Recent Strategies for High-Performing Indoor Perovskite Photovoltaics
Abstract
1. Introduction
2. Id-PPVs: Light Properties and Band Gap Optimization
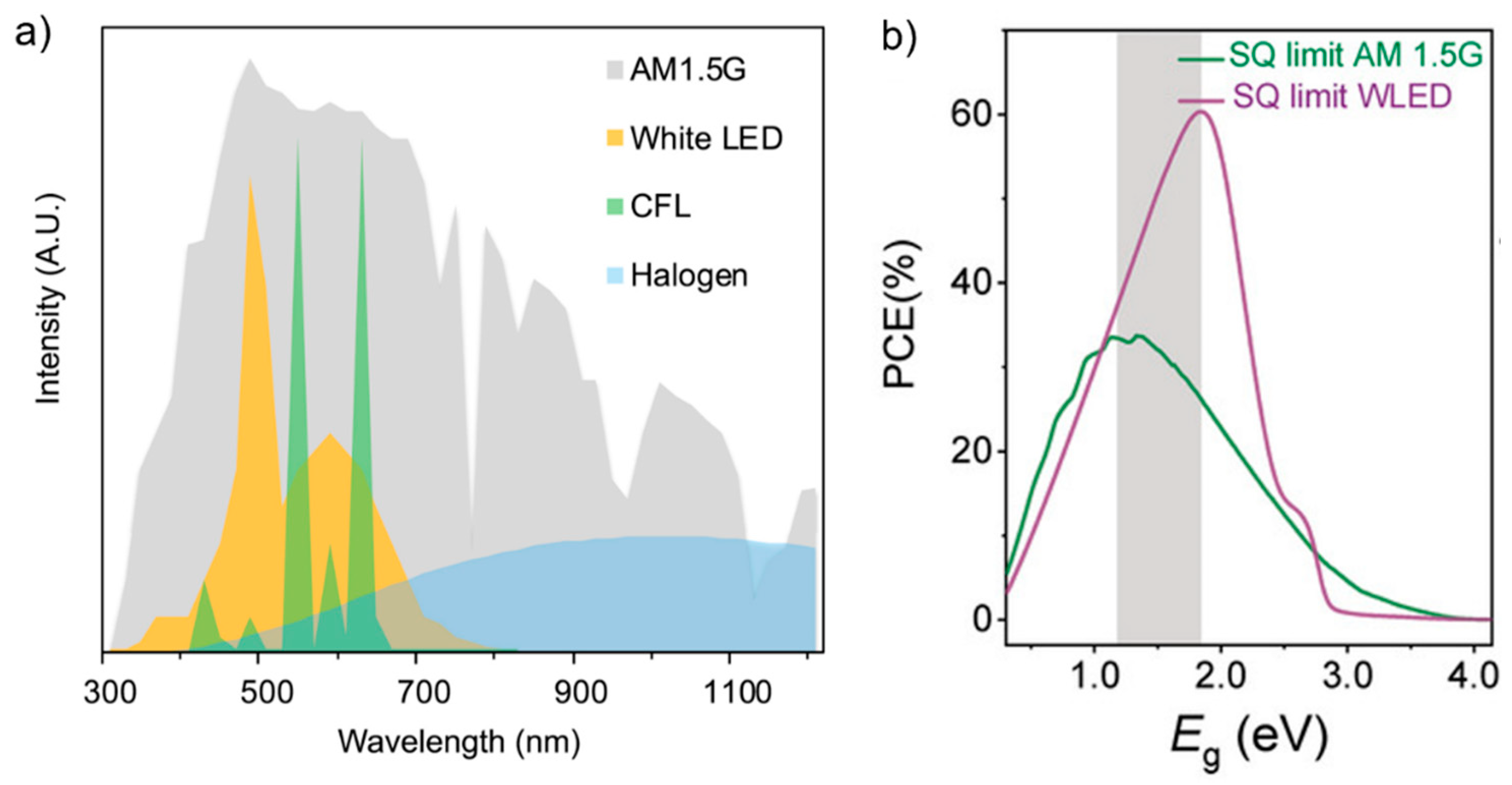
3. High Performance Id-PPVs
3.1. Perovskite Absorber Layer
3.1.1. Perovskite Bandgap Tuning
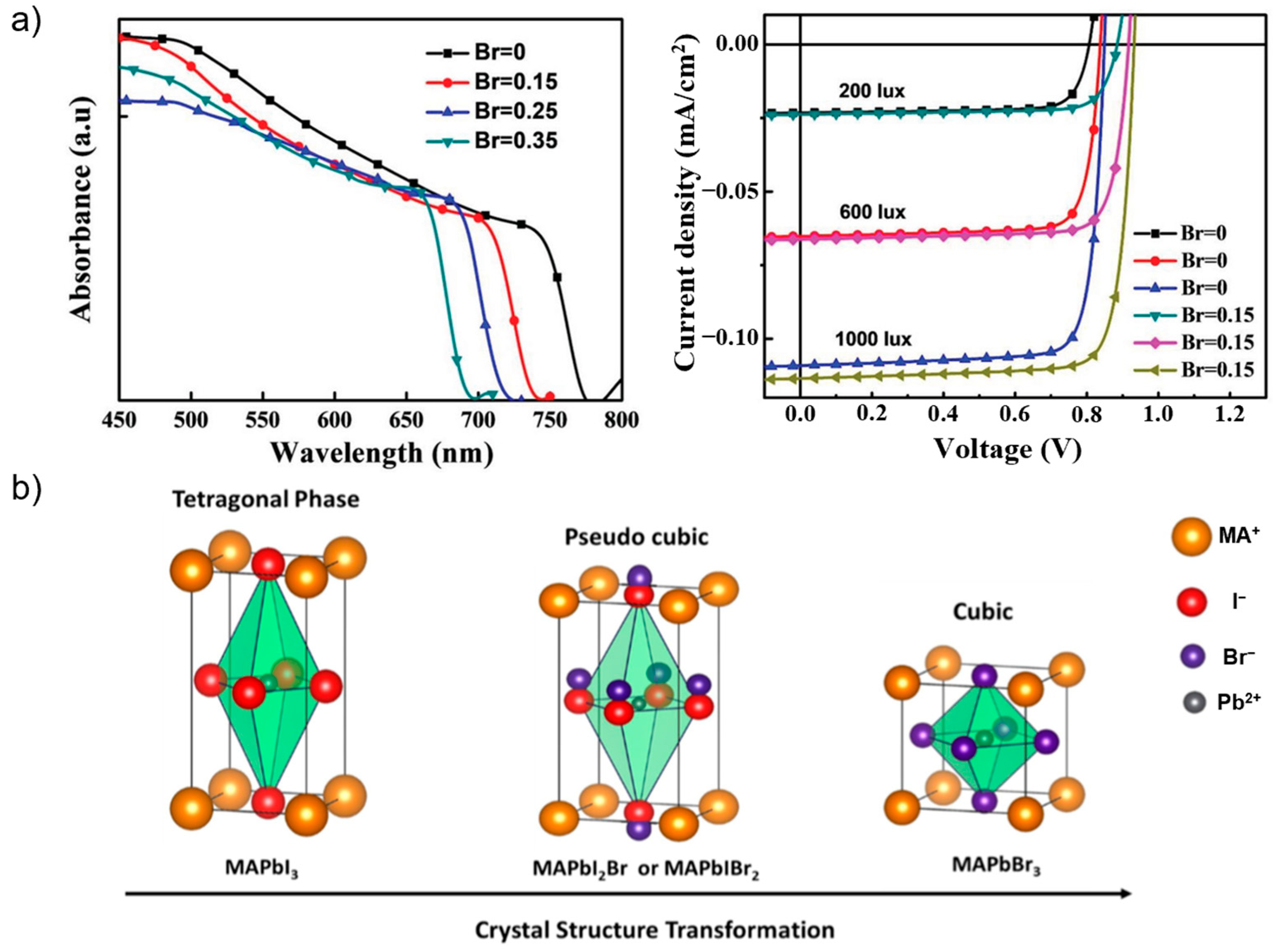
3.1.2. Perovskite Crystallization Kinetics
| Device | Perovskite Material | Key Technology | Bandgap (eV) | Intensity (lx) | Power Output (μWcm−2) | PCE (%) | Ref |
|---|---|---|---|---|---|---|---|
| n-i-p | FA0.90MA0.05Cs0.05PbI2.85Br0.15 | Optimized annealing method on flexible | 1.55 * | 1000 2 | - | 41.23 | [50] |
| n-i-p | Cs3Sb2ClxI9-x | Bionic perovskite passivation | 1.59 * | 1000 5 | - | 40.24 | [31] |
| n-i-p | (FAPbI3)0.97(MAPbBr3)0.03 | Bulk and surface defect passivation | 1.59 | 824.5 3 | - | 40.1 | [30] |
| p-i-n | MAPbI3 | Modified HTL | 1.50 # | 1000 3 | - | 39.2 | [37] |
| n-i-p | (MA0.91FA0.09)Pb(I0.94Br0.06) | Graft-type polymer HTL | 1.60 | 1000 5 | - | 38.2 | [51] |
| n-i-p | Cs0.05(FA0.85MA0.15)0.95 Pb(I0.85Br0.15)3 | Electron acceptor perovskite additives | - | 1000 5 | - | 37.9 | [52] |
| n-i-p | CsPbI2Br | Ionic liquid perovskite additives | 1.72 | 1000 2 | - | 37.24 | [53] |
| p-i-n | FA0.8Cs0.2Pb(I0.6Br0.4)3 | Bulk and surface defect passivation | 1.77 | 1000 4 | - | 37.18 | [32] |
| p-i-n | CH3NH3PbI2−xBrClx | Triple anion perovskite | 1.80 | 1000 5 | - | 36.2 | [44] |
| n-i-p | MAPbI3 | Heterojunction ETL | - | 1000 | 111.2 | 35.9 | [38] |
| p-i-n | Cs0.05(FA0.88MA0.12)0.95PbI3 | Alkali fluoride perovskite passivation | 1.55 | 1000 4 | 292 | 35.7 | [54] |
| n-i-p | (FA0.6MA0.4)0.9Cs0·1Pb(I0·6Br0.4)3 | PEA-halide additives | 1.75 | 1000 4 | 99.5 | 35.6 | [55] |
| p-i-n | MAPbI3 | Ionic liquid for ETL modification | 1.59 * | 1000 5 | - | 35.2 | [56] |
| n-i-p | MAPbI3 | Non-halide perovskite additives | 1.60 | 1000 5 | - | 34.86 | [49] |
| n-i-p | MAPbI3 | Bromine-doped perovskite | 1.68 * | 1000 1 | - | 34.5 | [41] |
| n-i-p | MAPbI3 | Blade coating | 1.59 * | 1000 4 | - | 33.8 | [57] |
| n-i-p | Cs0.05(FA0.6MA0.4)0.95Pb(I0.6Br0.4)3 | Perovskite passivation | 1.75 | 1000 2 | 89.26 | 33.42 | [35] |
| p-i-n | MAPbI3 | NiO-based HTL | - | 1000 2 | 98.4 | 32.88 | [58] |
| n-i-p | CsPbI2Br | Dopant-free polymer HTM | 1.91 | 1000 4 | 97.79 | 32.6 | [59] |
| n-i-p | FAPbI3 | Grain boundary stress release | 1.52 | 1062 5 | 106 | 31.85 | [60] |
| n-i-p | CsFAMA-based | Polymer-based perovskite passivation | 1.53 | 1000 4 | - | 30.73 | [61] |
| n-i-p | MAPbI3 | Doped SnO2 ETL | 1.60 # | 285 4 | - | 30.3 | [62] |
| p-i-n | MAPbI3 | Vacuum-based coating | - | 1000 5 | 94.9 | 30.1 | [63] |
3.2. Charge Transport Layer
3.2.1. Electron Transport Layer
3.2.2. HTL
3.3. Interface Engineering
3.4. Other Strategies
4. Towards Id-PPVs Commercialization
4.1. Human Friendly Id-PPVs
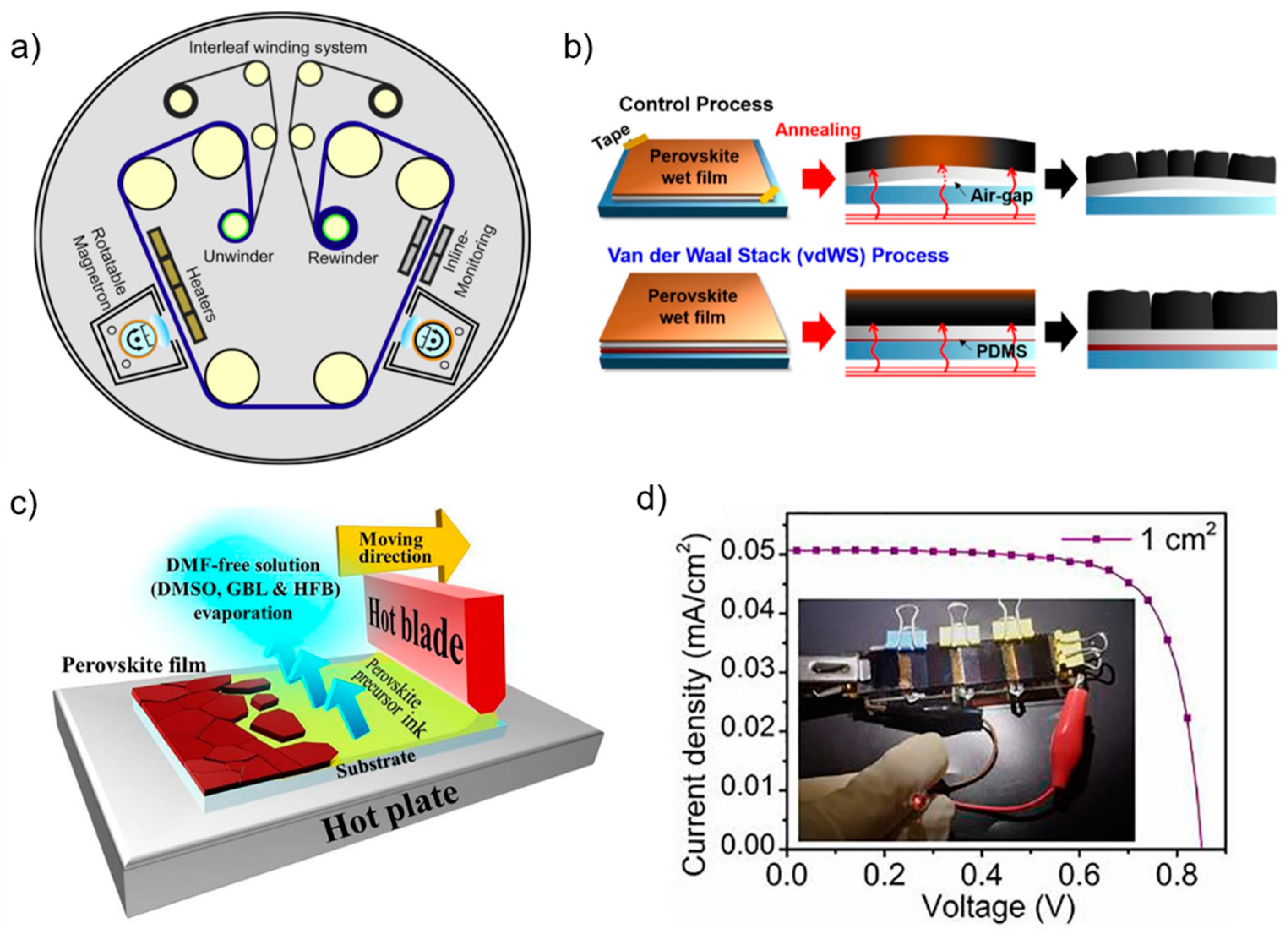
4.2. Flexible Id-PPVs

4.3. Large Area Id-PPVs
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Boveiri, H.R.; Khayami, R.; Elhoseny, M.; Gunasekaran, M. An efficient Swarm-Intelligence approach for task scheduling in cloud-based internet of things applications. J. Ambient. Intell. Humaniz. Comput. 2019, 10, 3469–3479. [Google Scholar] [CrossRef]
- Jabbar, W.A.; Kian, T.K.; Ramli, R.M.; Zubir, S.N.; Zamrizaman, N.S.M.; Balfaqih, M.; Shepelev, V.; Alharbi, S. Design and Fabrication of Smart Home With Internet of Things Enabled Automation System. IEEE Access 2019, 7, 144059–144074. [Google Scholar] [CrossRef]
- Yang, C.; Shen, W.; Wang, X. Applications of Internet of Things in manufacturing. In Proceedings of the 2016 IEEE 20th International Conference on Computer Supported Cooperative Work in Design (CSCWD), Nanchang, China, 4–6 May 2016; pp. 670–675. [Google Scholar]
- Elahi, H.; Munir, K.; Eugeni, M.; Atek, S.; Gaudenzi, P. Energy Harvesting towards Self-Powered IoT Devices. Energies 2020, 13, 5528. [Google Scholar] [CrossRef]
- Wang, Z.L.; Song, J. Piezoelectric nanogenerators based on zinc oxide nanowire arrays. Science 2006, 312, 242–246. [Google Scholar] [CrossRef]
- Choi, J.; Jung, I.; Kang, C.Y. A brief review of sound energy harvesting. Nano Energy 2019, 56, 169–183. [Google Scholar] [CrossRef]
- Wagih, M.; Weddell, A.S.; Beeby, S. Rectennas for Radio-Frequency Energy Harvesting and Wireless Power Transfer: A Review of Antenna Design [Antenna Applications Corner]. IEEE Antennas Propag. Mag. 2020, 62, 95–107. [Google Scholar] [CrossRef]
