In Vivo Study of Entero- and Hepatotoxicity of Silver Nanoparticles Stabilized with Benzyldimethyl-[3-myristoylamine)-propyl]ammonium Chloride (Miramistin) to CBF1 Mice upon Enteral Administration
Abstract
1. Introduction
2. Materials and Methods
2.1. AgNPs-BAC
- (a)
- Obtaining an ammonia complex of silver oxide:
- (b)
- Reducing diamminesilver(I) complex with glucose in the presence of BAC as a capping agent:
- (c)
- Purification of the AgNPs’ dispersion:
2.2. Transmission Electron Microscopy (TEM)
2.3. Electron Spectroscopy
2.4. Zeta Potential
2.5. Animals and Conditions
2.6. Experiment
2.7. Functional Status of the Animals
2.8. Hematological Examinations
2.9. Internal Organs Histology
2.10. Statistical Analysis
3. Results
3.1. Obtaining of AgNPs-BAC
3.2. Acute Experiment
3.2.1. Functional State of the Animals
3.2.2. Biochemical and Hematological Examinations
3.2.3. Macroscopic and Morphometric Examination of Internal Organs
3.2.4. Histological Examination
3.2.5. Subacute Experiment
3.2.6. Biochemical and Hematological Examination
3.2.7. Histological Examination
4. Discussion
4.1. Acute Experiment
4.2. Subacute Experiment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- White, R. A historical overview of the use of silver in wound management. Br. J. Community Nurs. 2001, 6, 3–8. [Google Scholar] [CrossRef]
- Klasen, H.J. A historical review of the use of silver in the treatment of burns. II. Renewed interest for silver. Burns 2000, 26, 131–138. [Google Scholar] [CrossRef]
- Lansdown, A. A Pharmacological and Toxicological Profile of Silver as an Antimicrobial Agent in Medical Devices. Adv. Pharmacol. Sci. 2010, 2010, 910686. [Google Scholar] [CrossRef]
- Lansdown, A.B. Silver in health care: Antimicrobial effects and safety in use. Curr. Probl. Dermatol. 2006, 33, 17–34. [Google Scholar] [PubMed]
- Zhang, W.; Qiao, X.; Chen, J. Synthesis of silver nanoparticles—Effects of concerned parameters in water/oil microemulsion. Mater. Sci. Eng. B 2007, 142, 1–15. [Google Scholar] [CrossRef]
- Li, Q.; Mahendra, S.; Lyon, D.Y.; Brunet, L.; Liga, M.V.; Li, D.; Alvarez, P.J.J. Antimicrobial nanomaterials for water disinfection and microbial control: Potential applications and implications. Water Res. 2008, 42, 4591–4602. [Google Scholar] [CrossRef]
- Lv, Y.; Liu, H.; Wang, Z.; Liu, S.; Hao, L.; Sang, Y.; Liu, D.; Wang, J.; Boughton, R.I. Silver nanoparticle-decorated porous ceramic composite for water treatment. J. Membr. Sci. 2009, 331, 50–56. [Google Scholar] [CrossRef]
- Cerkez, I.; Kocer, H.; Worley, S.; Broughton, R.; Huang, T. Multifunctional cotton fabric: Antimicrobial and durable press. J. Appl. Polym. Sci. 2012, 124, 4230–4238. [Google Scholar] [CrossRef]
- Fytianos, G.; Rahdar, A.; Kyzas, G. Nanomaterials in Cosmetics: Recent Updates. Nanomaterials 2020, 10, 979. [Google Scholar] [CrossRef] [PubMed]
- Marambio-Jones, C.; Hoek, E.M.V. A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J. Nanopart. Res. 2010, 12, 1531–1551. [Google Scholar] [CrossRef]
- Kim, Y.S.; Song, M.Y.; Park, J.D.; Song, K.S.; Ryu, H.R.; Chung, Y.H.; Chang, H.K.; Lee, J.H.; Oh, K.H.; Kelman, B.J.; et al. Subchronic oral toxicity of silver nanoparticles. Part. Fibre Toxicol. 2010, 7, 20. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Li, S.; Yue, X.; Lu, W. Review of Silver Nanoparticles (AgNPs)-Cellulose Antibacterial Composites. BioResources 2018, 13, 2150–2170. [Google Scholar] [CrossRef]
