In Vitro Effect of Replicated Porous Polymeric Nano-MicroStructured Biointerfaces Characteristics on Macrophages Behavior
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Method
2.2.1. Pyramid-Shaped Micropatterns PDMS Mold
2.2.2. Porous Microstructured PVDF Preparation
2.3. Microstructured PVDF Replica Topography Analysis by SEM and AFM
2.3.1. Scanning Electron Microscopy
2.3.2. Atomic Force Microscopy
2.4. Wettability and Surface Free Energy Characterization
2.4.1. Contact Angle (CA)
2.4.2. Surface Free Energy Measurements (SFE)
2.5. Replica PVDF–Chemical Profile: FTIR and XPS Analysis
2.5.1. Fourier-Transform Infrared Spectroscopy (FT-IR) Analysis
2.5.2. X-ray Photoelectron Spectroscopy
2.6. Biological Investigations of PVDF Replicated Surfaces
2.6.1. Sterilization Procedure
2.6.2. Cell Culture
2.6.3. In Vitro Macrophage Stimulation Assay
2.6.4. Cell Viability
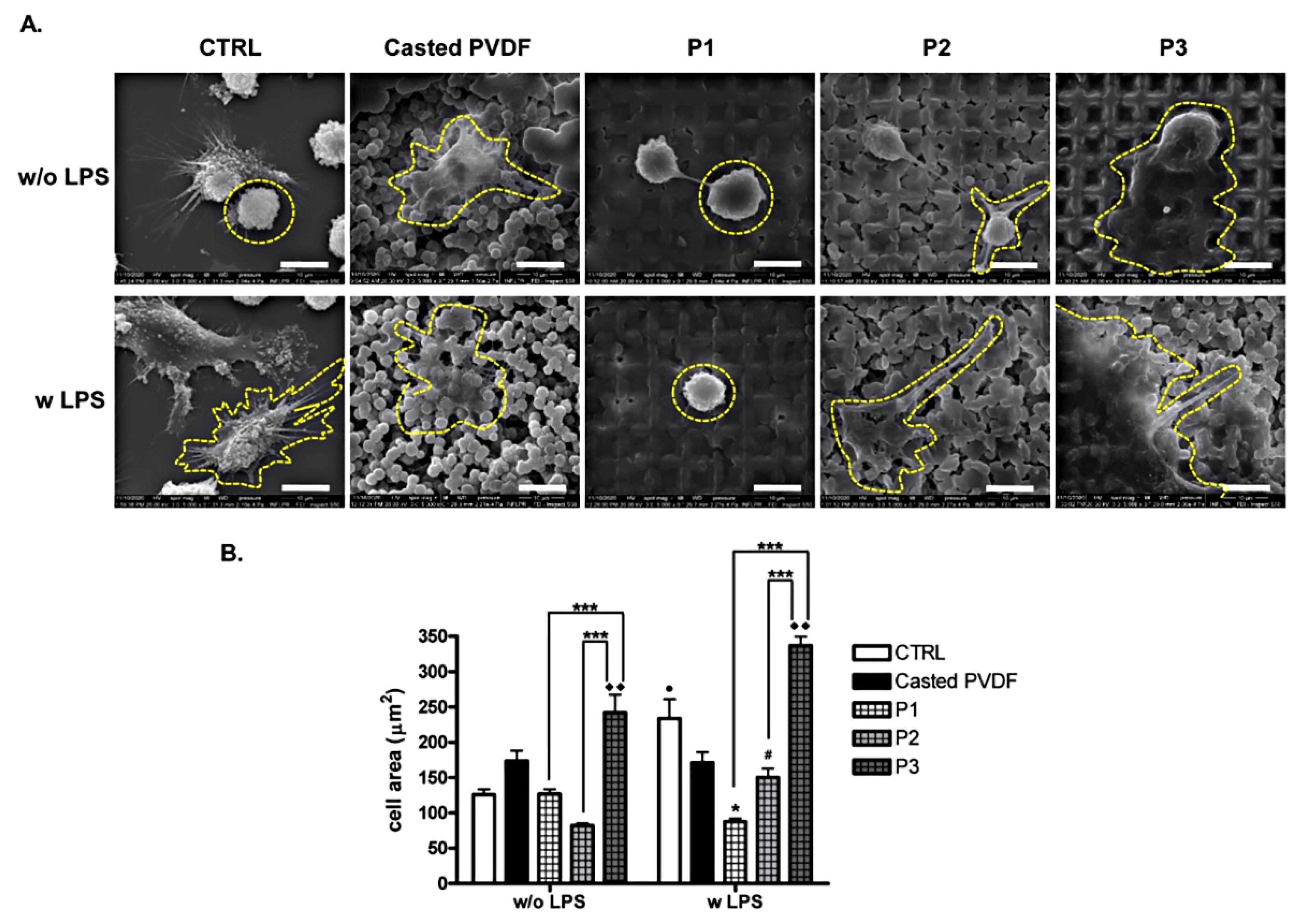
2.6.5. Cell Adhesion and Morphology
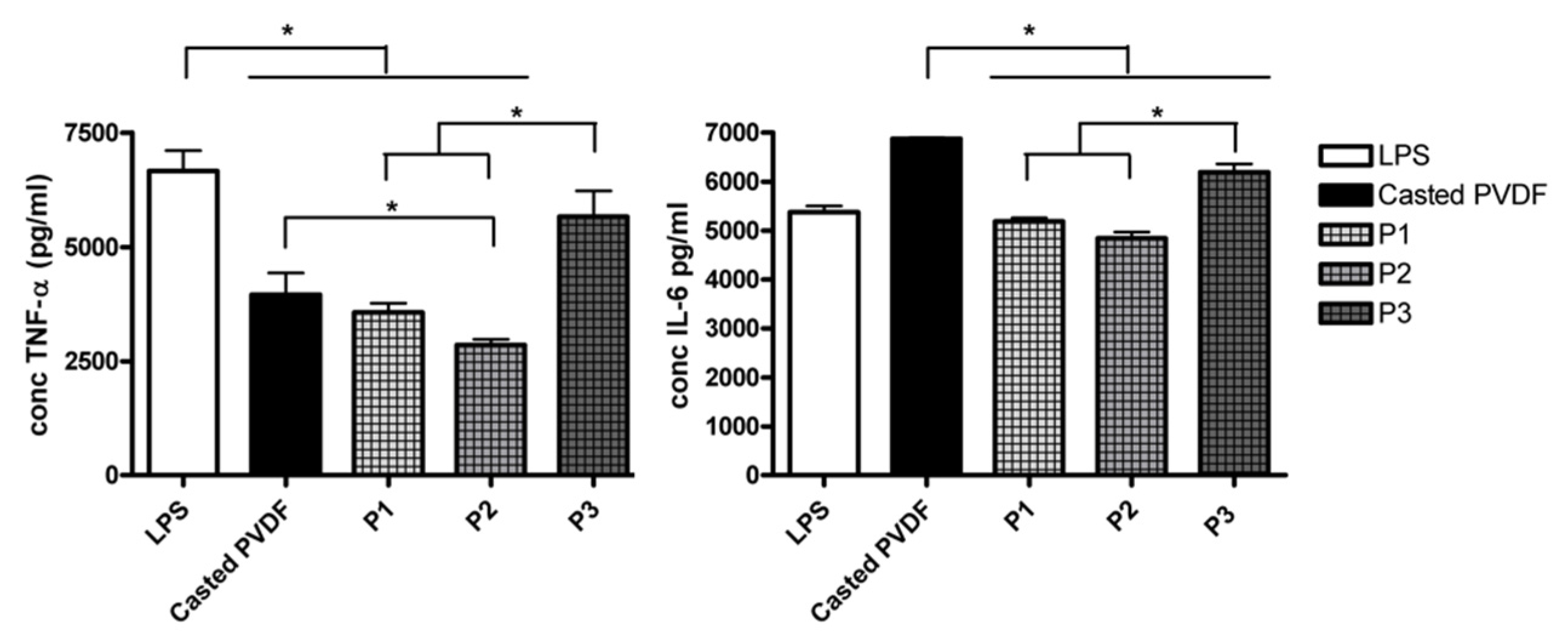
2.6.6. Cytokine Secretion
2.6.7. Statistical Analysis
3. Results and Discussions
3.1. PVDF-Replica Morphology Analysis
3.2. Chemical Characterization of the Replicated PVDF
3.3. Wettability
3.4. Biological In Vitro Investigations
3.4.1. Cell Viability
3.4.2. Cell Adhesion and Morphology
3.4.3. Cytokine Release
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maitz, M.F. Applications of synthetic polymers in clinical medicine. Biosurf. Biotribol. 2015, 1, 161–176. [Google Scholar] [CrossRef] [Green Version]
- Shen, L.; Feng, S.; Li, J. Surface modification of polyvinylidene fluoride (PVDF) membrane via radiation grafting: Novel mechanisms underlying the interesting enhanced membrane performance. Sci. Rep. 2017, 7, 2721. [Google Scholar] [CrossRef]
- Kleea, D.; Ademovic, Z.; Bosserhoff, A.; Hoecker, H.; Maziolis, G.; Erli, H.J. Surface modification of poly(vinylidenefluoride) to improve the osteoblast adhesion. Biomaterials 2003, 24, 3663–3670. [Google Scholar] [CrossRef]
