Near-Infrared Emission of HgTe Nanoplatelets Tuned by Pb-Doping
Abstract
1. Introduction
2. Materials and Methods
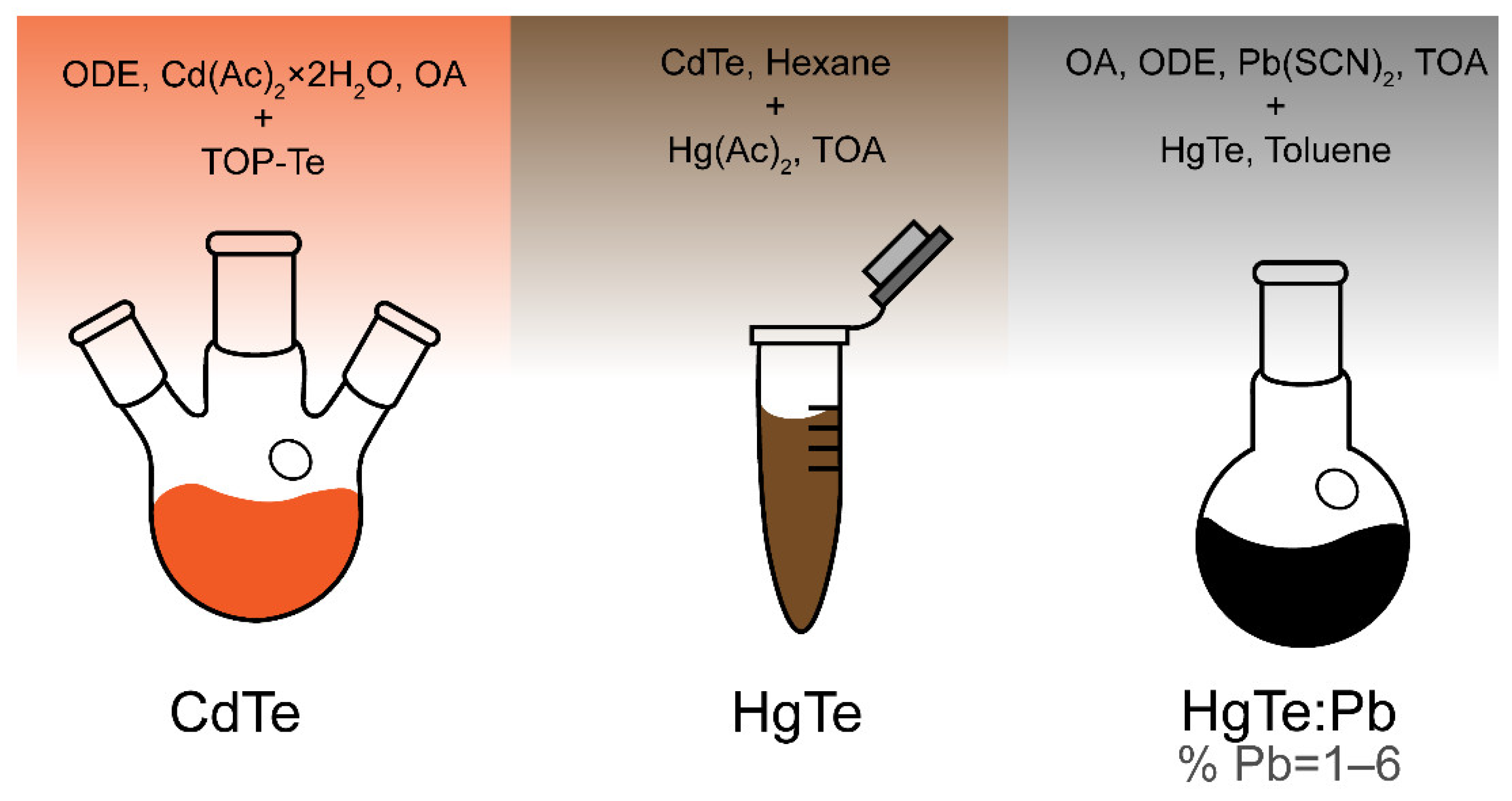
2.1. Synthesis of HgTe:Pb Nanoplatelets
2.2. Characterization
3. Results and Discussion
3.1. The Influence of Pb Doping on the Morphology of HgTe Nanoplatelets
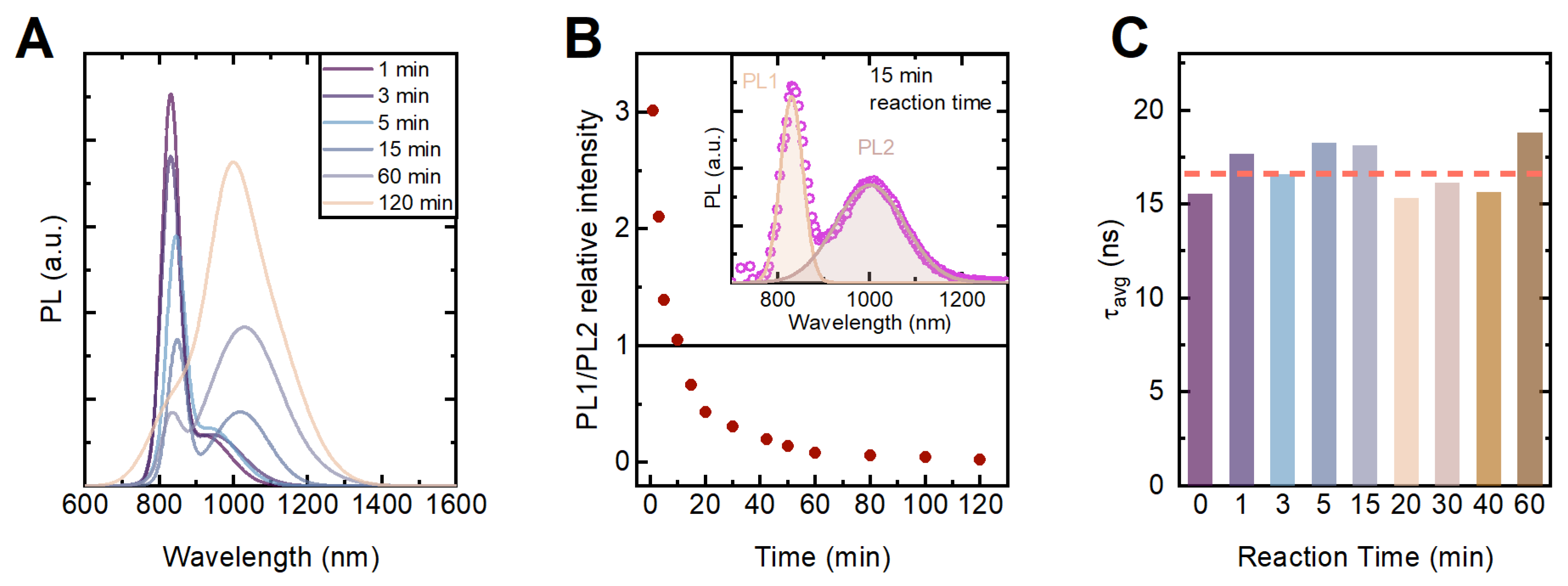
3.2. Spectral and Temporal Emission Parameters of the HgTe:Pb Nanoplatelets
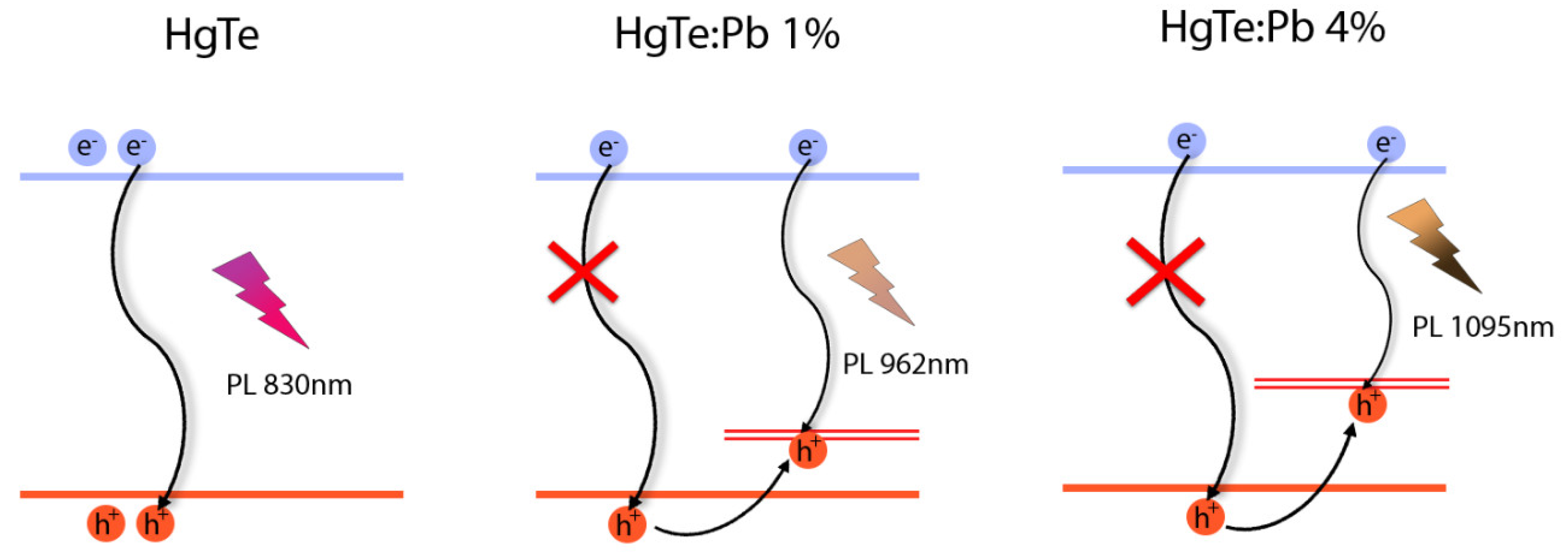
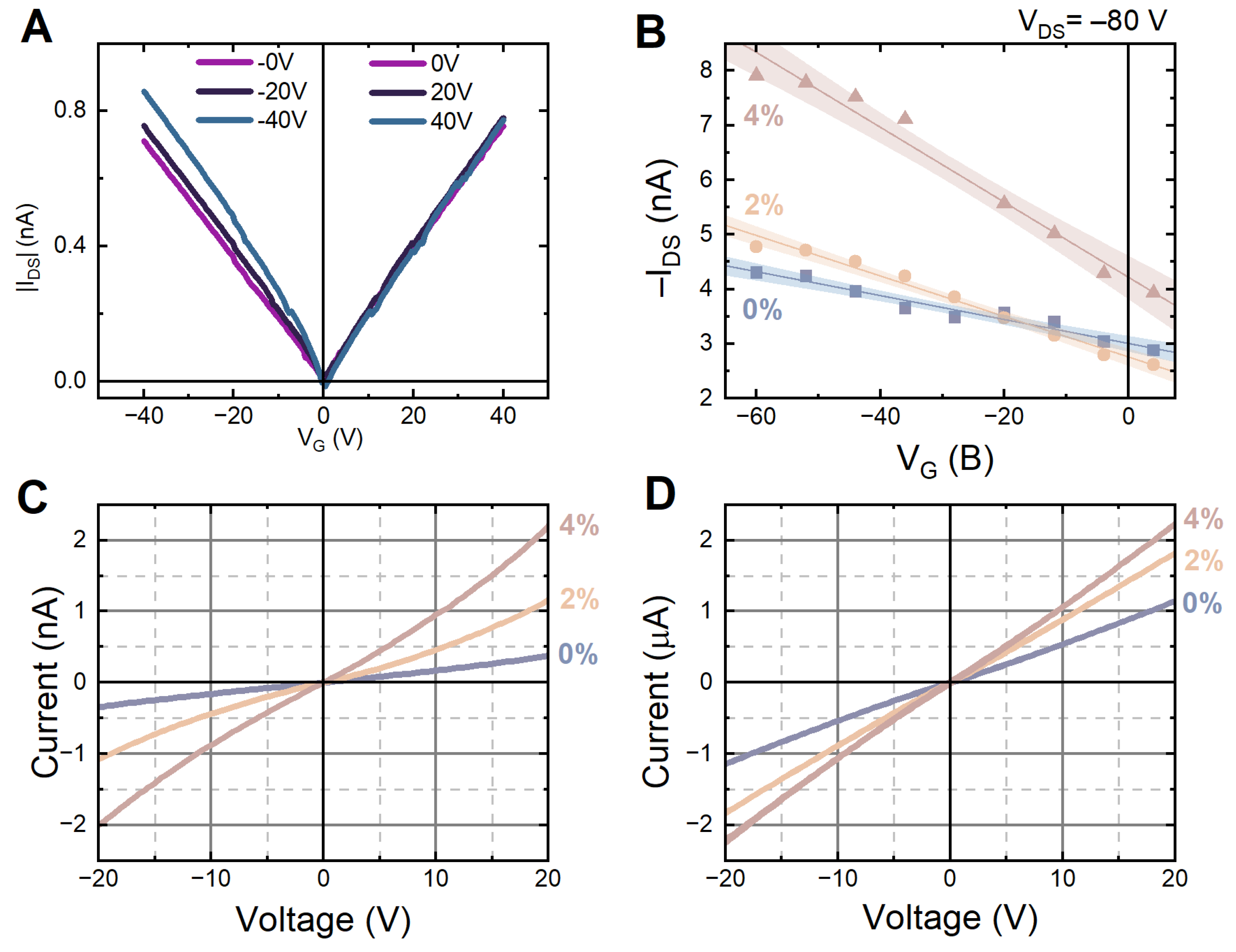
3.3. Electronic Properties of HgTe:Pb Nanoplatelets
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yao, J.; Li, P.; Li, L.; Yang, M. Biochemistry and Biomedicine of Quantum Dots: From Biodetection to Bioimaging, Drug Discovery, Diagnostics, and Therapy. Acta Biomater. 2018, 74, 36–55. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Karim, M.R.; Alharbi, H.F.; Aldokhayel, B.; Uzzaman, T.; Zahir, H. Cadmium Selenide Quantum Dots for Solar Cell Applications: A Review. Chem. Asian J. 2021, 16, 902–921. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Chen, R. Optical Properties and Applications of Two-Dimensional CdSe Nanoplatelets. InfoMat 2020, 2, 905–927. [Google Scholar] [CrossRef]
- Meerbach, C.; Tietze, R.; Voigt, S.; Sayevich, V.; Dzhagan, V.M.; Erwin, S.C.; Dang, Z.; Selyshchev, O.; Schneider, K.; Zahn, D.R.T.; et al. Brightly Luminescent Core/Shell Nanoplatelets with Continuously Tunable Optical Properties. Adv. Opt. Mater. 2019, 7, 1801478. [Google Scholar] [CrossRef]
