Glyconanomaterials for Human Virus Detection and Inhibition
Abstract
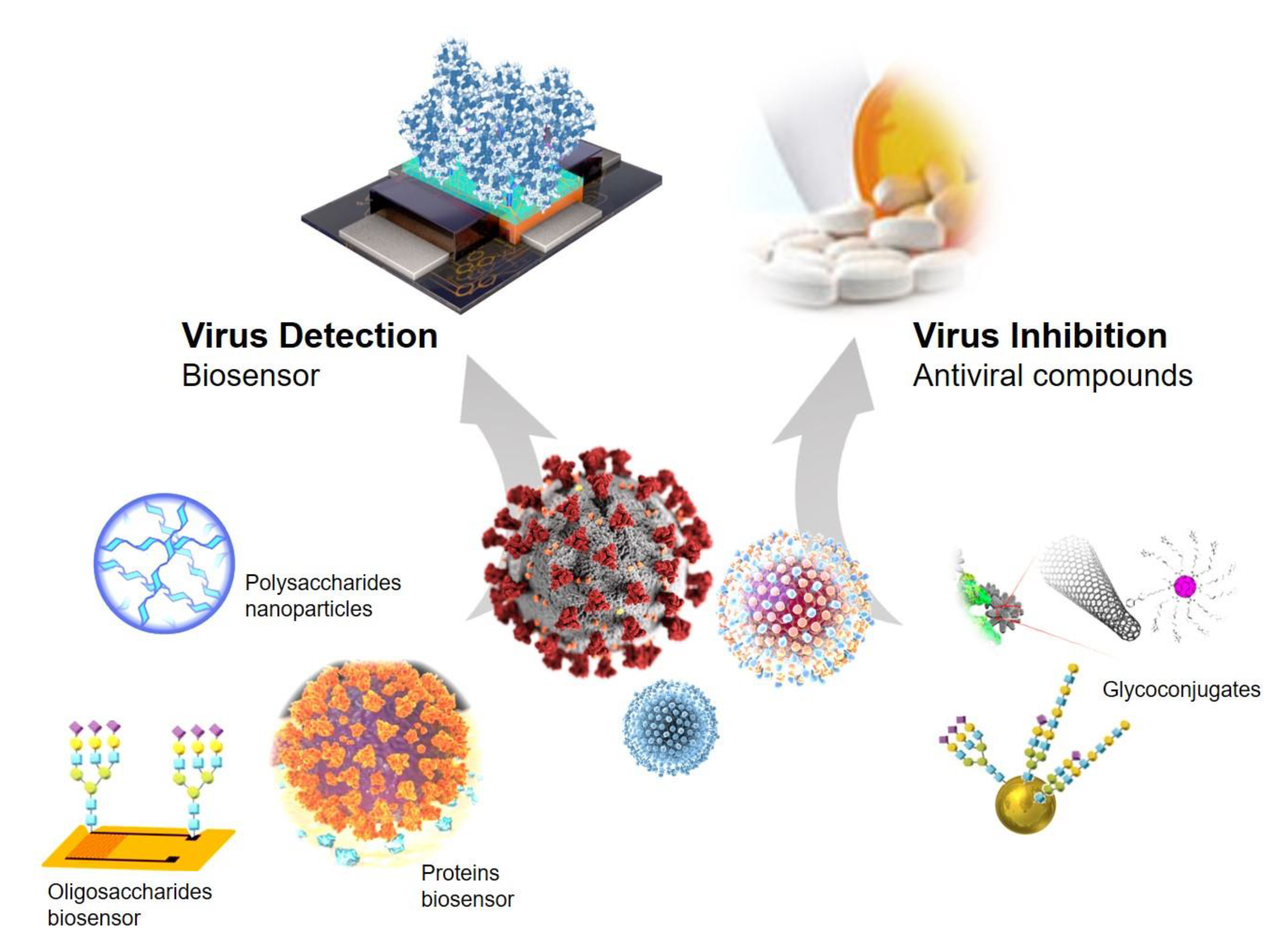
:1. Introduction
2. Proteins or Oligosaccharides Immobilized in Nanomaterials for Glycoprotein-Virus Detection
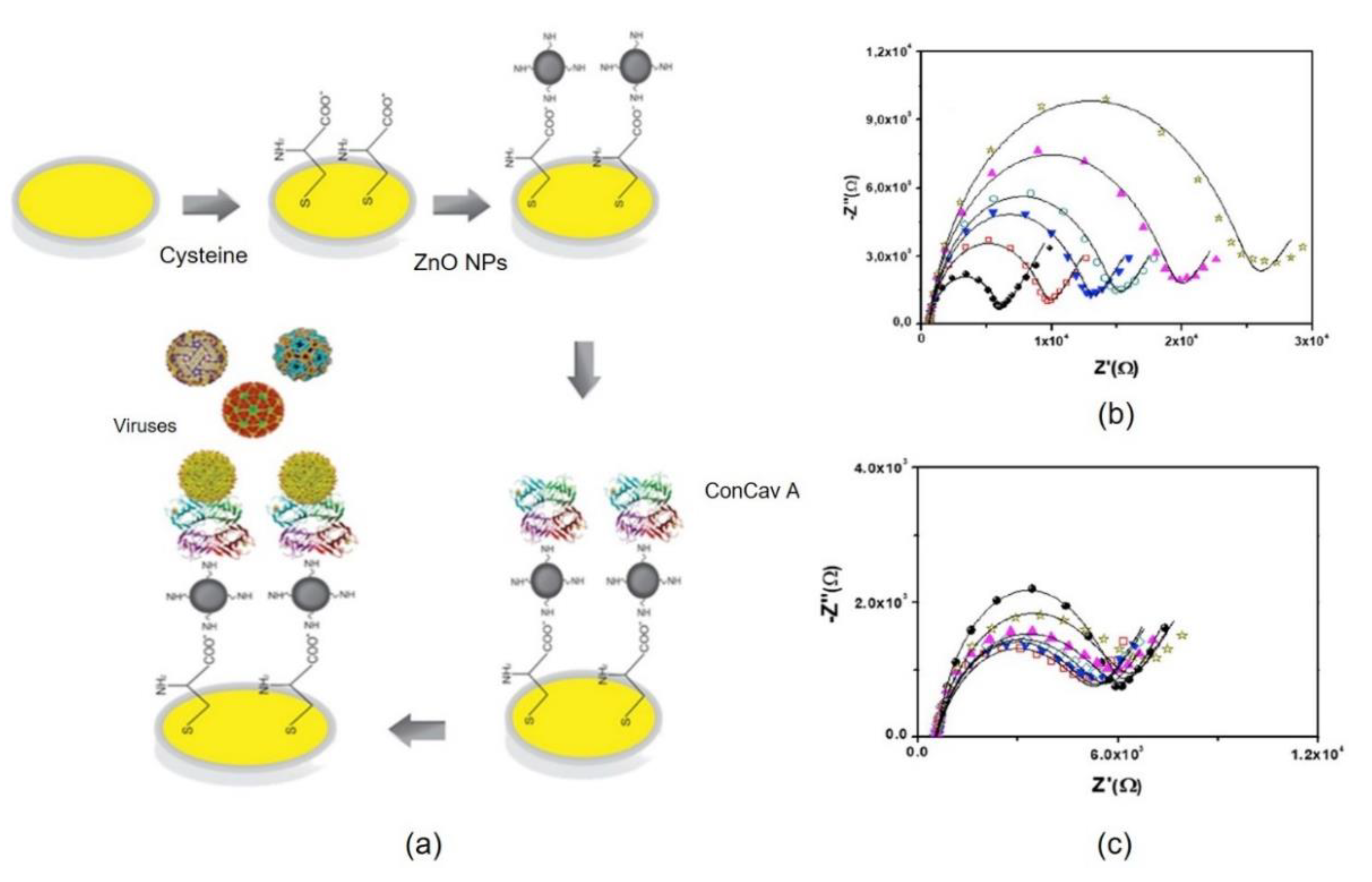
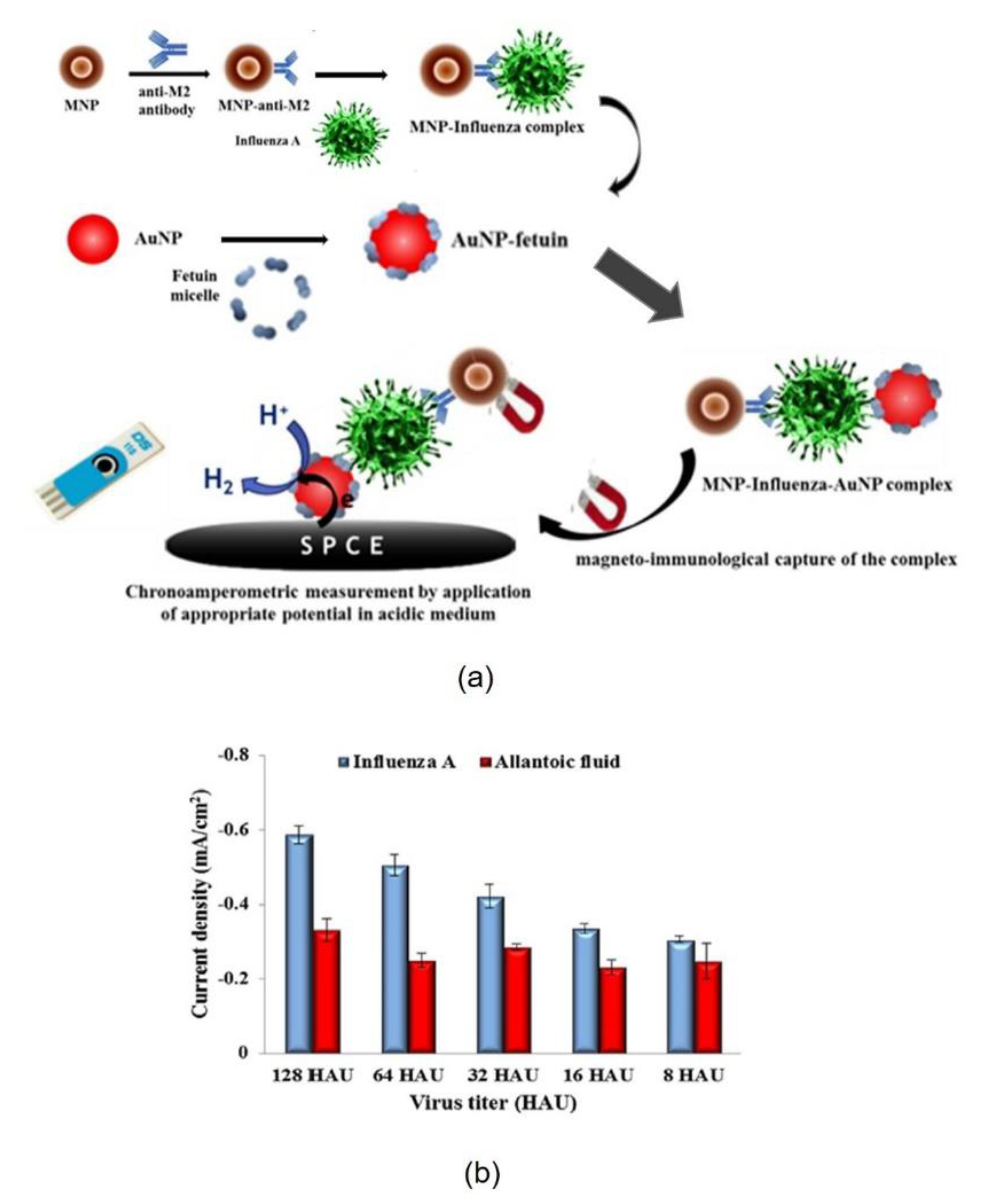
2.1. Proteins Immobilized in Nanomaterials
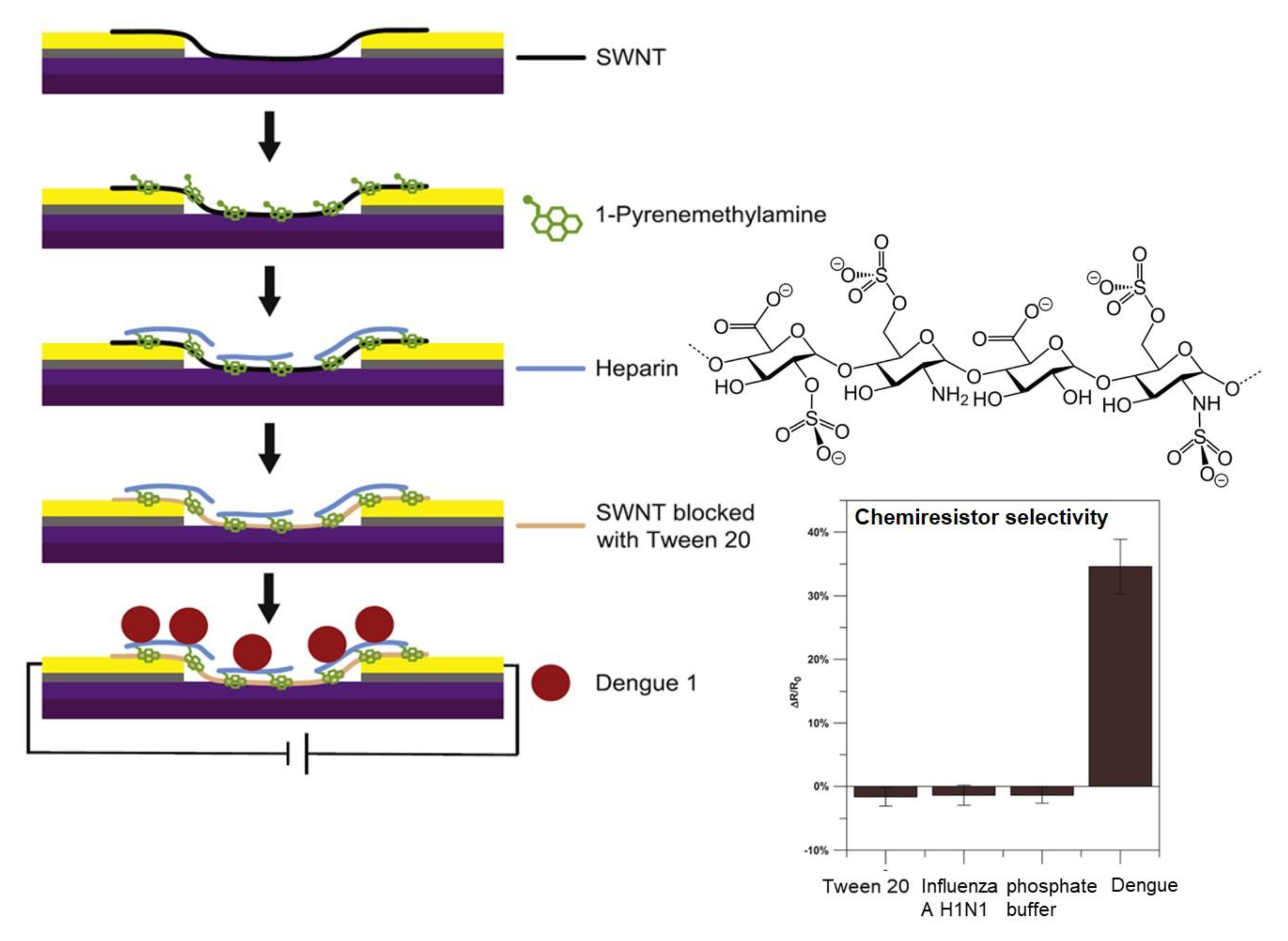
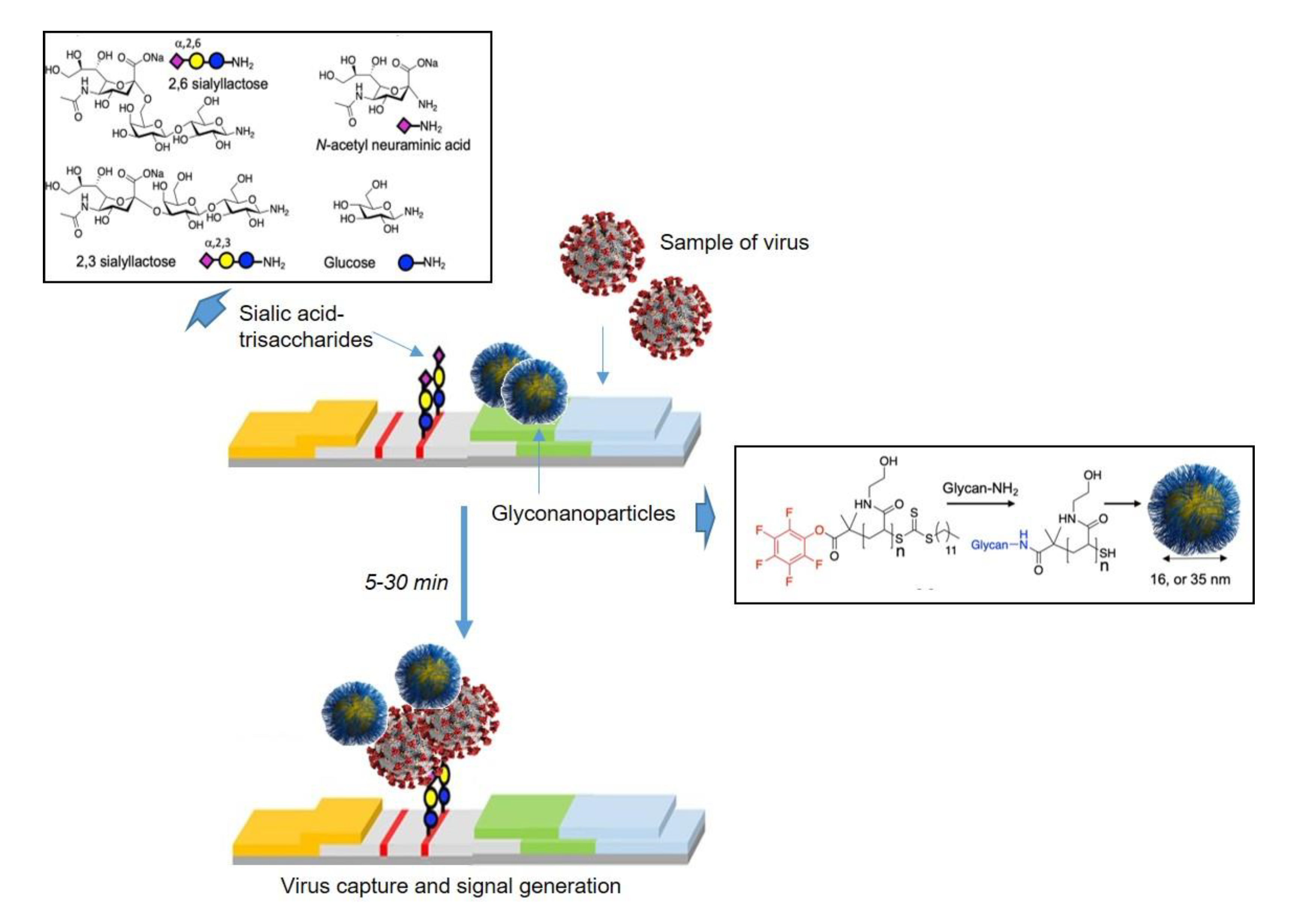
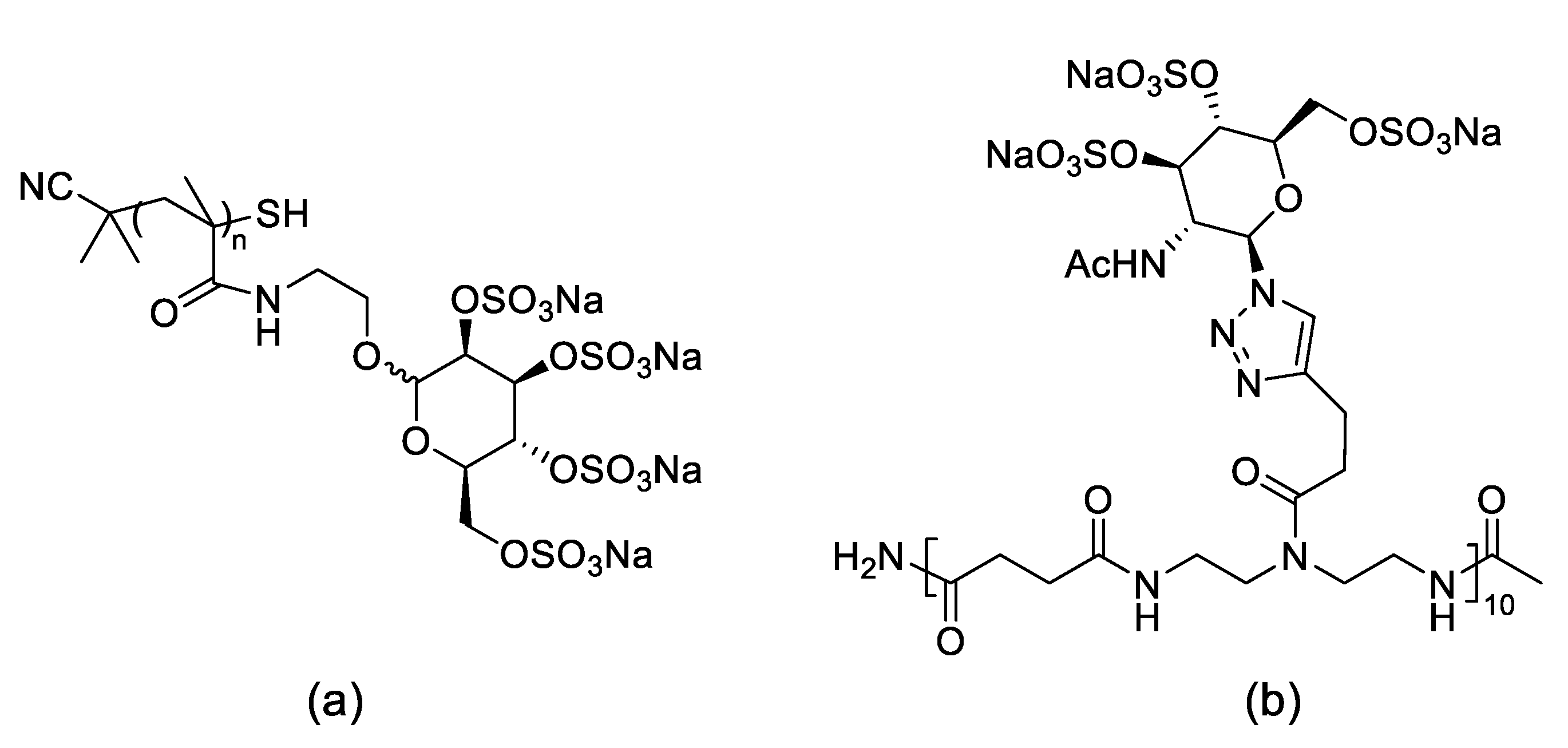
2.2. Oligosaccharides Immobilized in Nanomaterials
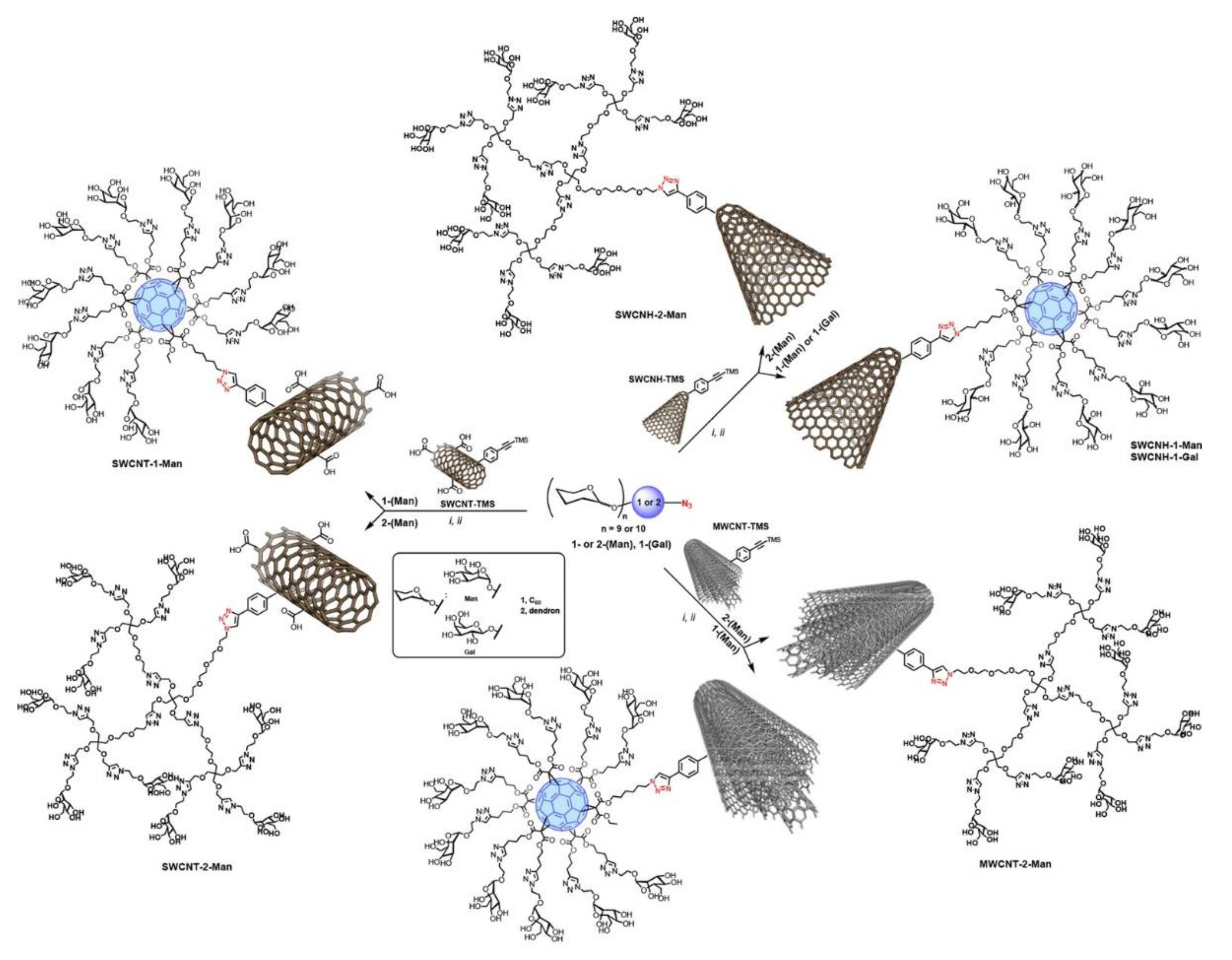
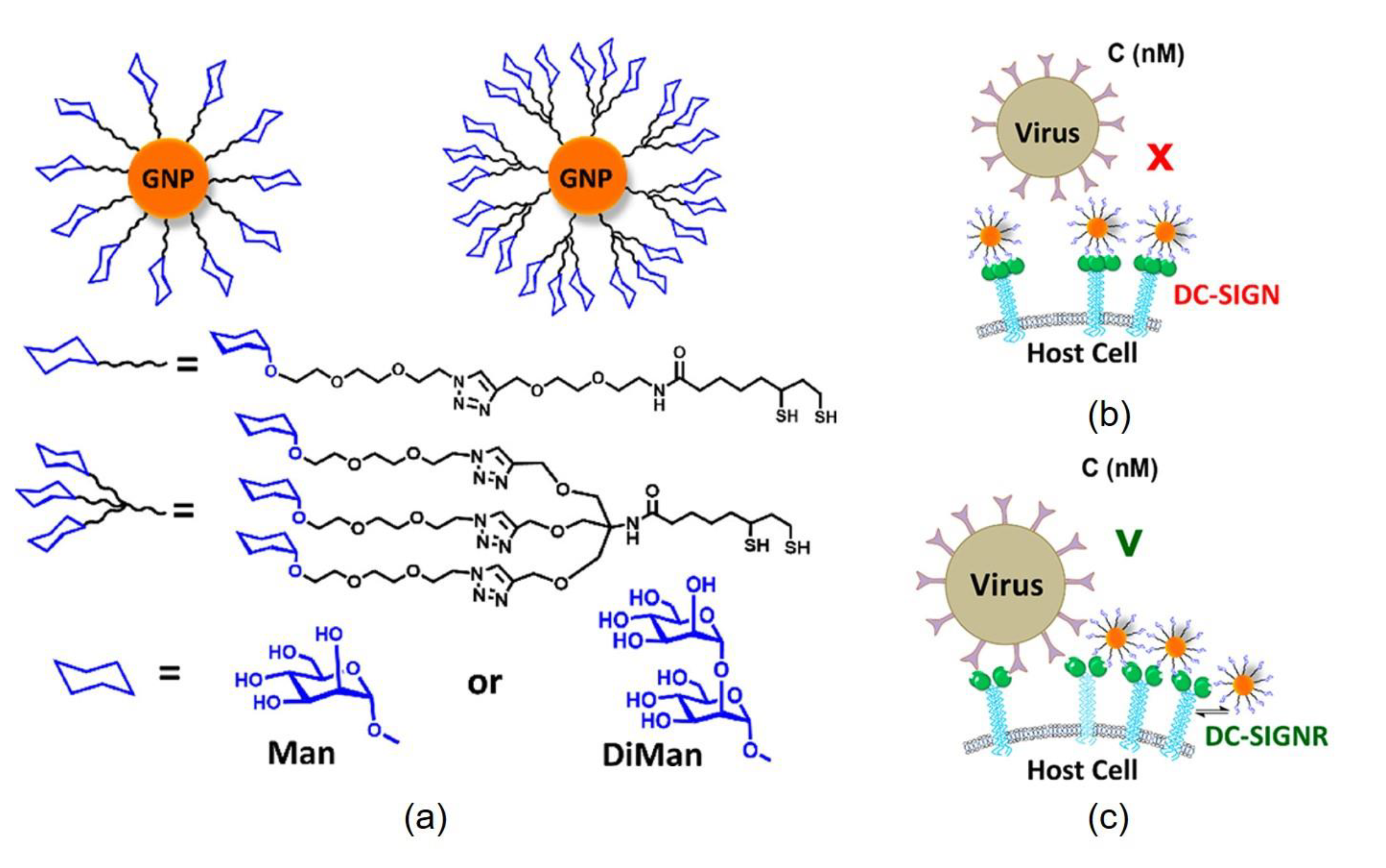
3. Glyconanoconjugates as Novel Antiviral Drugs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Liu, S.-L.; Wang, Z.-G.; Zhang, Z.-L.; Pang, D.-W. Tracking single viruses infecting their host cells using quantum dots. Chem. Soc. Rev. 2015, 45, 1211–1224. [Google Scholar] [CrossRef] [Green Version]
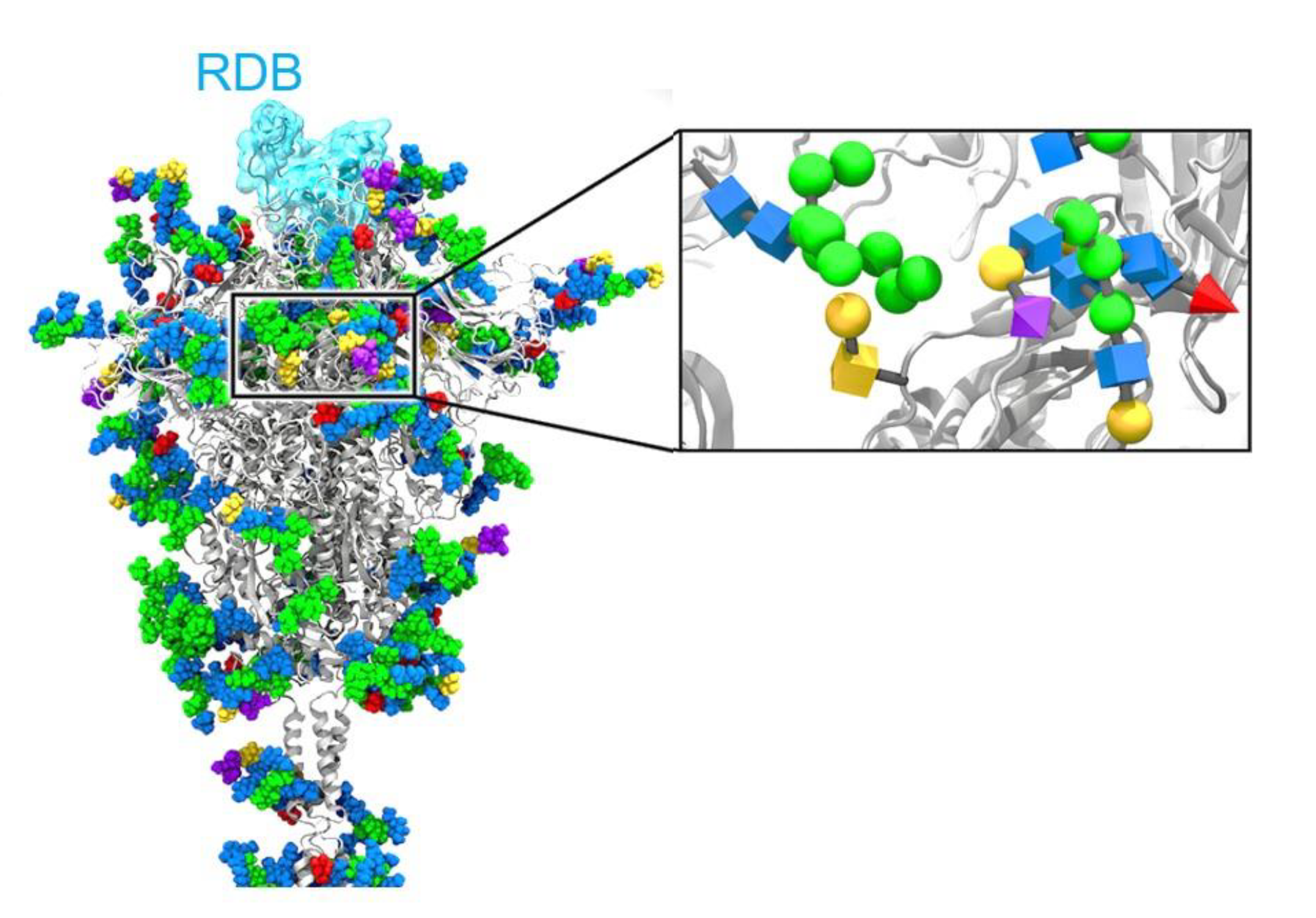
- Grant, O.C.; Montgomery, D.; Ito, K.; Woods, R.J. Analysis of the SARS-CoV-2 spike protein glycan shield reveals implications for immune recognition. Sci. Rep. 2020, 10, 1–11. [Google Scholar]
- Tate, M.D.; Job, E.R.; Deng, Y.-M.; Gunalan, V.; Maurer-Stroh, S.; Reading, P.C. Playing Hide and Seek: How Glycosylation of the Influenza Virus Hemagglutinin Can Modulate the Immune Response to Infection. Viruses 2014, 6, 1294–1316. [Google Scholar] [CrossRef] [Green Version]
- Baum, L.G.; Cobb, B.A. The direct and indirect effects of glycans on immune function. Glycobiology 2017, 27, 619–624. [Google Scholar] [CrossRef]
- Vigerust, D.J.; Shepherd, V.L. Virus glycosylation: Role in virulence and immune interactions. Trends Microbiol. 2007, 15, 211–218. [Google Scholar] [CrossRef]
