Thin Film and Nanostructured Pd-Based Materials for Optical H2 Sensors: A Review
Abstract
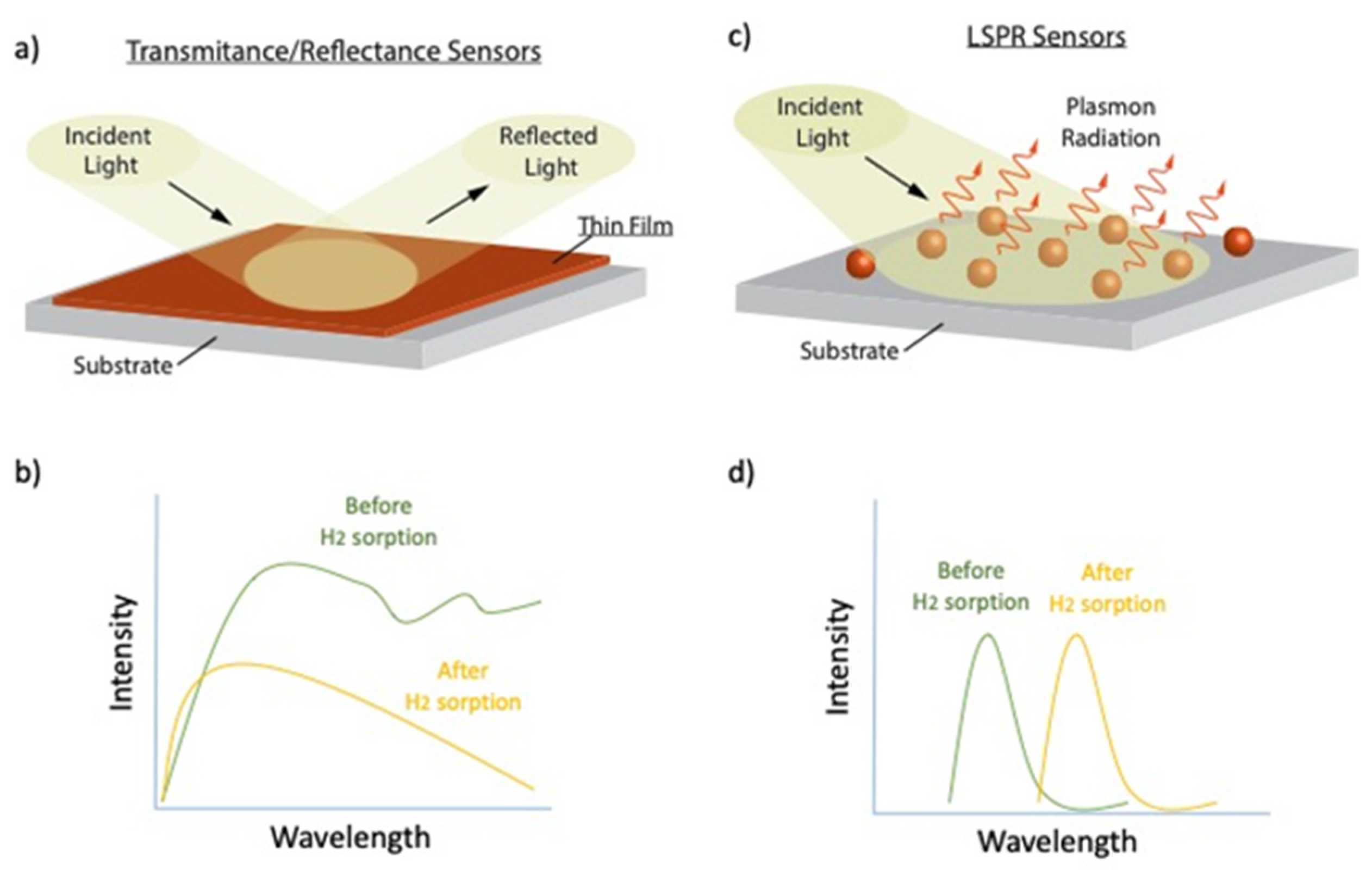
:1. Introduction
2. Theoretical Background
3. Pd-Based Thin Films for Optical H2 Sensors
4. Nanoparticle-Based Materials for Optical H2 Sensors
4.1. Materials Based on Aggregated Nanoparticles
4.2. Materials Based on Isolated Nanoparticles
5. Complex Nanostructured (CN) Materials for Optical H2 Sensors
6. Discussion
6.1. Addressing Limitations of Pd-Based of Optical H2 Sensing Materials
6.2. Manufacturing of Materials for Optical H2 Sensors
7. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hübert, T.; Boon-Brett, L.; Black, G.; Banach, U. Hydrogen sensors—A review. Sens. Actuators B Chem. 2011, 157, 329–352. [Google Scholar] [CrossRef]
- Tang, X.; Haddad, P.-A.; Mager, N.; Geng, X.; Reckinger, N.; Hermans, S.; Debliquy, M.; Raskin, J.-P. Chemically deposited palladium nanoparticles on graphene for hydrogen sensor applications. Sci. Rep. 2019, 9, 3653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sansone, F.J. Fuel cell hydrogen sensor for marine applications. Mar. Chem. 1992, 37, 3–14. [Google Scholar] [CrossRef]
- Leonardi, S.G.; Bonavita, A.; Donato, N.; Neri, G. Development of a hydrogen dual sensor for fuel cell applications. Int. J. Hydrogen Energy 2018, 43, 11896–11902. [Google Scholar] [CrossRef]
- Kumamoto, A.; Iseki, H.; Ono, R.; Oda, T. Measurement of minimum ignition energy in hydrogen-oxygen-nitrogen premixed gas by spark discharge. J. Phys. Conf. Ser. 2011, 301, 012039. [Google Scholar] [CrossRef] [Green Version]
- Ono, R.; Oda, T. Spark ignition of hydrogen-air mixture. J. Phys. Conf. Ser. 2008, 142, 012003. [Google Scholar] [CrossRef] [Green Version]
- Fedtke, P.; Wienecke, M.; Bunescu, M.-C.; Pietrzak, M.; Deistung, K.; Borchardt, E. Hydrogen sensor based on optical and electrical switching. Sens. Actuators B Chem. 2004, 100, 151–157. [Google Scholar] [CrossRef]
- Ren, Q.; Cao, Y.-Q.; Arulraj, D.; Liu, C.; Wu, D.; Li, W.-M.; Li, A.-D. Review—Resistive-type hydrogen sensors based on zinc oxide nanostructures. J. Electrochem. Soc. 2020, 167, 067528. [Google Scholar] [CrossRef]
- Kim, J.H.; Jeon, J.G.; Ovalle-Robles, R.; Kang, T.J. Aerogel sheet of carbon nanotubes decorated with palladium nanoparticles for hydrogen gas sensing. Int. J. Hydrogen Energy 2018, 43, 6456–6461. [Google Scholar] [CrossRef]
- Rashid, T.-R.; Phan, D.-T.; Chung, G.-S. Effect of ga-modified layer on flexible hydrogen sensor using zno nanorods decorated by pd catalysts. Sens. Actuators B Chem. 2014, 193, 869–876. [Google Scholar] [CrossRef]
- Xie, B.; Zhang, S.; Liu, F.; Peng, X.; Song, F.; Wang, G.; Han, M. Response behavior of a palladium nanoparticle array based hydrogen sensor in hydrogen–nitrogen mixture. Sens. Actuators A Phys. 2012, 181, 20–24. [Google Scholar] [CrossRef]
- Zhang, H.; Li, Z.; Liu, L.; Xu, X.; Wang, Z.; Wang, W.; Zheng, W.; Dong, B.; Wang, C. Enhancement of hydrogen monitoring properties based on Pd–SnO2 composite nanofibers. Sens. Actuators B Chem. 2010, 147, 111–115. [Google Scholar] [CrossRef]
