Hydrothermal Transformation of Eggshell Calcium Carbonate into Apatite Micro-Nanoparticles: Cytocompatibility and Osteoinductive Properties
Abstract
1. Introduction
2. Experimental Section
2.1. Materials and Methods
2.2. Physicochemical Characterization
2.3. Biological Tests
2.3.1. Cell Cultures
2.3.2. Cytocompatibility
2.3.3. Osteogenic Differentiation of m17.ASC
2.3.4. Alkaline Phosphatase (ALP) Staining and Quantitative Analysis
2.3.5. Alizarin Red Staining (ARS) and Quantitative Analysis
2.3.6. Statistical Analysis
3. Results
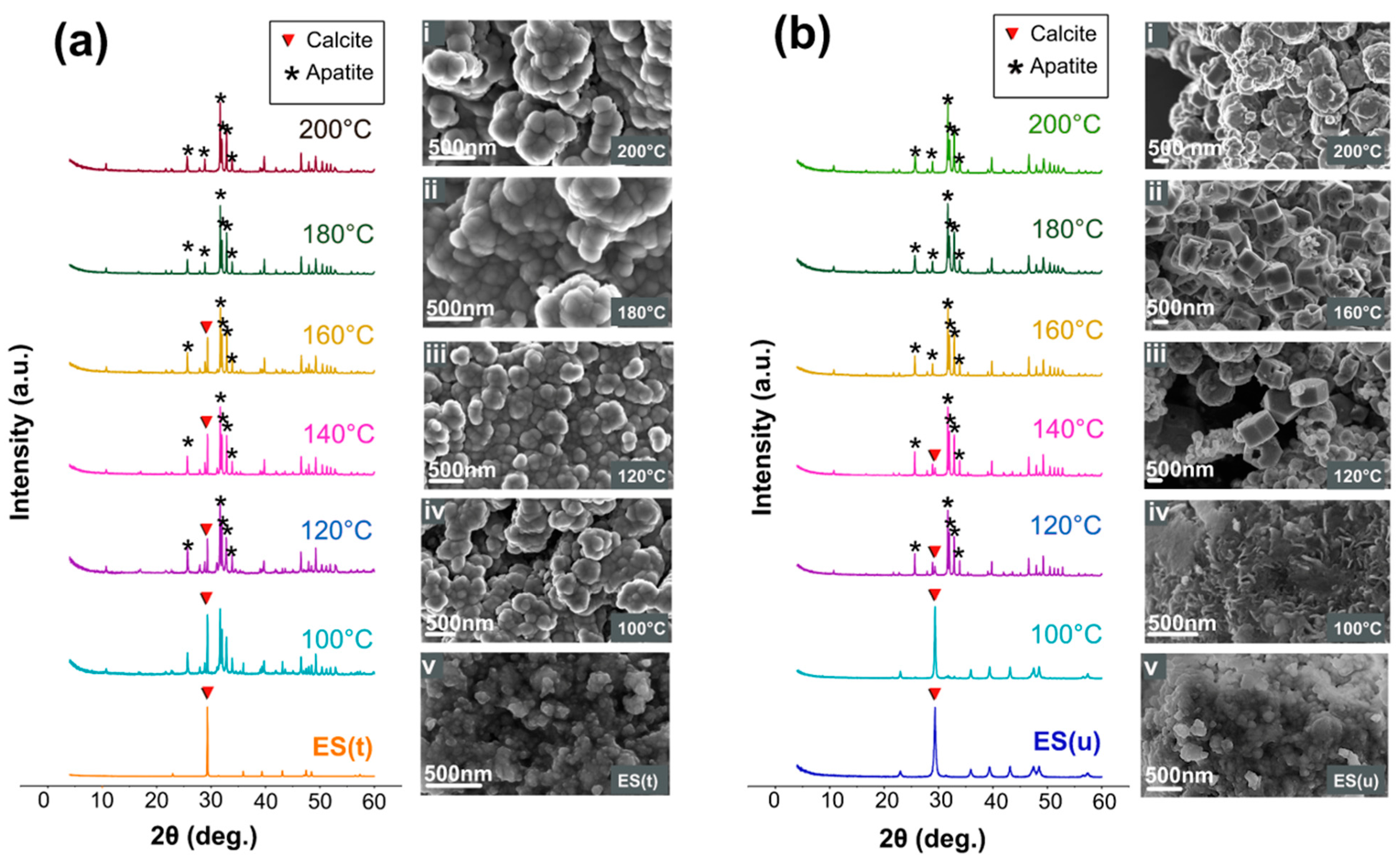
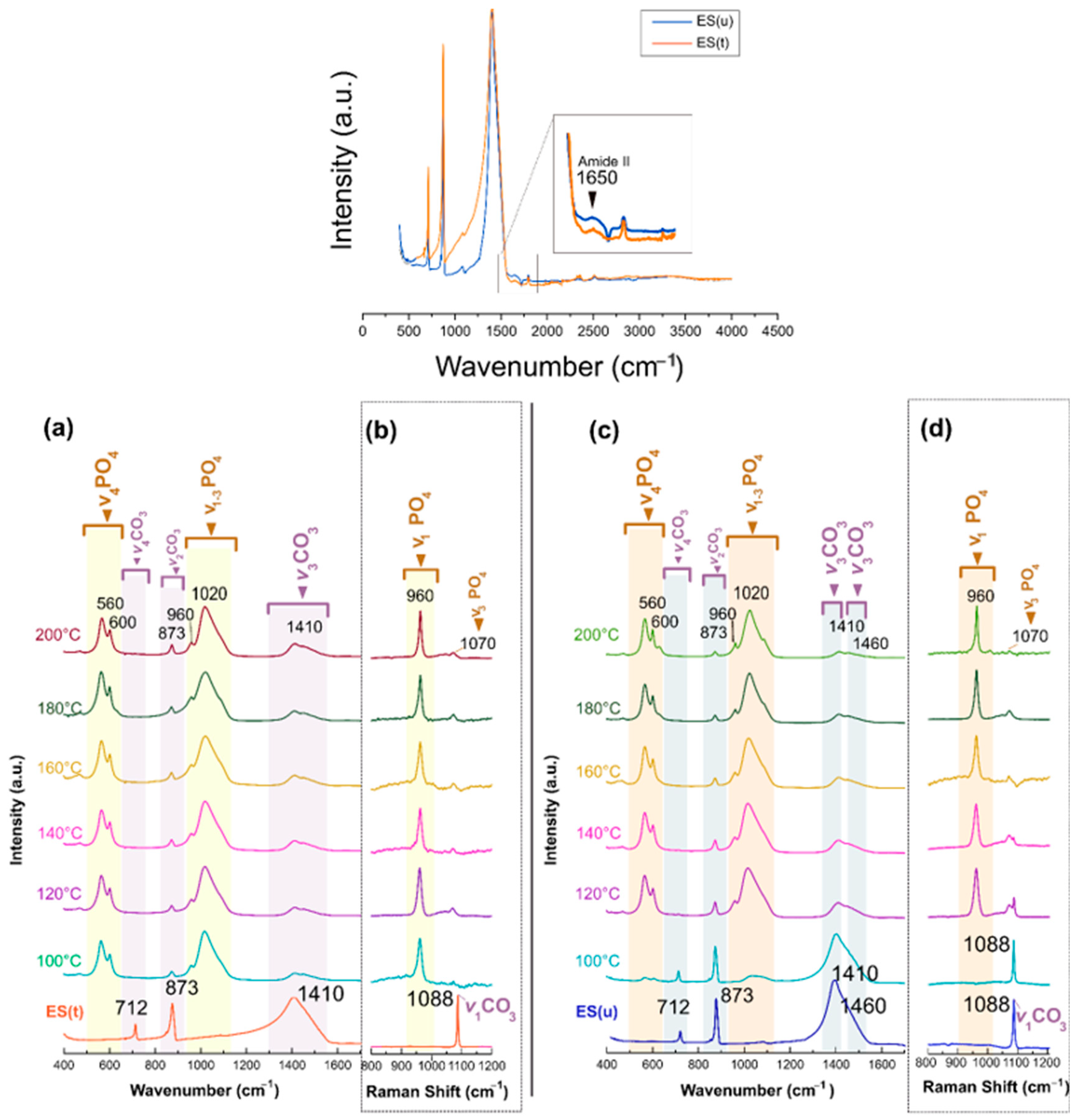
3.1. Crystallographic, Morphological, Compositional, and Spectroscopic Features
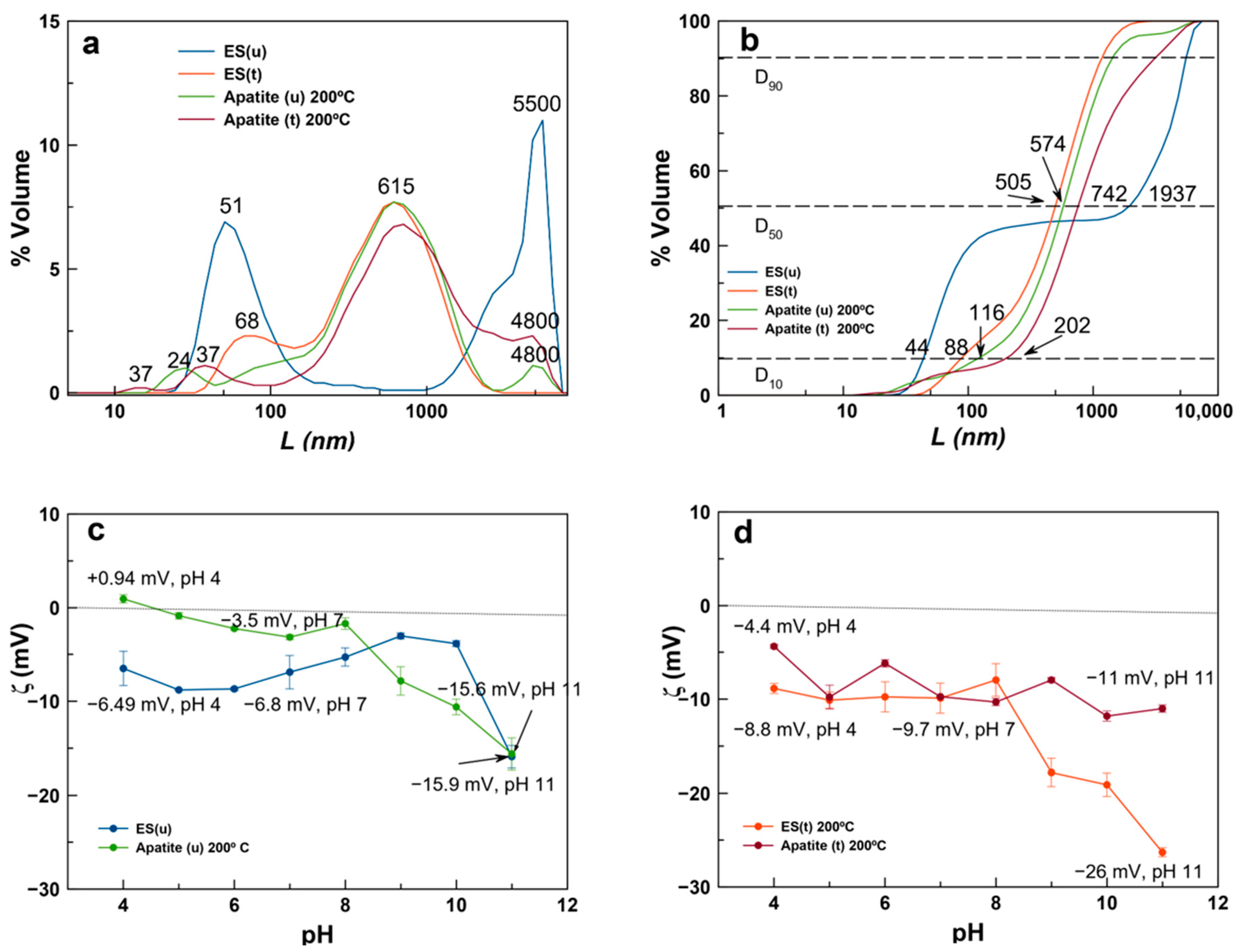
3.2. Crystal Size Distribution and ζ-Potential Versus pH of ES and Derived Ap Particles
3.3. Biological Tests
3.3.1. Cytocompatibility
3.3.2. Effect of Eggshell and Apatite Particles on the Osteogenesis and Mineralization of m17.ASC cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nys, Y.; Gautron, J.; Garcia-Ruiz, J.M.; Hincke, M.T. Avian Eggshell Mineralization; Biochemical and Functional Characterization of Matrix Proteins. Comptes Rendus Palevol 2004, 3, 549–562. [Google Scholar] [CrossRef]
- Hincke, M.; Gautron, J.; Rodriguez-Navarro, A.B.; McKee, M.D. The Eggshell: Structure and Protective Function. In Improving the Safety and Quality of Eggs and Egg Products: Egg Chemistry, Production and Consumption; Elsevier: Amsterdam, The Netherlands, 2011; pp. 151–182. ISBN 978-1-84569-754-9. [Google Scholar]
- Mann, K.; Macek, B.; Olsen, J.V. Proteomic Analysis of the Acid-Soluble Organic Matrix of the Chicken Calcified Eggshell Layer. Proteomics 2006, 6, 3801–3810. [Google Scholar] [CrossRef]
- Hernández-Hernández, A.; Vidal, M.L.; Gómez-Morales, J.; Rodríguez-Navarro, A.B.; Labas, V.; Gautron, J.; Nys, Y.; García Ruiz, J.M. Influence of Eggshell Matrix Proteins on the Precipitation of Calcium Carbonate (CaCO3). J. Cryst. Growth 2008, 310, 1754–1759. [Google Scholar] [CrossRef]
- Hernández-Hernández, A.; Gómez-Morales, J.; Rodríguez-Navarro, A.B.; Gautron, J.; Nys, Y.; García-Ruiz, J.M. Identification of Some Active Proteins in the Process of Hen Eggshell Formation. Cryst. Growth Des. 2008, 8, 4330–4339. [Google Scholar] [CrossRef]