- Altman, M.O.; Angel, M.; Košík, I.; Trovão, N.S.; Zost, S.J.; Gibbs, J.S.; Casalino, L.; Amaro, R.E.; Hensley, S.E.; Nelson, M.I.; et al. Human Influenza A Virus Hemagglutinin Glycan Evolution Follows a Temporal Pattern to a Glycan Limit. mBio 2019, 10, e00204-19. [Google Scholar] [CrossRef] [Green Version]
- Li, Q.; Wu, J.; Nie, J.; Zhang, L.; Hao, H.; Liu, S.; Zhao, C.; Zhang, Q.; Liu, H.; Nie, L.; et al. The impact of mutations in SARS-CoV-2 spike on viral infectivity and antigenicity. Cell 2020, 182, 1284–1294. [Google Scholar] [CrossRef]
- World Health Organization. World Health Organization COVID-19 Dashboard. Available online: https://covid19.who.int/ (accessed on 28 April 2021).
- Draz, M.S.; Vasan, A.; Muthupandian, A.; Kanakasabapathy, M.K.; Thirumalaraju, P.; Sreeram, A.; Krishnakumar, S.; Yogesh, V.; Lin, W.; Yu, X.G.; et al. Virus detection using nanoparticles and deep neural network–enabled smartphone system. Sci. Adv. 2020, 6, eabd5354. [Google Scholar] [CrossRef]
- Wood, C.S.; Thomas, M.R.; Budd, J.; Mashamba-Thompson, T.; Herbst, K.; Pillay, D.; Peeling, R.W.; Johnson, A.M.; McKendry, R.A.; Stevens, M.M. Taking connected mobile-health diagnostics of infectious diseases to the field. Nat. Cell Biol. 2019, 566, 467–474. [Google Scholar] [CrossRef] [Green Version]
- Draz, M.S.; Lakshminaraasimulu, N.K.; Krishnakumar, S.; Battalapalli, D.; Vasan, A.; Kanakasabapathy, M.K.; Sreeram, A.; Kallakuri, S.; Thirumalaraju, P.; Li, Y.; et al. Motion-Based Immunological Detection of Zika Virus Using Pt-Nanomotors and a Cellphone. ACS Nano 2018, 12, 5709–5718. [Google Scholar] [CrossRef]
