Cancer Cytotoxicity of a Hybrid Hyaluronan-Superparamagnetic Iron Oxide Nanoparticle Material: An In-Vitro Evaluation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
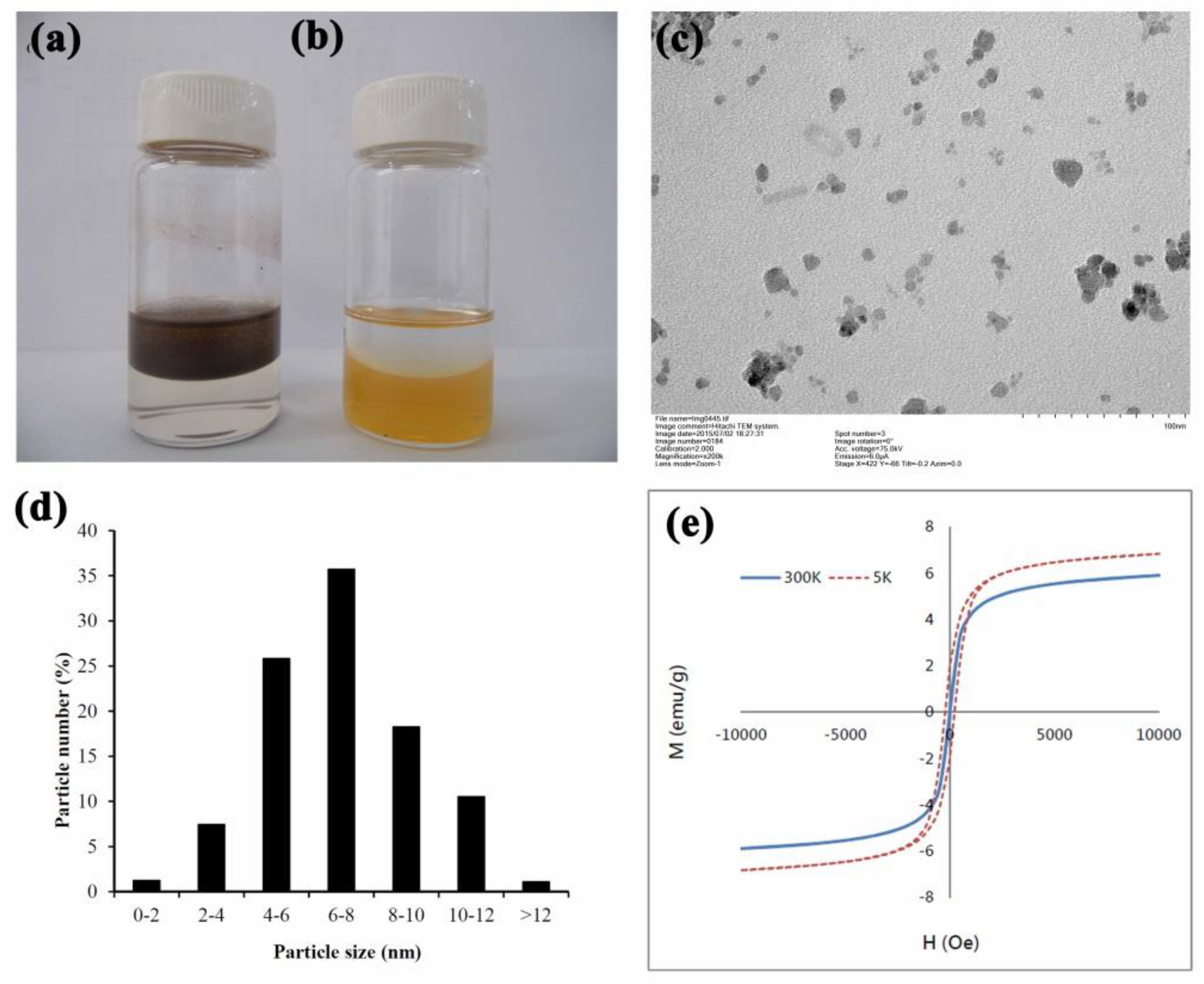
2.2. Preparation of Oleic-Acid Coated Fe3O4 Nanoparticles
2.3. Manufacture of LMWHA-SPIONs
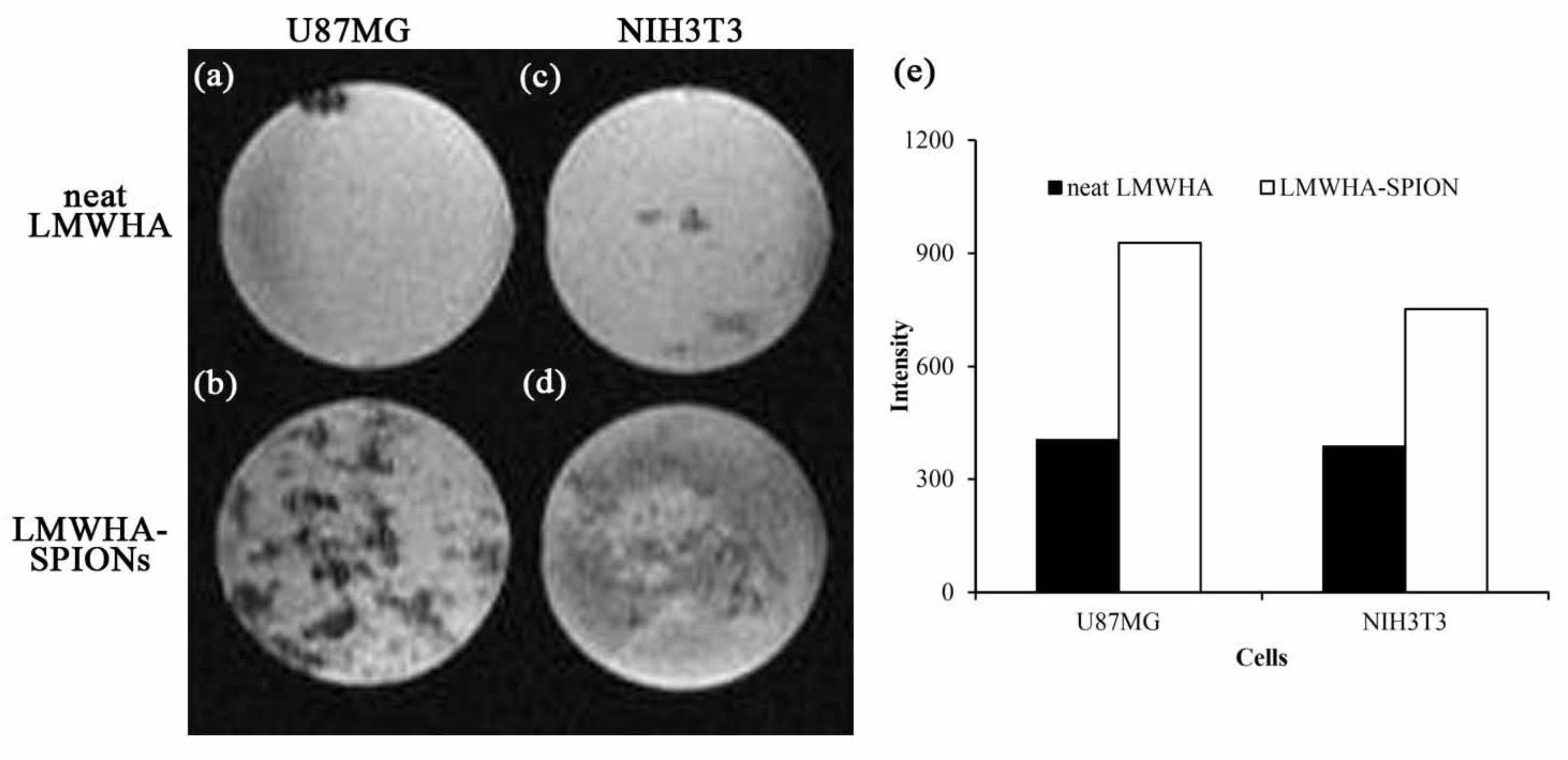
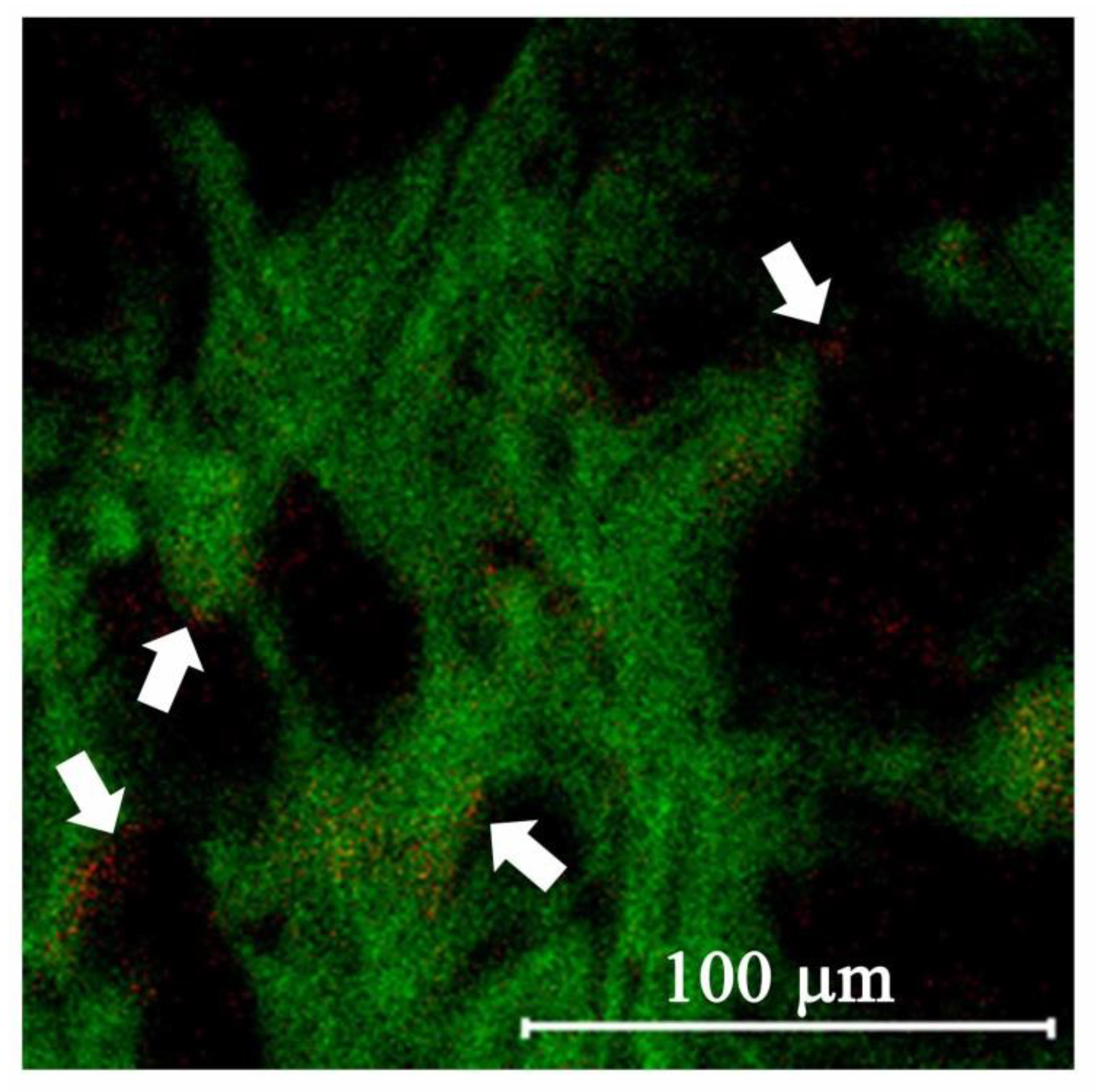
2.4. Targeting Ability of LMWHA-SPIONs
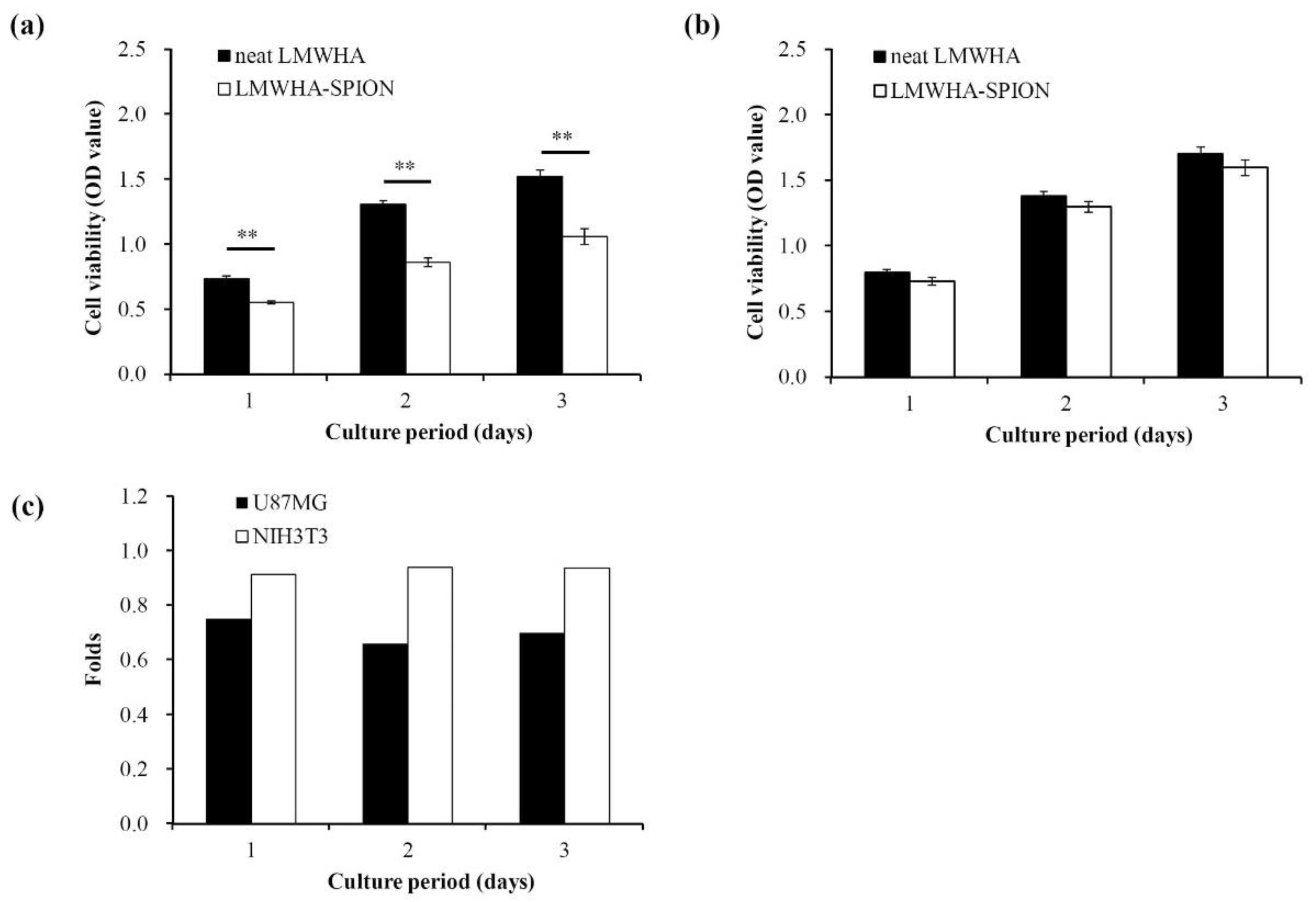
2.5. Viability of LMWHA-SPIONs Targeted Cells
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, K.; Choi, H.; Choi, E.S.; Park, M.H.; Ryu, J.H. Hyaluronic acid-coated nanomedicine for targeted cancer therapy. Pharmaceutics 2019, 11, 301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bayer, I.S. Hyaluronic acid and controlled release: A review. Molecules 2020, 25, 2649. [Google Scholar] [CrossRef] [PubMed]
- Trombino, S.; Servidio, C.; Curcio, F.; Cassano, R. Strategies for hyaluronic acid-based hydrogel design in drug delivery. Pharmaceutics 2019, 11, 407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toole, B.P. Hyaluronan-CD44 interactions in cancer: Paradoxes and possibilities. Clin. Cancer Res. 2009, 15, 7462–7468. [Google Scholar] [CrossRef] [Green Version]
- Kahmann, J.D.; O’Brien, R.; Werner, J.M.; Heinegard, D.; Ladbury, J.E.; Campbell, I.D.; Day, A.J. Localization and characterization of the hyaluronan-binding site on the link module from human TSG-6. Structure 2000, 8, 763–774. [Google Scholar] [CrossRef]
