Thickness-Dependent Photoelectrochemical Water Splitting Properties of Self-Assembled Nanostructured LaFeO3 Perovskite Thin Films
Abstract
1. Introduction
2. Materials and Methods
2.1. Thin Films Deposition
2.2. Characterization and Photoelectrochemical Measurements
3. Results
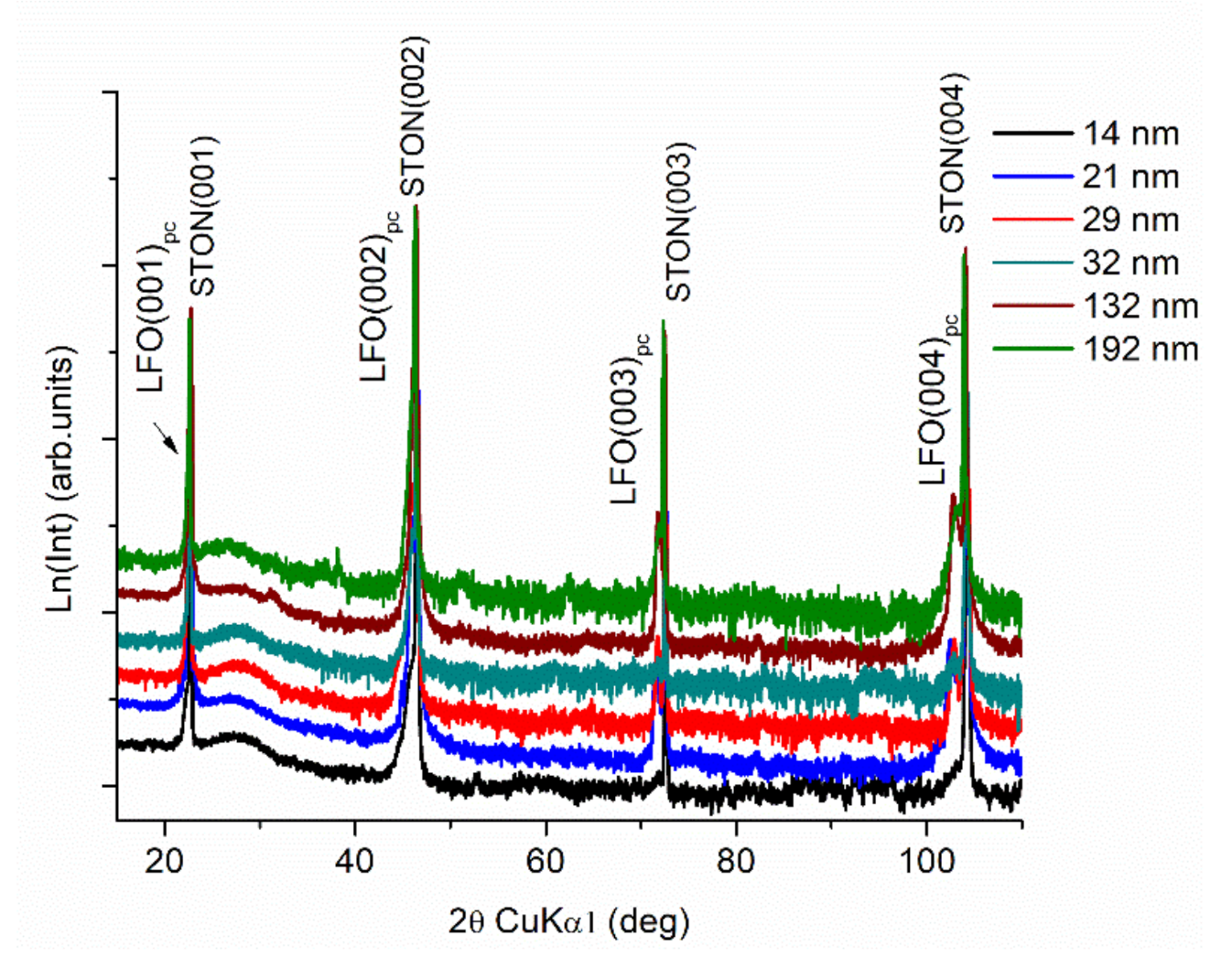
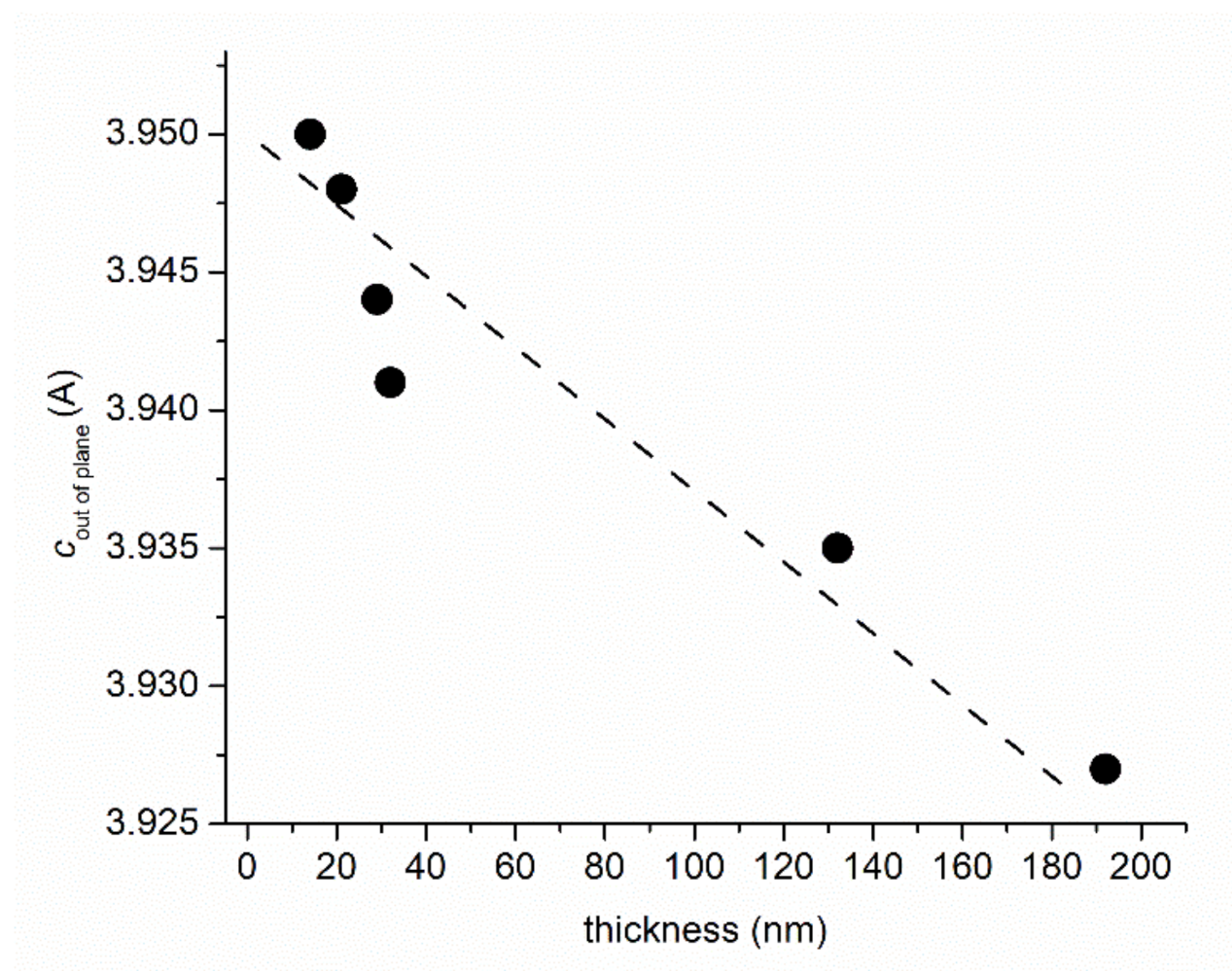
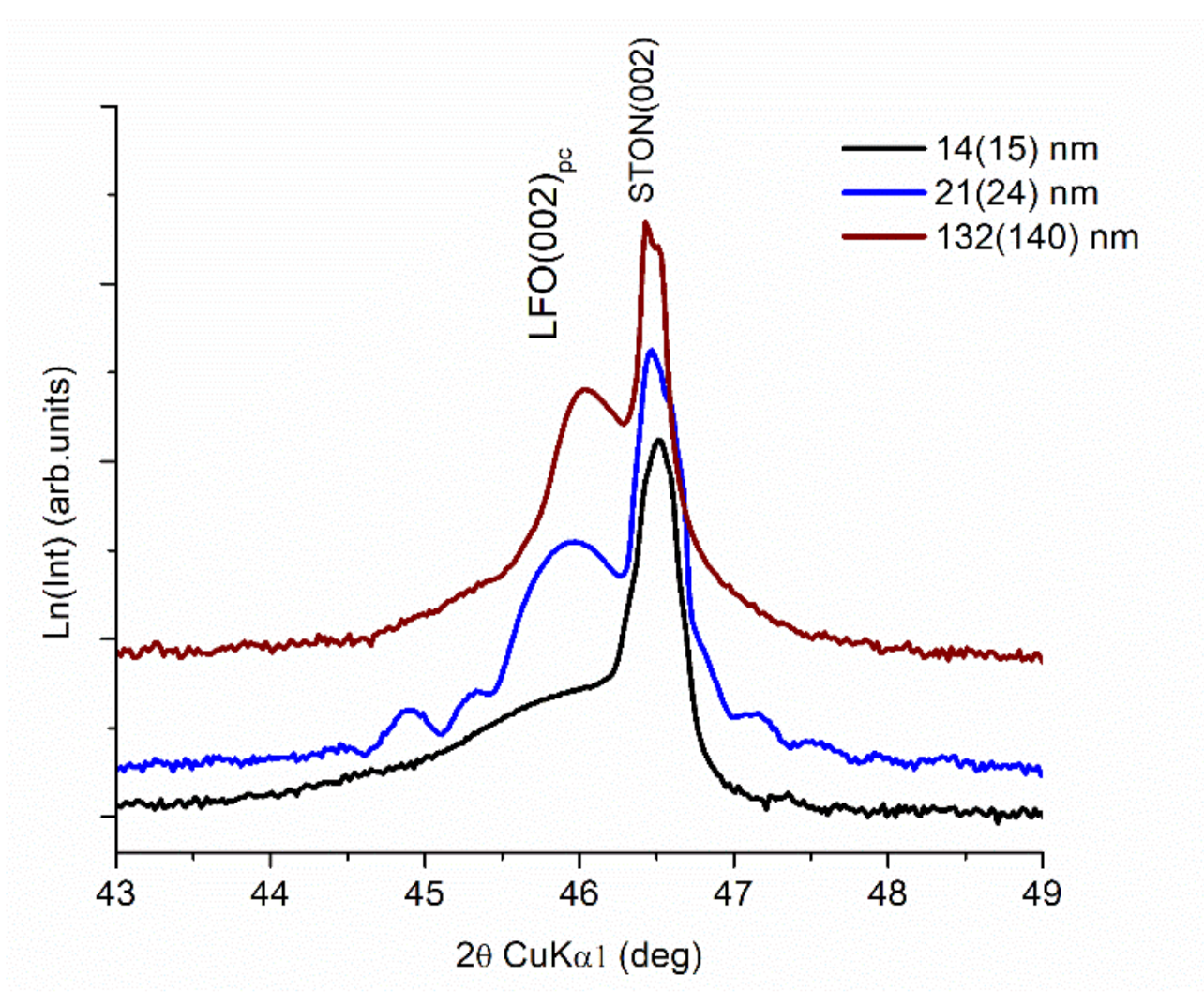
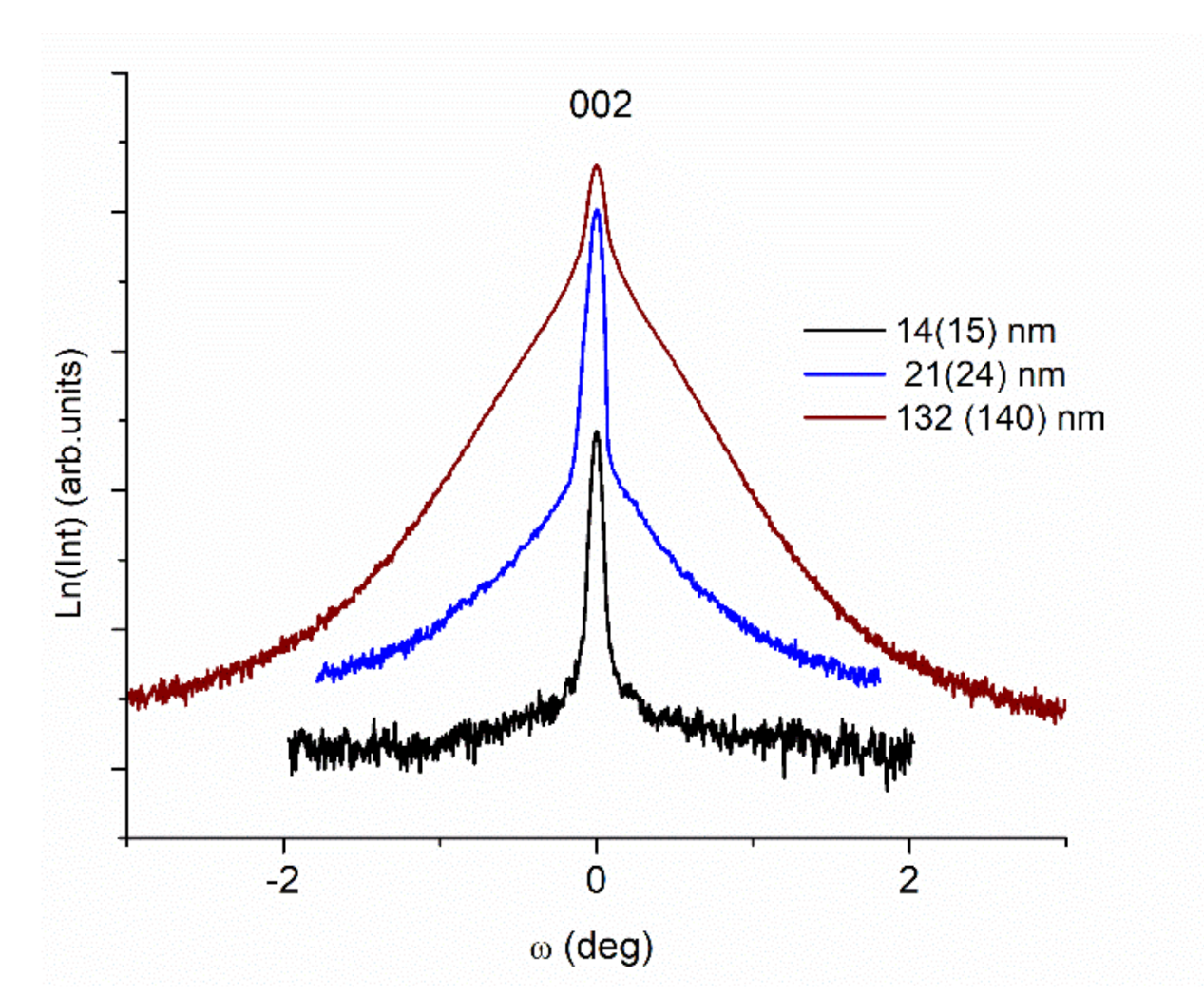
3.1. XRD Measurements
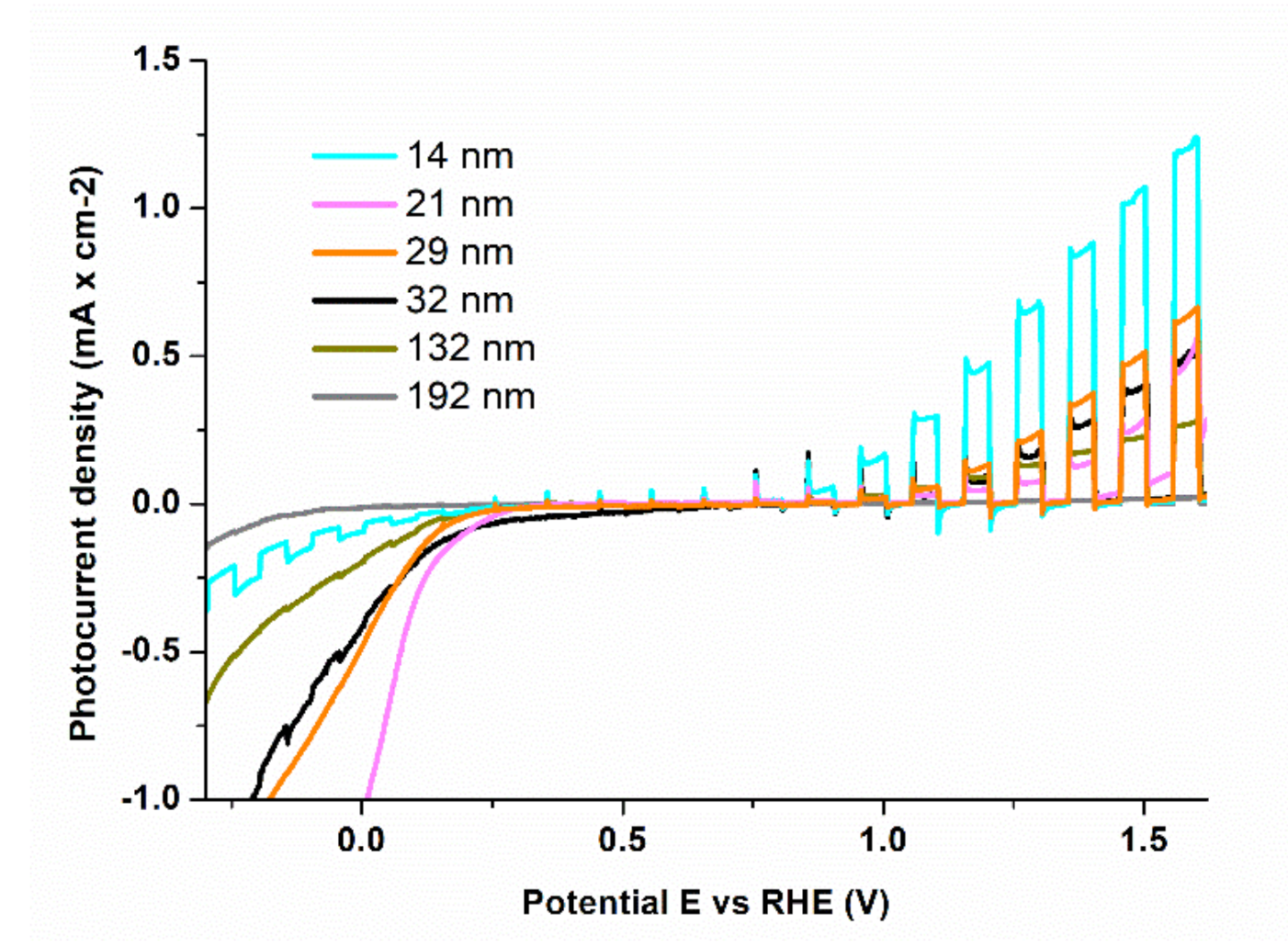
3.2. Photoelectrochemical (PEC) Measurements
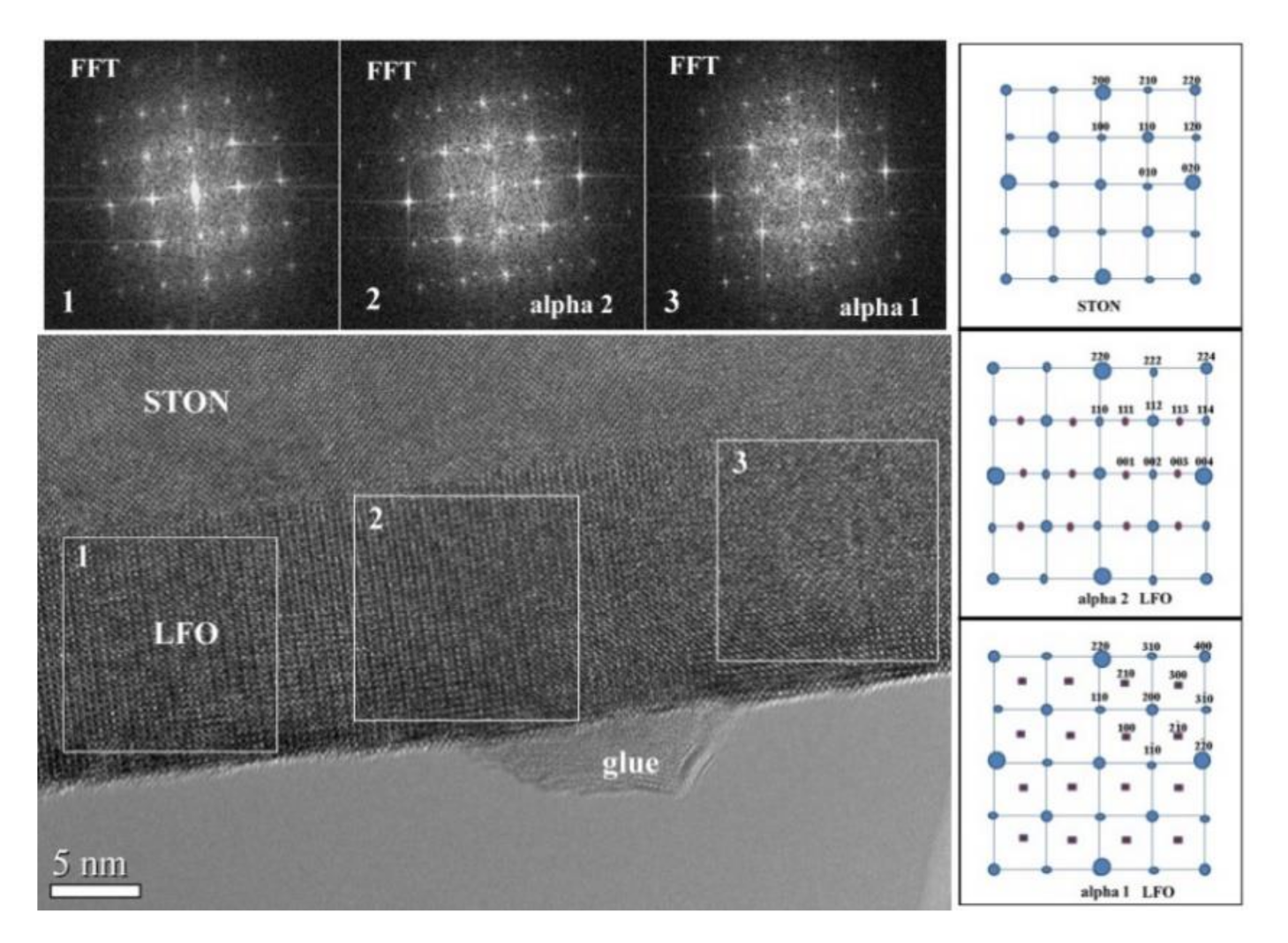
3.3. High-Resolution Electron Microscopy in Cross-Section (XTEM)
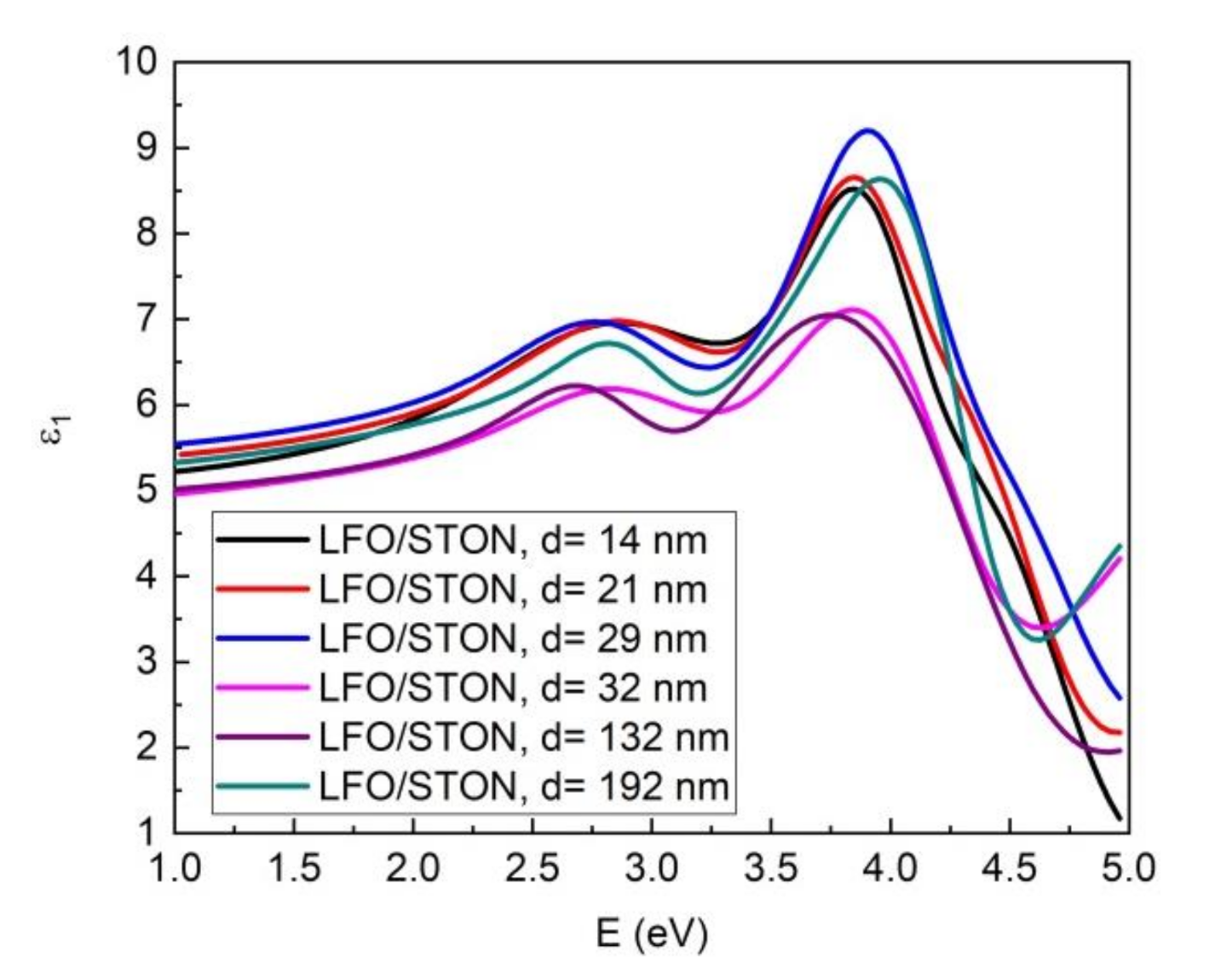
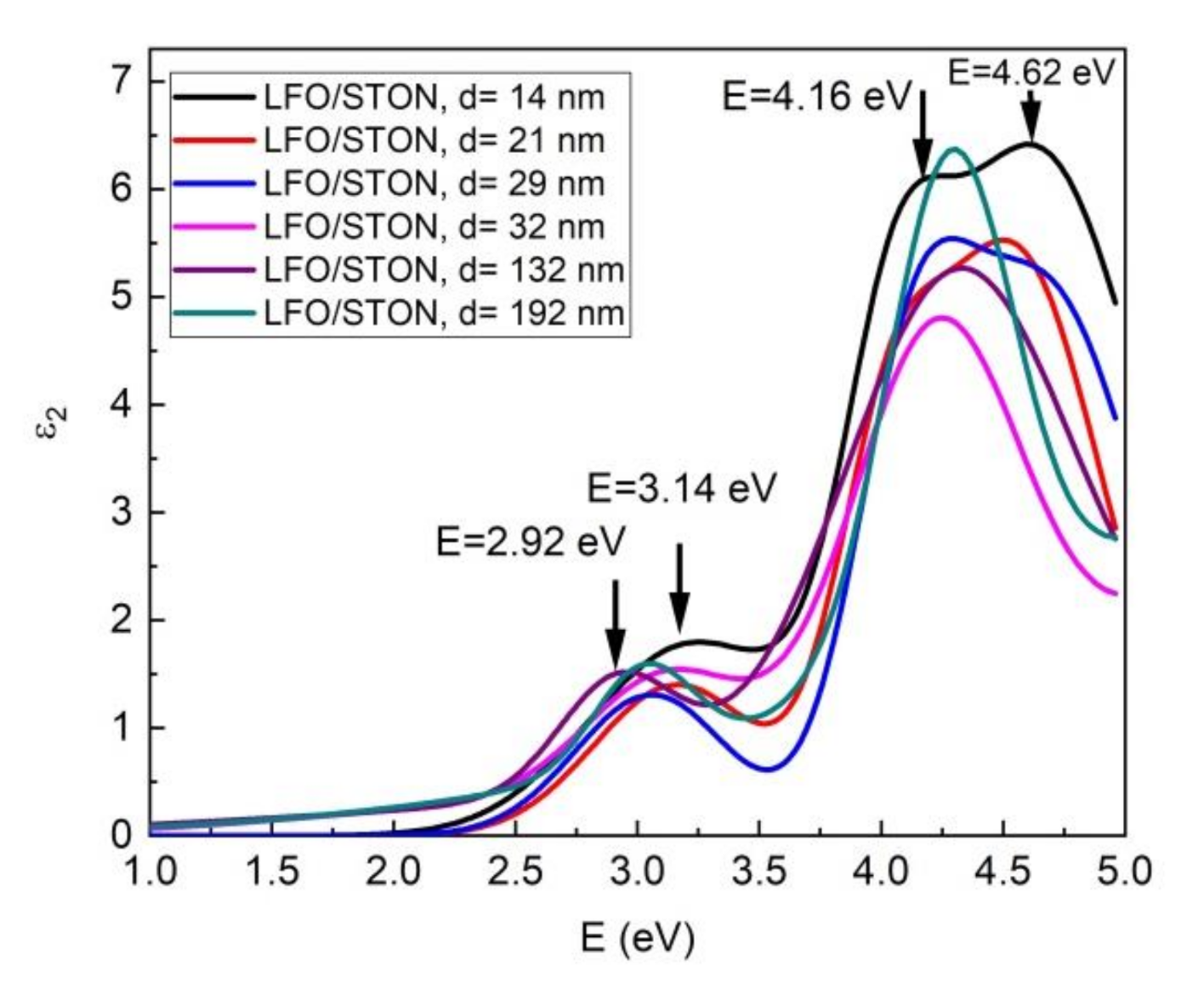
3.4. Optical Properties: Spectrometric Ellipsometry (SE)
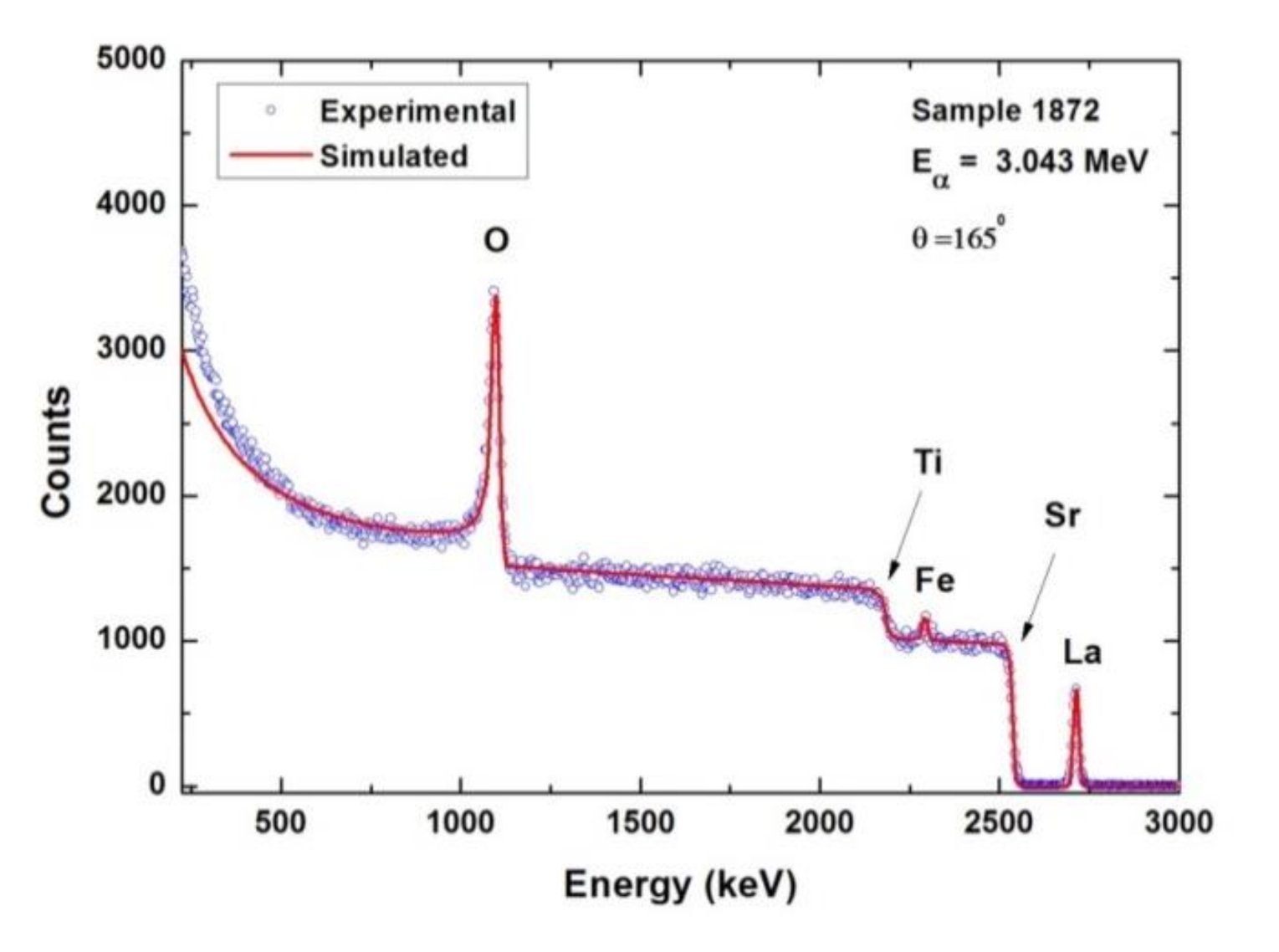
3.5. Stoichiometric Investigations: Rutherford Backscattering
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Turan, B.; Becker, J.-P.; Urbain, F.; Finger, F.; Rau, U.; Haas, S. Upscaling of Integrated Photoelectrochemical Water-Splitting Devices to Large Areas. Nat. Commun. 2016, 7, 12681. [Google Scholar] [CrossRef]
- Currao, A. Photoelectrochemical Water Splitting. CHIMIA Int. J. Chem. 2007, 61, 815–819. [Google Scholar] [CrossRef]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar Water Splitting Cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- De Chialvo, M.R.G.; Chialvo, A.C. Hydrogen Evolution Reaction: Analysis of the Volmer-Heyrovsky-Tafel Mechanism with a Generalized Adsorption Model. J. Electroanal. Chem. 1994, 372, 209–223. [Google Scholar] [CrossRef]
- Hu, C.; Zhang, L.; Gong, J. Recent Progress Made in the Mechanism Comprehension and Design of Electrocatalysts for Alkaline Water Splitting. Energy Environ. Sci. 2019, 12, 2620–2645. [Google Scholar] [CrossRef]
- Kim, J.S.; Kim, B.; Kim, H.; Kang, K. Recent Progress on Multimetal Oxide Catalysts for the Oxygen Evolution Reaction. Adv. Energy Mater. 2018, 8, 1702774. [Google Scholar] [CrossRef]