- Rashid, T.-R.; Phan, D.-T.; Chung, G.-S. A flexible hydrogen sensor based on pd nanoparticles decorated ZnO nanorods grown on polyimide tape. Sens. Actuators B Chem. 2013, 185, 777–784. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Z.; Jiang, T.; Xu, X.; Wang, C. Ultrasensitive hydrogen sensor based on pd0-loaded sno2 electrospun nanofibers at room temperature. ACS Appl. Mater. Interfaces 2013, 5, 2013–2021. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Dong, H.; Xia, S. Micromachined catalytic combustion type gas sensor for hydrogen detection. Micro Nano Lett. 2013, 8, 668–671. [Google Scholar] [CrossRef]
- Lupan, O.; Postica, V.; Hoppe, M.; Wolff, N.; Polonskyi, O.; Pauporté, T.; Viana, B.; Majérus, O.; Kienle, L.; Faupel, F.; et al. PdO/PdO2 functionalized zno: Pd films for lower operating temperature H2 gas sensing. Nanoscale 2018, 10, 14107–14127. [Google Scholar] [CrossRef]
- Buso, D.; Busato, G.; Guglielmi, M.; Martucci, A.; Bello, V.; Mattei, G.; Mazzoldi, P.; Post, M.L. Selective optical detection of H2 and CO with SiO2 sol–gel films containing Nio and Au nanoparticles. Nanotechnology 2007, 18, 475505. [Google Scholar] [CrossRef]
- Gu, H.; Wang, Z.; Hu, Y. Hydrogen gas sensors based on semiconductor oxide nanostructures. Sensors 2012, 12, 5517–5550. [Google Scholar] [CrossRef] [Green Version]
- Kaewsiri, D.; Inyawilert, K.; Wisitsoraat, A.; Tuantranont, A.; Phanichphant, S.; Liewhiran, C. Flame-spray-made ptox-functionalized Zn2SnO4 spinel nanostructures for conductometric H2 detection. Sens. Actuators B Chem. 2020, 316, 128132. [Google Scholar] [CrossRef]
- Inyawilert, K.; Wisitsoraat, A.; Tuantranont, A.; Phanichphant, S.; Liewhiran, C. Ultra-sensitive and highly selective H2 sensors based on fsp-made rh-substituted SnO2 sensing films. Sens. Actuators B Chem. 2017, 240, 1141–1152. [Google Scholar] [CrossRef]
- Brauns, E.; Morsbach, E.; Bäumer, M.; Lang, W. A Fast and Sensitive Catalytic Hydrogen Sensor Based on A Stabilized Nanoparticle Catalyst. In Proceedings of the 2013 Transducers & Eurosensors XXVII: The 17th International Conference on Solid-State Sensors, Actuators and Microsystems (TRANSDUCERS & EUROSENSORS XXVII), Barcelona, Spain, 16–20 June 2013; pp. 1178–1181. [Google Scholar]
- Harley-Trochimczyk, A.; Chang, J.; Zhou, Q.; Dong, J.; Pham, T.; Worsley, M.A.; Maboudian, R.; Zettl, A.; Mickelson, W. Catalytic hydrogen sensing using microheated platinum nanoparticle-loaded graphene aerogel. Sens. Actuators B Chem. 2015, 206, 399–406. [Google Scholar] [CrossRef] [Green Version]
- Pranti, A.S.; Loof, D.; Kunz, S.; Zielasek, V.; Bäumer, M.; Lang, W. Ligand-linked nanoparticles-based hydrogen gas sensor with excellent homogeneous temperature field and a comparative stability evaluation of different ligand-linked catalysts. Sensors 2019, 19, 1205. [Google Scholar] [CrossRef] [Green Version]
- Korotcenkov, G.; Han, S.D.; Stetter, J.R. Review of electrochemical hydrogen sensors. Chem. Rev. 2009, 109, 1402–1433. [Google Scholar] [CrossRef]
- Berndt, D.; Muggli, J.; Wittwer, F.; Langer, C.; Heinrich, S.; Knittel, T.; Schreiner, R. Mems-based thermal conductivity sensor for hydrogen gas detection in automotive applications. Sens. Actuators A Phys. 2020, 305, 111670. [Google Scholar] [CrossRef]
- Holleck, G.L. Diffusion and solubility of hydrogen in palladium and palladium—Silver alloys. J. Phys. Chem. 1970, 74, 503–511. [Google Scholar] [CrossRef]
- Boudart, M.; Hwang, H.S. Solubility of hydrogen in small particles of palladium. J. Catal. 1975, 39, 44–52. [Google Scholar] [CrossRef]
- Li, Y.; Cheng, Y.-T. Hydrogen diffusion and solubility in palladium thin films. Int. J. Hydrogen Energy 1996, 21, 281–291. [Google Scholar] [CrossRef]
