Tween® Preserves Enzyme Activity and Stability in PLGA Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. NP Formulation
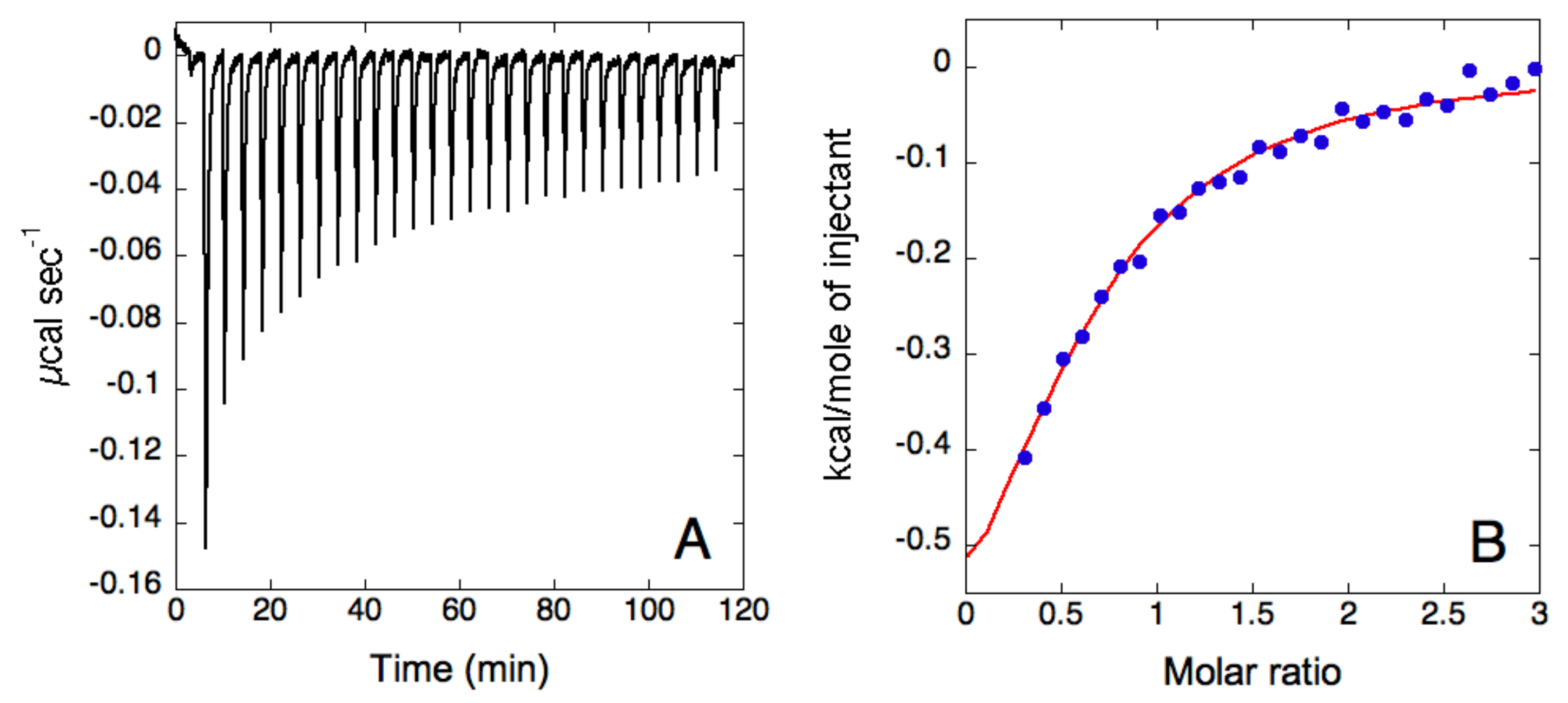
2.3. ITC Analysis
2.4. Size and Zeta Potential Analysis
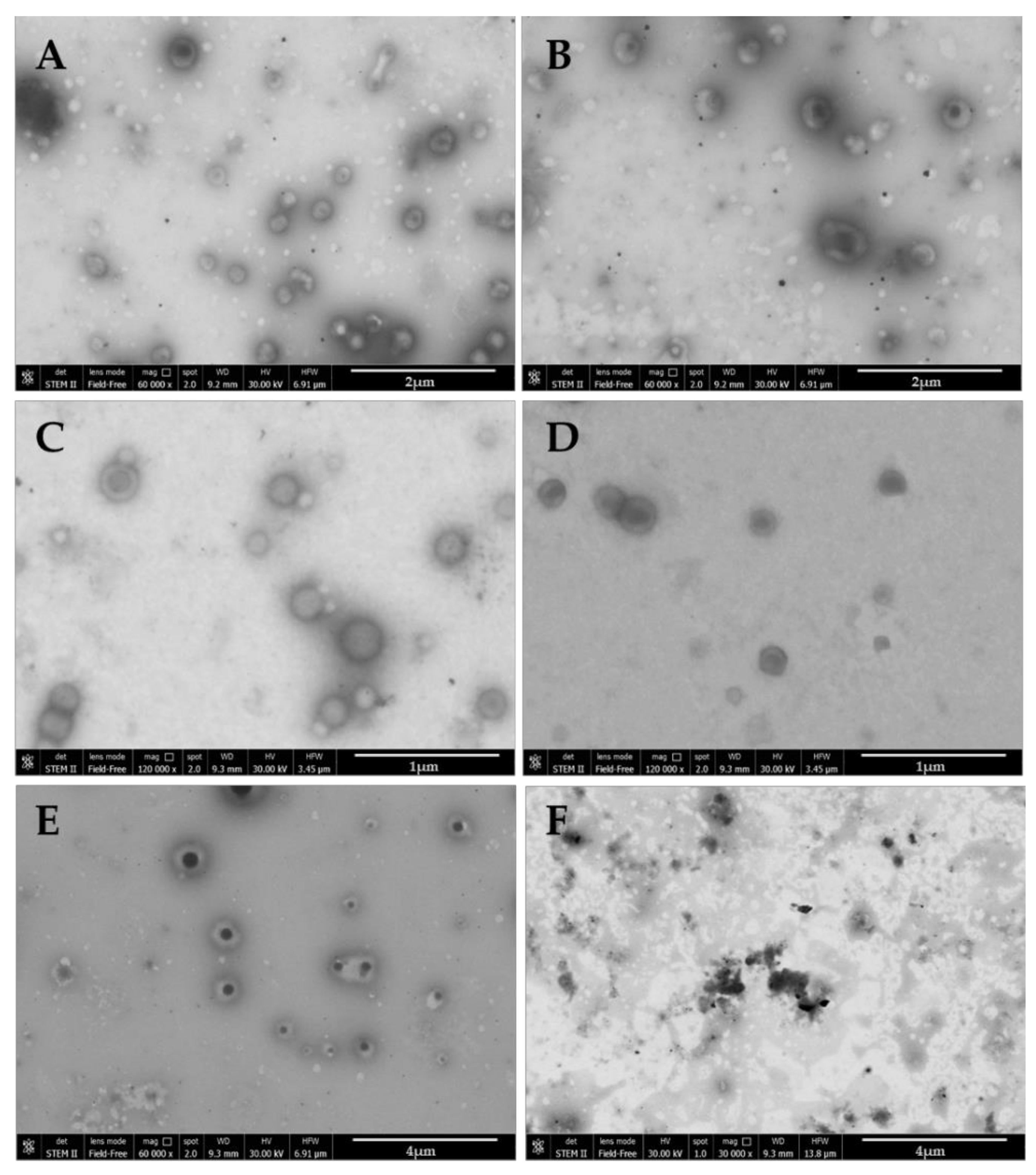
2.5. Microscopy Analysis
2.6. Weight Yield
2.7. Quantification of β-Glu in PLGA NPs
2.8. Quantification of Tween® in PLGA NPs
- -
- 0–5 min: 0–0% B
- -
- 5–10 min: 0–60% B
- -
- 10–15 min: 60–80% B
- -
- 15–17 min: 80–0% B
2.9. β-Glu Activity Assay
2.10. Enzyme Release Assay
2.11. Statistical Analysis
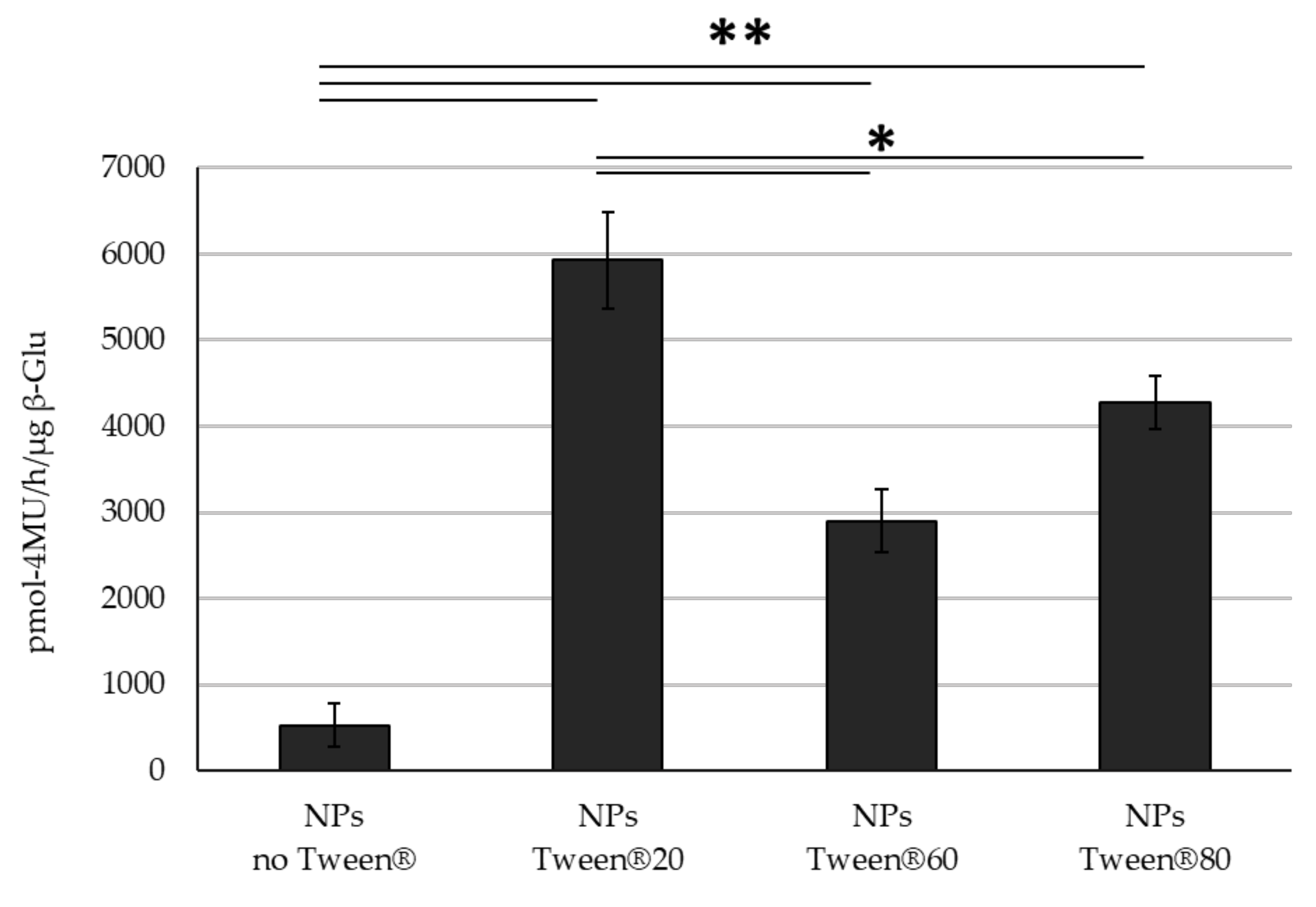
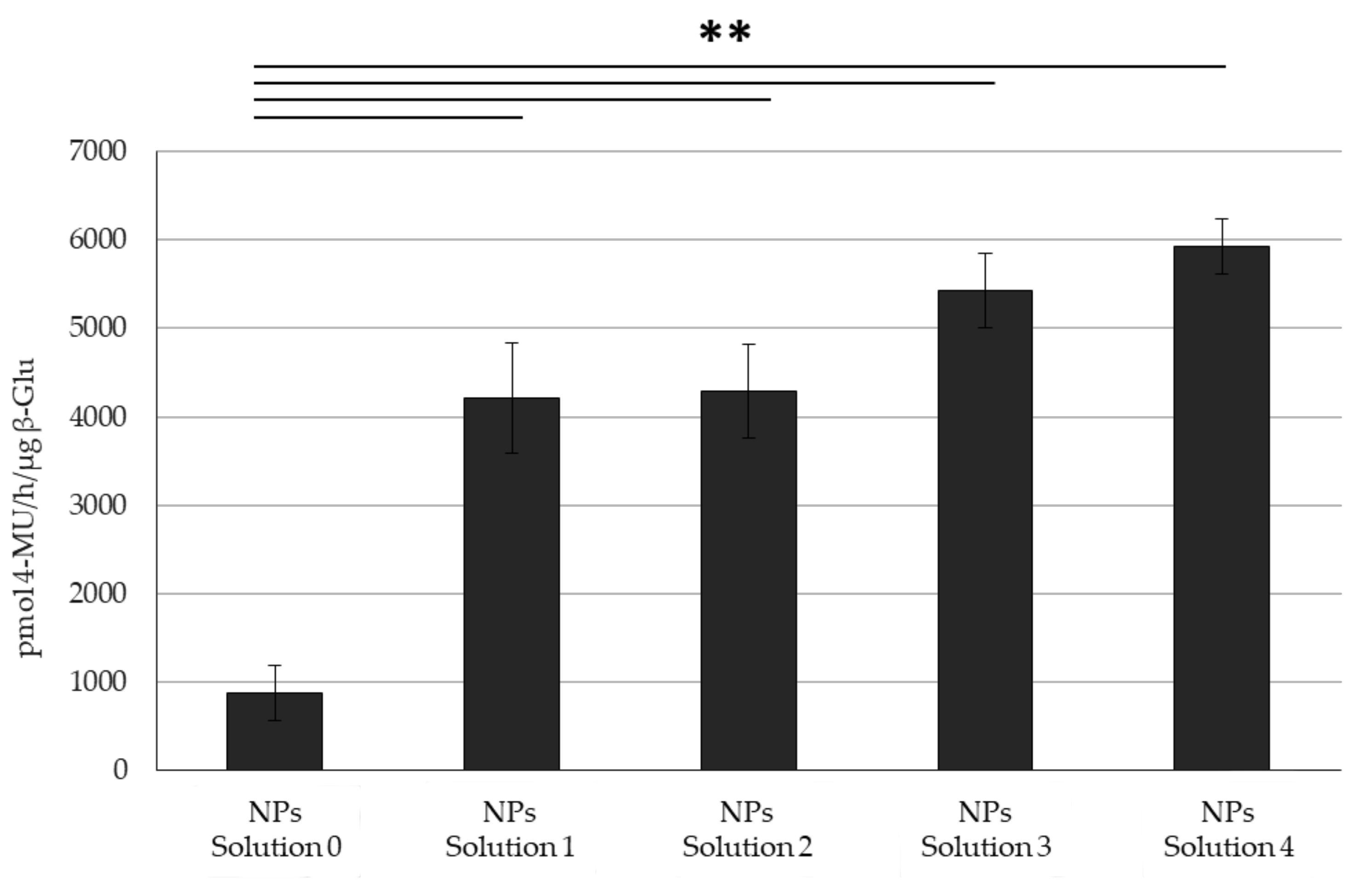
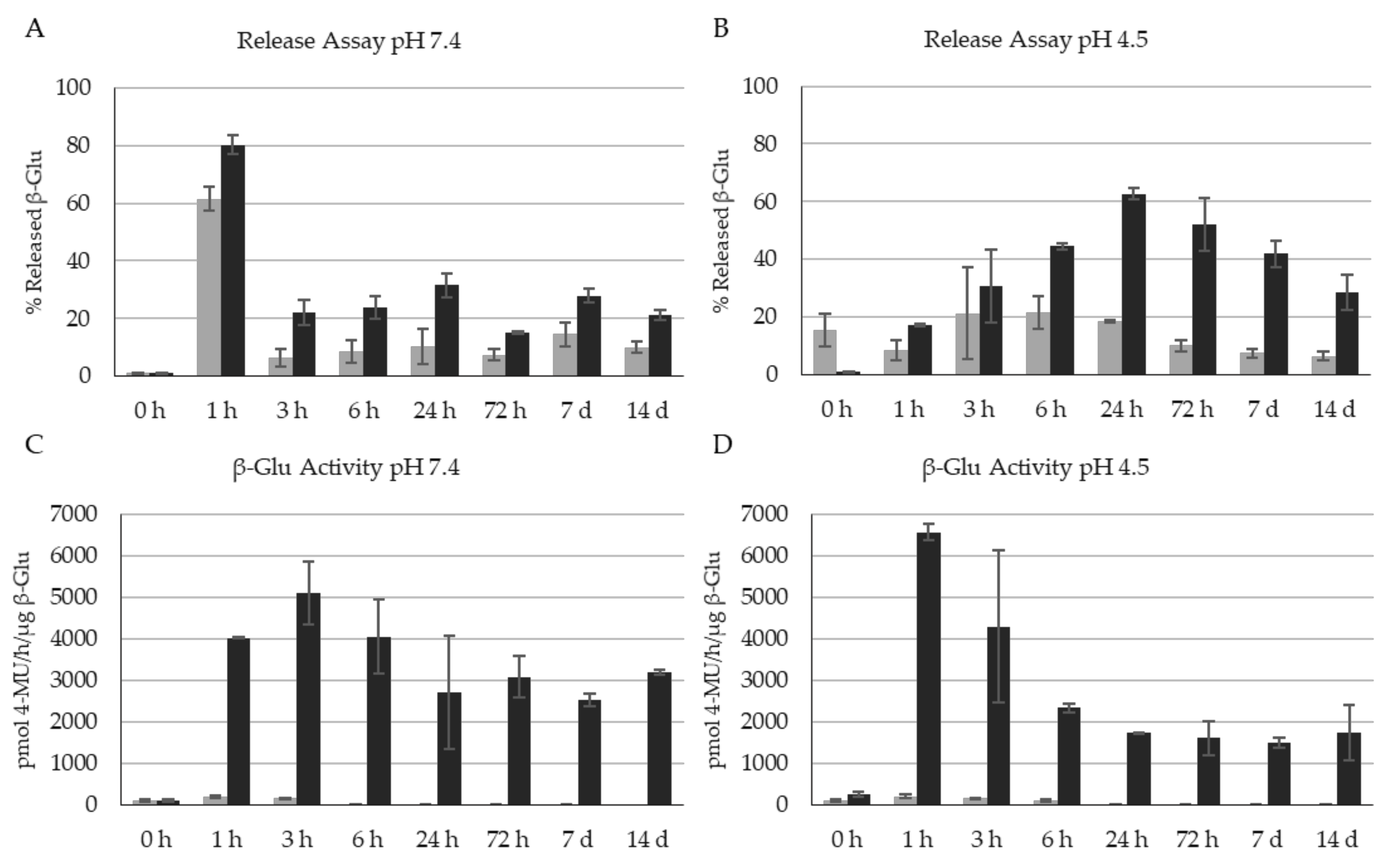
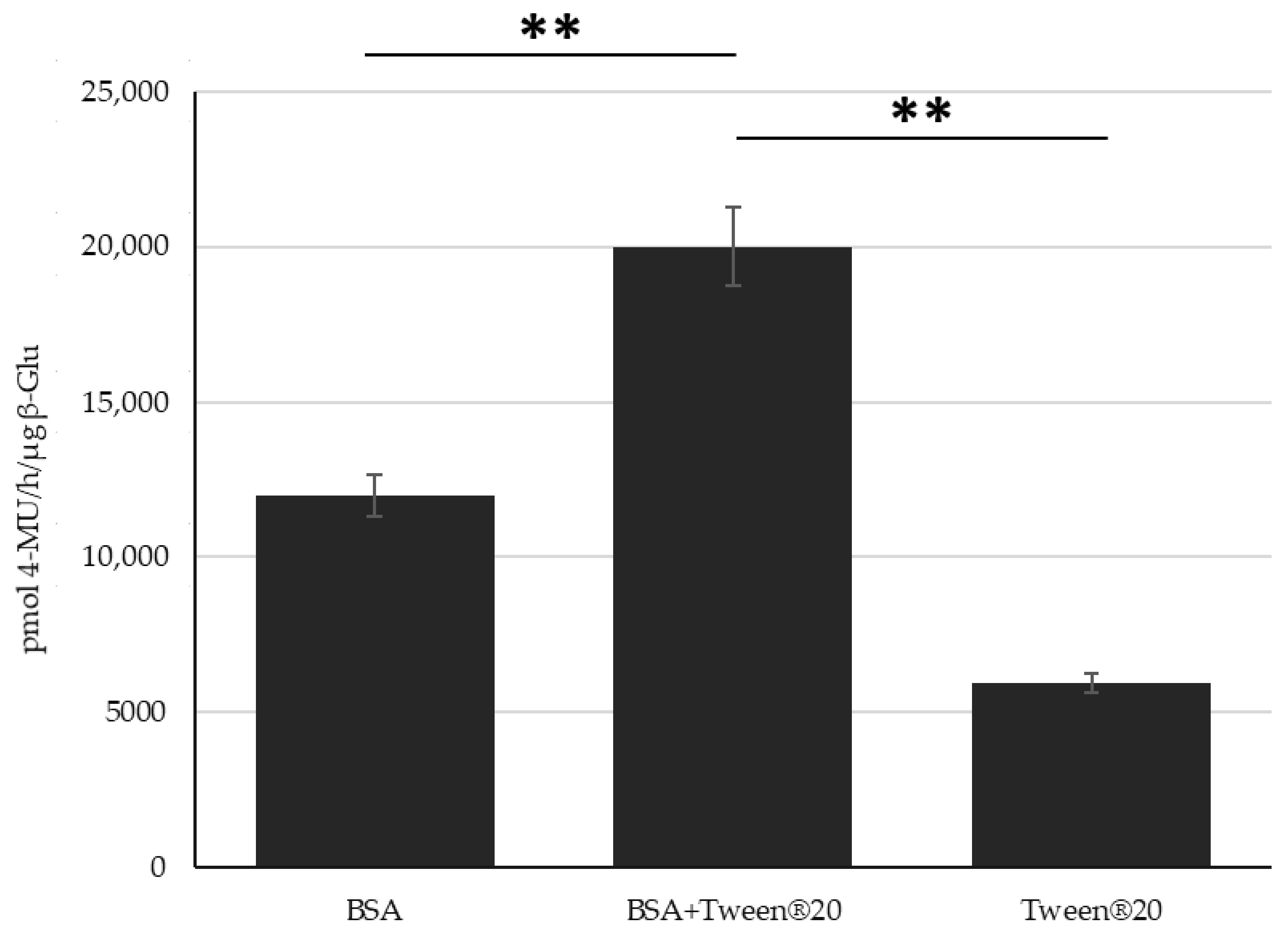
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Duskey, J.T.; Belletti, D.; Pederzoli, F.; Vandelli, M.A.; Forni, F.; Ruozi, B.; Tosi, G. Current Strategies for the Delivery of Therapeutic Proteins and Enzymes to Treat Brain Disorders. Int. Rev. Neurobiol. 2017, 137, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Rigon, L.; Salvalaio, M.; Pederzoli, F.; Legnini, E.; Duskey, J.T.; D’Avanzo, F.; De Filippis, C.; Ruozi, B.; Marin, O.; Vandelli, M.A.; et al. Targeting Brain Disease in MPSII: Preclinical Evaluation of IDS-Loaded PLGA Nanoparticles. Int. J. Mol. Sci. 2019, 20, 2014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mulvihill, J.J.; Cunnane, E.M.; Ross, A.M.; Duskey, J.T.; Tosi, G.; Grabrucker, A.M. Drug Delivery across the Blood–Brain Barrier: Recent Advances in the Use of Nanocarriers. Nanomedicine 2020, 15, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Tosi, G.; Duskey, J.T.; Kreuter, J. Nanoparticles as Carriers for Drug Delivery of Macromolecules across the Blood-Brain Barrier. Expert Opin. Drug Deliv. 2020, 17, 23–32. [Google Scholar] [CrossRef]
- Li, M. Enzyme Replacement Therapy: A Review and Its Role in Treating Lysosomal Storage Diseases. Pediatric Ann. 2018, 47, e191–e197. [Google Scholar] [CrossRef]
- Eisengart, J.B.; Jarnes, J.; Ahmed, A.; Nestrasil, I.; Ziegler, R.; Delaney, K.; Shapiro, E.; Whitley, C. Long-Term Cognitive and Somatic Outcomes of Enzyme Replacement Therapy in Untransplanted Hurler Syndrome. Mol. Genet. Metab. Rep. 2017, 13, 64–68. [Google Scholar] [CrossRef]
- Gaffke, L.; Pierzynowska, K.; Piotrowska, E.; Węgrzyn, G. How Close Are We to Therapies for Sanfilippo Disease? Metab. Brain Dis. 2018, 33, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Wiseman, J.A.; Meng, Y.; Nemtsova, Y.; Matteson, P.G.; Millonig, J.H.; Moore, D.F.; Sleat, D.E.; Lobel, P. Chronic Enzyme Replacement to the Brain of a Late Infantile Neuronal Ceroid Lipofuscinosis Mouse Has Differential Effects on Phenotypes of Disease. Mol. Ther.—Methods Clin. Dev. 2017, 4, 204–212. [Google Scholar] [CrossRef]
- Solovyeva, V.V.; Shaimardanova, A.A.; Chulpanova, D.S.; Kitaeva, K.V.; Chakrabarti, L.; Rizvanov, A.A. New Approaches to Tay-Sachs Disease Therapy. Front. Physiol 2018, 9, 1663. [Google Scholar] [CrossRef]
- Kumari, A.; Yadav, S.K.; Yadav, S.C. Biodegradable Polymeric Nanoparticles Based Drug Delivery Systems. Colloids Surf. B Biointerfaces 2010, 75, 1–18. [Google Scholar] [CrossRef]
- Wraith, J.E. Limitations of Enzyme Replacement Therapy: Current and Future. J. Inherit. Metab. Dis. 2006, 29, 442–447. [Google Scholar] [CrossRef]
