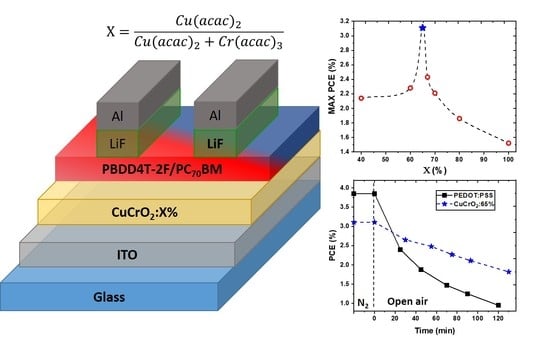
Optimized Stoichiometry for CuCrO2 Thin Films as Hole Transparent Layer in PBDD4T-2F:PC70BM Organic Solar Cells
Abstract
:1. Introduction
2. Material and Methods
2.1. Deposition of CuCrO2 Thin Films
2.2. Characterization of Thin Films
2.3. OSC Fabrication and Characterization
3. Results and Discussion
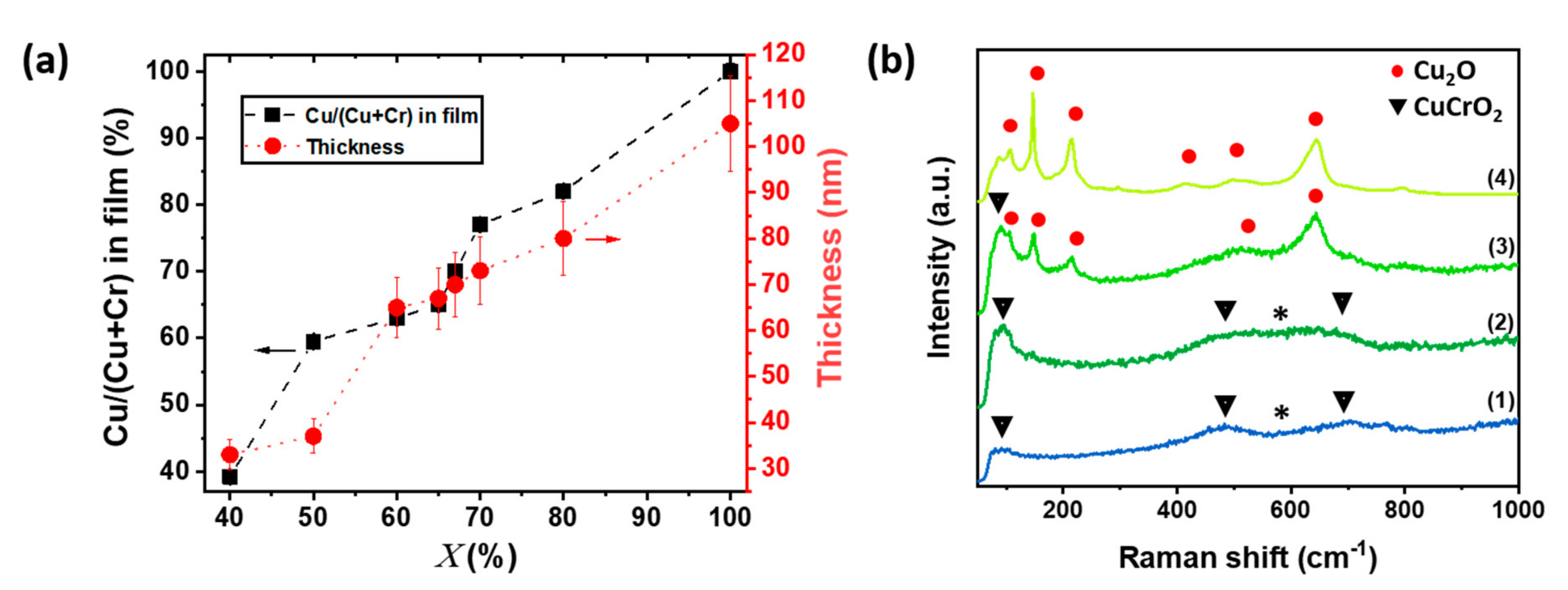
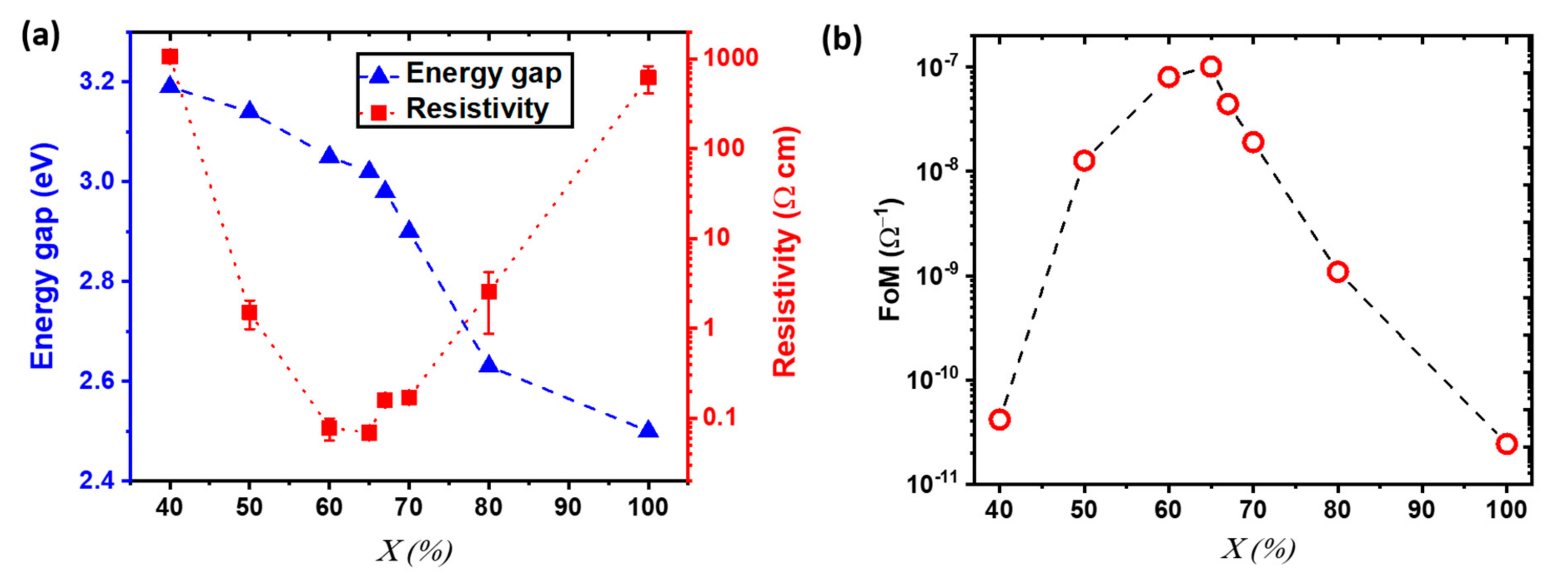
3.1. Characterization of Out of Stoichiometry CuCrO2 Thin Films
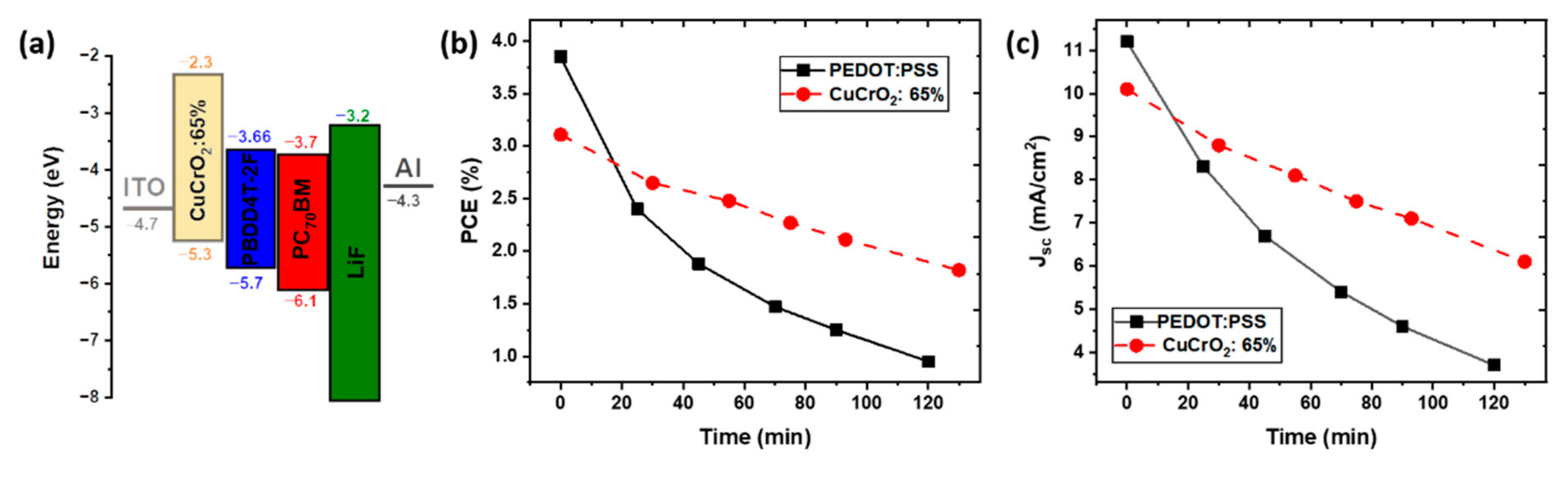
3.2. Integration of CuCrO2 Thin Films out of Stoichiometry as HTL in OSC
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bagher, A.M. Comparison of Organic Solar Cells and Inorganic Solar Cells. Int. J. Renew. Sustain. Energy 2014, 3, 53–58. [Google Scholar] [CrossRef]
- Cui, Y.; Yao, H.; Zhang, J.; Xian, K.; Zhang, T.; Hong, L.; Wang, Y.; Xu, Y.; Ma, K.; An, C.; et al. Single-Junction Organic Photovoltaic Cells with Approaching 18% Efficiency. Adv. Mater. 2020, 32, 1–7. [Google Scholar] [CrossRef]
- Colsmann, A.; Reinhard, M.; Kwon, T.H.; Kayser, C.; Nickel, F.; Czolk, J.; Lemmer, U.; Clark, N.; Jasieniak, J.; Holmes, A.B.; et al. Inverted Semi-Transparent Organic Solar Cells with Spray Coated, Surfactant Free Polymer Top-Electrodes. Sol. Energy Mater. Sol. Cells 2012, 98, 118–123. [Google Scholar] [CrossRef]
- Lian, H.; Jun, N.; Bolag, A.; Hexig, A.; Gerile, N.; Tegus, O.; Lin, S. Optimization of PEDOT:PSS Hole Transport Layer toward the Organic Solar Cells with High Fill Factor. Solid State Phenom. 2019, 288, 113–118. [Google Scholar] [CrossRef]
- Wakizaka, D.; Fushimi, T.; Ohkita, H.; Ito, S. Hole Transport in Conducting Ultrathin Films of PEDOT/PSS Prepared by Layer-by-Layer Deposition Technique. Polymer 2004, 45, 8561–8565. [Google Scholar] [CrossRef]
- Kemerink, M.; Timpanaro, S.; de Kok, M.M.; Meulenkamp, E.A.; Touwslager, F.J. Three-Dimensional Inhomogeneities in PEDOT:PSS Films. J. Phys. Chem. B 2004, 108, 18820–18825. [Google Scholar] [CrossRef]
- Huang, Q.; Evmenenko, G.A.; Dutta, P.; Lee, P.; Armstrong, N.R.; Marks, T.J. Covalently Bound Hole-Injecting Nanostructures. Systematics of Molecular Architecture, Thickness, Saturation, and Electron-Blocking Characteristics on Organic Light-Emitting Diode Luminance, Turn-on Voltage, and Quantum Efficiency. J. Am. Chem. Soc. 2005, 127, 10227–10242. [Google Scholar] [CrossRef] [PubMed]