- Wicke, E.; Brodowsky, H.; Züchner, H. Hydrogen in palladium and palladium alloys. In Hydrogen in Metals II: Application-Oriented Properties; Alefeld, G., Völkl, J., Eds.; Springer: Berlin/Heidelberg, Germany, 1978; pp. 73–155. [Google Scholar]
- Mirzaei, A.; Yousefi, H.R.; Falsafi, F.; Bonyani, M.; Lee, J.-H.; Kim, J.-H.; Kim, H.W.; Kim, S.S. An overview on how Pd on resistive-based nanomaterial gas sensors can enhance response toward hydrogen gas. Int. J. Hydrogen Energy 2019, 44, 20552–20571. [Google Scholar] [CrossRef]
- Arora, K.; Puri, N.K. Electrophoretically deposited nanostructured PdO thin film for room temperature amperometric H2 sensing. Vacuum 2018, 154, 302–308. [Google Scholar] [CrossRef]
- Öztürk, S.; Kılınç, N. Pd thin films on flexible substrate for hydrogen sensor. J. Alloys Compd. 2016, 674, 179–184. [Google Scholar] [CrossRef]
- Nugroho, F.A.A.; Darmadi, I.; Cusinato, L.; Susarrey-Arce, A.; Schreuders, H.; Bannenberg, L.J.; da Silva Fanta, A.B.; Kadkhodazadeh, S.; Wagner, J.B.; Antosiewicz, T.J.; et al. Metal–polymer hybrid nanomaterials for plasmonic ultrafast hydrogen detection. Nat. Mater. 2019, 18, 489–495. [Google Scholar] [CrossRef] [Green Version]
- Yue, S.; Hou, Y.; Wang, R.; Liu, S.; Li, M.; Zhang, Z.; Hou, M.; Wang, Y.; Zhang, Z. Cmos-compatible plasmonic hydrogen sensors with a detection limit of 40 ppm. Opt. Express 2019, 27, 19331–19347. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.A. Fiber optic sensor for hydrogen concentrations near the explosive limit. J. Electrochem. Soc. 1991, 138, L46. [Google Scholar] [CrossRef]
- Hamidi, S.M.; Ramezani, R.; Bananej, A. Hydrogen gas sensor based on long-range surface plasmons in lossy palladium film placed on photonic crystal stack. Opt. Mater. 2016, 53, 201–208. [Google Scholar] [CrossRef]
- Bannenberg, L.J.; Boelsma, C.; Asano, K.; Schreuders, H.; Dam, B. Metal hydride based optical hydrogen sensors. J. Phys. Soc. Jpn. 2020, 89, 051003. [Google Scholar] [CrossRef]
- Wadell, C.; Syrenova, S.; Langhammer, C. Plasmonic hydrogen sensing with nanostructured metal hydrides. ACS Nano. 2014, 8, 11925–11940. [Google Scholar] [CrossRef]
- Sturaro, M.; Zacco, G.; Zilio, P.; Surpi, A.; Bazzan, M.; Martucci, A. Gold nanodisks plasmonic array for hydrogen sensing at low temperature. Sensors 2019, 19, 647. [Google Scholar] [CrossRef] [Green Version]
- Darmadi, I.; Nugroho, F.A.A.; Langhammer, C. High-performance nanostructured palladium-based hydrogen sensors—Current limitations and strategies for their mitigation. ACS Sens. 2020, 5, 3306–3327. [Google Scholar] [CrossRef]
- Isaac, N.A.; Ngene, P.; Westerwaal, R.J.; Gaury, J.; Dam, B.; Schmidt-Ott, A.; Biskos, G. Optical hydrogen sensing with nanoparticulate Pd–Au films produced by spark ablation. Sens. Actuators B Chem. 2015, 221, 290–296. [Google Scholar] [CrossRef]
- Ndaya, C.C.; Javahiraly, N.; Brioude, A. Recent advances in palladium nanoparticles-based hydrogen sensors for leak detection. Sensors 2019, 19, 4478. [Google Scholar] [CrossRef] [Green Version]
- Syrenova, S.; Wadell, C.; Nugroho, F.A.A.; Gschneidtner, T.A.; Diaz Fernandez, Y.A.; Nalin, G.; Świtlik, D.; Westerlund, F.; Antosiewicz, T.J.; Zhdanov, V.P.; et al. Hydride formation thermodynamics and hysteresis in individual Pd nanocrystals with different size and shape. Nat. Mater. 2015, 14, 1236–1244. [Google Scholar] [CrossRef]
