Exploiting Blood Transport Proteins as Carborane Supramolecular Vehicles for Boron Neutron Capture Therapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Blood Transport Protein Structural Database
2.2. Docking and Refinement
2.3. MD Simulations
2.4. Molecular Mechanics/Generalized Born Surface Area (MM-GBSA) Calculations
3. Results and Discussion
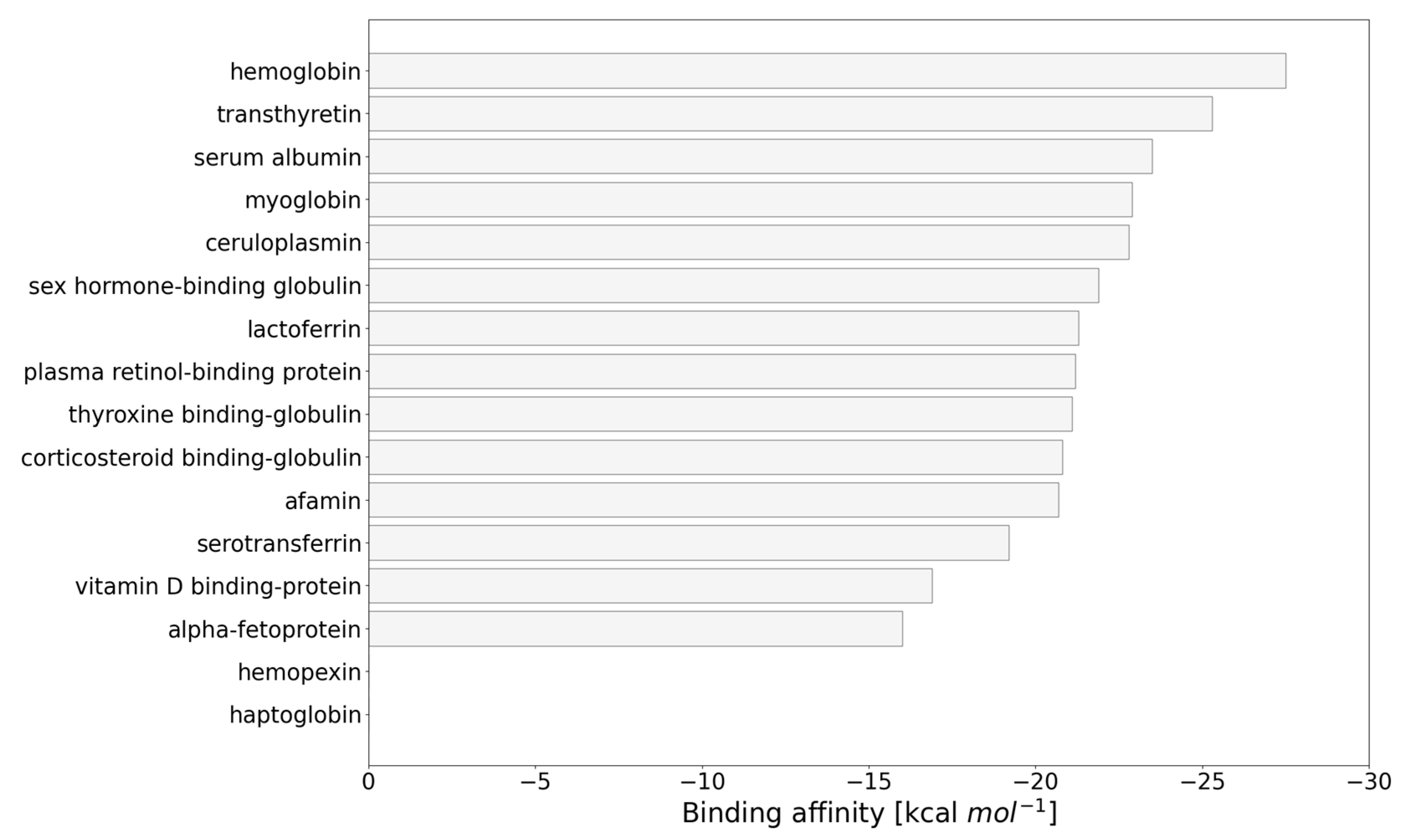
3.1. Identification of Blood Transport Proteins as Carriers for Carborane by Virtual Screening
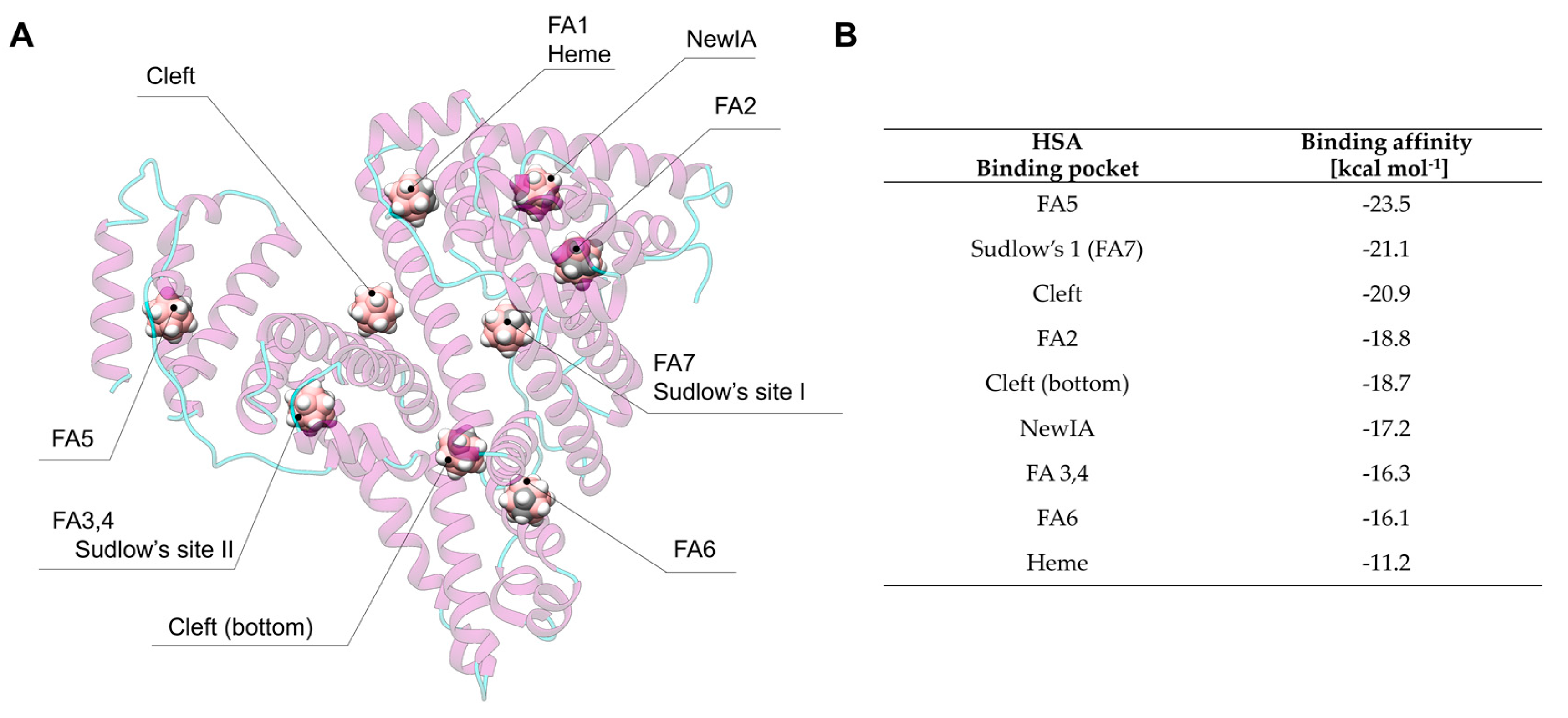
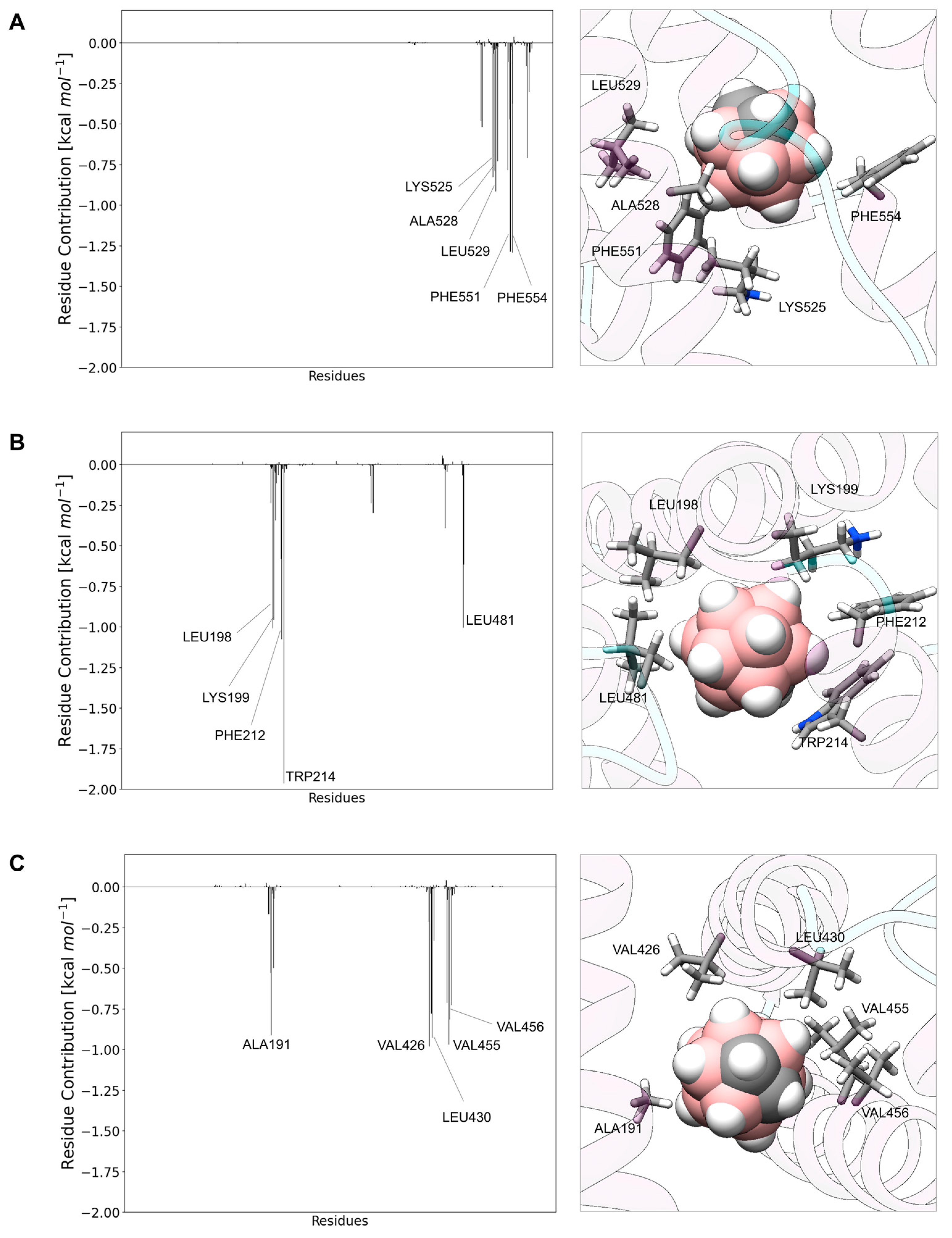
3.2. Carborane@HSA
3.3. Carborane@transthyretin
3.4. Carborane@hemoglobin
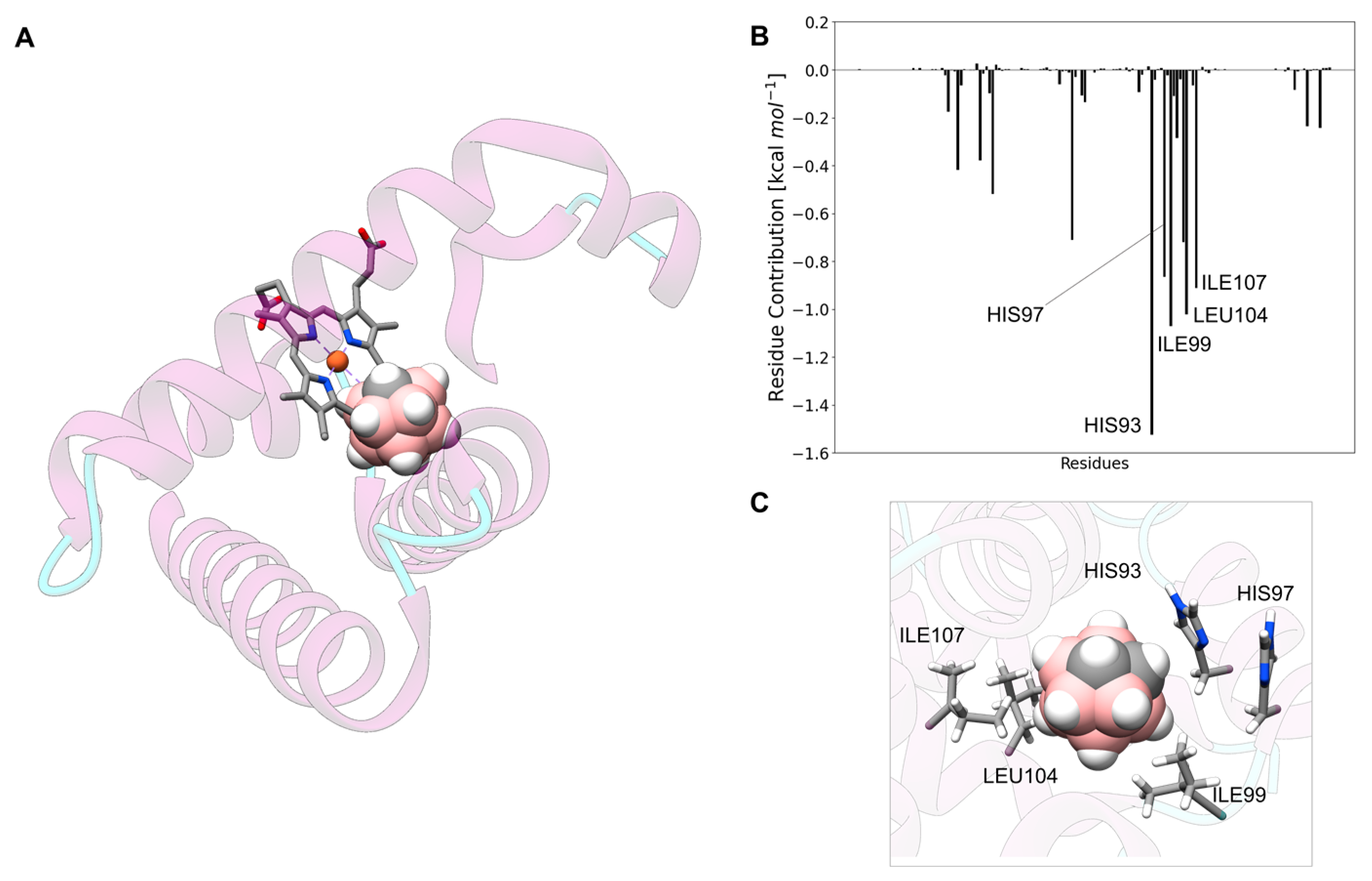
3.5. Carborane@myoglobin
3.6. Carborane@cerulosplamin
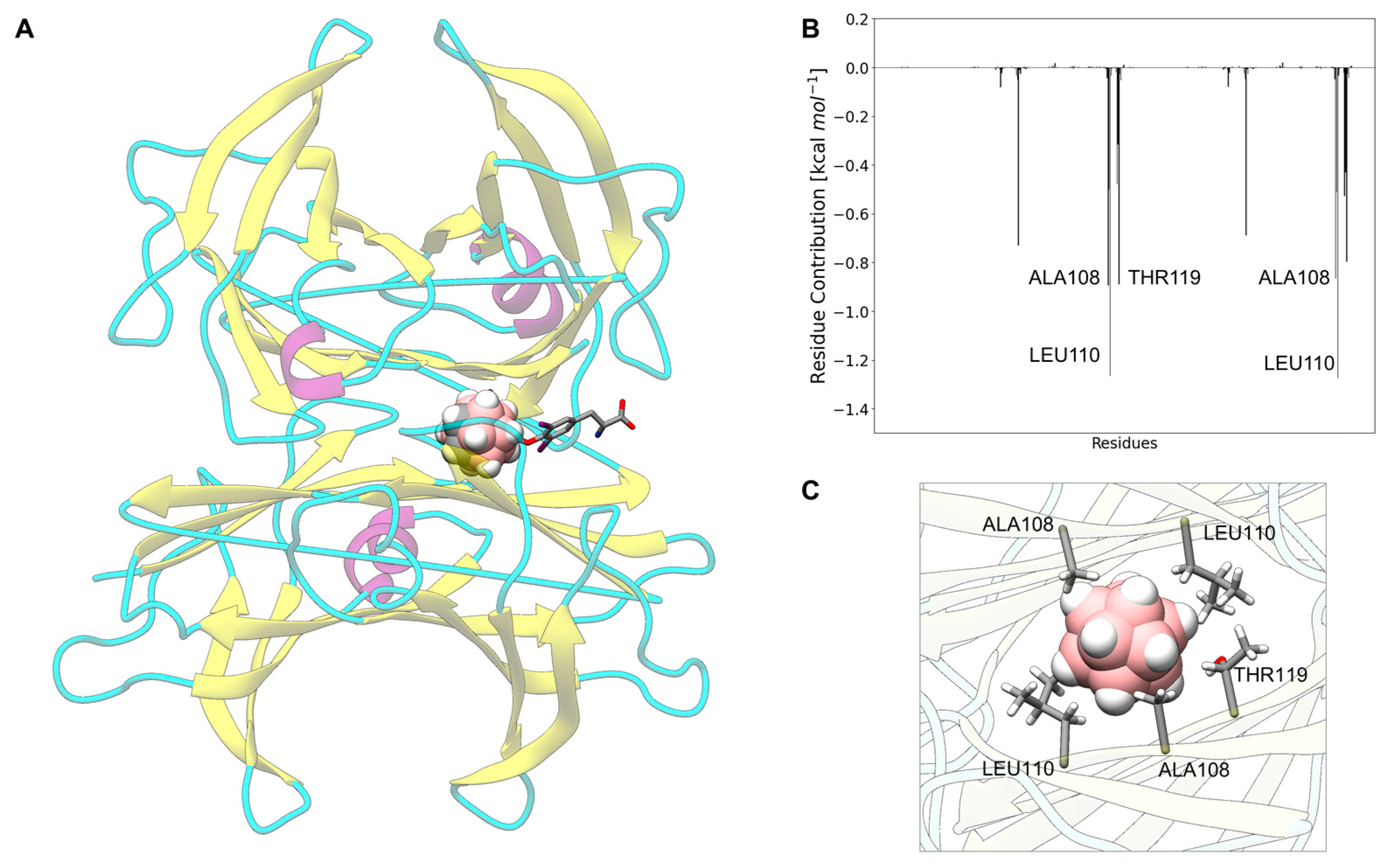
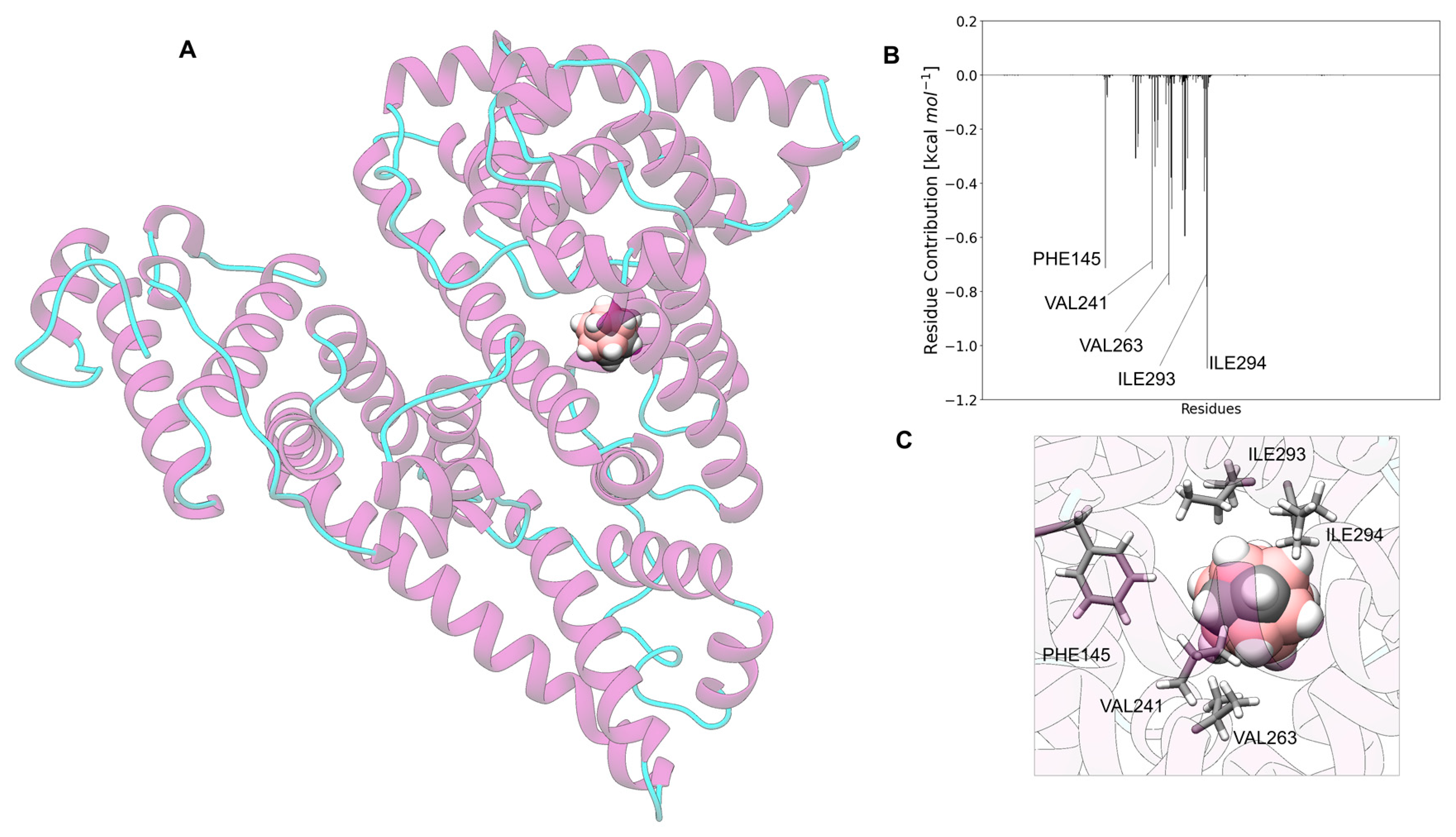
3.7. Carborane@sex Hormone-Binding Globulin
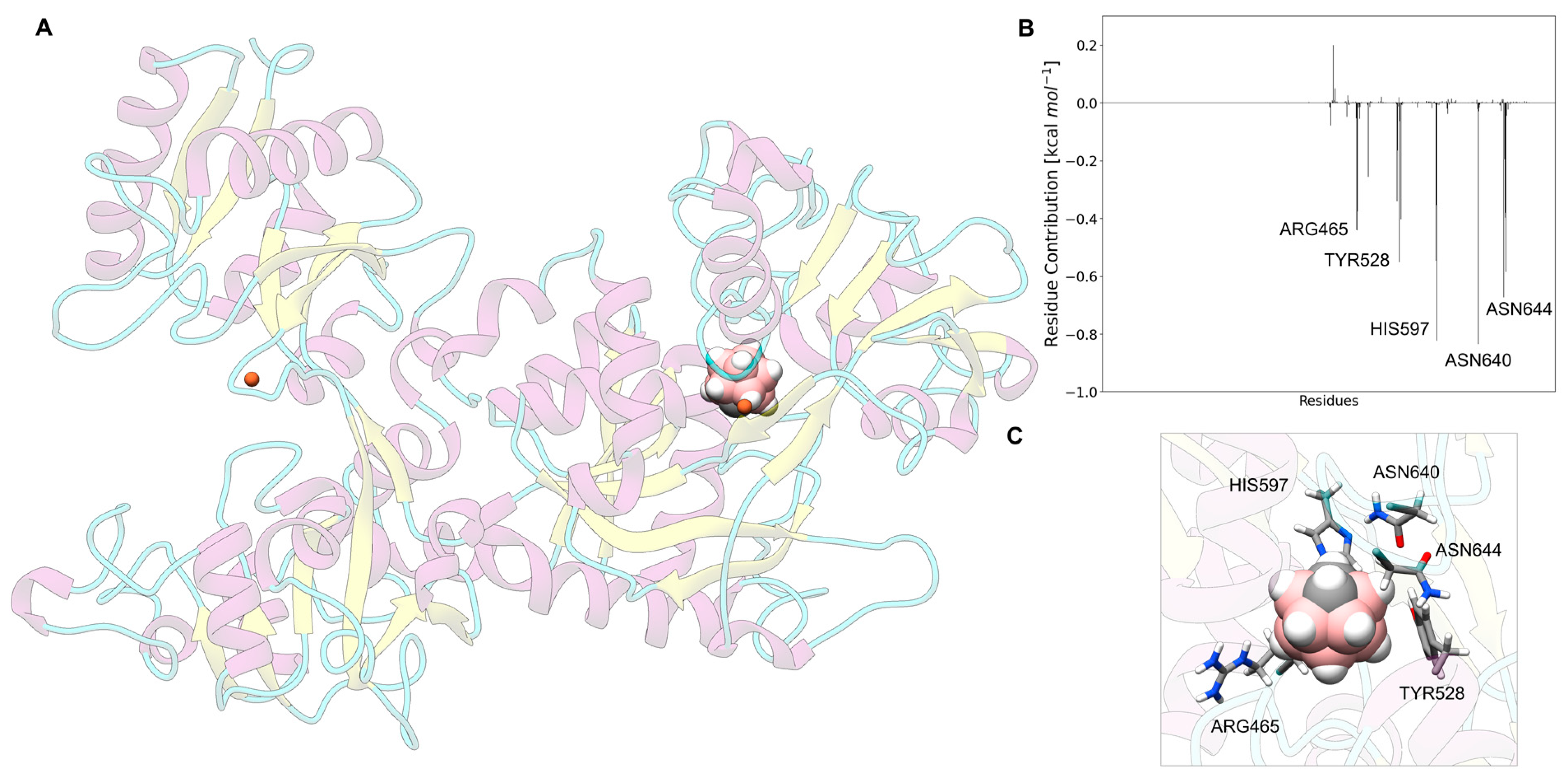
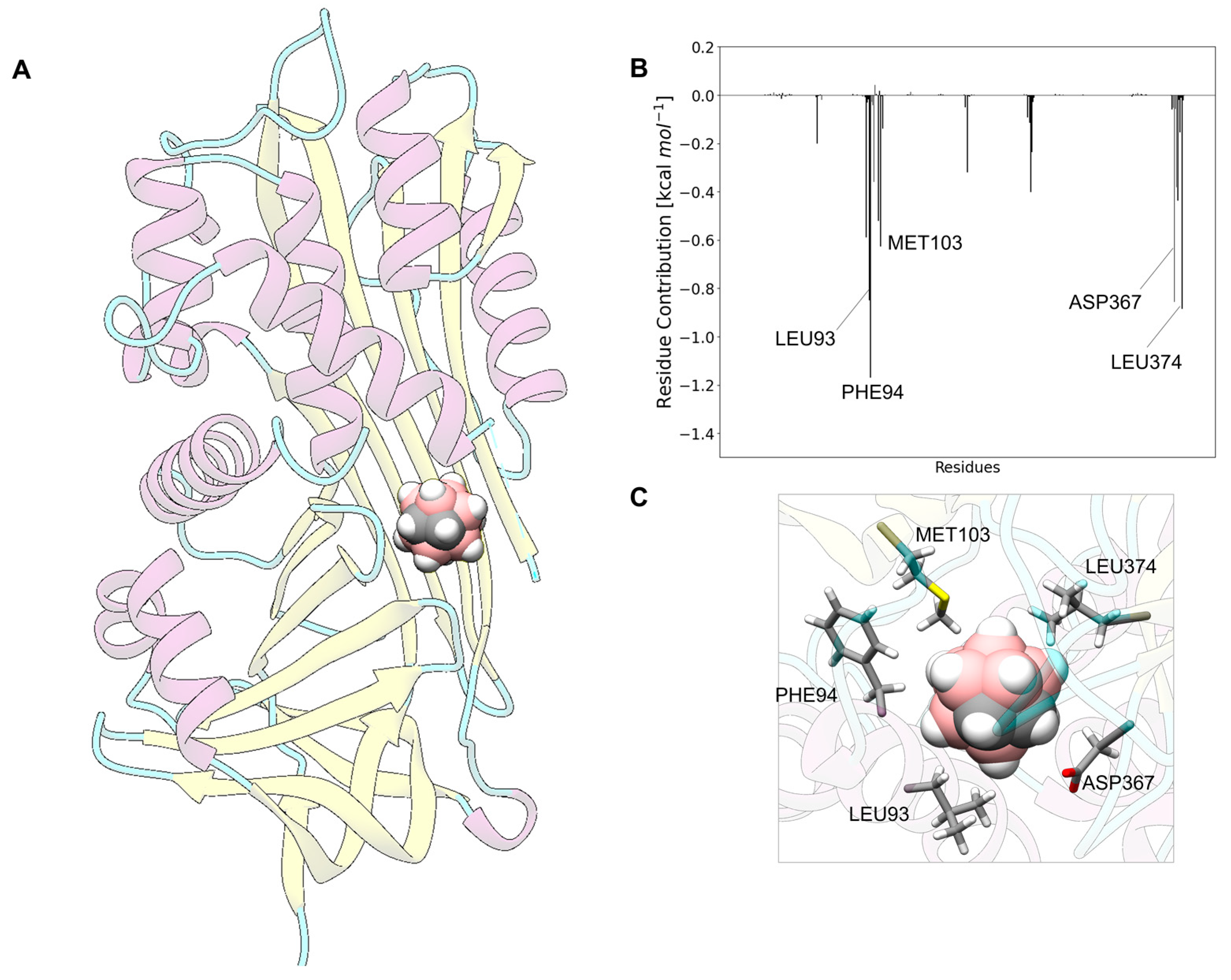
3.8. Carborane@lactotransferrin
3.9. Carborane@plasma Retinol-Binding Protein
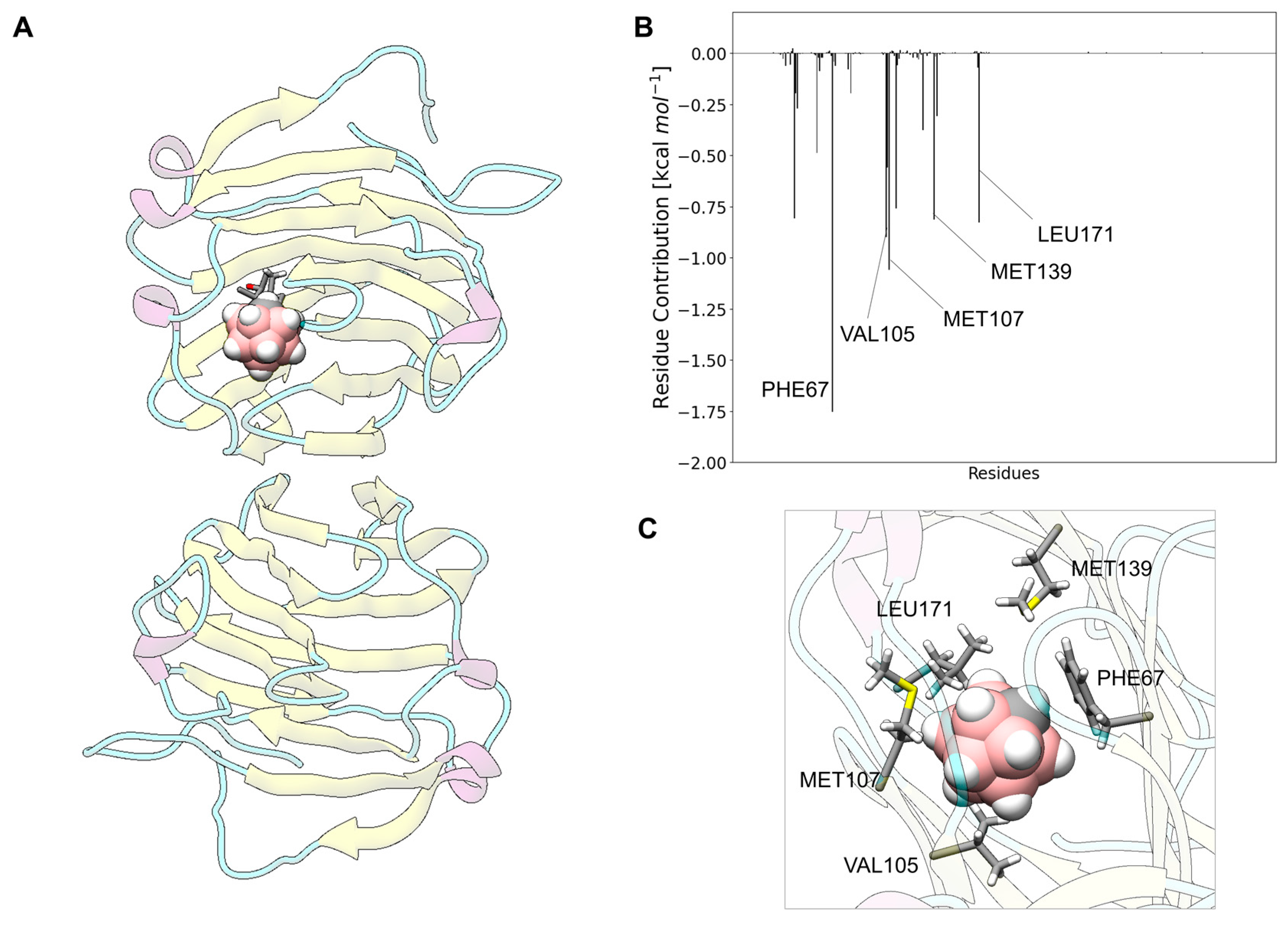
3.10. Carborane@thyroxine-Binding Globulin
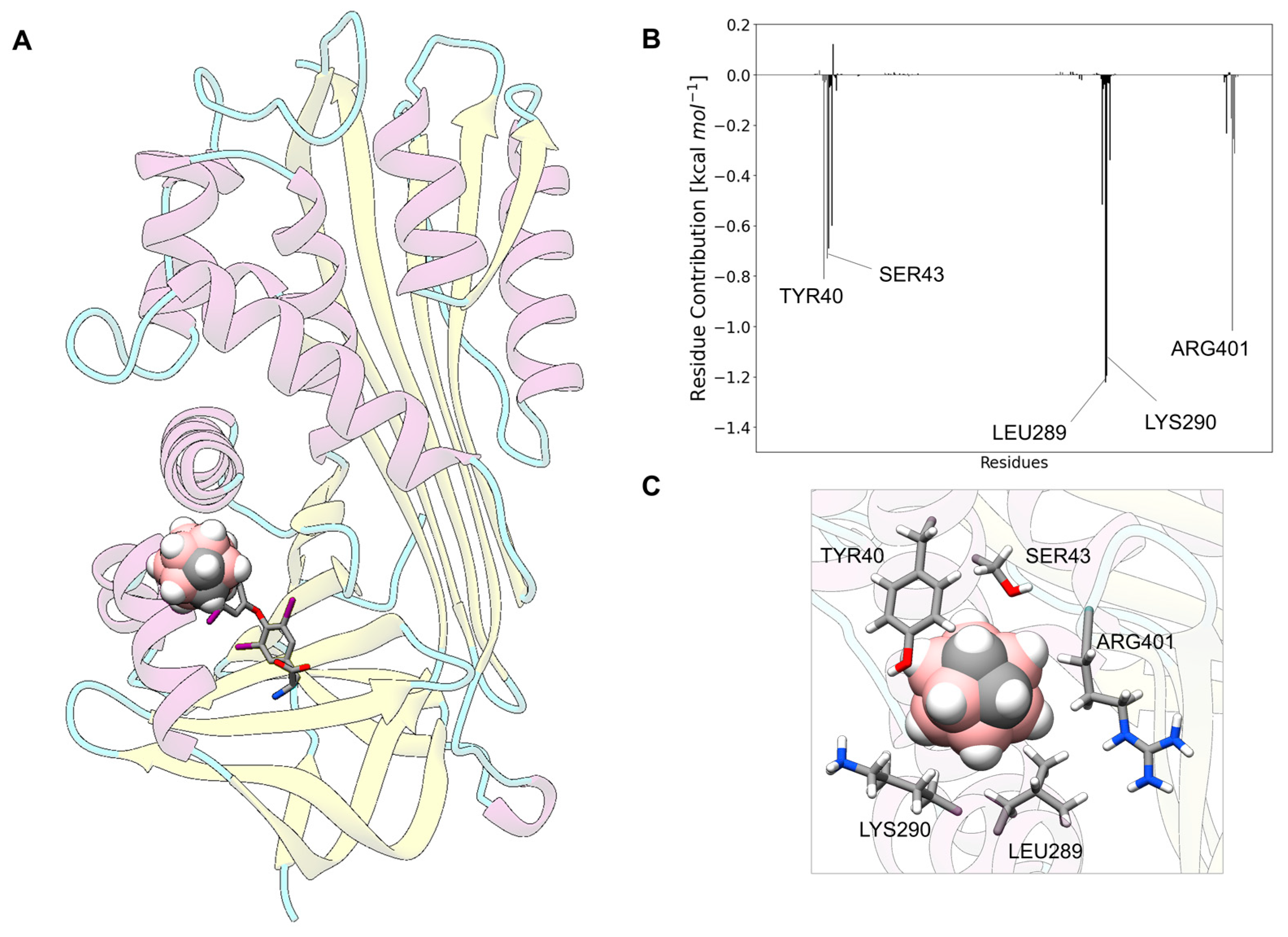
3.11. Carborane@corticosteroid-Binding Globulin
3.12. Carborane@afamin
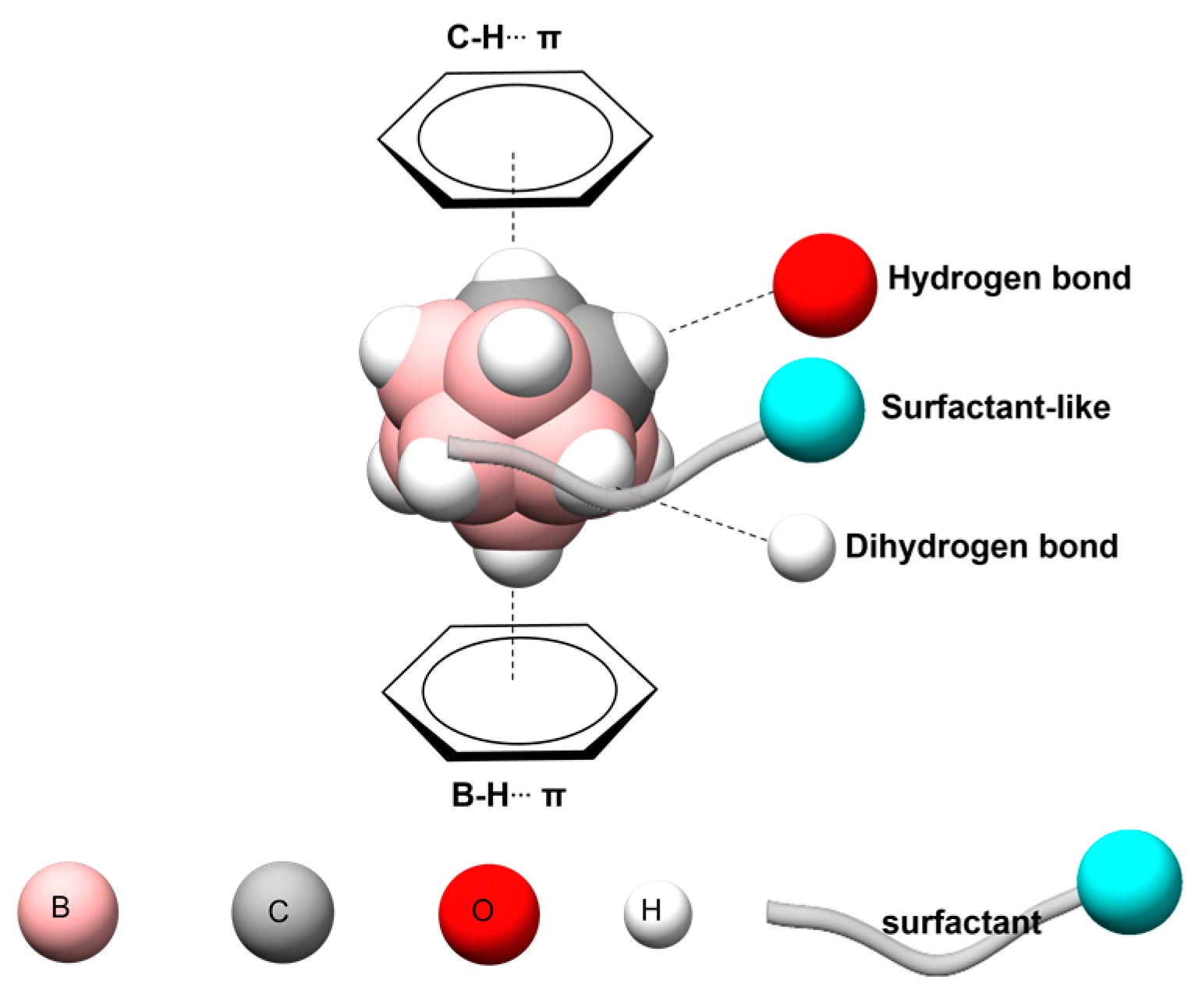
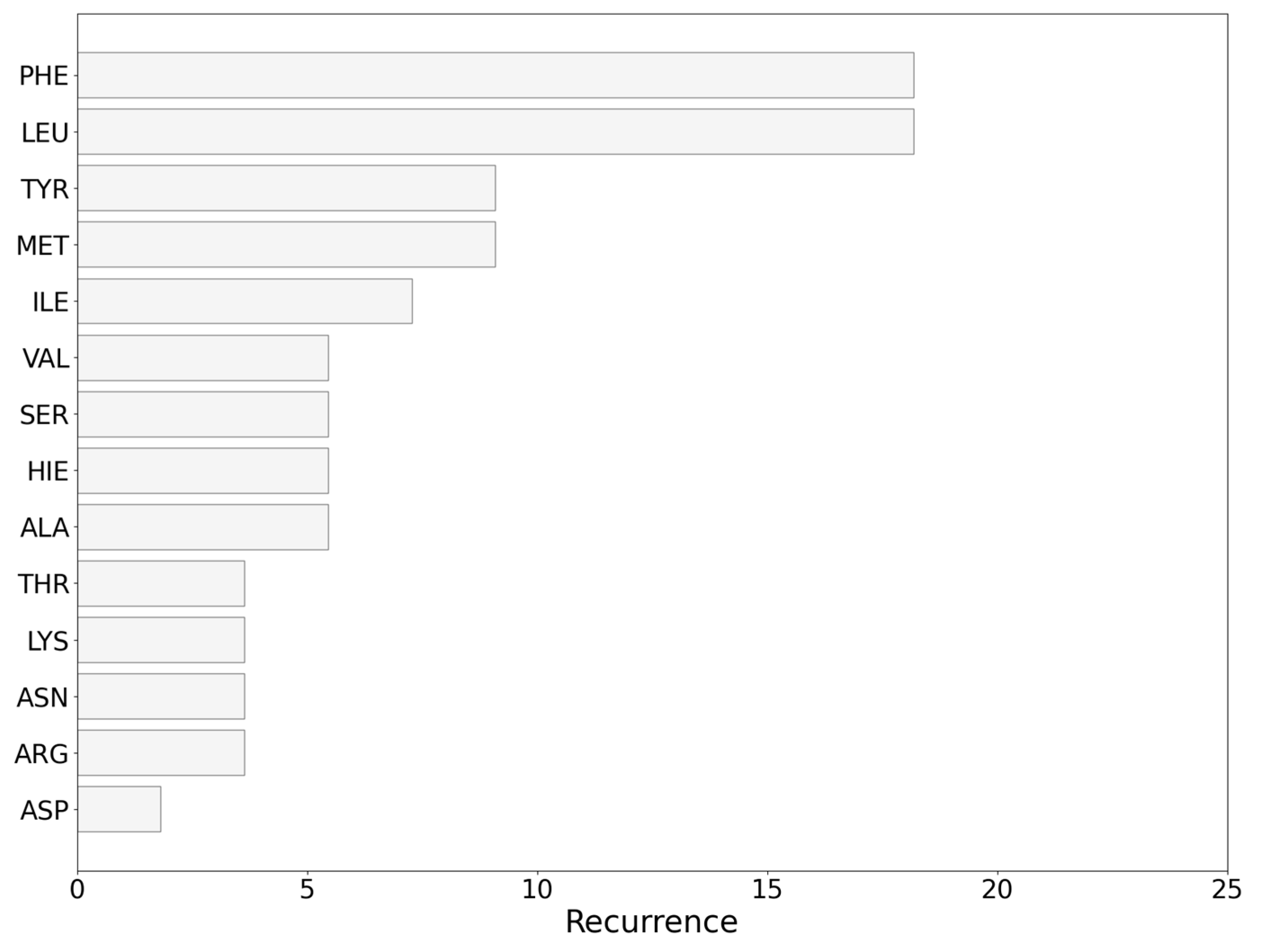
3.13. Analysis of the Nature of the Non-Covalent Interactions between the Amino Acids and the Carborane in the Protein-Binding Pockets
- (i)
- Hydrophobic interactions of the hydrophobic cage of the carborane with the aliphatic side chains of amino acids such as leucine (18%), methionine (9%), isoleucine (7%), valine (5%) and alanine (5%);
- (ii)
- BH-π and CH-π interactions of the hydridic B-H groups and acidic C-H groups of the carborane with aromatic amino acids such as phenylalanine (18%), tyrosine (9%) and histidine (5%).
- (iii)
- Formation of dihydrogen bonds with serine (5%), threonine (4%) and asparagine (4%), where serine and threonine use their hydroxylic moieties, while asparagine uses its amidic N-H to form the dihydrogen bonds with the hydridic B-H groups of the carborane;
- (iv)
- Surfactant-like interactions with lysine (4%) and arginine (4%) which wrap the hydrophobic cage of the carborane with their aliphatic chains, while their hydrophilic moieties interact with water;
- (v)
- Formation of classical hydrogen bonds with Asp (2.5%) where the carborane interacts with its acidic C-H groups with the carboxylate group of the amino acid.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malouff, T.D.; Seneviratne, D.S.; Ebner, D.K.; Stross, W.C.; Waddle, M.R.; Trifiletti, D.M.; Krishnan, S. Boron Neutron Capture Therapy: A Review of Clinical Applications. Front. Oncol. 2021, 11, 1820. [Google Scholar] [CrossRef] [PubMed]
- Murphy, N.; McCarthy, E.; Dwyer, R.; Farràs, P. Boron Clusters as Breast Cancer Therapeutics. J. Inorg. Biochem. 2021, 218, 111412. [Google Scholar] [CrossRef]
- Nuez-Martinez, M.; Pinto, C.I.G.; Guerreiro, J.F.; Mendes, F.; Marques, F.; Muñoz-Juan, A.; Xavier, J.A.M.; Laromaine, A.; Bitonto, V.; Protti, N.; et al. Cobaltabis(Dicarbollide) ([o-COSAN]−) as Multifunctional Chemotherapeutics: A Prospective Application in Boron Neutron Capture Therapy (BNCT) for Glioblastoma. Cancers 2021, 13, 6367. [Google Scholar] [CrossRef]
- Messner, K.; Vuong, B.; Tranmer, G.K. The Boron Advantage: The Evolution and Diversification of Boron’s Applications in Medicinal Chemistry. Pharmaceuticals 2022, 15, 264. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; Hosmane, N.S.; Zhu, Y. Boron Chemistry for Medical Applications. Molecules 2020, 25, 828. [Google Scholar] [CrossRef]
