Graphene Family Nanomaterials (GFN)-TiO2 for the Photocatalytic Removal of Water and Air Pollutants: Synthesis, Characterization, and Applications
Abstract
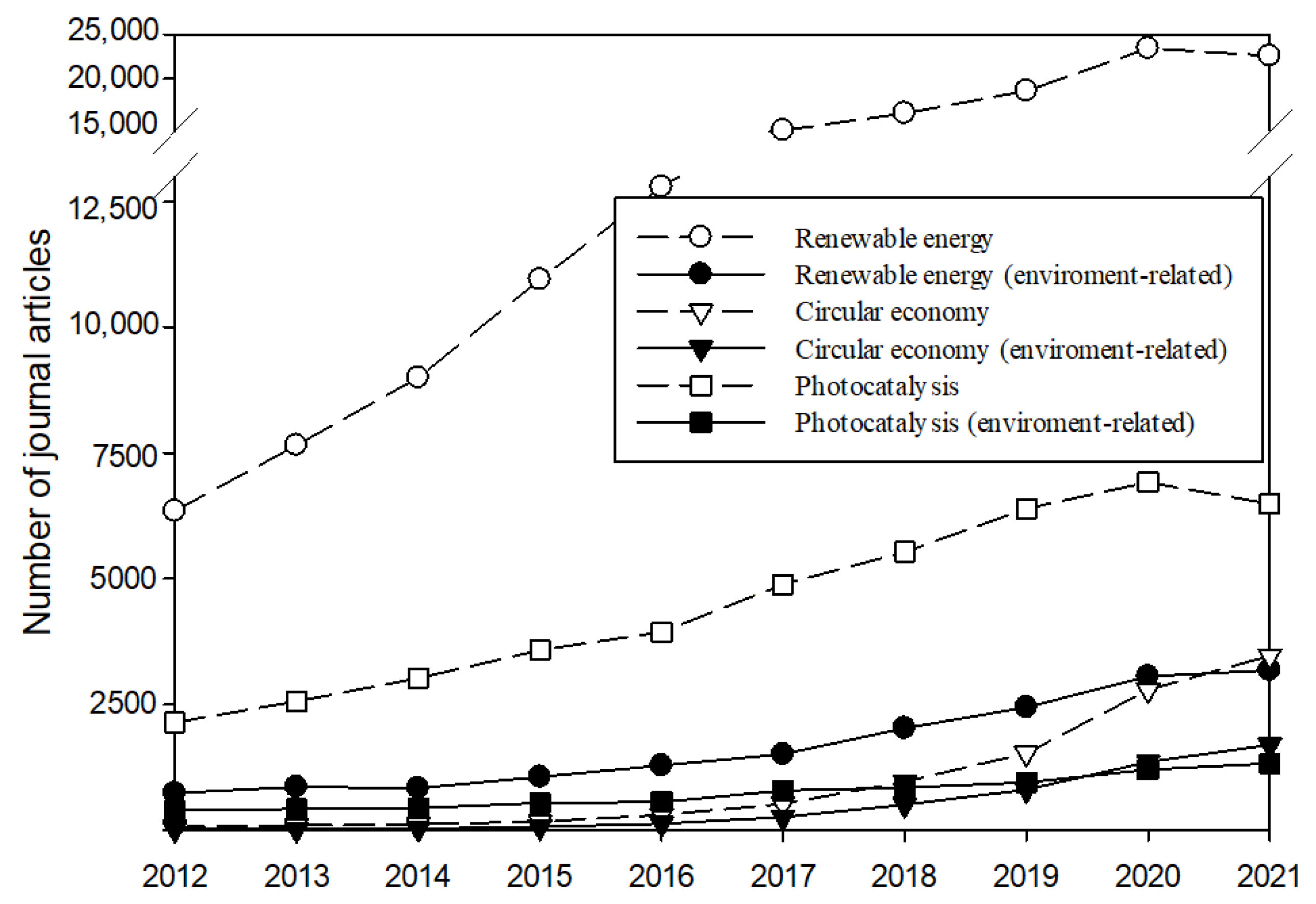
1. Introduction
2. TiO2
2.1. Background
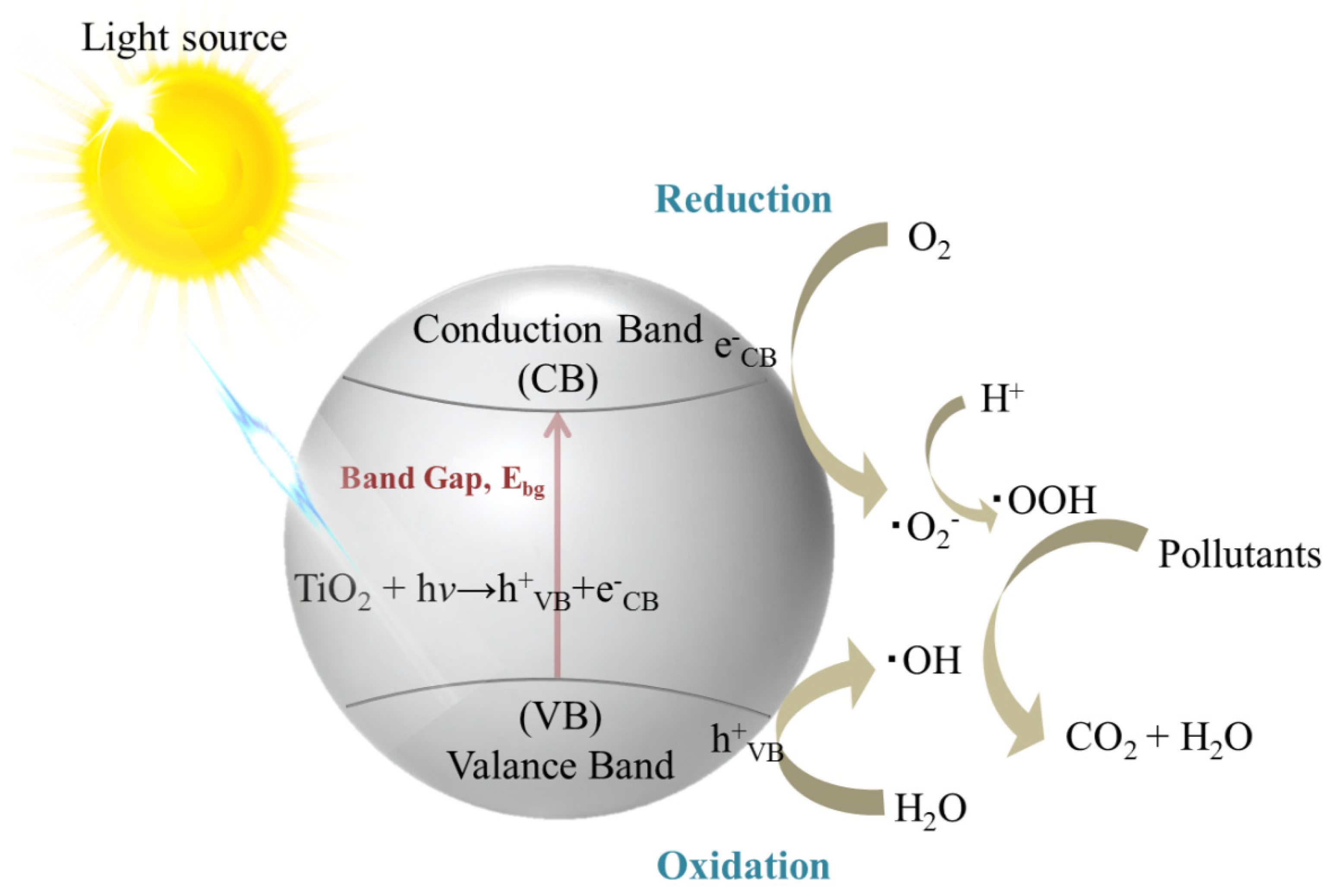
2.2. Photocatalysis
2.3. Synthesis
2.4. Properties between Different Polymorphs
3. Graphene Family Nanomaterials (GFN)
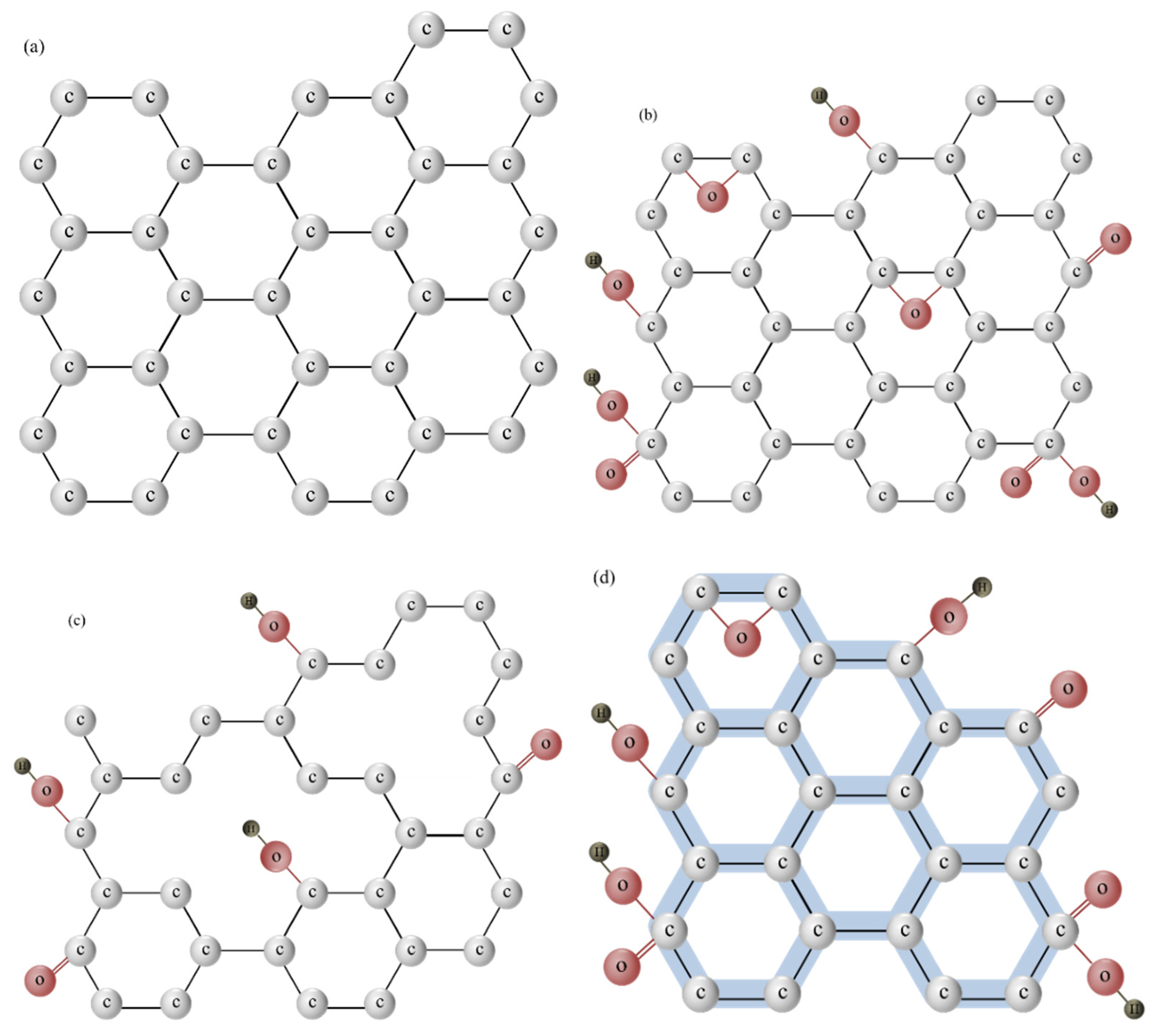
3.1. Graphene and Its Derivatives
3.2. Synthesis
3.3. Properties
| Properties | Graphene | GO | rGO | GQD |
|---|---|---|---|---|
| Functional group | No functional group | Epoxy, carboxyl, hydroxyl, and carboxyl | Epoxy, carboxyl, and hydroxyl | Epoxy, carbonyl, hydroxyl, and carboxyl |
| Nature | Hydrophobic | Hydrophilic | Hydrophilic | - |
| C:O ratio | No oxygen | 2-4 | 8-246 | 3 |
| d-spacing (nm) | 0.335 | 0.737 | 0.368 | 0.381 |
| Surface area (m2/g) | 2600 | 487 | 466 | - |
| Electron mobility (cm2V/s) | 10,000–50,000 | Insulator | 0.05–200 | - |
| Resistance (Ω) | 7200 | 0.514±0.236 | 2.01 ± 1.6 | - |
| Optics | 2.3% absorption(visible light) | - | ~20% adsorption (400–1800 nm) | - |
| Thermal conductivity (W/m·K) | ~5000 | 2.94 | 61.8 | - |
| Zeta potential (mV) | - | −33~−21.46 | −23.5~−26.5 | 8 |
| Young’s modulus | 1 | 0.2 | 0.25 | - |
| Reference | [79,103,104,105,106,107,108] | [103,104,109,110,111,112] | [77,103,104,110,113,114,115] | [103,104,113,116,117] |
4. GFN-TiO2
4.1. Synthesis
4.2. Characterization
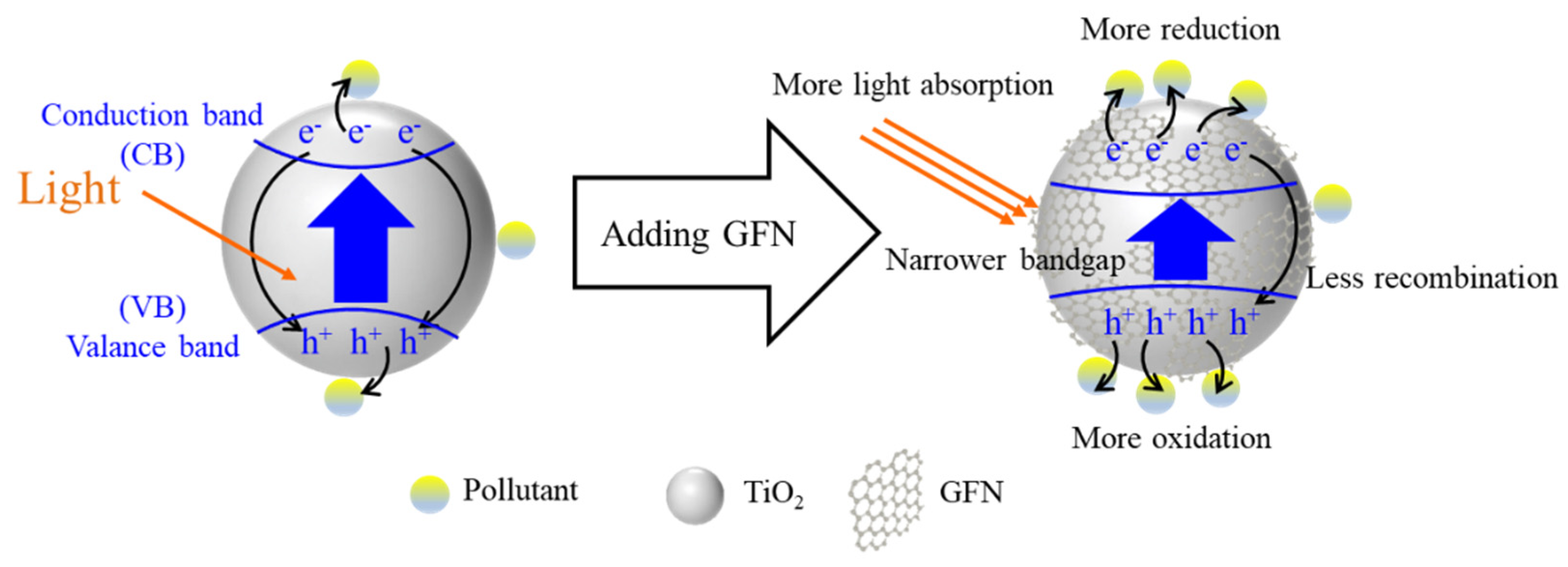
4.3. Photocatalysis Enhancement
5. Photocatalytic Removal of Pollutants
5.1. Water-Phase Pollutants
5.2. Air-Phase Pollutants
6. Conclusions and Future Work
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Prasad, S.; Singh, A.; Joshi, H. Ethanol as an alternative fuel from agricultural, industrial and urban residues. Resour. Conserv. Recycl. 2007, 50, 1–39. [Google Scholar] [CrossRef]
- Taghizadeh-Alisaraei, A.; Motevali, A.; Ghobadian, B. Ethanol production form date wastes: Adapted technologies, challenges, and global potential. Renew. Energy 2019, 143, 1094–1110. [Google Scholar] [CrossRef]
- Bai, Y.; Zhou, L.; Ma, K.; Zhou, H.; Xin, Y. Study on catalytic performances and reaction mechanisms of graphene electroactive membrane in wastewater treatment. Sep. Purif. Technol. 2019, 226, 278–285. [Google Scholar] [CrossRef]
- Shih, Y.-J.; Huang, C.-P.; Chan, Y.-H.; Huang, Y.-H. Electrochemical degradation of oxalic acid over highly reactive nano-textured γ-and α-MnO2/carbon electrode fabricated by KMnO4 reduction on loofah sponge-derived active carbon. J. Hazard. Mater. 2019, 379, 120759. [Google Scholar] [CrossRef] [PubMed]
- Wellia, D.V.; Xu, Q.C.; Sk, M.A.; Lim, K.H.; Lim, T.M.; Tan, T.T.Y. Experimental and theoretical studies of Fe-doped TiO2 films prepared by peroxo sol-gel method. Appl. Catal. A Gen. 2011, 401, 98–105. [Google Scholar] [CrossRef]
- Chen, W.-H.; Huang, J.-R.; Lin, C.-H.; Huang, C.-P. Catalytic degradation of chlorpheniramine over GO-Fe3O4 in the presence of H2O2 in water: The synergistic effect of adsorption. Sci. Total Environ. 2020, 736, 139468. [Google Scholar] [CrossRef]
- Lin, C.H.; Li, C.M.; Chen, C.H.; Chen, W.H. Removal of chlorpheniramine and variations of nitrosamine formation potentials in municipal wastewaters by adsorption onto the GO-Fe3O4. Environ. Sci. Pollut. Res. 2019, 26, 20701–20711. [Google Scholar] [CrossRef]
- Kisch, H. Semiconductor photocatalysis—Mechanistic and synthetic aspects. Angew. Chem. Int. Ed. 2013, 52, 812–847. [Google Scholar] [CrossRef] [PubMed]
- Ibhadon, A.; Fitzpatrick, P. Heterogeneous photocatalysis: Recent advances and applications. Catalysts 2013, 3, 189–218. [Google Scholar] [CrossRef]
- Gupta, S.M.; Tripathi, M. A review of TiO2 nanoparticles. Chin. Sci. Bull. 2011, 56, 1639. [Google Scholar] [CrossRef]
- Trouiller, B.; Reliene, R.; Westbrook, A.; Solaimani, P.; Schiestl, R.H. Titanium Dioxide Nanoparticles Induce DNA Damage and Genetic Instability In vivo in Mice. Cancer Res. 2009, 69, 8784–8789. [Google Scholar] [CrossRef]
- IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Carbon Black, Titanium Dioxide, and Talc; IARC: Lyon, France, 2010. [Google Scholar]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Frank, S.N.; Bard, A.J. Heterogeneous photocatalytic oxidation of cyanide ion in aqueous solutions at titanium dioxide powder. J. Am. Chem. Soc. 1977, 99, 303–304. [Google Scholar] [CrossRef]
- Frank, S.N.; Bard, A.J. Heterogeneous photocatalytic oxidation of cyanide and sulfite in aqueous solutions at semiconductor powders. J. Phys. Chem. 1977, 81, 1484–1488. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental applications of semiconductor photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Fox, M.A.; Dulay, M.T. Heterogeneous photocatalysis. Chem. Rev. 1993, 93, 341–357. [Google Scholar] [CrossRef]
- Carp, O.; Huisman, C.L.; Reller, A. Photoinduced reactivity of titanium dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Banerjee, S.; Pillai, S.C.; Falaras, P.; O’shea, K.E.; Byrne, J.A.; Dionysiou, D.D. New insights into the mechanism of visible light photocatalysis. J. Phys. Chem. Lett. 2014, 5, 2543–2554. [Google Scholar] [CrossRef]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.; Hamilton, J.W.; Byrne, J.A.; O’shea, K. A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl. Catal. B Environ. 2012, 125, 331–349. [Google Scholar] [CrossRef]
- Gaya, U.I.; Abdullah, A.H. Heterogeneous photocatalytic degradation of organic contaminants over titanium dioxide: A review of fundamentals, progress and problems. J. Photochem. Photobiol. C Photochem. Rev. 2008, 9, 1–12. [Google Scholar] [CrossRef]
- Banerjee, S.; Dionysiou, D.D.; Pillai, S.C. Self-cleaning applications of TiO2 by photo-induced hydrophilicity and photocatalysis. Appl. Catal. B Environ. 2015, 176, 396–428. [Google Scholar] [CrossRef]
- Keane, D.A.; McGuigan, K.G.; Ibáñez, P.F.; Polo-López, M.I.; Byrne, J.A.; Dunlop, P.S.; O’Shea, K.; Dionysiou, D.D.; Pillai, S.C. Solar photocatalysis for water disinfection: Materials and reactor design. Catal. Sci. Technol. 2014, 4, 1211–1226. [Google Scholar] [CrossRef]
- Fujishima, A. Photocatalytic and self-cleaning functions of TiO2 coatings. In Sustainable Energy and Environmental Technologies; World Scientific: Singapore, 2001; pp. 1–5. [Google Scholar]
- Chen, X.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications, and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.D.; Singh, D.; Saini, K.; Kant, C.; Sharma, V.; Jain, S.; Sharma, C. Sol–gel-derived super-hydrophilic nickel doped TiO2 film as active photo-catalyst. Appl. Catal. A Gen. 2006, 314, 40–46. [Google Scholar] [CrossRef]
- Catauro, M.; Tranquillo, E.; Dal Poggetto, G.; Pasquali, M.; Dell’Era, A.; Vecchio Ciprioti, S. Influence of the heat treatment on the particles size and on the crystalline phase of TiO2 synthesized by the sol-gel method. Materials 2018, 11, 2364. [Google Scholar] [CrossRef] [PubMed]