- Park, H.; Lee, D.; Park, G.; Park, S.; Khan, S.; Kim, J.; Kim, W. Energy harvesting using thermoelectricity for IoT (Internet of Things) and E-skin sensors. J. Phys. Energy 2019, 1, 042001. [Google Scholar] [CrossRef]
- Li, Y.; Grabham, N.J.; Beeby, S.P.; Tudor, M.J. The effect of the type of illumination on the energy harvesting performance of solar cells. Sol. Energy 2015, 111, 21–29. [Google Scholar] [CrossRef]
- Foti, M.; Tringali, C.; Battaglia, A.; Sparta, N.; Lombardo, S.; Gerardi, C. Efficient flexible thin film silicon module on plastics for indoor energy harvesting. Sol. Energy Mater. Sol. Cells 2014, 130, 490–494. [Google Scholar] [CrossRef]
- Cutting, C.L.; Bag, M.; Venkataraman, D. Indoor light recycling: A new home for organic photovoltaics. J. Mater. Chem. C 2016, 4, 10367–10370. [Google Scholar] [CrossRef]
- Cui, Y.; Wang, Y.; Bergqvist, J.; Yao, H.; Xu, Y.; Gao, B.; Yang, C.; Zhang, S.; Inganäs, O.; Gao, F.; et al. Wide-gap non-fullerene acceptor enabling high-performance organic photovoltaic cells for indoor applications. Nat. Energy 2019, 4, 768–775. [Google Scholar] [CrossRef]
- Bai, F.; Zhang, J.; Zeng, A.; Zhao, H.; Duan, K.; Yu, H.; Cheng, K.; Chai, G.; Chen, Y.; Liang, J.; et al. A highly crystalline non-fullerene acceptor enabling efficient indoor organic photovoltaics with high EQE and fill factor. Joule 2021, 5, 1231–1245. [Google Scholar] [CrossRef]
- Ma, R.; Zhou, K.; Sun, Y.; Liu, T.; Kan, Y.; Xiao, Y.; Dela Peña, T.A.; Li, Y.; Zou, X.; Xing, Z.; et al. Achieving high efficiency and well-kept ductility in ternary all-polymer organic photovoltaic blends thanks to two well miscible donors. Matter 2022, 5, 725–734. [Google Scholar] [CrossRef]
- Ma, R.; Yan, C.; Fong, P.W.-K.; Yu, J.; Liu, H.; Yin, J.; Huang, J.; Lu, X.; Yan, H.; Li, G. In situ and ex situ investigations on ternary strategy and co-solvent effects towards high-efficiency organic solar cells. Energy Environ. Sci. 2022, 15, 2479–2488. [Google Scholar] [CrossRef]
- Ma, R.; Yan, C.; Yu, J.; Liu, T.; Liu, H.; Li, Y.; Chen, J.; Luo, Z.; Tang, B.; Lu, X.; et al. High-Efficiency Ternary Organic Solar Cells with a Good Figure-of-Merit Enabled by Two Low-Cost Donor Polymers. ACS Energy Lett. 2022, 7, 2547–2556. [Google Scholar] [CrossRef]
- Haridas, R.; Velore, J.; Pradhan, S.C.; Vindhyasarumi, A.; Yoosaf, K.; Soman, S.; Unni, K.N.N.; Ajayaghosh, A. Indoor light-harvesting dye-sensitized solar cells surpassing 30% efficiency without co-sensitizers. Mater. Adv. 2021, 2, 7773–7787. [Google Scholar] [CrossRef]
- Venkatesan, S.; Lin, W.-H.; Hsu, T.-H.; Teng, H.; Lee, Y.-L. Indoor Dye-Sensitized Solar Cells with Efficiencies Surpassing 26% Using Polymeric Counter Electrodes. ACS Sustain. Chem. Eng. 2022, 10, 2473–2483. [Google Scholar] [CrossRef]
- Aftabuzzaman, M.; Sarker, S.; Lu, C.; Kim, H.K. In-depth understanding of the energy loss and efficiency limit of dye-sensitized solar cells under outdoor and indoor conditions. J. Mater. Chem. A 2021, 9, 24830–24848. [Google Scholar] [CrossRef]
- Kim, H.-S.; Lee, C.-R.; Im, J.-H.; Lee, K.-B.; Moehl, T.; Marchioro, A.; Moon, S.-J.; Humphry-Baker, R.; Yum, J.-H.; Moser, J.E.; et al. Lead Iodide Perovskite Sensitized All-Solid-State Submicron Thin Film Mesoscopic Solar Cell with Efficiency Exceeding 9%. Sci. Rep. 2012, 2, 591. [Google Scholar] [CrossRef]
- Min, H.; Lee, D.Y.; Kim, J.; Kim, G.; Lee, K.S.; Kim, J.; Paik, M.J.; Kim, Y.K.; Kim, K.S.; Kim, M.G.; et al. Perovskite solar cells with atomically coherent interlayers on SnO2 electrodes. Nature 2021, 598, 444–450. [Google Scholar] [CrossRef]
- Yoo, J.J.; Seo, G.; Chua, M.R.; Park, T.G.; Lu, Y.; Rotermund, F.; Kim, Y.K.; Moon, C.S.; Jeon, N.J.; Correa-Baena, J.P.; et al. Efficient perovskite solar cells via improved carrier management. Nature 2021, 590, 587–593. [Google Scholar] [CrossRef]
- Lee, G.; Kim, M.-C.; Choi, Y.W.; Ahn, N.; Jang, J.; Yoon, J.; Kim, S.M.; Lee, J.-G.; Kang, D.; Jung, H.S.; et al. Ultra-flexible perovskite solar cells with crumpling durability: Toward a wearable power source. Energy Environ. Sci. 2019, 12, 3182–3191. [Google Scholar] [CrossRef]
- Song, Z.N.; McElvany, C.L.; Phillips, A.B.; Celik, I.; Krantz, P.W.; Watthage, S.C.; Liyanage, G.K.; Apul, D.; Heben, M.J. A technoeconomic analysis of perovskite solar module manufacturing with low-cost materials and techniques. Energy Environ. Sci. 2017, 10, 1297–1305. [Google Scholar] [CrossRef]
