Recrystallization of CsPbBr3 Nanoparticles in Fluoropolymer Nonwoven Mats for Down- and Up-Conversion of Light
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Perovskite Nanoparticles
2.2. Synthesis of Nonwoven Mats with Perovskite Nanoparticles
2.3. Microscopic and Optical Characterization
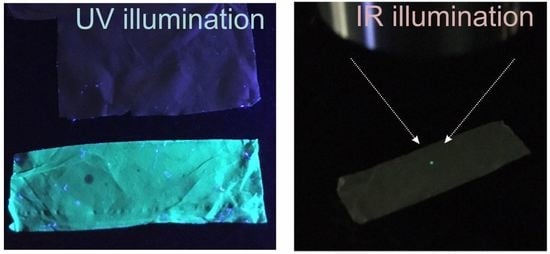
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sutherland, B.R.; Sargent, E.H. Perovskite photonic sources. Nat. Photonics 2016, 10, 295. [Google Scholar] [CrossRef]
- Makarov, S.; Furasova, A.; Tiguntseva, E.; Hemmetter, A.; Berestennikov, A.; Pushkarev, A.; Zakhidov, A.; Kivshar, Y. Halide-Perovskite Resonant Nanophotonics. Adv. Opt. Mater. 2019, 7, 1800784. [Google Scholar] [CrossRef]
- Green, M.A.; Ho-Baillie, A.; Snaith, H.J. The emergence of perovskite solar cells. Nat. Photonics 2014, 8, 506–514. [Google Scholar] [CrossRef]
- Lu, M.; Zhang, Y.; Wang, S.; Guo, J.; Yu, W.W.; Rogach, A.L. Metal halide perovskite light-emitting devices: Promising technology for next-generation displays. Adv. Funct. Mater. 2019, 29, 1902008. [Google Scholar] [CrossRef]
- Polushkin, A.S.; Tiguntseva, E.Y.; Pushkarev, A.P.; Makarov, S.V. Single-particle perovskite lasers: From material properties to cavity design. Nanophotonics 2020, 9, 599–610. [Google Scholar] [CrossRef]
- Luo, W.; Yan, L.; Liu, R.; Zou, T.; Zhang, S.; Liu, C.; Dai, Q.; Chen, J.; Zhou, H. High detectivity ITO/organolead halide perovskite Schottky photodiodes. Semicond. Sci. Technol. 2019, 34, 074004. [Google Scholar] [CrossRef]
- Soranyel, G.C.; Galian, R.E.; Pérez-Prieto, J. Maximizing the emissive properties of CH3NH3PbBr3 perovskite nanoparticles. J. Mater. Chem. A 2015, 3, 39187–39193. [Google Scholar]
- Ahmadi, M.; Wu, T.; Hu, B. A Review on Organic–Inorganic Halide Perovskite Photodetectors: Device Engineering and Fundamental Physics. Adv. Mater. 2017, 29, 1605242. [Google Scholar] [CrossRef]
- Zhang, F.; Zhong, H.; Chen, C.; Wu, X.-G.; Hu, X.; Huang, H.; Han, J.; Zou, B.; Dong, Y. Brightly Luminescent and Color-Tunable Colloidal CH3NH3PbX3 (X = Br, I, Cl) Quantum Dots: Potential Alternatives for Display Technology. Acs Nano 2015, 9, 4533–4542. [Google Scholar] [CrossRef]
- Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Krieg, F.; Caputo, R.; Hendon, C.H.; Yang, R.X.; Walsh, A.; Kovalenko, M.V. Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, and I): Novel Optoelectronic Materials Showing Bright Emission with Wide Color Gamut. Nano Lett. 2015, 15, 3692. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Susha, A.S.; Kershaw, S.V.; Hung, T.F.; Rogach, A.L. Control of emission color of high quantum yield CH3NH3PbBr3 perovskite quantum dots by precipitation temperature. Adv. Sci. 2015, 2, 1500194. [Google Scholar] [CrossRef] [PubMed]
- Berestennikov, A.S.; Li, Y.; Iorsh, I.V.; Zakhidov, A.A.; Rogach, A.L.; Makarov, S.V. Beyond quantum confinement: Excitonic nonlocality in halide perovskite nanoparticles with Mie resonances. Nanoscale 2019, 11, 6747–6754. [Google Scholar] [CrossRef]
- Choi, S.; Lee, H.; Ghaffari, R.; Hyeon, T.; Kim, D.-H. Recent Advances in Flexible and Stretchable Bio-Electronic Devices Integrated with Nanomaterials. Adv. Mater. 2016, 28, 4203. [Google Scholar] [CrossRef]