- Dubey, R.; Krishnamurthy, K.V.; Singh, S. Experimental studies of TiO2 nanoparticles synthesized by sol-gel and solvothermal routes for DSSCs application. Results Phys. 2019, 14, 102390. [Google Scholar] [CrossRef]
- Li, G.; Li, L.; Boerio-Goates, J.; Woodfield, B.F. High purity anatase TiO2 nanocrystals: Near room-temperature synthesis, grain growth kinetics, and surface hydration chemistry. J. Am. Chem. Soc. 2005, 127, 8659–8666. [Google Scholar] [CrossRef]
- Hirano, M.; Nakahara, C.; Ota, K.; Tanaike, O.; Inagaki, M. Photoactivity and phase stability of ZrO2-doped anatase-type TiO2 directly formed as nanometer-sized particles by hydrolysis under hydrothermal conditions. J. Solid State Chem. 2003, 170, 39–47. [Google Scholar] [CrossRef]
- Wang, L.Q.; Yang, X.N.; Zhao, X.L.; Zhang, R.J.; Yang, Y.L. Preparation of TiO2 nanoparticles in the solvothermal method. In Proceedings of the Key Engineering Materials; Trans Tech Publications Ltd.: Freienbach, Switzerland, 2011; pp. 1672–1677. [Google Scholar]
- Hong, S.-S.; Lee, M.S.; Park, S.S.; Lee, G.-D. Synthesis of nanosized TiO2/SiO2 particles in the microemulsion and their photocatalytic activity on the decomposition of p-nitrophenol. Catal. Today 2003, 87, 99–105. [Google Scholar] [CrossRef]
- Volz, H.G. Pigments, Inorganic Ullman’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Teleki, A.; Pratsinis, S.E.; Kalyanasundaram, K.; Gouma, P. Sensing of organic vapors by flame-made TiO2 nanoparticles. Sens. Actuators B Chem. 2006, 119, 683–690. [Google Scholar] [CrossRef]
- Jung, H.S.; Kim, H. Origin of Low Photocatalytic Activity of Rutile TiO2. Electron. Mater. Lett. 2009, 5, 73–76. [Google Scholar] [CrossRef]
- Luttrell, T.; Halpegamage, S.; Tao, J.G.; Kramer, A.; Sutter, E.; Batzill, M. Why is anatase a better photocatalyst than rutile?—Model studies on epitaxial TiO2 films. Sci. Rep. 2014, 4, 4043. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Gao, L.; Guo, J. Effects of calcination on the photocatalytic properties of nanosized TiO2 powders prepared by TiCl4 hydrolysis. Appl. Catal. B Environ. 2000, 26, 207–215. [Google Scholar] [CrossRef]
- Kesselman, J.M.; Shreve, G.A.; Hoffmann, M.R.; Lewis, N.S. Flux-matching conditions at TiO2 photoelectrodes: Is interfacial electron transfer to O2 rate-limiting in the TiO2-catalyzed photochemical degradation of organics? J. Phys. Chem. 1994, 98, 13385–13395. [Google Scholar] [CrossRef]
- Hanaor, D.A.; Sorrell, C.C. Review of the anatase to rutile phase transformation. J. Mater. Sci. 2011, 46, 855–874. [Google Scholar] [CrossRef]
- Sclafani, A.; Herrmann, J. Comparison of the photoelectronic and photocatalytic activities of various anatase and rutile forms of titania in pure liquid organic phases and in aqueous solutions. J. Phys. Chem. 1996, 100, 13655–13661. [Google Scholar] [CrossRef]
- Asahi, R.; Taga, Y.; Mannstadt, W.; Freeman, A.J. Electronic and optical properties of anatase TiO2. Phys. Rev. B 2000, 61, 7459. [Google Scholar] [CrossRef]
- Koelsch, M.; Cassaignon, S.; Minh, C.T.T.; Guillemoles, J.-F.; Jolivet, J.-P. Electrochemical comparative study of titania (anatase, brookite and rutile) nanoparticles synthesized in aqueous medium. Thin Solid Film. 2004, 451, 86–92. [Google Scholar] [CrossRef]
- Amtout, A.; Leonelli, R. Optical properties of rutile near its fundamental band gap. Phys. Rev. B 1995, 51, 6842. [Google Scholar] [CrossRef]
- Zhu, S.-C.; Xie, S.-H.; Liu, Z.-P. Nature of rutile nuclei in anatase-to-rutile phase transition. J. Am. Chem. Soc. 2015, 137, 11532–11539. [Google Scholar] [CrossRef]
- Zhang, H.; Banfield, J.F. Structural characteristics and mechanical and thermodynamic properties of nanocrystalline TiO2. Chem. Rev. 2014, 114, 9613–9644. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhou, P.; Liu, J.; Yu, J. New understanding of the difference of photocatalytic activity among anatase, rutile and brookite TiO2. Phys. Chem. Chem. Phys. 2014, 16, 20382–20386. [Google Scholar] [CrossRef] [PubMed]
- Cromer, D.T.; Herrington, K. The structures of anatase and rutile. J. Am. Chem. Soc. 1955, 77, 4708–4709. [Google Scholar] [CrossRef]
- Mo, S.-D.; Ching, W. Electronic and optical properties of three phases of titanium dioxide: Rutile, anatase, and brookite. Phys. Rev. B 1995, 51, 13023. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.L.; Liu, S.H.; Dai, Z.D.; He, Y.P.; Song, X.Z.; Tan, Z.Q. Titanium Dioxide: From Engineering to Applications. Catalysts 2019, 9, 191. [Google Scholar] [CrossRef]
- Hamal, D.B.; Klabunde, K.J. Synthesis, characterization, and visible light activity of new nanoparticle photocatalysts based on silver, carbon, and sulfur-doped TiO2. J. Colloid Interface Sci. 2007, 311, 514–522. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Suvarnaphaet, P.; Pechprasarn, S. Graphene-based materials for biosensors: A review. Sensors 2017, 17, 2161. [Google Scholar] [CrossRef]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and graphene oxide: Synthesis, properties, and applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef]
- Li, C.M.; Chen, C.H.; Chen, W.H. Different influences of nanopore dimension and pH between chlorpheniramine adsorptions on graphene oxide-iron oxide suspension and particle. Chem. Eng. J. 2017, 307, 447–455. [Google Scholar] [CrossRef]
- Bo, Z.; Shuai, X.R.; Mao, S.; Yang, H.C.; Qian, J.J.; Chen, J.H.; Yan, J.H.; Cen, K. Green preparation of reduced graphene oxide for sensing and energy storage applications. Sci. Rep. 2014, 4, 4684. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.B.; Zeng, T.H.; Hofmann, M.; Burcombe, E.; Wei, J.; Jiang, R.R.; Kong, J.; Chen, Y. Antibacterial Activity of Graphite, Graphite Oxide, Graphene Oxide, and Reduced Graphene Oxide: Membrane and Oxidative Stress. ACS Nano 2011, 5, 6971–6980. [Google Scholar] [CrossRef] [PubMed]
- Zheng, P.; Wu, N.Q. Fluorescence and Sensing Applications of Graphene Oxide and Graphene Quantum Dots: A Review. Chem. Asian J. 2017, 12, 2343–2353. [Google Scholar] [CrossRef] [PubMed]
- Lee, X.J.; Hiew, B.Y.Z.; Lai, K.C.; Lee, L.Y.; Gan, S.; Thangalazhy-Gopakumar, S.; Rigby, S. Review on graphene and its derivatives: Synthesis methods and potential industrial implementation. J. Taiwan Inst. Chem. Eng. 2019, 98, 163–180. [Google Scholar] [CrossRef]
- Wu, Z.S.; Ren, W.C.; Gao, L.B.; Liu, B.L.; Jiang, C.B.; Cheng, H.M. Synthesis of high-quality graphene with a pre-determined number of layers. Carbon 2009, 47, 493–499. [Google Scholar] [CrossRef]
- Shashurin, A.; Keidar, M. Synthesis of 2D materials in arc plasmas. J. Phys. D Appl. Phys. 2015, 48, 314007. [Google Scholar] [CrossRef]
- Kosynkin, D.V.; Higginbotham, A.L.; Sinitskii, A.; Lomeda, J.R.; Dimiev, A.; Price, B.K.; Tour, J.M. Longitudinal unzipping of carbon nanotubes to form graphene nanoribbons. Nature 2009, 458, 872. [Google Scholar] [CrossRef] [PubMed]