- Fagiolari, L.; Bella, F. Carbon-based materials for stable, cheaper and large-scale processable perovskite solar cells. Energy Environ. Sci. 2019, 12, 3437–3472. [Google Scholar] [CrossRef]
- Benitez-Rodriguez, J.F.; Chen, D.; Gao, M.; Caruso, R.A. Roll-to-Roll Processes for the Fabrication of Perovskite Solar Cells under Ambient Conditions. Solar RRL 2021, 5, 2100341. [Google Scholar] [CrossRef]
- Krishna, B.G.; Sundar Ghosh, D.; Tiwari, S. Progress in ambient air-processed perovskite solar cells: Insights into processing techniques and stability assessment. Solar Energy 2021, 224, 1369–1395. [Google Scholar] [CrossRef]
- You, J.; Hong, Z.; Yang, Y.; Chen, Q.; Cai, M.; Song, T.-B.; Chen, C.-C.; Lu, S.; Liu, Y.; Zhou, H.; et al. Low-Temperature Solution-Processed Perovskite Solar Cells with High Efficiency and Flexibility. ACS Nano 2014, 8, 1674–1680. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Cheng, H.M.; Silva, S.R.P. Critical review of recent progress of flexible perovskite solar cells. Mater. Today 2020, 39, 66–88. [Google Scholar] [CrossRef]
- He, X.; Chen, J.; Ren, X.; Zhang, L.; Liu, Y.; Feng, J.; Fang, J.; Zhao, K.; Liu, S. 40.1% Record Low-Light Solar-Cell Efficiency by Holistic Trap-Passivation using Micrometer-Thick Perovskite Film. Adv. Mater. 2021, 33, 2100770. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Li, X.-M.; Ma, C.; Yang, W.-F.; Cao, J.-J.; Igbari, F.; Wang, Z.-K.; Liao, L.-S.; Dong, C.; Li, X.M.; et al. Lycopene-Based Bionic Membrane for Stable Perovskite Photovoltaics. Adv. Funct. Mater. 2021, 31, 2011242. [Google Scholar] [CrossRef]
- Li, Y.; Li, R.; Lin, Q. Engineering the Non-Radiative Recombination of Mixed-Halide Perovskites with Optimal Bandgap for Indoor Photovoltaics. Small 2022, 18, 2202028. [Google Scholar] [CrossRef] [PubMed]
- Freunek, M.; Freunek, M.; Reindl, L.M. Maximum efficiencies of indoor photovoltaic devices. IEEE J. Photovolt. 2013, 3, 59–64. [Google Scholar] [CrossRef]
- Leary, G.; Switzer, G.; Kuntz, G.; Kaiser, T. Comparison of xenon lamp-based and led-based solar simulators. In Proceedings of the 2016 IEEE 43rd Photovoltaic Specialists Conference (PVSC), Portland, OR, USA, 5–10 June 2016; pp. 3062–3067. [Google Scholar]
- Lee, M.; Choi, E.; Soufiani, A.M.; Lim, J.; Kim, M.; Chen, D.; Green, M.A.; Seidel, J.; Lim, S.; Kim, J.; et al. Enhanced Hole-Carrier Selectivity in Wide Bandgap Halide Perovskite Photovoltaic Devices for Indoor Internet of Things Applications. Adv. Funct. Mater. 2021, 31, 2008908. [Google Scholar] [CrossRef]
- Ann, M.H.; Kim, J.; Kim, M.; Alosaimi, G.; Kim, D.; Ha, N.Y.; Seidel, J.; Park, N.; Yun, J.S.; Kim, J.H. Device design rules and operation principles of high-power perovskite solar cells for indoor applications. Nano Energy 2020, 68, 104321. [Google Scholar] [CrossRef]
- Li, N.; Feng, A.; Guo, X.; Wu, J.; Xie, S.; Lin, Q.; Jiang, X.; Liu, Y.; Chen, Z.; Tao, X. Engineering the Hole Extraction Interface Enables Single-Crystal MAPbI3 Perovskite Solar Cells with Efficiency Exceeding 22% and Superior Indoor Response. Adv. Energy Mater. 2022, 12, 2103241. [Google Scholar] [CrossRef]
- Noh, Y.W.; Jin, I.S.; Kim, K.S.; Park, S.H.; Jung, J.W. Reduced energy loss in SnO2/ZnO bilayer electron transport layer-based perovskite solar cells for achieving high efficiencies in outdoor/indoor environments. J. Mater. Chem. A 2020, 8, 17163–17173. [Google Scholar] [CrossRef]
- Mathews, I.; Kantareddy, S.N.; Buonassisi, T.; Peters, I.M. Technology and Market Perspective for Indoor Photovoltaic Cells. Joule 2019, 3, 1415–1426. [Google Scholar] [CrossRef]
- Wu, M.J.; Kuo, C.C.; Jhuang, L.S.; Chen, P.H.; Lai, Y.F.; Chen, F.C. Bandgap Engineering Enhances the Performance of Mixed-Cation Perovskite Materials for Indoor Photovoltaic Applications. Adv. Energy Mater. 2019, 9, 1901863. [Google Scholar] [CrossRef]
- Lim, J.W.; Kwon, H.; Kim, S.H.; You, Y.J.; Goo, J.S.; Ko, D.H.; Lee, H.J.; Kim, D.; Chung, I.; Kim, T.G.; et al. Unprecedentedly high indoor performance (efficiency > 34 %) of perovskite photovoltaics with controlled bromine doping. Nano Energy 2020, 75, 104984. [Google Scholar] [CrossRef]
- Raifuku, I.; Ishikawa, Y.; Chiang, Y.H.; Lin, P.Y.; Li, M.H.; Uraoka, Y.; Chen, P. Segregation-free bromine-doped perovskite solar cells for IoT applications. RSC Adv. 2019, 9, 32833–32838. [Google Scholar] [CrossRef]