- Ithurria, S.; Tessier, M.D.; Mahler, B.; Lobo, R.P.S.M.; Dubertret, B.; Efros, A.L. Colloidal Nanoplatelets with Two-Dimensional Electronic Structure. Nat. Mater. 2011, 10, 936–941. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, Y.; Yu, W.W. Near Infrared Emitting Quantum Dots: Synthesis, Luminescence Properties and Applications. J. Mater. Chem. C 2019, 7, 13662–13679. [Google Scholar] [CrossRef]
- Lesnyak, V.; George, C.; Genovese, A.; Prato, M.; Casu, A.; Ayyappan, S.; Scarpellini, A.; Manna, L. Alloyed Copper Chalcogenide Nanoplatelets via Partial Cation Exchange Reactions. ACS Nano 2014, 8, 8407–8418. [Google Scholar] [CrossRef] [PubMed]
- Skurlov, I.; Sokolova, A.; Galle, T.; Cherevkov, S.; Ushakova, E.; Baranov, A.; Lesnyak, V.; Fedorov, A.; Litvin, A. Temperature-Dependent Photoluminescent Properties of Pbse Nanoplatelets. Nanomaterials 2020, 10, 2570. [Google Scholar] [CrossRef]
- Saran, R.; Curry, R.J. Lead Sulphide Nanocrystal Photodetector Technologies. Nat. Photonics 2016, 10, 81–92. [Google Scholar] [CrossRef]
- Bouet, C.; Laufer, D.; Mahler, B.; Nadal, B.; Heuclin, H.; Pedetti, S.; Patriarche, G.; Dubertret, B. Synthesis of Zinc and Lead Chalcogenide Core and Core/Shell Nanoplatelets Using Sequential Cation Exchange Reactions. Chem. Mater. 2014, 26, 3002–3008. [Google Scholar] [CrossRef]
- Prudnikau, A.; Artemyev, M.; Molinari, M.; Troyon, M.; Sukhanova, A.; Nabiev, I.; Baranov, A.V.; Cherevkov, S.A.; Fedorov, A.V. Chemical Substitution of Cd Ions by Hg in CdSe Nanorods and Nanodots: Spectroscopic and Structural Examination. Mater. Sci. Eng. B 2012, 177, 744–749. [Google Scholar] [CrossRef]