- Mishra, N.; Boeckl, J.; Motta, N.; Iacopi, F. Graphene growth on silicon carbide: A review. Phys. Status Solidi 2016, 213, 2277–2289. [Google Scholar] [CrossRef]
- Dato, A.; Radmilovic, V.; Lee, Z.; Phillips, J.; Frenklach, M. Substrate-free gas-phase synthesis of graphene sheets. Nano Lett. 2008, 8, 2012–2016. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, R.; Wu, J.; Jiang, X.; Cao, P.; Hu, X.; Pan, T.; Qiu, C.; Yang, J.; Song, Y. Bottom-up fabrication of graphene on silicon/silica substrate via a facile soft-hard template approach. Sci. Rep. 2015, 5, 13480. [Google Scholar] [CrossRef]
- Yang, X.; Dou, X.; Rouhanipour, A.; Zhi, L.; Räder, H.J.; Müllen, K. Two-dimensional graphene nanoribbons. J. Am. Chem. Soc. 2008, 130, 4216–4217. [Google Scholar] [CrossRef] [PubMed]
- Brodie, B.C. On the Atomic Weight of Graphite. Philos. Trans. R. Soc. Lond. 1859, 149, 249–259. [Google Scholar]
- Staudenmaier, L. Verfahren zur Darstellung der Graphitsäure. Ber. Dtsch. Chem. Ges. 1898, 31, 1481–1499. [Google Scholar] [CrossRef]
- Hofmann, U.; König, E. Untersuchungen über Graphitoxyd. Z. Anorg. Allg. Chem. 1937, 234, 311–336. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Chen, J.; Yao, B.; Li, C.; Shi, G. An improved Hummers method for eco-friendly synthesis of graphene oxide. Carbon 2013, 64, 225–229. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, B.; Bulin, C.; Li, R.; Xing, R. High-efficient synthesis of graphene oxide based on improved hummers method. Sci. Rep. 2016, 6, 36143. [Google Scholar] [CrossRef]
- Zaaba, N.; Foo, K.; Hashim, U.; Tan, S.; Liu, W.-W.; Voon, C. Synthesis of graphene oxide using modified hummers method: Solvent influence. Procedia Eng. 2017, 184, 469–477. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved synthesis of graphene oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef]
- Peng, L.; Xu, Z.; Liu, Z.; Wei, Y.; Sun, H.; Li, Z.; Zhao, X.; Gao, C. An iron-based green approach to 1-h production of single-layer graphene oxide. Nat. Commun. 2015, 6, 5716. [Google Scholar] [CrossRef]
- Gómez-Navarro, C.; Weitz, R.T.; Bittner, A.M.; Scolari, M.; Mews, A.; Burghard, M.; Kern, K. Electronic transport properties of individual chemically reduced graphene oxide sheets. Nano Lett. 2007, 7, 3499–3503. [Google Scholar] [CrossRef]
- Zhu, Y.; Stoller, M.D.; Cai, W.; Velamakanni, A.; Piner, R.D.; Chen, D.; Ruoff, R.S. Exfoliation of graphite oxide in propylene carbonate and thermal reduction of the resulting graphene oxide platelets. ACS Nano 2010, 4, 1227–1233. [Google Scholar] [CrossRef]
- Stankovich, S.; Dikin, D.A.; Piner, R.D.; Kohlhaas, K.A.; Kleinhammes, A.; Jia, Y.; Wu, Y.; Nguyen, S.T.; Ruoff, R.S. Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 2007, 45, 1558–1565. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, H.; Shen, G.; Cheng, P.; Zhang, J.; Guo, S. Reduction of graphene oxide via L-ascorbic acid. Chem. Commun. 2010, 46, 1112–1114. [Google Scholar] [CrossRef] [PubMed]
- Stankovich, S.; Dikin, D.A.; Dommett, G.H.; Kohlhaas, K.M.; Zimney, E.J.; Stach, E.A.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Graphene-based composite materials. Nature 2006, 442, 282–286. [Google Scholar] [CrossRef]
- Fernandez-Merino, M.J.; Guardia, L.; Paredes, J.I.; Villar-Rodil, S.; Solis-Fernandez, P.; Martinez-Alonso, A.; Tascon, J.M.D. Vitamin C Is an Ideal Substitute for Hydrazine in the Reduction of Graphene Oxide Suspensions. J. Phys. Chem. C 2010, 114, 6426–6432. [Google Scholar] [CrossRef]
- Guo, H.-L.; Wang, X.-F.; Qian, Q.-Y.; Wang, F.-B.; Xia, X.-H. A green approach to the synthesis of graphene nanosheets. ACS Nano 2009, 3, 2653–2659. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Yan, L.; Bangal, P.R. Preparation of graphene by the rapid and mild thermal reduction of graphene oxide induced by microwaves. Carbon 2010, 48, 1146–1152. [Google Scholar] [CrossRef]
- Pan, D.Y.; Zhang, J.C.; Li, Z.; Wu, M.H. Hydrothermal Route for Cutting Graphene Sheets into Blue-Luminescent Graphene Quantum Dots. Adv. Mater. 2010, 22, 734. [Google Scholar] [CrossRef]
- Liang, W.X.; Bunker, C.E.; Sun, Y.P. Carbon Dots: Zero-Dimensional Carbon Allotrope with Unique Photoinduced Redox Characteristics. Acs Omega 2020, 5, 965–971. [Google Scholar] [CrossRef]
- Wang, Z.F.; Yu, J.F.; Zhang, X.; Li, N.; Liu, B.; Li, Y.Y.; Wang, Y.H.; Wang, W.X.; Li, Y.Z.; Zhang, L.C.; et al. Large-Scale and Controllable Synthesis of Graphene Quantum Dots from Rice Husk Biomass: A Comprehensive Utilization Strategy. ACS Appl. Mater. Interfaces 2016, 8, 1434–1439. [Google Scholar] [CrossRef] [PubMed]
- Van Tam, T.; Kang, S.G.; Babu, K.F.; Oh, E.-S.; Lee, S.G.; Choi, W.M. Synthesis of B-doped graphene quantum dots as a metal-free electrocatalyst for the oxygen reduction reaction. J. Mater. Chem. A 2017, 5, 10537–10543. [Google Scholar] [CrossRef]
- Zhang, J.; Ma, Y.-Q.; Li, N.; Zhu, J.-L.; Zhang, T.; Zhang, W.; Liu, B. Preparation of graphene quantum dots and their application in cell imaging. J. Nanomater. 2016, 2016, 9245865. [Google Scholar] [CrossRef][Green Version]
- Li, Y.; Shu, H.; Niu, X.; Wang, J. Electronic and optical properties of edge-functionalized graphene quantum dots and the underlying mechanism. J. Phys. Chem. C 2015, 119, 24950–24957. [Google Scholar] [CrossRef]
- Lu, J.; Yang, J.-X.; Wang, J.; Lim, A.; Wang, S.; Loh, K.P. One-pot synthesis of fluorescent carbon nanoribbons, nanoparticles, and graphene by the exfoliation of graphite in ionic liquids. ACS Nano 2009, 3, 2367–2375. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xin, X.; Lin, Z. Cu2ZnSnS4 nanocrystals and graphene quantum dots for photovoltaics. Nanoscale 2011, 3, 3040–3048. [Google Scholar] [CrossRef]
- Zheng, X.T.; Ananthanarayanan, A.; Luo, K.Q.; Chen, P. Glowing graphene quantum dots and carbon dots: Properties, syntheses, and biological applications. Small 2015, 11, 1620–1636. [Google Scholar] [CrossRef]
- Peng, J.; Zhao, Z.; Zheng, M.; Su, B.; Chen, X.; Chen, X. Electrochemical synthesis of phosphorus and sulfur co-doped graphene quantum dots as efficient electrochemiluminescent immunomarkers for monitoring okadaic acid. Sens. Actuators B Chem. 2020, 304, 127383. [Google Scholar] [CrossRef]
- Wang, L.; Li, W.; Wu, B.; Li, Z.; Pan, D.; Wu, M. Room-temperature synthesis of graphene quantum dots via electron-beam irradiation and their application in cell imaging. Chem. Eng. J. 2017, 309, 374–380. [Google Scholar] [CrossRef]
- Singh, R.; Kumar, R.; Singh, D.; Savu, R.; Moshkalev, S. Progress in microwave-assisted synthesis of quantum dots (graphene/carbon/semiconducting) for bioapplications: A review. Mater. Today Chem. 2019, 12, 282–314. [Google Scholar] [CrossRef]
- Hassan, M.; Haque, E.; Reddy, K.R.; Minett, A.I.; Chen, J.; Gomes, V.G. Edge-enriched graphene quantum dots for enhanced photo-luminescence and supercapacitance. Nanoscale 2014, 6, 11988–11994. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.U.; Jo, W.-K. FeWO4/g-C3N4 heterostructures decorated with N-doped graphene quantum dots prepared under various sonication conditions for efficient removal of noxious vapors. Ceram. Int. 2020, 46, 11346–11356. [Google Scholar] [CrossRef]
