Metal-Based Nanomaterials: Work as Drugs and Carriers against Viral Infections
Abstract
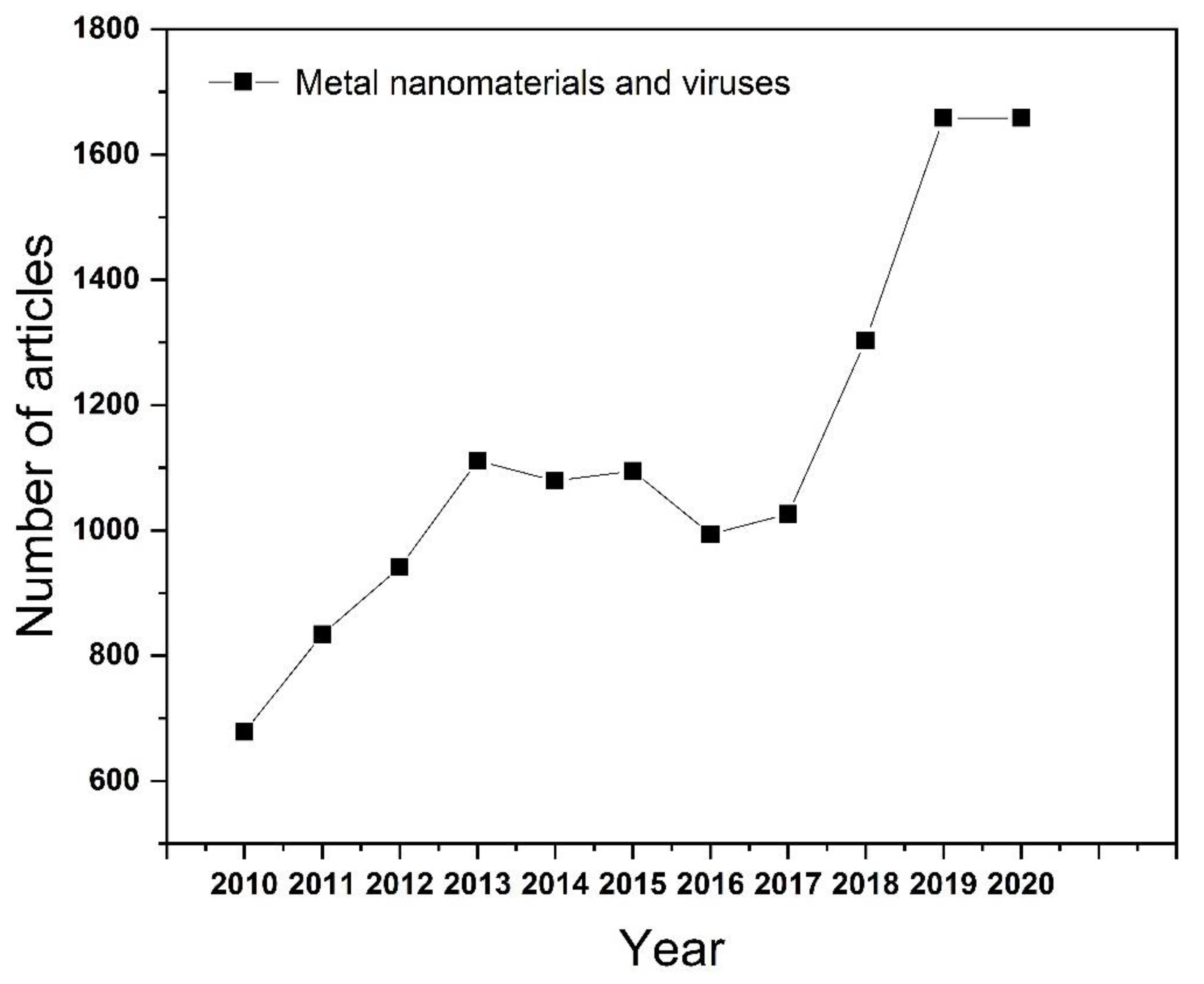
1. Introduction
2. Inhibition of Virus Infections
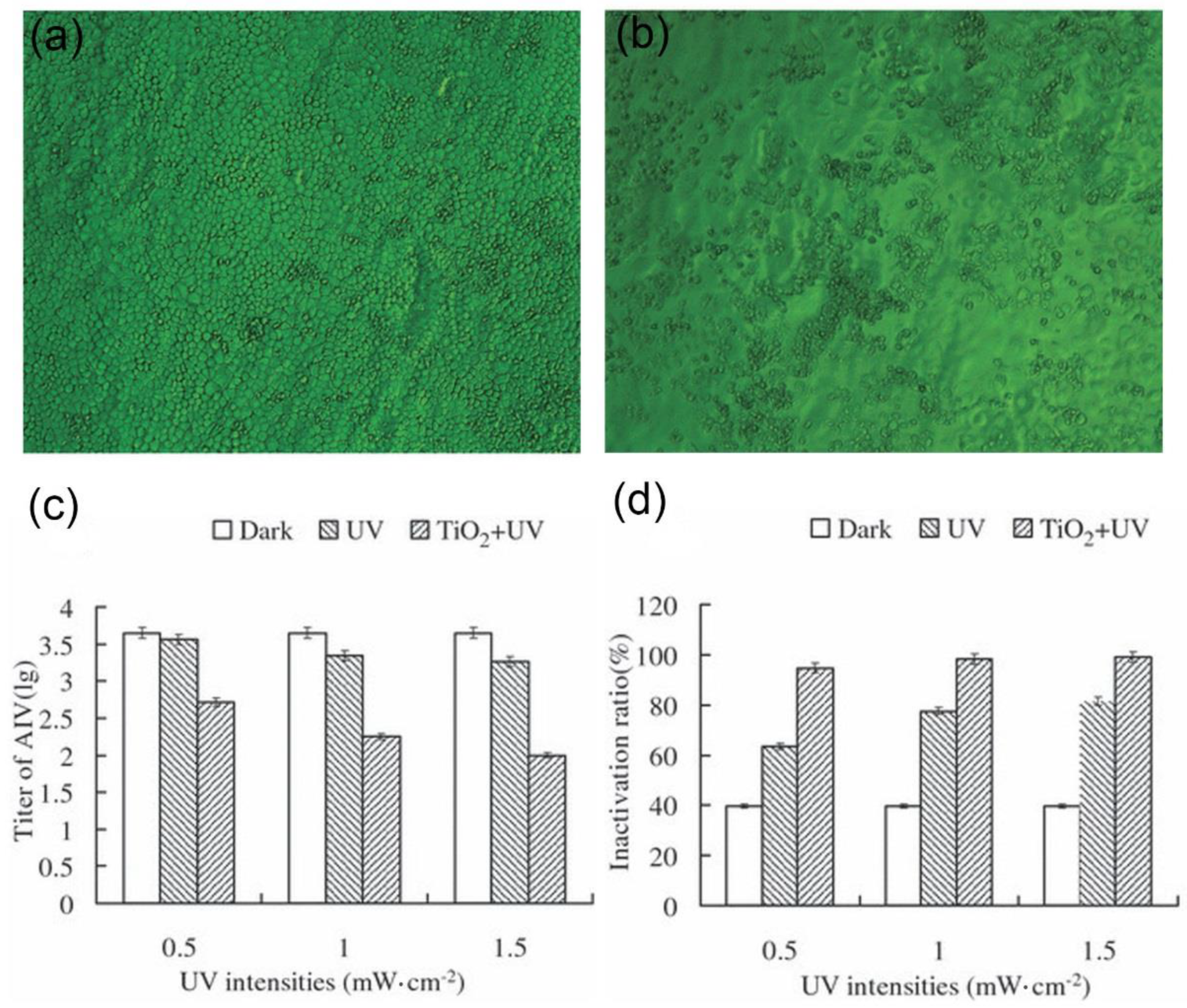
2.1. Direct Inactivation of the Virus
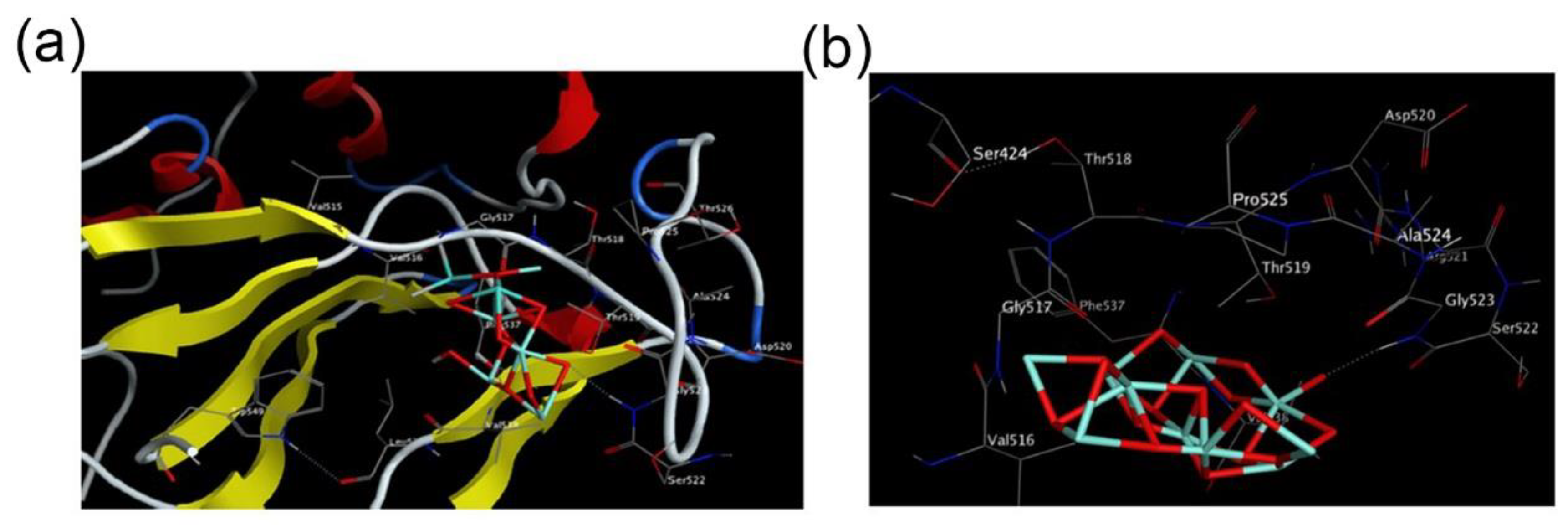
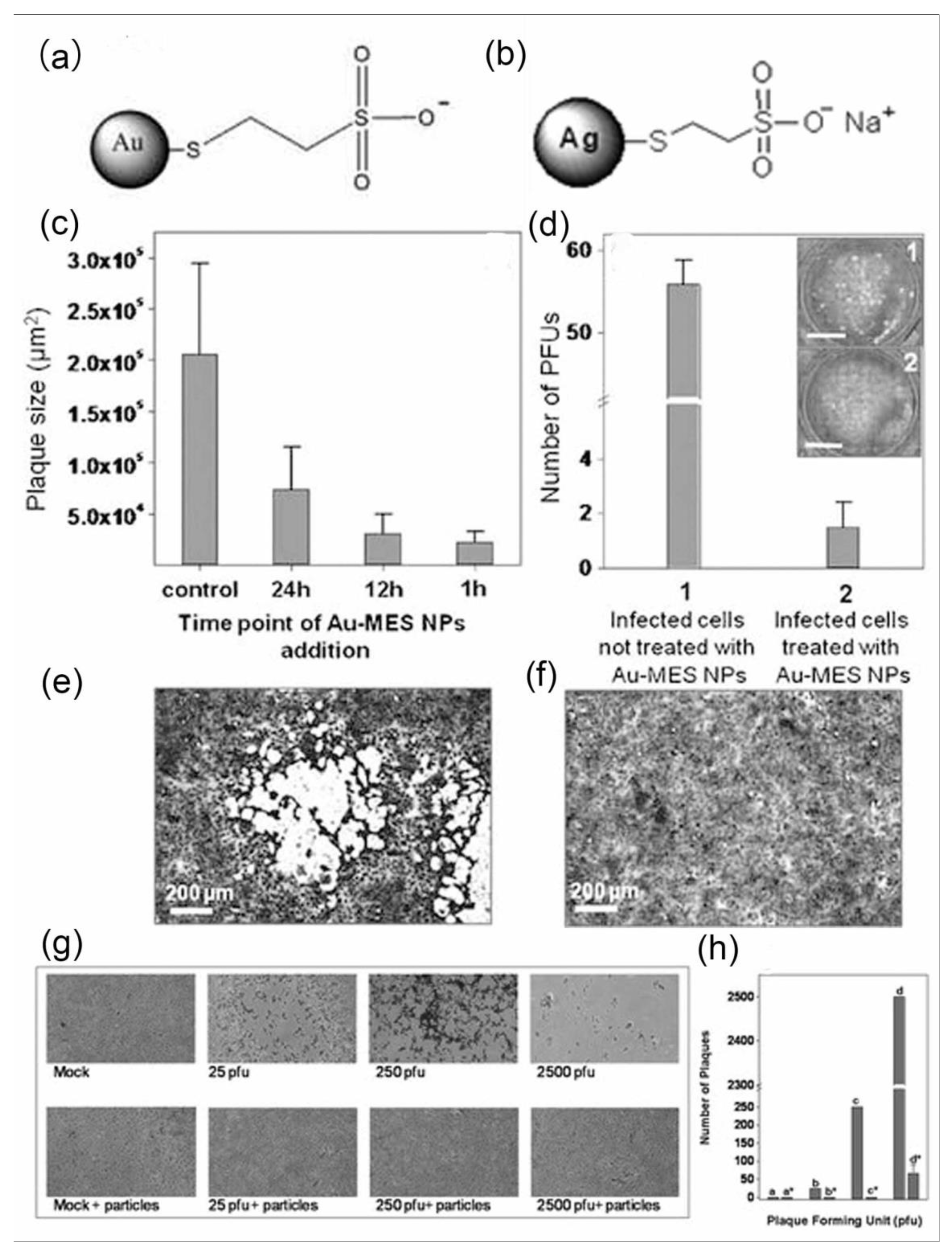
2.2. Inhibiting Virus Adsorption and Entry
2.3. Intracellular Virus Suppression
3. Loading Drug Synergy
3.1. Loading Drugs
3.2. Load Vaccine
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tan, M.; Bhadoria, A.S.; Cui, F. Estimating the proportion of people with chronic hepatitis B virus infection eligible for hepatitis B antiviral treatment worldwide: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2021, 6, 106–119. [Google Scholar] [CrossRef]
- Beerens, N.; Germeraad, E.A.; Venema, S.; Verheij, E.; Pritz-Verschuren, S.B.E.; Gonzales, J.L. Comparative pathogenicity and environmental transmission of recent highly pathogenic avian influenza H5 viruses. Emerg. Microbes Infect. 2021, 10, 97–108. [Google Scholar] [CrossRef]
- Cohen, M.S.; Chen, Y.Q.; McCauley, M.; Gamble, T.; Hosseinipour, M.C.; Kumarasamy, N.; Hakim, J.G.; Kumwenda, J.; Grinsztejn, B.; Pilotto, J.H.S.; et al. Prevention of HIV-1 infection with early antiretroviral therapy. N. Engl. J. Med. 2011, 365, 493–505. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.L.; Wang, X.G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.R.; Zhu, Y.; Li, B.; Huang, C.L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef]
- Shen, L.; Bao, N.; Yanagisawa, K.; Domen, K.; Gupta, A.; Grimes, C.A. Direct synthesis of ZnO nanoparticles by a solution-free mechanochemical reaction. Nanotechnology 2006, 17, 5117–5123. [Google Scholar] [CrossRef]
- Chen, Z.; Ren, W.; Gao, L.; Liu, B.; Pei, S.; Cheng, H.-M. Three-dimensional flexible and conductive interconnected graphene networks grown by chemical vapour deposition. Nat. Mater. 2011, 10, 424–428. [Google Scholar] [CrossRef]
- Kołodziejczak-Radzimska, A.; Jesionowski, T. Zinc Oxide—From Synthesis to Application: A Review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef]
- Pascu, A.; Stanciu, E.M.; Croitoru, C.; Roata, I.C.; Tierean, M.H. Carbon Nanoparticle-Supported Pd Obtained by Solar Physical Vapor Deposition. Adv. Mater. Sci. Eng. 2018, 2018, 1–7. [Google Scholar] [CrossRef]
- Rai, M.; Yadav, A.; Gade, A. CRC 675—Current Trends in Phytosynthesis of Metal Nanoparticles. Crit. Rev. Biotechnol. 2008, 28, 277–284. [Google Scholar] [CrossRef]
- Mohamed, H.; Afridi, S.; Khalil, A.T.; Zohra, T.; Alam, M.M.; Ikram, A.; Shinwari, Z.K.; Maaza, M. Phytosynthesis of BiVO4 nanorods using Hyphaene thebaica for diverse biomedical applications. AMB Express 2019, 9, 1–14. [Google Scholar] [CrossRef]
- Howes, P.D.; Chandrawati, R.; Stevens, M.M.; Stevens, M.M. Colloidal nanoparticles as advanced biological sensors. Science 2014, 346, 1247390. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.; Ouyang, S.; Bi, Y.; Umezawa, N.; Oshikiri, M.; Ye, J. Nano-photocatalytic materials: Possibilities and challenges. Adv. Mater. 2012, 24, 229–251. [Google Scholar] [CrossRef] [PubMed]
- Tale, B.; Nemade, K.R.; Tekade, P.V. Graphene based nano-composites for efficient energy conversion and storage in Solar cells and Supercapacitors: A Review. Polym. Plast. Technol. Mater. 2021, 60, 784–797. [Google Scholar]