- Zhang, L.-J.; Wang, S.; Xia, L.; Lv, C.; Tang, H.-W.; Liang, Z.; Xiao, G.; Pang, D.-W. Lipid-Specific Labeling of Enveloped Viruses with Quantum Dots for Single-Virus Tracking. mBio 2020, 11, e00135-20. [Google Scholar] [CrossRef]
- Castillo-Henríquez, L.; Brenes-Acuña, M.; Castro-Rojas, A.; Cordero-Salmerón, R.; Lopretti-Correa, M.; Vega-Baudrit, J.R. Biosensors for the Detection of Bacterial and Viral Clinical Pathogens. Sensors 2020, 20, 6926. [Google Scholar] [CrossRef]
- Varki, A. Biological roles of glycans. Glycobiology 2017, 27, 3–49. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Liu, X.; Chen, J.; Su, H.; Luo, Q.; Ye, J.; Tang, N.; Zhang, W.; Chen, W.; Ko, B.C.; et al. Heparin sulphate d-glucosaminyl 3-O-sulfotransferase 3B1 plays a role in HBV replication. Virology 2010, 406, 280–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Hao, Y.; Deng, D.; Xia, N. Nanomaterials-Based Colorimetric Immunoassays. Nanomaterials 2019, 9, 316. [Google Scholar] [CrossRef] [Green Version]
- Alhalaili, B.; Popescu, I.N.; Kamoun, O.; Alzubi, F.; Alawadhia, S.; Vidu, R. Nanobiosensors for the detection of novel c coronavirus 2019-nCoV and other pandemic/epidemic respiratory viruses: A Review. Sensors 2020, 20, 6591. [Google Scholar] [CrossRef]
- Simão, E.P.; Silva, D.B.; Cordeiro, M.T.; Gil, L.H.; Andrade, C.A.; Oliveira, M.D. Nanostructured impedimetric lectin-based biosensor for arboviruses detection. Talanta 2020, 208, 120338. [Google Scholar] [CrossRef]