- Cordeiro, C.M.M.; Hincke, M.T. Recent Patents on Eggshell: Shell and Membrane Applications. Recent Pat. Food Nutr. Agric. 2011, 3, 1–8. [Google Scholar] [CrossRef]
- Torres-Mansilla, A.; Hincke, M.; Voltes, A.; López-Ruiz, E.; Baldión, P.A.; Marchal, J.A.; Álvarez-Lloret, P.; Gómez-Morales, J. Eggshell Membrane as a Biomaterial for Bone Regeneration. Polymers 2023, 15, 1342. [Google Scholar] [CrossRef]
- Waheed, M.; Butt, M.S.; Shehzad, A.; Adzahan, N.M.; Shabbir, M.A.; Rasul Suleria, H.A.; Aadil, R.M. Eggshell Calcium: A Cheap Alternative to Expensive Supplements. Trends Food Sci. Technol. 2019, 91, 219–230. [Google Scholar] [CrossRef]
- Tsai, W.-T.; Hsien, K.-J.; Hsu, H.-C.; Lin, C.-M.; Lin, K.-Y.; Chiu, C.-H. Utilization of Ground Eggshell Waste as an Adsorbent for the Removal of Dyes from Aqueous Solution. Bioresour. Technol. 2008, 99, 1623–1629. [Google Scholar] [CrossRef]
- Baláž, M.; Ficeriová, J.; Briančin, J. Influence of Milling on the Adsorption Ability of Eggshell Waste. Chemosphere 2016, 146, 458–471. [Google Scholar] [CrossRef]
- Dawodu, F.A.; Ayodele, O.; Xin, J.; Zhang, S.; Yan, D. Effective Conversion of Non-Edible Oil with High Free Fatty Acid into Biodiesel by Sulphonated Carbon Catalyst. Appl. Energy 2014, 114, 819–826. [Google Scholar] [CrossRef]
- Anugrah, R.D.; Safahi, L. The Effect of Eggshell Organic Fertilizer on Vegetative Growth of Cayenne Pepper (Capsicum frutescens L). IOP Conf. Ser. Earth Environ. Sci. 2021, 755, 012001. [Google Scholar] [CrossRef]
- Balázsi, K.; Sim, H.-Y.; Choi, J.-Y.; Kim, S.-G.; Chae, C.-H.; Balázsi, C. Biogenic Nanosized Hydroxyapatite for Tissue Engineering Applications. In Proceedings of the International Symposium on Biomedical Engineering and Medical Physics, Riga, Latvia, 10–12 October 2012; Dekhtyar, Y., Katashev, A., Lancere, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 190–193. [Google Scholar]
- Gómez-Morales, J.; Iafisco, M.; Delgado-López, J.M.; Sarda, S.; Drouet, C. Progress on the Preparation of Nanocrystalline Apatites and Surface Characterization: Overview of Fundamental and Applied Aspects. Prog. Cryst. Growth Charact. Mater. 2013, 59, 1–46. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Nanosized and Nanocrystalline Calcium Orthophosphates. Acta Biomater. 2010, 6, 715–734. [Google Scholar] [CrossRef]