- Liang, G.F.; Wang, H.J.; Shi, H.; Wang, H.T.; Zhu, M.X.; Jing, A.H.; Li, J.H.; Li, G.D. Recent progress in the development of upconversion nanomaterials in bioimaging and disease treatment. J. Nanobiotechnol. 2020, 18, 154. [Google Scholar] [CrossRef]
- Morris, D.; Ansar, M.; Speshock, J.; Ivanciuc, T.; Qu, Y.; Casola, A.; Garofalo, R.P. Antiviral and Immunomodulatory Activity of Silver Nanoparticles in Experimental RSV Infection. Viruses 2019, 11, 732. [Google Scholar] [CrossRef]
- Halder, A.; Das, S.; Ojha, D.; Chattopadhyay, D.; Mukherjee, A. Highly monodispersed gold nanoparticles synthesis and inhibition of herpes simplex virus infections. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 89, 413–421. [Google Scholar] [CrossRef]
- Mishra, Y.K.; Adelung, R.; Röhl, C.; Shukla, D.; Spors, F.; Tiwari, V. Virostatic potential of micro–nano filopodia-like ZnO structures against herpes simplex virus-1. Antivir. Res. 2011, 92, 305–312. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.; Ou, C.; He, J.; Liu, L.; Shan, H.; Lei, C.; Hui, D.S.C.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Maduray, K.; Parboosing, R. Metal Nanoparticles: A Promising Treatment for Viral and Arboviral Infections. Biol. Trace Elem. Res. 2021, 199, 3159–3176. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.; Deshmukh, S.D.; Ingle, A.; Gupta, I.; Galdiero, M.; Galdiero, S. Metal nanoparticles: The protective nanoshield against virus infection. Crit. Rev. Microbiol. 2016, 42, 46–56. [Google Scholar] [CrossRef]
- Marija, M.; Aleksandar, A.; Jelena, M.; Tatjana, K.; Nebojsa, A. Antimicrobial Nanoarchitectonics || Nanoparticles in Antiviral Therapy. Antimicrob. Nanoarchitecton. 2017, 14, 383–410. [Google Scholar]
- Baram-Pinto, D.; Shukla, S.; Gedanken, A.; Sarid, R. Inhibition of HSV-1 attachment, entry, and cell-to-cell spread by functionalized multivalent gold nanoparticles. Small 2010, 6, 1044–1050. [Google Scholar] [CrossRef]
- Baram-Pinto, D.; Shukla, S.; Perkas, N.; Gedanken, A.; Sarid, R. Inhibition of Herpes Simplex Virus Type 1 Infection by Silver Nanoparticles Capped with Mercaptoethane Sulfonate. Bioconjug. Chem. 2009, 20, 1497–1502. [Google Scholar] [CrossRef]
- Ghaffari, H.; Tavakoli, A.; Moradi, A.; Tabarraei, A.; Bokharaei-Salim, F.; Zahmatkeshan, M.; Farahmand, M.; Javanmard, D.; Kiani, S.J.; Esghaei, M.; et al. Inhibition of H1N1 influenza virus infection by zinc oxide nanoparticles: Another emerging application of nanomedicine. J. Biomed. Sci. 2019, 26, 70. [Google Scholar] [CrossRef]
- Cui, H.; Jiang, J.; Gu, W.; Sun, C.J.; Wu, D.L.; Yang, T.; Yang, G.C. Photocatalytic Inactivation Efficiency of Anatase Nano-TiO2 Sol on the H9N2 Avian Influenza Virus. Photochem. Photobiol. 2010, 86, 1135–1139. [Google Scholar] [CrossRef]
- Mazurkova, N.A.; Spitsyna, Y.E.; Shikina, N.V.; Ismagilov, Z.R.; Zagrebel’nyi, S.N.; Ryabchikova, E.I. Interaction of titanium dioxide nanoparticles with influenza virus. Nanotechnol. Russ. 2010, 5, 417–420. [Google Scholar] [CrossRef]
- Tavakoli, A.; Hashemzadeh, M.S. Inhibition of herpes simplex virus type 1 by copper oxide nanoparticles. J. Virol. Methods 2020, 275, 113688. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Park, H.H.; Kim, S.Y.; Kim, S.J.; Woo, K.; Ko, G. Antiviral Properties of Silver Nanoparticles on a Magnetic Hybrid Colloid. Appl. Environ. Microbiol. 2014, 80, 2343–2350. [Google Scholar] [CrossRef]
