Mesoporous Iron(III)-Doped Hydroxyapatite Nanopowders Obtained via Iron Oxalate
Abstract
1. Introduction
2. Materials and Methods
2.1. Powders Synthesis
2.2. Powders Characterizations
2.3. DFT Calculations
2.4. Cytocompatibility Test
3. Results
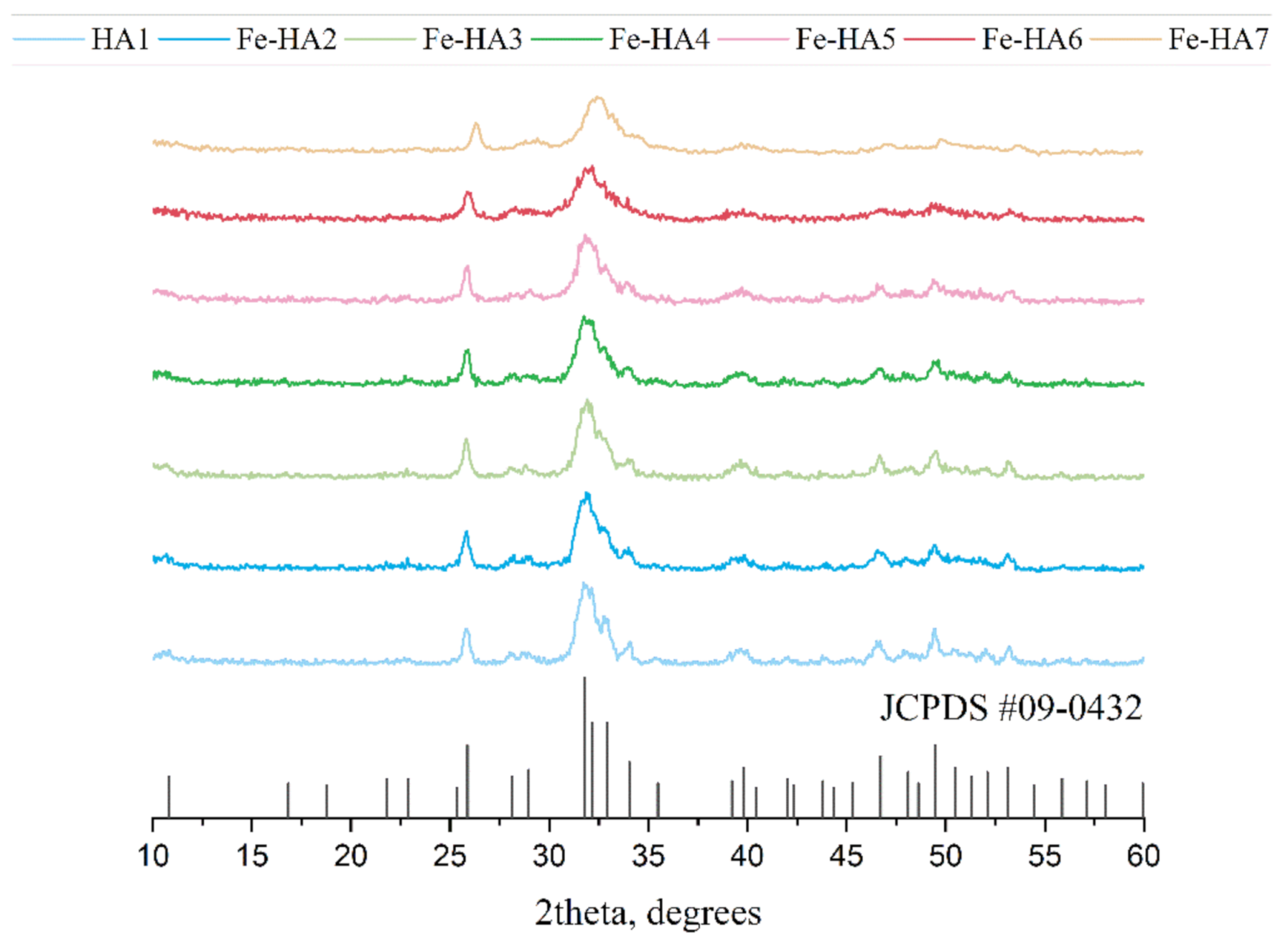
3.1. Powders Chemical and Phase Composition
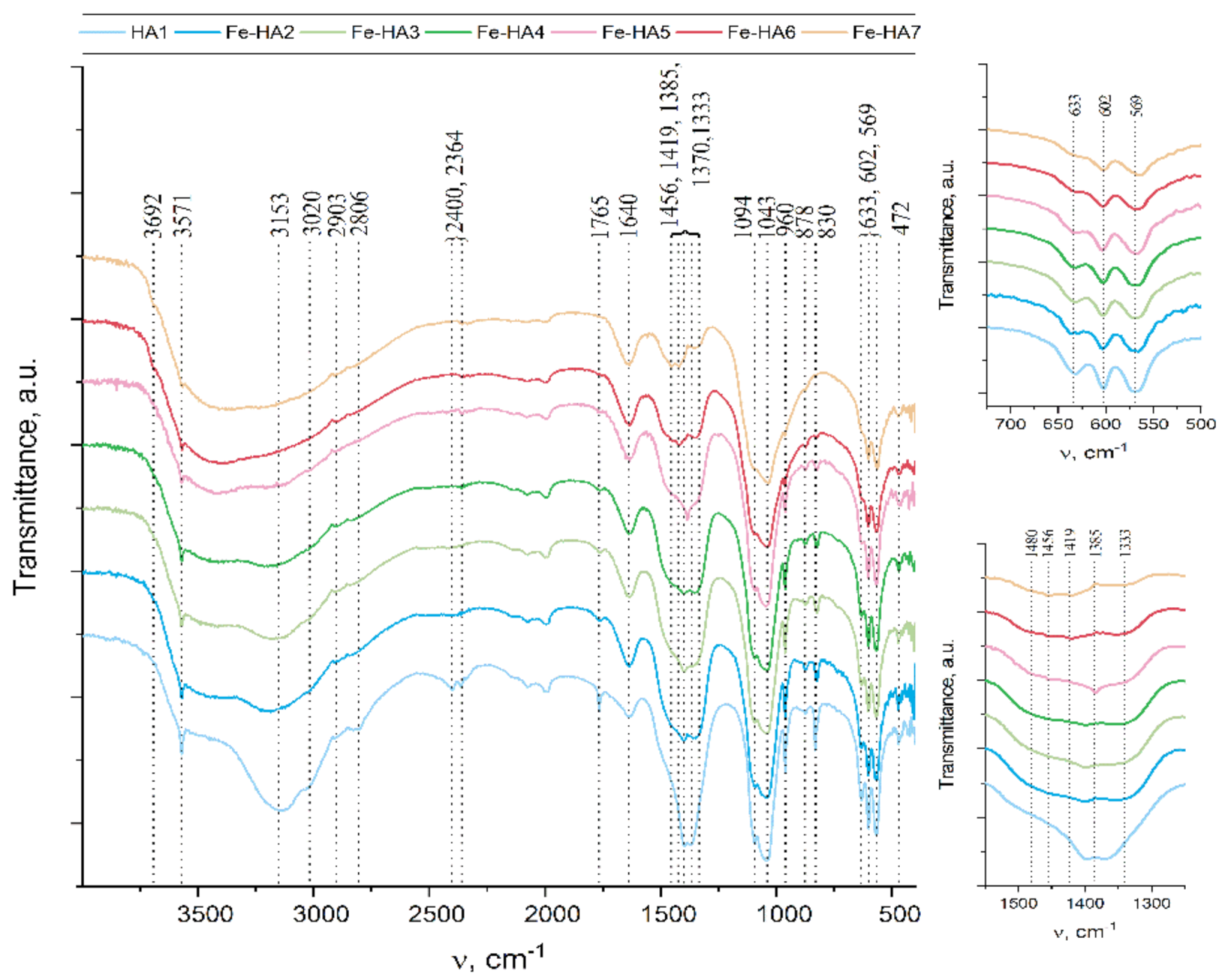
3.2. FTIR Investigation
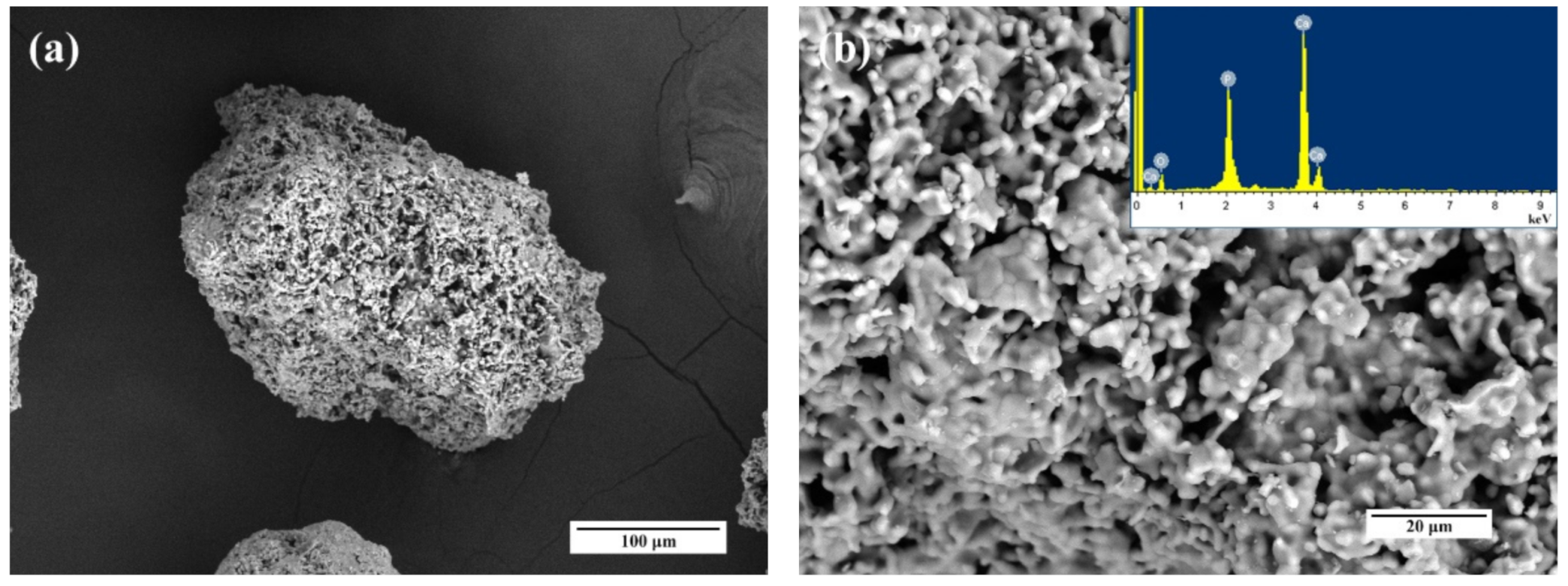
3.3. Microscopy Investigations
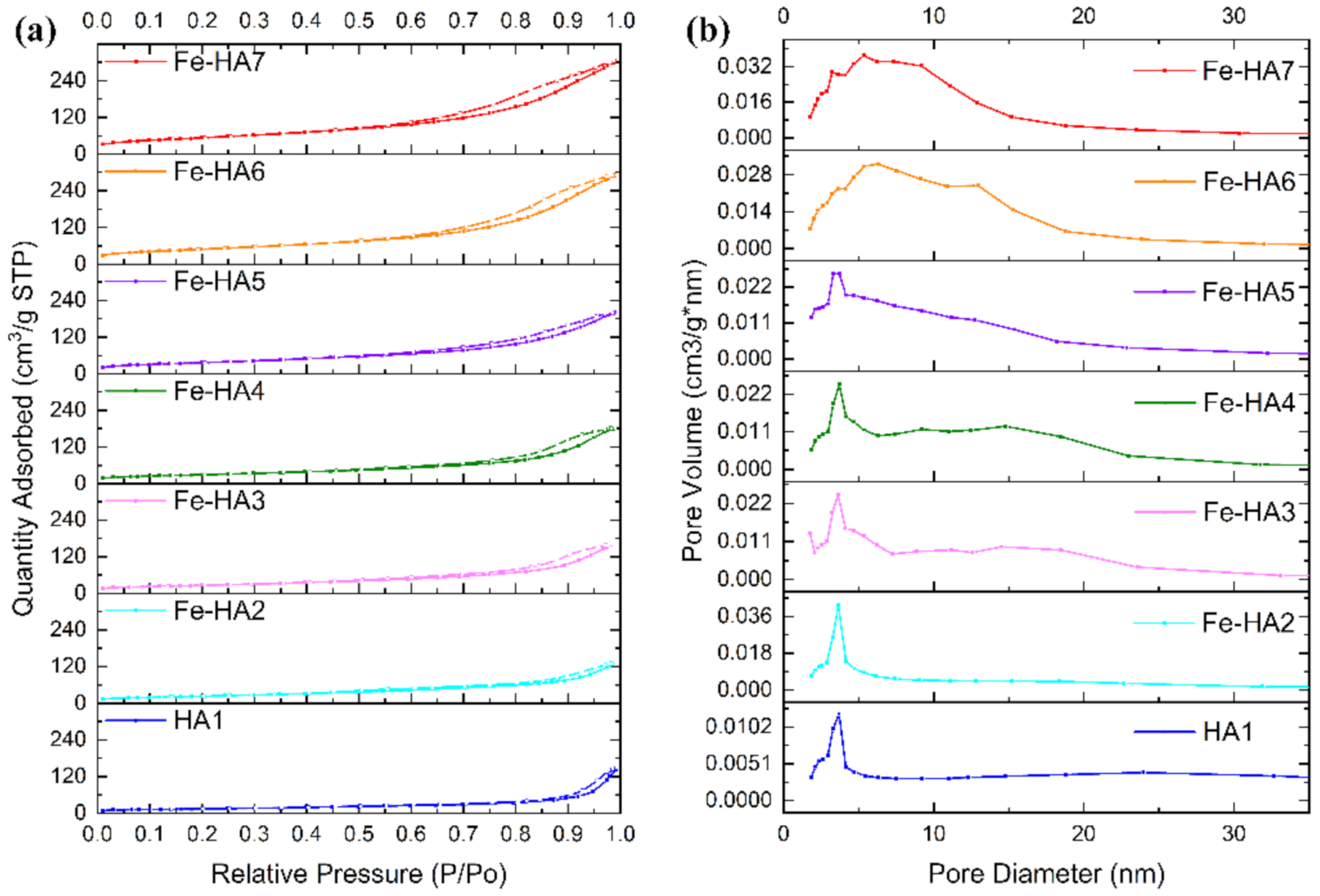
3.4. BET, Pore-Volume, and Adsorption Average Pore Width and Diameter for the Synthesized Samples
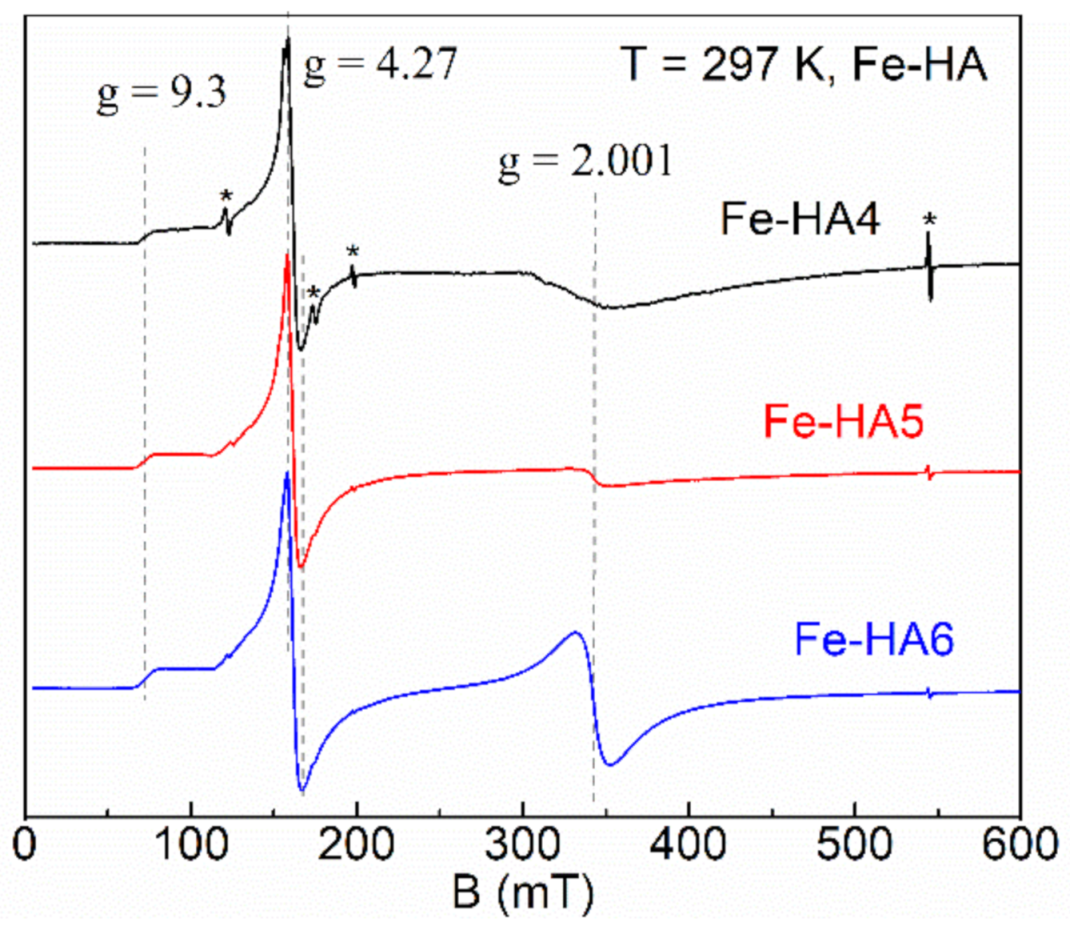
3.5. Electron Paramagnetic Resonance Spectroscopy
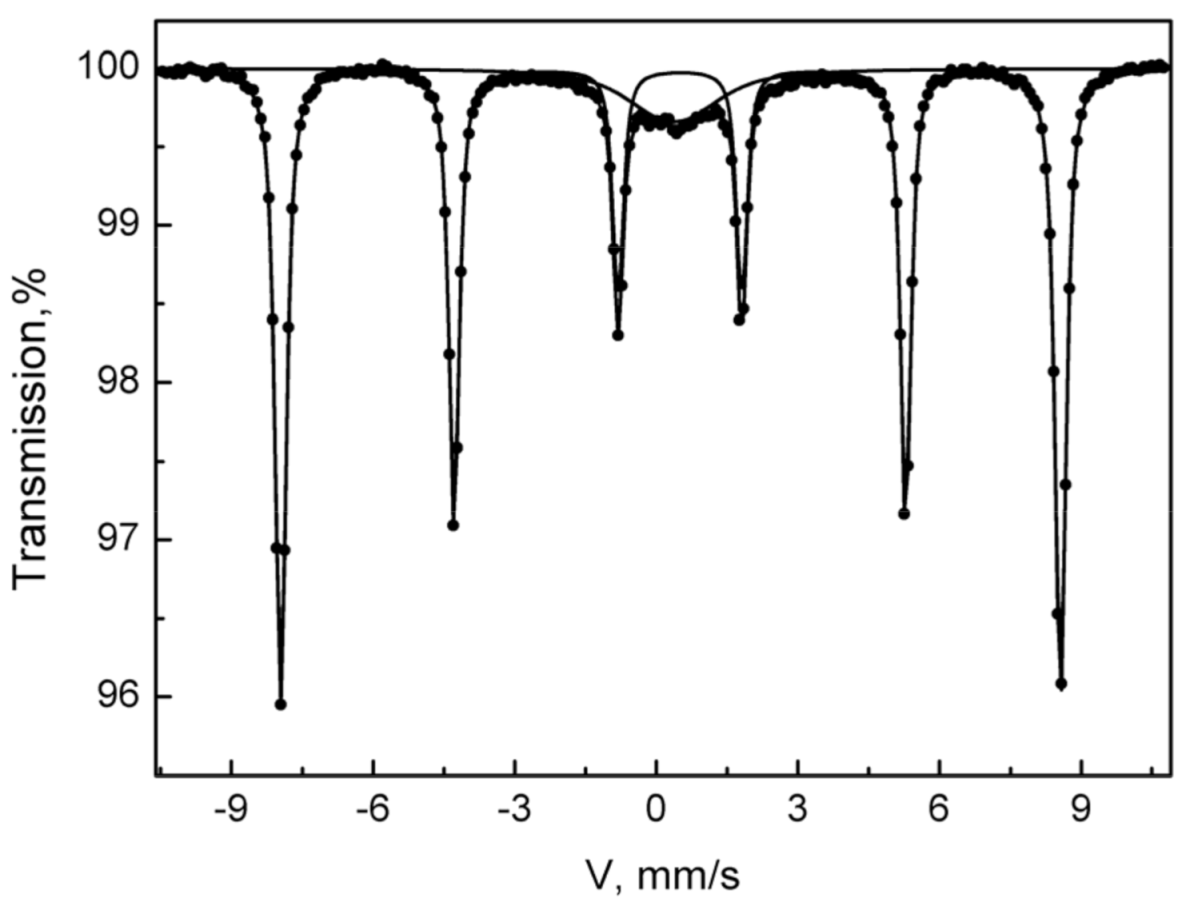
3.6. Mössbauer Spectroscopy
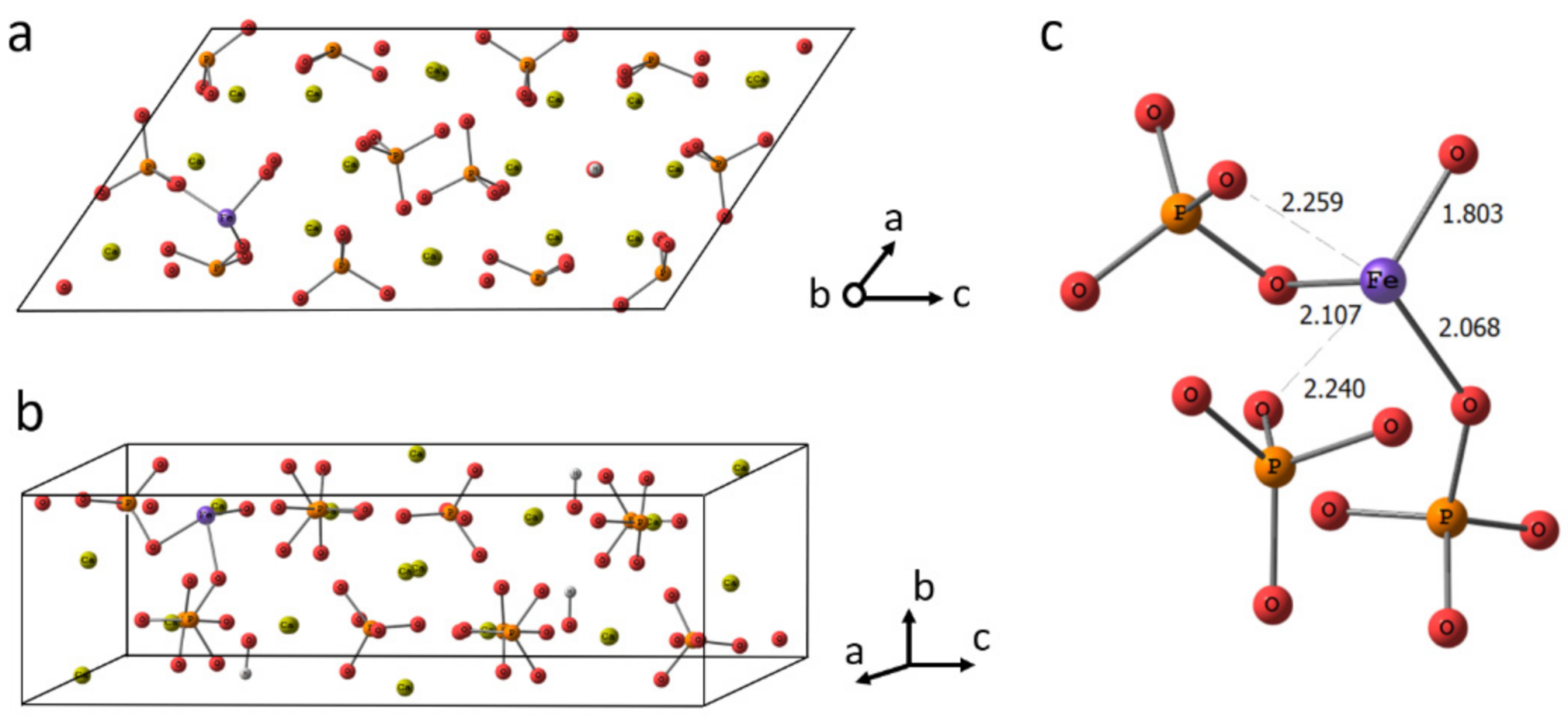
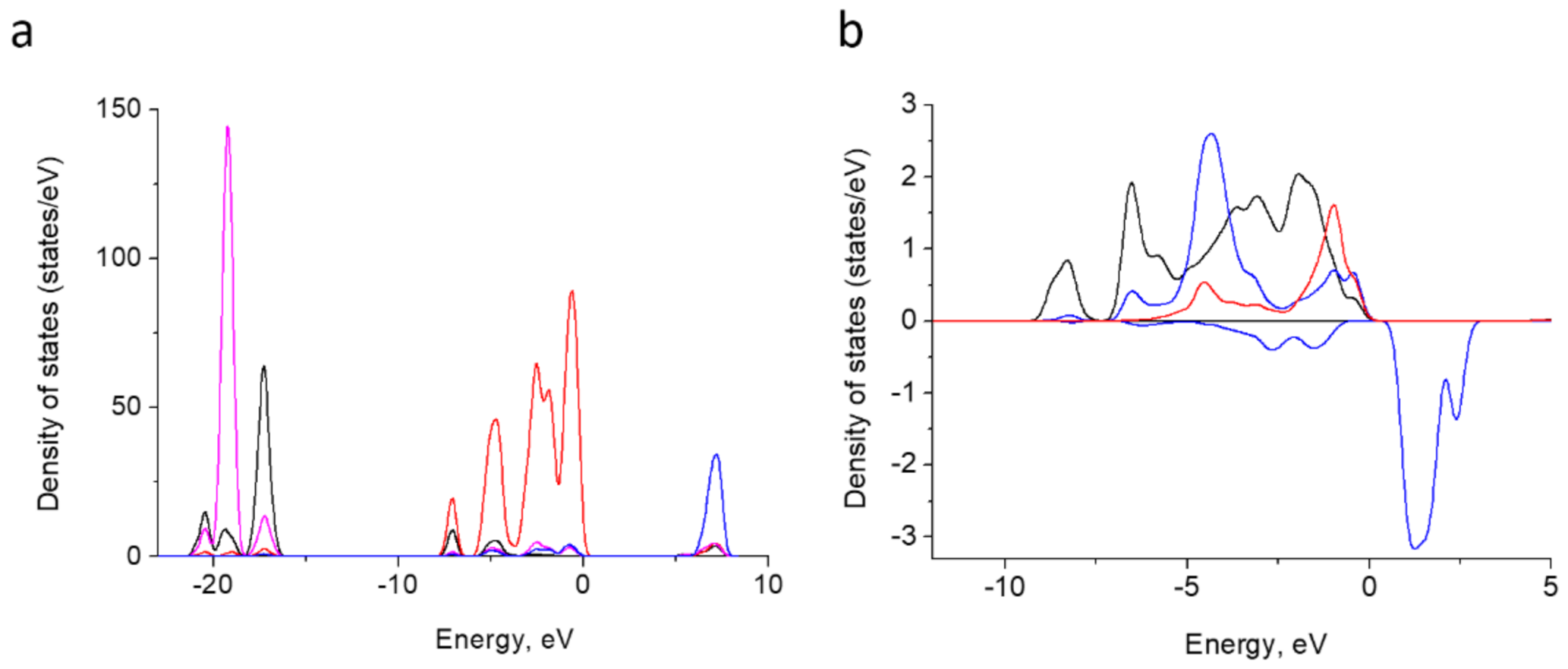
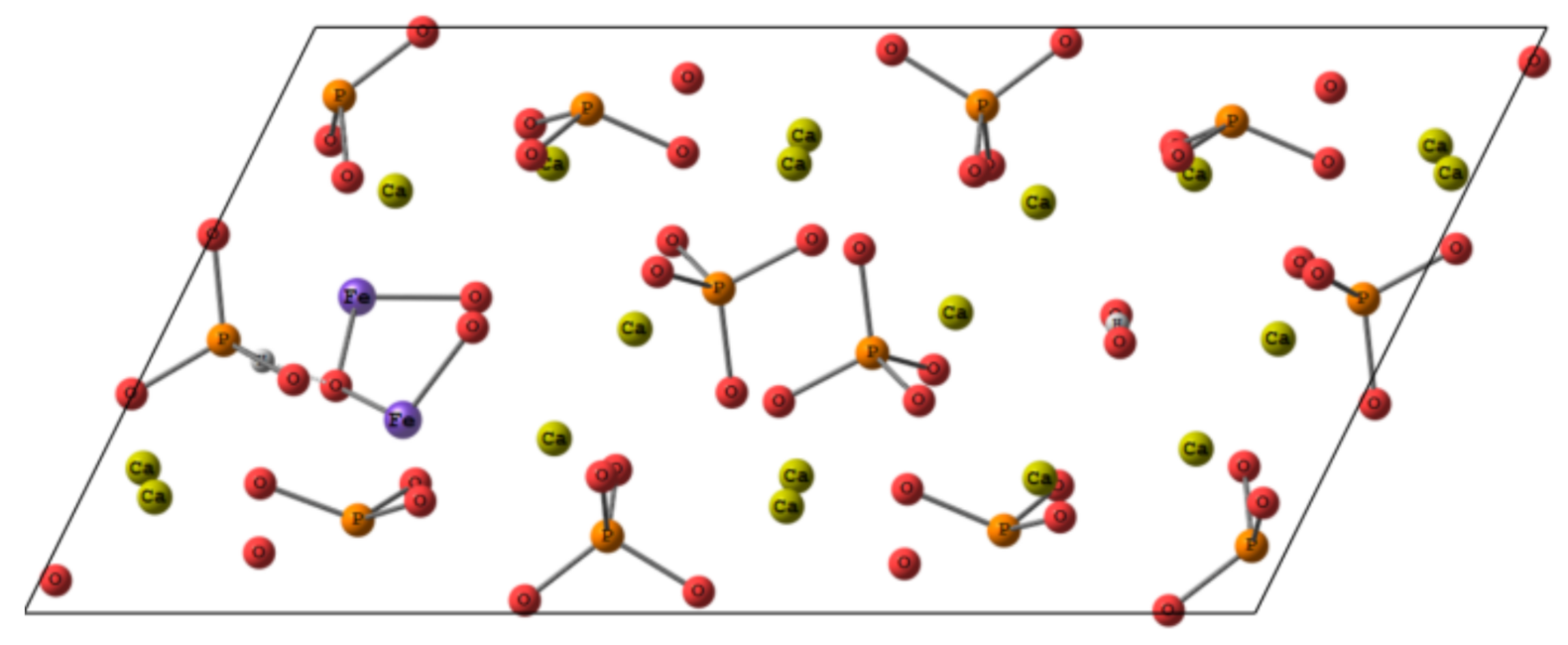
3.7. DFT Calculations
3.7.1. Geometry
3.7.2. Electron Structure
3.8. In Vitro Investigations
3.8.1. Granules Characterization
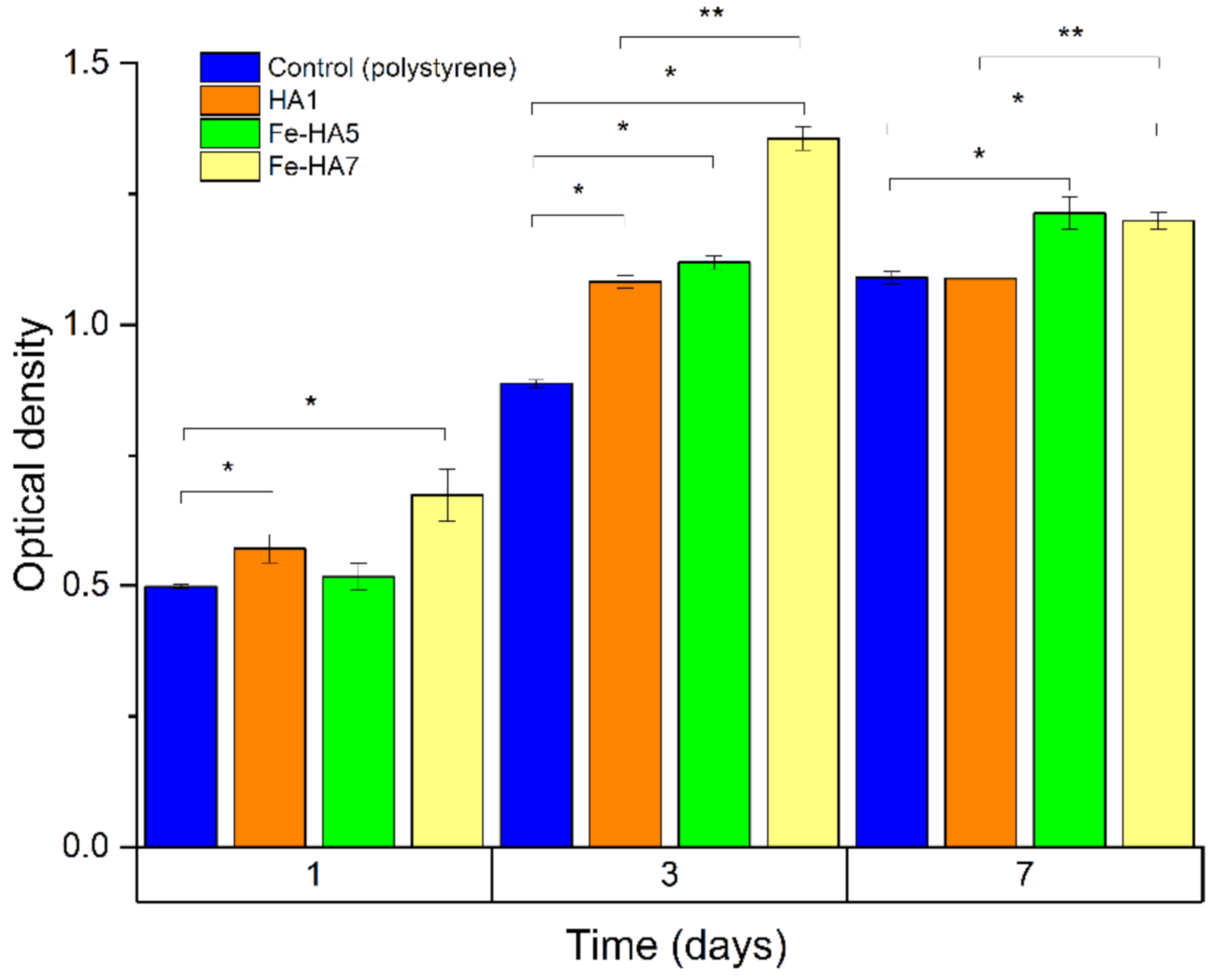
3.8.2. Cytocompatibility of Iron(III)-Doped HA
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Molino, G.; Palmieri, M.C.; Montalbano, G.; Fiorilli, S.; Vitale-Brovarone, C. Biomimetic and Mesoporous Nano-Hydroxyapatite for Bone Tissue Application: A Short Review. Biomed. Mater. 2020, 15. [Google Scholar] [CrossRef] [PubMed]
- Shamray, V.F.; Sirotinkin, V.P.; Smirnov, I.V.; Kalita, V.I.; Fedotov, A.Y.; Barinov, S.M.; Komlev, V.S. Structure of the Hydroxyapatite Plasma-Sprayed Coatings Deposited on Pre-Heated Titanium Substrates. Ceram. Int. 2017, 43, 9105–9109. [Google Scholar] [CrossRef]
- Ibrahim, M.; Labaki, M.; Giraudon, J.M.; Lamonier, J.F. Hydroxyapatite, a Multifunctional Material for Air, Water and Soil Pollution Control: A Review. J. Hazard. Mater. 2020, 383. [Google Scholar] [CrossRef] [PubMed]
- Nanocrystalline Hydroxyapatite: A Potent Material for Adsorption, Biological and Catalytic Studies|Elsevier Enhanced Reader. Available online: https://reader.elsevier.com/reader/sd/pii/S2214785318327500?token=F0F628C1B78F526EF2D7F9E209509D7AF8CD31A8DAB52EDC2DB28DD87FC1CD95D853B1EFC03D183CA897A2251704D507 (accessed on 15 February 2021).
- Zhou, H.; Yang, Y.; Yang, M.; Wang, W.; Bi, Y. Synthesis of Mesoporous Hydroxyapatite via a Vitamin C Templating Hydrothermal Route. Mater. Lett. 2018, 218, 52–55. [Google Scholar] [CrossRef]