- Jiao, Y.; Zheng, Y.; Jaroniec, M.; Qiao, S.Z. Design of Electrocatalysts for Oxygen- and Hydrogen-Involving Energy Conversion Reactions. Chem. Soc. Rev. 2015, 44, 2060–2086. [Google Scholar] [CrossRef]
- Seh, Z.W.; Kibsgaard, J.; Dickens, C.F.; Chorkendorff, I.; Nørskov, J.K.; Jaramillo, T.F. Combining Theory and Experiment in Electrocatalysis: Insights into Materials Design. Science 2017, 355, eaad4998. [Google Scholar] [CrossRef]
- Hou, Y.; Zhuang, X.; Feng, X. Electrocatalysts: Recent Advances in Earth-Abundant Heterogeneous Electrocatalysts for Photoelectrochemical Water Splitting (Small Methods 6/2017). Small Methods 2017, 1, 1700090. [Google Scholar] [CrossRef]
- Wolcott, A.; Smith, W.A.; Kuykendall, T.R.; Zhao, Y.; Zhang, J.Z. Photoelectrochemical Study of Nanostructured ZnO Thin Films for Hydrogen Generation from Water Splitting. Adv. Funct. Mater. 2009, 19, 1849–1856. [Google Scholar] [CrossRef]
- Kim, J.Y.; Magesh, G.; Youn, D.H.; Jang, J.-W.; Kubota, J.; Domen, K.; Lee, J.S. Single-Crystalline, Wormlike Hematite Photoanodes for Efficient Solar Water Splitting. Sci. Rep. 2013, 3, 2681. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Kim, T.L.; Lee, J.; Cho, S.Y.; Cha, J.; Jeong, S.Y.; An, H.; Kim, W.S.; Jung, Y.-S.; Park, J.; et al. Domain-Engineered BiFeO3 Thin-Film Photoanodes for Highly Enhanced Ferroelectric Solar Water Splitting. Nano Res. 2018, 11, 642–655. [Google Scholar] [CrossRef]
- Xu, H.-M.; Wang, H.; Shi, J.; Lin, Y.; Nan, C. Photoelectrochemical Performance Observed in Mn-Doped BiFeO3 Heterostructured Thin Films. Nanomaterials 2016, 6, 215. [Google Scholar] [CrossRef]
- Scarisoreanu, N.D.; Craciun, F.; Birjega, R.; Ion, V.; Teodorescu, V.S.; Ghica, C.; Negrea, R.; Dinescu, M. Joining Chemical Pressure and Epitaxial Strain to Yield Y-Doped BiFeO3 Thin Films with High Dielectric Response. Sci. Rep. 2016, 6, 25535. [Google Scholar] [CrossRef]