- Pfaff, F.; Glück, B.; Hoyer, T.; Rohländer, D.; Sauerbrei, A.; Zell, R. Tungsten carbide nanoparticles show a broad spectrum virucidal activity against enveloped and nonenveloped model viruses using a guideline-standardized in vitro test. Lett. Appl. Microbiol. 2019, 69, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Nayak, M.; Sahoo, G.C.; Pandey, K.; Sarkar, M.C.; Ansari, Y.; Das, V.N.R.; Topno, R.K.; Bhawna; Madhukar, M.; et al. Iron oxide nanoparticles based antiviral activity of H1N1 influenza A virus. J. Infect. Chemother. 2019, 25, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Abo-Zeid, Y.; Ismail, N.S.M.; Mclean, G.R.; Hamdy, N.M. A molecular docking study repurposes FDA approved iron oxide nanoparticles to treat and control COVID-19 infection. Eur. J. Pharm. Sci. 2020, 153, 105465. [Google Scholar] [CrossRef]
- Tyagi, M.; Rusnati, M.; Presta, M.; Giacca, M. Internalization of HIV-1 Tat Requires Cell Surface Heparan Sulfate Proteoglycans. J. Biol. Chem. 2001, 276, 3254–3261. [Google Scholar] [CrossRef]
- Vonnemann, J.; Sieben, C.; Wolff, C.; Ludwig, K.; Bottcher, C.; Herrmann, A.; Haag, R. Virus inhibition induced by polyvalent nanoparticles of different sizes. Nanoscale 2014, 6, 2353–2360. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Yeom, M.; Lee, T.; Kim, H.O.; Na, W.; Kang, A.; Lim, J.W.; Park, G.; Park, C.; Song, D.; et al. Porous gold nanoparticles for attenuating infectivity of influenza A virus. J. Nanobiotechnol. 2020, 18, 54. [Google Scholar] [CrossRef] [PubMed]
- Du, T.; Liang, J.; Dong, N.; Lu, J.; Fu, Y.Y.; Fang, L.R.; Xiao, S.B.; Han, H.Y. Glutathione-Capped Ag2S Nanoclusters Inhibit Coronavirus Proliferation through Blockage of Viral RNA Synthesis and Budding. ACS Appl. Mater. Interfaces. 2018, 10, 4369–4378. [Google Scholar] [CrossRef] [PubMed]
- Pang, C.; Zhang, P.; Mu, Y.; Ren, J.Z.; Zhao, B. Transformation and Cytotoxicity of Surface-Modified Silver Nanoparticles Undergoing Long-Term Aging. Nanomaterials 2020, 10, 2255. [Google Scholar] [CrossRef]
- Asok, A.; Kulkarni, A.R.; Gandhi, M.N. Defect-rich ZnO quantum dots as a potential multifunctional sunscreen and cosmetic active ingredient. Pure Appl. Chem. 2015, 87, 971–977. [Google Scholar] [CrossRef]
- Siddiqi, K.S.; Rahman, A.; Husen, A. Properties of Zinc Oxide Nanoparticles and Their Activity against Microbes. Nanoscale Res. Lett. 2018, 13, 141. [Google Scholar]
- Tavakoli, A.; Ataei-Pirkooh, A.; Sadeghi, G.M.M.; Bokharaei-Salim, F.; Sahrapour, P.; Kiani, S.J.; Moghoofei, M.; Farahmand, M.; Javanmard, D.; Monavari, S.H. Polyethylene glycol-coated zinc oxide nanoparticle an efficient nanoweapon to fight against herpes simplex virus type 1. Nanomedicine 2018, 13, 2675–2690. [Google Scholar] [CrossRef]
- Saeedi, T.; Alotaibi, H.F.; Prokopovich, P. Polymer colloids as drug delivery systems for the treatment of arthritis. Adv. Colloid Interface Sci. 2020, 285, 102273. [Google Scholar] [CrossRef]
- Patil-Sen, Y.; Torino, E.; De Sarno, F.; Ponsiglione, A.M.; Chhabria, V.; Ahmed, W.; Mercer, T. Biocompatible superparamagnetic core-shell nanoparticles for potential use in hyperthermia-enabled drug release and as an enhanced contrast agent. Nanotechnology 2020, 31, 375102. [Google Scholar] [CrossRef]
- Dos Apostolos, R.C.R.; Cipreste, M.F.; De Sousa, R.G.; De Sousa, E.M.B. Multifunctional hybrid nanosystems based on mesoporous silica and hydroxyapatite nanoparticles applied as potential nanocarriers for theranostic applications. J. Nanopart. Res. 2020, 22, 368. [Google Scholar] [CrossRef]