- Concolino, D.; Deodato, F.; Parini, R. Enzyme Replacement Therapy: Efficacy and Limitations. Ital. J. Pediatrics 2018, 44, 120. [Google Scholar] [CrossRef]
- Ries, M. Enzyme Replacement Therapy and beyond—in Memoriam Roscoe O. Brady, M.D. (1923–2016). J. Inherit. Metab. Dis. 2017, 40, 343–356. [Google Scholar] [CrossRef]
- Nelemans, L.C.; Gurevich, L. Drug Delivery with Polymeric Nanocarriers—Cellular Uptake Mechanisms. Materials 2020, 13, 366. [Google Scholar] [CrossRef] [Green Version]
- Alven, S.; Aderibigbe, B.A. Efficacy of Polymer-Based Nanocarriers for Co-Delivery of Curcumin and Selected Anticancer Drugs. Nanomaterials 2020, 10, 1556. [Google Scholar] [CrossRef]
- Abasian, P.; Ghanavati, S.; Rahebi, S.; Khorasani, S.N.; Khalili, S. Polymeric Nanocarriers in Targeted Drug Delivery Systems: A Review. Polym. Adv. Technol. 2020, 31, 2939–2954. [Google Scholar] [CrossRef]
- Venditti, I. Morphologies and Functionalities of Polymeric Nanocarriers as Chemical Tools for Drug Delivery: A Review. J. King Saud Univ.-Sci. 2019, 31, 398–411. [Google Scholar] [CrossRef]
- Avramović, N.; Mandić, B.; Savić-Radojević, A.; Simić, T. Polymeric Nanocarriers of Drug Delivery Systems in Cancer Therapy. Pharmaceutics 2020, 12, 298. [Google Scholar] [CrossRef] [Green Version]
- Duskey, J.T.; Baraldi, C.; Gamberini, M.C.; Ottonelli, I.; Da Ros, F.; Tosi, G.; Forni, F.; Vandelli, M.A.; Ruozi, B. Investigating Novel Syntheses of a Series of Unique Hybrid PLGA-Chitosan Polymers for Potential Therapeutic Delivery Applications. Polymers 2020, 12, 823. [Google Scholar] [CrossRef] [Green Version]
- Oddone, N.; Boury, F.; Garcion, E.; Grabrucker, A.M.; Martinez, M.C.; Da Ros, F.; Janaszewska, A.; Forni, F.; Vandelli, M.A.; Tosi, G.; et al. Synthesis, Characterization, and In Vitro Studies of an Reactive Oxygen Species (ROS)-Responsive Methoxy Polyethylene Glycol-Thioketal-Melphalan Prodrug for Glioblastoma Treatment. Front. Pharmacol. 2020, 11. [Google Scholar] [CrossRef]
- Belletti, D.; Riva, G.; Luppi, M.; Tosi, G.; Forni, F.; Vandelli, M.A.; Ruozi, B.; Pederzoli, F. Anticancer Drug-Loaded Quantum Dots Engineered Polymeric Nanoparticles: Diagnosis/Therapy Combined Approach. Eur. J. Pharm. Sci. 2017, 107, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Dinu, I.A.; Duskey, J.T.; Car, A.; Palivan, C.G.; Meier, W. Engineered Non-Toxic Cationic Nanocarriers with Photo-Triggered Slow-Release Properties. Polym. Chem. 2016, 7, 3451–3464. [Google Scholar] [CrossRef]
- Birolini, G.; Valenza, M.; Ottonelli, I.; Passoni, A.; Favagrossa, M.; Duskey, J.T.; Bombaci, M.; Vandelli, M.A.; Colombo, L.; Bagnati, R.; et al. Insights into Kinetics, Release, and Behavioral Effects of Brain-Targeted Hybrid Nanoparticles for Cholesterol Delivery in Huntington’s Disease. J. Control. Release 2021, 330, 587–598. [Google Scholar] [CrossRef] [PubMed]
- Musumeci, T.; Bonaccorso, A.; Carbone, C.; Impallomeni, G.; Ballistreri, A.; Duskey, J.T.; Puglisi, G.; Pignatello, R. Development and Biocompatibility Assessments of Poly(3-Hydroxybutyrate-Co-ε-Caprolactone) Microparticles for Diclofenac Sodium Delivery. J. Drug Deliv. Sci. Technol. 2020, 60, 102081. [Google Scholar] [CrossRef]