- Das, C.G.; Kumar, G.; Dhas, S.; Velu, K.; Govindaraju, K.; Joselin, J.; Jayaraman, B. Antibacterial activity of silver nanoparticles (biosynthesis): A short review on recent advances. Biocatal. Agric. Biotechnol. 2020, 27, 101593. [Google Scholar] [CrossRef]
- Mehrbod, P.; Motamed, N.; Tabatabaian, M.; Soleimani Estyar, R.; Amini, E.; Shahidi, M.; Kheiri, M.T. In Vitro Antiviral Effect of “Nanosilver” on Influenza Virus. DARU J. Pharm. Sci. 2015, 2015, 6. [Google Scholar]
- Kim, K.J.; Sung, W.S.; Suh, B.K.; Moon, S.K.; Choi, J.S.; Kim, J.G.; Lee, D.G. Antifungal activity and mode of action of silver nano-particles on Candida albicans. Biomet. Int. J. Role Met. Ions Biol. Biochem. Med. 2009, 22, 235–242. [Google Scholar] [CrossRef]
- Raut, R.W.; Mendhulkar, V.D.; Kashid, S.B. Photosensitized synthesis of silver nanoparticles using Withania somnifera leaf powder and silver nitrate. J. Photochem. Photobiol. B Biol. 2014, 132, 45–55. [Google Scholar] [CrossRef]
- Drogat, N.; Granet, R.; Sol, V.; Memmi, A.; Saad, N.; Klein Koerkamp, C.; Bressollier, P.; Krausz, P. Antimicrobial silver nanoparticles generated on cellulose nanocrystals. J. Nanopart. Res. 2011, 13, 1557–1562. [Google Scholar] [CrossRef]
- Boudreau, M.D.; Imam, M.S.; Paredes, A.M.; Bryant, M.S.; Cunningham, C.K.; Felton, R.P.; Jones, M.Y.; Davis, K.J.; Olson, G.R. Differential Effects of Silver Nanoparticles and Silver Ions on Tissue Accumulation, Distribution, and Toxicity in the Sprague Dawley Rat Following Daily Oral Gavage Administration for 13 Weeks. Toxicol. Sci. Off. J. Soc. Toxicol. 2016, 150, 131–160. [Google Scholar] [CrossRef]
- Loeschner, K.; Hadrup, N.; Qvortrup, K.; Larsen, A.; Gao, X.; Vogel, U.; Mortensen, A.; Lam, H.R.; Larsen, E.H. Distribution of silver in rats following 28 days of repeated oral exposure to silver nanoparticles or silver acetate. Part. Fibre Toxicol. 2011, 8, 18. [Google Scholar] [CrossRef]
- Honda, K.; Littman, D.R. The microbiota in adaptive immune homeostasis and disease. Nature 2016, 535, 75–84. [Google Scholar] [CrossRef]
- Thaiss, C.A.; Zmora, N.; Levy, M.; Elinav, E. The microbiome and innate immunity. Nature 2016, 535, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Shumakova, A.A.; Shipelin, V.A.; Efimochkina, N.R.; Minaeva, L.P.; Bykova, I.B.; Markova, Y.M.; Trushina, E.N.; Mustafina, O.K.; Gmoshinsky, I.V.; Khanferyan, R.A.; et al. Toxicological evaluation of colloidal nano-sized silver stabilized polyvinylpyrrolidone. IV. Influence on intestinal microbiota, immune indexes. Vopr. Pitan. 2016, 85, 27–35. [Google Scholar] [PubMed]
- Narciso, L.; Coppola, L.; Lori, G.; Andreoli, C.; Zjino, A.; Bocca, B.; Petrucci, F.; Di Virgilio, A.; Martinelli, A.; Tinari, A.; et al. Genotoxicity, biodistribution and toxic effects of silver nanoparticles after in vivo acute oral administration. NanoImpact 2020, 18, 100221. [Google Scholar] [CrossRef]
- Jeong, G.N.; Jo, U.B.; Ryu, H.Y.; Kim, Y.S.; Song, K.S.; Yu, I.J. Histochemical study of intestinal mucins after administration of silver nanoparticles in Sprague-Dawley rats. Arch. Toxicol. 2010, 84, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Dong, L.; Zhou, H.; Yan, B.; Chen, Y.; Cai, Y.; Liu, J. Coexposed nanoparticulate Ag alleviates the acute toxicity induced by ionic Ag+ in vivo. Sci. Total Environ. 2020, 723, 138050. [Google Scholar] [CrossRef]
