Development of New Natural Lipid-Based Nanoparticles Loaded with Aluminum-Phthalocyanine for Photodynamic Therapy against Melanoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Pre-Formulation Studies
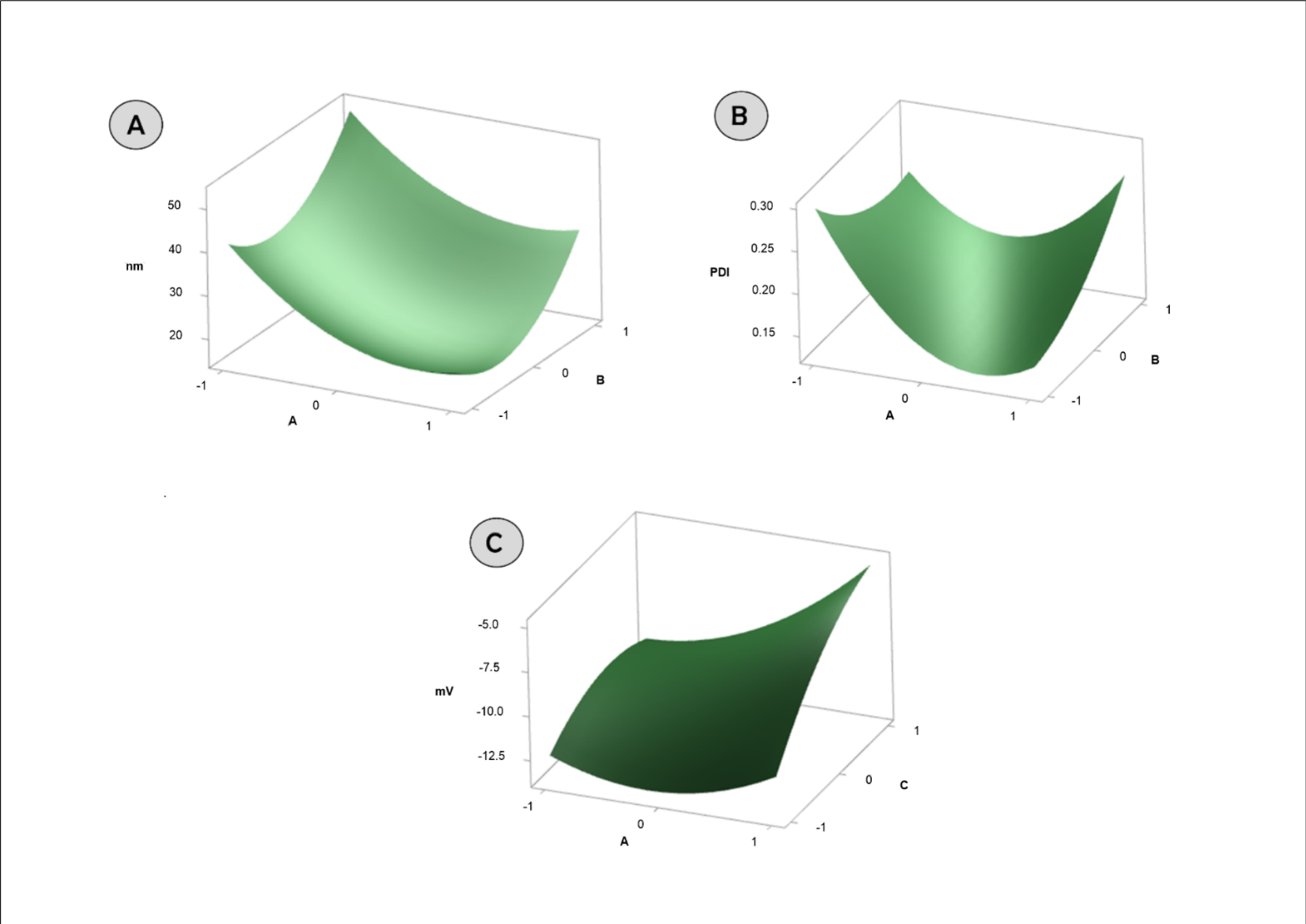
2.2. Box–Behnken
2.3. Evaluation of Encapsulation Efficiency (EE%)
2.4. Differential Scanning Calorimetry
2.5. Evaluation of Colloidal Properties
2.6. Analytical Methodology
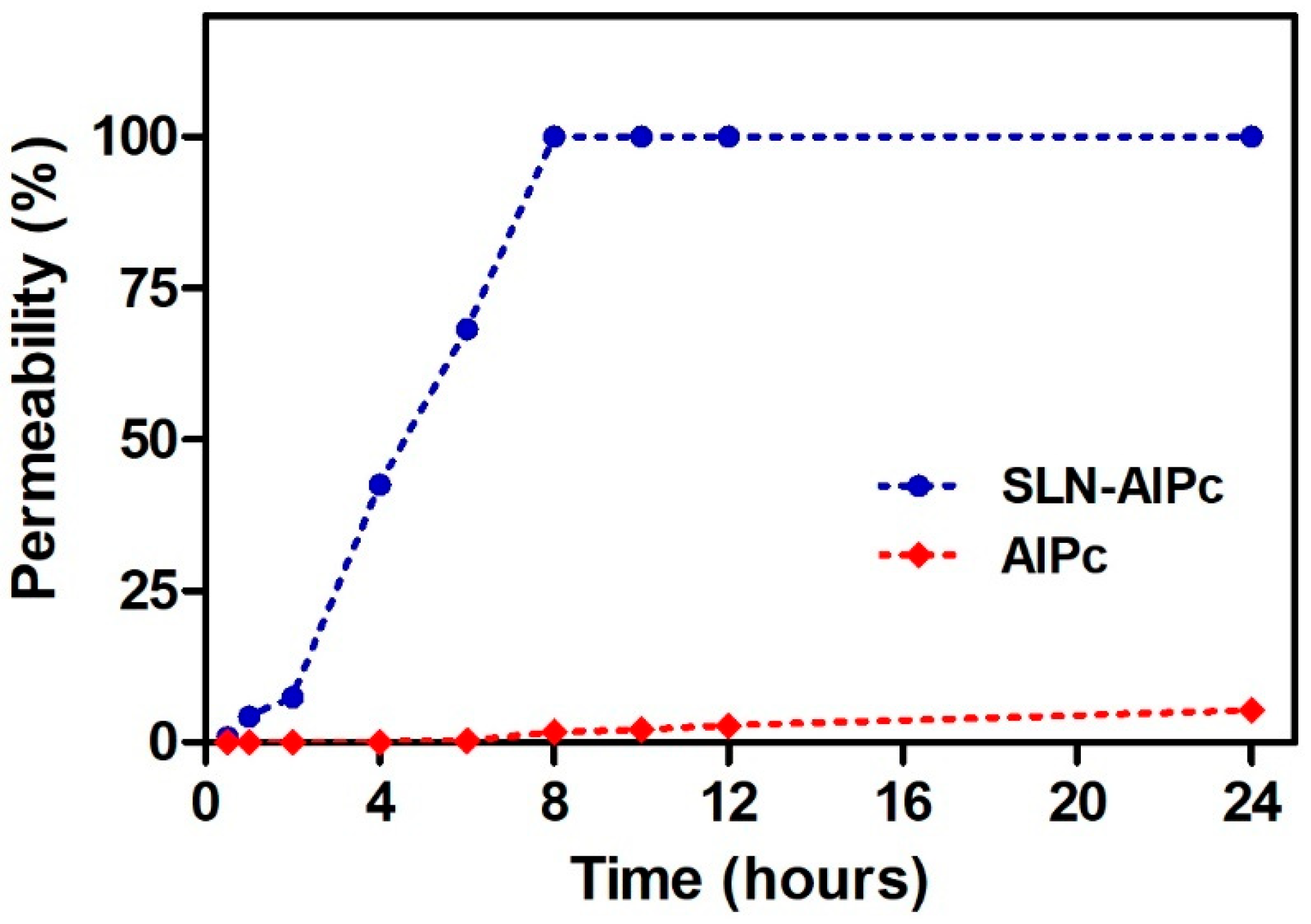
2.7. In Vitro Skin Permeation
2.8. Morphology of SLNs
2.9. Fourier-Transform InfraRed-FTIR
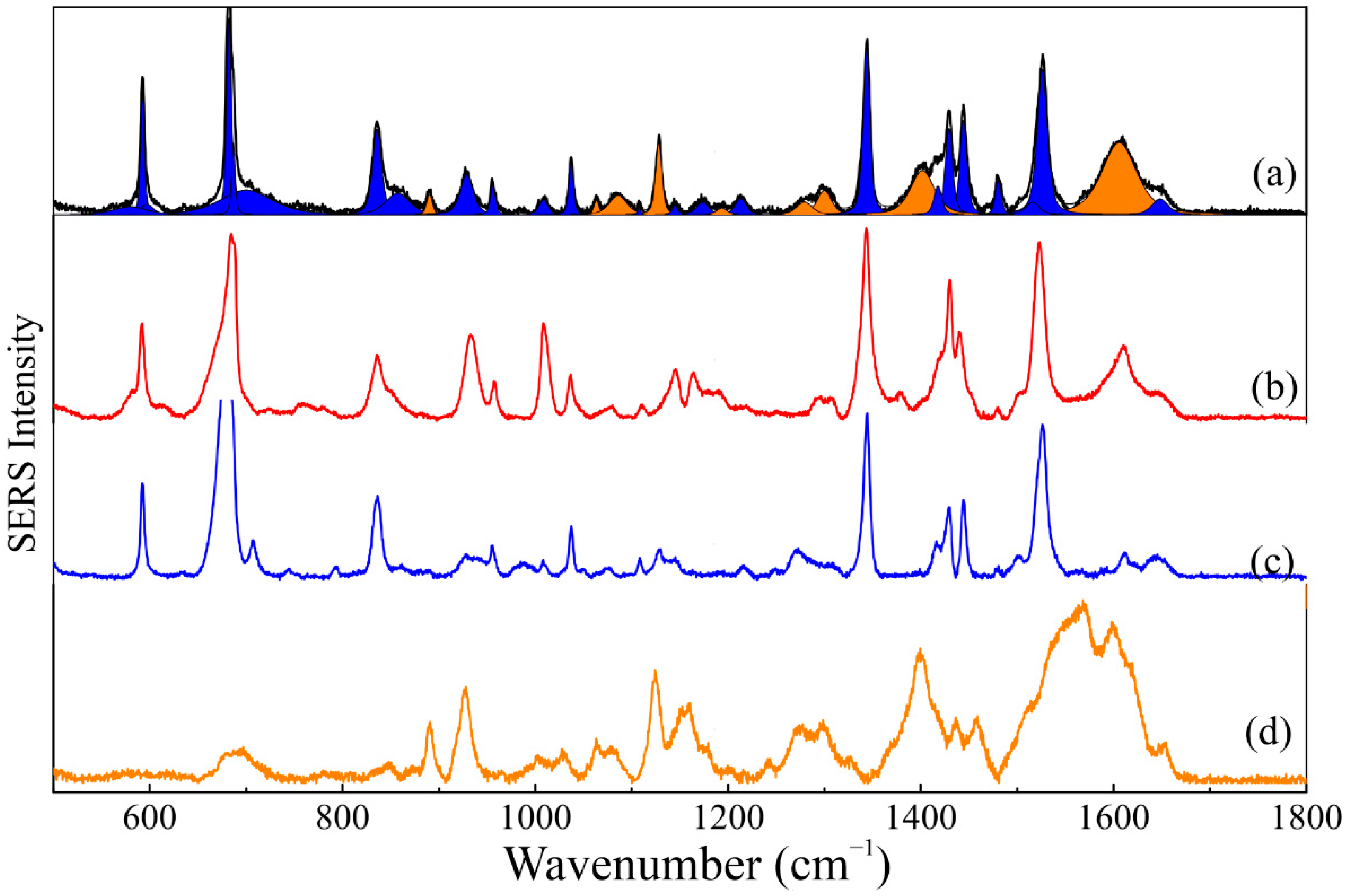
2.10. Raman Spectroscopy
2.11. Surface-Enhanced Raman Spectroscopy-SERS
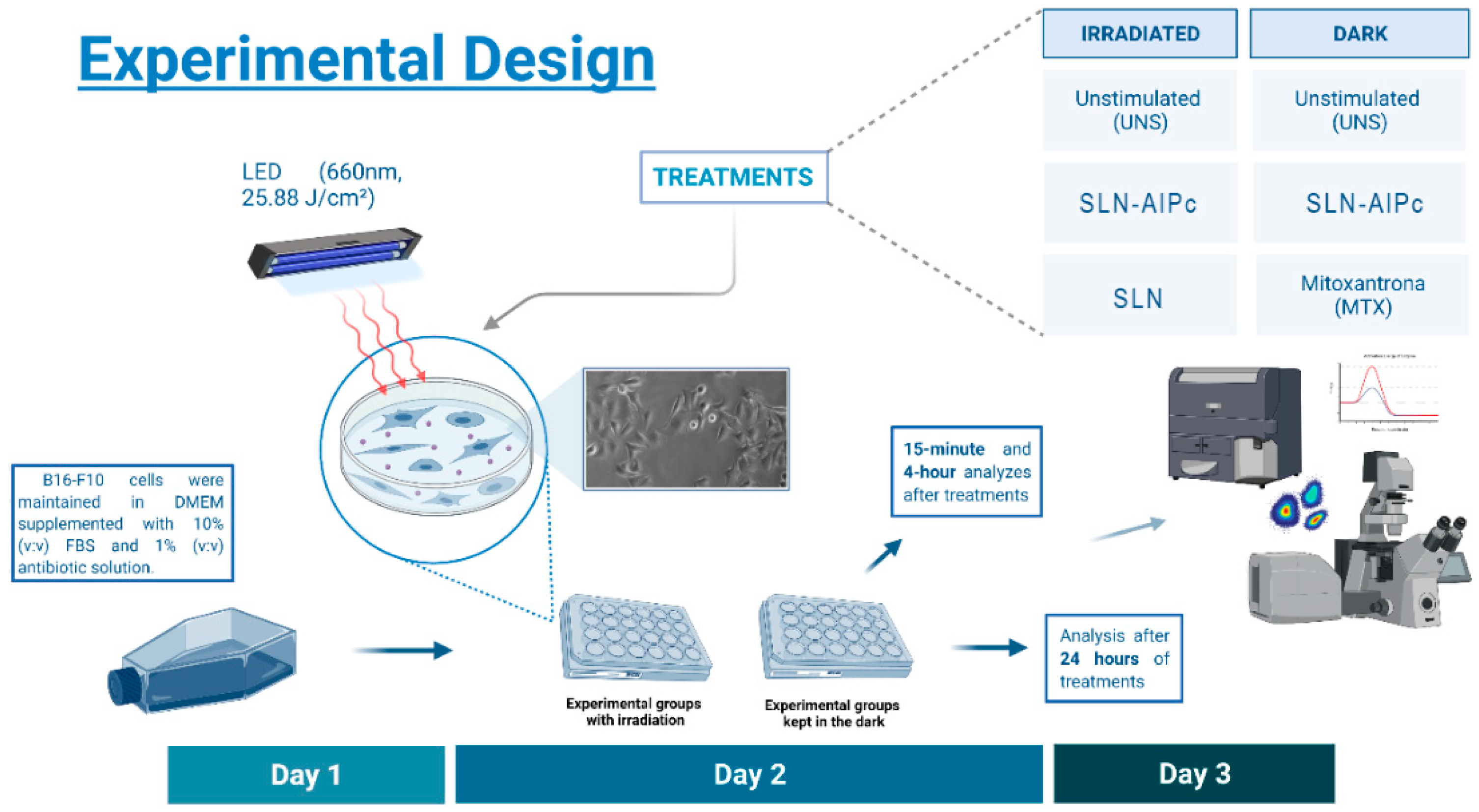
2.12. In Vitro Experimental Design
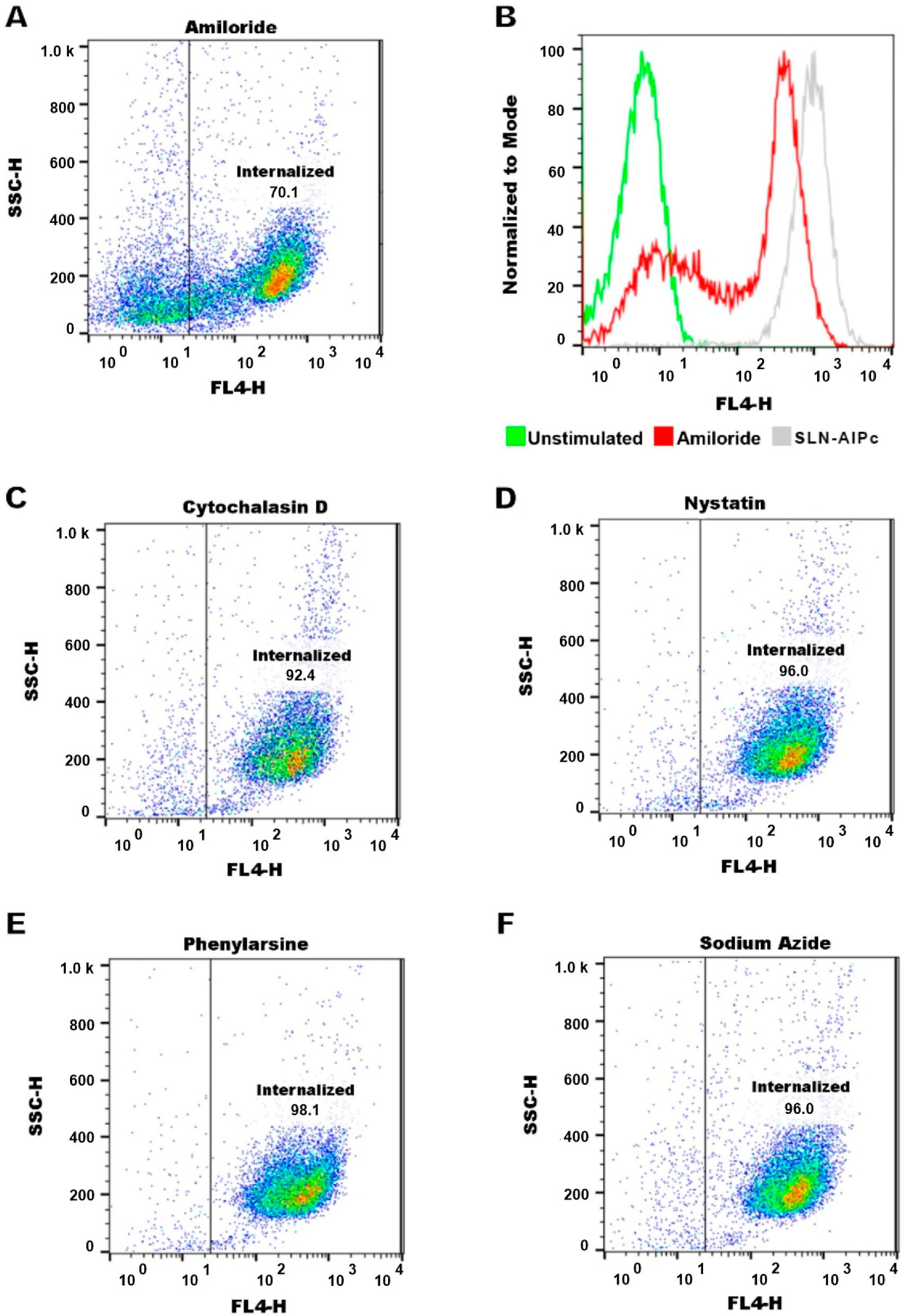
2.12.1. SLN-AlPc Internalization Assay and Endocytosis Pathway Analysis
2.12.2. Cell Viability and Cell Death Assays
2.12.3. B16-F10 Cell Morphology
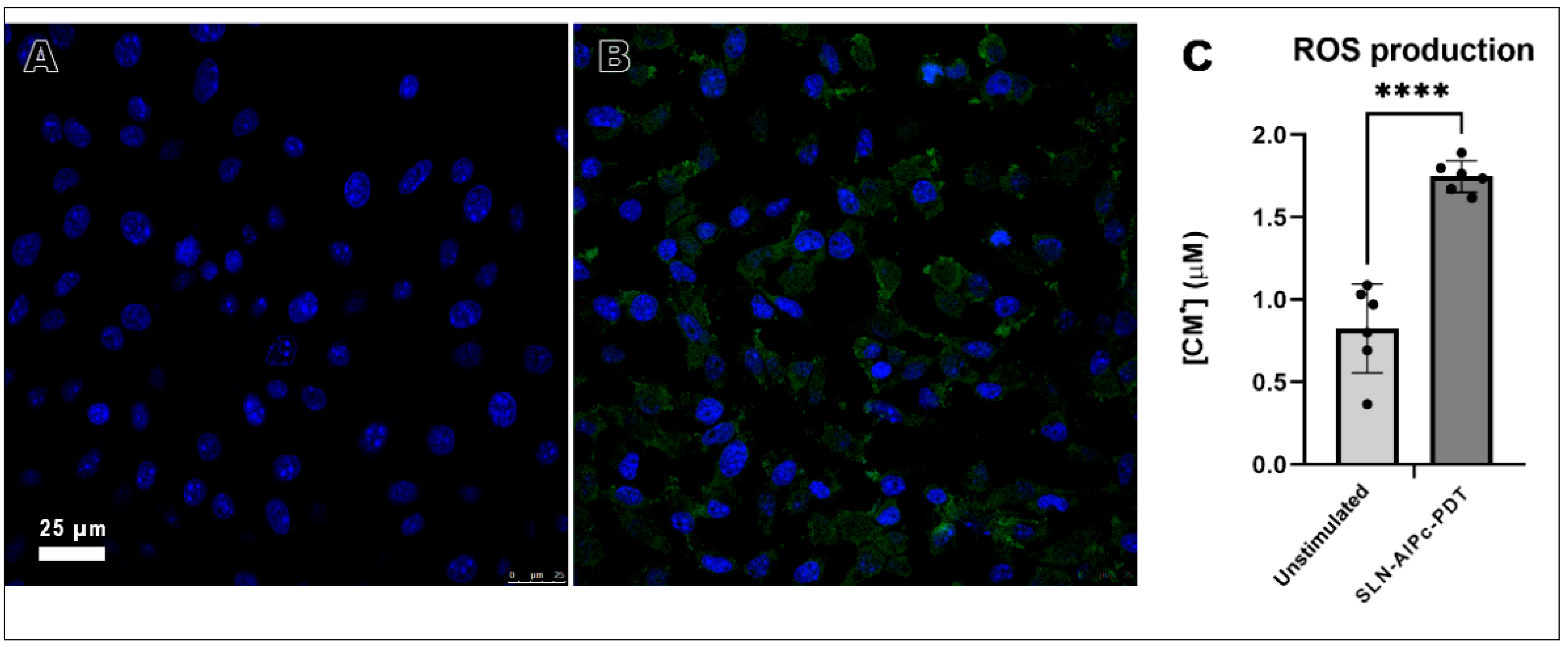
2.12.4. In Vitro ROS Production after SLN-AlPc-PDT
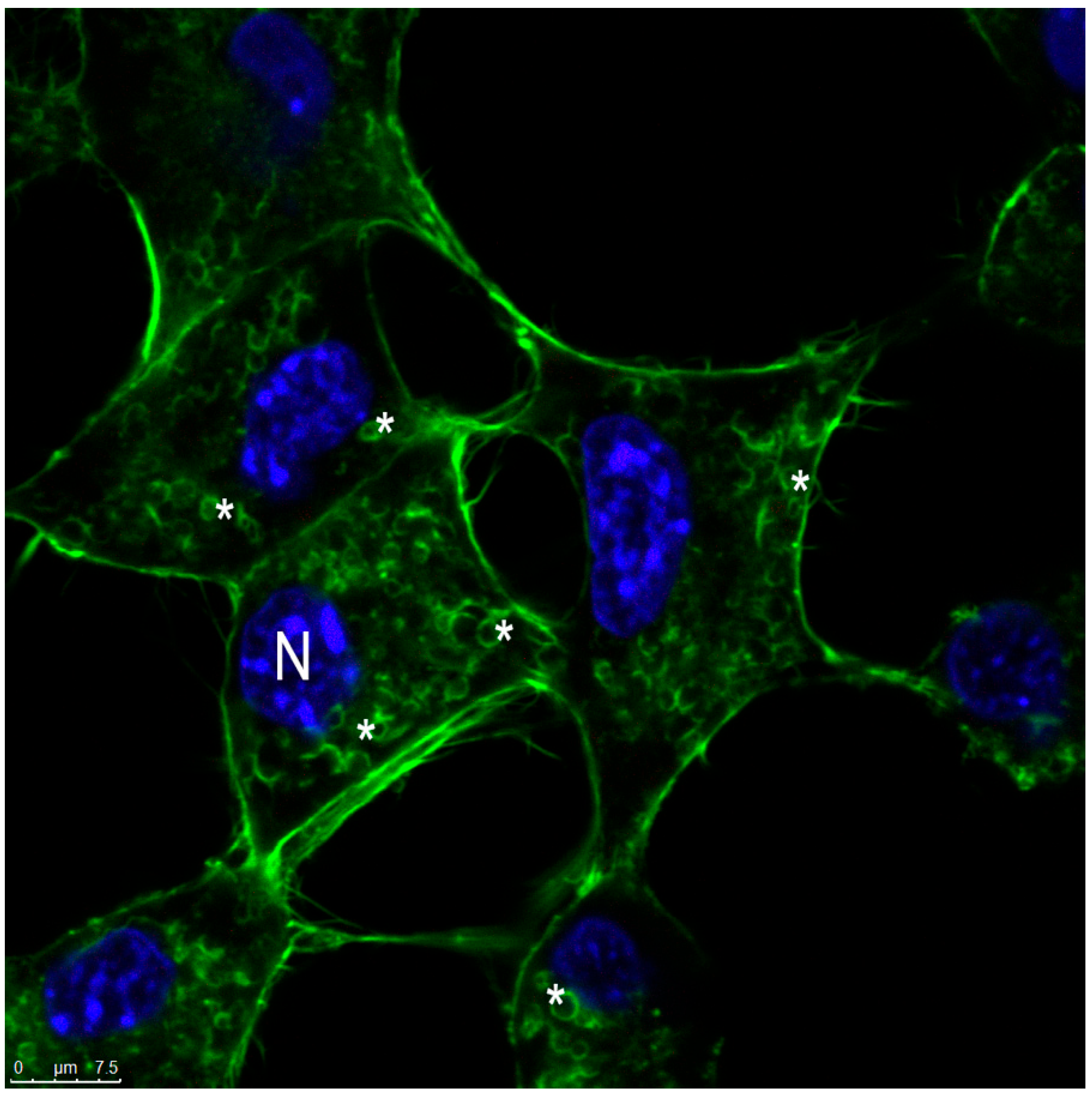
2.12.5. Actin Visualization
2.12.6. Western Blotting
3. Results and Discussion
3.1. Development and Optimization of Formulations
3.2. Differential Scanning Calorimetry
3.3. In Vitro Skin Permeation
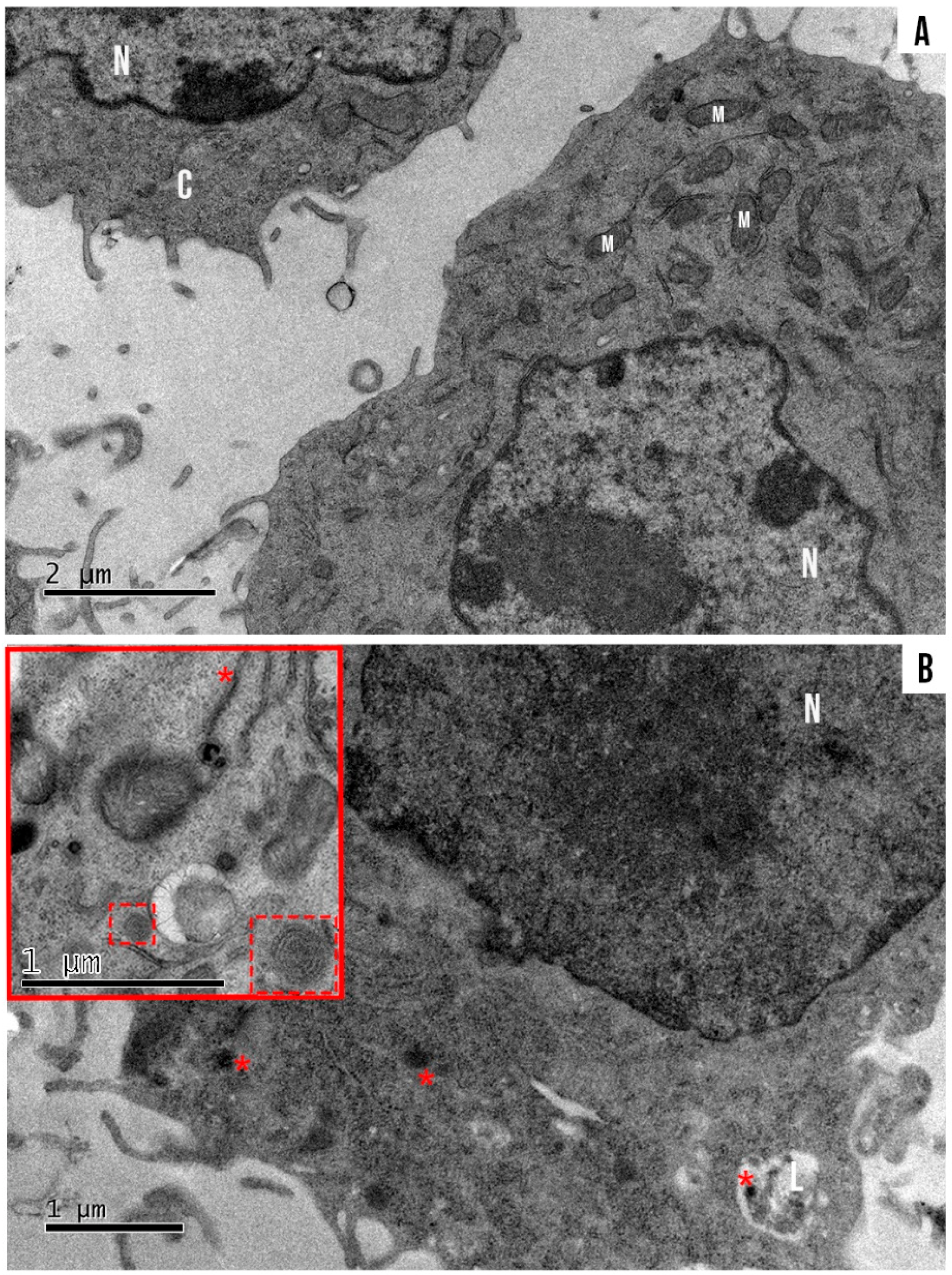
3.4. Morphology of SLN and SLN-AlPc
3.5. Spectroscopic Analysis
3.6. Photophysical Properties
3.7. Stability under Storage
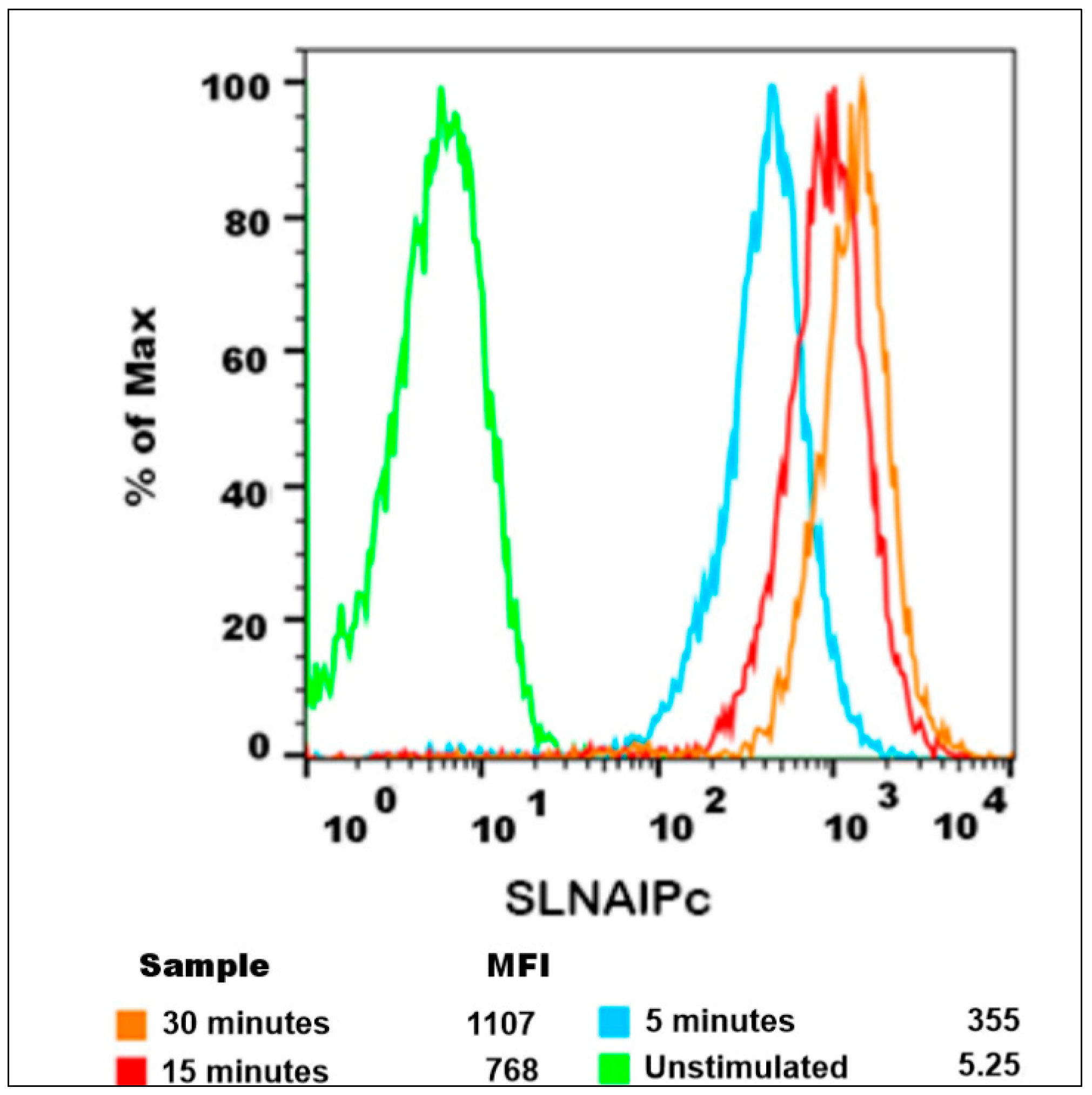
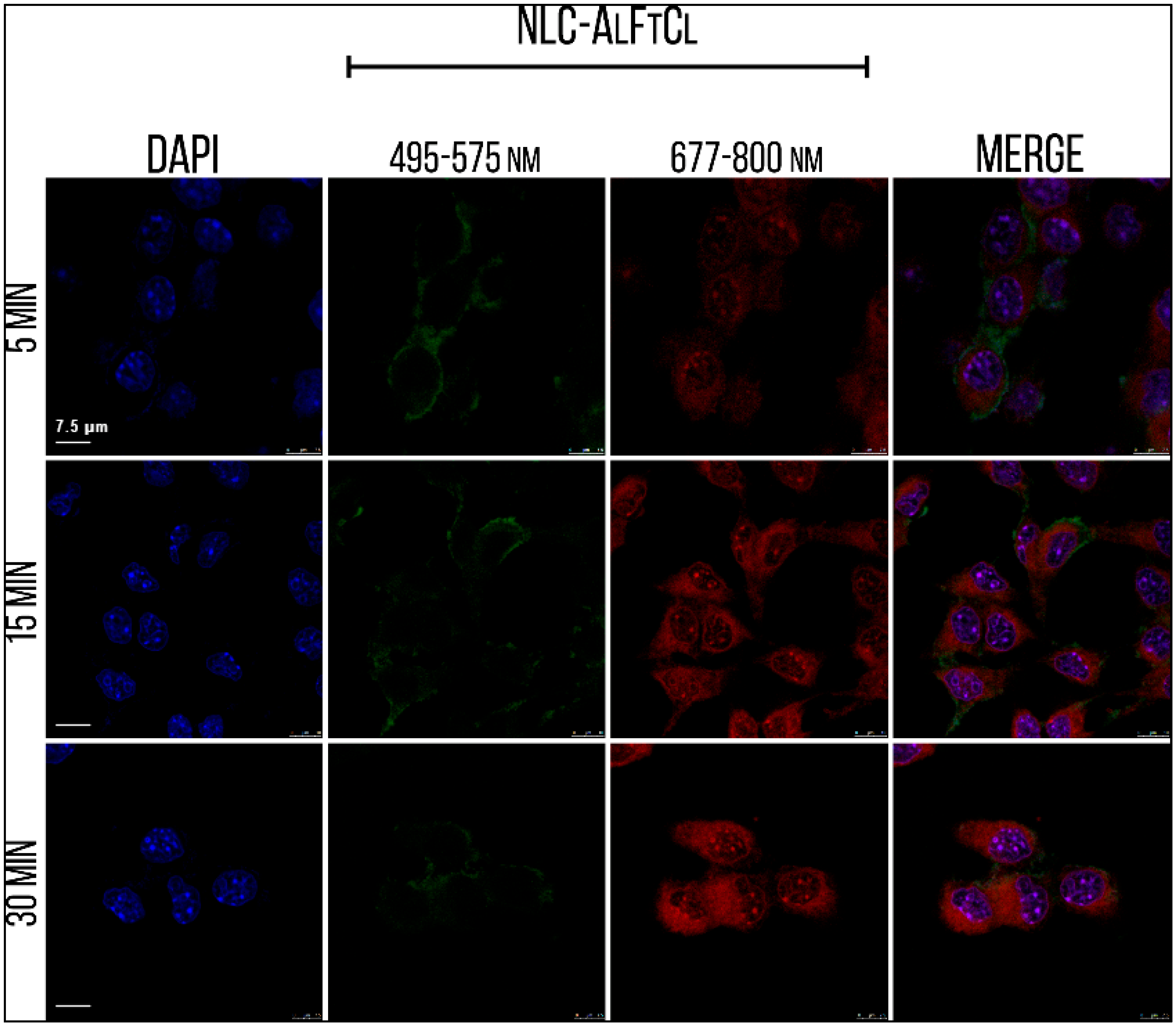
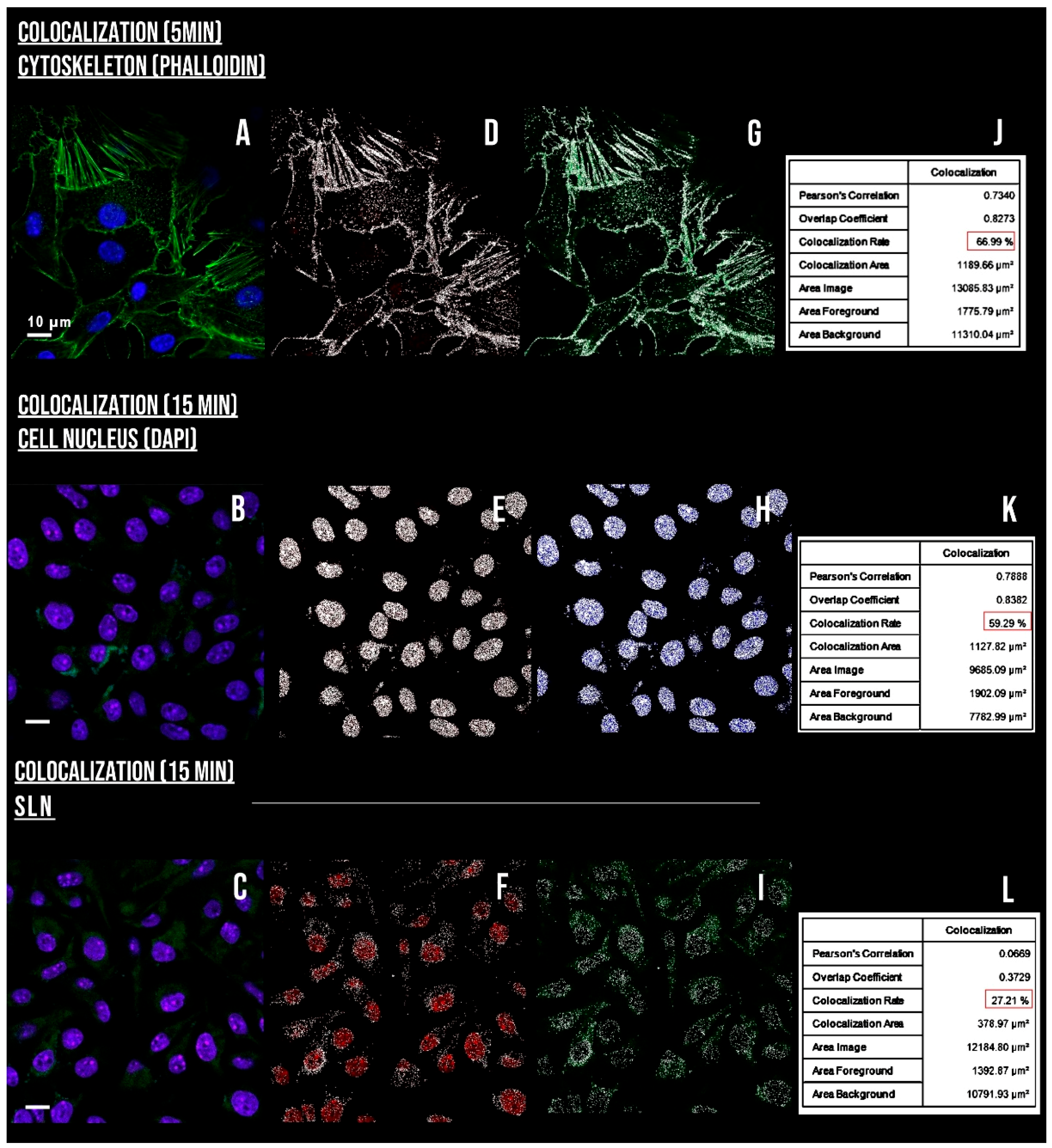
3.8. Internalization, Mechanisms of Endocytosis, and Subcellular Location of SLN-AlPc
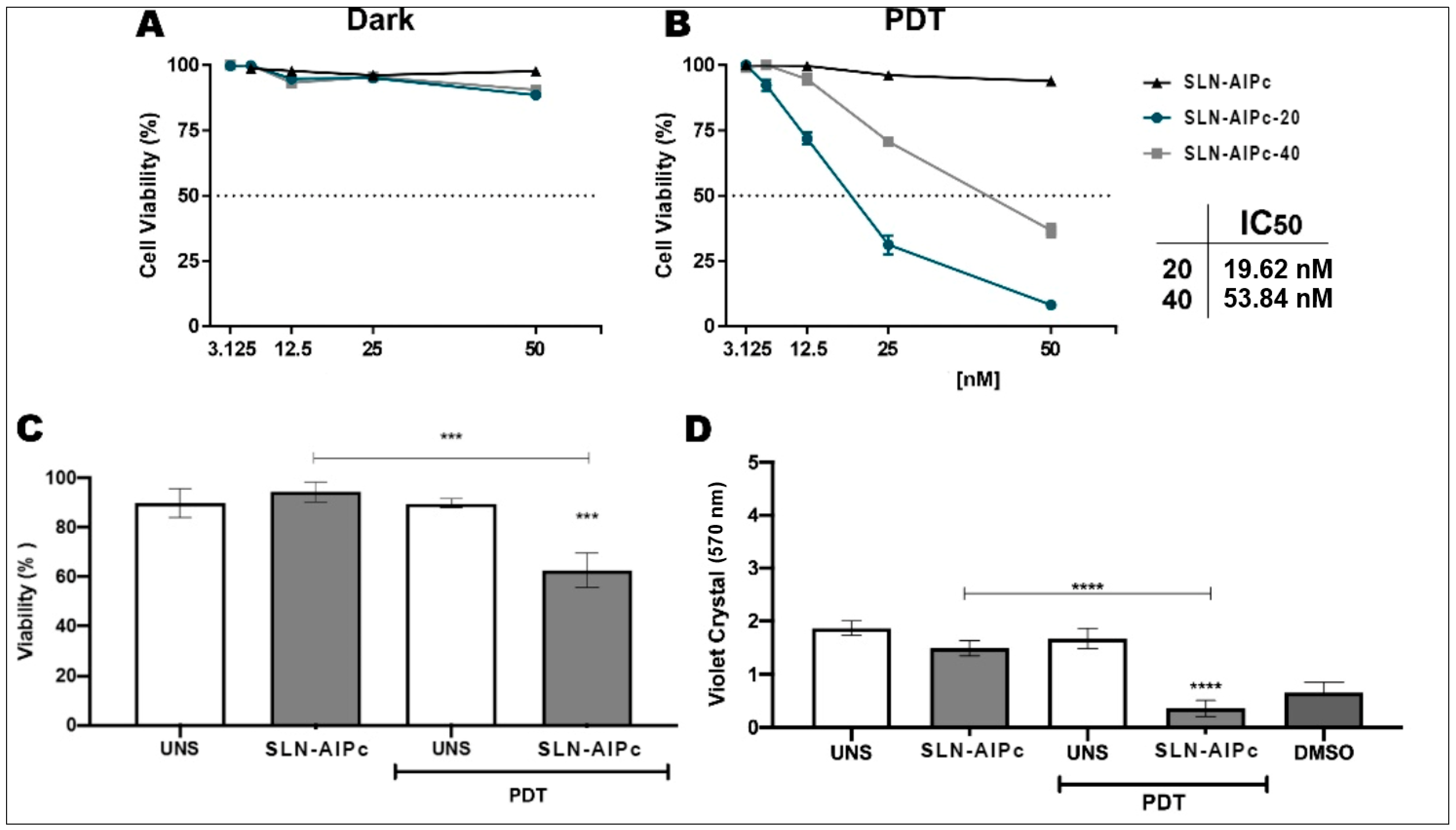
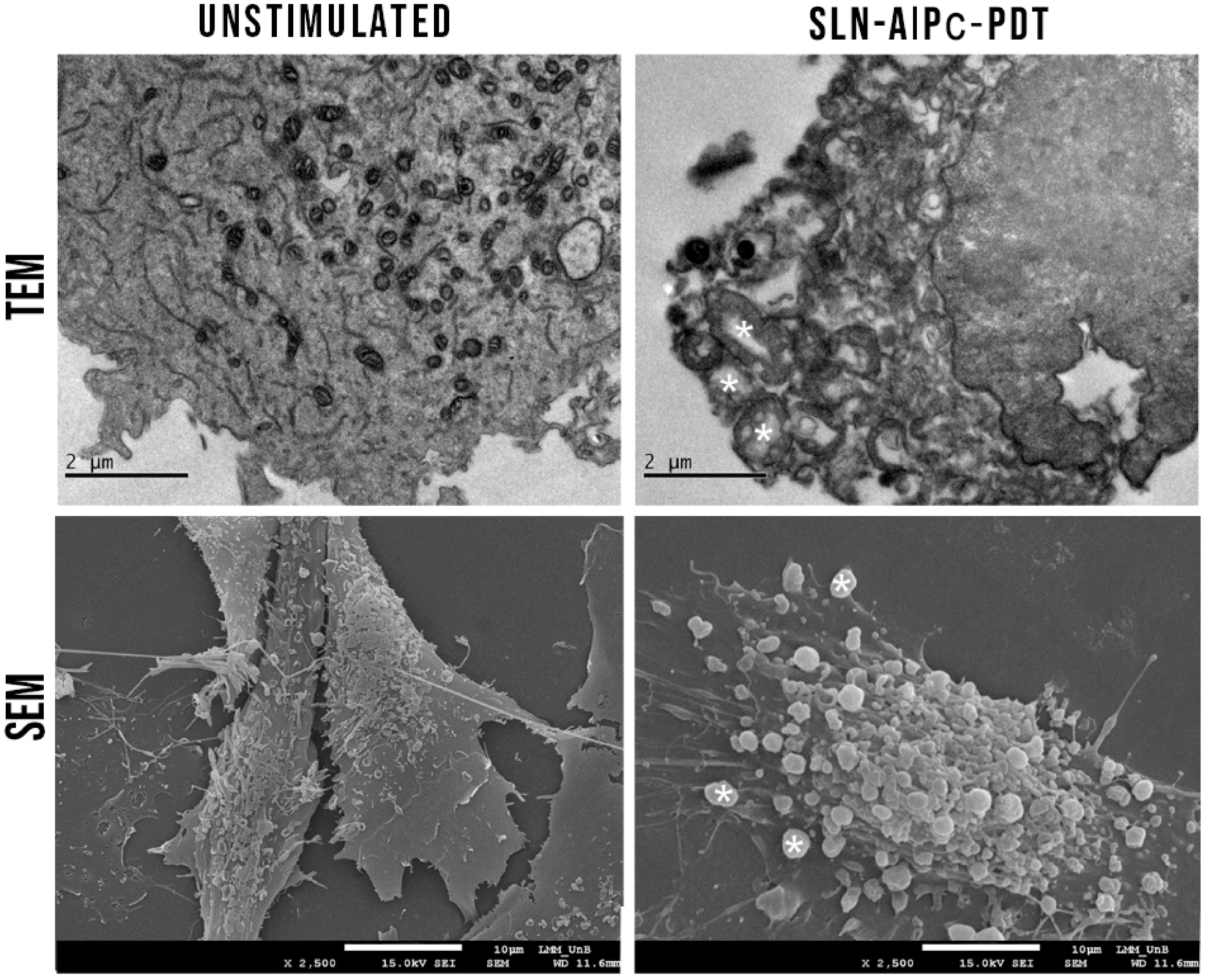
3.9. In Vitro Tests for the Assessment of Safety, Effectiveness, and Cell Death Analyses
3.10. SLN-AlPc-PDT Induces Apoptosis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hersey, P.; Zhang, X.D. How melanoma cells evade trail-induced apoptosis. Nat. Rev. Cancer 2001, 1, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Korbelik, M.J.; Banáth, K.M. Saw, Immunoregulatory cell depletion improves the efficacy of photodynamic therapy-generated cancer vaccines. Int. J. Mol. Sci. 2015, 16, 27005–27014. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Rashidi, L.H.; Yao, M.; Ma, L.; Chen, L.; Zhang, J.; Zhang, Y.; Chen, W. CuS nanoagents for photodynamic and photothermal therapies: Phenomena and possible mechanisms. Photodiagn. Photodyn. Ther. 2017, 19, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Yao, M.; Ma, L.; Hossu, M.; Han, X.; Juzenas, P.; Chen, W. X-ray-induced nanoparticle-based photodynamic therapy of cancer. Nanomedicine 2014, 9, 2339–2351. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, J. Using nanoparticles to enable simultaneous radiation and photodynamic therapies for cancer treatment. J. Nanosci. Nanotechnol. 2006, 6, 1159–1166. [Google Scholar] [CrossRef]
- Aghajanzadeh, M.; Zamani, M.; Kouchi, F.R.; Eixenberger, J.; Shirini, D.; Estrada, D.; Shirini, F. Synergic Antitumor Effect of Photodynamic Therapy and Chemotherapy Mediated by Nano Drug Delivery Systems. Pharmaceutics 2022, 14, 322. [Google Scholar] [CrossRef]
- Pandey, N.K.; Xiong, W.; Wang, L.; Chen, W.; Bui, B.; Yang, J.; Amador, E.; Chen, M.; Xing, C.; Athavale, A.A.; et al. Aggregation-induced emission luminogens for highly effective microwave dynamic therapy. Bioact. Mater. 2022, 7, 112–125. [Google Scholar] [CrossRef]
- Zhang, Q.; Guo, X.; Cheng, Y.; Chudal, L.; Pandey, N.K.; Zhang, J.; Ma, L.; Xi, Q.; Yang, G.; Chen, Y.; et al. Use of copper-cysteamine nanoparticles to simultaneously enable radiotherapy, oxidative therapy and immunotherapy for melanoma treatment. Signal Transduct. Target Ther. 2020, 5, 58. [Google Scholar] [CrossRef]
- Rodrigues, M.C. Photodynamic Therapy Based on Arrabidaea chica (Crajiru) Extract Nanoemulsion: In vitro Activity against Monolayers and Spheroids of Human Mammary Adenocarcinoma MCF-7 Cells. J. Nanomed. Nanotechnol. 2015, 6, 1000286. [Google Scholar] [CrossRef]
- Shrestha, S.; Wu, J.; Sah, B.; Vanasse, A.; Cooper, L.N.; Ma, L.; Li, G.; Zheng, H.; Chen, W.; Antosh, M.P. X-ray induced photodynamic therapy with copper-cysteamine nanoparticles in mice tumors. Proc. Natl. Acad. Sci. USA 2019, 116, 16823–16828. [Google Scholar] [CrossRef]
- Lloyd-Hughes, H.; Shiatis, A.E.; Pabari, A. Current and Future Nanotechnology Applications in the Management of Melanoma: A Review. J. Nanomed. Nanotechnol. 2015, 6, 1000334. [Google Scholar] [CrossRef]