- Kumar, G.S.; Karunakaran, G.; Girija, E.K.; Kolesnikov, E.; van Minh, N.; Gorshenkov, M.V.; Kuznetsov, D. Size and Morphology-Controlled Synthesis of Mesoporous Hydroxyapatite Nanocrystals by Microwave-Assisted Hydrothermal Method. Ceram. Int. 2018, 44, 11257–11264. [Google Scholar] [CrossRef]
- Liu, D.-M.; Troczynski, T.; Tseng, W.J. Water-Based Sol-gel Synthesis of Hydroxyapatite: Process Development. Biomaterials 2001, 22, 1721–1730. [Google Scholar] [CrossRef]
- da Silva, O.G.; Alves, M.M.; dos Santos, I.M.G.; Fonseca, M.G.; Jaber, M. Mesoporous Calcium Phosphate Using Casein as a Template: Application to Bovine Serum Albumin Sorption. Colloids Surf. B Biointerfaces 2017, 158, 480–487. [Google Scholar] [CrossRef]
- Javadinejad, H.R.; Saboktakin Rizi, M.; Aghababaei Mobarakeh, E.; Ebrahimian, M. Thermal Stability of Nano-Hydroxyapatite Synthesized via Mechanochemical Treatment. Arab. J. Sci. Eng. 2017, 42, 4401–4408. [Google Scholar] [CrossRef]
- Bakhtiari, L.; Javadpour, J.; Rezaie, H.R.; Erfan, M.; Mazinani, B.; Aminian, A. Pore Size Control in the Synthesis of Hydroxyapatite Nanoparticles: The Effect of Pore Expander Content and the Synthesis Temperature. Ceram. Int. 2016, 42, 11259–11264. [Google Scholar] [CrossRef]
- Mohammad, N.F.; Othman, R.; YEOH, F.Y. Controlling the Pore Characteristics of Mesoporous Apatite Materials: Hydroxyapatite and Carbonate Apatite. Ceram. Int. 2015, 41, 10624–10633. [Google Scholar] [CrossRef]
- Lett, J.A.; Sagadevan, S.; Prabhakar, J.J.; Hamizi, N.A.; Badruddin, I.A.; Johan, M.R.; Marlinda, A.R.; Wahab, Y.A.; Khan, T.M.Y.; Kamangar, S. Drug Leaching Properties of Vancomycin Loaded Mesoporous Hydroxyapatite as Bone Substitutes. Processes 2019, 7, 826. [Google Scholar] [CrossRef]
- Structural and Surface Morphological Change in Incorporation of Magnesium on Synthesised Nano Hydroxyapaptite. J. Environ. Nanotechnol. 2017, 6, 55–58. [CrossRef]
- Lee, I.H.; Lee, J.A.; Lee, J.H.; Heo, Y.W.; Kim, J.J. Effects of PH and Reaction Temperature on Hydroxyapatite Powders Synthesized by Precipitation. J. Korean Ceram. Soc. 2020, 57, 56–64. [Google Scholar] [CrossRef]
- Monmaturapoj, N. Nano-Size Hydroxyapatite Powders Preparation by Wet-Chemical Precipitation Route. J. Met. Mater. Miner. 2008, 18, 1. [Google Scholar]
