CrossRef]
- Aberl, J.; Brehm, M.; Fromherz, T.; Schuster, J.; Frigerio, J.; Rauter, P. SiGe quantum well infrared photodetectors on strained-silicon-on-insulator. Opt. Express 2019, 27, 32009–32018. [Google Scholar] [CrossRef]
- Wang, G.L.; Moeen, M.; Abedin, A.; Kolahdouz, M.; Luo, J.; Qin, C.L.; Zhu, H.L.; Yan, J.; Yin, H.Z.; Li, J.F.; et al. Optimization of SiGe selective epitaxy for source/drain engineering in 22 nm node complementary metal-oxide semiconductor (CMOS). J. Appl. Phys. 2013, 114, 123511. [Google Scholar] [CrossRef]
- Marris-Morini, D.; Vakarin, V.; Ramirez, J.M.; Liu, Q.; Ballabio, A.; Frigerio, J.; Montesinos, M.; Alonso-Ramos, C.; Roux, X.L.; Serna, S.; et al. Germanium-based integrated photonics from near- to mid-infrared applications. Nanophotonics 2018, 7, 1781–1793. [Google Scholar] [CrossRef]
- Vakarin, V.; Ye, W.N.; Ramírez, J.M.; Liu, Q.; Frigerio, J.; Ballabio, A.; Isella, G.; Vivien, L.; Alonso-Ramos, C.; Cheben, P.; et al. Ultra-wideband Ge-rich silicon germanium mid-infrared polarization rotator with mode hybridization flattening. Opt. Express 2019, 27, 9838–9847. [Google Scholar] [CrossRef]
- Mheen, B.; Song, Y.-J.; Kang, J.-Y.; Hong, S. Strained-SiGe Complementary MOSFETs Adopting Different Thicknesses of Silicon Cap Layers for Low Power and High Performance Applications. ETRI J. 2005, 27, 439–445. [Google Scholar] [CrossRef]
- Sedky, S.; Witvrouw, A.; Baert, K. Poly SiGe, a promising material for MEMS monolithic integration with the driving electronics. Sens. Actuators A 2002, 97–98, 503–511. [Google Scholar] [CrossRef]
- Scappucci, G.; Kloeffel, C.; Zwanenburg, F.A.; Loss, D.; Myronov, M.; Zhang, J.-J.; De Franceschi, S.; Katsaros, G.; Veldhorst, M. The germanium quantum information route. Nat. Rev. Mater. 2021, 6, 926–943. [Google Scholar] [CrossRef]
- Cecchi, S.; Gatti, E.; Chrastina, D.; Frigerio, J.; Gubler, E.M.; Paul, D.J.; Guzzi, M.; Isella, G. Thin SiGe virtual substrates for Ge heterostructures integration on silicon. J. Appl. Phys. 2014, 115, 093502. [Google Scholar] [CrossRef]
- Li, Y.S.; Sookchoo, P.; Cui, X.; Mohr, R.; Savage, D.E.; Foote, R.H.; Jacobson, R.B.; Sánchez-Pérez, J.R.; Paskiewicz, D.M.; Wu, X.; et al. Electronic Transport Properties of Epitaxial Si/SiGe Heterostructures Grown on Single-Crystal SiGe Nanomembranes. ACS Nano 2015, 9, 4891–4899. [Google Scholar] [CrossRef] [PubMed]
- Pillarisetty, R.; Chu-Kung, B.; Corcoran, S.; Dewey, G.; Kavalieros, J.; Kennel, H.; Kotlyar, R.; Le, V.; Lionberger, D.; Metz, M.; et al. High mobility strained germanium quantum well field effect transistor as the p-channel device option for low power (Vcc = 0.5 V) III–V CMOS architecture. In Proceedings of the 2010 International Electron Devices Meeting, San Francisco, CA, USA, 6–8 December 2010; pp. 6.7.1–6.7.4. [Google Scholar]
- Terashima, K.; Tajima, M.; Tatsumi, T. Near-band-gap photoluminescence of Si1−xGex alloys grown on Si(100) by molecular beam epitaxy. Appl. Phys. Lett. 1990, 57, 1925–1927. [Google Scholar] [CrossRef]
- Sturm, J.C.; Manoharan, H.; Lenchyshyn, L.C.; Thewalt, M.L.W.; Rowell, N.L.; Noël, J.P.; Houghton, D.C. Well-resolved band-edge photoluminescence of excitons confined in strained Si1−xGex quantum wells. Phys. Rev. Lett. 1991, 66, 1362–1365. [Google Scholar] [CrossRef]
