Bimetallic Nanowires on Laser-Patterned PEN as Promising Biomaterials
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Apparatus
2.2. Analytical Methods
2.3. Antibacterial Tests
2.4. Cytotoxicity
3. Results and Discussion
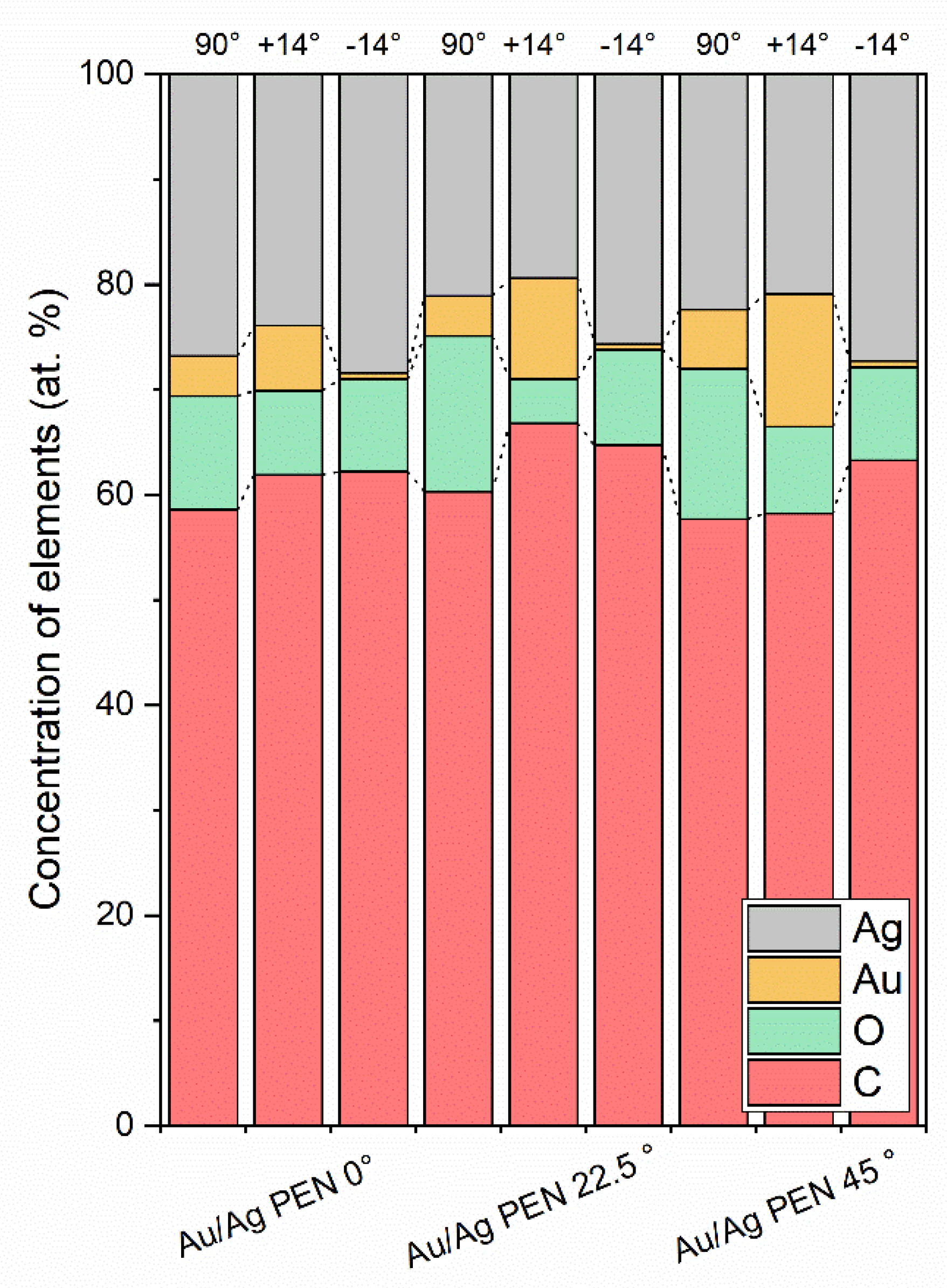
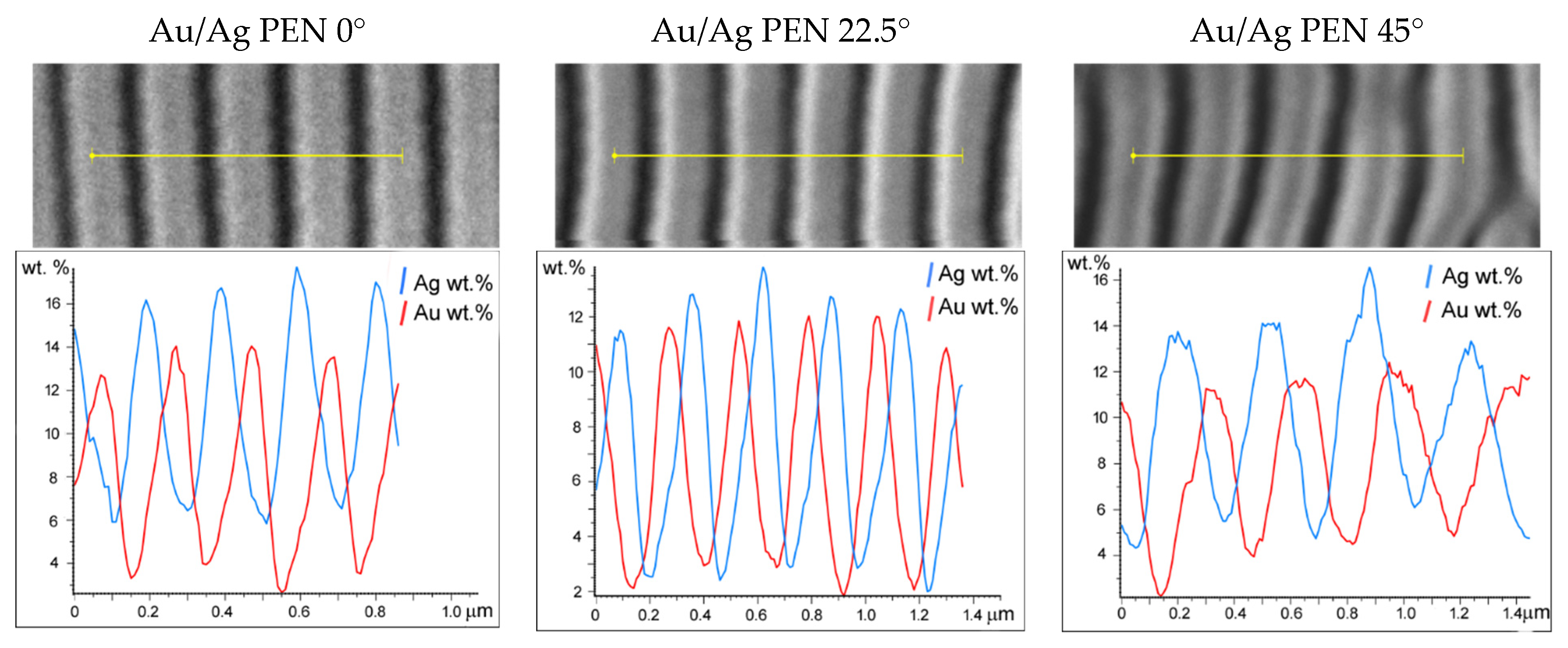
3.1. Surface Characterisation
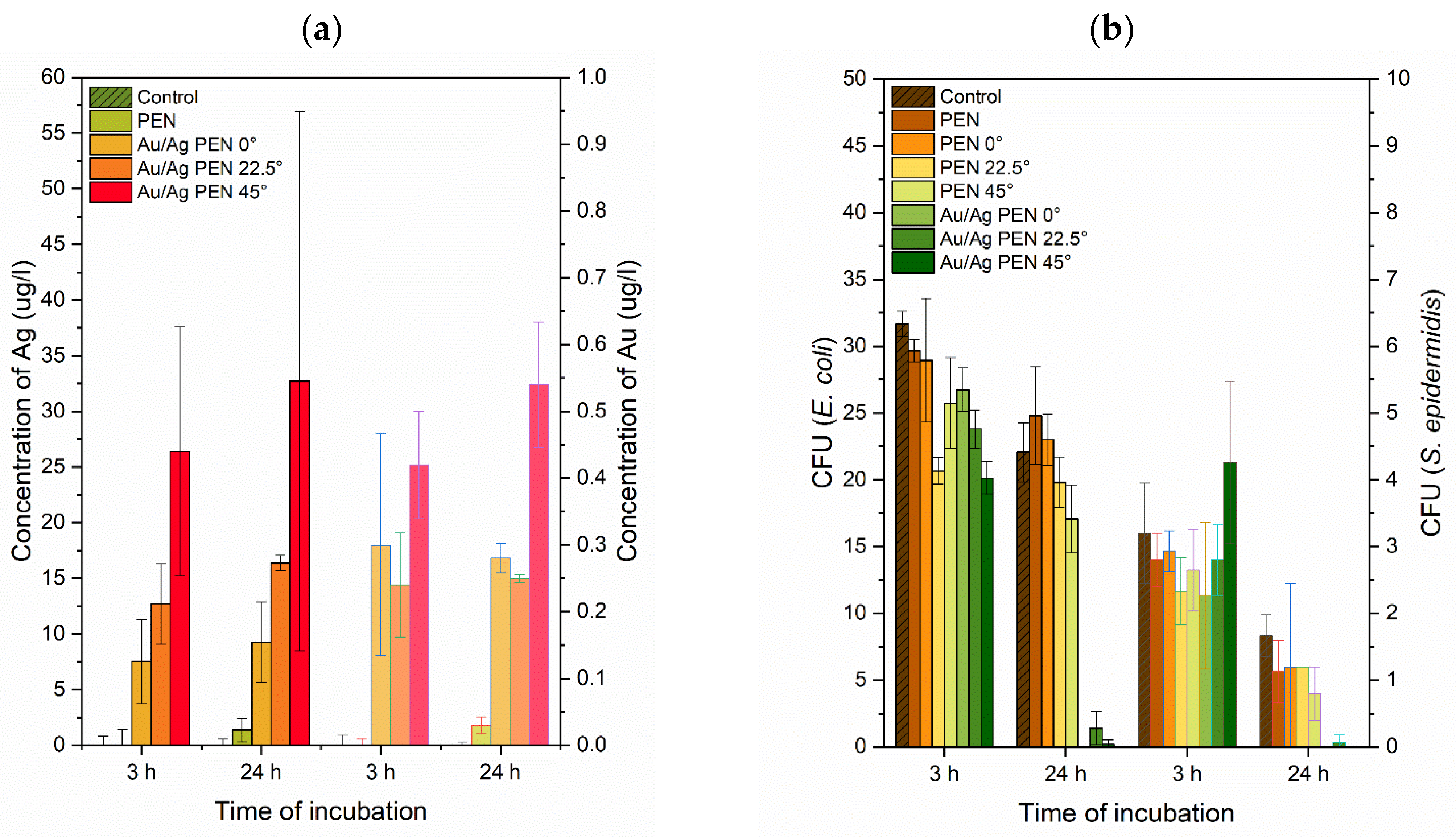
3.2. Release of Ag+ and Aun+
3.3. Antibacterial Tests
3.4. Cytotoxicity
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pryjmaková, J.; Kaimlová, M.; Hubáček, T.; Švorčík, V.; Siegel, J. Nanostructured Materials for Artificial Tissue Replacements. Int. J. Mol. Sci. 2020, 21, 2521. [Google Scholar] [CrossRef]
- Slepička, P.; Siegel, J.; Lyutakov, O.; Kasálková, N.S.; Kolská, Z.; Bačáková, L.; Švorčík, V. Polymer nanostructures for bioapplications induced by laser treatment. Biotechnol. Adv. 2018, 36, 839–855. [Google Scholar] [CrossRef] [PubMed]
- Aderson, J.M. Inflammatory Response to Implants. ASAIO J. 1988, 34, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Nicolle, L.E. Catheter associated urinary tract infections. Antimicrob. Resist. Infect. Control 2014, 3, 23. [Google Scholar] [CrossRef]
- Pavithra, D.; Doble, M. Biofilm formation, bacterial adhesion and host response on polymeric implants—Issues and prevention. Biomed. Mater. 2008, 3, 034003. [Google Scholar] [CrossRef]
- Fisher, L.E.; Hook, A.L.; Ashraf, W.; Yousef, A.; Barrett, D.A.; Scurr, D.J.; Chen, X.; Smith, E.F.; Fay, M.; Parmenter, C.D.; et al. Biomaterial modification of urinary catheters with antimicrobials to give long-term broadspectrum antibiofilm activity. J. Control. Release 2015, 202, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Chellamani, K.P.; Balaji, R.S.; Sudharsan, J. Antibacterial properties of allopathic drug loaded polycaprolactone nanomembrane. J. Acad. Ind. Res. 2013, 2, 341–344. [Google Scholar]