- Cai, W.; Chu, C.-C.; Liu, G.; Wang, Y.-X.J. Metal-Organic Framework-Based Nanomedicine Platforms for Drug Delivery and Molecular Imaging. Small 2015, 11, 4806–4822. [Google Scholar] [CrossRef]
- Li, Z.; Huang, H.; Tang, S.; Li, Y.; Yu, X.F.; Wang, H.Y.; Li, P.H.; Sun, Z.B.; Zhang, H.; Liu, C.L.; et al. Small gold nanorods laden macrophages for enhanced tumor coverage in photothermal therapy. Biomaterials 2016, 74, 144–154. [Google Scholar] [CrossRef]
- Zazo, H.; Colino, C.I.; Warzecha, K.T.; Hoss, M.; Gbureck, U.; Trautwein, C.; Tacke, F.; Lanao, J.M.; Bartneck, M. Gold Nanocarriers for Macrophage-Targeted Therapy of Human Immunodeficiency Virus. Macromol. Biosci. 2017, 17, 1600359. [Google Scholar] [CrossRef] [PubMed]
- Dungdung, R.; Bayal, M.; Valliyott, L.; Unniyampurath, U.; Nair, S.S.; Pilankatta, R. A slow, efficient and safe nanoplatform of tailored ZnS QD-mycophenolic acid conjugates for in vitro drug delivery against dengue virus 2 genome replication. Nanoscale Adv. 2020, 2, 5777–5789. [Google Scholar] [CrossRef]
- Nair, M.; Guduru, R.; Liang, P.; Hong, J.; Sagar, V.; Khizroev, S. Externally controlled on-demand release of anti-HIV drug using magneto-electric nanoparticles as carriers. Nat. Commun. 2013, 4, 1707. [Google Scholar] [CrossRef] [PubMed]
- Sadewasser, A.; Dietzel, E.; Michel, S.; Kluver, M.; Helfer, M.; Thelemann, T.; Klar, R.; Eickmann, M.; Becker, S.; Jaschinski, F. Anti-Niemann Pick C1 Single-Stranded Oligonucleotides with Locked Nucleic Acids Potently Reduce Ebola Virus Infection In Vitro. Mol. Ther. Nucleic Acids 2019, 16, 686–697. [Google Scholar] [CrossRef]
- Moelling, K.; Heinrich, J.; Matskevich, A.; Wittmer-Elzaouk, L.; Kwok, T. Silencing of viral RNAs by small double-stranded siDNA. Retrovirology 2009, 6, 58. [Google Scholar] [CrossRef][Green Version]
- Robaldo, L.; Berzal-Herranz, A.; Montserrat, J.M.; Iribarren, A.M. Activity of core-modified 10–23 DNAzymes against HCV. Chem. Med. Chem. 2014, 9, 2172–2177. [Google Scholar] [CrossRef]
- Ryoo, S.R.; Jang, H.; Kim, K.S.; Lee, B.; Kim, K.B.; Kim, Y.K.; Yeo, W.S.; Lee, Y.; Kim, D.E.; Min, D.H. Functional delivery of DNAzyme with iron oxide nanoparticles for hepatitis C virus gene knockdown. Biomaterials 2012, 33, 2754–2761. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liu, H.; Yang, S.H.; Wang, T.; Liu, C.; Cao, Y.C. Nanoparticle-based artificial RNA silencing machinery for antiviral therapy. Proc. Natl. Acad. Sci. USA 2012, 109, 12387–12392. [Google Scholar] [CrossRef] [PubMed]
- Kuschner, R.A.; Russell, K.L.; Abuja, M.; Bauer, K.M.; Faix, D.J.; Hait, H.; Henrick, J.; Jacobs, M.; Liss, A.; Lynch, J.A.; et al. A phase 3, randomized, double-blind, placebo-controlled study of the safety and efficacy of the live, oral adenovirus type 4 and type 7 vaccine, in U.S. military recruits. Vaccine 2013, 31, 2963–2971. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.K.; Pasetti, M.F.; Viret, J.-F.; Munoz, A.; Lagos, R.; Levine, M.M.; Campbell, J.D. A Clinical Study to Assess the Safety and Immunogenicity of Attenuated Measles Vaccine Administered Intranasally to Healthy Adults. Hum. Vaccin. 2014, 3, 54–58. [Google Scholar] [CrossRef][Green Version]
- Eswaran, S.P.; Praharaj, A.K.; Chander, Y.; Nagendra, A. Potency Titration of Oral Polio Vaccine by Estimation of Live Virus Content Using Tissue Culture Technique. Med. J. Armed Forces India 2003, 59, 105–107. [Google Scholar] [CrossRef][Green Version]