- Dufour, M.; Izquierdo, E.; Livache, C.; Martinez, B.; Silly, M.G.; Pons, T.; Lhuillier, E.; Delerue, C.; Ithurria, S. Doping as a Strategy to Tune Color of 2D Colloidal Nanoplatelets. ACS Appl. Mater. Interfaces 2019, 11, 10128–10134. [Google Scholar] [CrossRef]
- Knowles, K.E.; Nelson, H.D.; Kilburn, T.B.; Gamelin, D.R. Singlet-Triplet Splittings in the Luminescent Excited States of Colloidal Cu+:CdSe, Cu+:InP, and CuInS2 Nanocrystals: Charge-Transfer Configurations and Self-Trapped Excitons. J. Am. Chem. Soc. 2015, 137, 13138–13147. [Google Scholar] [CrossRef] [PubMed]
- Galle, T.; Samadi Khoshkhoo, M.; Martin-Garcia, B.; Meerbach, C.; Sayevich, V.; Koitzsch, A.; Lesnyak, V.; Eychmüller, A. Colloidal PbSe Nanoplatelets of Varied Thickness with Tunable Optical Properties. Chem. Mater. 2019, 31, 3803–3811. [Google Scholar] [CrossRef]
- Izquierdo, E.; Robin, A.; Keuleyan, S.; Lequeux, N.; Lhuillier, E.; Ithurria, S. Strongly Confined HgTe 2D Nanoplatelets as Narrow Near-Infrared Emitters. J. Am. Chem. Soc. 2016, 138, 10496–10501. [Google Scholar] [CrossRef] [PubMed]
- Parfenov, P.S.; Litvin, A.P.; Ushakova, E.V.; Fedorov, A.V.; Baranov, A.V.; Berwick, K. Note: Near Infrared Spectral and Transient Measurements of PbS Quantum Dots Luminescence. Rev. Sci. Instrum. 2013, 84, 116104. [Google Scholar] [CrossRef] [PubMed]