- Singh, R.; Parashar, M.; Sandhu, S.; Yoo, K.; Lee, J.J. The effects of crystal structure on the photovoltaic performance of perovskite solar cells under ambient indoor illumination. Sol. Energy 2021, 220, 43–50. [Google Scholar] [CrossRef]
- Cheng, R.; Chung, C.C.; Zhang, H.; Liu, F.; Wang, W.T.; Zhou, Z.; Wang, S.; Djurišić, A.B.; Feng, S.P. Tailoring Triple-Anion Perovskite Material for Indoor Light Harvesting with Restrained Halide Segregation and Record High Efficiency Beyond 36%. Adv. Energy Mater. 2019, 9, 1901980. [Google Scholar] [CrossRef]
- Kim, J.; Jang, J.H.; Choi, E.; Shin, S.J.; Kim, J.H.; Jeon, G.G.; Lee, M.; Seidel, J.; Kim, J.H.; Yun, J.S.; et al. Chlorine Incorporation in Perovskite Solar Cells for Indoor Light Applications. Cell Rep. Phys. Sci. 2020, 1, 100273. [Google Scholar] [CrossRef]
- Jin, I.S.; Kim, K.S.; Jung, J.W. CsCl-induced defect control of CsPbI2Br thin films for achieving open-circuit voltage of 1.33 V in all-inorganic perovskite solar cells. J. Power Sources 2021, 512, 230481. [Google Scholar] [CrossRef]
- Jin, I.S.; Parida, B.; Jung, J.W. Simultaneously enhanced efficiency and ambient stability of inorganic perovskite solar cells by employing tetramethylammonium chloride additive in CsPbI2Br. J. Mater. Sci. Technol. 2022, 102, 224–231. [Google Scholar] [CrossRef]
- Chen, J.; He, D.; Park, N.G. Methodologies for >30% Efficient Perovskite Solar Cells via Enhancement of Voltage and Fill Factor. Solar RRL 2021, 6, 2100767. [Google Scholar] [CrossRef]
- Dong, C.; Li, M.; Zhang, Y.; Wang, K.L.; Yuan, S.; Igbari, F.; Yang, Y.; Gao, X.; Wang, Z.K.; Liao, L.S. Lead Oxalate-Induced Nucleation Retardation for High-Performance Indoor and Outdoor Perovskite Photovoltaics. ACS Appl. Mater. Interfaces 2020, 12, 836–843. [Google Scholar] [CrossRef]
- Gong, O.Y.; Han, G.S.; Lee, S.; Seo, M.K.; Sohn, C.; Yoon, G.W.; Jang, J.; Lee, J.M.; Choi, J.H.; Lee, D.-K.; et al. Van der Waals Force-Assisted Heat-Transfer Engineering for Overcoming Limited Efficiency of Flexible Perovskite Solar Cells. ACS Energy Lett. 2022, 7, 2893–2903. [Google Scholar] [CrossRef]
- Opoku, H.; Kim, Y.H.; Lee, J.H.; Ahn, H.; Lee, J.J.; Baek, S.W.; Jo, J.W. A tailored graft-type polymer as a dopant-free hole transport material in indoor perovskite photovoltaics. J. Mater. Chem. A 2021, 9, 15294–15300. [Google Scholar] [CrossRef]
- Yang, F.; Su, Z.; Pascual, J.; Li, M.; Liu, H.; Qin, C.; Gao, X.; Li, G.; Li, Z.; Wang, Z. Enhancement of exciton separation in indoor perovskite photovoltaics by employing conjugated organic chromophores. J. Power Sources 2022, 520, 230785. [Google Scholar] [CrossRef]
- Du, Y.; Tian, Q.; Chang, X.; Fang, J.; Gu, X.; He, X.; Ren, X.; Zhao, K.; Liu, S. Ionic Liquid Treatment for Highest-Efficiency Ambient Printed Stable All-Inorganic CsPbI3 Perovskite Solar Cells. Adv. Mater. 2022, 34, 2106750. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xi, J.; Dong, H.; Ahn, N.; Zhu, Z.; Chen, J.; Li, P.; Zhu, X.; Dai, J.; Hu, Z.; et al. Impermeable inorganic “walls” sandwiching perovskite layer toward inverted and indoor photovoltaic devices. Nano Energy 2021, 88, 106286. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, J.; Wu, S.; Deng, X.; Li, F.; Liu, D.; Lee, C.C.C.; Lin, F.; Lei, D.; Chueh, C.C.; et al. Minimized surface deficiency on wide-bandgap perovskite for efficient indoor photovoltaics. Nano Energy 2020, 78, 105377. [Google Scholar] [CrossRef]
- Li, M.; Zhao, C.; Wang, Z.K.; Zhang, C.C.; Lee, H.K.H.; Pockett, A.; Barbé, J.; Tsoi, W.C.; Yang, Y.G.; Carnie, M.J.; et al. Interface Modification by Ionic Liquid: A Promising Candidate for Indoor Light Harvesting and Stability Improvement of Planar Perovskite Solar Cells. Adv. Energy Mater. 2018, 8, 1801509. [Google Scholar] [CrossRef]
- Bi, Z.; Xu, X.; Chen, X.; Zhu, Y.; Liu, C.; Yu, H.; Zheng, Y.; Troshin, P.A.; Guerrero, A.; Xu, G. High-performance large-area blade-coated perovskite solar cells with low ohmic loss for low lighting indoor applications. Chem. Eng. J. 2022, 446, 137164. [Google Scholar] [CrossRef]
- Saranin, D.; Komaricheva, T.; Luchnikov, L.; Muratov, D.S.; Le, T.S.; Karpov, Y.; Gostishchev, P.; Yurchuk, S.; Kuznetsov, D.; Didenko, S.; et al. Hysteresis-free perovskite solar cells with compact and nanoparticle NiO for indoor application. Sol. Energy Mater. Sol. Cells 2021, 227, 111095. [Google Scholar] [CrossRef]
