Fetotoxicity of Nanoparticles: Causes and Mechanisms
Abstract
1. Introduction
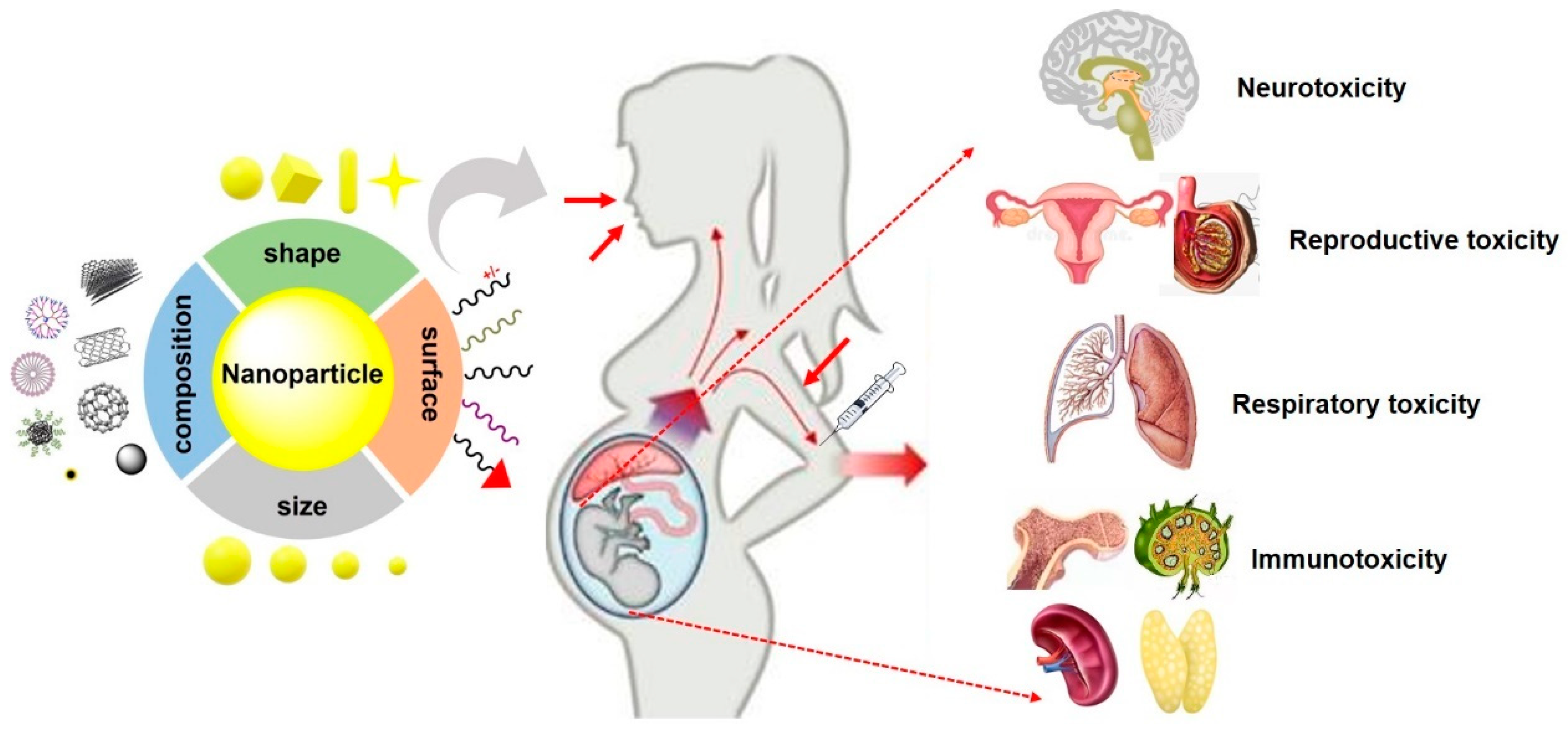
2. NPs-Induced Fetotoxicity
2.1. Adverse Prenatal Effects on Gestational Parameters
2.2. Neurotoxicity
2.3. Reproductive Toxicity
2.4. Immunotoxicity
2.5. Respiratory Toxicity
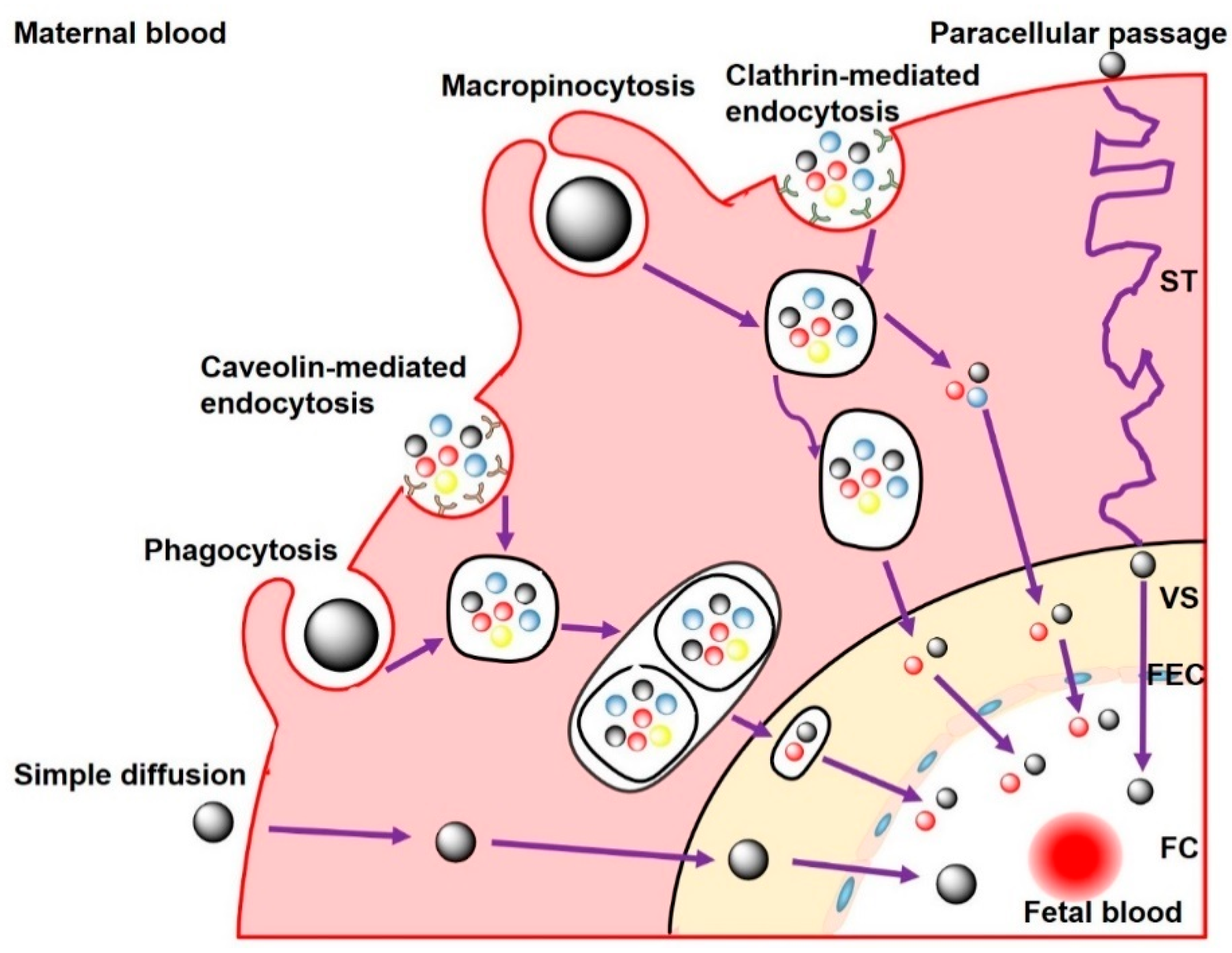
3. Transplacental Transfer of NPs
3.1. Maternal–Fetal NP Transfer
3.2. Transplacental Transport Mechanisms
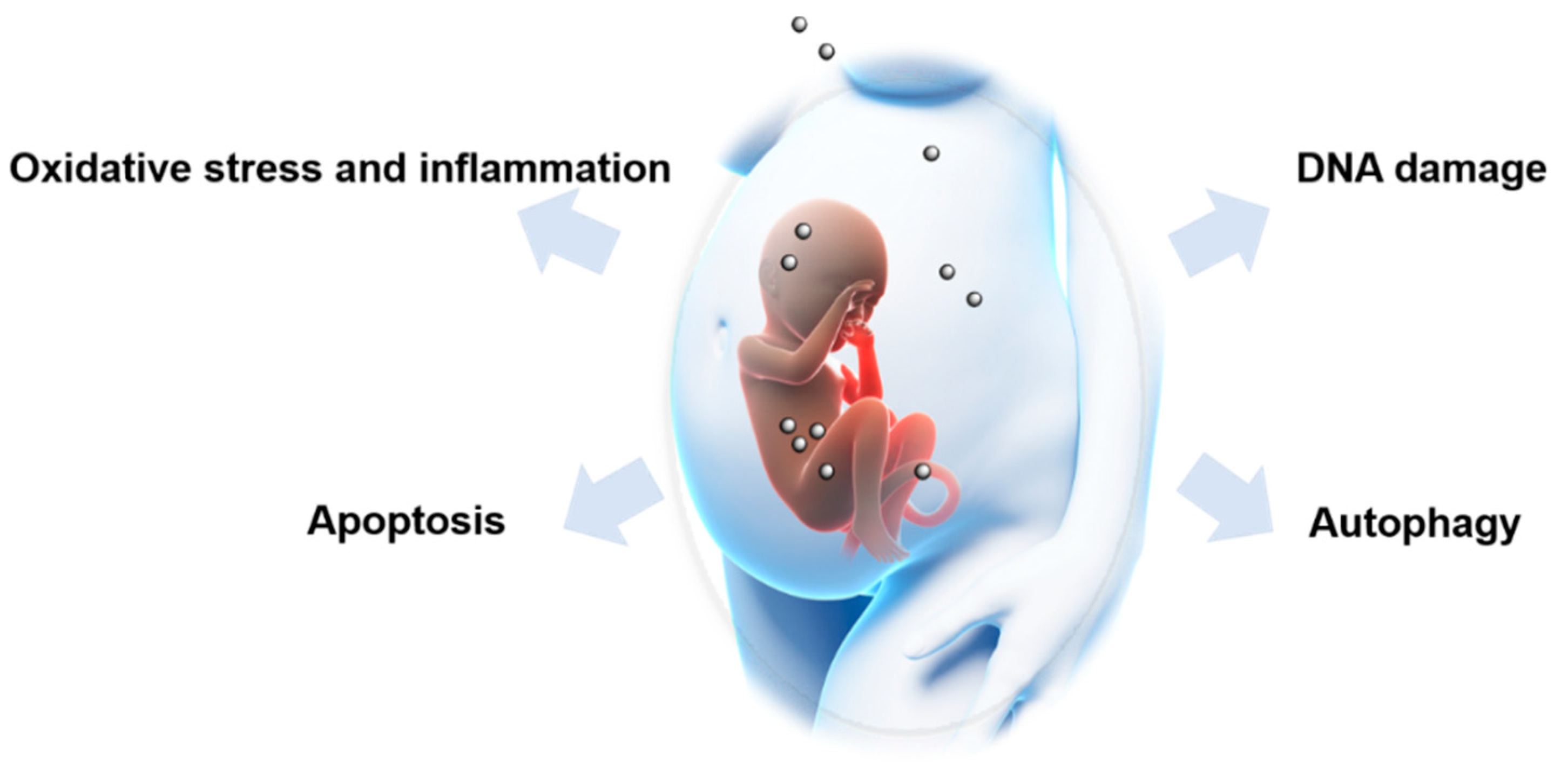
4. Molecular Mechanisms Involved in NP-Induced Fetotoxicity
4.1. Oxidative Stress and Inflammatory Responses
4.2. DNA Damage
4.3. Apotosis
4.4. Autophagy
5. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Petros, R.A.; DeSimone, J.M. Strategies in the design of nanoparticles for therapeutic applications. Nat. Rev. Drug Discov. 2010, 9, 615–627. [Google Scholar] [CrossRef]
- Bowman, D.M.; Van Calster, G.; Friedrichs, S. Nanomaterials and regulation of cosmetics. Nat. Nanotechnol. 2010, 5, 92. [Google Scholar] [CrossRef] [PubMed]
- Konstantatos, G.; Sargent, E.H. Nanostructured materials for photon detection. Nat. Nanotechnol. 2010, 5, 391–400. [Google Scholar] [CrossRef]
- Jia, J.; Li, F.; Zhai, S.; Zhou, H.; Liu, S.; Jiang, G.; Yan, B. Susceptibility of overweight mice to liver injury as a result of the ZnO nanoparticle-enhanced liver deposition of Pb2+. Environ. Sci. Technol. 2017, 51, 1775–1784. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, Y.; Yan, B. Nanotoxicity overview: Nano-threat to susceptible populations. Int. J. Mol. Sci. 2014, 15, 3671–3697. [Google Scholar] [CrossRef]
- Jia, J.; Li, F.; Zhou, H.; Bai, Y.; Liu, S.; Jiang, Y.; Jiang, G.; Yan, B. Oral exposure to silver nanoparticles or silver ions may aggravate fatty liver disease in overweight Mice. Environ. Sci. Technol. 2017, 51, 9334–9343. [Google Scholar] [CrossRef]
- Wei, Y.; Li, Y.; Jia, J.; Jiang, Y.; Zhao, B.; Zhang, Q.; Yan, B. Aggravated hepatotoxicity occurs in aged mice but not in young mice after oral exposure to zinc oxide nanoparticles. NanoImpact 2016, 3–4, 1–11. [Google Scholar] [CrossRef]
- Luyten, L.J.; Saenen, N.D.; Janssen, B.G.; Vrijens, K.; Plusquin, M.; Roels, H.A.; Debacq-Chainiaux, F.; Nawrot, T.S. Air pollution and the fetal origin of disease: A systematic review of the molecular signatures of air pollution exposure in human placenta. Environ. Res. 2018, 166, 310–323. [Google Scholar] [CrossRef]
- Barker, D.J. Fetal origins of coronary heart disease. BMJ 1995, 311, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Geng, Y.; Chen, J.; Gao, R.; Yu, C.; Yang, Z.; Chen, X.; Mu, X.; Liu, X.; He, J. Maternal exposure to CeO2 NPs during early pregnancy impairs pregnancy by inducing placental abnormalities. J. Hazard. Mater. 2020, 389, 1–10. [Google Scholar] [CrossRef]
- Teng, C.; Jia, J.; Wang, Z.; Yan, B. Oral co-exposures to zinc oxide nanoparticles and CdCl2 induced maternal-fetal pollutant transfer and embryotoxicity by damaging placental barriers. Ecotoxicol. Environ. Saf. 2020, 189, 109956. [Google Scholar] [CrossRef] [PubMed]
- Morales-Rubio, R.A.; Alvarado-Cruz, I.; Manzano-Leon, N.; Andrade-Oliva, M.D.; Uribe-Ramirez, M.; Quintanilla-Vega, B.; Osornio-Vargas, A.; De Vizcaya-Ruiz, A. In utero exposure to ultrafine particles promotes placental stress-induced programming of renin-angiotensin system-related elements in the offspring results in altered blood pressure in adult mice. Part. Fibre Toxicol. 2019, 16, 7. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Kuang, H.; Zhang, W.; Wei, H.; Xu, H. Quantum dots cause acute systemic toxicity in lactating rats and growth restriction of offspring. Nanoscale 2018, 10, 11564–11577. [Google Scholar] [CrossRef] [PubMed]
- Campagnolo, L.; Massimiani, M.; Vecchione, L.; Piccirilli, D.; Toschi, N.; Magrini, A.; Bonanno, E.; Scimeca, M.; Castagnozzi, L.; Buonanno, G.; et al. Silver nanoparticles inhaled during pregnancy reach and affect the placenta and the foetus. Nanotoxicology 2017, 11, 687–698. [Google Scholar] [CrossRef]
- Zhang, W.; Yang, L.; Kuang, H.; Yang, P.; Aguilar, Z.P.; Wang, A.; Fu, F.; Xu, H. Acute toxicity of quantum dots on late pregnancy mice: Effects of nanoscale size and surface coating. J. Hazard. Mater. 2016, 318, 61–69. [Google Scholar] [CrossRef]
- Yamashita, K.; Yoshioka, Y.; Higashisaka, K.; Mimura, K.; Morishita, Y.; Nozaki, M.; Yoshida, T.; Ogura, T.; Nabeshi, H.; Nagano, K.; et al. Silica and titanium dioxide nanoparticles cause pregnancy complications in mice. Nat. Nanotechnol. 2011, 6, 321–328. [Google Scholar] [CrossRef]
- Pietroiusti, A.; Massimiani, M.; Fenoglio, I.; Colonna, M.; Valentini, F.; Palleschi, G.; Camaioni, A.; Magrini, A.; Siracusa, G.; Bergamaschi, A.; et al. Low doses of pristine and oxidized single-wall carbon nanotubes affect mammalian embryonic development. ACS Nano 2011, 5, 4624–4633. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, C.; Huang, F.; Liu, X.; Wang, Z.; Yan, B. Breakthrough of ZrO2 nanoparticles into fetal brains depends on developmental stage of maternal placental barrier and fetal blood-brain-barrier. J. Hazard. Mater. 2021, 402, 1–11. [Google Scholar] [CrossRef]
- Zhou, Y.; Ji, J.; Chen, C.; Hong, F. Retardation of axonal and dendritic outgrowth is associated with the MAPK signaling pathway in offspring mice following maternal exposure to nanosized titanium dioxide. J. Agric. Food. Chem. 2019, 67, 2709–2715. [Google Scholar] [CrossRef]
- Morris-Schaffer, K.; Merrill, A.K.; Wong, C.; Jew, K.; Sobolewski, M.; Cory-Slechta, D.A. Limited developmental neurotoxicity from neonatal inhalation exposure to diesel exhaust particles in C57BL/6 mice. Part. Fibre Toxicol. 2019, 16, 1. [Google Scholar] [CrossRef]
- Hawkins, S.J.; Crompton, L.A.; Sood, A.; Saunders, M.; Boyle, N.T.; Buckley, A.; Minogue, A.M.; McComish, S.F.; Jimenez-Moreno, N.; Cordero-Llana, O.; et al. Nanoparticle-induced neuronal toxicity across placental barriers is mediated by autophagy and dependent on astrocytes. Nat. Nanotechnol. 2018, 13, 427–433. [Google Scholar] [CrossRef]
- Onoda, A.; Takeda, K.; Umezawa, M. Dose-dependent induction of astrocyte activation and reactive astrogliosis in mouse brain following maternal exposure to carbon black nanoparticle. Part. Fibre Toxicol. 2017, 14, 4. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, M.; Tainaka, H.; Oba, T.; Mizuo, K.; Umezawa, M.; Takeda, K. Maternal exposure to nanoparticulate titanium dioxide during the prenatal period alters gene expression related to brain development in the mouse. Part. Fibre Toxicol. 2009, 6, 20. [Google Scholar] [CrossRef]
- Skovmand, A.; Jensen, A.C.O.; Maurice, C.; Marchetti, F.; Lauvas, A.J.; Koponen, I.K.; Jensen, K.A.; Goericke-Pesch, S.; Vogel, U.; Hougaard, K.S. Effects of maternal inhalation of carbon black nanoparticles on reproductive and fertility parameters in a four-generation study of male mice. Part. Fibre Toxicol. 2019, 16, 13. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Hiyoshi, K.; Oshio, S.; Takano, H.; Takeda, K.; Ichinose, T. Effects of fetal exposure to carbon nanoparticles on reproductive function in male offspring. Fertil. Steril. 2010, 93, 1695–1699. [Google Scholar] [CrossRef] [PubMed]