- Holmer, M.W.A.; Bayliss, M.T.; Muir, H. Hyaluronic acid in human articular cartilage: Age-related changes in content and size. Biochem. J. 1988, 250, 435–441. [Google Scholar] [CrossRef] [Green Version]
- Mattheolabakis, G.; Milane, L.; Singh, A.; Amiji, M.M. Hyaluronic acid targeting of CD44 for cancer therapy: From receptor biology to nanomedicine. J. Drug Target. 2015, 23, 605–618. [Google Scholar] [CrossRef]
- Pedrosa, P.; Vinhas, R.; Fernandes, A.; Baptista, P.V. Gold nanotheranostics: Proof-of-concept or clinical tool? Nanomaterials 2015, 5, 1853–1879. [Google Scholar] [CrossRef] [Green Version]
- Arpicco, S.; Milla, P.; Stella, B.; Dosio, F. Hyaluronic acid conjugates as vectors for the active targeting of drugs, genes and nanocomposites in cancer treatment. Molecules 2014, 19, 3193–3230. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Li, J.; Sun, W.; Hu, Y.; Zhang, G.; Shen, M.; Shi, X. Hyaluronic acid-modified magnetic iron oxide nanoparticles for MR imaging of surgically induced endometriosis model in rats. PLoS ONE 2014, 9, e94718. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; He, Y.; Sun, W.; Luo, Y.; Cai, H.; Pan, Y.; Shen, M.; Xia, J.; Shi, X. Hyaluronic acid-modified hydrothermally synthesized iron oxide nanoparticles for targeted tumor MR imaging. Biomaterials 2014, 35, 3666–3677. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.E.; Kim, A.Y.; Saravanakumar, G.; Koo, H.; Kwon, I.C.; Choi, K.; Park, J.H.; Kim, K. Hyaluronidase-sensitive SPIONs for MR/optical dual imaging nanoprobes. Macromol. Res. 2011, 19, 861–867. [Google Scholar] [CrossRef]