- Guo, Z.; Jena, A.K.; Takei, I.; Ikegami, M.; Ishii, A.; Numata, Y.; Shibayama, N.; Miyasaka, T. Dopant-Free Polymer HTM-Based CsPbI2Br Solar Cells with Efficiency Over 17% in Sunlight and 34% in Indoor Light. Adv. Funct. Mater. 2021, 31, 4–6. [Google Scholar] [CrossRef]
- Chen, C.H.; Su, Z.H.; Lou, Y.H.; Yu, Y.J.; Wang, K.L.; Liu, G.L.; Shi, Y.R.; Chen, J.; Cao, J.J.; Zhang, L.; et al. Full-Dimensional Grain Boundary Stress Release for Flexible Perovskite Indoor Photovoltaics. Adv. Mater. 2022, 34, 2200320. [Google Scholar] [CrossRef]
- Zhu, X.; Dong, H.; Chen, J.; Xu, J.; Li, Z.; Yuan, F.; Dai, J.; Jiao, B.; Hou, X.; Xi, J.; et al. Photoinduced Cross Linkable Polymerization of Flexible Perovskite Solar Cells and Modules by Incorporating Benzyl Acrylate. Adv. Funct. Mater. 2022, 32, 2202408. [Google Scholar] [CrossRef]
- Bi, Z.; Zhang, S.; Thandapani, M.; Zhu, Y.; Zheng, Y.; Liem, N.Q.; Xiao, X.; Xu, G.; Guerrero, A.; Xu, X. High Shunt Resistance SnO2-PbO Electron Transport Layer for Perovskite Solar Cells Used in Low Lighting Applications. Adv. Sustain. Syst. 2021, 5, 2100120. [Google Scholar] [CrossRef]
- Chen, C.Y.; Lee, W.H.; Hsiao, S.Y.; Tsai, W.L.; Yang, L.; Lin, H.L.; Chou, H.J.; Lin, H.W. Vacuum-deposited perovskite photovoltaics for highly efficient environmental light energy harvesting. J. Mater. Chem. A 2019, 7, 3612–3617. [Google Scholar] [CrossRef]
- Yang, D.; Yang, R.; Ren, X.; Zhu, X.; Yang, Z.; Li, C.; Liu, S.F. Hysteresis-Suppressed High-Efficiency Flexible Perovskite Solar Cells Using Solid-State Ionic-Liquids for Effective Electron Transport. Adv. Mater. 2016, 28, 5206–5213. [Google Scholar] [CrossRef]
- Zhang, Y.; Fei, Z.; Gao, P.; Lee, Y.; Tirani, F.F.; Scopelliti, R.; Feng, Y.; Dyson, P.J.; Nazeeruddin, M.K. A Strategy to Produce High Efficiency, High Stability Perovskite Solar Cells Using Functionalized Ionic Liquid-Dopants. Adv. Mater. 2017, 29, 1702157. [Google Scholar] [CrossRef]
- Kim, H.; Lee, J.W.; Han, G.R.; Kim, S.K.; Oh, J.H. Synergistic Effects of Cation and Anion in an Ionic Imidazolium Tetrafluoroborate Additive for Improving the Efficiency and Stability of Half-Mixed Pb-Sn Perovskite Solar Cells. Adv. Funct. Mater. 2021, 31, 2008801. [Google Scholar] [CrossRef]
- Bai, S.; Da, P.; Li, C.; Wang, Z.; Yuan, Z.; Fu, F.; Kawecki, M.; Liu, X.; Sakai, N.; Wang, J.T.-W.; et al. Planar perovskite solar cells with long-term stability using ionic liquid additives. Nature 2019, 571, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Deng, X.; Wang, J.; Wang, S.; Niu, X.; Hao, F.; Ding, L. Ionic liquid reducing energy loss and stabilizing CsPbI2Br solar cells. Nano Energy 2021, 81, 105631. [Google Scholar] [CrossRef]
- Lee, H.K.H.; Barbé, J.; Meroni, S.M.P.; Du, T.; Lin, C.T.; Pockett, A.; Troughton, J.; Jain, S.M.; De Rossi, F.; Baker, J.; et al. Outstanding Indoor Performance of Perovskite Photovoltaic Cells—Effect of Device Architectures and Interlayers. Solar RRL 2019, 3, 1800207. [Google Scholar] [CrossRef]
- Lewis, A.; Troughton, J.R.; Smith, B.; McGettrick, J.; Dunlop, T.; De Rossi, F.; Pockett, A.; Spence, M.; Carnie, M.J.; Watson, T.M.; et al. In-depth analysis of defects in TiO2 compact electron transport layers and impact on performance and hysteresis of planar perovskite devices at low light. Sol. Energy Mater. Sol. Cells 2020, 209, 110448. [Google Scholar] [CrossRef]
- Sun, H.; Deng, K.; Jiang, Y.; Ni, J.; Xiong, J.; Li, L. Realizing Stable Artificial Photon Energy Harvesting Based on Perovskite Solar Cells for Diverse Applications. Small 2020, 16, 1906681. [Google Scholar] [CrossRef]
- Lee, K.M.; Lin, W.J.; Chen, S.H.; Wu, M.C. Control of TiO2 electron transport layer properties to enhance perovskite photovoltaics performance and stability. Org. Electron. 2020, 77, 105406. [Google Scholar] [CrossRef]
- Xiong, L.; Guo, Y.; Wen, J.; Liu, H.; Yang, G.; Qin, P.; Fang, G. Review on the Application of SnO2 in Perovskite Solar Cells. Adv. Funct. Mater. 2018, 28, 1802757. [Google Scholar] [CrossRef]
- Jagadamma, L.K.; Blaszczyk, O.; Sajjad, M.T.; Ruseckas, A.; Samuel, I.D.W. Efficient indoor p-i-n hybrid perovskite solar cells using low temperature solution processed NiO as hole extraction layers. Sol. Energy Mater. Sol. Cells 2019, 201, 110071. [Google Scholar] [CrossRef]
- Deng, D.; Guo, H.; Ji, B.; Wang, W.; Ma, L.; Luo, F. Size-selective catalysts in five functionalized porous coordination polymers with unsaturated zinc centers. New J. Chem. 2017, 41, 12611–12616. [Google Scholar] [CrossRef]