- Li, R.; Liu, Y.; Li, Z.; Shen, J.; Yang, Y.; Cui, X.; Yang, G. Bottom-up fabrication of single-layered nitrogen-doped graphene quantum dots through intermolecular carbonization arrayed in a 2D plane. Chem. Eur. J. 2016, 22, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.; Hai, X.; Chen, X.-W.; Wang, J.-H. Simultaneously fabrication of free and solidified N, S-doped graphene quantum dots via a facile solvent-free synthesis route for fluorescent detection. Talanta 2017, 168, 269–278. [Google Scholar] [CrossRef]
- Lu, J.; Yeo, P.S.E.; Gan, C.K.; Wu, P.; Loh, K.P. Transforming C 60 molecules into graphene quantum dots. Nat. Nanotechnol. 2011, 6, 247. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef]
- Nethravathi, C.; Rajamathi, J.T.; Ravishankar, N.; Shivakumara, C.; Rajamathi, M. Graphite oxide-intercalated anionic clay and its decomposition to graphene− inorganic material nanocomposites. Langmuir 2008, 24, 8240–8244. [Google Scholar] [CrossRef]
- Lin, C.H.; Chen, W.H. Influence of water, H2O2, H2SO4, and NaOH filtration on the surface characteristics of a graphene oxide-iron (GO-Fe) membrane. Sep. Purif. Technol. 2021, 262, 118317. [Google Scholar] [CrossRef]
- Carmalin, A.S.; Lima, E.C.; Allaudeen, N.; Rajan, S. Application of graphene based materials for adsorption of pharmaceutical traces from water and wastewater—A review. Desalination Water Treat. 2016, 57, 27573–27586. [Google Scholar] [CrossRef]
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater. Sci. 2011, 56, 1178–1271. [Google Scholar] [CrossRef]
- Bolotin, K.I.; Sikes, K.J.; Jiang, Z.; Klima, M.; Fudenberg, G.; Hone, J.; Kim, P.; Stormer, H. Ultrahigh electron mobility in suspended graphene. Solid State Commun. 2008, 146, 351–355. [Google Scholar] [CrossRef]
- Nair, R.R.; Blake, P.; Grigorenko, A.N.; Novoselov, K.S.; Booth, T.J.; Stauber, T.; Peres, N.M.; Geim, A.K. Fine structure constant defines visual transparency of graphene. Science 2008, 320, 1308. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-L.; Kudin, K.N.; McAllister, M.J.; Prud’homme, R.K.; Aksay, I.A.; Car, R. Oxygen-driven unzipping of graphitic materials. Phys. Rev. Lett. 2006, 96, 176101. [Google Scholar] [CrossRef] [PubMed]
- Seifvand, N.; Kowsari, E. Novel TiO2/graphene oxide functionalized with a cobalt complex for significant degradation of NOx and CO. RSC Adv. 2015, 5, 93706–93716. [Google Scholar] [CrossRef]
- Renteria, J.D.; Ramirez, S.; Malekpour, H.; Alonso, B.; Centeno, A.; Zurutuza, A.; Cocemasov, A.I.; Nika, D.L.; Balandin, A.A. Strongly anisotropic thermal conductivity of free-standing reduced graphene oxide films annealed at high temperature. Adv. Funct. Mater. 2015, 25, 4664–4672. [Google Scholar] [CrossRef]
- Baskoro, F.; Wong, C.-B.; Kumar, S.R.; Chang, C.-W.; Chen, C.-H.; Chen, D.W.; Lue, S.J. Graphene oxide-cation interaction: Inter-layer spacing and zeta potential changes in response to various salt solutions. J. Membr. Sci. 2018, 554, 253–263. [Google Scholar] [CrossRef]
- Suk, J.W.; Piner, R.D.; An, J.; Ruoff, R.S. Mechanical properties of monolayer graphene oxide. ACS Nano 2010, 4, 6557–6564. [Google Scholar] [CrossRef]
- Shen, J.; Zhu, Y.; Yang, X.; Zong, J.; Zhang, J.; Li, C. One-pot hydrothermal synthesis of graphene quantum dots surface-passivated by polyethylene glycol and their photoelectric conversion under near-infrared light. New J. Chem. 2012, 36, 97–101. [Google Scholar] [CrossRef]
- Becerril, H.A.; Mao, J.; Liu, Z.; Stoltenberg, R.M.; Bao, Z.; Chen, Y. Evaluation of solution-processed reduced graphene oxide films as transparent conductors. ACS Nano 2008, 2, 463–470. [Google Scholar] [CrossRef]
- Sadhukhan, S.; Ghosh, T.K.; Rana, D.; Roy, I.; Bhattacharyya, A.; Sarkar, G.; Chakraborty, M.; Chattopadhyay, D. Studies on synthesis of reduced graphene oxide (RGO) via green route and its electrical property. Mater. Res. Bull. 2016, 79, 41–51. [Google Scholar] [CrossRef]
- Wang, S.; Cole, I.S.; Zhao, D.; Li, Q. The dual roles of functional groups in the photoluminescence of graphene quantum dots. Nanoscale 2016, 8, 7449–7458. [Google Scholar] [CrossRef]
- Zhu, S.; Zhang, J.; Qiao, C.; Tang, S.; Li, Y.; Yuan, W.; Li, B.; Tian, L.; Liu, F.; Hu, R. Strongly green-photoluminescent graphene quantum dots for bioimaging applications. Chem. Commun. 2011, 47, 6858–6860. [Google Scholar] [CrossRef]
- Sun, Y.; Qi, Y.; Peng, B.; Li, W. NTCP-Reconstituted In Vitro HBV Infection System. In Hepatitis B Virus; Guo, H., Cuconati, A., Eds.; Humana Press: New York, NY, USA, 2016; Volume 1540. [Google Scholar]
- Neikov, O.D.; Naboychenko, S.S.; Yefimov, N.A. Handbook of Non-Ferrous Metal Powders: Technologies and Applications; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Tajik, S.; Dourandish, Z.; Zhang, K.; Beitollahi, H.; Le, Q.V.; Jang, H.W.; Shokouhimehr, M. Carbon and graphene quantum dots: A review on syntheses, characterization, biological and sensing applications for neurotransmitter determination. RSC Adv. 2020, 10, 15406–15429. [Google Scholar] [CrossRef]
- Zhang, Y.; Jiang, Z.; Huang, J.; Lim, L.Y.; Li, W.; Deng, J.; Gong, D.; Tang, Y.; Lai, Y.; Chen, Z. Titanate and titania nanostructured materials for environmental and energy applications: A review. RSC Adv. 2015, 5, 79479–79510. [Google Scholar] [CrossRef]
- Jiang, L.C.; Zhang, W.D. Electrodeposition of TiO2 nanoparticles on multiwalled carbon nanotube arrays for hydrogen peroxide sensing. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 2009, 21, 988–993. [Google Scholar]
- Cheng, Y.H.; Huang, Y.; Kanhere, P.D.; Subramaniam, V.P.; Gong, D.; Zhang, S.; Highfield, J.; Schreyer, M.K.; Chen, Z. Dual-Phase Titanate/Anatase with Nitrogen Doping for Enhanced Degradation of Organic Dye under Visible Light. Chem. Eur. J. 2011, 17, 2575–2578. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Liu, Z.; Pourpoint, F.; Armstrong, A.R.; Grey, C.P.; Bruce, P.G. Nanoparticulate TiO2 (B): An anode for lithium-ion batteries. Angew. Chem. 2012, 124, 2206–2209. [Google Scholar] [CrossRef]
- Rongan, H.; Haijuan, L.; Huimin, L.; Difa, X.; Liuyang, Z. S-scheme photocatalyst Bi2O3/TiO2 nanofiber with improved photocatalytic performance. J. Mater. Sci. Technol. 2020, 52, 145–151. [Google Scholar] [CrossRef]
- Xu, D.; Li, L.; He, R.; Qi, L.; Zhang, L.; Cheng, B. Noble metal-free RGO/TiO2 composite nanofiber with enhanced photocatalytic H2-production performance. Appl. Surf. Sci. 2018, 434, 620–625. [Google Scholar] [CrossRef]
- Wu, M.-C.; Wu, P.-Y.; Lin, T.-H.; Lin, T.-F. Photocatalytic performance of Cu-doped TiO2 nanofibers treated by the hydrothermal synthesis and air-thermal treatment. Appl. Surf. Sci. 2018, 430, 390–398. [Google Scholar] [CrossRef]