- Delore, V.; Salamand, C.; Marsh, G.; Arnoux, S.; Pepin, S.; Saliou, P. Long-term clinical trial safety experience with the inactivated split influenza vaccine, Vaxigrip®. Vaccine 2006, 24, 1586–1592. [Google Scholar] [CrossRef]
- Finke, S.; Karger, A.; Freuling, C.; Muller, T. Assessment of inactivated human rabies vaccines: Biochemical characterization and genetic identification of virus strains. Vaccine 2012, 30, 3603–3609. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhu, X.; Hu, Y.; Liang, M.; Sun, J.; Song, Y.F.; Yang, Q.; Ji, H.Q.; Zeng, G.; Song, L.F.; et al. Five-year antibody persistence in children after one dose of inactivated or live attenuated hepatitis A vaccine. Hum. Vaccin. Immunother. 2017, 13, 1–6. [Google Scholar] [CrossRef]
- Hernandez-Bernal, F.; Aguilar-Betancourt, A.; Aljovin, V.; Arias, G.; Valenzuela, C.; De Alejo, K.P.; Hernandez, K.; Oquendo, O.; Figueredo, N.; Figueroa, N.; et al. Comparison of four recombinant hepatitis B vaccines applied on an accelerated schedule in healthy adults. Hum. Vaccin. 2011, 7, 1026–1036. [Google Scholar] [CrossRef]
- Ellenberger, D.; Li, B.; Smith, J.; Yi, H.; Folks, T.; Robinson, H.; Butera, S. Optimization of a multi-gene HIV-1 recombinant subtype CRF02_AG DNA vaccine for expression of multiple immunogenic forms. Virology 2004, 319, 118–130. [Google Scholar] [CrossRef][Green Version]
- Huda, S.; Alam, M.A.; Sharma, P.K. Smart nanocarriers-based drug delivery for cancer therapy: An innovative and developing strategy. J. Drug Deliv. Sci. Technol. 2020, 60, 102018. [Google Scholar] [CrossRef]
- Wu, M.-X.; Yang, Y.-W. Metal-Organic Framework (MOF)-Based Drug/Cargo Delivery and Cancer Therapy. Adv. Mater. 2017, 29, 1606134. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, F.; Ju, E.; Liu, Z.; Chen, Z.W.; Ren, J.S.; Qu, X.G. Metal-Organic-Framework-Based Vaccine Platforms for Enhanced Systemic Immune and Memory Response. Adv. Funct. Mater. 2016, 26, 6454–6461. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, Q.; Wu, J.P.; Kirk, T.B.; Xu, J.K.; Liu, Z.H.; Xue, W. Reduction-Responsive Codelivery System Based on a Metal-Organic Framework for Eliciting Potent Cellular Immune Response. ACS Appl. Mater. Interfaces 2018, 10, 12463–12473. [Google Scholar] [CrossRef] [PubMed]
- Tao, W.; Hurst, B.L.; Shakya, A.K.; Uddin, M.J.; Ingrole, R.S.J.; Hernandez-Sanabria, M.; Arya, R.P.; Bimler, L.; Paust, S.; Tarbet, E.B.; et al. Consensus M2e peptide conjugated to gold nanoparticles confers protection against H1N1, H3N2 and H5N1 influenza A viruses. Antivir. Res. 2017, 141, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhu, W.; Luo, Y.; Wang, B.Z. Gold nanoparticles conjugating recombinant influenza hemagglutinin trimers and flagellin enhanced mucosal cellular immunity. Nanomedicine 2018, 14, 1349–1360. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Guzman, D.; Le Guen, P.; Villeret, B.; Sola, N.; Le Borgne, R.; Guyard, A.; Kemmel, A.; Crestani, B.; Sallenave, J.M.; Garcia-Verdugo, I. Silver nanoparticle-adjuvanted vaccine protects against lethal influenza infection through inducing BALT and IgA-mediated mucosal immunity. Biomaterials 2019, 217, 119308. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Mitchell, A.; Morcol, T.; Bell, S.J.D. Calcium Phosphate Nanoparticles Induce Mucosal Immunity and Protection against Herpes Simplex Virus Type 2. Clin. Diagn Lab. Immunol. 2002, 9, 1021–1024. [Google Scholar] [CrossRef]