- Pederzoli, F.; Ruozi, B.; Duskey, J.; Hagmeyer, S.; Sauer, A.K.; Grabrucker, S.; Coelho, R.; Oddone, N.; Ottonelli, I.; Daini, E.; et al. Nanomedicine against Aβ Aggregation by β–Sheet Breaker Peptide Delivery: In Vitro Evidence. Pharmaceutics 2019, 11, 572. [Google Scholar] [CrossRef] [Green Version]
- Puiggalí-Jou, A.; del Valle, L.J.; Alemán, C. Encapsulation and Storage of Therapeutic Fibrin-Homing Peptides Using Conducting Polymer Nanoparticles for Programmed Release by Electrical Stimulation. ACS Biomater. Sci. Eng. 2020, 6, 2135–2145. [Google Scholar] [CrossRef]
- Du, A.W.; Stenzel, M.H. Drug Carriers for the Delivery of Therapeutic Peptides. Biomacromolecules 2014, 15, 1097–1114. [Google Scholar] [CrossRef]
- Elsabahy, M.; Song, Y.; Eissa, N.G.; Khan, S.; Hamad, M.A.; Wooley, K.L. Morphologic Design of Sugar-Based Polymer Nanoparticles for Delivery of Antidiabetic Peptides. J. Control. Release 2021, 334, 1–10. [Google Scholar] [CrossRef]
- Tosi, G.; Pederzoli, F.; Belletti, D.; Vandelli, M.A.; Forni, F.; Duskey, J.T.; Ruozi, B. Nanomedicine in Alzheimer’s Disease: Amyloid Beta Targeting Strategy. Prog. Brain Res. 2019, 245, 57–88. [Google Scholar] [CrossRef]
- Liu, J.; Postupalenko, V.; Duskey, J.T.; Palivan, C.G.; Meier, W. PH-Triggered Reversible Multiple Protein–Polymer Conjugation Based on Molecular Recognition. J. Phys. Chem. B 2015, 119, 12066–12073. [Google Scholar] [CrossRef]
- Cózar-Bernal, M.J.; Holgado, M.A.; Arias, J.L.; Muñoz-Rubio, I.; Martín-Banderas, L.; Álvarez-Fuentes, J.; Fernández-Arévalo, M. Insulin-Loaded PLGA Microparticles: Flow Focusing versus Double Emulsion/Solvent Evaporation. J. Microencapsul. 2011, 28, 430–441. [Google Scholar] [CrossRef]
- Sigg, S.J.; Postupalenko, V.; Duskey, J.T.; Palivan, C.G.; Meier, W. Stimuli-Responsive Codelivery of Oligonucleotides and Drugs by Self-Assembled Peptide Nanoparticles. Biomacromolecules 2016, 17, 935–945. [Google Scholar] [CrossRef]
- Nussbaumer, M.G.; Duskey, J.T.; Rother, M.; Renggli, K.; Chami, M.; Bruns, N. Chaperonin–Dendrimer Conjugates for SiRNA Delivery. Adv. Sci. 2016, 3, 1600046. [Google Scholar] [CrossRef] [Green Version]
- Zou, W.; Liu, C.; Chen, Z.; Zhang, N. Preparation and Characterization of Cationic PLA-PEG Nanoparticles for Delivery of Plasmid DNA. Nanoscale Res. Lett. 2009, 4, 982–992. [Google Scholar] [CrossRef] [Green Version]
- Muro, S. New Biotechnological and Nanomedicine Strategies for Treatment of Lysosomal Storage Disorders. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2010, 2, 189–204. [Google Scholar] [CrossRef] [Green Version]
- Martinez, N.Y.; Andrade, P.F.; Durán, N.; Cavalitto, S. Development of Double Emulsion Nanoparticles for the Encapsulation of Bovine Serum Albumin. Colloids Surf. B Biointerfaces 2017, 158, 190–196. [Google Scholar] [CrossRef]
- Jahangiri, A.; Barghi, L. Polymeric Nanoparticles: Review of Synthesis Methods and Applications in Drug Delivery. J. Adv. Chem. Pharm. Mater. (JACPM) 2018, 1, 38–47. [Google Scholar]
- Mohanty, S.; Panda, S.; Purohit, D.; Si, S.C. A Comprehensive Review on PLGA-Based Nanoparticles Used for Rheumatoid Arthritis. Rese. J. Pharm. Technol. 2019, 12, 1481. [Google Scholar] [CrossRef]
- Xu, Y.; Kim, C.-S.; Saylor, D.M.; Koo, D. Polymer Degradation and Drug Delivery in PLGA-Based Drug–Polymer Applications: A Review of Experiments and Theories. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 1692–1716. [Google Scholar] [CrossRef]