- Ion, V.; Craciun, F.; Scarisoreanu, N.D.; Moldovan, A.; Andrei, A.; Birjega, R.; Ghica, C.; Di Pietrantonio, F.; Cannata, D.; Benetti, M.; et al. Impact of Thickness Variation on Structural, Dielectric and Piezoelectric Properties of (Ba,Ca)(Ti,Zr)O3 Epitaxial Thin Films. Sci. Rep. 2018, 8, 2056. [Google Scholar] [CrossRef] [PubMed]
- Scarisoreanu, N.D.; Craciun, F.; Moldovan, A.; Ion, V.; Birjega, R.; Ghica, C.; Negrea, R.F.; Dinescu, M. High Permittivity (1–x)Ba(Zr0.2Ti0.8)O3–x(Ba0.7Ca0.3)TiO3 (x = 0.45) Epitaxial Thin Films with Nanoscale Phase Fluctuations. ACS Appl. Mater. Interfaces 2015, 7, 23984–23992. [Google Scholar] [CrossRef] [PubMed]
- Haydous, F.; Scarisoreanu, N.D.; Birjega, R.; Ion, V.; Lippert, T.; Dumitrescu, N.; Moldovan, A.; Andrei, A.; Teodorescu, V.S.; Ghica, C.; et al. Rolling Dopant and Strain in Y-Doped BiFeO3 Epitaxial Thin Films for Photoelectrochemical Water Splitting. Sci. Rep. 2018, 8, 15826. [Google Scholar] [CrossRef]
- Takata, T.; Domen, K. Defect Engineering of Photocatalysts by Doping of Aliovalent Metal Cations for Efficient Water Splitting. J. Phys. Chem. C 2009, 113, 19386–19388. [Google Scholar] [CrossRef]
- Ikeda, S.; Hirao, K.; Ishino, S.; Matsumura, M.; Ohtani, B. Preparation of Platinized Strontium Titanate Covered with Hollow Silica and Its Activity for Overall Water Splitting in a Novel Phase-Boundary Photocatalytic System. Catal. Today 2006, 117, 343–349. [Google Scholar] [CrossRef]
- Kato, H.; Asakura, K.; Kudo, A. Highly Efficient Water Splitting into H2 and O2 over Lanthanum-Doped NaTaO3 Photocatalysts with High Crystallinity and Surface Nanostructure. J. Am. Chem. Soc. 2003, 125, 3082–3089. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, G.; Li, Y.; Teng, Y. Electronic structure and photocatalytic properties of copper-doped CaTiO3. Int. J. Hydrogen. Energy 2010, 35, 2713–2716. [Google Scholar] [CrossRef]
- Li, J.; Jia, L.; Fang, W.; Zeng, J. Enhancement of activity of LaNi0.7Cu0.3O3 for photocatalytic water splitting by reduction treatment at moderate temperature. Int. J. Hydrogen. Energy 2010, 35, 5270–5275. [Google Scholar] [CrossRef]