- Volkova, M.A.; Irza, A.V.; Chvala, I.A.; Frolov, S.F.; Drygin, V.V.; Kapczynski, D.R. Adjuvant effects of chitosan and calcium phosphate particles in an inactivated Newcastle disease vaccine. Avian Dis. 2014, 58, 46–52. [Google Scholar] [CrossRef]
- Knuschke, T.; Sokolova, V.; Rotan, O.; Wadwa, M.; Tenbusch, M.; Hansen, W.; Staeheli, P.; Epple, M.; Buer, J.; Westendorf, A.M. Immunization with biodegradable nanoparticles efficiently induces cellular immunity and protects against influenza virus infection. J. Immunol. 2013, 190, 6221–6229. [Google Scholar] [CrossRef]
- Rincon, V.; Rodriguez-Huete, A.; Lopez-Arguello, S.; Ibarra-Molero, B.; Sanchez-Ruiz, J.M.; Harmsen, M.M.; Mateu, M.G. Identification of the structural basis of thermal lability of a virus provides a rationale for improved vaccines. Structure. 2014, 22, 1560–1570. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, M.M.; Fijten, H.P.; Westra, D.F.; Dekker, A. Stabilizing effects of excipients on dissociation of intact (146S) foot-and-mouth disease virions into 12S particles during storage as oil-emulsion vaccine. Vaccine 2015, 33, 2477–2484. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yang, Y.; Lin, X.; Li, Z.J.; Ma, G.H.; Su, Z.G.; Zhang, S.P. A Novel Particulate Delivery System Based on Antigen-Zn(2+) Coordination Interactions Enhances Stability and Cellular Immune Response of Inactivated Foot and Mouth Disease Virus. Mol. Pharm. 2020, 17, 2952–2963. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.; Bhide, Y.; Marsman, S.; Holtrop, M.; Meijerhof, T.; De Vries-Idema, J.; De Haan, A.; Huckriede, A. Monophosphoryl Lipid A-Adjuvanted Virosomes with Ni-Chelating Lipids for Attachment of Conserved Viral Proteins as Cross-Protective Influenza Vaccine. Biotechnol. J. 2018, 13, e1700645. [Google Scholar] [CrossRef]
- Luzuriaga, M.A.; Welch, R.P.; Dharmarwardana, M.; Benjamin, C.E.; Li, S.B.; Dharmarwardana, M.; Benjamin, C.E.; Li, S.B.; Shahrivarkevishahi, A.; Popal, S.; et al. Enhanced Stability and Controlled Delivery of MOF-Encapsulated Vaccines and Their Immunogenic Response In Vivo. ACS Appl. Mater. Interfaces 2019, 11, 9740–9746. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, S.; Shahzad, K.; Mushtaq, S.; Ali, I.; Rafe, M.H.; Fazal-Ul-Karim, S.M. Antibacterial and antiviral potential of colloidal Titanium dioxide (TiO2) nanoparticles suitable for biological applications. Mater. Res. Express 2019, 6, 105409. [Google Scholar] [CrossRef]
- Bekele, A.Z.; Gokulan, K.; Williams, K.M.; Khare, S. Dose and Size-Dependent Antiviral Effects of Silver Nanoparticles on Feline Calicivirus, a Human Norovirus Surrogate. Foodborne Pathog. Dis. 2016, 13, 239–244. [Google Scholar] [CrossRef]
- Imai, K.; Ogawa, H.; Bui, V.N.; Inoue, H.; Fukuda, J.; Ohba, M.; Yamamoto, Y.; Nakamura, K. Inactivation of high and low pathogenic avian influenza virus H5 subtypes by copper ions incorporated in zeolite-textile materials. Antivir. Res. 2012, 93, 225–233. [Google Scholar] [CrossRef]
- Shionoiri, N.; Sato, T.; Fujimori, Y.; Nakayama, T.; Nemoto, M.; Matsunaga, T.; Tanaka, T. Investigation of the antiviral properties of copper iodide nanoparticles against feline calicivirus. J. Biosci. Bioeng. 2012, 113, 580–586. [Google Scholar] [CrossRef]