- Bannenberg, L.J.; Nugroho, F.A.A.; Schreuders, H.; Norder, B.; Trinh, T.T.; Steinke, N.-J.; van Well, A.A.; Langhammer, C.; Dam, B. Direct comparison of pdau alloy thin films and nanoparticles upon hydrogen exposure. ACS Appl. Mater. Interfaces 2019, 11, 15489–15497. [Google Scholar] [CrossRef] [Green Version]
- Wadell, C.; Nugroho, F.A.A.; Lidström, E.; Iandolo, B.; Wagner, J.B.; Langhammer, C. Hysteresis-free nanoplasmonic Pd–Au alloy hydrogen sensors. Nano Lett. 2015, 15, 3563–3570. [Google Scholar] [CrossRef] [PubMed]
- Cusinato, L.; Hellman, A. Structure and composition modification of ultrasmall palladium nanoparticles upon hydrogenation from first principles. J. Phys. Chem. C 2019, 123, 18609–18619. [Google Scholar] [CrossRef]
- Lueng, C.; Lupo, P.; Schefer, T.; Metaxas, P.J.; Adeyeye, A.O.; Kostylev, M. Sensitivity of ferromagnetic resonance in pdco alloyed films to hydrogen gas. Int. J. Hydrogen Energy 2019, 44, 7715–7724. [Google Scholar] [CrossRef] [Green Version]
- RaviPrakash, J.; McDaniel, A.H.; Horn, M.; Pilione, L.; Sunal, P.; Messier, R.; McGrath, R.T.; Schweighardt, F.K. Hydrogen sensors: Role of palladium thin film morphology. Sens. Actuators B Chem. 2007, 120, 439–446. [Google Scholar] [CrossRef]
- Giauque, W.F. The entropy of hydrogen and the third law of thermodynamics, the free energy and dissociation of hydrogen. J. Am. Chem. Soc. 1930, 52, 4816–4831. [Google Scholar] [CrossRef]
- Lee, Y.T.; Lee, J.M.; Kim, Y.J.; Joe, J.H.; Lee, W. Hydrogen gas sensing properties of pdo thin films with nano-sized cracks. Nanotechnology 2010, 21, 165503. [Google Scholar] [CrossRef] [PubMed]
- Corso, A.J.; Tessarolo, E.; Guidolin, M.; Della Gaspera, E.; Martucci, A.; Angiola, M.; Donazzan, A.; Pelizzo, M.G. Room-temperature optical detection of hydrogen gas using palladium nano-islands. Int. J. Hydrogen Energy 2018, 43, 5783–5792. [Google Scholar] [CrossRef]
- Szilágyi, P.Á.; Westerwaal, R.J.; van de Krol, R.; Geerlings, H.; Dam, B. Metal—Organic framework thin films for protective coating of Pd-based optical hydrogen sensors. J. Mater. Chem. C 2013, 1, 8146–8155. [Google Scholar] [CrossRef]
- Mooij, L.; Perkisas, T.; Pálsson, G.; Schreuders, H.; Wolff, M.; Hjörvarsson, B.; Bals, S.; Dam, B. The effect of microstructure on the hydrogenation of mg/fe thin film multilayers. Int. J. Hydrogen Energy 2014, 39, 17092–17103. [Google Scholar] [CrossRef]
- Ngene, P.; Westerwaal, R.J.; Sachdeva, S.; Haije, W.; de Smet, L.C.P.M.; Dam, B. Polymer-induced surface modifications of Pd-based thin films leading to improved kinetics in hydrogen sensing and energy storage applications. Angew. Chem. Int. Ed. 2014, 53, 12081–12085. [Google Scholar] [CrossRef]
- Gaspera, E.D.; Martucci, A. Sol-gel thin films for plasmonic gas sensors. Sensors 2015, 15, 16910–16928. [Google Scholar] [CrossRef] [Green Version]
- Fortunato, G.; Bearzotti, A.; Caliendo, C.; D’Amico, A. Hydrogen sensitivity of pd/sio2/si structure: A correlation with the hydrogen-induced modifications on optical and transport properties of α-phase Pd films. Sens. Actuators 1989, 16, 43–54. [Google Scholar] [CrossRef]
- Menumerov, E.; Marks, B.A.; Dikin, D.A.; Lee, F.X.; Winslow, R.D.; Guru, S.; Sil, D.; Borguet, E.; Hutapea, P.; Hughes, R.A.; et al. Sensing hydrogen gas from atmospheric pressure to a hundred parts per million with nanogaps fabricated using a single-step bending deformation. ACS Sens. 2016, 1, 73–80. [Google Scholar] [CrossRef]