- Bayón-Cordero, L.; Alkorta, I.; Arana, L. Application of Solid Lipid Nanoparticles to Improve the Efficiency of Anticancer drugs. Nanomaterials 2019, 9, 474. [Google Scholar] [CrossRef]
- Coelho, A.G.; dos Santos, W.R.P.; dos Santos, A.A.; da Silva, M.G.; Cunha, F.V.M.; Mendes, A.N.; Arcanjo, D.D.R. Plant-Derived Butters as Lipid Nanocarriers: A Systematic and Prospective Review. Recent Patents Nanotechnol. 2020, 14, 262–275. [Google Scholar] [CrossRef]
- Das, S.; Ng, W.K.; Tan, R.B.H. Are nanostructured lipid carriers (NLCs) better than solid lipid nanoparticles (SLNs): Development, characterizations and comparative evaluations of clotrimazole-loaded SLNs and NLCs? Eur. J. Pharm. Sci. 2012, 47, 139–151. [Google Scholar] [CrossRef]
- Kudarha, R.; Dhas, N.L.; Pandey, A.; Belgamwar, V.S.; Ige, P.P. Box-Behnken study design for optimization of bicalutamide-loaded nanostructured lipid carrier: Stability assessment. Pharm. Dev. Technol. 2015, 20, 608–618. [Google Scholar] [CrossRef]
- Araujo, V.H.S.; da Silva, P.B.; Szlachetka, I.O.; da Silva, S.W.; Fonseca-Santos, B.; Chorilli, M.; Ganassin, R.; de Oliveira, G.R.T.; da Rocha, M.C.O.; Fernandes, R.P.; et al. The influence of NLC composition on curcumin loading under a physicochemical perspective and in vitro evaluation. Colloids Surf. A Physicochem. Eng. Aspects 2020, 602, 125070. [Google Scholar] [CrossRef]
- Muehlmann, A.L.; Rodrigues, C.M.; Longo, P.J.; Garcia, P.M.; Py-Daniel, R.K.; Veloso, B.A.; de Souza, E.P.; da Silva, W.S.; Azevedo, B.R. Aluminium-phthalocyanine chloride nanoemulsions for anticancer photodynamic therapy: Development and in vitro activity against monolayers and spheroids of human mammary adenocarcinoma MCF-7 cells. J. Nanobiotechnol. 2015, 13, 1–11. [Google Scholar] [CrossRef]
- Vincenzo, B.; Riccio, F.; Luiz, A.; Nascimento, C.S.D.; Meneguin, A.B.; Rodero, C.F.; Santos, K.P.; Sábio, R.M.; de Annunzio, S.R.; Fontana, C.R.; et al. Solid Dispersions Incorporated into PVP Films for the Controlled Release of Trans-Resveratrol: Development Physicochemical and In Vitro Characterizations and In Vivo Cutaneous Anti-Inflammatory Evaluation. Pharmaceutics 2022, 6, 1149. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Da Rocha, M.C.O.; da Silva, P.B.; Radicchi, M.A.; Andrade, B.Y.G.; de Oliveira, J.V.; Venus, T.; Merker, C.; Estrela-Lopis, I.; Longo, J.P.F.; Báo, S.N. Docetaxel-loaded solid lipid nanoparticles prevent tumor growth and lung metastasis of 4T1 murine mammary carcinoma cells. J. Nanobiotechnol. 2020, 18, 43. [Google Scholar] [CrossRef]
- Gordillo-Galeano, A.; Mora-Huertas, C.E. Solid lipid nanoparticles and nanostructured lipid carriers: A review emphasizing on particle structure and drug release. Eur. J. Pharm. Biopharm. 2018, 133, 285–308. [Google Scholar] [CrossRef]
- Gomes, G.V.L.; Sola, M.R.; Rochetti, A.L.; Fukumasu, H.; Vicente, A.A.; Pinho, S.C. β-carotene and α-tocopherol coencapsulated in nanostructured lipid carriers of murumuru (Astrocaryum murumuru) butter produced by phase inversion temperature method: Characterisation, dynamic in vitro digestion and cell viability study. J. Microencapsul. 2019, 36, 43–52. [Google Scholar] [CrossRef]
- Campos, R.; Ollivon, M.; Marangoni, A.G. Molecular composition dynamics and structure of cocoa butter. Cryst. Growth Des. 2010, 10, 205–217. [Google Scholar] [CrossRef]
- Waghule, T.; Rapalli, V.K.; Singhvi, G.; Manchanda, P.; Hans, N.; Dubey, S.K.; Hasnain, M.S.; Nayak, A.K. Voriconazole loaded nanostructured lipid carriers based topical delivery system: QbD based designing, characterization, in-vitro and ex-vivo evaluation. J. Drug Deliv. Sci. Technol. 2019, 52, 303–315. [Google Scholar] [CrossRef]
- Schäfer-Korting, M.; Mehnert, W.; Korting, H.C. Lipid nanoparticles for improved topical application of drugs for skin diseases. Adv. Drug Deliv. Rev. 2007, 59, 427–443. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.K.; Kesharwani, R.; Kumar, V.; Patel, C.D.K. Lipid Nanoparticle Topical and Transdermal Delivery: A Review on Production, Penetration Mechanism to Skin. Int. J. Pharm. Investig. 2019, 9, 148–153. [Google Scholar] [CrossRef]