- Cheng, X.; Li, F.; Liang, L. Boron Neutron Capture Therapy: Clinical Application and Research Progress. Curr. Oncol. 2022, 29, 7868–7886. [Google Scholar] [CrossRef] [PubMed]
- Lamba, M.; Goswami, A.; Bandyopadhyay, A. A Periodic Development of BPA and BSH Based Derivatives in Boron Neutron Capture Therapy (BNCT). Chem. Commun. 2021, 57, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Zharkov, D.O.; Yudkina, A.V.; Riesebeck, T.; Loshchenova, P.S.; Mostovich, E.A.; Dianov, G.L. Boron-Containing Nucleosides as Tools for Boron-Neutron Capture Therapy. Am. J. Cancer Res. 2021, 11, 4668–4682. [Google Scholar]
- Gruzdev, D.A.; Levit, G.L.; Krasnov, V.P.; Charushin, V.N. Carborane-Containing Amino Acids and Peptides: Synthesis, Properties and Applications. Coord. Chem. Rev. 2021, 433, 213753. [Google Scholar] [CrossRef]
- Alamón, C.; Dávila, B.; García, M.F.; Sánchez, C.; Kovacs, M.; Trias, E.; Barbeito, L.; Gabay, M.; Zeineh, N.; Gavish, M.; et al. Sunitinib-Containing Carborane Pharmacophore with the Ability to Inhibit Tyrosine Kinases Receptors FLT3, KIT and PDGFR-β, Exhibits Powerful In Vivo Anti-Glioblastoma Activity. Cancers 2020, 12, 3423. [Google Scholar] [CrossRef] [PubMed]
- Barth, R.F.; Yang, W.; Al-Madhoun, A.S.; Johnsamuel, J.; Byun, Y.; Chandra, S.; Smith, D.R.; Tjarks, W.; Eriksson, S. Boron-Containing Nucleosides as Potential Delivery Agents for Neutron Capture Therapy of Brain Tumors. Cancer Res. 2004, 64, 6287–6295. [Google Scholar] [CrossRef] [PubMed]
- Leśnikowski, Z.J.; Paradowska, E.; Olejniczak, A.B.; Studzińska, M.; Seekamp, P.; Schüßler, U.; Gabel, D.; Schinazi, R.F.; Plešek, J. Towards New Boron Carriers for Boron Neutron Capture Therapy: Metallacarboranes and Their Nucleoside Conjugates. Bioorganic Med. Chem. 2005, 13, 4168–4175. [Google Scholar] [CrossRef] [PubMed]
- Smilowitz, H.M.; Slatkin, D.N.; Micca, P.L.; Miura, M. Microlocalization of Lipophilic Porphyrins: Non-Toxic Enhancers of Boron Neutron-Capture Therapy. Int. J. Radiat. Biol. 2013, 89, 611–617. [Google Scholar] [CrossRef]
- Miura, M.; Morris, G.M.; Hopewell, J.W.; Micca, P.L.; Makar, M.S.; Nawrocky, M.M.; Renner, M.W. Enhancement of the Radiation Response of EMT-6 Tumours by a Copper Octabromotetracarboranylphenylporphyrin. Br. J. Radiol. 2014, 85, 443–450. [Google Scholar] [CrossRef]
- Kawabata, S.; Yang, W.; Barth, R.F.; Wu, G.; Huo, T.; Binns, P.J.; Riley, K.J.; Ongayi, O.; Gottumukkala, V.; Vicente, M.G.H. Convection Enhanced Delivery of Carboranylporphyrins for Neutron Capture Therapy of Brain Tumors. J. Neurooncol. 2011, 103, 175–185. [Google Scholar] [CrossRef]
- Hu, K.; Yang, Z.; Zhang, L.; Xie, L.; Wang, L.; Xu, H.; Josephson, L.; Liang, S.H.; Zhang, M.R. Boron Agents for Neutron Capture Therapy. Coord. Chem. Rev. 2020, 405, 213139. [Google Scholar] [CrossRef]
- Imperio, D.; Panza, L. Sweet Boron: Boron-Containing Sugar Derivatives as Potential Agents for Boron Neutron Capture Therapy. Symmetry 2022, 14, 182. [Google Scholar] [CrossRef]
- Schwarze, B.; Gozzi, M.; Zilberfain, C.; Rüdiger, J.; Birkemeyer, C.; Estrela-Lopis, I.; Hey-Hawkins, E. Nanoparticle-Based Formulation of Metallacarboranes with Bovine Serum Albumin for Application in Cell Cultures. J. Nanoparticle Res. 2020, 22, 24. [Google Scholar] [CrossRef]
- Kawasaki, R.; Sasaki, Y.; Akiyoshi, K. Self-Assembled Nanogels of Carborane-Bearing Polysaccharides for Boron Neutron Capture Therapy. Chem. Lett. 2017, 46, 513–515. [Google Scholar] [CrossRef]