- Muszanska, A.K.; Rochford, E.T.J.; Gruszka, A.; Bastian, A.A.; Busscher, H.J.; Norde, W.; Van Der Mei, H.C.; Herrmann, A. Antiadhesive Polymer Brush Coating Functionalized with Antimicrobial and RGD Peptides to Reduce Biofilm Formation and Enhance Tissue Integration. Biomacromolecules 2014, 15, 2019–2026. [Google Scholar] [CrossRef]
- Roe, D.; Karandikar, B.; Bonn-Savage, N.; Gibbins, B.; Roullet, J.-B. Antimicrobial surface functionalization of plastic catheters by silver nanoparticles. J. Antimicrob. Chemother. 2008, 61, 869–876. [Google Scholar] [CrossRef]
- Siegel, J.; Kaimlová, M.; Vyhnálková, B.; Trelin, A.; Lyutakov, O.; Slepička, P.; Švorčík, V.; Veselý, M.; Vokatá, B.; Malinský, P.; et al. Optomechanical Processing of Silver Colloids: New Generation of Nanoparticle-Polymer Composites with Bactericidal Effect. Int. J. Mol. Sci. 2020, 22, 312. [Google Scholar] [CrossRef]
- Riveiro, A.; Maçon, A.L.B.; Del Val, J.; Comesaña, R.; Pou, J. Laser Surface Texturing of Polymers for Biomedical Applications. Front. Phys. 2018, 6, 16. [Google Scholar] [CrossRef]
- Allen, N.S. A study of the light absorption properties of polymer films using UV-visible derivative spectroscopy. Polym. Photochem. 1981, 1, 43–55. [Google Scholar] [CrossRef]
- Michaljaničová, I.; Slepička, P.; Rimpelova, S.; Kasálková, N.S.; Švorčík, V. Regular pattern formation on surface of aromatic polymers and its cytocompatibility. Appl. Surf. Sci. 2016, 370, 131–141. [Google Scholar] [CrossRef]