- Yang, X.; Li, J.; Liang, T.; Ma, C.; Zhang, Y.; Chen, H.; Hanagata, N.; Su, H.; Xu, M. Antibacterial activity of two-dimensional MoS2 sheets. Nanoscale 2014, 6, 10126–10133. [Google Scholar] [CrossRef]
- El Mahdy, M.M.; Eldin, T.A.; Aly, H.S.; Mohammed, F.F.; Shaalan, M.I. Evaluation of hepatotoxic and genotoxic potential of silver nanoparticles in albino rats. Exp. Toxicol. Pathol. Off. J. Ges. Toxikol. Pathol. 2015, 67, 21–29. [Google Scholar] [CrossRef]
- Park, E.-J.; Bae, E.; Yi, J.; Kim, Y.; Choi, K.; Lee, S.H.; Yoon, J.; Lee, B.C.; Park, K. Repeated-dose toxicity and inflammatory responses in mice by oral administration of silver nanoparticles. Environ. Toxicol. Pharmacol. 2010, 30, 162–168. [Google Scholar] [CrossRef]
- Zaitseva, N.; Zemlyanova, M.A.; Vasiliy, Z.; Dovbysh, A.A.; Gmoshinskii, I.; Khotimchenko, S.A.; Akafeva, T. Toxicological evaluation of nanosized colloidal silver, stabilized with polyvinylpyrrolidone, in 92–day experiment on rats. II. Internal organs morphology. Vopr. Pitan. 2016, 85, 47–55. [Google Scholar]
- Kim, Y.S.; Kim, J.S.; Cho, H.S.; Rha, D.S.; Kim, J.M.; Park, J.D.; Choi, B.S.; Lim, R.; Chang, H.K.; Chung, Y.H.; et al. Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in Sprague-Dawley rats. Inhalat. Toxicol. 2008, 20, 575–583. [Google Scholar] [CrossRef]
- Heydrnejad, M.S.; Samani, R.J.; Aghaeivanda, S. Toxic Effects of Silver Nanoparticles on Liver and Some Hematological Parameters in Male and Female Mice (Mus musculus). Biol. Trace Elem. Res. 2015, 165, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Wahhab, P.M. Modulation of hepatotoxicity, DNA fragmentation and gene expression of Solanum nigrum leaves extract in rats treated with silver nanoparticles. J. Appl. Pharm. Sci. 2017, 7, 25–35. [Google Scholar]
- Sallam, A.; Ahmed, M.; Hadeed, A.; El-Fotoh, M. The Protective Effect of Pomegranate Juice in Silver Nanoparticles Induced Hepatotoxicity in Mature Male Albino Mice. Zagazig Vet. J. 2017, 45, 289–295. [Google Scholar] [CrossRef]
- Abd El-Maksoud, E.M.; Lebda, M.A.; Hashem, A.E.; Taha, N.M.; Kamel, M.A. Ginkgo biloba mitigates silver nanoparticles-induced hepatotoxicity in Wistar rats via improvement of mitochondrial biogenesis and antioxidant status. Environ. Sci. Pollut. Res. 2019, 26, 25844–25854. [Google Scholar] [CrossRef]
- Nakkala, J.R.; Mata, R.; Raja, K.; Khub Chandra, V.; Sadras, S.R. Green synthesized silver nanoparticles: Catalytic dye degradation, in vitro anticancer activity and in vivo toxicity in rats. Mater. Sci. Eng. C 2018, 91, 372–381. [Google Scholar] [CrossRef]
- Roy, N.; Gaur, A.; Jain, A.; Bhattacharya, S.; Rani, V. Green synthesis of silver nanoparticles: An approach to overcome toxicity. Environ. Toxicol. Pharmacol. 2013, 36, 807–812. [Google Scholar] [CrossRef]
- Kim, Y.J.; Rahman, M.M.; Lee, S.M.; Kim, J.M.; Park, K.; Kang, J.H.; Seo, Y.R. Assessment of in vivo genotoxicity of citrated-coated silver nanoparticles via transcriptomic analysis of rabbit liver tissue. Int. J. Nanomed. 2019, 14, 393–405. [Google Scholar] [CrossRef]
- Hong, J.S.; Kim, S.; Lee, S.H.; Jo, E.; Lee, B.; Yoon, J.; Eom, I.C.; Kim, H.M.; Kim, P.; Choi, K.; et al. Combined repeated-dose toxicity study of silver nanoparticles with the reproduction/developmental toxicity screening test. Nanotoxicology 2014, 8, 349–362. [Google Scholar] [CrossRef]