- Johansson, H.K.L.; Hansen, J.S.; Elfving, B.; Lund, S.P.; Kyjovska, Z.O.; Loft, S.; Barfod, K.K.; Jackson, P.; Vogel, U.; Hougaard, K.S. Airway exposure to multi-walled carbon nanotubes disrupts the female reproductive cycle without affecting pregnancy outcomes in mice. Part. Fibre Toxicol. 2017, 14, 17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhang, Z.; Wu, Y.; Fu, K.; Chen, Y.; Li, W.; Chu, M. Systematic evaluation of graphene quantum dot toxicity to male mouse sexual behaviors, reproductive and offspring health. Biomaterials 2019, 194, 215–232. [Google Scholar] [CrossRef]
- Kyjovska, Z.O.; Boisen, A.M.; Jackson, P.; Wallin, H.; Vogel, U.; Hougaard, K.S. Daily sperm production: Application in studies of prenatal exposure to nanoparticles in mice. Reprod. Toxicol. 2013, 36, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; You, D.; Saravia, J.; Shen, H.; Cormier, S.A. Maternal exposure to combustion generated PM inhibits pulmonary Th1 maturation and concomitantly enhances postnatal asthma development in offspring. Part. Fibre Toxicol. 2013, 10, 29. [Google Scholar] [CrossRef]
- Chen, L.; Bennett, E.; Wheeler, A.J.; Lyons, A.B.; Woods, G.M.; Johnston, F.; Zosky, G.R. Maternal exposure to particulate matter alters early post-natal lung function and immune cell development. Environ. Res. 2018, 164, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Adamcakova-Dodd, A.; Monick, M.M.; Powers, L.S.; Gibson-Corley, K.N.; Thorne, P.S. Effects of prenatal inhalation exposure to copper nanoparticles on murine dams and offspring. Part. Fibre Toxicol. 2015, 12, 30. [Google Scholar] [CrossRef]
- El-Sayed, Y.S.; Shimizu, R.; Onoda, A.; Takeda, K.; Umezawa, M. Carbon black nanoparticle exposure during middle and late fetal development induces immune activation in male offspring mice. Toxicology 2015, 327, 53–61. [Google Scholar] [CrossRef]
- Yue, H.; Ji, X.; Li, G.; Hu, M.; Sang, N. Maternal exposure to PM2.5 affects fetal lung development at sensitive windows. Environ. Sci. Technol. 2020, 54, 316–324. [Google Scholar] [CrossRef]
- Yue, H.; Ji, X.; Ku, T.; Li, G.; Sang, N. Sex difference in bronchopulmonary dysplasia of offspring in response to maternal PM2.5 exposure. J. Hazard. Mater. 2020, 389, 122033. [Google Scholar] [CrossRef]
- Paul, E.; Franco-Montoya, M.L.; Paineau, E.; Angeletti, B.; Vibhushan, S.; Ridoux, A.; Tiendrebeogo, A.; Salome, M.; Hesse, B.; Vantelon, D.; et al. Pulmonary exposure to metallic nanomaterials during pregnancy irreversibly impairs lung development of the offspring. Nanotoxicology 2017, 11, 484–495. [Google Scholar] [CrossRef]
- Bove, H.; Bongaerts, E.; Slenders, E.; Bijnens, E.M.; Saenen, N.D.; Gyselaers, W.; Van Eyken, P.; Plusquin, M.; Roeffaers, M.B.J.; Ameloot, M.; et al. Ambient black carbon particles reach the fetal side of human placenta. Nat. Commun. 2019, 10, 3866. [Google Scholar] [CrossRef] [PubMed]
- Buerki-Thurnherr, T.; Schaepper, K.; Aengenheister, L.; Wick, P. Developmental toxicity of nanomaterials: Need for a better understanding of indirect effects. Chem. Res. Toxicol. 2018, 31, 641–642. [Google Scholar] [CrossRef]
- Dugershaw, B.B.; Aengenheister, L.; Hansen, S.S.K.; Hougaard, K.S.; Buerki-Thurnherr, T. Recent insights on indirect mechanisms in developmental toxicity of nanomaterials. Part. Fibre Toxicol. 2020, 17, 31. [Google Scholar] [CrossRef] [PubMed]
- Manangama, G.; Audignon-Durand, S.; Migault, L.; Gramond, C.; Zaros, C.; Teysseire, R.; Sentilhes, L.; Brochard, P.; Lacourt, A.; Delva, F. Maternal occupational exposure to carbonaceous nanoscale particles and small for gestational age and the evolution of head circumference in the French Longitudinal Study of Children—Elfe study. Environ. Res. 2020, 185, 109394. [Google Scholar] [CrossRef]
- Manangama, G.; Migault, L.; Audignon-Durand, S.; Gramond, C.; Zaros, C.; Bouvier, G.; Brochard, P.; Sentilhes, L.; Lacourt, A.; Delva, F. Maternal occupational exposures to nanoscale particles and small for gestational age outcome in the French Longitudinal Study of Children. Environ. Int. 2019, 122, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Valentino, S.A.; Tarrade, A.; Aioun, J.; Mourier, E.; Richard, C.; Dahirel, M.; Rousseau-Ralliard, D.; Fournier, N.; Aubriere, M.C.; Lallemand, M.S.; et al. Maternal exposure to diluted diesel engine exhaust alters placental function and induces intergenerational effects in rabbits. Part. Fibre Toxicol. 2016, 13, 39. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Li, X.; Suzuki, A.K.; Zhang, Y.; Fujitani, Y.; Nagaoka, K.; Watanabe, G.; Taya, K. Effects of exposure to nanoparticle-rich diesel exhaust on pregnancy in rats. J. Reprod. Dev. 2013, 59, 145–150. [Google Scholar] [CrossRef]
- Jackson, P.; Hougaard, K.S.; Boisen, A.M.; Jacobsen, N.R.; Jensen, K.A.; Moller, P.; Brunborg, G.; Gutzkow, K.B.; Andersen, O.; Loft, S.; et al. Pulmonary exposure to carbon black by inhalation or instillation in pregnant mice: Effects on liver DNA strand breaks in dams and offspring. Nanotoxicology 2012, 6, 486–500. [Google Scholar] [CrossRef]
- Fujitani, T.; Ohyama, K.-I.; Hirose, A.; Nishimura, T.; Nakae, D.; Ogata, A. Teratogenicity of multi-wall carbon nanotube (MWCNT) in ICR mice. J. Toxicol. Sci. 2012, 37, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Vidanapathirana, A.K.; Thompson, L.C.; Odom, J.; Holland, N.A.; Sumner, S.J.; Fennell, T.R.; Brown, J.M.; Wingard, C.J. Vascular tissue contractility changes following late gestational exposure to multi-walled carbon nanotubes or their dispersing vehicle in Sprague Dawley rats. J. Nanomed. Nanotechnol. 2014, 5. [Google Scholar] [CrossRef]
- Xu, S.; Zhang, Z.; Chu, M. Long-term toxicity of reduced graphene oxide nanosheets: Effects on female mouse reproductive ability and offspring development. Biomaterials 2015, 54, 188–200. [Google Scholar] [CrossRef]
- Chen, B.; Hong, W.; Yang, P.; Tang, Y.; Zhao, Y.; Aguilar, Z.P.; Xu, H. Nano zinc oxide induced fetal mice growth restriction, based on oxide stress and endoplasmic reticulum stress. Nanomaterials 2020, 10, 259. [Google Scholar] [CrossRef]
- Teng, C.; Jia, J.; Wang, Z.; Sharma, V.K.; Yan, B. Size-dependent maternal-fetal transfer and fetal developmental toxicity of ZnO nanoparticles after oral exposures in pregnant mice. Ecotoxicol. Environ. Saf. 2019, 182, 109439. [Google Scholar] [CrossRef]
- Lee, J.; Yu, W.J.; Song, J.; Sung, C.; Jeong, E.J.; Han, J.S.; Kim, P.; Jo, E.; Eom, I.; Kim, H.M.; et al. Developmental toxicity of intravenously injected zinc oxide nanoparticles in rats. Arch. Pharm. Res. 2016, 39, 1682–1692. [Google Scholar] [CrossRef]
- Hong, J.S.; Park, M.K.; Kim, M.S.; Lim, J.H.; Park, G.J.; Maeng, E.H.; Shin, J.H.; Kim, M.K.; Jeong, J.; Park, J.A.; et al. Prenatal development toxicity study of zinc oxide nanoparticles in rats. Int. J. Nanomed. 2014, 9, 159–171. [Google Scholar] [CrossRef][Green Version]
- Jo, E.; Seo, G.; Kwon, J.-T.; Lee, M.; Lee, B.C.; Eom, I.; Kim, P.; Choi, K. Exposure to zinc oxide nanoparticles affects reproductive development and biodistribution in offspring rats. J. Toxicol. Sci. 2013, 38, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Bowdridge, E.C.; Abukabda, A.B.; Engles, K.J.; McBride, C.R.; Batchelor, T.P.; Goldsmith, W.T.; Garner, K.L.; Friend, S.; Nurkiewicz, T.R. Maternal engineered nanomaterial inhalation during gestation disrupts vascular kisspeptin reactivity. Toxicol. Sci. 2019, 169, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Zhou, Y.; Zhao, X.; Sheng, L.; Wang, L. Maternal exposure to nanosized titanium dioxide suppresses embryonic development in mice. Int. J. Nanomed. 2017, 12, 6197–6204. [Google Scholar] [CrossRef] [PubMed]