- Ramachandran, B.; Ramachandra Rao, M.S. Chemical Pressure Effect on Optical Properties in Multiferroic Bulk BiFeO3. J. Appl. Phys. 2012, 112, 73516. [Google Scholar] [CrossRef]
- Ji, W.; Yao, K.; Lim, Y.-F.; Liang, Y.C.; Suwardi, A. Epitaxial Ferroelectric BiFeO3 Thin Films for Unassisted Photocatalytic Water Splitting. Appl. Phys. Lett. 2013, 103, 62901. [Google Scholar] [CrossRef]
- Li, F.; Liu, Y.; Liu, R.; Sun, Z.; Zhao, D.; Kou, C. Preparation of Ca-Doped LaFeO3 Nanopowders in a Reverse Microemulsion and Their Visible Light Photocatalytic Activity. Mater. Lett. 2010, 64, 223–225. [Google Scholar] [CrossRef]
- Li, S.; Jing, L.; Fu, W.; Yang, L.; Xin, B.; Fu, H. Photoinduced Charge Property of Nanosized Perovskite-Type LaFeO3 and Its Relationships with Photocatalytic Activity under Visible Irradiation. Mater. Res. Bull. 2007, 42, 203–212. [Google Scholar] [CrossRef]
- Celorrio, V.; Bradley, K.; Weber, O.J.; Hall, S.R.; Fermín, D.J. Photoelectrochemical Properties of LaFeO3 Nanoparticles. ChemElectroChem 2014, 1, 1667–1671. [Google Scholar] [CrossRef]
- Parida, K.M.; Reddy, K.H.; Martha, S.; Das, D.P.; Biswal, N. Fabrication of Nanocrystalline LaFeO3: An Efficient Sol–Gel Auto-Combustion Assisted Visible Light Responsive Photocatalyst for Water Decomposition. Int. J. Hydrogen. Energy 2010, 35, 12161–12168. [Google Scholar] [CrossRef]
- Natali Sora, I.; Fontana, F.; Passalacqua, R.; Ampelli, C.; Perathoner, S.; Centi, G.; Parrino, F.; Palmisano, L. Photoelectrochemical Properties of Doped Lanthanum Orthoferrites. Electrochim. Acta 2013, 109, 710–715. [Google Scholar] [CrossRef]
- Pawar, G.S.; Tahir, A.A. Unbiased Spontaneous Solar Fuel Production Using Stable LaFeO3 Photoelectrode. Sci. Rep. 2018, 8, 3501. [Google Scholar] [CrossRef]
- Zavaliche, F.; Zheng, H.; Mohaddes-Ardabili, L.; Yang, S.Y.; Zhan, Q.; Shafer, P.; Reilly, E.; Chopdekar, R.; Jia, Y.; Wright, P.; et al. Electric Field-Induced Magnetization Switching in Epitaxial Columnar Nanostructures. Nano Lett. 2005, 5, 1793–1796. [Google Scholar] [CrossRef]