- Fu, C.; Duan, X.; Cao, M.; Jiang, S.; Ban, X.; Guo, N.; Zhang, F.; Mao, J.; Huyan, T.; Shen, J. Targeted magnetic resonance imaging and modulation of hypoxia with multifunctional hyaluronic acid-mno2 nanoparticles in glioma. Adv. Healthc. Mater. 2019, 8, e1900047. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.P.; Yang, R.M.; Wang, L.; Li, N.N.; Qi, M.; Xu, X.D.; Wei, X.H.; Jiang, X.Q.; Zhang, L.M. Surface functionalization of superparamagnetic nanoparticles by an acid-liable polysaccharidebased prodrug for combinatorial monitoring and chemotherapy of hepatocellular carcinoma. RSC Adv. 2017, 7, 41919–41928. [Google Scholar] [CrossRef] [Green Version]
- El-Dakdouki, M.H.; El-Boubbou, K.; Zhu, D.C.; Huang, X. A simple method for the synthesis of hyaluronic acid coated magnetic nanoparticles for highly efficient cell labelling and in vivo imaging. RSC Adv. 2011, 1, 1449–1452. [Google Scholar] [CrossRef] [Green Version]
- Smejkalová, D.; Nešporová, K.; Huerta-Angeles, G.; Syrovátka, J.; Jirák, D.; Gálisová, A.; Velebný, V. Selective in vitro anticancer effect of superparamagnetic iron oxide nanoparticles loaded in hyaluronan polymeric micelles. Biomacromolecules 2014, 15, 4012–4020. [Google Scholar] [CrossRef]
- Wang, H.T.; Chou, P.C.; Wu, P.H.; Lee, C.M.; Fan, K.H.; Chang, W.J.; Lee, S.Y.; Huang, H.M. Physical and biological evaluation of low-molecular-weight hyaluronic acid-modified Fe3O4 nanoparticle for targeting MCF7 breast cancer cells. Polymers 2020, 12, 1094. [Google Scholar] [CrossRef]
- Huang, H.M.; Wu, P.H.; Chou, P.C.; Hsiao, W.T.; Wang, H.T.; Chiang, H.P.; Lee, C.M.; Wang, S.H.; Hsiao, Y.C. Enhancement of T2* weighted MRI imaging sensitivity of U87MG glioblastoma cells. Int. J. Nanomed. 2021, 16, 3789–3802. [Google Scholar] [CrossRef]