- Silva, W.M.; Ribeiro, H.; Taha-Tijerina, J.J. Potential Production of Theranostic Boron Nitride Nanotubes (64Cu-BNNTs) Radiolabeled by Neutron Capture. Nanomaterials 2021, 11, 2907. [Google Scholar] [CrossRef]
- Chiang, C.W.; Chien, Y.C.; Yu, W.J.; Ho, C.Y.; Wang, C.Y.; Wang, T.W.; Chiang, C.S.; Keng, P.Y. Polymer-Coated Nanoparticles for Therapeutic and Diagnostic Non-10B Enriched Polymer-Coated Boron Carbon Oxynitride (BCNO) Nanoparticles as Potent BNCT Drug. Nanomaterials 2021, 11, 2936. [Google Scholar] [CrossRef]
- Pitto-Barry, A. Polymers and Boron Neutron Capture Therapy (BNCT): A Potent Combination. Polym. Chem. 2021, 12, 2035–2044. [Google Scholar] [CrossRef]
- Takeuchi, I.; Kishi, N.; Shiokawa, K.; Uchiro, H.; Makino, K. Polyborane Encapsulated Liposomes Prepared Using PH Gradient and Reverse-Phase Evaporation for Boron Neutron Capture Therapy: Biodistribution in Tumor-Bearing Mice. Colloid Polym. Sci. 2018, 296, 1137–1144. [Google Scholar] [CrossRef]
- Xiong, H.; Wei, X.; Zhou, D.; Qi, Y.; Xie, Z.; Chen, X.; Jing, X.; Huang, Y. Amphiphilic Polycarbonates from Carborane-Installed Cyclic Carbonates as Potential Agents for Boron Neutron Capture Therapy. Bioconjugate Chem. 2016, 27, 2214–2223. [Google Scholar] [CrossRef]
- Lee, W.; Sarkar, S.; Ahn, H.; Kim, J.Y.; Lee, Y.J.; Chang, Y.; Yoo, J. PEGylated Liposome Encapsulating Nido-Carborane Showed Significant Tumor Suppression in Boron Neutron Capture Therapy (BNCT). Biochem. Biophys. Res. Commun. 2020, 522, 669–675. [Google Scholar] [CrossRef]
- Tsygankova, A.R.; Gruzdev, D.A.; Kanygin, V.V.; Guselnikova, T.Y.; Telegina, A.A.; Kasatova, A.I.; Kichigin, A.I.; Levit, G.L.; Mechetina, L.V.; Mukhamadiyarov, R.A.; et al. Liposomes Loaded with Lipophilic Derivative of Closo-Carborane as a Potential Boron Delivery System for Boron Neutron Capture Therapy of Tumors. Mendeleev. Commun. 2021, 31, 659–661. [Google Scholar] [CrossRef]
- Singh, B.; Kaur, G.; Singh, P.; Singh, K.; Kumar, B.; Vij, A.; Kumar, M.; Bala, R.; Meena, R.; Singh, A.; et al. Nanostructured Boron Nitride With High Water Dispersibility For Boron Neutron Capture Therapy. Sci. Rep. 2016, 6, 35535. [Google Scholar] [CrossRef]
- Ailuno, G.; Balboni, A.; Caviglioli, G.; Lai, F.; Barbieri, F.; Dellacasagrande, I.; Florio, T.; Baldassari, S. Boron Vehiculating Nanosystems for Neutron Capture Therapy in Cancer Treatment. Cells 2022, 11, 4029. [Google Scholar] [CrossRef]
- Sumitani, S.; Nagasaki, Y. Boron Neutron Capture Therapy Assisted by Boron-Conjugated Nanoparticles. Polym. J. 2012, 44, 522–530. [Google Scholar] [CrossRef]
- Dukenbayev, K.; Korolkov, I.V.; Tishkevich, D.I.; Kozlovskiy, A.L.; Trukhanov, S.V.; Gorin, Y.G.; Shumskaya, E.E.; Kaniukov, E.Y.; Vinnik, D.A.; Zdorovets, M.V.; et al. Fe3O4 Nanoparticles for Complex Targeted Delivery and Boron Neutron Capture Therapy. Nanomaterials 2019, 9, 494. [Google Scholar] [CrossRef]
- Ferreira, T.H.; Miranda, M.C.; Rocha, Z.; Leal, A.S.; Gomes, D.A.; Sousa, E.M.B. An Assessment of the Potential Use of BNNTs for Boron Neutron Capture Therapy. Nanomaterials 2017, 7, 82. [Google Scholar] [CrossRef]
- Oleshkevich, E.; Morancho, A.; Saha, A.; Galenkamp, K.M.O.; Grayston, A.; Crich, S.G.; Alberti, D.; Protti, N.; Comella, J.X.; Teixidor, F.; et al. Combining Magnetic Nanoparticles and Icosahedral Boron Clusters in Biocompatible Inorganic Nanohybrids for Cancer Therapy. Nanomedicine 2019, 20, 101986. [Google Scholar] [CrossRef]
- Ferrer-Ugalde, A.; Sandoval, S.; Pulagam, K.R.; Muñoz-Juan, A.; Laromaine, A.; Llop, J.; Tobias, G.; Núñez, R. Radiolabeled Cobaltabis(Dicarbollide) Anion-Graphene Oxide Nanocomposites for in Vivo Bioimaging and Boron Delivery. ACS Appl. Nano Mater. 2021, 4, 1613–1625. [Google Scholar] [CrossRef]