- Pietroiusti, A.; Vecchione, L.; Malvindi, M.A.; Aru, C.; Massimiani, M.; Camaioni, A.; Magrini, A.; Bernardini, R.; Sabella, S.; Pompa, P.P.; et al. Relevance to investigate different stages of pregnancy to highlight toxic effects of nanoparticles: The example of silica. Toxicol. Appl. Pharmacol. 2018, 342, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Blum, J.L.; Xiong, J.Q.; Hoffman, C.; Zelikoff, J.T. Cadmium associated with inhaled cadmium oxide nanoparticles impacts fetal and neonatal development and growth. Toxicol. Sci. 2012, 126, 478–486. [Google Scholar] [CrossRef]
- Vidanapathirana, A.K.; Thompson, L.C.; Herco, M.; Odom, J.; Sumner, S.J.; Fennell, T.R.; Brown, J.M.; Wingard, C.J. Acute intravenous exposure to silver nanoparticles during pregnancy induces particle size and vehicle dependent changes in vascular tissue contractility in Sprague Dawley rats. Reprod. Toxicol. 2018, 75, 10–22. [Google Scholar] [CrossRef]
- Austin, C.A.; Hinkley, G.K.; Mishra, A.R.; Zhang, Q.; Umbreit, T.H.; Betz, M.W.; Wildt, B.E.; Casey, B.J.; Francke-Carroll, S.; Hussain, S.M.; et al. Distribution and accumulation of 10 nm silver nanoparticles in maternal tissues and visceral yolk sac of pregnant mice, and a potential effect on embryo growth. Nanotoxicology 2016, 10, 654–661. [Google Scholar] [CrossRef]
- Ma, X.; Yang, X.; Wang, Y.; Liu, J.; Jin, S.; Li, S.; Liang, X.-J. Gold nanoparticles cause size-dependent inhibition of embryonic development during murine pregnancy. Nano Res. 2018, 11, 3419–3433. [Google Scholar] [CrossRef]
- Bara, N.; Eshwarmoorthy, M.; Subaharan, K.; Kaul, G. Mesoporous silica nanoparticle is comparatively safer than zinc oxide nanoparticle which can cause profound steroidogenic effects on pregnant mice and male offspring exposed in utero. Toxicol. Ind. Health 2018, 34, 507–524. [Google Scholar] [CrossRef]
- Di Bona, K.R.; Xu, Y.; Gray, M.; Fair, D.; Hayles, H.; Milad, L.; Montes, A.; Sherwood, J.; Bao, Y.; Rasco, J.F. Short- and long-term effects of prenatal exposure to iron oxide nanoparticles: Influence of surface charge and dose on developmental and reproductive toxicity. Int. J. Mol. Sci. 2015, 16, 30251–30268. [Google Scholar] [CrossRef]
- Stapleton, P.A.; Hathaway, Q.A.; Nichols, C.E.; Abukabda, A.B.; Pinti, M.V.; Shepherd, D.L.; McBride, C.R.; Yi, J.; Castranova, V.C.; Hollander, J.M.; et al. Maternal engineered nanomaterial inhalation during gestation alters the fetal transcriptome. Part. Fibre Toxicol. 2018, 15, 3. [Google Scholar] [CrossRef]
- Zalgeviciene, V.; Kulvietis, V.; Bulotiene, D.; Zurauskas, E.; Laurinaviciene, A.; Skripka, A.; Rotomskis, R. Quantum dots mediated embryotoxicity via placental damage. Reprod. Toxicol. 2017, 73, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Di Bona, K.R.; Xu, Y.; Ramirez, P.A.; DeLaine, J.; Parker, C.; Bao, Y.; Rasco, J.F. Surface charge and dosage dependent potential developmental toxicity and biodistribution of iron oxide nanoparticles in pregnant CD-1 mice. Reprod. Toxicol. 2014, 50, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Shirasuna, K.; Usui, F.; Karasawa, T.; Kimura, H.; Kawashima, A.; Mizukami, H.; Ohkuchi, A.; Nishimura, S.; Sagara, J.; Noda, T.; et al. Nanosilica-induced placental inflammation and pregnancy complications: Different roles of the inflammasome components NLRP3 and ASC. Nanotoxicology 2015, 9, 554–567. [Google Scholar] [CrossRef]
- Qi, W.; Bi, J.; Zhang, X.; Wang, J.; Wang, J.; Liu, P.; Li, Z.; Wu, W. Damaging effects of multi-walled carbon nanotubes on pregnant mice with different pregnancy times. Sci. Rep. 2014, 4, 4352. [Google Scholar] [CrossRef]
- Campagnolo, L.; Massimiani, M.; Palmieri, G.; Bernardini, R.; Sacchetti, C.; Bergamaschi, A.; Vecchione, L.; Magrini, A.; Bottini, M.; Pietroiusti, A. Biodistribution and toxicity of pegylated single wall carbon nanotubes in pregnant mice. Part. Fibre Toxicol. 2013, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Philbrook, N.A.; Walker, V.K.; Afrooz, A.R.; Saleh, N.B.; Winn, L.M. Investigating the effects of functionalized carbon nanotubes on reproduction and development in Drosophila melanogaster and CD-1 mice. Reprod. Toxicol. 2011, 32, 442–448. [Google Scholar] [CrossRef]
- Philbrook, N.A.; Winn, L.M.; Afrooz, A.R.; Saleh, N.B.; Walker, V.K. The effect of TiO2 and Ag nanoparticles on reproduction and development of Drosophila melanogaster and CD-1 mice. Toxicol. Appl. Pharmacol. 2011, 257, 429–436. [Google Scholar] [CrossRef]
- Reliene, R.; Hlavacova, A.; Mahadevan, B.; Baird, W.M.; Schiestl, R.H. Diesel exhaust particles cause increased levels of DNA deletions after transplacental exposure in mice. Mutat. Res. 2005, 570, 245–252. [Google Scholar] [CrossRef]
- Huang, X.; Zhang, F.; Sun, X.; Choi, K.Y.; Niu, G.; Zhang, G.; Guo, J.; Lee, S.; Chen, X. The genotype-dependent influence of functionalized multiwalled carbon nanotubes on fetal development. Biomaterials 2014, 35, 856–865. [Google Scholar] [CrossRef] [PubMed]
- Hougaard, K.S.; Jackson, P.; Kyjovska, Z.O.; Birkedal, R.K.; De Temmerman, P.J.; Brunelli, A.; Verleysen, E.; Madsen, A.M.; Saber, A.T.; Pojana, G.; et al. Effects of lung exposure to carbon nanotubes on female fertility and pregnancy. A study in mice. Reprod. Toxicol. 2013, 41, 86–97. [Google Scholar] [CrossRef]
- Lim, J.-H.; Kim, S.-H.; Lee, I.-C.; Moon, C.; Kim, S.-H.; Shin, D.-H.; Kim, H.-C.; Kim, J.-C. Evaluation of maternal toxicity in rats exposed to multi-wall carbon nanotubes during pregnancy. Environ. Health Toxicol. 2011, 26, e2011006. [Google Scholar] [CrossRef]
- Lim, J.H.; Kim, S.H.; Shin, I.S.; Park, N.H.; Moon, C.; Kang, S.S.; Kim, S.H.; Park, S.C.; Kim, J.C. Maternal exposure to multi-wall carbon nanotubes does not induce embryo-fetal developmental toxicity in rats. Birth Defects Res. B Dev. Reprod. Toxicol. 2011, 92, 69–76. [Google Scholar] [CrossRef]
- Hougaard, K.S.; Jackson, P.; Jensen, K.A.; Sloth, J.J.; Loeschner, K.; Larsen, E.H.; Birkedal, R.K.; Vibenholt, A.; Boisen, A.-M.Z.; Wallin, H.; et al. Effects of prenatal exposure to surface-coated nanosized titanium dioxide (UV-Titan). A study in mice. Part. Fibre Toxicol. 2010, 7. [Google Scholar] [CrossRef]
- Guillard, A.; Gaultier, E.; Cartier, C.; Devoille, L.; Noireaux, J.; Chevalier, L.; Morin, M.; Grandin, F.; Lacroix, M.Z.; Coméra, C.; et al. Basal Ti level in the human placenta and meconium and evidence of a materno-foetal transfer of food-grade TiO2 nanoparticles in an ex vivo placental perfusion model. Part. Fibre Toxicol. 2020, 17. [Google Scholar] [CrossRef]
- Allen, J.L.; Liu, X.; Pelkowski, S.; Palmer, B.; Conrad, K.; Oberdorster, G.; Weston, D.; Mayer-Proschel, M.; Cory-Slechta, D.A. Early postnatal exposure to ultrafine particulate matter air pollution: Persistent ventriculomegaly, neurochemical disruption, and glial activation preferentially in male mice. Environ. Health Perspect. 2014, 122, 939–945. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.L.; Conrad, K.; Oberdorster, G.; Johnston, C.J.; Sleezer, B.; Cory-Slechta, D.A. Developmental exposure to concentrated ambient particles and preference for immediate reward in mice. Environ. Health Perspect. 2013, 121, 32–38. [Google Scholar] [CrossRef]