- MacManus-Driscoll, J.L.; Zerrer, P.; Wang, H.; Yang, H.; Yoon, J.; Fouchet, A.; Yu, R.; Blamire, M.G.; Jia, Q. Strain Control and Spontaneous Phase Ordering in Vertical Nanocomposite Heteroepitaxial Thin Films. Nat. Mater. 2008, 7, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Moshnyaga, V.; Damaschke, B.; Shapoval, O.; Belenchuk, A.; Faupel, J.; Lebedev, O.I.; Verbeeck, J.; van Tendeloo, G.; Mücksch, M.; Tsurkan, V.; et al. Structural Phase Transition at the Percolation Threshold in Epitaxial (La0.7Ca0.3MnO3)1–X:(MgO)x Nanocomposite Films. Nat. Mater. 2003, 2, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Bi, L.; Kim, H.-S.; Dionne, G.F.; Ross, C.A.; Paik, H.; Park, Y.C. Orientation Control and Self-Assembled Nanopyramid Structure of LaFeO3 Films Epitaxially Grown on SrTiO3(001) Substrates. Appl. Phys. Lett. 2009, 95, 121908. [Google Scholar] [CrossRef]
- May, K.J.; Fenning, D.P.; Ming, T.; Hong, W.T.; Lee, D.; Stoerzinger, K.A.; Biegalski, M.D.; Kolpak, A.M.; Shao-Horn, Y. Thickness-Dependent Photoelectrochemical Water Splitting on Ultrathin LaFeO3 Films Grown on Nb:SrTiO3. J. Phys. Chem. Lett. 2015, 6, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Geller, S.; Wood, E.A. Crystallographic Studies of Perovskite-like Compounds. I. Rare Earth Orthoferrites and YFeO3, YCrO3, YAlO3. Acta Cryst. 1956, 9, 563–568. [Google Scholar] [CrossRef]
- Seo, J.W.; Fullerton, E.E.; Nolting, F.; Scholl, A.; Fompeyrine, J.; Locquet, J.-P. Antiferromagnetic LaFeO3 thin Films and Their Effect on Exchange Bias. J. Phys. Condens. Matter 2008, 20, 264014. [Google Scholar] [CrossRef]
- Scafetta, M.D.; May, S.J. Effect of Cation Off-Stoichiometry on Optical Absorption in Epitaxial LaFeO3 Films. Phys. Chem. Chem. Phys. 2017, 19, 10371–10376. [Google Scholar] [CrossRef]
- Williamson, G.K.; Hall, W.H. X-Ray Line Broadening from Filed Aluminium and Wolfram. Acta Metall. 1953, 1, 22–31. [Google Scholar] [CrossRef]