- Anand, A.; Madalaimuthu, J.P.; Schaal, M.; Otto, F.; Gruenewald, M.; Alam, S.; Fritz, T.; Schubert, U.S.; Hoppe, H. Why Organic Electronic Devices Comprising PEDOT:PSS Electrodes Should Be Fabricated on Metal Free Substrates. ACS Appl. Electron. Mater. 2021, 3, 929–943. [Google Scholar] [CrossRef]
- Bovill, E.; Yi, H.; Iraqi, A.; Lidzey, D.G. The Fabrication of Polyfluorene and Polycarbazole-Based Photovoltaic Devices Using an Air-Stable Process Route. Appl. Phys. Lett. 2014, 105, 1–6. [Google Scholar] [CrossRef]
- Gautam, K.; Singh, I.; Bhatnagar, P.K.; Peta, K.R. Single Mode Waveguiding Effect of ZnO Nanorods to Enhance the Luminance of Conjugated Polymer Based Light Emitting Diodes. J. Lumin. 2018, 204, 59–63. [Google Scholar] [CrossRef]
- Rasool, A.; Amiruddin, R.; Kossar, S.; Santhosh Kumar, M.C. Realization of In:ZnO/PEDOT:PSS Based Multifunctional Device for Ultraviolet (UV) Light Detection and Resistive Switching Memory Applications. J. Appl. Phys. 2020, 128, 044503. [Google Scholar] [CrossRef]
- Lee, S.J.; Pil Kim, H.; Mohd Yusoff, A.R.B.; Jang, J. Organic Photovoltaic with PEDOT:PSS and V2O5 Mixture as Hole Transport Layer. Sol. Energy Mater. Sol. Cells 2014, 120, 238–243. [Google Scholar] [CrossRef]
- De Jong, M.P.; van Ijzendoorn, L.J.; de Voigt, M.J.A. Stability of the Interface between Indium-Tin-Oxide and Poly(3,4-Ethylenedioxythiophene)/ Poly(Styrenesulfonate) in Polymer Light-Emitting Diodes. Appl. Phys. Lett. 2000, 77, 2255–2257. [Google Scholar] [CrossRef]
- Shrotriya, V.; Li, G.; Yao, Y.; Chu, C.W.; Yang, Y. Transition Metal Oxides as the Buffer Layer for Polymer Photovoltaic Cells. Appl. Phys. Lett. 2006, 88, 2–4. [Google Scholar] [CrossRef] [Green Version]
- Yu, S.; Yang, W.; Li, L.; Zhang, W. Improved Chemical Stability of ITO Transparent Anodes with a SnO2 Buffer Layer for Organic Solar Cells. Sol. Energy Mater. Sol. Cells 2016, 144, 652–656. [Google Scholar] [CrossRef]
- Norrman, K.; Madsen, M.V.; Gevorgyan, S.A.; Krebs, F.C. Degradation Patterns in Water and Oxygen of an Inverted Polymer Solar Cell. J. Am. Chem. Soc. 2010, 132, 16883–16892. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Uddin, A. Progress in Stability of Organic Solar Cells. Adv. Sci. 2020, 7, 1903259. [Google Scholar] [CrossRef] [Green Version]