- Woodward, N.C.; Haghani, A.; Johnson, R.G.; Hsu, T.M.; Saffari, A.; Sioutas, C.; Kanoski, S.E.; Finch, C.E.; Morgan, T.E. Prenatal and early life exposure to air pollution induced hippocampal vascular leakage and impaired neurogenesis in association with behavioral deficits. Transl. Psychiatry 2018, 8, 261. [Google Scholar] [CrossRef] [PubMed]
- Kulas, J.A.; Hettwer, J.V.; Sohrabi, M.; Melvin, J.E.; Manocha, G.D.; Puig, K.L.; Gorr, M.W.; Tanwar, V.; McDonald, M.P.; Wold, L.E.; et al. In utero exposure to fine particulate matter results in an altered neuroimmune phenotype in adult mice. Environ. Pollut. 2018, 241, 279–288. [Google Scholar] [CrossRef]
- Bernal-Melendez, E.; Lacroix, M.C.; Bouillaud, P.; Callebert, J.; Olivier, B.; Persuy, M.A.; Durieux, D.; Rousseau-Ralliard, D.; Aioun, J.; Cassee, F.; et al. Repeated gestational exposure to diesel engine exhaust affects the fetal olfactory system and alters olfactory-based behavior in rabbit offspring. Part. Fibre Toxicol. 2019, 16, 5. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Oshio, S.; Iwata, M.; Saburi, H.; Odagiri, T.; Udagawa, T.; Sugawara, I.; Umezawa, M.; Takeda, K. In utero exposure to a low concentration of diesel exhaust affects spontaneous locomotor activity and monoaminergic system in male mice. Part. Fibre Toxicol. 2010, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, M.; Onoda, A.; Korshunova, I.; Jensen, A.C.O.; Koponen, I.K.; Jensen, K.A.; Khodosevich, K.; Vogel, U.; Hougaard, K.S. Maternal inhalation of carbon black nanoparticles induces neurodevelopmental changes in mouse offspring. Part. Fibre Toxicol. 2018, 15, 36. [Google Scholar] [CrossRef]
- Onoda, A.; Takeda, K.; Umezawa, M. Dysregulation of major functional genes in frontal cortex by maternal exposure to carbon black nanoparticle is not ameliorated by ascorbic acid pretreatment. Sci. Total Environ. 2018, 634, 1126–1135. [Google Scholar] [CrossRef] [PubMed]
- Onoda, A.; Takeda, K.; Umezawa, M. Pretreatment with N-acetyl cysteine suppresses chronic reactive astrogliosis following maternal nanoparticle exposure during gestational period. Nanotoxicology 2017, 11, 1012–1025. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Wu, J.; Lai, X.; Zhang, Y.; Wei, L.; Liu, J.; Shao, L. Prenatal exposure to nanosized zinc oxide in rats: Neurotoxicity and postnatal impaired learning and memory ability. Nanomedicine 2017, 12, 777–795. [Google Scholar] [CrossRef]
- Notter, T.; Aengenheister, L.; Weber-Stadlbauer, U.; Naegeli, H.; Wick, P.; Meyer, U.; Buerki-Thurnherr, T. Prenatal exposure to TiO2 nanoparticles in mice causes behavioral deficits with relevance to autism spectrum disorder and beyond. Transl. Psychiatry 2018, 8, 193. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.; Zhou, Y.; Ji, J.; Zhuang, J.; Sheng, L.; Wang, L. Nano-TiO2 inhibits development of the central nervous system and its mechanism in offspring mice. J. Agric. Food. Chem. 2018, 66, 11767–11774. [Google Scholar] [CrossRef]
- Mohammadipour, A.; Fazel, A.; Haghir, H.; Motejaded, F.; Rafatpanah, H.; Zabihi, H.; Hosseini, M.; Bideskan, A.E. Maternal exposure to titanium dioxide nanoparticles during pregnancy; impaired memory and decreased hippocampal cell proliferation in rat offspring. Environ. Toxicol. Pharmacol. 2014, 37, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Chen, X.; Zhou, Z.; Lei, Y.; Ma, M.; Cao, R.; Sun, T.; Xu, J.; Huo, M.; Cao, R.; et al. Prenatal exposure to nanoparticulate titanium dioxide enhances depressive-like behaviors in adult rats. Chemosphere 2014, 96, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Tabatabaei, S.R.; Moshrefi, M.; Askaripour, M. Prenatal exposure to silver nanoparticles causes depression like responses in mice. Ind. J. Pharm. Sci. 2015, 77, 681–686. [Google Scholar] [CrossRef]
- Klocke, C.; Allen, J.L.; Sobolewski, M.; Mayer-Proschel, M.; Blum, J.L.; Lauterstein, D.; Zelikoff, J.T.; Cory-Slechta, D.A. Neuropathological consequences of gestational exposure to concentrated ambient fine and ultrafine particles in the mouse. Toxicol. Sci. 2017, 156, 492–508. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimzadeh Bideskan, A.; Mohammadipour, A.; Fazel, A.; Haghir, H.; Rafatpanah, H.; Hosseini, M.; Rajabzadeh, A. Maternal exposure to titanium dioxide nanoparticles during pregnancy and lactation alters offspring hippocampal mRNA BAX and Bcl-2 levels, induces apoptosis and decreases neurogenesis. Exp. Toxicol. Pathol. 2017, 69, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Hong, F.; Tian, Y.; Zhao, X.; Hong, J.; Ze, Y.; Wang, L. Nanoparticulate titanium dioxide-inhibited dendritic development is involved in apoptosis and autophagy of hippocampal neurons in offspring mice. Toxicol. Res. 2017, 6, 889–901. [Google Scholar] [CrossRef] [PubMed]
- Alimohammadi, S.; Hassanpour, S.; Moharramnejad, S. Effect of maternal exposure to zinc oxide nanoparticles on reflexive motor behaviors in mice offspring. Int. J. Pept. Res. Ther. 2019, 25, 1049–1056. [Google Scholar] [CrossRef]
- Alimohammadi, S.; Hosseini, M.S.; Behbood, L. Prenatal exposure to zinc oxide nanoparticles can induce depressive-like behaviors in mice offspring. Int. J. Pept. Res. Ther. 2019, 25, 401–409. [Google Scholar] [CrossRef]
- Naserzadeh, P.; Ghanbary, F.; Ashtari, P.; Seydi, E.; Ashtari, K.; Akbari, M. Biocompatibility assessment of titanium dioxide nanoparticles in mice fetoplacental unit. J. Biomed. Mater. Res. Part A 2018, 106, 580–589. [Google Scholar] [CrossRef]
- Engler-Chiurazzi, E.B.; Stapleton, P.A.; Stalnaker, J.J.; Ren, X.; Hu, H.; Nurkiewicz, T.R.; McBride, C.R.; Yi, J.; Engels, K.; Simpkins, J.W. Impacts of prenatal nanomaterial exposure on male adult Sprague-Dawley rat behavior and cognition. J. Toxicol. Environ. Health A 2016, 79, 447–452. [Google Scholar] [CrossRef]
- Ghaderi, S.; Tabatabaei, S.R.F.; Varzi, H.N.; Rashno, M. Induced adverse effects of prenatal exposure to silver nanoparticles on neurobehavioral development of offspring of mice. J. Toxicol. Sci. 2015, 40, 263–275. [Google Scholar] [CrossRef]
- Girardi, G.; Fraser, J.; Lennen, R.; Vontell, R.; Jansen, M.; Hutchison, G. Imaging of activated complement using ultrasmall superparamagnetic iron oxide particles (USPIO)--conjugated vectors: An in vivo in utero non-invasive method to predict placental insufficiency and abnormal fetal brain development. Mol. Psychiatry 2015, 20, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Mortamais, M.; Pujol, J.; Martinez-Vilavella, G.; Fenoll, R.; Reynes, C.; Sabatier, R.; Rivas, I.; Forns, J.; Vilor-Tejedor, N.; Alemany, S.; et al. Effects of prenatal exposure to particulate matter air pollution on corpus callosum and behavioral problems in children. Environ. Res. 2019, 178, 108734. [Google Scholar] [CrossRef] [PubMed]
- Kalkbrenner, A.E.; Windham, G.C.; Serre, M.L.; Akita, Y.; Wang, X.; Hoffman, K.; Thayer, B.P.; Daniels, J.L. Particulate matter exposure, prenatal and postnatal windows of susceptibility, and autism spectrum disorders. Epidemiology 2015, 26, 30–42. [Google Scholar] [CrossRef]