- Blanco, J.; Tomás-Hernández, S.; García, T.; Mulero, M.; Gómez, M.; Domingo, J.L.; Sánchez, D.J. Oral exposure to silver nanoparticles increases oxidative stress markers in the liver of male rats and deregulates the insulin signalling pathway and p53 and cleaved caspase 3 protein expression. Food Chem. Toxicol. 2018, 115, 398–404. [Google Scholar] [CrossRef]
- Osmanov, A.; Wise, A.; Denning, D.W. In vitro and in vivo efficacy of miramistin against drug-resistant fungi. J. Med. Microbiol. 2019, 68, 1047–1052. [Google Scholar] [CrossRef]
- Osmanov, A.; Farooq, Z.; Richardson, M.D.; Denning, D.W. The antiseptic Miramistin: A review of its comparative in vitro and clinical activity. FEMS Microbiol. Rev. 2020, 44, 399–417. [Google Scholar] [CrossRef] [PubMed]
- Agafonova, M.N.; Kazakova, R.R.; Lubina, A.P.; Zeldi, M.I.; Nikitina, E.V.; Balakin, K.V.; Shtyrlin, Y.G. Antibacterial activity profile of miramistin in in vitro and in vivo models. Microb. Pathog. 2020, 142, 104072. [Google Scholar] [CrossRef] [PubMed]
- Fromm-Dornieden, C.; Rembe, J.-D.; Schäfer, N.; Böhm, J.; Stuermer, E.K. Cetylpyridinium chloride and miramistin as antiseptic substances in chronic wound management—Prospects and limitations. J. Med. Microbiol. 2015, 64, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Danilova, T.A.; Danilina, G.A.; Adzhieva, A.A.; Minko, A.G.; Nikolaeva, T.N.; Zhukhovitskii, V.G.; Pronin, A.V. Effects of Miramistin and Phosprenil on Microbial Biofilms. Bull. Exp. Biol. Med. 2017, 163, 439–442. [Google Scholar] [CrossRef] [PubMed]
- Skogoreva, A.; Manzhurina, O.; Popova, O.; Semyonov, S.; Aristov, A. Improving treatment of subclinical cow mastitis using miramistin antiseptic agent. IOP Conf. Ser. Earth Environ. Sci. 2020, 422, 012045. [Google Scholar] [CrossRef]
- Krutyakov, Y.A.; Kudrinskiy, A.A.; Zherebin, P.M.; Yapryntsev, A.D.; Pobedinskaya, M.A.; Elansky, S.N.; Denisov, A.N.; Mikhaylov, D.M.; Lisichkin, G.V. Tallow amphopolycarboxyglycinate-stabilized silver nanoparticles: New frontiers in development of plant protection products with a broad spectrum of action against phytopathogens. Mater. Res. Express 2016, 3, 075403. [Google Scholar] [CrossRef]
- Gusev, A.; Kudrinsky, A.; Zakharova, O.; Klimov, A.; Zherebin, P.; Lisichkin, G.; Vasyukova, I.; Denisov, A.; Krutyakov, Y. Versatile synthesis of PHMB-stabilized silver nanoparticles and their significant stimulating effect on fodder beet (Beta vulgaris L.). Mater. Sci. Eng. C 2016, 62, 152–159. [Google Scholar] [CrossRef]
- Krutyakov, Y.A.; Zherebin, P.; Kudrinskiy, A.A.; Zubavichus, Y.V.; Presniakov, M.Y.; Yapryntsev, A.; Karabtseva, A.V.; Mikhaylov, D.M.; Lisichkin, G. New frontiers in water purification: Highly stable amphopolycarboxyglycinate-stabilized Ag-AgCl nanocomposite and its newly discovered potential. J. Phys. D Appl. Phys. 2016, 49, 375501. [Google Scholar] [CrossRef]
- Kudrinskiy, A.; Zherebin, P.; Gusev, A.; Shapoval, O.; Pyee, J.; Lisichkin, G.; Krutyakov, Y. New Relevant Descriptor of Linear QNAR Models for Toxicity Assessment of Silver Nanoparticles. Nanomaterials 2020, 10, 1459. [Google Scholar] [CrossRef]
- Vertelov, G.K.; Krutyakov, Y.A.; Efremenkova, O.V.; Olenin, A.Y.; Lisichkin, G.V. A versatile synthesis of highly bactericidal Myramistin® stabilized silver nanoparticles. Nanotechnology 2008, 19, 355707. [Google Scholar] [CrossRef]
- Krutyakov, Y.; Klimov, A.; Violin, B.; Kuzmin, V.; Ryzhikh, V.; Gusev, A.; Zakharova, O.; Lisichkin, G. Benzyldimethyl[3–(miristoylamino)-propyl]ammonium chloride stabilized silver nanoparticles (Argumistin™) in medicine: Results of clinical trials for treatment of infectious diseases of dogs and perspectives for humans. Eur. J. Nanomed. 2016, 8. [Google Scholar] [CrossRef]