- Barb, R.-A.; Hrelescu, C.; Dong, L.; Heitz, J.; Siegel, J.; Slepička, P.; Vosmanská, V.; Svorcik, V.; Magnus, B.; Marksteiner, R.; et al. Laser-induced periodic surface structures on polymers for formation of gold nanowires and activation of human cells. Appl. Phys. A 2014, 117, 295–300. [Google Scholar] [CrossRef]
- Siegel, J.; Slepička, P.; Heitz, J.; Kolská, Z.; Sajdl, P.; Švorčík, V. Gold nano-wires and nano-layers at laser-induced nano-ripples on PET. Appl. Surf. Sci. 2010, 256, 2205–2209. [Google Scholar] [CrossRef]
- Kaimlová, M.; Nemogová, I.; Kolarova, K.; Slepička, P.; Švorčík, V.; Siegel, J. Optimization of silver nanowire formation on laser processed PEN: Surface properties and antibacterial effects. Appl. Surf. Sci. 2019, 473, 516–526. [Google Scholar] [CrossRef]
- Suarasan, S.; Focsan, M.; Soritau, O.; Maniu, D.; Astilean, S. One-pot, green synthesis of gold nanoparticles by gelatin and investigation of their biological effects on Osteoblast cells. Colloids Surf. B Biointerfaces 2015, 132, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Ko, W.-K.; Heo, D.N.; Moon, H.-J.; Lee, S.J.; Bae, M.S.; Lee, J.B.; Sun, I.-C.; Jeon, H.B.; Park, H.K.; Kwon, I.K. The effect of gold nanoparticle size on osteogenic differentiation of adipose-derived stem cells. J. Colloid Interface Sci. 2015, 438, 68–76. [Google Scholar] [CrossRef]
- Peterbauer, T.; Yakunin, S.; Siegel, J.; Hering, S.; Fahrner, M.; Romanin, C.; Heitz, J. Dynamics of Spreading and Alignment of Cells Cultured In Vitro on a Grooved Polymer Surface. J. Nanomater. 2011, 2011, 413079. [Google Scholar] [CrossRef][Green Version]
- Crabtree, J.H.; Burchette, R.J.; ASiddiqi, R.; Huen, I.T.; Hadnott, L.L.; Fishman, A. The efficacy of silver-ion implanted catheters in reducing peritoneal dialysis-related infections. Perit. Dial. Int. 2003, 23, 368–374. [Google Scholar]