- Rajula, M.P.B.; Narayanan, V.; Venkatasubbu, G.D.; Mani, R.C.; Sujana, A. Nano-Hydroxyapatite: A Driving Force for Bone Tissue Engineering. J. Pharm. Bioallied Sci. 2021, 13, S11–S14. [Google Scholar] [CrossRef]
- Lara-Ochoa, S.; Ortega-Lara, W.; Guerrero-Beltrán, C.E. Hydroxyapatite Nanoparticles in Drug Delivery: Physicochemistry and Applications. Pharmaceutics 2021, 13, 1642. [Google Scholar] [CrossRef]
- Gómez-Morales, J.; Fernández-Penas, R.; Acebedo-Martínez, F.J.; Romero-Castillo, I.; Verdugo-Escamilla, C.; Choquesillo-Lazarte, D.; Esposti, L.D.; Jiménez-Martínez, Y.; Fernández-Sánchez, J.F.; Iafisco, M.; et al. Luminescent Citrate-Functionalized Terbium-Substituted Carbonated Apatite Nanomaterials: Structural Aspects, Sensitized Luminescence, Cytocompatibility, and Cell Uptake Imaging. Nanomaterials 2022, 12, 1257. [Google Scholar] [CrossRef]
- Cano Plá, S.M.; D’Urso, A.; Fernández-Sánchez, J.F.; Colangelo, D.; Choquesillo-Lazarte, D.; Ferracini, R.; Bosetti, M.; Prat, M.; Gómez-Morales, J. Biomimetic Citrate-Coated Luminescent Apatite Nanoplatforms for Diclofenac Delivery in Inflammatory Environments. Nanomaterials 2022, 12, 562. [Google Scholar] [CrossRef]
- Gómez-Morales, J.; Verdugo-Escamilla, C.; Fernández-Penas, R.; María Parra-Milla, C.; Drouet, C.; Maube-Bosc, F.; Oltolina, F.; Prat, M.; Fernando Fernández-Sánchez, J. Luminescent Biomimetic Citrate-Coated Europium-Doped Carbonated Apatite Nanoparticles for Use in Bioimaging: Physico-Chemistry and Cytocompatibility. RSC Adv. 2018, 8, 2385–2397. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, J.; Liu, J.; Zhang, Y.; Zhang, J.; Yang, R.; Meng, Z.; Gong, X.; Sui, L. Platinum nanoparticles enhance osteogenic differentiation of human dental follicle stem cells via scavenging ROS. Smart Mater. Med. 2023, 4, 621–638. [Google Scholar] [CrossRef]
- Nigar, F.; Johnston, A.-L.; Smith, J.; Oakley, W.; Islam, M.T.; Felfel, R.; Grant, D.; Lester, E.; Ahmed, I. Production of Nano Hydroxyapatite and Mg-Whitlockite from Biowaste-Derived Products via Continuous Flow Hydrothermal Synthesis: A Step towards Circular Economy. Materials 2023, 16, 2138. [Google Scholar] [CrossRef]