- Parfenov, P.S.; Litvin, A.P.; Baranov, A.V.; Veniaminov, A.V.; Ushakova, E.V. Calibration of the Spectral Sensitivity of Instruments for the near Infrared Region. J. Appl. Spectrosc. 2011, 78, 433–439. [Google Scholar] [CrossRef]
- Skurlov, I.D.; Onishchuk, D.A.; Parfenov, P.S.; Litvin, A.P. An Experimental Setup for Analysis of Weak Photoluminescence in the Near-Infrared Spectral Region. Opt. Spectrosc. 2018, 125, 756–759. [Google Scholar] [CrossRef]
- Martinet, Q.; Baronnier, J.; Girard, A.; Albaret, T.; Saviot, L.; Mermet, A.; Abecassis, B.; Margueritat, J.; Mahler, B. Ligand-Dependent Nano-Mechanical Properties of CdSe Nanoplatelets: Calibrating Nanobalances for Ligand Affinity Monitoring. Nanoscale 2021, 13, 8639–8647. [Google Scholar] [CrossRef]
- Dufour, M.; Qu, J.; Greboval, C.; Méthivier, C.; Lhuillier, E.; Ithurria, S. Halide Ligands to Release Strain in Cadmium Chalcogenide Nanoplatelets and Achieve High Brightness. ACS Nano 2019, 13, 5326–5334. [Google Scholar] [CrossRef] [PubMed]
- Livache, C.; Izquierdo, E.; Martinez, B.; Dufour, M.; Pierucci, D.; Keuleyan, S.; Cruguel, H.; Becerra, L.; Fave, J.L.; Aubin, H.; et al. Charge Dynamics and Optolectronic Properties in HgTe Colloidal Quantum Wells. Nano Lett. 2017, 17, 4067–4074. [Google Scholar] [CrossRef] [PubMed]
- Chu, A.; Martinez, B.; Ferré, S.; Noguier, V.; Gréboval, C.; Livache, C.; Qu, J.; Prado, Y.; Casaretto, N.; Goubet, N.; et al. HgTe Nanocrystals for SWIR Detection and Their Integration up to the Focal Plane Array. ACS Appl. Mater. Interfaces 2019, 11, 33116–33123. [Google Scholar] [CrossRef]