- Kim, D.-H.; Lu, N.; Ma, R.; Kim, Y.-S.; Kim, R.-H.; Wang, S.; Wu, J.; Won, S.M.; Tao, H.; Islam, A.; et al. Epidermal electronics. Science 2011, 333, 838. [Google Scholar] [CrossRef] [PubMed]
- Rogers, J.A.; Someya, T.; Huang, Y. Materials and mechanics for stretchable electronics. Science 2010, 327, 1603. [Google Scholar] [CrossRef]
- Choi, M.K.; Yang, J.; Kang, K.; Kim, D.C.; Choi, C.; Park, C.; Kim, S.J.; Chae, S.I.; Kim, T.-H.; Kim, J.H.; et al. Wearable red-green-blue quantum dot light-emitting diode array using high-resolution intaglio transfer printing. Nat. Commun. 2015, 6, 7149. [Google Scholar] [CrossRef]
- Kim, T.-H.; Lee, C.-S.; Kim, S.; Hur, J.; Lee, S.; Shin, K.W.; Yoon, Y.-Z.; Choi, M.K.; Yang, J.; Kim, D.-H.; et al. Fully Stretchable Optoelectronic Sensors Based on Colloidal Quantum Dots for Sensing Photoplethysmographic Signals. Acs Nano 2017, 11, 5992. [Google Scholar] [CrossRef]
- Son, D.; Chae, S.I.; Kim, M.; Choi, M.K.; Yang, J.; Park, K.; Kale, V.S.; Koo, J.H.; Choi, C.; Lee, M.; et al. Colloidal Synthesis of Uniform-Sized Molybdenum Disulfide Nanosheets for Wafer-Scale Flexible Nonvolatile Memory. Adv. Mater. 2016, 28, 9326. [Google Scholar] [CrossRef] [PubMed]
- Song, J.-K.; Son, D.; Kim, J.; Yoo, Y.J.; Lee, G.J.; Wang, L.; Choi, M.K.; Yang, J.; Lee, M.; Do, K.; et al. Wearable Force Touch Sensor Array Using a Flexible and Transparent Electrode. Adv. Funct. Mater. 2017, 27, 1605286. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, Y.; Huang, J.; Cai, J.; Zhu, J.; Yang, X.; Shen, J.; Li, C. Perovskite quantum dots encapsulated in electrospun fiber membranes as multifunctional supersensitive sensors for biomolecules, metal ions and pH. Nanoscale Horiz. 2017, 2, 225. [Google Scholar] [CrossRef]
- Xue, J.; Wu, T.; Dai, Y.; Xia, Y. Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications. Chem. Rev. 2019, 119, 5298. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.C.F. A Review of the Development of Basketry, Braiding, Crochet, and Felting towards the Creation of Seamless Fashion. Res. J. Text. Appar. 2004, 8, 65. [Google Scholar] [CrossRef]
- Komova, N.; Pavlov, A.M.; Serdobintsev, A.A.; Galushka, V.V.; Rusanova, T.Y. SERS-platforms based on electrospun nanofibers with embedded silver nanoparticles. In Saratov Fall Meeting 2018: Computations and Data Analysis: From Nanoscale Tools to Brain Functions; Postnov, D.E., Ed.; SPIE: Bellingham, WA, USA, 2019; p. 134. [Google Scholar]
- Tsai, P.C.; Chen, J.Y.; Ercan, E.; Chueh, C.C.; Tung, S.H.; Chen, W.C. Uniform Luminous Perovskite Nanofibers with Color-Tunability and Improved Stability Prepared by One-Step Core/Shell Electrospinning. Small 2018, 14, 1704379. [Google Scholar] [CrossRef]
- Ni, D.J.; Zhang, J.; Cao, Z.K.; Li, R.; Xu, T.F.; Sang, H.W.; Long, Y.Z. Supersensitive and reusable perovskite nanocomposite fiber paper for time-resolved single-droplet detection. J. Hazard. Mater. 2021, 403, 123959. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Fu, H.; Liao, H.; Liang, Z.; Ye, Y.; Zheng, J.; Yang, W. In situ synthesis of coaxial CsPbX 3@ polymer (X= Cl, Br, I) fibers with significantly enhanced water stability. J. Mater. Chem. C 2020, 8, 13972–13975. [Google Scholar] [CrossRef]