- Yaghoobi, N.; Faridi Majidi, R.; Faramarzi, M.A.; Baharifar, H.; Amani, A. Preparation, Optimization and Activity Evaluation of PLGA/Streptokinase Nanoparticles Using Electrospray. Adv. Pharm. Bull. 2017, 7, 131–139. [Google Scholar] [CrossRef] [Green Version]
- Hasanpour, A.; Esmaeili, F.; Hosseini, H.; Amani, A. Use of MPEG-PLGA Nanoparticles to Improve Bioactivity and Hemocompatibility of Streptokinase: In-Vitro and in-Vivo Studies. Mater. Sci. Eng. C 2021, 118, 111427. [Google Scholar] [CrossRef]
- Ding, D.; Zhu, Q. Recent Advances of PLGA Micro/Nanoparticles for the Delivery of Biomacromolecular Therapeutics. Mater. Sci. Eng. C 2018, 92, 1041–1060. [Google Scholar] [CrossRef]
- Mohammadpour, F.; Hadizadeh, F.; Tafaghodi, M.; Sadri, K.; Mohammadpour, A.H.; Kalani, M.R.; Gholami, L.; Mahmoudi, A.; Chamani, J. Preparation, in Vitro and in Vivo Evaluation of PLGA/Chitosan Based Nano-Complex as a Novel Insulin Delivery Formulation. Int. J. Pharm. 2019, 572, 118710. [Google Scholar] [CrossRef]
- Kaplan, M.A.; Sergienko, K.V.; Kolmakova, A.A.; Konushkin, S.V.; Baikin, A.S.; Kolmakov, A.G.; Sevostyanov, M.A.; Kulikov, A.V.; Ivanov, V.E.; Belosludtsev, K.N.; et al. Development of a Biocompatible PLGA Polymers Capable to Release Thrombolytic Enzyme Prourokinase. J. Biomater. Sci. Polym. Ed. 2020, 31, 1405–1420. [Google Scholar] [CrossRef]
- Salvalaio, M.; Rigon, L.; Belletti, D.; D’Avanzo, F.; Pederzoli, F.; Ruozi, B.; Marin, O.; Vandelli, M.A.; Forni, F.; Scarpa, M.; et al. Targeted Polymeric Nanoparticles for Brain Delivery of High Molecular Weight Molecules in Lysosomal Storage Disorders. PLoS ONE 2016, 11, e0156452. [Google Scholar] [CrossRef] [Green Version]
- Yun, X.; Maximov, V.D.; Yu, J.; Zhu, G.; Vertegel, A.A.; Kindy, M.S. Nanoparticles for Targeted Delivery of Antioxidant Enzymes to the Brain after Cerebral Ischemia and Reperfusion Injury. J. Cereb Blood Flow Metab. 2013, 33, 583–592. [Google Scholar] [CrossRef] [Green Version]
- Schuster, T.; Mühlstein, A.; Yaghootfam, C.; Maksimenko, O.; Shipulo, E.; Gelperina, S.; Kreuter, J.; Gieselmann, V.; Matzner, U. Potential of Surfactant-Coated Nanoparticles to Improve Brain Delivery of Arylsulfatase A. J. Control. Release 2017, 253, 1–10. [Google Scholar] [CrossRef]
- Pérez, C.; Castellanos, I.J.; Costantino, H.R.; Al-Azzam, W.; Griebenow, K. Recent Trends in Stabilizing Protein Structure upon Encapsulation and Release from Bioerodible Polymers. J. Pharm. Pharmacol. 2002, 54, 301–313. [Google Scholar] [CrossRef]
- Primavessy, D.; Günday Türeli, N.; Schneider, M. Influence of Different Stabilizers on the Encapsulation of Desmopressin Acetate into PLGA Nanoparticles. Eur. J. Pharm. Biopharm. 2017, 118, 48–55. [Google Scholar] [CrossRef]
- Van de Weert, M.; Hennink, W.E.; Jiskoot, W. Protein Instability in Poly(Lactic-Co-Glycolic Acid) Microparticles. Pharm. Res. 2000, 17, 1159–1167. [Google Scholar] [CrossRef]
- Fonte, P.; Soares, S.; Sousa, F.; Costa, A.; Seabra, V.; Reis, S.; Sarmento, B. Stability Study Perspective of the Effect of Freeze-Drying Using Cryoprotectants on the Structure of Insulin Loaded into PLGA Nanoparticles. Biomacromolecules 2014, 15, 3753–3765. [Google Scholar] [CrossRef] [PubMed]