- Siddique, S.; Banerjee, M.; Ray, M.R.; Lahiri, T. Attention-deficit hyperactivity disorder in children chronically exposed to high level of vehicular pollution. Eur. J. Pediatr. 2011, 170, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Klocke, C.; Allen, J.L.; Sobolewski, M.; Blum, J.L.; Zelikoff, J.T.; Cory-Slechta, D.A. Exposure to fine and ultrafine particulate matter during gestation alters postnatal oligodendrocyte maturation, proliferation capacity, and myelination. Neurotoxicology 2018, 65, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, S.; Ichinose, T.; Arashidani, K.; He, M.; Takano, H.; Shibamoto, T. Effects of fetal exposure to asian sand dust on development and reproduction in male offspring. Int. J. Environ. Res. Public Health 2016, 13, 1173. [Google Scholar] [CrossRef]
- Li, C.; Taneda, S.; Taya, K.; Watanabe, G.; Li, X.; Fujitani, Y.; Nakajima, T.; Suzuki, A.K. Effects of in utero exposure to nanoparticle-rich diesel exhaust on testicular function in immature male rats. Toxicol. Lett. 2009, 185, 1–8. [Google Scholar] [CrossRef]
- Kubo-Irie, M.; Uchida, H.; Mastuzawa, S.; Yoshida, Y.; Shinkai, Y.; Suzuki, K.; Yokota, S.; Oshio, S.; Takeda, K. Dose–dependent biodistribution of prenatal exposure to rutile-type titanium dioxide nanoparticles on mouse testis. J. Nanopart. Res. 2014, 16. [Google Scholar] [CrossRef]
- Takeda, K.S.; Ken-Ichiro, S.; Ishihara, A.; Kubo-Irie, M.; Fujimoto, R.; Tabata, M.; Oshio, S.; Nihei, Y.; Ihara, T.; Sugamata, M. Nanoparticles tranferred from pregnant mice to their offspring can damage the genital and cranial nerve systems. J. Health Sci. 2009, 55, 95–102. [Google Scholar] [CrossRef]
- Bourdon, M.; Torres-Rovira, L.; Monniaux, D.; Faure, C.; Levy, R.; Tarrade, A.; Rousseau-Ralliard, D.; Chavatte-Palmer, P.; Jolivet, G. Impact of a gestational exposure to diesel exhaust on offspring gonadal development: Experimental study in the rabbit. J. Dev. Orig. Health Dis. 2018, 9, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zeng, X.; Du, X.; Pan, K.; Song, L.; Song, W.; Xie, Y.; Zhao, J. Parental PM2.5 exposure-promoted development of metabolic syndrome in offspring is associated with the changes of immune microenvironment. Toxicol. Sci. 2019, 170, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Fedulov, A.V.; Leme, A.; Yang, Z.; Dahl, M.; Lim, R.; Mariani, T.J.; Kobzik, L. Pulmonary exposure to particles during pregnancy causes increased neonatal asthma susceptibility. Am. J. Respir. Cell Mol. Biol. 2008, 38, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, R.; Umezawa, M.; Okamoto, S.; Noda, A.; Uchiyama, M.; Tachibana, K.; Watanabe, S.; Ogawa, S.; Abe, R.; Takeda, K. Effect of maternal exposure to carbon black nanoparticle during early gestation on the splenic phenotype of neonatal mouse. J. Toxicol. Sci. 2014, 39, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Gregory, D.J.; Kobzik, L.; Yang, Z.; McGuire, C.C.; Fedulov, A.V. Transgenerational transmission of asthma risk after exposure to environmental particles during pregnancy. Am. J. Physiol. Cell. Mol. Physiol. 2017, 313, L395–L405. [Google Scholar] [CrossRef] [PubMed]
- Elbastawisy, Y.M.; Almasry, S.M. Histomorphological evaluation of maternal and neonatal distal airspaces after maternal intake of nanoparticulate titanium dioxide: An experimental study in Wistar rats. J. Mol. Histol. 2014, 45, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Rossant, J.; Cross, J.C. Placental development: Lessons from mouse mutants. Nat. Rev. Genet. 2001, 2, 538–548. [Google Scholar] [CrossRef]
- Coyne, C.B.; Lazear, H.M. Zika virus—reigniting the TORCH. Nat. Rev. Microbiol. 2016, 14, 707–715. [Google Scholar] [CrossRef]
- Enders, A.C.; Blankenship, T.N. Comparative placental structure. Adv. Drug Deliv. Rev. 1999, 38, 3–15. [Google Scholar] [CrossRef]
- Bongaerts, E.; Nawrot, T.S.; Van Pee, T.; Ameloot, M.; Bove, H. Translocation of (ultra)fine particles and nanoparticles across the placenta; a systematic review on the evidence of in vitro, ex vivo, and in vivo studies. Part. Fibre Toxicol. 2020, 17, 56. [Google Scholar] [CrossRef]
- Bai, X.; Wang, S.Q.; Yan, X.L.; Zhou, H.Y.; Zhan, J.H.; Liu, S.J.; Sharma, V.K.; Jiang, G.B.; Zhu, H.; Yan, B. Regulation of Cell Uptake and Cytotoxicity by Nanoparticle Core under the Controlled Shape, Size, and Surface Chemistries. ACS Nano 2020, 14, 289–302. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, C.; Liu, X.; Huang, F.; Wang, Z.; Yan, B. Oral intake of ZrO2 nanoparticles by pregnant mice results in nanoparticles’ deposition in fetal brains. Ecotoxicol. Environ. Saf. 2020, 202, 110884. [Google Scholar] [CrossRef]
- Tian, X.; Zhu, M.; Du, L.; Wang, J.; Fan, Z.; Liu, J.; Zhao, Y.; Nie, G. Intrauterine inflammation increases materno-fetal transfer of gold nanoparticles in a size-dependent manner in murine pregnancy. Small 2013, 9, 2432–2439. [Google Scholar] [CrossRef]
- Semmler-Behnke, M.; Lipka, J.; Wenk, A.; Hirn, S.; Schäffler, M.; Tian, F.; Schmid, G.; Oberdörster, G.; Kreyling, W.G. Size dependent translocation and fetal accumulation of gold nanoparticles from maternal blood in the rat. Part. Fibre Toxicol. 2014, 11, 33. [Google Scholar] [CrossRef] [PubMed]
- Muoth, C.; Großgarten, M.; Karst, U.; Ruiz, J.; Astruc, D.; Moya, S.; Diener, L.; Grieder, K.; Wichser, A.; Jochum, W.; et al. Impact of particle size and surface modifcation on gold nanoparticle penetration into human placental microtissues. Nanomedicine 2017, 12, 1119–1133. [Google Scholar] [CrossRef]
- Chu, M.; Wu, Q.; Yang, H.; Yuan, R.; Hou, S.; Yang, Y.; Zou, Y.; Xu, S.; Xu, K.; Ji, A.; et al. Transfer of quantum dots from pregnant mice to pups across the placental barrier. Small 2010, 6, 670–678. [Google Scholar] [CrossRef]
- Jia, J.; Wang, Z.; Yue, T.; Su, G.; Teng, C.; Yan, B. Crossing biological barriers by engineered nanoparticles. Chem. Res. Toxicol. 2020, 33, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Muoth, C.; Aengenheister, L.; Kucki, M.; Wick, P.; Buerki-Thurnherr, T. Nanoparticle transport across the placental barrier: Pushing the field forward! Nanomedicine 2016, 11, 941–957. [Google Scholar] [CrossRef] [PubMed]
- Keelan, J.A. Nanotoxicology: Nanoparticles versus the placenta. Nat. Nanotechnol. 2011, 6, 263–264. [Google Scholar] [CrossRef]
- Refuerzo, J.S.; Godin, B.; Bishop, K.; Srinivasan, S.; Shah, S.K.; Amra, S.; Ramin, S.M.; Ferrari, M. Size of the nanovectors determines the transplacental passage in pregnancy: Study in rats. Am. J. Obstet. Gynecol. 2011, 204, 546.e5–546.e9. [Google Scholar] [CrossRef]
- Grafmueller, S.; Manser, P.; Diener, L.; Diener, P.A.; Maeder-Althaus, X.; Maurizi, L.; Jochum, W.; Krug, H.F.; Buerki-Thurnherr, T.; von Mandach, U.; et al. Bidirectional transfer study of polystyrene nanoparticles across the placental barrier in an ex vivo human placental perfusion model. Environ. Health Perspect. 2015, 123, 1280–1286. [Google Scholar] [CrossRef]
- Kloet, S.K.; Walczak, A.P.; Louisse, J.; van den Berg, H.H.; Bouwmeester, H.; Tromp, P.; Fokkink, R.G.; Rietjens, I.M. Translocation of positively and negatively charged polystyrene nanoparticles in an in vitro placental model. Toxicol. In Vitro 2015, 29, 1701–1710. [Google Scholar] [CrossRef] [PubMed]