- Awada, H.; Mattana, G.; Tournebize, A.; Rodriguez, L.; Flahaut, D.; Vellutini, L.; Lartigau-Dagron, C.; Billon, L.; Bousquet, A.; Chambon, S. Surface Engineering of ITO Electrode with a Functional Polymer for PEDOT:PSS-Free Organic Solar Cells. Org. Electron. 2018, 57, 186–193. [Google Scholar] [CrossRef]
- Stadler, A. Transparent Conducting Oxides—An Up-To-Date Overview. Materials 2012, 5, 661–683. [Google Scholar] [CrossRef] [PubMed]
- Ratcliff, E.L.; Zacher, B.; Armstrong, N.R. Selective Interlayers and Contacts in Organic Photovoltaic Cells. J. Phys. Chem. Lett. 2011, 2, 1337–1350. [Google Scholar] [CrossRef]
- Irwin, M.D.; Buchholz, D.B.; Hains, A.W.; Chang, R.P.H.; Marks, T.J. P-Type Semiconducting Nickel Oxide as an Efficiency-Enhancing Anode Interfacial Layer in Polymer Bulk-Heterojunction Solar Cells. Proc. Natl. Acad. Sci. USA 2008, 105, 2783–2787. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Lee, Y.; Hsu, J.W.P. Sub-10 Nm Copper Chromium Oxide Nanocrystals as a Solution Processed p-Type Hole Transport Layer for Organic Photovoltaics. J. Mater. Chem. C 2016, 4, 3607–3613. [Google Scholar] [CrossRef]
- Dunlap-Shohl, W.A.; Daunis, T.B.; Wang, X.; Wang, J.; Zhang, B.; Barrera, D.; Yan, Y.; Hsu, J.W.P.; Mitzi, D.B. Room-Temperature Fabrication of a Delafossite CuCrO2 Hole Transport Layer for Perovskite Solar Cells. J. Mater. Chem. A 2018, 6, 469–477. [Google Scholar] [CrossRef]
- Gil, B.; Kim, J.; Yun, A.J.; Park, K.; Cho, J.; Park, M.; Park, B. CuCrO2 Nanoparticles Incorporated into PTAA as a Hole Transport Layer for 85 °C and Light Stabilities in Perovskite Solar Cells. Nanomaterials 2020, 10, 1669. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ibarra, V.; Barrera, D.; Xu, L.; Lee, Y.J.; Hsu, J.W.P. Solution Synthesized p-Type Copper Gallium Oxide Nanoplates as Hole Transport Layer for Organic Photovoltaic Devices. J. Phys. Chem. Lett. 2015, 6, 1071–1075. [Google Scholar] [CrossRef]
- Jeong, S.; Seo, S.; Shin, H. P-Type CuCrO2 Particulate Films as the Hole Transporting Layer for CH3NH3PbI3 Perovskite Solar Cells. RSC Adv. 2018, 8, 27956–27962. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Wang, H.; Zhu, H.; Chueh, C.C.; Chen, W.; Yang, S.; Jen, A.K.Y. Low-Temperature Solution-Processed CuCrO2 Hole-Transporting Layer for Efficient and Photostable Perovskite Solar Cells. Adv. Energy Mater. 2018, 8, 1702762. [Google Scholar] [CrossRef]