- Meng, A.; Zhang, J.; Xu, D.; Cheng, B.; Yu, J. Enhanced photocatalytic H2-production activity of anatase TiO2 nanosheet by selectively depositing dual-cocatalysts on {101} and {001} facets. Appl. Catal. B Environ. 2016, 198, 286–294. [Google Scholar] [CrossRef]
- Chen, J.; Wang, M.; Han, J.; Guo, R. TiO2 nanosheet/NiO nanorod hierarchical nanostructures: P–n heterojunctions towards efficient photocatalysis. J. Colloid Interface Sci. 2020, 562, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Safajou, H.; Khojasteh, H.; Salavati-Niasari, M.; Mortazavi-Derazkola, S. Enhanced photocatalytic degradation of dyes over graphene/Pd/TiO2 nanocomposites: TiO2 nanowires versus TiO2 nanoparticles. J. Colloid Interface Sci. 2017, 498, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wu, G.; Yuan, X.; Xie, T.; Zhang, L. Fabrication and optical properties of TiO2 nanowire arrays made by sol–gel electrophoresis deposition into anodic alumina membranes. J. Phys. Condens. Matter 2003, 15, 2917. [Google Scholar] [CrossRef]
- Makal, P.; Das, D. Self-doped TiO2 nanowires in TiO2-B single phase, TiO2-B/anatase and TiO2-anatase/rutile heterojunctions demonstrating individual superiority in photocatalytic activity under visible and UV light. Appl. Surf. Sci. 2018, 455, 1106–1115. [Google Scholar] [CrossRef]
- Al-Hajji, L.A.; Ismail, A.A.; Al-Hazza, A.; Ahmed, S.A.; Alsaidi, M.; Almutawa, F.; Bumajdad, A. Impact of calcination of hydrothermally synthesized TiO2 nanowires on their photocatalytic efficiency. J. Mol. Struct. 2020, 1200, 127153. [Google Scholar] [CrossRef]
- Dou, H.; Long, D.; Rao, X.; Zhang, Y.; Qin, Y.; Pan, F.; Wu, K. Photocatalytic Degradation Kinetics of Gaseous Formaldehyde Flow Using TiO2 Nanowires. ACS Sustain. Chem. Eng. 2019, 7, 4456–4465. [Google Scholar] [CrossRef]
- Lim, Y.W.L.; Tang, Y.; Cheng, Y.H.; Chen, Z. Morphology, crystal structure and adsorption performance of hydrothermally synthesized titania and titanate nanostructures. Nanoscale 2010, 2, 2751–2757. [Google Scholar] [CrossRef]
- Miao, L.; Tanemura, S.; Toh, S.; Kaneko, K.; Tanemura, M. Fabrication, characterization and Raman study of anatase-TiO2 nanorods by a heating-sol–gel template process. J. Cryst. Growth 2004, 264, 246–252. [Google Scholar] [CrossRef]
- Liu, B.; Wang, J.; Yang, J.; Zhao, X. Charge carrier interfacial transfer pathways from TiO2 and Au/TiO2 nanorod arrays to electrolyte and the association with photocatalysis. Appl. Surf. Sci. 2019, 464, 367–375. [Google Scholar] [CrossRef]
- Chen, W.; Wang, Y.; Liu, S.; Gao, L.; Mao, L.; Fan, Z.; Shangguan, W.; Jiang, Z. Non-noble metal Cu as a cocatalyst on TiO2 nanorod for highly efficient photocatalytic hydrogen production. Appl. Surf. Sci. 2018, 445, 527–534. [Google Scholar] [CrossRef]
- Ge, M.-Z.; Li, S.-H.; Huang, J.-Y.; Zhang, K.-Q.; Al-Deyab, S.S.; Lai, Y.-K. TiO2 nanotube arrays loaded with reduced graphene oxide films: Facile hybridization and promising photocatalytic application. J. Mater. Chem. A 2015, 3, 3491–3499. [Google Scholar] [CrossRef]
- Kasuga, T.; Hiramatsu, M.; Hoson, A.; Sekino, T.; Niihara, K. Formation of titanium oxide nanotube. Langmuir 1998, 14, 3160–3163. [Google Scholar] [CrossRef]
- Huy, T.H.; Bui, D.P.; Kang, F.; Wang, Y.-F.; Liu, S.-H.; Thi, C.M.; You, S.-J.; Chang, G.-M.; Pham, V.V. SnO2/TiO2 nanotube heterojunction: The first investigation of NO degradation by visible light-driven photocatalysis. Chemosphere 2019, 215, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Zhou, J.; Liu, Y.; Yang, Y.; Zheng, S.; Wang, Q. Constructing Ag2O nanoparticle modified TiO2 nanotube arrays for enhanced photocatalytic performances. J. Alloys Compd. 2020, 849, 156493. [Google Scholar] [CrossRef]
- Parida, K.M.; Mohapatra, L. Carbonate intercalated Zn/Fe layered double hydroxide: A novel photocatalyst for the enhanced photo degradation of azo dyes. Chem. Eng. J. 2012, 179, 131–139. [Google Scholar] [CrossRef]
- Tobajas, M.; Belver, C.; Rodriguez, J.J. Degradation of emerging pollutants in water under solar irradiation using novel TiO2-ZnO/clay nanoarchitectures. Chem. Eng. J. 2017, 309, 596–606. [Google Scholar] [CrossRef]
- Perera, S.D.; Mariano, R.G.; Vu, K.; Nour, N.; Seitz, O.; Chabal, Y.; Balkus, K.J., Jr. Hydrothermal synthesis of graphene-TiO2 nanotube composites with enhanced photocatalytic activity. ACS Catal. 2012, 2, 949–956. [Google Scholar] [CrossRef]
- Cho, T.-Y.; Han, C.-W.; Jun, Y.; Yoon, S.-G. Formation of artificial pores in nano-TiO 2 photo-electrode films using acetylene-black for high-efficiency, dye-sensitized solar cells. Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Wei, N.; Cui, H.; Wang, C.; Zhang, G.; Song, Q.; Sun, W.; Song, X.; Sun, M.; Tian, J. Bi2O3 nanoparticles incorporated porous TiO2 films as an effective p-n junction with enhanced photocatalytic activity. J. Am. Ceram. Soc. 2017, 100, 1339–1349. [Google Scholar] [CrossRef]
- Lv, X.; Wang, T.; Jiang, W. Preparation of Ag@AgCl/g-C3N4/TiO2 porous ceramic films with enhanced photocatalysis performance and self-cleaning effect. Ceram. Int. 2018, 44, 9326–9337. [Google Scholar] [CrossRef]
- Kim, S.; Chang, H.-K.; Kim, K.B.; Kim, H.-J.; Lee, H.-N.; Park, T.J.; Park, Y.M. Highly Porous SnO2/TiO2 Heterojunction Thin-Film Photocatalyst Using Gas-Flow Thermal Evaporation and Atomic Layer Deposition. Catalysts 2021, 11, 1144. [Google Scholar] [CrossRef]
- Low, F.W.; Lai, C.W.; Hamid, S.B.A. Surface modification of reduced graphene oxide film by Ti ion implantation technique for high dye-sensitized solar cells performance. Ceram. Int. 2017, 43, 625–633. [Google Scholar] [CrossRef]
- Jo, W.-K.; Kang, H.-J. Titanium dioxide–graphene oxide composites with different ratios supported by Pyrex tube for photocatalysis of toxic aromatic vapors. Powder Technol. 2013, 250, 115–121. [Google Scholar] [CrossRef]
- Chowdhury, S.; Parshetti, G.K.; Balasubramanian, R. Post-combustion CO2 capture using mesoporous TiO2/graphene oxide nanocomposites. Chem. Eng. J. 2015, 263, 374–384. [Google Scholar] [CrossRef]
- Zhang, C.; Chaudhary, U.; Lahiri, D.; Godavarty, A.; Agarwal, A. Photocatalytic activity of spark plasma sintered TiO2–graphene nanoplatelet composite. Scr. Mater. 2013, 68, 719–722. [Google Scholar] [CrossRef]
- Liang, D.; Cui, C.; Hu, H.; Wang, Y.; Xu, S.; Ying, B.; Li, P.; Lu, B.; Shen, H. One-step hydrothermal synthesis of anatase TiO2/reduced graphene oxide nanocomposites with enhanced photocatalytic activity. J. Alloys Compd. 2014, 582, 236–240. [Google Scholar] [CrossRef]
- Shen, J.; Yan, B.; Shi, M.; Ma, H.; Li, N.; Ye, M. One step hydrothermal synthesis of TiO2-reduced graphene oxide sheets. J. Mater. Chem. 2011, 21, 3415–3421. [Google Scholar] [CrossRef]
- Chang, B.Y.S.; Huang, N.M.; An’amt, M.N.; Marlinda, A.R.; Norazriena, Y.; Muhamad, M.R.; Harrison, I.; Lim, H.N.; Chia, C.H. Facile hydrothermal preparation of titanium dioxide decorated reduced graphene oxide nanocomposite. Int. J. Nanomed. 2012, 7, 3379. [Google Scholar]