- Chierchia, R.; Böttcher, T.; Heinke, H.; Einfeldt, S.; Figge, S.; Hommel, D. Microstructure of Heteroepitaxial GaN Revealed by X-Ray Diffraction. J. Appl. Phys. 2003, 93, 8918–8925. [Google Scholar] [CrossRef]
- Metzger, T.; Höpler, R.; Born, E.; Ambacher, O.; Stutzmann, M.; Stömmer, R.; Schuster, M.; Göbel, H.; Christiansen, S.; Albrecht, M.; et al. Defect Structure of Epitaxial GaN Films Determined by Transmission Electron Microscopy and Triple-Axis X-Ray Diffractometry. Philos. Mag. A 1998, 77, 1013–1025. [Google Scholar] [CrossRef]
- Vickers, M.E.; Kappers, M.J.; Datta, R.; McAleese, C.; Smeeton, T.M.; Rayment, F.D.G.; Humphreys, C.J. In-Plane Imperfections in GaN Studied by x-Ray Diffraction. J. Phys. D Appl. Phys. 2005, 38, A99–A104. [Google Scholar] [CrossRef]
- Xu, X.; Li, Y.; Liu, J.; Wei, H.; Liu, X.; Yang, S.; Wang, Z.; Wang, H. X-Ray Probe of GaN Thin Films Grown on InGaN Compliant Substrates. Appl. Phys. Lett. 2013, 102, 132104. [Google Scholar] [CrossRef]
- Nandi, R.; Appani, S.K.; Major, S.S. Vertically Aligned ZnO Nanorods of High Crystalline and Optical Quality Grown by Dc Reactive Sputtering. Mater. Res. Express 2016, 3, 095009. [Google Scholar] [CrossRef]
- Norton, D.P.; Park, C.; Budai, J.D.; Pennycook, S.J.; Prouteau, C. Plume-Induced Stress in Pulsed-Laser Deposited CeO2 Films. Appl. Phys. Lett. 1999, 74, 2134–2136. [Google Scholar] [CrossRef]
- Grepstad, J.K.; Takamura, Y.; Scholl, A.; Hole, I.; Suzuki, Y.; Tybell, T. Effects of Thermal Annealing in Oxygen on the Antiferromagnetic Order and Domain Structure of Epitaxial LaFeO3 Thin Films. Thin Solid Films 2005, 486, 108–112. [Google Scholar] [CrossRef]
- Andrei, F.; Boerasu, I.; Birjega, R.; Moldovan, A.; Dinescu, M.; Ion, V.; Mihailescu, C.; Scarisoreanu, N.D.; Leca, V. The Effects of the Oxygen Content on the Photoelectrochemical Properties of LaFeO3 Perovskite Thin Films Obtained by Pulsed Laser Deposition. Appl. Phys. A 2019, 125, 807. [Google Scholar] [CrossRef]