- Taghipour, B.; Yakhchali, M.; Haririan, I.; Tamaddon, A.M.; Samani, S.M. The Effects of Technical and Compositional Variables on the Size and Release Profile of Bovine Serum Albumin from PLGA Based Particulate Systems. Res. Pharm. Sci. 2014, 9, 407–420. [Google Scholar] [PubMed]
- Imamura, K.; Murai, K.; Korehisa, T.; Shimizu, N.; Yamahira, R.; Matsuura, T.; Tada, H.; Imanaka, H.; Ishida, N.; Nakanishi, K. Characteristics of Sugar Surfactants in Stabilizing Proteins During Freeze–Thawing and Freeze–Drying. J. Pharm. Sci. 2014, 103, 1628–1637. [Google Scholar] [CrossRef] [PubMed]
- Paillard-Giteau, A.; Tran, V.T.; Thomas, O.; Garric, X.; Coudane, J.; Marchal, S.; Chourpa, I.; Benoît, J.P.; Montero-Menei, C.N.; Venier-Julienne, M.C. Effect of Various Additives and Polymers on Lysozyme Release from PLGA Microspheres Prepared by an s/o/w Emulsion Technique. Eur. J. Pharm. Biopharm. 2010, 75, 128–136. [Google Scholar] [CrossRef]
- Rosa, G.D.; Iommelli, R.; La Rotonda, M.I.; Miro, A.; Quaglia, F. Influence of the Co-Encapsulation of Different Non-Ionic Surfactants on the Properties of PLGA Insulin-Loaded Microspheres. J. Control. Release 2000, 69, 283–295. [Google Scholar] [CrossRef]
- Duskey, J.T.; da Ros, F.; Ottonelli, I.; Zambelli, B.; Vandelli, M.A.; Tosi, G.; Ruozi, B. Enzyme Stability in Nanoparticle Preparations Part 1: Bovine Serum Albumin Improves Enzyme Function. Molecules 2020, 25, 4593. [Google Scholar] [CrossRef]
- Saraswat, M.; Reddy, P.Y.; Muthenna, P.; Reddy, G.B. Prevention of Non-Enzymic Glycation of Proteins by Dietary Agents: Prospects for Alleviating Diabetic Complications. Br. J. Nutr. 2008, 101, 1714–1721. [Google Scholar] [CrossRef] [Green Version]
- Pirooznia, N.; Hasannia, S.; Lotfi, A.S.; Ghanei, M. Encapsulation of Alpha-1 Antitrypsin in PLGA Nanoparticles: In Vitro Characterization as an Effective Aerosol Formulation in Pulmonary Diseases. J. Nanobiotechnol. 2012, 10, 20. [Google Scholar] [CrossRef] [Green Version]
- Iwai, J.; Ogawa, N.; Nagase, H.; Endo, T.; Loftsson, T.; Ueda, H. Effects of Various Cyclodextrins on the Stability of Freeze-Dried Lactate Dehydrogenase. J. Pharm. Sci. 2007, 96, 3140–3143. [Google Scholar] [CrossRef]
- Osman, R.; Kan, P.L.; Awad, G.; Mortada, N.; EL-Shamy, A.-E.; Alpar, O. Enhanced Properties of Discrete Pulmonary Deoxyribonuclease I (DNaseI) Loaded PLGA Nanoparticles during Encapsulation and Activity Determination. Int. J. Pharm. 2011, 408, 257–265. [Google Scholar] [CrossRef]
- Atkins, D.L.; Magana, J.R.; Sproncken, C.C.M.; van Hest, J.C.M.; Voets, I.K. Single Enzyme Nanoparticles with Improved Biocatalytic Activity through Protein Entrapment in a Surfactant Shell. Biomacromolecules 2021, 22, 1159–1166. [Google Scholar] [CrossRef]
- Hans, M.L.; Lowman, A.M. Biodegradable Nanoparticles for Drug Delivery and Targeting. Curr. Opin. Solid State Mater. Sci. 2002, 6, 319–327. [Google Scholar] [CrossRef]
- Astete, C.E.; Sabliov, C.M. Synthesis and Characterization of PLGA Nanoparticles. J. Biomater. Sci. Polym. Ed. 2006, 17, 247–289. [Google Scholar] [CrossRef]
- Arsiccio, A.; McCarty, J.; Pisano, R.; Shea, J.-E. Effect of Surfactants on Surface-Induced Denaturation of Proteins: Evidence of an Orientation-Dependent Mechanism. J. Phys. Chem. B 2018, 122, 11390–11399. [Google Scholar] [CrossRef]