- Guo, L.; Yuan, W.; Lu, Z.; Li, C. Polymer/nanosilver composite coatings for antibacterial applications. Colloids Surf. A Physicochem. Eng. Asp. 2013, 439, 69–83. [Google Scholar] [CrossRef]
- Mahmoodi, S.; Elmi, A.; Nezhadi, S.H. Copper Nanoparticles as Antibacterial Agents. J. Mol. Pharm. Org. Process. Res. 2018, 6, 1–7. [Google Scholar] [CrossRef]
- Panacek, A.; Kvitek, L.; Prucek, R.; Kolar, M.; Vecerova, R.; Pizurova, N.; Sharma, V.K.; Nevecna, T.; Zboril, R. Silver colloid nanoparticles: Synthesis, characterization, and their antibacterial activity. J. Phys. Chem. B 2006, 110, 16248–16253. [Google Scholar] [CrossRef]
- Zhang, Y.; Dasari, T.P.S.; Deng, H.; Yu, H. Antimicrobial Activity of Gold Nanoparticles and Ionic Gold. J. Environ. Sci. Heath Part C 2015, 33, 286–327. [Google Scholar] [CrossRef] [PubMed]
- Braydich-Stolle, L.; Hussain, S.; Schlager, J.J.; Hofmann, M.-C. In Vitro Cytotoxicity of Nanoparticles in Mammalian Germline Stem Cells. Toxicol. Sci. 2005, 88, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Asharani, P.V.; Mun, G.L.K.; Hande, M.P.; Valiyaveettil, S. Cytotoxicity and Genotoxicity of Silver Nanoparticles in Human Cells. ACS Nano 2009, 3, 279–290. [Google Scholar] [CrossRef]
- Polívková, M.; Štrublová, V.; Hubáček, T.; Rimpelová, S.; Švorčík, V.; Siegel, J. Surface characterization and antibacterial response of silver nanowire arrays supported on laser-treated polyethylene naphthalate. Mater. Sci. Eng. C 2017, 72, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Ton-That, C.; Shard, A.; Bradley, R.H. Thickness of Spin-Cast Polymer Thin Films Determined by Angle-Resolved XPS and AFM Tip-Scratch Methods. Langmuir 2000, 16, 2281–2284. [Google Scholar] [CrossRef]