- Zhang, H.; Fu, D.; Du, Z.; Fu, H.; Shao, G.; Yang, W.; Zheng, J. In situ growth of aligned CsPbBr3 nanorods in polymer fibers with tailored aspect ratios. Ceram. Int. 2020, 46, 18352–18357. [Google Scholar] [CrossRef]
- Jiang, D.H.; Kobayashi, S.; Jao, C.C.; Mato, Y.; Isono, T.; Fang, Y.H.; Kuo, C.C. Light down-converter based on luminescent nanofibers from the blending of conjugated rod-coil block copolymers and perovskite through electrospinning. Polymers 2020, 12, 84. [Google Scholar] [CrossRef]
- Jiang, D.H.; Tsai, Y.H.; Veeramuthu, L.; Liang, F.C.; Chen, L.C.; Lin, C.C.; Kuo, C.C. Novel ultra-stable and highly luminescent white light-emitting diodes from perovskite quantum dots—Polymer nanofibers through biaxial electrospinning. APL Mater. 2019, 7, 111105. [Google Scholar] [CrossRef]
- Cha, J.H.; Kim, H.; Lee, Y.; Kim, S.J.; Lee, M.W.; Kim, J.; Jung, D.Y. Nanoscale optical imaging of perovskite nanocrystals directly embedded in polymer fiber. Compos. Sci. Technol. 2019, 181, 107666. [Google Scholar] [CrossRef]
- Papagiorgis, P.G.; Manoli, A.; Alexiou, A.; Karacosta, P.; Karagiorgis, X.; Papaparaskeva, G.; Itskos, G. Robust hydrophobic and hydrophilic polymer fibers sensitized by inorganic and hybrid lead halide perovskite nanocrystal emitters. Front. Chem. 2019, 7, 87. [Google Scholar] [CrossRef]
- Liao, H.; Guo, S.; Cao, S.; Wang, L.; Gao, F.; Yang, Z.; Yang, W. A General Strategy for In Situ Growth of All-Inorganic CsPbX3 (X= Br, I, and Cl) Perovskite Nanocrystals in Polymer Fibers toward Significantly Enhanced Water/Thermal Stabilities. Adv. Opt. Mater. 2019, 6, 1800346. [Google Scholar] [CrossRef]
- Cardoso, V.; Correia, D.; Ribeiro, C.; Fernandes, M.; Lanceros-Méndez, S. Fluorinated Polymers as Smart Materials for Advanced Biomedical Applications. Polymers 2018, 10, 161. [Google Scholar] [CrossRef] [PubMed]
- Hühn, J.; Carrillo-Carrion, C.; Soliman, M.G.; Pfeiffer, C.; Valdeperez, D.; Masood, A.; Chakraborty, I.; Zhu, L.; Gallego, M.; Yue, Z.; et al. Selected Standard Protocols for the Synthesis, Phase Transfer, and Characterization of Inorganic Colloidal Nanoparticles. Chem. Mater. 2017, 29, 399–461. [Google Scholar]
- Prescher, C.; Prakapenka, V.B. DIOPTAS: A program for reduction of two-dimensional X-ray diffraction data and data exploration. High Press. Res. 2015, 35, 223–230. [Google Scholar] [CrossRef]
- Kubin, R.F.; Fletcher, A.N. Fluorescence quantum yields of some rhodamine dyes. J. Lumin. 1982, 27, 455–462. [Google Scholar] [CrossRef]
- Magde, D.; Wong, R.; Seybold, P.G. Fluorescence quantum yields and their relation to lifetimes of rhodamine 6G and fluorescein in nine solvents: Improved absolute standards for quantum yields. Photochem. Photobiol. 2002, 75, 327–334. [Google Scholar] [CrossRef]
- Dirin, D.N.; Cherniukh, I.; Yakunin, S.; Shynkarenko, Y.; Kovalenko, M.V. Solution-grown CsPbBr3 perovskite single crystals for photon detection. Chem. Mater. 2016, 28, 8470–8474. [Google Scholar] [CrossRef]
- Zhang, M.; Zheng, Z.; Fu, Q.; Chen, Z.; He, J.; Zhang, S.; Luo, W. Synthesis and single crystal growth of perovskite semiconductor CsPbBr3. J. Cryst. Growth 2017, 484, 37–42. [Google Scholar] [CrossRef]
- He, X.; Qiu, Y.; Yang, S. Fully-inorganic trihalide perovskite nanocrystals: A new research frontier of optoelectronic materials. Adv. Mater. 2018, 29, 1700775. [Google Scholar] [CrossRef]