- Sayhi, M.; Ouerghi, O.; Belgacem, K.; Arbi, M.; Tepeli, Y.; Ghram, A.; Anik, Ü.; Österlund, L.; Laouini, D.; Diouani, M.F. Electrochemical detection of influenza virus H9N2 based on both immunomagnetic extraction and gold catalysis using an immobilization-free screen printed carbon microelectrode. Biosens. Bioelectron. 2018, 107, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Parate, K.; Rangnekar, S.V.; Jing, D.; Mendivelso-Perez, D.L.; Ding, S.; Secor, E.B.; Smith, E.A.; Hostetter, J.; Hersam, M.C.; Claussen, J.C. Aerosol-Jet-Printed Graphene Immunosensor for Label-Free Cytokine Monitoring in Serum. ACS Appl. Mater. Interfaces 2020, 12, 8592–8603. [Google Scholar] [CrossRef]
- Loris, R.; Hamelryck, T.; Bouckaert, J.; Wyns, L. Legume lectin structure. Biochim. Biophys. Act. (BBA) Prot. Struct. Mol. Enzym. 1998, 1383, 9–36. [Google Scholar] [CrossRef]
- Wilder-Smith, A.; Murray, N.E.A.; Quam, M. Epidemiology of dengue: Past, present and future prospects. Clin. Epidemiol. 2013, 5, 299–309. [Google Scholar] [CrossRef] [Green Version]
- Kuno, G.; Chang, G.-J.J. Biological Transmission of Arboviruses: Reexamination of and New Insights into Components, Mechanisms, and Unique Traits as Well as Their Evolutionary Trends. Clin. Microbiol. Rev. 2005, 18, 608–637. [Google Scholar] [CrossRef] [Green Version]
- Sirohi, D.; Chen, Z.; Sun, L.; Klose, T.; Pierson, T.C.; Rossmann, M.G.; Kuhn, R.J. The 3.8 A resolution cryo-EM structure of Zika virus. Science 2016, 352, 467–470. [Google Scholar] [CrossRef] [Green Version]
- Carinelli, S.; Martí, M.; Alegret, S.; Pividori, M.I. Biomarker detection of global infectious diseases based on magnetic particles. New Biotechnol. 2015, 32, 521–532. [Google Scholar] [CrossRef]