- Garbo, C.; Sindilaru, M.; Carlea, A.; Tomoaia, G.; Almasan, V.; Petean, I.; Mocanu, A.; Horovitz, O.; Tomoaia-Cotisel, M. Synthesis and Structural Characterization of Novel Porous Zinc Substituted Nanohydroxyapatite Powders. Part. Sci. Technol. 2017, 35, 29–37. [Google Scholar] [CrossRef]
- Anwar, A.; Akbar, S. Novel Continuous Microwave Assisted Flow Synthesis of Nanosized Manganese Substituted Hydroxyapatite. Ceram. Int. 2018, 44, 10878–10882. [Google Scholar] [CrossRef]
- Rahavi, S.S.; Ghaderi, O.; Monshi, A.; Fathi, M.H. A Comparative Study on Physicochemical Properties of Hydroxyapatite Powders Derived from Natural and Synthetic Sources. Russ. J. Non-Ferr. Met. 2017, 58, 276–286. [Google Scholar] [CrossRef]
- Gol’Dberg, M.A.; Smirnov, V.V.; Ievlev, V.M.; Barinov, S.M.; Kutsev, S.V.; Shibaeva, T.v.; Shvorneva, L.I. Influence of Ripening Time on the Properties of Hydroxyapatite-Calcium Carbonate Powders. Inorg. Mater. 2012, 48, 181–186. [Google Scholar] [CrossRef]
- Goldberg, M.A.; Protsenko, P.V.; Smirnov, V.V.; Antonova, O.S.; Smirnov, S.V.; Konovalov, A.A.; Vorckachev, K.G.; Kudryavtsev, E.A.; Barinov, S.M.; Komlev, V.S. The Enhancement of Hydroxyapatite Thermal Stability by Al Doping. J. Mater. Res. Technol. 2020, 9, 76–88. [Google Scholar] [CrossRef]
- Goldberg, M.; Gafurov, M.; Makshakova, O.; Smirnov, V.; Komlev, V.; Barinov, S.; Kudryavtsev, E.; Sergeeva, N.; Achmedova, S.; Mamin, G.; et al. Influence of Al on the Structure and in Vitro Behavior of Hydroxyapatite Nanopowders. J. Phys. Chem. B 2019, 123. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Z.; Sun, W.; Xia, C. A Magnetically Recyclable Heterogeneous Catalyst: Cobalt Nano-Oxide Supported on Hydroxyapatite-Encapsulated γ-Fe2O3 Nanocrystallites for Highly Efficient Olefin Oxidation with H2O2. Catal. Commun. 2008, 10, 237–242. [Google Scholar] [CrossRef]
- Aliev, F.A.; Mukhamatdinov, I.I.; Sitnov, S.A.; Ziganshina, M.R.; Onishchenko, Y.V.; Sharifullin, A.V.; Vakhin, A.V. In-Situ Heavy Oil Aquathermolysis in the Presence of Nanodispersed Catalysts Based on Transition Metals. Processes 2021, 9, 127. [Google Scholar] [CrossRef]
- Lakhova, A.; Petrov, S.; Ibragimova, D.; Kayukova, G.; Safiulina, A.; Shinkarev, A.; Okekwe, R. Aquathermolysis of Heavy Oil Using Nano Oxides of Metals. J. Pet. Sci. Eng. 2017, 153, 385–390. [Google Scholar] [CrossRef]