- Aruna Rani, S.V.; Rajkumar, K.; Saravana Karthikeyan, B.; Mahalaxmi, S.; Rajkumar, G.; Dhivya, V. Micro-Raman Spectroscopy Analysis of Dentin Remineralization Using Eggshell Derived Nanohydroxyapatite Combined with Phytosphingosine. J. Mech. Behav. Biomed. Mater. 2023, 141, 105748. [Google Scholar] [CrossRef]
- Johnson Jeyakumar, S.; Sindhya, A.; Jothibas, M.; Pugalendhi, P.; Sathiyamoorthy, K. Preparation and Analysis of Pure and Surface Modified Nanohydroxyapatite Derived from Eggshells and Its In-Vitro Studies for Bone Graft Applications. Ceram. Int. 2023, 49, 18708–18727. [Google Scholar] [CrossRef]
- Hussain, S.; Sabiruddin, K. Synthesis of Eggshell Based Hydroxyapatite Using Hydrothermal Method. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1189, 012024. [Google Scholar] [CrossRef]
- Noviyanti, A.R.; Akbar, N.; Deawati, Y.; Ernawati, E.E.; Malik, Y.T.; Fauzia, R.P. A Novel Hydrothermal Synthesis of Nanohydroxyapatite from Eggshell-Calcium-Oxide Precursors. Heliyon 2020, 6, e03655. [Google Scholar] [CrossRef]
- Fernández-Penas, R.; Verdugo-Escamilla, C.; Triunfo, C.; Gärtner, S.; D’Urso, A.; Oltolina, F.; Follenzi, A.; Maoloni, G.; Cölfen, H.; Falini, G.; et al. Sustainable One-Pot Path to Transform Seashell Waste Calcium Carbonate to Osteoinductive Hydroxyapatite Micro-Nanoparticles. J. Mater. Chem. B 2023. [Google Scholar] [CrossRef]
- Ganot, P.; Loentgen, G.; Marin, F.; Plasseraud, L.; Allemand, D.; Tambutté, S. An Alternative and Effective Method for Extracting Skeletal Organic Matrix Adapted to the Red Coral Corallium Rubrum. Biol. Open 2022, 11, bio059536. [Google Scholar] [CrossRef]
- Zamperone, A.; Pietronave, S.; Merlin, S.; Colangelo, D.; Ranaldo, G.; Medico, E.; Di Scipio, F.; Berta, G.N.; Follenzi, A.; Prat, M. Isolation and Characterization of a Spontaneously Immortalized Multipotent Mesenchymal Cell Line Derived from Mouse Subcutaneous Adipose Tissue. Stem Cells Dev. 2013, 22, 2873–2884. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in Mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef]
- Sutthavas, P.; Schumacher, M.; Zheng, K.; Habibović, P.; Boccaccini, A.R.; van Rijt, S. Zn-Loaded and Calcium Phosphate-Coated Degradable Silica Nanoparticles Can Effectively Promote Osteogenesis in Human Mesenchymal Stem Cells. Nanomaterials 2022, 12, 2918. [Google Scholar] [CrossRef]