- Thompson, A.J.; de Vries, R.; Paulson, J.C. Virus recognition of glycan receptors. Curr. Opin. Virol. 2019, 34, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Wasik, D.; Mulchandani, A.; Yates, M.V. A heparin-functionalized carbon nanotube-based affinity biosensor for dengue virus. Biosens. Bioelectron. 2017, 91, 811–816. [Google Scholar] [CrossRef]
- Baker, A.N.; Richards, S.-J.; Guy, C.S.; Congdon, T.R.; Hasan, M.; Zwetsloot, A.J.; Gallo, A.; Lewandowski, J.R.; Stansfeld, P.J.; Straube, A.; et al. The SARS-COV-2 Spike protein binds sialic acdis and enables rapid detection in a lateral flow point of care diagnostic device. ACS Cent. Sci. 2020, 6, 2046–2052. [Google Scholar] [CrossRef]
- Okemoto-Nakamura, Y.; Someya, K.; Yamaji, T.; Saito, K.; Takeda, M.; Hanada, K. Poliovirus-nonsusceptible Vero cell line for the World Health Organization global action plan. Sci. Rep. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- WHO Situation Report—71. Coronavirus Disease 2019 (COVID-19). WHO, 2020. Available online: https://www.who.int (accessed on 11 April 2020).
- Tortorici, M.A.; Walls, A.C.; Lang, Y.; Wang, C.; Li, Z.; Koerhuis, D.; Boons, G.-J.; Bosch, B.-J.; Rey, F.A.; De Groot, R.J.; et al. Structural basis for human coronavirus attachment to sialic acid receptors. Nat. Struct. Mol. Biol. 2019, 26, 481–489. [Google Scholar] [CrossRef] [Green Version]
- Casalino, L.; Gaieb, Z.; Goldsmith, J.A.; Hjorth, C.K.; Dommer, A.C.; Harbison, A.M.; Fogarty, C.A.; Barros, E.P.; Taylor, B.C.; McLellan, J.S.; et al. Beyond Shielding: The Roles of Glycans in the SARS-CoV-2 Spike Protein. ACS Cent. Sci. 2020, 6, 1722–1734. [Google Scholar] [CrossRef]
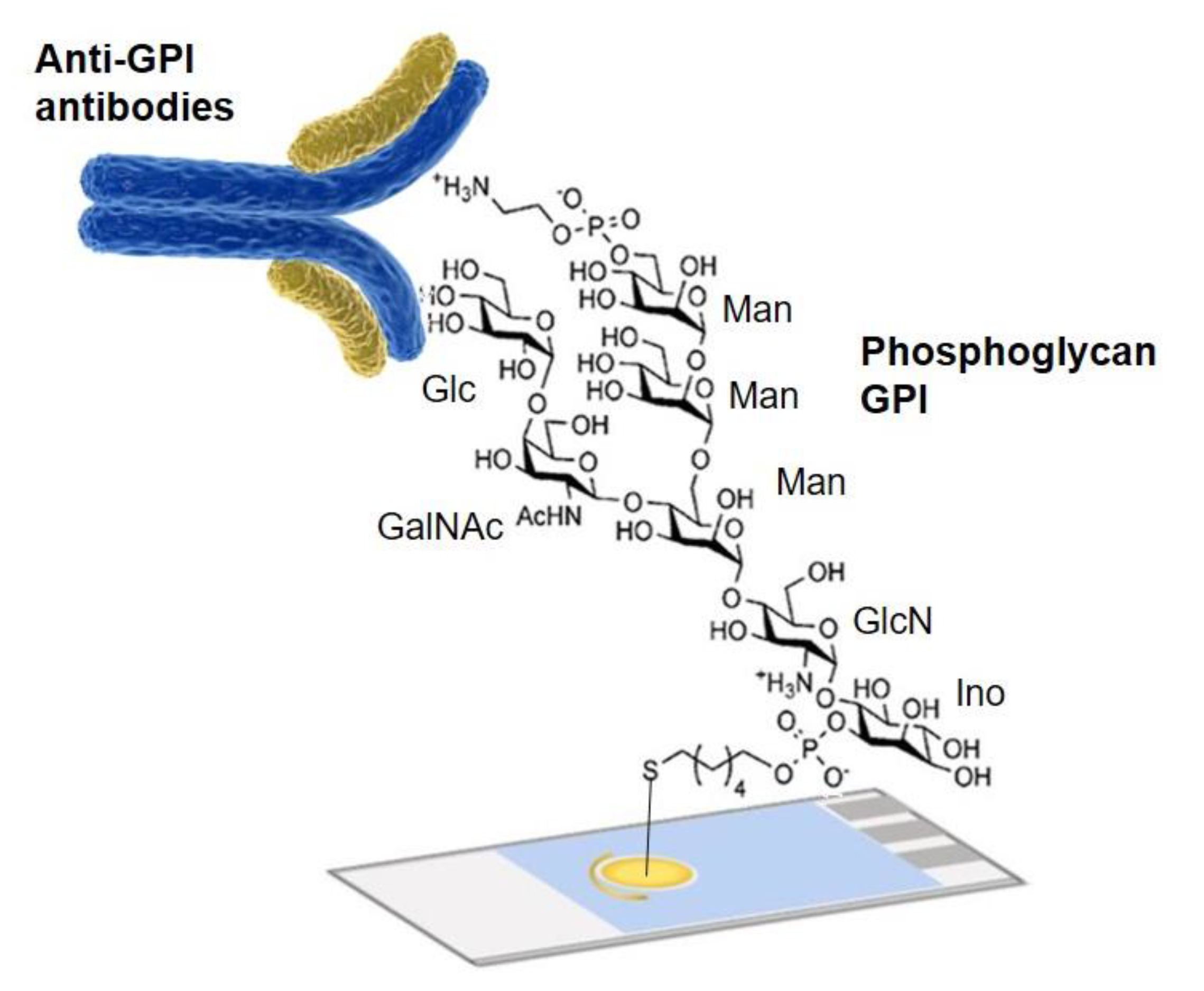