- Ashburn, P.; Bagnall, D. Silicon–Germanium: Properties, Growth and Applications. In Springer Handbook of Electronic and Photonic Materials; Kasap, S., Capper, P., Eds.; Springer: Boston, MA, USA, 2007; pp. 481–498. [Google Scholar]
- Oehme, M.; Werner, J.; Kirfel, O.; Kasper, E. MBE growth of SiGe with high Ge content for optical applications. Appl. Surf. Sci. 2008, 254, 6238–6241. [Google Scholar] [CrossRef]
- Kuan, T.S.; Iyer, S.S. Strain relaxation and ordering in SiGe layers grown on (100), (111), and (110) Si surfaces by molecular-beam epitaxy. Appl. Phys. Lett. 1991, 59, 2242–2244. [Google Scholar] [CrossRef]
- Alam, M.M.; Wagatsuma, Y.; Okada, K.; Hoshi, Y.; Yamada, M.; Hamaya, K.; Sawano, K. Critical thickness of strained Si1−xGex on Ge(111) and Ge-on-Si(111). Appl. Phys. Express 2019, 12, 081005. [Google Scholar] [CrossRef]
- Maydell, K.V.; Grunewald, K.; Kellermann, M.; Sergeev, O.; Klement, P.; Reininghaus, N.; Kilper, T. Microcrystalline SiGe Absorber Layers in Thin-film Silicon Solar Cells. Energy Procedia 2014, 44, 209–215. [Google Scholar] [CrossRef]
- Capellini, G.; Seta, M.D.; Busby, Y.; Pea, M.; Evangelisti, F.; Nicotra, G.; Spinella, C.; Nardone, M.; Ferrari, C. Strain relaxation in high Ge content SiGe layers deposited on Si. J. Appl. Phys. 2010, 107, 063504. [Google Scholar] [CrossRef]
- Qi, C.; Goncher, G.; Solanki, R.; Jordan, J. SiGe nanowire growth and characterization. Nanotechnology 2007, 18, 075302. [Google Scholar] [CrossRef]
- Potié, A.; Baron, T.; Latu-Romain, L.; Rosaz, G.; Salem, B.; Montès, L.; Gentile, P.; Kreisel, J.; Roussel, H. Controlled growth of SiGe nanowires by addition of HCl in the gas phase. J. Appl. Phys. 2011, 110, 024311. [Google Scholar] [CrossRef]
- Lew, K.-K.; Pan, L.; Dickey, E.C.; Redwing, J.M. Vapor–Liquid–Solid Growth of Silicon–Germanium Nanowires. Adv. Mater. 2003, 15, 2073–2076. [Google Scholar] [CrossRef]
- Hsu, Y.-J.; Lu, S.-Y.; Lin, Y.-F. One-Step Preparation of Coaxial CdS–ZnS and Cd1−xZnxS–ZnS Nanowires. Adv. Funct. Mater. 2005, 15, 1350–1357. [Google Scholar] [CrossRef]
- Akhtar, J.; Akhtar, M.; Malik, M.A.; O’Brien, P.; Raftery, J. A Single-Source Precursor Route to Unusual PbSe Nanostructures by a Solution–Liquid–Solid Method. J. Am. Chem. Soc. 2012, 134, 2485–2487. [Google Scholar] [CrossRef] [PubMed]
- Mathur, S.; Barth, S. Molecule-Based Chemical Vapor Growth of Aligned SnO2 Nanowires and Branched SnO2/V2O5 Heterostructures. Small 2007, 3, 2070–2075. [Google Scholar] [CrossRef]
- Sardar, K.; Dan, M.; Schwenzer, B.; Rao, C.N.R. A simple single-source precursor route to the nanostructures of AlN, GaN and InN. J. Mater. Chem. 2005, 15, 2175–2177. [Google Scholar] [CrossRef]
- Lobreyer, T.; Oberhammer, H.; Sundermeyer, W. Synthesis and Structure of Tetrasilylgermane, Ge(SiH3)4, and Other Silylgermanes. Angew. Chem. Int. Ed. Engl. 1993, 32, 586–587. [Google Scholar] [CrossRef]
- Hu, C.; Taraci, J.L.; Tolle, J.; Bauer, M.R.; Crozier, P.A.; Tsong, I.S.T.; Kouvetakis, J. Synthesis of Highly Coherent SiGe and Si4Ge Nanostructures by Molecular Beam Epitaxy of H3SiGeH3 and Ge(SiH3)4. Chem. Mater. 2003, 15, 3569–3572. [Google Scholar] [CrossRef]
- Lee, M.-S.; Bent, S.F. Bonding and Thermal Reactivity in Thin a-SiC:H Films Grown by Methylsilane CVD. J. Phys. Chem. B 1997, 101, 9195–9205. [Google Scholar] [CrossRef]
- Liu, C.W.; Sturm, J.C. Low temperature chemical vapor deposition growth of β-SiC on (100) Si using methylsilane and device characteristics. J. Appl. Phys. 1997, 82, 4558–4565. [Google Scholar] [CrossRef]