- Yan, J.; Yang, W.; Zhang, Q.; Yan, Y. Introducing Borane Clusters into Polymeric Frameworks: Architecture, Synthesis, and Applications. Chem. Commun. 2020, 56, 11720–11734. [Google Scholar] [CrossRef]
- Marfavi, A.; Kavianpour, P.; Rendina, L.M. Carboranes in Drug Discovery, Chemical Biology and Molecular Imaging. Nat. Rev. Chem. 2022, 6, 486–504. [Google Scholar] [CrossRef]
- Poater, J.; Viñas, C.; Bennour, I.; Escayola, S.; Solà, M.; Teixidor, F. Too Persistent to Give Up: Aromaticity in Boron Clusters Survives Radical Structural Changes. J. Am. Chem. Soc. 2020, 142, 9396–9407. [Google Scholar] [CrossRef]
- Farràs, P.; Juárez-Pérez, E.J.; Lepšík, M.; Luque, R.; Núñez, R. Metallacarboranes and Their Interactions: Theoretical Insights and Their Applicability. Chem. Soc. Rev. 2012, 41, 3445–3463. [Google Scholar] [CrossRef]
- Issa, F.; Kassiou, M.; Rendina, L.M. Boron in Drug Discovery: Carboranes as Unique Pharmacophores in Biologically Active Compounds. Chem. Rev. 2011, 111, 5701–5722. [Google Scholar] [CrossRef]
- Chen, Y.; Du, F.; Tang, L.; Xu, J.; Zhao, Y.; Wu, X.; Li, M.; Shen, J.; Wen, Q.; Cho, C.H.; et al. Carboranes as Unique Pharmacophores in Antitumor Medicinal Chemistry. Mol. Ther. Oncolytics 2022, 24, 400–416. [Google Scholar] [CrossRef]
- Barry, N.P.E.; Sadler, P.J. Dicarba-Closo-Dodecarborane-Containing Half-Sandwich Complexes of Ruthenium, Osmium, Rhodium and Iridium: Biological Relevance and Synthetic Strategies. Chem. Soc. Rev. 2012, 41, 3264–3279. [Google Scholar] [CrossRef]
- Fink, K.; Uchman, M. Boron Cluster Compounds as New Chemical Leads for Antimicrobial Therapy. Coord. Chem. Rev. 2021, 431, 213684. [Google Scholar] [CrossRef]
- Stockmann, P.; Gozzi, M.; Kuhnert, R.; Sárosi, M.B.; Hey-Hawkins, E. New Keys for Old Locks: Carborane-Containing Drugs as Platforms for Mechanism-Based Therapies. Chem. Soc. Rev. 2019, 48, 3497. [Google Scholar] [CrossRef]
- Fithroni, A.B.; Kobayashi, K.; Uji, H.; Ishimoto, M.; Akehi, M.; Ohtsuki, T.; Matsuura, E. Novel Self-Forming Nanosized DDS Particles for BNCT: Utilizing A Hydrophobic Boron Cluster and Its Molecular Glue Effect. Cells 2022, 11, 3307. [Google Scholar] [CrossRef]
- Takeuchi, I.; Nomura, K.; Makino, K. Hydrophobic Boron Compound-Loaded Poly(l-Lactide-Co-Glycolide) Nanoparticles for Boron Neutron Capture Therapy. Colloids Surf. B Biointerfaces 2017, 159, 360–365. [Google Scholar] [CrossRef]
- Alberti, D.; Protti, N.; Toppino, A.; Deagostino, A.; Lanzardo, S.; Bortolussi, S.; Altieri, S.; Voena, C.; Chiarle, R.; Crich, S.G.; et al. A Theranostic Approach Based on the Use of a Dual Boron/Gd Agent to Improve the Efficacy of Boron Neutron Capture Therapy in the Lung Cancer Treatment. Nanomedicine 2015, 11, 741–750. [Google Scholar] [CrossRef]
- Heber, E.M.; Kueffer, P.J.; Lee, M.W.; Hawthorne, M.F.; Garabalino, M.A.; Molinari, A.J.; Nigg, D.W.; Bauer, W.; Hughes, A.M.; Pozzi, E.C.C.; et al. Boron Delivery with Liposomes for Boron Neutron Capture Therapy (BNCT): Biodistribution Studies in an Experimental Model of Oral Cancer Demonstrating Therapeutic Potential. Radiat. Environ. Biophys. 2012, 51, 195–204. [Google Scholar] [CrossRef]
- Zhang, T.; Xu, D.; Yi, Y.; Wang, Y.; Cui, Z.; Chen, X.; Ma, Q.; Song, F.; Zhu, B.; Zhao, Z.; et al. Chitosan-Lactobionic Acid-Thioctic Acid-Modified Hollow Mesoporous Silica Composite Loaded with Carborane for Boron Neutron Capture Therapy of Hepatocellular Carcinoma. Mater. Des. 2022, 223, 111196. [Google Scholar] [CrossRef]