- Götze, S.; Azzouz, N.; Tsai, Y.H.; Groß, U.; Reinhardt, A.; Anish, C.; Seeberger, P.H.; Silva, D.V. Diagnosis of toxo-plasmosis using a synthetic glycosylphosphatidyl-inositol glycan. Angew. Chem. Int. Ed. 2014, 53, 13701–13705. [Google Scholar] [CrossRef]
- Saha, S.; Anilkumar, A.A.; Mayor, S. GPI-anchored protein organization and dynamics at the cell surface. J. Lipid Res. 2016, 57, 159–175. [Google Scholar] [CrossRef] [Green Version]
- Tsai, Y.-H.; Liu, X.; Seeberger, P.H. Chemical Biology of Glycosylphosphatidylinositol Anchors. Angew. Chem. Int. Ed. 2012, 51, 11438–11456. [Google Scholar] [CrossRef]
- Butler, D.L.; Gildersleeve, J.C. Abnormal antibodies to self-carbohydrates in SARS-CoV-2 infected patients. Europe PMC. bioRxiv 2020. [Google Scholar] [CrossRef]
- Echeverri, D.; Garg, M.; Silva, D.V.; Orozco, J. Phosphoglycan-sensitized platform for specific detection of anti-glycan IgG and IgM antibodies in serum. Talanta 2020, 217, 121117. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, T.; Fujita, M.; Maeda, Y. Biosynthesis, remodelling and functions of mammalian GPI-anchored proteins: Recent progress. J. Biochem. 2008, 144, 287–294. [Google Scholar] [CrossRef]
- Tashiro, H.; Brenner, M.K. Immunotherapy against cancer-related viruses. Cell Res. 2017, 27, 59–73. [Google Scholar] [CrossRef] [Green Version]
- Wannasarit, S.; Wang, S.; Figueiredo, P.; Trujillo, C.; Eburnea, F.; Simón-Gracia, L.; Correia, A.; Ding, Y.; Teesalu, T.; Liu, D.; et al. A virus-mimicking pH-responsive acetalated dextran-based membrane-active polymeric nanoparticle for intracellular delivery of antitumor therapeutics. Adv. Funct. Mater. 2019, 29, 1905352. [Google Scholar] [CrossRef]