- She, X.; Shen, Y.; Wang, J.; Jin, C. Pd films on soft substrates: A visual, high-contrast and low-cost optical hydrogen sensor. Light Sci. Appl. 2019, 8, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bannenberg, L.; Schreuders, H.; Dam, B. Tantalum-palladium: Hysteresis-free optical hydrogen sensor over 7 orders of magnitude in pressure with sub-second response. Adv. Funct. Mater. 2021, 31, 9. [Google Scholar]
- Lee, Y.-A.; Kalanur, S.S.; Shim, G.; Park, J.; Seo, H. Highly sensitive gasochromic H2 sensing by nano-columnar WO3-Pd films with surface moisture. Sens. Actuators B Chem. 2017, 238, 111–119. [Google Scholar] [CrossRef]
- Boudiba, A.; Roussel, P.; Zhang, C.; Olivier, M.-G.; Snyders, R.; Debliquy, M. Sensing mechanism of hydrogen sensors based on palladium-loaded tungsten oxide (Pd–WO3). Sens. Actuators B Chem. 2013, 187, 84–93. [Google Scholar] [CrossRef]
- Ahmad, M.Z.; Sadek, A.Z.; Yaacob, M.H.; Anderson, D.P.; Matthews, G.; Golovko, V.B.; Wlodarski, W. Optical characterisation of nanostructured Au/WO3 thin films for sensing hydrogen at low concentrations. Sens. Actuators B Chem. 2013, 179, 125–130. [Google Scholar] [CrossRef]
- Ngene, P.; Radeva, T.; Slaman, M.; Westerwaal, R.J.; Schreuders, H.; Dam, B. Seeing hydrogen in colors: Low-cost and highly sensitive eye readable hydrogen detectors. Adv. Funct. Mater. 2014, 24, 2374–2382. [Google Scholar] [CrossRef]
- Palmisano, V.; Filippi, M.; Baldi, A.; Slaman, M.; Schreuders, H.; Dam, B. An optical hydrogen sensor based on a Pd-capped mg thin film wedge. Int. J. Hydrogen Energy 2010, 35, 12574–12578. [Google Scholar] [CrossRef]
- Beni, T.; Yamasaku, N.; Kurotsu, T.; To, N.; Okazaki, S.; Arakawa, T.; Balčytis, A.; Seniutinas, G.; Juodkazis, S.; Nishijima, Y. Metamaterial for hydrogen sensing. ACS Sens. 2019, 4, 2389–2394. [Google Scholar] [CrossRef]
- Victoria, M.; Westerwaal, R.J.; Dam, B.; van Mechelen, J.L.M. Amorphous metal-hydrides for optical hydrogen sensing: The effect of adding glassy Ni–Zr to Mg–Ni–H. ACS Sens. 2016, 1, 222–226. [Google Scholar] [CrossRef]
- Bannenberg, L.J.; Boelsma, C.; Schreuders, H.; Francke, S.; Steinke, N.J.; van Well, A.A.; Dam, B. Optical hydrogen sensing beyond palladium: Hafnium and tantalum as effective sensing materials. Sens. Actuators B Chem. 2019, 283, 538–548. [Google Scholar] [CrossRef]
- Boelsma, C.; Bannenberg, L.J.; van Setten, M.J.; Steinke, N.J.; van Well, A.A.; Dam, B. Hafnium—An optical hydrogen sensor spanning six orders in pressure. Nat. Commun. 2017, 8, 15718. [Google Scholar] [CrossRef] [Green Version]
- Langhammer, C.; Zhdanov, V.P.; Zorić, I.; Kasemo, B. Size-dependent kinetics of hydriding and dehydriding of pd nanoparticles. Phys. Rev. Lett. 2010, 104, 135502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfeiffer, T.V.; Feng, J.; Schmidt-Ott, A. New developments in spark production of nanoparticles. Adv. Powder Technol. 2014, 25, 56–70. [Google Scholar] [CrossRef]
- Güntner, A.T.; Wied, M.; Pineau, N.J.; Pratsinis, S.E. Rapid and selective NH3 sensing by porous cubr. Adv. Sci. 2020, 7, 1903390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abegg, S.; Klein Cerrejon, D.; Güntner, A.T.; Pratsinis, S.E. Thickness optimization of highly porous flame-aerosol deposited WO3 films for NO2 sensing at ppb. Nanomaterials 2020, 10, 1170. [Google Scholar] [CrossRef] [PubMed]