- Fontananova, E.; Bahattab, M.A.; Aljlil, S.A.; Alowairdy, M.; Rinaldi, G.; Vuono, D.; Nagy, J.B.; Drioliac, E.; Di Profio, G. From hydrophobic to hydrophilic polyvinylidenefluoride (PVDF) membranes by gaining new insight into material’s properties. RSC Adv. 2015, 5, 56219–56231. [Google Scholar] [CrossRef]
- Saburi, E.; Abazari, M.F.; Hassannia, H. The use of mesenchymal stem cells in the process of treatment and tissue regeneration after recovery in patients with Covid-19. Gene 2021, 777, 145471. [Google Scholar] [CrossRef] [PubMed]
- Azadian, E.; Arjmand, B.; Khodaii, Z.; Ardeshirylajimi, A. A comprehensive overview on utilizing electromagnetic fields in bone regenerative medicine. Electromagn. Biol. Med. 2019, 1–20. [Google Scholar] [CrossRef]
- Wu, C.; Tang, Y.; Mao, B.; Zhao, K.; Cao, S.; Wu, Z. Rapid apatite induction of polarized hydrophilic HA/PVDF bio-piezoelectric coating on titanium surface. Surf. Coat. Technol. 2021, 405, 126510. [Google Scholar] [CrossRef]
- Damaraju, S.M.; Wu, S.; Jaffe, M.; Arinzeh, T.L. Structural changes in PVDF fibers due to electrospinning and its effect on biological function. Biomed. Mater. 2013, 8, 045007. [Google Scholar] [CrossRef] [PubMed]
- Szewczyk, P.K.; Metwally, S.; Krysiak, Z.J.; Kaniuk, Ł.; Karbowniczek, J.E.; Stachewicz, U. Enhanced osteoblasts adhesion and collagen formation on biomimetic polyvinylidene fluoride (PVDF) films for bone regeneration. Biomed. Mater. 2019, 14, 065006. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, L.N.; Neacsu, P.; Necula, M.G.; Bonciu, A.; Marascu, V.; Cimpean, A.; Moldovan, A.; Rotaru, A.; Dinca, V.; Dinescu, M. Induced Hydrophilicity and In Vitro Preliminary Osteoblast Response of Polyvinylidene Fluoride (PVDF) Coatings Obtained via MAPLE Deposition and Subsequent Thermal Treatment. Molecules 2020, 25, 582. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.; Li, W.; He, T.; Zhengnan, Z.; Qian, L.; Tan, G.; Ning, C. Polarization of an electroactive functional film on titanium for inducing osteogenic differentiation. Sci. Rep. 2016, 6, 35512. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, C.; Jenita, P.; Vitor, S.; Correia, V.; Miettinen, S.; Hytönen, P.; Lanceros-Méndez, S. Dynamic piezoelectric stimulation enhances osteogenic differentiation of human adipose stem cells. J. Biomed. Mater. Res. Part A 2014, 103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pärssinen, J.; Hammarén, H.; Rahikainen, R.; Sencadas, V.; Ribeiro, C.; Vanhatupa, S.; Miettinen, S.; Lanceros-Méndez, S.; Hytönen, V.P. Enhancement of adhesion and promotion of osteogenic differentiation of human adipose stem cells by poled electroactive poly(vinylidene fluoride). J. Biomed. Mater. Res. A 2015, 103, 919–928. [Google Scholar] [CrossRef] [Green Version]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ogle, M.E.; Segar, C.E.; Sridhar, S.; Botchwey, E.A. Monocytes and macrophages in tissue repair: Implications for immunoregenerative biomaterial design. Exp. Biol. Med. 2016, 241, 1084–1097. [Google Scholar] [CrossRef]
- Paul, N.E.; Skazik, C.; Harwardt, M.; Bartneck, M.; Denecke, B.; Klee, D.; Salber, J.; Zwadlo-Klarwasser, G. Topographical control of human macrophages by a regularly microstructured polyvinylidene fluoride surface. Biomaterials 2008, 29, 4056–4064. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.G.; Kim, S.H.; Kim, T.S. Polyvinylidene Fluoride Alters Inflammatory Responses by Activation-induced Cell Death in Macrophages. Immune Netw. 2017, 17, 402–409. [Google Scholar] [CrossRef] [Green Version]
- Malheiro, V.; Lehner, F.; Dinca, V.; Hoffmann, P.; Maniura-Weber, K. Convex and concave micro-structured silicone controls the shape, but not the polarization state of human macrophages. Biomater. Sci. 2016, 4, 1562–1573. [Google Scholar] [CrossRef] [Green Version]
- Schmidt, A.M.; Fagerer, S.R.; Jefimovs, K.; Buettner, F.; Marro, C.; Siringil, E.C.; Boehlen, K.L.; Pabst, M.; Ibáñez, A.J. Molecular phenotypic profiling of a Saccharomyces cerevisiae strain at the single-cell level. Analyst 2014, 139, 5709–5717. [Google Scholar] [CrossRef] [Green Version]
- Ibtisam, Y.A.; Muhammad, Y.; Mohd, H.H.J.; Haider, M.S. Effect of annealing process on the phase formation in poly(vinylidene fluoride) thin films. AIP Conf. Proc. 2014, 147, 1614. [Google Scholar] [CrossRef]