- Dahiya, P.; Kamal, R. Hyaluronic acid: A boon in periodontal therapy. N. Am. J. Med. Sci. 2013, 5, 309–315. [Google Scholar] [CrossRef]
- Korn, P.; Schulz, M.C.; Hintze, V.; Range, U.; Mai, R.; Eckelt, U.; Schnabelrauch, M.; Möller, S.; Becher, J.; Scharnweber, D.; et al. Chondroitin sulfate and sulfated hyaluronan-containing collagen coatings of titanium implants influence peri-implant bone formation in a minipig model. J. Biomed. Mater. Res. A 2014, 102, 2334–2344. [Google Scholar] [CrossRef]
- Sieger, D.; Korzinskas, T.; Jung, O.; Stojanovic, S.; Wenisch, S.; Smeets, R.; Barbeck, M. The addition of high doses of hyaluronic acid to a biphasic bone substitute decreases the proinflammatory tissue response. Int. J. Mol. Sci. 2019, 20, 1969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allison, D.D.; Grande-Allen, K.J. Review. Hyaluronan: A powerful tissue engineering tool. Tissue Eng. 2006, 12, 2131–2140. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.K.; Ye, H.J.; Lin, C.Y.; Lai, H.Y.; Chen, C.H.; Wang, S.H.; Chang, W.J.; Lee, S.Y.; Huang, H.M. Estimation of the effect of accelerating new bone formation of high and low molecular weight hyaluronic acid hybrid: An animal study. Polymers 2021, 13, 1708. [Google Scholar] [CrossRef] [PubMed]
- Zhong, L.; Liu, Y.; Xu, L.; Li, Q.; Zhao, D.; Li, Z.; Zhang, H.; Zhang, H.; Kan, Q.; Sun, J.; et al. Exploring the relationship of hyaluronic acid molecular weight and active targeting efficiency for designing hyaluronic acid-modified nanoparticles. Asian J. Pharm. Sci. 2019, 14, 521–530. [Google Scholar] [CrossRef]