- Martinez, B.; Livache, C.; Goubet, N.; Jagtap, A.; Cruguel, H.; Ouerghi, A.; Lacaze, E.; Silly, M.G.; Lhuillier, E. Probing Charge Carrier Dynamics to Unveil the Role of Surface Ligands in HgTe Narrow Band Gap Nanocrystals. J. Phys. Chem. C 2018, 122, 859–865. [Google Scholar] [CrossRef]
- Khan, A.H.; Pinchetti, V.; Tanghe, I.; Dang, Z.; Martín-García, B.; Hens, Z.; Van Thourhout, D.; Geiregat, P.; Brovelli, S.; Moreels, I. Tunable and Efficient Red to Near-Infrared Photoluminescence by Synergistic Exploitation of Core and Surface Silver Doping of CdSe Nanoplatelets. Chem. Mater. 2019, 31, 1450–1459. [Google Scholar] [CrossRef]
- Wei, R.; Tian, X.; Luo, H.; Liu, M.; Yang, Z.; Luo, Z.; Zhu, H.; Guo, H.; Li, J.; Qiu, J. Heavily Doped Semiconductor Colloidal Nanocrystals as Ultra-Broadband Switches for Near-Infrared and Mid-Infrared Pulse Lasers. ACS Appl. Mater. Interfaces 2019, 11, 40416–40423. [Google Scholar] [CrossRef]
- Sharma, A.; Sharma, M.; Gungor, K.; Olutas, M.; Dede, D.; Demir, H.V. Near-Infrared-Emitting Five-Monolayer Thick Copper-Doped CdSe Nanoplatelets. Adv. Opt. Mater. 2019, 7, 1900831. [Google Scholar] [CrossRef]
- Sharma, M.; Olutas, M.; Yeltik, A.; Kelestemur, Y.; Sharma, A.; Delikanli, S.; Guzelturk, B.; Gungor, K.; McBride, J.R.; Demir, H.V. Understanding the Journey of Dopant Copper Ions in Atomically Flat Colloidal Nanocrystals of CdSe Nanoplatelets Using Partial Cation Exchange Reactions. Chem. Mater. 2018, 30, 3265–3275. [Google Scholar] [CrossRef]