- Athanassiou, E.K.; Grass, R.N.; Stark, W.J. Chemical aerosol engineering as a novel tool for material science: From oxides to salt and metal nanoparticles. Aerosol Sci. Technol. 2010, 44, 161–172. [Google Scholar] [CrossRef]
- Pongthawornsakun, B.; Mekasuwandumrong, O.; Santos Aires, F.J.C.; Büchel, R.; Baiker, A.; Pratsinis, S.E.; Panpranot, J. Variability of particle configurations achievable by 2-nozzle flame syntheses of the au-pd-tio2 system and their catalytic behaviors in the selective hydrogenation of acetylene. Appl. Catal. A Gen. 2018, 549, 1–7. [Google Scholar] [CrossRef]
- Zong, Y.; Li, S.; Niu, F.; Yao, Q. Direct synthesis of supported palladium catalysts for methane combustion by stagnation swirl flame. Proc. Combust. Inst. 2015, 35, 2249–2257. [Google Scholar] [CrossRef]
- Pineau, N.J.; Keller, S.D.; Güntner, A.T.; Pratsinis, S.E. Palladium embedded in SnO2 enhances the sensitivity of flame-made chemoresistive gas sensors. Microchim. Acta 2020, 187, 96. [Google Scholar] [CrossRef]
- Singh, V.; Dhall, S.; Kaushal, A.; Mehta, B.R. Room temperature response and enhanced hydrogen sensing in size selected Pd-C core-shell nanoparticles: Role of carbon shell and Pd-C interface. Int. J. Hydrogen Energy 2018, 43, 1025–1033. [Google Scholar] [CrossRef]
- Watkins, W.L.; Borensztein, Y. Ultrasensitive and fast single wavelength plasmonic hydrogen sensing with anisotropic nanostructured Pd films. Sens. Actuators B Chem. 2018, 273, 527–535. [Google Scholar] [CrossRef] [Green Version]
- Darmadi, I.; Nugroho, F.A.A.; Kadkhodazadeh, S.; Wagner, J.B.; Langhammer, C. Rationally designed pdaucu ternary alloy nanoparticles for intrinsically deactivation-resistant ultrafast plasmonic hydrogen sensing. ACS Sens. 2019, 4, 1424–1432. [Google Scholar] [CrossRef] [PubMed]
- Wadell, C.; Langhammer, C. Drift-corrected nanoplasmonic hydrogen sensing by polarization. Nanoscale 2015, 7, 10963–10969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farnood, A.; Ranjbar, M.; Salamati, H. Localized surface plasmon resonance (lspr) detection of hydrogen gas by Pd2+/Au core/shell like colloidal nanoparticles. Int. J. Hydrogen Energy 2020, 45, 1158–1169. [Google Scholar] [CrossRef]
- Rodal-Cedeira, S.; Montes-García, V.; Polavarapu, L.; Solís, D.M.; Heidari, H.; La Porta, A.; Angiola, M.; Martucci, A.; Taboada, J.M.; Obelleiro, F.; et al. Plasmonic au@pd nanorods with boosted refractive index susceptibility and sers efficiency: A multifunctional platform for hydrogen sensing and monitoring of catalytic reactions. Chem. Mater. 2016, 28, 9169–9180. [Google Scholar] [CrossRef]
- Wy, Y.; Lee, S.; Wi, D.H.; Han, S.W. Colloidal clusters of bimetallic core–shell nanoparticles for enhanced sensing of hydrogen in aqueous solution. Part. Part. Syst. Charact. 2018, 35, 1700380. [Google Scholar] [CrossRef]
- Yip, H.K.; Zhu, X.; Zhuo, X.; Jiang, R.; Yang, Z.; Wang, J. Gold nanobipyramid-enhanced hydrogen sensing with plasmon red shifts reaching ≈140 nm at 2 vol% hydrogen concentration. Adv. Opt. Mater. 2017, 5, 1700740. [Google Scholar] [CrossRef]
- Luong, H.M.; Pham, M.T.; Madhogaria, R.P.; Phan, M.-H.; Larsen, G.K.; Nguyen, T.D. Bilayer plasmonic nano-lattices for tunable hydrogen sensing platform. Nano Energy 2020, 71, 104558. [Google Scholar] [CrossRef]