- Zhao, D.W.; Liu, P.; Sun, X.W.; Tan, S.T.; Ke, L.; Kyaw, A.K.K. An Inverted Organic Solar Cell with an Ultrathin Ca Electron-Transporting Layer and MoO3 Hole-Transporting Layer. Appl. Phys. Lett. 2009, 95, 200–203. [Google Scholar] [CrossRef]
- Nattestad, A.; Mozer, A.J.; Fischer, M.K.R.; Cheng, Y.-B.; Mishra, A.; Bäuerle, P.; Bach, U. Highly Efficient Photocathodes for Dye-Sensitized Tandem Solar Cells. Nat. Mater. 2009, 9, 31–35. [Google Scholar] [CrossRef]
- Kaya, I.C.; Akin, S.; Akyildiz, H.; Sonmezoglu, S. Highly Efficient Tandem Photoelectrochemical Solar Cells Using Coumarin6 Dye-Sensitized CuCrO2 Delafossite Oxide as Photocathode. Sol. Energy 2018, 169, 196–205. [Google Scholar] [CrossRef]
- Kavan, L. Electrochemistry and Dye-Sensitized Solar Cells. Curr. Opin. Electrochem. 2017, 2, 88–96. [Google Scholar] [CrossRef]
- Zhang, B.; Thampy, S.; Dunlap-Shohl, W.A.; Xu, W.; Zheng, Y.; Cao, F.-Y.; Cheng, Y.-J.; Malko, A.V.; Mitzi, D.B.; Hsu, J.W.P. Mg Doped CuCrO2 as Efficient Hole Transport Layers for Organic and Perovskite Solar Cells. Nanomaterials 2019, 9, 1311. [Google Scholar] [CrossRef] [Green Version]
- Bottiglieri, L.; Resende, J.; Weber, M.; Chaix-Pluchery, O.; Jiménez, C.; Deschanvres, J.-L. Out of Stoichiometry CuCrO2 Films as a Promising p-Type TCO for Transparent Electronics. Mater. Adv. 2021, 2, 4721–4732. [Google Scholar] [CrossRef]
- Crêpellière, J.; Popa, P.L.; Bahlawane, N.; Leturcq, R.; Werner, F.; Siebentritt, S.; Lenoble, D. Transparent Conductive CuCrO2 Thin Films Deposited by Pulsed Injection Metal Organic Chemical Vapor Deposition: Up-Scalable Process Technology for an Improved Transparency/Conductivity Trade-Off. J. Mater. Chem. C 2016, 4, 4278–4287. [Google Scholar] [CrossRef]
- Farrell, L.; Norton, E.; Smith, C.M.; Caffrey, D.; Shvets, I.V.; Fleischer, K. Synthesis of Nanocrystalline Cu Deficient CuCrO2—A High Figure of Merit p-Type Transparent Semiconductor. J. Mater. Chem. C 2016, 4, 126–134. [Google Scholar] [CrossRef] [Green Version]
- Rastogi, A.C.; Lim, S.H.; Desu, S.B. Structure and Optoelectronic Properties of Spray Deposited Mg Doped P-CuCrO2 Semiconductor Oxide Thin Films. J. Appl. Phys. 2008, 104, 023712. [Google Scholar] [CrossRef]