- Wang, H.T.; Chan, Y.H.; Feng, S.W.; Lo, Y.J.; Teng, N.C.; Huang, H.M. Development and biocompatibility tests of electrospun Poly-L-lactide nanofibrous membranes incorporating oleic acid coated-Fe3O4. J. Polym. Eng. 2014, 34, 241–245. [Google Scholar] [CrossRef]
- Huang, Y.C.; Huang, K.Y.; Lew, W.Z.; Fan, K.H.; Chang, W.J.; Huang, H.M. Gamma-irradiation-prepared low molecular weight hyaluronic acid promotes skin wound healing. Polymers 2019, 11, 1024. [Google Scholar] [CrossRef] [Green Version]
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic iron oxide nanoparticles (SPIONs): Development, surface modification and applications in chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46. [Google Scholar] [CrossRef] [Green Version]
- Willis, A.L.; Turro, N.J.; O’Brien, S. Spectroscopic characterization of the surface of iron oxide nanocrystals. Chem. Mater. 2005, 17, 5970–5975. [Google Scholar] [CrossRef]
- Zhang, L.; He, R.; Gu, H.C. Oleic acid coating on the monodisperse magnetite nanoparticles. Appl. Surf. Sci. 2006, 253, 2611–2617. [Google Scholar] [CrossRef]
- Cheng, F.Y.; Su, C.H.; Yang, Y.S.; Yeh, C.S.; Tsai, C.Y.; Wu, C.L.; Wu, M.T.; Shieh, D.B. Characterization of aqueous dispersions of Fe3O4 nanoparticles and their biomedical applications. Biomaterials 2005, 26, 729–738. [Google Scholar] [CrossRef]
- Ge, J.; Hu, Y.; Biasini, M.; Beyermann, W.P.; Yin, Y. Superparamagnetic magnetite colloidal nanocrystal clusters. Angew. Chem. Int. Ed. Engl. 2007, 46, 4342–4345. [Google Scholar] [CrossRef]