- Chang, X.-H.; Qin, W.-J.; Zhang, X.-Y.; Jin, X.; Yang, X.-G.; Dou, C.-X.; Ma, L.-F. Angle-Dependent Polarized Emission and Photoelectron Performance of Dye-Encapsulated Metal–Organic Framework. Inorg. Chem. 2021, 60, 10109–10113. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.H. Synthesis and structure of a zinc(II) coordination polymer assembled with 5-(3-carboxybenzyloxy)isophthalic acid and 1,2-bis(4-pyridyl)ethane. Z. Für Nat.-Sect. B J. Chem. Sci. 2022, 77, 561–564. [Google Scholar] [CrossRef]
- Li, J.X.; Zhang, Y.H.; Du, Z.X.; Feng, X. One-pot solvothermal synthesis of mononuclear and oxalate-bridged binuclear nickel compounds: Structural analyses, conformation alteration and magnetic properties. Inorg. Chim. Acta 2022, 530, 120697. [Google Scholar] [CrossRef]
- Li, J.X.; Xia, Y.Q.; Cheng, L.M.; Feng, X. One-pot hydrothermal synthesis of a mononuclear cobalt(II) complex and an organic-inorganic supramolecular adduct: Structures, properties and hirshfeld surface analyses. J. Solid State Chem. 2022, 313, 123271. [Google Scholar] [CrossRef]
- Li, J.X.; Xiong, L.Y.; Fu, L.L.; Bo, W.B.; Du, Z.X.; Feng, X. Structural diversity of Mn(II) and Cu(II) complexes based on 2-carboxyphenoxyacetate linker: Syntheses, conformation comparison and magnetic properties. J. Solid State Chem. 2022, 305, 122636. [Google Scholar] [CrossRef]
- Pham, H.D.; Jain, S.M.; Li, M.; Wang, Z.K.; Manzhos, S.; Feron, K.; Pitchaimuthu, S.; Liu, Z.; Motta, N.; Durrant, J.R.; et al. All-Rounder Low-Cost Dopant-Free D-A-D Hole-Transporting Materials for Efficient Indoor and Outdoor Performance of Perovskite Solar Cells. Adv. Electron. Mater. 2020, 6, 1900884. [Google Scholar] [CrossRef]
- Prasad, J.; Shao, Z.; Machhi, H.K.; Sharma, D.S.; Patel, V.K.; Pang, S.; Cui, G.; Soni, S.S. ‘V’ Shape A–D–A-Type Designed Small Hole Conductors for Efficient Indoor and Outdoor Staging from Solid Dye-Sensitized Solar Cells and Perovskite Solar Cells. Solar RRL 2021, 5, 2100206. [Google Scholar] [CrossRef]
- Yang, W.F.; Cao, J.J.; Chen, J.; Wang, K.L.; Dong, C.; Wang, Z.K.; Liao, L.S. Nicotinamide-Modified PEDOT:PSS for High Performance Indoor and Outdoor Tin Perovskite Photovoltaics. Solar RRL 2021, 5, 2100713. [Google Scholar] [CrossRef]
- Zhao, P.; Kim, B.J.; Jung, H.S. Passivation in perovskite solar cells: A review. Mater. Today Energy 2018, 7, 267–286. [Google Scholar] [CrossRef]
- Ke, W.; Xiao, C.; Wang, C.; Saparov, B.; Duan, H.-S.; Zhao, D.; Xiao, Z.; Schulz, P.; Harvey, S.P.; Liao, W.; et al. Employing Lead Thiocyanate Additive to Reduce the Hysteresis and Boost the Fill Factor of Planar Perovskite Solar Cells. Adv. Mater. 2016, 28, 5214–5221. [Google Scholar] [CrossRef]
- Chiang, Y.-H.; Li, M.-H.; Cheng, H.-M.; Shen, P.-S.; Chen, P. Mixed Cation Thiocyanate-Based Pseudohalide Perovskite Solar Cells with High Efficiency and Stability. ACS Appl. Mater. Interfaces 2017, 9, 2403–2409. [Google Scholar] [CrossRef]
- Pham, N.D.; Tiong, V.T.; Yao, D.; Martens, W.; Guerrero, A.; Bisquert, J.; Wang, H. Guanidinium thiocyanate selective Ostwald ripening induced large grain for high performance perovskite solar cells. Nano Energy 2017, 41, 476–487. [Google Scholar] [CrossRef]
- Ghosh, P.; Bruckbauer, J.; Trager-Cowan, C.; Krishnan Jagadamma, L. Crystalline grain engineered CsPbIBr2 films for indoor photovoltaics. Appl. Surf. Sci. 2022, 592, 152865. [Google Scholar] [CrossRef]
- Taylor, A.D.; Sun, Q.; Goetz, K.P.; An, Q.; Schramm, T.; Hofstetter, Y.; Litterst, M.; Paulus, F.; Vaynzof, Y. A general approach to high-efficiency perovskite solar cells by any antisolvent. Nat. Commun. 2021, 12, 1878. [Google Scholar] [CrossRef]
- Leijtens, T.; Prasanna, R.; Gold-Parker, A.; Toney, M.F.; McGehee, M.D. Mechanism of Tin Oxidation and Stabilization by Lead Substitution in Tin Halide Perovskites. ACS Energy Lett. 2017, 2, 2159–2165. [Google Scholar] [CrossRef]
- Yang, W.F.; Cao, J.J.; Dong, C.; Li, M.; Tian, Q.S.; Wang, Z.K.; Liao, L.S. Suppressed oxidation of tin perovskite by Catechin for eco-friendly indoor photovoltaics. Appl. Phys. Lett. 2021, 118, 023501. [Google Scholar] [CrossRef]
- Cao, J.J.; Lou, Y.H.; Yang, W.F.; Wang, K.L.; Su, Z.H.; Chen, J.; Chen, C.H.; Dong, C.; Gao, X.Y.; Wang, Z.K. Multifunctional potassium thiocyanate interlayer for eco-friendly tin perovskite indoor and outdoor photovoltaics. Chem. Eng. J. 2022, 433, 133832. [Google Scholar] [CrossRef]