- Zayats, A.V.; Smolyaninov, I.I.; Maradudin, A.A. Nano-optics of surface plasmon polaritons. Phys. Rep. 2005, 408, 131–314. [Google Scholar] [CrossRef]
- Strohfeldt, N.; Zhao, J.; Tittl, A.; Giessen, H. Sensitivity engineering in direct contact palladium-gold nano-sandwich hydrogen sensors [invited]. Opt. Mater. Express 2015, 5, 2525–2535. [Google Scholar] [CrossRef]
- Rochefort, A.; Abon, M.; Delichère, P.; Bertolini, J.C. Alloying effect on the adsorption properties of Pd50Cu50{111} single crystal surface. Surf. Sci. 1993, 294, 43–52. [Google Scholar] [CrossRef]
- Debauge, Y.; Abon, M.; Bertolini, J.C.; Massardier, J.; Rochefort, A. Synergistic alloying behaviour of Pd50Cu50 single crystals upon adsorption and CO-adsorption of CO and NO. Appl. Surf. Sci. 1995, 90, 15–27. [Google Scholar] [CrossRef]
- Matuschek, M.; Singh, D.P.; Jeong, H.-H.; Nesterov, M.; Weiss, T.; Fischer, P.; Neubrech, F.; Liu, N. Chiral plasmonic hydrogen sensors. Small 2018, 14, 1702990. [Google Scholar] [CrossRef]
- He, J.; Villa, N.S.; Luo, Z.; An, S.; Shen, Q.; Tao, P.; Song, C.; Wu, J.; Deng, T.; Shang, W. Integrating plasmonic nanostructures with natural photonic architectures in pd-modified morpho butterfly wings for sensitive hydrogen gas sensing. RSC Adv. 2018, 8, 32395–32400. [Google Scholar] [CrossRef] [Green Version]
- Sterl, F.; Strohfeldt, N.; Both, S.; Herkert, E.; Weiss, T.; Giessen, H. Design principles for sensitivity optimization in plasmonic hydrogen sensors. ACS Sens. 2020, 5, 917–927. [Google Scholar] [CrossRef]
- ElKabbash, M.; Sreekanth, K.V.; Alapan, Y.; Kim, M.; Cole, J.; Fraiwan, A.; Letsou, T.; Li, Y.; Guo, C.; Sankaran, R.M.; et al. Hydrogen sensing using thin-film perfect light absorber. ACS Photonics 2019, 6, 1889–1894. [Google Scholar] [CrossRef]
- Serhatlioglu, M.; Ayas, S.; Biyikli, N.; Dana, A.; Solmaz, M.E. Perfectly absorbing ultra thin interference coatings for hydrogen sensing. Opt. Lett. 2016, 41, 1724–1727. [Google Scholar] [CrossRef] [Green Version]
- Herkert, E.; Sterl, F.; Strohfeldt, N.; Walter, R.; Giessen, H. Low-cost hydrogen sensor in the ppm range with purely optical readout. ACS Sens. 2020, 5, 978–983. [Google Scholar] [CrossRef]
- Sirbuly, D.J.; Létant, S.E.; Ratto, T.V. Hydrogen sensing with subwavelength optical waveguides via porous silsesquioxane-palladium nanocomposites. Adv. Mater. 2008, 20, 4724–4727. [Google Scholar] [CrossRef]
- Song, H.; Luo, Z.; Liu, M.; Zhang, G.; Peng, W.; Wang, B.; Zhu, Y. Centrifugal deposited Au-Pd core-shell nanoparticle film for room-temperature optical detection of hydrogen gas. Sensors 2018, 18, 1448. [Google Scholar] [CrossRef] [Green Version]
- Monzón-Hernández, D.; Luna-Moreno, D.; Escobar, D.M.; Villatoro, J. Optical microfibers decorated with pdau nanoparticles for fast hydrogen sensing. Sens. Actuators B Chem. 2010, 151, 219–222. [Google Scholar] [CrossRef]
- Li, J.; Fan, R.; Hu, H.; Yao, C. Hydrogen sensing performance of silica microfiber elaborated with Pd nanoparticles. Mater. Lett. 2018, 212, 211–213. [Google Scholar] [CrossRef] [Green Version]
- Verma, N.; Delhez, R.; van der Pers, N.M.; Tichelaar, F.D.; Böttger, A.J. The role of the substrate on the mechanical and thermal stability of Pd thin films during hydrogen (de)sorption. Int. J. Hydrogen Energy 2021, 46, 4137–4153. [Google Scholar] [CrossRef]
- Prosser, J.H.; Brugarolas, T.; Lee, S.; Nolte, A.J.; Lee, D. Avoiding cracks in nanoparticle films. Nano Lett. 2012, 12, 5287–5291. [Google Scholar] [CrossRef]