- Lunca-Popa, P.; Botsoa, J.; Bahri, M.; Crêpellière, J.; Desgardin, P.; Audinot, J.N.; Wirtz, T.; Arl, D.; Ersen, O.; Barthe, M.F.; et al. Tuneable Interplay between Atomistic Defects Morphology and Electrical Properties of Transparent P-Type Highly Conductive off-Stoichiometric Cu-Cr-O Delafossite Thin Films. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Yu, R.S.; Wu, C.M. Characteristics of P-Type Transparent Conductive CuCrO2 Thin Films. Appl. Surf. Sci. 2013, 282, 92–97. [Google Scholar] [CrossRef]
- Wang, J.; Zheng, P.; Li, D.; Deng, Z.; Dong, W.; Tao, R.; Fang, X. Preparation of Delafossite-Type CuCrO2 Films by Sol-Gel Method. J. Alloys Compd. 2011, 509, 5715–5719. [Google Scholar] [CrossRef]
- Wang, J.; Daunis, T.B.; Cheng, L.; Zhang, B.; Kim, J.; Hsu, J.W.P. Combustion Synthesis of P-Type Transparent Conducting CuCrO2+x and Cu:CrOx Thin Films at 180 °C. ACS Appl. Mater. Interfaces 2018, 10, 3732–3738. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, C.V.; Migot, S.; Alhussein, A.; Jiménez, C.; Schuster, F.; Ghanbaja, J.; Sanchette, F. Structural and Microstructural Analysis of Bifunctional TiO2/Al-Zr Thin Film Deposited by Hybrid Process. Thin Solid Films 2020, 709, 138255. [Google Scholar] [CrossRef]
- Mincuzzi, G.; Palma, A.L.; DiCarlo, A.; Brown, T.M. Laser Processing in the Manufacture of Dye-Sensitized and Perovskite Solar Cell Technologies. ChemElectroChem 2016, 3, 9–30. [Google Scholar] [CrossRef]
- Nourdine, A.; Abdelli, M.; Charvin, N.; Flandin, L. Custom Synthesis of ZnO Nanowires for Efficient Ambient Air-Processed Solar Cells. ACS Omega 2021. [Google Scholar] [CrossRef]
- Ye, L.; Jing, Y.; Guo, X.; Sun, H.; Zhang, S.; Zhang, M.; Huo, L.; Hou, J. Remove the Residual Additives toward Enhanced Efficiency with Higher Reproducibility in Polymer Solar Cells. J. Phys. Chem. C 2013, 117, 14920–14928. [Google Scholar] [CrossRef]
- Yan, Y.; Li, W.; Cai, F.; Cai, J.; Huang, Z.; Gurney, R.S.; Liu, D.; Lidzey, D.G.; Pearson, A.J.; Wang, T. Correlating Nanoscale Morphology with Device Performance in Conventional and Inverted PffBT4T-2OD:PC71BM Polymer Solar Cells. ACS Appl. Energy Mater. 2018, 1, 3505–3512. [Google Scholar] [CrossRef]
- Han, M.; Wang, J.; Deng, Q.; Wang, J.; Li, W.; Zhang, P.; Li, C.; Hu, Z. Effect of Annealing Temperature on Structural, Optoelectronic Properties and Interband Transitions of CuCrO2 Nanocrystalline Films Prepared by the Sol-Gel Method. J. Alloys Compd. 2015, 647, 1028–1034. [Google Scholar] [CrossRef]