- Li, W.; Wang, F.; Feng, S.; Wang, J.; Sun, Z.; Li, B.; Li, Y.; Yang, J.; Elzatahry, A.A.; Xia, Y. Sol–gel design strategy for ultradispersed TiO2 nanoparticles on graphene for high-performance lithium ion batteries. J. Am. Chem. Soc. 2013, 135, 18300–18303. [Google Scholar] [CrossRef]
- Li, W.; Wang, F.; Liu, Y.; Wang, J.; Yang, J.; Zhang, L.; Elzatahry, A.A.; Al-Dahyan, D.; Xia, Y.; Zhao, D. General strategy to synthesize uniform mesoporous TiO2/graphene/mesoporous TiO2 sandwich-like nanosheets for highly reversible lithium storage. Nano Lett. 2015, 15, 2186–2193. [Google Scholar] [CrossRef]
- Williams, G.; Seger, B.; Kamat, P.V. TiO2-graphene nanocomposites. UV-assisted photocatalytic reduction of graphene oxide. Acs Nano 2008, 2, 1487–1491. [Google Scholar] [CrossRef]
- Li, B.; Zhang, X.; Li, X.; Wang, L.; Han, R.; Liu, B.; Zheng, W.; Li, X.; Liu, Y. Photo-assisted preparation and patterning of large-area reduced graphene oxide–TiO2 conductive thin film. Chem. Commun. 2010, 46, 3499–3501. [Google Scholar] [CrossRef]
- Cai, C.-J.; Xu, M.-W.; Bao, S.-J.; Ji, C.-C.; Lu, Z.-J.; Jia, D.-Z. A green and facile route for constructing flower-shaped TiO2 nanocrystals assembled on graphene oxide sheets for enhanced photocatalytic activity. Nanotechnology 2013, 24, 275602. [Google Scholar] [CrossRef]
- Štengl, V.; Bakardjieva, S.; Grygar, T.M.; Bludská, J.; Kormunda, M. TiO2-graphene oxide nanocomposite as advanced photocatalytic materials. Chem. Cent. J. 2013, 7, 41. [Google Scholar] [CrossRef]
- Wang, W.; Yu, J.; Xiang, Q.; Cheng, B. Enhanced photocatalytic activity of hierarchical macro/mesoporous TiO2–graphene composites for photodegradation of acetone in air. Appl. Catal. B Environ. 2012, 119, 109–116. [Google Scholar] [CrossRef]
- Jiang, G.; Lin, Z.; Chen, C.; Zhu, L.; Chang, Q.; Wang, N.; Wei, W.; Tang, H. TiO2 nanoparticles assembled on graphene oxide nanosheets with high photocatalytic activity for removal of pollutants. Carbon 2011, 49, 2693–2701. [Google Scholar] [CrossRef]
- Trapalis, A.; Todorova, N.; Giannakopoulou, T.; Boukos, N.; Speliotis, T.; Dimotikali, D.; Yu, J. TiO2/graphene composite photocatalysts for NOx removal: A comparison of surfactant-stabilized graphene and reduced graphene oxide. Appl. Catal. B Environ. 2016, 180, 637–647. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Z.-R.; Fu, X.; Xu, Y.-J. TiO2—Graphene nanocomposites for gas-phase photocatalytic degradation of volatile aromatic pollutant: Is TiO2− graphene truly different from other TiO2—Carbon composite materials? ACS Nano 2010, 4, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Lambert, T.N.; Chavez, C.A.; Hernandez-Sanchez, B.; Lu, P.; Bell, N.S.; Ambrosini, A.; Friedman, T.; Boyle, T.J.; Wheeler, D.R.; Huber, D.L. Synthesis and characterization of titania− graphene nanocomposites. J. Phys. Chem. C 2009, 113, 19812–19823. [Google Scholar] [CrossRef]
- Low, W.; Boonamnuayvitaya, V. Enhancing the photocatalytic activity of TiO2 co-doping of graphene–Fe3+ ions for formaldehyde removal. J. Environ. Manag. 2013, 127, 142–149. [Google Scholar] [CrossRef]
- Pallotti, D.; Gesuele, F.; Palomba, M.; Longo, A.; Carotenuto, G.; Maddalena, P.; Lettieri, S. Photoluminescence-based real-time monitoring of graphene oxide photoreduction: Demonstrations and application to graphene oxide/titanium dioxide composites. J. Lumin. 2017, 188, 129–134. [Google Scholar] [CrossRef]
- Li, K.; Xiong, J.; Chen, T.; Yan, L.; Dai, Y.; Song, D.; Lv, Y.; Zeng, Z. Preparation of graphene/TiO2 composites by nonionic surfactant strategy and their simulated sunlight and visible light photocatalytic activity towards representative aqueous POPs degradation. J. Hazard. Mater. 2013, 250, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Tri, N.L.M.; Jitae, K.; Van Thuan, D.; Huong, P.T.; Al Tahtamouni, T. Improved photocatalytic decomposition of methyl ethyl ketone gas from indoor air environment by using TiO2/graphene oxide. Mater. Res. Express 2019, 6, 105509. [Google Scholar]
- Najafi, M.; Kermanpur, A.; Rahimipour, M.R.; Najafizadeh, A. Effect of TiO2 morphology on structure of TiO2-graphene oxide nanocomposite synthesized via a one-step hydrothermal method. J. Alloys Compd. 2017, 722, 272–277. [Google Scholar] [CrossRef]
- Fotiou, T.; Triantis, T.M.; Kaloudis, T.; Pastrana-Martínez, L.M.; Likodimos, V.; Falaras, P.; Silva, A.N.M.; Hiskia, A. Photocatalytic Degradation of Microcystin-LR and Off-Odor Compounds in Water under UV-A and Solar Light with a Nanostructured Photocatalyst Based on Reduced Graphene Oxide–TiO2 Composite. Identification of Intermediate Products. Ind. Eng. Chem. Res. 2013, 52, 13991–14000. [Google Scholar] [CrossRef]
- Hu, X.; Zhao, Y.; Wang, H.; Cai, X.; Hu, X.; Tang, C.; Liu, Y.; Yang, Y. Decontamination of Cr (VI) by graphene oxide@ TiO2 in an aerobic atmosphere: Effects of pH, ferric ions, inorganic anions, and formate. J. Chem. Technol. Biotechnol. 2018, 93, 2226–2233. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-graphene composite as a high performance photocatalyst. ACS Nano 2009, 4, 380–386. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, Z.; Tang, Y.; Feng, X. Graphene encapsulated hollow TiO2 nanospheres: Efficient synthesis and enhanced photocatalytic activity. J. Mater. Chem. A 2013, 1, 3752–3756. [Google Scholar] [CrossRef]
- Wang, P.; Wang, J.; Wang, X.; Yu, H.; Yu, J.; Lei, M.; Wang, Y. One-step synthesis of easy-recycling TiO2-rGO nanocomposite photocatalysts with enhanced photocatalytic activity. Appl. Catal. B Environ. 2013, 132, 452–459. [Google Scholar] [CrossRef]
- Ng, Y.H.; Lightcap, I.V.; Goodwin, K.; Matsumura, M.; Kamat, P.V. To what extent do graphene scaffolds improve the photovoltaic and photocatalytic response of TiO2 nanostructured films? J. Phys. Chem. Lett. 2010, 1, 2222–2227. [Google Scholar] [CrossRef]
- Fernandez-Ibanez, P.; Polo-López, M.; Malato, S.; Wadhwa, S.; Hamilton, J.; Dunlop, P.; D’sa, R.; Magee, E.; O’shea, K.; Dionysiou, D. Solar photocatalytic disinfection of water using titanium dioxide graphene composites. Chem. Eng. J. 2015, 261, 36–44. [Google Scholar] [CrossRef]
- Yang, X.; Qin, J.; Jiang, Y.; Li, R.; Li, Y.; Tang, H. Bifunctional TiO2/Ag3 PO4/graphene composites with superior visible light photocatalytic performance and synergistic inactivation of bacteria. Rsc Adv. 2014, 4, 18627–18636. [Google Scholar] [CrossRef]
- Chang, Y.-N.; Ou, X.-M.; Zeng, G.-M.; Gong, J.-L.; Deng, C.-H.; Jiang, Y.; Liang, J.; Yuan, G.-Q.; Liu, H.-Y.; He, X. Synthesis of magnetic graphene oxide—TiO2 and their antibacterial properties under solar irradiation. Appl. Surf. Sci. 2015, 343, 1–10. [Google Scholar] [CrossRef]
- Cruz-Ortiz, B.R.; Hamilton, J.W.; Pablos, C.; Díaz-Jiménez, L.; Cortés-Hernández, D.A.; Sharma, P.K.; Castro-Alférez, M.; Fernández-Ibañez, P.; Dunlop, P.S.; Byrne, J.A. Mechanism of photocatalytic disinfection using titania-graphene composites under UV and visible irradiation. Chem. Eng. J. 2017, 316, 179–186. [Google Scholar] [CrossRef]