- Ribeiro, C.; Costa, C.; Correia, D. Electroactive poly(vinylidene fluoride)-based structures for advanced applications. Nat. Protoc. 2018, 13, 681–704. [Google Scholar] [CrossRef] [PubMed]
- Peichun, T.; Alisia, M.P.; Christophe, P.; Matthias, W.; Rob, G.H.L.; Detlef, L. Quantifying effective slip length over micropatterned hydrophobic surfaces. Phys. Fluids 2009, 21, 112002. [Google Scholar] [CrossRef] [Green Version]
- Wu, S. Calculation of Interfacial Tension in Polymer Systems. J. Polym. Sci. Part C 1971, 34, 19–30. [Google Scholar] [CrossRef]
- Shimizu, R.N.; Demarquette, N.R. Evaluation of Surface Energy of Solid Polymers Using Different Models. J. Appl. Polym. Sci. 2000, 76183. [Google Scholar] [CrossRef]
- Icriverzi, M.; Rusen, L.; Brajnicov, S.; Bonciu, A.; Dinescu, M.; Cimpean, A.; Evans, R.W.; Dinca, V.; Roseanu, A. Macrophage in vitro Response on Hybrid Coatings Obtained by Matrix Assisted Pulsed Laser Evaporation. Coatings 2019, 9, 236. [Google Scholar] [CrossRef] [Green Version]
- Alfarsi, M.A.; Hamlet, S.M.; Ivanovski, S. Titanium surface hydrophilicity modulates the human macrophage inflammatory cytokine response. J. Biomed. Mater. Res. Part A 2014, 102, 60–67. [Google Scholar] [CrossRef]
- Lanceros-Mendez, S.; Mano, J.F.; Costa, A.M.; Schmidt, V.H. FTIR and DSC studies of mechanically deformed β-PVDF films. J. Macromol. Sci. Part B 2001, 40, 517–527. [Google Scholar] [CrossRef] [Green Version]
- Wortmann, M.; Frese, N.; Mamun, A.; Trabelsi, M.; Keil, W.; Büker, B.; Javed, A.; Tiemann, M.; Moritzer, E.; Ehrmann, A.; et al. Chemical and Morphological Transition of Poly(acrylonitrile)/Poly(vinylidene Fluoride) Blend Nanofibers during Oxidative Stabilization and Incipient Carbonization. Nanomaterials 2020, 10, 1210. [Google Scholar] [CrossRef]
- Bai, H.; Wang, X.; Zhou, Y.; Zhang, L. Preparation and characterization of poly(vinylidene fluoride) composite membranes blended with nano-crystalline cellulose. Prog. Nat. Sci. Mater. Int. 2012, 22. [Google Scholar] [CrossRef] [Green Version]
- Park, Y.J.Y.; Kang, Y.S.; Park, C. Micropatterning of semicrystalline poly(vinylidene fluoride) (PVDF) solutions. Eur. Polym. J. 2005, 41, 1002–1012. [Google Scholar] [CrossRef]