- Ganassin, R.; da Silva, V.C.M.; Araujo, V.H.S.; Tavares, G.R.; da Silva, P.B.; Cáceres-Vélez, P.R.; Porcel, J.E.M.; Rodrigues, M.C.; Andreozzi, P.; Fernandes, R.P.; et al. Solid lipid nanoparticles loaded with curcumin: Development and in vitro toxicity against CT26 cells. Nanomedicine 2022, 17, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Mackay, M.E.; Tuteja, A.; Duxbury, P.M.; Hawker, C.J.; van Horn, B.; Guan, Z.; Chen, G.; Krishnan, R.S. General Strategies for Nanoparticle Dispersion. Science 2006, 311, 1740–1743. [Google Scholar] [CrossRef]
- De Souza, T.D.; Ziembowicz, F.I.; Müller, D.F.; Lauermann, S.C.; Kloster, C.L.; Santos, R.C.V.; Lopes, L.Q.S.; Ourique, A.F.; MacHado, G.; Villetti, M.A. Evaluation of photodynamic activity, photostability and in vitro drug release of zinc phthalocyanine-loaded nanocapsules. Eur. J. Pharm. Sci. 2016, 83, 88–98. [Google Scholar] [CrossRef]
- Silva-Junior, L.C.; Maia, G.; Passos, R.R.; de Souza, E.A.; Camara, G.A.; Giz, M.J. Analysis of the selectivity of PtRh/C and PtRhSn/C to the formation of CO2 during ethanol electrooxidation. Electrochim Acta 2013, 112, 612–619. [Google Scholar] [CrossRef]
- Almeida, E.D.P.; Dipieri, L.V.; Rossetti, F.C.; Marchetti, J.M.; Bentley, M.V.L.B.; Nunes, R.; Sarmento, V.H.; Valerio, M.E.G.; Júnior, J.J.R.; Montalvão, M.M.; et al. Skin permeation, biocompatibility and antitumor effect of chloroaluminum phthalocyanine associated to oleic acid in lipid nanoparticles. Photodiagn. Photodyn. Ther. 2018, 24, 262–273. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Solid lipid nanoparticles and nanostructured lipid carriers as novel drug delivery systems: Applications, advantages and disadvantages. Res. Pharm. Sci. 2018, 13, 288–303. [Google Scholar] [CrossRef]
- Garcês, A.; Amaral, M.H.; Lobo, J.M.S.; Silva, A.C. Formulations based on solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) for cutaneous use: A review. Eur. J. Pharm. Sci. 2018, 112, 159–167. [Google Scholar] [CrossRef]
- Almeida, A.J.; Souto, E. Solid lipid nanoparticles as a drug delivery system for peptides and proteins. Adv. Drug Deliv. Rev. 2007, 59, 478–490. [Google Scholar] [CrossRef]
- Chen, L.; Remondetto, G.E.; Subirade, M. Food protein-based materials as nutraceutical delivery systems. Trends Food Sci. Technol. 2006, 17, 272–283. [Google Scholar] [CrossRef]
- Yu, Q.; Hu, X.; Ma, Y.; Xie, Y.; Lu, Y.; Qi, J.; Xiang, L.; Li, F.; Wu, W. Lipids-based nanostructured lipid carriers (NLCs) for improved oral bioavailability of sirolimus. Drug Deliv. 2016, 23, 1469–1475. [Google Scholar] [CrossRef]