- Aengenheister, L.; Dugershaw, B.B.; Manser, P.; Wichser, A.; Schoenenberger, R.; Wick, P.; Hesler, M.; Kohl, Y.; Straskraba, S.; Suter, M.J.; et al. Investigating the accumulation and translocation of titanium dioxide nanoparticles with different surface modifications in static and dynamic human placental transfer models. Eur. J. Pharm. Biopharm. 2019, 142, 488–497. [Google Scholar] [CrossRef]
- Mu, Q.; Jiang, G.; Chen, L.; Zhou, H.; Fourches, D.; Tropsha, A.; Yan, B. Chemical basis of interactions between engineered nanoparticles and biological systems. Chem. Rev. 2014, 114, 7740–7781. [Google Scholar] [CrossRef]
- Wick, P.; Malek, A.; Manser, P.; Meili, D.; Maeder-Althaus, X.; Diener, L.; Diener, P.A.; Zisch, A.; Krug, H.F.; von Mandach, U. Barrier capacity of human placenta for nanosized materials. Environ. Health Perspect. 2010, 118, 432–436. [Google Scholar] [CrossRef]
- Buerki-Thurnherr, T.; von Mandach, U.; Wick, P. Knocking at the door of the unborn child: Engineered nanoparticles at the human placental barrier. Swiss Med. Wkly. 2012, 142, w13559. [Google Scholar] [CrossRef] [PubMed]
- Menezes, V.; Malek, A.; A Keelan, J. Nanoparticulate Drug Delivery in Pregnancy: Placental Passage and Fetal Exposure. Curr. Pharm. Biotechnol. 2011, 12, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Rattanapinyopituk, K.; Shimada, A.; Morita, T.; Sakurai, M.; Asano, A.; Hasegawa, T.; Inoue, K.; Takano, H. Demonstration of the clathrin- and caveolin-mediated endocytosis at the maternal-fetal barrier in mouse placenta after intravenous administration of gold nanoparticles. J. Vet. Med. Sci. 2014, 76, 377–387. [Google Scholar] [CrossRef]
- Ema, M.; Hougaard, K.S.; Kishimoto, A.; Honda, K. Reproductive and developmental toxicity of carbon-based nanomaterials: A literature review. Nanotoxicology 2016, 10, 391–412. [Google Scholar] [CrossRef] [PubMed]
- Jackson, P.; Halappanavar, S.; Hougaard, K.S.; Williams, A.; Madsen, A.M.; Lamson, J.S.; Andersen, O.; Yauk, C.; Wallin, H.; Vogel, U. Maternal inhalation of surface-coated nanosized titanium dioxide (UV-Titan) in C57BL/6 mice: Effects in prenatally exposed offspring on hepatic DNA damage and gene expression. Nanotoxicology 2013, 7, 85–96. [Google Scholar] [CrossRef]
- Balansky, R.; Longobardi, M.; Ganchev, G.; Iltcheva, M.; Nedyalkov, N.; Atanasov, P.; Toshkova, R.; De Flora, S.; Izzotti, A. Transplacental clastogenic and epigenetic effects of gold nanoparticles in mice. Mutat. Res. 2013, 751–752, 42–48. [Google Scholar] [CrossRef]
- Huang, J.P.; Hsieh, P.C.; Chen, C.Y.; Wang, T.Y.; Chen, P.C.; Liu, C.C.; Chen, C.C.; Chen, C.P. Nanoparticles can cross mouse placenta and induce trophoblast apoptosis. Placenta 2015, 36, 1433–1441. [Google Scholar] [CrossRef]
- Zhang, L.; Xie, X.; Zhou, Y.; Yu, D.; Deng, Y.; Ouyang, J.; Yang, B.; Luo, D.; Zhang, D.; Kuang, H. Gestational exposure to titanium dioxide nanoparticles impairs the placentation through dysregulation of vascularization, proliferation and apoptosis in mice. Int. J. Nanomed. 2018, 13, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Awaad, A.; Seleem, A.A. Histochemical changes in neonatal liver caused by vaginal instillation of magnetic nanoparticles in pregnant mice. Biotech. Histochem. 2016, 91, 48–62. [Google Scholar] [CrossRef] [PubMed]
- Fatemi, M.; Moshtaghian, J.; Ghaedi, K.; Jafari Dinani, N.; Naderi, G. Effects of Silver Nanoparticle on the Developing Liver of Rat Pups after Maternal Exposure. Iran. J. Pharm. Res. 2017, 16, 685–693. [Google Scholar] [PubMed]
- Onoda, A.; Umezawa, M.; Takeda, K.; Ihara, T.; Sugamata, M. Effects of maternal exposure to ultrafine carbon black on brain perivascular macrophages and surrounding astrocytes in offspring mice. PLoS ONE 2014, 9, e94336. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Liu, L.; Zhu, W.; Li, D.; Yang, L.; Duan, J.; Cai, Z.; Nie, Y.; Zhang, Y.; Gong, Q.; et al. Iron oxide nanoparticles promote macrophage autophagy and inflammatory response through activation of toll-like receptor-4 signaling. Biomaterials 2019, 203, 23–30. [Google Scholar] [CrossRef]
- Wu, L.; Zhang, Y.; Zhang, C.; Cui, X.; Zhai, S.; Liu, Y.; Li, C.; Zhu, H.; Qu, G.; Jiang, G.; et al. Tuning cell autophagy by diversifying carbon nanotube surface chemistry. ACS Nano 2014, 8, 2087–2099. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Jia, J.; Luo, Z.; Su, G.; Yue, T.; Yan, B. Remote induction of cell autophagy by 2D MoS2 nanosheets via perturbing cell surface receptors and mTOR pathway from outside of cells. ACS Appl. Mater. Interfaces 2019, 11, 6829–6839. [Google Scholar] [CrossRef]
- Zhou, X.; Yan, B. Induction of mTOR-dependent autophagy by WS2 nanosheets from both inside and outside of human cells. Nanoscale 2019, 11, 10684–10694. [Google Scholar] [CrossRef]
- Nakashima, A.; Aoki, A.; Kusabiraki, T.; Shima, T.; Yoshino, O.; Cheng, S.B.; Sharma, S.; Saito, S. Role of autophagy in oocytogenesis, embryogenesis, implantation, and pathophysiology of pre-eclampsia. J. Obstet. Gynaecol. Res. 2017, 43, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Nakashima, A. Review: The role of autophagy in extravillous trophoblast function under hypoxia. Placenta 2013, 34, S79–S84. [Google Scholar] [CrossRef]
- Nakashima, A.; Yamanaka-Tatematsu, M.; Fujita, N.; Koizumi, K.; Shima, T.; Yoshida, T.; Nikaido, T.; Okamoto, A.; Yoshimori, T.; Saito, S. Impaired autophagy by soluble endoglin, under physiological hypoxia in early pregnant period, is involved in poor placentation in preeclampsia. Autophagy 2013, 9, 303–316. [Google Scholar] [CrossRef]
- Saito, S.; Nakashima, A. A review of the mechanism for poor placentation in early-onset preeclampsia: The role of autophagy in trophoblast invasion and vascular remodeling. J. Reprod. Immunol. 2014, 101–102, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.; Zou, Z.; Sun, Q.; Luby-Phelps, K.; Cheng, P.; Hogan, R.N.; Gilpin, C.; Levine, B. Autophagy gene-dependent clearance of apoptotic cells during embryonic development. Cell 2007, 128, 931–946. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, S.; Kuma, A.; Murakami, M.; Kishi, C.; Yamamoto, A.; Mizushima, N. Autophagy is essential for preimplantation development of mouse embryos. Science 2008, 321, 117–120. [Google Scholar] [CrossRef]
- Gao, L.; Qi, H.B.; Kamana, K.C.; Zhang, X.M.; Zhang, H.; Baker, P.N. Excessive autophagy induces the failure of trophoblast invasion and vasculature: Possible relevance to the pathogenesis of preeclampsia. J. Hypertens. 2015, 33, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, V.; Jaiswal, M.K.; Mallers, T.; Katara, G.K.; Gilman-Sachs, A.; Beaman, K.D.; Hirsch, E. Altered autophagic flux enhances inflammatory responses during inflammation-induced preterm labor. Sci. Rep. 2015, 5, 9410. [Google Scholar] [CrossRef]
- Nakashima, A.; Higashisaka, K.; Kusabiraki, T.; Aoki, A.; Ushijima, A.; Ono, Y.; Tsuda, S.; Shima, T.; Yoshino, O.; Nagano, K.; et al. Autophagy is a new protective mechanism against the cytotoxicity of platinum nanoparticles in human trophoblasts. Sci. Rep. 2019, 9, 5478. [Google Scholar] [CrossRef]
- Broad, K.D.; Keverne, E.B. Placental protection of the fetal brain during short-term food deprivation. Proc. Natl. Acad. Sci. USA 2011, 108, 15237–15241. [Google Scholar] [CrossRef] [PubMed]