- Sarath Chandra, V.; Baskar, G.; Suganthi, R.V.; Elayaraja, K.; Ahymah Joshy, M.I.; Sofi Beaula, W.; Mythili, R.; Venkatraman, G.; Narayana Kalkura, S. Blood Compatibility of Iron-Doped Nanosize Hydroxyapatite and Its Drug Release. Acs Appl. Mater. Interfaces 2012, 4, 1200–1210. [Google Scholar] [CrossRef]
- Panseri, S.; Cunha, C.; D’Alessandro, T.; Sandri, M.; Giavaresi, G.; Marcacci, M.; Hung, C.T.; Tampieri, A. Intrinsically Superparamagnetic Fe-Hydroxyapatite Nanoparticles Positively Influence Osteoblast-like Cell Behaviour. J. Nanobiotechnol. 2012, 10, 32. [Google Scholar] [CrossRef]
- Ribeiro, T.P.; Monteiro, F.J.; Laranjeira, M.S. Duality of Iron (III) Doped Nano Hydroxyapatite in Triple Negative Breast Cancer Monitoring and as a Drug-Free Therapeutic Agent. Ceram. Int. 2020, 46, 16590–16597. [Google Scholar] [CrossRef]
- Laranjeira, M.S.; Moço, A.; Ferreira, J.; Coimbra, S.; Costa, E.; Santos-Silva, A.; Ferreira, P.J.; Monteiro, F.J. Different Hydroxyapatite Magnetic Nanoparticles for Medical Imaging: Its Effects on Hemostatic, Hemolytic Activity and Cellular Cytotoxicity. Colloids Surf. B Biointerfaces 2016, 146, 363–374. [Google Scholar] [CrossRef]
- Tampieri, A.; D’Alessandro, T.; Sandri, M.; Sprio, S.; Landi, E.; Bertinetti, L.; Panseri, S.; Pepponi, G.; Goettlicher, J.; Bañobre-López, M.; et al. Intrinsic Magnetism and Hyperthermia in Bioactive Fe-Doped Hydroxyapatite. Acta Biomater. 2012, 8, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, S.; Padmanabhan, V.P.; Kulandaivelu, R.; Sankara Narayanan Nellaiappan, T.S.; Sagadevan, S.; Paiman, S.; Mohammad, F.; Al-Lohedan, H.A.; Obulapuram, P.K.; Oh, W.C. Influence of Iron Doping towards the Physicochemical and Biological Characteristics of Hydroxyapatite. Ceram. Int. 2021, 47, 5061–5070. [Google Scholar] [CrossRef]
- Gu, L.; He, X.; Wu, Z. Mesoporous Fe3O4/Hydroxyapatite Composite for Targeted Drug Delivery. Mater. Res. Bull. 2014, 59, 65–68. [Google Scholar] [CrossRef]
- Abbasi Aval, N.; Pirayesh Islamian, J.; Hatamian, M.; Arabfirouzjaei, M.; Javadpour, J.; Rashidi, M.R. Doxorubicin Loaded Large-Pore Mesoporous Hydroxyapatite Coated Superparamagnetic Fe3O4 Nanoparticles for Cancer Treatment. Int. J. Pharm. 2016, 509, 159–167. [Google Scholar] [CrossRef]