- Michelin, K.; Wajner, A.; Goulart, L.S.; Fachel, Â.A.; Pereira, M.L.S.; de Mello, A.S.; Souza, F.T.S.; Pires, R.F.; Giugliani, R.; Coelho, J.C. Biochemical Study on β-Glucosidase in Individuals with Gaucher’s Disease and Normal Subjects. Clin. Chim. Acta 2004, 343, 145–153. [Google Scholar] [CrossRef]
- Belletti, D.; Grabrucker, A.M.; Pederzoli, F.; Menrath, I.; Vandelli, M.A.; Tosi, G.; Duskey, T.J.; Forni, F.; Ruozi, B. Hybrid Nanoparticles as a New Technological Approach to Enhance the Delivery of Cholesterol into the Brain. Int. J. Pharm. 2018, 543, 300–310. [Google Scholar] [CrossRef] [Green Version]
- Nayak, V.S.; Tan, Z.; Ihnat, P.M.; Russell, R.J.; Grace, M.J. Evaporative Light Scattering Detection Based HPLC Method for the Determination of Polysorbate 80 in Therapeutic Protein Formulations. J. Chromatogr. Sci. 2012, 50, 21–25. [Google Scholar] [CrossRef] [Green Version]
- Yoshii, H.; Buche, F.; Takeuchi, N.; Terrol, C.; Ohgawara, M.; Furuta, T. Effects of Protein on Retention of ADH Enzyme Activity Encapsulated in Trehalose Matrices by Spray Drying. J. Food Eng. 2008, 87, 34–39. [Google Scholar] [CrossRef]
- Bhatt, P.C.; Verma, A.; Al-Abbasi, F.A.; Anwar, F.; Kumar, V.; Panda, B.P. Development of Surface-Engineered PLGA Nanoparticulate-Delivery System of Tet1-Conjugated Nattokinase Enzyme for Inhibition of Aβ40 Plaques in Alzheimer’s Disease. Int. J. Nanomed. 2017, 12, 8749–8768. [Google Scholar] [CrossRef] [Green Version]
- Reddy, M.K.; Wu, L.; Kou, W.; Ghorpade, A.; Labhasetwar, V. Superoxide Dismutase-Loaded PLGA Nanoparticles Protect Cultured Human Neurons Under Oxidative Stress. Appl. Biochem. Biotechnol. 2008, 151, 565. [Google Scholar] [CrossRef] [Green Version]
- Parenti, G.; Pignata, C.; Vajro, P.; Salerno, M. New Strategies for the Treatment of Lysosomal Storage Diseases (Review). Int. J. Mol. Med. 2013, 31, 11–20. [Google Scholar] [CrossRef]
- Ratko, T.A.; Marbella, A.; Godfrey, S.; Aronson, N. Enzyme-Replacement Therapies for Lysosomal Storage Diseases; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2013. [Google Scholar]
- Ullman, J.C.; Arguello, A.; Getz, J.A.; Bhalla, A.; Mahon, C.S.; Wang, J.; Giese, T.; Bedard, C.; Kim, D.J.; Blumenfeld, J.R.; et al. Brain Delivery and Activity of a Lysosomal Enzyme Using a Blood-Brain Barrier Transport Vehicle in Mice. Sci. Transl. Med. 2020, 12. [Google Scholar] [CrossRef]
- Tang, R.; Kim, C.S.; Solfiell, D.J.; Rana, S.; Mout, R.; Velázquez-Delgado, E.M.; Chompoosor, A.; Jeong, Y.; Yan, B.; Zhu, Z.-J.; et al. Direct Delivery of Functional Proteins and Enzymes to the Cytosol Using Nanoparticle-Stabilized Nanocapsules. ACS Nano 2013, 7, 6667–6673. [Google Scholar] [CrossRef] [Green Version]
- Johnson, A.K.; Zawadzka, A.M.; Deobald, L.A.; Crawford, R.L.; Paszczynski, A.J. Novel Method for Immobilization of Enzymes to Magnetic Nanoparticles. J. Nanopart. Res. 2008, 10, 1009–1025. [Google Scholar] [CrossRef]
- Naeem, M.; Kim, W.; Cao, J.; Jung, Y.; Yoo, J.-W. Enzyme/PH Dual Sensitive Polymeric Nanoparticles for Targeted Drug Delivery to the Inflamed Colon. Colloids Surf. B Biointerfaces 2014, 123, 271–278. [Google Scholar] [CrossRef]
- Han, C.; Goodwine, J.; Romero, N.; Steck, K.S.; Sauer, K.; Doiron, A. Enzyme-Encapsulating Polymeric Nanoparticles: A Potential Adjunctive Therapy in Pseudomonas Aeruginosa Biofilm-Associated Infection Treatment. Colloids Surf. B Biointerfaces 2019, 184, 110512. [Google Scholar] [CrossRef]