- Rak, J.; Kaplánek, R.; Král, V. Solubilization and Deaggregation of Cobalt Bis(Dicarbollide) Derivatives in Water by Biocompatible Excipients. Bioorganic Med. Chem. Lett. 2010, 20, 1045–1048. [Google Scholar] [CrossRef]
- Rak, J.; Jakubek, M.; Kaplánek, R.; Matějíček, P.; Král, V. Cobalt Bis(Dicarbollide) Derivatives: Solubilization and Self-Assembly Suppression. Eur. J. Med. Chem. 2011, 46, 1140–1146. [Google Scholar] [CrossRef]
- Rak, J.; Dejlová, B.; Lampová, H.; Kaplánek, R.; Matějíček, P.; Cígler, P.; Král, V. On the Solubility and Lipophilicity of Metallacarborane Pharmacophores. Mol. Pharm. 2013, 10, 1751–1759. [Google Scholar] [CrossRef]
- Marconi, A.; Giugliano, G.; Di Giosia, M.; Marforio, T.D.; Trivini, M.; Turrini, E.; Fimognari, C.; Zerbetto, F.; Mattioli, E.J.; Calvaresi, M. Identification of Blood Transport Proteins to Carry Temoporfin: A Domino Approach from Virtual Screening to Synthesis and In Vitro PDT Testing. Pharmaceutics 2023, 15, 919. [Google Scholar] [CrossRef]
- Cantelli, A.; Malferrari, M.; Mattioli, E.J.; Marconi, A.; Mirra, G.; Soldà, A.; Marforio, T.D.; Zerbetto, F.; Rapino, S.; Di Giosia, M.; et al. Enhanced Uptake and Phototoxicity of C60@albumin Hybrids by Folate Bioconjugation. Nanomaterials 2022, 12, 3501. [Google Scholar] [CrossRef]
- Mattioli, E.J.; Ulfo, L.; Marconi, A.; Pellicioni, V.; Costantini, P.E.; Marforio, T.D.; Di Giosia, M.; Danielli, A.; Fimognari, C.; Turrini, E.; et al. Carrying Temoporfin with Human Serum Albumin: A New Perspective for Photodynamic Application in Head and Neck Cancer. Biomolecules 2023, 13, 68. [Google Scholar] [CrossRef]
- Marconi, A.; Mattioli, E.J.; Ingargiola, F.; Giugliano, G.; Marforio, T.D.; Prodi, L.; Di Giosia, M.; Calvaresi, M. Dissecting the Interactions between Chlorin E6 and Human Serum Albumin. Molecules 2023, 28, 2348. [Google Scholar] [CrossRef]
- Calvaresi, M.; Zerbetto, F. In Silico Carborane Docking to Proteins and Potential Drug Targets. J. Chem. Inf. Model. 2011, 51, 1882–1896. [Google Scholar] [CrossRef]
- Di Giosia, M.; Zerbetto, F.; Calvaresi, M. Incorporation of Molecular Nanoparticles inside Proteins: The Trojan Horse Approach in Theranostics. Acc. Mater. Res. 2021, 2, 594–605. [Google Scholar] [CrossRef]
- Frontera, A.; Bauzá, A. Closo -Carboranes as Dual CH⋯π and BH⋯π Donors: Theoretical Study and Biological Significance. Phys. Chem. Chem. Phys. 2019, 21, 19944–19950. [Google Scholar] [CrossRef]
- Zou, W.; Zhang, X.; Dai, H.; Yan, H.; Cremer, D.; Kraka, E. Description of an Unusual Hydrogen Bond between Carborane and a Phenyl Group. J. Organomet. Chem. 2018, 865, 114–127. [Google Scholar] [CrossRef]
- Saha, B.; Sharma, H.; Bhattacharyya, P.K. Nonclassical B-Hb⋯π Interaction in Diborane⋯localized-π Sandwiches: A DFT-D3 Study. Int. J. Quantum. Chem. 2019, 119, e25998. [Google Scholar] [CrossRef]
- Fanfrlík, J.; Brynda, J.; Kugler, M.; Lepšik, M.; Pospísilová, K.; Holub, J.; Hnyk, D.; Nekvinda, J.; Gruner, B.; Rezacova, P. B-H⋯π and C-H⋯π Interactions in Protein-Ligand Complexes: Carbonic Anhydrase II Inhibition by Carborane Sulfonamides. Phys. Chem. Chem. Phys. 2023, 25, 1728. [Google Scholar] [CrossRef]
- Zhang, X.; Dai, H.; Yan, H.; Zou, W.; Cremer, D. B−H···π Interaction: A New Type of Nonclassical Hydrogen Bonding. J. Am. Chem. Soc. 2016, 138, 4334–4337. [Google Scholar] [CrossRef]
- Di Giosia, M.; Soldà, A.; Seeger, M.; Cantelli, A.; Arnesano, F.; Nardella, M.I.; Mangini, V.; Valle, F.; Montalti, M.; Zerbetto, F.; et al. A Bio-Conjugated Fullerene as a Subcellular-Targeted and Multifaceted Phototheranostic Agent. Adv. Funct. Mater. 2021, 31, 2101527. [Google Scholar] [CrossRef]