- Kudrinskiy, A.A.; Krutyakov, Y.A.; Olenin, A.Y.; Romanovskaya, G.I.; Vasilyeva, S.Y.; Lisichkin, G.V. Sensitized fluorescence of silver nanoparticles in the presence of pyrene. J. Fluoresc. 2009, 19, 473–478. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals, 8th ed.; National Academies Press (US): Washington, DC, USA, 2011. [Google Scholar]
- State Standard of Russian Federation 33216-2014 Guidelines for Accommodation and Care of Animals. Species-Specific Provisions for Laboratory Rodents and Rabbits. 2014. Available online: http://docs.cntd.ru/document/1200127506 (accessed on 22 December 2020). (In Russian)
- Korzhevsky, D.E.; Gilyarov, A.V. Fundamentals of Histological Technique; SpetsLit Publishing House Ltd.: St. Perterburg, Russia, 2010; p. 95. (In Russian) [Google Scholar]
- Koptev, V.Y.; Leonova, M.A.; Balybina, N.Y.; Violin, B.V.; Kudrinsky, A.A.; Krutyakov, Y.A. The Influence of Argumistin® Preparation on Weight Gains and Bacterial Contamination in Broilers. Poultry 2015, 31–38. Available online: https://www.elibrary.ru/item.asp?id=24116586 (accessed on 27 November 2020). (In Russian).
- Krutyakov, Y.A.; Koptev, V.Y.; Kudrinsky, A.A.; Klimov, A.I.; Titova, M.A.; Balybina, N.Y.; Lisichkin, G.V. Determination of silver in tissues and organs of broiler chickens after oral and aerosol administration of an aqueous dispersion of silver nanoparticles. Hyg. Sanit. 2016, 95, 207–211. [Google Scholar] [CrossRef]
- van der Zande, M.; Vandebriel, R.J.; Van Doren, E.; Kramer, E.; Herrera Rivera, Z.; Serrano-Rojero, C.S.; Gremmer, E.R.; Mast, J.; Peters, R.J.; Hollman, P.C.; et al. Distribution, elimination, and toxicity of silver nanoparticles and silver ions in rats after 28–day oral exposure. ACS Nano 2012, 6, 7427. [Google Scholar] [CrossRef]
- Martins, A.D.C., Jr.; Azevedo, L.F.; de Souza Rocha, C.C.; Carneiro, M.F.H.; Venancio, V.P.; de Almeida, M.R.; Antunes, L.M.G. Evaluation of distribution, redox parameters, and genotoxicity inWistar rats co-exposed to silver and titanium dioxide nanoparticles. J. Toxicol. Environ. Health Part A 2017, 80, 1156. [Google Scholar] [CrossRef]
- Gmoshinskii, I.; Shipelin, V.; Khotimchenko, S. Nanomaterials in food products and their package: Comparative analysis of risks and advantages. Risk Anal. 2018, 134–142. [Google Scholar] [CrossRef]
- Li, J.; Tang, M.; Xue, Y. Review of the effects of silver nanoparticle exposure on gut bacteria. J. Appl. Toxicol. JAT 2019, 39, 27–37. [Google Scholar] [CrossRef]
- Recordati, C.; De Maglie, M.; Bianchessi, S.; Argentiere, S.; Cella, C.; Mattiello, S.; Cubadda, F.; Aureli, F.; D’Amato, M.; Raggi, A.; et al. Tissue distribution and acute toxicity of silver after single intravenous administration in mice: Nano-specific and size-dependent effects. Part. Fibre Toxicol. 2016, 13, 12. [Google Scholar] [CrossRef]
- Garcia, T.; Lafuente, D.; Blanco, J.; Sánchez, D.J.; Sirvent, J.J.; Domingo, J.L.; Gómez, M. Oral subchronic exposure to silver nanoparticles in rats. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2016, 92, 177–187. [Google Scholar] [CrossRef]
- Bergin, I.L.; Wilding, L.A.; Morishita, M.; Walacavage, K.; Ault, A.P.; Axson, J.L.; Stark, D.I.; Hashway, S.A.; Capracotta, S.S.; Leroueil, P.R.; et al. Effects of particle size and coating on toxicologic parameters, fecal elimination kinetics and tissue distribution of acutely ingested silver nanoparticles in a mouse model. Nanotoxicology 2016, 10, 352–360. [Google Scholar] [CrossRef]