- Cottingham, P.; Brutchey, R.L. On the crystal structure of colloidally prepared CsPbBr 3 quantum dots. Chem. Commun. 2016, 52, 5246–5249. [Google Scholar] [CrossRef]
- Lu, Z.; Li, Y.; Qiu, W.; Rogach, A.L.; Nagl, S. Composite Films of CsPbBr3 Perovskite Nanocrystals in a Hydrophobic Fluoropolymer for Temperature Imaging in Digital Microfluidics. ACS Appl. Mater. Interfaces 2020, 12, 19805–19812. [Google Scholar] [CrossRef]
- Li, Y.; Lv, Y.; Guo, Z.; Dong, L.; Zheng, J.; Chai, C.; Chen, N.; Lu, Y.; Chen, C. One-Step Preparation of Long-Term Stable and Flexible CsPbBr3 Perovskite Quantum Dots/Ethylene Vinyl Acetate Copolymer Composite Films for White Light-Emitting Diodes. ACS Appl. Mater. Interfaces 2018, 10, 15888–15894. [Google Scholar] [CrossRef]
- Manzi, A.; Tong, Y.; Feucht, J.; Yao, E.P.; Polavarapu, L.; Urban, A.S.; Feldmann, J. Resonantly enhanced multiple exciton generation through below-band-gap multi-photon absorption in perovskite nanocrystals. Nat. Commun. 2018, 9, 1–6. [Google Scholar] [CrossRef]
- Turren-Cruz, S.H.; Saliba, M.; Mayer, T.M.; Jua’rez-Santiesteban, H.; Mathew, X.; Nienhaus, L.; Tress, W.; Erodici, P.M.; Sher, J.M.; Bawendi, G.M.; et al. Enhanced charge carrier mobility and lifetime suppress hysteresis and improve efficiency in planar perovskite solar cells. Energy Environ. Sci. 2018, 11, 78–86. [Google Scholar] [CrossRef]
- Wehrenfennig, C.; Eperon, E.G.; Johnston, B.M.; Snaith, H.J.; Herz, M.L. High charge carrier mobilities and lifetimes in organolead trihalide perovskites. Adv. Mater. 2014, 26, 1584–1589. [Google Scholar] [CrossRef]
- Ščajev, P.; Miasojedovas, S.; Juršėnas, S. A carrier density dependent diffusion coefficient, recombination rate and diffusion length in MAPbI 3 and MAPbBr 3 crystals measured under one-and two-photon excitations. J. Mater. Chem. C 2020, 8, 10290–10301. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neplokh, V.; Markina, D.I.; Baeva, M.; Pavlov, A.M.; Kirilenko, D.A.; Mukhin, I.S.; Pushkarev, A.P.; Makarov, S.V.; Serdobintsev, A.A. Recrystallization of CsPbBr3 Nanoparticles in Fluoropolymer Nonwoven Mats for Down- and Up-Conversion of Light. Nanomaterials 2021, 11, 412. https://doi.org/10.3390/nano11020412
Neplokh V, Markina DI, Baeva M, Pavlov AM, Kirilenko DA, Mukhin IS, Pushkarev AP, Makarov SV, Serdobintsev AA. Recrystallization of CsPbBr3 Nanoparticles in Fluoropolymer Nonwoven Mats for Down- and Up-Conversion of Light. Nanomaterials. 2021; 11(2):412. https://doi.org/10.3390/nano11020412
Chicago/Turabian StyleNeplokh, Vladimir, Daria I. Markina, Maria Baeva, Anton M. Pavlov, Demid A. Kirilenko, Ivan S. Mukhin, Anatoly P. Pushkarev, Sergey V. Makarov, and Alexey A. Serdobintsev. 2021. "Recrystallization of CsPbBr3 Nanoparticles in Fluoropolymer Nonwoven Mats for Down- and Up-Conversion of Light" Nanomaterials 11, no. 2: 412. https://doi.org/10.3390/nano11020412
APA StyleNeplokh, V., Markina, D. I., Baeva, M., Pavlov, A. M., Kirilenko, D. A., Mukhin, I. S., Pushkarev, A. P., Makarov, S. V., & Serdobintsev, A. A. (2021). Recrystallization of CsPbBr3 Nanoparticles in Fluoropolymer Nonwoven Mats for Down- and Up-Conversion of Light. Nanomaterials, 11(2), 412. https://doi.org/10.3390/nano11020412