- Shangguan, M.; Lu, Y.; Qi, J.; Han, J.; Tian, Z.; Xie, Y.; Hu, F.; Yuan, H.; Wu, W. Binary lipids-based nanostructured lipid carriers for improved oral bioavailability of silymarin. J. Biomater. Appl. 2014, 28, 887–896. [Google Scholar] [CrossRef]
- García-Pinel, B.; Porras-Alcalá, C.; Ortega-Rodríguez, A.; Sarabia, F.; Prados, J.; Melguizo, C.; López-Romero, J.M. Lipid-based nanoparticles: Application and recent advances in cancer treatment. Nanomaterials 2019, 9, 638. [Google Scholar] [CrossRef]
- Lewis, R.N.A.H.; McElhaney, R.N. Membrane lipid phase transitions and phase organization studied by Fourier transform infrared spectroscopy. Biochim. Biophys. Acta (BBA) Biomembr. 2013, 1828, 2347–2358. [Google Scholar] [CrossRef]
- Da Silva, E.; Rousseau, D. Raman Spectroscopy for the Study of Molecular Order, Thermodynamics, and Solid-Liquid Transitions in Triacylglycerols; American Chemical Society: Washington, DC, USA, 2006. [Google Scholar] [CrossRef]
- Basova, T.V.; Kiselev, V.G.; Plyashkevich, V.A.; Cheblakov, P.B.; Latteyer, F.; Peisert, H.; Chassè, T. Orientation and morphology of chloroaluminum phthalocyanine films grown by vapor deposition: Electrical field-induced molecular alignment. Chem. Phys. 2011, 380, 40–47. [Google Scholar] [CrossRef]
- Zhang, J.; de Sousa, W.T.; da Silva, V.C.M.; Rodrigues, M.C.; Morais, J.A.V.; Song, J.-L.; Cheng, Z.-Q.; Longo, J.P.F.; Azevedo, R.B.; Jiang, C.-S.; et al. Synthesis and evaluation of new potential benzo[a]phenoxazinium photosensitizers for anticancer photodynamic therapy. Molecules 2018, 23, 1436. [Google Scholar] [CrossRef]
- Robertson, C.A.; Evans, D.H.; Abrahamse, H. Photodynamic therapy (PDT): A short review on cellular mechanisms and cancer research applications for PDT. J. Photochem. Photobiol. B 2009, 96, 1–8. [Google Scholar] [CrossRef]
- Morais, J.A.V.; Almeida, L.R.; Rodrigues, M.C.; Azevedo, R.B.; Muehlmann, L.A. The induction of immunogenic cell death by photodynamic therapy in B16F10 cells in vitro is effected by the concentration of the photosensitizer. Photodiagn. Photodyn. Ther. 2021, 35, 2392. [Google Scholar] [CrossRef]