- Herigstad, B.; Hamilton, M.; Heersink, J. How to optimize the drop plate method for enumerating bacteria. J. Microbiol. Methods 2001, 44, 121–129. [Google Scholar] [CrossRef]
- Novotna, Z.; Reznickova, A.; Rimpelova, S.; Vesely, M.; Kolska, Z.; Svorcik, V. Tailoring of PEEK bioactivity for improved cell interaction: Plasma treatment in action. RSC Adv. 2015, 5, 41428–41436. [Google Scholar] [CrossRef]
- Slepička, P.; Nedela, O.; Siegel, J.; Krajcar, R.; Kolska, Z.; Svorcik, V. Ripple polystyrene nano-pattern induced by KrF laser. Express Polym. Lett. 2014, 8, 459–466. [Google Scholar] [CrossRef]
- Slepička, P.; Chaloupka, A.; Sajdl, P.; Heitz, J.; Hnatowicz, V.; Švorčík, V. Angle dependent laser nanopatterning of poly (ethylene terephthalate) surfaces. Appl. Surf. Sci. 2011, 257, 6021–6025. [Google Scholar] [CrossRef]
- Bäuerle, D. Laser Processing and Chemistry, 3rd ed.; Springer-Verlag: Berlin/Heidelberg, Germany; New York, NY, USA, 2000. [Google Scholar]
- Csete, M.; Bor, Z. Laser-induced periodic surface structure formation on polyethylene-terephthalate. Appl. Surf. Sci. 1998, 133, 5–16. [Google Scholar] [CrossRef]
- Belardini, A.; Larciprete, M.C.; Centini, M.; Fazio, E.; Sibilia, C.; Bertolotti, M.; Toma, A.; Chiappe, D.; De Mongeot, F.B. Tailored second harmonic generation from self-organized metal nano-wires arrays. Opt. Express 2009, 17, 3603–3609. [Google Scholar] [CrossRef]
- Tyler, B.J.; Castner, D.G.; Ratner, B.D. Regularization: A stable and accurate method for generating depth profiles from angle-dependent XPS data. Surf. Interface Anal. 1989, 14, 443–450. [Google Scholar] [CrossRef]
- Vollath, D. Optical properties. In Nanoparticles-Nanocomposites-Nanomaterials: An Introduction for Beginners; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 181–228. [Google Scholar]