- Cantelli, A.; Malferrari, M.; Solda, A.; Simonetti, G.; Forni, S.; Toscanella, E.; Mattioli, E.J.; Zerbetto, F.; Zanelli, A.; Di Giosia, M.; et al. Human Serum Albumin−oligothiophene Bioconjugate: A Phototheranostic Platform for Localized Killing of Cancer Cells by Precise Light Activation. JACS Au 2021, 1, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Mei, K.; Guan, P.; Hu, X.; Zhao, Y.; Zhang, N.; Mei, K.; Guan, P.; Hu, X.; Zhao, Y.L. Protein-Based Artificial Nanosystems in Cancer Therapy. Small 2020, 16, 1907256. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.; Chen, H. Protein-Based Nanoplatforms for Tumor Imaging and Therapy. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1616. [Google Scholar] [CrossRef]
- Gou, Y.; Miao, D.; Zhou, M.; Wang, L.; Zhou, H.; Su, G. Bio-Inspired Protein-Based Nanoformulations for Cancer Theranostics. Front. Pharmacol. 2018, 9, 421. [Google Scholar] [CrossRef]
- Schaller, J.; Gerber, S.; Kämpfer, U.; Lejon, S.; Trachsel, C. Human Blood Plasma Proteins: Structure and Function; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2008; pp. 1–526. [Google Scholar] [CrossRef]
- Greco, G.; Ulfo, L.; Turrini, E.; Marconi, A.; Costantini, P.E.; Marforio, T.D.; Mattioli, E.J.; Di Giosia, M.; Danielli, A.; Fimognari, C.; et al. Light-Enhanced Cytotoxicity of Doxorubicin by Photoactivation. Cells 2023, 12, 392. [Google Scholar] [CrossRef]
- Shan, L.; Hao, Y.; Wang, S.; Korotcov, A.; Zhang, R.; Wang, T.; Califano, J.; Gu, X.; Sridhar, R.; Bhujwalla, Z.M.; et al. Visualizing Head and Neck Tumors in Vivo Using Near-Infrared Fluorescent Transferrin Conjugate. Mol. Imaging 2008, 7, 42–49. [Google Scholar] [CrossRef]
- Desai, N.; Trieu, V.; Damascelli, B.; Soon-Shiong, P. SPARC Expression Correlates with Tumor Response to Albumin-Bound Paclitaxel in Head and Neck Cancer Patients. Transl. Oncol. 2009, 2, 59–64. [Google Scholar] [CrossRef]
- Calabrese, G.; Daou, A.; Barbu, E.; Tsibouklis, J. Towards Carborane-Functionalised Structures for the Treatment of Brain Cancer. Drug Discov. Today 2018, 23, 63–75. [Google Scholar] [CrossRef]
- Berman, H.M.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.N.; Weissig, H.; Shindyalov, I.N.; Bourne, P.E. The Protein Data Bank. Nucleic Acids Res. 2000, 28, 235–242. [Google Scholar] [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly Accurate Protein Structure Prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef]
- Percy, A.J.; Chambers, A.G.; Smith, D.S.; Borchers, C.H. Standardized Protocols for Quality Control of MRM-Based Plasma Proteomic Workflows. J. Proteome Res. 2013, 12, 222–233. [Google Scholar] [CrossRef]
- Polanski, M.; Anderson, N.L. A List of Candidate Cancer Biomarkers for Targeted Proteomics. Biomark Insights 2006, 1. [Google Scholar] [CrossRef]
- Farrah, T.; Deutsch, E.W.; Omenn, G.S.; Campbell, D.S.; Sun, Z.; Bletz, J.A.; Mallick, P.; Katz, J.E.; Malmström, J.; Ossola, R.; et al. A High-Confidence Human Plasma Proteome Reference Set with Estimated Concentrations in PeptideAtlas. Mol. Cell. Proteom. 2011, 10, M110.006353. [Google Scholar] [CrossRef] [PubMed]
- Schneidman-Duhovny, D.; Inbar, Y.; Polak, V.; Shatsky, M.; Halperin, I.; Benyamini, H.; Barzilai, A.; Dror, O.; Haspel, N.; Nussinov, R.; et al. Taking Geometry to Its Edge: Fast Unbound Rigid (and Hinge-Bent) Docking. Proteins Struct. Funct. Genet. 2003, 52, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Calvaresi, M.; Zerbetto, F. Baiting Proteins with C60. ACS Nano 2010, 4, 2283–2299. [Google Scholar] [CrossRef]
- Calvaresi, M.; Zerbetto, F. Fullerene Sorting Proteins. Nanoscale 2011, 3, 2873–2881. [Google Scholar] [CrossRef]