- Rodrigues, M.C.; de Júnior, W.T.; Mundim, T.; Vale, C.L.C.; de Oliveira, J.V.; Ganassin, R.; Pacheco, T.J.A.; Morais, J.A.V.; Longo, J.P.F.; Azevedo, R.B.; et al. Induction of Immunogenic Cell Death by Photodynamic Therapy Mediated by Aluminum-Phthalocyanine in Nanoemulsion. Pharmaceutics 2022, 14, 196. [Google Scholar] [CrossRef]
- Jones, A.T. Macropinocytosis: Searching for an endocytic identity and role in the uptake of cell penetrating peptides. J. Cell. Mol. Med. 2007, 11, 670–684. [Google Scholar] [CrossRef]
- Garg, A.D.; Martin, S.; Golab, J.; Agostinis, P. Danger signalling during cancer cell death: Origins, plasticity and regulation. Cell Death Differ. 2014, 21, 26–38. [Google Scholar] [CrossRef]
- Garg, A.D.; Agostinis, P. ER stress, autophagy and immunogenic cell death in photodynamic therapy-induced anti-cancer immune responses. Photochem. Photobiol. Sci. 2014, 13, 474–487. [Google Scholar] [CrossRef]
- Rodrigues, M.C. Terapia Fotodinâmica Mediada por Extrato de Crajiru e Cloreto de Alumínio-Ftalocianina em Nanoemulsões no Tratamento de Câncer de Mama In Vitro; Instituto de Ciências Biológicas: Belo Horizonte, Brazil, 2014. [Google Scholar]



| Independent Variables | Levels | ||
|---|---|---|---|
| −1 | 0 | +1 | |
| A: Butter: Surfactant | 1:1 | 1:1.5 | 1:2 |
| B: AlPc (μM) | 20 | 40 | 80 |
| C: Temperature (°C) | 75 | 80 | 85 |
| Number of Experiment | A | B | C |
|---|---|---|---|
| 1 | 0 | 1 | −1 |
| 2 | −1 | 0 | 1 |
| 3 | −1 | −1 | 0 |
| 4 | 0 | −1 | −1 |
| 5 | 0 | 0 | 0 |
| 6 | 0 | 0 | 0 |
| 7 | 1 | 0 | 1 |
| 8 | −1 | 0 | −1 |
| 9 | 0 | −1 | 1 |
| 10 | −1 | 1 | 0 |
| 11 | 1 | −1 | 0 |
| 12 | 1 | 1 | 0 |
| 13 | 0 | 0 | 0 |
| 14 | 1 | 0 | −1 |
| 15 | 0 | 1 | 1 |
| Inhibitor | Concentration | Inhibition Pathway |
|---|---|---|
| Sodium azid | 100 mM | ATP-dependent pathways |
| Amiloride | 0.2 mM | Macropinocytosis |
| Cytochalasin D | 1 μM | Pinocytosis |
| Phenylarsine | 0.2 μM | Clathrin-mediated pathway |
| Nystatin | 20 μg/mL | Caveolin-mediated pathway |
| Sample | HD (nm) | PdI | ZP (mV) |
|---|---|---|---|
| SLN Murumuru | 27.5 ± 0.2 | 0.191 ± 0.001 | −11.80 ± 2.60 |
| SLN Babaçu | 100.6 ± 0.2 | 0.266 ± 0.005 | −10.40 ± 0.15 |
| SLN Bacuri | 536.4 ± 49.8 | 0.600 ± 0.039 | −22.30 ± 1.18 |