- Han, M.; Lu, Y.; Liu, Y.; Hu, Z.; Chen, G.; Jiang, K.; Zhang, J.; Li, W. Raman Scattering Measurements of Phonon Anharmonicity in the Delafossite CuGa1-xCrxO2 (0 ≤ x ≤ 1) Films. J. Raman Spectrosc. 2020, 51, 851–859. [Google Scholar] [CrossRef]
- Resende, J.; Chaix-Pluchery, O.; Rovezzi, M.; Malier, Y.; Renevier, H.; Nguyen, N.D.; Deschanvres, J.L.; Jiménez, C. Resilience of Cuprous Oxide under Oxidizing Thermal Treatments via Magnesium Doping. J. Phys. Chem. C 2019, 123, 8663–8670. [Google Scholar] [CrossRef]
- Lunca-Popa, P.; Afonso, J.; Grysan, P.; Crêpellière, J.; Leturcq, R.; Lenoble, D. Tuning the Electrical Properties of the P-Type Transparent Conducting Oxide Cu1-xCr1+xO2 by Controlled Annealing. Sci. Rep. 2018, 8, 7216. [Google Scholar] [CrossRef] [Green Version]
- Kawazoe, H.; Yasukawa, M.; Hyodo, H.; Kurita, M.; Yanagi, H.; Hosono, H. P-Type Electrical Conduction in Transparent Thin Films of CuAlO2. Nature 1997, 389, 939–942. [Google Scholar] [CrossRef]
- Raebiger, H.; Lany, S.; Zunger, A. Origins of the P-Type Nature and Cation Deficiency in Cu2O and Related Materials. Phys. Rev. B Condens. Matter Mater. Phys. 2007, 76, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Chikoidze, E.; Boshta, M.; Gomaa, M.; Tchelidze, T.; Daraselia, D.; Japaridze, D.; Shengelaya, A.; Dumont, Y.; Neumann-Spallart, M. Control of P-Type Conduction in Mg Doped Monophase CuCrO2 Thin Layers. J. Phys. D Appl. Phys. 2016, 49, 205107. [Google Scholar] [CrossRef]
- Sidik, U.; Kim, J.H.; Kim, H.-K.; Lee, H.Y.; Lee, J. Structures and Characteristics of Delafossite CuCr1−xO2 Thin Films Prepared by Pulsed Laser Deposition. Micro Nano Lett. 2014, 9, 854–857. [Google Scholar] [CrossRef]
- Chen, H.Y.; Chang, K.P.; Yang, C.C. Characterization of Transparent Conductive Delafossite-CuCr1-xO2 Films. Appl. Surf. Sci. 2013, 273, 324–329. [Google Scholar] [CrossRef]
- O’Sullivan, M.; Stamenov, P.; Alaria, J.; Venkatesan, M.; Coey, J.M.D. Magnetoresistance of CuCrO2-Based Delafossite Films. J. Phys. Conf. Ser. 2010, 200, 1–5. [Google Scholar] [CrossRef]
- Haacke, G. New Figure of Merit for Transparent Conductors. J. Appl. Phys. 1976, 47, 4086–4089. [Google Scholar] [CrossRef]