- Bamberger, D.; Hobernik, D.; Konhäuser, M.; Bros, M.; Wich, P.R. Surface modification of polysaccharide-based nano-particles with PEG and dextran and the effects on immune cell binding and stimulatory characteristics. Mol. Pharm. 2017, 14, 4403–4416. [Google Scholar] [CrossRef]
- Ciejka, J.; Wolski, K.; Nowakowska, M.; Pyrc, K.; Szczubiałka, K. Biopolymeric nano/microspheres for selective and reversible adsorptionof coronaviruses. Mater. Sci. Eng. C 2017, 76, 735–742. [Google Scholar] [CrossRef]
- Buck, C.; Thompson, C.D.; Roberts, J.N.; Müller, M.; Lowy, D.R.; Schiller, J.T. Carrageenan Is a Potent Inhibitor of Papillomavirus Infection. PLoS Pathog. 2006, 2, e69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Soria-Martinez, L.; Bauer, S.; Giesler, M.; Schelhaas, S.; Materlik, J.; Janus, K.A.; Pierzyna, P.; Becker, M.; Snyder, N.L.; Hartmann, L.; et al. Prophylactic Antiviral Activity of Sulfated Glycomimetic Oligomers and Polymers. J. Am. Chem. Soc. 2020, 142, 5252–5265. [Google Scholar] [CrossRef]
- Wesener, D.A.; Dugan, A.; Kiessling, L.L. Recognition of microbial glycans by soluble human lectins. Curr. Opin. Struct. Biol. 2017, 44, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Baribaud, F.; Doms, R.W.; Pöhlmann, S. The role of DC-SIGN and DC-SIGNR in HIV and Ebola virus infection: Can potential therapeutics block virus transmission and dissemination? Expert Opin. Ther. Targets 2002, 6, 423–431. [Google Scholar] [CrossRef]
- Rodríguez-Pérez, L.; Ramos-Soriano, J.; Pérez-Sánchez, A.; Illescas, B.M.; Muñoz, A.; Luczkowiak, J.; LaSala, F.; Rojo, J.; Delgado, R.; Martín, N. Nanocarbon-Based Glycoconjugates as Multivalent Inhibitors of Ebola Virus Infection. J. Am. Chem. Soc. 2018, 140, 9891–9898. [Google Scholar] [CrossRef]