| SLN Ucuuba | 140.8 ± 2.7 | 0.526 ± 0.006 | −21.60 ± 0.12 |
| Sample | HD (nm) | PdI | ZP (mV) | EE% |
|---|---|---|---|---|
| SLN | 55.53 ± 0.23 | 0.191 ± 0.001 | −11.80 ± 2.60 | 61.50 ± 0.67 |
| SLN optimized | 17.64 ± 0.21 | 0.173 ± 0.013 | −5.61 ± 0.93 | 66.40 ± 1.12 |
| Sample | Melting Point (°C) | ΔH (Jg−1) |
|---|---|---|
| Murumuru butter | 38.4 | 63.9 |
| AlPc | 175.1 | 194.3 |
| SLN | 96.3 | 1036.0 |
| SLN-AlPc | 93.3 | 873.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mello, V.C.; Araújo, V.H.S.; de Paiva, K.L.R.; Simões, M.M.; Marques, D.C.; da Silva Costa, N.R.; de Souza, I.F.; da Silva, P.B.; Santos, I.; Almeida, R.; et al. Development of New Natural Lipid-Based Nanoparticles Loaded with Aluminum-Phthalocyanine for Photodynamic Therapy against Melanoma. Nanomaterials 2022, 12, 3547. https://doi.org/10.3390/nano12203547
Mello VC, Araújo VHS, de Paiva KLR, Simões MM, Marques DC, da Silva Costa NR, de Souza IF, da Silva PB, Santos I, Almeida R, et al. Development of New Natural Lipid-Based Nanoparticles Loaded with Aluminum-Phthalocyanine for Photodynamic Therapy against Melanoma. Nanomaterials. 2022; 12(20):3547. https://doi.org/10.3390/nano12203547
Chicago/Turabian StyleMello, Victor Carlos, Victor Hugo Sousa Araújo, Karen Letycia Rodrigues de Paiva, Marina Mesquita Simões, Dafne Caroline Marques, Nelice Roberta da Silva Costa, Isadora Florêncio de Souza, Patricia Bento da Silva, Igor Santos, Raquel Almeida, and et al. 2022. "Development of New Natural Lipid-Based Nanoparticles Loaded with Aluminum-Phthalocyanine for Photodynamic Therapy against Melanoma" Nanomaterials 12, no. 20: 3547. https://doi.org/10.3390/nano12203547
APA StyleMello, V. C., Araújo, V. H. S., de Paiva, K. L. R., Simões, M. M., Marques, D. C., da Silva Costa, N. R., de Souza, I. F., da Silva, P. B., Santos, I., Almeida, R., Magalhães, K. G., da Silva, S. W., Santos, A. S., Veiga-Souza, F., Souza, P. E. N., Raddichi, M. A., Longo, J. P. F., de Araújo, J. T. C., Muehlmann, L. A., ... Báo, S. N. (2022). Development of New Natural Lipid-Based Nanoparticles Loaded with Aluminum-Phthalocyanine for Photodynamic Therapy against Melanoma. Nanomaterials, 12(20), 3547. https://doi.org/10.3390/nano12203547