- Avagliano, L.; Danti, L.; Doi, P.; Felis, S.; Guala, M.; Locatelli, A.; Maffeo, I.; Mecacci, F.; Plevani, C.; Simeone, S.; et al. Autophagy in placentas from acidotic newborns: An immunohistochemical study of LC3 expression. Placenta 2013, 34, 1091–1094. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, B.; Yao, M.; Dong, T.; Mao, Z.; Hang, B.; Han, X.; Lin, Z.; Bian, Q.; Li, M.; et al. Titanium dioxide nanoparticles induce proteostasis disruption and autophagy in human trophoblast cells. Chem. Biol. Interact. 2018, 296, 124–133. [Google Scholar] [CrossRef]
- Mao, Z.; Yao, M.; Li, Y.; Fu, Z.; Li, S.; Zhang, L.; Zhou, Z.; Tang, Q.; Han, X.; Xia, Y. MiR-96-5p and miR-101-3p as potential intervention targets to rescue TiO2 NP-induced autophagy and migration impairment of human trophoblastic cells. Biomater. Sci. 2018, 6, 3273–3283. [Google Scholar] [CrossRef]
- Goeden, N.; Velasquez, J.; Arnold, K.A.; Chan, Y.; Lund, B.T.; Anderson, G.M.; Bonnin, A. Maternal inflammation disrupts fetal neurodevelopment via increased placental output of serotonin to the fetal brain. J. Neurosci. 2016, 36, 6041–6049. [Google Scholar] [CrossRef]
- Kucki, M.; Aengenheister, L.; Diener, L.; Rippl, A.V.; Vranic, S.; Newman, L.; Vazquez, E.; Kostarelos, K.; Wick, P.; Buerki-Thurnherr, T. Impact of graphene oxide on human placental trophoblast viability, functionality and barrier integrity. 2D Mater. 2018, 5, 035014. [Google Scholar] [CrossRef]
- Juch, H.; Nikitina, L.; Reimann, S.; Gauster, M.; Dohr, G.; Obermayer-Pietsch, B.; Hoch, D.; Kornmueller, K.; Haag, R. Dendritic polyglycerol nanoparticles show charge dependent bio-distribution in early human placental explants and reduce hCG secretion. Nanotoxicology 2018, 12, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, P.A.; Minarchick, V.C.; Yi, J.; Engels, K.; McBride, C.R.; Nurkiewicz, T.R. Maternal engineered nanomaterial exposure and fetal microvascular function: Does the Barker hypothesis apply? Am. J. Obstet. Gynecol. 2013, 209, 227.e1–227.e11. [Google Scholar] [CrossRef] [PubMed]
- Koga, K.; Izumi, G.; Mor, G.; Fujii, T.; Osuga, Y. Toll-like receptors at the maternal-fetal interface in normal pregnancy and pregnancy complications. Am. J. Reprod. Immunol. 2014, 72, 192–205. [Google Scholar] [CrossRef]
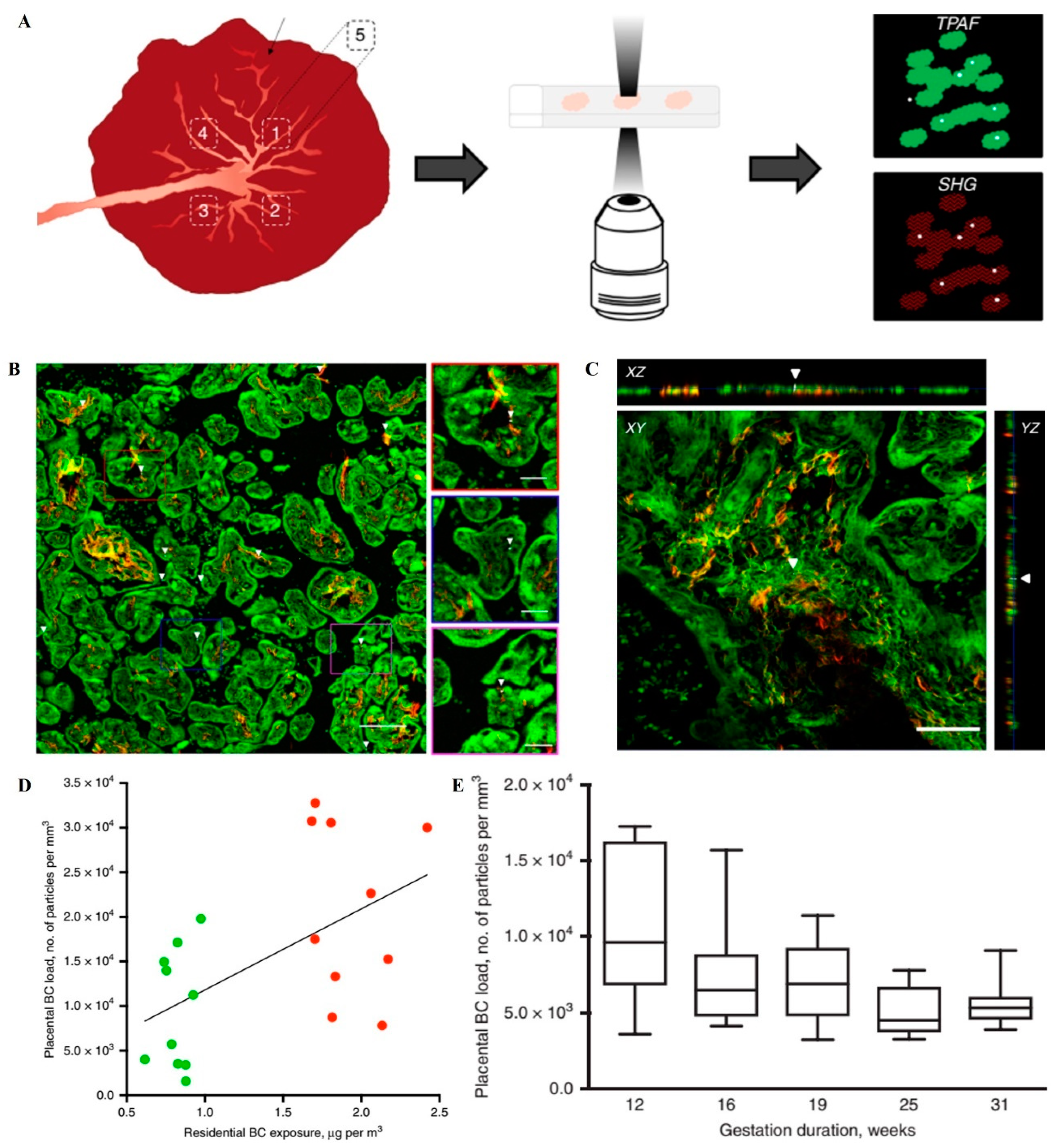
| NPs | Animals/Exposure Route | Fetotoxicities | Ref. |
|---|---|---|---|
| CeO2 NPs 3–5 nm | Mouse, i.v., 5 mg/kg, GD5, 6, and 7 | Decreased number and pups’ weight; increased fetal resorption rate | [10] |
| ZnO NPs 13 nm | Mouse, i.g., GD7-GD16 | Intrauterine growth retardation | [11] |
| Ultrafine particles | Mouse, intratracheal instillation, 400 μg/kg, GD7, 9, 11, 15 and 17 | Embryo reabsorption, decreased fetal weight and altered blood pressure in the offspring | [12] |
| QDs | Rat, inhalation exposure, 5,1 nmol/rat, GD5–GD19 | Growth restriction in offspring | [13] |
| Ag NPs 18–20 nm | Mouse, intranasal instillation, 640 μg/m3, GD1–GD15 | Increased number of resorbed fetuses | [14] |
| CdSe/ZnS QDs, 20 nm CdSe QDs, 15 nm | Mouse, i.v., 0.1 nmol/mouse, GD17 and 18 | Fetus malformation, hampered growth | [15] |
| SiO2 NPs, 70 nm TiO2 NPs, 35 nm | Mouse, i.v., SiO2 NPs: 0.2, 0.4, 0.8 mg/mouse, TiO2 NPs: 0.8 mg/mouse, GD16 and 17 | Smaller fetuses | [16] |
| p,o,uo-SWCNTs | Mouse, the retrobulbar injection, 0.01~30 μg/mouse, GD6 | Retarded limbs and snout development | [17] |
| Diesel engine exhaust (DEP), 69 nm | Rabbit, nose-only inhalation, 2 h/day, 5 days/week, GD3–GD27 | Growth retardation at mid gestation with decreased head length and umbilical pulse | [41] |
| NP-enriched DE, 22–27 nm | Rat, intranasal instillation, 148.86 μg/m3, 3.10 μg/m3, 5 h/day, GD1–GD19 | Increased fetal weight and decreased crown-rump length | [42] |
| CB NPs 14 nm | Mouse, intratracheal instillation, 11, 54, 268 μg/mouse, GD7, 10, 15 and 18 | Induced more DNA strand breaks in the liver of their offspring | [43] |
| MWCNTs | Mouse, i.p. or intratracheal instillation, 2, 3, 4, 5 mg/kg, GD9 | Increased the number of external malformation and skeletal malformation in fetuses | [44] |
| SWCNTs | Rat, intratracheal instillation or i.v., 100 μg/kg, GD17–GD19 | Induced vasoconstriction and reduced fetal growth | [45] |
| rGO | Mouse, i.v., 6.25, 12.5, 25 mg/kg, GD6 or 20 | Caused malformation in fetuses | [46] |
| ZnO NPs 30 nm | Mouse, i.g., 20, 60, 180, 540 mg/kg, GD11–GD18 | Fetal growth retardation, decreased fetal number | [47] |
| ZnO 13, 57 or 1900 nm | Mouse, i.g., GD7–GD16 | Decreased birth weight | [48] |
| ZnO NPs 44.2 nm | Rat, i.v., 5, 10, 20 mg/kg, GD7–GD21 | Increased the number of dead fetuses and decreased fetal weight | [49] |
| ZnO NPs 20 nm | Mouse, i.g., 100, 200, 400 mg/kg, GD5–GD19 | Significant decrease in fetal weight for 400 mg/kg exposure group | [50] |
| ZnO NPs 35 nm | Rat, i.g., 500 mg/kg, 2 weeks before mating to PND 4 | Reduced fetal weight and increased fetal resorption of pups | [51] |
| TiO2 NPs 21 nm | Rat, inhalation exposure, 12 mg/m3, 6 h/exposure, 6 days, GD11–GD16 | Significantly decreased pup’s weight and placental efficiency | [52] |
| TiO2 NPs 6.5 nm | Mouse, i.g., 25, 50, 100 mg/kg, GD1–GD18 | Inhibited the crown-rump length, fetal weight, the number of live fetuses and fetal skeleton development | [53] |
| SiO2 NPs 25, 60, 115 nm | Mouse, i.v., 3, 30, 200 μg/mouse, GD6, 13 and 17 | Decreased the resorbed number at the dose of 200 μg/mouse for 25 nm NPs | [54] |
| CdO NPs 11–15 nm | Mouse, inhalation exposure, 100 μg/m3, every other day or 230 μg/m3 daily, 2.5 h/day, GD5–GD17 | Decreased incidence of pregnancy, fetal length, and neonatal growth | [55] |
| Ag NPs 20, 110 nm | Rat, i.v., 200 μg/rat, GD17–GD19 | Fetal growth restriction | [56] |
| Ag NP 10 nm | Mouse, i.v., 66 μg/mouse, GD8–GD10 | Embryonic growth restriction | [57] |
| Au NPs 2, 15, 50 nm | Mouse, i.v., 2 mg/kg, or 0.5–10 mg/kg, GD4–GD6 | Disturb embryonic development in a size- and concentration-dependent manner. | [58] |
| ZnO NPs SiO2 NPs | Mouse, i.g., ZnO NPs: 50, 100, 300 mg/kg, SiO2 NPs: 50, 100 mg/kg, GD5–GD19 | Miscarriages and adversely affected the developing fetus | [59] |
| PEI-Fe2O3-NPs, 28 nm PAA-Fe2O3-NPs, 30 nm | Mouse, i.p., 10, 100 mg/kg, GD9, 10 and 11 | High dose exposure led to charge-dependent fetal loss, morphological alterations in uteri | [60] |
| TiO2 NPs 20 nm | Rat, inhalation exposure, 10 mg/m3, 6 h/exposure, 6 days, GD5–GD19 | Altered fetal epigenome | [61] |
| QDs 1.67, 2.59 or 3.21 nm | Mouse, i.p., 5, 10, 20 mg/kg, GD14 | Decreased survival rate, body length, body mass and disturbed ossification of limbs | [62] |
| Fe2O3 NPs 28–30 nm | Mouse, i.p., 10 mg/kg, GD10–GD17 | Increased fetal death | [63] |
| SiO2 NPs 70 nm | Mouse, i.v., 25, 40 mg/kg, GD13–GD14 | Pregnancy complications | [64] |
| o-MWCNTs | Mouse, i.v., 20 mg/kg, GD4, 11 and 15 | Induced maternal body weight gain and abortion rates dependent on pregnancy times | [65] |
| CNTs | Mouse, i.v., 10 μg/mouse, GD6 and 15 | Occasional teratogenic effects | [66] |
| fCNTs | Mouse, i.g., 10 mg/kg, GD9 | Increased the number of resorbed fetuses; fetal morphological and skeletal abnormalities | [67] |
| TiO2 NPs Ag NPs | Mouse, i.g., 10, 100, 1000 mg/kg, GD9 | Increase fetal mortality | [68] |