- Gunawidjaja, R.; Kharlampieva, E.; Choi, I.; Tsukruk, V.V. Bimetallic Nanostructures as Active Raman Markers: Gold-Nanoparticle Assembly on 1D and 2D Silver Nanostructure Surfaces. Small 2009, 5, 2460–2466. [Google Scholar] [CrossRef]
- Joo, H.-Y.; Kim, H.J.; Kim, S.J.; Kim, S.Y. Spectrophotometric analysis of aluminum nitride thin films. J. Vac. Sci. Technol. A 1999, 17, 862–870. [Google Scholar] [CrossRef]
- Murakami, D.; Jinnai, H.; Takahara, A. Wetting Transition from the Cassie-Baxter State to the Wenzel State on Textured Polymer Surfaces. Langmuir 2014, 30, 2061–2067. [Google Scholar] [CrossRef]
- Chernousova, S.; Epple, M. Silver as Antibacterial Agent: Ion, Nanoparticle, and Metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653. [Google Scholar] [CrossRef]
- Siegel, J.; Polívková, M.; Staszek, M.; Kolarova, K.; Rimpelova, S.; Švorčík, V. Nanostructured silver coatings on polyimide and their antibacterial response. Mater. Lett. 2015, 145, 87–90. [Google Scholar] [CrossRef]
- Chen, M.; Yu, Q.; Sun, H. Novel Strategies for the Prevention and Treatment of Biofilm Related Infections. Int. J. Mol. Sci. 2013, 14, 18488–18501. [Google Scholar] [CrossRef]
- PeŠŠková, V.; Kubies, D.; Hulejová, H.; Himmlová, L. The influence of implant surface properties on cell adhesion and proliferation. J. Mater. Sci. Mater. Med. 2007, 18, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.M.; Jiang, Z.; Bastmeyer, M.; Lahann, J. Physical Aspects of Cell Culture Substrates: Topography, Roughness, and Elasticity. Small 2012, 8, 336–355. [Google Scholar] [CrossRef] [PubMed]
- Marambio-Jones, C.; Hoek, E.M.V. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J. Nanoparticle Res. 2010, 12, 1531–1551. [Google Scholar] [CrossRef]