- Ahmed, L.; Rasulev, B.; Kar, S.; Krupa, P.; Mozolewska, M.A.; Leszczynski, J. Inhibitors or Toxins? Large Library Target-Specific Screening of Fullerene-Based Nanoparticles for Drug Design Purpose. Nanoscale 2017, 9, 10263–10276. [Google Scholar] [CrossRef]
- di Giosia, M.; Valle, F.; Cantelli, A.; Bottoni, A.; Zerbetto, F.; Calvaresi, M. C60 Bioconjugation with Proteins: Towards a Palette of Carriers for All PH Ranges. Materials 2018, 11, 691. [Google Scholar] [CrossRef]
- Calvaresi, M.; Furini, S.; Domene, C.; Bottoni, A.; Zerbetto, F. Blocking the Passage: C60 Geometrically Clogs K+ Channels. ACS Nano 2015, 9, 4827–4834. [Google Scholar] [CrossRef] [PubMed]
- Calvaresi, M.; Arnesano, F.; Bonacchi, S.; Bottoni, A.; Calò, V.; Conte, S.; Falini, G.; Fermani, S.; Losacco, M.; Montalti, M.; et al. C60@Lysozyme: Direct Observation by Nuclear Magnetic Resonance of a 1:1 Fullerene Protein Adduct. ACS Nano 2014, 8, 1871–1877. [Google Scholar] [CrossRef] [PubMed]
- Calvaresi, M.; Bottoni, A.; Zerbetto, F. Thermodynamics of Binding between Proteins and Carbon Nanoparticles: The Case of C60@Lysozyme. J. Phys. Chem. C 2015, 119, 28077–28082. [Google Scholar] [CrossRef]
- Marforio, T.D.; Mattioli, E.J.; Zerbetto, F.; Calvaresi, M. Fullerenes against COVID-19: Repurposing C60 and C70 to Clog the Active Site of SARS-CoV-2 Protease. Molecules 2022, 27, 1916. [Google Scholar] [CrossRef] [PubMed]
- Malarz, K.; Korzuch, J.; Marforio, T.D.; Balin, K.; Calvaresi, M.; Mrozek-Wilczkiewicz, A.; Musiol, R.; Serda, M. Identification and Biological Evaluation of a Water-Soluble Fullerene Nanomaterial as BTK Kinase Inhibitor. Int. J. Nanomed. 2023, 18, 1709–1724. [Google Scholar] [CrossRef] [PubMed]
- Di Giosia, M.; Valle, F.; Cantelli, A.; Bottoni, A.; Zerbetto, F.; Fasoli, E.; Calvaresi, M. Identification and Preparation of Stable Water Dispersions of Protein—Carbon Nanotube Hybrids and Efficient Design of New Functional Materials. Carbon 2019, 147, 70–82. [Google Scholar] [CrossRef]
- Berto, M.; Di Giosia, M.; Giordani, M.; Sensi, M.; Valle, F.; Alessandrini, A.; Menozzi, C.; Cantelli, A.; Gazzadi, G.C.; Zerbetto, F.; et al. Green Fabrication of (6,5)Carbon Nanotube/Protein Transistor Endowed with Specific Recognition. Adv. Electron. Mater. 2021, 7, 2001114. [Google Scholar] [CrossRef]
- Di Giosia, M.; Marforio, T.D.; Cantelli, A.; Valle, F.; Zerbetto, F.; Su, Q.; Wang, H.; Calvaresi, M. Inhibition of α-Chymotrypsin by Pristine Single-Wall Carbon Nanotubes: Clogging up the Active Site. J. Colloid. Interface Sci. 2020, 571, 174–184. [Google Scholar] [CrossRef]
- Calvaresi, M.; Hoefinger, S.; Zerbetto, F. Probing the Structure of Lysozyme–Carbon-Nanotube Hybrids with Molecular Dynamics. Chem. A Eur. J. 2012, 18, 4308–4313. [Google Scholar] [CrossRef]
- Wang, J.; Wolf, R.M.; Caldwell, J.W.; Kollman, P.A.; Case, D.A. Development and Testing of a General Amber Force Field. J. Comput. Chem. 2004, 25, 1157–1174. [Google Scholar] [CrossRef]
- Sárosi, M.B.; Lybrand, T.P. Molecular Dynamics Simulation of Cyclooxygenase-2 Complexes with Indomethacin Closo-Carborane Analogs. J. Chem. Inf. Model. 2018, 58, 1990–1999. [Google Scholar] [CrossRef] [PubMed]
- Roe, D.R.; Cheatham, T.E. PTRAJ and CPPTRAJ: Software for Processing and Analysis of Molecular Dynamics Trajectory Data. J. Chem. Theory Comput. 2013, 9, 3084–3095. [Google Scholar] [CrossRef]