| DEPs | Mouse, i.g., 31.25, 62.5, 125, 250, 500 mg/kg, GD11–GD16 | Increased the frequency of DNA deletions in fetus and offspring | [69] |
| NPs | Animals/Exposure Route | Neurotoxicity | Ref. |
|---|---|---|---|
| TiO2 NPs 6.5 nm | Mouse, i.g., 1.25, 2.5, 5 mg/kg, GD7–PND21 | Retarded axonal and dendritic outgrowth | [19] |
| DE | Mouse, s.c., 0.5, 1 mg/mL, GD5, 8, 11, 14 and 17 | Increased glial-fibrillary acidic protein level in the corpus callosum and cortex | [20] |
| CoCr NPs | Mouse, i.v., 0.12 mg/mouse, GD10 and 13 | Neurodevelopmental abnormalities with reactive astrogliosis and increased DNA damage in fetal hippocamus | [21] |
| CB NPs | Mouse, intranasal instillation, 2.9, 15, 73 μg/kg, GD5 and 9 | Reactive astrogliosis | [22] |
| TiO2 NPs 25–70 nm | Mouse, s.c., 1 μg/μL, 100 μL, GD7, 10, 13 and 16 | Changed gene expression related to neurotransmitters and psychiatric diseases in newborns | [23] |
| SWCNTs | Mouse, i.v., 2 mg/kg, GD11, 13, and 16 | Obvious brain deformity | [70] |
| TiO2 NPs 21 nm | Mouse, inhalation exposure, 42 mg/m3, 1 h/day, GD8–GD18 | Moderate neurobehavioral alterations in offspring mice | [74] |
| CB NPs 100–300 nm | Mouse, airway instillation, 0, 4.6, 37 mg/m3, 15 days, GD4–GD18 | Denaturation of perivascular macrophages and reactive astrocytes | [82] |
| CB NPs 84 nm | Mouse, intranasal instillation, 190 μg/kg, GD5 and 7 | Gene dysfunction in the frontal cortex in offspring mice | [83] |
| CB NPs 84.2 nm | Mouse, intranasal instillation, 95 μg/kg, GD5 and 9 | Astrogliosis in the offspring brain | [84] |
| ZnO NPs 30 nm | Rat, ig., 500 mg/kg, GD2–GD19 | Learning and memory impairment in the offspring brain | [85] |
| TiO2 NPs | Mouse, i.v., 100, 1000 μg, every second day, GD9 | Autism spectrum disorder-related behavioral deficits in the offspring | [86] |
| TiO2 NPs 10 nm | Rat, i.g., 100 mg/kg, GD2–GD21 | Impaired memory and decreased hippocampal cell proliferation in rat offspring | [88] |
| TiO2 NPs 5 nm | Rat, s.c., 1 μg/μL, 500 μL, GD7, 10, 13, 16 and 19 | Oxidative damage in the brain of newborn pups, and the depressive-like behaviors during adulthood | [89] |
| Ag NPs 10 nm | Mouse, s.c., 0.2, 2 mg/kg, once every three days, GD1–GD21 | Gender-specific depression-like behaviors in offspring | [90] |
| Ultrafine particles | Mouse, airway instillation, 92.69 μg/m3, 6 h/day, GD1–GD17 | Neurodevelopmental disorders in offspring | [91] |
| TiO2 NPs 6.5 nm | Rat, i.g., 100 mg/kg, GD2–GD21, PND2–PND21 | Impacted hippocampal neurogenesis and apoptosis in the offspring | [92] |
| TiO2 NPs 6.5 nm | Mouse, i.g., 1, 2, 3 mg/kg, GD1–PND21 | Inhibited dendritic outgrowth of hippocampal neurons in the offspring mice | [93] |
| ZnO NPs 20–40 nm | Mouse, s.c., 4 h/day, PND4–PND7, PND10–PND13 | Decreased ambulation score, hindlimb suspension score and degree of grip strength; increased degree of hindlimb foot angle | [94] |
| ZnO NPs 20–40 nm | Mouse, s.c., 0.5, 1 mg/mL, GD5, 8, 11, 14 and 17 | Depressive-like behaviors in offspring | [95] |
| TiO2 NPs 20 nm | Mouse, i.p., 2 mg/mL, GD11– GD16 | Decreased size and weight of fetus, a disrupted anatomical structure of the fetal brain, bulkier and abnormal shape of fetal liver | [96] |
| TiO2 NPs 170.9 nm | Rat, airway instillation, 10.4 mg/m3, 5 h/day, 4 days/week, GD7–GD20 | Induced psychological deficits in male adulthood rat | [97] |
| Ag NPs 10 nm | Mouse, s.c., 0.2, 2 mg/kg, once every three days, GD1–GD21 | Neurobehavioral disorders in the offspring | [98] |
| USPIO NPs | Mouse, i.v., 6.25, 12.5, 25 mg/kg, GD6 or 20 | Abnormal fetal neurodevelopment | [99] |
| NPs | Animals/Exposure Route | Fetotoxicities | Ref. |
|---|---|---|---|
| CB NPs 14 nm | Mouse, intranasal instillation, 200 μg/kg, GD8, GD15 | Alteration in reproductive function of male offspring | [25] |
| CB NPs 14 nm; | Mouse, intratracheal instillation, 268 μg/mouse, GD7, 10, 15 and 18 | Lowered sperm production | [28] |
| TiO2 NPs 21 nm | Mouse, intranasal instillation, 42 mg/m3, 1 h/day, 840 μg/mouse, GD8–GD18 | Lowered sperm production | [28] |
| SiO2 NPs ZnO NPs | Mouse, i.g., ZnO NPs: 0,50,100,300 mg/kg, SiO2 NPs: 0,50,250 mg/kg, GD15–GD19 | Prominent epithelial vacuolization, decreased seminiferous tubule diameter in testis | [59] |
| PEI-NPs 28 nm PAA-NPs 30 nm | Mouse, i.p., 10, 100 mg/kg, GD9, 10 and 11 | charge-dependent fetal loss, morphological alterations in uteri and testes of offspring | [60] |
| Asian sand dust | Mouse, intratracheal instillation, 200 μg/mouse, GD8 and 15 | Partial vacuolation of seminiferous tubules and low DSP in immature offspring | [104] |
| Nanoparticle-rich DE | Rat, inhalation exposure, 148.86 g/m3, 1.83 × 106 particles/cm3, GD2–GD20 | Endocrine disruption after birth and suppression in testicular function | [105] |
| TiO2 NPs 35 nm | Mouse, s.c., 0.5, 5, 50, 500 μg/mouse, GD5, 8, 11, 14 and 17 | A dose-dependent increase in the number of agglomerates in the offspring testes | [106] |
| TiO2 NPs 25–70 nm | Mouse, s.c., 1 mg/mL, 100 μL, GD3, 7, 10 and 14 | Decreased daily sperm production in offspring | [107] |
| NPs | Animals/Exposure Route | Fetotoxicities | Ref. |
|---|---|---|---|
| PMs | Mouse, oropharyngeal aspiration, 3 mg/kg, GD10, and 17 | Inhibition of the development of pulmonary T helper and T regulatory cells of the infant offspring | [29] |
| PMs | Mouse, intranasal instillation, 95 μg/kg, GD14, 16, and 18 | Inhibition of splenic T cell maturation in male offspring and alteration in early life immune development in a sex specific manner | [30] |
| Cu NPs | Mouse, inhalation exposure, 3.5 mg/m3, 4 h/day, GD4–GD20 | Altered expression of several Th1/Th2 or other immune response genes in pups’ spleens | [31] |
| CB NPs 14 nm | Mouse, intranasal instillation, 95 μg/kg, GD9 and 15 | Allergic or inflammatory effects in male offspring | [32] |
| PM2.5 | Moue, inhalation exposure, 8 h/day, 6 days/week, 16 weeks | Alteration in immune microenvironment | [109] |
| DEPs 14 nm | Mouse, intranasal instillation, 50 μg/mouse, GD14 | Increased allergic susceptibility in offspring | [110] |
| CB NPs 14 nm | Mouse, intranasal instillation, 95 μg/kg, GD9 and 15 | Suppressed development of immune system of the offspring mice | [111] |
| DEPs | Mouse, i.g., 31.25, 62.5, 125, 250, 500 mg/kg, GD11–GD16 | Increase in the frequency of DNA deletions in the mouse fetus and such genetic alterations in the offspring | [69] |
| DEPs | Mouse, intranasal instillation, 50 μg/mouse, GD14–GD15 | Triggered transgenerational transmission of asthma risk | [112] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, C.; Jiang, C.; Gao, S.; Liu, X.; Zhai, S. Fetotoxicity of Nanoparticles: Causes and Mechanisms. Nanomaterials 2021, 11, 791. https://doi.org/10.3390/nano11030791
Teng C, Jiang C, Gao S, Liu X, Zhai S. Fetotoxicity of Nanoparticles: Causes and Mechanisms. Nanomaterials. 2021; 11(3):791. https://doi.org/10.3390/nano11030791
Chicago/Turabian StyleTeng, Chuanfeng, Cuijuan Jiang, Sulian Gao, Xiaojing Liu, and Shumei Zhai. 2021. "Fetotoxicity of Nanoparticles: Causes and Mechanisms" Nanomaterials 11, no. 3: 791. https://doi.org/10.3390/nano11030791
APA StyleTeng, C., Jiang, C., Gao, S., Liu, X., & Zhai, S. (2021). Fetotoxicity of Nanoparticles: Causes and Mechanisms. Nanomaterials, 11(3), 791. https://doi.org/10.3390/nano11030791