- Zhao, H.; Saatchi, K.; Häfeli, U.O. Preparation of biodegradable magnetic microspheres with poly(lactic acid)-coated magnetite. J. Magn. Magn. Mater. 2009, 321, 1356–1363. [Google Scholar] [CrossRef]
- Shukla, S.; Jadaun, A.; Arora, V.; Sinha, R.K.; Biyani, N.; Jain, V.K. In vitro toxicity assessment of chitosan oligosaccharidecoated iron oxide nanoparticles. Toxicol. Rep. 2015, 2, 27–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kokesch-Himmelreich, J.; Schumacher, M.; Rohnke, M.; Gelinsky, M.; Janek, J. ToF-SIMS analysis of osteoblast-like cells and their mineralized extracellular matrix on strontium enriched bone cements. Biointerphases 2013, 8, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pour, M.D.; Ren, L.; Jennische, E.; Lange, S.; Ewing, A.G.; Malmberg, P. Mass spectrometry imaging as a novel approach to measure hippocampal zinc. J. Anal. At. Spectrom. 2019, 34, 1581–1587. [Google Scholar] [CrossRef] [Green Version]
- Jungnickel, H.; Laux, P.; Luch, A. Time-of-flight secondary ion mass spectrometry (ToF-SIMS): A new tool for the analysis of toxicological effects on single cell level. Toxics 2016, 4, 5. [Google Scholar] [CrossRef]
- Brunelle, A.; Touboul, D.; Laprevote, O. Biological tissue imaging with time-of-flight secondary ion mass spectrometry and cluster ion sources. J. Mass Spectrom. 2005, 40, 985–999. [Google Scholar] [CrossRef]
- Fletcher, J.S. Cellular imaging with secondary ion mass spectrometry. Analyst 2009, 134, 2204–2215. [Google Scholar] [CrossRef]