- Peng, Y.; Huq, T.N.; Mei, J.; Portilla, L.; Jagt, R.A.; Occhipinti, L.G.; MacManus-Driscoll, J.L.; Hoye, R.L.Z.; Pecunia, V. Lead-Free Perovskite-Inspired Absorbers for Indoor Photovoltaics. Adv. Energy Mater. 2021, 11, 2002761. [Google Scholar] [CrossRef]
- Singh, A.; Lai, P.T.; Mohapatra, A.; Chen, C.Y.; Lin, H.W.; Lu, Y.J.; Chu, C.W. Panchromatic heterojunction solar cells for Pb-free all-inorganic antimony based perovskite. Chem. Eng. J. 2021, 419, 129424. [Google Scholar] [CrossRef]
- Arivazhagan, V.; Gun, F.; Reddy, R.K.K.; Li, T.; Adelt, M.; Robertson, N.; Chen, Y.; Ivaturi, A. Indoor light harvesting lead-free 2-aminothiazolium bismuth iodide solar cells. Sustain. Energy Fuels 2022, 6, 3179–3186. [Google Scholar] [CrossRef]
- Yin, J.; Cui, J.; Zhou, H.; Cui, S.; Wang, C.; Guo, J.; Wei, J.; Zhang, X. Encapsulation of UV Glue, Hydrophobicity of Binder and Carbon Electrode Enhance the Stability of Organic–Inorganic Hybrid Perovskite Solar Cells up to 5 Years. Energy Technol. 2020, 8, 2000513. [Google Scholar] [CrossRef]
- Fu, Z.; Xu, M.; Sheng, Y.; Yan, Z.; Meng, J.; Tong, C.; Li, D.; Wan, Z.; Ming, Y.; Mei, A.; et al. Encapsulation of Printable Mesoscopic Perovskite Solar Cells Enables High Temperature and Long-Term Outdoor Stability. Adv. Funct. Mater. 2019, 29, 1809129. [Google Scholar] [CrossRef]
- Jiang, Y.; Qiu, L.; Juarez-Perez, E.J.; Ono, L.K.; Hu, Z.; Liu, Z.; Wu, Z.; Meng, L.; Wang, Q.; Qi, Y. Reduction of lead leakage from damaged lead halide perovskite solar modules using self-healing polymer-based encapsulation. Nat. Energy 2019, 4, 585–593. [Google Scholar] [CrossRef]
- Crespo-Quesada, M.; Pazos-Outón, L.M.; Warnan, J.; Kuehnel, M.F.; Friend, R.H.; Reisner, E. Metal-encapsulated organolead halide perovskite photocathode for solar-driven hydrogen evolution in water. Nat. Commun. 2016, 7, 12555. [Google Scholar] [CrossRef]
- Martins, J.; Emami, S.; Madureira, R.; Mendes, J.; Ivanou, D.; Mendes, A. Novel laser-assisted glass frit encapsulation for long-lifetime perovskite solar cells. J. Mater. Chem. A 2020, 8, 20037–20046. [Google Scholar] [CrossRef]
- Ramos, F.J.; Maindron, T.; Béchu, S.; Rebai, A.; Frégnaux, M.; Bouttemy, M.; Rousset, J.; Schulz, P.; Schneider, N. Versatile perovskite solar cell encapsulation by low-temperature ALD-Al2O3 with long-term stability improvement. Sustain. Energy Fuels 2018, 2, 2468–2479. [Google Scholar] [CrossRef]
- Yi, A.; Chae, S.; Won, S.; Jung, H.J.; Cho, I.H.; Kim, J.H.; Kim, H.J. Roll-transferred graphene encapsulant for robust perovskite solar cells. Nano Energy 2020, 77, 105182. [Google Scholar] [CrossRef]
- Lv, Y.; Zhang, H.; Liu, R.; Sun, Y.; Huang, W. Composite Encapsulation Enabled Superior Comprehensive Stability of Perovskite Solar Cells. ACS Appl. Mater. Interfaces 2020, 12, 27277–27285. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-M.; Suko, H.; Kim, K.; Han, J.; Lee, S.; Matsuo, Y.; Maruyama, S.; Jeon, I.; Daiguji, H. Multi-Walled Carbon Nanotube-Assisted Encapsulation Approach for Stable Perovskite Solar Cells. Molecules 2021, 26, 5060. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Kang, T.W.; Kim, S.H.; Jung, E.H.; Park, H.H.; Seo, J.; Lee, S.J. Antireflective, self-cleaning and protective film by continuous sputtering of a plasma polymer on inorganic multilayer for perovskite solar cells application. Sol. Energy Mater. Sol. Cells 2019, 191, 55–61. [Google Scholar] [CrossRef]
- Zhao, J.; Brinkmann, K.O.; Hu, T.; Pourdavoud, N.; Becker, T.; Gahlmann, T.; Heiderhoff, R.; Polywka, A.; Görrn, P.; Chen, Y.; et al. Self-Encapsulating Thermostable and Air-Resilient Semitransparent Perovskite Solar Cells. Adv. Energy Mater. 2017, 7, 1602599. [Google Scholar] [CrossRef]
- Brinkmann, K.O.; Zhao, J.; Pourdavoud, N.; Becker, T.; Hu, T.; Olthof, S.; Meerholz, K.; Hoffmann, L.; Gahlmann, T.; Heiderhoff, R.; et al. Suppressed decomposition of organometal halide perovskites by impermeable electron-extraction layers in inverted solar cells. Nat. Commun. 2017, 8, 13938. [Google Scholar] [CrossRef]
- Lee, J.; Kim, G.W.; Kim, M.; Park, S.A.; Park, T. Nonaromatic Green-Solvent-Processable, Dopant-Free, and Lead-Capturable Hole Transport Polymers in Perovskite Solar Cells with High Efficiency. Adv. Energy Mater. 2020, 10, 1902662. [Google Scholar] [CrossRef]