- Shelekhov, E.V.; Sviridova, T.A. Programs for X-ray Analysis of Polycrystals; Springer: Berlin/Heidelberg, Germany, 2000; Volume 42. [Google Scholar]
- Yashima, M.; Yonehara, Y.; Fujimori, H. Experimental Visualization of Chemical Bonding and Structural Disorder in Hydroxyapatite through Charge and Nuclear-Density Analysis. J. Phys. Chem. C 2011, 115, 25077–25087. [Google Scholar] [CrossRef]
- Biktagirov, T.; Gafurov, M.; Mamin, G.; Klimashina, E.; Putlayev, V.; Orlinskii, S. Combination of EPR Measurements and DFT Calculations to Study Nitrate Impurities in the Carbonated Nanohydroxyapatite. J. Phys. Chem. A 2014, 118, 1519–1526. [Google Scholar] [CrossRef]
- Vanderbilt, D. Rapid Communications Soft Self-Consistent Pseudopotentials in a Generalized Eigenvalue Formalism. Phys. Rev. B 1990, 41, 7892. [Google Scholar] [CrossRef]
- Giannozzi, P.; Baroni, S.; Bonini, N.; Calandra, M.; Car, R.; Cavazzoni, C.; Ceresoli, D.; Chiarotti, G.L.; Cococcioni, M.; Dabo, I.; et al. QUANTUM ESPRESSO: A Modular and Open-Source Software Project for Quantum Simulations of Materials. J. Phys. Condens. Matter 2009, 21. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865. [Google Scholar] [CrossRef]
- Monkhorst, H.J.; Pack, J.D. Special Points for Brillonin-Zone Integrations. Phys. Rev. B 1976, 13, 5188–5192. [Google Scholar] [CrossRef]
- Troullier, N.; Martins, J.L. Efficient Pseudopotentials for Plane-Wave Calculations. Phys. Rev. B 1991, 43, 1993–2006. [Google Scholar] [CrossRef]
- Jiang, M.; Terra, J.; Rossi, A.M.; Morales, M.A.; Baggio Saitovitch, E.M.; Ellis, D.E. (Formula Presented) Substitution in Hydroxyapatite: Theory and Experiment. Phys. Rev. B Condens. Matter Mater. Phys. 2002, 66, 1–15. [Google Scholar] [CrossRef]
- Komlev, V.S.; Barinov, S.M.; Koplik, E.V. A Method to Fabricate Porous Spherical Hydroxyapatite Granules Intended for Time-Controlled Drug Release. Biomaterials 2002, 23, 3449–3454. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Kongsri, S.; Janpradit, K.; Buapa, K.; Techawongstien, S.; Chanthai, S. Nanocrystalline Hydroxyapatite from Fish Scale Waste: Preparation, Characterization and Application for Selenium Adsorption in Aqueous Solution. Chem. Eng. J. 2013, 215–216, 522–532. [Google Scholar] [CrossRef]
- Li, Y.; Wang, S.; Zhang, Y.; Han, R.; Wei, W. Enhanced Tetracycline Adsorption onto Hydroxyapatite by Fe(III) Incorporation. J. Mol. Liq. 2017, 247, 171–181. [Google Scholar] [CrossRef]