- Ma, W.; Zhang, J.; Chen, S.; Wang, X. Crystalline Phase Formation of Poly(vinylidene fluoride) from Tetrahydrofuran/N,N-dimethylformamide Mixed Solutions. J. Macromol. Sci. Part B 2008, 47, 434–449. [Google Scholar] [CrossRef]
- Rinaldo, G., Jr. Determination of the α, β, and γ crystalline phases of poly(vinylidene fluoride) films prepared at different conditions. J. Appl. Polym. Sci. 2006, 100, 3272–3279. [Google Scholar] [CrossRef]
- Ning, C.; Zhou, Z.; Tan, G.; Zhu, Y.; Mao, C. Electroactive polymers for tissue regeneration: Developments and perspectives. Prog. Polym. Sci. 2018, 81, 144–162. [Google Scholar] [CrossRef]
- Rayahin, J.E.; Gemeinhart, R.A. Activation of Macrophages in Response to Biomaterials. Results Probl. Cell Differ. 2017, 62, 317–351. [Google Scholar] [CrossRef]
- Akashi, N.; Kuroda, S. Protein immobilization onto poly (vinylidene fluoride) microporous membranes activated by the atmospheric pressure low temperature plasma. Polymer 2014, 55, 2780–2791. [Google Scholar] [CrossRef] [Green Version]
- Ma, Z.; Lu, X.; Wu, C.; Gao, Q.; Zhao, L. Functional surface modification of PVDF membrane for chemical pulse cleaning. J. Membr. Sci. 2017, 524, 389–399. [Google Scholar] [CrossRef]
- Kaynak, A.; Mehmood, T.; Dai, X.J.; Magniez, K.; Kouzani, A. Study of Radio Frequency Plasma Treatment of PVDF Film Using Ar, O₂ and (Ar + O₂) Gases for Improved Polypyrrole Adhesion. Materials 2013, 6, 3482–3493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitsara, M.; Blanquer, A.; Murillo, G.; Humblot, V.; De Bragança, V.S.; Nogués, C.; Ibáñez, E.; Esteve, J.; Barrios, L. Permanently hydrophilic, piezoelectric PVDF nanofibrous scaffolds promoting unaided electromechanical stimulation on osteoblasts. Nanoscale 2019, 11, 8906–8917. [Google Scholar] [CrossRef]
- Chun, Y.W.; Wang, W.; Choi, J.; Nam, T.-H.; Lee, Y.-H.; Cho, K.-K.; Im, Y.-M.; Kim, M.; Gwon, Y.-H.; Kang, S.S.; et al. Control of macrophage responses on hydrophobic and hydrophilic carbon nanostructures. Carbon 2011, 49, 2092–2103. [Google Scholar] [CrossRef]