- Johnson, A.D.; Perrin, J.; Mucha, J.A.; Ibbotson, D.E. Kinetics of silicon carbide CVD: Surface decomposition of silacyclobutane and methylsilane. J. Phys. Chem. 1993, 97, 12937–12948. [Google Scholar] [CrossRef]
- Hewitt, S.B.; Tay, S.-P.; Tarr, N.G.; Boothroyd, A.R. Silicon carbide emitter diodes by LPCVD (low-pressure chemical vapour deposition) using di-tert-butylsilane. Can. J. Phys. 1992, 70, 946–948. [Google Scholar] [CrossRef]
- Poungoué Mbeunmi, A.B.; Arvinte, R.; Pelletier, H.; Jellite, M.; Arès, R.; Fafard, S.; Boucherif, A. Growth of Ge epilayers using iso-butylgermane (IBGe) and its memory effect in an III-V chemical beam epitaxy reactor. J. Cryst. Growth 2020, 547, 125807. [Google Scholar] [CrossRef]
- Jakomin, R.; Beaudoin, G.; Gogneau, N.; Lamare, B.; Largeau, L.; Mauguin, O.; Sagnes, I. p and n-type germanium layers grown using iso-butyl germane in a III-V metal-organic vapor phase epitaxy reactor. Thin Solid Films 2011, 519, 4186–4191. [Google Scholar] [CrossRef]
- Seifner, M.S.; Dijkstra, A.; Bernardi, J.; Steiger-Thirsfeld, A.; Sistani, M.; Lugstein, A.; Haverkort, J.E.M.; Barth, S. Epitaxial Ge0.81Sn0.19 Nanowires for Nanoscale Mid-Infrared Emitters. ACS Nano 2019, 13, 8047–8054. [Google Scholar] [CrossRef] [PubMed]
- Köstler, B.; Jungwirth, F.; Achenbach, L.; Sistani, M.; Bolte, M.; Lerner, H.-W.; Albert, P.; Wagner, M.; Barth, S. Mixed-Substituted Single-Source Precursors for Si1−xGex Thin Film Deposition. Inorg. Chem. 2022, 61, 17248–17255. [Google Scholar] [CrossRef] [PubMed]
- Mathur, S.; Barth, S.; Shen, H. Chemical Vapor Growth of NiGa2O4 Films: Advantages and Limitations of a Single Molecular Source. Chem. Vap. Depos. 2005, 11, 11–16. [Google Scholar] [CrossRef]
- Barth, S.; Seifner, M.S.; Bernardi, J. Growth of monocrystalline In2O3 nanowires by a seed orientation dependent vapour–solid–solid mechanism. J. Mater. Chem. C 2014, 2, 5747–5751. [Google Scholar] [CrossRef]
- Seifner, M.S.; Sistani, M.; Porrati, F.; Di Prima, G.; Pertl, P.; Huth, M.; Lugstein, A.; Barth, S. Direct Synthesis of Hyperdoped Germanium Nanowires. ACS Nano 2018, 12, 1236–1241. [Google Scholar] [CrossRef]
- Wagner, R.S.; Ellis, W.C. Vapor-Liquid-Solid Mechanism of Single Crystal Growth. Appl. Phys. Lett. 1964, 4, 89–90. [Google Scholar] [CrossRef]
- Gamalski, A.D.; Ducati, C.; Hofmann, S. Cyclic Supersaturation and Triple Phase Boundary Dynamics in Germanium Nanowire Growth. J. Phys. Chem. C 2011, 115, 4413–4417. [Google Scholar] [CrossRef]
- Maliakkal, C.B.; Mårtensson, E.K.; Tornberg, M.U.; Jacobsson, D.; Persson, A.R.; Johansson, J.; Wallenberg, L.R.; Dick, K.A. Independent Control of Nucleation and Layer Growth in Nanowires. ACS Nano 2020, 14, 3868–3875. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, V.; Senz, S.; Gösele, U. Diameter-Dependent Growth Direction of Epitaxial Silicon Nanowires. Nano Lett. 2005, 5, 931–935. [Google Scholar] [CrossRef]
- Hessel, C.M.; Heitsch, A.T.; Korgel, B.A. Gold Seed Removal from the Tips of Silicon Nanorods. Nano Lett. 2010, 10, 176–180. [Google Scholar] [CrossRef]
- Puglisi, R.A.; Bongiorno, C.; Borgh, G.; Fazio, E.; Garozzo, C.; Mannino, G.; Neri, F.; Pellegrino, G.; Scalese, S.; La Magna, A. Study on the Physico-Chemical Properties of the Si Nanowires Surface. Nanomaterials 2019, 9, 818. [Google Scholar] [CrossRef] [PubMed]