- Yue, L.; Zhao, W.; Wang, D.; Meng, M.; Zheng, Y.; Li, Y.; Qiu, J.; Yu, J.; Yan, Y.; Lu, P.; et al. Silver nanoparticles inhibit beige fat function and promote adiposity. Mol. Metab. 2019, 22, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gaillet, S.; Rouanet, J.M. Silver nanoparticles: Their potential toxic effects after oral exposure and underlying mechanisms—A review. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2015, 77, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Dziendzikowska, K.; Gromadzka-Ostrowska, J.; Lankoff, A.; Oczkowski, M.; Krawczyńska, A.; Chwastowska, J.; Sadowska-Bratek, M.; Chajduk, E.; Wojewódzka, M.; Dušinská, M.; et al. Time-dependent biodistribution and excretion of silver nanoparticles in male Wistar rats. J. Appl. Toxicol. JAT 2012, 32, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Kim, P.; Yoon, J.; Lee, B.; Choi, K.; Kil, K.H.; Park, K. Serum kinetics, distribution and excretion of silver in rabbits following 28 days after a single intravenous injection of silver nanoparticles. Nanotoxicology 2013, 7, 1120–1130. [Google Scholar] [CrossRef]
- Gmoshinski, I.V.; Shumakova, A.A.; Shipelin, V.A.; Maltsev, G.Y.; Khotimchenko, S.A. Influence of orally introduced silver nanoparticles on content of essential and toxic trace elements in organism. Nanotechnol. Russia 2016, 11, 646–652. [Google Scholar] [CrossRef]
- Hadrup, N.; Loeschner, K.; Bergström, A.; Wilcks, A.; Gao, X.; Vogel, U.; Frandsen, H.L.; Larsen, E.H.; Lam, H.R.; Mortensen, A. Subacute oral toxicity investigation of nanoparticulate and ionic silver in rats. Arch. Toxicol. 2012, 86, 543–551. [Google Scholar] [CrossRef]
- Cheraghi, J.; Hosseini, E.; Hoshmandfar, R.; Sahraei, R.; Farmany, A. In vivo effect of Silver Nanoparticles on serum, A.L.T.; AST and ALP activity in male and female mice. Adv. Environ. Biol. 2013, 7, 116–122. [Google Scholar]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.-H.; Park, S.J.; Lee, H.J.; Kim, S.H.; Park, Y.K.; Park, Y.H.; Hwang, C.-Y.; et al. Antimicrobial effects of silver nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 95–101. [Google Scholar] [CrossRef]
- Wang, Z.; Qu, G.; Su, L.; Wang, L.; Yang, Z.; Jiang, J.; Liu, S.; Jiang, G. Evaluation of the biological fate and the transport through biological barriers of nanosilver in mice. Curr. Pharm. Des. 2013, 19, 6691–6697. [Google Scholar] [CrossRef]
- Cui, X.; Bao, L.; Wang, X.; Chen, C. The Nano–Intestine Interaction: Understanding the Location-Oriented Effects of Engineered Nanomaterials in the Intestine. Small 2020, 16, 1907665. [Google Scholar] [CrossRef]
- Xu, M.; Yang, Q.; Xu, L.; Rao, Z.; Cao, D.; Gao, M.; Liu, S. Protein target identification and toxicological mechanism investigation of silver nanoparticles-induced hepatotoxicity by integrating proteomic and metallomic strategies. Part. Fibre Toxicol. 2019, 16, 46. [Google Scholar] [CrossRef] [PubMed]
- De Matteis, V.; Malvindi, M.A.; Galeone, A.; Brunetti, V.; Luca, E.; Kote, S.; Kshirsagar, P.; Sabella, S.; Bardi, G.; Pompa, P. Negligible particle-specific toxicity mechanism of silver nanoparticles: The role of Ag+ ion release in the cytosol. Nanomed. Nanotechnol. Biol. Med. 2014, 11, 731–739. [Google Scholar] [CrossRef] [PubMed]
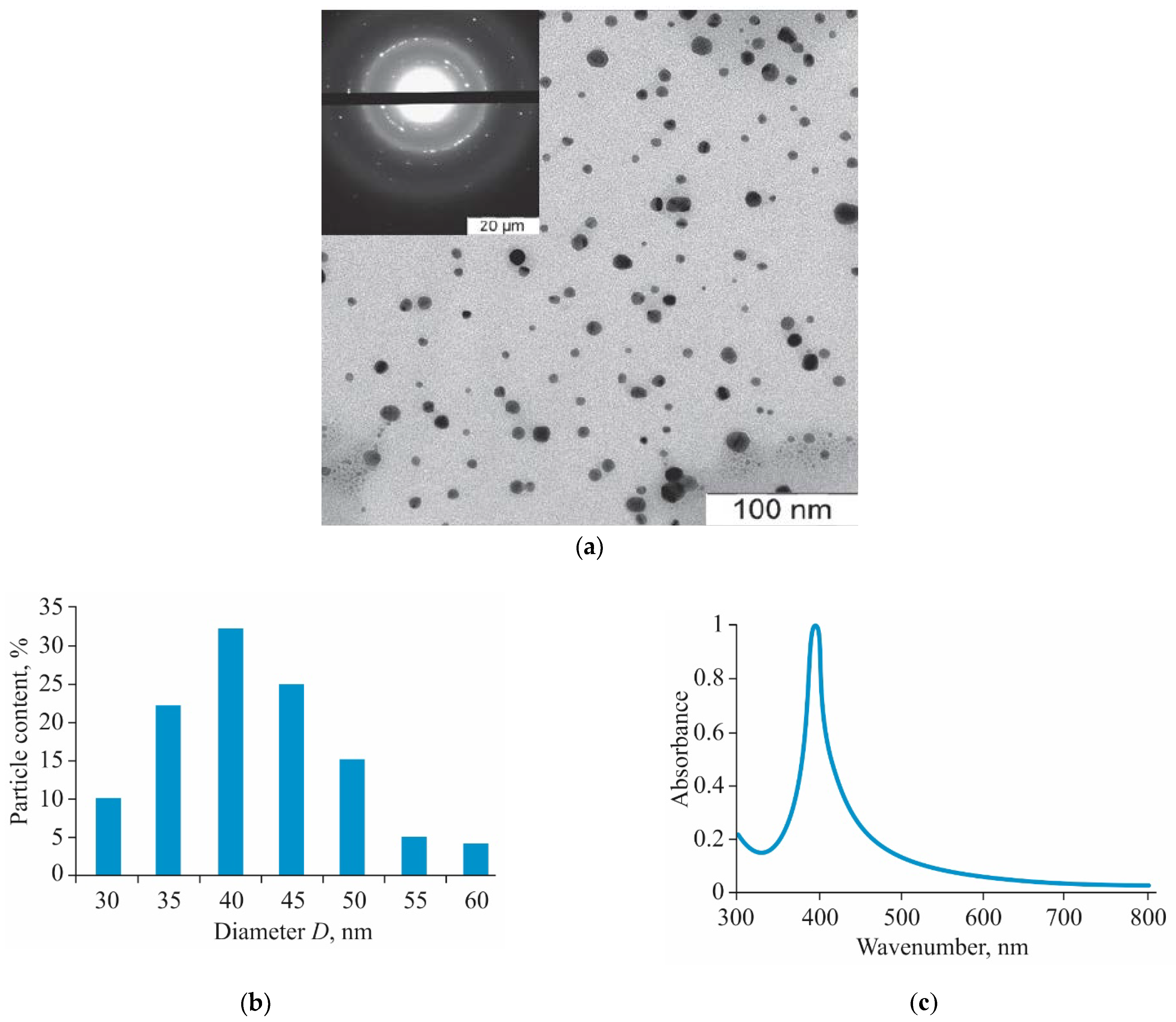