- Moradi, R.; Karimi-Sabet, J.; Shariaty-Niassar, M.; Koochaki, M.A. Preparation and Characterization of Polyvinylidene Fluoride/Graphene Superhydrophobic Fibrous Films. Polymers 2015, 7, 1444–1463. [Google Scholar] [CrossRef] [Green Version]
- Nurafiqah, R.; Wan, N.; Nuha, A.; Ahmad, I.; Juhana, J.; Zawati, H. Wettability and Surface Area Characteristic of PVDF Nanofibrous Composite Film. Mater. Today 2019, 19, 1413–1419. [Google Scholar] [CrossRef]
- Wenzel, R.N. Resistance of solid surfaces to wetting by water. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Harnett, E.M.; Alderman, J.; Wood, T. The surface energy of various biomaterials coated with adhesion molecules used in cell culture. Colloids Surf. B Biointerfaces 2007, 55, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Gentile, F.; Tirinato, L.; Battista, E.; Causa, F.; Liberale, C.; di Fabrizio, E.M.; Decuzzi, P. Cells preferentially grow on rough substrates. Biomaterials 2010, 31, 7205–7212. [Google Scholar] [CrossRef]
- Chanput, W.; Mes, J.J.; Wichers, H.J. THP-1 cell line: An in vitro cell model for immune modulation approach. Int. Immunopharmacol. 2014, 23, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Fletcher, A.; Niu, Y. Characterization of lipopolysaccharide-stimulated cytokine expression in macrophages and monocytes. Inflamm. Res. 2012, 61, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Starr, T.; Bauler, T.J.; Malik-Kale, P.; Steele-Mortimer, O. The phorbol 12-myristate-13-acetate differentiation protocol is critical to the interaction of THP-1 macrophages with Salmonella Typhimurium. PLoS ONE 2018, 13, e0193601. [Google Scholar] [CrossRef]
- Luu, T.U.; Gott, S.C.; Woo, B.W.; Rao, M.P.; Liu, W.F. Micro- and Nanopatterned Topographical Cues for Regulating Macrophage Cell Shape and Phenotype. ACS Appl. Mater. Interfaces 2015, 7, 28665–28672. [Google Scholar] [CrossRef] [Green Version]
- McWhorter, F.Y.; Wang, T.; Nguyen, P.; Chung, T.; Liu, W.F. Modulation of macrophage phenotype by cell shape. Proc. Natl. Acad. Sci. USA 2013, 110, 17253–17258. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Luu, T.U.; Chen, A.; Khine, M.; Liu, W.F. Topographical modulation of macrophage phenotype by shrink-film multi-scale wrinkles. Biomater. Sci. 2016, 4, 948–952. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.-S.; He, D.-Q.; Luo, D.; Wang, Y.; Yu, M.; Guan, B.; Liu, Y.A. Biomimetic Hierarchical Nanointerface Orchestrates Macrophage Polarization and Mesenchymal Stem Cell Recruitment to Promote Endogenous Bone Regeneration. ACS Nano 2019, 13, 6581–6595. [Google Scholar] [CrossRef] [PubMed]
- Makaremi, S.; Luu, H.; Boyle, J.P.; Zhu, Y.; Cerson, C.; Bowdish, D.M.E.; Moran-Mirabal, J.M. The Topography of Silica Films Modulates Primary Macrophage Morphology and Function. Adv. Mater. Interfaces 2019, 6, 1900677. [Google Scholar] [CrossRef]
- Miao, X.; Wang, D.; Xu, L.; Wang, J.; Zeng, D.; Lin, S.; Huang, C.; Liu, X.; Jiang, X. The response of human osteoblasts, epithelial cells, fibroblasts, macrophages and oral bacteria to nanostructured titanium surfaces: A systematic study. Int. J. Nanomed. 2020, 15, 2351–2352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Y.; Liang, H.; Liu, X.; Wu, J.; Yang, C.; Wong, M.T.; Kwan, K.Y.H.; Cheung, K.M.C.; Wu, S.; Yeung, W.K. Regulation of macrophage polarization through surface topography design to facilitate implant-to-bone osteointegration. Sci. Adv. 2021, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Stachelek, S.J.; Tomczyk, N.; Finley, M.J.; Composto, R.J.; Eckmann, D.M. Correlating macrophage morphology and cytokine production resulting from biomaterial contact. J. Biomed. Mater. Res A 2013, 101, 203–212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Refai, A.K.; Textor, M.; Brunette, D.M.; Waterfield, J.D. Effect of titanium surface topography on macrophage activation and secretion of proinflammatory cytokines and chemokines. J. Biomed. Mater. Res. A 2004, 70, 194–205. [Google Scholar] [CrossRef]
- Sussman, E.M.; Halpin, M.C.; Muster, J.; Moon, R.T.; Ratner, B.D. Porous implants modulate healing and induce shifts in local macrophage polarization in the foreign body reaction. Ann. Biomed. Eng. 2014, 42, 1508–1516. [Google Scholar] [CrossRef]