- Breckenfeld, E.; Chen, Z.; Damodaran, A.; Martin, L. Effects of Nonequilibrium Growth, Nonstoichiometry, and Film Orientation on the Metal-to-Insulator Transition in NdNiO3 Thin Films. ACS Appl. Mater. Interfaces 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Comes, R.B.; Smolin, S.Y.; Kaspar, T.C.; Gao, R.; Apgar, B.A.; Martin, L.W.; Bowden, M.E.; Baxter, J.B.; Chambers, S.A. Visible Light Carrier Generation in Co-Doped Epitaxial Titanate Films. Appl. Phys. Lett. 2015, 106, 092901. [Google Scholar] [CrossRef]
- Chen, Y.; Bagnall, D.M.; Koh, H.; Park, K.; Hiraga, K.; Zhu, Z.; Yao, T. Plasma Assisted Molecular Beam Epitaxy of ZnO on c -Plane Sapphire: Growth and Characterization. J. Appl. Phys. 1998, 84, 3912–3918. [Google Scholar] [CrossRef]
- Zanetti, S.M.; Leite, E.R.; Longo, E.; Varela, J.A. Preparation of Ferroelectric Bi-Layered Thin Films Using the Modified Polymeric Precursor Method. Mater. Res. 2001, 4, 157–162. [Google Scholar] [CrossRef]
- Rangel, J.H.G.; Bernardi, M.I.B.; Paskocimas, C.A.; Longo, E.; Varela, J.A. Study on the Orientation Degree of Pb1−xLaxTiO3 Thin Films by the Rocking Curve Technique and Its Morphological Aspects. Surf. Coat. Technol. 2007, 201, 6345–6351. [Google Scholar] [CrossRef]
- Yu, Q.; Meng, X.; Wang, T.; Li, P.; Liu, L.; Chang, K.; Liu, G.; Ye, J. A Highly Durable P-LaFeO3/n-Fe2O3 Photocell for Effective Water Splitting under Visible Light. Chem. Commun. 2015, 51, 3630–3633. [Google Scholar] [CrossRef]
- Peng, Q.; Wang, J.; Wen, Y.W.; Shan, B.; Chen, R. Surface Modification of LaFeO3 by Co-Pi Electrochemical Deposition as an Efficient Photoanode under Visible Light. RSC Adv. 2016, 6, 26192–26198. [Google Scholar] [CrossRef]
- Fujiwara, H. Spectroscopic Ellipsometry: Principles and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2007; ISBN 978-0470016084. [Google Scholar]
- Scafetta, M.D.; Cordi, A.M.; Rondinelli, J.M.; May, S.J. Band Structure and Optical Transitions in LaFeO3: Theory and Experiment. J. Phys. Condens. Matter 2014, 26, 505502. [Google Scholar] [CrossRef] [PubMed]
- SIMNRA—Software for Simulation of RBS, ERDA, NRA, MEIS and PIGE. Available online: https://www2.ipp.mpg.de/~mam/ (accessed on 30 April 2021).