- Miller, B.R.; McGee, T.D.; Swails, J.M.; Homeyer, N.; Gohlke, H.; Roitberg, A.E. MMPBSA.Py: An Efficient Program for End-State Free Energy Calculations. J. Chem. Theory Comput. 2012, 8, 3314–3321. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, G.D.; Cramer, C.J.; Truhlar, D.G. Parametrized Models of Aqueous Free Energies of Solvation Based on Pairwise Descreening of Solute Atomic Charges from a Dielectric Medium. J. Phys. Chem. 1996, 100, 19824–19839. [Google Scholar] [CrossRef]
- Hawkins, G.D.; Cramer, C.J.; Truhlar, D.G. Pairwise Solute Descreening of Solute Charges from a Dielectric Medium. Chem. Phys. Lett. 1995, 246, 122–129. [Google Scholar] [CrossRef]
- Ol’shevskaya, V.A.; Nikitina, R.G.; Savchenko, A.N.; Malshakova, M.V.; Vinogradov, A.M.; Golovina, G.V.; Belykh, D.V.; Kutchin, A.V.; Kaplan, M.A.; Kalinin, V.N.; et al. Novel Boronated Chlorin E6-Based Photosensitizers: Synthesis, Binding to Albumin and Antitumour Efficacy. Bioorganic Med. Chem. 2009, 17, 1297–1306. [Google Scholar] [CrossRef]
- Goszczyński, T.M.; Fink, K.; Kowalski, K.; Leśnikowski, Z.J.; Boratyński, J. Interactions of Boron Clusters and Their Derivatives with Serum Albumin. Sci. Rep. 2017, 7, 9800. [Google Scholar] [CrossRef]
- Jena, B.B.; Satish, L.; Mahanta, C.S.; Swain, B.R.; Sahoo, H.; Dash, B.P.; Satapathy, R. Interaction of Carborane-Appended Trimer with Bovine Serum Albumin: A Spectroscopic Investigation. Inorg. Chim. Acta 2019, 491, 52–58. [Google Scholar] [CrossRef]
- Fuentes, I.; Pujols, J.; Viñas, C.; Ventura, S.; Teixidor, F. Dual Binding Mode of Metallacarborane Produces a Robust Shield on Proteins. Chem. A Eur. J. 2019, 25, 12820–12829. [Google Scholar] [CrossRef]
- Julius, R.L.; Farha, O.K.; Chiang, J.; Perry, L.J.; Hawthorne, M.F. Synthesis and Evaluation of Transthyretin Amyloidosis Inhibitors Containing Carborane Pharmacophores. Proc. Natl. Acad. Sci. USA 2007, 104, 4808–4813. [Google Scholar] [CrossRef]



| Protein | PDB/UNIPROT ID | Protein Concentration |
|---|---|---|
| Albumin | 1N5U | 42 mg/mL [74] |
| Afamin | 6FAK | 35 μg/mL [74] |
| α -fetoprotein | 7YIM | 20 ng/mL [75] |
| Cerulosplasmin | 4ENZ | 64.9 μg/mL [74] |
| Corticosteroid-binding globulin | 2VDX | 1.2 μg/mL [76] |
| Haptoglobulin | 4X0L | 1.1 mg/mL [74] |
| Hemoglobin | 5HY8 | 41 μg/mL [74] |
| Hemopexin | P02790 | 257 μg/mL [74] |
| Lactotransferrin | 1CB6 | 270 ng/mL [74] |
| Myoglobin | 3RGK | 210 ng/mL [76] |
| Plasma retinol-binding protein | 5NU7 | 32 μg/mL [74] |
| Serotransferrin | 6SOY | 1.5 mg/mL [74] |
| Sex hormone-binding globulin | 6PYF | 260 ng/mL [76] |
| Thyroxine-binding globulin | 2RIV | 1.3 μg/mL [76] |
| Transthyretin | 4QXV | 109.6 μg/mL [74] |
| Vitamin D-binding protein | 1KW2 | 62.36 μg/mL [74] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marforio, T.D.; Mattioli, E.J.; Zerbetto, F.; Calvaresi, M. Exploiting Blood Transport Proteins as Carborane Supramolecular Vehicles for Boron Neutron Capture Therapy. Nanomaterials 2023, 13, 1770. https://doi.org/10.3390/nano13111770
Marforio TD, Mattioli EJ, Zerbetto F, Calvaresi M. Exploiting Blood Transport Proteins as Carborane Supramolecular Vehicles for Boron Neutron Capture Therapy. Nanomaterials. 2023; 13(11):1770. https://doi.org/10.3390/nano13111770
Chicago/Turabian StyleMarforio, Tainah Dorina, Edoardo Jun Mattioli, Francesco Zerbetto, and Matteo Calvaresi. 2023. "Exploiting Blood Transport Proteins as Carborane Supramolecular Vehicles for Boron Neutron Capture Therapy" Nanomaterials 13, no. 11: 1770. https://doi.org/10.3390/nano13111770
APA StyleMarforio, T. D., Mattioli, E. J., Zerbetto, F., & Calvaresi, M. (2023). Exploiting Blood Transport Proteins as Carborane Supramolecular Vehicles for Boron Neutron Capture Therapy. Nanomaterials, 13(11), 1770. https://doi.org/10.3390/nano13111770