- Jung, D.; Minami, I.; Patel, S.; Lee, J.; Jiang, B.; Yuan, Q.; Li, L.; Kobayashi, S.; Chen, Y.; Lee, K.-B.; et al. Incorporation of functionalized gold nanoparticles into nanofibers for enhanced attachment and differentiation of mammalian cells. J. Nanobiotechnol. 2012, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
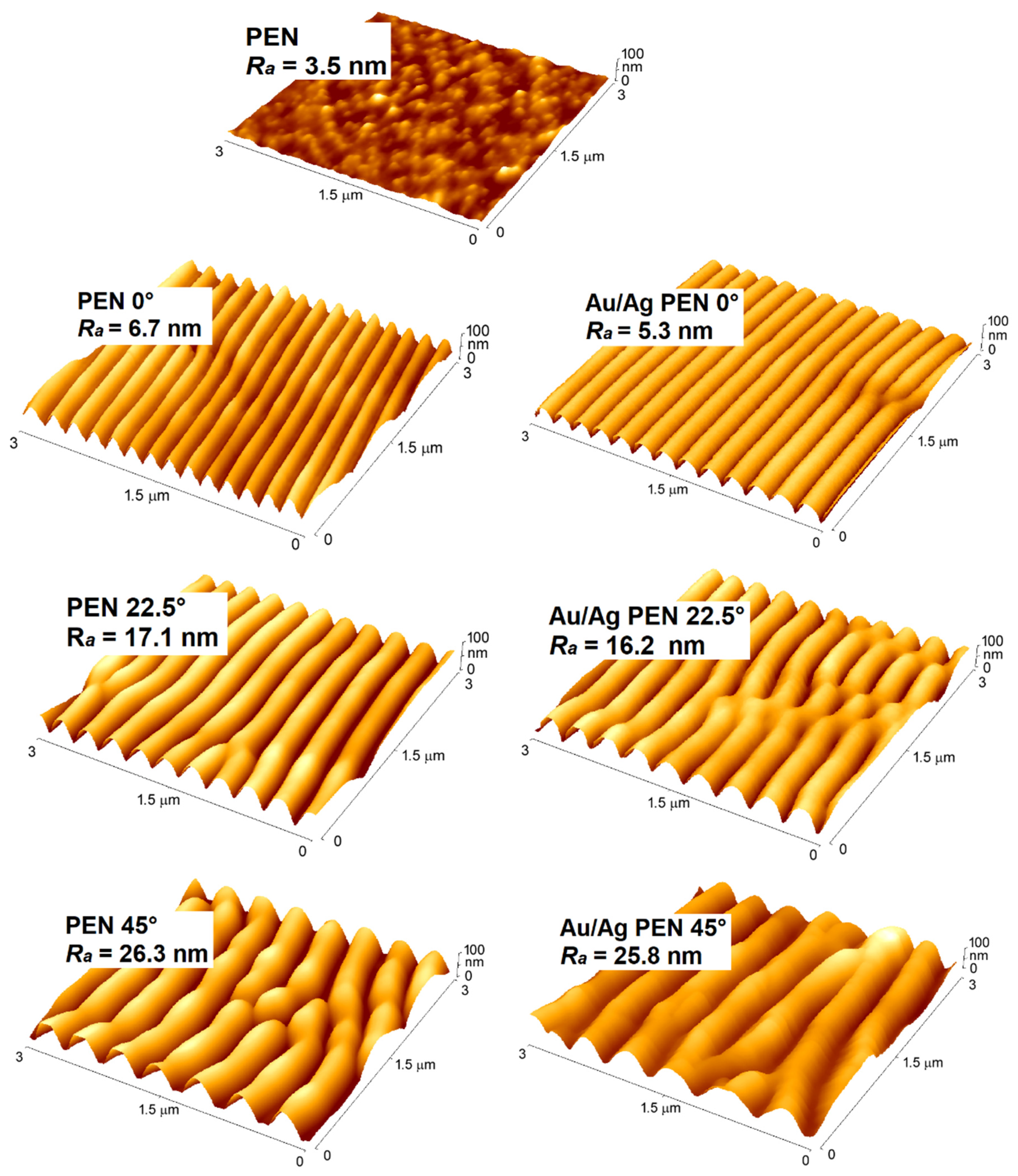





| Sample | Ra (nm) | Λ (nm) | h (nm) |
|---|---|---|---|
| PEN | 3.5 | - | - |
| PEN 0° | 6.7 | 212.4 | 24.8 |
| PEN 22.5° | 17.1 | 307.0 | 60.5 |
| PEN 45° | 26.3 | 398.0 | 94.8 |
| Au/Ag PEN 0° | 5.3 | 209.6 | 24.5 |
| Au/Ag PEN 22.5° | 16.2 | 303.4 | 59.4 |
| Au/Ag PEN 45° | 25.8 | 393.2 | 92.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pryjmaková, J.; Kaimlová, M.; Vokatá, B.; Hubáček, T.; Slepička, P.; Švorčík, V.; Siegel, J. Bimetallic Nanowires on Laser-Patterned PEN as Promising Biomaterials. Nanomaterials 2021, 11, 2285. https://doi.org/10.3390/nano11092285
Pryjmaková J, Kaimlová M, Vokatá B, Hubáček T, Slepička P, Švorčík V, Siegel J. Bimetallic Nanowires on Laser-Patterned PEN as Promising Biomaterials. Nanomaterials. 2021; 11(9):2285. https://doi.org/10.3390/nano11092285
Chicago/Turabian StylePryjmaková, Jana, Markéta Kaimlová, Barbora Vokatá, Tomáš Hubáček, Petr Slepička, Václav Švorčík, and Jakub Siegel. 2021. "Bimetallic Nanowires on Laser-Patterned PEN as Promising Biomaterials" Nanomaterials 11, no. 9: 2285. https://doi.org/10.3390/nano11092285
APA StylePryjmaková, J., Kaimlová, M., Vokatá, B., Hubáček, T., Slepička, P., Švorčík, V., & Siegel, J. (2021). Bimetallic Nanowires on Laser-Patterned PEN as Promising Biomaterials. Nanomaterials, 11(9), 2285. https://doi.org/10.3390/nano11092285