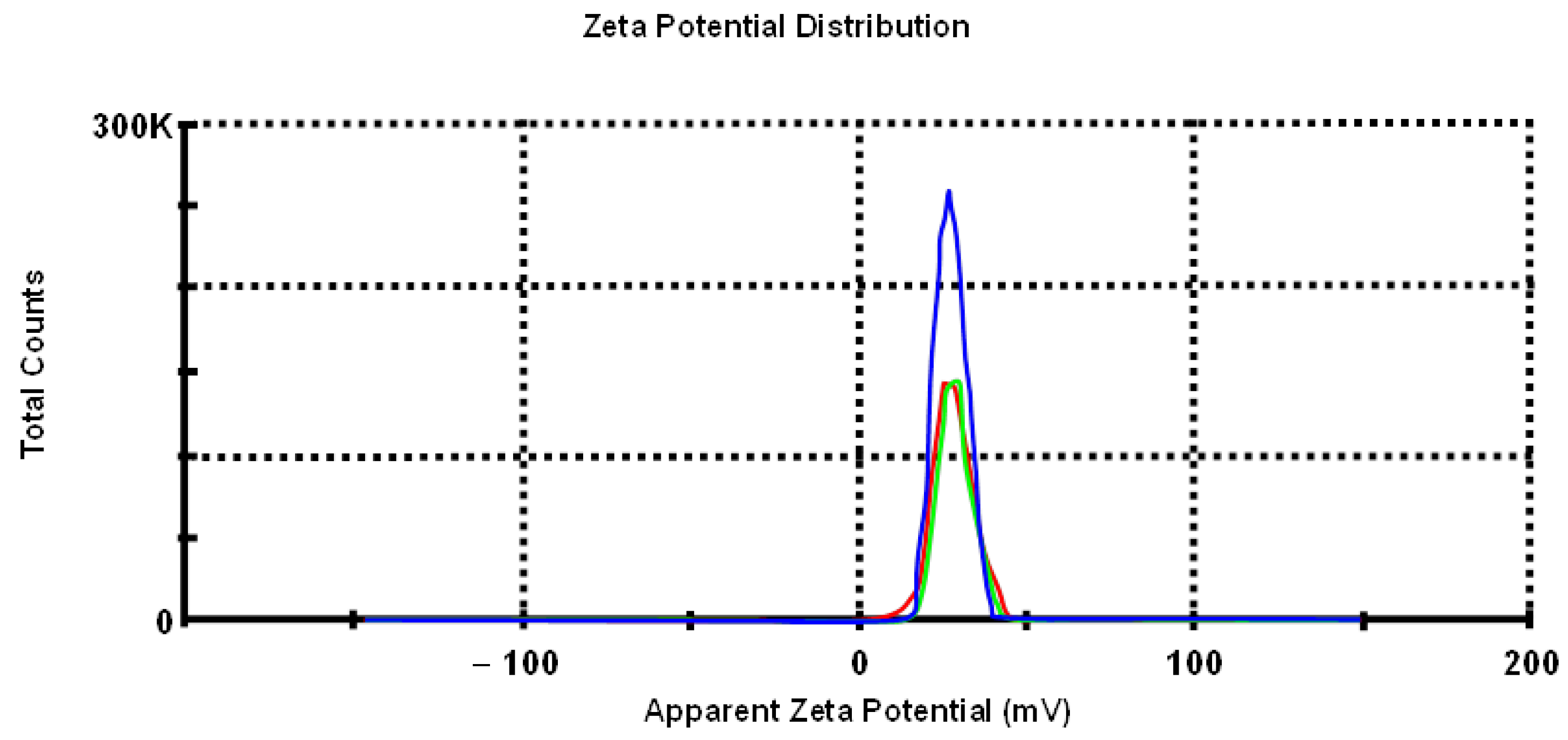
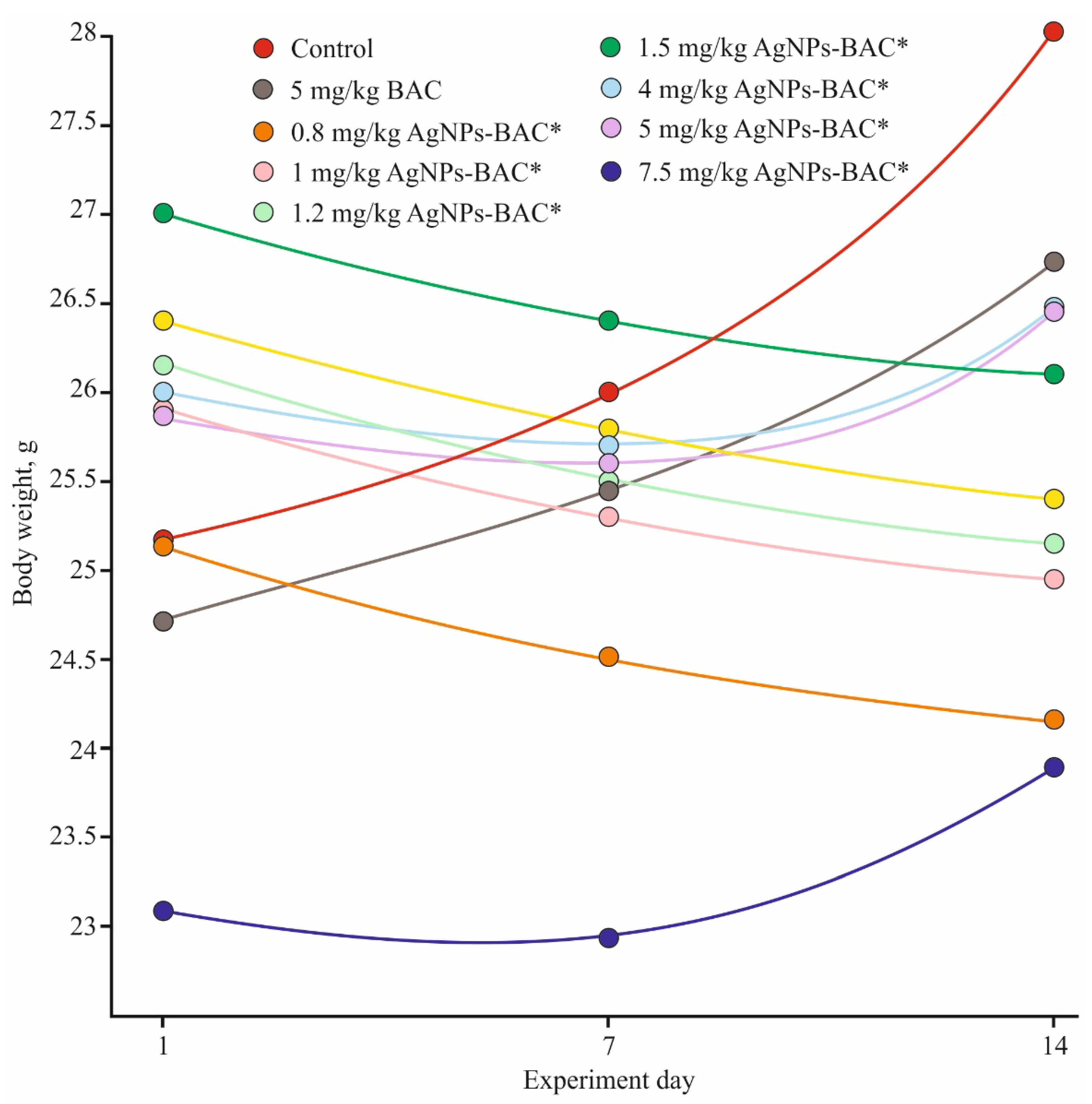
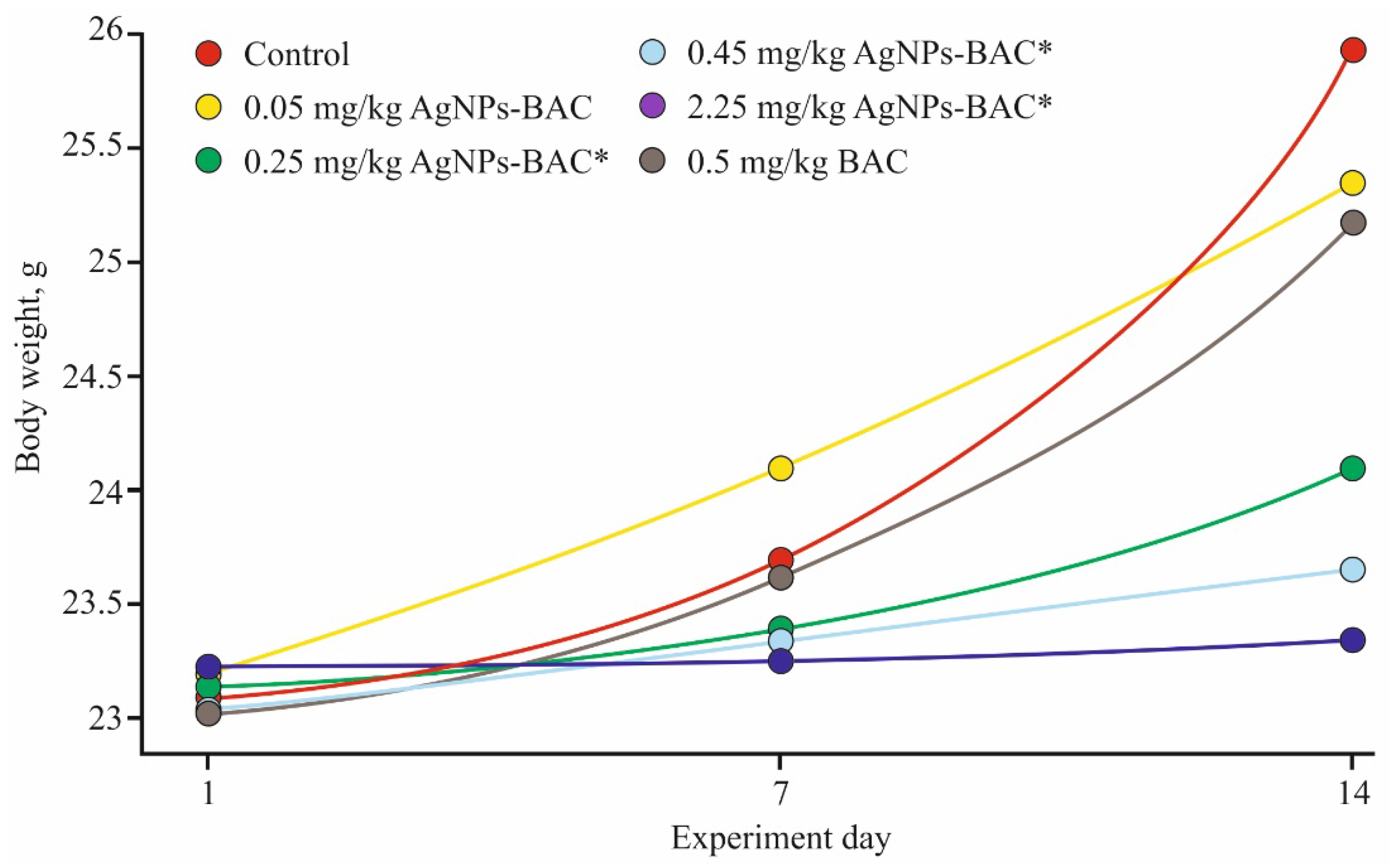
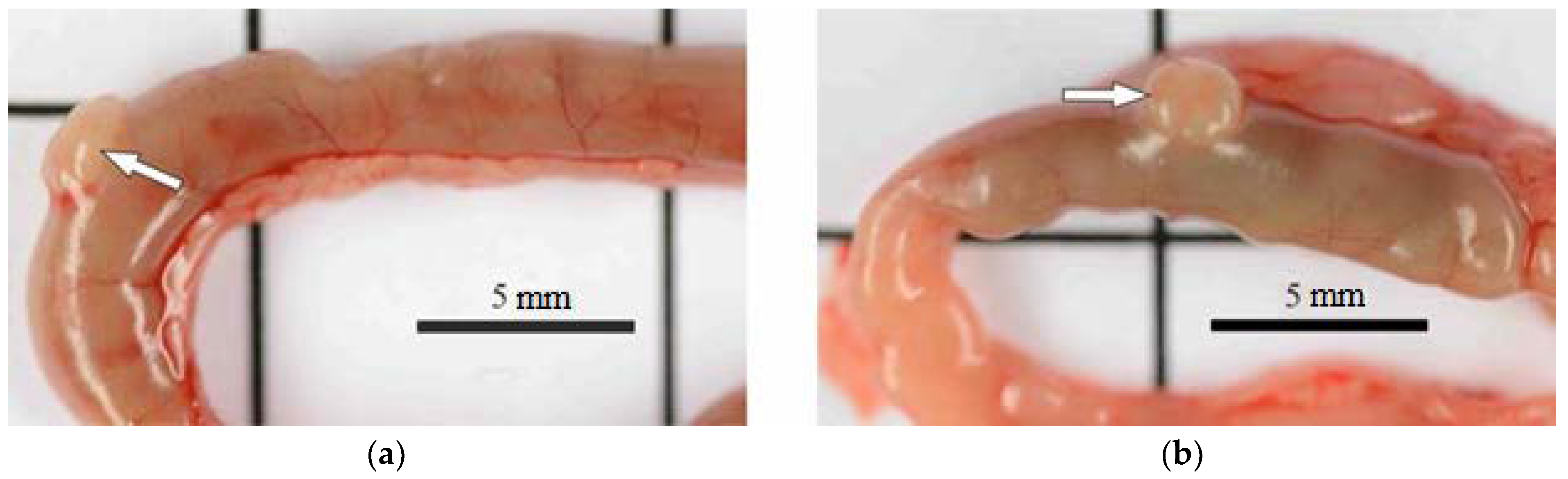
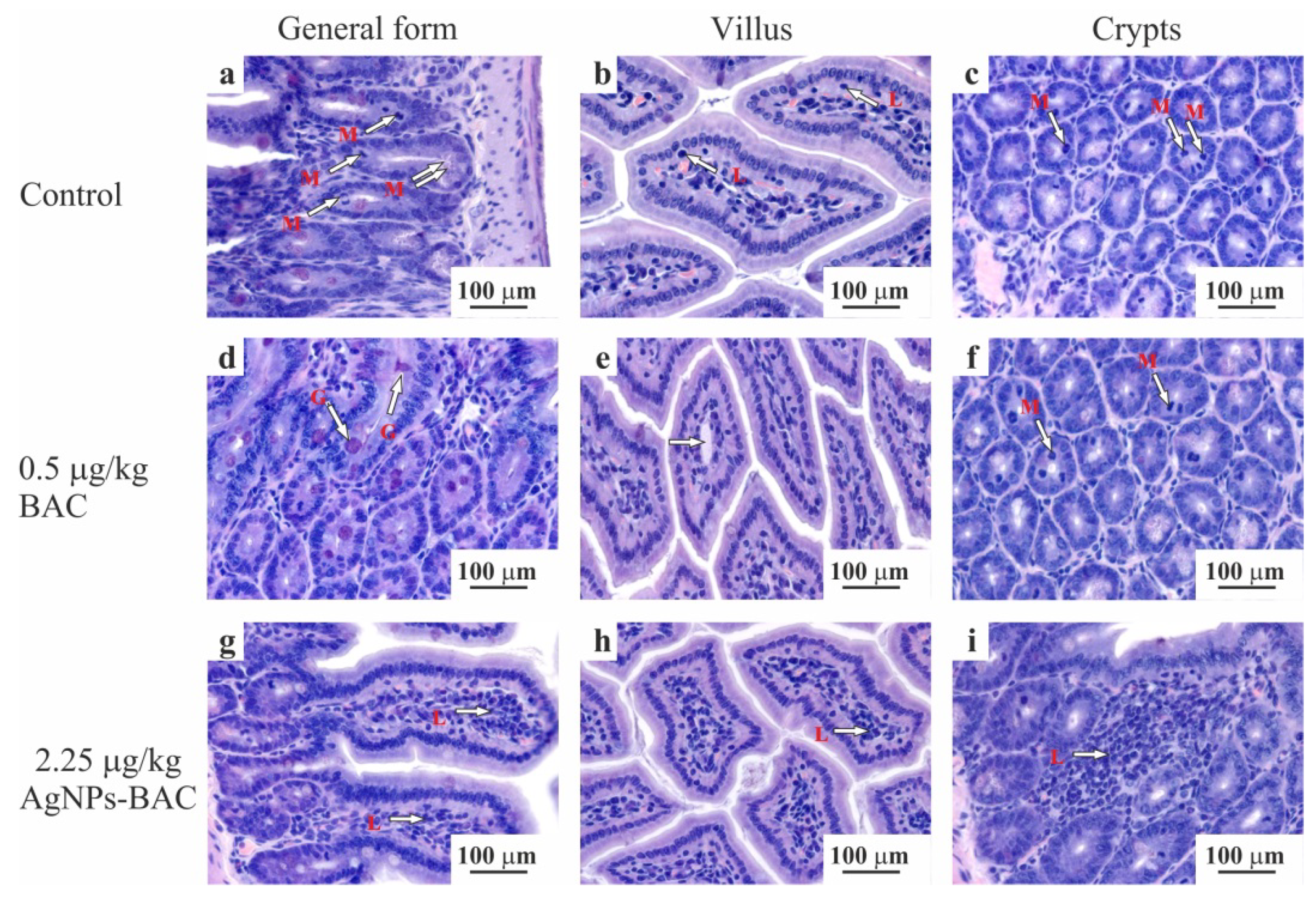
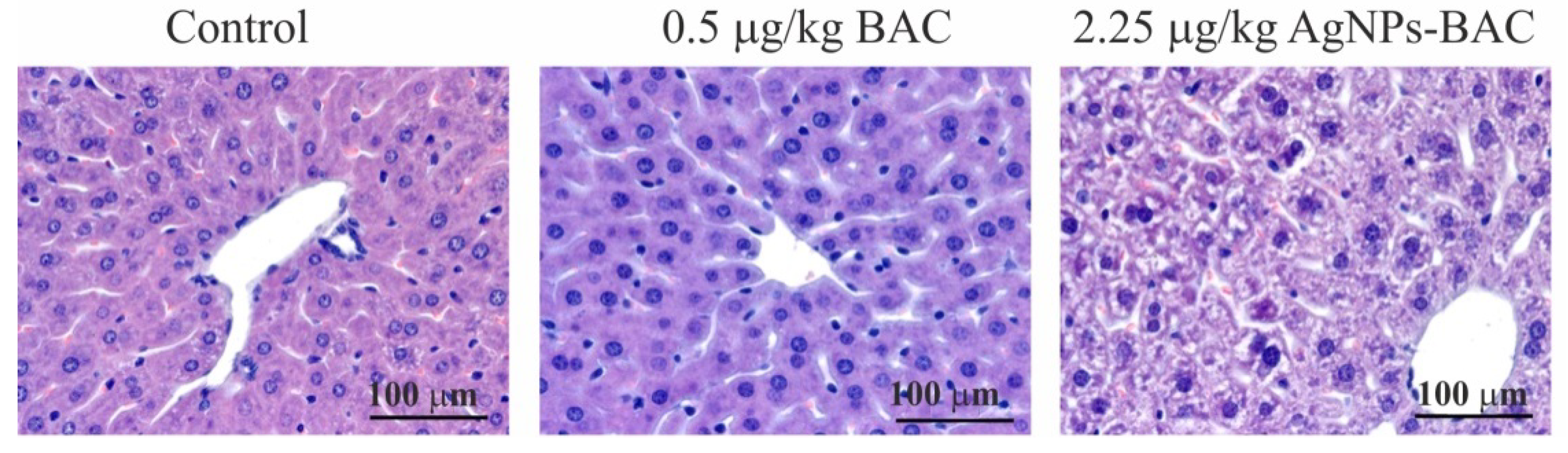
| Parameters/Group | 5 mg/kg Dose of AgNPs-BAC | 7.5 mg/kg Dose of AgNPs-BAC | 5 mg/kg Dose of BAC | Control |
|---|---|---|---|---|
| Red blood cells, ×1012 | 7.84 ± 0.48 | 7.24 ± 0.38 | 7.12± 0.39 | 6.95 ± 0.41 |
| White blood cells, ×109 | 3.74 ± 0.36 | 5.15 ± 0.64 | 5.17 ± 0.38 | 5.2 ± 0.59 |
| Haemoglobin, g/L | 128.07 ± 5.27 | 130.78 ± 8.26 | 127.46 ± 4.33 | 126.33 ± 3.23 |
| White blood cell differential | ||||
| Neutrophils: | ||||
| immature | 2.7 ± 0.75 | 1.2 ± 0.38 | 1.3 ± 0.52 | 1.16 ± 0.65 |
| band | 3.2 ± 0.43 | 3.9 ± 0.54 | 3.3 ± 1.2 | 4.1 ± 1.1 |
| segmented | 27.55 ± 2.3 | 16.4 ± 2.25 | 21.79 ± 2.8 | 21.33 ± 4.7 |
| Eosinophils | 0 | 0.6 ± 0.06 | 0.1 ± 0.01 | 0.1 ± 0.01 |
| Basophils | 0 | 0 | 0 | 0 |
| Monocytes | 0 | 0 | 0 | 0 |
| Lymphocyte | 66.55 ± 1.79 | 78.0 ± 2.18 | 76.0 ± 3.29 | 75.0 ± 5.05 |
| Aspartate transaminase, IU/L | 346.37 ± 7.1 * | 361.75 ± 4.63 * | 322.88 ± 5.78 | 319.83 ± 8.76 |
| Alanine transaminase, IU/L | 103.9 ± 0.55 * | 125.9 ± 0.53 * | 87.12 ± 0.94 | 87.12 ± 0.94 |
| Total protein, g/L | 54.71 ± 7.77 | 63.22 ± 2.68 | 56.32 ± 2.36 | 53.75 ± 1.56 |
| Albumin, g/L | 19.53 ± 0.88 | 23.83 ± 1.24 | 22.75 ± 0.76 | 22.05 ± 0.78 |
| Globulins, g/L | 35.48 ± 2.03 * | 37.9 ± 2.33 * | 32.44 ± 1.11 | 30.45 ± 1.12 |
| Albumin/Globulin | 0.56 ± 0.05 * | 0.66 ± 0.07 * | 0.7 ± 0.03 | 0.72 ± 0.03 |
| Urea, mmol/L | 12.05 ± 0.26 * | 14.62 ± 0.71 * | 9.2 ± 0.37 | 8.3 ± 0.35 |
| Creatinine, μmol/L | 38.75 ± 2.33 | 73.42 ± 8.13 | 55.53 ± 3.27 | 50.63 ± 5.24 |
| Group | Heart, % | Liver, % | Spleen, % | Kidney, % |
|---|---|---|---|---|
| Control | 0.39 ± 0.03 | 6.22 ± 0.2 | 0.89 ± 0.16 | 0.77 ± 0.02 |
| 5 mg/kg dose of BAC | 0.4 ± 0.02 | 6.03 ± 0.17 | 0.91 ± 0.15 | 0.79 ± 0.03 |
| 0.8 mg/kg dose of AgNPs-BAC | 0.41 ± 0.02 | 5.53 ± 0.27 | 0.67 ± 0.13 | 0.84 ± 0.07 |
| 1 mg/kg dose of AgNPs-BAC | 0.41 ± 0.01 | 5.73 ± 0.17 | 0.67 ± 0.09 | 0.79 ± 0.1 |
| 1.2 mg/kg dose of AgNPs-BAC | 0.41 ± 0.02 | 5.58 ± 0.23 | 0.65 ± 0.11 | 0.82 ± 0.07 |
| 1.5 mg/kg dose of AgNPs-BAC | 0.42 ± 0.02 | 5.55 ± 0.19 | 0.65 ± 0.09 | 0.84 ± 0.1 |
| 4 mg/kg dose of AgNPs-BAC | 0.42 ± 0.03 | 4.92 ± 0.22 * | 0.93 ± 0.06 | 0.86 ± 0.03 |
| 5 mg/kg dose of AgNPs-BAC | 0.41 ± 0.03 | 4.91 ± 0.19 * | 0.89 ± 0.07 | 0.81 ± 0.03 |
| 7.5 mg/kg dose of AgNPs-BAC | 0.42 ± 0.02 | 4.94 ± 0.19 * | 0.92 ± 0.07 | 0.86 ± 0.03 |
| Group | Total Protein, g/L | Albumin, g/L | Urea, mmol/L | Creatinine, μmol/L | ALT, UI/L | AST, UI/L |
|---|---|---|---|---|---|---|
| Control | 53.75 ± 1.26 | 22.05 ± 0.78 | 8.3 ± 0.35 | 50.63 ± 5.24 | 38.9 ± 1.6 | 66.56 ± 11.5 |
| 0.5 mg/kg dose of BAC | 52.63 ± 1.26 | 24.05 ± 0.63 | 8.41 ± 0.41 | 55.62 ± 6.21 | 39.9 ± 1.7 | 65.12 ± 10.3 |
| 0.05 mg/kg dose of AgNPs-BAC | 51.8 ± 1.7 | 27.7 ± 1.4 | 8.3 ± 0.6 | 67.12 ± 8.04 | 37.2 ± 1.4 | 64.7 ± 1.8 |
| 0.45 mg/kg dose of AgNPs-BAC | 52.0 ± 1.6 | 29.2 ± 1.1 | 8.5 ± 0.5 | 51.54 ± 6.45 | 40.4 ± 2.0 | 53.3 ± 1.7 |
| 0.25 mg/kg dose of AgNPs-BAC | 53.2 ± 1.9 | 28.7 ± 1.2 | 8.6 ± 0.5 | 52.42 ± 3.12 | 39.5 ± 1.8 | 64.1 ± 1.6 |
| 2.25 mg/kg dose of AgNPs-BAC | 51.65 ± 1.97 | 29.72 ± 1.3 | 8.74 ± 0.47 | 80.33 ± 18.02 | 9 ± 0.14 * | 3 ± 0.52 * |