- Lakowicz, J.R. (Ed.) Principles of Fluorescence Spectroscopy; Springer: Boston, MA, USA, 2006; ISBN 978-0-387-31278-1. [Google Scholar]
- Podzorov, V. Organic Single Crystals: Addressing the Fundamentals of Organic Electronics. MRS Bull. 2013, 38, 15–24. [Google Scholar] [CrossRef]
- Parfenov, P.S.; Bukhryakov, N.V.; Onishchuk, D.A.; Babaev, A.A.; Sokolova, A.V.; Litvin, A.P. Study of charge carrier mobility in PBS nanocrystal layers using field-effect transistors. Semiconductors 2022, 56, 175. [Google Scholar] [CrossRef]
- Zhang, S.; Hu, Y.; Hao, Q. Advances of Sensitive Infrared Detectors with HgTe Colloidal Quantum Dots. Coatings 2020, 10, 760. [Google Scholar] [CrossRef]
- Maulu, A.; Navarro-Arenas, J.; Rodríguez-Cantó, P.J.; Sánchez-Royo, J.F.; Abargues, R.; Suárez, I.; Martínez-Pastor, J.P. Charge transport in trap-sensitized infrared PbS quantum-dot-based photoconductors: Pros and cons. Nanomaterials 2018, 8, 677. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Buhro, W.E. Contrasting Ligand-Exchange Behavior of Wurtzite and Zinc-Blende Cadmium Telluride Nanoplatelets. Chem. Mater. 2021, 33, 1683–1697. [Google Scholar] [CrossRef]


| Pb/Hg Ratio | Thickness, nm | ID, A | IL, A | Photosensitivity | RL/RD | Resistivity pD, Om·m | μh, cm2/(V·s) |
|---|---|---|---|---|---|---|---|
| 0% | 340 | 3.6 × 10−10 | 1.1 × 10−6 | 0.9997 | 3060 | 11.3 × 106 | 7.0 × 10−7 |