- Seidel, Z.P.; Lee, C.T. Enhanced Activity of the Cellulase Enzyme β-Glucosidase upon Addition of an Azobenzene-Based Surfactant. ACS Sustain. Chem. Eng. 2020, 8, 1751–1761. [Google Scholar] [CrossRef]
- Otsuka, F.A.M.; Chagas, R.S.; Almeida, V.M.; Marana, S.R. Homodimerization of a Glycoside Hydrolase Family GH1 β-Glucosidase Suggests Distinct Activity of Enzyme Different States. Protein Sci. 2020, 29, 1879–1889. [Google Scholar] [CrossRef]
- Caramia, S.; Gatius, A.G.M.; dal Piaz, F.; Gaja, D.; Hochkoeppler, A. Dual Role of Imidazole as Activator/Inhibitor of Sweet Almond (Prunus Dulcis) β-Glucosidase. Biochem. Biophys. Rep. 2017, 10, 137–144. [Google Scholar] [CrossRef]
- Yusuf, M.; Khan, M.; Alrobaian, M.M.; Alghamdi, S.A.; Warsi, M.H.; Sultana, S.; Khan, R.A. Brain Targeted Polysorbate-80 Coated PLGA Thymoquinone Nanoparticles for the Treatment of Alzheimer’s Disease, with Biomechanistic Insights. J. Drug Deliv. Sci. Technol. 2021, 61, 102214. [Google Scholar] [CrossRef]
- Hodaei, D.; Baradaran, B.; Valizadeh, H.; Mohammadnejad, L.; Zakeri, P. The Effect of Tween Excipients on Expression and Activity of P-Glycoprotein in Caco-2 Cells. Pharm. Ind. 2014, 76, 788–794. [Google Scholar]
- Yang, S.; Liu, J.; Chen, Y.; Jiang, J. Reversal Effect of Tween-20 on Multidrug Resistance in Tumor Cells in Vitro. Biomed. Pharmacother. 2012, 66, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Dimitrijevic, D.; Shaw, A.J.; Florence, A.T. Effects of Some Non-Ionic Surfactants on Transepithelial Permeability in Caco-2 Cells. J. Pharm. Pharmacol. 2000, 52, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Scherließ, R. The MTT Assay as Tool to Evaluate and Compare Excipient Toxicity in Vitro on Respiratory Epithelial Cells. Int. J. Pharm. 2011, 411, 98–105. [Google Scholar] [CrossRef]
- Farkas, W.R.; Lorch, V.; Conover, W.R.; Al-Ansari, H.M.H.; Abney, L.K.; Painter, P.C.; Reyniers, J.P.; Congdon, C.C. Polysorbate Toxicity in Neonatal Rats and Mice. Pharmacol. Toxicol. 1991, 68, 154–156. [Google Scholar] [CrossRef]
- Chaturvedi, M.; Molino, Y.; Sreedhar, B.; Khrestchatisky, M.; Kaczmarek, L. Tissue Inhibitor of Matrix Metalloproteinases-1 Loaded Poly(Lactic-Co-Glycolic Acid) Nanoparticles for Delivery across the Blood–Brain Barrier. Int J. Nanomed. 2014, 9, 575–588. [Google Scholar] [CrossRef] [Green Version]
- Gelperina, S.; Maksimenko, O.; Khalansky, A.; Vanchugova, L.; Shipulo, E.; Abbasova, K.; Berdiev, R.; Wohlfart, S.; Chepurnova, N.; Kreuter, J. Drug Delivery to the Brain Using Surfactant-Coated Poly(Lactide-Co-Glycolide) Nanoparticles: Influence of the Formulation Parameters. Eur. J. Pharm. Biopharm. 2010, 74, 157–163. [Google Scholar] [CrossRef]
| Tween® 20:β-Glu* Solution n. | % v/v (Tween® 20/Water) ** | Tween® 20 (µmol) | Tween® 20:β-Glu (mol:mol) |
|---|---|---|---|
| 0 | 0 | 0 | 0 |
| 1 | 0.5 | 2.2 | 60:1 |
| 2 | 5.0 | 22.4 | 605:1 |
| 3 | 10.0 | 44.8 | 1209:1 |
| 4 | 20.0 | 89.6 | 2419:1 |
| Sample † | Z-Average nm ± S.D. | PDI ± S.D. | ζ-Pot mV ± S.D. | Yield% ± S.D. | LC% ± S.D. | EE% ± S.D. |
|---|---|---|---|---|---|---|
| Empty NPs | 190 ± 15 | 0.06 ± 0.01 | −20 ± 3 | 85.1 ± 3.1 | - | - |
| NPs β-Glu | 199 ± 25 | 0.09 ± 0.02 | −22 ± 6 | 82.5 ± 6.8 | 0.4 ± 0.1 | 3.9 ± 1.5 |