- Duta, L.; Oktar, F.N.; Stan, G.E.; Popescu-Pelin, G.; Serban, N.; Luculescu, C.; Mihailescu, I.N. Novel Doped Hydroxyapatite Thin Films Obtained by Pulsed Laser Deposition. Appl. Surf. Sci. 2013, 265, 41–49. [Google Scholar] [CrossRef]
- Mahene, W.L.; Gervas, C.; Hilonga, A.H.; Machunda, R.L. Synthesis and FTIR Characterization of Mg-Hydroxylapatite Derived from Dolostone with High Dolomite Mineral Content. Tanzan. J. Sci. 2020, 46, 661–672. [Google Scholar]
- Anee Kuriakose, T.; Kalkura, S.N.; Palanichamy, M.; Arivuoli, D.; Dierks, K.; Bocelli, G.; Betzel, C. Synthesis of Stoichiometric Nano Crystalline Hydroxyapatite by Ethanol-Based Sol-Gel Technique at Low Temperature. J. Cryst. Growth 2004, 263, 517–523. [Google Scholar] [CrossRef]
- Théorêt, A.; Sandorfy, C. Infrared Spectra and Crystalline Phase Transitions of Ammonium Nitrate. Can. J. Chem. 1964, 42, 57–62. [Google Scholar] [CrossRef]
- Landi, E.; Tampieri, A.; Celotti, G.; Vichi, L.; Sandri, M. Influence of Synthesis and Sintering Parameters on the Characteristics of Carbonate Apatite. Biomaterials 2004, 25, 1763–1770. [Google Scholar] [CrossRef] [PubMed]
- Deymier, A.C.; Nair, A.K.; Depalle, B.; Qin, Z.; Arcot, K.; Drouet, C.; Yoder, C.H.; Buehler, M.J.; Thomopoulos, S.; Genin, G.M.; et al. Protein-Free Formation of Bone-like Apatite: New Insights into the Key Role of Carbonation. Biomaterials 2017, 127, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Madupalli, H.; Pavan, B.; Tecklenburg, M.M.J. Carbonate Substitution in the Mineral Component of Bone: Discriminating the Structural Changes, Simultaneously Imposed by Carbonate in A and B Sites of Apatite. J. Solid State Chem. 2017, 255, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Elliott, J.C. Structure and Chemistry of the Apatites and Other Calcium Orthophosphates. Available online: https://books.google.ru/books?hl=ru&lr=&id=dksXBQAAQBAJ&oi=fnd&pg=PP1&dq=Elliott+J.+C.+Structure+and+chemistry+of+the+apatites+and+other+calcium+orthophosphates.+%E2%80%93+Elsevier,+2013&ots=xqOING4Oz0&sig=4h8qj5miONBTGVGxOrmM4AXhM9U&redir_esc=y#v=onepage&q=Elliott%20J.%20C.%20Structure%20and%20chemistry%20of%20the%20apatites%20and%20other%20calcium%20orthophosphates.%20%E2%80%93%20Elsevier%2C%202013&f=false (accessed on 26 February 2021).