- Compostella, F.; Pitirollo, O.; Silvestri, A.; Polito, L. Glyco-gold nanoparticles: Synthesis and applications. Beilstein J. Org. Chem. 2017, 13, 1008–1021. [Google Scholar] [CrossRef] [PubMed]
- Budhadev, D.; Poole, E.; Nehlmeier, I.; Liu, Y.; Hooper, J.; Kalverda, E.; Akshath, U.S.; Hondow, N.; Turnbull, W.B.; Pöhlmann, S.; et al. Glycan-Gold Nanoparticles as Multifunctional Probes for Multivalent Lectin–Carbohydrate Binding: Implications for Blocking Virus Infection and Nanoparticle Assembly. J. Am. Chem. Soc. 2020, 142, 18022–18034. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losada-Garcia, N.; Garcia-Sanz, C.; Andreu, A.; Velasco-Torrijos, T.; Palomo, J.M. Glyconanomaterials for Human Virus Detection and Inhibition. Nanomaterials 2021, 11, 1684. https://doi.org/10.3390/nano11071684
Losada-Garcia N, Garcia-Sanz C, Andreu A, Velasco-Torrijos T, Palomo JM. Glyconanomaterials for Human Virus Detection and Inhibition. Nanomaterials. 2021; 11(7):1684. https://doi.org/10.3390/nano11071684
Chicago/Turabian StyleLosada-Garcia, Noelia, Carla Garcia-Sanz, Alicia Andreu, Trinidad Velasco-Torrijos, and Jose M. Palomo. 2021. "Glyconanomaterials for Human Virus Detection and Inhibition" Nanomaterials 11, no. 7: 1684. https://doi.org/10.3390/nano11071684
APA StyleLosada-Garcia, N., Garcia-Sanz, C., Andreu, A., Velasco-Torrijos, T., & Palomo, J. M. (2021). Glyconanomaterials for Human Virus Detection and Inhibition. Nanomaterials, 11(7), 1684. https://doi.org/10.3390/nano11071684