- Sjövall, P.; Johansson, B.; Lausmaa, J. Localization of lipids in freeze-dried mouse brain sections by imaging TOF-SIMS. Appl. Surf. Sci. 2006, 252, 6966–6974. [Google Scholar] [CrossRef]
- Xiao, Y.D.; Paudel, R.; Liu, J.; Ma, C.; Zhang, Z.S.; Zhou, S.K. MRI contrast agents: Classification and application (Review). Int. J. Mol. Med. 2016, 38, 1319–1326. [Google Scholar] [CrossRef] [Green Version]
- Karimzadeh, I.; Aghazadeh, M.; Doroudi, T.; Ganjali, M.R.; Kolivand, P.H. Superparamagnetic iron oxide (Fe3O4) nanoparticles coated with peg/pei for biomedical applications: A facile and scalable preparation route based on the cathodic electrochemical deposition method. Adv. Phys. Chem. 2017, 2017, 9437487. [Google Scholar] [CrossRef] [Green Version]
- Qhattal, H.S.; Liu, X. Characterization of CD44-mediated cancer cell uptake and intracellular distribution of hyaluronan-grafted liposomes. Mol. Pharm. 2011, 8, 1233–1246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sieber, M.A.; Steger-Hartmann, T.; Lengsfeld, P.; Pietsch, H. Gadolinium-based contrast agents and NSF: Evidence from animal experience. J. Magn. Reson. Imaging 2009, 30, 1268–1276. [Google Scholar] [CrossRef] [PubMed]
- Ganau, M.; Syrmos, N.C.; D’Arco, F.; Ganau, L.; Chibbaro, S.; Prisco, L.; Ligarotti, G.K.I.; Ambu, R.; Soddu, A. Εnhancing contrast agents and radiotracers performance through hyaluronic acid-coating in neuroradiology and nuclear medicine. Hell. J. Nucl. Med. 2017, 20, 166–168. [Google Scholar]