- Zhang, K.H.L.; Du, Y.; Papadogianni, A.; Bierwagen, O.; Sallis, S.; Piper, L.F.J.; Bowden, M.E.; Shutthanandan, V.; Sushko, P.V.; Chambers, S.A. Perovskite Sr-Doped LaCrO3 as a New p-Type Transparent Conducting Oxide. Adv. Mater. 2015, 27, 5191–5195. [Google Scholar] [CrossRef]
- Asemi, M.; Ghanaatshoar, M. Conductivity Improvement of CuCrO2 Nanoparticles by Zn Doping and Their Application in Solid-State Dye-Sensitized Solar Cells. Ceram. Int. 2016, 42, 6664–6672. [Google Scholar] [CrossRef] [Green Version]
- Benko, F.A.; Koffyberg, F.P. Preparation and opto-electronic properties of semiconducting CuCrO2. Mater. Res. Bull. 1986, 21, 753–757. [Google Scholar] [CrossRef]
- Shibayama, N.; Fukumoto, S.; Sugita, H.; Kanda, H.; Ito, S. Influence of Transparent Conductive Oxide Layer on the Inverted Perovskite Solar Cell Using PEDOT:PSS for Hole Transport Layer. Mater. Res. Bull. 2018, 106, 433–438. [Google Scholar] [CrossRef]
- Fungura, F.; Lindemann, W.R.; Shinar, J.; Shinar, R. Carbon Dangling Bonds in Photodegraded Polymer:Fullerene Solar Cells. Adv. Energy Mater. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Zhang, S.; Qin, Y.; Uddin, M.A.; Jang, B.; Zhao, W.; Liu, D.; Woo, H.Y.; Hou, J. A Fluorinated Polythiophene Derivative with Stabilized Backbone Conformation for Highly Efficient Fullerene and Non-Fullerene Polymer Solar Cells. Macromolecules 2016, 49, 2993–3000. [Google Scholar] [CrossRef]
- Lee, K.W.; Kim, K.M.; Lee, J.; Amin, R.; Kim, B.; Park, S.K.; Lee, S.K.; Park, S.H.; Kim, H.J. A Two-Dimensional DNA Lattice Implanted Polymer Solar Cell. Nanotechnology 2011, 22, 375202. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, Y.; Li, Y. Challenges to the Stability of Active Layer Materials in Organic Solar Cells. Macromol. Rapid Commun. 2020, 41, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.B.; Tiwana, P.; Seidler, N.; Morse, G.E.; Lozman, O.R.; Johnston, M.B.; Herz, L.M. Effect of Ultraviolet Radiation on Organic Photovoltaic Materials and Devices. ACS Appl. Mater. Interfaces 2019, 11, 21543–21551. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Lee, J.; Jo, S.B.; Sin, D.H.; Ko, H.; Lee, H.; Lee, S.G.; Cho, K. Critical Factors Governing Vertical Phase Separation in Polymer-PCBM Blend Films for Organic Solar Cells. J. Mater. Chem. A 2016, 4, 15522–15535. [Google Scholar] [CrossRef]