- Athanasiadou, D.; Jiang, W.; Goldbaum, D.; Saleem, A.; Basu, K.; Pacella, M.S.; Böhm, C.F.; Chromik, R.R.; Hincke, M.T.; Rodríguez-Navarro, A.B.; et al. Nanostructure, Osteopontin, and Mechanical Properties of Calcitic Avian Eggshell. Sci. Adv. 2018, 4, eaar3219. [Google Scholar] [CrossRef]
- Nga, N.K.; Thuy Chau, N.T.; Viet, P.H. Facile Synthesis of Hydroxyapatite Nanoparticles Mimicking Biological Apatite from Eggshells for Bone-Tissue Engineering. Colloids Surf. B Biointerfaces 2018, 172, 769–778. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Anbalagan, G.; Pandi, S. Raman and Infrared Spectra of Carbonates of Calcite Structure. J. Raman Spectrosc. 2006, 37, 892–899. [Google Scholar] [CrossRef]
- Xu, B.; Hirsch, A.; Kronik, L.; Poduska, K.M. Vibrational Properties of Isotopically Enriched Materials: The Case of Calcite. RSC Adv. 2018, 8, 33985–33992. [Google Scholar] [CrossRef]
- Xu, B.; Poduska, K.M. Linking Crystal Structure with Temperature-Sensitive Vibrational Modes in Calcium Carbonate Minerals. Phys. Chem. Chem. Phys. 2014, 16, 17634–17639. [Google Scholar] [CrossRef]
- Kim, Y.; Caumon, M.-C.; Barres, O.; Sall, A.; Cauzid, J. Identification and Composition of Carbonate Minerals of the Calcite Structure by Raman and Infrared Spectroscopies Using Portable Devices. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 261, 119980. [Google Scholar] [CrossRef]
- Rey, C.; Marsan, O.; Combes, C.; Drouet, C.; Grossin, D.; Sarda, S. Characterization of Calcium Phosphates Using Vibrational Spectroscopies. In Advances in Calcium Phosphate Biomaterials; Springer Series in Biomaterials Science and Engineering; Springer: Berlin/Heidelberg, Germany, 2014; pp. 229–266. ISBN 978-3-642-53979-4. [Google Scholar]
- Grunenwald, A.; Keyser, C.; Sautereau, A.M.; Crubézy, E.; Ludes, B.; Drouet, C. Revisiting Carbonate Quantification in Apatite (Bio)Minerals: A Validated FTIR Methodology. J. Archaeol. Sci. 2014, 49, 134–141. [Google Scholar] [CrossRef]
- Kellum, J.A. Determinants of Blood PH in Health and Disease. Crit. Care 2000, 4, 6–14. [Google Scholar] [CrossRef][Green Version]
- Arnett, T.R. Extracellular PH Regulates Bone Cell Function. J. Nutr. 2008, 138, 415S–418S. [Google Scholar] [CrossRef]