- Chen, S.; Xiao, X.; Gu, H.; Huang, J. Iodine reduction for reproducible and high-performance perovskite solar cells and modules. Sci. Adv. 2022, 7, eabe8130. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, H.; Wu, X.; Zhao, D.; Zhang, S.; Zou, X.; Li, B.; Gao, D.; Li, Z.; Xia, X.; et al. Boosting the Fill Factor through Sequential Deposition and Homo Hydrocarbon Solvent toward Efficient and Stable All-Polymer Solar Cells. Adv. Energy Mater. 2022, 12, 2202729. [Google Scholar] [CrossRef]
- Schileo, G.; Grancini, G. Lead or no lead? Availability, toxicity, sustainability and environmental impact of lead-free perovskite solar cells. J. Mater. Chem. C 2021, 9, 67–76. [Google Scholar] [CrossRef]
- Miyasaka, T.; Kulkarni, A.; Kim, G.M.; Öz, S.; Jena, A.K. Perovskite Solar Cells: Can We Go Organic-Free, Lead-Free, and Dopant-Free? Adv. Energy Mater. 2020, 10, 1902500. [Google Scholar] [CrossRef]
- Park, S.Y.; Park, J.-S.; Kim, B.J.; Lee, H.; Walsh, A.; Zhu, K.; Kim, D.H.; Jung, H.S. Sustainable lead management in halide perovskite solar cells. Nat. Sustain. 2020, 3, 1044–1051. [Google Scholar] [CrossRef]
- Kim, B.J.; Kim, D.H.; Kwon, S.L.; Park, S.Y.; Li, Z.; Zhu, K.; Jung, H.S. Selective dissolution of halide perovskites as a step towards recycling solar cells. Nat. Commun. 2016, 7, 11735. [Google Scholar] [CrossRef]
- Zardetto, V.; Brown, T.M.; Reale, A.; Di Carlo, A. Substrates for flexible electronics: A practical investigation on the electrical, film flexibility, optical, temperature, and solvent resistance properties. J. Polym. Sci. Part B Polym. Phys. 2011, 49, 638–648. [Google Scholar] [CrossRef]
- Castro-Hermosa, S.; Lucarelli, G.; Top, M.; Fahland, M.; Fahlteich, J.; Brown, T.M. Perovskite Photovoltaics on Roll-To-Roll Coated Ultra-thin Glass as Flexible High-Efficiency Indoor Power Generators. Cell Rep. Phys. Sci. 2020, 1, 100045. [Google Scholar] [CrossRef]
- Mathews, I.; Kantareddy, S.N.R.; Sun, S.; Layurova, M.; Thapa, J.; Correa-Baena, J.P.; Bhattacharyya, R.; Buonassisi, T.; Sarma, S.; Peters, I.M. Self-Powered Sensors Enabled by Wide-Bandgap Perovskite Indoor Photovoltaic Cells. Adv. Funct. Mater. 2019, 29, 1904072. [Google Scholar] [CrossRef]
- Cheng, Y.; Peng, Y.; Jen, A.K.Y.; Yip, H.-L. Development and Challenges of Metal Halide Perovskite Solar Modules. Solar RRL 2022, 6, 2100545. [Google Scholar] [CrossRef]
- Park, J.; Bak, Y.E.; Lee, L.; Choi, H.; Khan, M.A.; Sung, M.M. Interdigitated Hierarchical Integration of an Efficient Lateral Perovskite Single-Crystal Solar Cell. ChemSusChem 2020, 13, 1882–1889. [Google Scholar] [CrossRef]
- Wojciechowski, K.; Forgács, D. Commercial Applications of Indoor Photovoltaics Based on Flexible Perovskite Solar Cells. ACS Energy Lett. 2022, 7, 3729–3733. [Google Scholar] [CrossRef]
- Kaltenbrunner, M.; Adam, G.; Głowacki, E.D.; Drack, M.; Schwödiauer, R.; Leonat, L.; Apaydin, D.H.; Groiss, H.; Scharber, M.C.; White, M.S.; et al. Flexible high power-per-weight perovskite solar cells with chromium oxide–metal contacts for improved stability in air. Nat. Mater. 2015, 14, 1032–1039. [Google Scholar] [CrossRef]

| Substrate | Key Technology | Lamp Type | Area (cm2) | Power Output (μWcm−2) | PCE (%) | Ref |
|---|---|---|---|---|---|---|
| FTO | Ionic liquid for ETL modification | FL | 9 | - | 35.2 | [56] |
| ITO | Non-halide perovskite additives | FL | 9 | - | 34.86 | [49] |
| ITO | NiO-based HTL | CWLED | 1 | 82.12 | 27.43 | [58] |
| ITO | Alkali fluoride perovskite passivation | WLED | 9 | - | 35.7 | [54] |
| FTO | Blade coating | WLED | 1 | - | 33.8 | [57] |
| PET/ITO | Polymer-based perovskite passivation | WLED | 24 | - | 30.73 | [61] |
| ITO | Halide for bandgap engineering | FL and LED | 1 | - | 17.89 | [40] |
| ITO | Vacuum-based coating | FL | 5.68 | 77.6 | 24.9 | [63] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mularso, K.T.; Jeong, J.-Y.; Han, G.S.; Jung, H.S. Recent Strategies for High-Performing Indoor Perovskite Photovoltaics. Nanomaterials 2023, 13, 259. https://doi.org/10.3390/nano13020259
Mularso KT, Jeong J-Y, Han GS, Jung HS. Recent Strategies for High-Performing Indoor Perovskite Photovoltaics. Nanomaterials. 2023; 13(2):259. https://doi.org/10.3390/nano13020259
Chicago/Turabian StyleMularso, Kelvian T., Ji-Young Jeong, Gill Sang Han, and Hyun Suk Jung. 2023. "Recent Strategies for High-Performing Indoor Perovskite Photovoltaics" Nanomaterials 13, no. 2: 259. https://doi.org/10.3390/nano13020259
APA StyleMularso, K. T., Jeong, J.-Y., Han, G. S., & Jung, H. S. (2023). Recent Strategies for High-Performing Indoor Perovskite Photovoltaics. Nanomaterials, 13(2), 259. https://doi.org/10.3390/nano13020259