- Giampiccolo, A.; Tobaldi, D.M.; Leonardi, S.G.; Murdoch, B.J.; Seabra, M.P.; Ansell, M.P.; Neri, G.; Ball, R.J. Sol gel graphene/TiO2 nanoparticles for the photocatalytic-assisted sensing and abatement of NO2. Appl. Catal. B Environ. 2019, 243, 183–194. [Google Scholar] [CrossRef]
- Xu, M.; Hu, X.; Wang, S.; Yu, J.; Zhu, D.; Wang, J. Photothermal effect promoting CO2 conversion over composite photocatalyst with high graphene content. J. Catal. 2019, 377, 652–661. [Google Scholar] [CrossRef]
- Dai, X.; Wang, Y.; Wang, X.; Tong, S.; Xie, X. Polarity on adsorption and photocatalytic performances of N-GR/TiO2 towards gaseous acetaldehyde and ethylene. Appl. Surf. Sci. 2019, 485, 255–265. [Google Scholar] [CrossRef]
- Roso, M.; Boaretti, C.; Pelizzo, M.G.; Lauria, A.; Modesti, M.; Lorenzetti, A. Nanostructured photocatalysts based on different oxidized graphenes for VOCs removal. Ind. Eng. Chem. Res. 2017, 56, 9980–9992. [Google Scholar] [CrossRef]
| Method | Mechanism | Phase of Formation | Pros and Cons | Reference |
|---|---|---|---|---|
| Sol-gel | Hydrolysis and condensation of TiCl4 or an organometallic compound | Amorphous and rutile | High purity, fine particle sizes, good size distribution, high surface areas, but the ease of agglomeration and long reaction time | [25,26,27,28] |
| Hydrothermal | Precipitation of TiO2 from aqueous solution at elevated temperature and pressure | Anatase and rutile | High crystallinity, low defects, fine particle size, good size distribution, limited agglomeration, control of crystal shape by temperature adjustment, but relatively higher costs | [25,29,30] |
| Solvothermal | Precipitation of TiO2 from organic solution at elevated temperature and pressure | Anatase and rutile | High crystallinity, low defects, suitability for materials unstable at high temperature, but organic solvents needed | [25,31] |
| Micelle and inverse micelle | Aggregation of TiO2 in a liquid colloid | Amorphous | High crystallinity, low defects, fine particle sizes, but relatively high costs and high crystallization temperatures | [25,32] |
| Flame pyrolysis | Combustion of TiCl4 with oxygen; used in industrial processes | Anatase and rutile | Rapid and mass production, but high energy needed and ease of rutile formation | [25,33,34] |
| Properties | Anatase | Brookite | Rutile |
|---|---|---|---|
| Crystal structure | Tetragonal | Orthorhombic | Tetragonal |
| Density (g/cm3) | 3.79 | 3.99 | 4.13 |
| Band gap (eV) | 3.2 a | ~3.2 b | 3.0 c |
| Light absorption (nm) | <390 | - | <415 |
| Dielectric constant | 6.04 | 7.89 | 6.62 |
| Lattice energy (kJ/mol) d | 24.75 | 18.53 | 0 |
| Surface enthalpy (J/m2) e | 1.34 | 1.66 | 1.93 |
| Photocatal. activity (mol/h) f | 3.5 × 10−5 | - | 1.1 × 10−5 |
| Effective electron mass (me*/m0) g | 0.0948 | 0.0949 | 1.4640 |
| Effective hole mass (mh*/m0) g | 0.1995 | 0.5620 | 0.4345 |
| Ti-O bond length (Å) h | 1.94 (shorter); 1.97 (longer) | 1.87–2.04 | 1.95 (shorter); 1.98 (longer) |
| O-Ti-O bond angle (degree) | 77.7; 92.6 | 77.0–105 | 81.2; 90.0 |
| Method | Major Approach | Pros and Cons | Cost | |
|---|---|---|---|---|
| Graphene | Mechanical exfoliation | Micro-mechanical cleavage, sonication, ball milling, and fluid dynamics | Straightforward and eco-friendly processes, fine product qualities, but relatively higher costs and limits of scalable production | High |
| Oxidative exfoliation-reduction | Chemical reduction, thermal reduction, and electrochemical reduction | Straightforward processes, cost-effectiveness, scalable production, but possible structural damage due to mal exfoliation, and potential use of hazardous chemicals | Low | |
| Liquid phase exfoliation | Sonication with proper solvents | Straightforward and eco-friendly processes (solvents recyclable), fine product qualities, scalable production, but parameters (e.g., solvent and ultra-sonication) critical to avoid physical deformation and defects | Moderate | |
| Chemical vapor deposition (CVD) | Thermal CVD, plasma-enhanced CVD, and thermal decomposition | Highly connected products with low defects and high surface areas, but relatively higher costs, limited yields, and high technical thresholds | Moderate | |
| Graphene oxide | Brodie | Graphite + H2CO3 (C/O ratio = 2.23) | Adjustable oxidation states, but potentials of long reaction time and production of explosive ClO2 and acid fog | Low |
| Staudenmaier | Graphite + HNO3 (fuming) + H2SO4 + KClO3 (C/O ratio = 2.52) | Adjustable oxidation state, but long reaction time and low temperatures to avoid exothermic reactions | Low | |
| Hofmann | Graphite + HNO3 + H2SO4 + KClO3 (C/O ratio = 2.52) | Low | ||
| Hummers | Graphite+NaNO3 +H2SO4+ KMnO4 (C/O ratio = 2.1-2.9) | Safe and fast reactions, but more parameters to control | Low | |
| Reduced graphene oxide | Chemical reduction | Various reductants | Fine product qualities, scalable production, but the potential of using hazardous reductants. Lower product qualities and removal of excess chemicals with the use of green reductants | Low |
| Thermal reduction | 1000–1100 °C for 30–45 s in the absence of air | Straightforward and eco-friendly processes, cost-effectiveness, but high capital costs and energy needed | Moderate | |
| Electrochemical reduction | The cathodic potential of 1–1.5 V | Low-defect products, rapid and eco-friendly processes, cost-effectiveness, but lower reduction levels and limited scalable production | Low | |
| Microwave and photo-reduction | Microwave reaction with visible or UV light | Fast reactions, no chemicals needed, and high yield efficiencies | Low | |
| Graphene quantum dot | Top-down | Hydrothermal synthesis, solvent thermal method, chemical oxidation, electrochemical exfoliation, electron beam lithography, microwave-assisted method, and ultra-sonication exfoliation | Scalable production, but difficulty of effective size control | High |
| Bottom-up | Soft template method, acid- and solvent-free synthesis, and metal catalysis | Effective size control, but long reaction time and limited scalable production | High |
| Dimension | Structure | Surface Area | Light Absorption Wavelength | Current Density | Reference |
|---|---|---|---|---|---|
| 0 | Nanoparticle (less than 100 nm) | 180–250 m2/g | Ultraviolet to infrared radiation | Not available | [121,122,123,124] |
| 1 | Nanofiber | 52–55 m2/g | <510 nm | 0.06 mA/cm2 | [125,126,127] |
| Nanowire | 61.5–92.6 m2/g | 250–540 nm | 1.6 mA/cm2 | [130,131,132,133,134] | |
| Nanorod | 104.6 m2/g | ~380 nm | 0.8 mA/cm2 | [135,136,137,138] | |
| Nanotube | 400 m2/g | <500 nm | 0.02 mA/cm2 | [139,140,141,142] | |
| 2 | Nanosheet | 31–146 m2/g | 200–900 nm | 0.03 mA/cm2 | [128,129] |
| 3 | Porous film | 36.4–70.8 m2/g | 200–700 nm | 18.54 mA/cm2 | [146,147,148,149] |
| Methods | Crystal Form | GFN Ratio | Pros and Cons | Reference |
|---|---|---|---|---|
| Ion implantation | Anatase | Not available | Fast production, few interfacial defects, great optical character, but high energy costs | [150] |