- Boedtkjer, E.; Pedersen, S.F. The Acidic Tumor Microenvironment as a Driver of Cancer. Annu. Rev. Physiol. 2020, 82, 103–126. [Google Scholar] [CrossRef]
- Riemann, A.; Ihling, A.; Thomas, J.; Schneider, B.; Thews, O.; Gekle, M. Acidic Environment Activates Inflammatory Programs in Fibroblasts via a CAMP-MAPK Pathway. Biochim. Biophys. Acta 2015, 1853, 299–307. [Google Scholar] [CrossRef]
- Liang, W.; Ding, P.; Li, G.; Lu, E.; Zhao, Z. Hydroxyapatite Nanoparticles Facilitate Osteoblast Differentiation and Bone Formation Within Sagittal Suture During Expansion in Rats. Drug Des. Dev. Ther. 2021, 15, 905–917. [Google Scholar] [CrossRef]
- Wang, Z.; Han, T.; Zhu, H.; Tang, J.; Guo, Y.; Jin, Y.; Wang, Y.; Chen, G.; Gu, N.; Wang, C. Potential Osteoinductive Effects of Hydroxyapatite Nanoparticles on Mesenchymal Stem Cells by Endothelial Cell Interaction. Nanoscale Res. Lett. 2021, 16, 67. [Google Scholar] [CrossRef] [PubMed]
- Yi, C.; Liu, D.; Fong, C.-C.; Zhang, J.; Yang, M. Gold Nanoparticles Promote Osteogenic Differentiation of Mesenchymal Stem Cells through P38 MAPK Pathway. ACS Nano 2010, 4, 6439–6448. [Google Scholar] [CrossRef]
- Ruiz-Agudo, E.; Putnis, C.V.; Putnis, A. Coupled Dissolution and Precipitation at Mineral–Fluid Interfaces. Chem. Geol. 2014, 383, 132–146. [Google Scholar] [CrossRef]
- Zeglinski, J.; Nolan, M.; Thompson, D.; Tofail, S.A.M. Reassigning the Most Stable Surface of Hydroxyapatite to the Water Resistant Hydroxyl Terminated (010) Surface. Surf. Sci. 2014, 623, 55–63. [Google Scholar] [CrossRef]
- Yoshimura, M.; Sujaridworakun, P.; Koh, F.; Fujiwara, T.; Pongkao, D.; Ahniyaz, A. Hydrothermal Conversion of Calcite Crystals to Hydroxyapatite. Mater. Sci. Eng. C 2004, 24, 521–525. [Google Scholar] [CrossRef]
- Wu, S.-C.; Tsou, H.-K.; Hsu, H.-C.; Hsu, S.-K.; Liou, S.-P.; Ho, W.-F. A Hydrothermal Synthesis of Eggshell and Fruit Waste Extract to Produce Nanosized Hydroxyapatite. Ceram. Int. 2013, 39, 8183–8188. [Google Scholar] [CrossRef]
- ISO 10993-5:2009; Biological Evaluation of Medical Devices — Part 5: Tests for In Vitro Cytotoxicity. International Organization for Standardization: Geneva, Switzerland, 2009. Available online: https://www.iso.org/standard/36406.html (accessed on 30 November 2022).