- Rutjes, A.W.; Jüni, P.; Costa, B.R.D.; Trelle, S.; Nüesch, E.; Reichenbach, S. Viscosupplementation for osteoarthritis of the knee: A systematic review and meta-analysis. Ann. Intern. Med. 2012, 157, 180–191. [Google Scholar] [CrossRef]
- Kiessling, M.K.; Klemke, C.D.; Kaminski, M.M.; Galani, I.E.; Krammer, P.H.; Gulow, K. Inhibition of constitutively activated nuclear factor-kappaB induces reactive oxygen species- and irondependent cell death in cutaneous T-cell lymphoma. Cancer Res. 2009, 69, 2365–2374. [Google Scholar] [CrossRef] [Green Version]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, Y.-L.; Liao, P.-B.; Wu, P.-H.; Chang, W.-J.; Lee, S.-Y.; Huang, H.-M. Cancer Cytotoxicity of a Hybrid Hyaluronan-Superparamagnetic Iron Oxide Nanoparticle Material: An In-Vitro Evaluation. Nanomaterials 2022, 12, 496. https://doi.org/10.3390/nano12030496
Chang Y-L, Liao P-B, Wu P-H, Chang W-J, Lee S-Y, Huang H-M. Cancer Cytotoxicity of a Hybrid Hyaluronan-Superparamagnetic Iron Oxide Nanoparticle Material: An In-Vitro Evaluation. Nanomaterials. 2022; 12(3):496. https://doi.org/10.3390/nano12030496
Chicago/Turabian StyleChang, Yen-Lan, Pei-Bang Liao, Ping-Han Wu, Wei-Jen Chang, Sheng-Yang Lee, and Haw-Ming Huang. 2022. "Cancer Cytotoxicity of a Hybrid Hyaluronan-Superparamagnetic Iron Oxide Nanoparticle Material: An In-Vitro Evaluation" Nanomaterials 12, no. 3: 496. https://doi.org/10.3390/nano12030496
APA StyleChang, Y.-L., Liao, P.-B., Wu, P.-H., Chang, W.-J., Lee, S.-Y., & Huang, H.-M. (2022). Cancer Cytotoxicity of a Hybrid Hyaluronan-Superparamagnetic Iron Oxide Nanoparticle Material: An In-Vitro Evaluation. Nanomaterials, 12(3), 496. https://doi.org/10.3390/nano12030496