- Hotchkiss, K.M.; Reddy, G.B.; Hyzy, S.L. Titanium surface characteristics, including topography and wettability, alter macrophage activation. Acta Biomater. 2016, 31, 425–434. [Google Scholar] [CrossRef] [Green Version]
- Barr, S.; Hill, E.; Bayat, A. Functional biocompatibility testing of silicone breast implants and a novel classification system based on surface roughness. J. Mech. Behav. Biomed. Mater. 2017, 75, 75–81. [Google Scholar] [CrossRef] [PubMed]

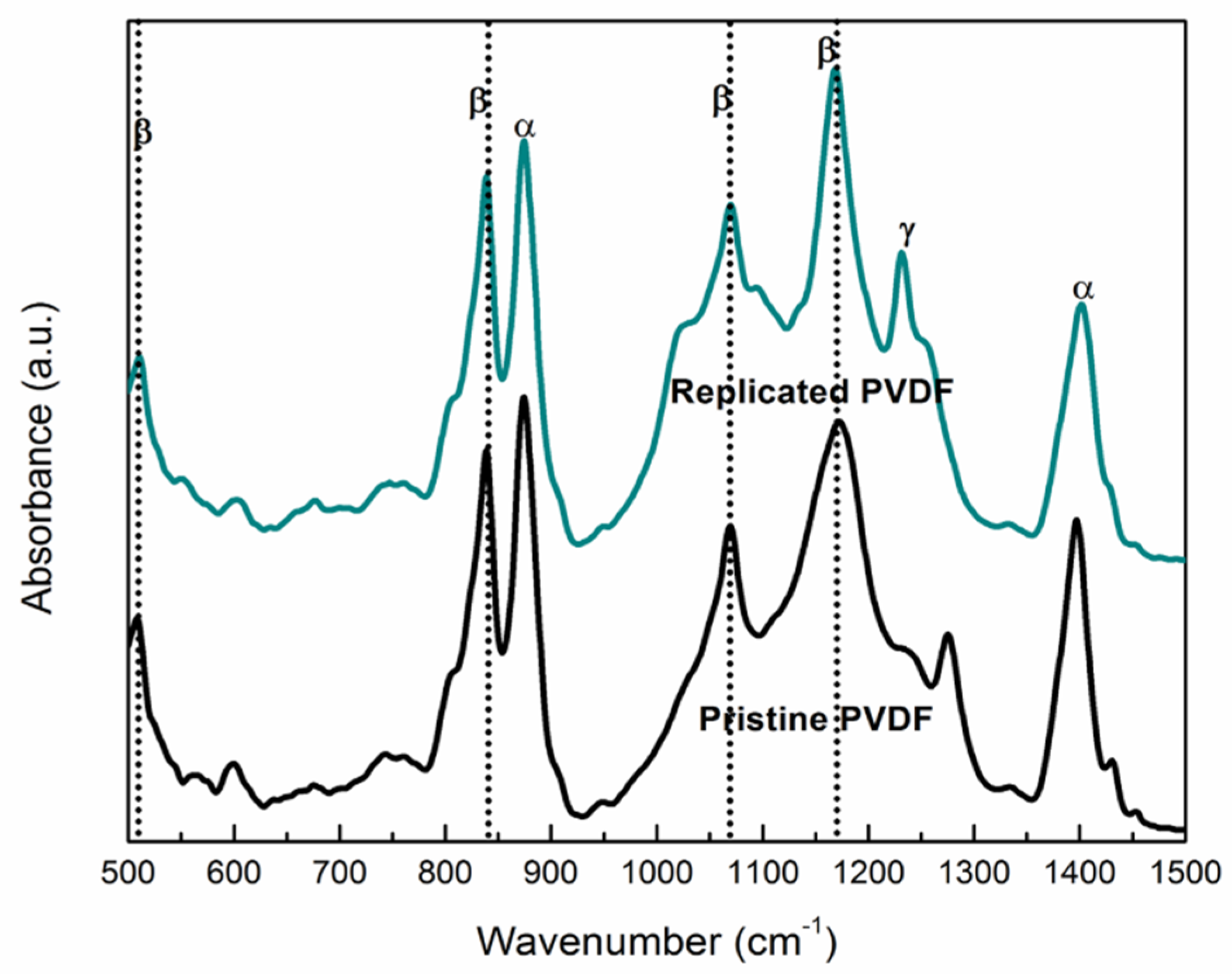
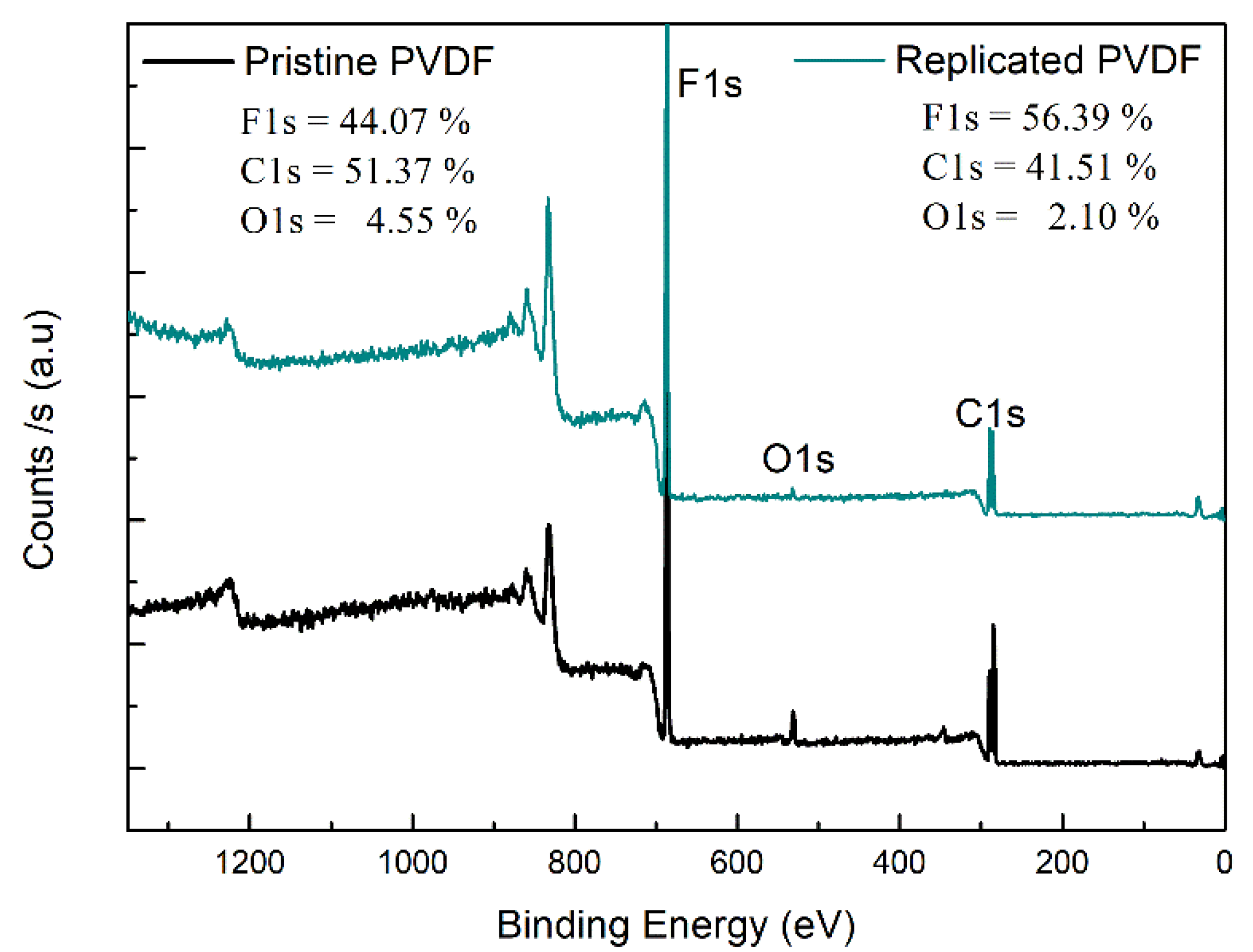
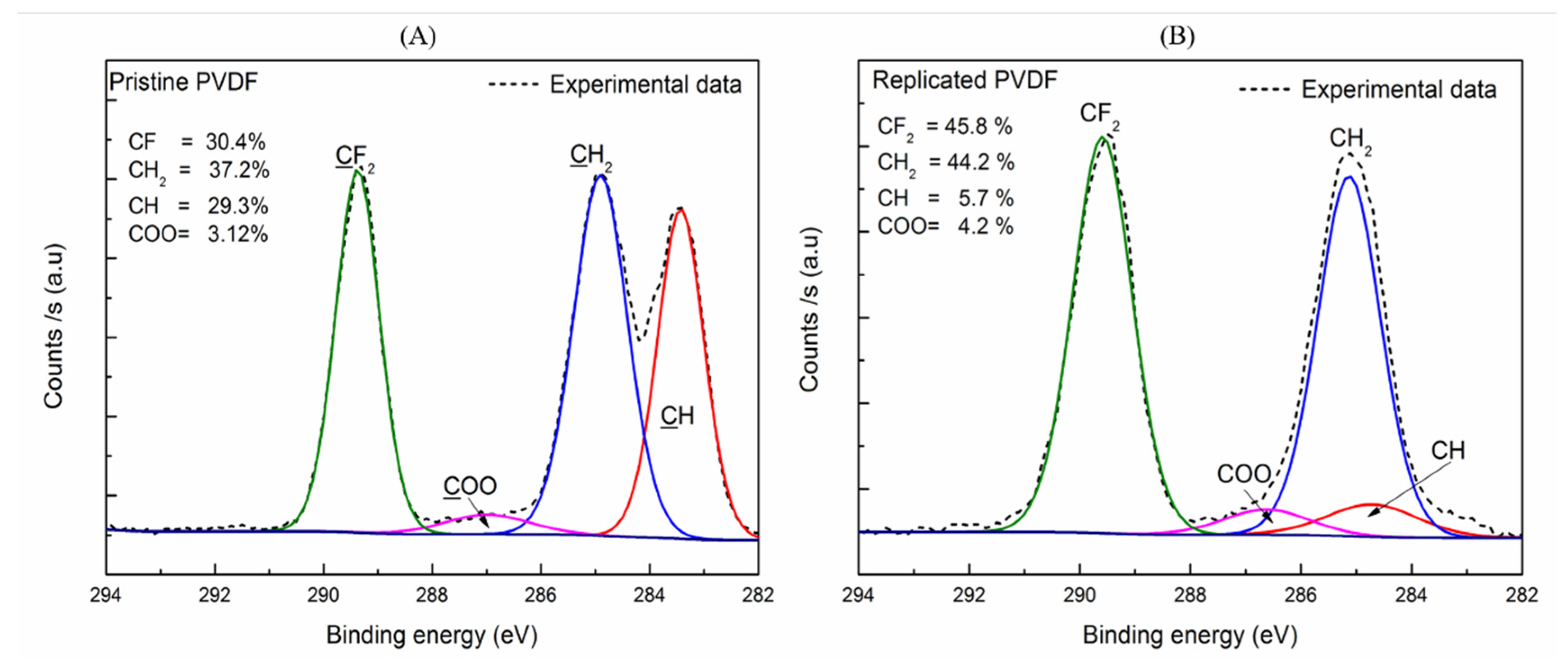
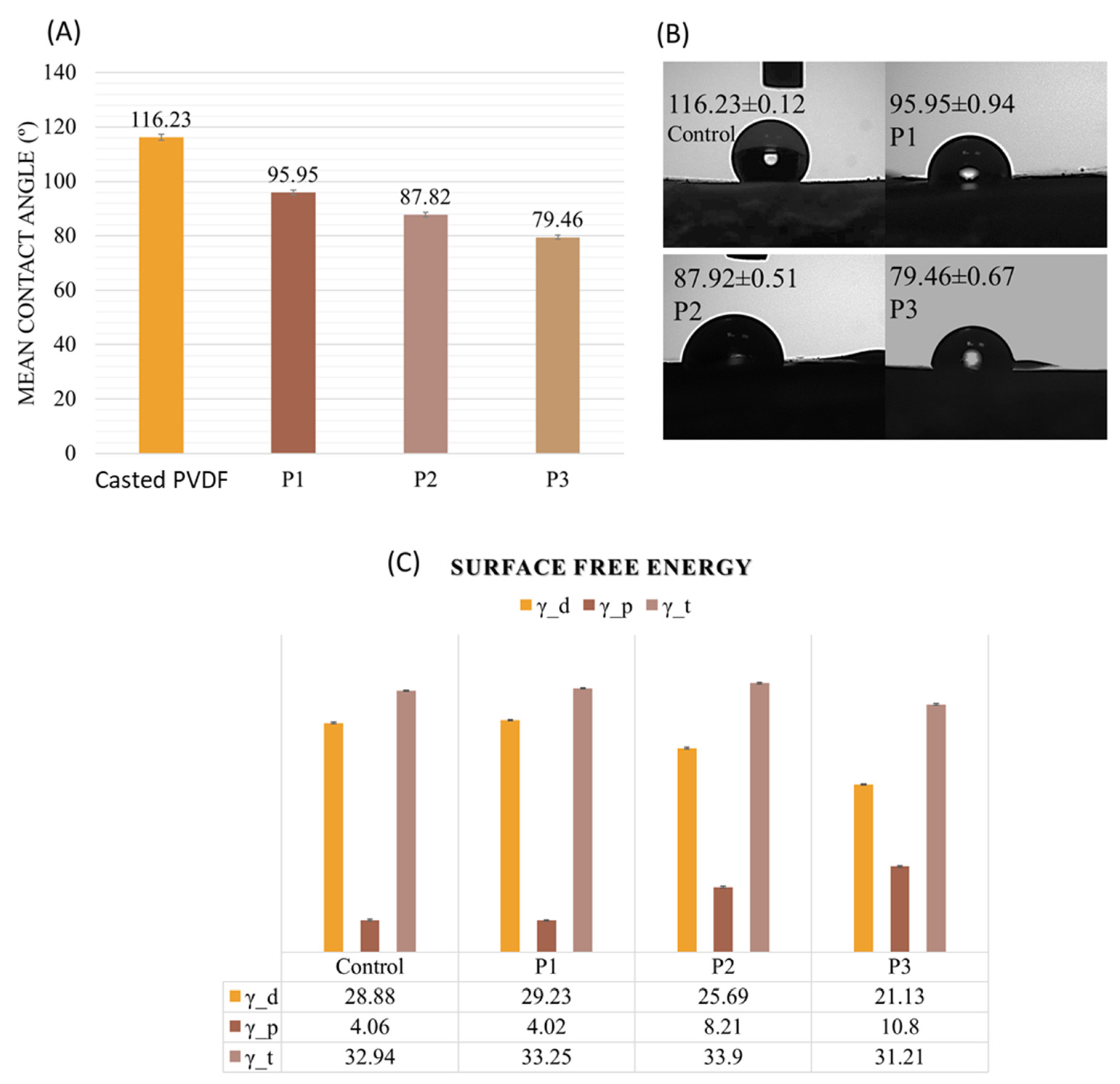

| β-Phase | Replicated PVDF |
|---|---|
| 839 cm−1 | related to C–C–C asymmetrical stretching vibration and CF stretching vibration (deformation of CF2) |
| 1071 and 1178 cm−1 | symmetrical stretching of CF2 group |
| γ-Phase | Replicated PVDF |
| 1233 cm−1 | γ- polymorph phase |
| PVDF | Atomic Ratio | |
|---|---|---|
| F/C | O/C | |
| Pristine | 0.86 | 0.08 |
| Replicated | 1.36 | 0.46 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dumitrescu, L.N.; Icriverzi, M.; Bonciu, A.; Roșeanu, A.; Moldovan, A.; Dinca, V. In Vitro Effect of Replicated Porous Polymeric Nano-MicroStructured Biointerfaces Characteristics on Macrophages Behavior. Nanomaterials 2021, 11, 1913. https://doi.org/10.3390/nano11081913
Dumitrescu LN, Icriverzi M, Bonciu A, Roșeanu A, Moldovan A, Dinca V. In Vitro Effect of Replicated Porous Polymeric Nano-MicroStructured Biointerfaces Characteristics on Macrophages Behavior. Nanomaterials. 2021; 11(8):1913. https://doi.org/10.3390/nano11081913
Chicago/Turabian StyleDumitrescu, Luminita Nicoleta, Madalina Icriverzi, Anca Bonciu, Anca Roșeanu, Antoniu Moldovan, and Valentina Dinca. 2021. "In Vitro Effect of Replicated Porous Polymeric Nano-MicroStructured Biointerfaces Characteristics on Macrophages Behavior" Nanomaterials 11, no. 8: 1913. https://doi.org/10.3390/nano11081913
APA StyleDumitrescu, L. N., Icriverzi, M., Bonciu, A., Roșeanu, A., Moldovan, A., & Dinca, V. (2021). In Vitro Effect of Replicated Porous Polymeric Nano-MicroStructured Biointerfaces Characteristics on Macrophages Behavior. Nanomaterials, 11(8), 1913. https://doi.org/10.3390/nano11081913