- Mercado, D.F.; Magnacca, G.; Malandrino, M.; Rubert, A.; Montoneri, E.; Celi, L.; Bianco Prevot, A.; Gonzalez, M.C. Paramagnetic Iron-Doped Hydroxyapatite Nanoparticles with Improved Metal Sorption Properties. A Bioorganic Substrates-Mediated Synthesis. Acs Appl. Mater. Interfaces 2014, 6, 3937–3946. [Google Scholar] [CrossRef]
- Singh, R.P.; Singh, J.P.; Singh, C.; Kaur, T.; Pal, A. Synthesis, Characterization and in-Vitro Bioactivity Evaluation of Mesoporous Ca10-XFex(PO4)6(OH)2 Nanorods-like Particles. Ceram. Int. 2020, 46, 12156–12164. [Google Scholar] [CrossRef]
- Guo, Y.P.; Yao, Y.B.; Guo, Y.J.; Ning, C.Q. Hydrothermal Fabrication of Mesoporous Carbonated Hydroxyapatite Microspheres for a Drug Delivery System. Microporous Mesoporous Mater. 2012, 155, 245–251. [Google Scholar] [CrossRef]
- Stafford, K.; Sing, W.; Sing, K.S.W. Reporting Physisorption Data for Gas/Solid Systems with Special Reference to the Determination of Surface Area and Porosity international union of pure and applied chemistry physical chemistry division commission on colloid and surface chemistry including catalysis subcommittee on reporting gas adsorption data* reporting physisorption data for gas/solid systems with Special Reference to the Determination of Surface Area and Porosity. Pure Appl. Chem. 1982, 54, 2201–2218. [Google Scholar] [CrossRef]
- Di Chen, J.; Wang, Y.J.; Wei, K.; Zhang, S.H.; Shi, X.T. Self-Organization of Hydroxyapatite Nanorods through Oriented Attachment. Biomaterials 2007, 28, 2275–2280. [Google Scholar] [CrossRef]
- Zhao, Y.F.; Ma, J. Triblock Co-Polymer Templating Synthesis of Mesostructured Hydroxyapatite. Microporous Mesoporous Mater. 2005, 87, 110–117. [Google Scholar] [CrossRef]
- Gabbasov, B.; Gafurov, M.; Starshova, A.; Shurtakova, D.; Murzakhanov, F.; Mamin, G.; Orlinskii, S. Conventional, Pulsed and High-Field Electron Paramagnetic Resonance for Studying Metal Impurities in Calcium Phosphates of Biogenic and Synthetic Origins. J. Magn. Magn. Mater. 2019, 470, 109–117. [Google Scholar] [CrossRef]
- Murzakhanov, F.; Gabbasov, B.; Iskhakova, K.; Voloshin, A.; Mamin, G.; Putlyaev, V.; Klimashina, E.; Fadeeva, I.; Fomin, A.; Barinov, S.; et al. Conventional Electron Paramagnetic Resonance for Studying Synthetic Calcium Phosphates with Metal Impurities (Mn2+, Cu2+, Fe3+). Magn. Reson. Solids 2017, 19, 1–10. [Google Scholar] [CrossRef]
- Abragam, A.; Bleaney, B. Electron Paramagnetic Resonance of Transition Ions. Available online: https://scholar.google.ru/scholar?cluster=11839537261188624540&hl=ru&as_sdt=2005&sciodt=0,5 (accessed on 26 February 2021).
- Sutter, B.; Wasowicz, T.; Howard, T.; Hossner, L.R.; Ming, D.W. Characterization of Iron, Manganese, and Copper Synthetic Hydroxyapatites by Electron Paramagnetic Resonance Spectroscopy. Soil Sci. Soc. Am. J. 2002, 66, 1359–1366. [Google Scholar] [CrossRef]
- Bou-Abdallah, F.; Chasteen, N.D. Spin Concentration Measurements of High-Spin (G′ = 4.3) Rhombic Iron(III) Ions in Biological Samples: Theory and Application. J. Biol. Inorg. Chem. 2008, 13, 15–24. [Google Scholar] [CrossRef]
- Singh, S.P.; Chakradhar, R.P.S.; Rao, J.L.; Karmakar, B. EPR, FTIR, Optical Absorption and Photoluminescence Studies of Fe2O3 and CeO2 Doped ZnO-Bi2O3-B2O3 Glasses. J. Alloys Compd. 2010, 493, 256–262. [Google Scholar] [CrossRef]
- Cnockaert, V.; Maes, K.; Bellemans, I.; Crivits, T.; Vrielinck, H.; Blanpain, B.; Verbeken, K. Quantification of the Fe3+ Concentration in Lead Silicate Glasses Using X-Band CW-EPR. J. Non-Cryst. Solids 2020, 536. [Google Scholar] [CrossRef]
- Herring, M.P.; Khachatryan, L.; Dellinger, B. Speciation of Iron (III) Oxide Nanoparticles and Other Paramagnetic Intermediates during High-Temperature Oxidative Pyrolysis of 1-Methylnaphthalene. World Acad. Sci. Eng. Technol. 2015, 9, 804–812. [Google Scholar]
- Domracheva, N.E.; Pyataev, A.V.; Manapov, R.A.; Gruzdev, M.S. Magnetic Resonance and Mössbauer Studies of Superparamagnetic γ-Fe2O3 Nanoparticles Encapsulated into Liquid-Crystalline Poly(Propylene Imine) Dendrimers. ChemPhysChem 2011, 12, 3009–3019. [Google Scholar] [CrossRef]
- Abdullin, A.F.; Pyataev, A.V.; Domracheva, N.E.; Gruzdev, M.S. Mössbauer Study of the Surface of Core-Shell Type Nanoparticles. J. Surf. Investig. 2016, 10, 35–38. [Google Scholar] [CrossRef]
- Matsunaga, K. First-Principles Study of Substitutional Magnesium and Zinc in Hydroxyapatite and Octacalcium Phosphate. J. Chem. Phys. 2008, 128. [Google Scholar] [CrossRef]
- Matsunaga, K.; Kuwabara, A. First-Principles Study of Vacancy Formation in Hydroxyapatite. Phys. Rev. B Condens. Matter Mater. Phys. 2007, 75. [Google Scholar] [CrossRef]
- Ferreira-Ermita, D.A.C.; Valente, F.L.; Carlo-Reis, E.C.; Araújo, F.R.; Ribeiro, I.M.; Cintra, C.C.V.; Borges, A.P.B. Characterization and in Vivo Biocompatibility Analysis of Synthetic Hydroxyapatite Compounds Associated with Magnetite Nanoparticles for a Drug Delivery System in Osteomyelitis Treatment. Results Mater. 2020, 5, 100063. [Google Scholar] [CrossRef]
- Tyapkin, P.Y.; Petrov, S.A.; Chernyshev, A.P.; Ancharov, A.I.; Sheludyakova, L.A.; Uvarov, N.F. Structural Features of Hydrate Forms of Iron(III) Oxalate. J. Struct. Chem. 2016, 57, 1134–1140. [Google Scholar] [CrossRef]