- Fujimori, Y.; Sato, T.; Hayata, T.; Nagao, T.; Nakayama, M.; Nakayama, T.; Sugamata, R.; Suzuki, K. Novel Antiviral Characteristics of Nanosized Copper(I) Iodide Particles Showing Inactivation Activity against 2009 Pandemic H1N1 Influenza Virus. Appl. Environ. Microbiol. 2012, 78, 951–955. [Google Scholar] [CrossRef]
- Moon, E.W.; Lee, H.W.; Rok, J.H.; Ha, J.H. Photocatalytic inactivation of viral particles of human norovirus by Cu-doped TiO2 non-woven fabric under UVA-LED wavelengths. Sci. Total Environ. 2020, 749, 141574. [Google Scholar] [CrossRef]
- Zhou, Y.; Jiang, X.; Tong, T.; Fang, L.R.; Wu, Y.; Liang, J.; Xiao, S. High antiviral activity of mercaptoethane sulfonate functionalized Te/BSA nanostars against arterivirus and coronavirus. RSC Adv. 2020, 10, 14161–14169. [Google Scholar] [CrossRef]
- Trigilio, J.; Antoine, T.E.; Paulowicz, I.; Mishra, Y.K.; Adelung, R.; Shukla, D. Tin Oxide Nanowires Suppress Herpes Simplex Virus-1 Entry and Cell-to-Cell Membrane Fusion. PLoS ONE 2012, 7, e48147. [Google Scholar] [CrossRef]
- Jeremiah, S.S.; Miyakawa, K.; Morita, T.; Yamaoka, Y.; Ryo, A. Potent antiviral effect of silver nanoparticles on SARS-CoV-2. Biochem. Biophys. Res. Commun. 2020, 533, 195–200. [Google Scholar] [CrossRef]
- Soto, E.R.; O’connell, O.; Dikengil, F.; Peters, P.J.; Clapham, P.R.; Ostroff, G.R. Targeted Delivery of Glucan Particle Encapsulated Gallium Nanoparticles Inhibits HIV Growth in Human Macrophages. J. Drug Deliv. 2016, 2016, 8520629. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-R.; Britigan, B.E.; Narayanasamy, P. Ga(III) Nanoparticles Inhibit Growth of both Mycobacterium tuberculosis and HIV and Release of Interleukin-6 (IL-6) and IL-8 in Coinfected Macrophages. Antimicrob. Agents Chemother. 2017, 61, e02505–e02516. [Google Scholar] [CrossRef] [PubMed]
- Du, T.; Zhang, J.; Li, C.; Song, T.; Liu, J.F.; Du, X.J.; Wang, S. Gold/Silver Hybrid Nanoparticles with Enduring Inhibition of Coronavirus Multiplication through Multisite Mechanisms. Bioconj. Chem. 2020, 31, 2553–2563. [Google Scholar] [CrossRef]
- Agnihothram, S.; Mullis, L.; Townsend, T.; Watanabe, F.; Mustafa, T.; Biris, A.; Manjanatha, M.; Azevedo, M. Titanium dioxide nanoparticles evoke proinflammatory response during Murine norovirus infection despite having minimal effects on virus replication. Int. J. Nanotechnol. Med. Eng. 2016, 1, 63–73. [Google Scholar]
- Du, T.; Cai, K.; Han, H.; Fang, L.; Liang, J.; Xiao, S. Probing the interactions of CdTe quantum dots with pseudorabies virus. Sci. Rep. 2015, 5, 16403. [Google Scholar] [CrossRef]
- Li, Y.; Lin, Z.; Zhao, M.; Xu, T.; Wang, C.; Hua, L.; Wang, H.; Xia, H.; Zhu, B. Silver Nanoparticle Based Codelivery of Oseltamivir to Inhibit the Activity of the H1N1 Influenza Virus through ROS-Mediated Signaling Pathways. ACS Appl. Mater. Interfaces 2016, 8, 24385–24393. [Google Scholar] [CrossRef]
- Levina, A.S.; Repkova, M.N.; Zarytova, V.F.; Mazurkova, N.A. TiO2∼DNA nanocomposites as efficient site-specific antiviral agents against influenza A virus in cell culture. In Proceedings of the 2015 IEEE 15th International Conference on Nanotechnology (IEEE-NANO), Rome, Italy, 27–30 July 2015; pp. 1509–1512. [Google Scholar]
- Moongraksathum, B.; Chien, M.Y.; Chen, Y.W. Antiviral and Antibacterial Effects of Silver-Doped TiO2 Prepared by the Peroxo Sol-Gel Method. J. Nanosci. Nanotechnol. 2019, 19, 7356–7362. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.; Zhou, J.; Song, Z.; Mei, H.; Zhou, M.; Fu, Z.; Han, H.; Zhao, L. Aptamer and RVG functionalized gold nanorods for targeted photothermal therapy of neurotropic virus infection in the mouse brain. Chem. Eng. J. 2021, 411, 128557. [Google Scholar] [CrossRef]