| 0.05 mg/kg Dose of AgNPs-BAC | 0.45 mg/kg Dose of AgNPs-BAC | 0.25 mg/kg Dose of AgNPs-BAC | 2.25 mg/kg Dose of AgNPs-BAC | Control | 0.5 mg/kg Dose of BAC | |
|---|---|---|---|---|---|---|
| Red blood cells, ×1012 | 6.96 ± 0.35 | 7.08 ± 0.49 | 6.98 ± 0.34 | 7.13 ± 0.29 | 6.95 ± 0.41 | 6.97 ± 0.36 |
| White blood cells, ×109 | 5.2 ± 0.36 | 3.98 ± 0.2 | 5.01 ± 0.35 | 3.67 ± 0.29 | 5.2 ± 0.59 | 4.8 ± 0.44 |
| Platelets, 103/μL | 734 ± 60 | 725 ± 62 | 736 ± 58 | 741 ± 44 | 732 ± 56 | 734 ± 57 |
| Haemoglobin, g/L | 128.92 ± 6.33 | 130.14 ± 3.48 | 129.01 ± 5.76 | 134.62 ± 4.77 | 126.33 ± 3.23 | 129.43 ± 3.11 |
| White blood cell differential, % | ||||||
| Neutrophils: | ||||||
| immature | 1.7 ± 0.75 | 1.6 ± 0.53 | 1.2 ± 0.38 | 1.36 ± 0.43 | 1.16 ± 0.65 | 1.3 ± 0.45 |
| band | 3.2 ± 0.43 | 3.2 ± 0.52 | 3.9 ± 0.54 | 2 ± 0.52 | 4.1 ± 1.1 | 3.6 ± 0.6 |
| segmented | 22.55 ± 2.3 | 22.15 ± 2.14 | 22.4 ± 2.25 | 22.09 ± 3.24 | 21.33 ± 4.7 | 22.16 ± 4.3 |
| Eosinophils | 0 | 0.36 ± 0.05 | 0.36 ± 0.06 | 0.36 ± 0.05 | 0.1 ± 0.01 | 0 |
| Basophils | 0 | 0 | 0 | 0 | 0 | 0 |
| Monocytes | 0 | 0 | 0 | 0 | 0 | 0 |
| Lymphocyte | 76.5 ± 1.79 | 74.31 ± 1.38 | 74.0 ± 2.18 | 74 ± 3.29 | 75.0 ± 5.05 | 73.0 ± 4.6 |
| Organ | Control | 0.05 mg/kg Dose of AgNPs-BAC | 0.25 mg/kg Dose of AgNPs-BAC | 0.45 mg/kg Dose of AgNPs-BAC | 2.25 mg/kg Dose of AgNPs-BAC | 0.5 mg/kg Dose of BAC |
|---|---|---|---|---|---|---|
| Kidneys,% | 0.77 ± 0.02 | 0.77 ± 0.03 | 0.78 ± 0.05 | 0.7 ± 0.07 | 0.71 ± 0.02 | 0.79 ± 0.03 |
| Liver,% | 6.22 ± 0.2 | 6.5 ± 0.39 | 5.86 ± 0.12 | 5.1 ± 0.26 * | 4.4 ± 0.09 * | 6.15 ± 0.12 |
| Heart,% | 0.39 ± 0.03 | 0.43 ± 0.03 | 0.37 ± 0.08 | 0.42 ± 0.02 | 0.42 ± 0.007 | 0.38 ± 0.08 |
| Spleen,% | 0.89 ± 0.16 | 0.65 ± 0.11 | 0.55 ± 0.4 | 0.46 ± 0.07 * | 0.41 ± 0.03 * | 0.82 ± 0.11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krutyakov, Y.A.; Kudrinskiy, A.A.; Kuzmin, V.A.; Pyee, J.; Gusev, A.A.; Vasyukova, I.A.; Zakharova, O.V.; Lisichkin, G.V. In Vivo Study of Entero- and Hepatotoxicity of Silver Nanoparticles Stabilized with Benzyldimethyl-[3-myristoylamine)-propyl]ammonium Chloride (Miramistin) to CBF1 Mice upon Enteral Administration. Nanomaterials 2021, 11, 332. https://doi.org/10.3390/nano11020332
Krutyakov YA, Kudrinskiy AA, Kuzmin VA, Pyee J, Gusev AA, Vasyukova IA, Zakharova OV, Lisichkin GV. In Vivo Study of Entero- and Hepatotoxicity of Silver Nanoparticles Stabilized with Benzyldimethyl-[3-myristoylamine)-propyl]ammonium Chloride (Miramistin) to CBF1 Mice upon Enteral Administration. Nanomaterials. 2021; 11(2):332. https://doi.org/10.3390/nano11020332
Chicago/Turabian StyleKrutyakov, Yurii A., Alexey A. Kudrinskiy, Vladimir A. Kuzmin, Jaeho Pyee, Alexander A. Gusev, Inna A. Vasyukova, Olga V. Zakharova, and Georgy V. Lisichkin. 2021. "In Vivo Study of Entero- and Hepatotoxicity of Silver Nanoparticles Stabilized with Benzyldimethyl-[3-myristoylamine)-propyl]ammonium Chloride (Miramistin) to CBF1 Mice upon Enteral Administration" Nanomaterials 11, no. 2: 332. https://doi.org/10.3390/nano11020332
APA StyleKrutyakov, Y. A., Kudrinskiy, A. A., Kuzmin, V. A., Pyee, J., Gusev, A. A., Vasyukova, I. A., Zakharova, O. V., & Lisichkin, G. V. (2021). In Vivo Study of Entero- and Hepatotoxicity of Silver Nanoparticles Stabilized with Benzyldimethyl-[3-myristoylamine)-propyl]ammonium Chloride (Miramistin) to CBF1 Mice upon Enteral Administration. Nanomaterials, 11(2), 332. https://doi.org/10.3390/nano11020332