- Tyapkin, P.Y.; Petrov, S.A.; Chernyshev, A.P.; Uvarov, N.F. Physicochemical Properties of Highly Dispersed Iron Oxides Formed Inside Mesoporous Silica. Russ. J. Gen. Chem. 2018, 88, 1066–1070. [Google Scholar] [CrossRef]
- Fadeeva, I.V.; Gafurov, M.R.; Kiiaeva, I.A.; Orlinskii, S.B.; Kuznetsova, L.M.; Filippov, Y.Y.; Fomin, A.S.; Davydova, G.A.; Selezneva, I.I.; Barinov, S.M. Tricalcium phosphate ceramics doped with silver, copper, zinc, and iron (III) ions in concentrations of less than 0.5 wt.% for bone tissue regeneration. BioNanoScience 2017, 7, 434–438. [Google Scholar] [CrossRef]
- Adah Nez, J.; Garcmh A-Labiano, F.; Fierro, V. Modelling for the High-Temperature Sulphation of Calcium-Based Sorbents with Cylindrical and Plate-like Pore Geometries. Chem. Eng. Sci. 2000, 55, 3665–3683. [Google Scholar] [CrossRef]
- Li, B.; Zhang, Y.; Zhou, X.; Liu, Z.; Liu, Q.; Li, X. Different Dye Removal Mechanisms between Monodispersed and Uniform Hexagonal Thin Plate-like MgAl-CO32--LDH and Its Calcined Product in Efficient Removal of Congo Red from Water. J. Alloys Compd. 2016, 673, 265–271. [Google Scholar] [CrossRef]
- Kamieniak, J.; Kelly, P.J.; Banks, C.E.; Doyle, A.M. Mechanical, PH and Thermal Stability of Mesoporous Hydroxyapatite. J. Inorg. Organomet. Polym. Mater. 2018, 28, 84–91. [Google Scholar] [CrossRef]
- Ullah, I.; Gloria, A.; Zhang, W.; Ullah, M.W.; Wu, B.; Li, W.; Domingos, M.; Zhang, X. Synthesis and Characterization of Sintered Sr/Fe-Modified Hydroxyapatite Bioceramics for Bone Tissue Engineering Applications. ACS Biomater. Sci. Eng. 2020, 6, 375–388. [Google Scholar] [CrossRef]





| Sample Code | Chemical Formula (Theoretical) | Chemical Formula (Experimental) | Fe Concentration, mol. % |
|---|---|---|---|
| HA1 | Ca10(PO4)6(OH)2 | Ca10(PO4)6(OH)2 | 0 |
| Fe-HA2 | Ca9.9925Fe0.005(PO4)6(OH)2 | Ca9.991Fe0.006(PO4)6(OH)2 | 0.01 |
| Fe-HA3 | Ca9.9625Fe0.025(PO4)6(OH)2 | Ca9.9625Fe0.025(PO4)6(OH)2 | 0.06 |
| Fe-HA4 | Ca9.925Fe0.05(PO4)6(OH)2 | Ca9.925Fe0.05(PO4)6(OH)2 | 0.11 |
| Fe-HA5 | Ca9.625Fe0.25(PO4)6(OH)2 | Ca9.665Fe0.23(PO4)6(OH)2 | 0.52 |
| Fe-HA6 | Ca9.25Fe0.5(PO4)6(OH)2 | Ca9.25Fe0.5(PO4)6(OH)2 | 1.11 |
| Fe-HA7 | Ca8.5Fe1(PO4)6(OH)2 | Ca8.38Fe1.08(PO4)6(OH)2 | 2.42 |
| Sample Code | a, nm | c, nm | c\a | V, nm3 | D, nm |
|---|---|---|---|---|---|
| Standard HA (JCPDC #09-0432) | 0.9418 | 0.6884 | 0.7309 | 0.5287 | - |
| HA1 | 0.9467 (7) | 0.6898 (6) | 0.7286 | 0.5354 | 13.8 (0.3) |
| Fe-HA2 | 0.9436 (12) | 0.6881 (11) | 0.7292 | 0.5306 | 13.4 (0.3) |
| Fe-HA3 | 0.9431 (6) | 0.6881 (8) | 0.7296 | 0.5307 | 13.7 (0.2) |
| Fe-HA4 | 0.9437 (2) | 0.6881 (2) | 0.7291 | 0.5308 | 13.2 (0.4) |
| Fe-HA5 | 0.9438 (2) | 0.6881 (2) | 0.7290 | 0.5308 | 12.8 (0.4) |
| Fe-HA6 | 0.9452 (28) | 0.6875 (5) | 0.7273 | 0.5319 | 10.7 (0.6) |
| Fe-HA7 | 0.9442 (18) | 0.6875(2) | 0.7234 | 0.5343 | 9.9 (0.2) |
| Sample | BET Specific Surface Area, m2/g | Total Pore Volume, cm3/g | BJH Adsorption Average Pore Diameter, nm |
|---|---|---|---|
| HA1 | 52.1 | 0.16 | 16.5 |
| Fe-HA2 | 85.2 | 0.18 | 8.5 |
| Fe-HA3 | 95.9 | 0.22 | 9.5 |
| Fe-HA4 | 105.7 | 0.26 | 9.9 |
| Fe-HA5 | 134.2 | 0.28 | 8.5 |
| Fe-HA6 | 178.2 | 0.42 | 8.9 |
| Fe-HA7 | 194.5 | 0.43 | 8.7 |
| Sample | Component | IS ±0.01 mm/s | ΔEQ (QS), ±0.01 mm/s | Hhf, T ± 0.1% | Spar, ±1% |
|---|---|---|---|---|---|
| Fe-HA7 | D1 | 0.37 | 0.7 | - | 13 |
| S1 | 0.37 | −0.2 | 51.3 | 87 |
| Materials | pH Value of Extract, CGM | MTT Test Results | ||
|---|---|---|---|---|
| OD, a.u. M (±m) | PVC, % | TI, % | ||
| HA1 | 7.7 | 0.454 (0.001) | 78.5 | 21.5 |
| HA-Fe5 | 7.4 | 0.502 (0.001) | 95.8 | 4.2 |
| HA-Fe7 | 7.8 | 0.515 (0.001) | 98.3 | 1.7 |
| Control (CGM) | 7.4 | 0.578 (0.001) | 100.0 | 0.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goldberg, M.A.; Gafurov, M.R.; Murzakhanov, F.F.; Fomin, A.S.; Antonova, O.S.; Khairutdinova, D.R.; Pyataev, A.V.; Makshakova, O.N.; Konovalov, A.A.; Leonov, A.V.; et al. Mesoporous Iron(III)-Doped Hydroxyapatite Nanopowders Obtained via Iron Oxalate. Nanomaterials 2021, 11, 811. https://doi.org/10.3390/nano11030811
Goldberg MA, Gafurov MR, Murzakhanov FF, Fomin AS, Antonova OS, Khairutdinova DR, Pyataev AV, Makshakova ON, Konovalov AA, Leonov AV, et al. Mesoporous Iron(III)-Doped Hydroxyapatite Nanopowders Obtained via Iron Oxalate. Nanomaterials. 2021; 11(3):811. https://doi.org/10.3390/nano11030811
Chicago/Turabian StyleGoldberg, Margarita A., Marat R. Gafurov, Fadis F. Murzakhanov, Alexander S. Fomin, Olga S. Antonova, Dinara R. Khairutdinova, Andrew V. Pyataev, Olga N. Makshakova, Anatoliy A. Konovalov, Alexander V. Leonov, and et al. 2021. "Mesoporous Iron(III)-Doped Hydroxyapatite Nanopowders Obtained via Iron Oxalate" Nanomaterials 11, no. 3: 811. https://doi.org/10.3390/nano11030811
APA StyleGoldberg, M. A., Gafurov, M. R., Murzakhanov, F. F., Fomin, A. S., Antonova, O. S., Khairutdinova, D. R., Pyataev, A. V., Makshakova, O. N., Konovalov, A. A., Leonov, A. V., Akhmedova, S. A., Sviridova, I. K., Sergeeva, N. S., Barinov, S. M., & Komlev, V. S. (2021). Mesoporous Iron(III)-Doped Hydroxyapatite Nanopowders Obtained via Iron Oxalate. Nanomaterials, 11(3), 811. https://doi.org/10.3390/nano11030811