- Ma, Q.; Rubenis, K.; Sirgurjónsson, Ò.; Hildebran, T.; Standal, T.; Zemjane, S.; Locs, J.; Loca, D.; Haugen, H.J. Eggshell-derived amorphous calcium phosphate: Synthesis, characterization and bio-functions as bone graft materials in novel 3D osteoblastic spheroids model. Smart Mater. Med. 2023, 4, 522–537. [Google Scholar] [CrossRef]
- Levingstone, T.J.; Herbaj, S.; Dunne, N.J. Calcium Phosphate Nanoparticles for Therapeutic Applications in Bone Regeneration. Nanomaterials 2019, 9, 1570. [Google Scholar] [CrossRef]
- Li, X.; Yang, X.; Liu, X.; He, W.; Huang, Q.; Li, S.; Feng, Q. Calcium carbonate nanoparticles promote osteogenesis compared to adipogenesis in human bone-marrow mesenchymal stem cells. Prog. Nat. Sci. Mater. Int. 2018, 28, 5–598. [Google Scholar] [CrossRef]
- Barradas, A.; Fernandes, H.; Groen, N.; Chin Chai, Y.; Schrooten, J.; van de Peppel, J.; van Leeuwen, J.; van Blitterswijk, C.; de Boer, J. A calcium-induced signaling cascade leading to osteogenic differentiation of human bone marrow-derived mesenchymal stromal cells. Biomaterials 2012, 33, 3205–3215. [Google Scholar] [CrossRef]
- Matsubara, T.; Kida, K.; Yamaguchi, A.; Hata, K.; Ichida, F.; Meguro, H. BMP2 regulates osterix through Msx2 and Runx2 during osteoblast differentiation. J. Biol. Chem. 2008, 283, 29119.e25. [Google Scholar] [CrossRef]
- Yang, X.; Li, Y.; Liu, X.; Zhang, R.; Feng, Q. In Vitro Uptake of Hydroxyapatite Nanoparticles and Their Effect on Osteogenic Differentiation of Human Mesenchymal Stem Cells. Stem Cells Int. 2018, 2018, e2036176. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, A.; González, A.; Planell, J.A.; Engel, E. Extracellular calcium modulates in vitro bone marrow-derived Flk-1 þ CD34þ progenitor cell chemotaxis and differentiation through a calcium-sensing receptor. Biochem. Biophys. Res. Commun. 2010, 393, 1–156e61. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Zhang, H.; Teng, J.; Huang, L.; Li, Y.; Sun, C. Involvement of calcium-sensing receptor in inhibition of lipolysis through intracellular cAMP and calcium pathways in human adipocytes. Biochem. Biophys. Res. Commun. 2011, 404, 393.e9. [Google Scholar] [CrossRef] [PubMed]
- Saidak, Z.; Brazier, M.; Kamel, S.; Mentaverri, R. Agonists and allosteric modulators of the Calcium-Sensing Receptor and Their Therapeutic Applications. Mol. Pharmacol. 2009, 76, 1131.e44. [Google Scholar] [CrossRef]
- Habraken, W.; Habibovic, P.; Epple, M.; Bohner, M. Calcium Phosphates in Biomedical Applications: Materials for the Future? Mater. Today 2016, 19, 69–87. [Google Scholar] [CrossRef]