| Colloidal blending process | Anatase or rutile | adjustable | Aging at room temperature and vacuum drying needed | [151,152] |
| Spark plasma sintering | Rutile | 1% v/v | Fast production, but high energy costs and increased rutile form | [153] |
| Hydrothermal method | Anatase | adjustable | Adjustable doping ratio, but high pressure needed | [154,155,156] |
| Sol-gel method | Anatase | 48% w/w | Aging at room temperature, long reaction time, and calcination needed | [157] |
| Hydrolysis | Anatase | 16% w/w | Great heterogeneous nucleation, but longer reaction time and calcination needed | [158] |
| UV-assisted photo-reduction | Not available | Not available | Fast production and few collapses during reduction, but extra light source needed | [159,160] |
| In-situ assembly | Anatase | Not available | No calcination and full anatase formation, but long synthesis time | [161,162] |
| Category | Technology | Description | Ref. |
|---|---|---|---|
| Morphology | SEM | Spherical and non-spherical (platelet- or flower-like) shapes were observed with low and high GFN contents, respectively. | [151,163,164,165,166,167] |
| TEM | A fine dispersion of TiO2 in GFN with low- and nano-dimensions was reported. | [163,165,166,167] | |
| AFM | The thickness of GFN-TiO2 was increased to a scale of μm after preparation. | [164] | |
| Chemical constitution | FTIR | The peak of Ti-O-Ti at 400–900 cm−1 was broadened or shifted by the influence of Ti-O-C. The signals of carbonyl and epoxy groups were reduced. | [151,165,168] |
| XPS | The formation of C-Ti, O=C-O-Ti, and C-O-Ti bonds in GFN-TiO2 was observed. | [163] [164] | |
| XRD | The signals due to the presence of anatase and rutile were reported. | [151,163,164,165,166,168] | |
| Raman | The signals of both TiO2 and GFN were reported. The D/G intensity ratio of GFN-TiO2 was higher than that of GFN. | [163,164,165] | |
| EPR | The formation of hydroxyl and superoxide radical species was observed in GFN-TiO2. | [166] | |
| Physicochemical properties | Zeta potential | The zeta potential of GFN-TiO2 ranged between those of GFN and TiO2. | [164] |
| TGA | The irregular mass loss occurred at high temperatures. | [164] | |
| BET | The surface area of GFN-TiO2 was significantly increased at a certain ratio of GFN to TiO2. | [151,163,164,165,168] | |
| ACM | The current density of GFN-TiO2 was significantly increased at a certain ratio of GFN to TiO. | [168] | |
| PL | The time dynamics of the TiO2-induced photoreduction of GO were observed. | [169] | |
| UV-Vis | A shift to larger wavelengths in the absorption edge was observed, indicating bandgap narrowing. | [151,164,165,166,168] |
| Materials | Average Size (nm) | Functional Group | Bandgap (eV) | Wavelength (nm) | Surface Area (m2/g) | Reference |
|---|---|---|---|---|---|---|
| Graphene-TiO2 | 3.8 | C-O, C=O, O=C-O, and O-Ti | NA 1 | 600 | 176 | [170] |
| Graphene-TiO2 | ~6 | C-O and O-C=O | NA | NA | 252 | [158] 2 |
| GO-TiO2 | NA | C-O, Ti-O-Ti, Ti-O-C, and OH | NA | ~800 | 69.2 | [151] |
| GO-Co-TiO2 | NA | C-O, C-N, O-C=O | 2.77 | 421 | 206 | [109] |
| GO-Ti | NA | NA | 2.9 | ~550 | 68.9 | [171] |
| rGO-TiO2 | 35 | NA | NA | ~360 | 212.75 | [172] |
| rGO-TiO2 | ~8 | NA | NA | NA | 229 | [157] 2 |
| Pollutant | Catalyst | Light Source | Removal | Ref. | |
|---|---|---|---|---|---|
| Inorganic | Cr(VI) (0.2 mM) | GO-TiO2 (0.5 g/L) | 254 nm, 20 W, UV lamp | 90% | [164] |
| Cr(VI)(10 mg/L) | GO-TiO2 (0.5 g/L) | 365 nm, 8 W, UV lamp | 99% | [174] | |
| Organic | Methylene blue (0.01 g/L) | Graphene-TiO2 (0.75 g/L) | 365 nm, 100 W, high-pressure Hg lamp >400 nm, 500W, Xe lamp | 85% 65% | [175] |
| Rhodamine B (20 mg/L) | Graphene-TiO2 (0.1 g/L) | 11 W, low-pressure Hg lamp | 91% | [176] | |
| Rhodamine B (20 mg/L) Norfloxacin (20 mg/L) Aldicarb (10.5 mg/L) | Graphene-TiO2 (1 g/L) | >400 nm, Xe lamp | 79.7% 86.2% 36.8% | [170] | |
| Malachite green oxalate (13.1 mg/L) | GO-TiO2 (0.2 g/L) | 450 W, water-cooled Hg lamp | 80% | [145] | |
| Phenol (10 mg/L) | rGO-TiO2 (5 g/L) | 310-400 nm, UV lamp | Not given | [177] | |
| 2,4-D (15 mM) | rGO-TiO2 (film) | <320 nm, 450 W, Xe lamp | ~87% | [178] | |
| Biological | E. coli (106 CFU/mL), F. solani spores (103 CFU/mL) | rGO-TiO2 (0.5 g/L) | Sunlight | ~100% | [179] |
| E. coli, S.aureus, S.typhi, P. aeruginosa, B. subtilis, B. pumilus (106 CFU/mL) | Graphene-Ag3PO4-TiO2 | >420 nm, 350 W, Xe lamp | ~100% | [180] | |
| E. coli (105–106 CFU/mL) | GO-TiO2 (0.2 g/L) | Xe lamp | ~100% | [181] | |
| E. coli (106 CFU/mL) | rGO-TiO2 (18 mg/L) | >285 nm, UV-visible light; >420 nm, visible light | ~100% | [182] |
| Pollutant | Catalyst | Light Source | Humidity or Flow Rate | Removal | Ref. | |
|---|---|---|---|---|---|---|
| Inorganic | NOx (1 ppm) | Graphene-TiO2 rGO-TiO2 | 15 W, UVA 8 W, visible light | 50% humidity, 3 L/min | 42% 49% | [165] |
| NOx (200 ppb) | Graphene-TiO2 | 280–780 nm, 300 W, solar lamp | 1 L/min | 77% | [183] | |
| CO (50 ppm) NOx (1 ppm) | Graphene-TiO2 | 8 W, UV lamp | 0.2 L/min | 46% 51% | [109] | |
| Organic | Acetone (300 ± 20 ppm) | Graphene-TiO2 | 365 nm, 15 W, UV lamp | 1 L/min | ~60% | [163] |
| Acetaldehyde (500 ppm) Ethylene (50 ppm) | Graphene-TiO2 | 260 W, fluorescent lamp 500 W, Xenon lamp | 20 cm3/min | ~82% ~90% | [185] | |
| Benzene (250 ppm) | Graphene-TiO2 | 254 nm, 4 W, UV lamp | 20 mL/min | 6.4% | [166] | |
| Formaldehyde (3000 ppm) | Graphene-TiO2 | 365 nm, 8 W, black light blue lamp >420 nm, 8 W, fluorescent lamp | Not specified | 50.3% 25.5% | [168] | |
| Methanol (4,000 ppm) | Graphene-TiO2 GO-TiO2 rGO-TiO2 | 254 nm, 16 W, UV lamp | 155 cm3/min | 80% 99% 99% | [186] | |
| BTEX (1 ppm) | GO-TiO2 | 400–720 nm, 8 W, daylight lamp | 55% humidity, 1 L/min | 96% | [151] | |
| MEKT (30 ppm) | GO-TiO2 | 80 W, Xe lamp | 40% humidity, 50 mL/min | 96.8% | [171] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, C.-H.; Chen, W.-H. Graphene Family Nanomaterials (GFN)-TiO2 for the Photocatalytic Removal of Water and Air Pollutants: Synthesis, Characterization, and Applications. Nanomaterials 2021, 11, 3195. https://doi.org/10.3390/nano11123195
Lin C-H, Chen W-H. Graphene Family Nanomaterials (GFN)-TiO2 for the Photocatalytic Removal of Water and Air Pollutants: Synthesis, Characterization, and Applications. Nanomaterials. 2021; 11(12):3195. https://doi.org/10.3390/nano11123195
Chicago/Turabian StyleLin, Chih-Hsien, and Wei-Hsiang Chen. 2021. "Graphene Family Nanomaterials (GFN)-TiO2 for the Photocatalytic Removal of Water and Air Pollutants: Synthesis, Characterization, and Applications" Nanomaterials 11, no. 12: 3195. https://doi.org/10.3390/nano11123195
APA StyleLin, C.-H., & Chen, W.-H. (2021). Graphene Family Nanomaterials (GFN)-TiO2 for the Photocatalytic Removal of Water and Air Pollutants: Synthesis, Characterization, and Applications. Nanomaterials, 11(12), 3195. https://doi.org/10.3390/nano11123195