| Materials | Size/nm | Synthetic Method | Antiviral Mechanism | Detection Methods | Antiviral Types | Ref. |
|---|---|---|---|---|---|---|
| TiO2 | 8 | Sonochemistry | Direct inactivating | Hemagglutination assay (HA) | Newcastle virus | [76] |
| TiO2 | 4–5 | Hydrolysis, Calcination | TCID50 | H3N2 | [26] | |
| TiO2 | 50 | Hydrothermal | SEM, Cytopathic effect (CPE), HA | H9N2 | [25] | |
| Fe2O3/Fe3O4 | / | / | Protein conformation analysis | COVID-19, Hepatitis C virus (HCV) | [31] | |
| Ag | 10, 75, 110 | Commercialization | TCID50 | Feline Calicivirus (FCV) | [77] | |
| Cu (II)-zeolite | / | Mechanochemistry | TCID50, HA/NA, RT-PCR, indirect fluorescent antibody (IFA) | H5N1, H5N3 | [78] | |
| CuI | 100–400 | Commercialization | Plaque assay | FCV | [79] | |
| CuI | 160 | Commercialization | Plaque assay, SDS-PAGE | H1N1 | [80] | |
| Cu-doped TiO2 | 20 | Sol-gel | qRT-PCR | Human norovirus | [81] | |
| Te/BSA | 57 ± 7.6 | Redox | TCID50, IFA | Porcine epidemic diarrhea virus | [82] | |
| SnO2 | 50–100 | Flame transport synthesis | Plaque assay, fluorescence imaging | Herpes simplex virus-1 | [83] | |
| ZnO | 100–500 | Flame transport synthesis | Reporter Lysis assay | Herpes simplex virus-1 | [17] | |
| Ag | 10 | Commercialization | TCID50, RT-qPCR, IFA | SARS-CoV-2 | [84] | |
| Ga | / | / | Plaque assay, fluorescence imaging, | HIV | [85] | |
| PEGylated ZnO-NPs | 16–20 | Commercialization | Intracellular inhibition (replication) | TCID50 | H1N1 | [24] |
| CuO | 40 | Commercialization | TCID50, IFA | Herpes simplex virus-1 | [27] | |
| Fe3O4 | 10–15 | Hydrothermal | TCID50, RT-PCR | H1N1 | [30] | |
| Ga(III) Nanoparticles | Mechanochemistry | TCID50 | HIV | [86] | ||
| Au@Ag | Seed-mediated growth | TCID50 | Coronavirus | [87] | ||
| TiO2 | 20–25 | / | RT-PCR, TCID50, alkaline single-cell gel electrophoresis (comet) | Noroviruses | [88] | |
| Ag2S | 3.7, 5.3 | Sol-gel | Intracellular inhibition (replication and release) | TCID50 | Porcine epidemic diarrhea virus | [35] |
| CdTe | 3.2 ± 0.8 | Hydrothermal | TCID50 | Pseudorabies virus | [89] | |
| Ag@OTV | 2 | Sonochemistry | Drug-loading | Caspase-3 activity, TEM | H1N1 | [90] |
| TiO2~DNA | ~5 | Hydrolysis synthesis | TCID50 | Influenza A virus | [91] | |
| Ag/TiO2 | 12–32 | Sol-gel | / | H1N1, enterovirus | [92] | |
| Au | 35 | Seed-mediated growth | Immunohistochemistry | Neurotropic virus | [93] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Yue, L.; Yang, Z.; Miao, Y.; Ouyang, R.; Hu, Y. Metal-Based Nanomaterials: Work as Drugs and Carriers against Viral Infections. Nanomaterials 2021, 11, 2129. https://doi.org/10.3390/nano11082129
Yang J, Yue L, Yang Z, Miao Y, Ouyang R, Hu Y. Metal-Based Nanomaterials: Work as Drugs and Carriers against Viral Infections. Nanomaterials. 2021; 11(8):2129. https://doi.org/10.3390/nano11082129
Chicago/Turabian StyleYang, Junlei, Lihuan Yue, Zhu Yang, Yuqing Miao, Ruizhuo Ouyang, and Yihong Hu. 2021. "Metal-Based Nanomaterials: Work as Drugs and Carriers against Viral Infections" Nanomaterials 11, no. 8: 2129. https://doi.org/10.3390/nano11082129
APA StyleYang, J., Yue, L., Yang, Z., Miao, Y., Ouyang, R., & Hu, Y. (2021). Metal-Based Nanomaterials: Work as Drugs and Carriers against Viral Infections. Nanomaterials, 11(8), 2129. https://doi.org/10.3390/nano11082129