| 2% | 590 | 1.2 × 10−9 | 1.8 × 10−6 | 0.9993 | 1500 | 5.9 × 106 | 1.5 × 10−6 |
| 4% | 435 | 2.2 × 10−9 | 2.2 × 10−6 | 0.9990 | 1000 | 2.4 × 106 | 3.8 × 10−6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sokolova, A.V.; Skurlov, I.D.; Babaev, A.A.; Perfenov, P.S.; Miropoltsev, M.A.; Danilov, D.V.; Baranov, M.A.; Kolesnikov, I.E.; Koroleva, A.V.; Zhizhin, E.V.; et al. Near-Infrared Emission of HgTe Nanoplatelets Tuned by Pb-Doping. Nanomaterials 2022, 12, 4198. https://doi.org/10.3390/nano12234198
Sokolova AV, Skurlov ID, Babaev AA, Perfenov PS, Miropoltsev MA, Danilov DV, Baranov MA, Kolesnikov IE, Koroleva AV, Zhizhin EV, et al. Near-Infrared Emission of HgTe Nanoplatelets Tuned by Pb-Doping. Nanomaterials. 2022; 12(23):4198. https://doi.org/10.3390/nano12234198
Chicago/Turabian StyleSokolova, Anastasiia V., Ivan D. Skurlov, Anton A. Babaev, Peter S. Perfenov, Maksim A. Miropoltsev, Denis V. Danilov, Mikhail A. Baranov, Ilya E. Kolesnikov, Aleksandra V. Koroleva, Evgeniy V. Zhizhin, and et al. 2022. "Near-Infrared Emission of HgTe Nanoplatelets Tuned by Pb-Doping" Nanomaterials 12, no. 23: 4198. https://doi.org/10.3390/nano12234198
APA StyleSokolova, A. V., Skurlov, I. D., Babaev, A. A., Perfenov, P. S., Miropoltsev, M. A., Danilov, D. V., Baranov, M. A., Kolesnikov, I. E., Koroleva, A. V., Zhizhin, E. V., Litvin, A. P., Fedorov, A. V., & Cherevkov, S. A. (2022). Near-Infrared Emission of HgTe Nanoplatelets Tuned by Pb-Doping. Nanomaterials, 12(23), 4198. https://doi.org/10.3390/nano12234198