| NPs_Tween® 20:β-Glu | 198 ± 33 | 0.09 ± 0.06 | −16 ± 7 | 75.9 ± 2.3 | 0.9 ± 0.1 | 7.8 ± 1.9 * |
| NPs_Tween® 60:β-Glu | 177 ± 44 | 0.05 ± 0.04 | −23 ± 7 | 78.2 ± 1.8 | 0.8 ± 0.1 | 5.6 ± 2.6 |
| NPs_Tween® 80:β-Glu | 208 ± 23 | 0.22 ± 0.03 | −20 ± 4 | 72.7 ± 5.4 | 0.5 ± 0.1 | 5.4 ± 2.2 |
| Tween® 20:β-Glu Solution n. | Z-Average nm ± S.D. | PDI ± S.D. | Peak 1 nm ± S.D. (% ± S.D.) | Peak 2 nm ± S.D. (% ± S.D.) | Peak 3 nm ± S.D. (% ± S.D.) | ζ-Pot mV ± S.D. |
|---|---|---|---|---|---|---|
| 0 | 792 ± 27 | 0.65 ± 0.11 | 78 ± 12 (80 ± 4) | 144 ± 45 (15 ± 5) | - | −2.21 ± 4 |
| 1 | 932 ± 225 | 0.79 ± 0.06 | 564 ± 200 (73 ± 11) | 1185 ± 3120 (20 ± 3) | 602 ± 915 (5 ± 3) | 2.27 ± 3 |
| 2 | 636 ± 23 | 0.71 ± 0.09 | 521 ± 275 (60 ± 5) | 36 ± 46 (38 ± 8) | 3470 ± 2453 (2 ± 2) | 2.12 ± 2 |
| 3 | 114 ± 60 | 0.35 ± 0.09 | 9 ± 1 (65 ± 7) | 529 ± 11 (34 ± 7) | 5125 ± 191 (8 ± 4) | 2.13 ± 1 |
| 4 | 116 ± 47 | 0.49 ± 0.14 | 281 ± 470 (59 ± 8) | 936 ± 935 (30 ± 9) | 3218 ± 1697 (10 ± 8) | 2.14 ± 1 |
| Z-Average nm ± S.D. | PDI ± S.D. | AFM Diameter nm ± S.D. | ζ-Pot mV ± S.D. | Yield% ± S.D. | LC% ± S.D. | EE% ± S.D. | |
|---|---|---|---|---|---|---|---|
| Empty NPs | 190 ± 15 | 0.06 ± 0.01 | 320 ± 47 | −20 ± 3 | 85.1 ± 3.1 | - | - |
| NPs_Solution0 | 199 ± 25 | 0.09 ± 0.02 | 311 ± 69 | −22 ± 6 | 82.5 ± 6.8 | 0.4 ± 0.1 | 3.9 ± 1.5 |
| NPs_Solution1 | 185 ± 24 | 0.08 ± 0.03 | 173 ± 41 | −16 ± 5 | 72.7 ± 5.5 | 1.1 ± 0.0 | 9.7 ± 1.2 |
| NPs_Solution2 | 188 ± 13 | 0.08 ± 0.01 | 142 ± 39 | −16 ± 10 | 73.9 ± 4.3 | 1.0 ± 0.1 | 11.1 ± 1.2 |
| NPs_Solution3 | 191 ± 23 | 0.08 ± 0.02 | 150 ± 26 | −16 ± 7 | 81.8 ± 3.6 | 0.8 ± 0.1 | 8.4 ± 4.0 |
| NPs_Solution4 | 198 ± 33 | 0.09 ± 0.06 | 270 ± 31 | −16 ± 7 | 75.9 ± 2.3 | 0.9 ± 0.1 | 7.8 ± 2.5 |
| Samples | Tween® 20 Content mg Tween® 20/100 mg NPs | β-Glu Content mg β-Glu/100 mg NPs | Tween® 20:β-Glu mol/mol |
|---|---|---|---|
| NPs_Solution1 | 9.4 ± 0.3 | 1.1 ± 0.0 | 844 |
| NPs_Solution2 | 9.3 ± 0.2 | 1.0 ± 0.1 | 872 |
| NPs_Solution3 | 9.3 ± 0.1 | 0.8 ± 0.1 | 1233 |
| NPs_Solution4 | 9.7 ± 0.6 | 0.9 ± 0.1 | 1560 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duskey, J.T.; Ottonelli, I.; Rinaldi, A.; Parmeggiani, I.; Zambelli, B.; Wang, L.Z.; Prud’homme, R.K.; Vandelli, M.A.; Tosi, G.; Ruozi, B. Tween® Preserves Enzyme Activity and Stability in PLGA Nanoparticles. Nanomaterials 2021, 11, 2946. https://doi.org/10.3390/nano11112946
Duskey JT, Ottonelli I, Rinaldi A, Parmeggiani I, Zambelli B, Wang LZ, Prud’homme RK, Vandelli MA, Tosi G, Ruozi B. Tween® Preserves Enzyme Activity and Stability in PLGA Nanoparticles. Nanomaterials. 2021; 11(11):2946. https://doi.org/10.3390/nano11112946
Chicago/Turabian StyleDuskey, Jason Thomas, Ilaria Ottonelli, Arianna Rinaldi, Irene Parmeggiani, Barbara Zambelli, Leon Z. Wang, Robert K. Prud’homme, Maria Angela Vandelli, Giovanni Tosi, and Barbara Ruozi. 2021. "Tween® Preserves Enzyme Activity and Stability in PLGA Nanoparticles" Nanomaterials 11, no. 11: 2946. https://doi.org/10.3390/nano11112946
APA StyleDuskey, J. T., Ottonelli, I., Rinaldi, A., Parmeggiani, I., Zambelli, B., Wang, L. Z., Prud’homme, R. K., Vandelli, M. A., Tosi, G., & Ruozi, B. (2021). Tween® Preserves Enzyme Activity and Stability in PLGA Nanoparticles. Nanomaterials, 11(11), 2946. https://doi.org/10.3390/nano11112946