- Benter, S.; Dubrovskii, V.G.; Bartmann, M.; Campo, A.; Zardo, I.; Sistani, M.; Stöger-Pollach, M.; Lancaster, S.; Detz, H.; Lugstein, A. Quasi One-Dimensional Metal–Semiconductor Heterostructures. Nano Lett. 2019, 19, 3892–3897. [Google Scholar] [CrossRef] [PubMed]
- Margalit, S.; Bar-Lev, A.; Kuper, A.B.; Aharoni, H.; Neugroschel, A. Oxidation of silicon-germanium alloys. J. Cryst. Growth 1972, 17, 288–297. [Google Scholar] [CrossRef]
- LeGoues, F.K.; Rosenberg, R.; Nguyen, T.; Himpsel, F.; Meyerson, B.S. Oxidation studies of SiGe. J. Appl. Phys. 1989, 65, 1724–1728. [Google Scholar] [CrossRef]
- Long, E.; Galeckas, A.; Kuznetsov, A.Y. Ge concentrations in pile-up layers of sub-100-nm SiGe films for nano-structuring by thermal oxidation. J. Vac. Sci. Technol. B 2012, 30, 041212. [Google Scholar] [CrossRef]
- Yu, P.Y.; Cardona, M. Fundamentals of Semiconductors; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Ren, S.-F.; Cheng, W.; Yu, P.Y. Microscopic investigation of phonon modes in SiGe alloy nanocrystals. Phys. Rev. B 2004, 69, 235327. [Google Scholar] [CrossRef]
- Lu, Q.; Adu, K.W.; Gutiérrez, H.R.; Chen, G.; Lew, K.-K.; Nimmatoori, P.; Zhang, X.; Dickey, E.C.; Redwing, J.M.; Eklund, P.C. Raman Scattering from Si1−xGex Alloy Nanowires. J. Phys. Chem. C 2008, 112, 3209–3215. [Google Scholar] [CrossRef]
- Sistani, M.; Staudinger, P.; Greil, J.; Holzbauer, M.; Detz, H.; Bertagnolli, E.; Lugstein, A. Room-Temperature Quantum Ballistic Transport in Monolithic Ultrascaled Al–Ge–Al Nanowire Heterostructures. Nano Lett. 2017, 17, 4556–4561. [Google Scholar] [CrossRef]
- Wind, L.; Sistani, M.; Böckle, R.; Smoliner, J.; Vukŭsić, L.; Aberl, J.; Brehm, M.; Schweizer, P.; Maeder, X.; Michler, J.; et al. Composition Dependent Electrical Transport in Si1−xGex Nanosheets with Monolithic Single-Elementary Al Contacts. Small 2022, 18, 2204178. [Google Scholar] [CrossRef]
- Park, S.J.; Jeon, D.-Y.; Sessi, V.; Trommer, J.; Heinzig, A.; Mikolajick, T.; Kim, G.-T.; Weber, W.M. Channel Length-Dependent Operation of Ambipolar Schottky-Barrier Transistors on a Single Si Nanowire. ACS Appl. Mater. Interfaces 2020, 12, 43927–43932. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Behrle, R.; Krause, V.; Seifner, M.S.; Köstler, B.; Dick, K.A.; Wagner, M.; Sistani, M.; Barth, S. Electrical and Structural Properties of Si1−xGex Nanowires Prepared from a Single-Source Precursor. Nanomaterials 2023, 13, 627. https://doi.org/10.3390/nano13040627
Behrle R, Krause V, Seifner MS, Köstler B, Dick KA, Wagner M, Sistani M, Barth S. Electrical and Structural Properties of Si1−xGex Nanowires Prepared from a Single-Source Precursor. Nanomaterials. 2023; 13(4):627. https://doi.org/10.3390/nano13040627
Chicago/Turabian StyleBehrle, Raphael, Vanessa Krause, Michael S. Seifner, Benedikt Köstler, Kimberly A. Dick, Matthias Wagner, Masiar Sistani, and Sven Barth. 2023. "Electrical and Structural Properties of Si1−xGex Nanowires Prepared from a Single-Source Precursor" Nanomaterials 13, no. 4: 627. https://doi.org/10.3390/nano13040627
APA StyleBehrle, R., Krause, V., Seifner, M. S., Köstler, B., Dick, K. A., Wagner, M., Sistani, M., & Barth, S. (2023). Electrical and Structural Properties of Si1−xGex Nanowires Prepared from a Single-Source Precursor. Nanomaterials, 13(4), 627. https://doi.org/10.3390/nano13040627