- Zhang, X.; Vecchio, K.S. Conversion of Natural Marine Skeletons as Scaffolds for Bone Tissue Engineering. Front. Mater. Sci. 2013, 7, 103–117. [Google Scholar] [CrossRef]
- Clarke, S.A.; Choi, S.Y.; McKechnie, M.; Burke, G.; Dunne, N.; Walker, G.; Cunningham, E.; Buchanan, F. Osteogenic Cell Response to 3-D Hydroxyapatite Scaffolds Developed via Replication of Natural Marine Sponges. J. Mater. Sci. Mater. Med. 2016, 27, 22. [Google Scholar] [CrossRef]
- Green, D.W.; Ben-Nissan, B.; Yoon, K.S.; Milthorpe, B.; Jung, H.S. Natural and Synthetic Coral Biomineralization for Human Bone Revitalization. Trends Biotechnol. 2017, 35, 43–54. [Google Scholar] [CrossRef]
- Chen, L.; Al-Bayatee, S.; Khurshid, Z.; Shavandi, A.; Brunton, P.; Ratnayake, J. Hydroxyapatite in Oral Care Products—A Review. Materials 2021, 14, 4865. [Google Scholar] [CrossRef] [PubMed]

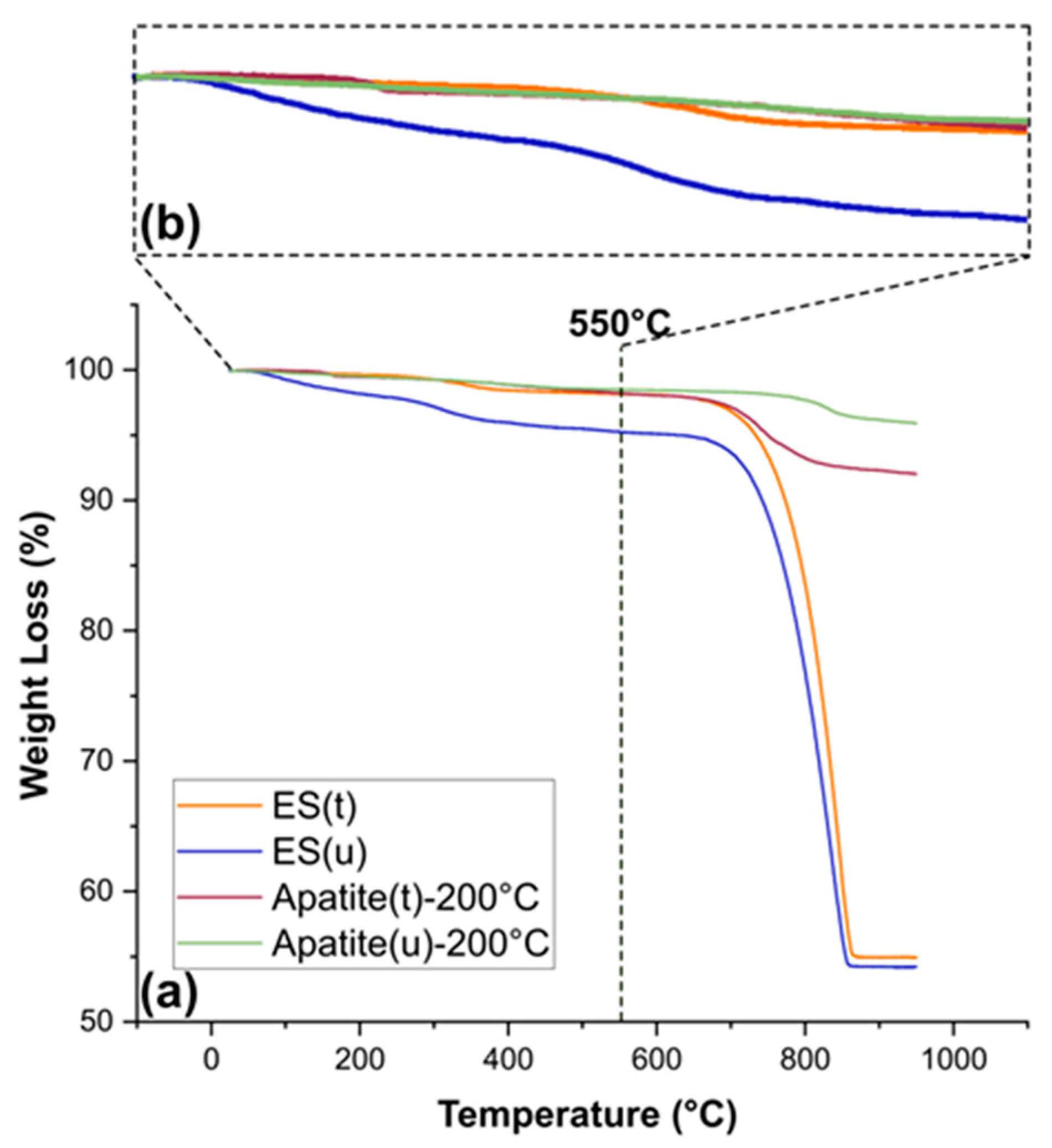

| ES(u) | ES(t) | Ap(u)-200 °C | Ap(t)-200 °C | |
|---|---|---|---|---|
| Water | 1.64 | 0.30 | 0.42 | 0.52 |
| OM | 3.09 | 1.55 | 1.07 | 1.26 |
| Mineral | 95.27 | 98.15 | 98.51 | 98.22 |
| Carbonate (550–850 °C) | - | - | 1.92 | 5.65 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres-Mansilla, A.; Álvarez-Lloret, P.; Fernández-Penas, R.; D’Urso, A.; Baldión, P.A.; Oltolina, F.; Follenzi, A.; Gómez-Morales, J. Hydrothermal Transformation of Eggshell Calcium Carbonate into Apatite Micro-Nanoparticles: Cytocompatibility and Osteoinductive Properties. Nanomaterials 2023, 13, 2299. https://doi.org/10.3390/nano13162299
Torres-Mansilla A, Álvarez-Lloret P, Fernández-Penas R, D’Urso A, Baldión PA, Oltolina F, Follenzi A, Gómez-Morales J. Hydrothermal Transformation of Eggshell Calcium Carbonate into Apatite Micro-Nanoparticles: Cytocompatibility and Osteoinductive Properties. Nanomaterials. 2023; 13(16):2299. https://doi.org/10.3390/nano13162299
Chicago/Turabian StyleTorres-Mansilla, Adriana, Pedro Álvarez-Lloret, Raquel Fernández-Penas, Annarita D’Urso, Paula Alejandra Baldión, Francesca Oltolina, Antonia Follenzi, and Jaime Gómez-Morales. 2023. "Hydrothermal Transformation of Eggshell Calcium Carbonate into Apatite Micro-Nanoparticles: Cytocompatibility and Osteoinductive Properties" Nanomaterials 13, no. 16: 2299. https://doi.org/10.3390/nano13162299
APA StyleTorres-Mansilla, A., Álvarez-Lloret, P., Fernández-Penas, R., D’Urso, A., Baldión, P. A., Oltolina, F., Follenzi, A., & Gómez-Morales, J. (2023). Hydrothermal Transformation of Eggshell Calcium Carbonate into Apatite Micro-Nanoparticles: Cytocompatibility and Osteoinductive Properties. Nanomaterials, 13(16), 2299. https://doi.org/10.3390/nano13162299