| D (nm) | Structural Data | |||||
|---|---|---|---|---|---|---|
| a out-of-plane (nm) | a in-plane (nm) | L┴ (nm) | ε┴ Microstrain (%) | αtilt (°) | LII (nm) | |
| 14(15) | 3.950 | 3.899 | 13 | 0.01 | 0.095 | 512 |
| 21(24) | 3.948 | 3.902 | 35 | 0.06 | 0.09 | 752 |
| 29 | 3.944 | n.det | 32 | 0.02 | 0.13 | 350 |
| 32 | 3.941 | 3.904 | 33 | 0.25 | 0.14 | 151 |
| 132(140) | 3.935 | n.det | 71 | 0.14 | 0.18 | 195 |
| 192 | 3.927 | n.det | 127 | 0.40 | 1.22 | 38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrei, F.; Ion, V.; Bîrjega, R.; Dinescu, M.; Enea, N.; Pantelica, D.; Mihai, M.D.; Maraloiu, V.-A.; Teodorescu, V.S.; Marcu, I.-C.; et al. Thickness-Dependent Photoelectrochemical Water Splitting Properties of Self-Assembled Nanostructured LaFeO3 Perovskite Thin Films. Nanomaterials 2021, 11, 1371. https://doi.org/10.3390/nano11061371
Andrei F, Ion V, Bîrjega R, Dinescu M, Enea N, Pantelica D, Mihai MD, Maraloiu V-A, Teodorescu VS, Marcu I-C, et al. Thickness-Dependent Photoelectrochemical Water Splitting Properties of Self-Assembled Nanostructured LaFeO3 Perovskite Thin Films. Nanomaterials. 2021; 11(6):1371. https://doi.org/10.3390/nano11061371
Chicago/Turabian StyleAndrei, Florin, Valentin Ion, Ruxandra Bîrjega, Maria Dinescu, Nicoleta Enea, Dan Pantelica, Maria Diana Mihai, Valentin-Adrian Maraloiu, Valentin Serban Teodorescu, Ioan-Cezar Marcu, and et al. 2021. "Thickness-Dependent Photoelectrochemical Water Splitting Properties of Self-Assembled Nanostructured LaFeO3 Perovskite Thin Films" Nanomaterials 11, no. 6: 1371. https://doi.org/10.3390/nano11061371
APA StyleAndrei, F., Ion, V., Bîrjega, R., Dinescu, M., Enea, N., Pantelica, D., Mihai, M. D., Maraloiu, V.-A., Teodorescu, V. S., Marcu, I.-C., & Scarisoreanu, N. D. (2021). Thickness-Dependent Photoelectrochemical Water Splitting Properties of Self-Assembled Nanostructured LaFeO3 Perovskite Thin Films. Nanomaterials, 11(6), 1371. https://doi.org/10.3390/nano11061371