- Chien, H.T.; Pilat, F.; Griesser, T.; Fitzek, H.; Poelt, P.; Friedel, B. Influence of Environmentally Affected Hole-Transport Layers on Spatial Homogeneity and Charge-Transport Dynamics of Organic Solar Cells. ACS Appl. Mater. Interfaces 2018, 10, 10102–10114. [Google Scholar] [CrossRef]

| HTL | Number of Devices | Jsc (mA/cm2) | Voc (V) | Rshunt (Ω cm2) | Rseries (Ω cm2) | FF (%) | PCE (%) |
|---|---|---|---|---|---|---|---|
| PEDOT:PSS | 12 | 10.4 ± 1.0 | 0.83 ± 0.01 | 166.9 ± 20.9 | 6.9 ± 1.6 | 43.3 ± 3.4 | 3.8 ± 0.7 |
| = 40% | 6 | 6.0 ± 0.8 | 0.74 ± 0.01 | 202.9 ± 17.0 | 108 ± 42 | 39 ± 1.6 | 1.8 ± 0.3 |
| = 50% | 4 | 4.16 ± 1.9 | 0.76 ± 0.01 | 282.3 ± 91.18 | 87 ± 67 | 34 ± 4.7 | 1.2 ± 0.7 |
| = 60% | 6 | 8.4 ± 0.6 | 0.73 ± 0.02 | 108.6 ± 14.0 | 46 ± 7 | 32 ± 2.1 | 2.0 ± 0.2 |
| = 65% | 12 | 10.0 ± 0.4 | 0.75 ± 0.02 | 113.6 ± 13.3 | 49 ± 10 | 37 ± 2.1 | 2.8 ± 0.2 |
| = 67% | 8 | 7.6 ± 2.1 | 0.73 ± 0.02 | 168.7 ± 80.6 | 98 ± 55 | 31 ± 4.4 | 2.2 ± 0.4 |
| = 70% | 10 | 6.6 ± 1.3 | 0.75 ± 0.02 | 137.1 ± 28.4 | 83 ± 29 | 31 ± 8.0 | 1.9 ± 0.2 |
| = 80% | 4 | 6.4 ± 0.8 | 0.77 ± 0.01 | 166.3 ± 19.5 | 75 ± 4 | 34 ± 0.1 | 1.7 ± 0.2 |
| = 100% | 6 | 3.7 ± 0.9 | 0.79 ± 0.01 | 358.8 ± 76.8 | 44 ± 17 | 39 ± 2.7 | 1.2 ± 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bottiglieri, L.; Nourdine, A.; Resende, J.; Deschanvres, J.-L.; Jiménez, C. Optimized Stoichiometry for CuCrO2 Thin Films as Hole Transparent Layer in PBDD4T-2F:PC70BM Organic Solar Cells. Nanomaterials 2021, 11, 2109. https://doi.org/10.3390/nano11082109
Bottiglieri L, Nourdine A, Resende J, Deschanvres J-L, Jiménez C. Optimized Stoichiometry for CuCrO2 Thin Films as Hole Transparent Layer in PBDD4T-2F:PC70BM Organic Solar Cells. Nanomaterials. 2021; 11(8):2109. https://doi.org/10.3390/nano11082109
Chicago/Turabian StyleBottiglieri, Lorenzo, Ali Nourdine, Joao Resende, Jean-Luc Deschanvres, and Carmen Jiménez. 2021. "Optimized Stoichiometry for CuCrO2 Thin Films as Hole Transparent Layer in PBDD4T-2F:PC70BM Organic Solar Cells" Nanomaterials 11, no. 8: 2109. https://doi.org/10.3390/nano11082109
APA StyleBottiglieri, L., Nourdine, A., Resende, J., Deschanvres, J.-L., & Jiménez, C. (2021). Optimized Stoichiometry for CuCrO2 Thin Films as Hole Transparent Layer in PBDD4T-2F:PC70BM Organic Solar Cells. Nanomaterials, 11(8), 2109. https://doi.org/10.3390/nano11082109