- Feng, J.; Guo, X.; Ramlawi, N.; Pfeiffer, T.V.; Geutjens, R.; Basak, S.; Nirschl, H.; Biskos, G.; Zandbergen, H.W.; Schmidt-Ott, A. Green manufacturing of metallic nanoparticles: A facile and universal approach to scaling up. J. Mater. Chem. A 2016, 4, 11222–11227. [Google Scholar] [CrossRef] [Green Version]
- Biskos, G.; Vons, V.; Yurteri, C.U.; Schmidt-Ott, A. Generation and sizing of particles for aerosol-based nanotechnology. KONA Powder Part. J. 2008, 26, 13–35. [Google Scholar] [CrossRef] [Green Version]
- Feng, J.; Ramlawi, N.; Biskos, G.; Schmidt-Ott, A. Internally mixed nanoparticles from oscillatory spark ablation between electrodes of different materials. Aerosol Sci. Technol. 2018, 52, 505–514. [Google Scholar] [CrossRef] [Green Version]










| Material Type | Sensing Material | Sensor Operating Concentration Range (ppm) | Limit of Detection (ppm) | Response Time (s) Parentheses Show the Concentration (in ppm) and Temperature | Recovery Time (s) |
|---|---|---|---|---|---|
| Thin film | Pd-capped Y thin films [63] | 5–103 | 5 | 10 (3 × 103) 25 (104) | 250 |
| Pd-capped Mg thin films [64] | 2 × 103–4 × 104 | na | 500 (4 × 104) | na | |
| Pd-capped Ta thin films [59,67] Room temperature operation of the above sensor | 10−2–105 | na 2 (room temperature) | 7 (3 × 103; 120 °C) <1 (room temperature) | 20 ~1 (room temperature) | |
| Pd-capped Hf thin flms [68] PdAu thin films [37,44] | 10−2–105 ~10−2–105 | na (10−2) na (10−2) | 4 (120 °C) a few seconds (28 °C) | 30 (120 °C) a few tens of seconds | |
| ANP-based | PdAu nanoparticulate material [41] | 103–106 | na (103) | <10 | <20 |
| Anisotropic nanostructured Pd thin films [78] | 1–105 | 10 | 0.6 (4 × 104) | na | |
| INP-based | PdAu nanodiscs on glass [45] | 103–106 | na (103) | <1 (4 × 104) | na |
| PdNPs/fused silica [51] | 104 and 5 × 104 (pulses) | na | 2 (5 × 104) | 5 | |
| PdAuCu nanoparticles [79] | 103–106 | 5 | 0.4 (4 × 104; 30 °C) | 5 | |
| PdNPs/SnO2 waveguide [96] | 8 × 103 to ~32 × 104 | na (5 × 103) | 3 (3 × 104) | 2 | |
| Au@Pd NPs/quartz [97] | 103–4 × 104 | na (103) | 4 (4 × 104) | 30 | |
| PdAuNPs@PTFE@PMMA [33] | 102–106 | 103 | 0.3 (4 × 104; 30 °C) | 4 | |
| CN | PdAu nanohelices [90] | 104–2.5 × 104 | na | <20 (103) | <80 |
| Morpho Butterfly@Pd nanostrips [91] | 10–4 × 104 | <10 | 50 (102) ~20 (104) | na | |
| PdAuNPs/optical fiber [98] | 8 × 103–6 × 104 | na (<104) | 2 (4 × 104) | 20 | |
| PdNPs-PMMA/optical fiber [99] | 2 × 103–× 104 | 35.8 | 5 (104) | na |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sousanis, A.; Biskos, G. Thin Film and Nanostructured Pd-Based Materials for Optical H2 Sensors: A Review. Nanomaterials 2021, 11, 3100. https://doi.org/10.3390/nano11113100
Sousanis A, Biskos G. Thin Film and Nanostructured Pd-Based Materials for Optical H2 Sensors: A Review. Nanomaterials. 2021; 11(11):3100. https://doi.org/10.3390/nano11113100
Chicago/Turabian StyleSousanis, Andreas, and George Biskos. 2021. "Thin Film and Nanostructured Pd-Based Materials for Optical H2 Sensors: A Review" Nanomaterials 11, no. 11: 3100. https://doi.org/10.3390/nano11113100
APA StyleSousanis, A., & Biskos, G. (2021). Thin Film and Nanostructured Pd-Based Materials for Optical H2 Sensors: A Review. Nanomaterials, 11(11), 3100. https://doi.org/10.3390/nano11113100






